Abstract
Vaccine-preventable diseases, such as measles, have been re-emerging in countries with moderate to high vaccine uptake. It is increasingly important to identify and close immunity gaps and increase coverage of routine childhood vaccinations, including two doses of the measles-mumps-rubella vaccine (MMR). Here, we present a simple cohort model relying on a Bayesian approach to evaluate the evolution of measles seroprevalence in Belgium using the three most recent cross-sectional serological survey data collections (2002, 2006 and 2013) and information regarding vaccine properties. We find measles seroprevalence profiles to be similar for the different regions in Belgium. These profiles exhibit a drop in seroprevalence in birth cohorts that were offered vaccination at suboptimal coverages in the first years after routine vaccination has been started up. This immunity gap is observed across all cross-sectional survey years, although it is more pronounced in survey year 2013. At present, the COVID-19 pandemic could negatively impact the immunization coverage worldwide, thereby increasing the need for additional immunization programs in groups of children that are impacted by this. Therefore, it is now even more important to identify existing immunity gaps and to sustain and reach vaccine-derived measles immunity goals.
Keywords: Measles elimination, Bayesian MCMC, Serial serological survey data, Immunity goals
1. Introduction
Measles is a highly infectious (childhood) disease, which was the leading cause of child morbidity and mortality globally prior to the introduction of measles-preventing vaccines [1]. Due to improvements in measles control and prevention, and the increment of widespread measles-containing vaccination, the reduction in global measles-related deaths is sustained [2]. Regardless of the immense progress, several large scale measles outbreaks have recently been reported in countries with moderate to high vaccine uptake. Countries experiencing outbreaks [3], [4], prior to the COVID-19 pandemic imposing severe restriction in terms of social interactions, include many European countries such as Romania (3,900 reported cases in 2019), France (2,637 in 2019), Italy (1,623 in 2019), Czech Republic (590 in 2019) and Poland (1,367 in 2019) [5], [6]. Moreover, in Ukraine there were over 57,000 measles cases in 2019 [7]. However, outbreaks are certainly not limited to Europe, and high case numbers have been reported from regions that had previously eliminated measles, such as, the United States of America (1,282 in 2019) [8]. In addition, the outbreak in Samoa and the Pacific Islands (5,707 in 2019), exacerbated by a decline in first (from 99% to 40%) and second dose (from 87% to 28%) MMR vaccine uptake between 2013 and 2018, was tragically accompanied by 83 deaths [9]. Even though two-dose combined measles-mumps-rubella (MMR) vaccination is well-established and vaccine uptake is considered moderate to high in many of these countries, these recent outbreaks have led to a thorough and critical investigation of the level (immunogenicity) and duration (persistence) properties and the effectiveness of the different MMR vaccine formulations used in practice [10], [11]. Outbreaks may occur as a result of a declining MMR vaccine uptake. Increasing levels of vaccine hesitancy in certain groups can lead to localized communities of unvaccinated individuals or susceptibility pockets [12], [13] thereby enlarging the population at risk of measles infection. Immunity gaps can also occur when the target uptake level for measles elimination and eradication, i.e. 95% coverage for both doses, is not reached [14]. In addition, primary vaccine failure and possibly waning of vaccine-induced immunity may lead to suboptimal protection against measles in highly-vaccinated populations [15]. Given that measles incidence worldwide surged in 2019 with the highest numbers of reported cases in 23 years [16], it is important to identify and close immunity gaps, strengthen essential immunization systems to increase two-dose coverage and to improve surveillance and preparedness to rapidly respond to outbreaks, as mentioned in the Immunization Agenda 2030 [17].
In Belgium, monovalent measles vaccination was present prior to 1985, at least to a small extent. Universal MMR vaccination was initiated in 1985 with a first dose of the measles-containing MMR vaccine for infants of one year. A second MMR dose was introduced in 1995, administered to children aged between 11 (in Flanders) and 13 (in Wallonia & Brussels Capital Region) years [18]. In Flanders, free-of-charge catch-up vaccination in adults born from 1970 onwards is integrated in the vaccination program for adults since September 2018 [19]. In Wallonia and Brussels Capital Region catch-up vaccination for adults and adolescents has been strongly promoted since 2017 [20] and onwards [21], but is only free of charge for children till 18 years old and for high school students till 20 years [22], but not for adults.
The yearly number of reported measles cases in Belgium fluctuated between 15 and 98 cases for the period between 2003 and 2010 [23]. In 2011, a large measles outbreak occurred in Belgium with a total of 674 reported cases. In 2018, Belgium received for the first time the status of interrupted endemic transmission, the first step towards measles elimination (i.e. the absence of endemic measles virus transmission in a defined geographic area for at least 12 months [6]). Although no transmission chain persisted for a period exceeding 12 months [24], the average yearly number of measles cases has been increasing since 2016, with 367 cases in 2017 (outbreak in the Walloon region) and 496 cases in 2019. In 2019, the WHO Regional Verification Committee pointed out the viral transmission of 51 consecutive weeks (so just under 12 months) and noted concern regarding the low MCV2 coverage [6]. As a result of the COVID-19 pandemic and mitigation measures taken across the world, the number of reported measles cases in Belgium decreased to 66 cases in 2020. A resurgence of measles and large-scale outbreaks could be expected when human behavior returns to pre-pandemic levels.
