Abstract
Patients with pre‐existing liver diseases are considered to have an increased risk of morbidity and mortality from any type of infection, including viruses. The aim of this work was to explore the implications of metabolic dysfunction‐associated fatty liver disease (MAFLD) and nonalcoholic fatty liver disease (NAFLD) definitions in coronavirus disease 2019 (COVID‐19) and to study the interaction between advanced fibrosis (AF) and each of these diseases in the death and intubation of patients hospitalized with COVID‐19. We performed a retrospective study with 359 patients hospitalized with confirmed COVID‐19 infection in a tertiary referral hospital who were admitted between April and June 2020. A multivariate Cox model was performed regarding the interaction of AF with MAFLD and NAFLD in the mortality and intubation of patients with COVID‐19. The death rate was statistically significantly higher in the MAFLD group compared to the control group (55% vs. 38.3%, p = 0.02). No significant difference was seen in the death rate between the NAFLD and control group. The MAFLD (44.09% vs. 20%, p = 0.001) and NAFLD (40.51% vs. 20%, p = 0.01) groups had statistically significantly higher intubation rates than the control group. A statistically significant interaction between NAFLD and AF was associated with an increase in mortality (p = 0.01), while a statistically significant interaction between MAFLD and AF was associated with an increased risk of mortality (p = 0.006) and intubation (p = 0.049). In the case of patients hospitalized with COVID‐19, our results indicate that the death rate was higher in the MAFLD group but not the NAFLD group compared to that in the control group. The intubation rates were higher in the NAFLD and MAFLD groups compared to rates in the control group, suggesting that both could be associated with COVID‐19 severity. In addition, we found interactions between AF with MAFLD and NAFLD.
The association of MAFLD and NAFLD with severe COVID‐19 disease and mortality could be related to the low proinflammatory state that has been associated with metabolic dysregulation. This is the first study that reports a significant interaction of NAFLD and MAFLD with AF in the deaths and intubations of patients hospitalized with COVID‐19.

INTRODUCTION
Liver health is critical for an effective immune response. The liver is a major source of proteins for the innate and adaptive immune responses (e.g., complement components and acute‐phase proteins responding to different proinflammatory cytokines). In addition, hepatocytes express different pattern recognition receptors, such as toll‐like receptors, cytoplasmic nucleotide‐binding oligomerization domain‐like receptors, and RNA helicase.[ 1 ]
Patients with pre‐existing liver diseases, such as cirrhosis and cancer, are considered to have an increased risk of morbidity and mortality from any type of infection, including viruses.[ 1 ] At the beginning of the coronavirus disease 2019 (COVID‐19) pandemic, it did not seem that hepatic damage could have a role in the death toll caused by COVID‐19. However, many reports started showing an association between COVID‐19 and alteration of liver function tests,[ 2 ] and patients with cirrhosis seemed to have a higher death rate.[ 3 ] Several studies have found an association between metabolic dysfunction‐associated fatty liver disease (MAFLD) and nonalcoholic fatty liver disease (NAFLD) with an increased risk of developing severe COVID‐19 disease. However, there is controversy whether NAFLD and MAFLD could be associated with a higher death rate from COVID‐19.[ 4 , 5 ]
MAFLD is one of the most common conditions worldwide, and it is estimated to be the most frequent chronic hepatic disease. In Mexico, MAFLD is a public health problem as it is present in more than 30% of its general population.[ 6 ] MAFLD was distinguished from NAFLD in 2020. One of the main aspects of MAFLD is that it focuses on metabolic dysregulation as the root of the condition. It also has positive criteria instead of being a diagnosis of exclusion.[ 7 , 8 ] MAFLD is characterized by “low‐grade chronic inflammation with endothelial dysfunction, changes in the function of monocytes, and altered antigen presentation.” In the case of COVID‐19, mechanisms involved in acute inflammation could overlap with the proinflammatory state of MAFLD, occasioning immune system dysregulation with excessive release of proinflammatory mediators (cytokine storm [CS]) that could increase the probability of organ damage or even multiple organ failure.[ 8 , 9 ]
Hepatic fibrosis is the final common result of injury to the liver. It occurs not only in patients with chronic liver disease but also in other conditions, such as obesity, diabetes mellitus, hypertension, and chronic kidney disease.[ 10 , 11 ] Hepatic fibrogenesis could progress to a stage called advanced fibrosis (AF), which is related to several clinical settings (including portal hypertension, variceal bleeding, hepatorenal syndrome, hepatic encephalopathy, and ascites).[ 12 ] The relation between liver fibrosis and death from COVID‐19 has been documented in few retrospective studies. High values for the fibrosis‐4 (FIB‐4) index for liver fibrosis have been related to a higher death rate, longer hospital stay, and higher need for mechanical ventilation.[ 13 , 14 ] However, different cut‐off points for the FIB‐4 index have been employed, leading to contradictory results, and some studies have not considered the most reported characteristics associated with the severity of COVID‐19 as confounding variables. Furthermore, an interaction of MAFLD and NAFLD with AF has not been considered.
The aim of this work was to explore the implications of MAFLD and NAFLD definitions in COVID‐19 and study the interaction between AF and each of these diseases in the death and intubation of patients hospitalized with COVID‐19.
MATERIALS AND METHODS
Patients and methods
We performed a retrospective study. All patients admitted to a tertiary referral hospital (Central Military Hospital [CMH], Secretaría De La Defensa Nacional) located in Mexico City, Mexico, with confirmed COVID‐19 infection from April 4, 2020, to June 24, 2020, were studied. COVID‐19 infection was confirmed using real‐time reverse transcription polymerase chain reaction on nasopharyngeal swabs. Epidemiological data (age, sex, body mass index [BMI], type 2 diabetes, hypertension, and obesity), outcomes (length of hospital stay, death, and intubation), and laboratory test results (hematological biometry, blood chemistry, and liver function tests) were obtained from the electronic health record (Digital Health System) of the CMH. We included the patients who died or were discharged between April 4, 2020, and June 24, 2020. Patients without liver function tests in the first 24 hours of hospitalization were excluded. In addition, patients with significant consumption of alcohol (>30 g/day for men and >20 g/day for women); known history of autoimmune liver disease, liver cancer, decompensated cirrhosis, platelet disorders, or myopathies; or incomplete data were excluded.
