Abstract
Aims
Experimental studies suggest that increased bone marrow (BM) activity is involved in the association between cardiovascular risk factors and inflammation in atherosclerosis. However, human data to support this association are sparse. The purpose was to study the association between cardiovascular risk factors, BM activation, and subclinical atherosclerosis.
Methods and results
Whole body vascular 18F-fluorodeoxyglucose positron emission tomography/magnetic resonance imaging (18F-FDG PET/MRI) was performed in 745 apparently healthy individuals [median age 50.5 (46.8–53.6) years, 83.8% men] from the Progression of Early Subclinical Atherosclerosis (PESA) study. Bone marrow activation (defined as BM 18F-FDG uptake above the median maximal standardized uptake value) was assessed in the lumbar vertebrae (L3–L4). Systemic inflammation was indexed from circulating biomarkers. Early atherosclerosis was evaluated by arterial metabolic activity by 18F-FDG uptake in five vascular territories. Late atherosclerosis was evaluated by fully formed plaques on MRI. Subjects with BM activation were more frequently men (87.6 vs. 80.0%, P = 0.005) and more frequently had metabolic syndrome (MetS) (22.2 vs. 6.7%, P < 0.001). Bone marrow activation was significantly associated with all MetS components. Bone marrow activation was also associated with increased haematopoiesis—characterized by significantly elevated leucocyte (mainly neutrophil and monocytes) and erythrocyte counts—and with markers of systemic inflammation including high-sensitivity C-reactive protein, ferritin, fibrinogen, P-selectin, and vascular cell adhesion molecule-1. The associations between BM activation and MetS (and its components) and increased erythropoiesis were maintained in the subgroup of participants with no systemic inflammation. Bone marrow activation was significantly associated with high arterial metabolic activity (18F-FDG uptake). The co-occurrence of BM activation and arterial 18F-FDG uptake was associated with more advanced atherosclerosis (i.e. plaque presence and burden).
Conclusion
In apparently healthy individuals, BM 18F-FDG uptake is associated with MetS and its components, even in the absence of systemic inflammation, and with elevated counts of circulating leucocytes. Bone marrow activation is associated with early atherosclerosis, characterized by high arterial metabolic activity. Bone marrow activation appears to be an early phenomenon in atherosclerosis development.
[Progression of Early Subclinical Atherosclerosis (PESA); NCT01410318].
Keywords: Subclinical atherosclerosis, Metabolic syndrome, Bone marrow, PET/MRI
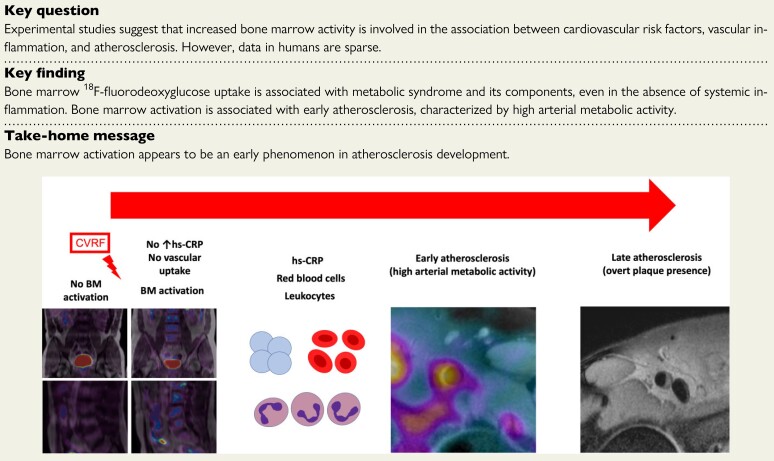
Structured Graphical Abstract
Structured Graphical Abstract.
The hypothesis of the natural history of the inflammatory process involving the atherosclerotic plaque formation. Bone marrow (BM) is implicated in the atherosclerotic process long before the appearance of acute cardiovascular events. Cardiovascular risk factors trigger BM activation, initially in the absence of systemic inflammation. As BM activation progresses, it is accompanied by an increase in haematopoietic progenitor cells and an associated increase in inflammatory markers. The next step in the process is arterial inflammation, leading to an increase in atherosclerotic burden.
See the editorial comment for this article ‘Mischief in the marrow: a root of cardiovascular evil’, by Peter Libby et al., https://doi.org/10.1093/eurheartj/ehac149.
Introduction
The association between inflammation and atherosclerosis is well established,1 and mechanistic studies have demonstrated that inflammation is an essential mediator of all stages of atherosclerosis, from initiation to progression and the development of thrombotic complications.2,3 Circulating immune cells play a critical role in the build-up of atherosclerotic plaques by adhering to activated endothelium and infiltrating the arterial wall to become lesional cells.4 This association has led to the study of various anti-inflammatory therapies in the last years, with encouraging results that justify the use of some of them such as low-dose colchicine in selected, high-risk patients.5
The bone marrow (BM) is the primary site of haematopoiesis, and the proliferation and migration of haematopoietic progenitors are regulated by various physiological and pathological stimuli.1,2 After an acute cardiovascular event, BM is activated by sympathetic signalling, triggering an increased haematopoiesis, and the release of progenitor cells that activate spleen production of monocytes, aggravating atherosclerosis progression.6–9 Experimental studies suggest that increased BM haematopoietic activity may be a central link between cardiometabolic risk factors and exacerbated inflammation in atherosclerosis. In mice, hypercholesterolaemia and low HDL-cholesterol levels associated with elevated haematopoietic activity with increased monocytosis and neutrophilia.10,11 Moreover, murine models of obesity present marked monocytosis and neutrophilia, associated with BM myeloid progenitor proliferation and expansion.12,13 Diabetes mellitus has also been associated with increased circulating neutrophils and monocytes, reflecting the expansion of BM myeloid progenitors.14,15 Hypertension, driven by an overactive sympathetic activation, deteriorates haematopoietic cell niche in the BM which can contribute to atherosclerosis.16 In humans, it has been suggested that chronic stress accelerates haematopoiesis, giving rise to higher levels of inflammatory cells that might contribute to the atherosclerotic process.17 In addition, haematopoietic stem cell division rates are increased in subjects with atherosclerosis,18 and it has been suggested that the haematopoietic system might be chronically affected in these subjects.19
Despite the extensive pre-clinical data, human data to support the association between BM haematopoietic activation and cardiovascular risk factors are sparse.
In some tissues, the high metabolic activity can be detected by imaging techniques such as 18F-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET/CT) imaging. This technique has been used to characterize BM activation after acute coronary syndrome,20,21 based on a higher 18F-FDG uptake related to an increased glucose consumption due to cellular proliferation, and vascular inflammation,20–22 which correlates with macrophage density as measured by histology and immunohistochemistry.23–25 Moreover, an association between amygdalar activity as evaluated by 18F-FDG PET/CT and cardiovascular disease events has been suggested to be mediated by increased BM activation.26 Hybrid 18F-FDG PET/magnetic resonance imaging (PET/MRI) is an accurate method for the detection of early atherosclerosis (characterized by high arterial metabolic activity) and for later stages of the disease (plaque detection), in asymptomatic middle-aged individuals.27 Here, we studied BM activation, detected by 18F-FDG PET/MRI in apparently healthy middle-aged individuals, and its association with cardiovascular risk factors and subclinical atherosclerosis.
Materials and methods
Study population
The study population consisted of participants in the PESA study [Progression of Early Subclinical Atherosclerosis—CNIC–Santander; (PESA) NCT01410318]28 who underwent whole body 18F-FDG PET/MRI.27 PESA is an observational prospective cohort study of 4184 asymptomatic employees at Santander Bank in Madrid. Participants were aged 40–54 years at enrolment (June 2010–February 2014). Exclusion criteria were previous cardiovascular disease, any condition reducing life expectancy or affecting study adherence, morbid obesity [body mass index (BMI) ≥ 40 kg/m2], or chronic kidney disease (estimated glomerular filtration rate <60 mL/min/1.73 m2). The main goal of the PESA study is to characterize atherosclerosis initiation and progression by means of serial multi-territory, multimodality non-invasive imaging,29 and paired biological sampling. A subgroup of PESA participants showing atherosclerosis on baseline vascular ultrasound, defined as being in the highest plaque tertile on vascular ultrasound and/or having any coronary artery calcification on CT, underwent baseline whole body 18F-FDG PET/MRI study to characterize arterial metabolic activity.27 Cardiovascular risk factors were assessed prospectively at enrolment using the 10-year risk algorithm based on Pooled Cohort Equations.30 Risk bands of <5, 5 to <7.5, and ≥7.5% were defined as low, intermediate, and high risk, respectively.31 A fasting blood test included blood count and biochemistry with the determination of systemic inflammation parameters.28 Blood count included leucocytes and their components (including neutrophils, lymphocytes, monocytes, eosinophils, and basophils), red blood cell count, haemoglobin, haematocrit, red blood cell width, and platelets. Leucocytosis was defined as a white blood cell count >10.5 × 103 cells/µL. Inflammation parameters included high-sensitivity C-reactive protein (hs-CRP), ferritin, erythrocyte sedimentation rate, fibrinogen, P-selectin, and vascular cell adhesion molecule-1 (VCAM-1). Insulin levels were also measured. Insulin resistance was measured by HOmeostatic Model Assessment for Insulin Resistance (HOMA-IR), calculated as [(fasting plasma glucose level × fasting insulin level)/405].32 In this study, metabolic syndrome (MetS) was defined when a participant met at least three of the following conditions: central obesity (waist circumference ≥88 cm in women and ≥102 cm in men);33 elevated plasma triglycerides (≥150 mg/dL); low plasma HDL-cholesterol (<40 mg/dL in men or <50 mg/dL in women); elevated fasting plasma glucose (≥100 mg/dL); and high blood pressure (systolic ≥130 mmHg and/or diastolic ≥85 mmHg).34 Dyslipidaemia was defined as total cholesterol ≥240 mg/dL, LDL-cholesterol ≥160 mg/dL, HDL-cholesterol <40 mg/dL, or use of lipid-lowering drugs.
The study protocol was approved by the institutional review board, and all participants provided written informed consent.
Hybrid positron emission tomography/magnetic resonance imaging acquisition protocol and image analysis
The vascular PET/MRI protocol has been published previously.27 In brief, the protocol included high-resolution black-blood MRI of the carotid, iliac, and femoral arteries and co-registered PET/MRI of the carotid arteries, thoracic aorta, infrarenal abdominal aorta, iliac, and femoral arteries. Ilio-femoral MRI attenuation maps and lower-body PET were acquired at the start of the protocol. Magnetic resonance imaging data were analysed with VP Diagnostics software version 2.1.0 (Seattle, WA, USA) and PET/MRI data were analysed with Philips Fusion Viewer version 2.0 (Philips Healthcare).
As previously described, attenuation correction used MRI attenuation maps with a three-class tissue (soft tissue, lung, and air) validated segmentation technique.35 A transverse 575 mm field of view was used, and images were generated with a voxel size of 4.0 mm × 4.0 mm × 4.0 mm. Specific templates for PET images were added to the attenuation maps to correct for attenuation effects of the scanner bed. For abdominal region, the coil has a minimal attenuation and was not included in the attenuation maps. Similarly, not corrected PET data were used to extend the attenuation maps beyond the magnetic resonance field of view limits and this information was integrated in attenuation correction maps for the final reconstruction.36
A total of six vascular territories per participant were analysed in MRI: left/right carotid arteries, left/right iliac arteries, and left/right femoral arteries. The presence, number, and plaque volumes were defined for each territory, and recorded as a surrogate of total plaque burden for each individual.
Lumbar vertebrae L3 and L4 were analysed in fused PET/MRI images.21 Quantitative 18F-FDG uptake was measured in multiple slices in the coronal axis by drawing 3D regions of interest encompassing the contour of each vertebra, excluding the cortical bone. The maximal standardized uptake value (SUVmax) was calculated in these regions of interest (calculated as decay-corrected tissue radioactivity divided by body weight and injected dose).37 For each participant, BM SUVmax was calculated as the mean SUVmax of the lumbar vertebrae (L3 and L4). Bone marrow activation was defined as BM SUVmax above the median value (1.9).
