Abstract
Sepsis is a leading cause of death in critically ill patients, primarily due to multiple organ failures. It is associated with a systemic inflammatory response that plays a role in the pathogenesis of the disease. Intestinal barrier dysfunction and bacterial translocation (BT) play pivotal roles in the pathogenesis of sepsis and associated organ failure. In this review, we describe recent advances in understanding the mechanisms by which the gut microbiome and BT contribute to the pathogenesis of sepsis. We also discuss several potential treatment modalities that target the microbiome as therapeutic tools for patients with sepsis.
Keywords: bacterial translocation, dysbiosis, gut microbiome, sepsis
Introduction
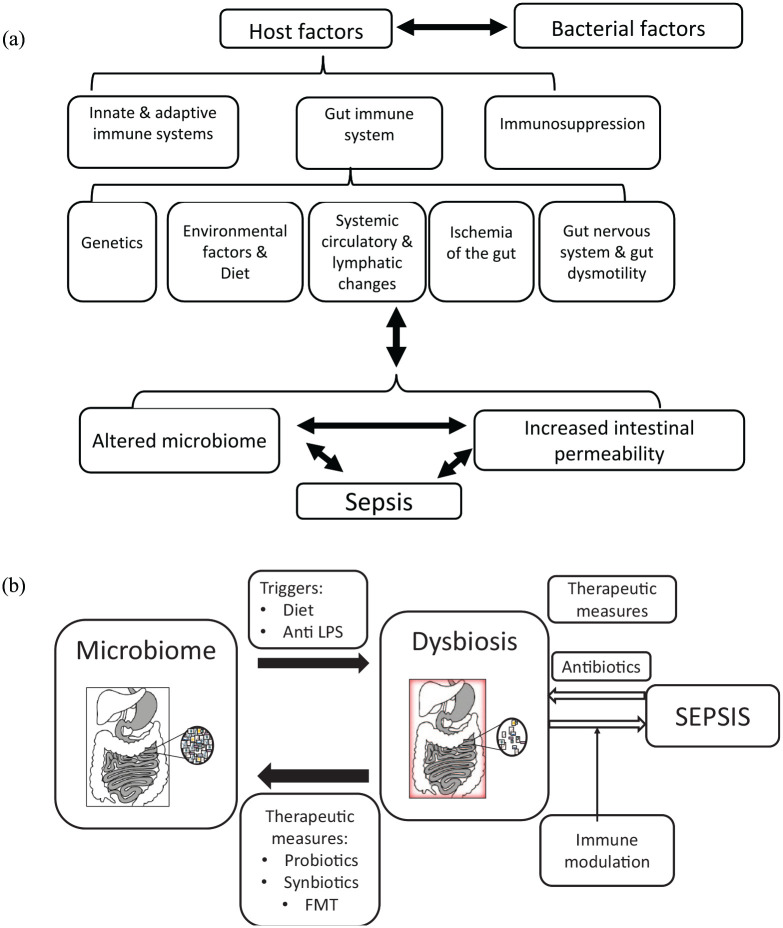
Sepsis is a life-threatening organ dysfunction associated with a dysregulated host response to infection. 1 It is a leading cause of death due to multiple organ failure (MOF) in critically ill patients. 2 Sepsis affects 300 million people annually and is a cause of death in more than 200,000 patients, making it the tenth most common cause of death in the United States. 3 The gut microbiome is associated with the pathogenesis of sepsis. Figure 1(a) shows a schematic diagram of several variables involved in the mechanism underlying sepsis development. Both bacteria-associated and host-related factors contribute to the development of gut barrier dysfunction and bacterial translocation (BT) in sepsis.4–7 In this review, we describe some of the recent advances in understanding the mechanisms through which the gut microbiome contributes to the pathogenesis of sepsis and discuss potential therapeutic measures for sepsis that target the microbiota.
Figure 1.
(a) Variables associated with the development of sepsis. Bacterial factors, including bacteria and host factors, include age, ethnicity, the immune system, diet, background diseases, and medications. Each factor represents a potential target for therapeutic interventions. The roles of dysbiosis and BT in the pathogenesis of sepsis are highlighted. (b) A schematic presentation of several potential therapeutic measures for sepsis, focusing on gut dysbiosis. A shift between a normal/healthy/low sepsis risk microbiome and an altered microbiome and a sepsis microbiome or that the altered microbiome (‘dysbiosis’) increases the risk of sepsis.
Dysbiosis is an imbalance of the gut microbiota in which the composition or function of the normal bacteria colonizing the gut is disrupted. 8
The cause of dysbiosis is multifactorial and involves a combination of genetic, dietary, stress, and disease factors (Figure 1(a)). Several genetic disorders were described in association with dysbiosis.9–11
Dysbiosis is associated with an impaired ability to maintain mucosal membrane function, contributing to systemic inflammation. 12 When dysbiosis occurs, toxins, bacterial endotoxins, bacteria, or debris can leak from the gut, along with food particles containing proteotoxins, such as gluten, casein, and zein, and heat-induced molecules, such as advanced glycation end products and advanced lipoxidation end products. 12
BT in sepsis
The gut is a source of systemic infection in critically ill patients due to increased BT associated with compromised barriers. Intestinal bacteria can be disseminated systemically via BT, moving through the mesenteric lymph nodes (MLNs) or activating the gut immune system.13,14 The intestinal barrier and BT dysfunction are involved in MOF, a complication of sepsis due to activation of the immune system and the secretion of pro-inflammatory cytokines.15–18 In BT, bacteria move through transcellular passages between enterocytes under the control of membrane pumps and via paracellular pathways due to the disruption of tight junctions.19,18 These mechanisms are potentiated by the intestinal barrier dysfunction, which is composed of physical, biochemical, and immunological factors. Endotoxins and antigens are transported from the gut into the circulation.20–22
Gut-origin sepsis is a process in which bacteria and bacteria-associated products incite a systemic response. These systemic responses are responsible for other clinical manifestations in critically ill patients with syndromes, such as acute respiratory distress syndrome (ARDS) and multiple organ dysfunction syndrome (MODS).23–25
BT can induce an inflammatory response, resulting in cytokine-mediated systemic inflammatory response syndrome (SIRS) and MODS. 15 The mechanisms associated with the development of SIRS and MODS are linked with bacterial mediators leaving the gut termed ‘danger particles’ or pathogen-associated molecular patterns (PAMPs). PAMPs stimulate the innate immune system. 26
Both viable bacteria and PAMPs can translocate and exert immunomodulatory effects that lead to sepsis.27–33 These factors lead to increased expression of nuclear factor kappa light-chain enhancer of activated B cell (NF-κB)-associated inflammatory transcription factors, 34 and the excess production of inflammatory cytokines associated with end-organ damage.35,36
Dysbiosis: malfunctioning of the gut microbiota and bacteria-dependent factors that determine virulence
Patients in the early stages of sepsis manifest differences in their microbiome composition.12,37 These patients show lower counts of Bifidobacterium and Lactobacillus species, which protect the normal gut microbiota, and higher counts of pathogenic facultative anaerobes, such as Staphylococcus sp. and Pseudomonas aeruginosa sp. 38 Similar changes have been described in patients post laparotomy. 38 Mice injected intraperitoneal with a fecal solution collected from C57BL/6 (B6) cecum developed polymicrobial sepsis, with a high mortality rate and significant bacterial dissemination. Hepatic dysfunction and systemic pro-inflammatory responses were noted, and their cytokine profiles, among them interleukin (IL)-1β, IL-6, and tumor necrosis factor-alpha (TNF-α), were correlated with the severity of sepsis. 39
Gram-negative bacteria release lipopolysaccharide (LPS) and other virulence factors that induce an inflammatory response. 40 They trigger the translocation of NF-κB to the nucleus, resulting in the upregulation of target adhesion molecules and cytokines, including IL-6, leading to leukocyte recruitment via increased E-selectin and intercellular adhesion molecules. 40 Immunogenic bacterial proteins, including proteins involved in cell adhesion, fimbria, oxidoreductase activity, proteolysis, antimicrobial resistance, and ion transport, were expressed in humans with Salmonella Paratyphi bacteremia and sepsis. 41
The interplay between the gut microbiome and the gut immune system
Sepsis and MOF result from recognizing virulent species by the innate immune system. 42 In sepsis, viable and non-viable pathological bacterial products translocate and promote a self-perpetuating circle of dysfunctional immune activation and systemic inflammation. 43
The innate immune system plays a role in regulating the composition of the microbiome, 44 and BT occurs more frequently in immunocompromised patients. Immune deficiencies and immunosuppressive therapies are associated with quantitative and qualitative changes in the intestinal microbiota, mucosal barrier permeability, and BT.18,45 Bloodstream infections that occur in patients with febrile neutropenia following cytotoxic chemotherapy are caused by BT. 46 Common gastrointestinal organisms are more prevalent in patients with neutropenia, suggesting that the BT of gut organisms causes central line-associated bloodstream infections in the setting of neutropenia.
In an animal model of sepsis, death was linked to a component of the innate immune system, the node-like receptor (NLR) apoptosis-inhibitory protein (Naip) 5-NLR4 (Naip5-Nlrc4) inflammasome. 42 The immune system is activated via pathways involving toll-like receptors (TLRs) and NF-κB. TLRs are transmembrane proteins that recognize microbial pathogens via the motifs mentioned above and invoke an intracellular signaling cascade that culminates in the activation of macrophages and the secretion of pro-inflammatory cytokines, promoting MODS. 47
In individuals with an impaired gastrointestinal barrier, LPS stimulates inflammatory reactions involved in the pathogenesis of sepsis.48,49 LPS originating in the intestinal lumen reaches the MLNs and circulatory system, causing endotoxemia, which leads to systemic inflammation.50,51 Immune suppression via a process in the MLNs affects the level of LPS in the abdominal aortic blood, thereby determining the degree of endotoxin translocation. 50 LPS can be eliminated in humans by inactivating LPS via LPS-binding molecules, enzymes that degrade LPS, or modifying the LPS-responding target cells.48,52,53 Primary or secondary defects in any of these can induce SIRS.
