Abstract
Introduction
Cerebral vasospasm (VSP) is the leading risk factor of neurological deterioration (i.e., delayed cerebral ischemia [DCI] and cerebral infarction) after aneurysmal subarachnoid hemorrhage (aSAH) and a cause of morbidity and mortality. The objective of this systematic literature review is to summarize the economic and humanistic burden of VSP and its related complications after aSAH.
Methods
A predefined protocol was designed, and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were followed. Systematic searches were conducted in MEDLINE, Embase, and Cochrane (in January 2021) to identify studies reporting economic and/or humanistic (i.e., health-related quality of life [HRQoL]) outcomes for patients with asymptomatic and symptomatic VSP after aSAH. Related conferences and additional sources were searched manually. Dual screening, data extraction, and qualitative analysis were conducted.
Results
Of 3818 abstracts identified for review, 43 full-text articles representing 42 single studies met the inclusion criteria and were included. Most studies (33) were observational; nine were randomized clinical trials (RCTs). Economic outcomes were reported in 31 studies, and alongside HRQoL outcomes in 4 studies; 7 studies reported HRQoL outcomes only. Forty studies were conducted in single countries, while only 2 RCTs were conducted in multiple countries. Patients diagnosed with VSP or DCI spent between 2.1 and 7.4 days longer in intensive care and between 4.7 and 17 days longer in hospital (total) compared with patients without VSP or DCI. A significantly higher cost burden of US$33,945 (2021 £26,712) was identified for patients with VSP and £9370 (2021 £13,733) for patients with DCI compared with patients without. Patients with DCI were also disadvantaged by being employed for 62 fewer days (during 24-month follow-up), with an estimated mean cost of £3821 (2021 £5600) for days off work. Poor HRQoL was associated with ≥ 1 days with VSP symptoms (odds ratio [OR]: 2.8, 95% confidence interval [CI]: 1.4–5.3), symptomatic VSP (OR: 1.9, 95% CI: 1.0–3.6), and DCI (OR: 2.3, 95% CI: 1.3–4.2), although this was not consistent across all studies. Symptomatic VSP and DCI were identified as significant risk factors for depressed mood (OR: 2.2, 95% CI: 1.0–4.9) and global cognitive impairment (OR: 2.3) at 12 months, respectively. The severity of VSP was a critical predictor of post-aSAH economic and humanistic burden. Similar trends in economic and humanistic burden were identified in the general aSAH patient population. Study design and patient heterogeneity precluded direct metaanalysis of the results.
Conclusion
A substantial direct and indirect economic burden is linked to VSP and its related complications after aSAH. Although limited evidence was identified for humanistic burden, these patients seem to suffer from poor HRQoL with long-lasting burden. Overall, there is an urgent need to understand better the concept of “burden of illness” of VSP and its related complications after aSAH.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40120-022-00348-6.
Keywords: Aneurysmal subarachnoid hemorrhage, Delayed cerebral ischemia, Economic burden, Health-related quality of life, Systematic review, Vasospasm
Plain Language Summary
Aneurysmal subarachnoid hemorrhage is a sudden, life-threatening emergency caused by bleeding in the subarachnoid space between the brain and skull. Vasospasm of the arteries surrounding the hemorrhage occurs in most patients and may lead to permanent brain damage. This study summarizes the published literature to describe the burden that patients may experience due to vasospasm and its related complications after aneurysmal subarachnoid hemorrhage, focusing on financial and life quality aspects. We show that the burden of vasospasm, and its related complications, is huge. Patients often experience reduced quality of life due to their poor health and are more likely to suffer from depression and intellectual impairment. There is also a substantial financial burden linked to vasospasm and its related complications, driven by the need for more intensive care, hospitalization, and higher investigative costs associated with the treatment and management of these patients. In addition, days off work and unemployment can cause a substantial indirect financial burden. Our study highlights the need for additional research to understand further the “burden of illness” of vasospasm and its related complications after aneurysmal subarachnoid hemorrhage.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40120-022-00348-6.
Key Summary Points
| Why carry out this study? |
| This is the first systematic literature review to summarize the economic and humanistic (quality of life) burden induced by cerebral vasospasm (VSP) and its related complications (namely delayed cerebral ischemia [DCI] and cerebral infarction) after aneurysmal subarachnoid hemorrhage (aSAH). |
| What was learned from the study? |
| The evidence showed a substantial incremental direct and indirect economic and humanistic burden in people who developed cerebral VSP and its related complications compared with those who did not. |
| The direct economic burden is mainly driven by a significant difference in intensive care and total hospitalization length of stay. |
| A single study revealed that people with DCI had a higher number of days out of work than those without DCI. |
| Although limited, evidence on humanistic outcomes showed that cerebral VSP and DCI are significant predictors of depressed mood and cognitive impairment at 12 months, respectively. |
| Early prevention of cerebral VSP occurrence and careful disease management are likely to reduce patients’ economic and humanistic burden after aSAH. |
| Further research is needed to confirm and further quantify the economic and humanistic burden of cerebral VSP and its related complications for patients after aSAH, especially after hospital discharge. |
Introduction
Aneurysmal subarachnoid hemorrhage (aSAH) is sudden life-threatening bleeding in the subarachnoid space between the brain and skull caused by the rupture of an aneurysm, a weak bulge formed on the wall of a cerebral artery [1, 2]. Approximately 85% of spontaneous SAH events (i.e., those not occurring as a result of trauma) are caused by a ruptured aneurysm [1]. Emergency intervention with endovascular coiling or microsurgical clipping is necessary to secure the aneurysm and prevent rebleeding.
aSAH has a global incidence of 5–9.1 cases per 100,000 person-years, with regional variation reported [1, 3–7]. The incidence of aSAH is 1.24 (95% confidence interval [CI]: 1.09–1.42) times higher in women than in men, and the risk of aSAH increases with age [3]. Mortality and morbidity rates following aSAH are high; at least 25% of patients will die within 48 h of the event, with death before hospital admission occurring in 10–15% of patients and in-hospital mortality reported in 20% of admitted patients, while around half of survivors will experience ongoing neurological deficit [8, 9].
Cerebral vasospasm (VSP) typically begins at around day 3 after aSAH onset, peaks in severity at days 8–11, and resolves by day 21 [10], with angiographic VSP (aVSP) being detectable in up to 70% of patients post-aSAH [11–14]. aVSP is an important contributor to delayed cerebral ischemia (DCI, also known as delayed ischemic neurological deficit) and cerebral infarction (CI). Other factors may also contribute to DCI or CI, including delayed cell apoptosis, microcirculatory dysfunction, microembolism, and cortical spreading depression [15]. Nonetheless, DCI is often regarded as being caused by VSP; 20–50% of patients with VSP will develop DCI between 4 and 14 days post-aSAH, and approximately 85% of patients with DCI have evidence of cerebral VSP [11, 16–19]. DCI is generally defined as the occurrence of focal neurologic impairment, or a decrease of at least 2 points on the Glasgow Coma Scale (GCS), that lasts for at least 1 h but is not apparent immediately after aneurysm occlusion or attributable to other causes after appropriate clinical, imaging, and laboratory assessments [20]. Approximately one-third of patients with aSAH will develop DCI-related neurological deterioration [11, 13, 21, 22].
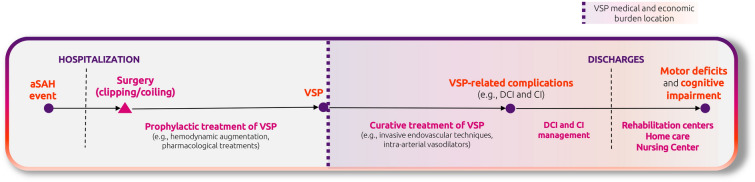
For patients who experience aSAH, acute downstream clinical complications of VSP, including DCI and CI, can cause a considerable long-term burden due to functional and cognitive impairment (Fig. 1). Several treatments for preventing or managing VSP-related complications have been tested in trials and adopted in clinical practice. To date, however, a single treatment that is effective in preventing such complications is lacking [18]. Hence, there is an urgent unmet therapeutic need in aSAH patients most at risk of cerebral VSP.
Fig. 1.
Patient pathway for VSP and its related complications following aSAH. aSAH, aneurysmal subarachnoid hemorrhage; CI, cerebral infarction; DCI, delayed cerebral ischemia; VSP, cerebral vasospasm
While previous publications have indicated that VSP and its related complications are associated with a substantial economic burden, this evidence largely comprises individual studies that are of limited generalizability due to differences in population characteristics, geographical locations, and clinical settings. To provide an up-to-date understanding of the economic and humanistic (i.e., health-related quality of life [HRQoL]) burden of cerebral VSP after aSAH, we systematically searched for, identified, and summarized the evidence from relevant randomized clinical trials (RCTs) and real-world evidence studies. The present paper reports the outcomes of this systematic literature review (SLR), focusing on the short- and long-term economic and HRQoL burden of VSP and its related complications after aSAH. A separate, future publication will address the clinical burden for this patient population.
Methods
Search Strategy
The SLR was conducted according to PRISMA 2020 guidelines [23]. A predefined protocol outlining the Population, Intervention, Comparators, Outcomes, and Study design (PICOS) criteria of the review was established (Table 1). Searches were developed on separate search terms for the disease (“subarachnoid hemorrhage” or “vasospasm”) and outcomes of interest. No time or language restrictions were applied in the search. However, editorials, conference abstracts, and narrative reviews were excluded.
Table 1.
