Abstract
Chronic pain (CP) represents a socio-economic burden for affected patients along with therapeutic challenges for currently available therapies. When conventional therapies fail, modulation of the affective pain matrix using reversible deep brain stimulation (DBS) or targeted irreversible thalamotomy by stereotactic radiosurgery (SRS) and magnetic resonance (MR)-guided focused ultrasound (MRgFUS) appear to be considerable treatment options. We performed a literature search for clinical trials targeting the affective pain circuits (thalamus, anterior cingulate cortex [ACC], ventral striatum [VS]/internal capsule [IC]). PubMed, Ovid, MEDLINE and Scopus were searched (1990–2021) using the terms “chronic pain”, “deep brain stimulation”, “stereotactic radiosurgery”, “radioneuromodulation”, “MR-guided focused ultrasound”, “affective pain modulation”, “pain attention”. In patients with CP treated with DBS, SRS or MRgFUS the somatosensory thalamus and periventricular/periaquaeductal grey was the target of choice in most treated subjects, while affective pain transmission was targeted in a considerably lower number (DBS, SRS) consisting of the following nodi of the limbic pain matrix: the anterior cingulate cortex; centromedian-parafascicularis of the thalamus, pars posterior of the central lateral nucleus and internal capsule/ventral striatum. Although DBS, SRS and MRgFUS promoted a meaningful and sustained pain relief, an effective, evidence-based comparative analysis is biased by heterogeneity of the observation period varying between 3 months and 5 years with different stimulation patterns (monopolar/bipolar contact configuration; frequency 10–130 Hz; intensity 0.8–5 V; amplitude 90–330 μs), source and occurrence of lesioning (radiation versus ultrasound) and chronic pain ethology (poststroke pain, plexus injury, facial pain, phantom limb pain, back pain). The advancement of neurotherapeutics (MRgFUS) and novel DBS targets (ACC, IC/VS), along with established and effective stereotactic therapies (DBS–SRS), increases therapeutic options to impact CP by modulating affective, pain-attentional neural transmission. Differences in trial concept, outcome measures, targets and applied technique promote conflicting findings and limited evidence. Hence, we advocate to raise awareness of the potential therapeutic usefulness of each approach covering their advantages and disadvantages, including such parameters as invasiveness, risk–benefit ratio, reversibility and responsiveness.
Keywords: Limbic pain network, Deep brain stimulation, Magnetic resonance-guided focussed ultrasound, Stereotactic radiosurgery, Centromedian-parafascicular, Ventral striatum/anterior limb of the internal capsule, Anterior cingulate cortex, Central lateral nucleus of the thalamus, Chronic pain
Key Summary Points
| Deep brain stimulation and lesioning procedures like stereotactic radiosurgery targeting mainly the somatosensory thalamic pain circuits yielded mixed results. |
| The ability to impact the affective domain of chronic pain received increased attention and a renaissance of deep brain stimulation for chronic pain treating the affective sphere of pain. |
| Incisionless techniques like magnetic resonance-guided focused ultrasound exhibited promising results and offer additional therapeutic options. |
| Other noninvasive (transcranial magnetic stimulation) and less-invasive cranial (motor cortex stimulation) and spinal neuromodulation (spinal cord stimulation) should be considered prior to cranial neuromodulation. |
Introduction
Despite tremendous advances in pharmacological, behavioural and physical therapies, chronic pain treatment remains challenging in a considerable proportion of individuals classified as refractory [1–3]. It has been estimated that up to 20–30% of patients with chronic pain perceive insufficient pain relief leading to enormous economical as well as psychosocial consequences for the affected individuals. Within the European Union alone, this is estimated to cost the European economy around 440 billion euros annually [2, 3]. To be able to provide alternative treatment strategies to patients with chronic pain and in view of the socio-economic impact, the development and implementation of novel methods, including magnetic resonance-guided focused ultrasound (MRgFUS), and further elaboration of established targets such as the centromedian-parafascicular complex of the thalamus (CmPf), the anterior cingulate cortex (ACC) and ventral striatum/anterior limb of the internal capsule (VS/ALIC) approaching affective brain circuits using stereotactic treatment strategies, such as deep brain stimulation (DBS) and stereotactic radiosurgery (SRS), deserves enhanced clinical scientific attention. In addition to improving drug therapies, functional stereotactic methods as treatment options are becoming increasingly established [4]. These therapeutic approaches originated from clinical observations made by Spiegel and Wycis during their search for therapeutic alternatives to frontal leucotomies [5], which, until then, had been regularly performed for the treatment of psychiatric disorders [6]. Over time, the thalamus gained recognition as a major relay station for both motor and somatosensory as well as viscero-sensory neural transmission, acting as a predisposing site to centrally impact neural pain transmission [7]. Consecutively, several human studies emerged in which the somatosensory properties of the thalamus, the nuclei ventromedialis (VPM) and ventrolateralis (VPL), were used as a target of DBS for the therapy of various pain symptoms (Fig. 1a). As a result of highly variable results as well as increasing options for drug pain management, approval for thalamic DBS for pain management was finally withdrawn in the USA in 1991, which led to a sharp decline in research efforts [4]. Despite this unjustified sceptical attitude towards thalamic DBS, the proportion of its use in the treatment of neurological diseases is steadily increasing because of continuously advanced electrode design (e.g. directional leads) and novel neuroimaging capabilities to detect both functional and structural cerebral foci [8, 9]. Observations derived from in-human DBS studies suggest that in addition to the predominantly targeted thalamic somatosensory VPL/VPM and periventricular/periaquaductal grey (PVG/PAG), limbic pain network associated DBS targets such as the medial thalamic nuclei (intralaminar nuclei of the thalamus; CmPf), the ACC and the ventral VS/ALIC have yielded a meaningful impact on chronic pain levels by modulating affective/cognitive pain pathways [9–14]. Initial studies observed that DBS of the ACC promoted its impact predominantly on the emotional appraisal of pain stimuli, similarly to what has been reported for CmPF-DBS [11]. In addition to invasive and reversible DBS, less-invasive ablative stereotactic thalamotomy using irradiation (SRS) or high-intensity focused ultrasound applied under MR thermometry guidance (MRgFUS) represents a potential alternative in the treatment of chronic pain by integrating high-resolution stereotactic navigation capabilities [15, 16].
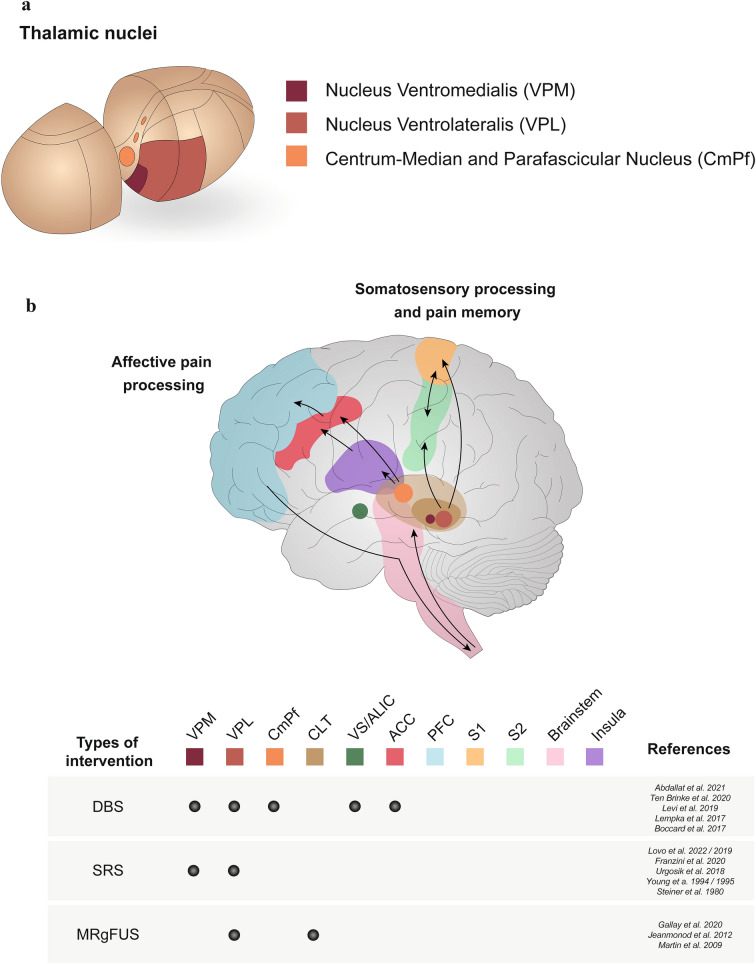
Fig. 1.
