Abstract
Introduction
Studies on complement activation have implicated a combination of the classical pathway (CP), lectin pathway (LP), and alternative pathway (AP) in triggering the terminal pathway (TP) for each common autoimmune glomerulonephritis (GN). Evaluating different pathways simultaneously may help identify whether one is preferentially activated and, consequently, which is best to target for each disease.
Methods
We followed 112 patients with focal segmental glomerular sclerosis (FSGS), membranous nephropathy (MN), IgA nephropathy (IgAN), lupus nephritis (LN), and antineutrophil cytoplasmic autoantibody-associated vasculitis (AAV) for a median duration of 22 (12–52) months. At the time of greatest clinical activity, we simultaneously evaluated urinary C3a (C3 convertase activity), C5a and sC5b-9 (TP), MASP-1 and MASP-2 (LP), C1q (CP), C4a (CP/LP), and Ba and Bb (AP). We evaluated the relation between activation fragments of the AP and CP/LP with the TP.
Results
Urinary complement biomarkers for each pathway were associated with the severity of proteinuria. Fragments of the TP were higher among patients with FSGS and MN compared with patients with IgAN, LN, and AAV. For the AP, urinary Ba level was lower in those with IgAN and LN compared with those with FSGS. For the CP/LP, urinary C4a, MASP-1, and MASP-2 levels were similar between diseases whereas urinary C1q levels were lower in those with LN. For each GN, independent associations existed between the activation markers of the AP and CP/LP with the degree of TP activation, except for the AP in AAV, although perhaps underpowered.
Conclusion
The AP and CP/LP contribute individually to the TP activation in autoimmune GN, and both seem to be valid potential therapeutic targets.
Keywords: autoimmune glomerulonephritis, complement pathway activation, monocyte chemoattractant protein-1, proteinuria, transforming growth factor β1
Graphical abstract
See Commentary on Page 939
Complement activation contributes to glomerular injury in autoimmune GN, including MN,1, 2, 3, 4, 5, 6, 7 IgAN,10, 11, 12, 13, 14, 15, 8, 9 LN,3,16, 17, 18, 19, 20 AAV,20, 21, 22, 23 and FSGS.3,5,24, 25, 26, 27
The AP, LP, and CP each generate C3 convertases, which cleave complement protein C3 into C3a and C3b and initiate tissue injury through C3b-mediated phagocytosis, formation of the membrane attack complex, and production of the C3a and C5a anaphylatoxins.28 At least 2 of the initiating branches of the complement system have been implicated in each of the common autoimmune glomerular diseases (see Supplementary Table S1). Nevertheless, their relative importance to one another in the pathogenesis of glomerular disease is unknown.5,24,27 Few of the cited studies have evaluated all pathways simultaneously, most focused on a single GN, and many used animal models or revised human pathology specimens with little clinical correlations. It is also uncertain how closely proteinuria, inflammation, and fibrosis are tied to complement activation rather than explained by other mechanisms.
The presence of C4a indicates activation of either the CP/LP whereas AP activation can be evidenced by the presence of factor B fragments (Ba and Bb). TP activation can be detected by measuring C3a, C5a, and soluble C5b-9 (sC5b-9). Identifying which complement pathway is preferentially activated may help focus future studies and lead to novel treatment modalities. Indeed, inhibiting the TP has already opened new therapeutic avenues for C3 glomerulopathy and AAV.29, 30, 31 Newer agents in investigation target 1 of the 3 initiating branches of the complement system.32,33
Consequently, we simultaneously evaluated complement pathway activation in patients with FSGS, MN, IgAN, LN, and AAV. We measured urinary C4a (CP/LP), MASP-1, MASP-2 (LP), C1q (CP), factor B fragments, Ba and Bb (AP), C3a (C3 convertase activity), C5a, and sC5b-9 (TP). We also evaluated urinary MCP-1 and TGF-β1 as surrogates of inflammation and fibrosis, respectively. We hypothesize that these urinary biomarkers will help identify which complement pathways are preferentially activated in each GN and that they will parallel disease severity, as evaluated by proteinuria, urinary MCP-1, and TGF-β1 levels.
Methods
Study Design
We performed an observational study in subjects with autoimmune GN recruited between 2006 and 2020 in 2 hospitals affiliated with the University of Montreal, Quebec, Canada. Each center’s ethics committee approved this study, and all participants provided informed consent. In doing so, the participants agreed to biobank urinary specimens with the possibility of subsequently evaluating other urinary biomarkers relevant to their disease. This work has been carried out in accordance with the Declaration of Helsinki.
Patients and Samples
We included all available individuals with biopsy-proven FSGS, MN, IgAN, LN, and AAV. Few patients with a history of diabetes were included in the study provided there were no signs of diabetic nephropathy. Renal biopsy was needed to be performed within 1 year of enrollment only when diabetes was preexisting, because diabetic nephropathy can result in renal complement activation.34 In the absence of diabetes, there was no time limitation between renal biopsy and enrollment. We collected blood pressure, serum creatinine, proteinuria, and antihypertensive and immunosuppressive drug use at each visit. A urinary void was taken at each visit. One part was sent for measurement of proteinuria with urinary creatinine, expressed in g/g of creatinine. The other part was frozen within 2 hours at −20 °C (rarely up to 6 hours after the end of a clinic). Once several samples were collected, these were thawed and then centrifuged at 200g for 10 minutes, aliquoted in multiple 0.4-ml vials, and stored at −80 °C until further processing. No protease inhibitor was used for storage. Finally, we obtained samples for comparison from patients without diabetes with chronic kidney disease (CKD) with <1 g/g proteinuria, clinically presumed to be secondary to nonglomerular diseases. These patients had also provided informed consent, and samples were handled identically.