The following three groups were formed: (1) patients without NAFLD or MAFLD (control); (2) patients with NAFLD not meeting the criteria for MAFLD; and (3) patients with MAFLD. These groups were also divided depending on the presence or absence of AF. MAFLD was diagnosed according to the international expert consensus statement as the presence of steatosis with a noninvasive method and one of the following: (a) overweight BMI (25–29.9 kg/m2); (b) type 2 diabetes; and (c) presence of two metabolic abnormalities (blood pressure >140/90 mm Hg, plasma triglycerides >150 mg/dL, plasma high‐density lipoprotein cholesterol [HDL‐C] <40 mg/dL in men and <50 mg/dL in women, and prediabetes).[ 7 ] Patients with hepatic steatosis but not meeting the criteria to diagnose MAFLD were included in the group with NAFLD.
In this study, hepatic steatosis was identified with a validated instrument with the following regression model (RM): 113.163 + 0.252 × ALT + 6.316 × BMI. A cut‐off value of 277.62 was established; at higher values, it has a sensitivity of 87.68% and a specificity of 90% in the identification of liver steatosis. We decided to use the RM instrument given the higher accuracy of the instrument identifying liver steatosis compared to the FibroScan (82.6% vs. 69.6%).[ 15 ] We had insufficient information to calculate other instruments of liver steatosis.
Fibrosis was evaluated with the FIB‐4 index using the following formula: FIB‐4 = [age × aspartate aminotransferase (AST)]/{platelets × [alanine aminotransferase (ALT)^1/2]}, and AF was identified with a FIB‐4 index value >2.67.[ 16 ]
Statistical analysis
Data are presented using mean (SD) and count (percentage [%]) for numerical and categorical variables, respectively. Numerical variables were compared using the two‐tailed t test or one‐way analysis of variance, depending on the number of compared groups. The assumption of the equality of variances across groups was assessed with Bartlett's test. The normal distribution of the residuals in each group was verified. Categorical variables were compared using Fisher's exact test. The death rate and intubation rate among the groups were compared, and a subgroup analysis was performed between the control group and each of the following three groups: NAFLD, MAFLD, and the overlap of MAFLD and NAFLD.
An analysis of the interaction of MAFLD and NAFLD with AF in the mortality and intubation of patients hospitalized with COVID‐19 was performed with a multivariate Cox proportional hazard regression (CPHR) model. The covariates used in the multivariate model were considered based on univariate analysis and by its clinical importance as the main factors for mortality and intubation described in this population.[ 17 , 18 ] The adjustment variables included age (continuous variable), sex, type 2 diabetes, hypertension, overweight (BMI, 25–29.9 kg/m2), and obesity (BMI, ≥30 kg/m2). Hazard ratios (HRs) were estimated as a measure of effect size of the variables included in the CPHR model. Time considered in the CPHR model was days of hospitalization to the outcomes. The outcomes studied were the occurrence of death or intubation during the hospitalization of patients with COVID‐19. In the death outcome, patients who recovered and were discharged from the hospital were censored; in the intubation outcome, patients who died during hospitalization or recovered and were discharged from the hospital without being intubated were censored. The goodness‐of‐fit of the CPHR models was estimated with the likelihood ratio chi‐square test. The proportional hazard assumption was evaluated with the Schoenfeld residuals method.
In the case of statistically significant interactions, a subgroup analysis among factors (MAFLD, NAFLD, and AF) was performed. Post hoc analysis was performed using 2 × 2 contingency tables with Fisher´s exact test. When comparing more than three groups, α was adjusted with the Bonferroni correction for categorical variables according to all the possible number of comparisons using the following inequality: adjusted p < Bonferroni‐adjusted α value = α/number of comparisons.
Statistical analyses were performed using IBM SPSS Statistics for Windows, version 27.0., whereas bar graphs and forest plots were elaborated in GraphPad Prism for Windows, version 8.4.0.
Ethical considerations
This study was approved (registration no. 066/2020) by the Research Committee at the CMH. The present study complies with the basic principles of human research following the Declaration of Helsinki. In this study, we used a de‐identified version of this data set, with the assent of the Research Committee at the CMH to process patient confidential data without explicit patient consent. All methods were carried out in accordance with relevant guidelines and regulations.
RESULTS
A total of 359 patients were included in the study. The mean age was 54.3 years (SD, 14.69 years), 257 (71.59%) were men, 79 (22.01%) had NAFLD, and 220 (61.21%) had MAFLD. The three most frequent comorbidities were overweight (33.43%), hypertension (15.32%), and type 2 diabetes (14.48%). The most frequent symptoms were fever (68.8%), dyspnea (64.9%), and cough (58.5%).
Of these patients, 141 (39.28%) were intubated and 185 (51.53%) died. The mean length of stay was 11.11 days (SD, 8.97). Nonsurvivors were older, with a mean age of 58.4 years (SD, 13.88 years); these patients had higher frequencies of both hypertension (20.54% vs. 9.77%, p = 0.005) and obesity (13.51% vs. 6.32%, p = 0.03) compared to survivors. In regard to the laboratory tests, nonsurvivors had higher levels of leukocytes 103/µL (mean, 11.05; SD, 5.14 vs. mean, 7.95; SD, 3.82; p = 0.001), neutrophils 103/µL (mean, 9.99; SD, 7.86 vs. mean, 6.5; SD, 3.9; p < 0.001), glucose mg/dL (mean, 175.05; SD, 108.43 vs. mean, 130.6; SD, 86.24; p < 0.001), urea mg/dL (mean, 65.96; SD, 48.69 vs. mean, 43.66; SD, 45.04; p < 0.001), triglycerides mg/dL (mean, 250.4; SD, 304.15 vs. mean, 216.75; SD, 150.1; p < 0.001), C‐reactive protein (CRP) mg/dL (mean, 188.93; SD, 135.39 vs. mean, 120.89; SD, 108.24; p < 0.001), and lactate dehydrogenase (LDH) UI/L (mean, 840.33; SD, 878.08 vs. mean, 560.06; SD, 348.02; p < 0.001) as well as lower levels of albumin g/dL (mean, 3; SD, 0.65 vs. mean, 3.67; SD, 2.45; p < 0.001) and HDL‐C mg/dL (mean, 22.55; SD, 9.39 vs. mean, 32.13; SD, 19.32; p < 0.001) (Table 1).
TABLE 1.