a Somatosensory and affective nuclei of the thalamus. Schematic diagram depicting the location and composition of the thalamic nuclei. The somatosensory thalamus includes the nuclei ventromedialis (VPM) and ventrolateralis (VPL); (yellow), and the centromedian-parafascicular complex of the thalamus (CmPf), which is located in internal medullary lamina (grey). b Projections of the somatosensory and affective pain circuits of the thalamus. Pain processing pathways are highlighted within the cerebral tissue. The somatosensory process and pain memory pathway originate from the brainstem and spinal cord via ascending fibres and project to the thalamic nuclei, which are found in the centre of the brain above the brainstem. Signals from the VPL project to the somatosensory cortex, located in the parietal lobe, and these signals are further exchanged between the primary (S1) and secondary (S2) regions. The affective pain processing pathway originates from the CmPF and is projected to the insula, located within the lateral sulcus, and is then transmitted to the anterior cingulate cortex (ACC) and prefrontal cortex (PFC). Finally, this signal is sent back to the brainstem and spinal cord via descending fibres. Currently available interventions, namely deep brain stimulation (DBS), stereotactic radiosurgery (SRS) and magnetic resonance-guided focused ultrasound (MRgFUS), targeting the different regions (CmPf, ACC, VS/ALIC, CLT) of the brain for the treatment of chronic pain are depicted
The comprehensive review aims to provide an update addressing DBS, SRS and MRgFUS targeting affective pain circuits of the brain (CmPf, ACC, VS/ALIC), in particular to summarize the neuroanatomical substrates of the intended targets, to compare the safety and effectiveness of different techniques, to discuss the pros/cons of previously completed and published in-human studies beyond the outcomes along with recommendations on what the neuromodulation community should address prospectively.
Methods
Search Strategy and Data Collection
To update and evaluate in-human studies in the field of stereotactic deep brain interventions defined as deep brain stimulation, radiosurgery and magnet resonance-guided focused ultrasound for patients with chronic pain, we conducted a comprehensive and structured literature search in the databases PubMed, Ovid, MEDLINE and Scopus between 1990 and 2021. In addition, we performed a manual library search using the terms “pain”, “chronic pain”, “pain and anxiety”, “affective pain modulation”, “deep brain stimulation”, “stereotactic radiosurgery”, “high-frequency focussed ultrasound”, “centromedian-parafascicular complex of the thalamus”, “intralaminar nuclei of the thalamus”, “central lateral nucleus of the thalamus”, “thalamotomy”, “anterior cingulate cortex”, “ventral striatum”, “anterior limb of the internal capsule” and “cingulotomy”.
Data sampling and analysis were performed in compliance with ethics guidelines. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Characteristics of Included Studies and Search Results from Literature
In-human studies (case reports, pilot studies, uncontrolled observational cohort trials, randomized controlled trials) targeting either the CmPf, the ACC or the VS/ALIC were included in this review. Study design, number of participants, outcome measures (safety, efficacy), stimulation and lesioning parameters, duration of treatment/observation and adverse events were recorded. The vast majority of studies consisted of uncontrolled observational cohort studies. Stereotactic techniques assessed were DBS (Table 1) and implant-free, ablative procedures (Table 2) using different thermal lesioning techniques (MRgFUS) and irradiation (SRS). Given the low number of randomized controlled trials, the clinical heterogeneity in stimulation patterns and duration of outcome measures along with the methodological diversity of the applied techniques across the assessed studies, we conceptualized a comprehensive review covering a summary of clinical findings published, the neuroanatomical basis for target justification and a potential guideline for future targeted clinical research.
Table 1.
Summary of in-human DBS studies published since 2017 (participants enrolled) targeting the affective sphere of pain (CmPf, ACC, VS/ALIC) for chronic pain
| References | No. | Study | DBS target | Indication | FU | Outcome | SAE/AE |
|---|---|---|---|---|---|---|---|
| Invasive DBS (reversible) | |||||||
| (Abdallat et al. 2021) [33] | 40 | PT | Unilateral VPM/VPL or CmPf alone | Facial pain | 63 months | Average pain relief 63 months > 50% in 10/18 patients | 11 hardware SAE |
| CRPS | No differences between VPM/VPL and CmPf | ||||||
| CPSP | |||||||
| Spinal cord lesion | |||||||
| Brachial plexus injury | |||||||
| Postherpetic pain | |||||||
| FBSS | |||||||
| Nerve lesions | |||||||
| Phantom limb | |||||||
| (Ten Brinke et al. 2020) [51] | 1 | CR | Unilateral VS/ALIC | CPSP | NA | Mean pain relief at the end of observation (VAS): 50% | NA |
| (Lempka et al. 2017) [12] | 10 | RCT | Bilateral VS/ALIC | CPSP | 24 months (6 months sham/verum + open-label phase 18 months) | Primary endpoint failure | Seizure (DBS “on”/“off”) |
| Improvement in affective pain scores | |||||||
| (Levi et al. 2019) [10] | 5 | PT | Bilateral ACC | Thalamic pain syndrome | 28 months | Mean pain relief 6 months (NRS): 37.9% | None |
| Mean pain relief 18 months (VAS): 35% | |||||||
| (Boccard et al. 2017) [43] | 22 | PT | Bilateral ACC | Neuropathic pain | 39 months | Mean pain relief 6 months (NRS): 60% | Seizure (DBS “on”/“off”) |
| Mean pain relief 12 months (VAS): 43% | |||||||
DBS deep brain stimulation, PT pilot study, RCT randomized controlled trial, CR case report, VPL nucleus ventrolateralis of the thalamus, VPM nucleus ventromedialis of the thalamus, CmPf centromedian-parafascicular complex of the thalamus, PVG periventricular grey, PAG periaqueductal grey, VC ventrocaudal nucleus of the thalamus, PLIC posterior limb of the capsula interna, ACC anterior cingulate cortex, CRPS complex regional pain syndrome, CPSP chronic post-stroke pain, FBSS failed back surgery syndrome, VAS visual analogue scale, NRS numeric rating scale, FU follow-up, SAE/AE serious adverse event, NA not applicable
Table 2.
