As case numbers of coronavirus disease 19 (COVID-19) increase, chronic symptoms, including those of autonomic dysfunction, are being reported with increasing frequency [1], leading to the diagnosis of post-acute sequelae of COVID-19 (PASC), or Long-COVID. In addition, small fiber neuropathy (SFN) has been reported after viral infections, including severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [2]. These associations have prompted our group to systematically perform autonomic testing and skin biopsies in a cohort of patients who have developed postural tachycardia syndrome (POTS) as a consequence of PASC (Long-COVID POTS). As part of this evaluation, all skin biopsy samples undergo immunohistochemical analysis of both intraepidermal nerve fiber density (IENFD) and phosphorylated α-synuclein (p-syn) [3], the pathological form of α-synuclein associated with the neurodegenerative diseases of Parkinson’s disease (PD), dementia with Lewy bodies (DLB), multiple system atrophy (MSA), and pure autonomic failure (PAF), as well as isolated REM sleep behavior disorder (iRBD), a prodromal manifestation of synucleinopathy for the majority of patients.
We report the presence of p-syn in five young patients with Long-COVID POTS (LC-POTS) but without prodromal features of neurodegenerative disease. All patients underwent autonomic reflex testing in our laboratory, including quantitative sudomotor axon reflex testing (QSART), heart rate variability with deep breathing, Valsalva maneuver, and a 10-min head-up tilt (HUT) at an angle of 70°. Beat-to-beat blood pressure (BP) was measured with finger plethysmography and confirmed with an automated cuff sphygmomanometer over the brachial artery. An intravenous catheter was placed, and supine and upright norepinephrine (NE) levels were collected. All pertinent medications were held five half-lives prior to testing. Patients underwent a complete neurological examination and comprehensive olfactory testing with the University of Pennsylvania Smell Identification Test (UPSIT)-40. Skin biopsy was performed at the C7 paraspinal level, distal thigh (10 mm distal to the iliac crest), and distal leg (10 mm proximal to the lateral malleolus). A multinational age- and gender-adjusted dataset was utilized for normative IENFD values [4].
Patient 1
A 19-year-old female with a history of anxiety developed sore throat, headache, congestion, and anosmia and was diagnosed with COVID in November 2020 on the basis of SARS-CoV-2 IgG. One month later, she developed panic attacks with hyperventilation, nausea and vomiting, fatigue despite sleeping 12 h nightly, orthostatic intolerance, and syncope. She denied dream-enacting behaviors or hyposmia. Neurological examination was normal. UPSIT demonstrated mild hyposmia (34/40). HUT demonstrated an exaggerated postural tachycardia with a heart rate (HR) increase of 60 bpm. Norepinephrine (NE) levels increased more than threefold on HUT, indicative of a hyperadrenergic response. Interleukin (IL)-6 levels were mildly elevated at 4.2 (normal < 4.0). The remainder of her laboratory workup, including tumor necrosis factor (TNF)-α, D-dimer, ferritin, creatine kinase (CK), antinuclear antibodies (ANA), erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), antiphospholipid antibodies, and ganglionic acetylcholine receptor antibodies, was normal. Skin biopsy demonstrated p-syn deposition at the distal thigh and distal leg (Fig. 1). IENFD was normal at all sites (Supplementary Table 1).
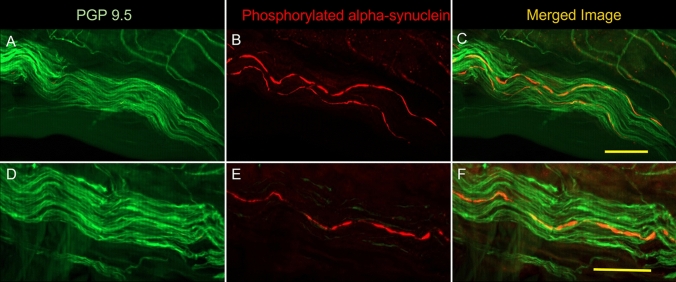
Fig. 1.
Examples of nerve fibers from two patients (A–C and D–F). On the left (A and D), nerve fibers surrounding a blood vessel are shown in green through immunostaining with protein gene product 9.5 (PGP9.5). In B and E, the presence of phosphorylated α-synuclein (P-SYN) is seen in red. In C and F, the merged image reveals the presence of intra-axonal phosphorylated α-synuclein in orange
Patient 2
A 38-year-old male with a history of hypertension developed shortness of breath and tachycardia and was diagnosed with COVID in November 2020 on the basis of nasopharyngeal RT-PCR. Over the next several weeks, he developed patchy subjective anhidrosis, subjective fever and chills, tachycardia, chest pains, orthostatic hypertension, and vasomotor pooling in his upper extremities only. He denied dream-enacting behaviors or hyposmia. Neurological examination was normal. UPSIT demonstrated mild hyposmia (30/40). HUT demonstrated an exaggerated postural tachycardia with an HR increase of 47 bpm. Laboratory workup as detailed above was notable for elevated anticardiolipin IgM antibodies at 13.6 MPL (normal < 12.5 MPL), elevated ferritin at 455 ng/mL (normal 30–400 ng/mL), and a positive ANA at 1:80 (homogeneous pattern). Skin biopsy demonstrated p-syn at the distal thigh and distal leg. IENFD was reduced at the distal thigh and distal leg (Supplementary Table 1).