Comparison of epidemiological data and baseline laboratory tests between survivor and nonsurvivor patients with COVID‐19
| Variable | Overall | Survivors | Nonsurvivors | p value |
|---|---|---|---|---|
| n = 359 | n = 174 | n = 185 | ||
| Characteristics | ||||
| Age in years, mean (SD) | 54.3 (14.69) | 50 (14.31) | 58.4 (13.88) | <0.001 a |
| SBP, mm Hg | 124.71 (26.71) | 122.73 (15.62) | 126.58 (33.39) | 0.1 |
| DBP, mm Hg | 73.21 (12‐9) | 74.46 (11.09) | 72.04 (14.33) | 0.07 |
| Sex (male) n (%) | 257 (71.59) | 123 (47.86) | 134 (52.14) | 0.7 b |
| Intubation | 141 (39.28) | 18 (12.77) | 123 (87.23) | <0.001 |
| Symptoms | ||||
| Fever, n (%) | 247 (68.8) | 122 (49.39) | 125 (50.61) | 0.6 b |
| Dyspnea | 233 (64.9) | 112 (64.37) | 121 (65.41) | 0.9 |
| Cough | 210 (58.5) | 103 (59.2) | 71 (40.8) | 0.8 |
| Myalgia | 151 (42.06) | 75 (43.1) | 76 (41.08) | 0.7 |
| Arthralgia | 146 (40.67) | 72 (41.38) | 74 (70) | 0.8 |
| Headache | 75 (20.89) | 39 (22.41) | 36 (19.46) | 0.5 |
| Pharyngalgia | 53 (14.76) | 30 (56.6) | 23 (43.4) | 0.2 |
| Chest pain | 36 (10.03) | 18 (10.34) | 18 (9.73) | 0.8 |
| Diarrhea | 23 (6.41) | 12 (6.9) | 11 (5.95) | 0.8 |
| Vomiting | 14 (3.9) | 10 (5.75) | 4 (2.16) | 0.1 |
| Anosmia | 6 (1.61) | 4 (2.3) | 2 (1.08) | 0.4 |
| Comorbilities | ||||
| Overweight n (%) | 120 (33.43) | 59 (33.91) | 61 (32.97) | 0.9 b |
| Hypertension | 55 (15.32) | 17 (9.77) | 38 (20.54) | 0.005 |
| Type 2 diabetes | 52 (14.48) | 21 (12.07) | 32 (17.3) | 0.1 |
| Obesity | 36 (10.03) | 11 (6.32) | 25 (13.51) | 0.03 |
| COPD | 11 (3.06) | 7 (4.02) | 4 (2.16) | 0.2 |
| Laboratory tests | ||||
| Hemoglobin in g/dL, mean (SD) | 14.36 (3.67) | 14.6 (4.32) | 14.1 (2.92) | 0.2a |
| Platelets, 103 | 238.97 (114.43) | 233.95 (110.6) | 243.7(118.01) | 0.4 |
| Leukocytes, 103/µL | 9.55 (4.8) | 7.95 (3.82) | 11.05 (5.14) | <0.001 |
| Lymphocytes, 103/µL | 1.12 (0.79) | 1.17 (0.81) | 1.06 (0.77) | 0.2 |
| Neutrophils, 103/µL | 8.34 (6.5) | 6.5 (3.9) | 9.99 (7.86) | <0.001 |
| Creatinine, mg/dL | 1.97 (6.58) | 2.21 (9.18) | 1.75 (2.22) | 0.5 |
| Glucose, mg/dL | 153.51 (101.01) | 130.6 (86.92) | 175.05 (108.43) | <0.001 |
| Urea, mg/dL | 55.18 (48.21) | 43.66 (45.04) | 65.96 (48.69) | <0.001 |
| ALT, UI/L | 55.23 (86.7) | 47.68 (33.08) | 62.32 (116.16) | 0.1 |
| AST, UI/L | 79.14 (368.8) | 48.11(28.79) | 108.32 (511.97) | 0.1 |
| Albumin, g/dL | 3.32 (1.79) | 3.67 (2.45) | 3 (0.65) | <0.001 |
| TB, mg/dL | 0.98 (3.97) | 0.67 (0.4) | 1.28 (5.48) | 0.1 |
| HDL‐C, mg/dL | 27.19 (15.77) | 32.13 (19.32) | 22.55 (9.39) | <0.001 |
| Triglycerides, mg/dL | 273.3 (214.78) | 216.75 (150.1) | 326.49 (250.4) | <0.001 |
| CRP, mg/dL | 156.96 (127.82) | 120.89 (108.24) | 188.93(135.39) | <0.001 |
| LDH, UI/L | 708.89 (696.1) | 560.09 (348.02) | 840.33 (878.08) | <0.001 |
| FIB‐4 score | 3.14 (7.54) | 2.12 (3.51) | 4.1 (9.86) | 0.01 |
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; COPD, chronic obstructive pulmonary disease; COVID‐19, coronavirus disease 2019; CRP, C‐reactive protein; DBP, diastolic blood pressure; FIB‐4, fibrosis‐4; HDL‐C, high‐density lipoprotein cholesterol; LDH, lactate dehydrogenase; SBP, systolic blood pressure; TB, total bilirubin.
Two‐tailed t test for independent measures.
Fisher´s exact test.
A statistically significant difference in the death rate was found between the MAFLD and NAFLD groups compared to the control group (54.18% vs. 38.33%, p = 0.03). The MAFLD group had a statistically significantly higher death rate compared to the control group (55% vs. 38.33%, p = 0.02). However, no differences were found between the NAFLD and control groups. Similarly, the intubation rate was significantly different between the MAFLD and NAFLD groups compared to the control group (43.14% vs. 20%, p < 0.001). The MAFLD (44.09% vs. 20%, p = 0.001) and NAFLD (40.51% vs. 20%, p = 0.01) groups had higher intubation rates compared to the control group (Figure 1). There were statistically significant differences in glucose, triglycerides, and HDL‐C among the groups. Patients with MAFLD had higher levels of glucose than the control and NAFLD groups, while patients with NAFLD and MAFLD had lower levels of HDL‐C and triglycerides compared to the control group (Table 2).
FIGURE 1.