We measured all complement proteins of interest once simultaneously in each individual using the urinary sample with the highest protein-to-creatinine ratio, with the exception of AAV for which we used the first available urinary sample at presentation with active vasculitis. Proteinuria and urinary C5a and sC5b-9 levels were also evaluated at the time of low clinical activity in these same patients, defined as the urinary sample with the lowest protein-to-creatinine ratio. The measurement of biomarkers at lowest clinical activity was done only if the patient was in remission at that time, defined by a proteinuria level ≤ 3.5 g/g for FSGS and MN and ≤1 g/g for IgAN and LN.35 Because all patients with AVV had a clinical remission, we used the sample with the lowest proteinuria level. The estimated glomerular filtration rate (eGFR) at the time of urinary sampling was estimated using the CKD-Epidemiology Collaboration formula.
Urinary Biomarker Measurements
We used human EIA Kits (MicroVue, Quidel Corp., San Diego, CA) to study the urinary complement fragments: C3a reflecting C3 convertase activity; C5a and sC5b-9 as evidence of TP activation; C4a as evidence of CP or LP activation; and Ba and Bb as evidence of AP activation. Although there is no activation of fragment differentiating CP from LP, we measured urinary C1q (Assay Pro, St. Charles, MO), a protein necessary for the initiation of CP, and urinary MASP-1 and MASP-2 (Cloud Clone Corp., Katy, TX), serine proteases essential for LP activation. Urine samples were initially diluted 1:5, with the exception of sC5b-9 and Ba, which were diluted 1:3 and 1:15, respectively. A few samples were further diluted to be within the detection limit set by each enzyme-linked immunosorbent assay. The assays’ lower sensitivity threshold for urinary C3a, C4a, C5a, Ba, Bb, sC5-9, MASP-1, MASP-2, and C1q was 0.06, 1.5, 0.05, 0.055, 90, 15, 0.45, 0.193, and 0.35 μg/l, respectively, and results are reported in μg/mmol of creatinine. Because urinary biomarker levels are likely influenced by the degree of proteinuria, we also considered values in μg/g proteinuria.5
Urinary MCP-1 and TGF-β1 have a prognostic value in autoimmune GN and can be used as surrogate markers.36, 37, 38, 39 They were measured using 2 Human Cytokine/Chemokine plex kit (Millipore, St. Charles, MO) on a multiplex platform (Eve Technologies Corp., Calgary, Alberta, Canada). The assays’ lower sensitivity thresholds were 2 and 10 ng/l for MCP-1 and TGF-β1, respectively. Samples were not diluted. We report the values of MCP-1 and TGF-β1 in ng/mmol of creatinine.
Statistical Analyses
Normally distributed variables are presented as means ± SDs and nonparametric variables as medians with their interquartile range. Correlations between urinary biomarkers were calculated using Spearman’s rho (ρ). Comparisons between groups of normally distributed, nonparametric, and percentage variables were done using one-way analysis of variance (subgroup analyses using the least significant difference method), Kruskal–Wallis test (subgroup analyses performed with the Mann-Whitney U test using the Bonferroni method for multiple comparisons), and the Pearson χ2 test, respectively.
To distinguish which complement pathway was independently associated with the production of the TP fragments, we performed univariate and multivariate linear regression analyses using CP/LP and AP activation fragments as covariates. The levels of urinary fragments were transformed using the square root, to respect the assumptions of linearity. For the dependent variable (TP), we chose C5a over C3a and sC5b-9 because the square root of C5a was relatively normal. For the AP, we chose Ba over Bb for similar reasons. We repeated the regression analyses by further adjusting for the confounding effect of proteinuria.
The 2-tailed P < 0.05 was considered statistically significant. Analyses were performed using the SPSS software (version 26, SPSS Inc., Chicago, IL).
Results
Patient Characteristics
A total of 112 patients participated in the study, in which there were 20 FSGS, 19 MN, 33 IgAN, 19 LN, and 21 AAV. Those with MN and IgAN were considered to be primary. Patient characteristics are presented in Table 1. Participants were followed in 22 (12–52) months. At the time of sampling at maximal clinical activity, the eGFR varied from 29 ± 16 in AAV to 77 ± 40 ml/min per 1.73 m2 in LN. Proteinuria was highest among participants with FSGS and MN (Table 1). Blood pressure was well controlled, and most patients were using renin–angiotensin system blockade, although the proportion of participants receiving immunosuppression varied according to the disease. There was a median of 3 (0–22), 7 (2–32), 8 (1–29), 3 (0–22), and 1 (0–3) months from the kidney biopsy to the urinary measurement at maximal clinical activity for FSGS, MN, IgAN, LN, and AAV, respectively. A total of 11%, 24%, 41%, and 24% were classes II, III, IV, and V of LN, respectively. The biomarker data set was complete, with the exception of 1 patient for whom not all urinary complement biomarkers were available and 10 participants for which urinary MCP-1 and TGF-β1 were missing. When comparing biomarkers at lowest clinical activity, 15 of 112 samples were missing: 9 because there was only 1 sample for that patient and 6 because the sample at lowest clinical activity was not in remission. We also analyzed urinary complement biomarkers in 13 individuals (9 men and 4 women) with nondiabetic CKD, clinically presumed not to be glomerular disease, with an age of 65 ± 16 years, eGFR of 30 ± 25 ml/min per 1.73 m2, and proteinuria level of 0.4 (0.2–0.7) g/g.
Table 1.
