Abstract
Cancer refers to the division of abnormal cells at an uncontrollable rate that possesses the ability to infiltrate and destroy normal tissues. It frequently spreads to normal tissues throughout the body, a condition known as metastasis, which is a significant concern. It is the second leading cause of mortality globally and treatment therapy can assist in improving survival rates. Exosomes are the extracellular vesicles secreted by several cells that act as messengers between cells. When engineered, exosomes act as promising drug delivery vehicles that help achieve targeted action at the tumour site and reduce the limitations of conventional treatments such as castration, chemotherapy, radiation, etc. The present review provides an overview of exosomes, the biogenesis, sources, isolation methods and characterization. The current status and applications of chemotherapeutic agents loaded, engineered exosomes in cancer treatment were convoluted.
Keywords: Exosomes, Drug delivery, Cancer chemotherapy, Targeted drug delivery, Clinical trials
Graphical abstract
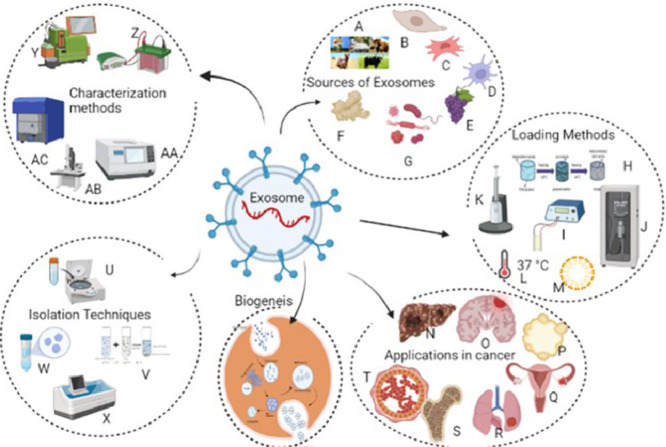
An overview of Exosome biogenesis, Sources, Isolation, Characterization, Loading and Applications. Sources of Exosomes: A-milk from various dairy animals; B,C- mesenchymal stem cells; d- Dendritic cells; E- plant sources grapes; F- Ginger; G- microbial sources; Isolation Techniques: U- Ultracentrifugation; V- Polymer precipitation; W- Size exclusion chromatography; X-Immune affinity techniques; Characterization Methods: Y-Flow Cytometry; Z- Western Blotting; AA- Nanoparticle tracking analysis;AB- Electron Microscopy; AC-Dynamic Light scattering; Loading methods: H- polymer addition; I- Electroporation; J- Sonication; K- Transfection; l-Incubation Technique; M- Surfactants; Applications in cancer: N—Hepatocarcinoma; O- brain cancer; P- Pancreatic cancer; Q- Ovarian cancer; R- Lung cancer; S- sarcoma,T- Breast cancer.
Introduction
Extracellular vesicles (EVs) are nano-sized membrane vesicles produced by many cell types [1]. Many unicellular and multicellular species have been shown to release EVs, indicating a conserved evolutionary process. Exosomes, micro vesicles, and apoptotic bodies are all examples of EVs released by mammalian cells that form the basis of this classification. Exosomes were first observed to release from the sheep reticulocytes by Pan et al., in 1983 [[2], [3]].With diameters ranging from 40 to 100 nm, exosomes are considered the smallest vesicle kind. They are produced by multivesicular endosomes (MVE) budding intraluminally and released when MVEs fuse with the plasma membrane.
Because Exosomes retain the original biological features of nano assemblies comprising biomacromolecules, cell-derived nanoparticles have attracted much attention. Exosomes, also known as small EVs, are nanoparticles that form naturally in cells and are discharged into other cells. Mesenchymal stem cells or malignant cells produce exosomes and lipid bilayers with a single-lamellar structure. Exosomes as nanocarriers offer several advantages. These advantages include their tiny size for penetration into deep tissues, slightly negative zeta potential for extended circulation, flexible cytoskeleton, and resemblance to cell membranes. Furthermore, some exosomes have a higher ability to bypass destruction or clearance by the immune system. Overall, because of their naturally biocompatible features, exosomes are appropriate natural nanocarriers for clinical usage [4].
Composition of exosomes and their cargo
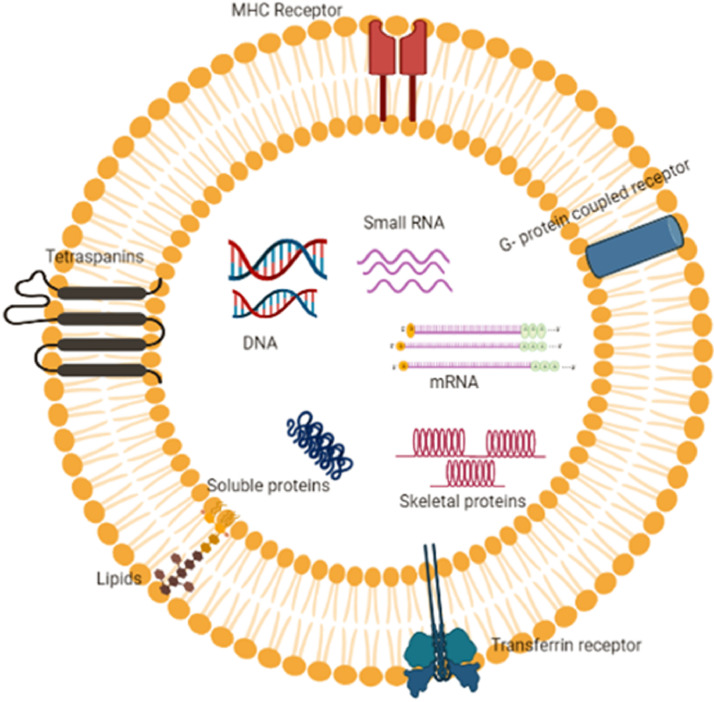
Exosomes are made up of biomolecular components that come together to form donor cells and then spread to other cells (Fig. 1). They are composed of lipids, proteins, and nucleic acids. Unlike the membranes of other EVs, the exosome membrane is mainly made up of lipid layers containing cholesterol, sphingolipid, ceramide and diacylglycerol [5]. A total of 63,100 proteins, 2838 miRNA bases, 1184 different lipids and 332,666 gene sequences were identified and available in Exocart [6]. Exosome surface proteins are influenced by their endosomal pathway and some of them, such as tumour-sensitive gene 101 (TSG101), heat shock proteins (HSC70), fusion proteins (flotillin and annexin) and tetraspanins (CD9, CD63, and CD81), can be employed as exosomal markers [7,8,9]. The composition of exosomes is summarized in Table 1.
Fig. 1.
Components and cargo activity of Exosomes; exosomes harbour surface proteins such as tetraspanins, MHC receptor, G-protein coupled receptor, Transferrin receptor and lipids. Also, internal components of the exosomes include DNA, RNA, mRNA, skeletal proteins, soluble proteins and chemical drugs.
Table 1.
Composition of exosomes.
| Composition | Types | Examples | References |
|---|---|---|---|
| Proteins | Cytosolic proteins, cell surface proteins, membrane associated proteins, enzymes, cytoskeletal proteins | fusion proteins, enzymes, chaperones, and MVBs formation-related proteins (e.g. CD9, CD63, CD81, CD147, Alix, TSG101, heat shock proteins (Hsp60), LAMP-1, Calnexin, PKM2, ANXA2, HSP90A1, SDCBP, YWHAE, LDHA, MSN, PDCP6IP, ANXA5, FASN, ACTN4, LDHB, ANXA1, HSPA1A and YWHA | [3] |
| Lipids | Cholesterol, Sphingolipids Ceramide, Glycerols | sphingomyelin, cholesterol, gangliosideGM3, desaturated lipids, phosphatidylserine, and ceramide, Diacyl glycerol, sterols | [10] |
| Nucleic acids | DNA, RNA | mtDNA, mRNAs, miRNAs, lncRNA | [11] |
| Metabolic products | carboxylic acids, amino acids, sugars, carnitines, biogenic amines,vitamins, cyclic alcohols | Pyruvic acid, lactic acid, fumaric acid, glutamic acid, α-keto glutaric acid | [12] |
Exosomes also contain nucleic acids such as double-stranded DNA (dsDNA), messenger RNAs (mRNAs) and micro-RNA (miRNAs). They may have varied biological activities depending on their cellular origin, owing to the wide range of chemicals they can transport. Transmembrane proteins such as tetraspanins (CD81, CD63, CD9), Alix and Tsg101 are all conserved proteins present in the exosomes, indicating that their biological activities are comparable [[13], [14], [15]] and can serve as potential markers for the identification of exosomes. Simultaneously, they have cell type-specific proteins representing their cellular origins and biological functions [16]. Donor cells exhibit physiological or pathological features in these nano assemblies. As a result, cancer cell-derived EVs are utilized to diagnose and treat cancer cells and they can also be used as anti-cancer medication delivery vehicles [17].
Source of exosomes
Exosomes are the extracellular vesicles that play a role in information transfer from one cell to another. These substances are produced by many cell types differing in their origin [18] and incudes [19] mesenchymal stem cells [20,21], dendritic cells [22], macrophages [23], blood [24], milk [24], bone marrow stem cells [25], saliva [26], cancer cells [27], plants [28,29], microbes [30,30]. A number of workers isolated exosomes from these different cell types and evaluated their role in cancer immunotherapy [11,[31], [32], [33]. Mendt and Sushrut reported a large-scale production of exosomes following GMP procedures from bone marrow stem cells targeted on oncogenic Kras [34]. Cancer cells generate and release more exosomes than normal cells and the compounds found in tumour cell-derived exosomes differ significantly from those found in normal cells.
Milk exosomes
Milk is commonly used as a source for exosomes and obtained from various species, including humans, bovine, porcine, rats, horses, wallabies, sheep, panda, and goat [35]. Milk exosomes from bovine sources are more commonly used than other sources and act as promising agents for exosomes isolation of exosomes [15,26,[35], [36], [37], [38], [39] with successful loading with both drugs and nucleic acids for target action. The presence of prions in bovine milk and proteinaceous compounds is a concern for the isolation of exosomes when using milk as a source.
Cancer stem cells
Cancer stem cells(CSCs) are known for their role in cell proliferation at tumour times and metastasis to other organ systems [40]. Exosomes are considered to be those messengers that mediate the conversion of non-tumour cells to tumour cells by carrying proteins and nucleic acids responsible for the regression of the tumour. HEK293 is an example of a cancer cell line used commercially to produce exosomes due to high productivity and neutral phenotype [41].
Mesenchymal stem cells
These are multipotent cells isolated from different human body sites such as bone marrow [32], adipose tissue, umblical cord, etc., that differentiate into their corresponding tissues. When MSCs are used as a potential source for exosome isolation, they produce relatively a large no of exosomal vesicles [42], with less immunogenicity, low phenotypic changes [43] and less rejection from the host [44]. For instance, the EVs secreted by SR4987 cells primed with paclitaxel (SR4987PTX) were found to transport active medicines and suppress the proliferation of human pancreatic adenocarcinoma cells in a dose-dependant manner [21]. Their use is limited due to the safety concerns arising from factors such as scalability. Also, media used for the growth of the cells, such as foetal bovine serum, is responsible for immunogenicity when retained in the final preparation.
Dendritic cells
Dendritic cells, the antigen-presenting cells in the body, are responsible for initiating antigen-specific immunity and tolerance [45]. The use of dendritic cells for exosome isolation offers advantages such as immune stimulation of Dendritic cells and stability during storage at freezing temperature for atleast six months. Dendritic cells-derived exosomes are being developed for clinical use in cancer vaccines development in phases I & II [46,47], maintenance immunotherapy after first line chemotherapy [48], tumour growth regression [49].
Plant cells
Clinical study of exosomes isolated from plant sources [29] such as aloe, barley, carrot, grapes [50,51] and ginger [52] etc., are used in different treatment protocols [53]. Several workers studied the role of exosomes secreted in plant cells and identified to protect the cells against pathogenic attack [[54], [55], [56]]. Isolation of such vesicles from plant cells [57] offers advantages in the immune free, which is a constraint in other sources of exosomes, increased cellular uptake, greater stability in the GI tract and targeted action [28]. Also, they can be isolated easily from parts such as pollen, seeds, leaves [58], juices or roots. Studies by Zhuang et al., using exosomes from grape seeds showed their activity in inhibiting brain tumour progression [59].
Microbial cells
Microorganisms such as bacteria, fungi and parasites are known to produce substances also known as outer membrane vesicles(OMV) [29] are known to have an important role in clinical therapeutics such as inflammation [60,61], enhanced T-cell regulatory effects [62], the virulence of the cells [30],antibiotic resistance [63], autoimmune disorders [62]. These OMVs exert these actions by encapsulating substances inside their vesicular structures such as enzymes [64], nucleic acids (DNA, RNA) and lipopolysaccharides [30].
Biogenesis of exosomes
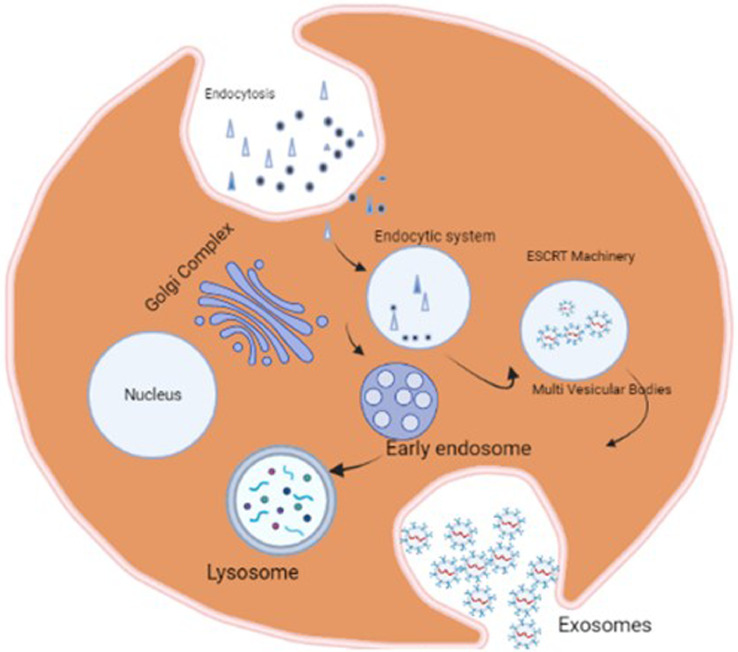
The biogenesis of exosomes generally begins with an invagination of the plasma membrane [65,66]. This process is believed to depend on the presence of an intracellular concentration of Calcium ions as reported by Savina et al., [67]. Using a High-throughput screening method, Datta et al., studied the influence of 4580 pharmacologically active substances on exosomes biosynthesis and found 12 such compounds that promote and inhibit the secretion of exosomes [68]. After the late endosomal stages, early endosomes develop into multivesicular bodies (MVBs). A second invagination of the exosome called "membrane inward budding" yields intraluminal vesicles (ILVs) inside the late endosomes in the middle of MVB maturation (Fig 2). When ILVs bud inward from late endosomes, cytoplasmic bioactive components such as proteins, lipids, and nucleic acids are loaded into the ILVs as payloads. Early endosomes are primarily produced by the endoplasmic reticulum or trans-Golgi in a few cases, independent of membrane invagination [18,69]. MVBs are fused with lysosomes for degradation or with the plasma membrane of parent cells to release multicomponents, including ILVs, into extracellular fluids [70]. The released materials are so called Exosomes. Exosomes can be produced spontaneously in two ways: the classical and direct methods [71]. Exosomes are synthesized and digested inside the cytoplasm as they transit between cells, carrying biological components taken from the Golgi apparatus or plasma membrane [72]. The classical method involves the selective removal of the plasma membrane from the cells via inward budding or endocytosis is the initial step in exosome formation. The lipid raft domains of the plasma membrane are principally responsible for the development of these endocytic vesicles. This mechanism is so efficient that a reticulocyte's plasma membrane can be recycled in as little as one hour [73]. In the direct method, multiple endocytic structures are then gathered to create early endosomes. After that, either these early endosomes are recycled into the plasma membrane or they are transformed into multivesicular bodies (MVB) with intraluminal vesicles (ILVs). MVBs eventually migrate and fuse with the plasma membrane, releasing exosomes into the extracellular environment. MVBs cargo could also be sent to lysosomes for destruction. Only MVBs with a high cholesterol content can fuse with the plasma membrane and produce exosomes, which is intriguing [74]. The endosomal sorting complex essential for transport (ESCRT) machinery is involved in the most well-studied exosome biogenesis route [40]. The ESCRT is a piece of complex protein machinery made up of four different proteins named ESCRT-0, -I, -II and -III that work together to help MVB production [75]. The ESCRT-0 identifies ubiquitinated proteins in the endosomal membrane, to put it briefly. Then, through interactions with Alix and Tsg101, ESCRT-I and -II are identified. They facilitate cargo sorting and intraluminal membrane budding, resulting in the formation of the ILV. Finally, ESCRT-III performs the membrane invagination and vesicle separation stage [76]. Following the development of the ILV, the ESCRT-III promotes cargo protein deubiquitination and with the help of the vacuolar protein sorting-associated protein 4, separates from the MVB (Vps4). The ESCRT machinery is regenerated after the vesicles have been formed. ILV can also be produced by mechanisms that are not dependant on ESCRT, such as the lipid raft-mediated and ceramide-dependant pathways [77]. Exosomes are made up of a double-layered lipid membrane that encases a small amount of cytosolic substance but does not contain any cytoplasmic organelles. The content of exosomes is directly related to the mother cell's physiological state [78].
Fig. 2.
Biogenesis of exosomes: Exosome biogenesis First, endocytosis can be mediated by either a clathrin-dependant or clathrin-independent pathway, which is often active at the lipid raft, which contains a variety of tumour-specific receptors and signalling proteins (e.g., growth factor receptors, oncoproteins), as well as common membrane proteins like tetraspanins. Exosome biogenesis can be accomplished in either an ESCRT-dependant or ESCRT-independent manner via the endosomal network. Exosomes (intraluminal vesicles) reveal inward budding of the multivesicular structures as a result (MVB).
Isolation and purification of exosomes
Exosomes, as they are secreted from a variety of cell types can be isolated through a number of techniques. The yield and purity of the exosomes is influenced by the type of isolation method adopted which also influences its interpretation [79]. The most commonly used method is Ultracentrifugation, the separation is based on the density gradient and size of the particles [80]. Centrifugation at high speeds at various time intervals helps in removal of the extraneous matter from the biological samples from exosomes. This method provides less damage to the integrity of membrane structure of exosomes but with low purity. This can be overcome with other techniques such as size based techniques [81], capture based techniques [82], polymer based [26] and micro fluidization [83] techniques for successful isolation and purification of exosomes. However, Ultracentrifugation techniques are still followed due to the advantages of less damage to the exosomes. The methods of isolation with advantages and disadvantages are summarized in the Table 2.