Summary of in-human studies using implant-free SRS for chronic pain
| References | PN | Design | Target | Indication | FU | Outcome | SAE/AE |
|---|---|---|---|---|---|---|---|
| Non-invasive lesioning (non-reversible) | |||||||
| Franzini et al. [55] | 8 | PT |
SRS Central lateral thalamotomy |
TDP | 24 months | 3.9 points relief in VAS/recurrence 2 patents after 36 months | None |
| BPI | |||||||
| CPSP | |||||||
| PHN | |||||||
| Young et al. [59] | 10 | PT |
SRS Intralaminar nuclei, the lateral portion of the medial dorsal nucleus and CmPf Unilateral lesion: 9 patents Bilateral lesion: 1 patent |
SD | NA | Excellent: 3 | None |
| PHN | Acceptable: 4 | ||||||
| SCI | Failure: 3 | ||||||
| TP | |||||||
| AD | |||||||
| CPSP | |||||||
| Young et al. [62] | 20 | PT | SRS/thalamotomy | Different origin | 22 months | Excellent/good pain relief |
1 death radiation necrosis |
| 140–180 Gy | (Not specified) | ||||||
| Young et al. [62] | 19 | PT | SRS | Different origin | 12 months | 27% pain free | Not reported |
| Intralaminar, mediodorsal, centromedian and parafascicular nuclei | 33% > 50% pain relief | ||||||
| Steiner et al. [60] | NA | PT | SRS | CCP | NA | Significant pain relief in 35% | None |
| Medial thalamotomy | |||||||
| Frighetto et al. [58] | 3 | PT | SRS | CPSP | NA | Significant pain relief | None |
| Medial thalamotomy | CCP | Recurrence after 4 months | |||||
SRS stereotactic radiosurgery, PT pilot study, RCT randomized controlled trial, CR case report, VPL nucleus ventrolateralis of the thalamus, CPSP chronic post-stroke pain, FU follow-up, SAE/AE serious adverse event, ICH intracranial haemorrhage, NP neuropathic pain, TN trigeminal neuralgia, CCP chronic cancer pain, TDP trigeminal deafferentation pain, AD anesthesia dolorosa, TP thalamic pain, SCI spinal cord injury, PHN postherpatic neuralgia, SD spinal disorders, BPI plexus brachialis injury, NA not applicable
Role of Thalamic Nuclei and Associated Affective Circuits in Pain Processing
Nociceptive impulses travel via fibre bundles, such as the tractus spinothalamicus, to the VPL and VPM, from which these lateral, sensory pain pathways continue to the primary, as well as secondary, somatosensory cortex (postcentral gyrus), enabling conscious (discriminative) perception of pain [17–20]. Experimental and in-human studies indicate that the intralaminar thalamic nuclei and the CmPf project to areas of the limbic system, such as the ACC, gaining access to descending pain-inhibiting tracts to the brainstem and spinal cord. This enables an affective-cognitive attribution of pain processing termed pain attention (Fig. 1a, b). The VPL displays a somatotopic organization and distribution, representing the head, the arm and the leg through distinct functional-structural borders within the VPL (12 mm lateral from the mid anterior/posterior commissural line = head; 13 mm lateral from the mid anterior/posterior commissural line = arm; 14 mm lateral from the mid anterior/posterior commissural line = leg). The VPL receives afferent input from the lemniscus system, globus pallidus and cerebellar nuclei (ncl. dentatus; ncl. interpositus; ncl. fastigius) and projects to the prefrontal cortex, orbitofrontal cortex and posterior parietal lobe (Fig. 1a).
The CmPf projects to the basal ganglia with the CM complex receiving fibres from the internal globus pallidal and the Pf complex receiving input from the brainstem. The CmPf itself projects to the prefrontal cortex, cingular cortex, entorhinal cortex and the parieto-temporal area. In particular, one projection includes a route the ncl. caudatus, the putamen, the globus pallidus back to the CmPf termed the Nauta–Mehler loop [17–20].
The reciprocal and dynamic interplay of the ACC with the thalamic nuclei (predominantly CmPf—less VPL/VPM), the amygdala, the insular, VS/ALIC and the dorsolateral prefrontal among others is critical for the short-term and long-term aversive sensory processing. Despite thalamic CmPf and ACC, the VS/ALIC (fronto-striatal circuits) and connecting the orbitofrontal cortex with thalamocortical relevant for emotion (anxiety), behaviour and reward have emerged as potential DBS targets (Fig. 1a, b) [11, 21–23]. Preclinical studies suggest that activation of these brain regions may in addition be of relevance for the transition from acute to chronic pain (long-term potentiation) and that the ACC may predominantly contribute to the short-term encoding of aversive stimuli, while in contrast the medial part of the thalamus may be relevant for the maintenance of an external stimuli (e.g. pain) [24–26]. On a synaptic level, ACC neurons promote neuroplasticity by upregulating N-methyl-d-aspartate (NMDA) receptors and increased glutamate release, which may represent a transmitter correlate of pain-associated mood alterations (anxiety, depression) [21–23]. Hence, it appears reasonable and may justify that DBS of the ACC, VS/ALIC and CmPf modulates the limbic circuits pain by impacting affective pain domains such as attention, anticipation, anxiety, anhedonia and depression. However, to apply both as potential targets (sensory and affective circuits) for cranial neuromodulation therapies simultaneously appears an underinvestigated approach (Fig. 1b).
Deep Brain Stimulation (Invasive Reversible)
The concept of using DBS for pain management, in which lesions in the thalamus are produced to alleviate the suffering of pain-stricken patients, has been applied for some decades. The use of DBS in the treatment of chronic pain originally represented a pioneering achievement, although the current widespread use of DBS in the treatment of movement disorders developed only secondarily to its original intended use for treatment of chronic pain.
DBS is an invasive but reversible brain stimulation technique involving a surgical stereotactic guided procedure permitting one to apply electric densities in defined deep-seeded brain circuits and it has been applied in broad variety of chronic pain disorders [complex regional pain syndrome (CRPS); chronic post-stroke pain (CPSP); facial pain (FP); phantom pain (PP); failed back surgery syndrome (FBSS); neuropathic pain (NP); trigeminal neuralgia (TN); chronic cancer pain CCP; trigeminal deafferentation pain (TDP); anesthesia dolorosa (AD); thalamic pain (TP); spinal cord injury (SCI); postherpetic neuralgia (PHN); persistent spinal pain syndrome (PSPS); plexus brachialis injury (PBI)] [27–30]. Reversible clinical DBS effects can be achieved by adjustment of specific stimulation parameters (amplitude μs, frequency Hz, contact configuration (mono/bipolar), intensity; current mA or voltage V). Several guidelines and recommendations exist that address stereotactic techniques, stimulation pattern and the potential value of trialing DBS effects using temporarily externalized DBS leads remain an issue of ongoing debate, thereby limiting level of evidence. The DBS system consists of three components: the implanted pulse generator, the lead and the electrode. The polarity of the system can be programmed as cathode, anode and neutral, and the configuration can be set to monopolar, bipolar, and multipolar in more advanced systems. Different recommendations and guidelines exist related to the operative protocol (awake versus asleep surgery; microelectrode recording versus neuroimage-guided implantation) [27–30]. However, the lack of evidence and negative RCT have led to the withdrawal of approvals for the therapy of chronic pain syndromes by the US Food and Drug Administration (FDA) along with an unjustified, sharp decline and scepticism regarding the usefulness of DBS (off-label) for the treatment of refractory chronic pain disorders [31].
Thalamic DBS Targeting the Limbic Pain Matrix (CmPf)
In order to provide an unbiased, robust data interpretation, Frizon et al. performed a systematic review assessing the impact of DBS on chronic pain syndromes covering the period from 1975 to 2017. Interestingly, according to their search criteria, DBS studies for cluster headache, a follow-up period below 3 months, combined DBS and motor cortex stimulation (MCS), primarily DBS target other than pain and no modern DBS leads implanted represented exclusion criteria [32]. A total of 228 DBS patients were extracted, of whom some were implanted using microelectrode recording (MER) for target verification, while DBS test stimulation was not performed systematically along with a wide range given for DBS trial failures ranging from 2% to 48%. The vast majority was implanted either in the sensory VPL/VPM alone and/or in combination with PAG/PVG. It is noteworthy that DBS effects occur earlier in PAG/PVG (opioid flash), while CmPF, VS/ALIC and ACC DBS promoted its effect over a longer time frame. Notably, in one case series (three patients), one single lead/one trajectory was inserted to approach and stimulate the CmPf and the PAG/PVG simultaneously. Under combined stimulation pattern, the patients perceived a marked pain relief, in particular the affective domains improved significantly. Consecutively, the patient’s choice was a combined stimulation pattern in all three cases, indicating a synergistic effect of PAG/PVG and CmPf on pain levels [32].