PICOS criteria
| Population | Patients (anµy age) with VSP-related complications following aSAH after clipping surgery or coiling |
| Interventions/comparators | Any treatment received in ICU and post-ICU setting or no intervention (for economic and humanistic burden) |
| Outcomes - Economic | During or post-hospitalization: |
| Direct costs | |
| Indirect costs | |
| Economic model results (e.g., ICER, LYG, cost per QALY) | |
| Hospitalization journey (e.g., length of stay) | |
| Resource use | |
| Outcomes - Humanistic | Utility, disutility scores |
| Health-related quality of life measures (e.g., SF-36, EQ5D) | |
| Loss of productivity | |
| Caregiver burden | |
| Study design | Clinical trials (RCTs, NRS) |
| Observational studies (any study design); real-world studies, hospital databases or chart reviews, economic or cost/utility studies (CEA, CUA, BIA, CMA), studies reporting on humanistic outcomes | |
| Excluded: commentaries, expert reviews, case reports | |
| Other considerations | Studies of aSAH survivors with subset of VSP patients to be included |
| Minimum sample size: ≥ 2 individuals | |
| Exclusion criteria | Studies not meeting the inclusion criteria outlined above |
aSAH, aneurysmal subarachnoid hemorrhage; BIA, budget impact analysis; CEA, cost-effectiveness analysis; CMA, cost-minimization analysis; CUA, cost-utility analysis; EQ-5D, EuroQoL 5-Dimensions; ICER, incremental cost-effectiveness ratio; ICU, intensive care unit; LYG, life-years gained; NRS, nonrandomized study; QALY, quality-adjusted life years; RCT, randomized controlled trial; SF-36, 36-item Short-Form Health Survey; VSP, vasospasm
MEDLINE, Embase, and the Cochrane Library were systematically searched on 8 January 2021 to identify peer-reviewed studies pertinent to the following economic and humanistic SLR research question: What is the economic and humanistic burden of VSP and its related complications after aSAH on patients, compared with patients without such complications?
Supplementary searches of relevant conference proceedings (held 2016–2020), key scientific societies, health technology assessment institutions, and bibliographies of pertinent SLRs were conducted.
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. See Supplementary Tables S1 and S2 for full details of the search strategy.
Study Selection
Duplicate references were removed, and the final list of abstracts was reviewed independently by two systematic reviewers against the SLR eligibility criteria. Where discrepancies arose, consensus between reviewers was reached by mutual consent or by a third reviewer. For studies selected for inclusion in the SLR, the double review process was repeated using the full-text articles.
During the full-text screening, the inclusion population criteria were expanded to consider studies that presented relevant outcomes for the general population of patients after aSAH if the study reported that a group of the recruited population was diagnosed with VSP. Therefore, the presentation of results for the economic and humanistic outcomes is structured in two separate parts, firstly presenting findings directly reported for patients with VSP and therefore answering our primary research question, and secondly, presenting findings from studies including a general patient population after aSAH (independently of VSP status) and therefore indirectly answering our research question.
Data Extraction
A bespoke extraction sheet (Supplementary Table S3) was developed, and all relevant data were extracted and validated. Information was collected regarding the study objective, study and patient characteristics, medication, and study outcomes (economic burden: direct/indirect costs, economic model results, and resource use; humanistic burden: utility/disutility scores, HRQoL measures, and caregiver burden). Details of consecutive recruitment of patients were also collected and considered in the potential bias of results.
To allow direct comparability of findings across studies, all costs were adjusted for inflation to year 2021 using the consumer price index (CPI) of each country (CPI Health index for UK; CPI for All Urban Consumers [CPI-U] Medical care services for USA seasonally adjusted). Costs were then converted into pound sterling (ISO code: GBP; symbol: £) to ease intercountry cost comparisons, using the exchange rate of the year in which the cost/data were collected. Where cost years spanned several years (e.g., 2012–2015), the last date (e.g., 2015) was used as the cost/data collection year. Costs were then rounded to the nearest whole number for reporting purposes, with equivalent costs in GBP inflated to 2021 and denoted as “2021 £.”
Due to the high heterogeneity in study design and patient characteristics between the studies, results are presented qualitatively as metaanalysis of the findings was not feasible.
Results
Included Studies
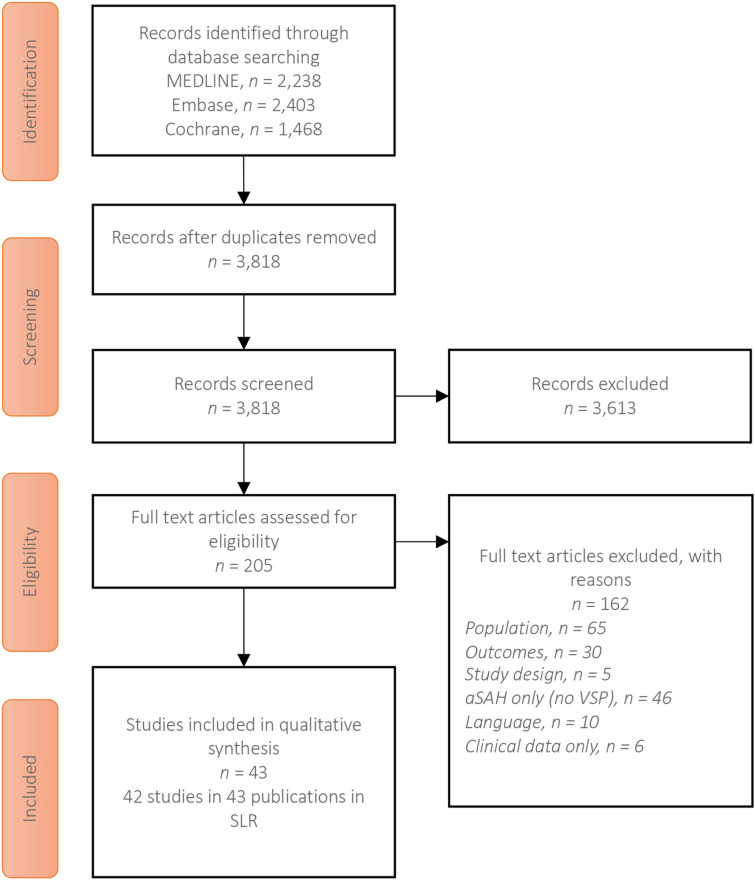
Searches in MEDLINE (2238 hits), Embase (2403 hits), and Cochrane (1468 hits), after the removal of 2291 duplicates, identified 3818 abstracts for review. From these, 205 full-text articles were assessed for eligibility and 162 articles were excluded, leaving 43 full-text articles representing 42 single studies meeting the inclusion criteria and included in this SLR (Fig. 2). Of note, Vetkas et al. [24] was linked to Vetkas et al. [25], which reported on the same population but did not provide any additional data, therefore no data were extracted. Vetkas et al. [25] did, however, meet the eligibility criteria for the SLR and was therefore included in the number of full-text articles identified. No additional relevant studies were identified from searches of conference proceedings or other sources.
Fig. 2.
PRISMA diagram showing the study selection process. aSAH, aneurysmal subarachnoid hemorrhage; SLR, systematic literature review; VSP, cerebral vasospasm
Nine studies [26–34] were linked to clinical trial reports, and 33 were observational [11, 24, 35–65] (see Supplementary Table S4 for details on study characteristics). Most reported data on economic outcomes only (31 studies) or alongside HRQoL outcomes (4 studies); 7 studies reported data on HRQoL outcomes only. No economic evaluations of treatments for VSP-related complications were identified.
Forty studies were conducted in single countries: 20 studies in the USA [11, 26, 30, 36, 39, 41–44, 46–48, 52, 56, 58–60, 63–65], and the remainder in Australia [49], Brazil [29], Canada [35], China [31], Estonia [24], Finland [50], Germany [40, 53, 54], India [28], Japan [33], New Zealand [51], Poland [61], Portugal [57], Turkey [37, 38], and the UK [32, 45, 55, 62]. Two RCTs were conducted in multiple countries: the CONSCIOUS-1 trial [27] in Austria, Canada, Finland, France, Germany, Israel, Italy, Sweden, Switzerland, the UK, and the USA, and another trial [34] in Canada, the Netherlands, and the USA.
Studies varied considerably in terms of the period of data coverage and length of data collection, from 9 months to 15.5 years for observational studies and from 14 months to 6 years for RCTs (although the reported data collection years may not coincide with the trial duration), with follow-up (when reported) ranging from 3 months [27, 31, 34] to 24 months [32].
The longest study was a retrospective cohort analysis of cost drivers for aneurysms using the US Value Driven Outcomes database, which reported data from a 15.5-year period (July 2001 to January 2017) [63]. Two RCTs [30, 31] and 13 observational studies [35–38, 40–42, 45, 46, 52, 56, 59, 63] collected data after 2010.
Patient numbers in the RCTs ranged from 33 [30] to 1644 [32]. Twenty-seven observational studies reported findings from samples of < 500 patients; the smallest study had only 14 patients [57]. Four observational studies of US national patient databases reported patient numbers ranging from 5353 to 74,356 [42, 46, 47, 52].
Patient Characteristics
Of the 18 studies that reported baseline Fisher grade, 14 studies [28–30, 32, 33, 39, 40, 43, 45, 53, 59, 60, 63, 65] included a patient population with high disease severity, with most patients falling into the Fisher grade 3 category, which has the highest risk for VSP, DCI, and resulting poor outcomes [66]. Approximately two-thirds or more of patients from 18 studies [24, 26, 28–30, 34, 36, 38, 40, 43, 48, 50, 54, 59–61, 63, 65] had a Hunt–Hess baseline score of ≤ 3 at hospital entry, corresponding to asymptomatic or minimal headache (grade 1), moderate-to-severe headache (grade 2), or drowsiness, confusion, or mild focal deficit (grade 3). Additionally, around half or more of patients from 4 studies [11, 39, 53, 55] had a Hunt–Hess baseline score of ≥ 3 at hospital entry (grade 4 = stupor, moderate to severe hemiparesis; grade 5 = deep coma). Similarly, patient baseline World Federation of Neurosurgical Societies (WFNS) grade varied considerably across the studies, from grade 1 (fully awake) to grade 4 and 5 (highest severity, with or without focal deficit) across 8 studies [27, 32, 38, 40, 45, 53, 55, 61], baseline modified Rankin scale (mRS) scores varied from no disability to severe disability across six studies [24, 30, 33, 48, 57, 61], and baseline GCS scores ranging from 3 (no responsiveness) to 15 (highest responsiveness) were reported across 4 RTCs [26, 27, 30, 31] and 10 observational studies [37, 38, 44, 51, 53, 55, 56, 59, 60, 64].
