Abstract
Methods of validation of formaldehyde decontamination of biological safety cabinets were compared. Decontamination of metal strips inoculated with Mycobacterium bovis, poliovirus, or Bacillus spp. spores was compared with the results obtained with three biological indicators. Conditions for successful decontamination, particularly relative humidity, were defined. The Attest 1291 biological indicator was the only biological indicator which was an aid in the detection of gross decontamination failure.
The decontamination and sterilization of heat- and moisture-sensitive items and the validation of this process have been long-standing problems. While formaldehyde gas has been used for over 100 years, the efficacy of this process remains controversial (11). The factors contributing to the effectiveness of decontamination by formaldehyde range from humidity and temperature levels to the medium to be decontaminated (1, 10).
Biological safety cabinets require decontamination before maintenance and repairs are performed, and the common practice of using spore strips and culture as validation is labor-intensive and time-consuming. To compare several indicator systems used within our institution, challenge organisms which exhibited reduced susceptibility to formaldehyde inactivation were tested. These included poliovirus, Mycobacterium bovis bacillus Calmette-Guérin (BCG) (2, 9), and also Bacillus spp. spores. Stainless steel strips were chosen as a carrier material to replicate the cabinet interior surface.
The commercially available biological indicator (BI) Attest 1262 and Attest 1264 systems contain a strip of paper inoculated with a specified number of Bacillus spp. spores and a pH-sensitive medium. The Attest Rapid Readout BI, Attest 1291, is incubated for 60 min, in contrast to the 24-h incubation required for other routinely used BIs; this is achieved by the detection of the enzymatic breakdown of a nonfluorescent substrate to a fluorescent product. These systems have been designed for the validation of steam or ethylene oxide sterilization.
Bacillus stearothermophilus ATCC 7953 and Bacillus subtilis subsp. niger ATCC 9372 bacteria were retrieved from BIs (B. stearothermophilus was retrieved from 3M Attest 1262 [lot 07.95.282], and B. subtilis was retrieved from 3M Attest 1264 [lot Sep 95 054]) and subcultured onto nutrient agar slopes (Medvet) and incubated at 56°C and 37°C, respectively.
The spores were harvested (12) and centrifuged with saline washes at 2,000 × g for 10 min, resuspended in sterile deionized water, stored at 4°C for 3 days (to lyse any remaining viable organisms) (7), and then stored in saline at 4°C. Test pieces were inoculated with 1.76 × 106 spores of B. subtilis and 4 × 105 spores of B. stearothermophilus (50-μl volumes) (numbers determined by viable counts). After treatment, the Bacillus test pieces were placed onto Columbia horse blood agar (Medvet) plates, and colonies in the imprint were counted after incubation. The strips were then placed into 5 ml of tryptone soy broth (Medvet) to confirm inactivation. Individual colonies on the plates were counted 24 and 48 h after inoculation. An additional test piece was included for the Bacillus spores, and the spores were enumerated by extraction in saline and viable counts of colonies on horse blood agar plates.
BCG (vaccine strain; CSL catalogue no. 0439.1301D [lot no. 0.439-50701]) was reconstituted and aseptically inoculated into Dubos medium (Medvet). Test pieces were inoculated with 1.39 × 106 organisms (50-μl volume at McFarland opacity standard 0.5) as determined by viable counts. Following treatment, the BCG-carrying test pieces were aseptically transferred to vials containing 1 ml of saline with 0.05% Tween 80 (Sigma) and vortexed to resuspend the organisms. Culture was performed on Middlebrook medium (Medvet) with incubation at 37°C in a 5% CO2–95% air atmosphere. Final colony counts were performed after 4 weeks of incubation.
Poliovirus type 1 Sabin strain (Picornoviridae) was obtained from the Virus Detection Laboratory, Institute of Medical and Veterinary Science (Adelaide, Australia). A 50% tissue culture infective dose (TCID50) (6) test with confluent Buffalo green monkey (BGM) cell monolayers was performed by using serial dilutions made in Dulbecco’s modified Eagle’s medium as the maintenance medium. The cytopathic effects due to the virus were recorded. The TCID50 titer of virus (TCID50/0.2 ml) was 1012.5. Test pieces were inoculated with 100 μl of this suspension. Following treatment, the poliovirus test pieces were vortexed in 1 ml of maintenance medium, and the supernatant was inoculated into culture for a TCID50 test and read daily for 7 days for cytopathic effects.