As part of this narrative review, we conducted a literature search on the topic since 2017 as a continuation to the work by Frizon et al. In total, we found seven clinical trials since the beginning of 2017 including the main indications with significant response rates in facial pain, post-stroke pain and chronic pain due to the brachial plexus lesion (Table 1). In thalamic DBS, most of the screened trials consisted of uncontrolled, small-scale cohort studies and mainly approached sensory-motor pain transmission by VPL/VPM DBS means. The largest DBS study assessing VPL/VPM versus CmPf with a considerable long-term follow- up was published most recently by Abdallat et al. and will be discussed in detail [33–37]. Most recently, the therapeutic impact of sensory versus affective thalamic cranial neuromodulation was determined in a large multifocal DBS series including 40 patients with pain and an extended mean follow-up period of 5 years (Table 1) [33]. The surgical procedures encompassed a two-step protocol with awake DBS lead implantation under intraoperative trial stimulation and microelectrode recording guidance, followed by a trial period with externalized leads assessing the effects of either VPL/VPM or CmPf. Depending on the trial outcome, permanent DBS/IPG implantation continued targeting sensory or affective thalamic nuclei. In, total, 33 out of 40 study participants perceived a marked pain relief (defined as at least 50%/30% pain reduction) and received permanent DBS therapy. From the 33 permanent implanted participants, 22 were CmPf responders versus 11 VPL/VPM responders, of whom 18 were available for an extended follow-up beyond 4 years. Outcome measures were changes in mean/average/maximum pain levels quantified by the visual analogue scale (VAS). Maximum pain threshold was decreased (50% reduction) in 8/18 and by 30% in 10/18, while average pain levels declined by 50% in 10/18 and by 30% in 16/18. However, these differences were statistically not significant when comparing VPL/VPM versus CmPf along with a less favourable responsiveness in patients with thalamic lesions. DBS stimulation patterns were similar in both targets (CmPf: 210 µs, 130 Hz, bipolar, 2–3 V versus VPL/VPM: 210 µs, 130 Hz, bipolar, 0.5–1.5 V). These findings were in line with previously published studies, supporting the therapeutic value of thalamic DBS for chronic pain [33].
Impact of ACC DBS on Affective Sphere of Pain
DBS targeting the VPL (representation of body parts anatomically segregated) evokes paraesthesia in the affected pain region of the body within a shorter latency, while CmPf, VS/ALIC and ACC evoke similar DBS effects quantified by affective-related pain domains occurring over a longer time period. Patient with chronic pain stimulated in the CmPf or ACC reported unchanged pain levels; however, attention and emotional effects (affective distancing to chronic pain stimuli) were observed in these cohorts of DBS patients [32, 38–43] (Table 1).
The dynamic and reciprocal interplay of the ACC with the thalamic nuclei was demonstrated in neuroimaging (structural/activity) studies. Davis and co-workers applied positron emission tomography (PET) before, during and after thalamic DBS and found an immediately increased ACC activation during DBS along with a delayed activation of the posterior part of the ACC after DBS. Notably, this study determined the thalamic-cingulate pathways relevant for the affective sphere of pain processing. Hence, it appears reasonable to target the ACC for chronic pain, which has been explored as an additional DBS target in the past by the Oxford Group [38, 44–46]. So far, findings from these studies indicate that ACC DBS is safe and efficient in targeting the affective sphere of pain with clinical effects occurring with a latency, other than the immediate response followed by PVG/PAG stimulation, although a lack of efficacy was observed partly in the long-term assessment. This study covered a similar large observational period (mean follow-up 5 years) and observed a potential side effect, as ACC DBS “on” as well as ACC DBS “off” evoked seizures in a considerable subset of participants (Table 1). Additional smaller case series confirmed these findings in five patients with thalamic pain over 18 months treated with ACC DBS with 35% decline of pain levels and significantly improved quality of life domains [10]. Despite the in-human use of ACC DBS, other cranial stimulation techniques targeting the ACC are currently under preclinical investigation probing focused ultrasound and optogenetic stimulation of the ACC for pain relief [47, 48].
VS/ALIC as an Additional DBS Target Modulating Affective Pain Processing
A randomized sham-controlled crossover trial determined the efficacy and safety of VS/ALIC DBS for chronic pain in nine patients with PSP over 24 months (6 months crossover sham/verum stimulation + open-label phase 18 months) (Table 1). The rationale for this target originated from observations made in patients with obsessive-compulsive disorder (OCD) and treatment-resistant depression (TRD) treated with VS/ALIC DBS, supporting the role of fronto-striatal circuits (projections from orbitofrontal to striatal-thalamic-cortical areas) in mood, emotion regulation and anxiety improvement, hence potentially able to impact the affective domain of pain (attention/anticipation). Outcome measures initially included mood-associated scores along with pain assessment. Although the primary and secondary endpoints failed (50% pain decline), an improvement of the affect-related sub-score was observed. Serious adverse events occurred, namely seizures, cognitive impairment and dysexecutive impairment [12–14].
Lempka and colleagues further used neuroimaging measures (functional magnetic resonance imaging; fMRI) and electrophysiological methodologies (magnetoencephalography; MEG) in the later stage in a subset of the study cohort, aiming to elaborate potential effects of VS/ALIC DBS objectively [13, 14]. Using heat stimuli in five patients with PSP and age-gender matched healthy controls, Lempka et al. assessed DBS under on/off conditions with DBS “on” significantly reducing prior heat-evoked activation of the thalamus, insula, operculum and the orbitofrontal area [13]. In the MEG analysis, ACC DBS (on) significantly changed event-related domains (N1/P2) during a specific task compared to DBS “off” and baseline [14]. To what extent these findings may serve as future biomarkers for cranial neuromodulation in pain remains largely unknown.
DBS Trial, Closed-Loop Stimulation Waveforms and Advanced Directional Leads for Chronic Pain: Where Do We Stand?
In the past, DBS trials were mainly performed in the treatment of movement disorders. However, novel DBS targets for chronic pain (ACC, VS/ALIC) have led to a renaissance of DBS trial stimulation for chronic pain. However, current available literature indicates that DBS test-stimulations using externalized leads have been investigated inconsistently [49]. In view of the recent developments in the field of DBS probing closed-loop stimulation along with the fact that biomarkers are lacking for chronic pain, externalized DBS leads offer the possibility to execute and quantify additional task-related measurements in order to elaborate potential central pain signatures (e.g. neurochemical, neuroimaging, electrophysiology) relevant for biomarker development [50]. Further DBS lead techniques permit one to focus stimulation/electrical density by using directional leads. Ten Brinke et al. investigated the therapeutic usefulness of such directional leads in a patient suffering from PSP, who was implanted in the sensory thalamus (VPL/VPM) and found that directional leads increased the precision and decreased stimulation-associated side effects [51]. However, large-scale controlled studies are needed comparing both types of DBS electrodes in chronic pain.
Although not within the scope of this review, it is noteworthy that radiofrequency ablation (RFA) operating between 350 and 500 kHz, as an invasive, ablative and non-reversible procedure, has been applied in a larger number of subjects with chronic pain [52, 53]. In a recent study [52], Rezaei Haddad and co-workers provided an update for RFA thalamotomy (VPL, VPM, CmPf, the pulvinar, the suprageniculate and posterior nuclei) and found a sustained pain suppression, a low-risk profile and no superiority of VPL/VPM compared to CmPf. However, most of the published trials were characterized by an uncontrolled observational cohort study design and heterogeneity in included patients and pain scales utilized [52].