In previous work, Hens et al. (2015) [25] estimated measles outbreak risk in Belgium while relying on readily available data including cross-sectional serological survey data, detailed vaccine coverage data and social contact data. More specifically, these authors used a multi-cohort approach to estimate the effective reproduction number over time at municipality level in Belgium with effective reproduction numbers above one indicating an outbreak risk in future years. Similar to the approach proposed by Hens et al. (2015) [25], Funk et al. (2019) [14] combined estimated immunity levels from serological data from 17 countries together with data on age-specific mixing behavior in order to derive contact-adjusted immunity levels. These immunity levels were then compared to case data from subsequent years to establish contact-adjusted immunity levels for measles elimination. These authors conclude that the immunity levels necessary for measles elimination are higher than previous guidance and that achieving high immunity in 5–9-year-olds is of great importance. A common feature in the aforementioned work is the use of cross-sectional serological data. In this paper, we focus on the combined estimation of vaccination coverage, exposure rates, and immunogenicity and persistence parameters from serial serological data in order to monitor the evolution of measles seroprevalence in Belgium. This is closely related to the cohort perspective in the work by Wood et al. (2015) [26], but we focus only on measles instead of on all three components of the MMR vaccine in this manuscript. Unlike previous work, in order to account for all relevant sources of uncertainty, our model is implemented in the Bayesian inferential paradigm allowing for the use of vaccine coverage data and prior information related to immunogenicity and persistence from a recent systematic review [11].
2. Materials and methods
2.1. Data sources
2.1.1. Serial serological survey data
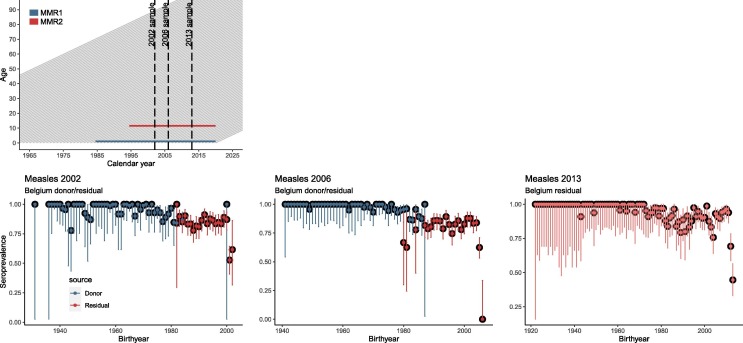
Belgian serial serological survey data on measles collected in 2002, 2006 and 2013 are available. Fig. 1 (panel 1) depicts a Lexis diagram showing the temporal evolution of the age of individuals in different birth cohorts, and important dates with regard to vaccine-induced immunization and serological data collections. Serological data in 2002 and 2006 are collected based on blood donor samples together with residual samples (i.e., left-over blood samples collected for other reasons, but available for subsequent testing with respect to the presence of measles antibodies) whereas the serology of 2013 only consists of residual samples [27], [28], [29]. The age distribution of the blood donor and residual samples are displayed in blue and red, respectively (see Fig. 1 , panel 2–4).
Fig. 1.
Panel 1: Lexis diagram showing the aging of (Belgian) birth cohorts born at different calendar times (gray oblique lines, one line per birth cohort), together with the timing of the serial serological survey data collections (in 2002, 2006 and 2013; black dashed vertical lines) and timing of MMR vaccination (horizontal blue and red lines). Panel 2–4: Observed measles seroprevalence (with 95% Clopper-Pearson confidence limits) for Belgium in 2002, 2006 and 2013 based on donor or residual samples. The age distribution of the donor and residual samples are displayed in blue and red respectively in panel 2–4. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
All analyses were performed at the WHO-accredited national reference laboratory for measles and rubella. For all three survey rounds the IgG Engygnost kist from Dade Behring/Siemens (Germany) was used as a reference (either directly of after standardization) with sensitivity 99.6% and specificity of 100% as provided by the manufacturer. Tisher et al. [30] demonstrated a sensitivity of 95.3% and specificity of 100%, using a plaque neutralization test (PNT) as a golden standard, and within a sero-epidemiological scope. Other studies also reported a slightly lower sensitivity (92%, 88%) for the EIA from Dade Behring in comparison to PNT [30]. IgG titres < 0.15 IU/ml and > 0.35 IU/ml (0.325 IU/ml in 2013) were considered sero-negative and sero-positive, respectively [27], [28]. Note, it is considered that presence of antibodies against measles as a proxy of immunity, although absence of antibodies does not equate to no protection. Cellular immunity also provides some degree of protection [31].
2.1.2. Vaccination coverage
Time-specific vaccination coverage estimates for Belgium (collected regionally, see Supplement 1 for more details) and for both recommended MMR doses are available. Such information is only available for a few years in which vaccination coverage studies were conducted. MMR vaccination coverages for the other years (i.e., after 1985, the start of MMR vaccination in Belgium) are inferred using a generalized linear model (glm) for binary outcome data (vaccinated: yes/no) with logit-link function (see Supplementary 1 for more details thereon). Although measles vaccination prior to 1985 is not explicitly accounted for, given the lack of reliable vaccination coverage information, exposure rates for birth cohorts born before 1984 are likely to be estimated to be too high. Nevertheless, we believe that the impact thereof is limited.