Information on aneurysm size was not widely reported among the included studies. Seventeen studies [24, 30, 32–34, 36, 38, 39, 49–51, 54, 57, 59, 62, 63, 65] reported information on aneurysm location, with aneurysms more likely (> 50% of patients) to be located anteriorly, although middle, internal, and posterior locations were also described.
Type of aneurysm management (via microsurgical clipping or endovascular coiling) was widely reported across 7 RCTs [28–34] and 28 observational studies [24, 35–39, 42–47, 49–59, 61–65]. All patients underwent microsurgical clipping in 3 RCTs [28, 29, 33] and 3 observational studies [49, 59, 61]; 1 RCT [31] and 2 observational studies [57, 62] included patients who had only received endovascular coiling. Other reported procedures included endovascular pipeline flow diversion [63], endovascular drainage [36], angioplasty [47], and wrapping [53].
Economic Burden
Resource Use: Length of Intensive Care Unit Stay by VSP Status
Intensive care unit (ICU) length of stay (LOS) was significantly longer in patients with VSP or DCI than in those without in 2 RCTs [27, 32] and 5 observational studies [11, 35, 39, 44, 64] (Table 2).
Table 2.
Length of hospital stay (days) by VSP status
| Author (date) Study ID |
Data collection | Treatment | Population | Total | ICU | |||
|---|---|---|---|---|---|---|---|---|
| Mean (SD) | Median | Mean (SD) | Median | |||||
| Clinical trials | ||||||||
|
Macdonald et al. (2012) [27] CONSCIOUS-1 |
2005–2006 | Clazosentan | Severe aVSP | 24.5 (–)* | – | 17α* | – | |
| No aVSP | 19.8 (–) | – | 11α | – | ||||
|
Rivero-Arias et al. (2009) [32] ISAT^ |
1997–2002 | Coiling/neurosurgical treatment | DCI, during first episode | 38.7 (56.4)* | – | 7.0 (5.7)* | – | |
| No DCI, during first episode | 23.6 (44.0) | – | 4.9 (5.9) | – | ||||
| Coiling/neurosurgical treatment | DCI, during subsequent episodes | – | – | 13 (0)* | – | |||
| No DCI, during subsequent episodes | – | – | 3.5 (4.9) | – | ||||
| Prevedello et al. (2006) [29] | 2000–2004 | HHH therapy + nimodipine | VSP | 32.1 (22.1)* | – | – | – | |
| No VSP | 15.1 (12.7) | – | – | – | ||||
| Magnesium sulfate | VSP | 19.4 (6.9) | – | – | – | |||
| No VSP | 19 (8.5) | – | – | – | ||||
| Observational studies | ||||||||
| Chotai et al. (2021) [42] | 2012–2015 | Mixed (coiling, clipping) | VSP | – | 20 (IQR: 15–26)* | – | – | |
| No VSP | – | 15 (IQR: 10–22) | – | – | ||||
| Abulhasan et al. (2018) [35] | 2010–2016 | Unspecified | VSP with DCI | – | * | – | * | |
| Alaraj et al. (2017) [36] | 2011–2014 | Mixed (coiling, clipping) | VSP | 24.1 (–)* | – | – | – | |
| No VSP | 15.9 (–) | – | – | – | ||||
| Drazin et al. (2015) [44] | – | Mixed (coiling, clipping) | VSP (clinical, with neurological deterioration) | – | – | * | – | |
| Chou et al. (2010) [43] | 1999–2004 | Mixed (coiling, clipping) | TCD-defined VSP | 21.3 (9.8)** | 18.5 (range: 5–54) | – | – | |
| No TCD-defined VSP | 16.6 (8.3) | 15 (range: 4–49) | – | – | ||||
| Mixed (coiling, clipping) | DCI | 23.5 (9)** | 22 (range: 10–50) | – | – | |||
| No DCI | 17.5 (9.1) | 15 (range: 4–54) | – | – | ||||
| Frontera et al. (2009) [11] | 1996–2002 | Unspecified | VSP | *† | – | *† | – | |
| Badjatia et al. (2005) [39] | 1995–2002 | Mixed (coiling, clipping) | Symptomatic VSP | 26 (13.8)* | – | 16.9 (7.9)* | – | |
| No aVSP | 20.2 (10.6) | – | 12.3 (5.7) | – | ||||
| Yundt et al. (1996) [64] | 1993–1994 | Mixed (clipping, not specified) | VSP | 28.4 (13.8) (nr) | – | 18.5 (9.7) (nr) | – | |
| No VSP | 17.0 (15.0) | – | 11.1 (11.3) | – | ||||
aSAH, aneurysmal subarachnoid hemorrhage; aVSP, angiographic vasospasm; DCI, delayed cerebral infarction; HHH, hypertension, hypervolemia, and hemodilution; IQR, interquartile range; SAH, subarachnoid hemorrhage; SD, standard deviation; TCD, transcranial Doppler; VSP, cerebral vasospasm
*p < 0.001; **p < 0.05; nr, p-value not reported. ^All data presented in the post hoc ISAT analysis refer to mean resource use per patient (SD). αFigures are taken as approximations from the graphs in the publication. †Significantly longer for VSP subgroup, but exact numbers not reported and only available in a 3D graph which does not allow for approximation of results
Specifically, patients with VSP or DCI spent an additional 2.1 days (patients with DCI) [32], 4.6 days (symptomatic VSP and clinical VSP with neurological deterioration) [39, 44], or 7.4 days (undefined VSP) [64] longer in the ICU compared with patients without VSP or DCI (all p < 0.001 where reported) (Table 2).
Three studies [11, 27, 35] did not report exact figures on mean length of ICU stay but provided results of comparative or regression-based analysis of ICU stay as a function of VSP status. One retrospective analysis showed that the association between VSP with DCI and increased ICU LOS remained statistically significant even after adjustment for the effect of age, gender, and admission grade [35]. Significantly prolonged ICU stays were also identified in patient cohorts with aVSP, transcranial Doppler (TCD)-defined VSP, symptomatic VSP, and DCI (Mann–Whitney U; p < 0.001) [11]. A post hoc analysis of the CONSCIOUS-1 trial reported significantly longer ICU stays for patients with severe or moderate aVSP than those with no aVSP (Kruskal–Wallis; p < 0.001 and p < 0.0005, respectively), even after adjustment for age and WFNS grade, but ICU duration was comparable between patients with mild or no aVSP [27].
Resource Use: Length of Overall Hospital Stay by VSP Status
Three RCTs [27, 29, 32] and 7 observational studies [11, 35, 36, 39, 42, 43, 64] reported overall LOS in hospital with VSP-specific data (Table 2). In all but 1 study (the experimental arm of a small RCT evaluating magnesium sulfate in the treatment of VSP [29]), patients with VSP or DCI spent significantly more time in hospital than those without VSP or DCI.
More specifically, results from RCTs showed that patients with VSP had 4.7 days (a post hoc analysis of the CONSCIOUS-1 trial [27]) to 17 days (in the control arm of the RCT evaluating magnesium sulfate to treat VSP [29]) longer total hospital stay than patients who did not experience VSP, and that patients with DCI spent an additional 15.1 days longer in hospital than those without DCI [32] (all p < 0.001) (Table 2).
Similar statistically significant trends in overall LOS were seen in observational studies, which showed an additional 4.7–11.4 days spent in hospital for patients with VSP than those without VSP (all p < 0.05 where reported) (Table 2). This trend was consistent regardless of cohort size, with comparable findings seen in a large observational study (n > 5000 patients with aSAH) using the US National Inpatient Sample database [42] and from a smaller US single-center study of 174 aSAH patients [36].
Discharge Disposition
The specific role of VSP in discharge disposition was only reported in one smaller study (n = 107) [44], which identified higher odds of an adverse discharge (i.e., discharge not to home) for patients with VSP after spontaneous SAH compared with those without VSP (adjusted odds ratio [OR]: 1.7, 95% CI: 0.56–5.07), although this association was not statistically significant (p = 0.35).
Readmission Due to VSP
Readmission rates due to VSP ranged from 0% to 5.6% in one study investigating the effectiveness of nimodipine therapy after aSAH (≤ 14 to 21 days) [56] and from 5.8% to 5.9% in aSAH patients managed with clipping or coiling [50]. A large (n > 12,000) observational study identified readmission rates due to cerebral VSP in patients discharged after hospitalization for aSAH of 6.9% within 30 days (p = 0.016) and 20.1% within 90 days, although this association was not statistically significant (p = 0.439) [52].
Costs: Direct Costs
Four studies [32, 42, 43, 64] with VSP-specific data identified a higher direct cost burden for patients with VSP or DCI when compared with patients without VSP or DCI (Table 3).
Table 3.