Characteristics of patients at time of maximal clinical activity
| Variable | FSGS | MN | IgAN | LN | AAV | P |
|---|---|---|---|---|---|---|
| n | 20 | 19 | 33 | 19 | 21 | |
| Female sex, % | 35 | 37 | 30 | 58 | 24 | 0.22 |
| Age (yr) | 56 ± 16 | 58 ± 13 | 43 ± 16 | 36 ± 11 | 65 ± 11 | <0.001 (FSGS, MN, FSGS > IgAN, LN) |
| eGFR, ml/min per 1.73 m2 | 41 ± 29 | 64 ± 34 | 58 ± 35 | 77 ± 40 | 29 ± 16 | <0.001 (FSGS < MN, LN; AAV < MN; AAV < IgAN < LN) |
| Proteinuria, g/g creatinine) | 5.3 (2.2–8.4) | 6.3 (4.2–11.2) | 2.3 (1.1–2.3) | 2.9 (0.4–6.8) | 1.5 (0.9–4.3) | See Figure 1 |
| Systolic blood pressure, mm Hg | 135 ± 21 | 132 ± 21 | 130 ± 16 | 126 ± 17 | 137 ± 21 | 0.42 |
| Diastolic blood pressure, mm Hg | 75 ± 9 | 74 ± 11 | 81 ± 11 | 79 ± 13 | 77 ± 12 | 0.22 |
| Antihypertensive drugs, n | 2 (1–3) | 2 (2–4) | 1 (1–2) | 1 (0–3) | 2 (1–3) | 0.23 |
| RASB, % | 84 | 79 | 85 | 53 | 48 | 0.006 (FSGS, MN, IgAN > LN, AAV) |
| Immunosuppression, % | 37 | 47 | 42 | 84 | 90 | <0.001 (FSGS, MN, IgAN < LN, AAV) |
| Diabetes, % | 0 | 11 | 12 | 5 | 14 | 0.49 |
| Cardiovascular disease, % | 10 | 0 | 0 | 5 | 0 | 0.16 |
AAV, antineutrophil cytoplasmic autoantibody-associated vasculitis; eGFR, estimated glomerular filtration rate; FSGS, focal segmental glomerulosclerosis; IgAN, IgA nephropathy; LN, lupus nephritis; MN, membranous nephropathy; RASB, renin-angiotensin system blockade.
Comparisons of normal, nonparametric and percentage data were done using 1-way analysis of variance, the Kruskal-Wallis, and the Pearson χ2 tests, respectively.
TP Activation
Urinary complement biomarkers were detected among all participants. The strongest intercorrelations between complement biomarkers existed between C3 convertase activity (C3a) and the TP activation fragments, C5a and sC5b-9 (all Spearman’s rho >0.8, P < 0.001). TP activation also correlated strongly with proteinuria (Supplementary Table S2). Significantly higher levels of C5a and sC5b-9 were observed among participants with FSGS and MN, compared with those with IgAN, LN, or AAV (Figure 1 and Supplementary Figure S1). These findings were similar when urinary complement levels were expressed as a ratio of proteinuria (Supplementary Figure S2). There was a marked reduction in urinary C5a and sC5b-9 for each GN during the period of lowest clinical disease activity, paralleling to proteinuria.
Figure 1.
Terminal pathway activation urinary fragments in autoimmune GN. Urinary terminal pathway fragments, expressed as a creatinine ratio. Median differences were evaluated with the Kruskal–Wallis test, whereas subgroup analyses were performed using the Mann-Whitney U test and Bonferroni P value thresholds. AAV, antineutrophil cytoplasmic autoantibody-associated vasculitis; FSGS, focal segmental glomerular sclerosis; IgAN, IgA nephropathy; LN, lupus nephritis; MN, membranous nephropathy; creat, creatinine; GN, glomerulonephritis; DB, diabetes; CKD, chronic kidney disease.
Subjects with nondiabetic CKD had much lower levels of TP activation compared with subjects with active autoimmune GN with urinary C5a levels of 0.04 (0.02–0.16) and sC5b-9 levels of 0.0 (0.0–2.7) μg/mmol of creatinine (Figure 1). By contrast, these levels were similar to periods of low disease activity.
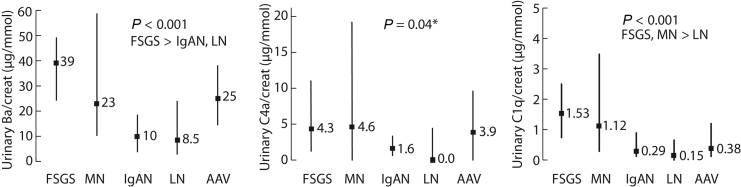
AP Activation
Urinary Ba level was higher among patients with FSGS compared with patients with IgAN and LN (Figure 2). Findings were similar when expressed as a proteinuria ratio (Supplementary Figure S2). Urinary Bb level was undetected in 35%, 47%, 58%, 84%, and 71% of FSGS, MN, IgAN, LN, and AAV, respectively (Supplementary Figure S1).
Figure 2.
Urinary Ba, C4a, and C1q in autoimmune GN. Expressed as a creatinine ratio. Median differences were evaluated with the Kruskal–Wallis test, whereas subgroup analyses were performed using the Mann-Whitney U test and Bonferroni P value thresholds. Although the Kruskal–Wallis tests were statistically significant for C4a, all subanalyses using the Bonferroni P value thresholds were not. AAV, antineutrophil cytoplasmic autoantibody-associated vasculitis; FSGS, focal segmental glomerulosclerosis; IgAN, IgA nephropathy; LN, lupus nephritis; MN, membranous nephropathy; creat, creatinine; GN, glomerulonephritis.
LP and CP Activation
Levels of urinary C4a, produced during activation of the LP or CP, did not significantly differ between the glomerular diseases, whether expressed as a creatinine (Figure 2) or proteinuria ratio (Supplementary Figure S2). To further explore the activation of LP and CP, respectively, levels of urinary MASP-1 and MASP-2 (LP) and urinary C1q (CP) were evaluated. Urinary C1q level was significantly lower in LN, compared with MN and FSGS (Figure 2 and Supplementary Figure S2). Urinary MASP-1 differed only between FSGS and LN. No difference was observed in urinary MASP-2 (Supplementary Figure S3). Findings were similar when expressed as a proteinuria ratio (data not found).