In mice with cecal ligation and perforation (CLP), a model of polymicrobial sepsis, perforation of the cecum allows the release of fecal material into the peritoneal cavity to generate an exacerbated immune response. In this model, LPS was positively correlated with the percentage of regulatory T lymphocytes and negatively correlated with the ratio between pro- and anti-inflammatory cells. 50 LPS altered the mechanisms related to defective hepatic ammonia detoxification in sepsis. 54 In LPS-treated rats, expression of the hepatocyte mitochondrial aquaporin-8 channel, the ammonia permeability of the inner mitochondrial membranes of the liver, and basal and glucagon-induced ureagenesis from ammonia were downregulated. 54
Within hours, the SIRS in sepsis shifts to an adaptive anti-inflammatory state with immunosuppression. 55 This anti-inflammatory phenotype is characterized by diminished expression of pro-inflammatory cytokine genes in response to TLR stimulation with bacterial LPS. 55 Translation of TNFα and IL-6 mRNA in endotoxin-adapted human monocytes is suppressed by a microRNA (miRNA)-based mechanism. TLR4-induced miRNA-146 modifies the subcellular localization of RNA-binding motif protein 4 (RBM4), resulting in the assembly of a translation-suppressor complex that disrupts the synthesis of TNFα and IL-6. Knockout of miRNA-146a results in serine-309 phosphorylation of RBM4 and its nuclear re-localization, which restores the TLR4-dependent synthesis of TNFα and IL-6, suggesting that miRNA-146a has a role in limiting an excessive acute inflammatory reaction. 55
Matrix metalloproteinase 7 (MMP7) is associated with activating alpha-defensins and broad-spectrum antimicrobial, anti-inflammatory peptides produced by Paneth cells in the gut. 49 MMP7 protects mice from LPS-induced intestinal permeability and mortality. LPS induces the activation of MMP7 in the small intestine, degranulation of Paneth cells, and promotes intestinal permeability. MMP7(–/–) mice are resistant to LPS-induced death, which is correlated with reduced cytokine levels and a decrease in LPS-induced BT to MLNs. 56 Alpha-defensins stimulate IL-6 release from macrophages and ileum explants in a TLR4-independent manner. 56
Follistatin-like protein 1 (FSTL-1) is overexpressed under inflammatory conditions characterized by elevated IL-1β. 57 FSTL-1 activates the mitochondrial electron transport chain increasing adenosine triphosphate (ATP) production.It is an essential potentiator of the NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome. 57 Serum concentrations of FSTL-1 are increased in patients with bacterial sepsis and mice administered LPS. 57
Studies have shown that the microbiome affects the immune system. Dysbiosis was shown to be associated with elevated plasma endotoxin levels and a higher rate of BT in the liver in rats with acute rejection following orthotropic liver transplantation. 45 Acute rejection is accompanied by a shift in gut microbiota composition toward Bacteroides and Ruminococcus. Bacteremia is a frequent complication of allogeneic hematopoietic stem cell transplantation. 58 These patients develop a shift in the bacterial populations of the gut and reduced microbial diversity. Commonly encountered organisms, including Enterococcus, Streptococcus, and Proteobacteria, account for more than 30% of the microbiota. Enterococcal domination increased the risk of vancomycin-resistant Enterococcus bacteremia, and Proteobacterial domination increased the risk of Gram-negative rod bacteremia. 58 The antibiotic treatment further affects the microbiota and the associated risk for sepsis. The risk of sepsis within 90 days after discharge from a previous hospital stay by type of antibiotic received during the previous stay was recently determined. The study showed that the risk of sepsis-associated exposure to our high-risk antibiotics was 65% higher than those without antibiotic exposure and was higher with increased quantities of antibiotics during hospitalization. 59
The interplay between the gut microbiome and the gut-associated nervous system affects BT
The enteric nervous system (ENS), which comprises multiple small ganglia that operate discretely but maintain the ability to communicate with each other to form the myenteric and submucosal neural plexuses, 60 is crucial for the proper functioning of the intestine. Enteric neurons express TLRs, suggesting that bacterial metabolites impact the ENS. Data from studies of neurodegenerative diseases and their relationship with the ENS and microbiome showed that the ENS functions like a relay system between the immune system, brain, and microbiome. In addition, the microbiome was shown to support the postnatal establishment of the ENS and play a role in maintaining homeostasis. 61
Altered gut motility contributes to BT, and intestinal obstruction and ileus are risk factors for BT.16,62 Patients with diseases that affect the ENS, such as Hirschsprung’s disease, showed increased rates of BT. 63 Multidirectional signaling between different components in the gut wall, spinal cord, and central nervous system impacts inflammation 2 . The interplay among the epithelial cells, mast cells, residential macrophages, glial cells, neurons, and smooth muscle cells within the gut wall involves intracellular signaling pathways, TLRs, and neuroactive substances, such as nitric oxide, prostaglandins, cytokines, chemokines, growth factors, tryptases, and hormones. 2
Intestinal ischemia and hyperdynamic circulation are associated with low systemic vascular resistance and altered gut permeability
Primary or secondary circulatory changes are associated with BT. During intestinal ischemia, inorganic phosphate, urea, and threonic acid were upregulated, while stearic acid, arabinose, xylose, glucose, and ribose acid were downregulated. 64 These molecular changes resulted from decreased gut microbiota metabolism, altered intestinal absorption, impaired renal function, and increased oxidative stress, which affected gut permeability. 64
Sepsis and cirrhosis are characterized by a hyperdynamic state of low systemic vascular resistance and the release of multiple mediators. 65 The similarity between the two conditions is related to the translocation of common bacterial products. 65 Permeability changes and BT influence intra-abdominal hypertension (IAH), 66 IAH-induced enterogenic endotoxemia, and subsequent MOF66,67 and alter the histology of the colonic mucosa, the expression of tight junction proteins, mucosal permeability, and the pro-oxidant–antioxidant balance. Acute exposure to elevated intra-abdominal pressure alters intestinal permeability and the pro-oxidant-antioxidant balance, intestinal mucosal injury, and subsequent gut-derived sepsis. 67 The gut-lymph hypothesis of MODS suggests that it occurs in high-risk patients due to gut injury and systemic spread of non-microbial, tissue-injurious factors that reach the systemic circulation via the intestinal lymphatics. 32
External factors impacting the gut microbiome
i. Various environmental insults alter the gut microbiome. 68 Age, ethnicity, the immune system, and diet also regulate the intestinal microbiota. 69 Mortality due to sepsis is linked to weak immune defense systems and an artificial environment associated with mechanical ventilation and the use of tubes, drains, intravascular lines, artificial nutrition, and extensive synthetic chemical drugs. These factors can reduce or eliminate the gut microbiota, impair immune function, increase systemic inflammation, and contribute to poor outcomes.3,12
ii. The ‘gut-origin sepsis’ hypothesis evolves from the notion that under stress, such as sepsis, trauma, burn, or shock, infectious complications originate from the gut microbiota.68,70,71 Following surgical trauma, dysregulated metabolic reactions and infections can occur, including hyperglycemia, insulin resistance, increased hepatic glucose production, and muscle protein breakdown, and these changes can become self-destructive, leading to further metabolic damage. 72 In survivors of significant burns, the number of beneficial bacteria, such as obligate anaerobes and Bifidobacterium, initially decreased but increased as their condition improved. 70 In contrast, in non-survivors, these bacteria decreased as gut failure and sepsis progressed. Several pathogenic bacteria, such as Pseudomonas aeruginosa, and fungi, such as Candida, increased only in non-survivors, whereas short-chain fatty acids, such as propionic and butyric acids, decreased. 70 Intestinal surgery in children disrupts the normal intestinal microbiota and barrier function, predisposing them to SIRS. 73
iii. Dietary patterns, including diets containing high fructose and high fat, and nutritional deficiencies affect intestinal permeability. 74 A western-style diet contributes to endotoxemia by causing changes in the intestinal barrier and altering the composition of the microbiota. 75 Approximately, 75% of the food consumed in Western diets does not benefit the microbiota of the lower gut. Much of the Western diet include refined carbohydrates, which are absorbed in the upper part of the GI tract. Food that reaches the large intestine is of limited value and contains fewer minerals, vitamins, and other nutrients essential for maintaining the microbiota. 12 Such a diet can lead to a microbiome of reduced size and diversit. 12
There are characteristic differences in the composition and activity of the gut microbiota between lean and obese individuals. 76 In obese people, a high-fat diet contributes to low-grade inflammation and the development of non-alcoholic fatty liver disease.77,78 In obese animal models caused by feeding either a high-fructose or high-fat diet, increased gut permeability, low-grade endotoxemia, and fatty liver were observed. 77 Germ-free mice did not develop obesity and liver damage. Following a fat diet, LPS can translocate from the gut into the circulatory system via direct diffusion due to increased gut permeability or be absorbed by enterocytes. 75 Obese patients with high postprandial hypertriglyceridemia increased serum LPS levels and chylomicron fraction following fat overload. 78 Postprandial LPS levels correlate with triglyceride levels. LPS binding to TLR4 activates an inflammatory cascade that alters insulin signaling. Chronic exposure to LPS contributes to weight gain and the development of type 2 diabetes mellitus (T2DM), 79 and obese and diabetic individuals had increased serum LPS levels. An increase in the number of gram-negative bacteria in the gut microbiota reduced gut mucosal integrity, and consumption of a high-fat diet increased serum LPS levels. 79
iv. The gastrointestinal tract is affected by antibiotics. Antibiotic treatment damages several mechanisms related to maintaining the gut barrier and homeostasis. In animals, antibiotic use created histopathological lesions, such as desquamation and epithelial tissue destruction. 80 Prior antibiotic exposure was associated with increased mortality in Gram-negative bacteria-induced sepsis. 81 The bacterial communities inhabiting the gut exist in a delicate balance, and antibiotics damage this balance by eliminating several key communities and promoting the colonization of antibiotic-resistant bacteria by exerting stress and eliminating sensitive strains. 82 Antibiotic treatment is associated with intestinal colonization and dominance of orally acquired antibiotic-resistant E. coli. 83 The spread of antibiotic-resistant bacteria has been attributed to impaired innate mucosal defenses caused by antibiotics. 84 Antibiotics modify the host immune system by altering the gut bacterial metabolites used for signal transmission from the microbiome to the gut immune system. Short-chain fatty acids (SCFAs), produced by bacteria through fermentation, have broad effects on enterocytes and play roles in various processes, including maintaining epithelial integrity, Treg differentiation, and the inflammatory response. 84 Antibiotics hinder this.