2.2. Model description
We use a Bayesian Markov Chain Monte Carlo (MCMC) approach to estimate a single exposure rate for cohorts born prior to 1984 (no MMR vaccination), different exposure rates for each birth cohort born in and after 1984, MMR vaccination coverage rates, and seroconversion and waning rates for the measles component in the combined MMR vaccine.
Here, we consider a cohort model describing the probability of being sero-negative for measles at calendar time , when born in year b, based on vaccination history and vaccination properties (i.e., prior vaccination coverage, seroconversion and waning after vaccination). First of all, for individuals born prior to the introduction of MMR vaccination (i.e., born prior to 1984), the probability of being sero-positive for measles is almost entirely induced through natural measles infection only (except for sero-positivity due to limited monovalent measles vaccination), which is presumed to induce lifelong humoral immunity [32]. Cohorts born after 1983 include individuals that are either fully vaccinated (with two MMR doses according to the MMR schedule highlighted above) or that only received the first MMR dose, depending on the survey year. The first MMR dose is assumed to be given exactly at the age of 1 year. However, depending on the vaccination coverage for MMR1 in year vaccination of only a part of the individuals in the birth cohort will take place. Moreover, not all of the sero-negative individuals at age 1 year seroconvert and gain humoral immunity against measles. Upon being seroconverted after MMR1 vaccination, a fraction will become sero-negative again after waning of humoral immunity [11]. Sero-negative individuals after potential MMR1 vaccination (i.e., unvaccinated children or those with primary vaccine failure) are either exposed to measles or not. The second dose is assumed to be administered at 11 (in Flanders) or 13 (in Brussels Capital Region & Wallonia) years of age. The probability of being sero-negative for measles is calculated based on whether individuals seroconverted after MMR1 and potentially waned thereafter (see Supplementary Table 2 for an overview of the different combinations). Maternal immunity is not accounted for. It is assumed that natural humoral immunity does not decline (or wane) over time. In contrast, we take into account that infection after waning of vaccine-induced humoral immunity is possible and that persons might thus be sero-negative after vaccination. For details, we refer the reader to Supplementary 2.
Parameters are estimated using a Bayesian Markov Chain Monte Carlo (MCMC) approach. This cohort model was implemented using the rstan package [33] R version 3.6.3 [34]. The No-U-Turn Samples (NUTS) algorithm was used to obtain four independent chains of 50,000 iterations, with 25,000 iterations used as burn-in and discarded from final inference. Informative normal priors are proposed for the seroconversion and waning rates, with distributional parameters derived based on the meta-analytic estimates reported by Schenk et al. (2021) [11]. Vague uniform priors are proposed for all exposure rates. Informative normal priors for the vaccination coverages are proposed based on the results of fitting a glm for binary data. All these parameters are restricted to fall between 0 and 1. In Supplementary Table 3 we present the prior distributions for the different model parameters.
Reported measles cases from 2003 until 2013 were retrieved from the European Centre for Disease Prevention and Control (ECDC) (Supplementary Fig. 2) [23]. The extracted data are available for age categories and are uniformly re-distributed in one-year age groups in order to get an estimate of cases by age (in years); and thus also by birth cohort for the specific year. The yearly number of cases for one birth cohort was calculated (based on the exposure duration (in years)). Furthermore, cases per birth year are standardized by cohort size and the fraction of sero-negative individuals in that specific birth cohort, while relying on the vaccine coverage point estimates from the fit.
3. Results
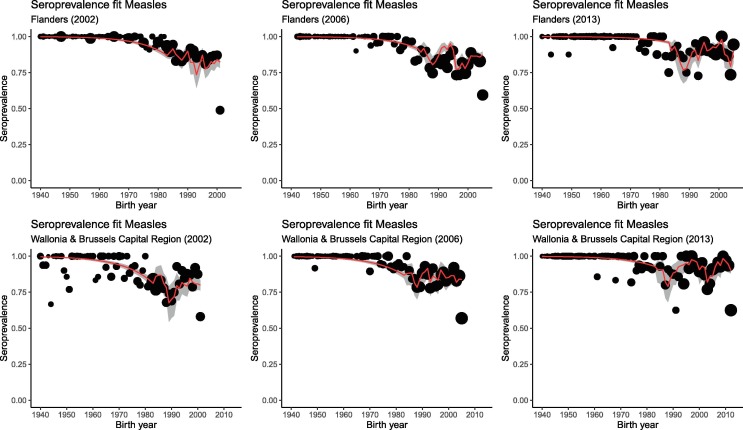
Fig. 2 presents the results from fitting the Bayesian cohort model to the available serological survey data. More specifically, the figure shows the estimated measles seroprevalence for Flanders and the combination of the Belgian regions Wallonia & Brussels Capital Region, by birth cohort (year) using the three serological surveys and relying on information with regard to the MMR vaccine properties. The red solid line represents the estimated seroprevalence curve together with pointwise 95% credible intervals (CrIs) shown in gray.
Fig. 2.
Estimated measles seroprevalence by birth cohort (year) for Flanders and Wallonia & Brussels Capital Region based on three Belgian serial serological survey data collections in 2002, 2006 and 2013 using the Bayesian cohort model.