Direct costs by VSP status
| Author (date) Study ID |
Data collection | Subgroup | n | Time point | Unit | Costs in local currency not inflated | Costs in GBP inflated to 2021 | ||
|---|---|---|---|---|---|---|---|---|---|
| Mean (SD) | Median (IQR) | Mean (SD) | Median (IQR) | ||||||
|
Rivero-Arias et al. (2009) [32] ISATa |
1997–2007 | With DCI | 403 | 24 months | GBP | 28,175* (26,773) | – | 41,293 (39,239) | – |
| No DCI | 1199 | 24 months | 18,805 (17,287) | – | 27,561 (25,336) | – | |||
| Chotai et al. (2021) [42]b | 2012–2015 | VSP | 1001 | – | USD | – |
101,735.42** (69,372.66–145,622.18) |
– |
80,059 (54,591–114,595) |
| No VSP | 4351 | – | USD | – |
67,790.77 (46,150.95–105,320.71) |
– |
53,347 (36,318–82,880) |
||
| Chou et al. (2010) [43]c | 1999–2004 | TCD-defined VSP | 116 | – | USD | 134,135† (62,859) |
118,699 (47,258–418,687) |
119,028 (55,780) |
105,331 (41,936–371,533) |
| No TCD-defined VSP | 73 | – | USD | 106,246 (47,360) |
91,181 (38,447–293,845) |
94,280 (42,026) |
80,912 (34,117–260,751) |
||
| DCI | 62 | – | USD | 150,101† (59,156) |
141,571 (77,340–418,687) |
133,196 (52,494) |
125,627 (68,630–371,533) |
||
| No DCI | 127 | – | USD | 110,310 (54,285) |
91,181 (38,447–309,413) |
97,887 (48,171) |
80,912 (34,117–274,566) |
||
| Yundt et al. (1996) [64]d | 1993–1994 | Surgically treated unruptured aneurysm | 28 | – | USD | 12,685 (8,783) | – | 22,233 (15,394) | – |
| aSAH | 42 | – | USD | 22,622 (20,897) | – | 39,650 (36,626) | – | ||
| SAH with VSP | 32 | – | USD | 38,415‡ (21,324) | – | 67,330 (37,375) | – | ||
| SAH negative angiogram | 10 | – | USD | 25,994 (33,475) | – | 45,560 (58,672) | – | ||
aSAH, aneurysmal subarachnoid hemorrhage; DCI, delayed cerebral infarction; GBP, British pound sterling; IQR, interquartile range; SAH, subarachnoid hemorrhage; SD, standard deviation; USD, US dollar; VSP, cerebral vasospasm
*p < 0.05 versus no DCI; **p < 0.001 versus no VSP; †p < 0.05 versus no VSP; ‡p < 0.001 versus surgically treated unruptured intracerebral aneurysm
aTreatment = Unspecified. bTreatment = Mixed (coiling, clipping). cTreatment = Mixed (coiling, clipping). Unadjusted inpatient costs. dTreatment = Mixed (clipping, not specified)
Specifically, the median total cost burden (cost years: 2012–2015) was significantly higher for patients with than those without VSP (US$101,735.42 versus US$67,790.77, respectively; an increase of US$33,945; p < 0.001) (2021 £80,059 versus £53,347, respectively; an increase of £26,712) [42], and patients with DCI were disadvantaged by a significantly higher difference of +£9370 (2021 £13,733) in total mean health costs per patient at 24-month follow-up (cost year: 2006) (p < 0.05 versus patients without DCI) [32]. Similarly, the mean total cost per patient was significantly higher for acute aSAH with VSP compared with surgically treated unruptured intracerebral aneurysm (p < 0.0001), and numerically higher compared with acute aSAH and acute SAH with negative angiogram [64] (Table 3).
Significantly higher unadjusted inpatient costs were also observed for patients with SAH and TCD-defined VSP or DCI than those without (cost year: 2006) [43]. After adjusting for potential confounding variables, adjusted total inpatient costs were 1.2 times significantly higher (p = 0.004) for patients with TCD-defined VSP than for those without (US$132,667 versus US$110,539, respectively; an increase of US$22,128) (2021 £117,726 versus £98,090, respectively; an increase of £19,636), and 1.3 times significantly higher (p < 0.001) for patients with DCI than for patients without (US$143,201 versus US$113,092, respectively; an increase of US$30,109) (2021 £127,073 versus £100,355, respectively; an increase of £26,718).
Costs: Indirect Costs
Estimates of indirect costs by VSP status were only reported in a post hoc analysis of the International Subarachnoid Aneurysm Trial (ISAT) (cost year: 2006) [32]. Despite the number of patients in paid employment being equally balanced prior to the initial hemorrhage (61.9% versus 58.9% for patients with DCI versus without DCI), patients with DCI were fully employed for significantly fewer days (62 days less) at 24-month follow-up (87 versus 149 days, respectively; p < 0.001). Patients with DCI were also disadvantaged by a higher mean cost for days off work of +£3821 (2021 £5600) than those without DCI (£27,583 versus £23,762, respectively) (2021 £40,426 versus £34,826, respectively).
General Patient Population after aSAH
For the general aSAH patient population (including some patients with VSP), the average ICU stay ranged from 10 to 19 days [27, 30, 40, 49, 60], and the average overall LOS in hospital varied between 7.75 and 20.9 days [26–28, 30, 31, 36, 37, 42, 45, 50, 63–65]. Overall, between 30% and 63% of aSAH patients were discharged home after receiving treatment [45, 47]. In terms of LOS after readmission, aSAH patients were rehospitalized for an average of 22.5–24.8 days within the first month and within the first 3 months [46].
The direct cost burden per patient in the general aSAH population was reported in 2 RCTs [26, 32] and 8 observational studies [36, 42, 43, 46, 47, 49, 60, 64] and ranged from a mean total cost of US$12,685 (2021 £22,233) in patients with surgically treated unruptured aneurysm [64] to a mean observed cost of US$421,942 (2021 £315,832) in patients with Hunt–Hess grade 4 [36]. Three studies reported a considerable indirect economic burden after aSAH, through reduced capacity to be employed or to return to previous full-time employment post-aSAH [24, 51, 57], with only 35% of aSAH survivors (mean follow-up of 4.5 years) returning to work despite generally acceptable neurologic recovery [24].
Humanistic Burden
Patient-Reported HRQoL
Patient-reported HRQoL for VSP-specific data was identified in 2 studies using the Short Form-36 (SF-36) Health Survey [38, 61]. In addition, 1 study each used the EuroQol 5-Dimensions (EQ-5D) [27], Sickness Impact Profile (SIP) [11], or measured HRQoL as cognitive impairment due to VSP-related DCI [58].
More specifically, the number of days with VSP symptoms (DVS) (≥ 1 DVS observed in 69 of 82 [84%] patients; mean DVS = 3.7 ± 2.4 days) was found to be a major risk factor for poorer HRQoL as measured by the SF-36 at 10–14 months post-aSAH (univariate OR: 2.7, 95% CI: 1.6–4.2; p < 0.001) [38]. The effect of VSP duration on HRQoL remained significant even after accounting for other factors such as treatment method, WFNS score, Hunt–Hess grade, or Fisher grade (multivariate OR: 2.8, 95% CI: 1.4–5.3; p = 0.002). Conversely, a retrospective study (n = 146) found no association between postoperative DCI after SAH and SF-36 scores at a mean follow-up of 6.5 years (p = 0.23–0.32) [61].
A post hoc analysis of the CONSCIOUS-1 trial identified significantly worse patient outcomes at 12 weeks after aSAH, measured by the EQ-5D total score and EQ-5D visual analog scale, among patients with severe aVSP versus patients with no aVSP (Kruskal–Wallis; both p < 0.0001) (Table 4) [27]. The EQ-5D total score was also reduced to a lesser extent in patients with moderate aVSP (Kruskal–Wallis; p = 0.034 versus patients with no aVSP), with no apparent differences between patients with mild or no aVSP.
Table 4.
VSP-specific populations reporting HRQoL using the EuroQol 5-Dimensions
| Author (date) Study ID |
Subgroup | Time point | Endpoint | n | Mean (95% CI) |
|---|---|---|---|---|---|
|
Macdonald et al. (2012) [27] CONSCIOUS-1 |
Severe aVSP | 12 weeks | Total | 73 | 0.38 (0.28–0.49)** |
| Moderate aVSP | 12 weeks | Total | 117 | 0.65 (0.59–0.70)* | |
| No aVSP | 12 weeks | Total | 169 | 0.74 (0.71–0.78) | |
| Severe aVSP | 12 weeks | VAS | 73 | 50.9 (42.9–58.8)** | |
| No aVSP | 12 weeks | VAS | 169 | 75.5 (72.7–78.4) |
aVSP, angiographic vasospasm; CI, confidence interval; HRQoL; health-related quality of life; n, number; VAS, visual analog scale
*p = 0.034 versus patients with no aVSP after adjustment for age and WFNS grade (Kruskal–Wallis one-way comparison across aVSP categories); **p < 0.0001 versus patients with no aVSP after adjustment for age and WFNS grade (Kruskal–Wallis one-way comparison across aVSP categories)
One study reported specifically on VSP populations using the SIP total score (dichotomized at the median value of the SAH study population) [11]. Both symptomatic VSP (OR: 1.9, 95% CI: 1.0–3.6; p = 0.048) and DCI (OR: 2.3, 95% CI: 1.3–4.2; p = 0.006) were associated with significant impairments in HRQoL (SIP ≥ 15.5) at 3 months after adjustment for disease and demographic factors. This was not seen in patients with aVSP (OR: 1.6, 95% CI: 0.9–2.9) or TCD-defined VSP (OR: 1.1, 95% CI: 0.7–1.8).
In one study that assessed cognitive impairment due to VSP-related DCI [58], patients who developed DCI had greater than two times higher odds of having global cognitive impairment at 12 months follow-up (OR: 2.3; p = 0.024), which remained significant after adjusting for age, education, and ethnicity (adjusted OR: 3.6, 95% CI 1.4–9.6; p = 0.01).
Depression and Emotional State
Patients with symptomatic VSP were significantly more likely to experience depressed mood (measured by the Center for Epidemiologic Studies Depression scale [CES-D]) 12 months after aSAH (OR: 2.2, 95% CI: 1.0–4.9; p = 0.044) compared with those without VSP in a prospective study of 216 patients [48].
A prospective, older study reported that VSP was the only complication that caused significant impairment in terms of patients’ temperament and emotionality at 12 months after SAH (p < 0.001) [51].
Carer Burden
No studies were identified that used VSP-specific data to assess carer burden in aSAH patients.