Driver of TP Activation
To evaluate which initiating pathway was responsible for the activation of the TP, we performed linear regression analyses between urinary Ba (AP) or C4a (CP/LP) and urinary C5a (TP) for each GN (Figure 3). A significant correlation was observed between the TP and both the CP/LP and AP in MN, IgAN, and LN (all P ≤ 0.005). In FSGS, the correlation between urinary Ba and C5a reached statistical significance (P = 0.003), whereas that between C4a and C5a was marginal (P = 0.058). Lastly, in AAV, a correlation between urinary C4a and C5a existed (P < 0.001) whereas that between Ba and C5a did not reach statistical significance (P = 0.091). We then performed multivariate linear regression analyses, which confirmed that urinary Ba and C4a levels independently predicted urinary C5a in FSGS, MN, IgAN, and LN. In AAV, only urinary C4a level significantly predicted TP activation, although we were underpowered to confidently exclude an association with AP (Table 2). We repeated these regression analyses by further adjusting for proteinuria. All associations remained significant with the exception of C4a in FSGS (Supplementary Table S3).
Figure 3.
Linear relationship between urinary C4a and Ba with C5a. AAV, antineutrophil cytoplasmic autoantibody-associated vasculitis; IgAN, IgA nephropathy; LN, lupus nephritis; MN, membranous nephropathy.
Table 2.
Independent associations between urinary levels of AP and CP/LP activation fragments and terminal pathway C5a
| Standardized β | P | |
|---|---|---|
| Focal and segmental glomerulosclerosis, r2: 0.52 | ||
| Ba | 0.59 | 0.003 |
| C4a | 0.36 | 0.047 |
| Membranous nephropathy, r2: 0.86 | ||
| Ba | 0.67 | <0.001 |
| C4a | 0.48 | <0.001 |
| IgA nephropathy, r2: 0.89 | ||
| Ba | 0.73 | <0.001 |
| C4a | 0.31 | <0.001 |
| Lupus nephritis, r2: 0.83 | ||
| Ba | 0.72 | <0.001 |
| C4a | 0.35 | 0.009 |
| ANCA-associated vasculitis, r2: 0.71 | ||
| Ba | 0.21 | 0.13 |
| C4a | 0.77 | <0.001 |
ANCA, antineutrophil cytoplasmic autoantibody; AP, alternative pathway; CP, classical pathway; LP, lectin pathway.
C4a and Ba correspond to CP/LP and AP activation fragments, respectively.
Correlations Between TP Activation and Proteinuria and Markers of Inflammation and Fibrosis
TP activation (urinary C5a) was significantly associated with the levels of proteinuria, urinary MCP-1, and TGF-β1 for each glomerular disease, except for AAV where urinary MCP-1 and TGF-β1 were perhaps driven by factors other than complement, although this analysis was perhaps underpowered (Supplementary Table S2). The results were almost identical on repeating the analyses using the anaphylatoxin C3a instead of C5a, except that, now, urinary C3a did correlate with urinary TGF-β1 in AAV (ρ = 0.47, P = 0.04).
Discussion
This study evaluated urinary components of the initiating and TPs of the complement system in autoimmune GN. Urinary complement cleavage products can be detected in a range of active glomerular disorders and have marked differences compared with nondiabetic CKD and paired samples at times of reduced GN activity. On the basis of these biomarkers, both AP and CP/LP were independently associated with the degree of TP activation for each studied GN except in AAV, where we were unable to find a relation between AP and TP. These associations also seemed independent of proteinuria. Finally, the C3a anaphylatoxin and TP activation correlated with proteinuria and markers of inflammation (urinary MCP-1) and fibrosis (urinary TGF-β1), which are surrogates of progressive disease.
Animal models have provided insight in the importance of the complement pathways and their complex interrelations. For instance, inactivation of AP in MN, FSGS, and LN animal models reduced glomerular disease development.6,7,40, 41, 42 In the LN MRL/lpr mouse model, MASP-1/3 deficiency confers protection against GN.43 Conversely, complement deficiencies can contribute to glomerular injury, further complexifying our understanding of complement in GN.44,45 Although animal studies have detailed many mechanisms of complement activation in glomerulopathies, corroborating which ones are most relevant in the clinical setting is lacking, as few studies have measured complement fragments in patients. Nevertheless, clinical trials have tested anticomplement therapies, including Avacopan, a C5a receptor antagonist, in AAV,30,31 and Narsoplimab, a MASP-2 inhibitor, in IgAN.32
Our objectives were to identify which initiating pathway(s) are independently associated with TP activation; gain insight in the correlations between complement activation and proteinuria, inflammation, and fibrosis; and finally help orient future studies on complement inhibition. This is the first study, to our knowledge, to evaluate all 3 complement activating pathways and the TP simultaneously. Our findings suggest that urinary C4a and Ba, specific to CP/LP and AP activation, respectively, both independently correlate with TP activation (urinary C5a), supporting previous pathology, animal and in vitro studies. In AAV, previous studies point toward pivotal roles for AP and CP/LP, and thus, we were most likely underpowered in our findings with only 21 patients.21,23 The elevated complement fragments found in FSGS were novel given that C3c is usually found scantly in sclerotic areas on biopsy. These unexpected findings warrant further investigation. We do not suggest measuring all these fragments in the clinical setting, nor does the present study address the value of repeated measurements.37 We performed these analyses because simultaneous measures of different complement biomarkers in multiple glomerulopathies, normalized for urinary creatinine or proteinuria, may provide useful information to other researchers addressing this promising field of investigation.