Patient 3
A 25-year-old female with no past medical history developed shortness of breath, tachycardia, fever, and orthostatic intolerance and syncope and was diagnosed with COVID in October 2020 on the basis of nasopharyngeal RT-PCR. Over the next several weeks, she developed worsening fatigue, heat intolerance, daily headaches, cognitive impairment, and hyperadrenergic surges with elevated BP and HR. She denied dream-enacting behaviors or hyposmia. Neurological examination was normal. UPSIT demonstrated mild hyposmia (31/40). HUT demonstrated an exaggerated postural tachycardia with a HR increase of 69 bpm. Laboratory workup as detailed above was notable for elevated TNF-α at 7.5 pg/mL (normal < 7.2 pg/mL) and prolonged activated partial thromboplastin time (aPTT) 42.7 s (normal 25.4–36.9 s). Skin biopsy demonstrated p-syn at C7 and the distal leg. IENFD was reduced at the distal leg (Supplementary Table 1).
Patient 4
A 43-year-old female with a history of psoriatic arthritis, fibromyalgia, and migraines developed nausea, vertigo, and anosmia and was diagnosed with COVID in April 2021 on the basis of SARS-CoV-2 IgG. Over the next several weeks, she developed orthostatic intolerance, palpitations, total body pruritis, and alternating constipation and diarrhea. She denied dream-enacting behaviors. Neurological examination was normal. UPSIT was normal. HUT demonstrated an exaggerated postural tachycardia with a HR increase of 32 bpm. Laboratory workup as detailed above was notable for elevated IL-6 at 4.7 pg/mL (normal < 4.0 pg/mL) and anticardiolipin IgM antibodies at 13.8 MPL (normal < 12.5 MPL). Skin biopsy demonstrated p-syn at all sites. IENFD was reduced at the distal thigh and distal leg.
Patient 5
A 41-year-old male with a history of migraines developed cough, anosmia, and extremity tingling and was diagnosed with COVID in May 2020 on the basis of SARS-CoV-2 IgG. Over the next several weeks, he developed orthostatic intolerance, cognitive impairment, and fatigue. He denied dream-enacting behaviors. Neurological examination and UPSIT were normal. HUT demonstrated an exaggerated postural tachycardia with a HR increase of 38 bpm. Laboratory workup as detailed above was notable for elevated D-dimer at 2.26 µg/mL (normal < 0.5 µg/mL). Skin biopsy demonstrated p-syn at C7 and the distal leg. IENFD was normal at all sites.
Discussion
We report the first cases, to our knowledge, of pathological cutaneous p-syn in patients with post-viral autonomic dysfunction after SARS-CoV-2 infection. All of our patients developed POTS after mild-to-moderate COVID-19; however, none of them reported prodromal symptoms of α-synucleinopathy prior to their diagnosis of COVID-19. Therefore, the association of p-syn in these five young adults, while intriguing, is of uncertain significance. Viral infections have been described as a potential trigger of neurodegenerative disease, most notably the post-encephalitic Parkinsonism of encephalitis lethargica after the influenza pandemic of 1918. More recently, several cases of Parkinsonism have been reported after COVID-19 [5]. In addition, a mechanistic study has demonstrated that the SARS-CoV-2 nucleocapsid protein (N-protein) might serve as a nucleus for the aggregation process of α-syn by accelerating amyloid fibril formation [6]. However, it should be noted that the authors of this study utilized a 140C α-synuclein mutation instead of the 129S mutation found in human disease, and a causal relationship between SARS-CoV-2 and neurodegenerative disease has not yet been identified. It should also be noted that our cases are not the first reports of p-syn in patients with POTS. In one small cohort of one of the authors (C.G.), cutaneous p-syn was reported on skin biopsy in 7/22 (32%) of POTS patients prior to the COVID-19 pandemic [7]. Those with a hypertensive response on HUT, as all of our patients had, were more likely to be p-syn positive.
As cutaneous p-syn has demonstrated itself as a highly sensitive and specific marker of the α-synucleinopathies [8], our patients’ results are unlikely to be false positives. Without a comparative biopsy prior to SARS-CoV-2 infection, it is unclear if the presence of p-syn is an incidental and unrelated finding, if the infection triggered accelerated aggregation of p-syn, or if the presence of p-syn predated the infection and predisposed our patients to the development of Long-COVID and/or POTS. One potential theory is that the pre-existing presence of phosphorylated α-synuclein could predispose individuals to more prolonged complications after COVID-19 infection. This would suggest that individuals with PD or other synucleinopathies might be at greater risk of complications [9]. Comparisons with other neurodegenerative diseases with similar morbidity would be required to determine the clinical relevance of this possibility.