The commensal microbiota is essential for keeping an intact intestinal barrier and for gut expression of non-defensin proteins. The stimulation of TLR promotes the expression of non-defensin proteins and can reverse the antibiotic-related reduction of gut defense. 81 Antibiotic-associated dysbiosis of the gut predisposes subjects to infections. It also underlies the systemic dissemination of antibiotic-resistant and commensal enterobacteria by promoting transcytosis across the layers of the gut epithelium. 83
Antibiotic treatment reduced the bactericidal-killing activity of the gut mucosa. It also amplified the translocation of Klebsiella pneumoniae and lowered the expression of non-defensin proteins. 81 TLR stimulation following antibiotic treatment increased the binding activity of NF-κB to DNA and the bactericidal activity in the gut mucosa. 81 Germ-free mice showed significant decreases in non-defensin proteins and the intestinal defense against pathogen translocation. 81
Therapeutic measures that target the gut microbiome
Most systemic effects arising from dysbiosis are attributed to the metabolites produced by commensal gut bacteria, which are essential for immune system function. Despite extensive research on this topic, no therapeutic intervention aimed at microbiome alteration has been implemented to treat or prevent sepsis. 85 However, several therapeutic measures to alter the gut microbiome to improve the prognosis of patients with sepsis are being explored. Several measures used for affecting BT and gut dysbiosis are described below.
Figure 1(b) shows a schematic representation of several potential therapeutic measures for sepsis.
Antibiotics
In sepsis, antibiotics act as a double-edged sword. Although they are a cornerstone of sepsis treatment, their effect on the microbiome may have detrimental consequences. 86 Targeted antibiotics that alter the microbiome have been studied. Azithromycin is a macrolide that inhibits bacterial protein synthesis and reduces biofilm formation. This antibiotic accumulates in phagocytes and is delivered at high concentrations to sites of infection. In chronic inflammatory disorders, it was shown to exert immunomodulatory effects on immune and epithelial cells by modulating the NF-κB inflammatory pathway, mucin release, surface receptor expression, macrophage phenotypes, and autophagy. 87 In a murine model of sepsis, azithromycin improved survival and attenuated the levels of various inflammatory cytokines, including IL-6, IL-1, and TNFα. 88 In humans, azithromycin decreased mortality and ventilator dependency in patients with sepsis-associated ARDS. 89 In patients with severe sepsis, azithromycin increased intensive care unit (ICU)-free days. 90
Selective digestive tract decontamination (SDD) and selective oropharyngeal decontamination (SOD) using enteral non-absorbable antimicrobials eradicated gram-negative bacteria, Staphylococcus aureus, and yeast colonization while leaving the anaerobic microbiome intact. 91 Using SDD in ICU settings reduced the incidence of respiratory tract infections, colonization of antibiotic-resistant bacteria, and improved survival.92–94 Despite the evidence of SDD efficacy, multiple studies have fueled controversy, 95 and SDD is not used in clinical practice because it may promote the emergence of antibiotic-resistant strains. 96 However, a Cochrane review suggested that a combination of topical and systemic prophylactic antibiotics reduces overall mortality and respiratory tract infections. 97
Probiotic, prebiotic, and synbiotic treatments
Recovery of the microbiome is essential for preventing long-lasting immune suppression after the initial bout of sepsis has ended. Probiotic, prebiotic, and synbiotic treatments have been explored for maintaining and repairing the gut microbiota. 68 These treatments may prevent and improve the prognosis of patients with sepsis. 98
Probiotics are viable, non-pathogenic microorganisms that confer benefits to the host by modifying the gut microbiota, promoting the local release of antimicrobial factors, maintaining gut barrier integrity, competing for epithelial adherence, preventing BT, and modulating the local immune response. 99 Probiotic-associated changes in the gut microbiota are correlated with disease outcomes. 100 The properties of probiotics are strain-specific, and beneficial effects have been observed for certain strains of Lactobacillus, Bifidobacterium, Saccharomyces, Enterococcus sp., Streptococcus sp., Pediococcus sp., Leuconostoc, Bacillus, and E. coli. 100 Probiotics are well tolerated and are believed to prevent disease progression by competing with native bacteria for nutrients and binding loci, producing bacteriocins that kill pathogens, increasing the production of IgA, improving mucosal immunity, and suppressing systemic inflammation. 98 In an animal pancreatitis model, the host-specific ileal microbiota was replaced with an ‘acute pancreatitis-associated microbiota’, which was reversed by administering probiotics. The microbiome of animals that received probiotics contained an abundance of a bacterial phylotype related to Clostridium lituseburense, which was correlated with reduced bacterial overgrowth, improved pancreatic pathology, and reduced pro-inflammatory cytokine levels. 101
The use of probiotics in critically ill patients has yielded mixed results. Several studies have shown that probiotics are associated with decreases in susceptibility to antibiotic-associated diarrhea, Clostridium difficile, ventilator-associated pneumonia, necrotizing enterocolitis (NEC), sepsis, MOF syndrome, and a shortened infection duration. 99 However, a meta-analysis did not show a beneficial effect on mortality or length of ICU stay. 102 Probiotic use in immunocompromised patients or patients with a leaky gut was associated with increased fungemia and bacteremia. 85 The use of Lactobacillus rhamnosus decreased ventilator-associated pneumonia in ICU patients.103,104 Probiotic administration prevented necrotizing enterocolitis (NEC) and decreased mortality in preterm infants; however, the level of evidence was deemed insufficient to recommend it as a routine treatment. 105 A recent large-scale multicenter trial did not show any benefit in using probiotics as a measure for decreasing ventilator-associated pneumonia or other infections among patients in ICU. 106 However, probiotics were beneficial in selected patient populations, requiring an individualized approach for sepsis. 107
Prebiotics are nutrients degraded by the gut microbiota that alter the function and composition of the microbiome. 108 These nutrients are non-digestible carbohydrates fermented by intestinal microbes, resulting in energy transfer. Digestion of these nutrients produces signaling metabolites, such as SCFAs and peptidoglycan, which affect the innate immune system. 109 Prebiotics can reduce inflammation, endotoxemia, and cytokine levels, thereby improving insulin resistance and glucose tolerance. 69 A diet rich in prebiotics is beneficial for managing T2DM for its positive impact on intestinal microbiota modulation. 69 Prebiotics, such as fermentable dietary fiber, increase glucagon-like peptide-1 and peptide YY and decrease ghrelin levels. In patients with T2DM, a macrobiotic diet improved fasting blood glucose, plasma lipid fractions, and plasma insulin levels. Different prebiotic molecules were tested in mouse models of sepsis, with positive results. Desaminotyrosine (DAT) maintained mucosal immune homeostasis and protected against barrier integrity. DAT attenuated dextran sodium sulfate-induced mucosal inflammation, and it protected mice treated with endotoxin to induce septic shock. 110 In an animal model, Xuanbai Chengqi decoction (XBCQ), a traditional Chinese medicine formulation, was protective against pulmonary infections, improved gut barrier function by increasing the amounts of occludin, a mucosal protein, improved the diversity of the gut microbiota, attenuated inflammatory markers in the lungs, and improved survival. 111 Finger millet arabinoxylan (FM–AX), a non-starch polysaccharide extracted from cereals, prevented metagenomic alterations in the cecum and endotoxemia in mice fed a high-fat diet. 112 In preterm infants, prebiotics decreased the incidence of sepsis, mortality, and length of hospital stay. 113
Synbiotic therapy employs a combination of probiotics and prebiotics and reduces septic complications. 68 Perioperative use of synbiotics reduced the incidence of bacteria in MLNs and blood. 114 Synbiotic nutrition reduced the incidence of postoperative sepsis in an elective general surgery setting. 115 A randomized controlled study in mechanically ventilated patients with sepsis showed that synbiotic treatment reduced ventilation-associated pneumonia but had no effect on mortality. 116 A meta-analysis of 13 randomized controlled trials including 962 patients who received synbiotics or probiotics showed a reduced incidence of postoperative sepsis in both groups. Subgroup analysis did not detect a reduction in the occurrence of urinary tract infections, pneumonia, or wound infections following surgery. 115 A randomized trial evaluating a synbiotic preparation of Lactobacillus plantarum and fructooligosaccharide in 4,500 healthy term infants showed a 40% decrease in the composite outcome of lower respiratory tract infection, sepsis, and death. 117
Several factors may have contributed to the lack of success of these approaches, including an unsuitable choice of probiotic species, low doses, and its use as a complement to antibiotic therapy rather than an alternative treatment. 3 In such approaches, the supplemented lactic acid bacteria were killed before reaching their target organs. At present, the routine use of these methods in critically ill patients is not recommended.14,99
Fecal microbial transplantation
Fecal microbial transplantation (FMT) is a method for altering the gut microbiota that may be superior to other methods due to the delivery of other organisms, viruses, fungi, and other metabolites in addition to commensal bacteria. FMT has been proven efficacious in treating recurrent and refractory Clostridium difficile (CDI) and several other dysbiosis-related conditions. 118 In a rat model of sepsis, FMT reduced mortality by 50%, prevented intestinal injury and mucosal atrophy, had antioxidant and anti-inflammatory effects, and inhibited NF-κB.119,120 Animal models restored bacterial communities in cecal crypts, replenished the gut epithelium, and protected gut stem cells. 98 There are several barriers to using FMT in critically ill patients treated with broad-spectrum antibiotics, as the treatment can inhibit proliferation of the transplanted bacteria.14,121 Therefore, the use of FMT in human subjects to treat sepsis remains anecdotal.