Whether triggering a specific pathway can set in motion the other initiating pathways is important to address. Although all 3 starting branches ultimately converge, 2 distinct C3 convertases, and, thus, 2 routes of TP activation exist. The CP and LP activation results in proteolytic cleavage of C2 and C4, which bind together to form the classical C3 convertase (C4bC2b), although the alternative C3 convertase (C3bBb) is specific to AP. Spontaneous activation of AP through C3 hydrolysis results in the production of C3b which binds to factor B. C3-bound factor B is cleaved by factor D to Bb and Ba fragments, catalyzing the formation of the alternative C3bBb convertase. Recent advances do establish crosstalk between the LP and the AP mediated by MASP-3, the exclusive activator of the AP’s factor D46; AP pathway activation can also be triggered by C3b production by CP/LP. The AP then acts as an amplification loop for further C3b production, leading to exponential TP activation and resulting in downstream inflammatory, phagocytic and lytic responses. Thus, the AP contributes to the CP and LP’s abilities to effectively defend the host against pathogens and maintain homeostasis47 and may explain the critical role of the AP observed in both animal and human studies of glomerular diseases.6,7,19, 20, 21, 22, 23, 24,40, 41, 42 Hence, primary AP activation may not be the sole mechanism by which factor B fragments are generated, as they may be produced by the AP’s amplification loop after CP/LP activation or by MASP-3.
Methodologic choices merit comment. We identified GN at the time of their greatest protein/creatinine ratio to improve the likelihood of identifying the predominant initiating complement pathway at the acme of disease, with the exception of AAV, where maximally active disease was considered to be initial presentation of their vasculitis. We opted for urinary over serum evaluation of complement activity because certain GN are renal-limited (e.g., primary MN and FSGS) and urinary biomarkers were previously found to predict disease.24,27,37,38,48 AP and CP/LP activation fragments were detected among the various GN; however, urinary biomarkers were higher in nephrotic-range glomerulopathies. Complement activation at the podocyte or in Bowman’s space may explain higher urinary levels observed in FSGS and MN. It does not imply that higher urinary measurements found in FSGS and MN equate to a greater complement burden, as complement fragments in IgAN, LN, and AAV may not be able to escape into the urine. Hence, levels of urinary complement fragments may not be comparable across glomerulopathies, and our findings do support further research. Indeed, there are trials revealing beneficial effects of complement inhibition in patients with IgAN and AAV, where we found lower activation fragments.30, 31, 32 Nevertheless, our results do suggest that complement inhibition should be considered in MN and FSGS. In addition, there is evidence suggesting heavy proteinuria stimulates tubular complement activation and may contribute to the higher levels of complement fragments.49 To address this, we did evaluate urinary biomarker levels expressed as a proteinuria ratio, yet results did not differ. Lastly, when correlating the TP with either AP or CP/LP activation, the TP anaphylatoxin C5a (11 kilodalton) was chosen because of its higher urinary detection rate compared with sC5b-9 (1030 kilodalton). Urinary Ba (30 kilodalton) was selected as the surrogate marker of AP activation as the larger Bb (60 kilodalton) was unfrequently detected in the urine, perhaps owing to lower urinary solubility or glomerular and/or tubular tissue fixation.46 Interestingly, levels of urinary sC5b-9 in active IgAN, LN, and AAV were similar to levels of 1.9 (0.5–10.4) found in a cohort of 83 subjects with overt diabetic nephropathy with an eGFR of 25 ± 9 ml/min per 1.72 m2 and a proteinuria level of 1.1 g/g.34 Levels of urinary complement fragments were much lower in individuals with nondiabetic CKD compared with those with active autoimmune GN.
Previous studies have evaluated levels of urinary C1q and MASP-1 in glomerular diseases.34 The serine proteases MASP-1 and MASP-2 activate the LP, whereas C1q is a recognition protein within the C1 complex which results in CP activation.46 Importantly, circulating MASP-1, MASP-2, and C1q are not cleavage fragments and thus do not confirm CP/LP activation. Furthermore, the MASP-1 and MASP-2 assays may not discriminate the inactive zymogen from its active form. This could explain why levels of urinary MASP-1 and MASP-2 were similar across GN. Urinary C1q, interestingly, was lowest among participants with LN possibly owing to excessive protein consumption or glomerular deposition during active LN.
Urinary MCP-1 and TGF-β1 are useful tools in evaluating disease activity and long-term outcomes in glomerular diseases.36, 37, 38, 39 Higher urinary levels of MCP-1 have been associated with active AAV, IgAN, and LN.38 Indeed, urinary MCP-1 was included as a secondary end point in trials investigating Avacopan in AAV,30,31 one of which observed a greater improvement in urinary MCP-1 among participants treated with Avacopan, militating for C5a/C5aR implication in MCP-1 secretion.30 TGF-β1 is a pleiotropic cytokine associated with the fibrogenic response after injury and subsequent progressive kidney disease.50 In vitro studies suggest that C5b-9–mediated sublytic injury is responsible for TGF-β1 secretion.51 Further supporting the role of complement in mediating progressive kidney disease comes from the LN MRL/lpr animal model in which inhibition of C3 activation resulted in the normalization of TGF-β1 and a reduction of glomerulosclerosis.52
Because multiple complement pathways may be activated simultaneously in glomerular diseases, an important question that arises is whether therapeutic approaches should focus on inhibiting the TP or specifically target an earlier complement pathway.53 Although likely more effective in controlling excessive complement activation, targeting the downstream TP comes at a cost, with a loss in important complement system functions. These include combating infections, linking the innate and adaptive immune responses, and clearing debris, apoptotic cells, and immune complexes.46 Targeting either the LP or AP would preserve the clearance capabilities of immune complexes and maintain a certain degree of host defense against pathogens.
Certain limitations must be underlined. The number of participants was modest, which may have precluded our ability to observe a significant correlation between AP and TP activation in AAV. More importantly, the lack of pathology correlations reduces the certainty of our findings. Biopsy immunohistochemistry could elucidate the role of chronic versus active lesions in the genesis of complement fragments.54 It could also reveal the localization of complement deposition (e.g., glomerular capillaries, podocyte, mesangial, tubular) and explain for the differences in urinary measurements observed between GN. Unfortunately, the patients were not enrolled at the time of renal diagnosis and, in some cases, the timing between the renal biopsy and urinary sampling was >1 year. The evolution for each patient, spontaneous or treatment-induced, during this time would undoubtedly affect urinary complement biomarkers. Finally, many subjects were receiving immunosuppressive treatments at the time of sampling, which may have influenced the results. It is possible that the maximal proteinuria does not correspond to the maximal immunologic activity when such treatments are given. Nevertheless, simultaneous immunohistologic localization of complement fragments would have addressed some limitations of urinary measurements, including confounding by filtration of circulating fragments.