General Patient Population after aSAH
For the broader aSAH population, poorer HRQoL (measured by the SF-36) was reported for up to 10–12 years after aSAH [24, 54, 55, 57], and aSAH patients rated as cognitively impaired due to VSP-related DCI at 12 months had significantly worse SIP physical, psychosocial, and total scores compared with patients who were unimpaired (all p ≤ 0.01) [48, 58]. Depression and emotional state were also severely affected in patients after aSAH [24, 48, 51, 57], with approximately half of SAH survivors experiencing clinically significant depressive symptoms at some point during the first year of recovery [48], and long-term clinically significant anxiety, depression, and posttraumatic stress symptoms reported at 10–12 years after aSAH [57]. After correcting for demographic factors, depressed mood (measured by the CES-D) predicted HRQoL (measured by SIP) over and above cognitive status and SAH severity [48]. Carer burden, measured using the caregiver disruption assessment form of the Neuropsychiatric Inventory Questionnaire, was also reported as very severe in 5 of 14 general aSAH patients who had survived aSAH 10–12 years previously [57].
Predictors of Economic and Humanistic Outcomes
Severity of VSP
Overall, comparative and regression-based analyses from multiple studies [11, 27, 32, 35, 41, 43, 52, 63] showed that the severity of VSP was a critical predictor of post-aSAH economic burden in terms of healthcare resource utilization (HCRU) and direct and indirect costs. For example, a significant relationship between the severity of aVSP and both ICU LOS (p < 0.0001) and total hospital LOS (p = 0.008) was reported in a post hoc analysis of the CONSCIOUS-1 trial [27]. Similarly, longer hospital and ICU stays were reported to be more pronounced for patients with symptomatic VSP or DCI than those with aVSP or TCD-defined VSP, although this was not tested statistically [11]. Cerebral VSP was significantly associated with 30-day readmission (p = 0.016) [52], and endovascular treatment of VSP was associated with greater odds of all-cause hospital readmission within 60 days (OR: 5.1; p = 0.001) and within 180 days (OR: 3.8; p = 0.001) [41]. In terms of direct costs, multivariate analysis using the US Value Driven Outcomes database found that VSP severity was an independent predictor of cost (β = 0.1, p = 0.02) as were the number of angiograms (β = 0.1, p = 0.02), disposition (β = 0.2, p = 0.001), year of treatment (β = 0.2, p < 0.001), procedure (β = 0.3, p < 0.001), and hospital LOS (β = 0.5, p < 0.001) [63]. The association between VSP severity and economic burden also appears to be significant independently of whether VSP was defined by TCD velocity or the presence of DCI, with the presence of either type of VSP significantly associated with higher inpatient costs and longer hospital stay [43].
VSP severity was also associated with HRQoL outcomes, with studies showing that the severity of VSP or the presence of DCI was a significant predictor of poorer HRQoL, independent of the tool used. Specifically, patients who experienced 1 or more days of VSP after aSAH had poorer HRQoL measured by the SF-36 [38], and patients with severe or moderate aVSP reported worse HRQoL measured by the EQ-5D [27] than patients with no VSP or aVSP, respectively. In addition, patients with symptomatic VSP or DCI had worse HRQoL measured by the SIP, whereas patients with aVSP or TCD-defined VSP did not [11], and patients with symptomatic VSP were more likely to have depressed mood measured by the CES-D at 12 months after aSAH [48]. In contrast, only one small study (n = 146) found no difference in HRQoL outcomes based on SF-36 scores at a mean follow-up of 6.5 years between patients who developed VSP-related DCI and those who did not [61], but it seems that patients with VSP-related DCI had significantly higher odds of global cognitive impairment at 12 months after aSAH [58].
Other Predictors
Other significant predictors of higher economic burden included older age [42, 47], poorer Glasgow Outcome Scale Extended scores [27], type of aneurysm treatment (i.e., clipping) [42], number of angiograms and CT scans [32], and type of hospital where patients were treated (i.e., hospitals performing angioplasty versus not) [47].
Higher HRQoL burden was generally predicted by several indicators of initial clinical status, including poorer GCS [55, 61], WFNS [55, 61], and Hunt–Hess [54, 55, 61] scores, and poorer Fisher grade [54, 55] (although a nonsignificant association with Fisher grade was reported in one study [61]).
Discussion
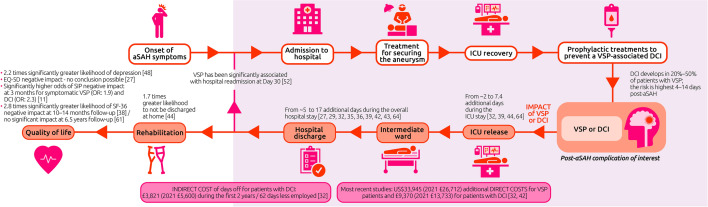
To our knowledge, this is the first SLR summarizing the economic and humanistic burden associated with VSP after aSAH. As expected, the burden of VSP and its related complications is huge, with a severe negative impact identified for both economic (direct and indirect costs, HCRU) and humanistic aspects, which not only affect the short term but may also extend over the longer term. A timeline to depict the patient journey is presented in Fig. 3.
Fig. 3.
Summary of the economic and humanistic burden for patients with VSP-related complications (i.e., VSP, DCI, and cerebral infarction) compared with patients without. The economic and humanistic burden evidences (highlighted in orange) are represented along the patient journey timeline. aSAH, aneurysmal subarachnoid hemorrhage; DCI, delayed cerebral ischemia; EQ-5D, EuroQoL 5-Dimensions tool for measuring quality of life; ICU, intensive care unit; OR, odds ratio; SF-36, 36-item Short-Form Health Survey; SIP, Sickness Impact Profile; VSP, cerebral vasospasm
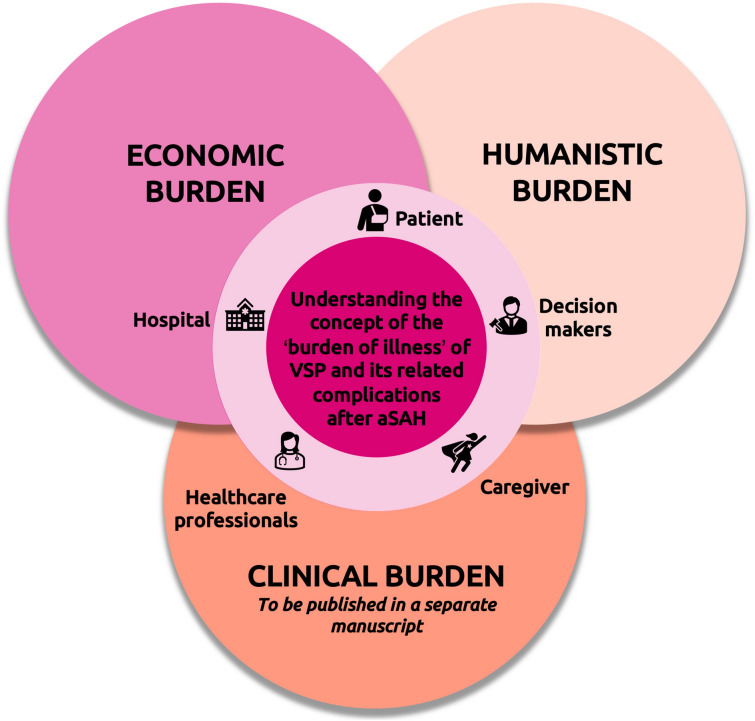
Our SLR advances knowledge on the concept of the “burden of illness” [67] of VSP and its related complications after aSAH, and develops the scientific basis for assessing the value of new targeted treatments for this condition (Fig. 4). Of note, the authors are currently preparing a separate publication that summarizes the findings from an SLR on the clinical burden of VSP and its related complications after aSAH, which will enable the synthesis of evidence across the three pillars (i.e., economic, humanistic, and clinical) of the “burden of illness”.
Fig. 4.
Three-petal schematic representing the three dimensions of the “burden of illness” of VSP and its related complications (i.e., delayed cerebral ischemia and cerebral infarction) after aSAH and impacted stakeholders (flower etamins). aSAH, aneurysmal subarachnoid hemorrhage; VSP, cerebral vasospasm
The substantial economic burden linked to VSP or VSP-related complications in the form of DCI was predominantly driven by longer ICU and hospital stays, readmissions, and additional costs of imaging and investigations associated with the treatment and management of patients. This was evident from studies showing a significantly higher total number of days spent in the ICU [11, 27, 32, 35, 39, 44, 64] and in hospital [11, 27, 29, 32, 35, 36, 39, 42, 43, 64] for patients with VSP or DCI compared with those without (2.1–7.4 and 4.7–17 additional days spent in the ICU and hospital, respectively; all p < 0.05 where reported). Conversely, similar hospitalization times for VSP and non-VSP aSAH patients were reported in the experimental arm of one RCT, although only 24 patients were included in the treatment group and this outcome should be viewed with caution [29]. Notably, the association between VSP and length of hospitalization remained significant independent of whether VSP was defined by TCD velocity or the presence of DCI [43], which may support the comparability of findings across the studies.
Patients who survive aSAH are at risk for complications that may require readmission to hospital [50, 52, 56]. Indeed, the occurrence of cerebral VSP after aSAH was significantly associated with hospital readmissions within 30 days of discharge [52]. VSP patients were also more likely to be adversely discharged (defined as discharge other than home), although this was not statistically significant [44].
A substantial direct and indirect economic burden of VSP and DCI was also identified, with a significantly higher total cost burden of US$33,945 (2021 £26,712) for patients with VSP [42] and £9370 (2021 £13,733) for patients with DCI [32], and, for patients with DCI, a significant reduction in the mean number of days in paid employment and an increased mean cost of £3821 (2021 £5600) for days off work over 2 years [32], compared with patients without VSP or DCI. Persistently higher inpatient costs, reflecting the provision of more intensive clinical care and longer hospital stays, were also reported for both TCD-defined VSP and DCI, despite adjustment for potential confounders [43]. Moreover, a post hoc analysis of the ISAT database showed that the devastating effects of aSAH are compounded if DCI also occurs, leading to significantly higher healthcare costs, driven in part by longer hospital stays and slower resumption of paid employment [32].