Statistical analysis
Normally distributed continuous variables are expressed as mean ± SD, whereas non-normally distributed variables are expressed as median (Q1–Q3). The distribution of continuous variables was assessed with graphical methods. Categorical variables are expressed as n (%). Differences between BM activation were assessed by Student’s t-test or Wilcoxon signed-rank test and χ 2 or Fisher exact test, for continuous and categorical variables, respectively, as appropriate. Linear trends across groups according to quintiles of 18F-FDG uptake were evaluated with an extension of the non-parametric Wilcoxon rank-sum test. For multivariate analysis, ordinal logistic regression models were performed. To evaluate the associations of BM activation in the presence of confounders, Model 1 (adjusting for age and sex) and Model 2 (adjusting for age, sex, glucose levels before PET/MRI, smoking, haemoglobin, and hs-CRP) were created. To evaluate the association between BM activation in the presence of vascular uptake associations and plaque volume (mm3) (0 and tertiles), several models were generated: Model 1 (qualitative), adjusting for age, sex, hypertension, dyslipidaemia, diabetes, smoking, family history of cardiovascular disease, and obesity; Model 2 (quantitative), adjusting for age, sex, systolic blood pressure, diastolic blood pressure, LDL-cholesterol, HDL-cholesterol, diabetes, smoking, dyslipidaemia treatment, family history of cardiovascular disease, and BMI; and Model 3, which is Model 1 but excluding dyslipidaemia treatment.
For all endpoints, differences were considered statistically significant at P-values < 0.05. Statistical analyses were performed using Stata software version 15 (StataCorp, College Station, TX, USA).
Results
A total of 946 PESA participants underwent whole body 18F-FDG PET/MRI at baseline. The mean 18F-FDG dose was 292.3 ± 11.1 MBq, and the radiation exposure was 5.6 ± 0.2 mSv. The mean start time after 18F-FDG injection was 106 ± 15 min for lower-body PET and 132.9 ± 19.9 min for upper-body PET. Reasons for non-completion were physical intolerance in upper-body studies (8 PETs and 51 MRIs), technical issues with MRI attenuation maps (97 initial PETs), and poor image quality (70 iliac MRIs). Complete 18F-FDG PET/MRI studies were available for 755 (79.8%) participants, and lumbar vertebrae BM images were of good quality for 745 participants (78.8% of the total sample who underwent PET/MRI); these participants constituted the population for the present study.
Baseline characteristics of subjects in relation to bone marrow activation
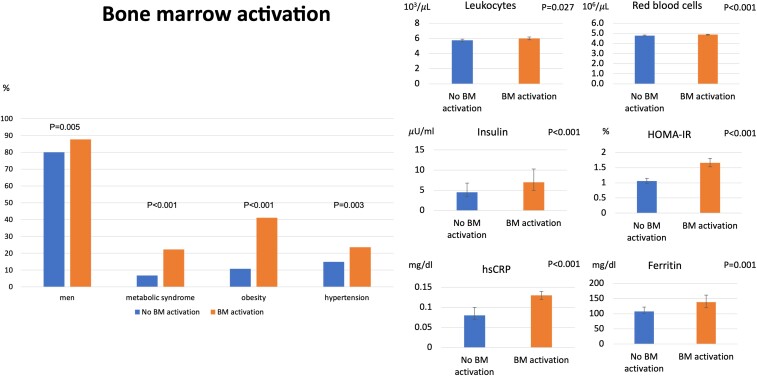
The median (Q1–Q3) age was 50.5 years (46.8–53.6) and 83.8% were men. Baseline characteristics are represented in Table 1. Participants with BM activation (Figure 1) were more frequently men (87.6 vs. 80% in those subjects without BM activation, P = 0.005) and more frequently had MetS (22.2 vs. 6.7%, P < 0.001). Bone marrow activation showed a significant association with central obesity (41.1 vs. 10.7%, P < 0.001), hypertension (23.5 vs. 14.9%, P = 0.003), higher plasma triglyceride levels (105.5 vs. 87 mg/dL, P < 0.001), lower HDL-cholesterol (44.5 vs. 48.4 mg/dL, P < 0.001), and higher fasting glucose (93 vs. 89 mg/dL, P < 0.001) (Figure 2). The BM activation and non-activation groups showed no differences in age, family history of cardiovascular disease, smoking, or total and LDL-cholesterol. Bone marrow activation group had higher levels of glycated haemoglobin (HbA1c, 5.5 vs. 5.4%, P = 0.007), and insulin resistance measured by HOMA-IR (1.7 vs. 1.1%, P < 0.001). Insulin levels were significantly increased in the group with BM activation (7 vs. 4.5 µU/mL, P < 0.001). Bone marrow activation was associated with significantly higher numbers of leucocytes (6.00 × 103 vs. 5.77 × 103 cells/µL in the group without BM activation, P = 0.027), especially neutrophils (3.4 × 103 vs. 3.2 × 103 cells/µL, P = 0.029) and red blood cell counts (4.9 × 106 vs. 4.8 × 106 cells/µL, P < 0.001). The BM activation group also showed significant elevation of the systemic inflammation markers, including hs-CRP (0.13 vs. 0.08 mg/dL, P < 0.001), ferritin (138.6 vs. 107.6 ng/dL, P = 0.001), fibrinogen (268.6 vs. 260.8 mg/dL, P = 0.03), P-selectin (139.7 vs. 129.2 ng/dL, P = 0.004), VCAM-1 (686.3 vs. 623.4 ng/mL, P = 0.025), and red blood cell distribution width (14.7 vs. 14.6%, P = 0.055). The main between-group differences in baseline characteristics are summarized in Figure 2.
Table 1.
Study population characteristics stratified by bone marrow activation (above or below-median 18F-fluorodeoxyglucose uptake)
| Total population | No bone marrow activation | Bone marrow activationa | P-value | |
|---|---|---|---|---|
| (n = 745) | (n = 375) | (n = 370) | ||
| Age, years | 50.5 (46.8–53.6) | 50.5 (47.0–53.8) | 50.5 (46.7–53.5) | 0.961 |
| Men | 624 (83.8) | 300 (80.0) | 324 (87.6) | 0.005 |
| Metabolic syndrome and components | ||||
| Metabolic syndrome | 107 (14.4) | 25 (6.7) | 82 (22.2) | <0.001 |
| Central obesity | 192 (25.8) | 40 (10.7) | 152 (41.1) | <0.001 |
| Triglycerides, mg/dL | 98 (72–131) | 87 (65–121) | 105 (77–140) | <0.001 |
| HDL-C, mg/dL | 46.5 ± 11.4 | 48.4 ± 11.6 | 44.5 ± 10.7 | <0.001 |
| Fasting glucose, mg/dL | 91 (85–97) | 89 (84–94) | 93 (87–99) | <0.001 |
| SBP, mmHg | 120.5 ± 12.3 | 118.7 ± 11.6 | 122.3 ± 12.6 | <0.001 |
| DBP, mmHg | 75.3 ± 9.1 | 73.7 ± 8.3 | 76.9 ± 9.5 | <0.001 |
| Other cardiovascular risk factors | ||||
| Family history of CV disease, n (%) | 154 (20.7) | 73 (19.5) | 81 (21.9) | 0.414 |
| Current smoking (%) | 197 (26.9) | 111 (30.1) | 86 (23.8) | 0.054 |
| Hypertension | 143 (19.2) | 56 (14.9) | 87 (23.5) | 0.003 |
| Dyslipidaemia | 440 (59.1) | 203 (54.1) | 237 (64.1) | 0.006 |
| Diabetes | 34 (4.6) | 13 (3.5) | 21 (5.7) | 0.149 |
| BMI, kg/m2 | 27.2 ± 3.5 | 25.5 ± 2.8 | 28.8 ± 3.2 | <0.001 |
| Weight, kg | 81.4 ± 13.3 | 75.5 ± 11.3 | 87.4 ± 12.4 | <0.001 |
| Waist circumference, cm | 93.8 ± 10.8 | 89.0 ± 9.6 | 98.7 ± 9.7 | <0.001 |
| Treatment | ||||
| Antihypertensive therapy | 100 (13.4) | 40 (10.7) | 60 (16.2) | 0.026 |
| Lipid-lowering therapy | 113 (15.2) | 50 (13.3) | 63 (17.0) | 0.160 |
| Antidiabetic therapy | 28 (3.8) | 11 (2.9) | 17 (4.6) | 0.233 |
| Biochemistry | ||||
| Total cholesterol, mg/dL | 208.3 ± 33.5 | 207.6 ± 32.3 | 209.0 ± 34.7 | 0.592 |
| LDL-C, mg/dL | 139.9 ± 30.1 | 139.1 ± 28.8 | 140.6 ± 31.4 | 0.488 |
| HbA1c, % | 5.5 (5.2–5.7) | 5.4 (5.2–5.7) | 5.5 (5.3–5.7) | 0.007 |
| HOMA-IR, % | 1.3 (0.9–2.1) | 1.1 (0.7–1.7) | 1.7 (1.0–2.5) | <0.001 |
| Insulin, µU/mL | 5.7 (3.9–8.2) | 4.5 (3.4–6.8) | 7.0 (5.0–10.3) | <0.001 |
| Inflammatory markers | ||||
| hs-CRP, mg/dL | 0.11 (0.06–0.19) | 0.08 (0.05–0.16) | 0.13 (0.07–0.23) | <0.001 |
| Ferritin, ng/mLb | 120.8 (63.0–204.0) | 107.6 (56.4–194.1) | 138.6 (74.8–214.1) | 0.001 |
| Erythrocyte sedimentation rate (1 h), mm | 5 (4–8) | 5 (4–7) | 6 (4–8) | 0.175 |
| Fibrinogen, mg/dL | 265.4 (236.5–295.1) | 260.8 (234.5–294.6) | 268.6 (239.6–299.2) | 0.030 |
| P-selectin, ng/mL | 134.7 (106.7–166.2) | 129.2 (104.5–160.6) | 139.7 (109.9–177.0) | 0.004 |
| Vascular cell adhesion molecule-1, ng/mL | 651.1 (519.6–820.0) | 623.4 (509.8–791.9) | 686.3 (540.6–849.0) | 0.025 |
| Blood count | ||||
| Leucocytes, 103 cells/µL | 5.87 (4.99–7.05) | 5.77 (4.94–6.95) | 6.00 (5.15–7.13) | 0.027 |
| Leucocytosis (>10.5 × 103 cells/µL) | 17 (2.3) | 8 (2.1) | 9 (2.4) | 0.785 |
| Red blood cell count, 106 cells/µL | 4.84 (4.59–5.08) | 4.78 (4.51–5.03) | 4.89 (4.64–5.12) | <0.001 |
| Red cell distribution width, % | 14.6 (14.0–15.2) | 14.6 (14.0–15.1) | 14.7 (14.0–15.2) | 0.055 |
| Haemoglobin, g/dL | 15.0 (14.3–15.7) | 14.9 (14.1–15.6) | 15.1 (14.4–15.8) | <0.001 |
| Haematocrit, % | 44.2 (41.9–46.3) | 43.9 (41.6–46.0) | 44.5 (42.5–46.5) | 0.002 |
| Platelet count, 103 cells/µL | 225 (198–257) | 226 (199–256) | 224 (194–258) | 0.686 |
| Segmented neutrophils, 103 cells/µL | 3.31 (2.73–4.23) | 3.21 (2.66–4.11) | 3.40 (2.78–4.36) | 0.029 |
| Lymphocytes, 103 cells/µL | 1.86 (1.56–2.22) | 1.84 (1.55–2.20) | 1.88 (1.57–2.22) | 0.465 |
| Monocytes, 103 cells/µL | 0.41 (0.34–0.52) | 0.41 (0.33–0.51) | 0.42 (0.34–0.52) | 0.201 |
| Eosinophils, 103 cells/µL | 0.13 (0.08–0.20) | 0.12 (0.08–0.20) | 0.13 (0.08–0.21) | 0.446 |
| Basophils, 103 cells/µL | 0.05 (0.03–0.07) | 0.05 (0.03–0.07) | 0.05 (0.03–0.06) | 0.689 |
| Neutrophil to lymphocyte ratio | 1.79 (1.43–2.30) | 1.75 (1.41–2.24) | 1.83 (1.46–2.36) | 0.178 |
| SCORE risk score | ||||
| Low (<1%) | 459 (64.2) | 245 (67.1) | 214 (61.1) | 0.095 |
| Intermediate (1–5%) | 254 (35.5) | 120 (32.9) | 134 (38.3) | 0.131 |
| High (>5%) | 2 (0.3) | 0 (0.0) | 2 (0.6) | 0.148 |
| ASCVD risk score | ||||
| Low | 369 (61.7) | 207 (66.1) | 162 (56.8) | 0.020 |
| Intermediate | 109 (18.2) | 50 (16.0) | 59 (20.7) | 0.135 |
| High | 120 (20.1) | 56 (17.9) | 64 (22.5) | 0.164 |
| Arterial uptake (18F-FDG) | ||||
| Presence of uptake | 358 (48.1) | 135 (36.0) | 223 (60.3) | <0.001 |
| Number of uptakes | 0 (0–2) | 0 (0–1) | 1 (0–2) | <0.001 |
| SUVmax arterial uptake | 1.38 (1.26–1.52) | 1.30 (1.20–1.41) | 1.46 (1.37–1.59) | <0.001 |
| Plaques by magnetic resonance | ||||
| Plaque presence | 671 (90.1) | 337 (89.9) | 334 (90.3) | 0.854 |
| Number of plaques | 3 (2–5) | 3 (1–5) | 3 (2–5) | 0.842 |
| Global plaque burden | 365.6 (175.6–706.9) | 348.5 (164.9–681.0) | 375.8 (192.4–751.2) | 0.313 |
Data are presented as n (%) or median (Q1–Q3).