In conclusion, our results suggest that both the AP and CP/LP contribute to TP activation in common autoimmune GN. This lends support to clinical trials investigating different complement pathway inhibition, either of the terminal or initiating branches, as possible therapeutic options in autoimmune GN.
Disclosure
VR has received honoraria as a consultant for Vertex Pharmaceuticals. ALL has received honoraria for advisory boards for Alexion and Astellas. All the other authors declared no competing interests.
Acknowledgments
This study was partly supported by grants from the Fonds de la recherche du Québec–Santé (#14395) and the Fondation de l’Hôpital du Sacré-Coeur de Montréal.
Footnotes
Table S1. Studies reporting the initiating pathway of complement activation in autoimmune glomerulonephritis.
Table S2. Correlation coefficients between terminal pathway activation (urinary C5a) and proteinuria and markers of inflammation and fibrosis.
Table S3. Independent associations between urinary levels of terminal pathway C5a with proteinuria, alternative and classic/lectin pathway activation fragments.
Figure S1. Urinary C3a et Bb in autoimmune glomerulonephritis.
Figure S2. Urinary C3a, C5a, sC5b9, Ba, C4a, and C1q expressed as a proteinuria ratio.
Figure S3. Urinary MASP-1 and MASP-2 in autoimmune glomerulonephritis.
STROBE Statement.
Supplementary Material
Table S1. Studies reporting the initiating pathway of complement activation in autoimmune glomerulonephritis.
Table S2. Correlation coefficients between terminal pathway activation (urinary C5a) and proteinuria and markers of inflammation and fibrosis.
Table S3. Independent associations between urinary levels of terminal pathway C5a with proteinuria, alternative and classic/lectin pathway activation fragments.
Figure S1. Urinary C3a et Bb in autoimmune glomerulonephritis.
Figure S2. Urinary C3a, C5a, sC5b9, Ba, C4a, and C1q expressed as a proteinuria ratio.
Figure S3. Urinary MASP-1 and MASP-2 in autoimmune glomerulonephritis.
STROBE Statement (PDF)
References
- 1.Hayashi N., Okada K., Matsui Y., et al. Glomerular mannose-binding lectin deposition in intrinsic antigen-related membranous nephropathy. Nephrol Dial Transplant. 2018;33:832–840. doi: 10.1093/ndt/gfx235. [DOI] [PubMed] [Google Scholar]
- 2.Segawa Y., Hisano S., Matsushita M., et al. IgG subclasses and complement pathway in segmental and global membranous nephropathy. Pediatr Nephrol. 2010;25:1091–1099. doi: 10.1007/s00467-009-1439-8. [DOI] [PubMed] [Google Scholar]
- 3.Lhotta K., Wurzner R., Konig P. Glomerular deposition of mannose-binding lectin in human glomerulonephritis. Nephrol Dial Transplant. 1999;14:881–886. doi: 10.1093/ndt/14.4.881. [DOI] [PubMed] [Google Scholar]
- 4.Drachenberg C.B., Papadimitriou J.C., Chandra P., et al. Epidemiology and pathophysiology of glomerular C4d staining in native kidney biopsies. Kidney Int Rep. 2019;4:1555–1567. doi: 10.1016/j.ekir.2019.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhang M.F., Huang J., Zhang Y.M., et al. Complement activation products in the circulation and urine of primary membranous nephropathy. BMC Nephrol. 2019;20:313. doi: 10.1186/s12882-019-1509-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Luo W., Olaru F., Miner J.H., et al. Alternative pathway is essential for glomerular complement activation and proteinuria in a mouse model of membranous nephropathy. Front Immunol. 2018;9:1433. doi: 10.3389/fimmu.2018.01433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schubart A., Anderson K., Mainolfi N., et al. Small-molecule factor B inhibitor for the treatment of complement-mediated diseases. Proc Natl Acad Sci U S A. 2019;116:7926–7931. doi: 10.1073/pnas.1820892116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Espinosa M., Ortega R., Sanchez M., et al. Association of C4d deposition with clinical outcomes in IgA nephropathy. Clin J Am Soc Nephrol. 2014;9:897–904. doi: 10.2215/CJN.09710913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Roos A., Rastaldi M.P., Calvaresi N., et al. Glomerular activation of the lectin pathway of complement in IgA nephropathy is associated with more severe renal disease. J Am Soc Nephrol. 2006;17:1724–1734. doi: 10.1681/ASN.2005090923. [DOI] [PubMed] [Google Scholar]
- 10.Medjeral-Thomas N.R., Troldborg A., Constantinou N., et al. Progressive IgA nephropathy is associated with low circulating mannan-binding lectin-associated serine protease-3 (MASP-3) and increased glomerular factor H-related protein-5 (FHR5) deposition. Kidney Int Rep. 2018;3:426–438. doi: 10.1016/j.ekir.2017.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Endo M., Ohi H., Ohsawa I., Fujita T., Matsushita M., Fujita T. Glomerular deposition of mannose-binding lectin (MBL) indicates a novel mechanism of complement activation in IgA nephropathy. Nephrol Dial Transplant. 1998;13:1984–1990. doi: 10.1093/ndt/13.8.1984. [DOI] [PubMed] [Google Scholar]
