Abstract
Purpose
Psoriasis is an immune-mediated chronic inflammatory disease. Metabolic syndrome (MetS) is characterized by central obesity, hypertension, dyslipidemia, diabetes and insulin resistance (IR). Increasing evidence indicates that psoriasis is associated with MetS. This study aimed to explore some metabolite indexes which could evaluate the severity or predict the risk of psoriasis patients associated with MetS.
Patients and methods
It was a case-control study conducted in Beijing Hospital of Traditional Chinese Medicine. Sixty healthy volunteers (HC), 100 patients with psoriasis (Ps), 100 patients with MetS (MetS) and 80 patients with both psoriasis and MetS (Ps+MetS) were entered between January 2016 and December 2018. Blood samples were taken after at least 12 hours fasting and the contents of trimethylamine N-oxide (TMAO), carnitine, choline and betaine in serum were measured by Liquid Chromatography Mass Spectrometry (LC-MS/MS). Besides, the serum levels of low-density lipoprotein (LDL), high-density lipoprotein (HDL), cholesterol (CHO), triglyceride (TG), blood glucose (BG), creatinine (Cr), urea nitrogen (BUN), uric acid (UA) were determined.
Results
The non-healthy groups had different degrees of dyslipidemia, Ps-MetS> Ps >MetS. Compared with HC, the Ps had a higher level of TG; The MetS had the lowest level of HDL; The Ps+Mets had the highest level of TG and CHO. The Ps and Ps+MetS both had high level of UA, but there was no difference between the two groups. As for intestinal metabolites, the Ps had significant differences in TMAO, carnitine, and betaine in comparison with HC. The MetS had the highest level of TMAO. There was positive correlation between PASI and TMAO and betaine.
Conclusions
TMAO and betaine could serve as indexes reflecting the severity of psoriasis. TG, CHO, LDL and UA could serve as risk factors of MetS in psoriatic patients.
Keywords: psoriasis, metabolic syndrome, TMAO
Introduction
Psoriasis is a chronic immune-mediated inflammatory skin disorder characterized by hyperkeratosis or dyskeratosis of keratinocytes, dilation of dermal papillary blood vessels and inflammatory cell infiltration. The global psoriasis prevalence rate is around 2–3% of the world population.1 At present, it is widely accepted that psoriasis is not solely a dermatological disease but a multi-system inflammatory disorder with multiple associated comorbidities, such as cardiovascular diseases,2,3 respiratory diseases,4,5 oral diseases,6,7 gastrointestinal diseases,8 chronic kidney disease, anxiety, depression, dementia, etc.9 Among all the comorbidities, it seemed that metabolic syndrome (MetS) was more prevalent. MetS is a cluster of metabolic abnormalities including hypertension, obesity, diabetes mellitus, etc.10–12 A meta-analysis demonstrated that psoriasis and MetS are intimately connected.13 Patients with psoriasis have a significantly higher risk of developing MetS than the general population.14 The other way around, MetS may prolong the course of psoriasis patients and make it more difficult for treatment.15 In addition, MetS could cause cardiovascular disease, which is the primary cause leading to the death of psoriasis patients.16–18 It seemed that there may be a shared mechanism between these two diseases and some biomarkers which could reflect the risk of MetS in psoriasis. For example, red cell distribution width (RDW) and mean platelet volume (MPV) could be employed as inexpensive, routinely obtained biomarkers in predicting myocardial infarction (MI), atrial fibrillation (AF), and chronic heart failure (CHF) in psoriasis and psoriatic arthritis.3 In addition, resistin was reported to be a link between psoriasis and increased cardiovascular risk.2 Therefore, it would be promising to explore such specific biomarkers.
In recent years, host-diet-microbiome interactions, playing a significant role in MetS, has become a hot research topic in psoriasis. Trimethylamine-N-oxide (TMAO) has attracted increasing interests among the gut-derived metabolic substances. Several dietary precursors of TMAO, including betaine, choline, phosphatidylcholine and L-carnitine,19 can be metabolized by specific gut microbiota to trimethylamine (TMA), converted into TMAO by hepatic flavin-containing monooxygenases (FMOs), particularly FMO3, and excreted via the kidneys from the circulation.20 TMAO has been considered as the early predictor of MetS.20 The high serum level of TMAO not only cause hypertension, dyslipidemia, renal failure, diabetes, but aggravate the formation and deterioration of coronary atherosclerotic plaques, eventually leading to an increase in the risk of serious cardiovascular events, such as myocardial infarction, stroke and heart failure.21
To figure out whether the TMAO could be the early predictors of psoriasis complicated with MetS, or even serve as an efficacy predictor, a case-control study was conducted. In this study, we aimed to investigate the serum level of TMAO, betaine, carnitine and choline in healthy people, patients with psoriasis, patients with MetS, and patients with both psoriasis and MetS. In addition, we investigated the correlations of TMAO with psoriasis area and severity index (PASI) and hypothesized that TMAO could serve as a biological marker of early metabolic derangement in subjects with psoriasis.