The severity of VSP appears to be a significant predictor of post-aSAH economic burden driving the difference in HCRU and costs between patients who experienced VSP or DCI after aSAH and those who did not [27, 32, 35, 41, 43, 52, 63]. In terms of direct costs, an analysis using the US Value Driven Outcomes database [63] identified VSP severity as a significant factor in the cost of care for patients with aSAH, although aneurysm procedure type and hospital LOS had larger β values.
The association between VSP severity and economic burden also appears to be significant independent of the way VSP has been diagnosed. This is shown by one study [43], which found that the presence of either TCD-defined VSP or DCI was significantly associated with higher inpatient costs and longer hospital stays. Both severe VSP, measured by a maximal TCD velocity of ≥ 180 cm/s, and DCI were associated with higher daily costs of inpatient care, reflecting the provision of more intensive care. Furthermore, the effect of VSP severity on total costs persisted even after adjusting for the positive association between inpatient costs and LOS.
Limited evidence was identified for humanistic outcomes, but these patients, independent of VSP status, tend, in general, to suffer from poor HRQoL with long-lasting burden. Studies show that VSP and DCI are significant risk factors for depressed mood and global cognitive impairment at 12 months, respectively. Specifically, patients with symptomatic VSP had a greater than two times higher odds of experiencing depressed mood during the first year of recovery after aSAH compared with those without [48], and patients who experienced VSP-related DCI experienced poorer cognitive orientation, attention, language, long-term memory, motor function, and verbal memory 1 year after aSAH, compared with patients without DCI [58]. In addition, one or more DVS was found to be a major risk factor for poor HRQoL, with an almost three times significantly higher likelihood of SF-36 negative impact at 10–14 months despite adjustment for confounding factors [38].
VSP-related complications after aSAH, including DCI and CI, can cause further deterioration of a patient’s neurological condition, with significant impairment in HRQoL measured by the SIP at 3 months reported in patients with DCI or symptomatic VSP, but not in patients with aVSP or TCD-defined VSP, after adjustment for disease and demographic factors [11]. Moreover, problems of psychosocial adaptation can be debilitating for patients who experience VSP after aSAH, with higher impairment on measures of temperament, emotion, and physical capabilities reported after 1 year [51].
Similar to economic findings, patients with severe VSP reported worst HRQoL and rated their health state lower than those with no VSP, although the association between the presence of DCI and patient-reported outcome measures was not consistent across all studies. Specifically, VSP severity predicted significantly poorer HRQoL (SF-36, EQ-5D, and SIP) and depressed mood at 12 months [11, 27, 38, 48], with the exception of one study that reported similar HRQoL outcomes (SF-36) in patients with, compared with those without, VSP-related DCI [61].
The broader aSAH population, which included patients with VSP or DCI, experienced similar economic burden to patients with VSP and its related complications. This can be seen for HCRU in terms of ICU and hospital LOS as well as LOS after readmission. In terms of direct and indirect costs, the mean total cost of treatment varied widely depending on the severity of aSAH. The uptake of paid employment following aSAH was also disrupted, causing considerable indirect economic burden [24, 51, 57].
Poorer HRQoL was also evident in this patient population, with negative impacts on HRQoL [24, 54, 55, 57], depression, and emotional state [48, 57] that were long-lasting. Considerable disruption to the caregiver was also evident [57].
One of the major strengths of this SLR is the systematic approach used to identify and collect the evidence in this disease area without restrictions on publication date, language, or country. Almost half of the included studies collected data after 2010, which may reflect current clinical practice (costs, HCRU) and changes in treatment management compared with studies that included older data. However, limitations of this SLR were also evident as most of the evidence retrieved was collected in single centers, thus precluding the generalizability of findings across different settings. In addition, evidence on humanistic outcomes for patients with VSP-related complications was sparse, restricting a crucial part of our understanding on the humanistic burden of the disease on patients and their carers.
In this SLR, most of the evidence was retrieved from observational studies, which may reflect routine clinical care. However, this SLR is limited by the variation across the studies in terms of the period of data coverage and length of data collection, patient selection criteria (i.e., age, severity at admission), and the definition of VSP, which pose challenges when comparing findings across studies. In addition, local hospital settings, VSP management, and unit costs of resources differed markedly across healthcare systems; therefore, caution is required when the findings from these studies are generalized to other settings. Studies also varied in terms of design and patient characteristics, which precluded direct metaanalysis of results.
A major limitation of this SLR is the complexity of factors contributing to DCI after aSAH, and the inability to differentiate between all-cause DCI and VSP-related DCI. Although both early brain injury and VSP can contribute to DCI development, VSP is likely to be the main causal factor for DCI.
This SLR has also identified several data gaps in the literature, highlighting the need for further research to assess the impact of VSP and its related complications on readmissions, quality of life, return to full-time employment, and the long-term impact of VSP.
Conclusions
Overall, the evidence showed that VSP and its related complications after aSAH severely impacted patients. There remains an urgent need to improve our understanding of the burden of VSP and its impact on HRQoL, its indirect burden on caregivers and work productivity, and its long-term impact. Understanding the concept of the “burden of illness” of VSP and its related complications after aSAH will provide the scientific basis for assessing novel targeted treatments. Ultimately, the development and approval of therapeutics that target and prevent VSP and its downstream complications will greatly enhance the armamentarium currently available to treating physicians.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
Funding
The Systematic Literature Review, the journal’s Rapid Service Fee, and medical writing services were funded by Idorsia Pharmaceuticals Ltd, Switzerland.
Medical Writing Assistance
Medical writing assistance was provided by Melanie Gatt (PhD), an independent medical writer. Support for this assistance was funded by Idorsia Pharmaceuticals Ltd.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Juliette C. Thompson, Eric J. Manalastas, and Grammati Sarri. All authors commented on all versions of the manuscript, and read and approved the final manuscript.
Prior Publication
The results of this systematic literature review were presented in a virtual format at Virtual ISPOR Europe 2021, Copenhagen, Denmark, November 30 to December 3, 2021 (POSA268, https://doi.org/10.1016/j.jval.2021.11.835).
Disclosures
Juliette C. Thompson, Eric J. Manalastas, Grammati Sarri, and Neil Hawkins from Visible Analytics Ltd received fees from Idorsia Pharmaceuticals Ltd for the conduct of the systematic literature review. François-Xavier Chalet is an employee of Idorsia Pharmaceutical Ltd. Darren A. Talbot is an employee and shareholder of Idorsia Pharmaceuticals Ltd. Since completion of the manuscript, Grammati Sarri has changed affiliation to visiting Lead Scientist, Cytel, London, UK.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
References
- 1.Macdonald RL, Schweizer TA. Spontaneous subarachnoid haemorrhage. Lancet. 2017;389(10069):655–666. doi: 10.1016/S0140-6736(16)30668-7. [DOI] [PubMed] [Google Scholar]
- 2.Aneurysmal DS, Hemorrhage S. Aneurysmal subarachnoid hemorrhage. J Neurosurg Anesthesiol. 2015;27(3):222–240. doi: 10.1097/ANA.0000000000000130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.de Rooij NK, Linn FH, van der Plas JA, Algra A, Rinkel GJ. Incidence of subarachnoid haemorrhage: a systematic review with emphasis on region, age, gender and time trends. J Neurol Neurosurg Psychiatry. 2007;78(12):1365–1372. doi: 10.1136/jnnp.2007.117655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Etminan N, Chang HS, Hackenberg K, de Rooij NK, Vergouwen MDI, Rinkel GJE, et al. Worldwide incidence of aneurysmal subarachnoid hemorrhage according to region, time period, blood pressure, and smoking prevalence in the population: a systematic review and meta-analysis. JAMA Neurol. 2019;76(5):588–597. doi: 10.1001/jamaneurol.2019.0006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bauer AM, Rasmussen PA. Treatment of intracranial vasospasm following subarachnoid hemorrhage. Front Neurol. 2014;5:72. doi: 10.3389/fneur.2014.00072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ingall T, Asplund K, Mahonen M, Bonita R. A multinational comparison of subarachnoid hemorrhage epidemiology in the WHO MONICA stroke study. Stroke. 2000;31(5):1054–1061. doi: 10.1161/01.str.31.5.1054. [DOI] [PubMed] [Google Scholar]