Table 2.
Different methods for isolation of exosomes.
| Method | Principle | Advantages | Disadvantages | References | ||
| Ultracentrifugation | Separation at high speeds at various time intervals | Simple Cost effective | Yields low purity exosomes Time consuming Low reproducibility | [79,80] | ||
| Size based separation i. Ultra-filtration ii Size exclusion chromatography (SEC) | Separation based on the size of the particle in the biological samples | Yield of exosomes is high, high purity | Not suitable for large volumesLoss of exosomes due to binding to membranes in filtrationLong run times in chromatography | [55] | ||
| Polymer based separation | Precipitation with poly ethylene glycol followed by centrifugation | Easy, scalabe technique | Moderate purity of exosomes | [26] | ||
| Microfluidization | Utilizes techniques such as microfluidic immune affinity and filtration techniques | Fast processes Easy separation Higher purity of exosomes | Expensive process Complicated equipment is required | [83] | ||
| Filtration | Ultra-filtration using membranes | Easy Inexpensive Used for large volume preparations | Deformation of particles, clogging of pores | [72] | ||
| Immunoaffinity | Selective antibody-mediated binding of vesicles with specific surface antigens. | Purity of exosomes is high | Expensive | [84] |
Characterization of exosomes
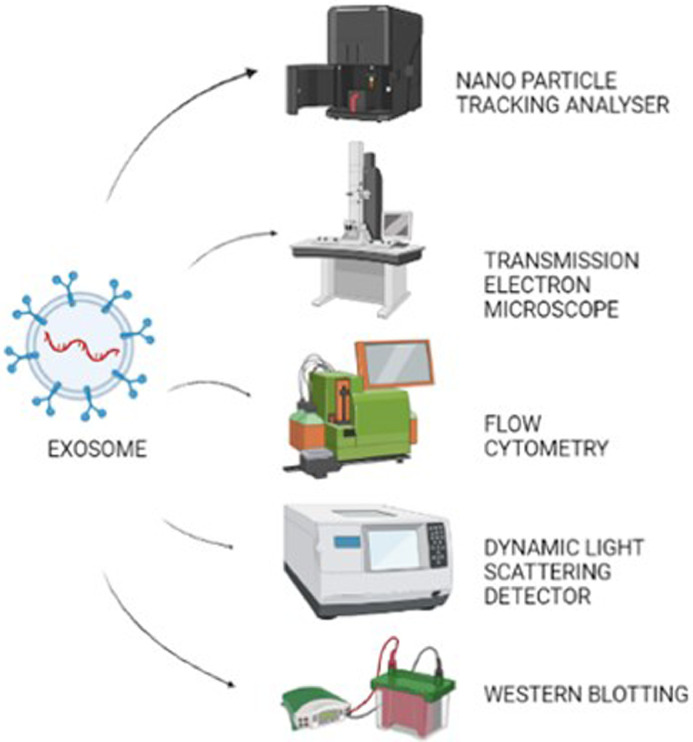
Following isolation, Exosomes are in general, characterized by different techniques [85] to understand their properties and functions. It includes techniques such as flow cytometry, nanoparticle tracking analysis, electrophoretic light scattering, protein quantification, electron microscopy and immuno blotting techniques such as Western blotting to study the properties of the exosomes that identify identifies the exosomes size range, structure and surface proteins [86]. The International Society of Extracellular Vesicles(ISEV) recommends the evaluation of extracellular vesicles by alteast two techniques [76,87]. These techniques suffer from constraints such as requirement of complicated equipment, low sensitivity and high requirement of chemicals makes the processes cost effective. Muller et al., discussed the importance of emerging techniques in the characterization of exomes together with improved techniques for their isolation and purification [88]. Some of the most commonly employed techniques for exosome characterization are explained below and shown in Fig. 3
Fig. 3.
Characterization techniques of Exosomes.
Nanoparticle tracking analysis
It is a combination of light scattering and Brownian motion of particles to analyse the exosome size. It is based on the laser illuminated microscopic [86] technique that tracks and counts each and every particle. This can also be replaced by florescence to label the exosomes with florescent molecules for easy identification. Electric field is finally applied in the suspension of exosomes that helps in determination of their zeta potential and polarity values through electrophoresis. Many researchers have used this technique for quantification of exosomes and studied their potential role in progression and inhibition of various disease conditions.
Electron microscopy
Scanning Electron Microscopy (SEM) and Transmission Electron Microscopy (TEM) are the Electron Microscopic techniques employed to determine the surface as well as size of the exosome particles [14]. These methods involve the application of laser beam on the thin film of the suspension coated with secondary electron sources such as Gold and their illumination results in morphological identification [37].
Flow cytometry
This method allows the phenotypic observation of exosomes [74,89,79]. In a hydrodynamically focused fluid stream, a flow cytometer drives cells and vesicles through a laser beam. The forward-scattered light is measured by one detector, which is situated in line with the laser beam (FSC). Other detectors measure fluorescence intensity and side-scattered light (SSC) perpendicular to the beam. Vesicles scatter light, which is important since scattering is commonly utilized as a trigger signal to alert an instrument that a vesicle is present [90].
Dynamic light scattering(DLS)
This technique also known as coherence spectroscopy is a physical technique used for the measurement of particle size and distribution of nano suspensions [86,91,92]. Light scattering by the particles is the principle involved in this technique to obtain the size of the particles. This method suffers from the drawback to identify the particle size in a heterogenic population where larger particles interfere with the size determination of smaller particles resulting in error in actual measurement.
Western Blotting
Immunoblotting or Western Blotting technique involves the affinity binding of a target antigen to its corresponding antibodies [93]. This method can be considered as destructive, the vesicles are lysed, denatured and reduced during sample preparation. This technique generally used for the detection of exosomes through identification of exosomal markers [94] such as CD9, CD63, TSG101etc., Because of its ease of use, wide accessibility and ability to detect exosomal surface proteins and interior proteins, Western blotting is one of the most widely used exosome investigation methods [93].
Role of exosomes in cancer
Extracellular vesicles(EVs) are released by both normal and cancer cells that vary in size, cargo and their distribution. In cancer, the low pH of the tumour environment and hypoxia promotes the enhanced exosomal delivery and their uptake into the tumour cells. Exosomes, in particular, promote autocrine/paracrine signalling, transforming stromal cells, causing angiogenesis and interacting with the immune system, all of which contribute to cancer progression [95], [96], [97].This is mediated by the transfer of nucleic acids that carry the tumourogenic gene to the non-cancer cells resulting in tumour progression. Modification of exosomes on its surface or cargo helps in treatment against cancer through targeted action.
Biomarkers
Exosomes as effective indicators for disease, particularly cancer, is a new area of investigation. Exosomal biomarker levels that are abnormal most likely suggest the existence of cancer or cancer in a later stage. Exosomes in blood analysis, as a set of promising biomarkers, may one day offer a minimally invasive path for cancer diagnosis, prognosis, and therapy [81]. Exosomal miRNA-103, tripartite motif-containing 3 protein, glypican-1 protein and hepatocyte growth factor-regulated tyrosine kinase substrate protein have all been suggested as potential markers for liver, gastric, pancreatic, and colon malignancies [3]. Yang Jin and associates conducted a clinical trial to study and investigate the sensitivity and specificity of serum exosome non coding RNA as biomarker to identify the onset of early lung cancer [98]. A cohort clinical study to predict the efficacy and toxicity of neoadjuvant chemoradiotherapy in rectal cancer treatment was conducted by Ji Zhu et al., using exosomal RNA as biomarkers [99]. Xiao et al., reported the application of miR-21 containing exosomes as potential source for identification of colorectal cancer [3]. Another clinical study conducted by Roberto A. Pazo-Cid and associates studied the prognostic and predictive value of tumour exosomes in gastric cancer patients [100]. An ongoing clinical study conducted by National Taiwan University Hospital, includes the study of exosomal proteins, the urine exosomal thyroglobulin, to diagnose thyroid follicular carcinoma before surgery [101]. Exosomal proteins, hence, play a role in angiogenesis, metastasis, tumour growth and illness, making them interesting biomarkers for cancer detection and prognosis.
Drug and nucleic acid delivery
The cells from which exosomes are separated, the production of exosome vesicles, cargo loading techniques, the selection of targeting biomolecules (e.g., peptides) on the surface, biodistribution and immune response are all important elements to consider in future translational applications involving exosomes [102]. Loading of small-molecule medications can be efficient, while there is potential for improvement in case of loading DNA/siRNA. Loading of nucleic acids into exosomes is achieved by many workers and provided evidence in targeted action in various diseases especially cancer [86,[103], [104], [105]]. Similar research using exosomes as delivery vehicle for anti-cancer drugs are also under pipeline and many studies provided the evidence of this approach.
Drug loading of exosomes
Drugs can be loaded into exosomes through two methods namely passive loading where diffusion is the principle mechanism of drug entrapment in exosomes and active loading where forceful loading of the drug occurs through reversible damage to cell membrane [11]. Incubation of drug with exosomes or exosomes secreting cells is a passive loading method, is an easy operating method without causing any damage to the integrity of exosomes. Increase in the diffusion efficiency of the drug are increased by active loading methods that includes physical treatment such as sonication, electroporation and surfactant treatment [106]. Fuhrmann et al., studied the various methods for drug loading into exosomes obtained from different cell lines and observed an increase incellular uptake of drug to an extent of 60% by electroporation technique in comparison with passive loading, saponin treatment, extrusion and hypotonic dialysis methods [107]. Kim et al., in their study loaded chemotherapeutic drugs like doxitaxel and paclitaxel using different methods such as passive loading, electroporation and sonication [108].
Incubation
It is the most commonly employed method of drug loading into exosomes. The technique involves mixing of the pure drug/ solution and exosome suspension at room temperature until a successful concentration of exosomes are loaded with the drug. The method may be again classified as active and passive where drug loading occurs after and before the isolation of exosomes. Yang et al., studied the cytotoxicity of drugs such as doxorubicin, paclitaxel through passive loading into exosomes [109]. This method is a non-invasive method and requires no energy for drug transport and as a result the loading efficiency of drug is low when compared to the other techniques of drug loading [110]. The factors that influence the passive loading includes the solubility of the drug, size of the drug and charge on the molecule [91,111]
Electroporation
It is technique that utilizes electric field to alter cell membranes for enhanced permeability [112]. This method is used for transfer of DNA using transfection method [113]. M. Lu and Y. Huang achieved loading of exosomes with nucleic acids by alteration of permeability of lipid membranes through electroporation technique [114]. This technique is also used by some workers for drug loading into exosomes such as paclitaxel, doxorubicin [110]. Liang et al., used the method of electroporation for loading of drug like paclitaxel and achieved a loading efficiency of 5.3% in comparison with other methods such as electroporation and passive loading [115]. The main disadvantage of this technique is damage to the structural integrity of the vesicles, formation of aggregates of exosomes [116], resulting in precipitation of adverse events.
Freeze thaw cycles
The freeze and thaw method take advantage of the creation of ice crystals that rupture the EV membrane momentarily, allowing medicinal chemicals to enter the EVs before the membrane is reconstituted [117]. When compared to sonication and extrusion-based approaches, this method demonstrates lower cargo loading. During drug inclusion, one to three freeze-thaw cycles were frequently done, which may hasten EV breakdown and aggregation [118]. Goh et al., studied the release characteristics of Doxorubicin in cell derived nano vesicles through loading with various techniques [118].
Sonication
This technique involves the application of sound waves and convert it to alter the cell membrane permeability of exosomes for efficient loading [119,120].A study showed that anti-cancer drug paclitaxel showed higher loading efficiency through sonification when compared to passive loading and electroporation technique [117]. The main disadvantage is it results in considerable variability in the structure of the exosome population along with significant change in size, membrane integrity and cellular uptake [121].
Surfactants
Use of saponins for loading drugs into exosomes is another proven method having better loading efficiencies. Saponins are known for their binding with membrane lipids such as cholesterol and their subsequent removal resulting in pore formation [120]. An example of this application is higher loading efficiencies of catalase enzyme into exosomes compared to incubation method [122]. A 11 fold increase in loading efficiency was observed using saponin treatment compare to passive loading [107]. In vivo haemolytic activity of saponins reported makes its use limited in terms of the concentration of the surfactant use in drug loading and purification after incubation with saponins [123].
Transfection
It is considered as an active loading method for loading of nucleic acids, proteins and drugs using transfection agents [123]. The advantage of using this technique is higher loading efficiencies of the drug but at the same time the toxicity and safety concern of the transfection agents makes the use limited [11]. Also, a chance of change in the genetic expression of exosomes is of major concern in determining its importance as drug loading technique.
Surface modification of exosomes
Exosomes are being studied as immunomodulators and therapeutic cargo carriers in large numbers. This expanding field of study is actively refining engineering solutions to improve efficacy and/or delivery. Exosomes that are naive have inferior targeting capabilities, are more difficult to isolate, have limited drug loading capacities, bioavailability difficulties, a short circulation half-life and a low concentration of functional surface receptors [124].To deliver exosome content effectively, the destination cell must be targeted specifically. This is mediated by the exosome's surface profile acting as a hallmark for the receiving cell type's uptake [69,125]. Exosomes can either induce signalling by directly engaging with extracellular receptors or release their cargo following fusing with the plasma membrane or internalization when they reach the target cell. As a result, a variety of engineering solutions have been devised, ranging from improving exosome uptake to optimizing cargo [126]. Engineered exosomes are modified in such a way that they can hold more components, such as drugs or miRNAs and have improved specificity and delivery efficacies. Engineered exosomes also have a number of advantages over native exosomes, including lower production costs, increased biocompatibility and a lack of undesired side effects such as cytotoxicity. Without the need for a doubt, intelligent engineering strategies for increasing the distribution of drugs, genes, heat shock proteins, and functional bioactive compounds to target tissues can improve the clinical application of exosomes. With the help of their surface ligands and receptors, they can also penetrate deeper into tissues [127].
Surface engineering
Exosomes can be surface changed despite the fact that they are natural vehicles. Surface engineering has the clear purpose of conferring cell type targeting specificity. Genetic engineering and chemical alteration are two methods of modification [128,129]. The gene sequence of a guiding protein or polypeptide is fused with that of a chosen exosomal membrane protein through genetic engineering. This method works well for displaying peptides and proteins on the surface, but it is limited to targeting genetically encodable patterns. Chemical modification enables the display of a wide range of natural and synthesized ligands via conjugation processes or lipid assembly.
Conjugation reactions can covalently and stably change exosomal surface proteins, however, the complexity of the exosome surface may limit the reaction's efficiency and the reaction typically lacks site specificity control. The vehicle's structure and function may also be compromised by covalent alteration. Exosomes can also have lipids or amphipathic molecules injected into their lipid bilayer, allowing their hydrophilic sections to be visible on the outside. This approach, which is based on lipid self-assembly, may also increase exosome toxicity. Polymer biohybrids prepared by surface modification of exosomes with cholesterol-modified DNA tethers using photo mediated atom transfer radical polymerization(ATRP) showed enhanced stability at different storage environments and proteolytic enzymes [130]. Tian et al., suggested a strategy for rapid and large scale production of exosomes using conjugation of c(RGDyK) peptide to of exosomal surface [125].
Genetic engineering
Exosome genetic engineering is a convenient way for giving exosomes new features. To begin, ligands or homing peptides are coupled to transmembrane proteins produced on exosome surfaces. Donor cells that have been transfected with plasmids encoding the fusion proteins then produce modified exosomes with targeting ligands on their surfaces [11]. LAMP-2B is the most targeted protein for modification of surface of the exosomes through genetic engineering [129,131,132]. Some co-workers engineered exosomes through modification of cell specific binding peptides as in case of rabies virus glycoprotein exosomes loaded with miRNA showing specificity to acetyl choline receptors [129]. In an another study integrin peptides of exosomes are modified to enhance the biodistribution and pharmacokinetic profiles of the exosomes [[132], [133], [134], [135], [136]]. tLyP-1 peptide is another such surface protein that targets the neuropilin receptor in non-small cell lung cancer [133,137,138]. Despite the effectiveness of engineering exosomes through this technique, it suffers from disadvantages like change in property of cells and exosomes and uneven loading of probe proteins [139].
Chemical modification
Chemical modification of exosomes offers techniques that used chemical substances to bind to the exosomes through various techniques such as click chemistry [137,140], formation of triazole linkage resulting in azide formation, linkage with PEG and disteroyl phosphoethanol amine analogs that makes the exosomes by pass their elimination by the immune system with prolonged plasma time for activity [141]. However, this technique causes instability of the exosomes and their aggregation during manipulation.
Membrane fusion
Yet another method of modification of exosomes for increased targeted action is the fusion of membrane of exosomes with compounds such as virus stimulating vesicles, hybridization with liposomes [142], drugs. Aptamer derived exosomes are used to target the cancer cells by delivering nucleic acid material such as RNA, miRNA and drugs substances to achieve targeted effect [143].
Pharmacokinetic characteristics and tumour distribution of naive exosomes
Naïve or naturally occurring exosomes produced by various cells in the body such as stem cells and cancer cells. A thorough study of the pharmacokinetics of exosomes, i.e., the in-vivo behaviour of exosomes, is critical for the clarification of the biological activities of exosomes and the practical implementation of exosome-based treatments. The critical elements in the pharmacokinetics of exosomes are their dispersion to organs and subsequent cellular absorption. Furthermore, it is thought that exosome uptake happens as a result of cellular recognition of the exosomes surface components. Exosomes formed naturally take on the physiological features of the cells from which they originate. When exosomes are treated in vivo, they demonstrate equal potency and a superior safety profile than the conventional cell therapy, implying that they could be used as cell-free medicines. Several attempts have been made to increase the usage of exosomes in various therapeutic domains by either designing exosomes or exosome-producing cells for loading API cargos and exosome targeting to specific tissues/cells144. The in vivo behaviour of administered exosomes is represented by its biodistribution and PK profile and determining these two parameters is critical for effective exosome-mediated new therapies development.
The first step in determining exosome pharmacokinetics is to assess tissue distribution, also known as exosome biodistribution. The biodistribution of exogenously delivered exosomes has been studied using a variety of labelling approaches [145]. Exosomes have been labelled with tiny lipophilic fluorescent dyes for in vivo tracking. Although the free dye released by exosomes might compromise the in vivo analysis reliability, this procedure is a good way to assess the localization of exosomes supplied to tissues [146,147]. In addition, dyes in the near IR region such as 1,10-dioctadecyl-3,3,30,30-tetramethylindodicarbocyanine perchlorate (DiD) and 1,10-dioctadecyltetramethylindotricarbocyanine iodide (DiR) are also used as markers to trace the biodistribution of exosomes by several workers [28,51,[148], [149], [150], [151]].