Stereotactic Radiosurgery (SRS)
SRS is a less-invasive, incisionless and implant-free method that utilizes radiation (140–180 Gy) to create very specific lesions in target tissues in groups of patients with pain not considered eligible for surgical treatment. On the basis of the principle of lesion surgery, SRS was applied in various pain disorders, mainly targeting sensory pain pathways (Fig. 1b). Current SRS procedures are highly precise owing to the development of alignable radioactive cobalt sources that reduce clinical complications.
Currently, preclinical and in-human studies suggest a therapeutic effect far beyond the lesion. This non-lesioning mechanism of action remains largely unknown, but may modulate structure and function of glia and neuron population on a molecular and cellular level relevant for neural transmission [54–62]. In this review, we discuss in detail the most recent publication addressing SRS for pain approaching various targets within the affective sphere of pain.
Franzini and co-workers targeted the posterior part of the central lateral thalamic nucleus (CLT) relevant for thalamo-cortical circuits of pain nucleus using SRS in eight patients with drug-resistant NP (four TDP, two BPI, one PSP, one PHP). Notably the same target was approached by MRgFUS. Bilateral CLT lesioning was performed in 3/8 and a unilateral thalamotomy was achieved in 5/8 utilizing a radiation dosage between 130 and 140 Gy. Within a mean observation period of 24 months, a significant responder rate (defined as at least 50% reduction) along with a significantly declined pain intensity (pre-VAS 9.4 versus post-VAS 5.5) was found after 12 months. No treatment-associated adverse events were recorded. However, pain recurrence was noticed after 24 months in two patients suffering from BPI and PSP types of chronic pain [55].
The CmPf was lesioned in another observational, uncontrolled cohort SRS study including 10 patients suffering from refractory TN, AD and CRPS with a median follow-up of 13 months. In line with previous GKRS pain trials, treatment effects occurred over an extended time (given average time 2 months). Pain outcome quantified by the VAS significantly decreased with 50% experiencing pain relief of at least 50% and 10% perceived pain decline of at least 30%, with 40% non-responder [56]. To facilitate therapeutic SRS effects, Lovo et al. conceptualized a multifocal radiation frame. This radioneuromodulation group advanced previously used protocols by targeting the CmPf and the retrogasserian (RGZ) simultaneously with a staged radiation pattern depending on the target (CmPf = 140 Gy versus RGZ = 90 Gy) in eight individuals with TN. Dual radioneuromodulation of the CmPf/RGZ exhibited immediate (24 h) therapeutic response (at least 50% reduction) in five out of eight study participants, which further (49 h) was extended towards a responder rate of 87.5% (7/8) with one subject reporting moderate relief (at least 30%) in a safe manner. Pain intensity declined markedly (pre-VAS 10 versus post-VAS 3) and was sustained after 4 months [57]. The safety and efficacy of CmPf SRS was revisited by an observational larger case series carried out by Urgosik and colleagues. CmPf was the target of choice in 30 patients with chronic pain (TN, TP, TDP, PP) treated with a single shot of 145 Gy. A responder rate of 43% (13/30) versus a non-responder rate 57% (17/30) was detected within a mean observation time of 3 months along with pain recurrence in four out of 13 previously responsive patients after 22–30 months [15]. These findings are summarized in Table 2.
Incisionless MR-Guided Focused Ultrasound (MRgFUS)
In the last decade, a new procedure using focused ultrasound under real-time MR thermometry guidance has been increasingly established and received FDA approval and CE mark for the treatment of unilateral essential tremor, tremor-dominant Parkinson’s disease and currently MRgFUS is under investigation for neuropsychiatric disorders (TRD, OCD) [63, 64]. Incisionless, implant-free MRgFUS uses high-frequency ultrasonic waves (650 kHz) to evoke thermal lesions in deep-seated brain structures under stereotactic guidance (helmet-like array with integrated stereotactic frame). The combination of a focused ultrasound (FUS) transducer with magnetic resonance imaging allows physicians to perform precise localized thermal lesioning along with clinical assessment of clinical effects as the patient remains awake during the procedure [63, 64]. MRgFUS permits intraprocedural motor and sensory (VPL) testing as temperatures between 40 and 45 °C induce reversible lesions and potentially decrease surgery-related complications (infection, haemorrhage and adverse events in adjunct anatomic structures). A total of three uncontrolled human studies targeting the sensory thalamic pathway have been conducted with a total number of 29 treated patients for chronic pain syndromes of different origin (trigeminal neuralgia, chronic neuropathic pain), and one study assessed pain levels in MRgFUS treated patients with different movement disorders [66, 67] (Fig. 1b).
In previous study of 12 patients with NP (unilaterally in five patients, bilaterally in seven patients) derived from central and/or peripheral origin, MRgFUS was applied with the CLT (pars posterior) as defined target, in particular the outflow pathways projecting to the ACC, creating lesions with a diameter between 3 and 4 mm and target variation of 1 mm. The decision to perform unilateral lesioning was based on several reasons: two patients had prior RFA, one bleeding due to ischemia/cavitation, one patient had a 100% decline of pain, one patient lack of compliance. The mean pain relief was 49% after 3 months in nine patients and further improved towards 57% pain reduction at 12 months in eight patients along with immediate effects observed in six patients. Post-procedure, drug intake was stopped or reduced in six patients. Despite the bleeding in one case, vestibular symptoms, head pain, nausea and vomiting were more frequently observed and recovered within days. Post-ablative neuroimaging analysis revealed seven out 18 lesions within the CLT, while the other lesions reached adjunct structures like the pulvinar or the CmPf of the thalamus. In a subset of eight patients, EEG recording at 3 and 12 months demonstrated a frequency band-specific spectral power amplitude decrease with patterns like those observed in healthy subjects [66].
The same group presented most recently novel MRgFUS data treating patients with drug-resistant trigeminal neuralgia (classic, idiopathic, secondary) with bilateral CLT MRgFUS [67]. MRgFUS was applied using the following treatment parameters: number of sonifications 15 ± 8, treatment duration 20–31 s, sonification power 1020 ± 236 W, peak temperatures 54–58 °C, lesion volume 153 ± 85 mm3. All patients were discharged after 24 h. Overall, a mean pain relief rate of 51% was achieved at 3 months, further increased to 71% pain reduction after 12 months and 78% pain decline after 50 months. The frequency of paroxysmal pain significantly declines from 84/day to 4/day post-sonication. No serious adverse events occurred with four patients stopping drug intake for pain [67]. Table 3 summarizes current published trials.
Table 3.
Summary of in-human studies using less-invasive, incisionless MRgFUS for chronic pain
| References | PN | Design | Target | Indication | FU | Outcome | SAE/AE |
|---|---|---|---|---|---|---|---|
| Non-invasive lesioning (non-reversible) | |||||||
| Gallay et al. [67] | 8 | PT | MRgFUS | TN | 53 months | Mean pain relief 3 months: 51% | None |
| Posterior part of the central lateral thalamic nucleus (peak 51–64 °C) | Mean pain relief 12 months: 71% | ||||||
| Mean pain relief 53 months: 78% | |||||||
| Jeanmonod et al. [66] | 12 | PT | MRgFUS | NP | 12 months | Mean pain relief 3 months: 49% | 1 ICH |
| Posterior part of the central lateral thalamic nucleus (peak 51–64 °C) | Mean pain relief 12 months: 57% | ||||||
| Gallay et al. [65] | 180 (treatments) | PT | MRgFUS | NP | 3 months | Not reported | None |
| Posterior part of the central lateral thalamic nucleus (peak 51–64 °C) | |||||||
| Martin et al. [64] | 9 | PT | MRgFUS | NP | NA | Mean pain relief after 2 days: 68% | None |
| VPL (peak temperature 51–60 °C) | |||||||
MRgFUS magnet resonance guided focused ultrasound, PT pilot study, RCT randomized controlled trial, CR case report, VPL nucleus ventrolateralis of the thalamus, CPSP chronic post-stroke pain, FU follow-up, SAE/AE serious adverse event, ICH intracranial haemorrhage, NP neuropathic pain, TN trigeminal neuralgia, NA not applicable
Conclusions
The list of currently available procedures illustrates the heterogeneity of cranial neuromodulation options in the treatment of various chronic pain syndromes. Primarily, the procedures can be categorized by their invasiveness and reversibility. In this regard, advantages of less-invasive therapeutic procedures include reduced operative risk and directly associated complications, such as infections and bleedings. The ability to modulate the sensory and affective brain networks is not solely a result of non-lesioning effects, as lesions may impact neural networks in a more permanent fashion.
