Abstract
Following acute gastroenteritis (AGE) due to bacteria, viruses, or protozoa, a subset of patients develop new onset Rome criteria positive irritable bowel syndrome (IBS), called postinfection IBS (PI-IBS). The pooled prevalence of PI-IBS following AGE was 11.5%. PI-IBS is the best natural model that suggests that a subset of patients with IBS may have an organic basis. Several factors are associated with a greater risk of development of PI-IBS following AGE including female sex, younger age, smoking, severity of AGE, abdominal pain, bleeding per rectum, treatment with antibiotics, anxiety, depression, somatization, neuroticism, recent adverse life events, hypochondriasis, extroversion, negative illness beliefs, history of stress, sleep disturbance, and family history of functional gastrointestinal disorders (FGIDs), currently called disorder of gut-brain interaction. Most patients with PI-IBS present with either diarrhea-predominant IBS or the mixed subtype of IBS, and overlap with other FGIDs, such as functional dyspepsia is common. The drugs used to treat non-constipation IBS may also be useful in PI-IBS treatment. Since randomized controlled trials on the efficacy of drugs to treat PI-IBS are rare, more studies are needed on this issue.
Keywords: Gastrointestinal disorders, Enteritis, Dysentery, COVID-19, Tropical sprue
INTRODUCTION
Functional gastrointestinal disorders (FGIDs), including irritable bowel syndrome (IBS), are common conditions globally, both in the community and in clinical practice.1 IBS is associated with significant impairment in quality of life, work productivity, psychological comorbidity, health-care utilization, and economic consequences.2 The pathogenesis of IBS is enigmatic.3 There has been a paradigm shift recently on the pathogenesis of IBS from psychosomatic disorder to micro-organic condition.3 The Rome Foundation experts recognize this in the Rome IV criteria for IBS published in 2016 in which IBS has been considered to be a disorder of “gut-brain interaction” rather than “brain-gut interaction.”4 Of the several pieces of evidence behind considering IBS to be a micro-organic disorder, the development of the condition following an infectious illness, called postinfection IBS (PI-IBS), is the most important one.3,5,6
PI-IBS is diagnosed when new onset Rome-criteria positive IBS develops in an individual after acute gastroenteritis (AGE) characterized by one or more of the following features, (1) diarrhea, (2) vomiting, (3) fever, and (4) a positive stool culture.6 However, these criteria may need to be modified considering the suggestion that the patients with coronavirus disease-19 (COVID-19), which may not always be associated with diarrhea, may also develop PI-IBS.7-9 The pathogenesis of PI-IBS is multifactorial, including the agent, host, and host-agent interaction-related factors.10 The current manuscript plans to review the (1) epidemiology, including risk factors, (2) etiology and pathogenesis, (3) clinical presentation, (4) investigations, (5) management, and (6) prognosis of PI-IBS. It also summarizes the future direction on the subject.
EPIDEMIOLOGY OF PI-IBS
1. Frequency of development of PI-IBS after AGE
As shown in Table 1, based on data from 29 studies, 3.7% to 85.5% of subjects with AGE due to different causes developed PI-IBS;10-39 this incidence rates were much higher than the development of IBS among the control population in 16 studies (0.3% to 23.3%).10 In a recent meta-analysis, the pooled prevalence of PI-IBS was 11.5% (2,217/21,421; 95% confidence interval, 8.2 to 15.8); the prevalence was comparable in the studies reporting prevalence at 3, 6, 12, 13–59, or ≥60 months after AGE.40 Wide variations in PI-IBS prevalence in different studies are related, at least in part, to the difference in the frequency of various risk factors for PI-IBS development in the diverse patient population.
Table 1.
Incidence, Etiology of Acute Gastroenteritis, and Risk Factors for Postinfection IBS in Cases and Controls
| Study (year) | Country | Cause of gastroenteritis | IBS in cases (%) | IBS in controls (%) | Risk factors for postinfection IBS |
|---|---|---|---|---|---|
| McKendrick et al. (1994)11 | UK | Salmonella | 12/38 (31.6) | No control | Severity of acute illness, vomiting and weight loss |
| Gwee et al. (1996)12 | UK | Shigella, Campylobacter, Salmonella | 20/75 (26.6) | No control | Anxiety, depression, somatization, and neurotic trait |
| Neal et al. (1997)14 | UK | Bacteria | 23/347 (6.6) | No control | Longer duration of diarrhea, younger age, and female sex |
| Gwee et al. (1999)13 | UK | - | 19/109 (17.4) | Psychological and rectal biopsy: 21 HS |
Psychological factors and persistent rectal inflammation |
| Rodríguez et al. (1999)15 | UK | Bacteria | 14/318 (4.4) | 2,027/58,4308 (0.3) | Not evaluated |
| Mearin et al. (2005)16 | Spain | Salmonella | 31/266 (11.6) | 5/333 (1.5) | No risk factor identified |
| Ilnyckyj et al. (2003)17 | Canada | Traveler’s diarrhea | 2/48 (4.2) | 1/61 (1.6) | Not evaluated |
| Dunlop et al. (2003)18 | UK | Campylobacter | 103/747 (13.8) | No control | Increased enterochromaffin cells in lamina propria and depression |
| Parry et al. (2003)19 | UK | Campylobacter, Salmonella | 18/108 (16.7) | 4/219 (1.9) | Not evaluated |
| Wang et al. (2004)34 | China | Shigella | 24/295 (8.1) | 2/243 (0.8) | Longer diarrhea, IL-1β mRNA expression and mast cell in ileum and rectosigmoid |
| Okhuysen et al. (2004)35 | USA | Traveler’s diarrhea | 60 (6) | No control | More diarrhea, medical consultation, and stool negative for the pathogen |
| Ji et al. (2005)36 | Korea | Shigellosis | 15/101 (14.8) | 6/102 (5.8) | Diarrhea duration |
| Parry et al. (2005)37 | UK | Bacteria | 16/107 (15) | No control | Smoking |
| Kim et al. (2006)20 | Korea | Shigella | 13/95 (13.6) | 4/105 (3.8) | Pre-existing FBD other than IBS |
| Marshall et al. (2006)21 | Canada | Escherichia coli, Campylobacter | 417/1,368 (30.5) | 71/701 (10.2) | Young age, female, bloody stools, weight loss, and long diarrhea |
| Borgaonkar et al. (2006)22 | Canada | Bacteria | 7/191 (3.7) | No control | Fever during gastroenteritis |
| Stermer et al. (2006)23 | Israel | Traveler’s diarrhea | 16/118 (13.6) | 7/287 (2.4) | Female gender, abdominal pain, long diarrhea, and antibiotic use |
| Marshall et al. (2007)24 | Canada | Viral diarrhea | 21/89 (23.6) | 1/29 (3.4) | Vomiting during gastroenteritis |
| Spence et al. (2007)25 | New Zealand | Campylobacter | 86/581 (14.8) | No control | Psychological comorbidity and lack or rest during gastroenteritis |
| Hanevik et al. (2009)26 | Norway | Giardia | 66/82 (80.5) | No control | Not evaluated |
| Zanini et al. (2012)27 | Italy | Norovirus | 40/186 (21.5) | 3/198 (1.5) | Not evaluated |
| Cremon et al. (2014)28 | Italy | Salmonella enterica subsp. enterica serovar Typhi | 75/204 (36.8) | 44/189 (23.3) | Anxiety and functional dyspepsia |
| Persson et al. (2015)29 | Norway | Giardia | 224/724 (32) | 96/847 (11.4) | Not evaluated |
| Wadhwa et al. (2016)30 | USA | Clostridium difficile | 52/205 (25) | No control | Longer infection duration, current anxiety, and higher BMI |
| Andresen et al. (2016)31 | Germany | Shiga-like toxin-producing E. coli | 98/389 (25.3) | No control | Higher somatization and anxiety scores |
| Dormond et al. (2016)38 | USA | Giardia lamblia | 16/80 (20.0) | 294 (5.4) | Axis I psychological disorders |
| Rahman et al. (2018)32 | Bangladesh | E. coli, Campylobacter, Vibrio cholerae, Salmonella, Shigella, Aeromonas | 57/345 (16.5) | 9/345 (2.6) | Dyspeptic symptoms, continuing bowel dysfunction, and weight loss |
| Parida et al. (2019)33 | India |
E. coli, Campylobacter, V. cholerae, Salmonella, Shigella, Yersinia enterocolitica |
35/136 (25.7) | No control | Younger age, prolonged duration of diarrhea and abdominal cramps |
| Iacob et al. (2020)39 | Romania | C. difficile, Salmonella, Campylobacter jejuni, rotavirus | 25/45 (55.5) | 6/45 (13.3) | Female gender, rotavirus and C. jejuni infection |
IBS, irritable bowel syndrome; HS, healthy subject; IL, interleukin; FBD, functional bowel disease; BMI, body mass index.