- 7.Hughes JD, Bond KM, Mekary RA, Dewan MC, Rattani A, Baticulon R, et al. Estimating the global incidence of aneurysmal subarachnoid hemorrhage: a systematic review for central nervous system vascular lesions and meta-analysis of ruptured aneurysms. World Neurosurg. 2018;115:430–447. doi: 10.1016/j.wneu.2018.03.220. [DOI] [PubMed] [Google Scholar]
- 8.Connolly ES, Jr, Rabinstein AA, Carhuapoma JR, Derdeyn CP, Dion J, Higashida RT, et al. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2012;43(6):1711–1737. doi: 10.1161/STR.0b013e3182587839. [DOI] [PubMed] [Google Scholar]
- 9.Stienen MN, Germans M, Burkhardt JK, Neidert MC, Fung C, Bervini D, et al. Predictors of in-hospital death after aneurysmal subarachnoid hemorrhage: analysis of a nationwide database (Swiss SOS [Swiss study on aneurysmal subarachnoid hemorrhage]) Stroke. 2018;49(2):333–340. doi: 10.1161/STROKEAHA.117.019328. [DOI] [PubMed] [Google Scholar]
- 10.Dorsch NW, King MT. A review of cerebral vasospasm in aneurysmal subarachnoid haemorrhage part I: incidence and effects. J Clin Neurosci. 1994;1(1):19–26. doi: 10.1016/0967-5868(94)90005-1. [DOI] [PubMed] [Google Scholar]
- 11.Frontera JA, Fernandez A, Schmidt JM, Claassen J, Wartenberg KE, Badjatia N, et al. Defining vasospasm after subarachnoid hemorrhage: what is the most clinically relevant definition? Stroke. 2009;40(6):1963–1968. doi: 10.1161/STROKEAHA.108.544700. [DOI] [PubMed] [Google Scholar]
- 12.Macdonald RL, Kassell NF, Mayer S, Ruefenacht D, Schmiedek P, Weidauer S, et al. Clazosentan to overcome neurological ischemia and infarction occurring after subarachnoid hemorrhage (CONSCIOUS-1): randomized, double-blind, placebo-controlled phase 2 dose-finding trial. Stroke. 2008;39(11):3015–3021. doi: 10.1161/STROKEAHA.108.519942. [DOI] [PubMed] [Google Scholar]
- 13.Macdonald RL. Delayed neurological deterioration after subarachnoid haemorrhage. Nat Rev Neurol. 2014;10(1):44–58. doi: 10.1038/nrneurol.2013.246. [DOI] [PubMed] [Google Scholar]
- 14.Crowley RW, Medel R, Dumont AS, Ilodigwe D, Kassell NF, Mayer SA, et al. Angiographic vasospasm is strongly correlated with cerebral infarction after subarachnoid hemorrhage. Stroke. 2011;42(4):919–923. doi: 10.1161/STROKEAHA.110.597005. [DOI] [PubMed] [Google Scholar]
- 15.Geraghty JR, Testai FD. Delayed cerebral ischemia after subarachnoid hemorrhage: beyond vasospasm and towards a multifactorial pathophysiology. Curr Atheroscler Rep. 2017;19(12):50. doi: 10.1007/s11883-017-0690-x. [DOI] [PubMed] [Google Scholar]
- 16.Dankbaar JW, Rijsdijk M, van der Schaaf IC, Velthuis BK, Wermer MJ, Rinkel GJ. Relationship between vasospasm, cerebral perfusion, and delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. Neuroradiology. 2009;51(12):813–819. doi: 10.1007/s00234-009-0575-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rabinstein AA, Weigand S, Atkinson JL, Wijdicks EF. Patterns of cerebral infarction in aneurysmal subarachnoid hemorrhage. Stroke. 2005;36(5):992–997. doi: 10.1161/01.STR.0000163090.59350.5a. [DOI] [PubMed] [Google Scholar]
- 18.Daou BJ, Koduri S, Thompson BG, Chaudhary N, Pandey AS. Clinical and experimental aspects of aneurysmal subarachnoid hemorrhage. CNS Neurosci Ther. 2019;25(10):1096–1112. doi: 10.1111/cns.13222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Muehlschlegel S. Subarachnoid Hemorrhage. Continuum (Minneap Minn) 2018;24(6):1623–1657. doi: 10.1212/CON.0000000000000679. [DOI] [PubMed] [Google Scholar]
- 20.Vergouwen MD, Vermeulen M, van Gijn J, Rinkel GJ, Wijdicks EF, Muizelaar JP, et al. Definition of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage as an outcome event in clinical trials and observational studies: proposal of a multidisciplinary research group. Stroke. 2010;41(10):2391–2395. doi: 10.1161/STROKEAHA.110.589275. [DOI] [PubMed] [Google Scholar]
- 21.Dorsch N. A clinical review of cerebral vasospasm and delayed ischaemia following aneurysm rupture. Acta Neurochir Suppl. 2011;110(Pt 1):5–6. doi: 10.1007/978-3-7091-0353-1_1. [DOI] [PubMed] [Google Scholar]
- 22.Hollingworth M, Jamjoom AAB, Bulters D, Patel HC. How is vasospasm screening using transcranial Doppler associated with delayed cerebral ischemia and outcomes in aneurysmal subarachnoid hemorrhage? Acta Neurochir (Wien) 2019;161(2):385–392. doi: 10.1007/s00701-018-3765-8. [DOI] [PubMed] [Google Scholar]
- 23.Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. J Clin Epidemiol. 2021;134:178–189. doi: 10.1016/j.jclinepi.2021.03.001. [DOI] [PubMed] [Google Scholar]
- 24.Vetkas A, Lepik T, Eilat T, Ratsep T, Asser T. Emotional health and quality of life after aneurysmal subarachnoid hemorrhage. Acta Neurochir (Wien) 2013;155(6):1107–1114. doi: 10.1007/s00701-013-1683-3. [DOI] [PubMed] [Google Scholar]
- 25.Vetkas A, Prans E, Koks S, Ratsep T, Asser T. Aneurysmal subarachnoid haemorrhage: effect of CRHR1 genotype on mental health-related quality of life. Sci Rep. 2020;10(1):724. doi: 10.1038/s41598-020-57527-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fountas KN, Machinis TG, Robinson JS, Sevin C, Fezoulidis NI, Castresana M, et al. The role of magnesium sulfate in the treatment of vasospasm in patients with spontaneous subarachnoid haemorrhage. Acta Neurochir Suppl. 2008;104:269–273. doi: 10.1007/978-3-211-75718-5_55. [DOI] [Google Scholar]
- 27.Macdonald RL, Hunsche E, Schuler R, Wlodarczyk J, Mayer SA. Quality of life and healthcare resource use associated with angiographic vasospasm after aneurysmal subarachnoid hemorrhage. Stroke. 2012;43(4):1082–1088. doi: 10.1161/STROKEAHA.111.634071. [DOI] [PubMed] [Google Scholar]
- 28.Mahajan C, Chouhan RS, Rath GP, Dash HH, Suri A, Chandra PS, et al. Effect of intraoperative brain protection with propofol on postoperative cognition in patients undergoing temporary clipping during intracranial aneurysm surgery. Neurol India. 2014;62(3):262–268. doi: 10.4103/0028-3886.136908. [DOI] [PubMed] [Google Scholar]
- 29.Prevedello DM, Cordeiro JG, de Morais AL, Saucedo NSJ, Chen IB, Araújo JC. Magnesium sulfate: role as possible attenuating factor in vasospasm morbidity. Surg Neurol. 2006;65(1):A1:14–S1:20. doi: 10.1016/j.surneu.2005.11.035. [DOI] [PubMed] [Google Scholar]
- 30.Raval RN, Small O, Magsino K, Chakravarthy V, Austin B, Applegate R, et al. Remote ischemic pre-conditioning in subarachnoid hemorrhage: a prospective pilot trial. Neurocrit Care. 2021;34(3):968–973. doi: 10.1007/s12028-020-01122-y. [DOI] [PubMed] [Google Scholar]
- 31.Ren C, Xu H, Xu G, Liu L, Liu G, Zhang Z, et al. Effect of intraoperative infusion of dexmedetomidine on postoperative recovery in patients undergoing endovascular interventional therapies: a prospective, randomized, controlled trial. Brain Behav. 2019;9(7):e01317. doi: 10.1002/brb3.1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rivero-Arias O, Wolstenholme J, Gray A, Molyneux AJ, Kerr RS, Yarnold JA, et al. The costs and prognostic characteristics of ischaemic neurological deficit due to subarachnoid haemorrhage in the United Kingdom. Evidence from the MRC International Subarachnoid Aneurysm Trial. J Neurol. 2009;256(3):364–373. doi: 10.1007/s00415-009-0034-z. [DOI] [PubMed] [Google Scholar]
- 33.Senbokuya N, Kinouchi H, Kanemaru K, Ohashi Y, Fukamachi A, Yagi S, et al. Effects of cilostazol on cerebral vasospasm after aneurysmal subarachnoid hemorrhage: a multicenter prospective, randomized, open-label blinded end point trial. J Neurosurg. 2013;118(1):121–130. doi: 10.3171/2012.9.JNS12492. [DOI] [PubMed] [Google Scholar]
- 34.Zwienenberg-Lee M, Hartman J, Rudisill N, Madden LK, Smith K, Eskridge J, et al. Effect of prophylactic transluminal balloon angioplasty on cerebral vasospasm and outcome in patients with Fisher grade III subarachnoid hemorrhage: results of a phase II multicenter, randomized, clinical trial. Stroke. 2008;39(6):1759–1765. doi: 10.1161/STROKEAHA.107.502666. [DOI] [PubMed] [Google Scholar]
- 35.Abulhasan YB, Alabdulraheem N, Schiller I, Rachel SP, Dendukuri N, Angle MR, et al. Health care-associated infections after subarachnoid hemorrhage. World Neurosurg. 2018;115:e393–e403. doi: 10.1016/j.wneu.2018.04.061. [DOI] [PubMed] [Google Scholar]
- 36.Alaraj A, Hussein AE, Esfahani DR, Amin-Hanjani S, Aletich VA, Charbel FT. Reducing length of stay in aneurysmal subarachnoid hemorrhage: a three year institutional experience. J Clin Neurosci. 2017;42:66–70. doi: 10.1016/j.jocn.2017.03.049. [DOI] [PubMed] [Google Scholar]