Based on Fig. 2, we clearly observe a drop in seroprevalence (or a decrease in humoral immunity against measles infection) for the birth cohorts born between 1985 and 1990, i.e., the group of individuals in which universal immunization with the combined MMR vaccine started and who were offered two doses before the 2002 survey. MMR1 uptake in these cohorts was suboptimal and even if efforts were done to give MMR2 vaccination, it does not systematically mean that is was completed with a second dose. Moreover, this decrease is observed across all cross-sectional survey years, although the extent is most pronounced in survey year 2013. Such gradual increase in sero-negativity for those birth cohorts (1985–1990) in the model-based seroprevalence fit is induced by the hypothesis of waning of humoral immunity. Moreover, the potential susceptibility gap (assuming that the absence of humoral immunity is indicative for the absence of protection against measles infection) is likely to enlarge, unless additional vaccination efforts targeting these birth cohorts are undertaken. Based on the empirical seroprevalence data, we observe a more complicated picture with regard to the evolution of the sero-negativity in these birth cohorts (Supplementary Fig. 3). Although the uncertainty surrounding the point estimates is large, a gradual decrease in sero-positivity from 2002 up to 2013 is only observed in some of the birth cohorts with complete MMR vaccination. Despite the unclear overall picture concerning waning of vaccine-induced humoral immunity, apparent fluctuations in sero-positivity rather than a decline in other birth cohorts throughout this short period can only be explained by exposure to natural infection or catch-up vaccination. Supplementary Fig. 4 displays the theoretical effect of waning of vaccine-induced humoral immunity with time since vaccination. More specifically, the evolution of sero-positivity in the presence of waning is shown for two birth cohorts, thereby relying on estimates obtained from the model. These projections assume the absence of a second MMR dose and boosting of humoral immunity levels due to exposure to the wild-type measles virus.
A small increase in seroprevalence is observed for birth cohorts from 1975 to 1980 in survey year 2013, which could be the result of the measles outbreak in 2011 [35].
Fig. 3 displays the estimated exposure rates for Flanders, and Wallonia & Brussels Capital Region. The exposure rate, defined as the probability to get infected with measles when the individual is not vaccinated/protected, prior to 1984 can be interpreted as the mean exposure rate over all previous years while other exposure rates are birth cohort-specific. The estimated exposure rates exhibit an increasing trend towards the younger birth cohorts, an evolution which is also observed in the standardized number of measles cases (per 100 susceptible individuals; gray bars in Fig. 3). Finally, although differences in exposure rates are present between Flanders and Wallonia & Brussels Capital Region, the overall trend seems to be similar across regions in Belgium.
Fig. 3.
Cohort-specific estimated exposure rates for Flanders (red) and Wallonia & Brussels Capital Region (blue) and in gray their pointwise 95% credible intervals, with standardized yearly incidence (bars) of reported measles cases per 100 susceptible individuals as reported between 2003 and 2013 by birth cohort (for the whole of Belgium). By standardizing, differences in vaccination coverage (in different years) and the effect of primary and secondary vaccine failure are accounted for. The reported cases by birth cohort are divided by the total number of susceptible individuals. Reported cases were extracted from the ECDC Surveillance Atlas of Infectious Diseases [23]. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
4. Discussion
In the past, immunity against measles was the result of recovering from natural infection. Despite natural immunity to measles being protective and long-lasting, measles infections led to an estimated 2.6 million deaths (mainly among young children) each year prior to the introduction of measles vaccines in 1963 [36]. Nowadays, populations around the world benefit from vaccine-induced protection against measles infection after vaccination with measles-containing vaccines including combined MMR vaccines. Between 2000 and 2018, measles vaccination prevented an estimated 23.2 million deaths with global measles deaths having decreased by 73% from 536,000 deaths in 2000 to 142,000 in 2018 [36]. Available serological data does not distinguish between natural and vaccine-induced humoral immunity. Neither does it provide insights in the protection offered by cellular immunity against measles infection after previous infection or vaccination, thereby making the interpretation of serological survey data difficult. More specifically, disentangling vaccine-induced and natural humoral immunity requires accommodating historical changes in disease incidence and vaccination coverage [37]. Currently, a significant portion of the population is protected as a result of either natural infection or vaccination. However, the proportion of population immunity gained via natural infection gradually decreases and currently the majority of population-level immunity is induced through vaccination. Indeed, the proportion of birth cohorts which have experienced intense natural measles circulation and exposure is gradually decreasing. This will ultimately lead to a population with only vaccine-derived immunity, which is known to be less persistent when compared to natural immunity [38]. Additionally, absence of boosting by re-exposure to the wild-type virus enhances the risk of waning, resulting in accumulating numbers of susceptible individuals and thus re-emerging (large-scale) measles outbreaks. Therefore, it is important to know if herd-immunity thresholds are still reached in highly vaccinated populations.