2. Risk factors for PI-IBS
Multiple factors have been identified to be associated with a greater risk of developing PI-IBS following AGE. The risk factors for the development of PI-IBS include female sex, younger age, smoking, severity of AGE (including stool frequency and diarrhea duration longer than 1 week), abdominal pain, bleeding per rectum, treatment with antibiotics, anxiety, depression, somatization, neuroticism, recent adverse life events, hypochondriasis, extroversion, negative illness beliefs, history of stress, sleep disturbance, and family history of FGIDs.10,33,40 In one of our studies, we found that the patients who continued to have lower and upper gastrointestinal (GI) symptoms referred to as chronic bowel dysfunction and dyspeptic symptoms during the initial period of follow-up after AGE were more likely to develop IBS and functional dyspepsia after the 6-month cutoff time point, which is required to diagnose these conditions as per the Rome criteria.32,41
In a Canadian study on PI-IBS, the authors developed and validated a risk score.42 For the risk score development, the authors used the risk factors mentioned above, including the demographic and psychological parameters and AGE-related parameters. In addition, they used weight loss of at least 10 lbs (4.5 kg) as one of the parameters. The authors found that if the score was low (score <42), 10% developed PI-IBS. In the intermediate (43–68) and high (>69) score groups, 35% and 60% of AGE patients developed PI-IBS, respectively.42 This scoring system was validated in an Indian cohort, which showed that with a cutoff value of >50, the sensitivity and specificity of the score to predict the development of PI-IBS at 6-month follow-up were 91.4% and 84.2%, respectively.33 Since the different cohorts of AGE patients reported are pretty heterogeneous concerning the above risk factors and the types of the organism causing the gastroenteritis episodes, the wide variation in the prevalence of PI-IBS ranging from 3.7% to 85.5% following AGE is not unexpected.10
ETIOLOGY AND PATHOGENESIS
1. Infections that are known to be followed by PI-IBS
As shown in Table 1, infection with bacterial, viral, and protozoal agents causing AGE has been reported to be followed by PI-IBS.11-39 Among bacteria, infection with organisms causing dysentery such as Shigella is more likely to result in PI-IBS than others.10 However, recently, even bacterial pathogens that conventionally do not invade the gut mucosa, such as Vibrio cholerae are also shown to cause PI-IBS.32,33 Protozoal agents such as Giardia lamblia have been associated with PI-IBS development with a high frequency.10,40 However, it is essential to mention that most PI-IBS studies following protozoal infection have been reported from two countries: Norway and Turkey.6,10 In some of these studies, a negative result on stool microscopy during follow-up was considered to denote eradication of giardiasis.26,29 However, it is well-known that the properly performed stool microscopy detect Giardia only in two-thirds of patients.43 Hence, some of these patients diagnosed with PI-IBS following Giardia infection might be suffering from chronic giardiasis. Since chronic giardiasis is known to cause malabsorption syndrome and chronic diarrhea,44 and PI-IBS is often diarrhea-predominant type, there might be diagnostic confusion between chronic giardiasis and PI-IBS. More adequately designed studies are needed on this issue.
PI-IBS less commonly follows viral diarrhea. As shown in Table 2, in the four case-control studies on PI-IBS following viral diarrhea, 0.4% to 12.5% of patients following viral diarrhea developed PI-IBS;24,27,45,46 however, the frequency among viral diarrhea patients was greater than that among the controls. The low frequency of PI-IBS in patients with viral diarrhea is expected; viral agents causing AGE are somewhat less invasive than bacterial agents such as Shigella.
Table 2.
Case-Control Studies on PI-IBS Following AGE Due to Viruses
| Study (year) | No. of AGE patients | PI-IBS in AGE patients, No. (%) | No. of controls | PI-IBS in controls, No. (%) |
|---|---|---|---|---|
| Porter et al. (2012)45 | 1,718 | 7 (0.4) | 6,875 | 42 (0.6) |
| Zanini et al. (2012)27 | 178 | 14 (7.8) | 198 | 3 (1.5) |
| Saps et al. (2009)46 | 44 | 4 (9.1) | 44 | 2 (4.5) |
| Marshall et al. (2007)24 | 87 | 11 (12.5) | 29 | 3 (10.3) |
| Total | 2,027 | 36 (1.7) | 7,146 | 50 (0.6) |
PI-IBS, postinfection irritable bowel syndrome; AGE, acute gastroenteritis.