Self-contained BIs from the 3M company (St. Paul, Minn.) containing B. stearothermophilus (Attest 1262, 7 × 105 spores, and Attest 1291, lot Nov 95 581, 1.8 × 106 spores) and B. subtilis (Attest 1264, 3.6 × 106 spores) were assessed in parallel with the biological challenge materials. They were activated upon retrieval and incubated according to the manufacturer’s directions. Cultures were checked daily. The Attest 1291 BI was read by fluorometric detection hourly for 7 h (8a), with continued incubation to check for growth.
Hot-air-sterilized stainless steel pieces (316-gauge satin finish; 2.0 cm by 1.6 cm by 0.09 cm) were the vehicle for the biological challenge materials. These strips were aseptically inoculated with the challenge organisms and air dried in a biological safety cabinet for 1 h. The dried test pieces were loosely enclosed in gamma-irradiated green kraft paper pulp by aseptic folding and sealing procedures.
The test materials were placed in a Gelman biological safety cabinet, class II (model BSC 1200), which was then decontaminated by using various concentrations of formaldehyde, various temperatures, and various relative humidities. A minimum of three determinations were made for each of the 12 sets of conditions studied.
A measured weight of paraformaldehyde powder (BDH catalog no. 294474L), mixed with a measured volume of deionized water to facilitate the alterations in relative humidity, was poured into a foil tray and placed onto a heating platform connected to an electrical timer. Heating to 160°C resulted in the thermal depolymerization of the paraformaldehyde powder to liberate formaldehyde gas within the sealed cabinet. Temperature was varied by use of the heating platform and electrical timers during decontamination.
The test pieces were positioned in four locations: under the tray within the cabinet, in the center back of the cabinet on the tray, within the cabinet on the side wall just below the filters, and on the outside of the cabinet above the metal grid beyond the exhaust filters. Attest BIs were also placed at these locations (held in place where necessary with Blu-Tac). The manufacturer of the Attest 1291 BIs, 3M, kindly made available the incubator (Attest 191; catalog no. 70-2005-9191-8) required for a period of this study.
The cabinet face and exhaust area were sealed with plastic sheeting and masking tape before decontamination was commenced. The cabinet fans were turned on for 5 min to circulate the formaldehyde gas after depolymerization (30 min after commencement of heating). After 15 h of decontamination, a second heating platform with a timer was activated and ammonia was liberated from ammonium carbonate powder (BDH catalog no. 100153W) to neutralize the formaldehyde vapors. A digital Amarell maximum and minimum temperature thermometer probe (Kreuzwertheim, Federal Republic of Germany) was positioned within the cabinet to measure the range of temperatures during decontamination. Relative humidity was measured with a Vaisola hygrometer (Helsinki, Finland).
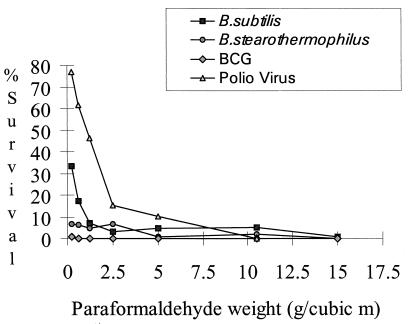
In experiments undertaken with B. subtilis spores as an indicator, a successful decontamination, reflected by a total kill, was observed for seven pieces (of a total of 144 pieces). The decontamination conditions which were optimal for all of the biological challenge materials were 66% relative humidity, a minimum temperature of 28°C, and 10.5 g of paraformaldehyde/m3 (Fig. 1). Formaldehyde was more efficacious in killing the spores of B. stearothermophilus than those of B. subtilis (Fig. 1). This organism showed a poor survival rate, exhibiting up to a 102 reduction in viability following drying and 15-h incubation. The mean survival for untreated controls was 3.7 × 103, with a standard deviation of 3.1 × 103. Decontamination was observed in a total of 16 instances, and when test results were expressed as a percentage of the untreated control value, the data showed that B. stearothermophilus was killed more readily than B. subtilis. When BCG was used as an indicator, decontamination at low levels of formaldehyde was observed. No recovery of viable organisms, plated at a 1:10 dilution, was evident at concentrations of 2.5 g of paraformaldehyde/m3 and above, regardless of temperature or relative humidity (Fig. 1). Poliovirus was not recovered when the optimal decontamination conditions were used. A cytotoxic effect for a control not containing virus was observed at 15 g of paraformaldehyde/m3, and for this reason data in relation to virus inactivation at this concentration were not validated and were not included in the analysis. In all other cases, no toxic effect was observed for these controls. Poliovirus was a useful challenge material for evaluation of decontamination but was inactivated at formaldehyde levels insufficient for decontamination of bacterial spores.
FIG. 1.
Percent survival of test organisms after decontamination with various concentrations of formaldehyde. Survival was measured as a percentage of the growth (CFU or TCID50) of that for the untreated control for each test organism. The data were for test pieces placed inside the cabinet on the side wall. (The relative humidity was <58%, and the temperature was <27°C.)