Materials and Methods
Study Population
This is a case-control study conducted in Beijing Hospital of Traditional Chinese Medicine, Capital Medical University from January 2016 to December 2018. According to diagnostic criteria for psoriasis vulgaris22 and MetS,23 60 healthy volunteers from physical examination center as healthy control (HC), 100 patients with psoriasis (Ps) and 80 patients with both psoriasis and MetS (Ps + MetS) from the department of dermatology, 100 patients with MetS (MetS) from department of ulcerative vascular surgery, including both in- and out-patients were entered the study. All subjects were interviewed by one dermatologist and underwent a thorough history and physical examination. Sociodemographic data including sex, age, educational status and marital status were recorded for each subject.
Serological Assays
A 5 ml blood sample was taken at least 12 hours fasting and then was centrifuged to separate serum, and finally stored at −80 °C for subsequent analysis. The levels of TMAO and three related metabolites (betaine, choline and carnitine) were determined by Liquid Chromatography-Electrospray Ionization-Tandem Mass Spectrometry (LC-ESI-MS/MS). Proteins were precipitated with methanol (serum: methanol, 1:4, v/v); samples were vortex-mixed for 2 min, centrifuged at 12,000 g for 10 min at 4 °C; then supernatants were collected and used for analysis with a 5500 Q-TRAP hybrid/triple quadrupole linear ion trap mass spectrometer (AB Sciex, Framingham, MA, USA) with electrospray ionization (ESI) in positive mode.
In addition, low-density lipoprotein (LDL), high-density lipoprotein (HDL), cholesterol (CHO), triglyceride (TG), blood glucose (BG), creatinine (Cr), urea nitrogen (BUN), and uric acid (UA) in serum were measured using an automatic biochemical analyzer (Roche P800, Switzerland).
Scoring Criteria of PASI
The PASI score was used to evaluate the severity of psoriasis-like skin lesion, which was calculated as follows:24
PASI (head) = 0.1 (erythema + infiltration + desquamation) × lesion area;
PASI (upper limb) = 0.2 (erythema + infiltration + desquamation) × lesion area;
PASI (trunk) = 0.3 (erythema + infiltration + desquamation) × lesion area;
PASI (lower limb) = 0.4 (erythema + infiltration + desquamation) × lesion area;
PASI total score= PASI (head) + PASI (upper limb) + PASI (lower limb) + PASI (trunk).
The severity of each symptom was assessed comparing with reference pictures. The specific scoring details are shown in Table 1.
Table 1.
Scoring Criteria of PASI
| Symptoms | 0 | 1 | 2 | 3 | 4 |
|---|---|---|---|---|---|
| Erythema | None | Slightly reddish | Red, not deep | Markedly reddened | Extremely reddened |
| Scales | None | Very fine scales covering<50% | Fine scales covering>50% | Coarse scales covering>50% | Very coarse scales covering>50% |
| Thickness | None | Mild 0.25 mm | Moderate 0.5 mm | Severe 1 mm | Very severe 1.25 mm |
| Area | 0=None | 1=1%-9% | 2=10%-29% | 3=30–49% | |
| 4=50–69% | 5=70–89% | 6=90–100% |
Note: PASI adapted from: Fredriksson T, Pettersson U. Severe psoriasis – oral therapy with a new retinoid. Dermatologica. 1978;157(4):238-244.46
Abbreviation: PASI, Psoriasis Area and Severity Index.
Ethical Approval
The work has been carried out in accordance with the Code of Ethics of the World Medical Association for experiments in humans and approved by Research Ethical Committee of Beijing Hospital of Traditional Chinese Medicine, Capital Medical University (2016BL-006-01). This study was registered in Chinese Clinical Trial Registry (ChiCTR-INR-16007941). The process and purpose of the whole study were explained to participants and informed consent was obtained.
In compliance with the informed consent signed with the patient, we will share all data related to this article, and hide the patients’ personal information completely.
Data Sharing Statement
No additional data are available. The data that supports the findings of this article are available by email at any time. The data is available from in the manuscript can provided by Xinwei Guo and Yeping Qin.
Statistical Analysis
All the results were performed by IBM SPSS version 22.0 for Windows (SPSS, Inc., Chicago, IL, USA). Data were presented in the form of means (M) ± standard deviations (SD). Non-parametric tests were applied for further analysis for the data doesn’t have a normal distribution. Differences across groups were evaluated by one-factor analysis of variance (ANOVA) and differences between two means were examined using the Mann–Whitney U test (Mann–Whitney test). Pearson’s correlation coefficient (Pearson’s r) was calculated to examine the linear correlation between PASI scores and intestinal metabolites (TMAO, betaine, choline, carnitine). A P-value of < 0.05 was considered statistically significant.