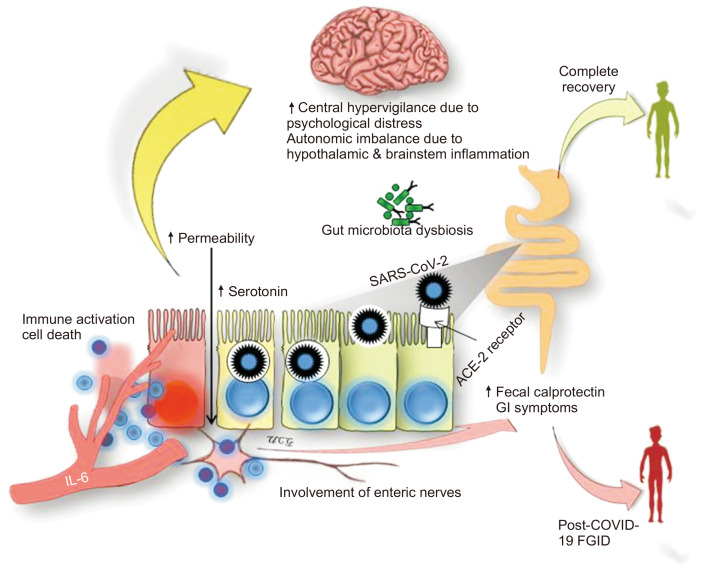
An important issue is whether infection with severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), an RNA virus, originating from the Wuhan city, China, in December 2019, resulting in the devastating pandemic of COVID-19, can cause PI-FGIDs?7 This is not entirely unexpected as angiotensin-converting enzyme-2 (ACE-2) receptors, the binding site of SARS-CoV-2, are also present in intestinal epithelial cells; the virus has been detected in feces in about half of the patients, GI symptoms, including diarrhea occur in about one-fifth of patients, fecal calprotectin, a marker of GI inflammation, and mucosal serotonin are raised in patients with COVID-19, macroscopic as well as histological evidence of GI mucosal injury occur in them, and gut microbiota dysbiosis has been shown to occur in patients with COVID-19.7-9,47-55 Moreover, the SARS-CoV-2 virus has an affinity to involve central and peripheral nervous systems as evidenced by anosmia and ageusia occurrence in 15% to 30% of patients and autopsy findings of marked inflammation of olfactory bulb, cerebrum, and brainstem.53,54 Patients with COVID-19 have also been shown to increased intestinal permeability.56 Moreover, the psychological stress due to the COVID-19 pandemic may be associated with central hypervigilance.55 Fig. 1 summarizes all these putative pathophysiological mechanisms, which may be related to the development of post-COVID-19 FGIDs. Though based on these potential pathophysiological mechanisms, we hypothesized on a possibility of an inevitable surge of post-COVID-19 FGIDs;7 it is yet to be proved by a published study.
Fig. 1.
Putative pathophysiological mechanism of post-coronavirus disease 2019 (COVID-19) functional gastrointestinal disorders (FGIDs), currently called disorders of the gut-brain interaction (DGBI). Severe acute respiratory distress syndrome coronavirus-2 (SARS-CoV-2) affects the gastrointestinal (GI) tract after entering via the angiotensin-converting enzyme-2 (ACE-2) receptor, which leads to immune activation (including increase in inflammatory markers such as interleukin-6 [IL-6]), damage to intestinal mucosa associated with GI symptoms, the presence of SARS-CoV-2 ribonucleic acid in feces, abnormal intestinal permeability, increased fecal calprotectin, increased mucosal serotonin, and gut microbiota dysbiosis. Increased visceral sensitivity at the gut level due to enteric nervous system involvement and at the central level due to psychological stress may contribute to the development of post-COVID-19 FGIDs/DGBI.
2. Pathogenesis of PI-IBS
The pathogenesis of PI-IBS involves agent, host, and host-agent interaction-related factors. As shown in Tables 1 and 2, the reported frequency of PI-IBS development is highest with protozoa, intermediate with bacteria, and lowest with the viral agents.10 Several host factors interplay with the infectious agent leading to protracted long-term inflammation of the gut, which might be compounded by inadequate T regulatory responses controlling the inflammation.10 Chronic inflammation and altered intestinal permeability have been shown in patients with diarrhea-predominant IBS (IBS-D), including PI-IBS.10,57 In a case-control study in a cohort of 21 AGE patients due to Campylobacter and 12 controls, enteroendocrine cells, CD3, CD4, and CD8 lymphocyte counts in lamina propria and intraepithelial lymphocytes and small intestinal permeability were increased in PI-IBS patients than the controls.58 In a recent systematic review and meta-analysis, small intestinal permeability was more often abnormal among patients with PI-IBS (4/4 studies) and IBS-D (9/13 studies) than those with constipation-predominant IBS (IBS-C; 2/7 studies).59 Lack of down-regulation in the inflammatory response following acute GI infection contributes to the development of PI-IBS. In a study on eight PI-IBS patients, seven with AGE not developing PI-IBS and 18 healthy controls, the patients with PI-IBS exhibited increased inflammatory cell infiltrate and higher levels of interleukin (IL)-1β (a pro-inflammatory cytokine) mRNA in rectal biopsies by reverse transcriptase-polymerase chain reaction even 3 months after AGE compared to the other groups.60 PI-IBS patients have also been shown to have increased levels of peripheral IL-6, pleiotropic cytokine activating inflammatory nuclear factor (NF)-kB transcription pathway, compared to the healthy controls.61,62 Host factors are essential determinant of the protracted inflammatory response following GI infection. Polymorphisms in several pro- and anti-inflammatory cytokine genes increasing and decreasing their responses, respectively, have been shown to play an important role. The Canadian Walkerton cohort polymorphism study revealed a trend towards an association between single nucleotide polymorphisms in the gene involved in the immune and gut epithelial barrier pathways located in TLR9 (encoding pattern recognition receptor [rs352139 and P545P]) and CDH1 (encoding a tight junction protein [rs16260, -C160A], and IL-6 (rs1800795, -G174C) and development of PI-IBS.63 In another study, TNF alpha polymorphism was associated with PI-IBS following Campylobacter jejuni infection.64 We found that patients with IBS-D and PI-IBS had higher rectal mucosal serotonin and polymorphism in the serotonin re-uptake protein gene than controls.65
Gut microbiota dysbiosis is another crucial contributor to the development of PI-IBS. Gut microbiota gets altered during the acute phase in patients with AGE66 that may last long after the AGE episode.67 PI-IBS patients have been shown to have unique gut microbiota signature compared to the other IBS patients.68 De Palma et al.69 successfully colonized mice with fecal microbiota either from IBS-D patients or healthy subjects; the mice receiving microbiota from IBS-D patients as opposed to those receiving from healthy subjects developed IBS phenotypes such as alteration in gut transit, anxiety-like behavior, abnormal intestinal permeability, higher CD3 positive cells in intestinal crypts, C3, CXR3, NF-kB, and β defensin. Gut microbiota in patients with IBS has been shown to have reduced diversity akin to patients with non-PI-IBS.67 Relatively greater abundances of microbes of Bacteroidetes phylum (including Bacteroides and Prevotella) and a relative reduction in those of the phylum Firmicutes such as Clostridia are shown in patients with PI-IBS than controls.62 However, more data are needed to evaluate whether such alterations in the gut microbiota are the causes or effects of AGE and PI-IBS; these patients might have modified their diet, received antibiotics, and altered gut motility, which are known to alter the gut microbiota.70-72 However, since gut microbiota influences gut motility, visceral hypersensitivity, immune response, psychological dysfunction, sleep disturbances, serotonin, and bile acid metabolism, which are associated with FGIDs, including IBS, the role of gut microbiota dysbiosis in the pathogenesis of PI-IBS cannot be over-estimated.70-72
3. Clinical presentation and diagnosis of PI-IBS
The criteria laid down by the Rome Foundation Working Team on PI-IBS suggest that this condition should be diagnosed when new onset Rome-criteria positive IBS develops in an individual after AGE characterized by two or more of the following features; diarrhea, vomiting, and fever (when stool culture is not available).6 However, it is essential to note that though the Rome Foundation Working Team on PI-IBS suggested using Rome IV criteria for diagnosis of PI-IBS, considering the low sensitivity of Rome IV criteria compared to the Rome III criteria to diagnose IBS,1,73 the latter criteria may also be acceptable.