Over the last years/decades, we have witnessed an increase in measles incidence worldwide [39], [40], [41]. Although it was previously claimed that there was no readily available evidence that showed that waning of vaccine-derived immunity plays a significant role in the resurgence of measles [32], more recent systematic evidence does show a notable amount of waning of humoral immunity levels. For example, 20 years after MMR vaccination, the expected loss of sero-positivity equals 16.4% (95% CI 9.5–27.4) based on an annual waning rate of 0.009 (95% CI 0.005–0.016) as estimated by Schenk et al. (2021) [11]. Despite the potential negative effect of waning on outbreak risk, the impact thereof on the overall sero-negativity profile at population-level is currently difficult to observe directly, especially in the presence of ongoing measles circulation. Moreover, the increase of measles incidence and sustained measles transmission is believed to be mainly attributable to insufficiently high vaccine-induced immunization within populations, i.e., sub-optimal vaccination coverage levels below the critical vaccination coverage threshold or the presence of clusters of unimmunised people. However, current immunity gaps might widen even further as a result of the aforementioned secondary vaccine failure. Despite the availability of only a limited number of eligible studies in their review and meta-analysis, Hughes et al. (2020) [15] show a decreasing trend in two dose vaccine effectiveness as time since the first dose of measles-containing vaccines increases in eliminating settings. The authors underline the importance of conducting more vaccine effectiveness studies to monitor what would happen in a community relying solely on vaccine-induced immunity [15]. Information about the vaccination status of measles cases could be used to unravel the contribution of primary/secondary vaccine failure on the one hand and suboptimal vaccination coverage on the other hand to measles outbreaks. Unfortunately, incomplete registration (including unknown status mainly by the adult population) of the vaccination status of measles cases in Belgium currently hampers such an assessment.
Attaining herd immunity levels for measles is paramount to reaching the elimination goals of measles by 2030, as measles incidence reached the highest reported numbers in 2019 [16]. On top of that, vaccine refusal in European countries, and elsewhere, is increasing, potentially even worsened as a result of mistrust and vaccine hesitancy with regard to COVID-19 vaccination. Vaccine-derived immunity will need to be sustained and increased to reach those elimination goals, potentially through additional vaccination efforts in those birth cohorts that received their two MMR doses first (i.e., in the oldest birth cohorts eligible for MMR vaccination). At present, the COVID-19 pandemic could negatively impact the immunization coverage worldwide [42], thereby even increasing the need for additional immunization programs in groups of children that are impacted by this. Accordingly, measles elimination is a fragile state that needs to be continuously maintained because of the likelihood of measles importations [37]. This is exemplified by the current COVID-19 pandemic in which the emergence of new variants of concern (i.e., variants of the original SARS-CoV-2 strain) leads to large scale outbreaks throughout Europe by “traveling” from one place to another at a rapid speed. Furthermore, given that SARS-CoV-2 is considerably less contagious than measles (e.i., basic reproduction number 2–4 versus 12–18 for measles) and Belgium is an interactive country, measles importations are likely to occur and the risk of outbreaks is large following importation. In the past, at least 15% and 10% of measles cases were probably imported in Belgium in 2018 and 2019, respectively [24], [43].
In this paper we monitor existing immunity gaps, as we expect an upswing in measles outbreaks in the foreseeable future. We present a simple cohort model relying on available serological data from three cross-sectional surveys conducted in a highly vaccinated Belgian population and vaccination coverage data to evaluate the evolution of measles seroprevalence in Belgium. Although an assessment of the three components of the MMR vaccine would be interesting (see, e.g., Wood et al., 2015 [26]), especially given shared features such as vaccine uptake, we focus in this manuscript on measles separately. A combined study is considered as future research. We have looked at seroprevalence levels in this study, however an evaluation of measles IgG levels itself [44], [45] would possible be more profound. This is considered as future research. Looking at the seroprevalence for measles, both Flanders and Wallonia & Brussels Capital Region share similar seroprevalence profiles. The proposed model allows for the estimation of time-varying exposure, and accommodates primary and secondary vaccine failure effects on the seroprevalence profile, accounting for the variability in a Bayesian perspective. Although, in general, for measles, the contributing factor of waning of vaccine-induced immunity is considered small to moderate as compared to insufficient immunization of persons (i.e., vaccination coverages that are too low), the proposed model relies on such information through informative priors for seroconversion and waning parameters [11]. Consequently, the cohort model disentangles the extent of primary and secondary vaccine failure from exposure to measles circulation in the population. Based on the recent resurgence in measles cases throughout Europe, and more specifically in Belgium as well, the model seems to capture the current increasing trend in exposure affecting in particular the youngest birth cohorts. A loss of sero-positivity after vaccination has been observed in a recent systematic review and meta-analysis [11], thereby focusing on persistence of humoral immunity at the individual level. At a population level, however, the biological waning process is typically accompanied with boosting events in case of measles circulation, an increase in humoral immunity as a result of catch-up vaccination campaigns, immigration and emigration of individuals, etc. This interplay makes it difficult to disentangle these different contributions to the overall sero-positivity observed for a specific birth cohort at a given calendar time. From a policy perspective, it is important to mention that waning of humoral immunity is related to the MMR vaccine itself, whereas vaccination coverage is something which could be improved through measles vaccination campaigns and campaigns oriented towards increasing awareness regarding the consequences of a lowering vaccine uptake over time. Although a 95% vaccination coverage would aid in attaining herd immunity, an even higher vaccination coverage could mitigate the potential issue of waning of vaccine-induced humoral immunity.