In the present case series, our sample size is a significant limitation, as is the lack of follow-up. Thus, caution is paramount in interpreting these findings. At the time of this writing, we have performed skin biopsies on a total of eight patients with LC-POTS, yielding a p-syn positivity rate of 63%. It is interesting to note that 4/5 of our positive patients had evidence of mild hyposmia on olfactory testing, and cutaneous p-syn has been associated with hyposmia in other synuclein cohorts, specifically iRBD [10]. Most of our patients also had evidence of ongoing immunological dysfunction as evidenced by abnormal inflammatory markers, a common finding in Long-COVID [11]. Finally, it should be noted that two of our three patients with reduced IENFD did not describe the typical painful symptoms of SFN. In these patients, it is possible that the small fiber loss observed on microscopic analysis may reflect a reactive process of different pathophysiologic mechanism than that of SFN. This has been previously noted in many of the conditions associated with p-syn deposition, including PD, MSA, and PAF [12]. Our findings should be viewed at this stage as preliminary only, and larger cohorts and longitudinal follow-up of patients with Long-COVID will be necessary to interpret these results more accurately.
Supplementary Information
Below is the link to the electronic supplementary material.
Funding:
This work was supported by grant funding from Dysautonomia International.
Declarations
Conflict of interest
M. G. Miglis reports grant funding from Dysautonomia International, serves on the medical advisory board of Dysautonomia International (unpaid), and reports personal compensation for consulting work with MED-IQ, 2nd MD, and Infinite MD. C. Gibbons has served as a scientific advisor for CND Life Sciences, has stock options with CND Life Sciences, and has received personal compensation for his editorial activities (Associate Editor) with Autonomic Neuroscience Basic and Clinical. R. Shaik and J. Seliger report nothing to disclose.
Ethics
This study has been approved by the Stanford institutional review board and has therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
References
- 1.Larsen NW, Stiles LE, Miglis MG. Preparing for the long-haul: autonomic complications of COVID-19. Auton Neurosci. 2021;235:102841. doi: 10.1016/j.autneu.2021.102841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Abrams RMC, Simpson DM, Navis A, et al. Small fiber neuropathy associated with SARS-CoV-2 infection. Muscle Nerve. 2021 doi: 10.1002/MUS.27458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang N, Garcia J, Freeman R, Gibbons CH. Phosphorylated alpha-synuclein within cutaneous autonomic nerves of patients with Parkinson’s disease: the implications of sample thickness on results. J Histochem Cytochem. 2020;68:669–678. doi: 10.1369/0022155420960250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Provitera V, Gibbons CH, Wendelschafer-Crabb G, et al. A multi-center, multinational age- and gender-adjusted normative dataset for immunofluorescent intraepidermal nerve fiber density at the distal leg. Eur J Neurol. 2016;23:333–338. doi: 10.1111/ene.12842. [DOI] [PubMed] [Google Scholar]
- 5.Merello M, Bhatia KP, Obeso JA. SARS-CoV-2 and the risk of Parkinson’s disease: facts and fantasy. Lancet Neurol. 2021;20:94. doi: 10.1016/S1474-4422(20)30442-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Semerdzhiev SA, Fakhree MAA, Segers-Nolten I, et al. Interactions between SARS-CoV-2 N-protein and α-synuclein accelerate amyloid formation. ACS Chem Neurosci. 2022;13:143–150. doi: 10.1021/ACSCHEMNEURO.1C00666/SUPPL_FILE/CN1C00666_SI_001.PDF. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Levine TD, Bellaire B, Gibbons C, Freeman R. Cutaneous alpha-synuclein deposition in postural tachycardia patients. Ann Clin Transl Neurol. 2021;8:908–917. doi: 10.1002/ACN3.51347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Donadio V. Skin nerve α-synuclein deposits in Parkinson’s disease and other synucleinopathies: a review. Clin Auton Res. 2019;29:577–585. doi: 10.1007/S10286-018-0581-4. [DOI] [PubMed] [Google Scholar]
- 9.Khoshnood RJ, Zali A, Tafreshinejad A, et al. Parkinson’s disease and COVID-19: a systematic review and meta-analysis. Neurol Sci. 2022;43:775–783. doi: 10.1007/s10072-021-05756-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Doppler K, Jentschke H-M, Schulmeyer L, et al. Dermal phospho-alpha-synuclein deposits confirm REM sleep behaviour disorder as prodromal Parkinson’s disease. Acta Neuropathol. 2017;133:535–545. doi: 10.1007/s00401-017-1684-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Phetsouphanh C, Darley DR, Wilson DB, et al. Immunological dysfunction persists for 8 months following initial mild-to-moderate SARS-CoV-2 infection. Nat Immunol. 2022;23:210–216. doi: 10.1038/s41590-021-01113-x. [DOI] [PubMed] [Google Scholar]
- 12.Donadio V, Incensi A, Rizzo G, et al. Skin biopsy may help to distinguish multiple system atrophy-Parkinsonism from Parkinson’s disease with orthostatic hypotension. Mov Disord. 2020;35:1649–1657. doi: 10.1002/mds.28126. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.