As Rome criteria require that the symptoms must have started at least 6-month before and the patients must be symptomatic during the last 3 months, many patients continuing to fulfill the Rome criteria for IBS following AGE would not be diagnosed as IBS unless they wait for 6 months to meet the criteria.4,6,41 Hence, we proposed that if the symptoms of IBS are present during the follow-up after AGE, but the subjects did not fulfill the duration criteria suggested by the Rome Foundation, the condition be called chronic bowel dysfunction,32 which the Rome Foundation Working Team has accepted.6
An essential clinical clue to the possibility of PI-IBS comes from the targeted question to the patients “When did the symptom start?” If the patient recalls the date or month, the clinicians should be alert about the possibility of PI-IBS. Most patients with PI-IBS have either IBS-D or mixed type of IBS.40 PI-IBS may have other overlapping FGIDs. In one of our earlier studies, of 66 of 345 AGE patients developing PI-FGIDs, 16 (24%) had overlapping functional dyspepsia.32 Patients may also have other extra-GI symptoms such as psychological comorbidity, sleep dysfunction similar to the other non-PI-IBS.
INVESTIGATIONS FOR PATIENTS WITH PI-IBS
The investigations in patients with PI-IBS include routine hematology, blood biochemistry, C-reactive protein, fecal calprotectin, and stool microscopy to look for protozoa, such as Giardia.6 However, it is essential to mention that in the presence of severe symptoms, significant weight loss, anorexia, local epidemiology, and the other clinical clues (based even on the results of the initial tests), investigations to rule out other causes of chronic diarrhea such as celiac disease, tropical sprue, microscopic and collagenous colitis, and inflammatory bowel disease are warranted.6 Since about one-tenth of patients with Rome-criteria defined, PI-IBS may have post-infectious malabsorption syndrome (tropical sprue) in tropical countries and visitors to these endemic regions, the investigations for tropical sprue should be undertaken if clinically indicated.32 PI-IBS and tropical sprue are spectrum disorders. The investigations for tropical sprue include D-xylose test, fecal fat excretion, serum vitamin B12 estimation, and endoscopic duodenal biopsy.32 Since a third of patients with tropical sprue and about a fifth of patients with IBS-D have small intestinal bacterial overgrowth (SIBO), a glucose hydrogen breath test may be done as a specialized investigation to diagnose SIBO in patients with PI-IBS.74-76 Pimentel’s group found that anti-cytolethal distending toxin B and anti-vinculin antibodies are helpful in diagnosing IBS-D in general and PI-IBS, in particular among patients with diarrhea.77 Hence, these may be helpful in diagnosis in selected patients.
TREATMENT AND PROGNOSIS OF PI-IBS
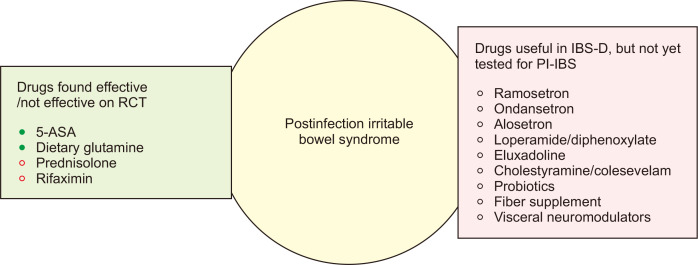
The data on the treatment of PI-IBS are scanty. Hence, currently, these patients are treated in the same way as any IBS-D patient.6,78 Fig. 2 outlines the drugs which have been found useful or not in the treatment of PI-IBS and the drugs that have been established in the treatment of IBS-D but have not been evaluated for PI-IBS.6,78 The name of drugs for the treatment of IBS-D and their dosages are summarized in Table 3. Though 5-aminosalicylic acid was not found useful in IBS-D in randomized controlled trials, it benefited the subset of patients with PI-IBS.79 In a randomized controlled trial from China, dietary glutamine was found useful in PI-IBS patients.80 Prednisolone and rifaximin were not found useful in PI-IBS.81-83 However, the rifaximin study had several limitations, such as a small sample size, low dose of rifaximin (550 mg twice daily), and use of early peak-criteria on lactulose hydrogen breath test for diagnosis of SIBO, which is known to have poor specificity.82 Since restriction of fermentable oligo-di-mono-saccharide and polyol (FODMAP) diet has been shown helpful in IBS, it is also expected to help patients with PI-IBS.3,78,84 However, there is no study yet on the low FODMAP diet in the management of PI-IBS. Visceral neuromodulators, particularly the tricyclic anti-depressants, are expected to be helpful in the management of PI-IBS as these tend to reduce diarrhea by their anticholinergic activity.3,78 However, no study has been conducted on this issue in patients with PI-IBS. Since other subtypes of IBS such as mixed IBS and even rarely constipation-predominant IBS may occur after AGE, the management of different subtypes of IBS may also be applicable in patients with PI-IBS.3,78
Fig. 2.
Potentially useful drugs in the treatment of PI-IBS. In the light green box, the drugs that have been evaluated in RCTs are listed (filled green circles indicate the drugs that were found to be effective in the RCTs, and empty red circles show those that were found to be ineffective). The pink box lists the drugs that were found to be effective in diarrhea-predominant IBS but have not yet been evaluated in PI-IBS.
5-ASA, 5-aminosalicylic acid; RCT, randomized controlled trial; IBS, irritable bowel syndrome; IBS-D, diarrhea-predominant IBS; PI-IBS, postinfection IBS.
Table 3.