There are several limitations to the model. First, we focus on the absence of antibodies which does not equate to absence of protection. Cellular immunity also provides some degree of protection. Second, we assume that a second MMR dose has qualitatively the same effect as the first one. Moreover, the seroconversion and waning probability are assumed to be the same after the first and second dose. Serologicial surveys are usually categorized into sero-positive and -negative based on manufacturer’s cut-off values of the ELISA test. Bolotin et al. [46] reflect on the correlate of protection for measles (i.e., 120 mIU/ml), which is mainly based on the work of Chen et al. [47] using a neutralization essay. The ELISA assays using in the seroprevalence studies included in our study used a higher cut-off (150–325/350 mIU/ml) to not overestimate protection, as an ELISA test may also detect non-neutralising antibodies. Still, it cannot be excluded that a binary classification into seropositive and –negative individuals based on their antibody titer levels causes some misclassification, as was illustrated before by studies reported in the review by Bolotin et al. [46] as well as other modelling studies focusing on dichotomous seroprevalence data (e.g. Bollaerts et al. [48]). Next, there is plenty of uncertainty concerning the vaccination coverage estimates, as available data points are scarce, which might result in over- or underestimation of vaccination coverages. However, in the Bayesian MCMC approach, this uncertainty is accounted for in the estimation of the underlying seroprevalence. Finally, the model presumes the exposure rate to be constant for birth cohorts (born prior to 1984) without universal vaccination. Although this is a simplification of reality, observed seroprevalences are very high in those cohorts due to previous exposure to circulating measles virus, and the overall exposure rate is to be considered as an average exposure for those cohorts in the past.
Despite these limitations, the cohort model seems to describe the evolution of measles seroprevalence in Belgium quite well and points out where the possible immunity gaps might be situated in the future. Belgium has undertaken several steps to decrease immunity gaps by introducing catch-up campaigns and advancing MMR2 to 6 years (in Flanders) and 7–9 years (in Wallonia & Brussels Capital Region). Until MMR2 vaccination reaches a coverage of at least 95%, outbreaks are to be expected in Belgium. The results of this paper underline, once more, the necessity of continuing to administer two doses, to improve the registration of vaccination status, and consider administering an additional dose to individuals with ambiguous vaccination status. Besides reaching and maintaining high vaccination coverage levels in young infants and children, the question remains whether waning of vaccine-induced humoral immunity might pose a predicament in the near future. Even though it is not possible to obtain conclusive evidence of waning based on empirical data, it is something that requires close surveillance in the future. More research is needed on the frequency and timing of MMR vaccination in general as well as more studies examining the degree of waning in the absence of circulating measles. Although this exercise focuses on Belgium, such immunization efforts are relevant and important for other countries across Europe with moderate to high MMR vaccine uptake while facing a similar measles outbreak risk.
Author contributors
JS, SA and NH designed the study and NH conceived the project. JS and SA performed data analysis, data interpretation and writing of the first draft of the manuscript. JS made the tables and figures. All authors contributed to, reviewed, and approved the final version of the manuscript.
Declaration of Competing Interest
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Niel Hens reports financial support was provided by European Research Council. Julie Schenk reports financial support was provided by European Research Council. Niel Hens reports a relationship with GSK. Niel Hens reports a relationship with Pfizer Inc. Niel Hens reports a relationship with Johnson & Johnson. Heidi Theeten reports a relationship with Pfizer Inc.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.vaccine.2022.05.009.
Appendix A. Supplementary data
The following are the Supplementary data to this article:
References
- 1.WHO. Measles vaccines: WHO position paper – April 2017. 2017 [22/10/2021]. Available from: http://www.who.int/wer.
- 2.Dabbagh A., Laws R.L., Steulet C., Dumolard L., Mulders M.N., Kretsinger K., et al. Progress toward regional measles elimination — worldwide, 2000–2017. MMWR Morb Mortal Wkly Rep. 2018;67(47):1323–1329. doi: 10.15585/mmwr.mm6747a6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dixon M.G., Ferrari M., Antoni S., Li X.i., Portnoy A., Lambert B., et al. Progress toward regional measles elimination — worldwide, 2000–2020. MMWR Morb Mortal Wkly Rep. 2021;70(45):1563–1569. doi: 10.15585/mmwr.mm7045a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Patel M.K., Goodson J.L., Alexander J.P., Kretsinger K., Sodha S.V., Steulet C., et al. Progress toward regional measles elimination — worldwide, 2000–2019. MMWR Morb Mortal Wkly Rep. 2020;69(45):1700–1705. doi: 10.15585/mmwr.mm6945a6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.WHO. Measles resported cases and incidence 2021 [07/04/2022]. Available from: https://immunizationdata.who.int/pages/incidence/measles.html.
- 6.WHO. 9th Meeting of the European Regional Verification Commission for Measles and Rubella Elimination (RVC) (2021). 2021.
- 7.WHO. Strengthening response to measles outbreak in Ukraine. 2020.
- 8.CDC. Measles Cases and Outbreaks. 2021 [22/10/2021]. Available from: https://www.cdc.gov/measles/cases-outbreaks.html.
