Abstract
Essential oils (EOs) are complex mixtures of volatile compounds extracted from different parts of plants by different methods. There is a large diversity of these natural substances with varying properties that lead to their common use in several areas. The agrochemical, pharmaceutical, medical, food, and textile industry, as well as cosmetic and hygiene applications are some of the areas where EOs are widely included. To overcome the limitation of EOs being highly volatile and reactive, microencapsulation has become one of the preferred methods to retain and control these compounds. This review explores the techniques for extracting essential oils from aromatic plant matter. Microencapsulation strategies and the available technologies are also reviewed, along with an in-depth overview of the current research and application of microencapsulated EOs.
Keywords: essential oils, extraction techniques, microencapsulation, controlled release, microcapsules, pharmacology
1. Introduction
Essential oils (EOs) are liquid products present in plants and can be defined as complex natural mixtures of volatile secondary lipophilic metabolites that give plants and spices their essence and colour [1,2]. These compounds can be obtained by hydrodistillation, solvent extraction, and supercritical CO2 extraction, among other methods that can also be used for essential oil extraction [1]. These oils can be extracted from different parts of the plant, such as the flowers, leaves, stems, roots, fruits, and bark, and have different biological and pharmaceutical properties [3]. Due to their versatile nature, the oils can be utilised for several purposes, from contact toxicant and fumigant to attractive or repellent applications [4]. However, there are many factors that affect the chemical composition of essential oils, including genetic variation, type or variety of plants, plant nutrition, fertilizer applications, geographical location of the plant, climate, seasonal variations, stress during growth or maturation, as well as post-harvest drying and storage.
EOs that have antimicrobial properties are alternatives to the use of antibiotics and chemical additives [5]. As they have been used worldwide in many industries, their prices differ due to the supply of raw materials, issues related to harvesting, climate factors, and extraction yields. Some of these EOs also have antioxidant properties, with studies reporting that EOs from celery, citronella, cloves, oregano, parsley, tarragon, and thyme seeds were able to inhibit 50% of the 2,2-diphenylpicliryl-hydrazil (DPPH) radical elimination activity [6]. The application of EOs as antioxidants has been evaluated in different types of foods, and research is currently being conducted to optimise the process [7].
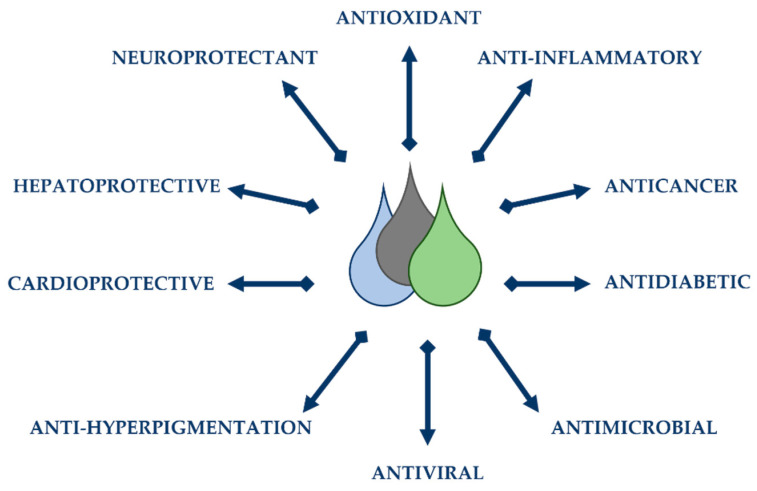
Essential oils have gained renewed interest in several areas over the years. Its use was expanded to the medical field due to its biocidal activities (bactericides, viricides, and fungicides) and medicinal properties [8]. The use of natural compounds has become popular in the food industry, with EOs being used as preservatives and food additives due to their antioxidant and antimicrobial properties and pleasant flavour. EOs are included in the composition of many dosage forms in pharmaceutical products. Studies have been carried out on the many biological activities of essential oils (Figure 1) and their components, and particular interests have also been established to elucidate their modes of action, allowing for the improved and targeted intervention in new drugs [9].
Figure 1.
Biological activities of essential oils.
EOs are unstable and highly susceptible to changes caused by external factors, such as light, temperature, oxygen, and humidity [10]. The high volatility and reactivity of these compounds represent challenges for the application of essential oils in several industries [11]. To overcome these limitations, the microencapsulation technique is often used to maintain the functional and biological characteristics of these compounds and to control their release [12].
Microencapsulation is a technology based on the coating of solid, liquid, or gaseous particles through an encapsulating agent that acts as a barrier, completely isolating the core material from the external environment [13]. Most microcapsules have a diameter within the range 1–1000 µm [14]. The shell material can be a film of a natural, semi-synthetic, or synthetic polymer and its choice has a key role in the stability of core material [15]. Arabic gum, agar, alginate, proteins, and dextrins are some of the materials used as encapsulating agents in the microencapsulation process [16].
Due to the enormous interest of the scientific community and the industry in the microencapsulation of active substances, several microencapsulation methods have been developed over time. Encapsulation processes are usually divided into three main categories: physicochemical, mechanical, and chemical processes [17]. In this review article, some of these methods, which are used in EO microencapsulation, are described and an outlook of scientific works developed in this area is approached.
2. Essential Oils
Essential oils are defined, according to the European Pharmacopoeia, as an “odorous product, usually of complex composition, obtained from a botanically defined plant raw material by steam distillation, dry distillation, or a suitable mechanical process without heating. EOs are usually separated from the aqueous phase by a physical process that does not significantly affect their composition” [18]. EOs are extracted from aromatic plant materials such as oily aromatic liquids, and they can be biosynthesised in different plant organs as secondary metabolites, such as flowers, herbs, buds, leaves, fruits, branches, bark, zest, seeds, wood, rhizomes, and roots.
Essential oils are complex mixtures of highly volatile aromatic compounds named after the plant from which they are derived. Within the different species of plants, only 10% contain EOs and are called aromatic plants. These natural products exert the function of protecting the plants, guaranteeing the growth of the plant and the propagation of species. Essential oil provides the essence, odour, or flavour of the plant and some of the functions that it performs in plants can also be made in living organisms [19]. EOs are generally liquid at room temperature and are hydrophobic (immiscible with water) and lipophilic (miscible with other oils and organic solvents) substances [20]. In general, essential oils are a mixture of compounds with their own physicochemical characteristics that, when combined, give the oil a particular odour. The different aroma of oils is fundamentally due to variations in the volatility and relative concentration of its constituents [21].
2.1. Chemistry of Essential Oils
The chemical composition of EOs can be complex due to the number of different components, which can have promising chemical and biological properties [22]. Essential oils are complex mixtures that can contain over 300 different compounds. Most EOs are characterised by two or three main components in reasonably high concentrations (20–70%) compared to other components present in small amounts [8]. The organic constituents have a low molecular weight, and their vapour pressure (at atmospheric pressure and at room temperature) is high enough for them to be partially in vapour state [23].
Chemically, EOs mainly belong to two classes of compounds: terpenes and phenylpropanoids (Table 1). The terpene family is predominant, and phenylpropanoids, when they appear, are responsible for the characteristic odour and taste [24].
Table 1.
Composition of compounds found in essential oils [29].
| Essential Oil Compounds | ||
|---|---|---|
| Classes | Constituents | |
| Terpenes | Monoterpene | (−)-Camphene, p-cymene, (+)-limonene, β-ocimene α-phellandrene, α-pinene, α-terpinene, terpinoleneorange, |
| Sesquiterpene | (−)-β-isabolene, α-cadinene, β-caryophyllene, α-copaene, β-elemene, α-farnesene, α-humulene, α-zingiberene |
|
| Phenylpropanoids | (E)-Anethole, cinnamaldehyde, cinnamic acid cinnamic alcohol, eugenol, methyleugenol, myristicin |
|
2.1.1. Terpenoids
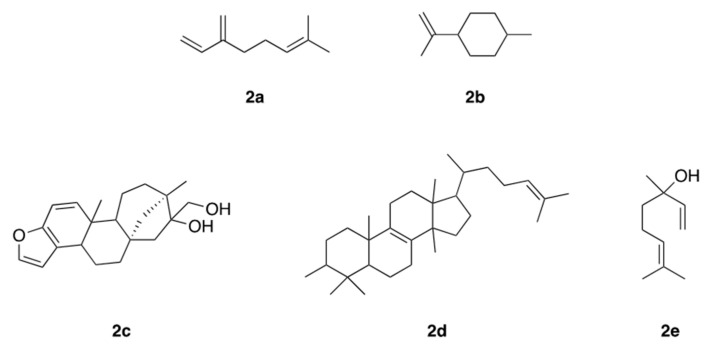
Terpenes, also called terpenoids, constitute the largest class of natural products with several structurally diversified known compounds [25]. Their structures contain carbon skeletons and are formed by isoprene units, being classified according to the number of these units that compose their structure. They can be classified as hemiterpenes (1 isoprene unit; 5 carbons), monoterpenes (2 isoprene units; 10 carbons), sesquiterpenes (3 isoprene units; 15 carbons), diterpenes (4 isoprene units; 20 carbons), triterpenes (6 isoprene units; 30 carbons), and tetraterpenes (8 isoprene units; 40 carbons), among others. Monoterpenes and sesquiterpenes are mostly found in volatile essential oils. Terpenes can present aromatic, aliphatic, and cyclic structures and can contain oxygen atoms, being called terpenoids (Figure 2) [26].
Figure 2.
Structures of terpenes and terpenoids: acyclic monoterpenes (2a), cyclic monoterpenes (2b), diterpenes (2c), triterpenes (2d), and terpenoids (2e).
2.1.2. Phenylpropanoids
Phenylpropanoids are natural substances commonly found in plants and consist of a six-carbon aromatic ring joined to a three-carbon side chain. This side chain contains a double bond and the aromatic ring may be substituted. These compounds are biosynthesised from shikimic acid, which forms the basic units of cinnamic and p-coumaric acids. These units, through enzymatic reductions, produce propenylbenzenes and/or allylbenzenes and, through oxidations with side chain degradation, generate aromatic aldehydes [27,28].
2.2. Extraction Methods
Aromatic herbs or parts thereof, such as leaves, flowers, bark, seeds, and fruits, are subjected to extraction processes after being collected at specific stages of maturity and stored under controlled conditions (light, temperature, and humidity).
Extraction techniques are essentially divided into classical and conventional methods and innovative methods. Classical methods are based on the distillation of water by heating, to extract the EOs from the plant matter. Hydrodistillation, steam distillation, hydrodiffusion, organic solvent extraction, and cold pressing are some of these methods. New extraction technologies have been developed in order to overcome some of the disadvantages of conventional methods. Methods such as ultrasound-assisted extraction and microwave-assisted extraction use energy sources that make the process more environmentally friendly. On the other hand, methods such as supercritical fluid extraction and subcritical liquid extraction allow the non-polar components from the material to be extracted [30].
2.2.1. Hydrodistillation
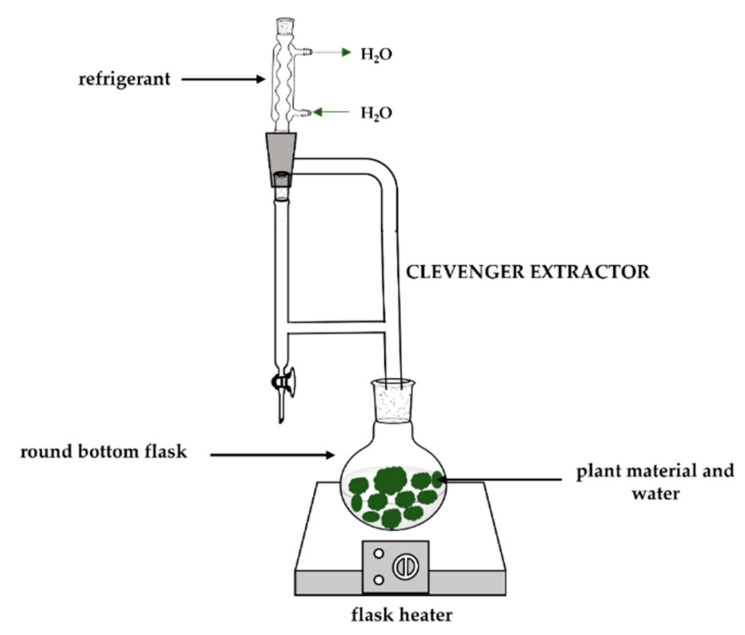
Hydrodistillation is the oldest and simplest method for extracting OEs. This method is characterised by direct contact between the solvent and the plant material, that is, the raw material is submerged in boiling water (Figure 3) [31].
Figure 3.
Schematic representation of hydrodistillation.
In this procedure, the cell walls are broken, and the oil is evaporated together with the water, and then condensed into a mixture of water vapour and volatile compounds of vegetable raw material. However, these two phases (volatile compounds and water) are immiscible, rendering possible an additional separation according to the difference in density [32]. This technique is inexpensive, but, at the same time, it is not selective because of the waste of large amounts of the compound in the solvent (part of the extract can be lost in the aqueous phase) and can provide low yields [33,34]. Despite being the oldest method, hydrodistillation is still used today for extracting oils from different matrices. Essential oils from Rosmarinus officinalis L. [35], Ziziphora clinopodioides L. [36], Citrus latifolia Tanaka [37], and Zingiber officinale [38] are some of the medicinal plants where EOs can be extracted by hydrodistillation.
2.2.2. Steam Distillation
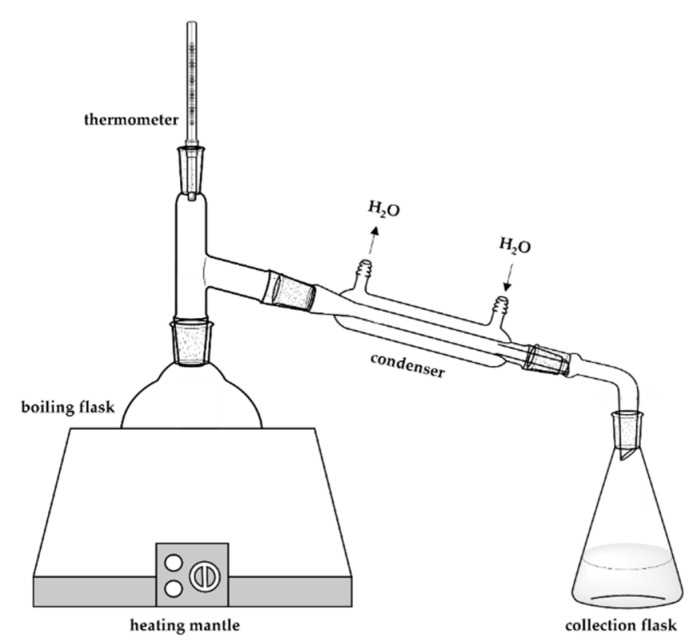
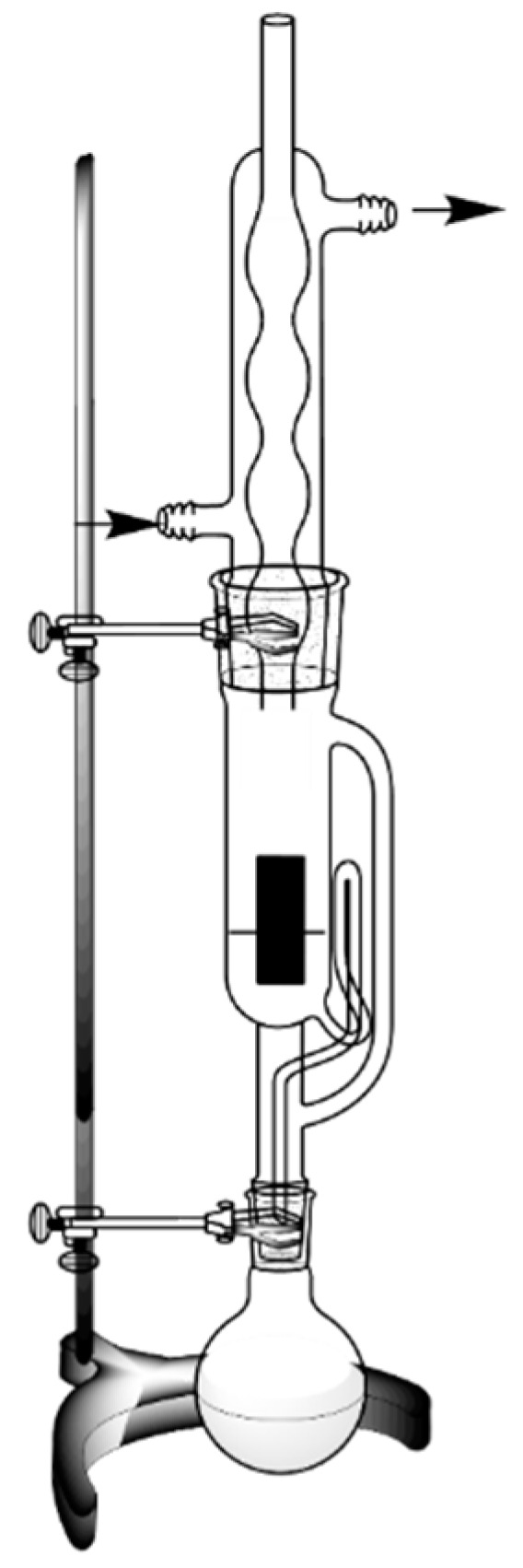
Steam distillation is one of the preferred methods of extracting EOs. The extraction procedure is based on the same principles as hydrodistillation. The difference essentially lies in the absence of contact between the substrate to be extracted and the water, which causes a reduction in the extraction time.
The sample is placed in a column where the bottom part is connected to a flask with water under heating (Figure 4). The top part is connected to a condenser, where the steam produced passes through the sample, taking essential oils to the condenser. This process causes the condensation of the water–oil mixture, and this mixture can be separated by liquid–liquid extraction [39].
Figure 4.
Experimental setup used in steam distillation.
This method is applied commercially and on a large scale in the extraction of essential oils from hops [40] and in the extraction of several EOs such as lavender [41] and patchouli essential oil [42].
2.2.3. Organic Solvent Extraction
Some essential oils (such as rose and jasmine) have low thermal stability and are unable to withstand high temperatures. In these cases, organic solvents that have a low boiling temperature, are chemically inert, and have low cost can be used.
In organic solvent extraction, the sample is placed in contact with the organic solvent (which can be hexane, benzene, toluene, or petroleum ether, among others) for a period that allows the transfer of the soluble content of the sample. The extracted matrix is concentrated by evaporating the solvent present in the liquid phase. This method allows the sample to be permanently in contact with a quantity of fresh solvent and, at the end of the process, it is not necessary to carry out filtration, as long as there are high yields [34]. Solvent extraction is the most-used conventional method in the cosmetic industry [43,44,45]. Figure 5 represents the extraction of organic solvents through a Soxhlet extraction [32,46].
Figure 5.
Schematic representation of organic solvent extraction using the Soxhlet method.
2.2.4. Cold Pressing
Essential oils are mechanically removed by cold pressing, where the oil glands are broken and volatile oils are released. In this process, an aqueous emulsion is formed, where the oil present can be obtained through centrifugation, decantation, or fractional distillation [13]. The cold pressing method is essentially used to extract oils from citrus fruits [47,48,49].
2.2.5. Supercritical Fluid Extraction (SCFE)
Supercritical fluid extraction is an efficient, environmentally friendly, and clean technique for isolating EOs. In this technique, supercritical fluids are used as extraction agents due to the supercritical state of fluids, conferring excellent characteristics for the extraction process, such as low viscosity, high density (close to that of a liquid), and high diffusivity (high penetration power).
Several substances can be used as supercritical solvents, such as water, carbon dioxide (CO2), methane, ethylene, and ethane. However, CO2 is the most-used solvent due to its critical point being easily reached (low temperature and pressure, 31.2 °C and 72.9 atm, respectively), low toxicity and reactivity, low cost, and non-flammability. After selecting the ideal temperature and pressure for extraction, supercritical fluid passes through the sample and the oils are dissolved and extracted. Subsequently, the extraction solution is maintained at a pressure below the critical point and as the pressure decreases, the supercritical fluid passes to the gaseous state and loses its solvating capacity, being recycled [50]. This method is increasingly used commercially, being applied in the extraction of EOs from the leaves of laurel [51], rosemary [52], sage [53], flowering plants [54], and horseradish tree [55].
2.2.6. Microwave-Assisted Extraction (MAE)
Due to the need to use more ecological and energy-efficient extraction methods, microwave-assisted extraction has become an alternative to conventional methods. The sample is placed in a microwave reactor without any solvent, where the electromagnetic energy that is converted into heat increases the internal temperature of sample cells due to the evaporation of the moisture present. The internal pressure increases, the glands rupture, and the essential oil is released [56]. Several EOs were extracted from plant matrices through this technique, such as orange [57], laurel [58], lemon [59], mint [60], rosemary [61], and basil [62].
2.2.7. Ultrasound-Assisted Extraction (UAE)
Ultrasound energy allows the intensification of EO extraction [63]. Therefore, it is usually combined with other extraction techniques in order to accelerate the extraction process and increase the speed of mass transfer. The sample is submerged in a solvent while being subjected to ultrasound. This method, through rapid solvent movements, induces a mechanical vibration of the walls and membranes of the sample that causes the release of essential oils. In some areas, it is already considered a large-scale application method, such as in the medical and food industry, where it is used to increase the quality of the extracted substrate, reduce working time, and increase the yield [64].
In general, alternative methods have emerged to overcome some of the disadvantages and limitations of conventional methods. Traditional methods have long extraction times (4 to 6 h), high energy consumption, and use solvents that increase environmental pollution. Furthermore, they can cause chemical changes to the EOs that are thermally unstable, causing a decrease in the quality of the extracted oils and changes in the chemical nature of compounds. The ‘greener’ alternatives are more sustainable and economical due to reduced water and energy consumption and reduced CO2 emissions. However, these methods are not easily accessible, and the initial investment is higher.
Therefore, currently, the hydrodistillation method continues to be the most-used extraction technique in laboratory due to its accessibility, simplicity, and lower cost [46]. Table 2 summarises the advantages and disadvantages of the different EO extraction methods.
Table 2.