- 12.Zhu L., Zhai Y.L., Wang F.M., et al. Variants in complement factor H and complement factor H-related protein genes, CFHR3 and CFHR1, affect complement activation in IgA nephropathy. J Am Soc Nephrol. 2015;26:1195–1204. doi: 10.1681/ASN.2014010096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Medjeral-Thomas N.R., Lomax-Browne H.J., Beckwith H., et al. Circulating complement factor H-related proteins 1 and 5 correlate with disease activity in IgA nephropathy. Kidney Int. 2017;92:942–952. doi: 10.1016/j.kint.2017.03.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tortajada A., Gutierrez E., Goicoechea de Jorge E., et al. Elevated factor H-related protein 1 and factor H pathogenic variants decrease complement regulation in IgA nephropathy. Kidney Int. 2017;92:953–963. doi: 10.1016/j.kint.2017.03.041. [DOI] [PubMed] [Google Scholar]
- 15.Bao L., Haas M., Quigg R.J. Complement factor H deficiency accelerates development of lupus nephritis. J Am Soc Nephrol. 2011;22:285–295. doi: 10.1681/ASN.2010060647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sato N., Ohsawa I., Nagamachi S., et al. Significance of glomerular activation of the alternative pathway and lectin pathway in lupus nephritis [published correction appears in Lupus. 2011;20:1455] Lupus. 2011;20:1378–1386. doi: 10.1177/0961203311415561. [DOI] [PubMed] [Google Scholar]
- 17.Troldborg A., Thiel S., Trendelenburg M., et al. The lectin pathway of complement activation in patients with systemic lupus erythematosus. J Rheumatol. 2018;45:1136–1144. doi: 10.3899/jrheum.171033. [DOI] [PubMed] [Google Scholar]
- 18.Alperin J.M., Ortiz-Fernandez L., Sawalha A.H. Monogenic lupus: a developing paradigm of disease. Front Immunol. 2018;9:2496. doi: 10.3389/fimmu.2018.02496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Song D., Guo W.Y., Wang F.M., et al. Complement alternative pathways activation in patients with lupus nephritis. Am J Med Sci. 2017;353:247–257. doi: 10.1016/j.amjms.2017.01.005. [DOI] [PubMed] [Google Scholar]
- 20.Gou S.J., Yuan J., Chen M., Yu F., Zhao M.H. Circulating complement activation in patients with anti-neutrophil cytoplasmic antibody-associated vasculitis. Kidney Int. 2013;83:129–137. doi: 10.1038/ki.2012.313. [DOI] [PubMed] [Google Scholar]
- 21.Hilhorst M., van Paassen P., van Rie H., et al. Complement in ANCA-associated glomerulonephritis. Nephrol Dial Transplant. 2017;32:1302–1313. doi: 10.1093/ndt/gfv288. [DOI] [PubMed] [Google Scholar]
- 22.Xing G.Q., Chen M., Liu G., et al. Complement activation is involved in renal damage in human antineutrophil cytoplasmic autoantibody associated pauci-immune vasculitis. J Clin Immunol. 2009;29:282–291. doi: 10.1007/s10875-008-9268-2. [DOI] [PubMed] [Google Scholar]
- 23.Xing G.Q., Chen M., Liu G., Zheng X., E J., Zhao M.H. Differential deposition of C4d and MBL in glomeruli of patients with ANCA-negative pauci-immune crescentic glomerulonephritis. J Clin Immunol. 2010;30:144–156. doi: 10.1007/s10875-009-9344-2. [DOI] [PubMed] [Google Scholar]
- 24.Thurman J.M., Wong M., Renner B., et al. Complement activation in patients with focal segmental glomerulosclerosis. PLoS One. 2015;10 doi: 10.1371/journal.pone.0136558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.van de Lest N.A., Zandbergen M., Wolterbeek R., et al. Glomerular C4d deposition can precede the development of focal segmental glomerulosclerosis. Kidney Int. 2019;96:738–749. doi: 10.1016/j.kint.2019.04.028. [DOI] [PubMed] [Google Scholar]
- 26.Heybeli C., Oktan M.A., Yildiz S., et al. Mesangial C4d deposition is independently associated with poor renal survival in patients with primary focal segmental glomerulosclerosis. Clin Exp Nephrol. 2019;23:650–660. doi: 10.1007/s10157-018-01688-0. [DOI] [PubMed] [Google Scholar]
- 27.Huang J., Cui Z., Gu Q.H., et al. Complement activation profile of patients with primary focal segmental glomerulosclerosis. PLoS One. 2020;15 doi: 10.1371/journal.pone.0234934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sarma J.V., Ward P.A. The complement system. Cell Tissue Res. 2011;343:227–235. doi: 10.1007/s00441-010-1034-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bomback A.S., Smith R.J., Barile G.R., et al. Eculizumab for dense deposit disease and C3 glomerulonephritis. Clin J Am Soc Nephrol. 2012;7:748–756. doi: 10.2215/CJN.12901211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jayne D.R.W., Bruchfeld A.N., Harper L., et al. Randomized trial of C5a receptor inhibitor Avacopan in ANCA-associated vasculitis. J Am Soc Nephrol. 2017;28:2756–2767. doi: 10.1681/ASN.2016111179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Merkel P.A., Niles J., Jimenez R., et al. Adjunctive treatment with Avacopan, an oral C5a receptor inhibitor, in patients with antineutrophil cytoplasmic antibody-associated vasculitis. ACR Open Rheumatol. 2020;2:662–671. doi: 10.1002/acr2.11185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lafayette R.A., Rovin B.H., Reich H.N., et al. Safety, tolerability and efficacy of Narsoplimab, a novel MASP-2 inhibitor for the treatment of IgA nephropathy. Kidney Int Rep. 2020;5:2032–2041. doi: 10.1016/j.ekir.2020.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zipfel P.F., Wiech T., Rudnick R., Fonso S., Person F., Skerka C. Complement inhibitors in clinical trials for glomerular diseases. Front Immunol. 2019;10:2166. doi: 10.3389/fimmu.2019.02166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pelletier K., Bonnefoy A., Chapdelaine H., et al. Clinical value of complement activation biomarkers in overt diabetic nephropathy. Kidney Int Rep. 2019;4:797–805. doi: 10.1016/j.ekir.2019.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kidney Disease: Improving Global Outcomes Glomerular Diseases Work Group KDIGO 2021 clinical practice guideline for the management of glomerular diseases. Kidney Int. 2021;100:S1–S276. doi: 10.1016/j.kint.2021.05.021. [DOI] [PubMed] [Google Scholar]