- 9.Craig A.T., Heywood A.E., Worth H. Measles epidemic in Samoa and other Pacific islands. Lancet Infect Dis. 2020;20(3):273–275. doi: 10.1016/S1473-3099(20)30053-0. [DOI] [PubMed] [Google Scholar]
- 10.Nic Lochlainn L.M., de Gier B., van der Maas N., Strebel P.M., Goodman T., van Binnendijk R.S., et al. Immunogenicity, effectiveness, and safety of measles vaccination in infants younger than 9 months: a systematic review and meta-analysis. Lancet Infect Dis. 2019;19(11):1235–1245. doi: 10.1016/S1473-3099(19)30395-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schenk J., Abrams S., Theeten H., Van Damme P., Beutels P., Hens N. Immunogenicity and persistence of trivalent measles, mumps, and rubella vaccines: a systematic review and meta-analysis. Lancet Infect Dis. 2021;21(2):286–295. doi: 10.1016/S1473-3099(20)30442-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gardner L., Dong E., Khan K., Sarkar S. Persistence of US measles risk due to vaccine hesitancy and outbreaks abroad. Lancet Infect Dis. 2020;20(10):1114–1115. doi: 10.1016/S1473-3099(20)30522-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kuylen E., Willem L., Broeckhove J., Beutels P., Hens N. Clustering of susceptible individuals within households can drive measles outbreaks: an individual-based model exploration. Sci Rep. 2020;10(1) doi: 10.1038/s41598-020-76746-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Funk S., Knapp J.K., Lebo E., Reef S.E., Dabbagh A.J., Kretsinger K., et al. Combining serological and contact data to derive target immunity levels for achieving and maintaining measles elimination. BMC Med. 2019;17(1) doi: 10.1186/s12916-019-1413-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hughes S.L., Bolotin S., Khan S., Li Y.e., Johnson C., Friedman L., et al. The effect of time since measles vaccination and age at first dose on measles vaccine effectiveness - A systematic review. Vaccine. 2020;38(3):460–469. doi: 10.1016/j.vaccine.2019.10.090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.WHO. Worldwide measles deaths climb 50% from 2016 to 2019 claiming over 207 500 lives in 2019. 2020 [18/03/2021]. Available from: https://www.who.int/news/item/12-11-2020-worldwide-measles-deaths-climb-50-from-2016-to-2019-claiming-over-207-500-lives-in-2019.
- 17.WHO. Implementing the Immunization Agenda 2030 2021. Available from: https://www.who.int/teams/immunization-vaccines-and-biologicals/strategies/ia2030.
- 18.Lernout T., Theeten H., Hens N., Braeckman T., Roelants M., Hoppenbrouwers K., et al. Timeliness of infant vaccination and factors related with delay in Flanders. Belgium Vaccine. 2014;32(2):284–289. doi: 10.1016/j.vaccine.2013.10.084. [DOI] [PubMed] [Google Scholar]
- 19.De Schutter I. Vaccinatie tegen mazelen, bof en rubella bij volwassenen 2018. Available from: https://www.zorg-en-gezondheid.be/sites/default/files/atoms/files/brief%20mazelenvaccinatie%20september%202018%20%28002%29.pdf.
- 20.La rougeole : l’unique protection est la vaccination. 2020 [15/02/2022]. Available from: https://www.vaccination-info.be/la-rougeole-lunique-protection-est-la-vaccination/.
- 21.Adultes. 2021 [15/02/2022]. Available from: https://www.vaccination-info.be/adultes/.
- 22.Permentier B. Le programme de vaccination de la federation Wallonie-Bruxelles pour les SPSE et le CPMS-WBE. 2021 [15/02/2022]. Available from: https://www.e-vax.be/VaccHelp/help/pdf/memo_rentree_vaccination_spse_cpms.pdf.
- 23.ECDC. Disease Data from ECDC Surveillance Atlas for Measles. 2022 [27/01/2022]. Available from: https://www.ecdc.europa.eu/en/measles/surveillance-and-disease-data/atlas.
- 24.Grammens T, Cornelissen L, Hutse V, Abady M, Theeten H, Wyndham-Thomas C, et al. Epidemiologische surveillance van mazelen Morbillivirus-2019. 2019.
- 25.Hens N., Abrams S., Santermans E., Theeten H., Goeyvaerts N., Lernout T., et al. Assessing the risk of measles resurgence in a highly vaccinated population: Belgium anno 2013. Euro Surveill. 2015;20(1) doi: 10.2807/1560-7917.es2015.20.1.20998. [DOI] [PubMed] [Google Scholar]
- 26.Wood JG, Goeyvaerts N, MacIntyre CR, Menzies RI, McIntyre PB, Hens N. Estimating vaccine coverage from serial trivariate serologic data in the presence of waning immunity. Epidemiology (Cambridge, Mass). 2015;26(3):381-9. Epub 2015/03/13. doi: 10.1097/ede.0000000000000278. PubMed PMID: 25764249 [DOI] [PubMed]
- 27.Andrews N., Tischer A., Siedler A., Pebody R.G., Barbara C., Cotter S., et al. Towards elimination: measles susceptibility in Australia and 17 European countries. Bull World Health Organ. 2008;86(3):197–204. doi: 10.2471/blt.07.041129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Theeten H., Hutse V., Hens N., Yavuz Y., Hoppenbrouwers K., Beutels P., et al. Are we hitting immunity targets? The 2006 age-specific seroprevalence of measles, mumps, rubella, diphtheria and tetanus in Belgium. Epidemiol Infect. 2011;139(4):494–504. doi: 10.1017/S0950268810001536. [DOI] [PubMed] [Google Scholar]
- 29.Litzroth A., Suin V., Wyndham-Thomas C., Quoilin S., Muyldermans G., Vanwolleghem T., et al. Low hepatitis C prevalence in Belgium: implications for treatment reimbursement and scale up. BMC Public Health. 2019;19(1) doi: 10.1186/s12889-018-6347-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tischer A., Andrews N., Kafatos G., Nardone A., Berbers G., Davidkin I., et al. Standardization of measles, mumps and rubella assays to enable comparisons of seroprevalence data across 21 European countries and Australia. Epidemiol Infect. 2007;135(5):787–798. doi: 10.1017/S0950268807008266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Plotkin S.A. Complex correlates of protection after vaccination. Clin Infect Dis. 2013;56(10):1458–1465. doi: 10.1093/cid/cit048. [DOI] [PubMed] [Google Scholar]
- 32.Strebel P.M., Papania M.J., Fiebelkorn A.P., Halsey N.A. Measles vaccine. Vaccines Sixth Edition ed: Elsevier. 2013:352–387. [Google Scholar]
- 33.Carpenter B., Gelman A., Hoffman M.D., Lee D., Goodrich B., Betancourt M., et al. Stan: A Probabilistic Programming Language. J Stat Softw. 2017;76(1) doi: 10.18637/jss.v076.i01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Team R. R: A Language and Environment for Statistical Computing. 2021. Available from: https://www.r-project.org/.