To summarize, as soon as their entry into the blood, the exosomes are rapidly cleared by the reticuloendothelial system, reaches the liver, resulting in its rapid clearance from the body [151]. Also, the localization of exosomes i.e., targeting action is dependant on factors such as its origin, membrane composition, types of cells exosomes are targeted and pathophysiological conditions of the host [144]. Amin Mirzaaghasi et al., studied the biodistribution and pharmacokinetics of exosomes in comparison with liposomes in a mouse model of sepsis and observed the localization of exosomes in the lungs and prolonged plasma concentration due to liver dysfunction [94]. Several studies on the biodistribution of exosomes using dyes and markers showed the uptake of the exosomes followed their origin [130] and at the same time exosomes can't be used in all cases of cancer as they found to contribute a niche formation by delivering tumourogenic factors to healthy tissues [152], [153], [154] and cases of thrombosis and haemostatic perturbations in cancer patients [155].
Application of drug loaded exosomes in cancer therapy
Cancer is the leading cause of death worldwide after ischaemic heart disease [156]. A number of treatment methods are followed to achieve a breakthrough in the treatment strategy [157]. Differential expression of exosomal proteins results in a variety of events i.e., upregulation of proteins subcutaneous transplantation tumour models, orthotopic tumour models and advanced metastatic tumour models modulates the tumour microenvironment whereas the downregulated proteins results in tumour cell growth, survival and drug resistance in tumour cells. Drug delivery through exosomes through modifications of surface proteins as well as loading strategies is one such treatment strategy aimed at targeted delivery of the drug and at the same time reduce the side effects due to chemotherapy. Some of the applications of exosomes indelivering drug to the tumour sites in various cancers are summarized below
Liver carcinoma
Tuying Yong et al., developed exosome-biomimetic nanoparticles having the potential to be used as drug carriers to improve anticancer efficacy [158]. Biocompatible exosomes sheathed in porous silicon nanoparticles (PSiNPs) are developed for targeted cancer chemotherapy using Doxorubicin (Dox). After incubation with DOX@PSiNPs, DOX@E-PSiNPs are exocytosed from tumour cells. DOX@E-PSiNPs exhibited enhanced tumour accumulation, tumour penetration and cross-reactive cellular uptake by bulk cancer cells and cancer stem cells (CSCs) following intravenous injection, resulting in increased in vivo Dox enrichment in total tumour cells and side population cells. DOX@E-PSiNPs also demonstrated significant anticancer and CSCs killing activity in. Hongzhao Qi et al., synthesized and evaluated the tumour inhibitory effects of Doxorubicin exosomes through anchoring supra magnetic properties [159]. The exosomes isolated from blood were blended with supra magnetic ferric oxide (carboxyl functionalized Fe3O4 nanoparticles), assessed for their anti tumour activity on H22 cell lines through exposure to magnetic field. It was observed that the magnetically modified exosomes with Dox showed excellent in vitro cytotoxicity in the presence of magnetic field and can expand the area of its applications.
Pulmonary cancer
The lungs are the most common sites of metastasis and tumour relapse following chemotherapy and accounts for 30–55% [160]. Exosomes (exo) were loaded with Paclitaxel (PTX), an anti-cancer drug commonly used against lung cancer and transported to pulmonary metastases [160] by Kim et al. A variety of cancer forms, including lung cancer, have been demonstrated to overexpress the sigma receptor, a membrane-bound protein. Morse et al., ran a phase I clinical trial to explore the safety, feasibility, and efficacy of delivering autologous dendritic cell-loaded exosomes (Dex) loaded with antigenic HLA-presented peptides of melanoma-associated antigen (MAGE) to patients with advanced non-small-cell lung cancer (NSCLC) [16,161]. MAGE gene products are of particular relevance because of their widespread expression in various tumors and their ability to activate tumour-specific cytotoxic T-lymphocyte (CTL) responses against MAGE-expressing tumour cells culminating in tumour cell lysis. Antigen-specific CTLs produced by MAGE gene-derived peptides are particularly effective in preventing and treating a variety of tumors, and MAGE has thus been exploited as a tumour target [162]. Kim et al., studied the drug loading efficiencies of the exosomes with drugs Doxorubicin and Paclitaxel and their uptake against the multi-drug-resistant (MDR) tumour cells. Confocal studies revealed a significant accumulation of exosomes in cancer cells, as well as a limited uptake of liposomes and nanoparticles [108]. Exosomes were taken up approximately 30 times better than synthetic nanoparticles, implying that PTX loaded into exosomes can be effectively delivered to tumour cells in therapeutically sufficient amounts [163]. These findings demonstrate the benefits of exosome-based delivery systems over conventional synthetic nanocarriers. In another study by Agarwal et al., exosomes isolated from bovine milk were used for investigating the possibilities of loading paclitaxel through oral administration [164]. Both pure drug (PAC) and exosome paclitaxel(EXOPAC) showed excellent stabilities in simulated gastro intestinal fluids and storage at −80°C. Furthermore, it was also observed that the orally delivered EXOPAC showed a significant antitumor effect against human lung xenografts of nude mice, with a substantial reduction in systemic and immunogenic toxicities compared to i.v. delivered Paclitaxel.
Breast carcinoma
Breast cancer commonly found in women than men is a heterogenic disease that results in death worldwide [33]. Tian et al., in their study, observed the effects of modified exosomes with Doxorubicin (Dox) on mice with tumors of breast cancer [132]. Immature dendritic cells(iDC) were used for the expression of exosomes which, when later engineered with αv integrin-specific iRGD peptide (CRGDKGPDC) that helped in targeting the tumour cells resulted in enhanced drug penetration [133] and reduction in tumour aggression when compared to its pure drug(Doxorubicin) alone or combination of drug-loaded pure exosomes. Also, the study showed a decrease in the tissue toxicity of Doxorubicin which is its major side effect in tumour treatment [165]. Another study conducted by Yu et al., on triple negative breast cancer (TNBC) employed exosomes loadedwith erastin proved to be a powerful platform for TNBC chemotherapy [111]. Exosomes derived from TNBC cell line and lung fibroblasts, labelled with folate marker are used to enhance the solubility and reduce renal toxicity of the drug erastin. The results showed a better inhibition on the proliferation and migration of the tumour cells with increased uptake efficiency of erastin and ferroptosis that is necessary in killing the tumour cells. Han et al., in their study proved the anti-tumour activity of Taxol on the breast cancer cell lines through apoptosis of cells by loading into exosomes derived from natural killer cells of the defence system [92]. A similar work with the drug Doxorubicin was carried out by Gomari et al., to evaluate its efficiencies in both pure and exosome loaded form [166]. Their study supported that the exosome loaded Doxorubicin bioavailability can be increased with significant reduction in side effects.
T-cell leukamia
Fabrication of exosomes with aptamers such as sgc8 and their study for their targeted action on T-leukamia cell lines showed enhanced cellular accumulation with reduced cytotoxicity [167]. Jianmei Zou and their associates observed the changes in the drug accumulation and cytotoxicity through fabrication with aptamer such as 5-Carboxyfluorescein (FAM) labeled diacyllipid-DNA probes for improving the availability of cancer drugs. In their study cellular uptake and accumulation studies of anticancer drug Doxorubicin [168] are conducted using various uptake inhibitors such as sodium azide, dynasore [169], chlorpromazine [170] etc., and observed that the cellular accumulation of mostanticancer drugs occurred through active transport and also reduction in cytotoxicity of fabricated exosomes when compared to pure drug exosome combination or pure drug alone. Another study by Dongmei Sun et al., observed the increase in accumulation of curcumin loaded exosomes by tenfold when compared to the pure drug alone [171]. The cytotoxic effect of the combination was studied on CD11b+Gr-1+ cells in the lungs of mice and found to cause an increase in apoptosis of the cells with increase in uptake of curcumin exosomes.
Skin cancer
Pinto et al., used mesenchymal cell derived exosomes to overcome the disadvantages in photodynamic therapy (PDT) in peritoneal carcinomatosis for targeted drug delivery using photosensitizer such as loaded extracellular vesicles [17,172]. The study conducted on murine models showed outstanding tumour selectivity, minimal off target accumulation of photosensitizer, reduction in death due to PDT, improvement in direct cytotoxic antitumor effectiveness with indirect modulation of the tumour microenvironment promoting an inflammatory immune response in peritoneal metastasis (PM) and T- lymphocyte activation against the tumour. By increasing the exposure time of the biological targets, the exosome delivery system was shown to actually enhance the tumoricidal effect of Acridine Orange (AO) by Elisabetta Iessi and co workers [173]. The results showed macrophage exosomes, MØExo-AO was taken up much more quickly within tumour tissues and was retained for much longer than free AO, achieving significant tumoricidal efficacy in 3D conditions such as spheroids. This has the potential for clinical application in cancer patients, as it uses the tumor's inherent acidity against it by delivering and releasing larger amounts of chemotherapeutic drugs while sparing normal tissues. An even more advantage that has already been exploited at the local level is photodynamic therapy (PDT), i.e., the ability to purposefully stimulate this agent by exposing it to light of the appropriate wavelength.
Pancreatic cancer
Being one of the most lethal cancers, pancreatic cancer has a 5 year survival rate of about 6% with a median survival rate accounting to about 6 months [174]. Mutations in KRAS have been linked to pancreas, lung and colon cancer, amongst others and oncogenic KRAS mutations, as well as activation of downstream effectors like MEK, Akt and Erk, amongst others, are sufficient drivers of pancreas cancer. A sound rationale for targeting Ras for cancer treatment emerged, but Ras has remained largely unsolvable. There was some efficacy reported with methodologies developed to target oncogenic Kras using siRNA molecules, but these approaches may have been limited by a lack of specificity and inefficient delivery. Despite this, a recent clinical study found that siG12D-LODERTM was well-tolerated and had potential efficacy in patients with locally advanced pancreas cancer. Costa-Silva et al., emphasised the significant repercussions of EV contact with immune system components [175]. They discovered that when Kupffer cells in the liver were exposed to pancreatic carcinoma-derived exosomes, these cells secreted transforming growth factor-beta and hepatic stellate cells produced fibronectin. This environment encouraged the entry of bone marrow-derived macrophages, which released a macrophage movement inhibitory factor that has been linked to pancreatic cancer cell liver metastasis. Sushrut Kamerkar et al., show that engineered iExosomes can control advanced PDAC in mice and that this method is clinically viable [176]. Carlos et al., provided new evidence of ability of curcumin's anti-cancer functions form one pancreatic cancer cell to another cancer cell through exosomal transportation [70]. The study also showed the curcumin function is not limited to cancerous cells but also extends to the tumour microenvironment. Also, the evaluation of efficacy of curcumin exosomes on pancreatic cell lines PANC-1 and MIA PaCa-2 cell and found significant reduction in viability of the tumour cells when compared with the pure drug. L. Pascucci et al., demonstrated the antitumour activity of engineered exosomes from mesenchymal stem cells primed with Palcitaxel showed a strong resistance to Paclitaxel toxicity and inhibit the proliferation of CFPAC-1tumour cells in a dose dependant manner [177].
Ovarian cancer
Ovarian cancer, a major type of gynaecological cancers with a 5 year survival rate of 44% is a case of concern of death in women worlwide [178]. Triptolide(TP), a herbal drug is famous for its anti-inflammatory and immunosuppressant action. It also possess anti-tumour activity but limited due to poor solubility apart from reproductive, hepato and nephrotoxicities [179,180]. Huan Liu et al., loaded this anticancer drug into the tumour cell derived exosomes observed the cytotoxic and autophagy effects of TP-Exosomes on SKOV3 cells were weaker than those of free drug, but tumour cell proliferation and tumour growth inhibition were stronger. Furthermore, TP-Exosomes are toxic to the liver and spleen. To summarise, TP-Exos may be a promising treatment for ovarian cancer, but they must be further optimised to reduce liver and spleen damage.
Brain tumour
A main constraint in treatment of brain tumours is the low efficiency of drug delivery to the glial cells [181] due to the presence of blood brain barrier. Zhuang et al., in a study studied the effect of curcumin loaded exosomes on the glial cells using a non-invasive route of administration such as intra nasal to treat inflammation related to brain cells [182]. The study conducted using three inflammation mediated models one of them utilizing GL26 tumour cells where the drug loaded exosomes were found to significantly delayed brain tumour growth with rapid uptake of the drug into the glial cells and subsequent induced apoptosis of the cancer cells. This strategy may be considered as a novel therapeutic approach in treatment of neurological disorders. Yang et al., in a study observed the transport of drugs such as Doxorubicin and Paclitaxel across the blood brain barrier [109]. The study conducted using Zebra fish models(xenotransplant brain cancer models) showed a significant decrease in cancer metastasis when drugs are blended into exosomes rather than alone. The study also used a dye, a fluorescent compound Rhodamine to label the drugs for observing the selective uptake of the drugs into the brain which occurred when the drugs are loaded into exosomes. Jia et al., engineered exosomes through conjugation with neuropilin-1-targeted peptide (RGERPPR, RGE) by click chemistry after loading with Doxorubicin and supra magnetic iron oxide nanoparticles to target brain cells in glioma [137]. The observations showed an increase in uptake efficiency of the engineered exosomes with higher therapeutic functions due to synergistic effect, providing a new approach for improving the diagnosis and treatment of brain tumour.
Prostate cancer
Prostate cancer, the common cancer amongst men having a high rate of mortality [183]. H. Saari et al., studied the effectiveness of the drug paclitaxel through its loading in cancer cell derived exosomes [184]. The study progressed with the separation of exosomes through centrifugation from the prostate cell lines such as LNCaP and PC-3 followed by loading with paclitaxel drug that showed enhanced cytotoxicity of the combination than with the pure drug alone. The study also showed no significant leakage of the drug from the vesicular system that explained its enhanced cytotoxic effect. Smyth et al., studied the biodistribution and delivery efficacy of exosomes with that of liposomes using Doxorubicin in tumour proliferation [154]. The observations showed increase in cellular retention of exosomal Doxorubicin at the tumour site when compared with liposomal Doxorubicin and suggests that exosomes are better drug delivery vehicles than liposomes. Another study conducted by Alcayaga-Miranda et al., demonstrated the ability of menstrual derived mesenchymal cells derived exosomes tumorogenic activity by blocking tumour induced angiogenesis by formation of free radicals [185,185]. In a study conducted by Coccè and his associates, the anti-tumour efficiency of mesenchymal stromal cells were evaluated through priming with anti-cancer drug paclitaxel [186]. The secretome of the mesenchymal stromal cells lack anti-tumour activity but a marked increase in antitumour activity after priming with paclitaxel was observed.
Osteo sarcoma
Bone cancer, a relatively rare than the other types of cancer has a 5 year survival rate of 75% [27]. Wei et al., in a study observed the efficiency of doxorubicin in osteosarcoma MG63 cell lines [187]. The results showed an increase in the cytotoxic effect on the tumour cells with reduction in toxicity of the myocardial cells that is attributed to the interaction of membrane proteins of both tumour and exosomes. The various drugs used for loading into exosomes, their loading techniques are summarized in the Table 3.
Table 3.
An overview of drugs used in cancer treatment.
| Drug | Source of exosome | Loading technique | Cancer | Application | References |
| Doxorubicin | Tumour cells | Porous silicon nanoparticles | subcutaneous transplantation tumour models, orthotopic tumour models and advanced metastatic tumour models of liver | enhanced tumour accumulation, tumour penetration, and cross-reactive cellular uptake by bulk cancer cells and cancer stem cells | [158] |
| Doxorubicin | Blood | Blending with supra magnetic particles | Liver carcinoma | excellent in vitro cytotoxicity in the presence of magnetic field | [86] |
| Paclitaxel | Macrophages | Various loading techniques | Lung cancer | Over expression of sigma receptor | [160] |
| peptides of melanoma-associated antigen (MAGE) | Dendritic cells | Various loading techniques | Lung cancer | MAGE gene-derived peptides are particularly effective in preventing and treating a variety of tumors | [188] |
| Doxorubicin Paclitaxel | Cell lines | Various loading techniques | Lung cancer | exoPTX holds significant potential for the delivery of various chemotherapeutics to treat drug resistant cancers | [160] |
| Paclitaxel | Bovine milk | Passive loading | Lung cancer | significant antitumor effect against human lung xenografts of nude mice | [164] |
| Doxorubicin | Immature dendritic cells | Incubation | Breast cancer | Enhanced drug penetration and reduction in tumour aggression | [132] |
| Erastin | Cancer cells and fibroblasts | PEGylation | Breast cancer | better inhibition on the proliferation and migration of the tumour cells with increased uptake efficiency of erastin and ferroptosis | [111] |
| Taxol | Natural Killer cells | Electroporation | Breast cancer | Drug-loaded Exos can effectively inhibit proliferation and induce apoptosis of tumour cells, thereby exerting an anti-tumour effect. | [92] |
| Doxorubicin | Mesenchymal cells | Electroporation | Breast cancer | Increased bioavailability with significant reduction in side effects | [136] |
| Doxorubicin | Immature Dendritic cells | Electroporation | T-cell leukamia | Enhanced cellular accumulation and reduced toxicity | [132] |
| Doxorubicin | Various sources | Various methods | T-cell leukamia | Enhanced cellular accumulation and reduced toxicity | [189] |
| Curcumin | Plant based exosomes | Various methods | Colorectal cancer | Ongoing clinical trials to study the efficacy of curcumin in cancer treatment | [190] |
| Curcumin | Monocytes | Passive loading, freeze thawing | T-cell leukamia | Enhanced stability of the drug with 10 fold higher accumulation | [168] |
| meta(-tetrahydroxyphenyl)-chlorin (mTHPC) | Mesenchymal cells | Passive loading | Skin cancer | enhancement of tumoral selectivity in comparison to the free drug and to the liposomal formulation | [172] |
| Acridine Orange | Macrophages and tumour cells | Passive loading | Skin cancer | Increase in cell uptake of drug, decrease in cytotoxicity | [173] |
| Curcumin | Tumour cells | Passive loading | Pancreatic Cancer | Significant reduction in viability of tumour cells compared with the drug | [70] |
| Paclitaxel | Mesenchymal stem cells | Passive loading | Pancreatic Cancer | Increase in resistance of cells towards drug toxicity, inhibition of proliferation | [186] |
| Triptolide | Tumour cells | Ultra Sonication | Ovarian cancer | Stronger inhibition of tumour proliferation and growth | [179] |
| Curcumin | Various cells | Passive loading | Brain cancer | Significant delay in tumour growth | [137] |
| Doxorubicin and Paclitaxel | Tumour cells | Passive loading | Brain tumour | Significant reduction in cancer metastais | [109] |
| Doxorubicin combined with supra magnetic particles | Tumour cells | Electroporation | Brain tumour | Increased drug accumulation inside the brain and synergistic effect | [168] |
| Paclitaxel | Cancer cells | 0Passive loading | Prostate cancer | Enhanced cytotoxicity when compared with pure drug alone | [184] |
| Paclitaxel | Mesenchymal stem cells | Passive loading | Prostate cancer | A significant anti cancer activity is observed compared to the pure drug alone | [185] |
| Poryphyrins | Various cell lines | Electroporation, passive loading and saponin treatment | Skin cancer | increase in cytotoxicity of the porphyrin loaded exosomes with increase in hydrophobicity of porphyrins | [107] |
| Doxorubicin | Bone marrow stem cells | Passive loading | Bone cancer | enhanced cellular uptake efficiency and anti-tumour effect in osteosarcoma MG63 cell line | [25] |
Exosomes as anti-cancer vaccines
Exosomes also serve as a tool in the preparation of anti-cancer vaccines. They stimulate the T-cell mediated immune response against the tumour cells [4]. Dendritic cells act as strong adjuvants in initiating the T-cell mediated immune response [191,192]. Laurence Zitvogel et al., demonstrated the application of exosomes as cancer vaccines [193]. They demonstrated that the single intradermal injection of tumour peptide loaded Dendritic cell derived exosomes induced a potent immune response in nude mouse with a sharp rise in T-cell immunity resulting in tumour growth delay or complete eradication. BS Sundarasetty et al., developed vaccine using a Lentivirus vector programmed Dendritic cells that proved to have greater specificity on melanoma cells [194]. Bernard Escudier and co-workers reported the feasibility and safety of Dendritic cell derived exosomes based vaccination for lung cancer [188]. MHC class I and II peptide loaded exosomes in stage III and IV cancer patients showed better response and stabilization in cancer patients. In another phase II clinical trials conducted by Gustave Roussy and Curie developed an Immunotherapeutic method for cancer vaccination using metronomic cyclophosphamide and vaccination with Dex (dendritc cell derived exosomes) [195]. Phase I results of their study shown the safety and feasibility of administration of Dex and stimulation of T-cell response have to be reported with a progression free survival rate of atleast 4 months. A study conducted by Damo et al., in vaccine preparation using Dexo(Dendritic cell derived Exosomes) stimulated with TLR- 3 ligand, loaded with antigen OVA followed by maturation with poly (I:C) showed promising anti-cancer activity with high stimulation of immune cells [196]. In another study, Zhen Lu proved the efficacy of exosomal vaccines. Exosomes produced from α-fetoprotein Dendritic cells showed a antigen specific immune response and significant tumour suppression in nude mice models and HCC (hepato cellular carcinoma) mice models [197].