Bone marrow activation was defined when the mean BM SUVmax was above the median value (SUVmax 1.9).
Measured in 622 of 745 individuals.
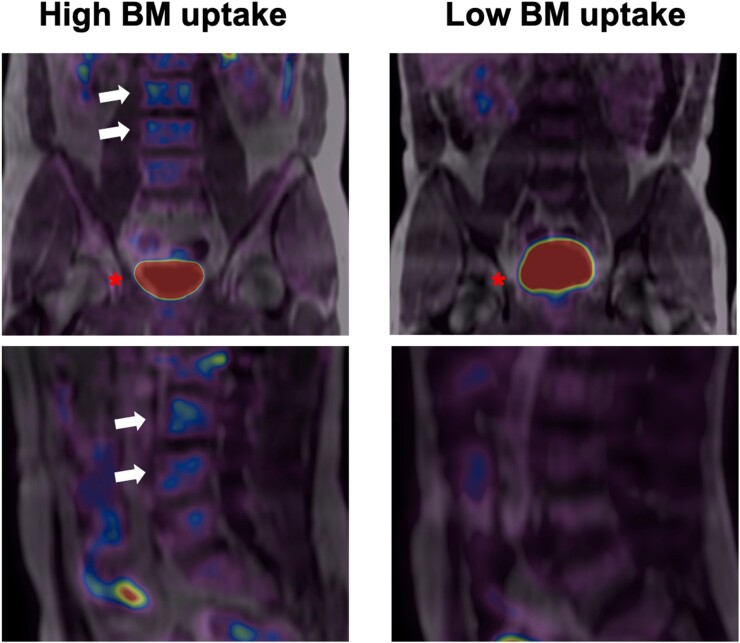
Figure 1.
Bone marrow uptake. Representative baseline 18F-fluorodeoxyglucose positron emission tomography/magnetic resonance imaging scans from participants. The left panel shows fused 18F-fluorodeoxyglucose positron emission tomography/magnetic resonance imaging in coronal (upper) and sagittal (lower) views. L3 and L4 vertebrae (white arrows) present high 18F-fluorodeoxyglucose uptake (visualized in blue). The right panel shows the same coronal and sagittal views; 18F-fluorodeoxyglucose uptake is not visualized. The bladder is visualized in red in both upper panels (red asterisk).
Figure 2.
Population with bone marrow activation. Characteristics of participants with baseline bone marrow 18F-fluorodeoxyglucose uptake compared with the group without bone marrow uptake. Participants with bone marrow 18F-fluorodeoxyglucose uptake were more frequently male and had a higher prevalence of metabolic syndrome and its components, central obesity, hypertension, and altered glucose metabolism. The bone marrow uptake group also had higher levels of systemic inflammatory markers (high-sensitivity C-reactive protein and ferritin) and showed higher counts of leucocytes and red blood cells.
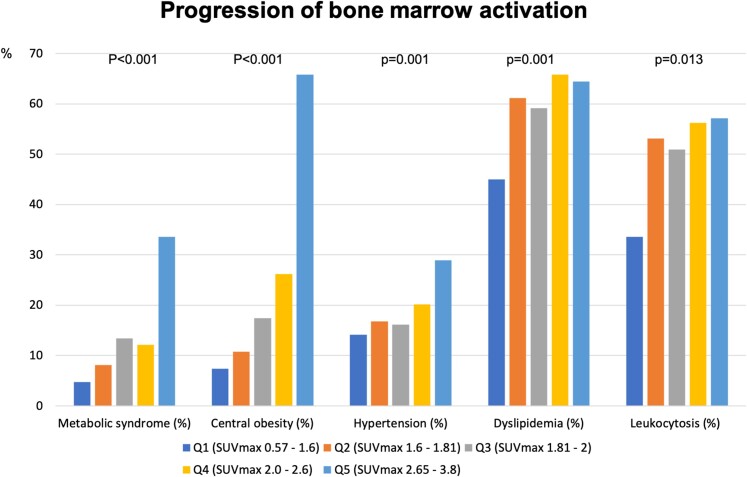
To explore the association between the degree of BM activation and participant characteristics, we divided the population into BM-uptake quintiles according to SUVmax. The characteristics of each BM-uptake subgroup are presented in Table 2. The higher the degree of BM activation (i.e. the higher the BM-uptake quintile), the higher the percentage of men and the more frequent the presence of central obesity. The same pattern of increase across quintiles was observed for the frequency of MetS; hypertension; low HDL; and elevated fasting glucose, HbA1c, and HOMA-IR.
Table 2.
Study population characteristics stratified by bone marrow activation (18F-fluorodeoxyglucose uptake) quintile
| BM-uptake Q1 | BM-uptake Q2 | BM-uptake Q3 | BM-uptake Q4 | BM-uptake Q5 | P for trend | |
|---|---|---|---|---|---|---|
| (n = 149) | (n = 149) | (n = 149) | (n = 149) | (n = 149) | ||
| Mean BM SUVmax | 0.57–1.6 | 1.6–1.81 | 1.81–2.0 | 2.0–2.3 | 2.3–3.8 | |
| Age, years | 50.0 (46.7–53.0) | 51.7 (47.5–54.3) | 49.9 (45.6–52.8) | 50.5 (47.0–53.8) | 50.9 (47.1–53.8) | 0.431 |
| Men | 105 (70.5) | 128 (85.9) | 128 (85.9) | 134 (89.9) | 129 (86.6) | <0.001 |
| Metabolic syndrome and components | ||||||
| Metabolic syndrome | 7 (4.7) | 12 (8.1) | 20 (13.4) | 18 (12.1) | 50 (33.6) | <0.001 |
| Central obesity | 11 (7.4) | 16 (10.7) | 27 (18.1) | 40 (26.8) | 98 (65.8) | <0.001 |
| Triglycerides, mg/dL | 81 (61–113) | 96 (72–128) | 101 (68–128) | 103 (73–133) | 107 (88–147) | <0.001 |
| HDL-C, mg/dL | 50.8 ± 12.5 | 46.3 ± 11.3 | 47.0 ± 10.6 | 45.5 ± 10.7 | 42.8 ± 10.2 | <0.001 |
| Fasting glucose, mg/dL | 87 (83–92) | 90 (83–95) | 91 (84–98) | 93 (88–97) | 94 (87–101) | <0.001 |
| SBP, mmHg | 117.6 ± 12.4 | 119.8 ± 11.6 | 120.5 ± 11.3 | 121.2 ± 11.8 | 123.5 ± 13.5 | <0.001 |
| DBP, mmHg | 73.1 ± 9.1 | 74.3 ± 8.1 | 75.1 ± 7.9 | 75.4 ± 9.2 | 78.6 ± 10.1 | <0.001 |
| Other cardiovascular risk factors | ||||||
| Family history of CV disease | 28 (18.8) | 26 (17.4) | 30 (20.1) | 38 (25.5) | 32 (21.5) | 0.201 |
| Current smoking | 47 (32.2) | 45 (30.6) | 31 (21.2) | 32 (22.1) | 42 (28.6) | 0.176 |
| Hypertension | 21 (14.1) | 25 (16.8) | 24 (16.1) | 30 (20.1) | 43 (28.9) | 0.001 |
| Dyslipidaemia | 67 (45.0) | 91 (61.1) | 88 (59.1) | 98 (65.8) | 96 (64.4) | 0.001 |
| Diabetes | 4 (2.7) | 2 (1.3) | 11 (7.4) | 6 (4.0) | 11 (7.4) | 0.026 |
| BMI, kg/m2 | 24.7 ± 3.0 | 25.9 ± 2.5 | 26.7 ± 2.7 | 28.1 ± 2.7 | 30.5 ± 3.2 | <0.001 |
| Weight, kg | 71.9 ± 12.1 | 77.3 ± 10.1 | 79.6 ± 10.6 | 85.2 ± 10.9 | 93.1 ± 12.0 | <0.001 |
| Waist circumference, cm | 85.7 ± 10.0 | 90.7 ± 8.9 | 92.9 ± 8.5 | 96.7 ± 8.5 | 103.0 ± 9.5 | <0.001 |
| Treatment | ||||||
| Antihypertensive therapy | 16 (10.7) | 15 (10.1) | 18 (12.1) | 21 (14.1) | 30 (20.1) | 0.010 |
| Lipid-lowering therapy | 14 (9.4) | 20 (13.4) | 25 (16.8) | 32 (21.5) | 22 (14.8) | 0.043 |
| Antidiabetic therapy | 3 (2.0) | 1 (0.7) | 11 (7.4) | 5 (3.4) | 8 (5.4) | 0.057 |
| Biochemistry | ||||||
| Total cholesterol, mg/dL | 205.8 ± 30.3 | 209.3 ± 34.2 | 210.0 ± 31.2 | 210.8 ± 36.4 | 205.6 ± 35.1 | 0.837 |
| LDL-C, mg/dL | 136.4 ± 28.3 | 141.2 ± 29.9 | 141.9 ± 27.0 | 142.3 ± 32.9 | 137.5 ± 31.8 | 0.583 |
| HbA1c, % | 5.4 (5.2–5.6) | 5.4 (5.1–5.6) | 5.5 (5.2–5.7) | 5.5 (5.2–5.8) | 5.5 (5.3–5.7) | 0.005 |
| HOMA-IR, % | 0.9 (0.6–1.3) | 1.2 (0.8–1.9) | 1.2 (0.8–1.9) | 1.5 (1.0–2.1) | 2.0 (1.3–2.9) | <0.001 |
| Insulin, µU/mL | 4.0 (2.8–5.8) | 5.1 (3.8–7.5) | 5.3 (3.8–7.5) | 6.3 (4.3–8.6) | 8.2 (6.0–11.5) | <0.001 |
| Inflammatory markers | ||||||
| hs-CRP, mg/dL | 0.07 (0.04–0.13) | 0.10 (0.05–0.18) | 0.10 (0.05–0.20) | 0.12 (0.07–0.21) | 0.15 (0.08–0.28) | <0.001 |
| Ferritin, ng/mLa | 90.2 (40.9–152.4) | 127.7 (71.3–198.5) | 122.4 (69.0–224.6) | 137.5 (74.5–200.2) | 154.9 (81.6–253.0) | <0.001 |
| Erythrocyte sedimentation rate (1 h), mm | 5 (4–8) | 5 (4–7) | 5 (4–7) | 6 (4–8) | 6 (4–8) | 0.277 |