It is noteworthy that the response rates presented are heterogeneous and biased by measurement of different stimulation parameters, aetiology of pain disorders, patient selection, occurrence of observed therapeutic effects, dose–response relationship and patient preference. There is consensus in many studies that patient stratification is required to precisely select effective therapies. In contrast, meaningful responsiveness was observed predominantly in subsets of chronic pain disorders (phantom pain, deafferentation pain, brachial plexus injuries), while other pain phenotypes appeared less suitable and failed to achieve sustained pain relief.
Technical progress over the course of the last two decades in stimulator (brain sensing) and electrode (directional leads) technologies along with closed-loop systems requires a biomarker and has been investigated in wide range of DBS indications such as movement disorders, epilepsy and neuropsychiatric disorders. However, such biomarker development represents a key barrier to overcome for chronic pain, as there currently are no reliable, established electrophysiological and neuroimaging biomarkers useful for directional DBS programming in chronic pain. In addition, open questions must be addressed: How does one stratify potential responders among patients with refractory pain? How does a sensory versus an affective responder differ? Which target of the affective sphere of pain should we use for which pain disorder (CmPf, ACC, VS/ALIC)? Is multifocal stimulation targeting sensory and affective brain structures a solution? However, prior to central neurostimulation, non-invasive non-lesioning brain stimulation techniques (TMS, tDCS, tACS) and less-invasive spinal cord stimulation, dorsal root ganglion stimulation and motor cortex stimulation should be considered.
Acknowledgements
We thank Antonio Garcia for providing the scientific illustration.
Funding
This work did not receive any funding from the private and/or institutional sector. No funding was received for the publication of this article.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
Thomas M. Kinfe conducted the study, drafted the manuscript, and edited and approved the final version. Michael Buchfelder, Andreas Stadlbauer, Martin Nüssel, Yining Zhao, Constantin Knorr, Alessandro del Vecchio and Martin Regensburger edited the manuscript. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work, and have given their approval for this version to be published.
Disclosures
Thomas M. Kinfe works as a consultant for Abbott (formerly St. Jude Medical, Inc.) and Medtronic Inc. Yining Zhao is a participant of the neuromodulation in pain fellowship sponsored by Abbott Inc. Michael Buchfelder, Andreas Stadlbauer, Martin Nüssel, Constantin Knorr, Alessandro del Vecchio and Martin Regensburger have nothing to disclose.
Compliance with Ethics Guidelines
Data sampling and analysis were performed in compliance with ethics guidelines. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Footnotes
Martin Nüssel and Yining Zhao contributed equally to this work.
References
- 1.Cohen SP, Vase L, Hooten WM. Chronic pain: an update on burden, best practices, and new advances. Lancet. 2021;397:2082–2097. doi: 10.1016/s0140-6736(21)00393-7. [DOI] [PubMed] [Google Scholar]
- 2.Leadley RM, Armstrong N, Lee YC, Allen A, Kleijnen J. Chronic diseases in the European Union: the prevalence and health cost implications of chronic pain. J Pain Palliat Care Pharmacother. 2012;26:310–325. doi: 10.3109/15360288.2012.736933. [DOI] [PubMed] [Google Scholar]
- 3.Karra R, Holten-Rossing S, Mohammed D, Parmeggiani L, Heine M, Namnun OC. Unmeet needs in the management of functional impairment in patients with chronic pain: a multnational survey. Pain Management. 2021;11(3):303–314. doi: 10.2217/pmt-2020-0098. [DOI] [PubMed] [Google Scholar]
- 4.Singleton WGB, Ashida R, Patel NK. Deep brain stimulation for facial pain. Prog Neurol Surg. 2020;35:141–161. doi: 10.1159/000509734. [DOI] [PubMed] [Google Scholar]
- 5.Spiegel EA, Wycis HT. Present status of stereoencephalotomies for pain relief. Confin Neurol. 1966;27:7–17. doi: 10.1159/000103927. [DOI] [PubMed] [Google Scholar]
- 6.Rzesnitzek L, Hariz M, Krauss JK. Psychosurgery in the history of stereotactic functional neurosurgery. Stereotact Funct Neurosurg. 2020;98:241–247. doi: 10.1159/000508167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gustin SM, Wrigley PJ, Youssef AM, McIndoe L, Wilcox SL, Rae CD. Thalamic activity and biochemical changes in individuals with neuropathic pain after spinal cord injury. Pain. 2014;155:1027–1036. doi: 10.1016/j.pain.2014.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Krack P, Volkmann J, Tinkhauser G, Deuschl G. Deep brain stimulation in movement disorders: from experimental surgery to evidence-based therapy. Mov Disord. 2019;34:1795–1810. doi: 10.1002/mds.27860. [DOI] [PubMed] [Google Scholar]
- 9.Moisset X, Lanteri-Minet M, Fontaine D. Neurostimulation methods in the treatment of chronic pain. J Neural Transm. 2020;127:673–686. doi: 10.1007/s00702-019-02092-y. [DOI] [PubMed] [Google Scholar]
- 10.Levi V, Cordella R, D’Ammando A, Tringali G, Dones I, Messina G. Dorsal anterior cingulate cortex (ACC) deep brain stimulation (DBS): a promising surgical option for the treatment of refractory thalamic pain syndrome (TPS) Acta Neurochir (Wien) 2019;161:1579–1588. doi: 10.1007/s00701-019-03975-5. [DOI] [PubMed] [Google Scholar]
- 11.Xiao X, Zhang YQ. A new perspective on the anterior cingulate cortex and affective pain. Neurosci Behav Rev. 2018;90:200–211. doi: 10.1016/j.neubiorev.2018.03.022. [DOI] [PubMed] [Google Scholar]
- 12.Lempka SF, Malone DA, Jr, Hu B, et al. Randomized clinical trial of deep brain stimulation for poststroke pain. Ann Neurol. 2017;81(5):653–663. doi: 10.1002/ana.24927. [DOI] [PubMed] [Google Scholar]
- 13.Jones SE, Lempka SF, Gopalakrishnan R, et al. Functional magnetic resonance imaging correlates of ventral striatal deep brain stimulation for poststroke pain. Neuromodulation. 2021;24(2):259–264. doi: 10.1111/ner.13247. [DOI] [PubMed] [Google Scholar]
- 14.Gopalakrishnan R, Burgess RC, Malone DA, et al. Deep brain stimulation of the ventral striatal area for poststroke pain syndrome: a magnetoencephalography study. J Neurophysiol. 2018;119(6):2118–2128. doi: 10.1152/jn.00830.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Urgosik D, Liscak R. Medial Gamma Knife thalamotomy for intractable pain. J Neurosurg. 2018;129:72–76. doi: 10.3171/2018.7.GKS181583. [DOI] [PubMed] [Google Scholar]
- 16.Monteith SJ, Medel R, Kassell NF, Wintermark M, Eames M, Snell J. Transcranial magnetic resonance-guided focused ultrasound surgery for trigeminal neuralgia: a cadaveric and laboratory feasibility study. J Neurosurg. 2013;118(2):319–328. doi: 10.3171/2012.10.JNS12186. [DOI] [PubMed] [Google Scholar]
- 17.Krauss JK, Volkmann J. Tiefe hirnstimulation. Heidelberg: Steinkopff. 2004. 10.1007/978-3-7985-1956-5..