Manufacturing strategies and limitations
Exosomes are recognized by the cells from which they originate as well as their tailored qualities. Their microenvironment, culture and dissociation systems, on the other hand, control their efficient and reliable creation. Isolation and characterization are also the key criterion required as downstream processes in their manufacture [69]. This procedure is usually broken down into three steps
-
I
Cell and debris removal
-
II
Condition medium concentration and
-
III
Purification
Depending on the source of isolation of exosomes, the exosomes differ in the growth requirements and capabilities of exosome production [198,199]. These include growth factors, oxygen requirements, cell density, size and size distribution of exosomes [53] etc., When selecting a medium for the growth of exosomes, the media is generally made free of xeno products i.e., free from animal derived products which in normal conditions are responsible for contamination and instability of exosomes. These conditions facilitate the implementation of GMP in commercial exosomal production [200]. The commercial production generally accompanies the usage of either conical flasks as in the case of static systems or bioreactors for large scale and continuous production.
Sart et al., in their study of exosomes identified the role of micro carriers in stirred tank bioreactors that provided maximum surface area [201]. Another study by Yan and Wu, emphasized the use of stirred tank bioreactors in hollow fibre perfusion bioreactor for industrial production of exosomes [202].
After cultivation of desired exosomes, the important task is the cell and debris removal. This is achieved through the process of ultracentrifugation in commercial process for static cultures [203]. Other techniques include density gradient, precipitation, liquid chromatography and size exclusion chromatography which have various applications. Tangential filtration, a newly developed technique from principles of tangential flow [204] and membrane filtration is a gentle, effaceable technique than ultracentrifugation that helps in prevention of cake formation. Scaling up of production of exosomes to plant scale requires modification of the existing lab techniques employed that may have influence on the CMC (Chemistry Manufacturing and Control) timelines of the exosomes and corresponding GMP requirements. Chen et al., observed the differences in these attributes during scale up when the source of exosomes have been differed [205]. Brindley and Panchalingam in their studies observed the influence of shear stress on the structure of the vesicles in large bioreactors with modifications in cellular phenotypes [206,207]. This problem can be reduced by employing multiple layer flasks or layered flasks in small scale but again a constraint in large scale production. A commercially available Integra CELLine™ system that utilizes different compartments for cells and media with a continuous nutrient flow without causing damage to exosomes [208]. Secondly, the medium used in the cultivation such as Foetal bovine sera are rich in exosomes and contaminants act as constraints for good manufacturing practices of exosome production. Further, the loading efficiencies also are influenced by the source of exosomes in the production. The final step in manufacturing is the purification step that again is influenced by the heterogenicity of the exosome population. Different techniques are employed for this stage where Ultra-centrifugation technique [209] in combination with other methods such as size exclusion chromatography, sucrose gradient techniques are employed to get higher concentrations of purity of exosomes as ultra-centrifugation followed by size exclusion technique enables elution of larger particles such as exosomes while retaining smaller particles such as non-vesicular proteins and RNA. The characterization of the exosomes throughout production is a requisite to confirm its safety, efficacy and quantification. These include surface protein profiling [210], Western blotting technique, liquid chromatography, mass spectrophotometry, size analysis by scanning and transmission electron microscopy, concentration analysis by nano particle analysis, particle count by flow cytometry etc.,
Also, the storage conditions for pure exosomes are a challenge for large volume preparations for further processes like translation and loading cargo into exosomes. These include storage temperature, buffer and pH that influences their stability [211].
Exosome under clinical investigations
Use of exosomes for various treatments is under several stages of clinical trials. A total of 118 clinical trials [212,213] at various stages are ongoing by the year February 2022. Of the total studies, 58 studies are ongoing in cancer treatment at various stages [214]. Around 18 protocols occurring in phase 1 clinical trials [212] in which 9 happens to target various cancers [215]. 11 studies using exosomes have made their success in phase 1 trials and are now 3 protocols for cancer treatment are in their Phase 2 study [216]. In the phase 3 trials 3 such delivery systems using exosomes for targeted therapy made their position in clinical treatment [217]. The following Table 4 lists out the ongoing clinical trials in cancer using exosome mediated therapy.
Table 4.
Clinical trials of exosomes in various cancer studies.
| Sl no | - NCT number | Title of project | Condition | - Clinical Trial Phase | References |
| 1. | NCT04939324 | Molecular Profiling of Exosomes in tumour-draining Vein of Early-staged Lung Cancer (ExOnSite-Pro) | Lung Cancer Exosomes Non Small Cell Lung Cancer | Pre-clinical phase | [218] |
| 2. | NCT03542253 | Combined Diagnosis of CT and Exosome in Early Lung Cancer | Early Lung Cancer | Pre-clinical phase | [219] |
| 3. | NCT03821909 | Acquisition of Portal Venous CTCs and Exosomes From Patients With Pancreatic Cancer by EUS | Pancreatic Cancer | Pre-clinical phase | [220] |
| 4. | NCT01779583 | Circulating Exosomes as Potential Prognostic And Predictive Biomarkers In Advanced Gastric Cancer Patients ("EXO-PPP Study") | Gastric Cancer | Pre-clinical phase | [100] |
| 5. | NCT01344109 | A Pilot Study of tumour-derived Exosomes as Diagnostic and Prognostic Markers in Breast Cancer Patients Receiving Neoadjuvant Chemotherapy | Breast Neoplasms | Pre-clinical phase | [221] |
| 6. | NCT02393703 | Interrogation of Exosome-mediated Intercellular signalling in Patients With Pancreatic Cancer | Pancreatic Cancer Benign Pancreatic Disease | Pre-clinical phase | [222] |
| 7. | NCT02702856 | Clinical Validation of a Urinary Exosome Gene Signature in Men Presenting for Suspicion of Prostate Cancer | Prostate cancer | Pre-clinical phase | [223] |
| 8. | NCT02147418 | Exosome Testing as a Screening Modality for Human Papillomavirus-Positive Oropharyngeal Squamous Cell Carcinoma | Oropharyngeal Cancer | Pre-clinical phase | [224] |
| 9. | NCT03738319 | Non-coding RNA in the Exosome of the Epithelia Ovarian Cancer | High Grade Serous Carcinoma•Ovarian Cancer •Exosomes •Prognosis •Early Diagnosis | Pre-clinical phase | [225] |
| 10. | NCT01668849 | Edible Plant Exosome Ability to Prevent Oral Mucositis Associated With ChemoradiationTreatment of Head and Neck Cancer | Dietary Supplement: Grape extract •Drug: Lortab, Fentanyl patch, mouthwash | Pre-clinical phase | [226] |
| 11. | NCT02890849 | Clinical Research for the Consistency Analysis of PD-L1 in Cancer Tissue and Plasma Exosome | •NSCLC | Pre-clinical phase | [227] |
| 12. | NCT04394572 | Identification of New Diagnostic ProteinMarkers for Colorectal Cancer | Colorectal Cancer | Pre-clinical phase | [228] |
| 13. | NCT03109873 | Metformin Hydrochloride in Affecting Cytokinesand Exosomes in Patients With Head and Neck Cancer | •Larynx •Lip •Oral Cavity •Pharynx | Early Phase 1 | [229] |
| 14. | NCT01159288 | Trial of a Vaccination WithTumor Antigenloaded Dendritic Cell-derived Exosomes | •Non Small Cell Lung Cancer | Phase 2 | [195] |
| 15. | NCT04960956 | Glycosylation of Exosomes in Prostate and Urothelial Carcinoma | •Prostate Cancer •Urothelial Carcinoma | Pre-clinical phase | [230] |
| 16. | NCT04288141 | A Study to Measure the Expression of the HER2-HER3 Dimer in Tumour and Blood(Exosomes) Samples From Patients With HER2 Positive Breast Cancer Receiving HER2Targeted Therapies | •HER2-positive Breast Cancer | Pre-clinical phase | [231] |
| 17. | NCT03974204 | Analyses of Exosomes in the Cerebrospinal Fluid for Breast Cancer Patients With Suspicionof Leptomeningeal Metastasis | Breast Cancer •Leptomeningeal Metastasis | Pre-clinical phase | [232] |
| 18. | NCT04529915 | Multicenter Clinical Research for Early Diagnosis of Lung Cancer Using Blood Plasma Derived Exosome | Lung Cancer | Pre-clinical phase | [233] |
| 19. | NCT03032913 | Diagnostic Accuracy of Circulating tumour Cells (CTCs) and Onco-exosome Quantification in the Diagnosis of Pancreatic Cancer - PANCCTC | •Pancreatic Ductal Adenocarcinoma (PDAC) | Pre-clinical phase | [234] |
| 20. | NCT01294072 | Study Investigating the Ability of Plant Exosomes to Deliver Curcumin to Normal andColon Cancer Tissue | •Colon Cancer | Phase 1 | [235] |
| 21. | NCT03874559 | Exosomes in Rectal Cancer | Rectal cancer | Pre-clinical phase | [236] |
| 22. | NCT04167722 | How Does Prostate Cancer Metastasize? Studying the Role of Secreted Packages(Exosomes) From Fat Tissue in Lean and Obese Patients | Prostate Cancer •Obesity | Pre-clinical phase | [237] |
| 23. | NCT02869685 | Clinical Research for the Consistency Analysis of PD-L1 in Lung Cancer Tissue and Plasma Exosome Before and After Radiotherapy | •NSCLC | Pre-clinical phase | [227] |
| 24. | NCT04629079 | Improving the Early Detection of Lung Cancer by Combining Exosomal Analysis of Hypoxia With Standard of Care Imaging | •Lung Cancer | Pre-clinical phase | [238] |
| 25. | NCT04227886 | Study on Predictive Biomarkers of Neoadjuvant Chemoradiotherapy for Rectal Cancer | •Rectal Neoplasm MalignantCarcinoma •Chemoradiotherapy •Neoadjuvant Therapy •Predictive Biomarkers •Adenocarcinoma | Pre-clinical phase | [99] |
| 26. | NCT03581435 | A Study of Circulating Exosome Proteomics In Gallbladder Carcinoma Patients | •Proteinosis •Gallbladder Carcinoma | Pre-clinical phase | [239] |
| 27. | NCT03102268 | ncRNAs in Exosomes of Cholangiocarcinoma | •Cholangiocarcinoma •Benign Biliary stricture | Pre-clinical phase | [240] |
| 28. | NCT03608631 | iExosomes in Treating Participants With Metastatic Pancreas Cancer With KrasG12D Mutation | KRAS NP_004976.2:p.G12D •Metastatic Pancreatic Adenocarcinoma •Pancreatic Ductal Adenocarcinoma •Stage IV Pancreatic Cancer AJCC v8 | Phase 1 | [241] |
| 29. | NCT04053855 | Evaluation of Urinary Exosomes Presence From Clear Cell Renal CellCarcinoma | Clear Cell Renal Cell Carcinoma | Pre-clinical phase | [242] |
| 30. | NCT04453046 | Hemopurifier Plus Pembrolizumab in Head and Neck Cancer | •Squamous Cell Carcinoma of the Head and Neck | Pre-clinical phase | [243] |
| 31. | NCT05218759 | Exosomes Detection for the Prediction of theEfficacy and Adverse Reactions of Anlotinib in Patients With Advanced NSCLC | Non-Small Cell Lung Cancer | Pre-clinical phase | [244] |
| 32. | NCT03811600 | Exosomes Implication in PD1-PD-L1 Activation in OSAS | Sleep Apnea syndromes,Obstructive •Cancer | Pre-clinical phase | [245] |
| 33. | NCT04499794 | The Study of Exosome EML4-ALK Fusion in NSCLC Clinical Diagnosis and Dynamic Monitoring | Untreated Advanced NSCLC Patients •FISH Identified ALK FusionPositive or Negative | Pre-clinical phase | [246] |
| 34. | NCT03830619 | Serum Exosomal Long Noncoding RNAs as Potential Biomarkers for Lung Cancer Diagnosis | Lung Cancer (Diagnosis | Pre-clinical phase | [98] |
| 35. | NCT04427475 | Prediction of Immunotherapeutic Effect of Advanced Non-small Cell Lung Cancer | NSCLC Patients | Pre-clinical phase | [247] |
| 36. | NCT04530890 | Interest of Circulating tumour DNA in Digestive and gynaecologic/Breast Cancer | •Breast Cancer •Digestive Cancer •gynaecologic Cancer •Circulating tumour DNA •Exosomes | Pre-clinical phase | [248] |
| 37. | NCT05101655 | Construction of Microfluidic Exosome Chip for Diagnosis of Lung Metastasis of Osteosarcoma | •Osteosarcoma •Pulmonary Metastases | Pre-clinical phase | [249] |
| 38. | NCT03800121 | Study of Exosomes in Monitoring Patients With Sarcoma (EXOSARC) | •Sarcoma | Pre-clinical phase | [250] |
| 39. | NCT03108677 | Circulating Exosome RNA in Lung Metastasesof Primary High-Grade Osteosarcoma | Lung Metastases •Osteosarcoma | Pre-clinical phase | [251] |
| 40. | NCT03895216 | Identification and Characterization of Predictive Factors of Onset of Bone Metastases in CancerPatients | •Bone Metastases | Pre-clinical phase | [252] |
| 41. | NCT04720599 | Clinical Evaluation of ExoDxProstate(IntelliScore) in Men Presenting for Initial Prostate Biopsy | Urologic Cancer | Pre-clinical phase | [253] |
| 42. | NCT02310451 | Study of Molecular Mechanisms Implicated in the Pathogenesis of Melanoma.Role of Exosomes | •Metastatic Melanoma • | Pre-clinical phase | [254] |
| 43. | NCT04747574 | Evaluation of the Safety of CD24-Exosomes inPatients With COVID-19 Infection | SARS-CoV-2 | Phase 1 - | [255] |
| 44. | NCT04636788 | Circulating Extracellular Exosomal Small RNA as Potential Biomarker for Human PancreaticCancer | •Pancreas Adenocarcinoma | Pre-clinical phase | [256] |
| 45. | NCT04356300 | Exosome of Mesenchymal Stem Cells for Multiple Organ Dysfuntion Syndrome After Surgical Repaire of Acute Type A Aortic Dissection | •Multiple Organ Failure | Pre-clinical phase | [257] |
| 46. | NCT03985696 | Exosomes and Immunotherapy in Non-HodgkinB-cell Lymphomas | Lymphoma, B-cell, Aggressive Non-Hodgkin (B-NHL) | Pre-clinical phase | [258] |
| 47. | NCT04852653 | A Prospective Feasibility Study Evaluating Extracellular Vesicles Obtained by Liquid Biopsy for Neoadjuvant Treatment Response Assessment in Rectal Cancer | Multiple Organ Failure | Pre-clinical phase | [259] |
| 48. | NCT04357717 | ExoDx Prostate Evaluation in Prior NegativeProstate Biopsy Setting | •Prostate Cancer | Pre-clinical phase | [260] |
| 49. | NCT04653740 | Omic Technologies to Track Resistance to Palbociclib in Metastatic Breast Cancer | Advanced Breast Cancer | Pre-clinical phase | [261] |
| 50. | NCT04948437 | Urinary Exosomal Biomarkers of Thyroglobulin and Galectin-3 for Prognosis and Follow-up in Patients of Thyroid Cancer | Thyroid Cancer •Papillary Thyroid Cancer •Follicular Thyroid Cancer | Pre-clinical phase | [101] |
| 51. | NCT03488134 | Predicting Prognosis and Recurrence of Thyroid Cancer Via New Biomarkers, Urinary Exosomal Thyroglobulin and Galectin-3 | •Thyroid Cancer | Pre-clinical phase | [262] |
| 52. | NCT04298398 | Impact of Group Psychological Interventions on Extracellular Vesicles in People Who HadCancer | Cancer | Pre-clinical phase | [263] |
| 53. | NCT02862470 | Anaplastic Thyroid Cancer and FollicularThyroid Cancer-derived Exosomal AnalysisVia Treatment of Lovastatin and Vildagliptin and Pilot Prognostic Study Via Urine Exosomal Biological Markers in Thyroid Cancer Patients | Thyroid Cancer | Pre-clinical phase | [264] |
| 54. | NCT05192694 | Evaluation of Fapi-pet in Prostate Cancer. | •Prostate Cancer | Pre-clinical phase | [265] |
| 55. | NCT03493984 | Plant Exosomes and Patients Diagnosed With Polycystic Ovary Syndrome (PCOS) 17 | •Other: Ginger exosomes •Other: Aloe exosomes •Other: Placebo | Pre-clinical phase | [266] |
| 56. | NCT03228277 | Olmutinib Trial in T790M (+) NSCLC Patients Detected by Liquid Biopsy Using BALFExtracellular Vesicular DNA | Non Small Cell Lung Cancer | •Drug: Olmutinib Phase: Phase 2 | [267] |
| 57. | NCT04182893 | Clinical Study of ctDNA and Exosome Combined Detection to Identify Benign and Malignant Pulmonary Nodules | Pulmonary Nodules | Pre-clinical phase | [268] |
Conclusion and future perspectives
Cancers are the leading cause of death world-wide. Various treatments are employed for the reduction of tumour progression and improving survival rate. Exosomes are natural vesicles secreted from different cells in the body that carry information from one cell to another. Engineering the exosomes through modification of surface membranes or loading of chemotherapeutic agents have shown significant results in reducing tumour progression and metastasis. The observations from various workers have proved the ability of exosomes as delivery vehicles for chemotherapeutic agents and can be considered as a potential drug delivery system in near future. Exosomes are the vesicular structures secreted by cells responsible for transfer of nucleic acids and other cargo between two cellular junctions. Fabrication of the exosomes with aptamers, drug substances enhance exosomal activity in terms of cell proliferation and tumour regression with reduced organ toxicities. According to the report submitted to US FDA, 108 clinical trials were being conducted at different phases using exosomes as the drug delivery vehicles [269]. Studies of exosomes loaded with drugs proved as promising agents in chemotherapy of cancer and expansion of research in this area is the requisite.