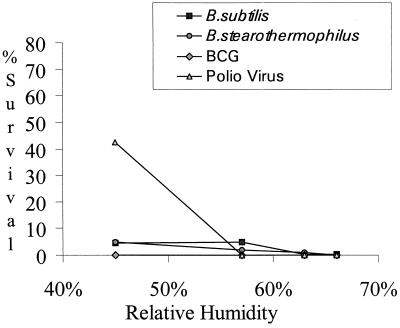
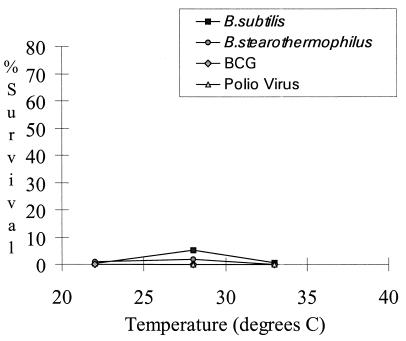
Increasing the relative humidity within the cabinet during decontamination resulted in increased inactivation of the challenge organisms (Fig. 2). For BCG, relative humidity was not a significant parameter, as this organism was killed at very low levels of formaldehyde. While the effectiveness of formaldehyde decontamination increased with the elevation of relative humidity (4, 10), condensation of water was evident within the cabinet when decontamination conditions exceeded 63% relative humidity. As formaldehyde has an affinity for water (5), excessive elevation of relative humidity results in a lowering of the levels of formaldehyde gas available for decontamination. The survival of bacterial spores varied marginally over the temperature range examined, but poliovirus and BCG were both inactivated at the levels of formaldehyde tested (Fig. 3).
FIG. 2.
Percent survival of test organisms after decontamination at various relative humidities. Paraformaldehyde weights were ≥5 g/m3, and the temperature was approximately 25°C.
FIG. 3.
Percent survival of test organisms with variation of temperature during decontamination with paraformaldehyde at ≥10.5 g/m3 and a relative humidity of approximately 58%.
Statistical analysis of the results for each challenge organism by means of the Kruskal-Wallis nonparametric test (5% significance level) indicated that the population medians were affected by the variations of formaldehyde level, relative humidity, and temperature.
Data for BIs and test pieces (Table 1) located beyond the exhaust filters indicated that this location was the most difficult to decontaminate. While pathogens are removed by the filters, these filters must be replaced at the end of their useful lives by maintenance staff and the decontamination procedure should ensure that these are safe to handle. Placing a strip beyond the filters was intended to provide a measure of formaldehyde penetration of the filter area. While the results obtained for the indicators at 1 h were less sensitive than those for the test pieces, they offer a means of rapid detection of gross decontamination failure.
TABLE 1.
Survival of test organisms on strips positioned at different test locations during decontaminationa
| Location of strip | B. subtilis (CFU) | B. stearothermophilus (CFU) | BCG (CFU) | Poliovirus (TCID50/0.2 ml) |
|---|---|---|---|---|
| Under the cabinet tray | 15,000 | 47.4 | <10 | 0 |
| Cabinet tray | 20,000 | 46 | <10 | 0 |
| Cabinet side | 21,000 | 73.8 | <10 | 0 |
| Beyond the exhaust filters | 19,000 | 293.2 | <10 | 100.66 |
Data are averages of three determinations. Decontamination conditions were as follows: paraformaldehyde, 10.6 g/m3; relative humidity, 57%; and temperature, 28°C.
As formaldehyde has a poor penetration capacity (4), the effect of the kraft paper pulp enclosures was tested. Test pieces inoculated with the Bacillus sp. spores, resting in an open petri dish within the cabinet, showed that the use of the paper did not affect the results obtained (data not shown). Challenge organisms inoculated onto paper are more readily killed than those on nonabsorbent surfaces (3). However, given the additional challenge offered due to the design of the self-contained BIs in the steam sterilization process (8), it was considered that this may also be true in regard to formaldehyde decontamination. The spores in the self-contained BIs Attest 1262 and Attest 1264 were killed at low levels of formaldehyde (Table 2). The results for Attest 1291 Rapid Readout BIs correlated more closely with decontamination documented by inactivation of biological challenge materials (Table 2).
TABLE 2.