- 37.Alay GH, Postaci NA, Aytac I, Acar F, Ornek D, Dikmen B. Retrospective analysis of anesthetic management in the cerebral aneurysm treatment: issues in the course of endovascular versus surgical treatment. Kuwait Med J. 2020;52(2):169–174. [Google Scholar]
- 38.Ali A, Tanirgan G, Sabanci PA, Sivrikoz N, Abdullah T, Sencer A, et al. Relation of gray-white matter ratio with long-term cognitive functions and quality of life in patients with mild to moderate aneurysmal subarachnoid hemorrhage: a prospective observational study. Acta Neurochir (Wien) 2018;160(1):181–189. doi: 10.1007/s00701-017-3374-y. [DOI] [PubMed] [Google Scholar]
- 39.Badjatia N, Topcuoglu MA, Buonanno FS, Smith EE, Nogueira RG, Rordorf GA, et al. Relationship between hyperglycemia and symptomatic vasospasm after subarachnoid hemorrhage. Crit Care Med. 2005;33(7):1603–1609. doi: 10.1097/01.ccm.0000168054.60538.2b. [DOI] [PubMed] [Google Scholar]
- 40.Bercker S, Winkelmann T, Busch T, Laudi S, Lindner D, Meixensberger J. Hydroxyethyl starch for volume expansion after subarachnoid haemorrhage and renal function: results of a retrospective analysis. PLoS ONE. 2018;13(2):e0192832. doi: 10.1371/journal.pone.0192832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chatrath A, Soldozy S, Sokolowski JD, Burke RM, Schultz JG, Rannigan ZC, et al. Endovascular and surgical treatment is predictive of readmission risk after aneurysmal subarachnoid hemorrhage. World Neurosurg. 2020;142:e494–e501. doi: 10.1016/j.wneu.2020.07.079. [DOI] [PubMed] [Google Scholar]
- 42.Chotai S, Patel PD, Liles C, Chen H, Shannon CN, Froehler MT, et al. Impact of neurovascular comorbidities and complications on outcomes after procedural management of intracranial aneurysm: part 2, ruptured intracranial aneurysm. World Neurosurg. 2021;146:e270–e312. doi: 10.1016/j.wneu.2020.10.091. [DOI] [PubMed] [Google Scholar]
- 43.Chou CH, Reed SD, Allsbrook JS, Steele JL, Schulman KA, Alexander MJ. Costs of vasospasm in patients with aneurysmal subarachnoid hemorrhage. Neurosurgery. 2010;67(2):345–351. doi: 10.1227/01.NEU.0000371980.08391.71. [DOI] [PubMed] [Google Scholar]
- 44.Drazin D, Rosner J, Nuno M, Alexander MJ, Schievink WI, Palestrant D, et al. Type of admission is associated with outcome of spontaneous subarachnoid hemorrhage. Int J Stroke. 2015;10(4):529–533. doi: 10.1111/ijs.12005. [DOI] [PubMed] [Google Scholar]
- 45.Harris L, Hill CS, Elliot M, Fitzpatrick T, Ghosh A, Vindlacheruvu R. Comparison between outcomes of endovascular and surgical treatments of ruptured anterior communicating artery aneurysms. Br J Neurosurg. 2021;35(3):313–318. doi: 10.1080/02688697.2020.1812517. [DOI] [PubMed] [Google Scholar]
- 46.Hoffman H, Jalal MS, Chin LS. A propensity score-matched comparison of readmission rates associated with microsurgical clipping and endovascular treatment of ruptured intracranial aneurysms. J Stroke Cerebrovasc Dis. 2020;29(5):104696. doi: 10.1016/j.jstrokecerebrovasdis.2020.104696. [DOI] [PubMed] [Google Scholar]
- 47.Khatri R, Tariq N, Vazquez G, Suri MF, Ezzeddine MA, Qureshi AI. Outcomes after nontraumatic subarachnoid hemorrhage at hospitals offering angioplasty for cerebral vasospasm: a national level analysis in the United States. Neurocrit Care. 2011;15(1):34–41. doi: 10.1007/s12028-010-9423-5. [DOI] [PubMed] [Google Scholar]
- 48.Kreiter KT, Rosengart AJ, Claassen J, Fitzsimmons BF, Peery S, Du YE, et al. Depressed mood and quality of life after subarachnoid hemorrhage. J Neurol Sci. 2013;335(1–2):64–71. doi: 10.1016/j.jns.2013.08.024. [DOI] [PubMed] [Google Scholar]
- 49.Morgan MK, Jonker B, Finfer S, Harrington T, Dorsch NW. Aggressive management of aneurysmal subarachnoid haemorrhage based on a papaverine angioplasty protocol. J Clin Neurosci. 2000;7(4):305–308. doi: 10.1054/jocn.1999.0224. [DOI] [PubMed] [Google Scholar]
- 50.Niskanen M, Koivisto T, Ronkainen A, Rinne J, Ruokonen E. Resource use after subarachnoid hemorrhage: comparison between endovascular and surgical treatment. Neurosurgery. 2004;54(5):1081–1086. doi: 10.1227/01.neu.0000119350.80122.43. [DOI] [PubMed] [Google Scholar]
- 51.Ogden JA, Mee EW, Henning M. A prospective study of psychosocial adaptation following subarachnoid haemorrhage. Neuropsychol Rehabil. 1994;4(1):7–30. doi: 10.1080/09602019408401453. [DOI] [Google Scholar]
- 52.Rumalla K, Smith KA, Arnold PM, Mittal MK. Subarachnoid hemorrhage and readmissions: national rates, causes, risk factors, and outcomes in 16,001 hospitalized patients. World Neurosurg. 2018;110:e100–e111. doi: 10.1016/j.wneu.2017.10.089. [DOI] [PubMed] [Google Scholar]
- 53.Sakr Y, Dunisch P, Santos C, Matthes L, Zeidan M, Reinhart K, et al. Poor outcome is associated with less negative fluid balance in patients with aneurysmal subarachnoid hemorrhage treated with prophylactic vasopressor-induced hypertension. Ann Intensive Care. 2016;6(1):25. doi: 10.1186/s13613-016-0128-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Scharbrodt W, Stein M, Schreiber V, Boker DK, Oertel MF. The prediction of long-term outcome after subarachnoid hemorrhage as measured by the Short Form-36 Health Survey. J Clin Neurosci. 2009;16(11):1409–1413. doi: 10.1016/j.jocn.2009.01.011. [DOI] [PubMed] [Google Scholar]
- 55.Soehle M, Chatfield DA, Czosnyka M, Kirkpatrick PJ. Predictive value of initial clinical status, intracranial pressure and transcranial Doppler pulsatility after subarachnoid haemorrhage. Acta Neurochir (Wien) 2007;149(6):575–583. doi: 10.1007/s00701-007-1149-6. [DOI] [PubMed] [Google Scholar]
- 56.Sokolowski JD, Chen CJ, Soldozy S, Mastorakos P, Burke RM, Nguyen JM, et al. Nimodipine after aneurysmal subarachnoid hemorrhage: fourteen-day course for patients that meet criteria for early hospital discharge. Clin Neurol Neurosurg. 2021;200:106299. doi: 10.1016/j.clineuro.2020.106299. [DOI] [PubMed] [Google Scholar]
- 57.Sousa L, Antunes A, Mendes T, Reimao S, Neto LL, Campos J. Long-term neuropsychiatric and neuropsychological sequelae of endovascularly treated aneurysmal subarachnoid hemorrhage. Acta Med Port. 2019;32(11):706–713. doi: 10.20344/amp.10894. [DOI] [PubMed] [Google Scholar]
- 58.Springer MV, Schmidt JM, Wartenberg KE, Frontera JA, Badjatia N, Mayer SA. Predictors of global cognitive impairment 1 year after subarachnoid hemorrhage. Neurosurgery. 2009;65(6):1043–1050. doi: 10.1227/01.NEU.0000359317.15269.20. [DOI] [PubMed] [Google Scholar]
- 59.Strickland BA, Mert M, Ravina K, Chartrain A, Rennert RC, Bakhsheshian J, et al. Discrepancy in neurologic outcomes following aneurysmal subarachnoid hemorrhage as a function of socioeconomic class. World Neurosurg. 2020;138:e787–e794. doi: 10.1016/j.wneu.2020.03.087. [DOI] [PubMed] [Google Scholar]
- 60.Suarez JI, Shannon L, Zaidat OO, Suri MF, Singh G, Lynch G, et al. Effect of human albumin administration on clinical outcome and hospital cost in patients with subarachnoid hemorrhage. J Neurosurg. 2004;100(4):585–590. doi: 10.3171/jns.2004.100.4.0585. [DOI] [PubMed] [Google Scholar]
- 61.Szmuda T, Słoniewski P, Waszak PM, Kindrachuk M, Olijewski W. Short- and long-term outcome of surgically treated ruptured internal carotid artery aneurysms. Acta Neuropsychologica. 2013;11(4):403–417. doi: 10.5604/17307503.1090469. [DOI] [Google Scholar]
- 62.Taylor CJ, Robertson F, Brealey D, O'Shea F, Stephen T, Brew S, et al. Outcome in poor grade subarachnoid hemorrhage patients treated with acute endovascular coiling of aneurysms and aggressive intensive care. Neurocrit Care. 2011;14(3):341–347. doi: 10.1007/s12028-010-9377-7. [DOI] [PubMed] [Google Scholar]
- 63.Wilde H, Twitchell S, Reese J, Guan J, Eli IM, Karsy M, et al. Evaluation of disease severity and treatment intensity as cost drivers for ruptured intracranial aneurysms. Acta Neurochir (Wien) 2020;162(1):157–167. doi: 10.1007/s00701-019-04153-3. [DOI] [PubMed] [Google Scholar]
- 64.Yundt KD, Dacey RG, Jr, Diringer MN. Hospital resource utilization in the treatment of cerebral aneurysms. J Neurosurg. 1996;85(3):403–409. doi: 10.3171/jns.1996.85.3.0403. [DOI] [PubMed] [Google Scholar]
- 65.Zaidat OO, Ionita CC, Hussain SI, Alexander MJ, Friedman AH, Graffagnino C. Impact of ruptured cerebral aneurysm coiling and clipping on the incidence of cerebral vasospasm and clinical outcome. J Neuroimaging. 2009;19(2):144–149. doi: 10.1111/j.1552-6569.2008.00285.x. [DOI] [PubMed] [Google Scholar]
- 66.Fisher CM, Kistler JP, Davis JM. Relation of cerebral vasospasm to subarachnoid hemorrhage visualized by computerized tomographic scanning. Neurosurgery. 1980;6(1):1–9. doi: 10.1227/00006123-198001000-00001. [DOI] [PubMed] [Google Scholar]
- 67.National Institute for Health and Care Excellence (NICE) Value Based Assessment of Health Technologies (Consultation Paper). 2022. https://www.nice.org.uk/Media/Default/About/what-we-do/NICE-guidance/NICE-technology-appraisals/VBA-TA-Methods-Guide-for-Consultation.pdf. Accessed 17 Jan 2022.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.