Advantages and disadvantages of each essential oil extraction method [46].
| Type of Method | Method | Advantages | Disadvantages |
|---|---|---|---|
| Conventional | Hydrodistillation |
|
|
| Steam distillation |
|
|
|
| Organic solvent extraction |
|
|
|
| Cold pressing |
|
|
|
| Innovative | Supercritical fluid extraction |
|
|
| Microwave-assisted extraction |
|
|
|
| Ultrasound-assisted extraction |
|
|
2.3. Essential Oils Application
2.3.1. Essential Oils in Plants
EOs are stored in specific parts of plants, acting in extraordinarily different ways. Some aromatic plants have been widely explored due to their properties, such as bay laurel (Laurus nobilis). This plant is an aromatic tree, and laurel oil is extracted from the dry leaves and branches, appearing as a greenish yellow liquid with a powerful medicinal odour. In addition to being used in cuisine, the laurel tree leaves are used in medicine for having antioxidant [65], antibacterial [65,66], and antifungal [22] properties. According to the literature, laurel has also been proven to be an insect repellent [67,68]. However, it can cause dermatitis in some individuals, and due to the possible narcotic properties attributed to methyleugenol, this oil should be used in moderation.
Cymbopogon nardus, commonly known as citronella, is an aromatic and perennial herb. Citronella oil can also be produced from Java or Maha Pengiri citronella (C. winterianus) [69]. Citronella leaves are used for their aromatic and medicinal value in many cultures, such as in the treatment of fever, intestinal parasites, and digestive and menstrual problems, as well as for use as an insect stimulant and repellent [69,70,71,72]. Citronella is also used in traditional Chinese medicine for rheumatic pain, and it has antifungal [73], antioxidant, and antibacterial [74] properties. It is non-toxic and non-irritating, but it can cause dermatitis in some people [69].
Regarding the medical properties of hops (Humulus lupulus), these are better known for treatments associated with nerves, insomnia, nervous tension, neuralgia, and for sexual neurosis in both sexes [5]. It has antibacterial [75,76], antifungal [76], anti-cancer [76,77], and repellent [78,79] properties. In China, it is used for pulmonary tuberculosis and cystitis treatment. It can also be used to make beer. It is non-toxic and non-irritating, but it can cause sensitivity in some individuals, and people with depression should avoid this oil [76].
Lemon balm (Melissa officinalis) is a herbaceous perennial from the mint family and it has antibacterial, antifungal [80], sedative, antipyretic, antispasmodic, anti-hypertensive, anti-Alzheimer, and antiseptic properties [81]. In addition to the treatment of several gastrointestinal, liver, and nervous system disorders, it has also been reported that lemon balm is useful in the treatment of asthma, bronchitis, coughs, and several pains [82]. Furthermore, this plant is notably marked by its antimicrobial applications in different medicines, exemplified by its use in insect bites (wasps and bees) and poisonous or infectious bites [81,83].
Azadirachta indica, better known as neem, is an ancient tree that has been used for centuries for the most varied purposes. The plant provides a great number of secondary metabolites with biological activity. The plant has gained great importance in several areas, such as agriculture, livestock, and medicine [84]. It is used as an insecticide [85], antiviral [86], antibacterial [87], and antimicrobial [88], among others. Neem oil is very effective for acne, psoriasis, and eczema treatments, but it can also be applied as a support in the treatment of topical fungal or viral conditions, such as nail fungus, athlete’s foot, warts, or wounds. The natural antihistamines contained in neem oil are effective in relieving the itching and burning caused by, for example, bee, mosquito, and spider bites. The main constituent of neem is azadiractin, found in the leaves, fruits, and seeds.
Mentha pulegium, better known as pennyroyal or mint (Brazil), is one of the best-known species of the genus Mentha. Pennyroyal extracts are good insect repellents [89,90,91,92]. There are several studies that show that these extracts also have other properties, such as antimicrobial [92,93,94], antioxidant [92,93,95], antibacterial [95,96], and anti-tumour [96] uses. It is still current in the British Herbal Pharmacopoeia, indicated for flatulent dyspepsia, intestinal colic, common cold, delayed menstruation, skin rashes, and gout [69].
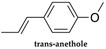
Illicium verum, popularly known as star anise, is a plant considered a spice for medicinal and culinary use. The extraction of Illicium verum has carminative, stomach, stimulating, and diuretic properties and is used as a pharmaceutical supplement [97]. The extracted shikimic acid is one of the main ingredients of the antiviral drug Tamiflu® (oseltamivir) that is used to treat symptoms caused by avian influenza [98]. It has also been reported to have antimicrobial properties [99] and antioxidant properties [100], as well as significant anti-cancer potential [101]. There are studies in which star anise has been used as an insect repellent, such as for the Indian flour moth (P. interpunctella larvae) [102,103]. The main constituent of star anise is trans-anethole (Table 3) (80–90%) and when used in large doses, it is narcotic and slows down circulation, which can lead to brain disorders [69].
Table 3.
Essential oil plants, species, and main components, and their molecular structure and biological properties.
| Plant | Species | EO Major Components | Chemical Structures of EOs Components | Some Biological Properties |
|---|---|---|---|---|
| Bay Laurel | Laurus nobilis | Cineol, pinene, linalool, terpineol acetate, methyleugenol |
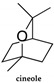
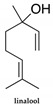
|
Antioxidant, antibacterial, antifungal, insect repellent |
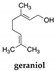
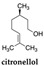
| Citronella | Cymbopogon nardus | geraniol, citronellal, geranyl acetate, limonene, camphene |
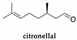
|
Antimicrobial, antifungal, antioxidant, antibacterial, insect and stimulate repellent |
| Hops | Humulus lupulus | Humulene, myrcene, caryophyllene, farnesene |
|
Antibacterial, antifungal, anti-cancer, repellent |
| Lemon Balm | Melissa officinalis | Geraniol, citral, citronellol, eugenol, linalyl acetate |
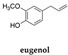
|
Antibacterial, antifungal, antimicrobial, sedative, antipyretic, antispasmodic, anti-hypertensive, anti-Alzheimer, antiseptic |
| Neem | Azadirachta indica | Azadiractin |
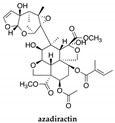
|
Insecticide, antiviral, antibacterial, antimicrobial |
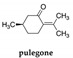
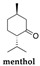
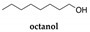
| Pennyroyal | Mentha Pulegium | Pulegone, menthol, iso-mentone, octanol, piperitenone, trans-iso-pulegone |
|
Insect repellent, antimicrobial, antioxidant, antibacterial, anti-tumour |
| Star Anise | Illicium verum | Trans-anethole |
|
Antimicrobial, antioxidant, diuretic, anti-cancer potential, insect repellent |
| Valerian | Valeriana officinalis | Borneol, camphene, α and β-pinene, valeranone, valerenol |

|
Antibacterial, antimicrobial, antifungal, antioxidative, sedative |
Valerian is a perennial flowering plant with many chemical constituents, with more than 150 constituents identified in its essential oil [104]. Regarding therapeutic indications, it is advisable for people with nervous agitation, mild anxiety, and difficulties in sleeping [105,106]. There are also studies that demonstrate its antibacterial [107] and antimicrobial [108] properties. It is non-toxic, non-irritating, and can cause sensitisation. Table 3 describes the main components of the EOs present in plants, as well as their chemical structures and some of the biological properties.
2.3.2. Pharmacological and Medical Applications
Essential oils have a wide range of biological properties, and there has been a growing interest in clinical applications (Table 4). Some of the properties include the chemo-preventive effects of cancer [109], antifungal [110], antiviral [111], antimicrobial, analgesic, anti-inflammatory [112], and antiparasitic activities [113].
Table 4.
Some pharmacological actions of essential oils [114].
| Condition | Essential Oil |
|---|---|
| Anxiety, agitation, stress, challenging behaviours | Angelica, labdanum, bergamot, sweet orange, palmarosa, lavender, basil, geranium, and valerian |
| End-of-life agitation | Lavender, sandalwood, and frankincense |
| Fatigue | Labdanum, grapefruit, coriander, citronella, black peppermint, gully gum, spearmint, geranium, Scots pine, clary, and ginger |
| Insomnia | Angelica, lemon, mandarin, sweet orange, lavender, lemon balm, myrtle, basil, sweet marjoram, and valerian |
| Mental exhaustion, burnout | Peppermint, basil, and everlasting |
| Memory loss | May Chang, peppermint, and rosemary |
| Pain management | Gully gum, lavender, German chamomile, sweet marjoram, rosemary, and ginger |
An extensive range of EOs have antibacterial activity against Gram-positive and Gram-negative bacteria, along with antifungal properties. These compounds have been studied and have shown very promising results in salmonella, staphylococci, and other oral pathogens, and can be an alternative to antibiotics providing they are properly studied for these effects [115,116]. EOs that have shown antibacterial potential are basil [117], manuka oil (more potent among eucalyptus oil, rosmarinus, lavandula, and tea tree) [118], melaleuca oil [119], and essential leaf oils P. undulatum and Hedychium gardnerianum.
With regard to antifungal activity, melaleuca oil showed positive results for all of its constituents, especially against dermatophytes and filamentous fungi [120]. In a reported study, germinated conidia of Aspergillus niger were more susceptible to non-germinated ones, with EOs of Melaleuca ericifolia, Melaleuca armillaris, Melaleuca leucadendron, and Melaleuca styphelioides exhibiting good activity against this fungus [120]. These same oils were evaluated for their antiviral activity in African green monkey kidney cells through the plaque reduction assay in the herpes simplex virus type 1 [121]. Other plants that have good antifungal activity are M. piperita, Brassica nigra, Angelica archangelica, Cymbopogon nardus, Skimmia laureola, Artemisia sieberi, and Cuminum cyminum [122,123,124,125,126,127]
Regarding antioxidant activity, the essential oil of the seeds of Nigella sativa L. showed considerable activity in the elimination of hydroxyl radicals. The essential oil of M. armillaris has marked antioxidant potential, changing the parameters of superoxide dismutase, and improving the concentrations of vitamin E and vitamin C [128]. However, there have been promising insect repellency/toxicity results from the essential oils of Nepeta parnassica, in Culex pipiens molestus [129]. Geranial, neral, geraniol, nerol, and trans-anethole are well established to stimulate the estrogenic response, and citrus (a combination of geraniol, nerol, and eugenol) is effective in replacing [3H] 17β-estradiol at the oestrogen receptors in recombinant yeast cells [130,131].
Cancer is a growing health problem worldwide and is the second leading cause of death. Essential oil constituents play an important role in cancer prevention and treatment as alternatives to synthetic drugs. Mechanisms such antioxidant, antimutagenic, and antiproliferative properties, enhancement of immune function and surveillance, enzyme induction and enhancing detoxification, modulation of multidrug resistance, and the synergistic mechanism of EO constituents are accountable for their chemo-preventive properties [132]. It has been reported that mitochondrial damage and apoptosis/necrosis in the yeast Saccharomyces cerevisiae were reduced by essential oils [133]. Recently, some studies demonstrated that certain EOs exhibited antimutagenicity towards mutations caused by UV light [8]. Jaganathan et al. reported that the active constituent eugenol from Syzygium aromaticum (cloves), nutmeg, basil, cinnamon, and bay leaves showed antiproliferative activity against several cancer cell lines in animal models [134].
In addition to medicinal and pharmacological applications, essential oils are used in perfumes, cosmetics, hygiene products, disinfectants, repellents, candles, phytochemicals, preservatives, and food additives.
2.3.3. Food Applications
In food, cosmetics, and personal care products, EOs are used as a natural aroma due to their chemical properties. In the food industry, EOs are being used as a food preservative because one of the main concerns is the preservation of food to prolong its useful life, ensuring safety and quality [11]. An expiration date is defined as the period of time during which the food product will remain safe. This ensures the maintenance of sensory, chemical, physical, microbiological, and functional characteristics. For example, spices can be encapsulated to extend their shelf life, maintain their properties, and inhibit reactions with other compounds [135]. Cinnamaldehyde, the aromatic agent present in cinnamon, has antimicrobial properties, and when encapsulated can slow the growth of yeasts in bakery products. Thus, the use of cinnamon in encapsulated form allows the product to be flavoured without interfering with the leavening process [136].
As the unpleasant taste and instability limit the application of EOs, the use of these encapsulated compounds can allow their application for several purposes. One of them is the intensification of the flavour of food products, where capsules can be used that release the product quickly when introduced into the mouth [20].
The packaging has the function of delaying deterioration, maintaining the quality and safety of packaged foods. For the packaging material to be satisfactory, it must be inert and scratch resistant, and must not allow molecular transfer to or from the packaging materials. Active packaging technologies extend the shelf life and control the quality of food products, decreasing microbial, biochemical, and enzymatic reactions through different strategies, such as adding chemical additives/preservatives, removing oxygen, controlling humidity and/or temperature, or a combination of these [137]. Oregano oil contains a high amount of carvacrol and is considered one of the most active plant extracts against pathogens due to its antimicrobial activity. Therefore, it has been used to preserve a variety of foods such as pizza, fresh beef [138], and cheddar cheese [139]. For the same purpose, limonene is reported for the preservation of strawberries [140], rosemary in chicken breast cuts [141], and cinnamon in pastries [142].
2.3.4. Cosmetic and Cleaning Applications
In the detergent and cosmetics industry, microcapsules of essential oils are used in many products such as perfumes, creams, and deodorants where the controlled release of EOs is essential, increasing the duration of fragrance and the properties of the EOs [45]. Aroma ingredients such as patchouli (Pogostemoncablin), citronella (Cymbopogon winterianus), sandalwood (Santalum álbum), bergamot (Citrusaurantium), rosemary (Rosmarinus officinalus), mint (Mentha piperita), and vetiver (Chrysopogon zizanioides) are frequently used [4]. Regarding the EOs from flowers, Lavandula officinalis, rose, jasmine, tuberose, narcissus, and gardenia are those most commonly exploited for cosmetic applications [143]. Products such as detergents, soaps, shampoos, and softeners are largely produced using these natural compounds.
Over the years, EOs have also been used against nosocomial infections, as a cleaning liquid for disinfecting equipment and medical surfaces [9], or as an aerosol in operating rooms and waiting rooms to limit contamination [10].
2.3.5. Agrochemical Applications
The loss of quality of agricultural products is caused by the presence of insect pests. The presence of these pests leads to reduced quality, low yield, and economic losses. Furthermore, human and animal health is compromised due to the production of carcinogenic secondary metabolites. To overcome this problem, chemical insecticides were used to excess. Despite being highly efficient, their overuse caused physiological resistance in several insect species and irreversible damage to the environment. Essential oils have emerged as a natural plant alternative to protect agricultural products from pests [144]. The use of EOs has intensified, mainly in gardens and homes, for pest control (Table 5), being important due to their toxic (pesticide) effect. EOs can be inhaled, ingested, or absorbed through the skin of insects. Monoterpenoids are an important group of chemical compounds in essential oils that interfere with the octopaminergic system of insects, which represent a target for insect control. As vertebrates do not have octopamine receptors, most chemicals in EOs are relatively safe to use. The special regulatory status together with the availability of essential oils has made the commercialisation of EO-based pesticides possible. Microencapsulation technology is used to produce these natural pesticides in order to mimic chemical compartmentalisation in plants, by protecting essential oils from degradation [145].
Table 5.
Pests/pesticides and their corresponding essential oil [145].
| Pests | Essential Oil |
|---|---|
| Ants | Peppermint, mint |
| Aphids | Cedar, hyssop, peppermint, mint |
| Beetles | Peppermint, thyme |
| Caterpillars | Peppermint, mint |
| Mites | Lavender, lemongrass, sage, thyme |
| Fleas | Peppermint, lemon grass, mint, lavender |
| Flies | Lavender, mint, rosemary, sage |
| Mosquitoes | Patchouli, mint |
| Lice | Cedar, peppermint, mint |
| Moths | Cedar, hyssop, lavender, peppermint, mint |
| Slugs | Cedar, hyssop, pine |
| Snails | Cedar, pine, patchouli |
| Spiders | Peppermint, mint |
| Ticks | Lavender, lemongrass, sage, thyme |
2.3.6. Textile Applications
Essential oils are used in medical and technical fabrics. The technique used in industrial processes is encapsulation, which is used to give finishes and properties to textiles that were not possible or economical. The main application for encapsulation is durable fragrances and skin softeners, and other applications may include insect repellents, dye, vitamins, microbial agents, and phase-change materials, and medical applications, such as antibiotics, hormones, and other medications.
The functionalisation of textiles with EOs with anti-mosquito repellent properties is a revolutionary way to protect human beings from insect bites and, thus, from many diseases such as malaria and dengue [146]. Plants, whose OEs have been reported to have repellent properties, include citronella, cedar, geranium, pine, cinnamon, basil, thyme, garlic, and mint. Khanna et al. performed the synthesis of a modified cyclodextrin host (β-CD CA) for inclusion complexation with the essential oils of cedarwood, clove, eucalyptus, peppermint, lavender, and jasmine for the assessment of repellent efficacy against Anopheles Stephensi in cotton. It was concluded that jasmine EO is the weakest against mosquitoes, as it worked as an attractant simulating flower nectar. Eucalyptus and clove are the feeding deterrents. On the other hand, lavender and peppermint are potential mosquito repellents, and cedarwood is an effective mosquito killer [147]. Soroh et al. reported that textiles treated with the Litsea and lemon EO microemulsion showed potential mosquito-repellent properties [148]. In general, citronella remains the most promising as an insect repellent and, therefore, is the most-incorporated EO in tissue functionalisation for this purpose. Specos et al. demonstrated citronella essential oil’s mosquito-repellent action, especially against Aedes aegypti [148]. Microcapsules with citronella are commonly incorporated into matrices such as cotton and polyester [149]. Another report determined that bio-based citronella oil has a better insect repellent effect than synthetic agents. Sariisik et al. concluded that, after washing, the insect repellent activity of the printing and coating method was increased, and the fabrics still showed repellency after five washing cycles [150].
3. Microencapsulation
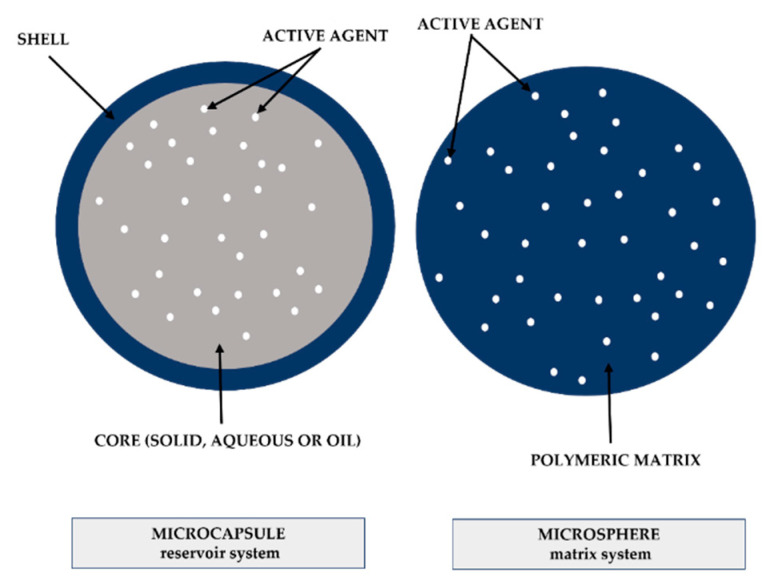
Microencapsulation is the protection of small solid, liquid, or gaseous particles through a coating system (1–1000 mm) [151]. The encapsulated material is called the core and the material that forms the coating of particle is the wall or encapsulating agent [152]. Wall material can be a natural, synthetic, or semi-synthetic polymeric coating. In this technology, microparticles are formed, which can be classified in relation to their size and morphology, according to the encapsulating agent and microencapsulation method used [153].
Microparticles can be distinguished according to their form: they are classified as a reservoir-type system, ‘microcapsules’, when the core (encapsulated material) is concentrated in the central region, coated by a continuous wall material (encapsulating agent); or a monolithic system, ‘microspheres’, when the active agent (core) is dispersed in a matrix system (Figure 6). In general, the main difference is that in microspheres, part of the encapsulated material is exposed on the surface of the microparticle [154].
Figure 6.
Schematic representation of a microcapsule and a microsphere.
The physicochemical characteristics of the microcapsule are defined by the encapsulating agent and the active agent. The wall material must form a cohesive film that bonds with the encapsulated material [155]. Several materials can be used for the coating, with proteins, carbohydrates, and lipids being frequently used. Furthermore, the materials must be chemically compatible and the encapsulating agent chemically inert, so as not to react with the core [156].
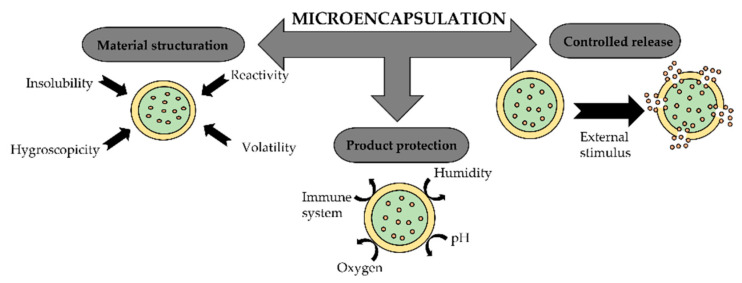
Microencapsulation technologies achieve several objectives (Figure 7) and they are particularly used to protect the core active agent’s sensitivity to oxygen, light, and moisture, or to prevent interaction with other compounds. However, the most important reason for encapsulating an active agent is to obtain a controlled release [157].
Figure 7.
Objectives of microencapsulation.
The process of defining a microencapsulation system is mainly dependent on the purpose of the microcapsules. Characteristics such as shape, size, permeability, biodegradability, or biocompatibility are defined depending on the application of this material. Other physical and mechanical properties of the microcapsule, such as strength and flexibility, must also be defined [158].
One of the great advantages of microencapsulation is the mechanism of the controlled, sustained, or targeted release of the active agent. This release can occur at a certain defined time or not, through a mechanism of diffusion through or rupture of the wall. The release can be activated through temperature variations, solubility, pH changes, or even the biodegradability of the wall material [159].