CRediT authorship contribution statement
Anita Raj Panigrahi: Writing – original draft. Lankalapalli Srinivas: Conceptualization, Supervision. Jagadeesh Panda: Writing – review & editing.
Declaration of Competing Interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledments
The authors acknowledge GITAM Institute of Pharmacy, GITAM deemed to be University for providing necessary financial support through seed grant, facilities and support in collecting information and review. A special thanks to Biorender platform for creating the original figures for the article.
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.tranon.2022.101439.
Appendix. Supplementary materials
References
- 1.Zaborowski M.P., Balaj L., Breakefield X.O., Lai C.P.K. Extracellular vesicles: composition, biological relevance, and methods of study. Bioscience. 2015;65:783–797. doi: 10.1093/biosci/biv084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pan B.T., Johnstone R.M. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor. Cell. 1983;33(3):967–978. doi: 10.1016/0092-8674(83)90040-5. [DOI] [PubMed] [Google Scholar]
- 3.Xiao Y., Zhong J., Zhong B., Huang J., Jiang L., Jiang Y., Zhong T. Exosomes as potential sources of biomarkers in colorectal cancer. Cancer Lett. 2020;476(September 2019):13–22. doi: 10.1016/j.canlet.2020.01.033. [DOI] [PubMed] [Google Scholar]
- 4.Luan X., Sansanaphongpricha K., Myers I., Chen H., Yuan H., Sun D. Engineering exosomes as refined biological nanoplatforms for drug delivery. Acta Pharmacol. Sin. 2017;38(6):754–763. doi: 10.1038/aps.2017.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen H., Wang L., Zeng X., Schwarz H., Nanda H.S., Peng X., Zhou Y. Exosomes, a new star for targeted delivery. Front. Cell Dev. Biol. 2021;9(October) doi: 10.3389/fcell.2021.751079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mathivanan S., Simpson R.J. ExoCarta: a compendium of exosomal proteins and RNA. Proteomics. 2009;9(21):4997–5000. doi: 10.1002/pmic.200900351. [DOI] [PubMed] [Google Scholar]
- 7.Kalluri R. The biology and function of exosomes in cancer. J. Clin. Invest. 2016;126(4):1208–1215. doi: 10.1172/JCI81135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kibria, G., Ramos, E.K., Wan, Y., Gius, D.R., & Liu H. Exosomes as a drug delivery system in cancer therapy: potential and challenges. 2019;15(9):3625–3633. doi: 10.1021/acs.molpharmaceut.8b00277.Exosomes. [DOI] [PMC free article] [PubMed]
- 9.Antimisiaris S.G., Mourtas S., Marazioti A. Exosomes and exosome-inspired vesicles for targeted drug delivery. Pharmaceutics. 2018;10(4) doi: 10.3390/pharmaceutics10040218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chen S., Lv M., Fang S., Ye W., Gao Y., Xu Y. Poly(I:C) enhanced anti-cervical cancer immunities induced by dendritic cells-derived exosomes. Int. J. Biol. Macromol. 2018;113:1182–1187. doi: 10.1016/j.ijbiomac.2018.02.034. [DOI] [PubMed] [Google Scholar]
- 11.Fu S., Wang Y., Xia X., Zheng J.C. Exosome engineering: current progress in cargo loading and targeted delivery. NanoImpact. 2020;20(August) doi: 10.1016/j.impact.2020.100261. [DOI] [Google Scholar]
- 12.Liang Y., Duan L., Lu J., Xia J. Engineering exosomes for targeted drug delivery. Theranostics. 2021;11(7):3183–3195. doi: 10.7150/THNO.52570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Andreu Z., Yáñez-Mó M. Tetraspanins in extracellular vesicle formation and function. Front. Immunol. 2014;5(SEP):1–12. doi: 10.3389/fimmu.2014.00442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dabrowska S., Del Fattore A., Karnas E., Frontczak-Baniewicz M., Kozlowska H., Muraca M., Lukomska B. Imaging of extracellular vesicles derived from human bone marrow mesenchymal stem cells using fluorescent and magnetic labels. Int. J. Nanomed. 2018;13:1653–1664. doi: 10.2147/IJN.S159404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vaswani K., Koh Y.Q., Almughlliq F.B., Peiris H.N., Mitchell M.D. A method for the isolation and enrichment of purified bovine milk exosomes. Reprod. Biol. 2017;17(4):341–348. doi: 10.1016/j.repbio.2017.09.007. [DOI] [PubMed] [Google Scholar]
- 16.Giacobino C., Canta M., Fornaguera C., Borrós S., Cauda V. Extracellular vesicles and their current role in cancer immunotherapy. Cancers (Basel) 2021;13(9) doi: 10.3390/cancers13092280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jeong H., Park W., Kim D., Na K. Dynamic nanoassemblies of nanomaterials for cancer photomedicine. Adv Drug Deliv Rev. 2021;177 doi: 10.1016/j.addr.2021.113954. [DOI] [PubMed] [Google Scholar]
- 18.Kim J., Song Y., Park C.H., Choi C. Platform technologies and human cell lines for the production of therapeutic exosomes. Extracell Vesicles Circ. Nucleic Acids. 2021:3–17. doi: 10.20517/evcna.2020.01. Published online. [DOI] [Google Scholar]
- 19.Hernandez-Oller L., Seras-Franzoso J., Andrade F., Rafael D., Abasolo I., Gener P., Schwartz S. Extracellular vesicles as drug delivery systems in cancer. Pharmaceutics. 2020;12(12):1–20. doi: 10.3390/pharmaceutics12121146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Weng Z., Zhang B., Wu C., Yu F., Han B., Li B., Li L. Therapeutic roles of mesenchymal stem cell-derived extracellular vesicles in cancer. J. Hematol. Oncol. 2021;14(1):1–22. doi: 10.1186/s13045-021-01141-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang X., Tu H., Yang Y., Fang L., Wu Q., Li J. Mesenchymal stem cell-derived extracellular vesicles: roles in tumor growth, progression, and drug resistance. Stem Cells Int. 2017 doi: 10.1155/2017/1758139. 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Samuel M., Gabrielsson S. Personalized medicine and back–allogeneic exosomes for cancer immunotherapy. J. Intern. Med. 2021;289(2):138–146. doi: 10.1111/joim.12963. [DOI] [PubMed] [Google Scholar]
- 23.Hou Y., Liu Y., Liang S., Ding R., Mo S., Yan D., Li D. The novel target:exosoms derived from M2 macrophage. Int. Rev. Immunol. 2021;40(3):183–196. doi: 10.1080/08830185.2020.1800687. [DOI] [PubMed] [Google Scholar]
- 24.Koh Y.Q., Peiris H.N., Vaswani K., Meier S., Burke C.R., Macdonald K.A., Mitchell M.D. Characterization of exosomes from body fluids of dairy cows. J. Anim. Sci. 2017;95(9):3893. doi: 10.2527/jas2017.1727. [DOI] [PubMed] [Google Scholar]
- 25.Bruno S., Collino F., Deregibus M.C., Grange C., Tetta C., Camussi G. Microvesicles derived from human bone marrow mesenchymal stem cells inhibit tumor growth. Stem Cells Dev. 2013;22(5):758–771. doi: 10.1089/scd.2012.0304. [DOI] [PubMed] [Google Scholar]
- 26.Kandimalla R., Aqil F., Tyagi N., Gupta R. Milk exosomes: a biogenic nanocarrier for small molecules and macromolecules to combat cancer. Am. J. Reprod. Immunol.. Published online. 2020:0–2. doi: 10.1111/aji.13349. [DOI] [PubMed] [Google Scholar]
- 27.Li S. The basic characteristics of extracellular vesicles and their potential application in bone sarcomas. J. Nanobiotechnol. 2021;19(1):1–12. doi: 10.1186/s12951-021-01028-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dad H.A., Gu T.W., Zhu A.Q., Huang L.Q., Peng L.H. Plant exosome-like nanovesicles: emerging therapeutics and drug delivery nanoplatforms. Mol. Ther. 2021;29(1):13–31. doi: 10.1016/j.ymthe.2020.11.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Schuh C.M.A.P., Cuenca J., Alcayaga-Miranda F., Khoury M. Exosomes on the border of species and kingdom intercommunication. Transl. Res. 2019;210:80–98. doi: 10.1016/j.trsl.2019.03.008. [DOI] [PubMed] [Google Scholar]
- 30.Jha C., Ghosh S., Gautam V., Malhotra P., Ray P. In vitro study of virulence potential of Acinetobacter baumannii outer membrane vesicles. Microb. Pathog. 2017;111:218–224. doi: 10.1016/j.micpath.2017.08.048. [DOI] [PubMed] [Google Scholar]
- 31.Zhao W., Hou X., Vick O.G., Dong Y. RNA delivery biomaterials for the treatment of genetic and rare diseases. Biomaterials. 2019;217(April) doi: 10.1016/j.biomaterials.2019.119291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mittal S., Gupta P., Chaluvally-Raghavan P., Pradeep S. Emerging role of extracellular vesicles in immune regulation and cancer progression. Cancers. 2020;12(12):1–16. doi: 10.3390/cancers12123563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bondhopadhyay B., Sisodiya S., Alzahrani F.A., Bakhrebah M.A., Chikara A., Kasherwal V., Exosomes Hussain S. A forthcoming era of breast cancer therapeutics. Cancers (Basel) 2021;13(18):1–18. doi: 10.3390/cancers13184672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mendt M., Kamerkar S., Sugimoto H., McAndrews K.M., Wu C.C., Gagea M., Kalluri R. Generation and testing of clinical-grade exosomes for pancreatic cancer. JCI insight. 2018;3(8):1–22. doi: 10.1172/jci.insight.99263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sedykh S., Kuleshova A., Nevinsky G. Milk exosomes: perspective agents for anticancer drug delivery. Int. J. Mol. Sci. 2020;21(18):1–16. doi: 10.3390/ijms21186646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Li B., Hock A., Wu R.Y., Minich A., Botts S.R., Lee C., Pierro A. Bovine milk-derived exosomes enhance goblet cell activity and prevent the development of experimental necrotizing enterocolitis. PLoS ONE. 2019;14(1):1–12. doi: 10.1371/journal.pone.0211431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Admyre C., Johansson S.M., Qazi K.R., Filén J.-.J., Lahesmaa R., Norman M., Gabrielsson S. Exosomes with immune modulatory features are present in human breast milk. J. Immunol. 2007;179(3):1969–1978. doi: 10.4049/jimmunol.179.3.1969. [DOI] [PubMed] [Google Scholar]
- 38.Yamauchi M., Shimizu K., Rahman M., Ishikawa H., Takase H., Ugawa S., Inoshima Y. Efficient method for isolation of exosomes from raw bovine milk. Drug Dev. Ind. Pharm. 2019;45(3):359–364. doi: 10.1080/03639045.2018.1539743. [DOI] [PubMed] [Google Scholar]
- 39.Del Pozo-Acebo L., de las Hazas M.C.L., Tomé-Carneiro J., Gil-Cabrerizo P., San-Cristobal R., Busto R. Dávalos A. Bovine milk-derived exosomes as a drug delivery vehicle for mirna-based therapy. Int. J. Mol. Sci. 2021;22(3):1–19. doi: 10.3390/ijms22031105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dai J., Su Y., Zhong S., Cong L., Liu B., Yang J., Jiang Y. Exosomes: key players in cancer and potential therapeutic strategy. Signal Transduct Target Ther. 2020;5(1) doi: 10.1038/s41392-020-00261-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ferguson S.W., Nguyen J. Exosomes as therapeutics: the implications of molecular composition and exosomal heterogeneity. J. Control Release. 2016;228:179–190. doi: 10.1016/j.jconrel.2016.02.037. [DOI] [PubMed] [Google Scholar]
- 42.Yeo R.W.Y., Lai R.C., Zhang B., Tan S.S., Yin Y., Teh B.J., Lim S.K. Mesenchymal stem cell: an efficient mass producer of exosomes for drug delivery. Adv. Drug Deliv. Rev. 2013;65(3):336–341. doi: 10.1016/j.addr.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 43.Norman P Spack, Daniel E., Shumer N.J.N. The phenotypic effects of exosomes secreted from distinct cellular sources: a comparative study based on miRNA composition. Physiol. Behav. 2017;176(12):139–148. doi: 10.1208/s12248-018-0227-4.The. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhang S., Hou Y., Yang J., Xie D., Jiang L., Hu H., Zhang Q. Application of mesenchymal stem cell exosomes and their drug-loading systems in acute liver failure. J. Cell Mol. Med. 2020;24(13):7082–7093. doi: 10.1111/jcmm.15290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pitt Jonathan M., André Fabrice, Amigorena Sebastian, Soria Jean-Charles, Alexander Eggermont G.K. LZ. Dendritic cell-derived exosomes for cancer therapy. J Clin Invest. 2016;126(4):1224–1232. doi: 10.1172/JCI81137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Pitt J.M., André F., Amigorena S., Soria J.C., Eggermont A., Kroemer G., Zitvogel L. Dendritic cell-derived exosomes for cancer therapy. J. Clin. Invest. 2016;126(4):1224–1232. doi: 10.1172/JCI81137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Viaud S., Terme M., Flament C., Taieb J., André F., Novault S., Chaput N. Dendritic cell-derived exosomes promote natural killer cell activation and proliferation: a role for NKG2D ligands and IL-15Rα. PLoS ONE. 2009;4(3) doi: 10.1371/journal.pone.0004942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Besse B., Charrier M., Lapierre V., Dansin E., Lantz O., Planchard D., Chaput N. Dendritic cell-derived exosomes as maintenance immunotherapy after first line chemotherapy in NSCLC. Oncoimmunology. 2016;5(4) doi: 10.1080/2162402X.2015.1071008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Dai S., Wei D., Wu Z., Zhou X., Wei X., Huang H., Li G. Phase I clinical trial of autologous ascites-derived exosomes combined with GM-CSF for colorectal cancer. Mol Ther. 2008;16(4):782–790. doi: 10.1038/mt.2008.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ju S., Mu J., Dokland T., Zhuang X., Wang Q., Jiang H., Zhang H.G. Grape exosome-like nanoparticles induce intestinal stem cells and protect mice from DSS-induced colitis. Mol. Ther. 2013;21(7):1345–1357. doi: 10.1038/mt.2013.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wang Q., Ren Y., Mu J., Egilmez N.K., Zhuang X., Deng Z., Zhang H.G. Grapefruit-derived nanovectors use an activated leukocyte trafficking pathway to deliver therapeutic agents to inflammatory tumor sites. Cancer Res. 2015;75(12):2520–2529. doi: 10.1158/0008-5472.CAN-14-3095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhuang X., Deng Z.B., Mu J., Zhang L., Yan J., Miller D., Zhang H.G. Ginger-derived nanoparticles protect against alcohol-induced liver damage. J. Extracell. Vesicles. 2015;4(1):1–18. doi: 10.3402/jev.v4.28713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Chen Y.S., Lin E.Y., Chiou T.W., Harn H.J. Exosomes in clinical trial and their production in compliance with good manufacturing practice. Tzu Chi Med. J. 2020;32(2):113–120. doi: 10.4103/tcmj.tcmj_182_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Samuel M., Bleackley M., Anderson M., Mathivanan S. Extracellular vesicles including exosomes in cross kingdom regulation: a viewpoint from plant-fungal interactions. Front. Plant Sci. 2015;6(September):1–5. doi: 10.3389/fpls.2015.00766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Chukhchin D.G., Bolotova K., Sinelnikov I., Churilov D., Novozhilov E. Exosomes in the phloem and xylem of woody plants. Planta. 2020;251(1):1–14. doi: 10.1007/s00425-019-03315-y. [DOI] [PubMed] [Google Scholar]
- 56.Hansen L.L., Nielsen M.E. Plant exosomes: using an unconventional exit to prevent pathogen entry? J. Exp. Bot. 2017;69(1):59–68. doi: 10.1093/jxb/erx319. [DOI] [PubMed] [Google Scholar]
- 57.An Q., Van Bel A.J.E., Hückelhoven R. Do plant cells secrete exosomes derived from multivesicular bodies? Plant Signal Behav. 2007;2(1):4–7. doi: 10.4161/psb.2.1.3596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Rutter B.D., Innes R.W. Extracellular vesicles isolated from the leaf apoplast carry stress-response proteins. Plant Physiol. 2017;173(1):728–741. doi: 10.1104/pp.16.01253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhuang X., Teng Y., Samykutty A., Mu J., Deng Z., Zhang L., Zhang H.G. Grapefruit-derived nanovectors delivering therapeutic miR17 through an intranasal route inhibit brain tumor progression. Mol. Ther. 2016;24(1):96–105. doi: 10.1038/mt.2015.188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lapinet J.A., Scapini P., Calzetti F., Pérez O., Cassatella M.A. Gene expression and production of tumor necrosis factor alpha, interleukin-1β (IL-1β), IL-8, macrophage inflammatory protein 1α (MIP-1α), MIP-1β, and gamma interferon-inducible protein 10 by human neutrophils stimulated with group B meningococcal outer me. Infect. Immun. 2000;68(12):6917–6923. doi: 10.1128/IAI.68.12.6917-6923.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cecil J.D., O'Brien-Simpson N.M., Lenzo J.C., Holden J.A., Singleton W., Perez-Gonzalez A., Reynolds E.C. Outer membrane vesicles prime and activate macrophage inflammasomes and cytokine secretion in vitro and in vivo. Front. Immunol. 2017;8(AUG):1–22. doi: 10.3389/fimmu.2017.01017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Shen Y., Torchia M.L.G., Lawson G.W., Karp C.L., Ashwell J.D., Mazmanian S.K. Outer membrane vesicles of a human commensal mediate immune regulation and disease protection. Cell Host Microbe. 2012;12(4):509–520. doi: 10.1016/j.chom.2012.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Grenier D., Bertrand J., Mayrand D. Porphyromonas gingivalis outer membrane vesicles promote bacterial resistance to chlorhexidine. Oral Microbiol. Immunol. 1995;10(5):319–320. doi: 10.1111/j.1399-302X.1995.tb00161.x. [DOI] [PubMed] [Google Scholar]