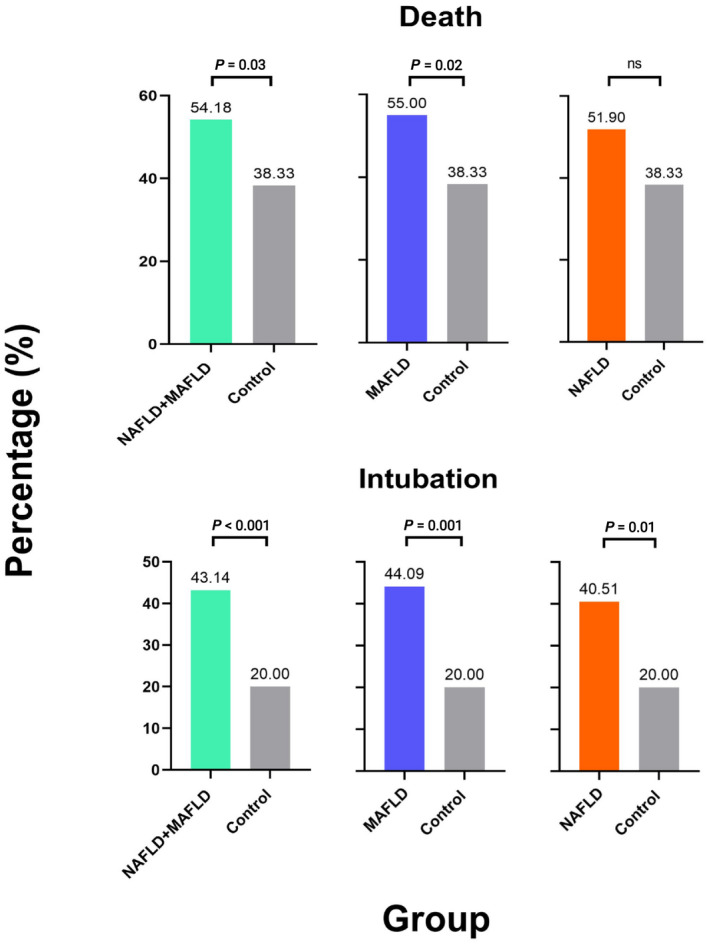
Bar graphs representing subgroup analysis of death and intubation among the groups. MAFLD, metabolic dysfunction‐associated fatty liver disease; NAFLD, nonalcoholic fatty liver disease; ns, not significant
TABLE 2.
Comparison of epidemiological data and baseline laboratory tests between control and patients with NAFLD and MAFLD with COVID‐19
| Variable | Control | NAFLD | MAFLD | p value |
|---|---|---|---|---|
| n = 60 | n = 79 | n = 220 | ||
| Characteristics | ||||
| Age in years, mean (SD) | 55.96 (17.48) | 54.56 (14) | 53.87 (14.13) | 0.6 a |
| Length of stay in days, mean (SD) | 11.18 (7.77) | 11 (9.16) | 11.14 (9.24) | 0.9 |
| SBP, mm Hg, mean (SD) | 124.76 (42.1) | 123.23 (19.2) | 125.23 (22.88) | 0.8 |
| DBP, mm Hg, mean (SD) | 71.1 (10.23) | 71.79 (13.06) | 74.3 (13.42) | 0.1 |
| Sex (male) n (%) | 42 (70) | 62 (78.48) | 153 (69.55) | 0.3 b |
| Death n (%) | 23 (38.33) | 41 (51.9) | 121 (55) | 0.07 |
| Intubated n (%) | 12 (20) | 32 (40.51) | 97 (44.09) | 0.002 |
| Symptoms, n (%) | ||||
| Fever | 39 (65) | 59 (74.68) | 149 (67.73) | 0.4 b |
| Dyspnea | 41 (68.33) | 53 (67.09) | 139 (63.18) | 0.7 |
| Cough | 39 (65) | 49 (62.03) | 122 (55.45) | 0.3 |
| Myalgia | 23 (38.33) | 35 (44.3) | 93 (42.27) | 0.7 |
| Arthralgia | 19 (31.67) | 33 (41.77) | 94 (42.73) | 0.3 |
| Headache | 13 (21.67) | 14 (17.72) | 48 (21.82) | 0.7 |
| Pharyngalgia | 12 (20) | 16 (20.25) | 25 (11.36) | 0.06 |
| Chest pain | 7 (11.67) | 5 (6.33) | 24 (10.91) | 0.4 |
| Diarrhea | 6 (10) | 3 (3.8) | 14 (6.36) | 0.3 |
| Vomiting | 1 (1.67) | 1 (1.27) | 12 (5.45) | 0.2 |
| Anosmia | 1 (1.67) | 1 (1.27) | 4 (1.82) | 1 |
| Comorbilities, n (%) | ||||
| Overweight | 3 (5) | 0 (0) | 117 (53.18) | <0.001 b |
| Hypertension | 9 (15) | 3 (3.8) | 43 (19.55) | 0.002 |
| Type 2 diabetes | 8 (13.33) | 0 (0) | 45 (20.45) | <0.001 |
| Obesity | 0 (0) | 6 (7.59) | 30 (13.64) | 0.002 |
| COPD | 1 (1.67) | 1 (1.27) | 9 (4.09) | 0.6 |
| Laboratory tests, mean (SD) | ||||
| Hemoglobin, mg/dL | 14.14 (6.81) | 14.45 (2.31) | 14.39 (2.75) | 0.8 a |
| Platelets, 103/µL | 256.76 (139.59) | 242.45 (106.17) | 232.87 (109.59) | 0.3 |
| Leukocytes, 103/µL | 8.73 (4.86) | 9.62 (4.55) | 9.75 (4.87) | 0.3 |
| Lymphocytes, 103/µL | 1.05 (0.43) | 0.98 (0.62) | 1.18 (0.91) | 0.1 |
| Neutrophils, 103/µL | 7.16 (4.93) | 8.38 (4.78) | 8.65 (7.34) | 0.2 |
| Creatinine, mg/dL | 2.4 (4.14) | 1.15 (1.22) | 2.15 (8.09) | 0.4 |
| Glucose, mg/dL | 139.4 (91.53) | 123.54 (65.01) | 168.1(110.91) | 0.001 |
| Urea, mg/dL | 57.21 (58.85) | 48.51 (45.06) | 57.03 (46.06) | 0.3 |
| ALT, UI/L | 27.56 (16.58) | 67.54 (49.99) | 58.38 (105.12) | 0.01 |
| AST, UI/L | 40.2 (19.38) | 69.98 (54.67) | 93.4 (469.62) | 0.5 |
| Albumin, mg/dL | 3.79 (4.02) | 3.29 (0.67) | 3.19 (0.70) | 0.07 |
| TB, mg/dL | 0.74 (0.46) | 0.83 (0.56) | 1.11 (5.04) | 0.7 |
| HDL‐ C, mg/dL | 39.91 (29.91) | 26.94 (9.83) | 23.81 (8.68) | <0.001 |
| Triglycerides, mg/dL | 410.08 (353.6) | 230.76 (121.66) | 251.27 (173.74) | <0.001 |
| CRP, mg/dL | 150.23 (137) | 149.49 (124.2) | 161.64 (126.84) | 0.7 |
| LDH, UI/L | 567 (403.03) | 682.59 (414.09) | 754.3 (819.67) | 0.1 |
| FIB‐4 score | 3.45 (7.03) | 3.17 (7.42) | 3.05 (7.75) | 0.9 |
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; COPD, chronic obstructive pulmonary disease; COVID‐19, coronavirus disease 2019; CRP, C‐reactive protein; DBP, diastolic blood pressure; FIB‐4, fibrosis‐4; HDL‐C, high‐density lipoprotein cholesterol; LDH, lactate dehydrogenase; MAFLD, metabolic dysfunction‐associated fatty liver disease; NAFLD, nonalcoholic fatty liver disease; SBP, systolic blood pressure; TB, total bilirubin.