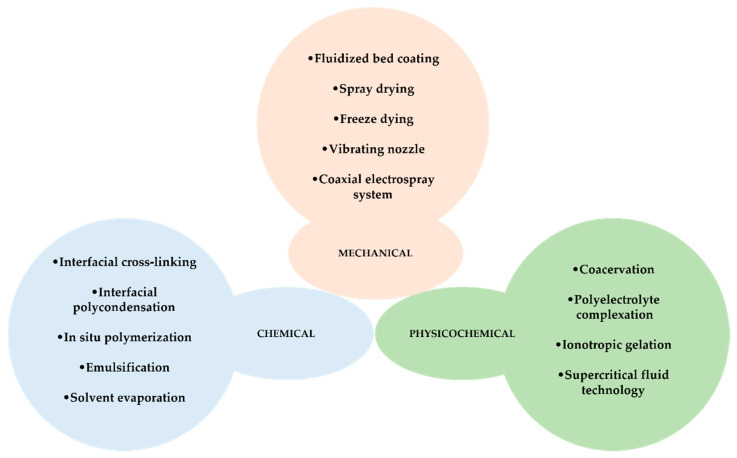
Depending on the nature of the interaction of the encapsulating and encapsulated material, microencapsulation methods can be distinguished as chemical, physicochemical, and mechanical (Figure 8) [160]. In general, a microencapsulation method must be fast, easy, reproducible, and easily scalable for industry. The most-used microencapsulation methods are spray drying and coacervation, and these approaches will be mentioned in more detail below.
Figure 8.
Main microencapsulation methods.
3.1. Emulsification
Emulsification is a fundamental step in oil microencapsulation, being used in a wide variety of food and pharmaceutical products. It is applied for the encapsulation of bioactive substances in aqueous solutions, which can be used directly in liquid or dried (spray-or freeze-drying) to form powders.
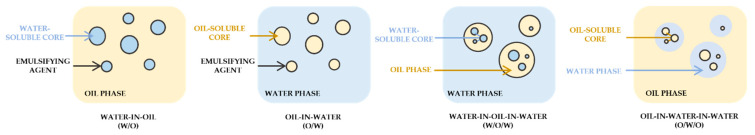
An emulsion consists of at least two immiscible liquids, with one of the liquids being dispersed as small spherical drops in the other. As can be seen in Figure 9, there are four systems, consisting of:
Oil-in-water emulsion (O/W);
Water-in-oil emulsion (W/O);
Oil-in-water-in-oil emulsion (O/W/O);
Water-in-oil-in-water emulsion (W/O/W).
Figure 9.
Illustration of emulsion systems.
In these systems, the droplet diameters can vary from 0.1 to 100 μm [161] and have been extensively revised by scientists [162]. The O/W emulsion consists of small oil droplets that are dispersed in an aqueous medium, being the droplets wrapped in a thin interfacial layer. Its advantages are the ease of preparation and low cost, with some disadvantages such as physical instability and limited control [162]. Through modifications of the emulsifiers, features can be added, such as the use of Maillard reaction products. These products can increase encapsulation efficiency and are able to protect the microencapsulation oil and other oils from oxidation [163].
A straightforward method for obtaining small droplets with a stratum size distribution is the evaporation/extraction of the emulsifying substance. This method is used in the preparation of biodegradable and non-biodegradable polymeric microparticles and in the microencapsulation of a wide variety of liquid and solid materials [164]. However, it is an expensive method with a low encapsulation efficiency, leading to residual solvent amounts [165].
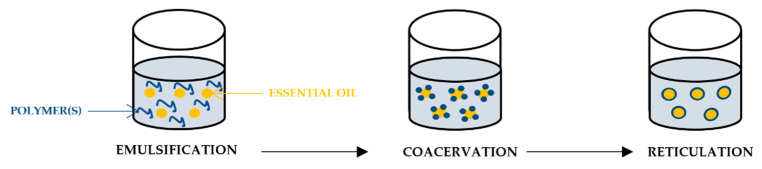
3.2. Coacervation
Coacervation is one of the most widely used microencapsulation techniques. The technique is based on oppositely charged polyelectrolyte polymers that interact and form a wall covering the active agent. The coacervation process can be classified as simple and complex if one or two (or more) polymers are used, respectively. Generally, this technique is defined by the separation of two liquid phases in a colloidal solution, where one phase is rich in polymer (coacervated phase) and the other phase does not contain polymer (equilibrium phase) [46].
Complex coacervation involves the interaction of two oppositely charged colloids, where the neutralisation of charges induces a phase separation. A polysaccharide and a protein are usually used as the different polymers. Wall material systems that are most widely investigated include gelatin/gum arabic, gelatin/alginate, gelatin/glutaraldehyde, gelatin/chitosan and gelatin/carboxymethyl cellulose [166].
In the process of the microencapsulation of hydrophobic materials (Figure 10), the emulsification of the encapsulated agent in an aqueous solution containing two different polymers occurs, usually at a temperature and pH above the gel and isoelectric point of the protein. Then, the separation into two liquid phases (polymer-rich phase and aqueous phase) follows, which results from the electrostatic interaction of the polymers. Subsequently, a microcapsule wall is formed as the deposition of the polymer-rich phase occurs around the hydrophobic particles of the active agent, due to controlled cooling below the gelation temperature. Finally, the microcapsule walls harden through the addition of a crosslinking agent [167].
Figure 10.
Schematic illustration of the coacervation method.
Simple coacervation has advantages over complex coacervation in terms of the associated cost, as cheap inorganic salts are used to induce the separation phase, while expensive hydrocolloids are applied in the complex method. Furthermore, complex coacervation is more sensitive to small variations in pH. However, compared to other microencapsulation methods, complex coacervation is a simple, scalable, inexpensive, reproducible, and solvent-free method, enabling its industrial use [166].
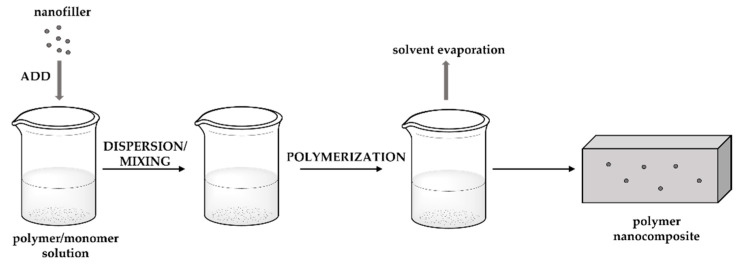
3.3. In Situ Polymerisation
In situ polymerisation (Figure 11) is based on the formation of a wall through the addition of a reagent inside or outside the core material [168], becoming one of the most-used methods in the preparation of microcapsules and functional fibres. Polymerisation takes place in the continuous phase and not on both sides of the interface between the core material and the continuous phase. Microcapsule formation occurs through an oil emulsion in a solution of melamine–formaldehyde resin and a sonication process to emulsify the oil in the aqueous phase. Then, resin is added under agitation and the pH is adjusted, with the formation of shells, thus promoting the reaction of the melamine with the formaldehyde at the interface of the oil droplets. This type of microcapsule is used in fragrances, insect repellents, food packaging, and footwear. The microcapsules result in smooth surface morphologies and are able to preserve the encapsulated scented oils for a sufficient period of time. They also have good thermal and controlled release properties [168,169].
Figure 11.
Schematic illustration in situ polymerisation method (adapted from [168]).
Using a polymer as a microcapsule wrapper is considered a good addition due to its high strength and stability [170]. On the other hand, using a copolymer to prepare microcapsules with a low molecular weight of formaldehyde–melamine avoids the toxicity of formaldehyde [171].
In situ polymerisation is a method of rapid and easy expansion [172] and, at the same time, provides high encapsulation efficiency. However, the polymerisation reaction is difficult to control [173] and requires a large amount of solvent, making the monomers non-biodegradable and/or non-biocompatible [174].
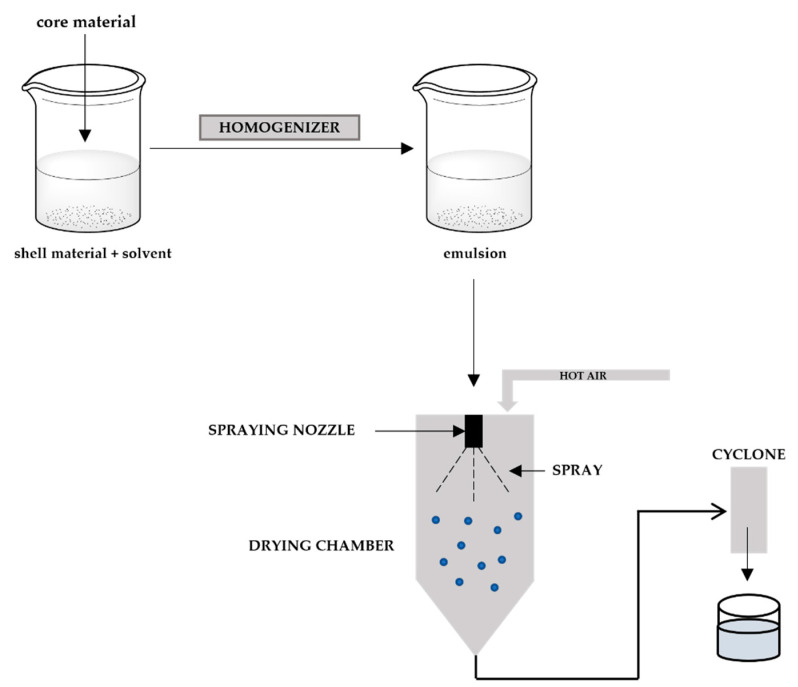
3.4. Spray Drying
Spray drying is the most-used technology in the microencapsulation of essential oils. It is mainly used on an industrial scale, as it allows simple, reproducible, continuous, and low-cost production. Being used more frequently in the food industry, this process is also utilised in the cosmetics, pesticides, and pharmaceutical industries [175]. This technique allows encapsulated and powdered Eos to be obtained due to the ability to dry them in just one operation. In this process, the atomisation of emulsions occurs in a drying chamber with relatively high temperatures, where the evaporation of the solvent takes place and, consequently, microcapsules are formed [176].
The spray-drying technique involves four steps (Figure 12), where the preparation of dispersion first occurs, i.e., the wall materials are dissolved in water with agitation and controlled temperature. Still in the same step, the addition of the EOs follows and, if necessary, the emulsifier can be added. Afterwards, the dispersion is homogenised to be injected into the equipment through an atomising nozzle, where small droplets are formed. In the third step, emulsion atomisation occurs, where the formed droplets enter the drying chamber with a flow of hot air present. Finally, the dehydration of the atomised microparticles is done through the evaporation of the solvent, which dries the microparticles, which can them be recovered in the form of powder in a collector or filter [177].
Figure 12.
Schematic representation of spray drying.
The main limitations of this technique are related to the wall material, which must have good water solubility, and to the number of encapsulating agents available. In addition, some materials may be sensitive to the high temperatures introduced in the atomisation process. In addition, the production of microcapsules in fine powder form can cause agglomeration and an additional process may be required [166].
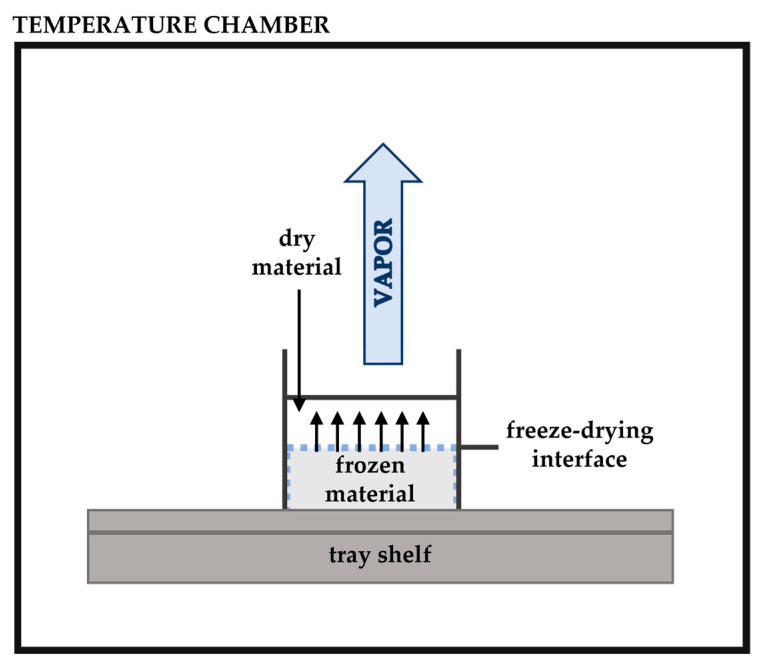
3.5. Freeze Drying
Freeze drying, also known as lyophilisation, is a simple process (Figure 13) that is used to dehydrate most materials sensitive to heat and aromas such as oils. Sublimation is the major principle involved in this drying process, where water passes directly from a solid state to a vapour state without passing through the liquid state. Before starting this process, the oil is dissolved in water and frozen [178]. Afterwards, the pressure is reduced and heat is added to allow the frozen water to sublimate the material directly from the solid phase to the gas phase. Freeze-dried materials appear to have the maximum retention of volatile compounds compared to spray drying, and this technique is used to microencapsulate some oils, with high yields [179]. This method helps to better preserve the EO content in many types of herbs and spices compared with other preservation techniques [180]. Lyophilisation is simple and easy to operate, showing that lyophilised samples are more resistant to oxidation and less efficient in microencapsulation [181]. The process disadvantages include high energy use, long processing time, and high production costs [182].
Figure 13.
Schematic diagram of a freeze dryer (adapted from [163]).
3.6. Supercritical Fluid (SCF) Technology
Many pharmaceutical, cosmetic, and food industries use supercritical fluid technology (Figure 14) to form the microcapsules of essential oils due to their inherent advantages. The use of a wide variety of materials that produce controlled particle sizes and morphologies, the easy solvent removal, the non-degradation of the product, and being a non-toxic method are some of the many advantages of SCF technology.
Figure 14.
Flow chart of supercritical fluid technology.
The methods used for supercritical fluids are the precipitation of gas anti-solvent, particles of saturated gas solutions, the extraction of fluid emulsions, and the rapid expansion of supercritical solutions [183,184]. The supercritical solvent impregnation process has proven to be successful in a wide variety of substances (essential oils, fragrances, active pharmaceutical compounds, and dyes) and matrices (wood, polymers, cotton, and contact lenses).
An alternative to spray drying (that degrades oils at high temperatures) is impregnation with supercritical solvent, as it is an ecological process where supercritical carbon dioxide is used as a green solvent.
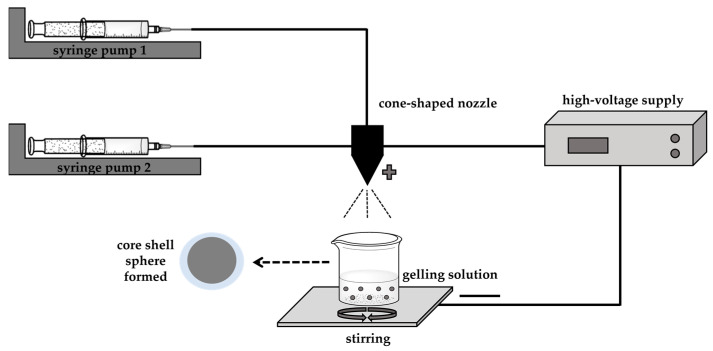
3.7. Coaxial Electrospray System
The food, cosmetic, and pharmaceutical industries use a new technology to encapsulate oils, called coaxial electrospraying (Figure 15) [185,186]. This system is used in two phases, with external and internal solutions being sprayed coaxially and simultaneously through two feed channels separated by a nozzle.
Figure 15.
Schematic representation of microencapsulation process by coaxial electrospraying (adapted from [144]).
In the electrospray process, the Taylor cone is composed of a core-shell structure that is formed at the top of the spray nozzle, ending up with the polymeric solution encapsulating the internal liquid. This method is distinguished by its ease and efficiency, and the maximum speed of the core material. The coaxial electrospray system provides a uniform size distribution, a high encapsulation efficiency, and an effective protection of bioactivity. However, the encapsulation efficiency and the stability of the microcapsules are affected by the wall materials [186]. Furthermore, controlling the process in coaxial electrospraying is difficult to some extent [187].
In experimental terms, the reported work on coaxial electrospray is based on individual laboratory experiments, consisting of specific combinations of materials and empirical process parameters. The fabrication of polymeric microparticles and nanoparticles is hampered by the lack of standard protocols. Regarding the collection of particles, the methodology cannot facilitate the hardening of the shell or maintain the morphology of particle, or even prevent its aggregation. On the theoretical side, many existing process models are empirical or semi-quantitatively empirical. The simulated results are not enough for the quantitative control of the process, as numerical simulations, such as computational fluid dynamics modelling, have been used to simulate the formation of the liquid cone and atomisation in a single axial electrospray process [188]. In summary, more experimental and theoretical study is needed to better understand the physical nature of coaxial electrospray and to provide quantitative guidance for process control.
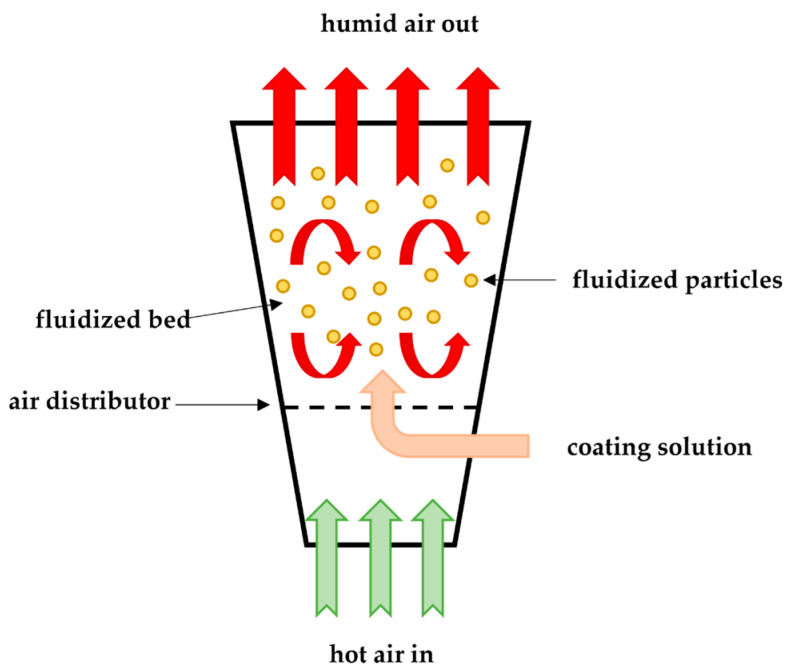
3.8. Fluidized Bed Coating
Fluidised bed coating is one of the most efficient coating methods, in which the ingredients can be mixed, granulated, and dried in the same container. Consequently, the handling and processing time of the material is reduced. This approach was recently used to encapsulate fish oil by spraying and coating it (Figure 16) [189]. Fluidised bed coating is carried out by suspending the solid particles of the core material by an air stream under controlled temperature and humidity and then sprayed, building, over time, a thin layer on the surface of the suspended particles. This material must have an acceptable viscosity for atomisation, and the pumping should be able to form an appropriate film and be thermally stable [190].
Figure 16.
Schematic representation of bottom spray fluidised bed coating process (adapted from [166]).
There are several methods used in fluidised bed coating, including top spray, bottom spray, and tangential spray methods. In the top spray system, the coating solution is sprayed in the opposite direction with air in the fluid bed. The opposite flows lead to an increase in the efficiency of encapsulation and the prevention of agglomerates formation, achieving microcapsules with a size between 2 and 100 μm. The bottom spray, known as the Wurster system, uses a coating chamber that has a cylindrical steel nozzle (used to spray the coating material) and a cribriform bottom plate, coating small particles (100 μm). This multilayer coating procedure helps to reduce particle defects, although it is a time-consuming process. On the other hand, tangential spray consists of a coating chamber with a rotating bottom of the same diameter as the chamber. During the process, the drum is raised to create a space between the edge of the chamber and the drum. A tangential nozzle is placed above the rotating drum, where the coating material is released. Then, the particles move through the space into the spray zone and are finally encapsulated [191]. During this process, there are three mechanical forces, namely, centrifugal force, lifting force, and gravity.
The particles to be coated must be spherical and dense, and must have a narrow size distribution and perfect fluidity, with the non-spherical particles having the largest possible surface area and requiring more coating material.
This technique has a low operating cost and a high thermal efficiency process, allowing total temperature control. However, it can be time consuming, which becomes a disadvantage [173].
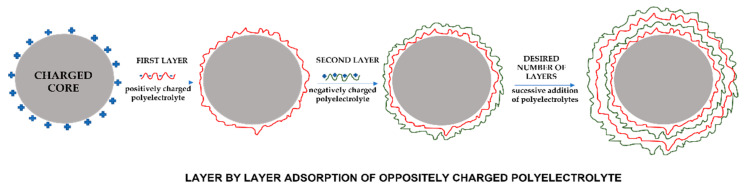
3.9. Layer-by-Layer Self-Assembly
Layer-by-layer (LbL) is a relatively simple and promising technique for the encapsulation, stabilisation, storage, and release of several active compounds [192]. This method consists of alternating the adsorption of oppositely charged wall materials through many intermolecular interactions onto a charged substrate (Figure 17). The microcapsules have good chemical and mechanical stability through a formation mechanism constituted by irreversible electrostatic interactions that allow the adsorption of successive layers of polyelectrolytes [193]. The adsorption of the layers is normally carried out by immersing the suspension in alternate solutions of cationic and anionic polymers, with washing processes being carried out after the deposition of each layer [194]. This technique has significant advantages over other microencapsulation methods, because it allows the control of the permeability, morphology, composition, size, and wall thickness of the microcapsules by adjusting the number of layers and experimental conditions [195]. Controlling these parameters allows a better adaptation of the microcapsule to its functionality in the target application. However, most LbL systems have some restrictions in terms of biocompatibility [196].
Figure 17.
Layer-by-layer (LbL) self-assembly microcapsules (adapted from [197]).
4. Microencapsulation of Essential Oils
Microencapsulation is an alternative that can be utilised to overcome several limitations in the application of essential oils. This application is profoundly affected by the high volatility and chemically unstable nature of EOs [198]. In addition, EOs are compounds that can be easily degraded due to interactions with other chemical components and exposure to several factors such as light, temperature, and oxygen [166].