Drugs (Including the Dosages) Used in the Treatment of Diarrhea-Predominant Irritable Bowel Syndrome (IBS) That May Also Be Useful in Postinfection IBS
| Drug class | Agents | Dose |
|---|---|---|
| Opiates | Loperamide | 2–4 mg, 4 times per day |
| Diphenoxylate | 2.5–5 mg, 4 times per day | |
| Eluxadoline | 100 mg twice daily | |
| Bile acid modulators | Cholestyramine | 4 g daily or up to 4 times per day |
| Colestipol | 4 g daily or up to 4 times per day | |
| Colesevelam | 1,875 mg up to twice daily | |
| Obeticholic acid | 25 mg per day | |
| 5HT-3 receptor antagonists | Alosetron | 0.5–1 mg twice daily |
| Ondansetron | 2–8 mg twice daily | |
| Ramosetron | 5 µg per day | |
| Gut microbiota manipulators | ||
| Probiotics | Lactobacillus sp., Saccharomyces boulardii | |
| Antibiotics | Rifaximin | 550 mg thrice daily for 14 days |
| Fiber supplements | Calcium polycarbophil | 5–10 g daily |
| Psyllium | 10–20 g daily | |
| Pectin | 2 Capsules daily before meal |
The studies showed that the prognosis of PI-IBS patients is generally good as almost half of them recover during the long-term follow-up.85,86 A Korean study showed that though the rates of IBS increased till 3-year, it reduced thereafter. Among the PI-IBS patients, post-viral PI-IBS is more often transient than post-bacterial IBS.87
CONCLUSION AND FUTURE DIRECTIONS
PI-IBS is a disorder of gut-brain interaction (previously called FGID), which undoubtedly proved that some patients with FGIDs are not entirely functional.3,4 PI-IBS may occur following bacterial, protozoal, and viral AGE.10 Two recent studies, one from Bangladesh and the other from India showed that cholera may be followed by the development of PI-IBS.32,33 A pooled prevalence of PI-IBS was 11.5% after an episode of AGE.40 Most patients with PI-IBS either present as IBS-D or mixed subtype of IBS.40 Overlap of PI-IBS with other FGIDs, mainly, functional dyspepsia is common.32 One-fifth of patients with Rome criteria positive PI-IBS may have PI-malabsorption, popularly known as tropical sprue.32 It is believed that the development of PI-IBS may follow COVID-19.7 However, studies are urgently needed to evaluate the frequency, spectrum, and risk factors for the development of PI-IBS, if any, following COVID-19. Since most patients with PI-IBS presents with the non-constipation type of IBS, the drugs used to treat non-constipation IBS may also be useful in PI-IBS treatment. However, most of these drugs have not been evaluated for the treatment of IBS. Of the four drugs evaluated by randomized controlled trial, prednisolone and rifaximin were found ineffective in PI-IBS treatment.81-83 Dietary glutamine was found useful in the treatment of PI-IBS in a Chinese study.80 5-Aminosalicylic acid undergoing randomized controlled trial was not found useful in IBS-D patients.79 However, in the subgroup of patients with PI-IBS, 5-aminosalicylic acid was found useful in IBS-D patients.79 More studies are needed to evaluate the efficacy of various drugs in the treatment of PI-IBS.
Footnotes
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
REFERENCES
- 1.Sperber AD, Bangdiwala SI, Drossman DA, et al. Worldwide prevalence and burden of functional gastrointestinal disorders, results of Rome Foundation Global Study. Gastroenterology. 2021;160:99–114. doi: 10.1053/j.gastro.2020.04.014. [DOI] [PubMed] [Google Scholar]
- 2.Gwee KA, Ghoshal UC, Chen M. Irritable bowel syndrome in Asia: pathogenesis, natural history, epidemiology, and management. J Gastroenterol Hepatol. 2018;33:99–110. doi: 10.1111/jgh.13987. [DOI] [PubMed] [Google Scholar]
- 3.Ghoshal UC. Marshall and Warren Lecture 2019: a paradigm shift in pathophysiological basis of irritable bowel syndrome and its implication on treatment. J Gastroenterol Hepatol. 2020;35:712–721. doi: 10.1111/jgh.15032. [DOI] [PubMed] [Google Scholar]
- 4.Drossman DA, Hasler WL. Rome IV-functional GI disorders: disorders of gut-brain interaction. Gastroenterology. 2016;150:1257–1261. doi: 10.1053/j.gastro.2016.03.035. [DOI] [PubMed] [Google Scholar]
- 5.Barbara G, Feinle-Bisset C, Ghoshal UC, et al. The intestinal microenvironment and functional gastrointestinal disorders. Gastroenterology. 2016;150:1305–1318. doi: 10.1053/j.gastro.2016.02.028. [DOI] [PubMed] [Google Scholar]
- 6.Barbara G, Grover M, Bercik P, et al. Rome Foundation Working Team report on post-infection irritable bowel syndrome. Gastroenterology. 2019;156:46–58. doi: 10.1053/j.gastro.2018.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schmulson M, Ghoshal UC, Barbara G. Managing the inevitable surge of post-COVID-19 functional gastrointestinal disorders. Am J Gastroenterol. 2021;116:4–7. doi: 10.14309/ajg.0000000000001062. [DOI] [PubMed] [Google Scholar]
- 8.Kumar A, Arora A, Sharma P, et al. Gastrointestinal and hepatic manifestations of corona virus disease-19 and their relationship to severe clinical course: a systematic review and meta-analysis. Indian J Gastroenterol. 2020;39:268–284. doi: 10.1007/s12664-020-01058-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ghoshal UC, Ghoshal U, Mathur A, et al. The spectrum of gastrointestinal symptoms in patients with coronavirus disease-19: predictors, relationship with disease severity, and outcome. Clin Transl Gastroenterol. 2020;11:e00259. doi: 10.14309/ctg.0000000000000259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ghoshal UC, Rahman MM. Post-infection irritable bowel syndrome in the tropical and subtropical regions: Vibrio cholerae is a new cause of this well-known condition. Indian J Gastroenterol. 2019;38:87–94. doi: 10.1007/s12664-019-00959-2. [DOI] [PubMed] [Google Scholar]
- 11.McKendrick MW, Read NW. Irritable bowel syndrome: post salmonella infection. J Infect. 1994;29:1–3. doi: 10.1016/S0163-4453(94)94871-2. [DOI] [PubMed] [Google Scholar]
- 12.Gwee KA, Graham JC, McKendrick MW, et al. Psychometric scores and persistence of irritable bowel after infectious diarrhoea. Lancet. 1996;347:150–153. doi: 10.1016/S0140-6736(96)90341-4. [DOI] [PubMed] [Google Scholar]