One‐way analysis of variance.
Fisher´s exact test.
The CPHR found statistically significant interactions between AF and MAFLD (p = 0.05) in the intubation outcome. For the death outcome, AF had a statistically significant interaction with both NAFLD (p = 0.01) and MAFLD (p = 0.006) (Table 3). When groups were divided according to the presence of AF, there were statistically significant global differences in mortality (p < 0.001) and intubation (p = 0.002) among them. Bonferroni‐adjusted α established the statistically significant level at adjusted p < 0.0033 for the subgroup analysis. Statistically significant differences in intubation rates were found between the control group with the MAFLD (16.3% vs. 39.9%, adjusted p = 0.003) and MAFLD + AF (16.3% vs. 53.7 %, adjusted p < 0.001) groups. In addition, the subgroup analysis found differences in mortality between the control group with the NAFLD + AF (adjusted p = 0.002) and MAFLD + AF (adjusted p < 0.001) groups (Figure 2; Table 4).
TABLE 3.
Cox proportional hazards regression model analysis of interactions between NAFLD and MAFLD with AF in death and intubation
| AF | NAFLD | Interaction AF × NAFLD | MAFLD | Interaction AF × MAFLD | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| coef (SE) | p value | coef (SE) | p value | coef (SE) | p value | coef (SE) | p value | coef (SE) | p value | |
| Intubation | 0.25 (0.19) | 0.1 | 0.76( 0.34) | 0.02 | 0.14 (0.33) | 0.6 | 1.02 (0.32) | 0.002 | 0.41 (0.21) | 0.049 |
| Death | 0.46 (0.16) | 0.004 | 0.44 (0.27) | 0.9 | 0.65 (0.26) | 0.01 | 0.56 (0.25) | 0.02 | 0.5 (0.18) | 0.006 |
Note: Adjusted to age, sex, and the comorbidities hypertension, type 2 diabetes, overweight, and obesity.
Abbreviations: AF, advanced fibrosis; coef, coefficient; SE, standard error; MAFLD, metabolic dysfunction‐associated fatty liver disease; NAFLD, nonalcoholic fatty liver disease.
FIGURE 2.
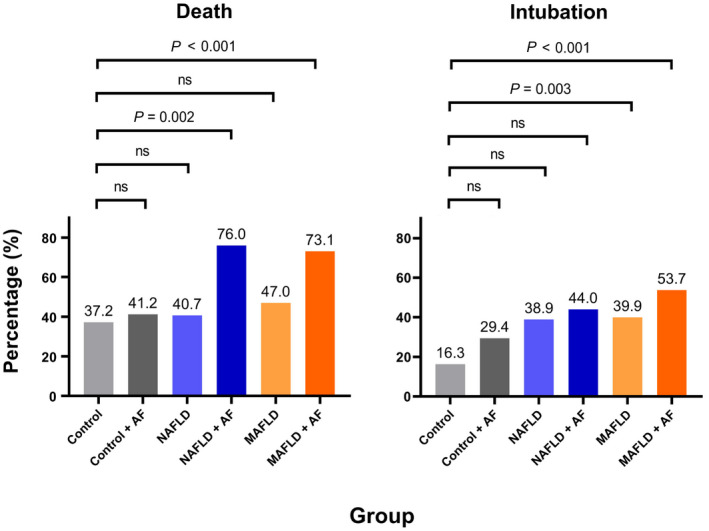
Bar graphs representing post hoc analysis of the association of NAFLD and MAFLD with mortality and intubation compared to the control group when AF was considered. Bonferroni correction for α (0.05) was performed considering all possible comparisons among groups. A significant result is when the adjusted p value is less that Bonferroni‐adjusted α = 0.0033. AF, advanced fibrosis; MAFLD, metabolic dysfunction‐associated fatty liver disease; NAFLD, nonalcoholic fatty liver disease; ns, not significant
TABLE 4.
Comparison of death and intubation rates among groups with advanced fibrosis
| Group | Survivors n = 174 | Nonsurvivors n = 185 | p value |
|---|---|---|---|
| Controla | 27 (15.52) | 16 (8.65) | <0.001* |
| Control and AFabc | 10 (5.75) | 7 (3.71) | |
| NAFLDa | 32 (18.39) | 22 (11.89) | |
| NAFLD and AFbc | 6 (3.45) | 19(10.27) | |
| MAFLDab | 81 (46.55) | 72 (38.92) | |
| MAFLD and AFc | 18 (10.34) | 49 (26.49) |
| Nonintubated n = 218 | Intubated n = 141 | p value | |
|---|---|---|---|
| Controla | 36 (16.5) | 7 (4.96) | 0.004 |
| Control and AFabc | 13 (5.5) | 5 (3.55) | |
| NAFLDabc | 33 (15.14) | 21 (14.89) | |
| NAFLD and AFabc | 14 (6.42) | 11 (7.8) | |
| MAFLDbc | 92 (42.2) | 61 (43.26) | |
| MAFLD and AFc | 31 (14.22) | 36 (25.53) |
Abbreviations: AF, advanced fibrosis; MAFLD, metabolic dysfunction‐associated fatty liver disease; NAFLD, nonalcoholic fatty liver disease.
a–cDifferent supercript letters indicate differences between groups (Bonferroni‐adjusted p < 0.0033).
Significant using Fisher´s exact test.
Adjusted and unadjusted CPHR models are shown in Figure 3. The adjusted model found that patients with NAFLD (adjusted HR, 2.15; 95% confidence interval [CI], 1.1–4.21; p = 0.02) and MAFLD (adjusted HR, 2.79; 95% CI, 1.46–5.31; p = 0.002) had a higher risk of intubation compared to patients in the control group. Patients with MAFLD had an increased risk of death compared to the control group (adjusted HR, 1.75; 95% CI, 1.07–2.88; p = 0.02). Between the adjustment covariables, only age was associated with intubation (adjusted HR, 1.02; 95% CI, 1.01–1.02; p < 0.001) and death (adjusted HR, 1.02; 95% CI, 1.01–1.03; p < 0.001).