Anti-LPS measures
Endotoxin from gram-negative bacteria, LPS, activates TLRs and stimulates the inflammatory processes in sepsis and other conditions.51–53 Several LPS-binding molecules have been studied for their potential to prevent or alleviate excess inflammatory responses. 98 The role of LPS in gut-derived sepsis was studied in neutropenic rats fed a preparation of P. aeruginosa to induce sepsis. The animals were administered colostrum or hyperimmune colostrum produced by vaccinating pregnant cows against a conserved portion of LPS, which increased the levels of antibodies against LPS. The use of both regular and antibody-enriched colostrum was associated with increased survival and decreased amounts of bacteria in extra-intestinal sites. 122 Anti-LPS antibodies reduce the inflammatory response that induces metabolic disease by altering the ‘leaky’ mucosal membrane and removing endotoxin. 123 Anti-inflammatory LPS (A-LPS), produced by certain microbes, such as Bacteroides, was shown to help prevent and treat LPS-induced inflammatory response by altering the balance between A-LPS and pro-inflammatory LPS (P-LPS). 124 The results of therapeutic interventions aimed at removing LPS by immunoglobulins or extracorporeal means are controversial, 48 and human trials for sepsis have confirmed positive effects on several secondary parameters but not on morbidity or survival. 48
Dietary measures
Dietary adjustment is a relatively simple way of altering the intestinal microbiota. 125 Dietary changes can modulate the composition of the gut microbiota and improve gut mucosal integrity, decrease the occurrence of endotoxemia and postprandial inflammatory effects, and lead to adequate insulin signaling.74,79 Parenteral nutrition, elemental enteral nutrition (EN), and eco-immuno-nutrition (EIN) hastened the recovery of patients with severe pancreatitis and alleviated the associated SIRS.126,127 EN decreased endotoxin, TNFα, IL-6, and BT levels and enhanced IL-10 expression. 127 Pancreatic sepsis, MOF, and mortality were lower in the EN and EIN-treated groups. EN improved the intestinal mucosal barrier, promoted mucosal repair, and stabilized the intestinal microbiome. Early enteral nutrition (EEN) in patients with sepsis inhibited an excessive immune response, shortened the duration of mechanical ventilation, and the hospital stay, but had no effect on 28-day mortality. 128
Dietary fiber has anti-inflammatory properties. Fiber supplementation improved outcomes by inducing microbial changes that regulate inflammatory metabolites. 129 Fiber supplementation in critically ill patients increased the abundance of SCFA-producing bacteria without increasing the occurrence of diarrhea or abdominal distention. 130 Immune cells utilize large amounts of glutamine, most of which derives from the degradation of muscle protein. 131 In a model using samples from healthy children, glutamine promoted HSP70 and TNFα release. 132 The effects depend on the degree of inflammation, and glutamine administration to patients with sepsis enhanced immune competence and improved the catabolic phase of septic patients with malnutrition.131,133
Immune modulatory and anti-inflammatory therapies
Immunomodulatory drugs indirectly affect the gut microbiota, contributing to their beneficial effects on sepsis. 134 In germ-free mice subjected to inflammatory stimulation, mice treated with gut microbiome samples collected from patients treated successfully with methotrexate for rheumatoid arthritis showed a lessor immune response. 135 However, the clinical use of steroids and other immunosuppressive agents in patients with sepsis is controversial.136,137 Immune modulatory therapy with a combination of dexamethasone and thymosin alpha1 altered dendritic cell (DC) function and enhanced the resistance of endotoxemic mice to BT and secondary infections, improving their outcomes. 138 Mice treated with this combination showed improved survival, a decrease in BT to extra-intestinal organs, and an enhanced ability to eradicate secondary infections by reversing changes in DC during endotoxemia. 138 Failure to respond to steroid therapy in sepsis is caused by decreased expression and function of glucocorticoid receptors (GRs), and GR expression and translocation were decreased in septic animals. 139 Leflunomide has immunomodulatory and anti-inflammatory effects, which mitigate the host response to BT, and leflunomide-mediated prevention of protein and lipid peroxidation was observed in septic bowel tissue in rats. 140
Thymic stromal lymphopoietin (TSLP), a member of the IL-2 cytokine family, has multiple effects on immune cells. 141 The short form of TLSP is linked to the homeostasis of the gut barrier in a healthy state. However, its long form is overexpressed in an inflammatory state, whereas the short form is downregulated. 142 Most studies showed a decreased inflammatory response, the clinical outcomes varied. 143
Amino-procalcitonin (NPCT) is a peptide derived from the prohormone procalcitonin. NPCT underlies the pathogenesis of acute lung injury in sepsis and is a risk factor for mortality. Treatment with anti-NPCT reduced the inflammatory cytokine expression during sepsis and prevented the nuclear NF-κB translocation in tissues while increasing the anti-inflammatory cytokine IL-10 in mice. 144 The humanized anti-PcrV IgG antigen-binding fragment KB001 inhibited toxin translocation, with demonstrated safety and a favorable pharmacokinetic profile, and thus has potential as a nonantibiotic strategy to reduce inflammation and damage in P. aeruginosa-related pneumonia. 145
Colchicine, an anti-inflammatory drug, is an inhibitor of mitosis and microtubule assembly.146–149 By binding to soluble non-polymerized tubulin heterodimers, it forms tight colchicine-tubulin complexes, thereby inhibiting the polymerization of microtubules. 150 Microtubules are involved in the function of both adaptive and innate immune systems, making them potential therapeutic targets for immune modulation. 149 Colchicine inhibits microtubule rearrangement and interferes with the function of the immune system. It inhibits the recruitment, migration, and chemotaxis of neutrophils. Colchicine reduces neutrophil deformability by lowering microtubule levels, thus altering the ability of neutrophils to migrate via small pores, which is crucial for extravasation in response to inflammatory signals. 151 The colchicine-tubulin complex attenuates macrophage NLRP inflammasome arrangement and activation. 152
A new artificial intelligence-based system is being developed for analyzing data on the function of microtubules of gut cells and data on microbiota metabolites, ENS, and the gut immune cells. The system will provide a method for improving the response to the currently used measures targeting BT and dysbiosis to reduce the hyperinflammatory response associated with sepsis, similar to other disorders.153–160
Summary
The human gut is a complex organ involved in multiple physiological processes. The gut immune system and microbiota play essential roles in health and disease,51,154,161–167 and their roles in sepsis have been established. BT and several other microbiota-linked mechanisms contribute to sepsis and subsequent MOF development. Bacterial-associated, host and environment-dependent mechanisms are involved in the pathogenesis of sepsis. These findings provide potential new therapeutic approaches for sepsis. In these types of therapies, the microbiome is targeted, directly or indirectly, to restore its homeostatic states, thus reducing the damage caused by the aggravated inflammatory processes that ensue. Since the treatment for sepsis has not changed dramatically in recent years, and the morbidity and mortality rates remain high, there is a need for new therapeutic targets.
Footnotes
Abbreviations: BT: bacterial translocation; MOF: multiple organ failure; MLN: mesenteric lymph nodes; ARDS: acute respiratory distress syndrome; LPS: lipopolysaccharide; SIRS: systemic inflammatory response syndrome; MODS: multiple organ dysfunction syndrome; SBP: spontaneous bacterial translocation; PAMPs pathogen-associated molecular pattern; IL: interleukin; TNFα: tumor necrosis factor-alpha; NF-κB: nuclear factor kappa-light-chain enhancer of activated B cell; TTSS: type III secretion system; OMVs: outer membrane vesicles; TLR: Toll-like receptor; CLP: cecal ligation and perforation; TMED7: transmembrane emp24 protein transport domain containing 7; miRNA: microRNA; MMP7: Matrix metalloproteinase 7; FSTL-1: follistatin-like protein 1; NLRP: nod-like receptor family, pyrin domain containing 3; ENS: enteric nervous system; IAH: intra-abdominal hypertension; SCFA: short-chain fatty acids; EN: enteral nutrition; EIN: eco-immuno-nutrition; HSP: heat shock protein; DC: dendritic cells; GR: glucocorticoid response; NPCT: amino-procalcitonin
Author contributions: Assaf Potruch: Data curation; Writing – original draft; Writing – review & editing.