Essential oils can be “trapped” in microcapsules, which act as micro-reservoirs, ensuring excellent protection [199]. The encapsulation process, where small particles are enclosed in solid carriers to increase their protection, has the ability to reduce evaporation, promote easier handling, and control the release of essential oils during storage and application [199]. Furthermore, through microencapsulation, it is possible to change the appearance of EOs (which behave like a powder), without changing their structure and properties [177].
In EO microencapsulation, the first step is often to emulsify or disperse the essential oils in an aqueous solution of a wall material, which also acts as an emulsifier. This process happens because the EOs exist in liquid form at room temperature. Then, the resulting microcapsules must be dried under controlled conditions, so that the loss of the encapsulated material by volatilisation is reduced [177]. One of the areas that has also aroused interest in the microencapsulation of EOs is in the agrochemical industry. Yang et al. prepared and characterised microcapsules based on polyurea, containing essential oils as an active agent for possible applications in the controlled release of agrochemical compounds [200]. The microcapsules were synthesised by O/W emulsion interfacial polymerisation and the synthetic conditions that showed the best results were used to encapsulate four essential oils (lemongrass, lavender, sage, and thyme), capable of interfering with the seed germination and root elongation of some plants. In cases of pest control, biological pesticides must be more effective than synthetic pesticides.
Bagle et al. reported success in encapsulating neem oil, an effective biological insecticide, in phenol formaldehyde (PF) microcapsules [201]. The microcapsules were obtained using an in situ polymerisation process in an O/W emulsion and their size was determined using a particle size analyser. Controlled release was monitored by measuring optical observations in the UV range. Figure 18 shows scanning electron microscopy (SEM) micrographs of PF microcapsules containing neem oil. It was possible to visualise that the PF microcapsules were spherical and globular, with diameters between 30 and 50 µm at 400–500 rpm. The microcapsules’ surface was considered quite smooth and can be useful regarding the protection and sustained release of the neem oil inside.
Figure 18.
SEM micrographs (a–c) of phenol formaldehyde microcapsules containing neem oil [201].
The chemical constitution of synthesised microcapsules was confirmed by Fourier-transform infrared spectroscopy (FTIR), and it was found to be a good thermal stability of MCs needed for the long-term preservation of the core, and it was concluded that neem oil can be better preserved in PF microcapsules.
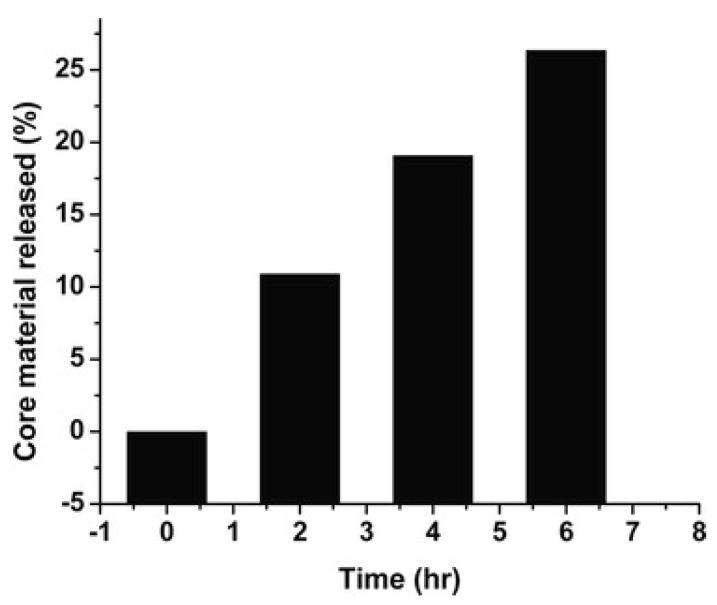
The controlled release behaviour of PF microcapsules containing neem oil was studied and the experimental data are shown in Figure 19. A release of about 30% was observed after 6 h, confirmed by the decrease in absorbance over time.
Figure 19.
Controlled release of core material over time [201].
Like neem oil, other essential oils also have insecticidal properties, such as Rosmarinus officinalis and Zataria multiflora (Lamiaceae), that can be used as pesticides for stored-product pests. In the study carried out by Ahsaei et al., these oils were encapsulated in octenyl succinic anhydride (OSA) starch to test their insecticidal activity against Tribolium confusum. The microcapsules were obtained using an O/W emulsion and dried using the spray drying technique [202]. The solid formulations were characterised by particle size, encapsulation efficiency, and water activity. The release rate under storage conditions was measured over a period of 40 days, and the insecticidal activity against T. confusum was determined using specific bioassays. It was concluded that the encapsulation efficiency depends directly on the surfactant-to-oil ratio. Regarding the morphology of microcapsules loaded with OEs, SEM micrographs reveal the presence of oval and spherical microcapsules with irregular surfaces. The microcapsules appear to be devoid of cracks or fractures, which is an advantageous feature for protecting the oil. The results also showed an optimised release of pesticides from controlled release formulations, which maximises their biological activity for a longer time.
The food sector is probably the sector where the microencapsulation of essential oils is most explored, with the encapsulation of flavours being one of the great interests of this industry. Flavours are necessary for some foods, to promote consumer satisfaction and the consumption of those products. Nevertheless, the flavour stability in foods has been a challenge for this sector in order to achieve quality and acceptability.
For the encapsulation of a flavour, Fernandes et al. evaluated, by spray drying, the effects of the partial or total substitution of arabic gum with modified starch, maltodextrin, and inulin in the encapsulation of rosemary essential oil [203].
Regarding the characterisation of microcapsules, moisture content, wettability and solubility, density and apparent density, and oil retention was determined. From SEM observations (Figure 20), the authors found that there was no evidence of cracking in the particles using any of the encapsulating formulations, ensuring low gas permeability and thus better protecting the EO of rosemary. Differences were observed in the surface of each type of particle, showing that the particles have a spherical shape. It was concluded that the total substitution of arabic gum with modified starch or a mixture of modified starch and maltodextrin did not affect the efficiency of the encapsulation, increasing the possibility of developing new formulations of encapsulants. With the addition of inulin, the oil retention of particles decreased. However, the combination of modified starch and inulin was shown to be a viable substitute for arabic gum in foods.
Figure 20.
Scanning electron micrographs of the particles containing rosemary essential oil [203]. (A): arabic gum; (B): arabic gum/maltodextrin; (C): arabic gum/inulin; (D): starch; (E): modified starch/maltodextrin; (F): modified starch/inulin.
A group of researchers compared the release properties of three different microcapsules, namely gelatin microcapsules loaded with holy basil essential oil (HBEO) (designated as UC), UC coated with aluminium carboxymethylcellulose (CC), and UC coated with aluminium compound carboxymethyl cellulose–beeswax (CB) [204]. To be applied as a feed additive, the HBEO was encapsulated in order to be a potential alternative to antibiotic growth promoters (AGP). However, its benefits depend on the available amount in the gastrointestinal tract.
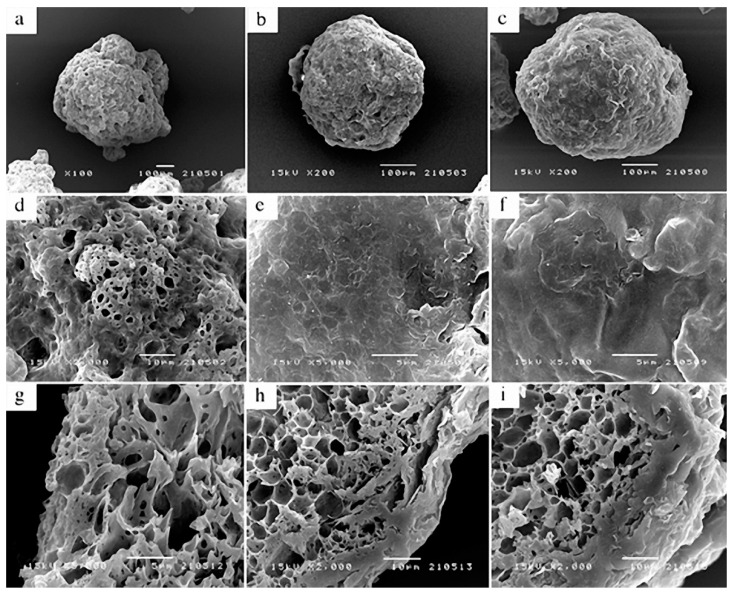
The SEM technique was used to characterise the internal and external factors of the microcapsule surface morphology. According to Figure 21, UC microcapsules (Figure 21a) are almost spherical in shape and after coating, the CC (Figure 21b) and CB (Figure 21c) microcapsules are more spherical. Upon magnification of these micrographs, it was possible to verify that UC microcapsules have a spongy structure (Figure 21d) and that CC (Figure 21e) and CB (Figure 21) microcapsules are denser. When cut transversely, UC microcapsules seem to have a gelatinous morphology (Figure 21g), whereas the CC (Figure 21h) and CB microcapsules (Figure 21i) reveal a thicker and more compact outer coating layer with a honeycomb structure. This method of encapsulation demonstrated an effective process for improving HBEO efficacy for pathogen reduction in the distal region of the intestine.
Figure 21.
SEM micrographs of UC, CC, and CB gelatin-based microcapsules: (a) whole UC; (b) whole CC; (c) whole CB; (d) external surface of UC; (e) external surface of CC; (f) external surface of CB; (g) inner edge of UC; (h) inner edge of CC; (i) inner edge of CB [204].
Regarding food safety, the use of antimicrobial packaging materials offers the potential to retard the growth rate of spoilage microorganisms. The physical and antimicrobial properties of nanofibres manufactured for active packaging systems were studied by Munhuweyi et al. [205]. Microcapsules and active nanofibres derived from the precipitation of β-cyclodextrin (β-CD) with essential oils of cinnamon and oregano were developed and their antifungal activity in vitro against Botrytis sp. was examined. To induce microencapsulation, the solutions were subjected to co-precipitation. It was verified that cinnamon microcapsules have greater antimicrobial efficacy when compared to oregano. As food preservatives, this microencapsulation system could have promising applications in the development of active packaging systems.
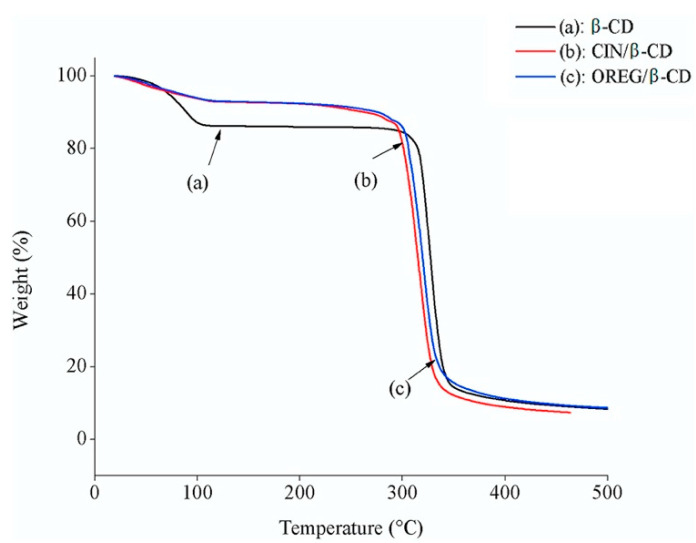
Using the thermogravimetric analysis (TGA) technique, the initial weight loss for simple β-CD occurred at ~100 °C and the greatest weight loss at ~330 °C (Figure 22a). The degradation temperature of β-CD in the CIN/β-CD and OREG/β-CD complexes decreased from ~330 °C to ~270 °C (Figure 22b,c). Comparing the TGA curves, there is a difference between them, demonstrating the presence of chemical and guest molecule interaction in the complex.
Figure 22.
TGA curves of (a) plain β-cyclodextrin (β-CD), (b) microencapsulated cinnamon (CIN/β-CD), and (c) oregano (OREG/β-CD) [205].
Using the simple coacervation method, Leimann et al. encapsulated lemongrass, which is known for its broad spectrum antimicrobial activity [206]. Poly(vinyl alcohol) crosslinked with glutaraldehyde was used as the wall-forming polymer. The influence of the agitation rate and the fraction of oil volume on the microcapsule size distribution was evaluated. Sodium dodecyl sulphate (SDS) and poly(vinyl pyrrolidone) were tested to prevent the agglomeration of microcapsules during the process. The microcapsules did not show agglomeration when 0.03% by weight of SDS was used. The composition and antimicrobial properties of the encapsulated oil were determined, demonstrating that the microencapsulation process did not deteriorate the encapsulated essential oil.
Cyclodextrins (CDs) are important supramolecular microcapsule hosts in foods and other fields, and the essential oil of Laurus nobilis (LEO) has natural antioxidant properties in food due to its main constituents being terpenic alcohols and phenols. For these reasons, Li et al. isolated LEO by microwave-assisted hydrodistillation [207]. The authors prepared chitosan (CS) microcapsules loaded with citrus essential oils (CEOs: D-limonene, linalool, a-terpinene, myrcene, and a-pinene) using six different emulsifiers (Tween 20, Tween 40, Tween 60, Tween 60/Tween 20/Span 80 1:1, Tween 20/sodium dodecyl benzene sulfonate (SDBS) 1:1, Span 80) through an emulsion gelation technique [208]. After preparing β-cyclodextrin (β-CD) microcapsules and their derivatives, several affecting factors were examined in detail.
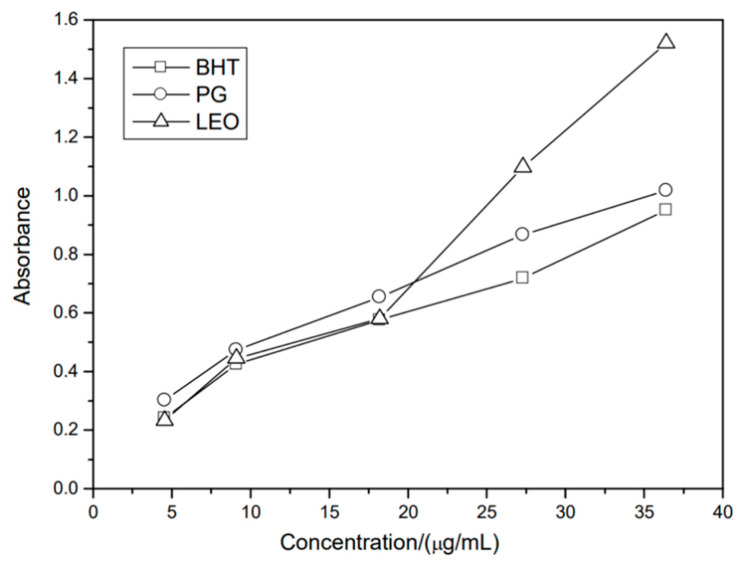
Figure 23 shows the total antioxidant activity of LEO. LEO caused Mo (VI) to be deoxidised to become Mo (V) through a mechanism of total antioxidant activity. Mo (V) exhibits maximum absorption at 695 nm and has a stronger antioxidant activity; the greater the concentration of Mo (V) solution, the greater the absorbency of the solution. With the increase in absorbance of the solutions, there was an increase in the concentrations of the sample, causing the antioxidant activity to increase significantly.
Figure 23.
Absorbance of antioxidants in different concentrations of 2,6-ditert-butylphenol (BHT, square), propyl gallate (PG, circle) and Laurus nobilis essential oil (LEO, triangle) [207].
The microcapsules were analysed and the results indicate that the choice of emulsifier significantly affects the size and effectiveness of incorporating the microcapsules.
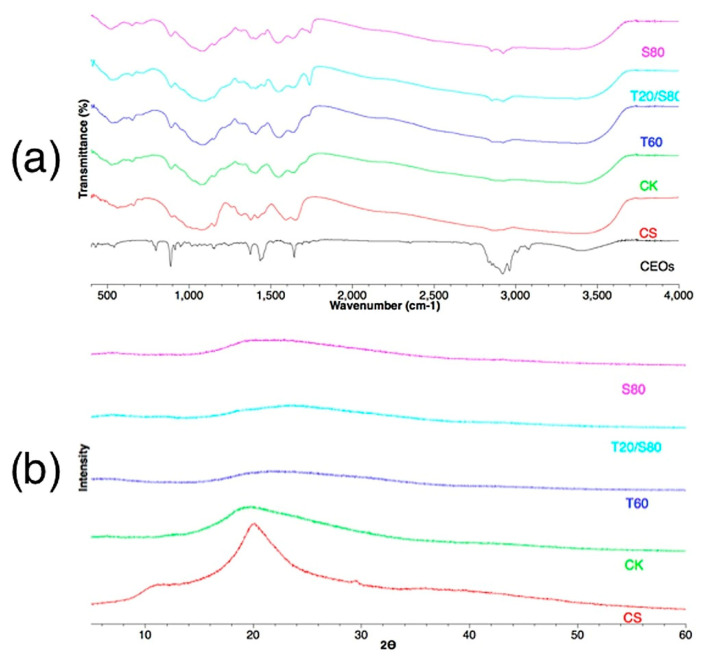
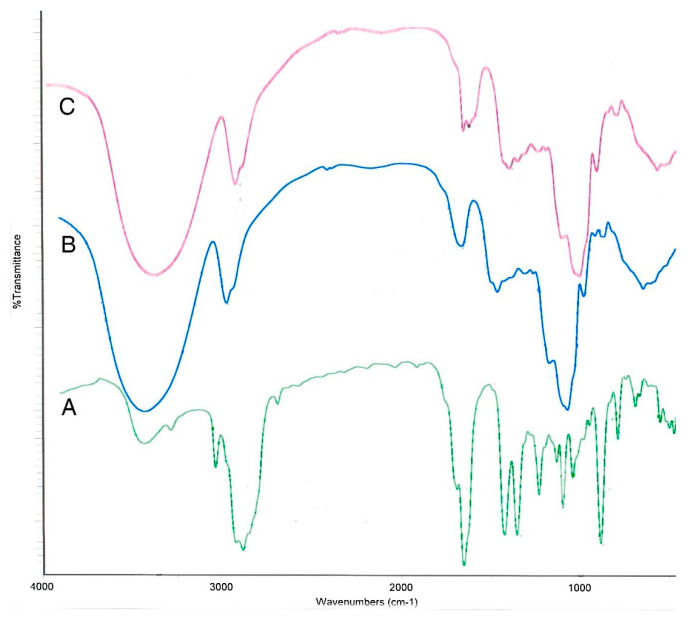
Figure 24a presents the FTIR spectra observed in CS, CEOs, and four groups of microcapsules prepared with different emulsifiers. In the CEOs curve, the peak at 886 cm−1 corresponds to the absorption of limonene. The strong methylene/methyl band occurs at 1435 cm−1, and at 1646 cm−1, the C=O stretching vibration appears. Peaks corresponding to the asymmetric and symmetrical modes of the CH2 elongation vibration appear for Span 80 and Tween 60, and the new connections can be seen at 2922 cm−1. Through these results, it was possible to observe that the CEOs were incorporated in the microcapsules, showing benefits for inhibiting them from oxidation and volatilisation.
Figure 24.
(a) FTIR spectroscopy, (b) X-ray diffraction of pure chitosan (CS), control group (CK), Tween 60, Tween 20/Span 80, and Span 80 [208].
A second step in the characterisation of the microcapsules was the analysis of the crystallographic structure. Through X-ray diffraction (XRD) analysis (Figure 24b), it was possible to observe that CS exhibits a diffraction pattern with a broad band centred at 2θ 20°, thus indicating the existence of an amorphous structure. Comparing the CS with the microcapsule groups, the latter exhibit a significant reduction in this broad band. This reduction in intensity is due to the destruction of the CS structure, which can be attributed to a change in the arrangement of the molecules in the crystalline chain.
To develop a new use of functional EOs, Karimi Sani et al. studied the influence of process parameters on the characteristics of microencapsulated essential oil Melissa officinalis using whey protein isolate (WPI) and sodium caseinate (NaCS). The impacts of these variables were examined using the response surface methodology. Smaller particle sizes were obtained for higher amounts of WPI with the lowest level of applied sonication power. The results of the desirability function indicate that the maximum amount of WPI with an ultrasound power of 50 W led to the smallest particle size and the lowest zeta potential and turbidity. In this study, the ultrasonic technique showed potential in the use of milk proteins to produce microparticles with OEs. The obtained results showed that the microcapsules loaded with Melissa officinalis can preserve the bioactive compounds and induce flavour stability, enabling their use in food formulations and pharmaceutical products.
Mehran et al. carried out a study of the microencapsulation of spearmint essential oil (SEO), using a mixture of inulin and arabic gum as wall material in order to be used in the food and pharmaceutical industry [209]. The technique used for the formation of the microcapsules was spray drying. The microcapsules were characterised for oil retention, encapsulation efficiency, hygroscopicity, and carbon content, having as ideal conditions 35% solid wall, 4% essential oil concentration, and 110 °C inlet temperature, with maximum retention of 91% of oil. To confirm that the SEO was encapsulated, this group of researchers used differential scanning calorimetry (DSC) and FTIR characterisation techniques.
The infrared spectra of pure SEO, pure matrix (containing inulin and arabic gum), and microcapsules are shown in Figure 25. In the SEO spectrum, the characteristic peaks at 801 cm−1 and 894 cm−1 are ascribed to =CH vibrations. The C-O-C elongation corresponds to the peak at 1109 cm−1 and the C=O elongation corresponds to the peak 1675 cm−1. In the matrix spectrum, a wide band at 3392 cm−1 is related to the hydroxylated group. In relation to the peak at 1030 cm−1, it can be associated with the strong absorption bands of the C-O-C elongation. In the microcapsule spectrum, it can be observed that it is quite similar to the matrix, and that the peaks related to the SEO disappear or are absent, which may be related to the overlap of the peaks of the matrix and SEO due to the low weight fraction of SEO in the total weight of the microcapsules. Through this spectrum, it was possible to verify the successful encapsulation of the SEO (peaks at 1673 cm−1 and 900 cm−1).
Figure 25.
FTIR spectra of (A) pure SEO, (B) pure matrix, and (C) inulin and arabic gum-based microcapsules [209].