| Fibrinogen, mg/dL | 255.4 (230.1–286.6) | 266.2 (236.5–296.1) | 265.7 (235.3–294.6) | 264.4 (239.9–299.2) | 277.2 (249.8–302.8) | 0.001 |
| P-selectin, ng/mL | 125.0 (102.8–156.1) | 130.5 (105.9–166.2) | 135.6 (106.9–166.7) | 135.3 (106.7–167.2) | 144.1 (115.4–180.5) | 0.002 |
| VCAM-1, ng/mL | 615.3 (503.8–777.6) | 625.1 (499.1–790.5) | 681.2 (530.4–859.5) | 674.0 (518.6–849.0) | 680.8 (555.1–821.7) | 0.027 |
| Blood count | ||||||
| Leucocytes, 103 cells/µL | 5.59 (4.91–6.72) | 5.93 (4.95–7.07) | 5.80 (4.91–6.75) | 5.98 (5.17–7.13) | 6.20 (5.35–7.48) | 0.001 |
| Leucocytosis (>10.5 × 103 cells/µL) | 0 (0) | 5 (3.4) | 4 (2.7) | 4 (2.7) | 4 (2.7) | 0.225 |
| Red blood cell count, 106 cells/µL | 4.71 (4.38–4.99) | 4.83 (4.60–5.05) | 4.85 (4.59–5.05) | 4.90 (4.67–5.10) | 4.90 (4.65–5.15) | <0.001 |
| Red cell distribution width, % | 14.6 (14.0–15.2) | 14.6 (14.0–15.2) | 14.5 (13.9–15.1) | 14.7 (13.9–15.2) | 14.8 (14.2–15.2) | 0.089 |
| Haemoglobin, g/dL | 14.7 (13.8–15.3) | 15.1 (14.4–15.7) | 15.0 (14.2–15.6) | 15.1 (14.6–15.8) | 15.2 (14.5–15.8) | <0.001 |
| Haematocrit, % | 43.0 (40.9–45.6) | 44.5 (41.9–46.4) | 44.2 (41.7–46.0) | 44.6 (42.9–46.6) | 44.6 (42.8–46.7) | <0.001 |
| Platelets, 103 cells/µL | 228 (199–253) | 224 (196–256) | 222 (196–259) | 227 (200–257) | 222 (193–262) | 0.831 |
| Segmented neutrophils, 103 cell/µL | 3.13 (2.60–3.77) | 3.34 (2.72–4.26) | 3.19 (2.71–3.92) | 3.48 (2.61–4.42) | 3.48 (2.97–4.50) | 0.001 |
| Lymphocytes, 103 cells/µL | 1.83 (1.62–2.16) | 1.86 (1.55–2.17) | 1.86 (1.54–2.23) | 1.82 (1.55–2.22) | 1.91 (1.64–2.28) | 0.136 |
| Monocytes, 103 cells/µL | 0.39 (0.32–0.49) | 0.41 (0.33–0.53) | 0.41 (0.33–0.50) | 0.42 (0.35–0.51) | 0.43 (0.35–0.53) | 0.019 |
| Eosinophils, 103 cells/µL | 0.12 (0.08–0.20) | 0.12 (0.08–0.20) | 0.12 (0.07–0.21) | 0.13 (0.08–0.19) | 0.14 (0.09–0.23) | 0.361 |
| Basophils, 103 cells/µL | 0.05 (0.03–0.07) | 0.05 (0.03–0.06) | 0.04 (0.03–0.06) | 0.05 (0.03–0.06) | 0.05 (0.03–0.07) | 0.128 |
| Neutrophil to lymphocyte ratio | 1.66 (1.37–2.15) | 1.81 (1.46–2.25) | 1.74 (1.37–2.30) | 1.82 (1.42–2.45) | 1.88 (1.51–2.27) | 0.048 |
| SCORE risk score | ||||||
| Low (<1%) | 106 (72.1) | 88 (59.5) | 102 (73.9) | 82 (57.8) | 81 (57.9) | 0.003 |
| Intermediate (1–5%) | 41 (27.9) | 60 (40.5) | 35 (25.4) | 59 (41.5) | 59 (42.1) | 0.002 |
| High (>5%) | 0 (0.0) | 0 (0.0) | 1 (0.7) | 1 (0.7) | 0 (0) | 0.538 |
| ASCVD risk score | ||||||
| Low | 96 (73.8) | 69 (56.1) | 84 (70.0) | 57 (54.3) | 63 (52.5) | 0.001 |
| Intermediate | 17 (13.1) | 24 (19.5) | 18 (15.0) | 22 (20.9) | 28 (23.3) | 0.047 |
| High | 17 (13.1) | 30 (24.4) | 18 (15.0) | 26 (24.8) | 29 (24.2) | 0.040 |
| Arterial uptake (18F-FDG) | ||||||
| Presence of uptake | 37 (24.8) | 59 (39.6) | 73 (49.0) | 82 (55.0) | 107 (71.8) | <0.001 |
| Number of uptakes | 0 (0–0) | 0 (0–1) | 0 (0–1) | 1 (0–2) | 1 (0–2) | <0.001 |
| SUVmax arterial uptake | 1.26 (1.14–1.32) | 1.33 (1.24–1.43) | 1.37 (1.27–1.47) | 1.41 (1.32–1.51) | 1.58 (1.46–1.69) | <0.001 |
| Plaques by magnetic resonance | ||||||
| Plaque presence | 127 (85.2) | 136 (91.3) | 138 (92.6) | 134 (89.9) | 136 (91.3) | 0.166 |
| Number of plaques | 3 (1–5) | 3 (2–5) | 3 (2–5) | 3 (2–5) | 3 (2–5) | 0.855 |
| Global plaque burden, mm3 | 348.5 (164.9–669.9) | 362.3 (166.8–699.5) | 370.2 (177.8–688.8) | 372.5 (175.6–753.5) | 371.4 (204.8–753.3) | 0.365 |
Data are presented as n (%) or median (Q1–Q3).
Measured in 622 of 745 individuals.
The increase in BM activation was also associated with elevated numbers of leucocytes (mainly neutrophils, with an increased neutrophil to lymphocyte ratio), and red blood cells, with numbers increasing progressively across quintiles. The distribution of relevant participant characteristics stratified by BM-uptake quintile is shown in Figure 3. The circulating systemic inflammation markers, hs-CRP, ferritin, fibrinogen, P-selectin, and VCAM-1 also steadily increased across BM-uptake quintiles (Table 2).
Figure 3.
Progression of bone marrow activation. Prevalence of metabolic syndrome, central obesity, hypertension, dyslipidaemia, and leucocytosis stratified by quintiles of bone marrow 18F-fluorodeoxyglucose uptake.
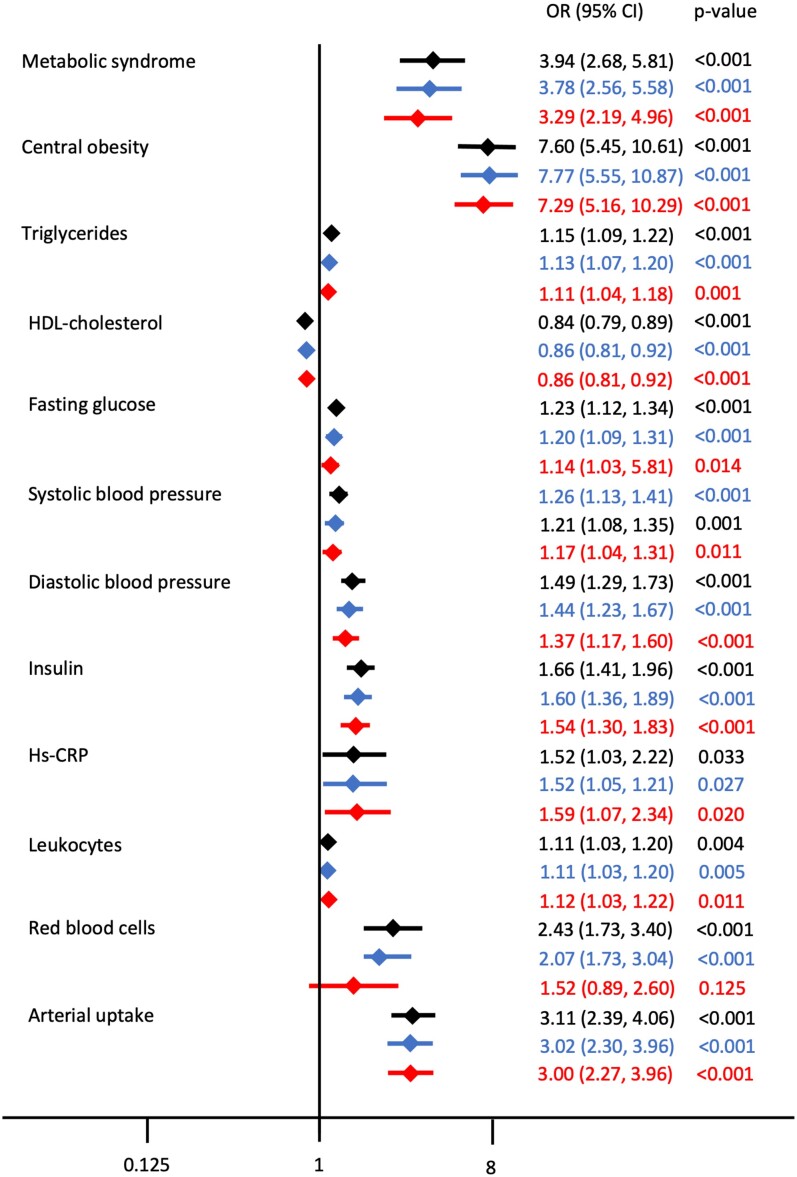
When adjusted for Model 1 (age and sex) and Model 2 (age, sex, glucose levels before PET/MRI, smoking, haemoglobin, and hs-CRP), BM activation remained significantly associated with MetS and its components, insulin levels, hs-CRP, leucocytes, and arterial uptake (Figure 4).
Figure 4.
Unadjusted and adjusted associations for different factors with bone marrow activation. Associations between different factors with bone marrow activation, expressed as odds ratio with its 95% confidence interval. The unadjusted estimates are presented in black, the estimates adjusted for Model 1 (adjusted for age and sex) are presented in blue, and the estimates adjusted for Model 2 (adjusted for age, sex, glucose levels before positron emission tomography/magnetic resonance imaging, smoking, haemoglobin, and high-sensitivity C-reactive protein) are presented in red.
When BM uptake was evaluated as a continuous variable, MetS and its components (particularly central obesity) and the presence of arterial uptake had the largest effect size on BM activation (see Supplementary material online, Table S1); this was consistent with the analysis of BM-uptake quintiles.