FIGURE 3.
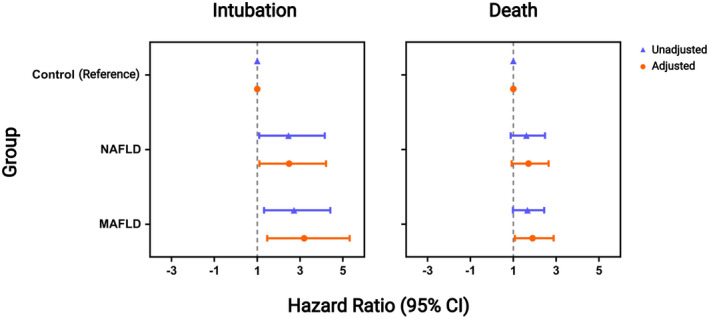
Forest plot with the unadjusted and adjusted hazard ratios for MAFLD and NAFLD relative to the control group. Adjusted models include age, sex, and the comorbidities hypertension, type 2 diabetes, overweight, and obesity. Age was considered a continuous variable. The model for the death outcome had a global likelihood chi‐square test with p = 0.001 and global Schoenfeld residual tests chi‐square with p = 0.3. The model for the intubation outcome had a global likelihood chi‐square test with p = 0.001 and global Schoenfeld residual tests chi‐square with p = 0.8. CI, confidence interval; MAFLD, metabolic dysfunction‐associated fatty liver disease; NAFLD, nonalcoholic fatty liver disease
Adjusted and unadjusted CPHR models when groups were divided according to the presence of AF are presented in Figure 4. Patients with NAFLD (adjusted HR, 2.62; 95% CI, 1.1–6.21; p = 0.02), MAFLD (adjusted HR, 3.09; 95% CI, 1.36–7.02; p = 0.007), and MAFLD + AF (adjusted HR, 3.81; 95% CI, 1.65–8.81; p = 0.002) were associated with an increased risk of intubation compared to patients in the control group. Patients with NAFLD + AF (adjusted HR, 2.44; 95% CI, 1.21–4.9; p = 0.01) and MAFLD + AF (adjusted HR, 2.25; 95% CI, 1.23–4.12; p = 0.008) were associated with a higher risk of death.
FIGURE 4.
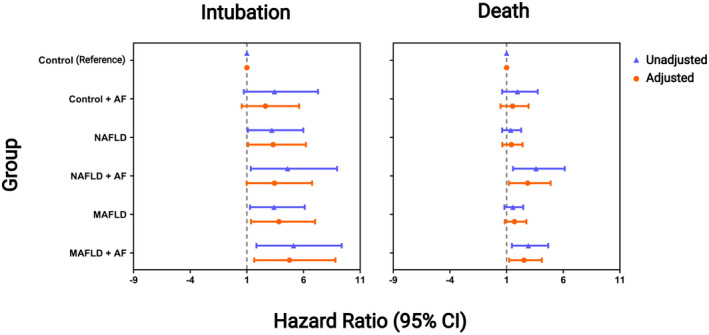
Forest plot with the unadjusted and adjusted hazard ratios for MAFLD and NAFLD relative to the control group when groups were divided according to the presence of advanced fibrosis. Adjusted models include age, sex, and the comorbidities hypertension, type 2 diabetes, overweight, and obesity. Age was considered a continuous variable. The model for the death outcome had a global likelihood chi‐square test with p < 0.001 and global Schoenfeld residual tests chi‐square with p = 0.1. The model for the intubation outcome had a global likelihood chi‐square test with p = 0.007 and global Schoenfeld residual tests chi‐square with p = 0.8. AF, advanced fibrosis; CI, confidence interval; MAFLD, metabolic dysfunction‐associated fatty liver disease; NAFLD, nonalcoholic fatty liver disease
DISCUSSION
In the case of patients hospitalized with COVID‐19, our results indicate that the death rate was higher in the MAFLD group but not the NAFLD group compared to that in the control group. The intubation rates were higher in the NAFLD and MAFLD group compared to the rate in the control group. In addition, we found a statistically significant interaction of NAFLD and MAFLD with AF that increased the risk of mortality and intubation of the patients. Age was a determinant variable for mortality and intubation in patients hospitalized with COVID‐19.
There are few comparable studies in methodology and the studied population. However, our results agree with the meta‐analysis performed by Hegyi and colleagues[ 19 ] that found patients with NAFLD were associated with a more severe course of COVID‐19 disease (intensive care unit [ICU] admission and clinical severity); however, they were unable to evaluate mortality as an outcome. A meta‐analysis by Singh and colleagues[ 20 ] showed that patients with NAFLD had an increased risk for severe COVID‐19 infection (by clinical criteria) and ICU admission with no difference in mortality compared to the non‐NAFLD group.
In the case of patients with MAFLD, the meta‐analysis by Sharma and Kumar[ 21 ] found that MAFLD was associated with a 4‐fold to 6‐fold increase in COVID‐19 severity compared to patients without MAFLD. Likewise, it was found in the same study that patients with MAFLD did not have an increased risk of dying from COVID‐19. The meta‐analysis by Tao and colleagues[ 22 ] also found that the risk of severe COVID‐19 increased with MAFLD and that there was no association of MAFLD with death from COVID‐19. However, in both meta‐analyses, there was heterogeneity in the criteria for diagnosing MAFLD among the different studies, and multivariate models were not considered.
Our results agree with the international literature suggesting that both NAFLD and MAFLD may increase the risk of severity in COVID‐19. The association of MAFLD and NAFLD with severe COVID‐19 could be related to the low proinflammatory state that has been associated with metabolic dysregulation. Severe COVID‐19 has been associated with a proinflammatory state called a CS. A CS is composed of the activation of inflammasomes and excessive release of interleukin (IL)‐6 and IL‐1B into the bloodstream by macrophages; this could lead to organ damage or even circulatory collapse.[ 23 ]
The liver has the largest macrophage (Kupffer cells) reservoir in the body, and it is known that Kupffer cells shift to proinflammatory M1 polarization in response to weight gain and insulin resistance, which are two important factors in NAFLD and MAFLD, favoring a chronic low proinflammatory state.[ 9 , 24 ] Consequently, the proinflammatory state in MAFLD or NAFLD could overlap with the acute proinflammatory state caused by COVID‐19 infection, resulting in immune system dysregulation with excessive release of proinflammatory mediators (i.e., CS).