- 64.Ciofu O., Beveridge T.J., Kadurugamuwa J., Walther-Rasmussen J., Høiby N. Chromosomal β-lactamase is packaged into membrane vesicles and secreted from Pseudomonas aeruginosa. J. Antimicrob. Chemother. 2000;45(1):9–13. doi: 10.1093/jac/45.1.9. [DOI] [PubMed] [Google Scholar]
- 65.Sun Z., Zhang J., Li J., Li M., Ge J., Wu P., Qian H. Roles of mesenchymal stem cell-derived exosomes in cancer development and targeted therapy. Stem Cells Int. 2021:2021. doi: 10.1155/2021/9962194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Fu P., Zhang J., Li H., Mak M., Xu W., Tao Z. Extracellular vesicles as delivery systems at nano-/micro-scale. Adv. Drug Deliv. Rev. 2021;(xxxx) doi: 10.1016/j.addr.2021.113910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Savina A., Furlán M., Vidal M., Colombo M.I. Exosome release is regulated by a calcium-dependent mechanism in K562 cells. J. Biol. Chem. 2003;278(22):20083–20090. doi: 10.1074/jbc.M301642200. [DOI] [PubMed] [Google Scholar]
- 68.Datta A., Kim H., McGee L., Johnson A.E., Talwar S., Marugan J., Abdel-Mageed A.B. High-Throughput screening identified selective inhibitors of exosome biogenesis and secretion: a drug repurposing strategy for advanced cancer. Sci. Rep. 2018;8(1):1–13. doi: 10.1038/s41598-018-26411-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Perocheau D., Touramanidou L., Gurung S., Gissen P., Baruteau J. Clinical applications for exosomes: are we there yet? Br. J. Pharmacol. 2021;178(12):2375–2392. doi: 10.1111/bph.15432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Osterman C.J.D., Lynch J.C., Leaf P., Gonda A., Bennit H.R.F., Griffiths D. & Wall NR. Curcumin modulates pancreatic adenocarcinoma cell-derived exosomal function. PLoS ONE. 2015;10(7):1–17. doi: 10.1371/journal.pone.0132845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Carmen Gutierrezz-Millan, Clara Calvo Diaz JML and CIC. advances in exosomes-based drug delivery systems _ Enhanced Reader.pdf. [DOI] [PubMed]
- 72.Yakimchuk K. Exosomes: isolation methods and specific markers. Mater. Methods. 2015;5:1–17. doi: 10.13070/mm.en.5.1450. [DOI] [Google Scholar]
- 73.Fisher W.S., Tchounwou C., Ewert K.K., Wei S., Roberts L. Exosomes are secreted at distinct densities in human cancer cells and are promising vectors for hydrophobic drug delivery. Biophys. J. 2021;120(3):273a. doi: 10.1016/j.bpj.2020.11.1740. [DOI] [Google Scholar]
- 74.Alzhrani G.N., Alanazi S.T., Alsharif S.Y., Albalawi A.M., Alsharif A.A., Abdel-Maksoud M.S., Elsherbiny N. Exosomes: isolation, characterization, and biomedical applications. Cell Biol. Int. 2021;45(9):1807–1831. doi: 10.1002/cbin.11620. [DOI] [PubMed] [Google Scholar]
- 75.Zhang H., Lyden D. Asymmetric-flow field-flow fractionation technology for exomere and small extracellular vesicle separation and characterization. Nat. Protoc. 2019;14(4):1027–1053. doi: 10.1038/s41596-019-0126-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Müller Bark J., Kulasinghe A., Amenábar J.M., Punyadeera C. Exosomes in cancer. Adv. Clin. Chem. 2021;101:1–40. doi: 10.1016/bs.acc.2020.06.006. [DOI] [PubMed] [Google Scholar]
- 77.Gilligan K.E., Dwyer R.M. Engineering exosomes for cancer therapy. Int. J. Mol. Sci. 2017;18(6) doi: 10.3390/ijms18061122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Severic M., Ma G., Pereira S.G.T., Ruiz A., Cheung C.C.L., WT Al-Jamal. Genetically-engineered anti-PSMA exosome mimetics targeting advanced prostate cancer in vitro and in vivo. J. Control Release. 2021;330:101–110. doi: 10.1016/j.jconrel.2020.12.017. [DOI] [PubMed] [Google Scholar]
- 79.Ludwig N., Whiteside T.L., Reichert T.E. Challenges in exosome isolation and analysis in health and disease. Int. J. Mol. Sci. 2019;20(19) doi: 10.3390/ijms20194684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zhu L., Sun H.T., Wang S., Huang S.L., Zheng Y., Wang C.Q., Dong Q.Z. Isolation and characterization of exosomes for cancer research. J. Hematol. Oncol. 2020;13(1):1–24. doi: 10.1186/s13045-020-00987-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Li P., Kaslan M., Lee S.H., Yao J., Gao Z. Progress in exosome isolation techniques. Theranostics. 2017;7(3):789–804. doi: 10.7150/thno.18133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yang D., Zhang W., Zhang H., Zhang F., Chen L., Ma L., Wang T. Progress, opportunity, and perspective on exosome isolation - Efforts for efficient exosome-based theranostics. Theranostics. 2020;10(8):3684–3707. doi: 10.7150/thno.41580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Liga A., Vliegenthart A.D.B., Oosthuyzen W., Dear J.W., Kersaudy-Kerhoas M. Exosome isolation: a microfluidic road-map. Lab Chip. 2015;15(11):2388–2394. doi: 10.1039/c5lc00240k. [DOI] [PubMed] [Google Scholar]
- 84.Kadiu I., Narayanasamy P., Dash P.K., Zhang W., Gendelman H.E. Biochemical and biologic characterization of exosomes and microvesicles as facilitators of HIV-1 infection in macrophages. J. Immunol. 2012;189(2):744–754. doi: 10.4049/jimmunol.1102244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Koritzinsky E.H., Street J.M., Star R.A., Yuen P.S.T. Quantification of exosomes. J. Cell Physiol. 2017;232(7):1587–1590. doi: 10.1002/jcp.25387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Patil S.M., Sawant S.S., Kunda N.K. Exosomes as drug delivery systems: a brief overview and progress update. Eur. J. Pharm. Biopharm. 2020;154:259–269. doi: 10.1016/j.ejpb.2020.07.026. [DOI] [PubMed] [Google Scholar]
- 87.Lötvall, J., Hill, A.F., Hochberg, F., Buzás, E.I., Vizio, D.D., Gardiner, C., Théry C. Minimal experimental requirements for definition of extracellular vesicles and their functions. Published online 2014. doi: 10.3402/jev.v3.26913. [DOI] [PMC free article] [PubMed]
- 88.Carr B., Malloy A., Warren J. Nanoparticle tracking analysis. Innov. Pharm. Technol. 2008;44(26):38–40. doi: 10.12681/osj.22598. [DOI] [Google Scholar]
- 89.Deb A., Gupta S., Mazumder P.B. Exosomes: a new horizon in modern medicine. Life Sci. 2021;264(September 2020) doi: 10.1016/j.lfs.2020.118623. [DOI] [PubMed] [Google Scholar]
- 90.Van Der Pol E., Van Gemert M.J.C., Sturk A., Nieuwland R., Van Leeuwen T.G. Single vs. swarm detection of microparticles and exosomes by flow cytometry. J. Thromb. Haemost. 2012;10(5):919–930. doi: 10.1111/j.1538-7836.2012.04683.x. [DOI] [PubMed] [Google Scholar]
- 91.Vashisht M., Rani P., Sunita Onteru SK, Singh D. Curcumin primed exosomes reverses LPS-induced pro-inflammatory gene expression in. Buffalo Granulosa Cells. 2018;119 doi: 10.1002/jcb.26309. Vol. [DOI] [PubMed] [Google Scholar]
- 92.Han D., Wang K., Zhang T., Gao G.C., Xu H. Natural killer cell-derived exosome-entrapped paclitaxel can enhance its anti-tumor effect. Eur. Rev. Med. Pharmacol. Sci. 2020;24(10):5703–5713. doi: 10.26355/eurrev_202005_21362. [DOI] [PubMed] [Google Scholar]
- 93.Doyle L., Wang M. Overview of extracellular vesicles, their origin, composition, purpose, and methods for exosome isolation and analysis. Cells. 2019;8(7):727. doi: 10.3390/cells8070727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Mirzaaghasi A., Han Y., Ahn S.H., Choi C., Park J.H. Biodistribution and pharmacokinectics of liposomes and exosomes in a mouse model of sepsis. Pharmaceutics. 2021;13(3):1–9. doi: 10.3390/pharmaceutics13030427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Nicolini A., Ferrari P., Biava P.M. Exosomes and cell communication: from tumour-derived exosomes and their role in tumour progression to the use of exosomal cargo for cancer treatment. Cancers (Basel) 2021;13(4):1–24. doi: 10.3390/cancers13040822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Jabbari N., Akbariazar E., Feqhhi M., Rahbarghazi R., Rezaie J. Breast cancer-derived exosomes: tumor progression and therapeutic agents. J. Cell Physiol. 2020;235(10):6345–6356. doi: 10.1002/jcp.29668. [DOI] [PubMed] [Google Scholar]
- 97.Tai Y.L., Chen K.C., Hsieh J.T., Shen T.L. Exosomes in cancer development and clinical applications. Cancer Sci. 2018;109(8):2364–2374. doi: 10.1111/cas.13697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Yang Jin, professor, Wuhan union hospital C. Serum exosomal long noncoding RNAs as potential biomarkers for lung cancer diagnosis. Published online 2021:1–5.
- 99.Zhu Ji. Published online; 2020. Fudan University Shanghai Cancer Center Shanghai C. Study on Predictive Biomarkers of Neoadjuvant Chemoradiotherapy For Rectal Cancer; pp. 1–9. [Google Scholar]
- 100.Pazo-Cid, Roberto A., Medical oncology Department MSUH, Zaragoza S. Exosomes as potential prognostic and predictive biomarkers in advanced gastric cancer patients (" EXO-PPP Study ") (EXO-PPP). (Cdc):1–7.
- 101.WANG, CHIH-YUAN, PEI-JIE HUANG NTUH. Exosomes urinary exosomal biomarkers of thyroglobulin and galectin-3 for prognosis and follow-up in patients of thyroid cancer. Published online 2021:1–8.
- 102.Inamdar S., Nitiyanandan R., Rege K. Emerging applications of exosomes in cancer therapeutics and diagnostics. Bioeng. Transl. Med. 2017;2(1):70–80. doi: 10.1002/btm2.10059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Limoni S.K., Moghadam M.F., Moazzeni S.M., Gomari H., Salimi F. Engineered exosomes for targeted transfer of siRNA to HER2 positive breast cancer cells. Appl. Biochem. Biotechnol. 2019;187(1):352–364. doi: 10.1007/s12010-018-2813-4. [DOI] [PubMed] [Google Scholar]
- 104.Park O., Choi E.S., Yu G., Kim J.Y., Kang Y.Y., Jung H., Mok H. Efficient delivery of tyrosinase related protein-2 (TRP2) peptides to lymph nodes using serum-derived exosomes. Macromol. Biosci. 2018;18(12):6–11. doi: 10.1002/mabi.201800301. [DOI] [PubMed] [Google Scholar]
- 105.Zhao L., Gu C., Gan Y., Shao L., Chen H., Zhu H. Exosome-mediated siRNA delivery to suppress postoperative breast cancer metastasis. J. Control Release. 2020;318:1–15. doi: 10.1016/j.jconrel.2019.12.005. [DOI] [PubMed] [Google Scholar]
- 106.Zhang X., Zhang H., Gu J., Zhang J., Shi H., Qian H., Santos H.A. Engineered extracellular vesicles for cancer therapy. Adv. Mater. 2021;33(14):1–25. doi: 10.1002/adma.202005709. [DOI] [PubMed] [Google Scholar]
- 107.Fuhrmann G., Serio A., Mazo M., Nair R., Stevens M.M. Active loading into extracellular vesicles significantly improves the cellular uptake and photodynamic effect of porphyrins. J. Control Release. 2015;205:35–44. doi: 10.1016/j.jconrel.2014.11.029. [DOI] [PubMed] [Google Scholar]
- 108.Myung Soo Kim, Matthew J. Haney, Yuling Zhao, Vivek Mahajan, Irina Dey- gen, Natalia L. Klyachko, Eli Inskoe, Aleksandr Piroyan, Marina Sokolsky, Onyi Okolie, Shawn D. Hingtgen, Alexander V. KabanovEVB. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomed. Nanotechnol., Biol. Med.. 2016;12(3):655–664. doi: 10.1016/J.NANO.2015.10.012. [DOI] [PMC free article] [PubMed]
- 109.ang T., Martin P., Fogarty B., Brown A., Schurman K., Phipps R., Bai S. Exosome delivered anticancer drugs across the blood-brain barrier for brain cancer therapy in Danio Rerio. Pharm. Res. 2015;32(6):2003–2014. doi: 10.1007/s11095-014-1593-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Wang J., Chen D., Ho E.A. Challenges in the development and establishment of exosome-based drug delivery systems. J. Control Release. 2021;329:894–906. doi: 10.1016/j.jconrel.2020.10.020. [DOI] [PubMed] [Google Scholar]
- 111.Yu M., Gai C., Li Z., Ding D., Zheng J., Zhang W., Li W. Targeted exosome-encapsulated erastin induced ferroptosis in triple negative breast cancer cells. Cancer Sci. 2019;110(10):3173–3182. doi: 10.1111/cas.14181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Tsong T.Y. Electroporation of cell membranes. Biophysical journal. 1991;60(2):297–306. doi: 10.1016/S0006-3495(91)82054-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Peng H., Ji W., Zhao R., Yang J., Lu Z., Li Y., Zhang X. Exosome: a significant nano-scale drug delivery carrier. J. Mater. Chem. B. 2020;8(34):7591–7608. doi: 10.1039/d0tb01499k. [DOI] [PubMed] [Google Scholar]
- 114.Lu M., Huang Y. Bioinspired exosome-like therapeutics and delivery nanoplatforms. Biomaterials. 2020;242(February) doi: 10.1016/j.biomaterials.2020.119925. [DOI] [PubMed] [Google Scholar]
- 115.Liang G., Zhu Y., Ali D.J., Tian T., Xu H., Si K., Xiao Z. Engineered exosomes for targeted co-delivery of miR-21 inhibitor and chemotherapeutics to reverse drug resistance in colon cancer. J. Nanobiotechnol. 2020;18(1):1–15. doi: 10.1186/s12951-019-0563-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Johnsen K.B., Gudbergsson J.M., Skov M.N., Christiansen G., Gurevich L., Moos T., Duroux M. Evaluation of electroporation-induced adverse effects on adipose-derived stem cell exosomes. Cytotechnology. 2016;68(5):2125–2138. doi: 10.1007/s10616-016-9952-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Sun H., Burrola S., Wu J., Ding W.Q. Extracellular vesicles in the development of cancer therapeutics. Int. J. Mol. Sci. 2020;21(17):1–22. doi: 10.3390/ijms21176097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Goh W.J., Lee C.K., Zou S., Woon E.C.Y., Czarny B., Pastorin G. Doxorubicin-loaded cell-derived nanovesicles: an alternative targeted approach for anti-tumor therapy. Int. J. Nanomed. 2017;12:2759–2767. doi: 10.2147/IJN.S131786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Salarpour S., Forootanfar H., Pournamdari M., Ahmadi-Zeidabadi M., Esmaeeli M., Pardakhty A. Paclitaxel incorporated exosomes derived from glioblastoma cells: comparative study of two loading techniques. DARU, J. Pharm. Sci. 2019;27(2):533–539. doi: 10.1007/s40199-019-00280-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Sancho-Albero M., Encabo-Berzosa M.D.M., Beltrán-Visiedo M., Fernández-Messina L., Sebastián V., Sánchez-Madrid F., Martín-Duque P. Efficient encapsulation of theranostic nanoparticles in cell-derived exosomes: leveraging the exosomal biogenesis pathway to obtain hollow gold. Nanoparticle-Hybrids. 2019;11 doi: 10.1039/c9nr06183e. [DOI] [PubMed] [Google Scholar]
- 121.Nizamudeen Z.A., Xerri R., Parmenter C., Suain K., Markus R., Chakrabarti L. & Sottile V. Low-power sonication can alter extracellular vesicle size and properties. Cells. Published online. 2021 doi: 10.3390/cells10092413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Luan X., Sansanaphongpricha K., Myers I., Chen H., Yuan H., Sun D. Engineering exosomes as refined biological nanoplatforms for drug delivery. Nature Publishing Group. 2017;38 doi: 10.1038/aps.2017.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Xi X.M., Chen-Meng Xia SJ, Lu R. Drug loading techniques for exosome-based drug delivery systems. Pharmazie. 2021;76(2–3):61–67. doi: 10.1691/ph.2021.0128. [DOI] [PubMed] [Google Scholar]
- 124.Ullah M., Qian N.P.M., Yannarelli G. Advances in innovative exosome-technology for real time monitoring of viable drugs in clinical translation, prognosis and treatment response. Oncotarget. 2021;12(11):1029–1031. doi: 10.18632/ONCOTARGET.27927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Tian T., Zhang H.X., He C.P., Fan S., Zhu Y.L., Qi C., Gao J. Surface functionalized exosomes as targeted drug delivery vehicles for cerebral ischemia therapy. Biomaterials. 2018;150:137–149. doi: 10.1016/j.biomaterials.2017.10.012. [DOI] [PubMed] [Google Scholar]
- 126.Bell B.M., Kirk I.D., Hiltbrunner S., Gabrielsson S., Bultema J.J. Designer exosomes as next-generation cancer immunotherapy. Nanomed. Nanotechnol., Biol. Med.. 2016;12(1):163–169. doi: 10.1016/j.nano.2015.09.011. [DOI] [PubMed] [Google Scholar]
- 127.Zhao Z., Zlokovic B.V. Remote control of BBB: a tale of exosomes and microRNA. Cell Res. 2017;27(7):849–850. doi: 10.1038/cr.2017.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Yang Y., Hong Y., Cho E., Kim G.B., Kim I.S. Extracellular vesicles as a platform for membrane-associated therapeutic protein delivery. J. Extracell. Vesicles. 2018;7(1) doi: 10.1080/20013078.2018.1440131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Alvarez-Erviti L., Seow Y., Yin H., Betts C., Lakhal S., Wood M.J.A. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol. 2011;29(4):341–345. doi: 10.1038/nbt.1807. [DOI] [PubMed] [Google Scholar]