Association between bone marrow activation and early atherosclerosis
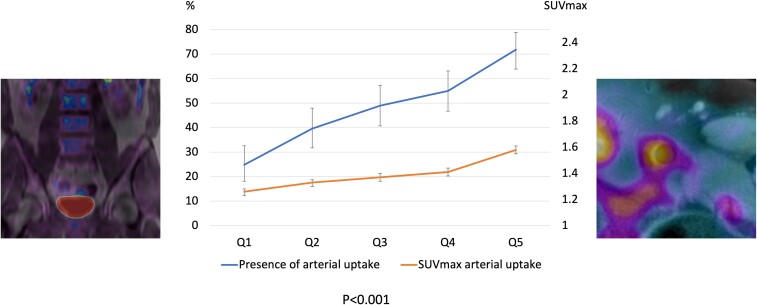
Arterial 18F-FDG uptake is a surrogate for high vascular metabolic activity due to macrophage accumulation, the precursor of atherosclerosis.27,38 Bone marrow activation was significantly associated with the presence of vascular 18F-FDG uptake (60 vs. 36%, P < 0.001). Participants with BM activation also had more sites of vascular 18F-FDG uptakes, and higher degree of uptake (vascular SUVmax) (Tables 1 and 2 and Figure 5).
Figure 5.
Relationship between bone marrow and vascular 18F-fluorodeoxyglucose uptake. The left and right panels show representative positron emission tomography/magnetic resonance imaging analysis of 18F-fluorodeoxyglucose uptake in lumbar vertebrae and vascular tissue, respectively. The chart shows increases in the presence of vascular uptake (left Y-axis) and in vascular-uptake SUVmax (right Y-axis) with increasing bone marrow uptake quintile.
We next explored the association between BM activation and vascular 18F-FDG uptake. Participants with BM activation and vascular 18F-FDG uptake tended to be older (51.2 vs. 49.4 years among those with BM activation and no vascular uptake, P < 0.001) and more frequently presented with MetS (26.9 vs. 15%, P = 0.007) (Table 3). The co-occurrence of BM activation and vascular 18F-FDG uptake showed a significant association with central obesity (48.9 vs. 29.3% in participants with BM activation without vascular 18F-FDG uptake, P < 0.001), smoking (28.6 vs. 16.2%, P = 0.007), higher total cholesterol (211.9 vs. 204.5 mg/dL, P = 0.045), lower HDL-cholesterol (43.1 vs. 46.7 mg/dL, P = 0.001), higher triglyceride (112 vs. 92 mg/dL, P < 0.001), and higher fasting glucose levels (94 vs. 92 mg/dL, P = 0.013), as well as with lower rates of individuals assigned to a low-risk group in the SCORE algorithm. There was no between-group difference in age, family history of cardiovascular disease, and LDL-cholesterol. The co-occurrence of BM activation and vascular 18F-FDG uptake was associated with elevated levels of HbA1c (5.5 vs. 5.4%, P = 0.042), plasma insulin (7.6 vs. 5.8 µU/mL, P < 0.001) and insulin resistance measured by HOMA-IR (1.9 vs. 1.4%, P < 0.001). Co-occurring BM activation and vascular 18F-FDG uptake were associated with significantly elevated numbers of leucocytes (6.22 × 103 vs. 5.83 × 103 cells/µL in the BM activation group without vascular 18F-FDG uptake, P = 0.023), especially monocytes (0.43 × 103 vs. 0.40 × 103 cells/µL, P = 0.007) and of red blood cells (4.91 × 106 vs. 4.86 × 106 cells/µL, P = 0.038). This trend was accompanied by significantly elevated markers of systemic inflammation in the BM activation plus vascular 18F-FDG uptake group, including hs-CRP (0.15 vs. 0.11 mg/dL, P < 0.001).
Table 3.
Characteristics of population with bone marrow activation according to the presence or absence of vascular 18F-fluorodeoxyglucose uptake
| BM activation without vascular uptake | BM activation and vascular uptake | P-value | |
|---|---|---|---|
| (n = 147) | (n = 223) | ||
| Age, years | 49.4 (45.4–52.9) | 51.2 (48.0–54.1) | <0.001 |
| Men | 123 (83.7) | 201 (90.1) | 0.065 |
| Metabolic syndrome and its components | |||
| Metabolic syndrome | 22 (15.0) | 60 (26.9) | 0.007 |
| Central obesity | 43 (29.3) | 108 (48.9) | <0.001 |
| Triglycerides, mg/dL | 92 (68–128) | 112 (86–147) | <0.001 |
| HDL-C, mg/dL | 46.7 ± 11.7 | 43.1 ± 9.8 | 0.001 |
| Fasting glucose, mg/dL | 92 (85–98) | 94 (88–101) | 0.013 |
| SBP, mmHg | 120.4 ± 11.4 | 123.6 ± 13.2 | 0.018 |
| DBP, mmHg | 75.6 ± 8.7 | 77.8 ± 9.9 | 0.029 |
| Other cardiovascular risk factors | |||
| Family history of CV disease | 29 (19.7) | 52 (23.3) | 0.414 |
| Current smoking | 23 (16.2) | 63 (28.6) | 0.007 |
| Hypertension | 31 (21.1) | 56 (25.1) | 0.372 |
| Dyslipidaemia | 85 (57.8) | 152 (68.2) | 0.043 |
| Diabetes | 6 (4.1) | 15 (6.7) | 0.282 |
| BMI, kg/m2 | 27.9 ± 3.3 | 29.5 ± 3.0 | <0.001 |
| Weight, kg | 84.1 ± 12.3 | 89.5 ± 12.1 | <0.001 |
| Waist circumference, cm | 95.4 ± 9.3 | 100.8 ± 9.3 | <0.001 |
| Treatment | |||
| Antihypertensive therapy | 22 (15.0) | 38 (17.0) | 0.596 |
| Lipid-lowering therapy | 25 (17.0) | 38 (17.0) | 0.993 |
| Antidiabetic therapy | 6 (4.1) | 11 (4.9) | 0.702 |
| Biochemistry | |||
| Total cholesterol, mg/dL | 204.5 ± 31.3 | 211.9 ± 36.5 | 0.045 |
| LDL-C, mg/dL | 137.1 ± 27.1 | 143.0 ± 33.7 | 0.077 |
| HOMA-IR, % | 1.4 (1.0–2.1) | 1.9 (1.2–2.9) | <0.001 |
| HbA1c, % | 5.4 (5.2–5.7) | 5.5 (5.3–5.8) | 0.042 |
| Insulin, µU/mL | 5.8 (4.3–8.0) | 7.6 (5.4–11.5) | <0.001 |
| Inflammatory markers | |||
| hs-CRP, mg/dL | 0.11 (0.05–0.18) | 0.15 (0.08–0.29) | <0.001 |
| Ferritin, ng/mLa | 131.38 (61.13–207.27) | 151.23 (81.63–224.11) | 0.390 |
| Erythrocyte sedimentation rate (1 h), mm | 5 (4–7) | 6 (4–8) | 0.084 |
| Fibrinogen, mg/dL | 264.4 (237.3–291.5) | 273.1 (240.9–302.8) | 0.178 |
| P-selectin, ng/mL | 139.9 (111.0–177.0) | 139.3 (107.7–177.2) | 0.981 |
| Vascular cell adhesion molecule-1, ng/mL | 645.2 (512.4–861.2) | 712.4 (557.1–838.8) | 0.379 |
| Blood count | |||
| Leucocytes, 103 cells/µL | 5.83 (4.99–6.89) | 6.22 (5.23–7.33) | 0.023 |
| Leucocytosis (>10.5 × 103 cells/µL) | 2 (1.4) | 7 (3.1) | 0.277 |
| Red blood cell count, 106 cells/µL | 4.86 (4.59–5.05) | 4.91 (4.68–5.18) | 0.038 |
| Red cell distribution width, % | 14.6 (14.0–15.2) | 14.7 (14.0–15.3) | 0.130 |
| Haemoglobin, g/dL | 15.0 (14.4–15.6) | 15.1 (14.5–15.9) | 0.107 |
| Haematocrit, % | 44.3 (41.6–46.1) | 44.7 (42.9–46.7) | 0.047 |
| Platelet count, 103 cells/µL | 228 (194–259) | 221 (193–258) | 0.452 |
| Segmented neutrophils,103 cell/µL | 3.30 (2.69–4.17) | 3.45 (2.86–4.50) | 0.072 |
| Lymphocytes,103 cells/µL | 1.83 (1.55–2.13) | 1.92 (1.58–2.29) | 0.097 |
| Monocytes, (103 cells/µL | 0.40 (0.32–0.49) | 0.43 (0.36–0.54) | 0.007 |
| Eosinophils,103 cells/µL | 0.12 (0.08–0.19) | 0.14 (0.08–0.23) | 0.326 |
| Basophils,103 cells/µL | 0.04 (0.03–0.06) | 0.05 (0.03–0.07) | 0.085 |
| Neutrophil to lymphocyte ratio | 1.79 (1.42–2.36) | 1.85 (1.48–2.38) | 0.498 |
| SCORE risk score | |||
| Low (<1%) | 100 (71.9) | 114 (54.0) | 0.001 |
| Intermediate (1−5%) | 39 (28.1) | 95 (45.0) | 0.001 |
| High (>5%) | 0 (0.0) | 2 (1.0) | 0.250 |
| ASCVD risk score | |||
| Low | 83 (72.8) | 79 (46.2) | <0.001 |
| Intermediate | 15 (13.2) | 44 (25.7) | 0.010 |
| High | 16 (14.0) | 48 (28.1) | 0.005 |
| Plaques by magnetic resonance | |||
| Plaque presence | 122 (83.0) | 212 (95.1) | <0.001 |
| Number of plaques | 2 (1–4) | 4 (2–5) | <0.001 |
| Global plaque burden, mm3 | 284.5 (125.2–519.5) | 448.4 (229.4–819.0) | <0.001 |
| Plaques by 2D vascular ultrasound | |||
| Plaque presence | 117 (81.2) | 203 (94.4) | <0.001 |
| Number of plaques | 2 (1–4.5) | 4 (2–7) | <0.001 |
| Plaques by 3D vascular ultrasound | |||
| Plaque presence | 90 (67.2) | 180 (87.4) | <0.001 |
| Global plaque burden, mm3 | 30.3 (0–128.6) | 100.8 (37.8–214.4) | <0.001 |
Data are presented as n (%) or median (Q1–Q3).
Measured in 622 of 745 individuals.