The double barrier release system is a method used for essential oils that have antifungal activity, even against drug-resistant fungi. However, there are some limitations due to the sensitivity to pH, temperature, and light. Adepu et al. encapsulated three essential oils (thymol, eugenol, and carvacrol) in a polylactic acid shell with high encapsulation efficiency to achieve their synergistic antifungal activity using the coacervation phase separation method. These were incorporated into bacterial cellulose (a nanofibre fibrous hydrogel) [210]. An antifungal test was performed on the Candida albicans fungus model (a cause of common oral and vaginal infections). Another test was carried out—a transvaginal drug release study in vitro—to compare the release of microcapsules like colloids and composites, where the latter exhibited a controlled release. Through several studies, such as the SEM technique, it was found that the average size and size distribution of the microcapsules depends on the concentration of the used polymer (poly(lactic acid)) and surfactant (poloxamer).
SEM images of BC loaded with microcapsules demonstrate a regular distribution and spherical shape, appearing to be well separated and stable in the stages of the preparation process (Figure 26). From the highest magnification image, it was observed that the microcapsules were anchored to the nanofibre matrix.
Figure 26.
Low- and high-magnification SEM micrographs of (a,a’) BC-PLA1.5-Pol5.0, (b,b’) BC-PLA1.5-Pol2.5, (c,c’) BC-PLA3.0-Pol5.0, and (d,d’) BCPLA3.0-Pol2.5 [210].
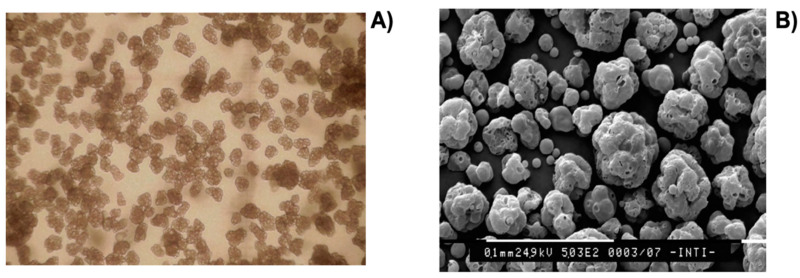
Repellent essential oils are becoming increasingly widespread due to their low toxicity and customer approval. Its application in textile materials has been widely developed. To optimise their application efficiency, it is important to develop long-lasting repellent textiles using OEs. Specos et al. obtained citronella-loaded gelatin microcapsules through the complex coacervation method, which were applied to cotton fabrics in order to study the repellent effectiveness of the obtained fabrics [70]. The release of citronella by the treated tissues was monitored and the repellent activity evaluated by exposing a human hand and arm covered with the treated tissues to Aedes aegypti mosquitoes.
It was found that the tissues treated with citronella microcapsules present greater and more lasting protection against insects in comparison to fabrics sprayed with an ethanol solution of essential oil. Repellent textiles were obtained by filling cotton fabrics with microcapsule sludge, using a conventional drying method. This methodology does not require additional investments for the textile finishing industries, which is a desirable factor in developing countries. Figure 27A shows the morphology of blackberry-type microcapsules in a fresh paste with diameters ranging from 25 to 100 µm, while Figure 27B shows SEM micrographs of spray-dried microcapsules revealing two types of structures, with small spherical units of less than 10 µm and clusters ranging from 25 to 100 µm.
Figure 27.
(A) Optical micrographs of gelatin microcapsules containing citronella essential oil (100× magnification) and (B) SEM microphotographs of spray-dried microcapsules containing citronella essential oil (500× magnification) [70].
In 2016, Ribeiro et al. investigated the functionalisation of photocatalytic titanium dioxide nanoparticles on the surface of polymeric microcapsules as a way to control the release of citronella by solar radiation, thus obtaining a release of a repellent without mechanical intervention [211]. These authors used a modified hydrothermal sol-gel method to synthesise TiO2 nanoparticles. Through several characterisation techniques, these authors were able to observe the surface of the microcapsules and the release efficiency. Using in vitro biological assays with live mosquitoes, the controlled release repellence effect of these photocatalytic microcapsules was reinforced by the inhibition of these vectors. According to the results, it was shown that functionalising the microcapsules with photocatalytic nanoparticles on the surface, and then exposing them to ultraviolet radiation, effectively increased the emission of citronella into the air, repelling mosquitoes. Table 6 shows an overview of illustrative examples of EO microencapsulation oils, wall materials, and microencapsulation methods with industrial importance.
Table 6.
Overview of essential oil microencapsulation, methods, wall materials, and industrial applications.
| Microencapsulation Method | Wall Material(s) | Essential Oil/Source | Applications | Reference |
|---|---|---|---|---|
| Emulsification | Hydroxypropyl methyl cellulose/ chitosan/silica |
Peppermint oil | Medical | [212] |
| Polydopamine | Turpentine | Agrochemical | [213] | |
| β-cyclodextrin | Thyme | Food | [214] | |
| β-cyclodextrin/sugar beet pectin | Garlic | Food | [215] | |
| Ionic Gelation | Cassava starch/poly(butylene adipate-co-terephthalate) | Oregano | Food | [216] |
| Simple Coacervation | Gelatin | Basil | Agrochemical | [217] |
| Poly(vinyl alcohol) | Lemongrass | Food and pharmaceutical | [206] | |
| Complex Coacervation | Gelatin/gum arabic | Citronella | Anti-mosquito textile | [149] |
| Gelatin/sodium alginate | Citronella | Anti-mosquito textile | [218] | |
| Gelatin/gum arabic | Lavender | Cosmetics | [219] | |
| Chitosan/gum arabic/maltodextrin | Peppermint | Cosmetics | [220] | |
| Chitosan/k-carrageenan | Pimenta dioica | Food | [221] | |
| Gelatin/chia mucilage | Oregano | Food | [222] | |
| Mung bean protein isolate/apricot peel pectin | Rose | Food | [223] | |
| Sichuan pepper seed soluble dietary fibre/soybean protein isolate | Sichuan pepper | Food | [224] | |
| Whey protein isolate/arabic gum | Orange | Food | [225] | |
| Interfacial Polymerization |
Polyurea | Lemongrass, lavender, sage and thyme | Agrochemical | [200] |
| In situ polymerisation | Silicon dioxide/poly(melamine formaldehyde) | Cinnamon | Agrochemical | [226] |
| Melamine/formaldehyde | Thyme | Food | [169] | |
| Melamine/formaldehyde | Lavandin and tea tree | Paints | [227] | |
| Extrusion | Sodium alginate | Nutmeg | Pharmaceutical | [228] |
| Sodium alginate | Rosemary | Agrochemical | [229] | |
| Spray Drying | Acacia gum | Citronella | Cosmetics | [230] |
| Palm trunk/ chitosan | Ginger | Food | [231] | |
| Maltodextrin | Citrus | Food | [232] | |
| Gum arabic/maltodextrin/sodium alginate/whey protein concentrate | Juniper berry |
Food | [233] | |
| Whey protein isolate/maltodextrin/sodium alginate |
Cinnamon | Food | [234] | |
| Hydroxypropyl methyl cellulose/maltodextrin | Oregano | Food | [235] | |
| Gelatin/arabic gum | Citronella | Textile | [70] | |
| Gum arabic/starch/maltodextrin/inulin | Rosemary | Food | [203] | |
| Gum arabic/modified starch | Black pepper | Food | [236] | |
| Gum arabic/maltodextrin/whey protein isolate |
Basil | Food | [237] | |
| Freeze-drying | Urushiol | Not mentioned | Medical and pharmaceutical | [238] |
| β-cyclodextrin | Litsea cubeba | Cosmetics and pharmaceutical | [239] | |
| Maltodextrin/gelation | Lemongrass | Cosmetics, pharmaceutical and food | [240] | |
| Gum arabic/collagen hydrolysate |
Origanum onites L. |
Environmental | [241] | |
| Whey protein isolate/carboxymethylcellulose |
Orange | Food | [242] | |
| Supercritical Fluid Technology |
Starch | Oregano | Food | [243] |
| n-octenyl succinic/modified starches | Lavandin | Agrochemical | [244] | |
| Modified starch | Limonene | Food | [245] |
5. Conclusions
This review summarises different types of EO structures and describes their extraction and application methodology. In addition, different techniques for microencapsulating essential oils are described and some reports are presented to provide a basis for research and industrial development.
As described in this paper, EOs are used in several applications in the pharmaceutical, cosmetic, agricultural, and food industries, as they are natural metabolites produced by plants with interesting properties. Furthermore, EOs are being explored as an alternative to synthetic products due to their ecological factors and the fact that their characteristics are different from the corresponding synthetic product. For example, synthesised oil may have the same odour as natural oils, but may not have the same therapeutic characteristics.
Currently, there is growing interest in the application of EO microencapsulation, making it an effective and important tool in the preparation of high-quality products, improving their chemical, oxidative, and thermal stability. Besides these advantages, the shelf life, biological activity, functional activity, controlled release, physicochemical properties, and general quality of oils can also be improved with microencapsulation technology.
Based on the scientific studies available and presented throughout this paper, it can be concluded that the microencapsulation of EOs is an emerging trend for industrial applications. However, this development has limitations, such as the low diversity of wall materials and their incompatibility with microencapsulation methods. Many of the encapsulating agents available present a high cost for production on an industrial scale. In future research, microencapsulation must also be directed to encapsulate a different mixture of oils by different techniques, in order to disguise the flavour of the oils and to improve safety, quality, and nutritional value.
Funding
Vânia I. Sousa and Joana F. Parente are grateful to the Project ReleaseME-POCI-01-0247-FEDER-033268, for their research grants from the Agência Nacional de Inovação, co-funded by the European Regional Development Fund (ERDF), through the Operational Programme for Competitiveness and Internationalisation (COMPETE 2020), under the PORTUGAL 2020 Partnership Agreement. Juliana F. Marques and Marta A. Forte are grateful to the Fundação para a Ciência e Tecnologia (FCT) of Portugal for their Ph.D. grants, SFRH/BD/112868/2015 and PD/BD/128491/2017, respectively. The authors also acknowledge the funding from FCT/PIDDAC through the Strategic Funds project reference UIDB/04650/2020-2023. This research was funded by the project Repel+: New solutions for mosquito repellence as an application for malaria control (project number 47036) from the Agência Nacional de Inovação, co-funded by the European Regional Development Fund (ERDF), through the Operational Programme for Competitiveness and Internationalisation (COMPETE 2020), under the PORTUGAL 2020 Partnership Agreement.
Data Availability Statement
The raw/processed data required to reproduce these findings cannot be shared at this time due to technical or time limitations.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Benchaar C., Greathead H. Essential oils and opportunities to mitigate enteric methane emissions from ruminants. Anim. Feed Sci. Technol. 2011;166–167:338–355. doi: 10.1016/j.anifeedsci.2011.04.024. [DOI] [Google Scholar]
- 2.Benchaar C., Calsamiglia S., Chaves A.V., Fraser G.R., Colombatto D., McAllister T.A., Beauchemin K.A. A review of plant-derived essential oils in ruminant nutrition and production. Anim. Feed Sci. Technol. 2008;145:209–228. doi: 10.1016/j.anifeedsci.2007.04.014. [DOI] [Google Scholar]
- 3.Raut J.S., Karuppayil S.M. A status review on the medicinal properties of essential oils. Ind. Crops Prod. 2014;62:250–264. doi: 10.1016/j.indcrop.2014.05.055. [DOI] [Google Scholar]
- 4.Lubbe A., Verpoorte R. Cultivation of medicinal and aromatic plants for specialty industrial materials. Ind. Crop. Prod. 2011;34:785–801. doi: 10.1016/j.indcrop.2011.01.019. [DOI] [Google Scholar]
- 5.daCunha A.P., Nogueira M.T., Rodrigues Roque O., Barroso J.M.G. Plantas Aromáticas e Óleos Essenciais: Composição e Aplicações. Fundação Calouste Gulbenkian; Lisbon, Portugal: 2012. [Google Scholar]
- 6.Teixeira B., Marques A., Ramos C., Neng N.R., Nogueira J.M.F., Saraiva J.A., Nunes M.L. Chemical composition and antibacterial and antioxidant properties of commercial essential oils. Ind. Crops Prod. 2013;43:587–595. doi: 10.1016/j.indcrop.2012.07.069. [DOI] [Google Scholar]
- 7.Sacchetti G., Maietti S., Muzzoli M., Scaglianti M., Manfredini S., Radice M., Bruni R. Comparative evaluation of 11 essential oils of different origin as functional antioxidants, antiradicals and antimicrobials in foods. Food Chem. 2005;91:621–632. doi: 10.1016/j.foodchem.2004.06.031. [DOI] [Google Scholar]
- 8.Bakkali F., Averbeck S., Averbeck D., Idaomar M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008;46:446–475. doi: 10.1016/j.fct.2007.09.106. [DOI] [PubMed] [Google Scholar]
- 9.Warnke P.H., Becker S.T., Podschun R., Sivananthan S., Springer I.N., Russo P.A.J., Wiltfang J., Fickenscher H., Sherry E. The battle against multi-resistant strains: Renaissance of antimicrobial essential oils as a promising force to fight hospital-acquired infections. J. Cranio-Maxillofac. Surg. 2009;37:392–397. doi: 10.1016/j.jcms.2009.03.017. [DOI] [PubMed] [Google Scholar]
- 10.De Billerbeck V.G. Huiles essentielles et bactéries résistantes aux antibiotiques. Phytotherapie. 2007;5:249–253. doi: 10.1007/s10298-007-0265-z. [DOI] [Google Scholar]
- 11.Burt S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004;94:223–253. doi: 10.1016/j.ijfoodmicro.2004.03.022. [DOI] [PubMed] [Google Scholar]
- 12.Nakatsu T., Lupo A.T., Chinn J.W., Kang R.K.L. Biological activity of essential oils and their constituents. Stud. Nat. Prod. Chem. 2000;21:571–631. doi: 10.1016/S1572-5995(00)80014-9. [DOI] [Google Scholar]
- 13.Dima C., Dima S. Essential oils in foods: Extraction, stabilization, and toxicity. Curr. Opin. Food Sci. 2015;5:29–35. doi: 10.1016/j.cofs.2015.07.003. [DOI] [Google Scholar]
- 14.Hogenbom J., Jones A., Wang H.V., Pickett L.J., Faraone N. Synthesis and characterization of β-cyclodextrin-essential oil inclusion complexes for tick repellent development. Polymers. 2021;13:1892. doi: 10.3390/polym13111892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ponce Cevallos P.A., Buera M.P., Elizalde B.E. Encapsulation of cinnamon and thyme essential oils components (cinnamaldehyde and thymol) in β-cyclodextrin: Effect of interactions with water on complex stability. J. Food Eng. 2010;99:70–75. doi: 10.1016/j.jfoodeng.2010.01.039. [DOI] [Google Scholar]
- 16.Saberi-Riseh R., Moradi-Pour M., Mohammadinejad R., Thakur V.K. Biopolymers for biological control of plant pathogens: Advances in microencapsulation of beneficial microorganisms. Polymers. 2021;13:1938. doi: 10.3390/polym13121938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lengyel M., Kállai-Szabó N., Antal V., Laki A.J., Antal I. Microparticles, microspheres, and microcapsules for advanced drug delivery. Sci. Pharm. 2019;87:20. doi: 10.3390/scipharm87030020. [DOI] [Google Scholar]
- 18.Council of Europe, editor. European Pharmacopoeia. 10th ed. EDQM; Strasbourg, France: 2021. European Directorate for the Quality of Medicines & HealthCare (EDQM) [Google Scholar]
- 19.Svoboda K.P., Greenaway R.I. Investigation of volatile oil glands of Satureja hortensis L. (summer savory) and phytochemical comparison of different varieties. Int. J. Aromather. 2003;13:196–202. doi: 10.1016/S0962-4562(03)00038-9. [DOI] [Google Scholar]
- 20.Dajic Stevanovic Z., Sieniawska E., Glowniak K., Obradovic N., Pajic-Lijakovic I. Natural Macromolecules as Carriers for Essential Oils: From Extraction to Biomedical Application. Front. Bioeng. Biotechnol. 2020;8:1–24. doi: 10.3389/fbioe.2020.00563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Figueiredo A.C., Pedro L.G., Barroso J.G. Potencialidades e Aplicações das Plantas Aromáticas e Medicinais. Plant Biotechnology Center—Faculty of Sciences of the University of Lisbon; Lisbon, Portugal: 2007. Plantas aromáticas e medicinais: Factores que afetam a produção. [Google Scholar]
- 22.Peixoto L.R., Rosalen P.L., Ferreira G.L.S., Freires I.A., de Carvalho F.G., Castellano L.R., de Castro R.D. Antifungal activity, mode of action and anti-biofilm effects of Laurus nobilis Linnaeus essential oil against Candida spp. Arch. Oral Biol. 2017;73:179–185. doi: 10.1016/j.archoralbio.2016.10.013. [DOI] [PubMed] [Google Scholar]
- 23.Eslahi H., Fahimi N., Sardarian A.R. Chemical Composition of Essential Oils: Chemistry, Safety and Applications. In: Hashemi S.M.B., Khaneghah A.M., de Souza Sant’Ana A., editors. Essential Oils in Food Processing: Chemistry, Safety and Applications. Wiley; Hoboken, NJ, USA: 2017. pp. 119–171. [Google Scholar]
- 24.Dhifi W., Bellili S., Jazi S., Bahloul N., Mnif W. Essential Oils’ Chemical Characterization and Investigation of Some Biological Activities: A Critical Review. Medicines. 2016;3:25. doi: 10.3390/medicines3040025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chang T.H., Hsieh F.L., Ko T.P., Teng K.H., Liang P.H., Wang A.H.J. Structure of a heterotetrameric geranyl pyrophosphate synthase from mint (Mentha piperita) reveals intersubunit regulation. Plant Cell. 2010;22:454–467. doi: 10.1105/tpc.109.071738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Noriega P. Terpenes in Essential Oils: Bioactivity and Applications. In: Perveen S., Al-Taweel A.M., editors. Terpenes and Terpenoids—Recent Advances. Licensee IntechOpen; London, UK: 2020. [Google Scholar]
- 27.Neelam, Khatkar A., Sharma K.K. Phenylpropanoids and its derivatives: Biological activities and its role in food, pharmaceutical and cosmetic industries. Crit. Rev. Food Sci. Nutr. 2020;60:2655–2675. doi: 10.1080/10408398.2019.1653822. [DOI] [PubMed] [Google Scholar]
- 28.Liu H.-W.B., Begley T.P. Comprehensive Natural Products III. 3rd ed. Elsevier; Amsterdam, The Netherlands: 2020. [Google Scholar]
- 29.Grassmann J., Elstner E.F. Essential Oils—Properties and Uses. In: Caballero B., editor. Encyclopedia of Food Sciences and Nutrition. Academic Press; Cambridge, MA, USA: 2003. [Google Scholar]
- 30.Stratakos A., Koidis A. Essential Oils in Food Preservation, Flavor and Safety. In: Preedy V.R., editor. Methods for Extracting Essential Oils. Academic Press; Cambridge, MA, USA: 2016. pp. 31–38. [Google Scholar]
- 31.Silva M.G.F. Bachelor’s Thesis. Federal Technological University of Paraná; Londrina, Brazil: Dec, 2011. Atividade antioxidante e antimicrobiana in vitro de óleos essenciais e extratos hidroalcóolicos de manjerona (Origanum majorana L.) e manjericão (Ocimum basilicum L.) Graduation Conclusion Work. [Google Scholar]
- 32.Sartor R.B. Modelagem, Simulação e Otimização de uma Unidade Industrial de Extração de Óleos Essenciais por Arraste a Vapor. Universidade Federal do Rio Grande do Sul; Porto Alegre, Brazil: 2009. [Google Scholar]
- 33.Masango P. Cleaner production of essential oils by steam distillation. J. Clean. Prod. 2005;13:833–839. doi: 10.1016/j.jclepro.2004.02.039. [DOI] [Google Scholar]
- 34.Luque de Castro M.D., García-Ayuso L.E. Soxhlet extraction of solid materials: An outdated technique with a promising innovative future. Anal. Chim. Acta. 1998;369:1–10. doi: 10.1016/S0003-2670(98)00233-5. [DOI] [Google Scholar]