Asaf Schwartz: Data curation; Writing – original draft; Writing – review & editing.
Yaron Ilan: Conceptualization; Data curation; Validation; Writing – review & editing.
Conflict of interest statement: The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iD: Yaron Ilan  https://orcid.org/0000-0003-0802-1220
https://orcid.org/0000-0003-0802-1220
Contributor Information
Assaf Potruch, Department of Medicine, Faculty of Medicine, Hebrew University and Hadassah Medical Center, Jerusalem, Israel.
Asaf Schwartz, Department of Medicine, Faculty of Medicine, Hebrew University and Hadassah Medical Center, Jerusalem, Israel.
Yaron Ilan, Department of Medicine, Faculty of Medicine, Hebrew University and Hadassah Medical Center, Kiryat Hadassah, P.O.B 12000, Jerusalem IL-91120, Israel.
References
- 1. Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016; 315: 801–810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. De Winter BY, De Man JG. Interplay between inflammation, immune system and neuronal pathways: effect on gastrointestinal motility. World J Gastroenterol 2010; 16: 5523–5535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Bengmark S. Pro- and synbiotics to prevent sepsis in major surgery and severe emergencies. Nutrients 2012; 4: 91–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Lee CC, Feng Y, Yeh YM, et al. Gut dysbiosis, bacterial colonization and translocation, and neonatal sepsis in very-low-birth-weight preterm infants. Front Microbiol 2021; 12: 746111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Doudakmanis C, Bouliaris K, Kolla C, et al. Bacterial translocation in patients undergoing major gastrointestinal surgery and its role in postoperative sepsis. World J Gastrointest Pathophysiol 2021; 12: 106–114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Panpetch W, Chancharoenthana W, Bootdee K, et al. Lactobacillus rhamnosus L34 attenuates gut translocation-induced bacterial sepsis in murine models of leaky gut. Infect Immun 2018; 86: e00700-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Ikeda M, Shimizu K, Ogura H, et al. Hydrogen-rich saline regulates intestinal barrier dysfunction, dysbiosis, and bacterial translocation in a murine model of sepsis. Shock 2018; 50: 640–647. [DOI] [PubMed] [Google Scholar]
- 8. Walker WA. Chapter 25 – dysbiosis. In: Floch MH, Ringel Y, Allan Walker W. (eds) The microbiota in gastrointestinal pathophysiology. Boston, MA: Academic Press, 2017, pp. 227–232. [Google Scholar]
- 9. Alshehri D, Saadah O, Mosli M, et al. Dysbiosis of gut microbiota in inflammatory bowel disease: current therapies and potential for microbiota-modulating therapeutic approaches. Bosn J Basic Med Sci 2021; 21: 270–283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Brown J, Robusto B, Morel L. Intestinal dysbiosis and tryptophan metabolism in autoimmunity. Front Immunol 2020; 11: 1741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Kirby TO, Ochoa-Reparaz J, Roullet JB, et al. Dysbiosis of the intestinal microbiome as a component of pathophysiology in the inborn errors of metabolism. Mol Genet Metab 2021; 132: 1–10. [DOI] [PubMed] [Google Scholar]
- 12. Bengmark S. Nutrition of the critically ill – emphasis on liver and pancreas. Hepatobiliary Surg Nutr 2012; 1: 25–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Nagpal R, Yadav H. Bacterial translocation from the gut to the distant organs: an overview. Ann Nutr Metab 2017; 71(Suppl. 1): 11–16. [DOI] [PubMed] [Google Scholar]
- 14. Ilan Y. Why targeting the microbiome is not so successful: can randomness overcome the adaptation that occurs following gut manipulation? Clin Exp Gastroenterol 2019; 12: 209–217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Puleo F, Arvanitakis M, Van Gossum A, et al. Gut failure in the ICU. Semin Respir Crit Care Med 2011; 32: 626–638. [DOI] [PubMed] [Google Scholar]
- 16. Winslow MM, Gallo EM, Neilson JR, et al. The calcineurin phosphatase complex modulates immunogenic B cell responses. Immunity 2006; 24: 141–152. [DOI] [PubMed] [Google Scholar]
- 17. Berg RD, Garlington AW. Translocation of certain indigenous bacteria from the gastrointestinal tract to the mesenteric lymph nodes and other organs in a gnotobiotic mouse model. Infect Immun 1979; 23: 403–411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Vaishnavi C. Translocation of gut flora and its role in sepsis. Indian J Med Microbiol 2013; 31: 334–342. [DOI] [PubMed] [Google Scholar]
- 19. MacFie J. Current status of bacterial translocation as a cause of surgical sepsis. Br Med Bull 2005; 71: 1–11. [DOI] [PubMed] [Google Scholar]
- 20. O’Boyle CJ, MacFie J, Mitchell CJ, et al. Microbiology of bacterial translocation in humans. Gut 1998; 42: 29–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Sedman PC, Macfie J, Sagar P, et al. The prevalence of gut translocation in humans. Gastroenterology 1994; 107: 643–649. [DOI] [PubMed] [Google Scholar]
- 22. Bauer M. The liver-gut-axis: initiator and responder to sepsis. Curr Opin Crit Care 2022; 28: 216–220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Cuevas P, De la Maza LM, Gilbert J, et al. The lung lesion in four different types of shock in rabbits. Arch Surg 1972; 104: 319–322. [DOI] [PubMed] [Google Scholar]
- 24. Souza DG, Vieira AT, Soares AC, et al. The essential role of the intestinal microbiota in facilitating acute inflammatory responses. J Immunol 2004; 173: 4137–4146. [DOI] [PubMed] [Google Scholar]
- 25. Rush BF, Jr, Redan JA, Flanagan JJ, Jr, et al. Does the bacteremia observed in hemorrhagic shock have clinical significance? A study in germ-free animals. Ann Surg 1989; 210: 342–345; discussion 346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Bianchi ME. DAMPs, PAMPs and alarmins: all we need to know about danger. J Leukoc Biol 2007; 81: 1–5. [DOI] [PubMed] [Google Scholar]
- 27. Anitha M, Vijay-Kumar M, Sitaraman SV, et al. Gut microbial products regulate murine gastrointestinal motility via Toll-like receptor 4 signaling. Gastroenterology 2012; 143: 1006–1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Wells JM, Rossi O, Meijerink M, et al. Epithelial crosstalk at the microbiota-mucosal interface. Proc Natl Acad Sci U S A 2011; 108(Suppl. 1): 4607–4614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Hammer HF. Gut microbiota and inflammatory bowel disease. Dig Dis 2011; 29: 550–553. [DOI] [PubMed] [Google Scholar]
- 30. Damman CJ, Miller SI, Surawicz CM, et al. The microbiome and inflammatory bowel disease: is there a therapeutic role for fecal microbiota transplantation? Am J Gastroenterol 2012; 107: 1452–1459. [DOI] [PubMed] [Google Scholar]
- 31. Iliev ID, Funari VA, Taylor KD, et al. Interactions between commensal fungi and the C-type lectin receptor Dectin-1 influence colitis. Science 2012; 336: 1314–1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Deitch EA. Gut-origin sepsis: evolution of a concept. Surgeon 2012; 10: 350–356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Lamprecht G, Heininger A. [Current aspects of sepsis caused by bacterial translocation]. Zentralbl Chir 2012; 137: 274–278. [DOI] [PubMed] [Google Scholar]
- 34. Ward NS, Casserly B, Ayala A. The compensatory anti-inflammatory response syndrome (CARS) in critically ill patients. Clin Chest Med 2008; 29: 617–625, viii. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Hatakeyama N, Matsuda N. Mechanisms of inflammatory response and organ dysfunction: organ-protective strategy by anesthetics. Curr Pharm Des 2014; 20: 5766–5778. [DOI] [PubMed] [Google Scholar]
- 36. Coldewey SM, Rogazzo M, Collino M, et al. Inhibition of IkappaB kinase reduces the multiple organ dysfunction caused by sepsis in the mouse. Dis Model Mech 2013; 6: 1031–1042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Liu Z, Li N, Fang H, et al. Enteric dysbiosis is associated with sepsis in patients. FASEB J 2019; 33: 12299–12310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Shimizu K, Ogura H, Goto M, et al. Altered gut flora and environment in patients with severe SIRS. J Trauma 2006; 60: 126–133. [DOI] [PubMed] [Google Scholar]
- 39. Shrum B, Anantha RV, Xu SX, et al. A robust scoring system to evaluate sepsis severity in an animal model. BMC Res Notes 2014; 7: 233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Soult MC, Lonergan NE, Shah B, et al. Outer membrane vesicles from pathogenic bacteria initiate an inflammatory response in human endothelial cells. J Surg Res 2013; 184: 458–466. [DOI] [PubMed] [Google Scholar]
- 41. Alam MM, Tsai LL, Rollins SM, et al. Identification of in vivo-induced bacterial proteins during human infection with Salmonella enterica serotype Paratyphi A. Clin Vaccine Immunol 2013; 20: 712–719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Ayres JS, Trinidad NJ, Vance RE. Lethal inflammasome activation by a multidrug-resistant pathobiont upon antibiotic disruption of the microbiota. Nat Med 2012; 18: 799–806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Bruns T, Zimmermann HW, Stallmach A. Risk factors and outcome of bacterial infections in cirrhosis. World J Gastroenterol 2014; 20: 2542–2554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Levy M, Thaiss CA, Elinav E. Metagenomic cross-talk: the regulatory interplay between immunogenomics and the microbiome. Genome Medicine 2015; 7: 1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Xie Y, Luo Z, Li Z, et al. Structural shifts of fecal microbial communities in rats with acute rejection after liver transplantation. Microb Ecol 2012; 64: 546–554. [DOI] [PubMed] [Google Scholar]