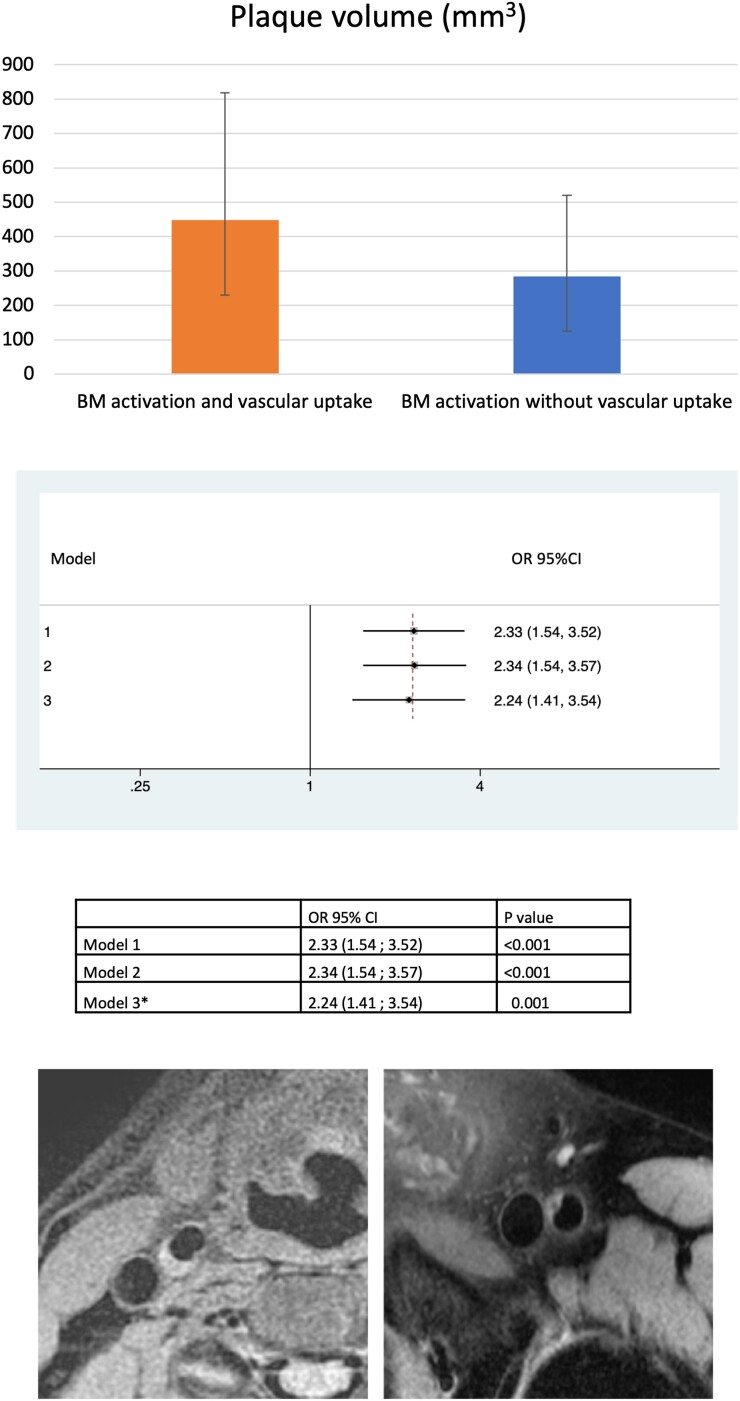
More advanced stages of atherosclerosis are characterized by full-grown plaques. We observed that subjects with BM activation plus vascular 18F-FDG uptake had a higher prevalence of plaques than those with BM activation but without vascular 18F-FDG uptake (95.1 vs. 83%, P < 0.001). Similarly, the BM activation plus vascular 18F-FDG uptake group had more plaques (4 vs. 2, P < 0.001) and a higher plaque burden (448.4 vs. 284.5 mm3, P < 0.001). These associations remained significant after adjusting for classical risk factors (odds ratio 2.33, 95% confidence interval 1.54–3.52, P < 0.001 in the fully adjusted model) (Figures 6 and 7). This association was consistent when atherosclerosis was evaluated by 2D and 3D vascular ultrasound; subjects with BM activation plus 18F-FDG uptake had a higher prevalence and number of plaques and higher plaque burden when compared with those with BM activation but without vascular uptake.
Figure 6.
Bone marrow activation in the presence of vascular 18F-fluorodeoxyglucose uptake is associated with higher atherosclerotic plaque volume. Participants with co-occurring bone marrow activation and vascular 18F-fluorodeoxyglucose uptake had a significantly higher plaque burden than those with bone marrow activation but no vascular 18F-fluorodeoxyglucose uptake. The upper panel shows atherosclerotic plaque volume (mm3) in the group with bone marrow activation and vascular uptake (orange bar) and in the group with bone marrow activation without vascular uptake (blue bar). The mid-panel shows the comparison of adjusted odds ratios and 95% confidence interval for the different models. *In Model 3, 105 participants taking lipid-lowering therapies were eliminated. The lower panel shows representative magnetic resonance images of atherosclerotic plaques.
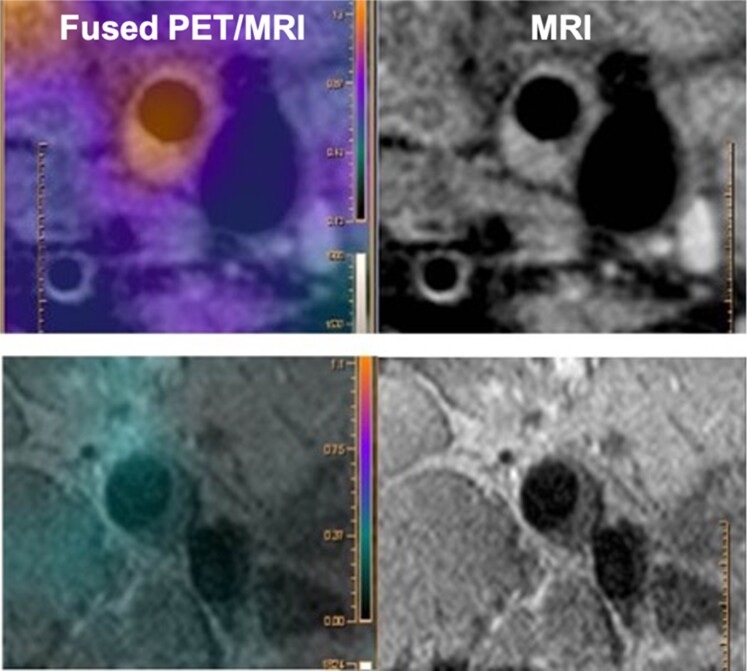
Figure 7.
Positron emission tomography/magnetic resonance imaging analysis of the atherosclerotic plaque 18F-fluorodeoxyglucose uptake. The upper row shows a carotid atherosclerotic plaque with 18F-fluorodeoxyglucose uptake. The lower row shows a femoral atherosclerotic plaque without 18F-fluorodeoxyglucose uptake.
Bone marrow activation in the absence of systemic inflammation
To assess whether BM activation occurred as part of a systemic inflammatory reaction, we studied the subgroup of 402 participants showing no systemic inflammation (below-median hs-CRP). The characteristics of this subpopulation stratified by BM activation are summarized in Table 4. Participants with BM activation were more frequently male (86.4 vs. 75.8%, P = 0.009), and were more frequently positive for MetS (17.9 vs. 3.8%, P < 0.001) and its components, hypertension (25.3 vs. 12.9%, P = 0.001), and diabetes (7.4 vs. 2.5%, P = 0.020). This group also showed higher elevations in fasting glucose (92 vs. 88 mg/dL, P < 0.001), and insulin resistance index (HOMA-IR 1.54 vs. 0.97%, P < 0.001) and plasma insulin (6.1 vs. 4.3 µU/mL, P < 0.001); and presented lower levels of HDL-cholesterol (46.1 vs. 49.7 mg/dL, P = 0.002) and higher levels of triglycerides (97 vs. 83 mg/dL, P = 0.010). The BM activation group showed a higher elevation of erythropoiesis (red blood cell count 4.90 × 106 vs. 4.76 × 106 cells/µL, P = 0.001); however, leucocyte numbers did not differ between inflammation-free participants with and without BM activation. In this subgroup without systemic inflammation, BM activation was significantly associated with higher arterial metabolic activity: more prevalence of vascular 18F-FDG uptake (51.9 vs. 31.2%, P < 0.001), and more sites of vascular 18F-FDG uptake and higher vascular SUVmax.
Table 4.
Bone marrow activation in the subpopulation without systemic inflammation (below-median high-sensitivity C-reactive protein)
| Total | No bone marrow uptake | Bone marrow uptakeb | P-value | |
|---|---|---|---|---|
| (n = 402) | CRP < median | CRP < median | ||
| (n = 240) | (n = 162) | |||
| Age, years | 50.1 (46.7–53.2) | 50.1 (46.2–53.2) | 50.3 (46.7–53.2) | 0.657 |
| Men | 322 (80.1) | 182 (75.8) | 140 (86.4) | 0.009 |
| Metabolic syndrome and its components | ||||
| Metabolic syndrome | 38 (9.5) | 9 (3.8) | 29 (17.9) | <0.001 |
| Central obesity | 73 (18.2) | 17 (7.1) | 56 (34.6) | <0.001 |
| Triglycerides, mg/dL | 88 (64–118) | 83 (62–115) | 97 (71–128) | 0.010 |
| HDL-C, mg/dL | 48.2 ± 11.4 | 49.7 ± 11.4 | 46.1 ± 11.1 | 0.002 |
| Fasting glucose, mg/dL | 89 (84–96) | 88 (83–94) | 92 (85–98) | <0.001 |
| SBP, mmHg | 119.4 ± 11.8 | 117.7 ± 11.3 | 121.9 ± 12.1 | <0.001 |
| DBP, mmHg | 74.6 ± 8.9 | 72.9 ± 8.4 | 77.2 ± 9.1 | <0.001 |
| Other cardiovascular risk factors | ||||
| Family history of CV disease | 88 (21.9) | 50 (20.8) | 38 (23.5) | 0.533 |
| Current smoking | 92 (23.3) | 65 (27.4) | 27 (17.1) | 0.017 |
| Hypertension | 72 (17.9) | 31 (12.9) | 41 (25.3) | 0.001 |
| Dyslipidaemia | 218 (54.2) | 118 (49.2) | 100 (61.7) | 0.013 |
| Diabetes | 18 (4.5) | 6 (2.5) | 12 (7.4) | 0.020 |
| BMI, kg/m2 | 26.4 ± 3.3 | 25.1 ± 2.8 | 28.2 ± 3.1 | <0.001 |
| Weight, kg | 79.0 ± 13.4 | 74.0 ± 11.4 | 86.4 ± 12.8 | <0.001 |
| Waist circumference, cm | 91.5 ± 10.8 | 87.4 ± 9.5 | 97.5 ± 9.7 | <0.001 |
| Treatment | ||||
| Antihypertensive therapy | 50 (12.4) | 23 (9.6) | 27 (16.7) | 0.035 |
| Lipid-lowering therapy | 59 (14.7) | 31 (12.9) | 28 (17.3) | 0.225 |
| Antidiabetic therapy | 15 (3.7) | 5 (2.1) | 10 (6.2) | 0.034 |
| Biochemistry | ||||
| Total cholesterol, mg/dL | 205.6 ± 33.3 | 206.2 ± 30.9 | 204.7 ± 36.6 | 0.646 |
| LDL-C, mg/dL | 137.5 ± 30.2 | 137.7 ± 28.0 | 137.2 ± 33.2 | 0.869 |
| HbA1c, % | 5.4 (5.2–5.7) | 5.4 (5.2–5.7) | 5.4 (5.2–5.7) | 0.404 |
| HOMA-IR, % | 1.15 (0.77–1.86) | 0.97 (0.67–1.52) | 1.54 (0.98–2.23) | <0.001 |
| Insulin, µU/mL | 5.1 (3.6–7.2) | 4.3 (3.2–6.2) | 6.1 (4.3–8.2) | <0.001 |
| Inflammatory markers | ||||
| hs-CRP, mg/dL | 0.06 (0.04–0.08) | 0.06 (0.04–0.08) | 0.07 (0.04–0.08) | 0.080 |
| Ferritin, ng/mLa | 107.7 (56.5–197.9) | 101.7 (46.2–187.5) | 122.8 (70.5–227.4) | 0.015 |
| Erythrocyte sedimentation rate (1 h), mm | 5 (4–6) | 5 (4–6) | 5 (2–7) | 0.311 |
| Fibrinogen, mg/dL | 252.0 (229.4–275.4) | 252.5 (229.8–276.1) | 250.9 (226.6–275.1) | 0.926 |
| P-selectin, ng/mL | 131.1 (103.4–162.0) | 125.8 (103.2–160.2) | 139.3 (103.7–168.0) | 0.071 |
| Vascular cell adhesion molecule-1, ng/mL | 638.3 (512.8–816.6) | 620.4 (507.8–788.8) | 699.9 (514.4–862.5) | 0.113 |
| Blood count | ||||
| Leucocytes, 103 cells/µL | 5.59 (4.86–6.64) | 5.55 (4.83–6.73) | 5.63 (4.93–6.47) | 0.575 |
| Leucocytosis, (>10.5 × 103 cells/µL) | 2 (0.5) | 2 (0.8) | 0 (0) | 0.244 |
| Red blood cells, 106 cells/µL | 4.81 (4.51–5.06) | 4.76 (4.46–5.02) | 4.90 (4.62–5.15) | 0.001 |
| Red cell distribution width, % | 14.5 (14.0–15.1) | 14.5 (14.0–15.0) | 14.6 (14.0–15.2) | 0.245 |
| Haemoglobin, g/dL | 14.9 (14.0–15.6) | 14.8 (13.9–15.5) | 15.1 (14.2–15.9) | 0.002 |
| Haematocrit, % | 43.9 (41.6–46.3) | 43.5 (41.1–45.8) | 44.5 (42.2–46.9) | 0.005 |
| Platelets, 103 cells/µL | 219 (192–255) | 224 (196–256) | 214 (189–244) | 0.074 |
| Segmented neutrophils, 103 cell/µL | 3.10 (2.56–3.80) | 3.09 (2.55–3.88) | 3.12 (2.59–3.77) | 0.839 |
| Lymphocytes, 103 cells/µL | 1.83 (1.54–2.17) | 1.82 (1.52–2.18) | 1.87 (1.55–2.16) | 0.471 |
| Monocytes, 103 cells/µL | 0.39 (0.33–0.48) | 0.39 (0.33–0.48) | 0.39 (0.33–0.47) | 0.678 |
| Eosinophils number (1000 cells/µL) | 0.12 (0.08–0.19) | 0.11 (0.08–0.19) | 0.12 (0.07–0.20) | 0.547 |
| Eosinophils, 103 cells/µL | 0.04 (0.03–0.06) | 0.05 (0.03–0.06) | 0.04 (0.02–0.06) | 0.094 |
| Neutrophil to lymphocyte ratio | 1.68 (1.37–2.10) | 1.71 (1.37–2.13) | 1.66 (1.36–2.08) | 0.431 |
| SCORE risk score | ||||
| Low (<1%) | 265 (68.6) | 165 (70.2) | 100 (66.2) | 0.410 |
| Intermediate (1–5%) | 121 (31.4) | 70 (29.8) | 51 (33.8) | 0.410 |
| High (>5%) | 0 (0.0) | 0 (0.0) | 0 (0.0) | — |
| ASCVD risk score | ||||
| Low | 216 (66.9) | 139 (69.5) | 77 (62.6) | 0.201 |
| Intermediate | 55 (17.0) | 33 (16.5) | 22 (17.9) | 0.748 |
| High | 52 (16.1) | 28 (14.0) | 24 (19.5) | 0.191 |
| Plaques by magnetic resonance | ||||
| Plaque presence | 357 (88.8) | 213 (88.8) | 144 (88.9) | 0.965 |
| Number of plaques | 3 (1–5) | 3 (1–5) | 3 (1–4) | 0.708 |
| Global plaque burden | 328.7 (162.4–616.0) | 321.6 (150.2–604.3) | 347.3 (180.2–628.0) | 0.647 |
| Arterial uptake (18F-FDG) | ||||
| Presence of uptake | 159 (39.5) | 75 (31.2) | 84 (51.9) | <0.001 |
| Number of uptakes | 0 (0–1) | 0 (0–1) | 1 (0–2) | <0.001 |
| SUVmax arterial uptake | 1.33 (1.23–1.45) | 1.28 (1.18–1.37) | 1.44 (1.32–1.54) | <0.001 |
Data are presented as n (%) or median (Q1–Q3).