However, in this study, we found that patients with MAFLD, in addition to having a higher risk of severity, have a statistically significantly higher risk of dying from COVID‐19. Among the main causes of this situation are the high heterogeneity of criteria for diagnosing MAFLD among the studies; in the present study, we tried to adhere as closely as possible to the criteria of the international group of experts. The MAFLD definition has more importance in metabolic risk and is therefore superior in detecting people at increased risk of dying from COVID‐19. For instance, it has been estimated that patients with MAFLD have a higher risk of atherosclerotic cardiovascular disease compared to those with NAFLD.[ 25 ] In addition, patients with MAFLD have been found to have a higher prevalence of colorectal adenomas.[ 26 ]
This is the first study that reports a significant interaction of NAFLD and MAFLD with AF in the deaths and intubations of patients hospitalized with COVID‐19. However, this finding agrees with Calapod and colleagues[ 27 ] who showed that patients with NAFLD and a FIB‐4 index >3.25 had longer hospital stays, more admissions to the ICU, and higher death rates; and with Targher and colleagues[ 28 ] who found that patients with MAFLD with significant AF had a higher likelihood of having severe COVID‐19 disease. In addition, Campos et al.[ 29 ] reported that patients with MAFLD and liver fibrosis had associations with intubation, mortality, and acute kidney injury. Our results contradict the study of Lopez‐Mendez et al.[ 30 ] who reported no association between AF and NAFLD with mortality. However, their cut‐off point for AF was different from the international consensus (FIB‐4 index >2.67), and their method was a logistic regression that was not adjusted for age and sex, increasing the possibility of bias.
Liver fibrosis is a process initiated by the activation of stellate cells, followed by release of profibrotic and proinflammatory mediators. The key in the activation of stellate cells in MAFLD or NAFLD is the perpetuation of the proinflammatory stimuli by the Kupffer cells.[ 31 ] Despite having a profibrotic effect, stellate cells can also promote the production of adhesion proteins and chemokines, which result in the recruitment of inflammatory cells and the amplification of proinflammatory stimuli.[ 32 ] Presumably, the activation of stellate cells could synergize with the M1 polarization of macrophages in the acute inflammatory state caused by COVID‐19, leading to a more severe presentation of the disease with higher mortality. Our results may indicate that both NAFLD and MAFLD, when significant fibrosis is present, are at a statistically significant increase of death during hospitalization for COVID‐19.
Our study has a number of strengths. First is that MAFLD was diagnosed according to international expert consensus and not only as the presence of liver steatosis, and to the best of our knowledge, few papers have correctly classified MAFLD. Second, we were able to adjust the risk to the main determinants in mortality reported for COVID‐19 in a Mexican population. Third, the sample size exploring MAFLD was one of the largest reported in Mexico. Fourth, the patients analyzed were admitted in the first months of the pandemic and therefore did not receive drugs with evidence for reducing mortality, limiting their management to supportive measures.
Our study also had a number of limitations. First, the limitations of this study are those inherent to retrospective studies. Second, our study population consisted only of a Mexican population treated in Mexico, so the results cannot be generalized to other populations. Third, we have insufficient data to calculate the risk of severity and ICU admission. Fourth, the data correspond to the first months of the pandemic when treatments were less standardized, including the criteria for mechanical ventilation. Fifth, there were limitations of use of the FIB‐4 score to determine fibrosis and the inability to stage fibrosis rather than reliance on a single cutoff. Sixth, patients with decompensated cirrhosis were excluded.
In conclusion, in the case of patients hospitalized with COVID‐19, our results indicate that the death rate was higher in the MAFLD group but not the NAFLD group compared to that in the control group. The intubation rates were higher in the NAFLD and MAFLD groups compared to rates in the control group, suggesting that both could be associated with COVID‐19 severity. In addition, AF may additively affect the prognosis of patients with NAFLD and MAFLD. Further studies should focus on finding the pathophysiological mechanisms for the interactions found between AF with MAFLD and NAFLD.
CONFLICT OF INTEREST
All authors declare no conflict of interests.
ACKNOWLEDGMENTS
Vázquez‐Medina is indebted to “Dirección General de Calidad y Educación en Salud” for fellowship that enabled him to pursue social service of the Bachelor of Médico Cirujano y Partero.
Vázquez‐Medina MU, Cerda‐Reyes E, Galeana‐Pavón A, López‐Luna CE, Ramírez‐Portillo PM, Ibañez‐Cervantes G, et al. Interaction of metabolic dysfunction‐associated fatty liver disease and nonalcoholic fatty liver disease with advanced fibrosis in the death and intubation of patients hospitalized with coronavirus disease 2019. Hepatol Commun. 2022;6:2000–2010. 10.1002/hep4.1957
REFERENCES
- 1. Marjot T, Webb GJ, Barritt AS, Moon AM, Stamataki Z, Wong VW, et al. COVID‐19 and liver disease: mechanistic and clinical perspectives. Nat Rev Gastroenterol Hepatol. 2021;18:348–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Boregowda U, Aloysius MM, Perisetti A, Gajendran M, Bansal P, Goyal H. Serum activity of liver enzymes is associated with higher mortality in COVID‐19: a systematic review and meta‐analysis. Front Med (Lausanne). 2020;7:431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Iavarone M, D'Ambrosio R, Soria A, Triolo M, Pugliese N, Del Poggio P, et al. High rates of 30‐day mortality in patients with cirrhosis and COVID‐19. J Hepatol. 2020;73:1063–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Pan L, Huang P, Xie X, Xu J, Guo D, Jiang Y. Metabolic associated fatty liver disease increases the severity of COVID‐19: a meta‐analysis. Dig Liver Dis. 2021;53:153–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Zhou Y‐J, Zheng KI, Wang X‐B, Sun Q‐F, Pan K‐H, Wang T‐Y, et al. Metabolic‐associated fatty liver disease is associated with severity of COVID‐19. Liver Int. 2020;40:2160–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Bernal‐Reyes R, Castro‐Narro G, Malé‐Velázquez R, Carmona‐Sánchez R, González‐Huezo MS, García‐Juárez I, et al. The Mexican consensus on nonalcoholic fatty liver disease. Rev Gastroenterol Mex (Engl Ed). 2019;84:69–99. [DOI] [PubMed] [Google Scholar]