- 46. Steinberg JP, Robichaux C, Tejedor SC, et al. Distribution of pathogens in central line-associated bloodstream infections among patients with and without neutropenia following chemotherapy: evidence for a proposed modification to the current surveillance definition. Infect Control Hosp Epidemiol 2013; 34: 171–175. [DOI] [PubMed] [Google Scholar]
- 47. Seki R. Toll-like receptor ligand-induced liver injury in D-galactosamine-sensitized mice: differences between TLR7/8 and TLR9 ligands, cytokine patterns, and cross-tolerance induction by TLR2 ligand pretreatment. J Immunol Res 2017; 2017: 9653793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Buttenschoen K, Radermacher P, Bracht H. Endotoxin elimination in sepsis: physiology and therapeutic application. Langenbecks Arch Surg 2010; 395: 597–605. [DOI] [PubMed] [Google Scholar]
- 49. Pendyala S, Walker JM, Holt PR. A high-fat diet is associated with endotoxemia that originates from the gut. Gastroenterology 2012; 142: 1100–1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Yu W, Du H, Fu Q, et al. The influence of Th1/Th2 and CD4+ regulatory t cells of mesenteric lymph nodes on systemic lipopolysaccharide. Pol J Pathol 2014; 65: 125–129. [DOI] [PubMed] [Google Scholar]
- 51. Ben Ya’acov A, Lichtenstein Y, Zolotarov L, et al. The gut microbiome as a target for regulatory T cell-based immunotherapy: induction of regulatory lymphocytes by oral administration of anti-LPS enriched colostrum alleviates immune mediated colitis. BMC Gastroenterol 2015; 15: 154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Mizrahi M, Shabat Y, Ben Ya’acov A, et al. Alleviation of insulin resistance and liver damage by oral administration of Imm124-E is mediated by increased Tregs and associated with increased serum GLP-1 and adiponectin: results of a phase I/II clinical trial in NASH. J Inflamm Res 2012; 5: 141–150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Adar T, Ben Ya’acov A, Lalazar G, et al. Oral administration of immunoglobulin G-enhanced colostrum alleviates insulin resistance and liver injury and is associated with alterations in natural killer T cells. Clin Exp Immunol 2012; 167: 252–260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Soria LR, Marrone J, Molinas SM, et al. Lipopolysaccharide impairs hepatocyte ureagenesis from ammonia: involvement of mitochondrial aquaporin-8. FEBS Lett 2014; 588: 1686–1691. [DOI] [PubMed] [Google Scholar]
- 55. Brudecki L, Ferguson DA, McCall CE, et al. MicroRNA-146a and RBM4 form a negative feed-forward loop that disrupts cytokine mRNA translation following TLR4 responses in human THP-1 monocytes. Immunol Cell Biol 2013; 91: 532–540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Vandenbroucke RE, Vanlaere I, Van Hauwermeiren F, et al. Pro-inflammatory effects of matrix metalloproteinase 7 in acute inflammation. Mucosal Immunol 2014; 7: 579–588. [DOI] [PubMed] [Google Scholar]
- 57. Chaly Y, Fu Y, Marinov A, et al. Follistatin-like protein 1 enhances NLRP3 inflammasome-mediated IL-1beta secretion from monocytes and macrophages. Eur J Immunol 2014; 44: 1467–1479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Taur Y, Xavier JB, Lipuma L, et al. Intestinal domination and the risk of bacteremia in patients undergoing allogeneic hematopoietic stem cell transplantation. Clin Infect Dis 2012; 55: 905–914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Baggs J, Jernigan JA, Halpin AL, et al. Risk of subsequent sepsis within 90 days after a hospital stay by type of antibiotic exposure. Clin Infect Dis 2018; 66: 1004–1012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Furness JB. The enteric nervous system and neurogastroenterology. Nat Rev Gastroenterol Hepatol 2012; 9: 286–294. [DOI] [PubMed] [Google Scholar]
- 61. Kabouridis PS, Lasrado R, McCallum S, et al. Microbiota controls the homeostasis of glial cells in the gut lamina propria. Neuron 2015; 85: 289–295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Sen V, Uluca U, Ece A, et al. Role of Ankaferd on bacterial translocation and inflammatory response in an experimental rat model of intestinal obstruction. Int J Clin Exp Med 2014; 7: 2677–2686. [PMC free article] [PubMed] [Google Scholar]
- 63. Hagl CI, Klotz M, Wink E, et al. Temporal and regional morphological differences as a consequence of FGF-2 deficiency are mirrored in the myenteric proteome. Pediatr Surg Int 2008; 24: 49–60. [DOI] [PubMed] [Google Scholar]
- 64. Fahrner R, Beyoğlu D, Beldi G, et al. Metabolomic markers for intestinal ischemia in a mouse model. J Surg Res 2012; 178: 879–887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Vincent JL, Gustot T. Sepsis and cirrhosis: many similarities. Acta Gastroenterol Belg 2010; 73: 472–478. [PubMed] [Google Scholar]
- 66. Stegall MD, Raghavaiah S, Gloor JM. The (re)emergence of B cells in organ transplantation. Curr Opin Organ Transplant 2010; 15: 451–455. [DOI] [PubMed] [Google Scholar]
- 67. Leng Y, Zhang K, Fan J, et al. Effect of acute, slightly increased intra-abdominal pressure on intestinal permeability and oxidative stress in a rat model. PLoS ONE 2014; 9: e109350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Shimizu K, Ogura H, Asahara T, et al. Probiotic/synbiotic therapy for treating critically ill patients from a gut microbiota perspective. Dig Dis Sci 2013; 58: 23–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Fallucca F, Porrata C, Fallucca S, et al. Influence of diet on gut microbiota, inflammation and type 2 diabetes mellitus. First experience with macrobiotic Ma-Pi 2 diet. Diabetes Metab Res Rev 2014; 30(Suppl. 1): 48–54. [DOI] [PubMed] [Google Scholar]
- 70. Shimizu K, Ogura H, Asahara T, et al. Gut microbiota and environment in patients with major burns – a preliminary report. Burns 2015; 41: e28–e33. [DOI] [PubMed] [Google Scholar]
- 71. Correia MI, Liboredo JC, Consoli ML. The role of probiotics in gastrointestinal surgery. Nutrition 2012; 28: 230–234. [DOI] [PubMed] [Google Scholar]
- 72. Hartl WH, Jauch KW. Metabolic self-destruction in critically ill patients: origins, mechanisms and therapeutic principles. Nutrition 2014; 30: 261–267. [DOI] [PubMed] [Google Scholar]
- 73. Chawla BK, Teitelbaum DH. Profound systemic inflammatory response syndrome following non-emergent intestinal surgery in children. J Pediatr Surg 2013; 48: 1936–1940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Teixeira TF, Collado MC, Ferreira CL, et al. Potential mechanisms for the emerging link between obesity and increased intestinal permeability. Nutr Res 2012; 32: 637–647. [DOI] [PubMed] [Google Scholar]
- 75. Moreira AP, Texeira TF, Ferreira AB, et al. Influence of a high-fat diet on gut microbiota, intestinal permeability and metabolic endotoxaemia. Br J Nutr 2012; 108: 801–809. [DOI] [PubMed] [Google Scholar]
- 76. Molinaro F, Paschetta E, Cassader M, et al. Probiotics, prebiotics, energy balance, and obesity: mechanistic insights and therapeutic implications. Gastroenterol Clin North Am 2012; 41: 843–854. [DOI] [PubMed] [Google Scholar]
- 77. Frazier TH, DiBaise JK, McClain CJ. Gut microbiota, intestinal permeability, obesity-induced inflammation, and liver injury. JPEN J Parenter Enteral Nutr 2011; 35(5 Suppl.): 14S–20S. [DOI] [PubMed] [Google Scholar]
- 78. Clemente-Postigo M, Queipo-Ortuño MI, Murri M, et al. Endotoxin increase after fat overload is related to postprandial hypertriglyceridemia in morbidly obese patients. J Lipid Res 2012; 53: 973–978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Boroni Moreira AP, de Cássia Gonçalves Alfenas R. The influence of endotoxemia on the molecular mechanisms of insulin resistance. Nutr Hosp 2012; 27: 382–390. [DOI] [PubMed] [Google Scholar]
- 80. Shi Y, Zhai Q, Li D, et al. Restoration of cefixime-induced gut microbiota changes by Lactobacillus cocktails and fructooligosaccharides in a mouse model. Microbiol Res 2017; 200: 14–24. [DOI] [PubMed] [Google Scholar]
- 81. Wu YY, Hsu CM, Chen PH, et al. Toll-like receptor stimulation induces nondefensin protein expression and reverses antibiotic-induced gut defense impairment. Infect Immun 2014; 82: 1994–2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Zhang S, Chen D-C. Facing a new challenge: the adverse effects of antibiotics on gut microbiota and host immunity. Chin Med J 2019; 132: 1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Yu LC, Shih YA, Wu LL, et al. Enteric dysbiosis promotes antibiotic-resistant bacterial infection: systemic dissemination of resistant and commensal bacteria through epithelial transcytosis. Am J Physiol Gastrointest Liver Physiol 2014; 307: G824–G835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Brandl K, Plitas G, Mihu CN, et al. Vancomycin-resistant enterococci exploit antibiotic-induced innate immune deficits. Nature 2008; 455: 804–807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Kullberg RF, Wiersinga WJ, Haak BW. Gut microbiota and sepsis: from pathogenesis to novel treatments. Curr Opin Gastroenterol 2021; 37; 578–585. [DOI] [PubMed] [Google Scholar]