Biological indicator results
| Decontamination conditions: relative humidity (%)/temp (°C)/para-formaldehyde (g/m3) | No. positive/total no.
|
||
|---|---|---|---|
| Attest 1262 for B. stearo-thermophilus at 24 h | Attest 1264 for B. subtilis at 24 h | Attest 1291 for B. stearo-thermophilus at 1 h | |
| 45/22/0.25 | 12/12 | 12/12 | 4/4 |
| 50/22/0.65 | 8/12 (12/12 at 48 h) | 9/12 | 4/4 |
| 52/22/1.25 | 6/12 (12/12 at 48 h) | 3/12 | 4/4 |
| 50/23/2.5 | 0/12 | 0/12 | Not tested |
| 45/24/5.0 | 0/12 | 0/12 | 2/8 (located beyond the exhaust filter) |
| 50/23/5.0 | 0/12 | 0/12 | 0/4 (2/4 at 3 h) |
| 57/28/10.5 | 0/12 | 0/12 | 0/8 (4/8 at 3 h) |
| 66/28/10.5 | 0/12 | 0/12 | 0/8 (2/8 at 2 h; 8/8 at 3 h) |
The difficulties in obtaining effective decontamination by using formaldehyde gas demonstrated in this study are consistent with earlier reports (3, 5). Bacterial spores inoculated onto stainless steel were the most resistant to decontamination. However, this method of validating decontamination is not feasible when rapid verification of decontamination is required. While the Attest 1291 BI result did not parallel the inactivation of all the challenge organisms, in cabinets where the presence of sporulating pathogens is unlikely it offers an aid in detecting incomplete decontamination.
ACKNOWLEDGMENTS
Special thanks for advice, discussion, and encouragement are given to P. Lee and the staff of the Media Production Unit, R. Lumb of the Tuberculosis Laboratory, B. Winter of the Antibiotic/Infection Control Laboratory, and M. Rapaic and D. Devonshire-Gill of the Virus Detection Laboratory, Infectious Diseases Laboratories, and to the staff of the Medtech Engineering-Metrology Laboratory, Institute of Medical and Veterinary Science.
REFERENCES
- 1.Ackland N R, Hinton M R, Denmeade K R. Controlled formaldehyde fumigation system. Appl Environ Microbiol. 1980;39:480–487. doi: 10.1128/aem.39.3.480-487.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Duguid J P, Marmion B P, Swain R H A, editors. Mackie and McCartney medical microbiology. 13th ed. Edinburgh, Scotland: Churchill Livingstone Publishers; 1978. p. 460. [Google Scholar]
- 3.Everall P H, Morris C A, Oliver P R, Becker J F. Problems in the disinfection of class 1 microbiological safety cabinets. J Clin Pathol. 1982;35:698–705. doi: 10.1136/jcp.35.7.698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hoffman R K, Spiner D R. Effect of relative humidity on the penetrability and sporicidal activity of formaldehyde. Appl Microbiol. 1970;20:616–619. doi: 10.1128/am.20.4.616-619.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lach V H. A study of conventional formaldehyde fumigation methods. J Appl Bacteriol. 1990;68:471–477. doi: 10.1111/j.1365-2672.1990.tb02898.x. [DOI] [PubMed] [Google Scholar]
- 6.Lennett L E, Schmidt N J. Diagnostic procedures for viral, rickettsial and chlamydial infections. 5th ed. Washington, D.C: American Public Health Association; 1979. p. 100. [Google Scholar]
- 7.Long S K, Williams O B. Method for removal of vegetative cells from bacterial spore preparations. J Bacteriol. 1958;76:332. doi: 10.1128/jb.76.3.332-332.1958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McCormick P. Biological indicators. Infect Control Hosp Epidemiol. 1988;9:504–507. doi: 10.1086/645761. [DOI] [PubMed] [Google Scholar]
- 8a.3M. Attest Rapid Readout biological indicators product profile. St. Paul, Minn: 3M; 1996. [Google Scholar]
- 9.Scott E M, Groman S P. In: Sterilisation, disinfection and preservation. 4th ed. Block S S, editor. Philadelphia, Pa: Lea and Febiger Publishers; 1991. p. 635. [Google Scholar]
- 10.Spiner D R, Hoffman R K. Effect of relative humidity on formaldehyde decontamination. Appl Microbiol. 1971;22:1138–1140. doi: 10.1128/am.22.6.1138-1140.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Taylor L A, Barbeito M S, Gremillion G G. Paraformaldehyde for surface sterilization and detoxification. Appl Microbiol. 1969;17:614–618. doi: 10.1128/am.17.4.614-618.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wright A M, Hoxey E V, Soper C J, Davies D J G. Biological indicators for low temperature steam and formaldehyde sterilization: the effect of defined media on sporulation, growth index and formaldehyde resistance of spores of Bacillus stearothermophilus strains. J Appl Bacteriol. 1995;79:432–438. doi: 10.1111/j.1365-2672.1995.tb03158.x. [DOI] [PubMed] [Google Scholar]