- 36.Camilla R., Brachemi S., Pichette V., et al. Urinary monocyte chemotactic protein 1: marker of renal function decline in diabetic and nondiabetic proteinuric renal disease. J Nephrol. 2011;24:60–67. doi: 10.5301/jn.2010.1458. [DOI] [PubMed] [Google Scholar]
- 37.Khalili M., Bonnefoy A., Genest D.S., Quadri J., Rioux J.P., Troyanov S. Clinical use of complement, inflammation, and fibrosis biomarkers in autoimmune glomerulonephritis. Kidney Int Rep. 2020;5:1690–1699. doi: 10.1016/j.ekir.2020.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tam F.W.K., Ong A.C.M. Renal monocyte chemoattractant protein-1: an emerging universal biomarker and therapeutic target for kidney diseases? Nephrol Dial Transplant. 2020;35:198–203. doi: 10.1093/ndt/gfz082. [DOI] [PubMed] [Google Scholar]
- 39.Goumenos D.S., Tsakas S., El Nahas A.M., et al. Transforming growth factor-beta(1) in the kidney and urine of patients with glomerular disease and proteinuria. Nephrol Dial Transplant. 2002;17:2145–2152. doi: 10.1093/ndt/17.12.2145. [DOI] [PubMed] [Google Scholar]
- 40.Xiao H., Schreiber A., Heeringa P., Falk R.J., Jennette J.C. Alternative complement pathway in the pathogenesis of disease mediated by anti-neutrophil cytoplasmic autoantibodies. Am J Pathol. 2007;170:52–64. doi: 10.2353/ajpath.2007.060573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sekine H., Kinser T.T., Qiao F., et al. The benefit of targeted and selective inhibition of the alternative complement pathway for modulating autoimmunity and renal disease in MRL/lpr mice. Arthritis Rheum. 2011;63:1076–1085. doi: 10.1002/art.30222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lenderink A.M., Liegel K., Ljubanovic D., et al. The alternative pathway of complement is activated in the glomeruli and tubulointerstitium of mice with adriamycin nephropathy. Am J Physiol Ren Physiol. 2007;293:F555–F564. doi: 10.1152/ajprenal.00403.2006. [DOI] [PubMed] [Google Scholar]
- 43.Machida T., Sakamoto N., Ishida Y., Takahashi M., Fujita T., Sekine H. Essential roles for mannose-binding lectin-associated serine protease-1/3 in the development of lupus-like glomerulonephritis in MRL/lpr mice. Front Immunol. 2018;9:1191. doi: 10.3389/fimmu.2018.01191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Botto M., Dell’Agnola C., Bygrave A.E., et al. Homozygous C1q deficiency causes glomerulonephritis associated with multiple apoptotic bodies. Nat Genet. 1998;19:56–59. doi: 10.1038/ng0598-56. [DOI] [PubMed] [Google Scholar]
- 45.Einav S., Pozdnyakova O.O., Ma M., Carroll M.C. Complement C4 is protective for lupus disease independent of C3. J Immunol. 2002;168:1036–1041. doi: 10.4049/jimmunol.168.3.1036. [DOI] [PubMed] [Google Scholar]
- 46.Dobó J., Kocsis A., Gál P. Be on target: strategies of targeting alternative and lectin pathway components in complement-mediated diseases. Front Immunol. 2018;9:1851. doi: 10.3389/fimmu.2018.01851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Murphy K., Weaver C. Garland Science; 2016. Janeway’s Immunobiology. [Google Scholar]
- 48.Li J., Chen B., Gao C., et al. Clinical and pathological features of idiopathic membranous nephropathy with focal segmental sclerosis. BMC Nephrol. 2019;20:467. doi: 10.1186/s12882-019-1641-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ogrodowski J.L., Hebert L.A., Sedmak D., et al. Measurement of SC5b-9 in urine in patients with the nephrotic syndrome. Kidney Int. 1991;40:1141–1147. doi: 10.1038/ki.1991.326. [DOI] [PubMed] [Google Scholar]
- 50.Tsakas S., Goumenos D.S. Accurate measurement and clinical significance of urinary transforming growth factor-beta1. Am J Nephrol. 2006;26:186–193. doi: 10.1159/000093178. [DOI] [PubMed] [Google Scholar]
- 51.Gao L., Qiu W., Wang Y., Xu W., Xu J., Tong J. Sublytic complement C5b-9 complexes induce thrombospondin-1 production in rat glomerular mesangial cells via PI3-k/Akt: association with activation of latent transforming growth factor-beta1. Clin Exp Immunol. 2006;144:326–334. doi: 10.1111/j.1365-2249.2006.03069.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bao L., Zhou J., Holers V.M., Quigg R.J. Excessive matrix accumulation in the kidneys of MRL/lpr lupus mice is dependent on complement activation. J Am Soc Nephrol. 2003;14:2516–2525. doi: 10.1097/01.asn.0000089831.96794.0b. [DOI] [PubMed] [Google Scholar]
- 53.Alghadban S., Kenawy H.I., Dudler T., et al. Absence of the lectin activation pathway of complement ameliorates proteinuria-induced renal injury. Front Immunol. 2019;10:2238. doi: 10.3389/fimmu.2019.02238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Dumont C., Merouani A., Ducruet T., et al. Clinical relevance of membrane attack complex deposition in children with IgA nephropathy and Henoch-Schonlein purpura. Pediatr Nephrol. 2020;35:843–850. doi: 10.1007/s00467-019-04445-x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.