- 86. Suez J, Zmora N, Zilberman-Schapira G, et al. Post-antibiotic gut mucosal microbiome reconstitution is impaired by probiotics and improved by autologous FMT. Cell 2018; 174: 1406–1423.e16. [DOI] [PubMed] [Google Scholar]
- 87. Parnham MJ, Erakovic Haber V, Giamarellos-Bourboulis EJ, et al. Azithromycin: mechanisms of action and their relevance for clinical applications. Pharmacol Ther 2014; 143: 225–245. [DOI] [PubMed] [Google Scholar]
- 88. Patel A, Joseph J, Periasamy H, et al. Azithromycin in combination with ceftriaxone reduces systemic inflammation and provides survival benefit in a murine model of polymicrobial sepsis. Antimicrob Agents Chemother 2018; 62: e00752-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Kawamura K, Ichikado K, Takaki M, et al. Efficacy of azithromycin in sepsis-associated acute respiratory distress syndrome: a retrospective study and propensity score analysis. Springerplus 2016; 5: 1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Afshar M, Foster CL, Layden JE, et al. Azithromycin use and outcomes in severe sepsis patients with and without pneumonia. J Crit Care 2016; 32: 120–125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Oostdijk EA, de Smet AM, Kesecioglu J, et al. The role of intestinal colonization with gram-negative bacteria as a source for intensive care unit-acquired bacteremia. Crit Care Med 2011; 39: 961–966. [DOI] [PubMed] [Google Scholar]
- 92. Stoutenbeek CP, van Saene HK, Miranda DR, et al. The effect of selective decontamination of the digestive tract on colonisation and infection rate in multiple trauma patients. Intensive Care Med 1984; 10: 185–192. [DOI] [PubMed] [Google Scholar]
- 93. De Jonge E, De Wilde R, Juffermans N, et al. Carriage of antibiotic-resistant Gram-negative bacteria after discontinuation of selective decontamination of the digestive tract (SDD) or selective oropharyngeal decontamination (SOD). Critical Care 2018; 22: 243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Plantinga NL, de Smet AMGA, Oostdijk EAN, et al. Selective digestive and oropharyngeal decontamination in medical and surgical ICU patients: individual patient data meta-analysis. Clin Microbiol Infect 2018; 24: 505–513. [DOI] [PubMed] [Google Scholar]
- 95. Hurley JC. Selective digestive decontamination is neither safe nor efficacious for critically ill patients. Crit Care Med 2020; 48: 732–735. [DOI] [PubMed] [Google Scholar]
- 96. Reis Miranda D, Citerio G, Perner A, et al. Use of selective digestive tract decontamination in European intensive cares: the ifs and whys. Minerva Anestesiol 2015; 81: 734–742. [PubMed] [Google Scholar]
- 97. Minozzi S, Pifferi S, Brazzi L, et al. Topical antibiotic prophylaxis to reduce respiratory tract infections and mortality in adults receiving mechanical ventilation. Cochrane Database Syst Rev 2021; 1: CD000022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Bassetti M, Bandera A, Gori A. Therapeutic potential of the gut microbiota in the management of sepsis. In: Vincent J-L. (ed.) Annual update in intensive care and emergency medicine. Heidelberg: Springer, 2020, pp. 293–304. [Google Scholar]
- 99. Theodorakopoulou M, Perros E, Giamarellos-Bourboulis EJ, et al. Controversies in the management of the critically ill: the role of probiotics. Int J Antimicrob Agents 2013; 42(Suppl.): S41–S44. [DOI] [PubMed] [Google Scholar]
- 100. Fijan S. Microorganisms with claimed probiotic properties: an overview of recent literature. Int J Environ Res Public Health 2014; 11: 4745–4767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Gerritsen J, Timmerman HM, Fuentes S, et al. Correlation between protection against sepsis by probiotic therapy and stimulation of a novel bacterial phylotype. Appl Environ Microbiol 2011; 77: 7749–7756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Manzanares W, Lemieux M, Langlois PL, et al. Probiotic and synbiotic therapy in critical illness: a systematic review and meta-analysis. Critical Care 2016; 19: 262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Morrow LE, Kollef MH, Casale TB. Probiotic prophylaxis of ventilator-associated pneumonia: a blinded, randomized, controlled trial. Am J Respir Crit Care Med 2010; 182: 1058–1064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Weng H, Li JG, Mao Z, et al. Probiotics for preventing ventilator-associated pneumonia in mechanically ventilated patients: a meta-analysis with trial sequential analysis. Front Pharmacol 2017; 8: 717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Mihatsch WA, Braegger CP, Decsi T, et al. Critical systematic review of the level of evidence for routine use of probiotics for reduction of mortality and prevention of necrotizing enterocolitis and sepsis in preterm infants. Clin Nutr 2012; 31: 6–15. [DOI] [PubMed] [Google Scholar]
- 106. Johnstone J, Meade M, Lauzier F, et al. Effect of probiotics on incident ventilator-associated pneumonia in critically ill patients: a randomized clinical trial. JAMA 2021; 326: 1024–1033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Miller WD, Keskey R, Alverdy JC. Sepsis and the microbiome: a vicious cycle. J Infect Dis 2021; 223: S264–S269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Walker AW, Ince J, Duncan SH, et al. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J 2011; 5: 220–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Stinson LF, Payne MS, Keelan JA. Planting the seed: origins, composition, and postnatal health significance of the fetal gastrointestinal microbiota. Crit Rev Microbiol 2017; 43: 352–369. [DOI] [PubMed] [Google Scholar]
- 110. Wei Y, Gao J, Kou Y, et al. The intestinal microbial metabolite desaminotyrosine is an anti-inflammatory molecule that modulates local and systemic immune homeostasis. FASEB J 2020; 34: 16117–16128. [DOI] [PubMed] [Google Scholar]
- 111. Mu S, Zhang J, Du S, et al. Gut microbiota modulation and anti-inflammatory properties of Xuanbai Chengqi decoction in septic rats. J Ethnopharmacol 2021; 267: 113534. [DOI] [PubMed] [Google Scholar]
- 112. Sarma SM, Singh DP, Singh P, et al. Finger millet arabinoxylan protects mice from high-fat diet induced lipid derangements, inflammation, endotoxemia and gut bacterial dysbiosis. Int J Biol Macromol 2018; 106: 994–1003. [DOI] [PubMed] [Google Scholar]
- 113. Chi C, Buys N, Li C, et al. Effects of prebiotics on sepsis, necrotizing enterocolitis, mortality, feeding intolerance, time to full enteral feeding, length of hospital stay, and stool frequency in preterm infants: a meta-analysis. Eur J Clin Nutr 2019; 73: 657–670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Yokoyama Y, Nishigaki E, Abe T, et al. Randomized clinical trial of the effect of perioperative synbiotics versus no synbiotics on bacterial translocation after oesophagectomy. Br J Surg 2014; 101: 189–199. [DOI] [PubMed] [Google Scholar]
- 115. Kinross JM, Markar S, Karthikesalingam A, et al. A meta-analysis of probiotic and synbiotic use in elective surgery: does nutrition modulation of the gut microbiome improve clinical outcome? JPEN J Parenter Enteral Nutr 2013; 37: 243–253. [DOI] [PubMed] [Google Scholar]
- 116. Shimizu K, Yamada T, Ogura H, et al. Synbiotics modulate gut microbiota and reduce enteritis and ventilator-associated pneumonia in patients with sepsis: a randomized controlled trial. Critical Care 2018; 22: 239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Haak BW, Prescott HC, Wiersinga WJ. Therapeutic potential of the gut microbiota in the prevention and treatment of sepsis. Front Immunol 2018; 9: 2042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Cammarota G, Ianiro G, Tilg H, et al. European consensus conference on faecal microbiota transplantation in clinical practice. Gut 2017; 66: 569–580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Assimakopoulos SF, Papadopoulou I, Bantouna D, et al. Fecal microbiota transplantation and hydrocortisone ameliorate intestinal barrier dysfunction and improve survival in a rat model of cecal ligation and puncture-induced sepsis. Shock 2021; 55: 666–675. [DOI] [PubMed] [Google Scholar]
- 120. Cheng S, Ma X, Geng S, et al. Fecal microbiota transplantation beneficially regulates intestinal mucosal autophagy and alleviates gut barrier injury. mSystems 2018; 3: e00137-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Klingensmith NJ, Coopersmith CM. Fecal microbiota transplantation for multiple organ dysfunction syndrome. Crit Care 2016; 20: 398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Cross AS, Opal SM, Palardy JE, et al. A pilot study of an anti-endotoxin Ig-enriched bovine colostrum to prevent experimental sepsis. Innate Immun 2021; 27: 266–274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Piya MK, Harte AL, McTernan PG. Metabolic endotoxaemia: is it more than just a gut feeling. Curr Opin Lipidol 2013; 24: 78–85. [DOI] [PubMed] [Google Scholar]
- 124. Lin TL, Shu CC, Chen YM, et al. Like cures like: pharmacological activity of anti-inflammatory lipopolysaccharides from gut microbiome. Front Pharmacol 2020; 11: 554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Tilg H, Moschen AR. Food, immunity, and the microbiome. Gastroenterology 2015; 148: 1107–1119. [DOI] [PubMed] [Google Scholar]
- 126. Krupnick AS, Morris JB. The long-term results of resection and multiple resections in Crohn’s disease. Semin Gastrointest Dis 2000; 11: 41–51. [PubMed] [Google Scholar]