- 18.Herrero MT, Barcia C, Navarro JM. Functional anatomy of thalamus and basal ganglia. Childs Nerv Syst. 2002;18(8):386–404. doi: 10.1007/s00381-002-0604-1. [DOI] [PubMed] [Google Scholar]
- 19.Robertson RT, Kaitz SS. Thalamic connections with limbic cortex. I. Thalamocortical projections. J Comp Neurol. 1981;195(3):501–525. doi: 10.1002/cne.901950308. [DOI] [PubMed] [Google Scholar]
- 20.Kaitz SS, Robertson RT. Thalamic connections with limbic cortex. II. Corticothalamic projections. J Comp Neurol. 1981;195(3):527–545. doi: 10.1002/cne.901950309. [DOI] [PubMed] [Google Scholar]
- 21.Price DD. Psychological and neural mechanisms of the affective dimension of pain. Science. 2000;288(5472):1769–1772. doi: 10.1126/science.288.5472.1769. [DOI] [PubMed] [Google Scholar]
- 22.Tim VPB, Graham LC, Bong-Kiun K, Min Z. Synaptic plasticity in the anterior cingulate cortex in acute and chronic pain. Nat Rev Neurosci. 2016;17(8):485–496. doi: 10.1038/nrn.2016.68. [DOI] [PubMed] [Google Scholar]
- 23.Xiao X, Ding M, Zhang YQ. Role of the anterior cingulate cortex in translational pain research. Neurosci Bull. 2021;37:405–422. doi: 10.1007/s12264-020-00615-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chai SC, Kung JC, Shyu BC. Roles of the anterior cingulate cortex and medial thalamus in short-term and long-term aversive information processing. Mol Pain. 2010;6:42. doi: 10.1186/1744-8069-6-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Becker S, Gandhi W, Schweinhardt P. Cerebral interactions of pain and reward and their relevance for chronic pain. Neurosci Lett. 2012;520(2):182–187. doi: 10.1016/j.neulet.2012.03.013. [DOI] [PubMed] [Google Scholar]
- 26.Duncan GH, Kupers RC, Marchand S, Villemure JG, Gybels JM, Bushnell MC. Stimulation of human thalamus for pain relief: possible modulatory circuits revealed by positron emission tomography. Neurophysiology. 1998;80(6):3326–3330. doi: 10.1152/jn.1998.80.6.3326. [DOI] [PubMed] [Google Scholar]
- 27.Krauss JK, Lipsman N, Aziz T, Boutet A, Brown P, Chang JW. Technology of deep brain stimulation: current status and future directions. Nat Rev Neurol. 2021;17:75–87. doi: 10.1038/s41582-020-00426-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Aum DJ, Tierney TS. Deep brain stimulation: foundations and future trends. Front Biosci (Landmark Ed) 2018;23:162–182. doi: 10.2741/4586. [DOI] [PubMed] [Google Scholar]
- 29.Ben-Haim S, Mirzadeh Z, Rosenberg WS. Deep brain stimulation for intractable neuropathic facial pain. Neurosurg Focus. 2018;45:E15. doi: 10.3171/2018.5.FOCUS18160. [DOI] [PubMed] [Google Scholar]
- 30.Farrell SM, Green A, Aziz T. The current state of deep brain stimulation for chronic pain and its context in other forms of neuromodulation. Brain Sci. 2018;8:158. doi: 10.3390/brainsci8080158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Slavin KV, Isagulayn ED, Rzaev DA. Deep brain stimulation for chronic pain: time to reconsider the skeptical attitude? Brain Sci. 2020;10:772. doi: 10.3390/brainsci10110772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Frizon LA, Yamamoto EA, Nagel SJ, Simonson MT, Hogue O, Machado AG. Deep brain stimulation for pain in the modern era: a systematic review. Neurosurgery. 2020;86:191–202. doi: 10.1093/neuros/nyy552. [DOI] [PubMed] [Google Scholar]
- 33.Abdallat M, Saryyeva A, Blahak C, Wolf ME, Weigel R, Loher TJ. Centromedian-parafascicular and somatosensory thalamic deep brain stimulation for treatment of chronic neuropathic pain: a contemporary series of 40 patients. Biomedicines. 2021;2021:9. doi: 10.3390/biomedicines9070731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kashanian A, DiCesare JAT, Rohatgi P, Albano L, Krahl SE, Bari F. Case series: deep brain stimulation for facial pain. Oper Neurosurg (Hagerstown) 2020;19:510–517. doi: 10.1093/ons/opaa170. [DOI] [PubMed] [Google Scholar]
- 35.Polanski WH, Zolal A, Klein J, Kitzler HH, Schackert G, Eisner W. Somatosensory functional MRI tractography for individualized targeting of deep brain stimulation in patients with chronic pain after brachial plexus injury. Acta Neurochir (Wien) 2019;161:2485–2490. doi: 10.1007/s00701-019-04065-2. [DOI] [PubMed] [Google Scholar]
- 36.Abreu V, Vaz R, Rebelo V, Rosas MJ, Chamadoira C, Gillies MJ. Thalamic deep brain stimulation for neuropathic pain: efficacy at three years’ follow-up. Neuromodulation. 2017;20:504–513. doi: 10.1111/ner.12620. [DOI] [PubMed] [Google Scholar]
- 37.Abreu V, Vaz R, Chamadoira C, Rebelo V, Reis C, Costa F. Thalamic deep brain stimulation for post-traumatic neuropathic limb pain: efficacy at five years’ follow-up and effective volume of activated brain tissue. Neurochirurgie. 2021;68(1):52–60. doi: 10.1016/j.neuchi.2021.06.006. [DOI] [PubMed] [Google Scholar]
- 38.Davis KD, Taub E, Duffner F, et al. Activation of the anterior cingulate cortex by thalamic stimulation in patients with chronic pain: a positron emission tomography study. J Neurosurg. 2000;92(1):64–69. doi: 10.3171/jns.2000.92.1.0064. [DOI] [PubMed] [Google Scholar]
- 39.Boccard SGJ, Fitzgerald JJ, Pereira EAC, Moir L, Van Hartevelt TJ, Kringelbach ML. Targeting the affective component of chronic pain: a case series of deep brain stimulation of the anterior cingulate cortex. Neurosurgery. 2014;74:627–628. doi: 10.1227/NEU.0000000000000321. [DOI] [PubMed] [Google Scholar]
- 40.Russo JF, Sheth SA. Deep brain stimulation of the dorsal anterior cingulate cortex for the treatment of chronic neuropathic pain. Neurosurg Focus. 2015;38(6):E11. doi: 10.3171/2015.3.FOCUS1543. [DOI] [PubMed] [Google Scholar]
- 41.Boccard SGJ, Fernandes HM, Jbabdi S, Van Hartevelt TJ, Kringelbach ML, Quaghebeur G. Tractography study of deep brain stimulation of the anterior cingulate cortex in chronic pain: key to improve the targeting. World Neurosurg. 2016;86:361–363. doi: 10.1016/j.wneu.2015.08.065. [DOI] [PubMed] [Google Scholar]
- 42.Boccard SGJ, Pereira EAC, Moir L, Van-Hartevelt TJ, Kringelbach ML, FitzGerald JJ. Deep brain stimulation of the anterior cingulate cortex: targeting the affective component of chronic pain. NeuroReport. 2014;25:83–88. doi: 10.1097/WNR.0000000000000039. [DOI] [PubMed] [Google Scholar]
- 43.Boccard SGJ, Prangnell SJ, Pycroft L, Cheeran B, Moir L, Pereira EAC. Long-term results of deep brain stimulation of the anterior cingulate cortex for neuropathic pain. World Neurosurg. 2017;106:625–637. doi: 10.1016/j.wneu.2017.06.173. [DOI] [PubMed] [Google Scholar]