Measured in 622 of 745 individuals.
Bone marrow activation was defined when the mean BM SUVmax was above the median value (SUVmax 1.9).
Discussion
The present study analysed BM activation in a population of apparently healthy individuals from the PESA study who underwent whole body 18F-FDG PET/MRI. The main study findings are as follows: (i) BM activation is associated with MetS and its individual components, as well as with elevated numbers of leucocytes, and systemic inflammation; (ii) BM activation is associated with high arterial metabolic activity (high arterial 18F-FDG uptake), a surrogate for macrophage infiltration (i.e. the precursor of atherosclerosis); (iii) the association between BM activation and MetS, and between BM activation and arterial 18F-FDG uptake is maintained even in the absence of systemic inflammation; and (iv) co-occurring BM activation and vascular 18F-FDG uptake was significantly associated with more advanced stages of atherosclerosis (higher plaque prevalence, number of plaques, and plaque burden). To the best of our knowledge, this is the first demonstration of an association between BM activation and metabolic factors linked to atherosclerosis in humans.
The relationship between BM activation and acute cardiovascular events has been described extensively, both in experimental models and in clinical studies. In mice, acute myocardial infarction is followed by activation of the sympathetic nervous system that increases BM activation signals. These stimuli trigger the release of progenitor cells from the BM and activate monocyte production in the spleen,6,7 which together with local plaque macrophage proliferation produce a rapid turnover that facilitates atherosclerosis progression.39
Experimental studies have demonstrated that increased haematopoietic activity in BM plays a central role in the association between cardiovascular risk factors, vascular inflammation, and atherosclerosis formation. Studies in mice have demonstrated that low HDL-cholesterol levels and hypercholesterolaemia are associated with an increase in BM myelopoietic activity which leads to increased neutrophilia and monocytosis.10,11 Elevated HDL-cholesterol levels have an anti-atherogenic role based on the suppression of BM myeloid proliferation. In conditions such as obesity, inflamed adipose tissue increases BM haematopoietic cells proliferation, leading to an exacerbated inflammation and associated disease processes.12,13 Hyperglycaemia and diabetes have been associated with an increased production of inflammatory myeloid cells in the BM, which exacerbate diabetes mellitus-associated complications including atherosclerosis.14,15 Sympathetic activation present in hypertension has been demonstrated to modulate BM haematopoiesis with the increase of myeloid cells and contributing to atherosclerosis and cardiovascular disease.
However, evidence of the association between BM metabolic activity and cardiovascular risk factors in humans is lacking.16
In this PESA subcohort of middle-aged healthy participants, BM activation indexed as lumbar vertebrae 18F-FDG uptake was associated with the presence of MetS and its components, with higher frequencies detected for central obesity, hypertension, low HDL-C, triglycerides, and altered glucose metabolism in these participants. Bone marrow activation was also associated with increased haematopoiesis and systemic inflammation, assessed from circulating hs-CRP, ferritin, fibrinogen, P-selectin, and VCAM-1 levels. Notably, the association between BM activation and MetS was maintained even in the absence of systemic inflammation (Table 4). The activation of BM in the presence of MetS even in the absence of systemic inflammation suggests that an association between them exists. Indeed, cardiovascular risk factors, including lifestyle factors, have been shown to contribute to haematopoiesis activation.40 In line with this finding, we found an association between BM activation and increased haematopoiesis, even in the absence of systemic inflammation (Table 4). The increase in leucocyte numbers and red blood cell counts was slight but still significant. Bone marrow activation showed a significant association with high arterial metabolic activity (a precursor of atherosclerosis) indexed by arterial 18F-FDG uptake. These results suggest that BM activation is an early phenomenon occurring in response to MetS that contributes to the early stages of atherosclerosis (Structured Graphical Abstract). It has been shown that BM activation causes the release of haematopoietic progenitors into the blood stream as a response to different stimuli including acute cardiovascular events.6 In the absence of such an event, as in the asymptomatic PESA population studied here, BM activation in response to cardiovascular risk factors triggers the release of haematopoietic progenitors and starts the inflammatory process that leads to atherosclerosis initiation and progression. In this regard, haematopoietic progenitor cells mobilized from the BM during the atherosclerotic process and seeded in the spleen, contribute to leucocyte production.40
Metabolic syndrome has been associated with several lipid abnormalities such as elevated triglyceride levels, low HDL-cholesterol levels, and increased proportion of small dense LDL particles, despite optimal LDL-cholesterol levels.41,42 Consistent with this, in our population, the group with BM activation presented higher triglyceride and lower HDL-cholesterol levels, but there was no association with LDL-cholesterol and total cholesterol levels.
In our population, we have observed a myeloid-bias haematopoiesis in patients with BM activation (Table 1). This is consistent with experimental findings, which have demonstrated that cardiovascular risk factors are linked to an increased BM myeloid proliferation.10–13
Bone marrow activation is associated with early atherosclerosis, characterized by high arterial metabolic activity (arterial 18F-FDG uptake). Moreover, BM activation in the presence of arterial 18F-FDG uptake is associated with more advanced stages of atherosclerosis, characterized by higher plaque burden, suggesting that high arterial metabolic activity is a prerequisite to trigger atherogenesis.
Taken together, our results suggest that, in the presence of cardiovascular risk factors, mainly those associated with MetS, there is an increase in BM metabolic activity that contributes to early atherosclerosis by increasing inflammatory cell proliferation. Further research is needed to define the mechanisms involved in this process in order to identify targets to prevent subclinical atherosclerosis progression and its clinical complications.
Study limitations
The study was subject to selection bias because the subcohort undergoing whole body 18F-FDG PET/MRI was selected from the total PESA cohort based on the presence of subclinical atherosclerosis on vascular ultrasound; this design could also lead to collider bias, which also applies when looking at the non-inflammation subgroup. However, if we had studied participants with no evidence of atherosclerosis, associations might have been even stronger. Moreover, the cross-sectional nature of the study precludes a definite conclusion about causal relationship between risk factors, BM activation, and atherosclerosis. Based on pathology studies, the progression of areas with 18F-FDG uptake to fully grown plaques is thought to be part of the natural history of atherosclerosis, but it has not been formally demonstrated yet.
The availability of data on markers of systemic inflammation allows us to validate that the association between cardiometabolic risk factors and increased BM activity is not simply a reflection of systemic inflammation. Around 20% of participants invited to participate declined enrolment or had MRI contraindications, comparable to similar studies.43 As previously reported,27 the first 100 PET studies could not be used due to inaccuracies in MRI-based attenuation and reconstruction; however, feasibility was almost 100% thereafter.
Conclusions
In middle-aged apparently healthy individuals, BM activation, identified as 18F-FDG uptake, is associated with cardiovascular risk factors, mainly MetS, and its components. This association is present even in the absence of systemic inflammation. Bone marrow activation is associated with early atherosclerosis, characterized by high arterial metabolic activity (18F-FDG uptake).
Supplementary material
Supplementary material is available at European Heart Journal online.
Supplementary Material
Acknowledgements
Simon Bartlett provided English editing. Braulio Pérez Asenjo and Belén Arroyo helped with imaging acquisition and interpretation.
Contributor Information
Ana Devesa, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; Cardiology Department, IIS-Fundación Jiménez Díaz University Hospital, Madrid, Spain; Icahn School of Medicine at Mount Sinai, New York, NY, USA.
Manuel Lobo-González, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain.
Juan Martínez-Milla, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; Cardiology Department, IIS-Fundación Jiménez Díaz University Hospital, Madrid, Spain.
Belén Oliva, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain.
Inés García-Lunar, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; Cardiology Department, Hospital Ramón y Cajal, Madrid, Spain; CIBER de Enfermedades Cardiovasculares (CIBERCV), Madrid, Spain.
Annalaura Mastrangelo, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain.
Samuel España, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; Departamento de Estructura de la Materia, Física Térmica y Electrónica, Universidad Complutense de Madrid, IdISSC, Madrid, Spain.
Javier Sanz, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; Icahn School of Medicine at Mount Sinai, New York, NY, USA.
José M Mendiguren, Banco de Santander, Madrid, Spain.
Hector Bueno, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; CIBER de Enfermedades Cardiovasculares (CIBERCV), Madrid, Spain; Cardiology Department, Hospital Universitario 12 de Octubre, and i+12 Research Institute, Madrid, Spain.
Jose J Fuster, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; CIBER de Enfermedades Cardiovasculares (CIBERCV), Madrid, Spain.
Vicente Andrés, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; CIBER de Enfermedades Cardiovasculares (CIBERCV), Madrid, Spain.
Antonio Fernández-Ortiz, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; CIBER de Enfermedades Cardiovasculares (CIBERCV), Madrid, Spain; Hospital Clínico San Carlos, Universidad Complutense, IdISSC, Madrid, Spain.
David Sancho, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain.
Leticia Fernández-Friera, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; Hospital Universitario HM Montepríncipe-CIEC, Madrid, Spain.
Javier Sanchez-Gonzalez, Philips Healthcare, Madrid, Spain.
Xavier Rossello, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; CIBER de Enfermedades Cardiovasculares (CIBERCV), Madrid, Spain; Cardiology Department, Hospital Universitari Son Espases-IDISBA, Palma de Mallorca, Spain.