- 13.Gwee KA, Leong YL, Graham C, et al. The role of psychological and biological factors in postinfective gut dysfunction. Gut. 1999;44:400–406. doi: 10.1136/gut.44.3.400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Neal KR, Hebden J, Spiller R. Prevalence of gastrointestinal symptoms six months after bacterial gastroenteritis and risk factors for development of the irritable bowel syndrome: postal survey of patients. BMJ. 1997;314:779–782. doi: 10.1136/bmj.314.7083.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rodríguez LA, Ruigómez A. Increased risk of irritable bowel syndrome after bacterial gastroenteritis: cohort study. BMJ. 1999;318:565–566. doi: 10.1136/bmj.318.7183.565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mearin F, Pérez-Oliveras M, Perelló A, et al. Dyspepsia and irritable bowel syndrome after a Salmonella gastroenteritis outbreak: one-year follow-up cohort study. Gastroenterology. 2005;129:98–104. doi: 10.1053/j.gastro.2005.04.012. [DOI] [PubMed] [Google Scholar]
- 17.Ilnyckyj A, Balachandra B, Elliott L, Choudhri S, Duerksen DR. Post-traveler's diarrhea irritable bowel syndrome: a prospective study. Am J Gastroenterol. 2003;98:596–599. doi: 10.1111/j.1572-0241.2003.07297.x. [DOI] [PubMed] [Google Scholar]
- 18.Dunlop SP, Jenkins D, Neal KR, Spiller RC. Relative importance of enterochromaffin cell hyperplasia, anxiety, and depression in postinfectious IBS. Gastroenterology. 2003;125:1651–1659. doi: 10.1053/j.gastro.2003.09.028. [DOI] [PubMed] [Google Scholar]
- 19.Parry SD, Stansfield R, Jelley D, et al. Does bacterial gastroenteritis predispose people to functional gastrointestinal disorders? A prospective, community-based, case-control study. Am J Gastroenterol. 2003;98:1970–1975. doi: 10.1111/j.1572-0241.2003.07664.x. [DOI] [PubMed] [Google Scholar]
- 20.Kim HS, Kim MS, Ji SW, Park H. The development of irritable bowel syndrome after Shigella infection: 3 year follow-up study. Korean J Gastroenterol. 2006;47:300–305. [PubMed] [Google Scholar]
- 21.Marshall JK, Thabane M, Garg AX, et al. Incidence and epidemiology of irritable bowel syndrome after a large waterborne outbreak of bacterial dysentery. Gastroenterology. 2006;131:445–450. doi: 10.1053/j.gastro.2006.05.053. [DOI] [PubMed] [Google Scholar]
- 22.Borgaonkar MR, Ford DC, Marshall JK, Churchill E, Collins SM. The incidence of irritable bowel syndrome among community subjects with previous acute enteric infection. Dig Dis Sci. 2006;51:1026–1032. doi: 10.1007/s10620-006-9348-1. [DOI] [PubMed] [Google Scholar]
- 23.Stermer E, Lubezky A, Potasman I, Paster E, Lavy A. Is traveler's diarrhea a significant risk factor for the development of irritable bowel syndrome? A prospective study. Clin Infect Dis. 2006;43:898–901. doi: 10.1086/507540. [DOI] [PubMed] [Google Scholar]
- 24.Marshall JK, Thabane M, Borgaonkar MR, James C. Postinfectious irritable bowel syndrome after a food-borne outbreak of acute gastroenteritis attributed to a viral pathogen. Clin Gastroenterol Hepatol. 2007;5:457–460. doi: 10.1016/j.cgh.2006.11.025. [DOI] [PubMed] [Google Scholar]
- 25.Spence MJ, Moss-Morris R. The cognitive behavioural model of irritable bowel syndrome: a prospective investigation of patients with gastroenteritis. Gut. 2007;56:1066–1071. doi: 10.1136/gut.2006.108811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hanevik K, Dizdar V, Langeland N, Hausken T. Development of functional gastrointestinal disorders after Giardia lamblia infection. BMC Gastroenterol. 2009;9:27. doi: 10.1186/1471-230X-9-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zanini B, Ricci C, Bandera F, et al. Incidence of post-infectious irritable bowel syndrome and functional intestinal disorders following a water-borne viral gastroenteritis outbreak. Am J Gastroenterol. 2012;107:891–899. doi: 10.1038/ajg.2012.102. [DOI] [PubMed] [Google Scholar]
- 28.Cremon C, Stanghellini V, Pallotti F, et al. Salmonella gastroenteritis during childhood is a risk factor for irritable bowel syndrome in adulthood. Gastroenterology. 2014;147:69–77. doi: 10.1053/j.gastro.2014.03.013. [DOI] [PubMed] [Google Scholar]
- 29.Persson R, Wensaas KA, Hanevik K, Eide GE, Langeland N, Rortveit G. The relationship between irritable bowel syndrome, functional dyspepsia, chronic fatigue and overactive bladder syndrome: a controlled study 6 years after acute gastrointestinal infection. BMC Gastroenterol. 2015;15:66. doi: 10.1186/s12876-015-0296-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wadhwa A, Al Nahhas MF, Dierkhising RA, et al. High risk of post-infectious irritable bowel syndrome in patients with Clostridium difficile infection. Aliment Pharmacol Ther. 2016;44:576–582. doi: 10.1111/apt.13737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Andresen V, Löwe B, Broicher W, et al. Post-infectious irritable bowel syndrome (PI-IBS) after infection with Shiga-like toxin-producing Escherichia coli (STEC) O104:H4: a cohort study with prospective follow-up. United European Gastroenterol J. 2016;4:121–131. doi: 10.1177/2050640615581113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rahman MM, Ghoshal UC, Sultana S, et al. Long-term gastrointestinal consequences are frequent following sporadic acute infectious diarrhea in a tropical country: a prospective cohort study. Am J Gastroenterol. 2018;113:1363–1375. doi: 10.1038/s41395-018-0208-3. [DOI] [PubMed] [Google Scholar]
- 33.Parida PK, Mishra D, Pati GK, et al. A prospective study on incidence, risk factors, and validation of a risk score for post-infection irritable bowel syndrome in coastal eastern India. Indian J Gastroenterol. 2019;38:134–142. doi: 10.1007/s12664-019-00943-w. [DOI] [PubMed] [Google Scholar]
- 34.Wang LH, Fang XC, Pan GZ. Bacillary dysentery as a causative factor of irritable bowel syndrome and its pathogenesis. Gut. 2004;53:1096–1101. doi: 10.1136/gut.2003.021154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Okhuysen PC, Jiang ZD, Carlin L, Forbes C, DuPont HL. Post-diarrhea chronic intestinal symptoms and irritable bowel syndrome in North American travelers to Mexico. Am J Gastroenterol. 2004;99:1774–1778. doi: 10.1111/j.1572-0241.2004.30435.x. [DOI] [PubMed] [Google Scholar]
- 36.Ji S, Park H, Lee D, Song YK, Choi JP, Lee SI. Post-infectious irritable bowel syndrome in patients with Shigella infection. J Gastroenterol Hepatol. 2005;20:381–386. doi: 10.1111/j.1440-1746.2005.03574.x. [DOI] [PubMed] [Google Scholar]
- 37.Parry SD, Barton JR, Welfare MR. Factors associated with the development of post-infectious functional gastrointestinal diseases: does smoking play a role? Eur J Gastroenterol Hepatol. 2005;17:1071–1075. doi: 10.1097/00042737-200510000-00010. [DOI] [PubMed] [Google Scholar]