- 44.Caston RM, Smith EH, Davis TS, Rolston JD. The cerebral localization of pain: anatomical and functional considerations for targeted electrical therapies. J Clin Med. 2020;9(6):1945. doi: 10.3390/jcm9061945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kim D, Chae Y, Park H-J, Lee I-S. Effects of chronic pain treatment on altered functional and metabolic activities in the brain: a systematic review and meta-analysis of functional neuroimaging studies. Front Neurosci. 2021;15:684926. doi: 10.3389/fnins.2021.684926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yang S, Bie X, Wang Y, Li J, Wang Y, Sun X. Image features of resting-state functional magnetic resonance imaging in evaluating poor emotion and sleep quality in patients with chronic pain under artificial intelligence algorithm. Contrast Media Mol Imaging. 2022;2022:5002754. doi: 10.1155/2022/5002754.eCollection. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Feng X, Niu L, Long M, et al. Transcranial ultrasound of the anterior cingulate cortex reduces neuropathic pain in mice. Evid Based Compl Alternat Med. 2021 doi: 10.1155/2021/6510383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Moon HC, Park YS. Optogenetic stimulation of the anterior cingulate cortex modulates the pain processing in neuropathic pain: a review. J Mol Neurosci. 2022;72(1):1–8. doi: 10.1007/s12031-021-01898-4. [DOI] [PubMed] [Google Scholar]
- 49.Shirvalkar P, Sellers KK, Schmitgen A, et al. A deep brain stimulation trial period for treating chronic pain. J Clin Med. 2020;9:3155. doi: 10.3390/jcm9103155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mirza KB, Golden CT, Nikolic K, Toumazou C. Closed-loop implantable therapeutic neuromodulation systems based on neurochemical monitoring. Front Neurosci. 2019;13:808. doi: 10.3389/fnins.2019.00808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ten Brinke TR, Arnts H, Schuurman R, van den Munckhof P. Directional sensory thalamus deep brain stimulation in poststroke refractory pain. BMJ Case Rep. 2020;2020:13. doi: 10.1136/bcr-2019-233254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Stern MA, Isbaine F, Qiu D, Riley JP, Boulis NM, Gross RE. Radiofrequency ablation through previously effective deep brain stimulation leads for Parkinson disease: a retrospective series. World Neurosurg. 2020;144:e750–e765. doi: 10.1016/j.wneu.2020.09.060. [DOI] [PubMed] [Google Scholar]
- 53.Rezaei Haddad A, Hayley J, Mostofi A, Brown M, Pereira E. Stereotactic radiofrequency thalamotomy for cancer pain: a systematic review. World Neurosurg. 2021;151:225–234.e6. doi: 10.1016/j.wneu.2021.04.075. [DOI] [PubMed] [Google Scholar]
- 54.Régis J. Radiosurgery as neuromodulation therapy! In: Chernov M, Hayashi M, Ganz J, Takakura K, editors. Gamma knife neurosurgery in the management of intracranial disorders. Acta Neurochirurgica Supplement, vol 116. Vienna: Springer. 2013. 10.1007/978-3-7091-1376-9_19.
- 55.Franzini A, Attuati L, Zaed I, Moosa S, Stravato A, Navarria P. Gamma Knife central lateral thalamotomy for the treatment of neuropathic pain. J Neurosurg. 2020;2020:1–9. doi: 10.3171/2020.4.JNS20558. [DOI] [PubMed] [Google Scholar]
- 56.Lovo EE, Torres B, Campos F, et al. Stereotactic gamma ray radiosurgery to the centromedian and parafascicular complex of the thalamus for trigeminal neuralgia and other complex pain syndromes. Cureus. 2019;11(12):e6421. doi: 10.7759/cureus.6421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lovo EE, Moreira A, Barahona KC, et al. Radioneuromodulation by dual-target irradiation in pain crisis from trigeminal neuralgia. Cureus. 2022;14(1):e20971. doi: 10.7759/cureus.20971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Frighetto L, De Salles A, Wallace R, Ford J, Selch M, Cabatan-Awang C. Linear accelerator thalamotomy. Surg Neurol. 2004;62:104–106. doi: 10.1016/j.surneu.2003.08.033. [DOI] [PubMed] [Google Scholar]
- 59.Young RF, Jacques DS, Rand RW, Copcutt BR. Medial thalamotomy with the Leksell Gamma Knife for treatment of chronic pain. Acta Neurochir Suppl. 1994;62:105–110. doi: 10.1007/978-3-7091-9371-6_22. [DOI] [PubMed] [Google Scholar]
- 60.Steiner L, Forster D, Leksell L, et al. Gammathalamotomy in intractable pain. Acta Neurochir. 1980;52:173–184. doi: 10.1007/BF01402072. [DOI] [PubMed] [Google Scholar]
- 61.Young RF, Vermeulen SS, Grimm P, Posewitz AE, Jacques DB, Rand RW. Gamma Knife thalamotomy for the treatment of persistent pain. Stereotact Funct Neurosurg. 1995;64(1):172–181. doi: 10.1159/000098777. [DOI] [PubMed] [Google Scholar]
- 62.Young RF, Jacques DS, Rand RW, Copcutt BC, Vermeulen SS, Posewitz AE. Technique of stereotactic medial thalamotomy with the Leksell Gamma Knife for treatment of chronic pain. Neurol Res. 1995;17:59–65. doi: 10.1080/01616412.1995.11740287. [DOI] [PubMed] [Google Scholar]
- 63.Kinfe T, Stadlbauer A, Winder K, Hurlemann R, Buchfelder M. Incisionless MR-guided focused ultrasound: technical considerations and current therapeutic approaches in psychiatric disorders. Expert Rev Neurother. 2020;20(7):687–696. doi: 10.1080/14737175.2020.1779590. [DOI] [PubMed] [Google Scholar]
- 64.Martin E, Jeanmonod D, Morel A, Zadicario E, Werner B. High-intensity focused ultrasound for noninvasive functional neurosurgery. Ann Neurol. 2009;66:858–861. doi: 10.1002/ana.21801. [DOI] [PubMed] [Google Scholar]
- 65.Gallay MN, Moser D, Jeanmonod D. Safety and accuracy of incisionless transcranial MR-guided focused ultrasound functional neurosurgery: single-center experience with 253 targets in 180 treatments. J Neurosurg. 2018;2018:1–10. doi: 10.3171/2017.12.JNS172054. [DOI] [PubMed] [Google Scholar]
- 66.Jeanmonod D, Werner B, Morel A, Michels L, Zadicario E, Schiff G. Transcranial magnetic resonance imaging-guided focused ultrasound: noninvasive central lateral thalamotomy for chronic neuropathic pain. Neurosurg Focus. 2012;32:E1. doi: 10.3171/2011.10.FOCUS11248. [DOI] [PubMed] [Google Scholar]
- 67.Gallay MN, Moser D, Jeanmonod D. MR-guided focused ultrasound central lateral thalamotomy for trigeminal neuralgia. Single center experience. Front Neurol. 2020;11:271. doi: 10.3389/fneur.2020.00271. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.