- 35.Sabbe M, Hue D, Hutse V, Goubau P. Measles resurgence in Belgium from January to mid-April 2011: a preliminary report. 2011 2011/4//. Report No. [PubMed]
- 36.WHO. Measles. 2019 [19/01/2022]. Available from: https://www.who.int/en/news-room/fact-sheets/detail/measles.
- 37.Winter AK, Martinez ME, Cutts FT, Moss WJ, Ferrari MJ, McKee A, et al. Benefits and Challenges in Using Seroprevalence Data to Inform Models for Measles and Rubella Elimination. J Infect Dis 2018;218(3):355-64. Epub 2018/03/22. doi: 10.1093/infdis/jiy137. PubMed PMID: 29562334; PubMed Central PMCID: PMCPMC6049004. [DOI] [PMC free article] [PubMed]
- 38.Bianchi F.P., Mascipinto S., Stefanizzi P., De Nitto S., Germinario C., Tafuri S. Long-term immunogenicity after measles vaccine vs. wild infection: an Italian retrospective cohort study. Human Vaccines and Immunotherapeutics. 2021;17(7):2078–2084. doi: 10.1080/21645515.2020.1871296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bernard H., Fischer R., Wildner M. Ongoing measles outbreak in southern Bavaria, Germany. Euro surveillance : bulletin Europeen sur les maladies transmissibles = Eur Commun Dis Bull. 2008;13(1):2–3. [PubMed] [Google Scholar]
- 40.Siani A. Measles outbreaks in Italy: A paradigm of the re-emergence of vaccine-preventable diseases in developed countries. Prev Med. 2019;121:99–104. doi: 10.1016/j.ypmed.2019.02.011. [DOI] [PubMed] [Google Scholar]
- 41.Smithson R, Irvine N, Hutton C, Doherty L, Watt A. Spotlight on measles 2010: ongoing measles outbreak in Northern Ireland following an imported case, September-October 2010. Euro surveillance : bulletin Europeen sur les maladies transmissibles = Eur Commun Dis Bull 2010;15(43). doi: 10.2807/ese.15.43.19698-en. [DOI] [PubMed]
- 42.Bramer C.A., Kimmins L.M., Swanson R., Kuo J., Vranesich P., Jacques-Carroll L.A., et al. Decline in child vaccination coverage during the COVID-19 pandemic - Michigan care improvement registry, May 2016-May 2020. MMWR Morb Mortal Wkly Rep. 2020;69(20):630–631. doi: 10.15585/mmwr.mm6920e1. [DOI] [PubMed] [Google Scholar]
- 43.Cornelissen L, Hutse V, Theeten H, Wyndham-Thomas C. Epidemiologische surveillance van mazelen Morbillivirus- 2018. 2018.
- 44.Marchi S., Monti M., Viviani S., Remarque E.J., Esposito S., Principi N., et al. Immunity to measles in Italian children and adolescents: a persistent problem in view of measles elimination. J Public Health. 2021;43(4):e601–e609. doi: 10.1093/pubmed/fdaa153. [DOI] [PubMed] [Google Scholar]
- 45.Marchi S., Remarque E.J., Viviani S., Rizzo C., Monteverde Spencer G.T., Coluccio R., et al. Measles immunity over two decades in two large Italian Regions: How far is the elimination goal? Vaccine. 2021;39(40):5928–5933. doi: 10.1016/j.vaccine.2021.08.001. [DOI] [PubMed] [Google Scholar]
- 46.Bolotin S., Hughes S.L., Gul N., Khan S., Rota P.A., Severini A., et al. What is the evidence to support a correlate of protection for measles? A systematic review. J Infect Dis. 2020;221(10):1576–1583. doi: 10.1093/infdis/jiz380. [DOI] [PubMed] [Google Scholar]
- 47.Chen R.T., Markowitz L.E., Albrecht P., Stewart J.A., Mofenson L.M., Preblud S.R., et al. Measles antibody: reevaluation of protective titers. J Infect Dis. 1990;162(5):1036–1042. doi: 10.1093/infdis/162.5.1036. [DOI] [PubMed] [Google Scholar]
- 48.Bollaerts K., Aerts M., Shkedy Z., Faes C., Van der Stede Y., Beutels P., et al. Estimating the population prevalence and force of infection directly from antibody titres. Statistical Modelling. 2012;12(5):441–462. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.