- 35.Elyemni M., Louaste B., Nechad I., Elkamli T., Bouia A., Taleb M., Chaouch M., Eloutassi N. Extraction of Essential Oils of Rosmarinus officinalis L. by Two Different Methods: Hydrodistillation and Microwave Assisted Hydrodistillation. Sci. World J. 2019;2019:3659432. doi: 10.1155/2019/3659432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rezaei K., Hashemi N.B., Sahraee S. Phytopharmaceuticals. Wiley; Hoboken, NJ, USA: 2021. Use of Hydrodistillation as a Green Technology to Obtain Essential Oils From Several Medicinal Plants Belonging to Lamiaceae (Mint) Family; pp. 59–75. [DOI] [Google Scholar]
- 37.Atti-Santos A.C., Rossato M., Serafini L.A., Cassel E., Moyna P. Extraction of essential oils from lime (Citrus latifolia tanaka) by hydrodistillation and supercritical carbon dioxide. Brazilian Arch. Biol. Technol. 2005;48:155–160. doi: 10.1590/S1516-89132005000100020. [DOI] [Google Scholar]
- 38.Baldin V.P., Bertin de Lima Scodro R., Mariano Fernandez C.M., Ieque A.L., Caleffi-Ferracioli K.R., Dias Siqueira V.L., de Almeida A.L., Gonçalves J.E., Garcia Cortez D.A., Cardoso R.F. Ginger essential oil and fractions against Mycobacterium spp. J. Ethnopharmacol. 2019;244:112095. doi: 10.1016/j.jep.2019.112095. [DOI] [PubMed] [Google Scholar]
- 39.Fitriady M.A., Sulaswatty A., Agustian E., Salahuddin, Aditama D.P.F. Steam distillation extraction of ginger essential oil: Study of the effect of steam flow rate and time process. AIP Conf. Proc. 2017;1803:020032-1–020032-10. doi: 10.1063/1.4973159. [DOI] [Google Scholar]
- 40.Jeliazkova E., Zheljazkov V.D., Kačániova M., Astatkie T., Tekwani B.L. Sequential elution of essential oil constituents during steam distillation of hops (Humulus lupulus L.) and influence on oil yield and antimicrobial activity. J. Oleo Sci. 2018;67:871–883. doi: 10.5650/jos.ess17216. [DOI] [PubMed] [Google Scholar]
- 41.Gavahian M., Chu Y.H. Ohmic accelerated steam distillation of essential oil from lavender in comparison with conventional steam distillation. Innov. Food Sci. Emerg. Technol. 2018;50:34–41. doi: 10.1016/j.ifset.2018.10.006. [DOI] [Google Scholar]
- 42.Donelian A., Carlson L.H.C., Lopes T.J., Machado R.A.F. Comparison of extraction of patchouli (Pogostemon cablin) essential oil with supercritical CO2 and by steam distillation. J. Supercrit. Fluids. 2009;48:15–20. doi: 10.1016/j.supflu.2008.09.020. [DOI] [Google Scholar]
- 43.Guzmán E., Lucia A. Essential Oils and Their Individual Components in Cosmetic Products. Cosmetics. 2021;8:114. doi: 10.3390/cosmetics8040114. [DOI] [Google Scholar]
- 44.Sharmeen J.B., Mahomoodally F.M., Zengin G., Maggi F. Essential oils as natural sources of fragrance compounds for cosmetics and cosmeceuticals. Molecules. 2021;26:666. doi: 10.3390/molecules26030666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Carvalho I.T., Estevinho B.N., Santos L. Application of microencapsulated essential oils in cosmetic and personal healthcare products—A review. Int. J. Cosmet. Sci. 2016;38:109–119. doi: 10.1111/ics.12232. [DOI] [PubMed] [Google Scholar]
- 46.Asbahani A.E., Miladi K., Badri W., Sala M., Addi E.H.A., Casabianca H., Mousadik A.E., Hartmann D., Jilale A., Renaud F.N.R., et al. Essential oils: From extraction to encapsulation. Int. J. Pharm. 2015;483:220–243. doi: 10.1016/j.ijpharm.2014.12.069. [DOI] [PubMed] [Google Scholar]
- 47.Li G., Xiang S., Pan Y., Long X., Cheng Y., Han L., Zhao X. Effects of Cold-Pressing and Hydrodistillation on the Active Non-volatile Components in Lemon Essential Oil and the Effects of the Resulting Oils on Aging-Related Oxidative Stress in Mice. Front. Nutr. 2021;8:1–15. doi: 10.3389/fnut.2021.689094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Menichini F., Tundis R., Bonesi M., De Cindio B., Loizzo M.R., Conforti F., Statti G.A., Menabeni R., Bettini R., Menichini F. Chemical composition and bioactivity of Citrus medica L. cv. Diamante essential oil obtained by hydrodistillation, cold-pressing and supercritical carbon dioxide extraction. Nat. Prod. Res. 2011;25:789–799. doi: 10.1080/14786410902900085. [DOI] [PubMed] [Google Scholar]
- 49.Ferhat M.A., Meklati B.Y., Chemat F. Comparison of different isolation methods of essential oil from Citrus fruits: Cold pressing, hydrodistillation and microwave ‘dry’ distillation. Flavour Fragr. J. 2007;22:494–504. doi: 10.1002/ffj.1829. [DOI] [Google Scholar]
- 50.Yousefi M., Rahimi-Nasrabadi M., Pourmortazavi S.M., Wysokowski M., Jesionowski T., Ehrlich H., Mirsadeghi S. Supercritical fluid extraction of essential oils. TrAC—Trends Anal. Chem. 2019;118:182–193. doi: 10.1016/j.trac.2019.05.038. [DOI] [Google Scholar]
- 51.De Corato U., Maccioni O., Trupo M., Di Sanzo G. Use of essential oil of Laurus nobilis obtained by means of a supercritical carbon dioxide technique against post harvest spoilage fungi. Crop Prot. 2010;29:142–147. doi: 10.1016/j.cropro.2009.10.012. [DOI] [Google Scholar]
- 52.Irmak S., Solakyildirim K., Hesenov A., Erbatur O. Study on the stability of supercritical fluid extracted rosemary (Rosmarinus Offcinalis L.) essential oil. J. Anal. Chem. 2010;65:899–906. doi: 10.1134/S1061934810090030. [DOI] [Google Scholar]
- 53.Glisic S., Ivanovic J., Ristic M., Skala D. Extraction of sage (Salvia officinalis L.) by supercritical CO2: Kinetic data, chemical composition and selectivity of diterpenes. J. Supercrit. Fluids. 2010;52:62–70. doi: 10.1016/j.supflu.2009.11.009. [DOI] [Google Scholar]
- 54.Uquiche E., Cirano N., Millao S. Supercritical fluid extraction of essential oil from Leptocarpha rivularis using CO2. Ind. Crops Prod. 2015;77:307–314. doi: 10.1016/j.indcrop.2015.09.001. [DOI] [Google Scholar]
- 55.Zhao S., Zhang D. Supercritical fluid extraction and characterisation of Moringa oleifera leaves oil. Sep. Purif. Technol. 2013;118:497–502. doi: 10.1016/j.seppur.2013.07.046. [DOI] [Google Scholar]
- 56.Martínez-Abad A., Ramos M., Hamzaoui M., Kohnen S., Jiménez A., Garrigós M.C. Optimisation of sequential microwave-assisted extraction of essential oil and pigment from lemon peels waste. Foods. 2020;9:1493. doi: 10.3390/foods9101493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ferhat M.A., Meklati B.Y., Smadja J., Chemat F. An improved microwave Clevenger apparatus for distillation of essential oils from orange peel. J. Chromatogr. A. 2006;1112:121–126. doi: 10.1016/j.chroma.2005.12.030. [DOI] [PubMed] [Google Scholar]
- 58.Bayramoglu B., Sahin S., Sumnu G. Extraction of essential oil from laurel leaves by using microwaves. Sep. Sci. Technol. 2009;44:722–733. doi: 10.1080/01496390802437271. [DOI] [Google Scholar]
- 59.Lucchesi M.E., Smadja J., Bradshaw S., Louw W., Chemat F. Solvent free microwave extraction of Elletaria cardamomum L.: A multivariate study of a new technique for the extraction of essential oil. J. Food Eng. 2007;79:1079–1086. doi: 10.1016/j.jfoodeng.2006.03.029. [DOI] [Google Scholar]
- 60.Filly A., Fernandez X., Minuti M., Visinoni F., Cravotto G., Chemat F. Solvent-free microwave extraction of essential oil from aromatic herbs: From laboratory to pilot and industrial scale. Food Chem. 2014;150:193–198. doi: 10.1016/j.foodchem.2013.10.139. [DOI] [PubMed] [Google Scholar]
- 61.Okoh O.O., Sadimenko A.P., Afolayan A.J. Comparative evaluation of the antibacterial activities of the essential oils of Rosmarinus officinalis L. obtained by hydrodistillation and solvent free microwave extraction methods. Food Chem. 2010;120:308–312. doi: 10.1016/j.foodchem.2009.09.084. [DOI] [Google Scholar]
- 62.Lucchesi M.E., Chemat F., Smadja J. Solvent-free microwave extraction of essential oil from aromatic herbs: Comparison with conventional hydro-distillation. J. Chromatogr. A. 2004;1043:323–327. doi: 10.1016/j.chroma.2004.05.083. [DOI] [PubMed] [Google Scholar]
- 63.Sandhu H.K., Sinha P., Emanuel N., Kumar N., Sami R., Khojah E., Al-Mushhin A.A.M. Effect of ultrasound-assisted pretreatment on extraction efficiency of essential oil and bioactive compounds from citrus waste by-products. Separations. 2021;8:244. doi: 10.3390/separations8120244. [DOI] [Google Scholar]
- 64.Zheng S., Zhang G., Wang H.J., Long Z., Wei T., Li Q. Progress in ultrasound-assisted extraction of the value-added products from microorganisms. World J. Microbiol. Biotechnol. 2021;37:1–14. doi: 10.1007/s11274-021-03037-y. [DOI] [PubMed] [Google Scholar]
- 65.Ramos C., Teixeira B., Batista I., Matos O., Serrano C., Neng N.R., Nogueira J.M.F., Nunes M.L., Marques A. Antioxidant and antibacterial activity of essential oil and extracts of bay laurel Laurus nobilis Linnaeus (Lauraceae) from Portugal. Nat. Prod. Res. 2012;26:518–529. doi: 10.1080/14786419.2010.531478. [DOI] [PubMed] [Google Scholar]
- 66.Dadalioǧlu I., Evrendilek G.A. Chemical compositions and antibacterial effects of essential oils of Turkish oregano (Origanum minutiflorum), bay laurel (Laurus nobilis), Spanish lavender (Lavandula stoechas L.), and fennel (Foeniculum vulgare) on common foodborne pathogens. J. Agric. Food Chem. 2004;52:8255–8260. doi: 10.1021/jf049033e. [DOI] [PubMed] [Google Scholar]
- 67.Shaaya E., Ravid U., Paster N., Juven B., Zisman U., Pissarev V. Fumigant toxicity of essential oils against four major stored-product insects. J. Chem. Ecol. 1991;17:499–504. doi: 10.1007/BF00982120. [DOI] [PubMed] [Google Scholar]
- 68.Cosimi S., Rossi E., Cioni P.L., Canale A. Bioactivity and qualitative analysis of some essential oils from Mediterranean plants against stored-product pests: Evaluation of repellency against Sitophilus zeamais Motschulsky, Cryptolestes ferrugineus (Stephens) and Tenebrio molitor (L.) J. Stored Prod. Res. 2009;45:125–132. doi: 10.1016/j.jspr.2008.10.002. [DOI] [Google Scholar]
- 69.Lawless J. The Encyclopedia of Essential Oils: The Complete Guide to the Use of Aromatic Oils In Aromatherapy, Herbalism, Health, and Well Being. Conari Press; Newburyport, MA, USA: 2013. [Google Scholar]
- 70.Specos M.M.M., García J.J., Tornesello J., Marino P., Vecchia D.M., Tesoriero D.M.V., Hermida L.G. Microencapsulated citronella oil for mosquito repellent finishing of cotton textiles. Trans. R. Soc. Trop. Med. Hyg. 2010;104:653–658. doi: 10.1016/j.trstmh.2010.06.004. [DOI] [PubMed] [Google Scholar]
- 71.Sakulku U., Nuchuchua O., Uawongyart N., Puttipipatkhachorn S., Soottitantawat A., Ruktanonchai U. Characterization and mosquito repellent activity of citronella oil nanoemulsion. Int. J. Pharm. 2009;372:105–111. doi: 10.1016/j.ijpharm.2008.12.029. [DOI] [PubMed] [Google Scholar]
- 72.Tawatsin A., Wratten S.D., Scott R.R., Thavara U., Techadamrongsin Y. Repellency of volatile oils from plants against three mosquito vectors. J. Vectory Ecol. 2001;26:76–82. [PubMed] [Google Scholar]
- 73.Nakahara K., Alzoreky N.S., Yoshihashi T., Nguyen H.T.T., Trakoontivakorn G. Chemical Composition and Antifungal Activity of Essential Oil from Cymbopogon nardus (Citronella Grass) Jpn. Agric. Res. Q. 2003;37:249–252. doi: 10.6090/jarq.37.249. [DOI] [Google Scholar]
- 74.Andrade M.A., Das Graças Cardoso M., Batista L.R., Mallet A.C.T., Machado S.M.F. Essential oils of Cinnamomum zeylanicum, Cymbopogon nardus and Zingiber officinale: Composition, antioxidant and antibacterial activities. Rev. Ciência Agronômica. 2012;43:399–408. doi: 10.1590/S1806-66902012000200025. [DOI] [Google Scholar]
- 75.Simpson W.J., Smith A.R.W. Factors affecting antibacterial activity of hop compounds and their derivatives. J. Appl. Bacteriol. 1992;72:327–334. doi: 10.1111/j.1365-2672.1992.tb01843.x. [DOI] [PubMed] [Google Scholar]
- 76.Zanoli P., Zavatti M. Pharmacognostic and pharmacological profile of Humulus lupulus L. J. Ethnopharmacol. 2008;116:383–396. doi: 10.1016/j.jep.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 77.Miranda C.L., Stevens J.F., Helmrich A., Henderson M.C., Rodriguez R.J., Yang Y.H., Deinzer M.L., Barnes D.W., Buhler D.R. Antiproliferative and cytotoxic effects of prenylated flavonoids from hops (Humulus lupulus) in human cancer cell lines. Food Chem. Toxicol. 1999;37:271–285. doi: 10.1016/S0278-6915(99)00019-8. [DOI] [PubMed] [Google Scholar]
- 78.Reher T., Van Kerckvoorde V., Verheyden L., Wenseleers T., Beliën T., Bylemans D., Martens J.A. Evaluation of hop (Humulus lupulus) as a repellent for the management of Drosophila suzukii. Crop Prot. 2019;124:104839. doi: 10.1016/j.cropro.2019.05.033. [DOI] [Google Scholar]
- 79.Bedini S., Flamini G., Girardi J., Cosci F., Conti B. Not just for beer: Evaluation of spent hops (Humulus lupulus L.) as a source of eco-friendly repellents for insect pests of stored foods. J. Pest Sci. 2015;88:583–592. doi: 10.1007/s10340-015-0647-1. [DOI] [Google Scholar]
- 80.Abdel-Naime W.A., Fahim J.R., Fouad M.A., Kamel M.S. Antibacterial, antifungal, and GC–MS studies of Melissa officinalis. S. Afr. J. Bot. 2019;124:228–234. doi: 10.1016/j.sajb.2019.05.011. [DOI] [Google Scholar]
- 81.Shakeri A., Sahebkar A., Javadi B. Melissa officinalis L.—A review of its traditional uses, phytochemistry and pharmacology. J. Ethnopharmacol. 2016;188:204–228. doi: 10.1016/j.jep.2016.05.010. [DOI] [PubMed] [Google Scholar]
- 82.Moradkhani H., Sargsyan E., Bibak H., Naseri B., Sadat-Hosseini M., Fayazi-Barjin A., Meftahizade H. Melissa officinalis L., a valuable medicine plant: A review. J. Med. Plants Res. 2010;4:2753–2759. [Google Scholar]
- 83.Vitalini S., Tomè F., Fico G. Traditional uses of medicinal plants in Valvestino (Italy) J. Ethnopharmacol. 2009;121:106–116. doi: 10.1016/j.jep.2008.10.005. [DOI] [PubMed] [Google Scholar]
- 84.Biswas K., Banerjee R.K., Bandyopadhyay U., Chattopadhyay I. Biological activities and medicinal properties of Neem (Azadirachta indica) Curr. Sci. 2001;82:1336–1445. [Google Scholar]
- 85.Schmutterer H. Properties and Potencial of Natural Pesticides from Neem Tree, Azadirachta Indica. Anu. Rev. Entomol. 1990;35:271–297. doi: 10.1146/annurev.en.35.010190.001415. [DOI] [PubMed] [Google Scholar]
- 86.Rao A., Sukumar S., Paramasivam T., Kamalakshi S., Parashuraman M., Shantha A.R. Study of antiviral activity of tender leaves of Margosa tree (Melia azadericta) on vaccinia and variola virus: A preliminary report. Indian J Med Res. 1969;57:495–502. [PubMed] [Google Scholar]
- 87.Chopra I., Gupta K., Nazir B. Preliminary study of anti-bacterial substances from Melia azidirachta. Indian J. Med. Res. 1952;40:511–515. [PubMed] [Google Scholar]
- 88.Almas K. The antimicrobial effects of extracts of Azadirachta indica (Neem) and Salvadora persica (Arak) chewing sticks. Indian J. Dent. Res. 1999;10:23–26. [PubMed] [Google Scholar]
- 89.Salem N., Sriti J., Bachrouch O., Msaada K., Khammassi S., Hammami M., Selmi S., Boushih E., Ouertani M., Hachani N., et al. Phenological stage effect on phenolic composition and repellent potential of Mentha pulegium against Tribolium castaneum and Lasioderma serricorne. Asian Pac. J. Trop. Biomed. 2018;8:207–216. doi: 10.4103/2221-1691.231283. [DOI] [Google Scholar]
- 90.Salem N., Bachrouch O., Sriti J., Msaada K., Khammassi S., Hammami M., Selmi S., Boushih E., Koorani S., Abderraba M., et al. Fumigant and repellent potentials of Ricinus communis and Mentha pulegium essential oils against Tribolium castaneum and Lasioderma serricorne. Int. J. Food Prop. 2018;20:2899–2913. doi: 10.1080/10942912.2017.1382508. [DOI] [Google Scholar]
- 91.Soares S.F., Borges L.M.F., de Sousa Braga R., Ferreira L.L., Louly C.C.B., Tresvenzol L.M.F., de Paula J.R., Ferri P.H. Repellent activity of plant-derived compounds against Amblyomma cajennense (Acari: Ixodidae) nymphs. Vet. Parasitol. 2010;167:67–73. doi: 10.1016/j.vetpar.2009.09.047. [DOI] [PubMed] [Google Scholar]
- 92.Brahmi F., Abdenour A., Bruno M., Silvia P., Alessandra P., Danilo F., Drifa Y.G., Fahmi E.M., Khodir M., Mohamed C. Chemical composition and in vitro antimicrobial, insecticidal and antioxidant activities of the essential oils of Mentha pulegium L. and Mentha rotundifolia (L.) Huds growing in Algeria. Ind. Crops Prod. 2016;88:96–105. doi: 10.1016/j.indcrop.2016.03.002. [DOI] [Google Scholar]
- 93.Teixeira B., Marques A., Ramos C., Batista I., Serrano C., Matos O., Neng N.R., Nogueira J.M.F., Saraiva J.A., Nunes M.L. European pennyroyal (Mentha pulegium) from Portugal: Chemical composition of essential oil and antioxidant and antimicrobial properties of extracts and essential oil. Ind. Crops Prod. 2012;36:81–87. doi: 10.1016/j.indcrop.2011.08.011. [DOI] [Google Scholar]
- 94.Ait-Ouazzou A., Lorán S., Arakrak A., Laglaoui A., Rota C., Herrera A., Pagán R., Conchello P. Evaluation of the chemical composition and antimicrobial activity of Mentha pulegium, Juniperus phoenicea, and Cyperus longus essential oils from Morocco. Food Res. Int. 2012;45:313–319. doi: 10.1016/j.foodres.2011.09.004. [DOI] [Google Scholar]
- 95.Khaled-Khodja N., Boulekbache-Makhlouf L., Madani K. Phytochemical screening of antioxidant and antibacterial activities of methanolic extracts of some Lamiaceae. Ind. Crops Prod. 2014;61:41–48. doi: 10.1016/j.indcrop.2014.06.037. [DOI] [Google Scholar]
- 96.Pehlivan Karakaş F., Yildirim A., Türker A. Biological screening of various medicinal plant extracts for antibacterial and antitumor activities. Turk. J. Biol. 2012;36:641–652. doi: 10.3906/biy-1203-16. [DOI] [Google Scholar]
- 97.Lee S.W., Li G., Lee K.S., Jung J.S., Xu M.L., Seo C.S., Chang H.W., Kim S.K., Song D.K., Son J.K. Preventive Agents against Sepsis and New Phenylpropanoid Glucosides from the Fruits of Illicium verum. Planta Med. 2003;69:861–864. doi: 10.1055/s-2003-43210. [DOI] [PubMed] [Google Scholar]
- 98.Ohira H., Torii N., Aida T.M., Watanabe M., Smith R.L. Rapid separation of shikimic acid from Chinese star anise (Illicium verum Hook. f.) with hot water extraction. Sep. Purif. Technol. 2009;69:102–108. doi: 10.1016/j.seppur.2009.07.005. [DOI] [Google Scholar]
- 99.Yang J.F., Yang C.H., Chang H.W., Yang C.S., Wang S.M., Hsieh M.C., Chuang L.Y. Chemical composition and antibacterial activities of Illicium verum against antibiotic-resistant pathogens. J. Med. Food. 2010;13:1254–1262. doi: 10.1089/jmf.2010.1086. [DOI] [PubMed] [Google Scholar]
- 100.Padmashree A., Roopa N., Semwal A.D., Sharma G.K., Agathian G., Bawa A.S. Star-anise (Illicium verum) and black caraway (Carum nigrum) as natural antioxidants. Food Chem. 2007;104:59–66. doi: 10.1016/j.foodchem.2006.10.074. [DOI] [Google Scholar]
- 101.Xin S., Xiongmin L., Chunlin F., Qiuxia L. Extraction, characterization and antitumor effect of the polysaccharides from star anise (Illicium verum Hook. f.) J. Med. Plants Res. 2010;4:2666–2673. doi: 10.5897/JMPR09.438. [DOI] [Google Scholar]
- 102.Lee J.S., Park M.A., Yoon C.S., Na J.H., Han J. Characterization and Preservation Performance of Multilayer Film with Insect Repellent and Antimicrobial Activities for Sliced Wheat Bread Packaging. J. Food Sci. 2019;84:3194–3203. doi: 10.1111/1750-3841.14823. [DOI] [PubMed] [Google Scholar]
- 103.Park M.A., Chang Y., Choi I., Bai J., Ja-hyun N., Han J. Development of A Comprehensive Biological Hazard-Proof Packaging Film with Insect-Repellent, Antibacterial, and Antifungal Activities. J. Food Sci. 2018;83:3035–3043. doi: 10.1111/1750-3841.14397. [DOI] [PubMed] [Google Scholar]