- 127. Wang G, Wen J, Xu L, et al. Effect of enteral nutrition and ecoimmunonutrition on bacterial translocation and cytokine production in patients with severe acute pancreatitis. J Surg Res 2013; 183: 592–597. [DOI] [PubMed] [Google Scholar]
- 128. Liu Y, Zhao W, Chen W, et al. Effects of early enteral nutrition on immune function and prognosis of patients with sepsis on mechanical ventilation. J Intensive Care Med 2020; 35: 1053–1061. [DOI] [PubMed] [Google Scholar]
- 129. Sawicki CM, Livingston KA, Obin M, et al. Dietary fiber and the human gut microbiota: application of evidence mapping methodology. Nutrients 2017; 9: 125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Moron R, Galvez J, Colmenero M, et al. The importance of the microbiome in critically ill patients: role of nutrition. Nutrients 2019; 11: 3002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Oudemans-van Straaten HM, van Zanten AR. Glutamine supplementation in the critically ill: friend or foe? Crit Care 2014; 18: 143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Marino LV, Pathan N, Meyer R, et al. The effect of 2 mMol glutamine supplementation on HSP70 and TNF-alpha release by LPS stimulated blood from healthy children. Clin Nutr 2015; 34: 1195–1201. [DOI] [PubMed] [Google Scholar]
- 133. Koksal GM, Erbabacan E, Tunali Y, et al. The effects of intravenous, enteral and combined administration of glutamine on malnutrition in sepsis: a randomized clinical trial. Asia Pac J Clin Nutr 2014; 23: 34–40. [DOI] [PubMed] [Google Scholar]
- 134. Cohen I, Ruff WE, Longbrake EE. Influence of immunomodulatory drugs on the gut microbiota. Transl Res 2021; 233: 144–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Nayak RR, Alexander M, Deshpande I, et al. Methotrexate impacts conserved pathways in diverse human gut bacteria leading to decreased host immune activation. Cell Host Microbe 2021; 29: 362–377.e11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Ramanan M, Cohen J, Venkatesh B. Steroids and sepsis: the debate continues. Int Anesthesiol Clin 2019; 57: 17–30. [DOI] [PubMed] [Google Scholar]
- 137. Batzofin BM, Sprung CL, Weiss YG. The use of steroids in the treatment of severe sepsis and septic shock. Best Pract Res Clin Endocrinol Metab 2011; 25: 735–743. [DOI] [PubMed] [Google Scholar]
- 138. Xiang XS, Li N, Zhao YZ, et al. Combination therapy with thymosin alpha1 and dexamethasone helps mice survive sepsis. Inflammation 2014; 37: 402–416. [DOI] [PubMed] [Google Scholar]
- 139. Bergquist M, Nurkkala M, Rylander C, et al. Expression of the glucocorticoid receptor is decreased in experimental Staphylococcus aureus sepsis. J Infect 2013; 67: 574–583. [DOI] [PubMed] [Google Scholar]
- 140. Ozturk E, Surucu M, Karaman A, et al. Protective effect of leflunomide against oxidative intestinal injury in a rodent model of sepsis. J Surg Res 2014; 187: 610–615. [DOI] [PubMed] [Google Scholar]
- 141. Ziegler SF, Roan F, Bell BD, et al. The biology of thymic stromal lymphopoietin (TSLP). Adv Pharmacol 2013; 66: 129–155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Bjerkan L, Sonesson A, Schenck K. Multiple functions of the new cytokine-based antimicrobial peptide thymic stromal lymphopoietin (TSLP). Pharmaceuticals 2016; 9: 41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Bergmann CB, Beckmann N, Salyer CE, et al. Potential targets to mitigate trauma-or sepsis-induced immune suppression. Front Immunol 2021; 12: 622601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Tavares E, Maldonado R, Miñano FJ. Immunoneutralization of endogenous aminoprocalcitonin attenuates sepsis-induced acute lung injury and mortality in rats. Am J Pathol 2014; 184: 3069–3083. [DOI] [PubMed] [Google Scholar]
- 145. Sawa T, Ito E, Nguyen VH, et al. Anti-PcrV antibody strategies against virulent Pseudomonas aeruginosa. Hum Vaccin Immunother 2014; 10: 2843–2852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. Forkosh E, Kenig A, Ilan Y. Introducing variability in targeting the microtubules: review of current mechanisms and future directions in colchicine therapy. Pharmacol Res Perspect 2020; 8: e00616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Ilan Y. Randomness in microtubule dynamics: an error that requires correction or an inherent plasticity required for normal cellular function. Cell Biol Int 2019; 43: 739–748. [DOI] [PubMed] [Google Scholar]
- 148. Ilan-Ber T, Ilan Y. The role of microtubules in the immune system and as potential targets for gut-based immunotherapy. Mol Immunol 2019; 111: 73–82. [DOI] [PubMed] [Google Scholar]
- 149. Ilan Y. Microtubules: from understanding their dynamics to using them as potential therapeutic targets. J Cell Physiol 2019; 234: 7923–7937. [DOI] [PubMed] [Google Scholar]
- 150. Ben-Chetrit E. Colchicine. In: Hashkes PJ, Laxer RM, Simon A. (eds) Textbook of autoinflammation. Heidelberg: Springer, 2019, pp. 729–749. [Google Scholar]
- 151. Paschke S, Weidner AF, Paust T, et al. Technical advance: inhibition of neutrophil chemotaxis by colchicine is modulated through viscoelastic properties of subcellular compartments. J Leukoc Biol 2013; 94: 1091–1096. [DOI] [PubMed] [Google Scholar]
- 152. Slobodnick A, Shah B, Krasnokutsky S, et al. Update on colchicine, 2017. Rheumatology 2018; 57: i4–i11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153. Ishay Y, Potruch A, Schwartz A, et al. A digital health platform for assisting the diagnosis and monitoring of COVID-19 progression: an adjuvant approach for augmenting the antiviral response and mitigating the immune-mediated target organ damage. Biomed Pharmacother 2021; 143: 112228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Ishay Y, Potruch A, Weksler-Zangen S, et al. Augmented antiviral T cell immunity by oral administration of IMM-124E in preclinical models and a phase I/IIa clinical trial: a method for the prevention and treatment of COVID-19. Drug Dev Res. Epub ahead of print 1 October 2021. DOI: 10.1002/ddr.21890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Ilan Y. Improving global healthcare and reducing costs using second-generation artificial intelligence-based digital pills: a market disruptor. Int J Environ Res Public Health 2021; 18: 811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156. Ilan Y. Second-generation digital health platforms: placing the patient at the center and focusing on clinical outcomes. Front Digit Health 2020; 2: 569178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157. Khoury T, Ishay Y, Rotnemer-Golinkin D, et al. A synergistic effect of Ambroxol and Beta-Glucosylceramide in alleviating immune-mediated hepatitis: a novel immunomodulatory non-immunosuppressive formulation for treatment of immune-mediated disorders. Biomed Pharmacother 2020; 132: 110890. [DOI] [PubMed] [Google Scholar]
- 158. Gelman R, Bayatra A, Kessler A, et al. Targeting SARS-CoV-2 receptors as a means for reducing infectivity and improving antiviral and immune response: an algorithm-based method for overcoming resistance to antiviral agents. Emerg Microbes Infect 2020; 9: 1397–1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159. Ilan Y. Overcoming randomness does not rule out the importance of inherent randomness for functionality. J Biosci 2019; 44: 132. [PubMed] [Google Scholar]
- 160. Khoury T, Ilan Y. Introducing patterns of variability for overcoming compensatory adaptation of the immune system to immunomodulatory agents: a novel method for improving clinical response to anti-TNF therapies. Front Immunol 2019; 10: 2726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161. Ilan Y. Immune rebalancing by oral immunotherapy: a novel method for getting the immune system back on track. J Leukoc Biol 2019; 105: 463–472. [DOI] [PubMed] [Google Scholar]
- 162. Ilan Y, Shailubhai K, Sanyal A. Immunotherapy with oral administration of humanized anti-CD3 monoclonal antibody: a novel gut-immune system-based therapy for metaflammation and NASH. Clin Exp Immunol 2018; 193: 275–283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163. Lalazar G, Zigmond E, Weksler-Zangen S, et al. Oral administration of beta-glucosylceramide for the treatment of insulin resistance and nonalcoholic steatohepatitis: results of a double-blind, placebo-controlled trial. J Med Food 2017; 20: 458–464. [DOI] [PubMed] [Google Scholar]
- 164. Ilan Y, Gingis-Velitski S, Ben Ya’aco A, et al. A plant cell-expressed recombinant anti-TNF fusion protein is biologically active in the gut and alleviates immune-mediated hepatitis and colitis. Immunobiology 2017; 222: 544–551. [DOI] [PubMed] [Google Scholar]
- 165. Ilan Y. Review article: novel methods for the treatment of non-alcoholic steatohepatitis – targeting the gut immune system to decrease the systemic inflammatory response without immune suppression. Aliment Pharmacol Ther 2016; 44: 1168–1182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166. Ilan Y. Oral immune therapy: targeting the systemic immune system via the gut immune system for the treatment of inflammatory bowel disease. Clin Transl Immunology 2016; 5: e60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167. Israeli E, Zigmond E, Lalazar G, et al. Oral mixture of autologous colon-extracted proteins for the Crohn’s disease: a double-blind trial. World J Gastroenterol 2015; 21: 5685–5694. [DOI] [PMC free article] [PubMed] [Google Scholar]