- 38.Dormond M, Gutierrez RL, Porter CK. Giardia lamblia infection increases risk of chronic gastrointestinal disorders. Trop Dis Travel Med Vaccines. 2016;2:17. doi: 10.1186/s40794-016-0030-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Iacob T, Țățulescu DF, Lupșe MS, Dumitrașcu DL. Post-infectious irritable bowel syndrome after a laboratory-proven enteritis. Exp Ther Med. 2020;20:3517–3522. doi: 10.3892/etm.2020.9018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Klem F, Wadhwa A, Prokop LJ, et al. Prevalence, risk factors, and outcomes of irritable bowel syndrome after infectious enteritis: a systematic review and meta-analysis. Gastroenterology. 2017;152:1042–1054. doi: 10.1053/j.gastro.2016.12.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lacy BE, Mearin F, Chang L, et al. Bowel disorders. Gastroenterology. 2016;150:1393–1407. doi: 10.1053/j.gastro.2016.02.031. [DOI] [PubMed] [Google Scholar]
- 42.Thabane M, Simunovic M, Akhtar-Danesh N, Marshall JK. Development and validation of a risk score for post-infectious irritable bowel syndrome. Am J Gastroenterol. 2009;104:2267–2274. doi: 10.1038/ajg.2009.302. [DOI] [PubMed] [Google Scholar]
- 43.Jangra M, Dutta U, Shah J, et al. Role of polymerase chain reaction in stool and duodenal biopsy for diagnosis of giardiasis in patients with persistent/chronic diarrhea. Dig Dis Sci. 2020;65:2345–2353. doi: 10.1007/s10620-019-06042-2. [DOI] [PubMed] [Google Scholar]
- 44.Ghoshal UC, Mehrotra M, Kumar S, et al. Spectrum of malabsorption syndrome among adults & factors differentiating celiac disease & tropical malabsorption. Indian J Med Res. 2012;136:451–459. [PMC free article] [PubMed] [Google Scholar]
- 45.Porter CK, Faix DJ, Shiau D, Espiritu J, Espinosa BJ, Riddle MS. Postinfectious gastrointestinal disorders following norovirus outbreaks. Clin Infect Dis. 2012;55:915–922. doi: 10.1093/cid/cis576. [DOI] [PubMed] [Google Scholar]
- 46.Saps M, Pensabene L, Turco R, Staiano A, Cupuro D, Di Lorenzo C. Rotavirus gastroenteritis: precursor of functional gastrointestinal disorders? J Pediatr Gastroenterol Nutr. 2009;49:580–583. doi: 10.1097/MPG.0b013e31819bcbd2. [DOI] [PubMed] [Google Scholar]
- 47.Wan Y, Shang J, Graham R, Baric RS, Li F. Receptor recognition by the novel coronavirus from Wuhan: an analysis based on decade-long structural studies of SARS coronavirus. J Virol. 2020;94:e00127–20. doi: 10.1128/JVI.00127-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wong MC, Huang J, Lai C, Ng R, Chan FK, Chan PK. Detection of SARS-CoV-2 RNA in fecal specimens of patients with confirmed COVID-19: a meta-analysis. J Infect. 2020;81:e31–e38. doi: 10.1016/j.jinf.2020.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yeoh YK, Zuo T, Lui GC, et al. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut. 2021;70:698–706. doi: 10.1136/gutjnl-2020-323020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zuo T, Zhang F, Lui GC, et al. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology. 2020;159:944–955. doi: 10.1053/j.gastro.2020.05.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Effenberger M, Grabherr F, Mayr L, et al. Faecal calprotectin indicates intestinal inflammation in COVID-19. Gut. 2020;69:1543–1544. doi: 10.1136/gutjnl-2020-321388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ha S, Jin B, Clemmensen B, et al. Serotonin is elevated in COVID-19-associated diarrhoea. Gut. 2021;70:2015–2017. doi: 10.1136/gutjnl-2020-323542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Reza-Zaldívar EE, Hernández-Sapiéns MA, Minjarez B, et al. Infection mechanism of SARS-COV-2 and its implication on the nervous system. Front Immunol. 2021;11:621735. doi: 10.3389/fimmu.2020.621735.1522e5bc505c4017a8639701d8463bc8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Schurink B, Roos E, Radonic T, et al. Viral presence and immunopathology in patients with lethal COVID-19: a prospective autopsy cohort study. Lancet Microbe. 2020;1:e290–e299. doi: 10.1016/S2666-5247(20)30144-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Twenge JM, Joiner TE. Mental distress among U.S. adults during the COVID-19 pandemic. J Clin Psychol. 2020;76:2170–2182. doi: 10.1002/jclp.23064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cardinale V, Capurso G, Ianiro G, Gasbarrini A, Arcidiacono PG, Alvaro D. Intestinal permeability changes with bacterial translocation as key events modulating systemic host immune response to SARS-CoV-2: a working hypothesis. Dig Liver Dis. 2020;52:1383–1389. doi: 10.1016/j.dld.2020.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ghoshal UC, Ranjan P. Post-infectious irritable bowel syndrome: the past, the present and the future. J Gastroenterol Hepatol. 2011;26 Suppl 3:94–101. doi: 10.1111/j.1440-1746.2011.06643.x. [DOI] [PubMed] [Google Scholar]
- 58.Spiller RC, Jenkins D, Thornley JP, et al. Increased rectal mucosal enteroendocrine cells, T lymphocytes, and increased gut permeability following acute Campylobacter enteritis and in post-dysenteric irritable bowel syndrome. Gut. 2000;47:804–811. doi: 10.1136/gut.47.6.804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hanning N, Edwinson AL, Ceuleers H, et al. Intestinal barrier dysfunction in irritable bowel syndrome: a systematic review. Therap Adv Gastroenterol. 2021;14:1756284821993586. doi: 10.1177/1756284821993586.948a5793b5cb42018f4877a867660e28 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gwee KA, Collins SM, Read NW, et al. Increased rectal mucosal expression of interleukin 1beta in recently acquired post-infectious irritable bowel syndrome. Gut. 2003;52:523–526. doi: 10.1136/gut.52.4.523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Jalanka-Tuovinen J, Salojärvi J, Salonen A, et al. Faecal microbiota composition and host-microbe cross-talk following gastroenteritis and in postinfectious irritable bowel syndrome. Gut. 2014;63:1737–1745. doi: 10.1136/gutjnl-2013-305994. [DOI] [PubMed] [Google Scholar]
- 62.Downs IA, Aroniadis OC, Kelly L, Brandt LJ. Postinfection irritable bowel syndrome: the links between gastroenteritis, inflammation, the microbiome, and functional disease. J Clin Gastroenterol. 2017;51:869–877. doi: 10.1097/MCG.0000000000000924. [DOI] [PubMed] [Google Scholar]