- 7. Eslam M, Sanyal AJ, George J; International Consensus Panel . MAFLD: a consensus‐driven proposed nomenclature for metabolic associated fatty liver disease. Gastroenterology. 2020;158:1999–2014.e1. [DOI] [PubMed] [Google Scholar]
- 8. Portincasa P, Krawczyk M, Smyk W, Lammert F, Di Ciaula A. COVID‐19 and non‐alcoholic fatty liver disease: two intersecting pandemics. Eur J Clin Invest. 2020;50:e13338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Vasques‐Monteiro IML, Souza‐Mello V. Coronavirus disease 2019 severity in obesity: metabolic dysfunction‐associated fatty liver disease in the spotlight. World J Gastroenterol. 2021;27:1738–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Lomonaco R, Godinez Leiva E, Bril F, Shrestha S, Mansour L, Budd J, et al. Advanced liver fibrosis is common in patients with type 2 diabetes followed in the outpatient setting: the need for systematic screening. Diabetes Care. 2021;44:399–406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Yamamura S, Kawaguchi T, Nakano D, Tomiyasu Y, Yoshinaga S, Doi Y, et al. Profiles of advanced hepatic fibrosis evaluated by FIB‐4 index and shear wave elastography in health checkup examinees. Hepatol Res. 2020;50:199‐213. [DOI] [PubMed] [Google Scholar]
- 12. Ginès P, Graupera I, Lammert F, Angeli P, Caballeria L, Krag A, et al. Screening for liver fibrosis in the general population: a call for action. Lancet Gastroenterol Hepatol. 2016;1:256–60. [DOI] [PubMed] [Google Scholar]
- 13. Sterling RK, Oakes T, Gal TS, Stevens MP, deWit M, Sanyal AJ. The fibrosis‐4 index is associated with need for mechanical ventilation and 30‐day mortality in patients admitted with corona virus disease 2019. J Infect Dis. 2020;222:1794–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Park JG, Kang MK, Lee YR, Song JE, Kim NY, Kweon YO, et al.; Daegu‐Gyeongbuk Liver Study Group . Fibrosis‐4 index as a predictor for mortality in hospitalised patients with COVID‐19: a retrospective multicentre cohort study. BMJ Open. 2020;10:e041989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Feng G, He NA, Zhou Y‐F, Li X‐P, Niu C, Liu M‐L, et al. A simpler diagnostic formula for screening nonalcoholic fatty liver disease. Clin Biochem. 2019;64:18–23. [DOI] [PubMed] [Google Scholar]
- 16. Ibáñez‐Samaniego L, Bighelli F, Usón C, Caravaca C, Fernández Carrillo C, Romero M, et al. Elevation of liver fibrosis index FIB‐4 is associated with poor clinical outcomes in patients with COVID‐19. J Infect Dis. 2020;222:726–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. TDe la Torre GM, Montelongo‐Mercado EA, Noyola‐Villalobos HF, García‐Ruiz A, Hernández‐Díaz S, Santiago‐Torres M, et al. Epidemiology of hospitalized patients with COVID‐19 in a tertiary care hospital. Gac Med Mex. 2021;157:237–44. [DOI] [PubMed] [Google Scholar]
- 18. Najera H, Ortega‐Avila AG. Health and institutional risk factors of COVID‐19 mortality in Mexico, 2020. Am J Prev Med. 2021;60:471–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Hegyi PJ, Váncsa S, Ocskay K, Dembrovszky F, Kiss S, Farkas N, et al. Metabolic associated fatty liver disease is associated with an increased risk of severe COVID‐19: a systematic review with meta‐analysis. Front Med (Lausanne). 2021;8:626425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Singh A, Hussain S, Antony B. Non‐alcoholic fatty liver disease and clinical outcomes in patients with COVID‐19: a comprehensive systematic review and meta‐analysis. Diabetes Metab Syndr. 2021;15:813–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Sharma P, Kumar A. Metabolic dysfunction associated fatty liver disease increases risk of severe Covid‐19. Diabetes Metab Syndr. 2020;14:825–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Tao Z, Li Y, Cheng B, Zhou T, Gao Y. Risk of severe COVID‐19 increased by metabolic dysfunction‐associated fatty liver disease. J Clin Gastroenterol. 2021;55:830–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Ragab D, Salah Eldin H, Taeimah M, Khattab R, Salem R. The COVID‐19 cytokine storm; what we know so far. Front Immunol. 2020;11:1446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Luo W, Xu Q, Wang Q, Wu H, Hua J. Effect of modulation of PPAR‐γ activity on Kupffer cells M1/M2 polarization in the development of non‐alcoholic fatty liver disease. Sci Rep. 2017;7:44612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Tsutsumi T, Eslam M, Kawaguchi T, Yamamura S, Kawaguchi A, Nakano D, et al. MAFLD better predicts the progression of atherosclerotic cardiovascular risk than NAFLD: generalized estimating equation approach. Hepatol Res. 2021;51:1115–28. [DOI] [PubMed] [Google Scholar]
- 26. Fukunaga S, Nakano D, Kawaguchi T, Eslam M, Ouchi A, Nagata T, et al. Non‐obese MAFLD is associated with colorectal adenoma in health check examinees: a multicenter retrospective study. Int J Mol Sci. 2021;22:5462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Calapod OP, Marin AM, Onisai M, Tribus LC, Pop CS, Fierbinteanu‐Braticevici C. The impact of increased Fib‐4 score in patients with type II diabetes mellitus on Covid‐19 disease prognosis. Medicina (Kaunas). 2021;57:434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Targher G, Mantovani A, Byrne CD, Wang X‐B, Yan H‐D, Sun Q‐F, et al. Risk of severe illness from COVID‐19 in patients with metabolic dysfunction‐associated fatty liver disease and increased fibrosis scores. Gut. 2020;69:1545–7. [DOI] [PubMed] [Google Scholar]
- 29. Campos‐Murguía A, Román‐Calleja BM, Toledo‐Coronado IV, González‐Regueiro JA, Solís‐Ortega AA, Kúsulas‐Delint D, et al. Liver fibrosis in patients with metabolic associated fatty liver disease is a risk factor for adverse outcomes in COVID‐19. Dig Liver Dis. 2021;53:525–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lopez‐Mendez I, Aquino‐Matus J, Gall S‐B, Prieto‐Nava JD, Juarez‐Hernandez E, Uribe M, et al. Association of liver steatosis and fibrosis with clinical outcomes in patients with SARS‐CoV‐2 infection (COVID‐19). Ann Hepatol. 2021;20:100271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Friedman SL. Molecular regulation of hepatic fibrosis, an integrated cellular response to tissue injury. J Biol Chem. 2000;275:2247–50. [DOI] [PubMed] [Google Scholar]
- 32. Sherman MH. Stellate cells in tissue repair, inflammation, and cancer. Annu Rev Cell Dev Biol. 2018;34:333–55. [DOI] [PubMed] [Google Scholar]