- 130.Lathwal S., Yerneni S.S., Boye S., Muza U.L., Takahashi S., Sugimoto N., Matyjaszewski K. Engineering exosome polymer hybrids by atom transfer radical polymerization. Proc. Natl. Acad. Sci. U S A. 2021;118(2):1–11. doi: 10.1073/pnas.2020241118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Bai J., Duan J., Liu R., Du Y., Luo Q., Cui Y., Lu W. Engineered targeting tLyp-1 exosomes as gene therapy vectors for efficient delivery of siRNA into lung cancer cells. Asian J. Pharm. Sci. 2020;15(4):461–471. doi: 10.1016/j.ajps.2019.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Tian Y., Li S., Song J., Ji T., Zhu M., Anderson G.J., Nie G. A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials. 2014;35(7):2383–2390. doi: 10.1016/j.biomaterials.2013.11.083. [DOI] [PubMed] [Google Scholar]
- 133.Sugahara K.N., Teesalu T., Prakash Karmali P., Ramana Kotamraju V., Agemy L., Greenwald D.R., Ruoslahti E. Coadministration of a tumor-penetrating peptide enhances the efficacy of cancer drugs. Science (80) 2010;328(5981):1031–1035. doi: 10.1126/science.1183057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Kondo T., Oka T., Sato H., Shinnou Y., Washio K. Aptamer-functionalized exosomes: elucidating the cellular uptake mechanism and the potential for cancer-targeted chemotherapy. Int. J. Oncol. 2019;35(3):2383–2390. doi: 10.3892/ijo. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Zhu Z., Wen Y., Xuan C., Chen Q., Xiang Q., Wang J., Zhao Z. Identifying the key genes and microRNAs in prostate cancer bone metastasis by bioinformatics analysis. FEBS Open Bio. 2020;10(4):674–688. doi: 10.1002/2211-5463.12805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Gong C., Tian J., Wang Z., Gao Y., Wu X., Ding X., Gao S. Functional exosome-mediated co-delivery of doxorubicin and hydrophobically modified microRNA 159 for triple-negative breast cancer therapy. J. Nanobiotechnol. 2019;17(1):1–18. doi: 10.1186/s12951-019-0526-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Jia G., Han Y., An Y., Ding Y., He C., Wang X., Tang Q. Vol. 178. Elsevier Ltd; 2018. (NRP-1 Targeted and Cargo-Loaded Exosomes Facilitate Simultaneous Imaging and Therapy of Glioma in Vitro and in Vivo). [DOI] [PubMed] [Google Scholar]
- 138.Sun S., Lei Y., Li Q., Wu Y., Zhang L., Mu P.P., Gao D.S. Neuropilin-1 is a glial cell line-derived neurotrophic factor receptor in glioblastoma. Oncotarget. 2017;8(43):74019–74035. doi: 10.18632/oncotarget.18630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Yi Y.W., Lee J.H., Kim S.Y., Pack C.G., Ha D.H., Park S.R., Cho B.S. Advances in analysis of biodistribution of exosomes by molecular imaging. Int. J. Mol. Sci. 2020;21(2) doi: 10.3390/ijms21020665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Jafari D., Shajari S., Jafari R., Mardi N., Gomari H., Ganji F., Samadikuchaksaraei A. Designer exosomes: a new platform for biotechnology therapeutics. BioDrugs. 2020;34(5):567–586. doi: 10.1007/s40259-020-00434-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Revia R.A., Stephen Z.R., Zhang M. Theranostic nanoparticles for RNA-based cancer treatment. Acc. Chem. Res. 2019;52(6):1496–1506. doi: 10.1021/acs.accounts.9b00101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Sato Y.T., Umezaki K., Sawada S., Mukai S.A., Sasaki Y., Harada N., Akiyoshi K. Engineering hybrid exosomes by membrane fusion with liposomes. Sci. Rep. 2016;6(June 2015):1–11. doi: 10.1038/srep21933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Chen Y., Deng Y., Zhu C., Xiang C. Anti prostate cancer therapy: aptamer-functionalized, curcumin and cabazitaxel co-delivered, tumor targeted lipid-polymer hybrid nanoparticles. Biomed. Pharmacother. 2020;127(200) doi: 10.1016/j.biopha.2020.110181. [DOI] [PubMed] [Google Scholar]
- 144.Choi H., Choi Y., Yim H.Y., Mirzaaghasi A., Yoo J.K., Choi C. Biodistribution of exosomes and engineering strategies for targeted delivery of therapeutic exosomes. Tissue Eng. Regen. Med. 2021;18(4):499–511. doi: 10.1007/s13770-021-00361-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Morishita M., Takahashi Y., Nishikawa M., Takakura Y. Pharmacokinetics of exosomes—an important factor for elucidating the biological roles of exosomes and for the development of exosome-based therapeutics. J. Pharm. Sci. 2017;106(9):2265–2269. doi: 10.1016/j.xphs.2017.02.030. [DOI] [PubMed] [Google Scholar]
- 146.Grange C., Tapparo M., Bruno S., Chatterjee D., Quesenberry P.J., Tetta C., Camussi G. Biodistribution of mesenchymal stem cell-derived extracellular vesicles in a model of acute kidney injury monitored by optical imaging. Int. J. Mol. Med. 2014;33(5):1055–1063. doi: 10.3892/ijmm.2014.1663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Kotmakçı M., Çetintaş V.B. Extracellular vesicles as natural nanosized delivery systems for small-molecule drugs and genetic material: steps towards the future nanomedicines. J. Pharm. Pharm. Sci. 2015;18(3):396–413. doi: 10.18433/J36W3X. [DOI] [PubMed] [Google Scholar]
- 148.Saleh A.F., Lázaro-Ibáñez E., Forsgard M.A.M., Shatnyeva O., Osteikoetxea X., Karlsson F., Edmunds N. Extracellular vesicles induce minimal hepatotoxicity and immunogenicity. Nanoscale. 2019;11(14):6990–7001. doi: 10.1039/c8nr08720b. [DOI] [PubMed] [Google Scholar]
- 149.Qiao L., Hu S., Huang K., Su T., Li Z., Vandergriff A., Cheng K. Tumor cell-derived exosomes home to their cells of origin and can be used as Trojan horses to deliver cancer drugs. Theranostics. 2020;10(8):3474–3487. doi: 10.7150/thno.39434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Mohammadi S., Yousefi F., Shabaninejad Z., Movahedpour A., Mahjoubin Tehran M., Shafiee A., Mirzaei H. Exosomes and cancer: from oncogenic roles to therapeutic applications. IUBMB Life. 2020;72(4):724–748. doi: 10.1002/iub.2182. [DOI] [PubMed] [Google Scholar]
- 151.Parada N., Romero-Trujillo A., Georges N., Alcayaga-Miranda F. Camouflage strategies for therapeutic exosomes evasion from phagocytosis. J. Adv. Res. 2021;31:61–74. doi: 10.1016/j.jare.2021.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Suetsugu A., Honma K., Saji S., Moriwaki H., Ochiya T., Hoffman R.M. Imaging exosome transfer from breast cancer cells to stroma at metastatic sites in orthotopic nude-mouse models. Adv. Drug Deliv. Rev. 2013;65(3):383–390. doi: 10.1016/j.addr.2012.08.007. [DOI] [PubMed] [Google Scholar]
- 153.Jung K.O., Jo H., Yu J.H., Gambhir S.S., Pratx G. Development and MPI tracking of novel hypoxia-targeted theranostic exosomes. Biomaterials. 2018;177:139–148. doi: 10.1016/j.biomaterials.2018.05.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Smyth Tyson, Kullberg Max, Malik Noeen, Smith-Jones Peter, Graner Michael W. TJA. Biodistribution and delivery efficiency of unmodified tumor- derived exosomes. Physiol. Behav. 2017;176(3):139–148. doi: 10.1016/j.jconrel.2014.12.013. doi:10.1016/j.jconrel.2014.12.013.Biodistribution. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Leal A.C., Mizurini D.M., Gomes T., Rochael N.C., Saraiva E.M., Dias M.S., Monteiro R.Q. Tumor-derived exosomes induce the formation of neutrophil extracellular traps: implications for the establishment of cancer-associated thrombosis. Sci. Rep. 2017;7(1):1–12. doi: 10.1038/s41598-017-06893-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Pérez-Herrero E., Fernández-Medarde A. Advanced targeted therapies in cancer: drug nanocarriers, the future of chemotherapy. Eur. J. Pharm. Biopharm. 2015;93(March):52–79. doi: 10.1016/j.ejpb.2015.03.018. [DOI] [PubMed] [Google Scholar]
- 157.National Cancer Institute . 2015. Types of Cancer treatment. Nih. Published online. 1-4. http://www.cancer.gov/aboutcancer/%5Cntreatment/types/chemotherapy. [Google Scholar]
- 158.Yong T., Zhang X., Bie N., Zhang H., Zhang X., Li F., Yang X. Tumor exosome-based nanoparticles are efficient drug carriers for chemotherapy. Nat. Commun. 2019;10(1) doi: 10.1038/s41467-019-11718-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Qi H., Liu C., Long L., Ren Y., Zhang S., Chang X., Kang C. Blood exosomes endowed with magnetic and targeting properties for cancer therapy. ACS Nano. 2016;10(3):3323–3333. doi: 10.1021/acsnano.5b06939. [DOI] [PubMed] [Google Scholar]
- 160.Kim M.S., Haney M.J., Zhao Y., Yuan D., Deygen I., Klyachko N.L., Batrakova E.V. Engineering macrophage-derived exosomes for targeted paclitaxel delivery to pulmonary metastases: in vitro and in vivo evaluations. Nanomedicine Nanotechnology. Biol. Med. 2018;14(1):195–204. doi: 10.1016/j.nano.2017.09.011. [DOI] [PubMed] [Google Scholar]
- 161.Morse M.A., Garst J., Osada T., Khan S., Hobeika A., Clay T.M., Lyerly H.K. A phase I study of dexosome immunotherapy in patients with advanced non-small cell lung cancer. J. Transl. Med. 2005;3:1–8. doi: 10.1186/1479-5876-3-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Nicholson H., Mesangeau C., McCurdy C.R., Bowen W.D. Sigma-2 receptors play a role in cellular metabolism: stimulation of glycolytic hallmarks by CM764 in human SK-N-SH neuroblastomas. J. Pharmacol. Exp. Ther. 2016;356(2):232–243. doi: 10.1124/jpet.115.228387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Kim Myung Soo, Haney Matthew J., Zhao Yuling, Mahajan Vivek, Deygen Irina, Klyachko Natalia L., Inskoe Eli, Piroyan Aleksandr, Sokolsky Marina, Okolie Onyi, Hingtgen Shawn D., Kabanov Alexander V., Batrakova Elena V. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Physiol. Behav. 2017;176(3):139–148. doi: 10.1016/j.nano.2015.10.012. doi:10.1016/j.nano.2015.10.012.Development. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Agrawal, A.K., Aqil, F., Jeyabalan, J., Spencer, W.A., Beck, J., Gachuki, B.W., Gupta R.C. Milk-derived exosomes for oral delivery of paclitaxel. Nanomed. Nanotechnol., Biol. Med.2017;13(5):1627–1636. doi: 10.1016/j.nano.2017.03.001. [DOI] [PubMed]
- 165.Singal P k. D.Sc, Iliskovic N. Doxorubıcın-induced cardiomyopathy. New Engl. J. Med. Curr. 1998;339(13):900–905. doi: 10.1056/NEJM199809243391307. [DOI] [PubMed] [Google Scholar]
- 166.Gomari H., Moghadam M.F., Soleimani M. Targeted cancer therapy using engineered exosome as a natural drug delivery vehicle. Onco. Targets Ther. 2018;11:5753–5762. doi: 10.2147/OTT.S173110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Zou J., Shi M., Liu X., Jin C., Xing X., Qiu L., Tan W. Aptamer-functionalized exosomes: elucidating the cellular uptake mechanism and the potential for cancer-targeted chemotherapy. Anal. Chem. 2019;91(3):2425–2430. doi: 10.1021/acs.analchem.8b05204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Johnsen K.B., Gudbergsson J.M., Skov M.N., Pilgaard L., Moos T., Duroux M. A comprehensive overview of exosomes as drug delivery vehicles - Endogenous nanocarriers for targeted cancer therapy. Biochim. Biophys. Acta - Rev. Cancer. 2014;1846(1):75–87. doi: 10.1016/j.bbcan.2014.04.005. [DOI] [PubMed] [Google Scholar]
- 169.Macia E., Ehrlich M., Massol R., Boucrot E., Brunner C., Dynasore Kirchhausen T. A cell-permeable inhibitor of dynamin. Dev. Cell. 2006;10(6):839–850. doi: 10.1016/j.devcel.2006.04.002. [DOI] [PubMed] [Google Scholar]
- 170.dosSantos T., Varela J., Lynch I., Salvati A., Dawson K.A. Effects of transport inhibitors on the cellular uptake of carboxylated polystyrene nanoparticles in different cell lines. PLoS ONE. 2011;6(9) doi: 10.1371/journal.pone.0024438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Sun D., Zhuang X., Xiang X., Liu Y., Zhang S., Liu C., Zhang H.G. A novel nanoparticle drug delivery system: the anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. Mol. Ther. 2010;18(9):1606–1614. doi: 10.1038/mt.2010.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Gazeau F., Pocard M., Pinto A., Marangon I., Méreaux J., Nicolás-Boluda A., Silva A.K.A. Immune reprogramming precision photodynamic therapy of peritoneal metastasis by scalable stem-cell-derived extracellular vesicles. ACS Nano. 2021;15(2):3251–3263. doi: 10.1021/acsnano.0c09938. [DOI] [PubMed] [Google Scholar]
- 173.Iessi E., Logozzi M., Lugini L., Azzarito T., Federici C., Spugnini E.P., Fais S. Acridine orange/exosomes increase the delivery and the effectiveness of acridine orange in human melanoma cells: a new prototype for theranostics of tumors. J. Enzyme Inhib. Med. Chem. 2017;32(1):648–657. doi: 10.1080/14756366.2017.1292263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Costa-Silva, B., Aiello, N., Ocean, A. et al Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat. Cell Bio. 2015;17(1):816–826. doi: 10.1038/ncb3169. In this issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Vader P., Mol E.A., Pasterkamp G., Schiffelers R.M. Extracellular vesicles for drug delivery. Adv. Drug Deliv. Rev. 2016;106:148–156. doi: 10.1016/j.addr.2016.02.006. [DOI] [PubMed] [Google Scholar]
- 176.Kamerkar Sushrut, LeBleu Valerie S., Sugimoto Hikaru, Yang Sujuan, Ruivo Carolina F., Lee Sonia A.Melo J.Jack. RK. Exosomes facilitate therapeutic targeting of oncogenic kras in pancreatic cancer. Physiol. Behav. 2016;176(3):139–148. doi: 10.1038/nature22341. doi:10.1038/nature22341.Exosomes. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Pascucci L., Coccè V., Bonomi A., Ami D., Ceccarelli P., Ciusani E. Pessina A. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: a new approach for drug delivery. J. Control Release. 2014;192:262–270. doi: 10.1016/j.jconrel.2014.07.042. [DOI] [PubMed] [Google Scholar]
- 178.Li X., Liu Y., Zheng S., Zhang T., Wu J., Sun Y., Liu G. Role of exosomes in the immune microenvironment of ovarian cancer. Oncol. Lett. 2021;21(5):1–17. doi: 10.3892/OL.2021.12638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Liu H., Shen M., Zhao D., Ru D., Duan Y., Ding C., Li H. The effect of riptolide-loaded exosomes on the proliferation and apoptosis of human ovarian cancer SKOV3 Cells. Biomed. Res. Int. 2019 doi: 10.1155/2019/2595801. 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Fan D., Guo Q., Shen J., Zheng K., Lu C., Zhang G. He X. The effect of triptolide in rheumatoid arthritis: from basic research towards clinical translation. Int. J. Mol. Sci. 2018;19(2):1–17. doi: 10.3390/ijms19020376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Druzhkova T.A., Yakovlev A.A. Exosome drug delivery through the blood–brain barrier: experimental approaches and potential applications. Neurochem. J. 2018;12(3):195–204. doi: 10.1134/s1819712418030030. [DOI] [Google Scholar]
- 182.Zhuang X., Xiang X., Grizzle W., Sun D., Zhang S., Axtell R.C., Zhang H.G. Treatment of brain inflammatory diseases by delivering exosome encapsulated anti-inflammatory drugs from the nasal region to the brain. Mol. Ther. 2011;19(10):1769–1779. doi: 10.1038/mt.2011.164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Lorenc T., Klimczyk K., Michalczewska I., Słomka M. 2020. Ijms-21-02118-V2 (1).Pdf. Published online. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Saari H., Lázaro-Ibáñez E., Viitala T., Vuorimaa-Laukkanen E., Siljander P., Yliperttula M. Microvesicle- and exosome-mediated drug delivery enhances the cytotoxicity of Paclitaxel in autologous prostate cancer cells. J. Control Release. 2015;220:727–737. doi: 10.1016/j.jconrel.2015.09.031. [DOI] [PubMed] [Google Scholar]
- 185.Alcayaga-Miranda F., González P.L., Lopez-Verrilli A., Varas-Godoy M., Aguila-Díaz C., Contreras L., Khoury M. Prostate tumor-induced angiogenesis is blocked by exosomes derived from menstrual stem cells through the inhibition of reactive oxygen species. Oncotarget. 2016;7(28):44462–44477. doi: 10.18632/oncotarget.9852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Coccè V., Franzè S., Brini A.T., Giannì A.B., Pascucci L., Ciusani E. Pessina A. In vitro anticancer activity of extracellular vesicles (evs) secreted by gingival mesenchymal stromal cells primed with paclitaxel. Pharmaceutics. 2019;11(2) doi: 10.3390/pharmaceutics11020061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Wei H., Chen J., Wang S., Fu F., Zhu X., Wu C., Lin J. A nanodrug consisting ofdoxorubicin and exosome derived frommesenchymal stem cells for osteosarcoma treatment in vitro. Int. J. Nanomed. 2019;14:8603–8610. doi: 10.2147/IJN.S218988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Escudier B., Dorval T., Chaput N., André F., Caby M.P., Novault S., Zitvogel L. Vaccination of metastatic melanoma patients with autologous dendritic cell (DC) derived-exosomes: results of the first phase 1 clinical trial. J. Transl. Med. 2005;3:1–13. doi: 10.1186/1479-5876-3-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Kanchanapally R., Deshmukh S.K., Chavva S.R., Tyagi N., Srivastava S.K., Patel G.K., Singh S. Drug-loaded exosomal preparations from different cell types exhibit distinctive loading capability, yield, and antitumor efficacies: a comparative analysis. Int. J. Nanomed. 2019;14:531–541. doi: 10.2147/IJN.S191313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Sinha D., Roy S., Saha P., Chatterjee N., Bishayee A. Trends in research on exosomes in cancer progression and anticancer therapy. Cancers (Basel) 2021;13(2):1–31. doi: 10.3390/cancers13020326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.McGovern N., Schlitzer A., Janela B., Ginhoux F. Protocols for the Identification and Isolation of Antigen-Presenting Cells in Human and Mouse Tissues. 2016;1423 doi: 10.1007/978-1-4939-3606-9_12. [DOI] [PubMed] [Google Scholar]