- 63.Villani AC, Lemire M, Thabane M, et al. Genetic risk factors for post-infectious irritable bowel syndrome following a waterborne outbreak of gastroenteritis. Gastroenterology. 2010;138:1502–1513. doi: 10.1053/j.gastro.2009.12.049. [DOI] [PubMed] [Google Scholar]
- 64.Swan C, Duroudier NP, Campbell E, et al. Identifying and testing candidate genetic polymorphisms in the irritable bowel syndrome (IBS): association with TNFSF15 and TNFα. Gut. 2013;62:985–994. doi: 10.1136/gutjnl-2011-301213. [DOI] [PubMed] [Google Scholar]
- 65.Kumar S, Ranjan P, Mittal B, Ghoshal UC. Serotonin transporter gene (SLC6A4) polymorphism in patients with irritable bowel syndrome and healthy controls. J Gastrointestin Liver Dis. 2012;21:31–38. [PubMed] [Google Scholar]
- 66.Gorbach SL, Neale G, Levitan R, Hepner GW. Alterations in human intestinal microflora during experimental diarrhoea. Gut. 1970;11:1–6. doi: 10.1136/gut.11.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Sundin J, Rangel I, Fuentes S, et al. Altered faecal and mucosal microbial composition in post-infectious irritable bowel syndrome patients correlates with mucosal lymphocyte phenotypes and psychological distress. Aliment Pharmacol Ther. 2015;41:342–351. doi: 10.1111/apt.13055. [DOI] [PubMed] [Google Scholar]
- 68.Jalanka J, Salonen A, Fuentes S, de Vos WM. Microbial signatures in post-infectious irritable bowel syndrome: toward patient stratification for improved diagnostics and treatment. Gut Microbes. 2015;6:364–369. doi: 10.1080/19490976.2015.1096486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.De Palma G, Lynch MD, Lu J, et al. Transplantation of fecal microbiota from patients with irritable bowel syndrome alters gut function and behavior in recipient mice. Sci Transl Med. 2017;9:eaaf6397. doi: 10.3410/f.727365371.793533515. [DOI] [PubMed] [Google Scholar]
- 70.Ghoshal UC, Shukla R, Ghoshal U, Gwee KA, Ng SC, Quigley EM. The gut microbiota and irritable bowel syndrome: friend or foe? Int J Inflam. 2012;2012:151085. doi: 10.1155/2012/151085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ghoshal UC, Ghoshal U. Small intestinal bacterial overgrowth and other intestinal disorders. Gastroenterol Clin North Am. 2017;46:103–120. doi: 10.1016/j.gtc.2016.09.008. [DOI] [PubMed] [Google Scholar]
- 72.Wei L, Singh R, Ro S, Ghoshal UC. Gut microbiota dysbiosis in functional gastrointestinal disorders: underpinning the symptoms and pathophysiology. JGH Open. 2021;5:976–987. doi: 10.1002/jgh3.12528.18949b32040c445aabbe4b6c17bc2422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Goyal O, Nohria S, Dhaliwal AS, et al. Prevalence, overlap, and risk factors for Rome IV functional gastrointestinal disorders among college students in northern India. Indian J Gastroenterol. 2021;40:144–153. doi: 10.1007/s12664-020-01106-y. [DOI] [PubMed] [Google Scholar]
- 74.Ghoshal UC, Ghoshal U, Ayyagari A, et al. Tropical sprue is associated with contamination of small bowel with aerobic bacteria and reversible prolongation of orocecal transit time. J Gastroenterol Hepatol. 2003;18:540–547. doi: 10.1046/j.1440-1746.2003.03006.x. [DOI] [PubMed] [Google Scholar]
- 75.Ghoshal UC, Gwee KA. Post-infectious IBS, tropical sprue and small intestinal bacterial overgrowth: the missing link. Nat Rev Gastroenterol Hepatol. 2017;14:435–441. doi: 10.1038/nrgastro.2017.37. [DOI] [PubMed] [Google Scholar]
- 76.Ghoshal UC, Nehra A, Mathur A, Rai S. A meta-analysis on small intestinal bacterial overgrowth in patients with different subtypes of irritable bowel syndrome. J Gastroenterol Hepatol. 2020;35:922–931. doi: 10.1111/jgh.14938. [DOI] [PubMed] [Google Scholar]
- 77.Rezaie A, Park SC, Morales W, et al. Assessment of anti-vinculin and anti-cytolethal distending toxin B antibodies in subtypes of irritable bowel syndrome. Dig Dis Sci. 2017;62:1480–1485. doi: 10.1007/s10620-017-4585-z. [DOI] [PubMed] [Google Scholar]
- 78.Ghoshal UC. Pros and cons while looking through an Asian window on the Rome IV criteria for irritable bowel syndrome: pros. J Neurogastroenterol Motil. 2017;23:334–340. doi: 10.5056/jnm17020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lam C, Tan W, Leighton M, et al. A mechanistic multicentre, parallel group, randomised placebo-controlled trial of mesalazine for the treatment of IBS with diarrhoea (IBS-D) Gut. 2016;65:91–99. doi: 10.1136/gutjnl-2015-309122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zhou Q, Verne ML, Fields JZ, et al. Randomised placebo-controlled trial of dietary glutamine supplements for postinfectious irritable bowel syndrome. Gut. 2019;68:996–1002. doi: 10.1136/gutjnl-2017-315136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Dunlop SP, Jenkins D, Neal KR, et al. Randomized, double-blind, placebo-controlled trial of prednisolone in post-infectious irritable bowel syndrome. Aliment Pharmacol Ther. 2003;18:77–84. doi: 10.1046/j.1365-2036.2003.01640.x. [DOI] [PubMed] [Google Scholar]
- 82.Harris LA. Rifaximin for irritable bowel syndrome (IBS) in Gulf War veterans: losing the battle but winning the war? Dig Dis Sci. 2019;64:609–610. doi: 10.1007/s10620-019-05505-w. [DOI] [PubMed] [Google Scholar]
- 83.Tuteja AK, Talley NJ, Stoddard GJ, Verne GN. Double-blind placebo-controlled study of rifaximin and lactulose hydrogen breath test in Gulf War veterans with irritable bowel syndrome. Dig Dis Sci. 2019;64:838–845. doi: 10.1007/s10620-018-5344-5. [DOI] [PubMed] [Google Scholar]
- 84.Mehtab W, Agarwal A, Singh N, Malhotra A, Makharia GK. All that a physician should know about FODMAPs. Indian J Gastroenterol. 2019;38:378–390. doi: 10.1007/s12664-019-01002-0. [DOI] [PubMed] [Google Scholar]
- 85.Neal KR, Barker L, Spiller RC. Prognosis in post-infective irritable bowel syndrome: a six year follow up study. Gut. 2002;51:410–413. doi: 10.1136/gut.51.3.410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Jung IS, Kim HS, Park H, Lee SI. The clinical course of postinfectious irritable bowel syndrome: a five-year follow-up study. J Clin Gastroenterol. 2009;43:534–540. doi: 10.1097/MCG.0b013e31818c87d7. [DOI] [PubMed] [Google Scholar]
- 87.Thabane M, Marshall JK. Post-infectious irritable bowel syndrome. World J Gastroenterol. 2009;15:3591–3596. doi: 10.3748/wjg.15.3591. [DOI] [PMC free article] [PubMed] [Google Scholar]