Borja Ibanez, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; Cardiology Department, IIS-Fundación Jiménez Díaz University Hospital, Madrid, Spain; CIBER de Enfermedades Cardiovasculares (CIBERCV), Madrid, Spain.
Valentin Fuster, Centro Nacional de Investigaciones Cardiovasculares (CNIC), c/Melchor Fernández Almagro 3, Madrid 28029, Spain; Icahn School of Medicine at Mount Sinai, New York, NY, USA.
Funding
The PESA study is funded by the CNIC and Santander Bank. The present study was partially funded by an intramural grant CNIC-Severo Ochoa to D.S. and B.I. B.I. is supported by the European Commission (H2020-HEALTH 945118 and ERC-CoG 819775). The CNIC is supported by the ISCIII, the Ministry of Science and Innovation, and the Pro CNIC Foundation. CNIC is a Severo Ochoa Center of Excellence (CEX2020-001041-S).
Conflict of interest: J.S.-G. is a Philips employee. All other authors declare no conflicts of interest.
References
- 1. Libby P. Inflammation in atherosclerosis. Nature 2002;420:868–874. [DOI] [PubMed] [Google Scholar]
- 2. Libby P, Loscalzo J, Ridker PM, Farkouh ME, Hsue PY, Fuster V, et al. Inflammation, immunity, and infection in atherothrombosis: JACC review topic of the week. J Am Coll Cardiol 2018;72:2071–2081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Poller WC, Nahrendorf M, Swirski FK. Hematopoiesis and cardiovascular disease. Circ Res 2020;126:1061–1085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Swirski FK, Libby P, Aikawa E, Alcaide P, Luscinskas FW, Weissleder R, et al. Ly-6Chi monocytes dominate hypercholesterolemia-associated monocytosis and give rise to macrophages in atheromata. J Clin Invest 2007;117:195–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Visseren FLJ, Mach F, Smulders YM, Carballo D, Koskinas KC, Bäck M, et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J 2021;42:3227–3337. [DOI] [PubMed] [Google Scholar]
- 6. Dutta P, Courties G, Wei Y, Leuschner F, Gorbatov R, Robbins C, et al. Myocardial infarction accelerates atherosclerosis. Nature 2012;487:325–329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Cremer S, Schloss M, Vinegoni C, Brody F, Zhang S, Rohde D, et al. Diminished reactive hematopoiesis and cardiac inflammation in a mouse model of recurrent myocardial infarction. J Am Coll Cardiol 2020;75:901–915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Courties G, Herisson F, Sager HB, Heidt T, Ye Y, Wei Y, et al. Ischemic stroke activates hematopoietic bone marrow stem cells. Circ Res 2015;116:407–417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Courties G, Frodermann V, Honold L, Zheng Y, Herisson F, Schloss MJ, et al. Glucocorticoids regulate bone marrow B lymphopoiesis after stroke. Circ Res 2019;124:1372–1385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Tolani S, Pagler TA, Murphy AJ, Bochem AE, Abramowicz S, Welch C, et al. Hypercholesterolemia and reduced HDL-C promote hematopoietic stem cell proliferation and monocytosis: studies in mice and FH children. Atherosclerosis 2013;229:79–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Yvan-Charvet L, Pagler T, Gautier EL, Avagyan S, Siry RL, Han S, et al. ATP-binding cassette transporters and HDL suppress hematopoietic stem cell proliferation. Science 2010;328:1689–1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Nagareddy PR, Kraakman M, Masters SL, Stirzaker RA, Gorman DJ, Grant RW, et al. Adipose tissue macrophages promote myelopoiesis and monocytosis in obesity. Cell Metab 2014;19:821–835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Singer K, DelProposto J, Lee Morris D, Zamarron B, Mergian T, Maley N, et al. Diet-induced obesity promotes myelopoiesis in hematopoietic stem cells. Mol Metab 2014;3:664–675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Nagareddy PR, Murphy AJ, Stirzaker RA, Hu Y, Yu S, Miller RG, et al. Hyperglycemia promotes myelopoiesis and impairs the resolution of atherosclerosis. Cell Metab 2013;17:695–708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Hoyer FF, Zhang X, Coppin E, Vasamsetti SB, Modugu G, Schloss MJ, et al. Bone marrow endothelial cells regulate myelopoiesis in diabetes mellitus. Circulation 2020;142:244–258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Al-Sharea A, Lee MKS, Whillas A, Michell DL, Shihata WA, Nicholls AJ, et al. Chronic sympathetic driven hypertension promotes atherosclerosis by enhancing hematopoiesis. Haematologica 2019;104:456–467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Heidt T, Sager HB, Courties G, Dutta P, Iwamoto Y, Zaltsman A, et al. Chronic variable stress activates hematopoietic stem cells. Nat Med 2014;20:754–758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Heyde A, Rohde D, McAlpine CS, Zhang S, Hoyer FF, Gerold JM, et al. Increased stem cell proliferation in atherosclerosis accelerates clonal hematopoiesis. Cell 2021;184:1348–1361.e22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. van der Valk FM, Kuijk C, Verweij SL, Stiekema LCA, Kaiser Y, Zeerleder S, et al. Increased haematopoietic activity in patients with atherosclerosis. Eur Heart J 2017;38:425–432. [DOI] [PubMed] [Google Scholar]
- 20. Emami H, Singh P, Macnabb M, Vucic E, Lavender Z, Rudd JHF, et al. Splenic metabolic activity predicts risk of future cardiovascular events: demonstration of a cardiosplenic axis in humans. JACC Cardiovasc Imaging 2015;8:121–130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Kim EJ, Kim S, Kang DO, Seo HS. Metabolic activity of the spleen and bone marrow in patients with acute myocardial infarction evaluated by 18F-fluorodeoxyglucose positron emission tomograpic imaging. Circ Cardiovasc Imaging 2014;7:454–460. [DOI] [PubMed] [Google Scholar]
- 22. Tahara N, Kai H, Yamagishi Si, Mizoguchi M, Nakaura H, Ishibashi M, et al. Vascular inflammation evaluated by [18F]-fluorodeoxyglucose positron emission tomography is associated with the metabolic syndrome. J Am Coll Cardiol 2007;49:1533–1539. [DOI] [PubMed] [Google Scholar]
- 23. Nahrendorf M, Frantz S, Swirski FK, Mulder WJM, Randolph G, Ertl G, et al. Imaging systemic inflammatory networks in ischemic heart disease. J Am Coll Cardiol 2015;65:1583–1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Taqueti VR, Di Carli MF, Jerosch-Herold M, Sukhova GK, Murthy VL, Folco EJ, et al. Increased microvascularization and vessel permeability associate with active inflammation in human atheromata. Circ Cardiovasc Imaging 2014;7:920–929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Teague HL, Ahlman MA, Alavi A, Wagner DD, Lichtman AH, Nahrendorf M, et al. Unraveling vascular inflammation: from immunology to imaging. J Am Coll Cardiol 2017;70:1403–1412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Tawakol A, Ishai A, Takx RAP, Figueroa AL, Ali A, Kaiser Y, et al. Relation between resting amygdalar activity and cardiovascular events: a longitudinal and cohort study. Lancet 2017;389:834–845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Fernández-Friera L, Fuster V, López-Melgar B, Oliva B, Sánchez-González J, Macías A, et al. Vascular inflammation in subclinical atherosclerosis detected by hybrid PET/MRI. J Am Coll Cardiol 2019;73:1371–1382. [DOI] [PubMed] [Google Scholar]
- 28. Fernández-Ortiz A, Jiménez-Borreguero LJ, Peñalvo JL, Ordovás JM, Mocoroa A, Fernández-Friera L, et al. The progression and early detection of subclinical atherosclerosis (PESA) study: rationale and design. Am Heart J 2013;166:990–998. [DOI] [PubMed] [Google Scholar]
- 29. Ibanez B, Fernández-Ortiz A, Fernández-Friera L, García-Lunar I, Andrés V, Fuster V. Progression of Early Subclinical Atherosclerosis (PESA) study. J Am Coll Cardiol 2021;78:156–179. [DOI] [PubMed] [Google Scholar]
- 30. Fernández-Friera L, Peñalvo JL, Fernández-Ortiz A, Ibañez B, López-Melgar B, Laclaustra M, et al. Prevalence, vascular distribution, and multiterritorial extent of subclinical atherosclerosis in a middle-aged cohort the PESA (Progression of Early Subclinical Atherosclerosis) study. Circulation 2015;131:2104–2113. [DOI] [PubMed] [Google Scholar]
- 31. Arnett DK, Blumenthal RS, Albert MA, Buroker AB, Goldberger ZD, Hahn EJ, et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2019;74:e177–e232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Pisprasert V, Ingram KH, Lopez-Davila MF, Munoz AJ, Garvey WT. Limitations in the use of indices using glucose and insulin levels to predict insulin sensitivity: impact of race and gender and superiority of the indices derived from oral glucose tolerance test in African Americans. Diabetes Care 2013;36:845–853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. World Health Organization . Obesity: Preventing and Managing the Global Epidemic: Report of a WHO Consultation. Geneva: WHO; 1999. https://apps.who.int/iris/handle/10665/42330. [PubMed] [Google Scholar]
- 34. Swarup S, Goyal A, Grigorova Y, Zeltser R. Metabolic syndrome. StatPearls Publ, 2020. https://www.ncbi.nlm.nih.gov/books/NBK459248/. [PubMed] [Google Scholar]
- 35. Schulz V, Torres-Espallardo I, Renisch S, Hu Z, Ojha N, Börnert P, et al. Automatic, three-segment, MR-based attenuation correction for whole-body PET/MR data. Eur J Nucl Med Mol Imaging 2011;38:138–152. [DOI] [PubMed] [Google Scholar]
- 36. Mota-Cobian A, Alonso-Farto JC, Fernández-Friera L, Sánchez-González J, López-Melgar B, Jiménez-Borreguero LJ, et al. The effect of tissue-segmented attenuation maps on PET quantification with a special focus on large arteries. Rev Esp Med Nucl Imagen Mol (Engl Ed) 2018;37:94–102. [DOI] [PubMed] [Google Scholar]
- 37. Bucerius J, Hyafil F, Verberne HJ, Slart RHJA, Lindner O, Sciagra R, et al. Position paper of the cardiovascular committee of the European Association of Nuclear Medicine (EANM) on PET imaging of atherosclerosis. Eur J Nucl Med Mol Imaging 2016;43:780–792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Abdelbaky A, Corsini E, Figueroa AL, Fontanez S, Subramanian S, Ferencik M, et al. Focal arterial inflammation precedes subsequent calcification in the same location. Circ Cardiovasc Imaging 2013;6:747–754. [DOI] [PubMed] [Google Scholar]
- 39. Ye YX, Calcagno C, Binderup T, Courties G, Keliher EJ, Wojtkiewicz GR, et al. Imaging macrophage and hematopoietic progenitor proliferation in atherosclerosis. Circ Res 2015;117:835–845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Nahrendorf M, Swirski FK. Lifestyle effects on hematopoiesis and atherosclerosis. Circ Res 2015;116:884–894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Paredes S, Fonseca L, Ribeiro L, Ramos H, Oliveira JC, Palma I. Novel and traditional lipid profiles in metabolic syndrome reveal a high atherogenicity. Sci Rep 2019;9:11792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Ginsberg HN, Zhang YL, Hernandez-Ono A. Metabolic syndrome: focus on dyslipidemia. Obesity (Silver Spring) 2006;14:41S–49S. [DOI] [PubMed] [Google Scholar]
- 43. Fayad ZA, Mani V, Woodward M, Kallend D, Abt M, Burgess T, et al. Safety and efficacy of dalcetrapib on atherosclerotic disease using novel non-invasive multimodality imaging (dal-PLAQUE): a randomised clinical trial. Lancet 2011;378:1547–1559. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.