- 104.Brown J. Valeriana officinalis. Aust. J. Herb. Med. 2012;24:136–139. doi: 10.1007/978-3-540-71095-0_10944. [DOI] [Google Scholar]
- 105.Joanne Barnes Herbal Medicines: A Guide for Health Care Professionals. 2nd ed. Pharmaceutical Press; London, UK: 1996. [Google Scholar]
- 106.Grant K.L. Patient education and herbal dietary supplements. Am. J. Health Pharm. 2000;57:1997–2003. doi: 10.1093/ajhp/57.21.1997. [DOI] [PubMed] [Google Scholar]
- 107.Wang J., Zhao J., Liu H., Zhou L., Liu Z., Wang J., Han J., Yu Z., Yang F. Chemical analysis and biological activity of the essential oils of two valerianaceous species from China: Nardostachys chinensis and valeriana officinalis. Molecules. 2010;15:6411–6422. doi: 10.3390/molecules15096411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Khademian R., Karimzadeh F., Moradi P., Asghari B. Anti-microbial Properties of Valeriana officinalis, Satureja bachtiarica and Thymus daenensis Methanolic Extracts against Helicobacter pylori. J. Pharm. Res. Int. 2019;26:1–7. doi: 10.9734/jpri/2019/v26i630153. [DOI] [Google Scholar]
- 109.Holubec H., Baines A.T., Bayse J.L., Waite S., Bhattacharyya A.K., Clark L.C., Payne C.M., Earnest D.L., Nelson M.A. Chemoprevention of colon carcinogenesis by dietary selenomethionine. Gastroenterology. 1998;114:A612. [Google Scholar]
- 110.Trombetta D., Castelli F., Sarpietro M.G., Venuti V., Cristani M., Daniele C., Saija A., Mazzanti G., Bisignano G. Mechanisms of antibacterial action of three monoterpenes. Antimicrob. Agents Chemother. 2005;49:2474–2478. doi: 10.1128/AAC.49.6.2474-2478.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Khan M.T.H., Ather A., Thompson K.D., Gambari R. Extracts and molecules from medicinal plants against herpes simplex viruses. Antivir. Res. 2005;67:107–119. doi: 10.1016/j.antiviral.2005.05.002. [DOI] [PubMed] [Google Scholar]
- 112.Peana A.T., D’Aquila P.S., Panin F., Serra G., Pippia P., Moretti M.D.L. Anti-inflammatory activity of linalool and linalyl acetate constituents of essential oils. Phytomedicine. 2002;9:721–726. doi: 10.1078/094471102321621322. [DOI] [PubMed] [Google Scholar]
- 113.Ziegler H.L., Jensen T.H., Christensen J., Stærk D., Hägerstrand H., Sittie A.A., Olsen C.E., Staalsø T., Ekpe P., Jaroszewski J.W. Possible artefacts in the in vitro determination of antimalarial activity of natural products that incorporate into lipid bilayer: Apparent antiplasmodial activity of dehydroabietinol, a constituent of Hyptis suaveolens. Planta Med. 2002;68:547–549. doi: 10.1055/s-2002-32548. [DOI] [PubMed] [Google Scholar]
- 114.Sienkiewicz M., Głowacka A., Poznańska-Kurowska K., Kaszuba A., Urbaniak A., Kowalczyk E. The effect of clary sage oil on staphylococci responsible for wound infections. Postep. Dermatol. I Alergol. 2015;32:21–26. doi: 10.5114/pdia.2014.40957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Fujita K.I., Chavasiri W., Kubo I. Anti-Salmonella Activity of Volatile Compounds of Vietnam Coriander. Phyther. Res. 2015;29:1081–1087. doi: 10.1002/ptr.5351. [DOI] [PubMed] [Google Scholar]
- 116.Wan J., Wilcock A., Coventry M.J. The effect of essential oils of basil on the growth of Aeromonas hydrophila and Pseudomonas fluorescens. J. Appl. Microbiol. 1998;84:152–158. doi: 10.1046/j.1365-2672.1998.00338.x. [DOI] [PubMed] [Google Scholar]
- 117.Takarada K., Kimizuka R., Takahashi N., Honma K., Okuda K., Kato T. A comparison of the antibacterial efficacies of essential oils against oral pathogens. Oral Microbiol. Immunol. 2004;19:61–64. doi: 10.1046/j.0902-0055.2003.00111.x. [DOI] [PubMed] [Google Scholar]
- 118.Hammer K.A., Dry L., Johnson M., Michalak E.M., Carson C.F., Riley T.V. Susceptibility of oral bacteria to Melaleuca alternifolia (tea tree) oil in vitro. Oral Microbiol. Immunol. 2003;18:389–392. doi: 10.1046/j.0902-0055.2003.00105.x. [DOI] [PubMed] [Google Scholar]
- 119.Hammer K.A., Carson C.F., Riley T.V. Antifungal activity of the components of Melaleuca alternifolia (tea tree) oil. J. Appl. Microbiol. 2003;95:853–860. doi: 10.1046/j.1365-2672.2003.02059.x. [DOI] [PubMed] [Google Scholar]
- 120.Deans S.G., Ritchie G. Antibacterial properties of plant essential oils. Int. J. Food Microbiol. 1987;5:165–180. doi: 10.1016/0168-1605(87)90034-1. [DOI] [Google Scholar]
- 121.Samber N., Khan A., Varma A., Manzoor N. Synergistic anti-candidal activity and mode of action of Mentha piperita essential oil and its major components. Pharm. Biol. 2015;53:1496–1504. doi: 10.3109/13880209.2014.989623. [DOI] [PubMed] [Google Scholar]
- 122.Mejía-Garibay B., Palou E., López-Malo A. Composition, Diffusion, and Antifungal Activity of Black Mustard (Brassica nigra) Essential Oil When Applied by Direct Addition or Vapor Phase Contact. J. Food Prot. 2015;78:843–848. doi: 10.4315/0362-028X.JFP-14-485. [DOI] [PubMed] [Google Scholar]
- 123.Prakash B., Singh P., Goni R., Raina A.K.P., Dubey N.K. Efficacy of Angelica archangelica essential oil, phenyl ethyl alcohol and α- terpineol against isolated molds from walnut and their antiaflatoxigenic and antioxidant activity. J. Food Sci. Technol. 2015;52:2220–2228. doi: 10.1007/s13197-014-1278-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Trindade L.A., de Araújo Oliveira J., de Castro R.D., de Oliveira Lima E. Inhibition of adherence of C. albicans to dental implants and cover screws by Cymbopogon nardus essential oil and citronellal. Clin. Oral Investig. 2015;19:2223–2231. doi: 10.1007/s00784-015-1450-3. [DOI] [PubMed] [Google Scholar]
- 125.Barkatullah, Ibrar M., Muhammad N., De Feo V. Chemical composition and biological activities of the essential oil of Skimmia laureola leaves. Molecules. 2015;20:4735–4745. doi: 10.3390/molecules20034735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Mahboubi M., Kazempour N. The antifungal activity of Artemisia sieberi essential oil from different localities of Iran against dermatophyte fungi. J. Mycol. Med. 2015;25:65–71. doi: 10.1016/j.mycmed.2015.02.042. [DOI] [PubMed] [Google Scholar]
- 127.Baratta M.T., Damien H.J., Deans S.G., Biondi D.M., Ruberto G. Chemical composition, antimicrobial and antioxidative activity of laurel, sage, rosemary, oregano and coriander essential oils. J. Essent. Oil Res. 1998;10:618–627. doi: 10.1080/10412905.1998.9700989. [DOI] [Google Scholar]
- 128.Gkinis G., Tzakou O., Iliopoulou D., Roussis V. Chemical Composition and Biological Activity of Nepeta parnassica Oils and Isolated Nepetalactones. Z. Für Nat. C. 2003;58:681–686. doi: 10.1515/znc-2003-9-1015. [DOI] [PubMed] [Google Scholar]
- 129.Grunfeld E., Gresty M.A. Relationship between motion sickness, migraine and menstruation in crew members of a “round the world” yacht race. Brain Res. Bull. 1998;47:433–436. doi: 10.1016/S0361-9230(98)00099-9. [DOI] [PubMed] [Google Scholar]
- 130.Howes M.-J.R., Houghton P.J., Barlow D.J., Pocock V.J., Milligan S.R. Assessment of estrogenic activity in some common essential oil constituents. J. Pharm. Pharmacol. 2010;54:1521–1528. doi: 10.1211/002235702216. [DOI] [PubMed] [Google Scholar]
- 131.Ali B., Al-Wabel N.A., Shams S., Ahamad A., Khan S.A., Anwar F. Essential oils used in aromatherapy: A systemic review. Asian Pac. J. Trop. Biomed. 2015;5:601–611. doi: 10.1016/j.apjtb.2015.05.007. [DOI] [Google Scholar]
- 132.Bhalla Y., Gupta V.K., Jaitak V. Anticancer activity of essential oils: A review. J. Sci. Food Agric. 2013;93:3643–3653. doi: 10.1002/jsfa.6267. [DOI] [PubMed] [Google Scholar]
- 133.Edris A.E. Pharmaceutical and Therapeutic Potentials of Essential Oils and Their Individual Volatile Constituents: A Review. Phyther. Res. 2007;21:308–323. doi: 10.1002/ptr.2072. [DOI] [PubMed] [Google Scholar]
- 134.Jaganathan S.K., Supriyanto E. Antiproliferative and Molecular Mechanism of Eugenol-Induced Apoptosis in Cancer Cells. Molecules. 2012;17:6290–6304. doi: 10.3390/molecules17066290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Gupta S., Khan S., Muzafar M., Kushwaha M., Yadav A.K., Gupta A.P. Encapsulation: Entrapping essential oil/flavors/aromas in food. In: Grumezescu A.M., editor. Encapsulations. Elsevier; Amsterdam, The Netherlands: 2016. pp. 229–268. [Google Scholar]
- 136.Reis D.R., Ambrosi A., Luccio M. Di Encapsulated essential oils: A perspective in food preservation. Future Foods. 2022 doi: 10.1016/j.fufo.2022.100126. [DOI] [Google Scholar]
- 137.Ribeiro-Santos R., Andrade M., Sanches-Silva A. Application of encapsulated essential oils as antimicrobial agents in food packaging. Curr. Opin. Food Sci. 2017;14:78–84. doi: 10.1016/j.cofs.2017.01.012. [DOI] [Google Scholar]
- 138.Zinoviadou K.G., Koutsoumanis K.P., Biliaderis C.G. Physico-chemical properties of whey protein isolate films containing oregano oil and their antimicrobial action against spoilage flora of fresh beef. Meat Sci. 2009;82:338–345. doi: 10.1016/j.meatsci.2009.02.004. [DOI] [PubMed] [Google Scholar]
- 139.Suppakul P., Sonneveld K., Bigger S.W., Miltz J. Efficacy of polyethylene-based antimicrobial films containing principal constituents of basil. LWT—Food Sci. Technol. 2008;41:779–788. doi: 10.1016/j.lwt.2007.06.006. [DOI] [Google Scholar]
- 140.Vu K.D., Hollingsworth R.G., Leroux E., Salmieri S., Lacroix M. Development of edible bioactive coating based on modified chitosan for increasing the shelf life of strawberries. Food Res. Int. 2011;44:198–203. doi: 10.1016/j.foodres.2010.10.037. [DOI] [Google Scholar]
- 141.De Melo A.A.M., Geraldine R.M., Silveira M.F.A., Lopes M.C., Silva C., Fernandes T.H., Oliveira A.N. De Microbiological Quality and Other Characteristics of Refrigerated Chicken Meat in Contact with Cellulose Acetate-based Film Incorporated with Rosemary Essential Oil. Braz. J. Microbiol. 2012;16:1419–1427. doi: 10.1590/S1517-83822012000400025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Souza A.C., Goto G.E.O., Mainardi J.A., Coelho A.C.V., Tadini C.C. Cassava starch composite films incorporated with cinnamon essential oil: Antimicrobial activity, microstructure, mechanical and barrier properties. LWT—Food Sci. Technol. 2013;54:346–352. doi: 10.1016/j.lwt.2013.06.017. [DOI] [Google Scholar]
- 143.Aburjai T., Natsheh F.M. Plants Used in Cosmetics. Phyther. Res. 2003;17:987–1000. doi: 10.1002/ptr.1363. [DOI] [PubMed] [Google Scholar]
- 144.Minozzo M., Steffens J., Backes G.T., Paroul N., Cansian R.L. Biological potential and microencapsulation of Cinnamomum cassia essential oil as an alternative for pest control in stored maize. Res. Soc. Dev. 2021;10:e530101422334. doi: 10.33448/rsd-v10i14.22334. [DOI] [Google Scholar]
- 145.Malešević V.K., Vaštag Ž., Radulović-Popović L., Senka M.P., Periĉin-Starĉević I. Microencapsulation technology and essential oil pesticides for food plant production. In: Preedy V.R., editor. Essential Oils in Food Preservation, Flavor and Safety. Academic Press; Cambridge, MA, USA: 2016. pp. 123–129. [Google Scholar]
- 146.Govindarajan M., Sivakumar R. Laboratory evaluation of Indian medicinal plants as repellents against malaria, dengue, and filariasis vector mosquitoes. Parasitol. Res. 2015;114:601–612. doi: 10.1007/s00436-014-4222-0. [DOI] [PubMed] [Google Scholar]
- 147.Khanna S., Chakraborty J.N. Mosquito repellent activity of cotton functionalized with inclusion complexes of β-cyclodextrin citrate and essential oils. Fash. Text. 2018;5:1–18. doi: 10.1186/s40691-017-0125-x. [DOI] [Google Scholar]
- 148.Soroh A., Owen L., Rahim N., Masania J., Abioye A., Qutachi O., Goodyer L., Shen J., Laird K. Microemulsification of essential oils for the development of antimicrobial and mosquito repellent functional coatings for textiles. J. Appl. Microbiol. 2021;131:2808–2820. doi: 10.1111/jam.15157. [DOI] [PubMed] [Google Scholar]
- 149.Bezerra F.M., Carmona O.G., Carmona C.G., Lis M.J., de Moraes F.F. Controlled release of microencapsulated citronella essential oil on cotton and polyester matrices. Cellulose. 2016;23:1459–1470. doi: 10.1007/s10570-016-0882-5. [DOI] [Google Scholar]
- 150.Sariişik M., Kartal G.E., Erkan G., Etkeser S. Alternative methods for transferring mosquito repellent capsules containing bio-based citronella oil to upholstery fabrics: Coating and printing. J. Coat. Technol. Res. 2022;19:323–336. doi: 10.1007/s11998-021-00529-2. [DOI] [Google Scholar]
- 151.Singh M.N., Hemant K.S.Y., Ram M., Shivakumar H.G. Microencapsulation: A promising technique for controlled drug delivery. Res. Pharm. Sci. 2010;5:65–77. [PMC free article] [PubMed] [Google Scholar]
- 152.Gonçalves A., Estevinho B.N., Rocha F. Microencapsulation of vitamin A: A review. Trends Food Sci. Technol. 2016;51:76–87. doi: 10.1016/j.tifs.2016.03.001. [DOI] [Google Scholar]
- 153.Dubey R., Shami T.C., Rao K.U.B. Microencapsulation Technology and Applications. Def. Sci. J. 2009;59:82–95. doi: 10.14429/dsj.59.1489. [DOI] [Google Scholar]
- 154.Paulo F., Santos L. Design of experiments for microencapsulation applications: A review. Mater. Sci. Eng. C. 2017;77:1327–1340. doi: 10.1016/j.msec.2017.03.219. [DOI] [PubMed] [Google Scholar]
- 155.Mehta N., Kumar P., Verma A.K., Umaraw P., Kumar Y., Malav O.P., Sazili A.Q., Lorenzo J.M. Microencapsulation as a Noble Technique for the Application of Bioactive Compounds in the Food Industry: A Comprehensive Review. Appl. Sci. 2022;12:1424. doi: 10.3390/app12031424. [DOI] [Google Scholar]
- 156.Nesterenko A., Alric I., Silvestre F., Durrieu V. Vegetable proteins in microencapsulation: A review of recent interventions and their effectiveness. Ind. Crops Prod. 2013;42:469–479. doi: 10.1016/j.indcrop.2012.06.035. [DOI] [Google Scholar]
- 157.Petrusic S., Koncar V. Controlled release of active agents from microcapsules embedded in textile structures. In: Koncar V., editor. Smart Textiles and Their Applications. Elsevier; Amsterdam, The Netherlands: 2016. pp. 89–114. [Google Scholar]
- 158.Zhang Y., Rochefort D. Characterisation and applications of microcapsules obtained by interfacial polycondensation. J. Microencapsul. 2012;29:636–649. doi: 10.3109/02652048.2012.676092. [DOI] [PubMed] [Google Scholar]
- 159.Hoyos-Leyva J.D., Bello-Pérez L.A., Alvarez-Ramirez J., Garcia H.S. Microencapsulation using starch as wall material: A review. Food Rev. Int. 2018;34:148–161. doi: 10.1080/87559129.2016.1261298. [DOI] [Google Scholar]
- 160.Jamekhorshid A., Sadrameli S.M., Farid M. A review of microencapsulation methods of phase change materials (PCMs) as a thermal energy storage (TES) medium. Renew. Sustain. Energy Rev. 2014;31:531–542. doi: 10.1016/j.rser.2013.12.033. [DOI] [Google Scholar]
- 161.Fang Z., Bhandari B. Encapsulation of polyphenols—A review. Trends Food Sci. Technol. 2010;21:510–523. doi: 10.1016/j.tifs.2010.08.003. [DOI] [Google Scholar]
- 162.Mcclements D.J., Decker E.A., Park Y., Weiss J. Structural design principles for delivery of bioactive components in nutraceuticals and functional foods. Crit. Rev. Food Sci. Nutr. 2009;49:577–606. doi: 10.1080/10408390902841529. [DOI] [PubMed] [Google Scholar]
- 163.Augustin M.A., Sanguansri L., Bode O. Maillard reaction products as encapsulants for fish oil powders. J. Food Sci. 2006;71:E25–E32. doi: 10.1111/j.1365-2621.2006.tb08893.x. [DOI] [Google Scholar]
- 164.Li M., Rouaud O., Poncelet D. Microencapsulation by solvent evaporation: State of the art for process engineering approaches. Int. J. Pharm. 2008;363:26–39. doi: 10.1016/j.ijpharm.2008.07.018. [DOI] [PubMed] [Google Scholar]
- 165.O’Donnell P.B., McGinity J.W. Preparation of microspheres by the solvent evaporation technique. Adv. Drug Deliv. Rev. 1997;28:25–42. doi: 10.1016/S0169-409X(97)00049-5. [DOI] [PubMed] [Google Scholar]
- 166.Bakry A.M., Abbas S., Ali B., Majeed H., Abouelwafa M.Y., Mousa A., Liang L. Microencapsulation of Oils: A Comprehensive Review of Benefits, Techniques, and Applications. Compr. Rev. Food Sci. Food Saf. 2016;15:143–182. doi: 10.1111/1541-4337.12179. [DOI] [PubMed] [Google Scholar]
- 167.Timilsena Y.P., Akanbi T.O., Khalid N., Adhikari B., Barrow C.J. Complex coacervation: Principles, mechanisms and applications in microencapsulation. Int. J. Biol. Macromol. 2019;121:1276–1286. doi: 10.1016/j.ijbiomac.2018.10.144. [DOI] [PubMed] [Google Scholar]
- 168.Hwang J., Kim J., Wee Y., Yun J., Jang H., Kim S., Ryu H. Preparation and characterization of melamine-formaldehyde resin microcapsules containing fragrant oil. Biotechnol. Bioprocess Eng. 2006;11:332–336. doi: 10.1007/BF03026249. [DOI] [Google Scholar]
- 169.Chung S.K., Seo J.Y., Lim J.H., Park H.H., Yea M.J., Park H.J. Microencapsulation of essential oil for insect repellent in food packaging system. J. Food Sci. 2013;78:E709–E714. doi: 10.1111/1750-3841.12111. [DOI] [PubMed] [Google Scholar]
- 170.Sánchez-Navarro M.M., Cuesta-Garrote N., Arán-Áis F., Orgilés-Barceló C. Microencapsulation of melaleuca alternifolia (tea tree) oil as biocide for footwear applications. J. Dispers. Sci. Technol. 2011;32:1722–1727. doi: 10.1080/01932691.2011.616126. [DOI] [Google Scholar]
- 171.Long Y., York D., Zhang Z., Preece J.A. Microcapsules with low content of formaldehyde: Preparation and characterization. J. Mater. Chem. 2009;19:6882–6887. doi: 10.1039/b902832c. [DOI] [Google Scholar]
- 172.Bansode S.S., Banarjee S.K., Gaikwad D.D., Jadhav S.L., Thorat R.M. Microencapsulation: A review. Int. J. Pharm. Sci. Rev. Res. 2010;1:38–43. [Google Scholar]
- 173.Lam P.L., Gambari R. Advanced progress of microencapsulation technologies: In vivo and in vitro models for studying oral and transdermal drug deliveries. J. Control. Release. 2014;178:25–45. doi: 10.1016/j.jconrel.2013.12.028. [DOI] [PubMed] [Google Scholar]
- 174.Huang H.J., Chen X.D., Yuan W.K. Microencapsulation based on emulsification for producing pharmaceutical products: A literature review. Dev. Chem. Eng. Miner. Process. 2006;14:515–544. doi: 10.1002/apj.5500140318. [DOI] [Google Scholar]
- 175.Estevinho B.N., Rocha F., Santos L., Alves A. Microencapsulation with chitosan by spray drying for industry applications—A review. Trends Food Sci. Technol. 2013;31:138–155. doi: 10.1016/j.tifs.2013.04.001. [DOI] [Google Scholar]
- 176.Gharsallaoui A., Roudaut G., Chambin O., Voilley A., Saurel R. Applications of spray-drying in microencapsulation of food ingredients: An overview. Food Res. Int. 2007;40:1107–1121. doi: 10.1016/j.foodres.2007.07.004. [DOI] [Google Scholar]
- 177.Mohammed N.K., Tan C.P., Manap Y.A., Muhialdin B.J., Hussin A.S.M. Spray Drying for the Encapsulation of Oils—A Review. Molecules. 2020;25:3873. doi: 10.3390/molecules25173873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Hazarika U., Gosztola B. Lyophilization and its Effects on the Essential Oil Content and Composition of Herbs and Spices—A Review. Acta Sci. Pol. Technol. Aliment. 2020;19:467–473. doi: 10.17306/J.AFS.2020.0853. [DOI] [PubMed] [Google Scholar]
- 179.Gardeli C., Evageliou V., Poulos C., Yanniotis S., Komaitis M. Drying of fennel plants: Oven, freeze drying, effect of freeze-drying time, and use of biopolymers. Dry. Technol. 2010;28:542–549. doi: 10.1080/07373931003622321. [DOI] [Google Scholar]