- 192.Saxena M., Bhardwaj N. Turbocharging vaccines: emerging adjuvants for dendritic cell based therapeutic cancer vaccines. Curr. Opin. Immunol. 2017;47:35–43. doi: 10.1016/j.coi.2017.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Laurence Zitvogel, Armelle Regnault, Anne Lozier, Joseph Wolfers, Caroline Flament, Danielle Tenza, Paola Ricciardi-Castagnoli G.R. & SA. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell derived exosomes. Published online 1998. [DOI] [PubMed]
- 194.Sundarasetty B.S., Chan L., Darling D., Giunti G., Farzaneh F., Schenck F. Stripecke R. Lentivirus-induced “Smart” dendritic cells: pharmacodynamics and GMP-compliant production for immunotherapy against TRP2-positive melanoma. Gene Ther. 2015;22(9):707–720. doi: 10.1038/gt.2015.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Roussy Gustave. Cancer Campus GP, Study. Trial of a Vaccination With Tumor Antigen-loaded Dendritic Cell-derived Exosomes (CSET 1437) 2018:10–14. Published online. [Google Scholar]
- 196.Damo M., Wilson D.S., Simeoni E., Hubbell J.A. TLR-3 stimulation improves anti-tumor immunity elicited by dendritic cell exosome-based vaccines in a murine model of melanoma. Sci. Rep. 2015;5(July):1–15. doi: 10.1038/srep17622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Lu Z., Zuo B., Jing R., Gao X., Rao Q., Liu Z., Yin H.F. Dendritic cell-derived exosomes elicit tumor regression in autochthonous hepatocellular carcinoma mouse models. J. Hepatol. 2017;67(4):739–748. doi: 10.1016/j.jhep.2017.05.019. [DOI] [PubMed] [Google Scholar]
- 198.Gao Jin, Dong ZW Xinyue. Generation, purification and engineering of extracellular vesicles and their biomedical applications. Physiol. Behav. 2017;176(3):139–148. doi: 10.1016/j.ymeth.2019.11.012.Generation. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Wan Z., Zhao L., Lu F., Gao X., Dong Y., Zhao Y., Liu L. Mononuclear phagocyte system blockade improves therapeutic exosome delivery to the myocardium. Theranostics. 2020;10(1):218–230. doi: 10.7150/thno.38198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Lamparski H.G., Metha-Damani A., Yao J.Y., Patel S., Hsu D.H., Ruegg C., Le Pecq J.B. Production and characterization of clinical grade exosomes derived from dendritic cells. J. Immunol. Methods. 2002;270(2):211–226. doi: 10.1016/S0022-1759(02)00330-7. [DOI] [PubMed] [Google Scholar]
- 201.Sart S., Tsai A.C., Li Y., Ma T. Three-dimensional aggregates of mesenchymal stem cells: cellular mechanisms, biological properties, and applications. Tissue Eng. - Part B Rev. 2014;20(5):365–380. doi: 10.1089/ten.teb.2013.0537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Yan L., Wu X. Exosomes produced from 3D cultures of umbilical cord mesenchymal stem cells in a hollow-fiber bioreactor show improved osteochondral regeneration activity. Cell Biol. Toxicol. 2020;36(2):165–178. doi: 10.1007/s10565-019-09504-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Linares R., Tan S., Gounou C., Arraud N., Brisson A.R. High-speed centrifugation induces aggregation of extracellular vesicles. J. Extracell. Vesicles. 2015;4(1):1–7. doi: 10.3402/jev.v4.29509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Busatto S., Vilanilam G., Ticer T., Lin W.L., Dickson D.W., Shapiro S., Wolfram J. Tangential flow filtration for highly efficient concentration of extracellular vesicles from large volumes of fluid. Cells. 2018;7(12) doi: 10.3390/cells7120273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Chen T.S., Arslan F., Yin Y., Tan S.S., Lai R.C., Choo A.B.H., Lim S.K. Enabling a robust scalable manufacturing process for therapeutic exosomes through oncogenic immortalization of human ESC-derived MSCs. J. Transl. Med. 2011;9(1):47. doi: 10.1186/1479-5876-9-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Brindley D., Moorthy K., Lee J.H., Mason C., Kim H.W., Wall I. Bioprocess forces and their impact on cell behavior: implications for bone regeneration therapy. J. Tissue Eng. 2011;2(1):1–13. doi: 10.4061/2011/620247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Panchalingam K.M., Jung S., Rosenberg L., Behie L.A. Bioprocessing strategies for the large-scale production of human mesenchymal stem cells: a review Mesenchymal Stem/Stromal Cells - An update. Stem Cell Res. Ther. 2015;6(1):1–10. doi: 10.1186/s13287-015-0228-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Mitchell J.P., Court J., Mason M.D., Tabi Z., Clayton A. Increased exosome production from tumour cell cultures using the Integra CELLine culture system. J. Immunol. Methods. 2008;335(1–2):98–105. doi: 10.1016/j.jim.2008.03.001. [DOI] [PubMed] [Google Scholar]
- 209.Lee Y.X.F., Johansson H., Wood M.J.A., El Andaloussi S. Considerations and implications in the purification of extracellular vesicles – a cautionary tale. Front. Neurosci. 2019;13(October) doi: 10.3389/fnins.2019.01067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Duijvesz D., Burnum-Johnson K.E., Gritsenko M.A., Hoogland A.M., Vredenbregt-van Den Berg M.S., Willemsen R., Jenster G. Proteomic profiling of exosomes leads to the identification of novel biomarkers for prostate cancer. PLoS ONE. 2013;8(12):1–10. doi: 10.1371/journal.pone.0082589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Yamashita T., Takahashi Y., Takakura Y. Possibility of exosome-based therapeutics and challenges in production of exosomes eligible for therapeutic application. Biol. Pharm. Bull. 2018;41(6):835–842. doi: 10.1248/bpb.b18-00133. [DOI] [PubMed] [Google Scholar]
- 212.ClinicalTrials. gov Search Results 02/16/2022. 2022;(1):1–5.
- 213.Medicine U. NL of. ClinicalTrials.gov. 2022;(Cdc):1–10.
- 214.ClinicalTrials. gov Search Results 02/16/2022 cancer. 2022;(1):1–18.
- 215.ClinicalTrials. gov Search Results 02/16/2022 cancer phase 1. 2022;(1):3–5.
- 216.ClinicalTrials. gov Search Results 02/16/2022 phase 2. 2022;(1):2–5.
- 217.ClinicalTrials. gov Search Results 02/16/2022 phase 3. 2022;(1):5216562.
- 218.Tricard, Jérémy, University hospital L. Molecular profiling of exosomes in tumor-draining vein of early-staged lung cancer. (Cdc):1–7. 2022.
- 219.Gongjianping S.A.H. Combined Diagnosis of CT and Exosome in Early Lung Cancer. 2018;(Cdc):1–10. [Google Scholar]
- 220.Zhang, Shu, Ying Lv NDTH the AH of NUMS, Nanjing, Jiangsu C. Acquisition of portal venous CTCs and exosomes from patients with pancreatic cancer by EUS (CTCs). (Cdc):1–7.
- 221.Susan E. Eubanks, RN, M.S.N., OCN, clinical trials manager LWJCC. A pilot study of tumor-derived exosomes as diagnostic and prognostic markers in breast can. (Cdc):1–6.
- 222.William Jarnagin PKMSKCC, New York, New York US. interrogation of exosome-mediated intercellular signaling in patients with pancreatic cancer. 2018;(Cdc):1–10.
- 223.Roger Tun Exosome diagnostics I. Clinical validation of a urinary exosome gene signature in men presenting for suspicion of prostate cancer. Published online 2016:1–6.
- 224.Cowan Andrew, Parks Valerie. 2014. RN U of NMCC, Albuquerque, New Mexico US. Exosome Testing As a Screening Modality For Human Papillomavirus-Positive Oropharyngeal Squamous Cell Carcinoma; pp. 1–7. Published online. [Google Scholar]
- 225.Li Lei. Peking union medical college hospital, Beijing C. coding RNA in the Exosome of the Epithelia Ovarian Cancer. 2018:6–11. Published online. [Google Scholar]
- 226.Rebecca Redman U of L & JGBCC, Information. Edible Plant Exosome Ability to Prevent Oral Mucositis Associated With Chemoradiation Treatment of Head and Neck Cancer. Published online 2021:1–7.
- 227.Jianguo Sun X.H. of C. Clinical Research for the Consistency Analysis of PD-L1 in Lung Cancer Tissue and Plasma Exosome Before and After Radiotherapy (RadImm02). Published online 2016:1–5.
- 228.Olivier Bouché, CHU de Reims F. Identification of new diagnostic protein markers for colorectal cancer. Published online 2020:1–6.
- 229.Adam Luginbuhl SKCC at TJ. Metformin hydrochloride in affecting cytokines and exosomes in patients with head and neck cancer. Published online 2020:1–8.
- 230.Roberto Pili U at BIU. Glycosylation of exosomes in prostate and urothelial carcinoma. Published online 2021:13–18.
- 231.A Study to Measure the Expression of the HER2-HER3 dimer in tumour and blood (Exosomes) samples from patients with HER2 positive breast cancer receiving HER2 targeted therapies. Case Med. Res. 2020 doi: 10.31525/ct1-nct04288141. Published online. [DOI] [Google Scholar]
- 232.Emilie LE RHUN MCOL. Analyses of exosomes in the cerebrospinal fluid for breast cancer patients with suspicion of leptomeningeal metastasis . Published online 2021:1–8.
- 233.Hyun Koo Kim, Guro-gu K. Multicenter clinical research for early diagnosis of lung cancer using blood plasma derived exosome. Published online 2021:1–7.
- 234.Etienne BUSCAIL. University Hospital B, Information, Chu de Bordeaux Bordeaux F; 2018. Diagnostic Accuracy of Circulating Tumor Cells (CTCs) and Onco-exosome Quantification in the Diagnosis of Pancreatic Cancer - PANC-CTC (PANC-CTC) pp. 1–6. Published online. [Google Scholar]
- 235.Grald W. Dryden, Jr. U of L. study investigating the ability of plant exosomes to deliver curcumin to normal and colon cancer tissue. Published online 2011:1–8.
- 236.Andrew Hoover U of KMC. Exosomes in Rectal Cancer. Published online 2019:2–7.
- 237.Tam Joseph, Ward Becky. Charing cross hospital, imperial college healthcare NHS Trust, London UK. How Does Prostate Cancer Metastasize ? Studying the Role of Secreted Packages (Exosomes) From Fat Tissue in Lean and Obese Patients (EXOPRO) 2019:1–7. Published online. [Google Scholar]
- 238.Win, Thida, Borthwick Research Unit LH, Stevenage U.K. Improving the early detection of lung cancer by combining exosomal analysis of hypoxia with standard of care imaging (LungExoDETECT). Published online 2020:2–7.
- 239.yingbin liu, Xinhua Hospital, Shanghai C. A study of circulating exosome proteomics in gallbladder carcinoma patients. Published online 2018:1–6.
- 240.Lin Miao TSH of NMU. ncRNAs in exosomes of cholangiocarcinoma. Published online 2017:1–6.
- 241.Center SPMDAC. iExosomes in Treating Participants With Metastatic Pancreas Cancer With KrasG12D Mutation. Published online 2018:1–8.
- 242.MOTTET Nicolas, Hocine HABCHI. Centre Hospitalier Universitaire de Saint Etienne; Chu Saint-Etienne F: 2019. Evaluation of Urinary Exosomes Presence From Clear Cell Renal Cell Carcinoma; pp. 1–8. Published online. [Google Scholar]
- 243.Dan Zandberg, Jennifer Ruth, RN, Lisa Boswell U.H.C.C., Pittsburgh, Pennsylvania US. Hemopurifier plus pembrolizumab in head and neck cancer. Published online 2020:1–10.
- 244.Aiqin Gu, Shanghai Chest Hospital, Shanghai C. Exosomes detection for the prediction of the efficacy and adverse reactions of anlotinib in patients with advanced NSCLC. Published online 2022:1–7.
- 245.University Hospital A, Study. Exosomes implication in PD1-PD-L1 activation in OSAS (ExoSAS). Published online 2020:1–6.
- 246.Yutao Liu CA of MS. The study of exosome EML4-ALK fusion in NSCLC clinical diagnosis and dynamic monitoring. Published online 2020:2–7.
- 247.Jialei Wang C hospital FU, Shanghai C. Prediction of immunotherapeutic effect of advanced non-small cell lung cancer. Published online 2020:1–7.
- 248.EVRARD, Camille, CHU POitiers Poitiers F. Interest of circulating tumor DNA in digestive and gynecologic /breast cancer. Published online 2022:1–6.
- 249.Ruijin Hospital Shanghai Jiao Tong University School of Medicine . Published online; Shanghai C: 2021. Construction of Microfluidic Exosome Chip for Diagnosis of Lung Metastasis of Osteosarcoma; pp. 1–6. [Google Scholar]
- 250.Alice HERVIEU, Emilie REDERSTORFF, CGFL. Study of exosomes in monitoring patients with sarcoma (EXOSARC) (EXOSARC). Published online 2021:1–7.
- 251.Shen Yuhui. 2017. Ruijin Hospital Shanghai Jiao Tong University School of Medicine hai, Shanghai C. Circulating Exosome RNA in Lung Metastases of Primary High-Grade Osteosarcoma; pp. 1–7. Published online. [Google Scholar]
- 252.Gianluca Giavaresi, Istituto Ortopedico Rizzoli, Bagheria, Palermo I. Identification and characterization of predictive factors of onset of bone metastases in cancer patients (PreMetOn). Published online 2019:1–7.
- 253.Mikkel Noerholm Exosome diagnostics I. Clinical evaluation of ExoDx prostate (intelliscore) in men presenting for initial prostate biopsy. Published online 2021:1–6.
- 254.Henri MONTAUDIE, Centre, hospitalier universitaire de nice F. Study of molecular mechanisms implicated in the pathogenesis of melanoma . Role of Exosomes (EXOSOMES). Published online 2016:1–7.
- 255.Eli Sprecher, MD, Tel-Aviv Sourasky Medical Center I. cancer | exosomes evaluation of the safety of CD24-exosomes in patients with COVID-19 infection. Published online 2021:1–8.
- 256.Bin Cheng, Huazhong University of Science and Technology & Tongji Hospital, Tongji medical college, Wuhan, Hubei C. Circulating extracellular exosomal small RNA as potential biomarker for human pancreatic cancer. Published online 2020:1–6.
- 257.Liang-wan Chen, Union Hospital FMU. Exosome of mesenchymal stem cells for multiple organ dysfuntion syndrome after surgical repaire of acute type A aortic dissection. Published online 2020:1–7.
- 258.Julie ABRAHAM, Danielle TROUTAUD, University Hospital L. Exosomes and Immunotherapy in Non-Hodgkin B-cell Lymphomas (ExoReBLy). Published online 2020:1–6.
- 259.A Prospective Feasibility Study Evaluating Extracellular Vesicles obtained by liquid biopsy for neoadjuvant treatment response assessment in rectal cancer - full text view, ClinicalTrials.gov.pdf. 2022.
- 260.Sonia Kumar, exosome diagnostics I. Exodx prostate evaluation in prior negative prostate biopsy setting. Published online 2021:1–6.
- 261.HAJJAJI, Nawale NH, Marie MV VANSEYMORTIER COL, Lille F. Omic technologies to track resistance to palbociclib in metastatic breast cancer (OMERIC). Published online 2020:1–7.
- 262.Hospital NTU, Information. Predicting prognosis and recurrence of thyroid cancer via new biomarkers, urinary exosomal thyroglobulin and galectin-3. Published online 2021:1–7.
- 263.Instituto Portugues de Oncologia Francisco. Published online; 2020. Gentil, Porto & Polytechnic Institute of Porto, VTT Technical Research Centre, Finland University of Oulu, Linnaeus University EC. Impact of Group Psychological Interventions on Extracellular Vesicles in People Who Had Cancer (MindGAP-P) pp. 1–11. [Google Scholar]
- 264.Anaplastic Thyroid Cancer and follicular thyroid cancer-derived exosomal analysis via treatment of lovastatin and vildagliptin and pilot prognostic study via urine exosomal biological markers in thyroid cancer patients - full text view - clinicaltrial.pdf. 2022.
- 265.Antti Rannikko HUCH. Evaluation of Fapi-pet in Prostate Cancer . (FAPIPETPCA). Published online 2022:1–7.
- 266.Henry Bohler . Published online; 2021. University of Louisville, Kentucky US. Plant Exosomes and Patients Diagnosed With Polycystic Ovary Syndrome; pp. 1–7. [Google Scholar]
- 267.Kye Young Lee KUMC & HPCL . Published online; 2019. Olmutinib Trial in T790M (+) NSCLC Patients Detected By Liquid Biopsy Using BALF Extracellular Vesicular DNA; pp. 1–7. [Google Scholar]
- 268.Sun Jiayuan, Hospital Shanghai Chest, Shangai C. Published online; 2019. Clinical Study of ctDNA and Exosome Combined Detection to Identify Benign and Malignant Pulmonary Nodules (ctDNA) pp. 6–11. [Google Scholar]
- 269.Published online; 2022. ClinicalTrials.gov Search Results 01/28/2022; pp. 1–9. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.