- 180.Vujanović M.D., Đurović S.D., Radojković M.M. Chemical Composition of Essential Oils of Elderberry (Sambucus Nigra L.) Flowers and Fruits. Acta Period. Technol. 2021;52:229–237. doi: 10.2298/APT2152229V. [DOI] [Google Scholar]
- 181.de Araújo J.S.F., de Souza E.L., Oliveira J.R., Gomes A.C.A., Kotzebue L.R.V., da Silva Agostini D.L., de Oliveira D.L.V., Mazzetto S.E., da Silva A.L., Cavalcanti M.T. Microencapsulation of sweet orange essential oil (Citrus aurantium var. dulcis) by liophylization using maltodextrin and maltodextrin/gelatin mixtures: Preparation, characterization, antimicrobial and antioxidant activities. Int. J. Biol. Macromol. 2020;143:991–999. doi: 10.1016/j.ijbiomac.2019.09.160. [DOI] [PubMed] [Google Scholar]
- 182.Sebaaly C., Greige-Gerges H., Stainmesse S., Fessi H., Charcosset C. Effect of composition, hydrogenation of phospholipids and lyophilization on the characteristics of eugenol-loaded liposomes prepared by ethanol injection method. Food Biosci. 2016;15:1–10. doi: 10.1016/j.fbio.2016.04.005. [DOI] [Google Scholar]
- 183.Cocero M.J., Martín Á., Mattea F., Varona S. Encapsulation and co-precipitation processes with supercritical fluids: Fundamentals and applications. J. Supercrit. Fluids. 2009;47:546–555. doi: 10.1016/j.supflu.2008.08.015. [DOI] [Google Scholar]
- 184.Jyothi N.V.N., Prasanna P.M., Sakarkar S.N., Prabha K.S., Ramaiah P.S., Srawan G.Y. Microencapsulation techniques, factors influencing encapsulation efficiency. J. Microencapsul. 2010;27:187–197. doi: 10.3109/02652040903131301. [DOI] [PubMed] [Google Scholar]
- 185.Zhang S., Kawakami K., Yamamoto M., Masaoka Y., Kataoka M., Yamashita S., Sakuma S. Coaxial electrospray formulations for improving oral absorption of a poorly water-soluble drug. Mol. Pharm. 2011;8:807–813. doi: 10.1021/mp100401d. [DOI] [PubMed] [Google Scholar]
- 186.Koo S.Y., Cha K.H., Song D.G., Chung D., Pan C.H. Microencapsulation of peppermint oil in an alginate-pectin matrix using a coaxial electrospray system. Int. J. Food Sci. Technol. 2014;49:733–739. doi: 10.1111/ijfs.12358. [DOI] [Google Scholar]
- 187.Zhang L., Huang J., Si T., Xu R.X. Coaxial electrospray of microparticles and nanoparticles for biomedical applications. Expert Rev. Med. Devices. 2012;9:595–612. doi: 10.1586/erd.12.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Lastow O., Balachandran W. Numerical simulation of electrohydrodynamic (EHD) atomization. J. Electrostat. 2006;64:850–859. doi: 10.1016/j.elstat.2006.02.006. [DOI] [Google Scholar]
- 189.Anwar S.H., Kunz B. The influence of drying methods on the stabilization of fish oil microcapsules: Comparison of spray granulation, spray drying, and freeze drying. J. Food Eng. 2011;105:367–378. doi: 10.1016/j.jfoodeng.2011.02.047. [DOI] [Google Scholar]
- 190.Teunou E., Poncelet D. Fluid-Bed Coating. In: Onwulata C., editor. Encapsulated and Powdered Foods. Taylor & Francis; Abingdon, UK: 2005. p. 18. [Google Scholar]
- 191.Desai K.G.H., Jin Park H. Recent developments in microencapsulation of food ingredients. Dry. Technol. 2005;23:1361–1394. doi: 10.1081/DRT-200063478. [DOI] [Google Scholar]
- 192.Sadovoy A.V., Lomova M.V., Antipina M.N., Braun N.A., Sukhorukov G.B., Kiryukhin M.V. Layer-by-layer assembled multilayer shells for encapsulation and release of fragrance. ACS Appl. Mater. Interfaces. 2013;5:8948–8954. doi: 10.1021/am401871u. [DOI] [PubMed] [Google Scholar]
- 193.Xie Y.L., Wang M.J., Yao S.J. Preparation and characterization of biocompatible microcapsules of sodium cellulose sulfate/chitosan by means of layer-by-layer self-assembly. Langmuir. 2009;25:8999–9005. doi: 10.1021/la9014338. [DOI] [PubMed] [Google Scholar]
- 194.Ge L., Tan X., Sheng R., Xiao J. Layer-by-layer self-assembly of giant polyelectrolyte microcapsules templated by microbubbles as potential hydrophilic or hydrophobic drug delivery system. Colloids Interface Sci. Commun. 2022;47 doi: 10.1016/j.colcom.2022.100603. [DOI] [Google Scholar]
- 195.Zhang Z., Zhang S., Su R., Xiong D., Feng W., Chen J. Controlled Release Mechanism and Antibacterial Effect of Layer-By-Layer Self-Assembly Thyme Oil Microcapsule. J. Food Sci. 2019;84:1427–1438. doi: 10.1111/1750-3841.14610. [DOI] [PubMed] [Google Scholar]
- 196.Pan H.M., Subramanian A., Ochs C.J., Dewavrin J.Y., Beyer S., Trau D.W. Edible polyelectrolyte microcapsules with water-soluble cargo assembled in organic phase. RSC Adv. 2014;4:35163–35166. doi: 10.1039/C4RA04750H. [DOI] [Google Scholar]
- 197.Verma G., Hassan P.A. Self assembled materials: Design strategies and drug delivery perspectives. Phys. Chem. Chem. Phys. 2013;15:17016–17028. doi: 10.1039/c3cp51207j. [DOI] [PubMed] [Google Scholar]
- 198.Aguiar M.C.S., das Graças Fernandes da Silva M.F., Fernandes J.B., Forim M.R. Evaluation of the microencapsulation of orange essential oil in biopolymers by using a spray-drying process. Sci. Rep. 2020;10:1–11. doi: 10.1038/s41598-020-68823-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Martins I.M., Barreiro M.F., Coelho M., Rodrigues A.E. Microencapsulation of essential oils with biodegradable polymeric carriers for cosmetic applications. Chem. Eng. J. 2014;245:191–200. doi: 10.1016/j.cej.2014.02.024. [DOI] [Google Scholar]
- 200.Scarfato P., Avallone E., Iannelli P., De Feo V., Acierno D. Synthesis and characterization of polyurea microcapsules containing essential oils with antigerminative activity. J. Appl. Polym. Sci. 2007;105:3568–3577. doi: 10.1002/app.26420. [DOI] [Google Scholar]
- 201.Bagle A.V., Jadhav R.S., Gite V.V., Hundiwale D.G., Mahulikar P.P. Controlled release study of phenol formaldehyde microcapsules containing neem oil as an insecticide. Int. J. Polym. Mater. Polym. Biomater. 2013;62:421–425. doi: 10.1080/00914037.2012.719142. [DOI] [Google Scholar]
- 202.Ahsaei S.M., Rodríguez-Rojo S., Salgado M., Cocero M.J., Talebi-Jahromi K., Amoabediny G. Insecticidal activity of spray dried microencapsulated essential oils of Rosmarinus officinalis and Zataria multiflora against Tribolium confusum. Crop Prot. 2020;128:104996. doi: 10.1016/j.cropro.2019.104996. [DOI] [Google Scholar]
- 203.Fernandes R.V.D.B., Borges S.V., Botrel D.A. Gum arabic/starch/maltodextrin/inulin as wall materials on the microencapsulation of rosemary essential oil. Carbohydr. Polym. 2014;101:524–532. doi: 10.1016/j.carbpol.2013.09.083. [DOI] [PubMed] [Google Scholar]
- 204.Chitprasert P., Sutaphanit P. Holy basil (ocimum sanctum linn.) Essential oil delivery to swine gastrointestinal tract using gelatin microcapsules coated with aluminum carboxymethyl cellulose and beeswax. J. Agric. Food Chem. 2014;62:12641–12648. doi: 10.1021/jf5019438. [DOI] [PubMed] [Google Scholar]
- 205.Munhuweyi K., Caleb O.J., van Reenen A.J., Opara U.L. Physical and antifungal properties of β-cyclodextrin microcapsules and nanofibre films containing cinnamon and oregano essential oils. LWT—Food Sci. Technol. 2018;87:413–422. doi: 10.1016/j.lwt.2017.09.012. [DOI] [Google Scholar]
- 206.Leimann F.V., Gonçalves O.H., Machado R.A.F., Bolzan A. Antimicrobial activity of microencapsulated lemongrass essential oil and the effect of experimental parameters on microcapsules size and morphology. Mater. Sci. Eng. C. 2009;29:430–436. doi: 10.1016/j.msec.2008.08.025. [DOI] [Google Scholar]
- 207.Li R., Jiang S., Jiang Z.T. β-Cyclodextrin supramolecular microcapsules and antioxidant activity of Laurus nobilis essential oil. J. Essent. Oil-Bear. Plants. 2017;20:1511–1524. doi: 10.1080/0972060X.2017.1354724. [DOI] [Google Scholar]
- 208.Li Y., Wu C., Wu T., Wang L., Chen S., Ding T., Hu Y. Preparation and characterization of citrus essential oils loaded in chitosan microcapsules by using different emulsifiers. J. Food Eng. 2018;217:108–114. doi: 10.1016/j.jfoodeng.2017.08.026. [DOI] [Google Scholar]
- 209.Mehran M., Masoum S., Memarzadeh M. Microencapsulation of Mentha spicata essential oil by spray drying: Optimization, characterization, release kinetics of essential oil from microcapsules in food models. Ind. Crops Prod. 2020;154:112694. doi: 10.1016/j.indcrop.2020.112694. [DOI] [Google Scholar]
- 210.Adepu S., Khandelwal M. Bacterial cellulose with microencapsulated antifungal essential oils: A novel double barrier release system. Materialia. 2020;9:100585. doi: 10.1016/j.mtla.2020.100585. [DOI] [Google Scholar]
- 211.Ribeiro A.D., Marques J., Forte M., Correia F.C., Parpot P., Oliveira C., Pereira A.I., Andrade L., Azenha C., Mendes A., et al. Microencapsulation of citronella oil for solar-activated controlled release as an insect repellent. Appl. Mater. Today. 2016;5:90–97. doi: 10.1016/j.apmt.2016.09.003. [DOI] [Google Scholar]
- 212.Lai H., Liu Y., Huang G., Chen Y., Song Y., Ma Y.Q., Yue P. Fabrication and antibacterial evaluation of peppermint oil-loaded composite microcapsules by chitosan-decorated silica nanoparticles stabilized Pickering emulsion templating. Int. J. Biol. Macromol. 2021;183:2314–2325. doi: 10.1016/j.ijbiomac.2021.05.198. [DOI] [PubMed] [Google Scholar]
- 213.Tang C., Li Y., Pun J., Mohamed Osman A.S., Tam K.C. Polydopamine microcapsules from cellulose nanocrystal stabilized Pickering emulsions for essential oil and pesticide encapsulation. Colloids Surf. A Physicochem. Eng. Asp. 2019;570:403–413. doi: 10.1016/j.colsurfa.2019.03.049. [DOI] [Google Scholar]
- 214.Cai C., Ma R., Duan M., Lu D. Preparation and antimicrobial activity of thyme essential oil microcapsules prepared with gum arabic. RSC Adv. 2019;9:19740–19747. doi: 10.1039/C9RA03323H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Emadzadeh B., Ghorani B., Naji-Tabasi S., Charpashlo E., Molaveisi M. Fate of β-cyclodextrin-sugar beet pectin microcapsules containing garlic essential oil in an acidic food beverage. Food Biosci. 2021;42:101029. doi: 10.1016/j.fbio.2021.101029. [DOI] [Google Scholar]
- 216.de Medeiros J.A.S., Blick A.P., Galindo M.V., Alvim I.D., Yamashita F., Ueno C.T., Shirai M.A., Grosso C.R.F., Corradini E., Sakanaka L.S. Incorporation of Oregano Essential Oil Microcapsules in Starch-Poly (Butylene Adipate Co-Terephthalate) (PBAT) Films. Macromol. Symp. 2019;383:1800052. doi: 10.1002/masy.201800052. [DOI] [Google Scholar]
- 217.Ngamekaue N., Chitprasert P. Effects of beeswax-carboxymethyl cellulose composite coating on shelf-life stability and intestinal delivery of holy basil essential oil-loaded gelatin microcapsules. Int. J. Biol. Macromol. 2019;135:1088–1097. doi: 10.1016/j.ijbiomac.2019.06.002. [DOI] [PubMed] [Google Scholar]
- 218.De Matos E.F., Scopel B.S., Dettmer A. Citronella essential oil microencapsulation by complex coacervation with leather waste gelatin and sodium alginate. J. Environ. Chem. Eng. 2018;6:1989–1994. doi: 10.1016/j.jece.2018.03.002. [DOI] [Google Scholar]
- 219.Xiao Z., Liu W., Zhu G., Zhou R., Niu Y. Production and characterization of multinuclear microcapsules encapsulating lavender oil by complex coacervation. Flavour Fragr. J. 2014;29:166–172. doi: 10.1002/ffj.3192. [DOI] [Google Scholar]
- 220.Baiocco D., Preece J.A., Zhang Z. Microcapsules with a fungal chitosan-gum Arabic-maltodextrin shell to encapsulate health-beneficial peppermint oil. Food Hydrocoll. Health. 2021;1:100016. doi: 10.1016/j.fhfh.2021.100016. [DOI] [Google Scholar]
- 221.Dima C., Cotârlet M., Alexe P., Dima S. Microencapsulation of essential oil of pimento [Pimenta dioica (L) Merr.] by chitosan/k-carrageenan complex coacervation method. Innov. Food Sci. Emerg. Technol. 2014;22:203–211. doi: 10.1016/j.ifset.2013.12.020. [DOI] [Google Scholar]
- 222.Hernández-Nava R., López-Malo A., Palou E., Ramírez-Corona N., Jiménez-Munguía M.T. Encapsulation of oregano essential oil (Origanum vulgare) by complex coacervation between gelatin and chia mucilage and its properties after spray drying. Food Hydrocoll. 2020;109:106077. doi: 10.1016/j.foodhyd.2020.106077. [DOI] [Google Scholar]
- 223.Qiu L., Zhang M., Adhikari B., Chang L. Microencapsulation of rose essential oil in mung bean protein isolate-apricot peel pectin complex coacervates and characterization of microcapsules. Food Hydrocoll. 2022;124:107366. doi: 10.1016/j.foodhyd.2021.107366. [DOI] [Google Scholar]
- 224.Chen K., Zhang M., Adhikari B., Wang M. Microencapsulation of Sichuan pepper essential oil in soybean protein isolate-Sichuan pepper seed soluble dietary fiber complex coacervates. Food Hydrocoll. 2022;125:107421. doi: 10.1016/j.foodhyd.2021.107421. [DOI] [Google Scholar]
- 225.Rojas-Moreno S., Cárdenas-Bailón F., Osorio-Revilla G., Gallardo-Velázquez T., Proal-Nájera J. Effects of complex coacervation-spray drying and conventional spray drying on the quality of microencapsulated orange essential oil. J. Food Meas. Charact. 2018;12:650–660. doi: 10.1007/s11694-017-9678-z. [DOI] [Google Scholar]
- 226.Li Y., Liu J., He X., Kong D., Zhou C., Wu H., Yang Z., Yang Z., Hu Y. Preparation of Cinnamon Oil-Loaded Antibacterial Composite Microcapsules by In Situ Polymerization of Pickering Emulsion Templates. Macromol. Mater. Eng. 2020;305:1900851. doi: 10.1002/mame.201900851. [DOI] [Google Scholar]
- 227.Revuelta M.V., Bogdan S., Gámez-Espinosa E., Deyá M.C., Romagnoli R. Green antifungal waterborne coating based on essential oil microcapsules. Prog. Org. Coat. 2021;151:106101. doi: 10.1016/j.porgcoat.2020.106101. [DOI] [Google Scholar]
- 228.Matulyte I., Kasparaviciene G., Bernatoniene J. Development of new formula microcapsules from nutmeg essential oil using sucrose esters and magnesium aluminometasilicate. Pharmaceutics. 2020;12:628. doi: 10.3390/pharmaceutics12070628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Dolçà C., Ferrándiz M., Capablanca L., Franco E., Mira E., López F., García D. Microencapsulation of Rosemary Essential Oil by Co-Extrusion/Gelling Using Alginate as a Wall Material. J. Encapsulation Adsorpt. Sci. 2015;5:121–130. doi: 10.4236/jeas.2015.53010. [DOI] [Google Scholar]
- 230.Yingngam B., Kacha W., Rungseevijitprapa W., Sudta P., Prasitpuriprecha C., Brantner A. Response surface optimization of spray-dried citronella oil microcapsules with reduced volatility and irritation for cosmetic textile uses. Powder Technol. 2019;355:372–385. doi: 10.1016/j.powtec.2019.07.065. [DOI] [Google Scholar]
- 231.Maulidna, Wirjosentono B., Tamrin, Marpaung L. Microencapsulation of ginger-based essential oil (Zingiber cassumunar roxb) with chitosan and oil palm trunk waste fiber prepared by spray-drying method. Case Stud. Therm. Eng. 2020;18:100606. doi: 10.1016/j.csite.2020.100606. [DOI] [Google Scholar]
- 232.Ren W., Tian G., Zhao S., Yang Y., Gao W., Zhao C., Zhang H., Lian Y., Wang F., Du H., et al. Effects of spray-drying temperature on the physicochemical properties and polymethoxyflavone loading efficiency of citrus oil microcapsules. LWT—Food Sci. Technol. 2020;133:109954. doi: 10.1016/j.lwt.2020.109954. [DOI] [Google Scholar]
- 233.Bajac J., Nikolovski B., Lončarević I., Petrović J., Bajac B., Đurović S., Petrović L. Microencapsulation of juniper berry essential oil (Juniperus communis L.) by spray drying: Microcapsule characterization and release kinetics of the oil. Food Hydrocoll. 2022;125:107430. doi: 10.1016/j.foodhyd.2021.107430. [DOI] [Google Scholar]
- 234.Hu Q., Li X., Chen F., Wan R., Yu C.W., Li J., McClements D.J., Deng Z. Microencapsulation of an essential oil (cinnamon oil) by spray drying: Effects of wall materials and storage conditions on microcapsule properties. J. Food Process. Preserv. 2020;44:e14805. doi: 10.1111/jfpp.14805. [DOI] [Google Scholar]
- 235.Asensio C.M., Paredes A.J., Martin M.P., Allemandi D.A., Nepote V., Grosso N.R. Antioxidant Stability Study of Oregano Essential Oil Microcapsules Prepared by Spray-Drying. J. Food Sci. 2017;82:2864–2872. doi: 10.1111/1750-3841.13951. [DOI] [PubMed] [Google Scholar]
- 236.Shaikh J., Bhosale R., Singhal R. Microencapsulation of black pepper oleoresin. Food Chem. 2006;94:105–110. doi: 10.1016/j.foodchem.2004.10.056. [DOI] [Google Scholar]
- 237.Ozdemir N., Bayrak A., Tat T., Yanık Z.N., Altay F., Halkman A.K. Fabrication and characterization of basil essential oil microcapsule-enriched mayonnaise and its antimicrobial properties against Escherichia coli and Salmonella Typhimurium. Food Chem. 2021;359:129940. doi: 10.1016/j.foodchem.2021.129940. [DOI] [PubMed] [Google Scholar]
- 238.Qi Z., Zhou H., Xue X., Zhang C., Chen H., Yuan H., Wang C. Fabrication of epoxy sustained-release coatings loaded with urushiol microcapsules containing essential oil for inhibition on drug-resistant Helicobacter pylori and Staphylococcus aureus. Prog. Org. Coat. 2021;161:106459. doi: 10.1016/j.porgcoat.2021.106459. [DOI] [Google Scholar]
- 239.Yang Y.H., Li X.Z., Zhang S. Preparation methods and release kinetics of: Litsea cubeba essential oil microcapsules. RSC Adv. 2018;8:29980–29987. doi: 10.1039/C8RA05769A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Martins W.S., de Araújo J.S.F., Feitosa B.F., Oliveira J.R., Kotzebue L.R.V., Agostinic D.L.S., Oliveira D.L.V., Mazzetto S.E., Cavalcanti M.T., daSilva A.L. Lemongrass (Cymbopogon citratus DC. Stapf) essential oil microparticles: Development, characterization, and antioxidant potential. Food Chem. 2021;355:129644. doi: 10.1016/j.foodchem.2021.129644. [DOI] [PubMed] [Google Scholar]
- 241.Ocak B. Gum arabic and collagen hydrolysate extracted from hide fleshing wastes as novel wall materials for microencapsulation of Origanum onites L. essential oil through complex coacervation. Environ. Sci. Pollut. Res. 2020;27:42727–42737. doi: 10.1007/s11356-020-10201-8. [DOI] [PubMed] [Google Scholar]
- 242.Rojas-Moreno S., Osorio-Revilla G., Gallardo-Velázquez T., Cárdenas-Bailón F., Meza-Márquez G. Effect of the cross-linking agent and drying method on encapsulation efficiency of orange essential oil by complex coacervation using whey protein isolate with different polysaccharides. J. Microencapsul. 2018;35:165–180. doi: 10.1080/02652048.2018.1449910. [DOI] [PubMed] [Google Scholar]
- 243.Almeida A.P., Rodríguez-Rojo S., Serra A.T., Vila-Real H., Simplicio A.L., Delgadilho I., Beirão Da Costa S., Beirão Da Costa L., Nogueira I.D., Duarte C.M.M. Microencapsulation of oregano essential oil in starch-based materials using supercritical fluid technology. Innov. Food Sci. Emerg. Technol. 2013;20:140–145. doi: 10.1016/j.ifset.2013.07.009. [DOI] [Google Scholar]
- 244.Varona S., Kareth S., Martín Á., Cocero M.J. Formulation of lavandin essential oil with biopolymers by PGSS for application as biocide in ecological agriculture. J. Supercrit. Fluids. 2010;54:369–377. doi: 10.1016/j.supflu.2010.05.019. [DOI] [Google Scholar]
- 245.Machado L.C., Pelegati V.B., Oliveira A.L. Study of simple microparticles formation of limonene in modified starch using PGSS—Particles from gas-saturated suspensions. J. Supercrit. Fluids. 2016;107:260–269. doi: 10.1016/j.supflu.2015.09.023. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The raw/processed data required to reproduce these findings cannot be shared at this time due to technical or time limitations.