Results
Clinical Characteristics of the Study Population
The general data of study subjects in four groups are summarized in Table 2. The mean age of four groups was 47 ± 14.75, 41.33 ± 12.51, 61.19 ± 12.51 and 43.65 ± 14.52 separately. Except for the MetS, no significant difference was observed for age. Most of the patients with MetS were from department ulcerative vascular surgery and had a history of type 2 diabetes mellitus (T2DM), because abnormal lipid metabolism and abnormal glucose metabolism are present in type 2 diabetes mellitus (T2DM).
Table 2.
Clinical Characteristics of the Study Population
| Characteristics | HC n=60 |
Ps n=100 |
MetS n=100 |
Ps+MetS n=80 |
|---|---|---|---|---|
| Gender | ||||
| Male (%) | 31(51.7) | 68(68) | 67(67) | 58(72.6) |
| Female (%) | 29(48.3) | 32(32) | 33(33) | 22(27.4) |
| Age (M ± SD) | 47 ± 14.75 | 41.33 ± 12.51 | 61.19 ± 12.51 | 43.65 ± 14.52 |
| Biochemical index (M ± SD) | ||||
| LDL (mmol/L) | 2.45 ± 0.66 | 2.73 ± 0.73 | 2.53 ± 0.89 | 2.89 ± 1.23* |
| HDL (mmol/L) | 1.25 ± 0.30 | 1.17 ± 0.30 | 1.11 ± 0.28** | 1.17 ± 0.31 |
| CHO (mmol/L) | 4.23 ± 0.81 | 4.45 ± 0.93 | 4.21 ± 1.10 | 4.69 ± 0.99* |
| TG (mmol/L) | 1.18 ± 0.47 | 1.60 ± 1.10** | 1.51 ± 0.97 | 2.25 ± 1.48***### |
| BG (mmol/L) | 4.94 ± 0.75 | 5.05 ± 0.79 | 7.05 ± 2.27***### | 5.57 ± 1.89* |
| Cr (μmol/L) | 61.35±13.67 | 65.32 ± 15.81 | 78.56 ± 25.6***### | 65.11 ± 13.75 |
| UA (μmol/L) | 324.85±99.314.00 | 391.76±102.63*** | 349.98±114.98## | 387.42±125.43** |
| BUN(mmol/L) | ± 1.09 | 4.51 ± 1.43 | 6.96 ± 2.37***### | 4.04 ± 1.31 |
Notes: *P< 0.05, **P< 0.01, ***P< 0.01, compared to HC. ##P< 0.01, ###P< 0.01, compared to Ps.
Abbreviations: HC, healthy volunteers; Ps, psoriasis; MetS, metabolic syndrome; Ps+MetS, psoriasis with metabolic syndrome.
Comparisons of biochemical indices including lipid metabolic index, blood glucose metabolism index and kidney function index among groups were presented in Table 2. In terms of lipid metabolism, several relevant biochemical indicators, including TG, CHO, HDL, and LDL were measured. Compared with HC, the Ps had higher level of TG (P< 0.01); The MetS showed lower level of HDL (P< 0.01); And the Ps+MetS presented higher level of TG (P< 0.001), CHO (P< 0.05) and LDL (P< 0.01). Pairwise comparisons revealed that statistical differences across three non-healthy groups. The TG (P< 0.001), LDL (P< 0.05) and CHO (P< 0.01) showed highest level in Ps+MetS. Although the means of four lipid metabolism indexes in Ps were higher than MetS, no differences were observed between them. As for glycemic metabolism, the MetS had the highest BG level (P< 0.001), and significant difference was also observed between HC and Ps+MetS (P< 0.05). Beyond the above-mentioned, several parameters, including Cr, BUN, UA about kidney function were also determined. Compared with HC, the Ps and Ps+MetS both had higher level of UA (P< 0.01); The MetS showed higher level of BUN (P< 0.001) and Cr (P< 0.001). Among the three non-healthy groups, the MetS presented the highest level of Cr and BUN (P< 0.001). While for the UA, the Ps had a higher level than MetS. And there were no statistical differences between Ps and Ps+MetS.
The Levels of Serum Metabolites and Correlation Between the Metabolites and PASI Score
The statistical comparisons of intestine metabolites in the four groups were also carried out in Table 3. Compared with healthy controls, TMAO levels were significantly higher in Ps (P< 0.001) and MetS (P< 0.001); Although there was a little increasing tendency in Ps+MetS, no difference was observed. For the other three related metabolites, pairwise group comparisons revealed significant findings. In comparison with healthy, the lower carnitine level (P < 0.001) and higher betaine level (P< 0.001) were measured in Ps. Compared with psoriatic patients, the Ps+MetS presented higher level of carnitine (P< 0.001), lower level of betaine (P< 0.001) and choline (P< 0.001), and the MetS had lower level of betaine (P< 0.001). Besides, noticeable statistical differences were observed between MetS and Ps+MetS in levels of these three metabolites, namely carnitine (P< 0.001), betaine (P< 0.001), and choline (P< 0.001).
Table 3.
Intestinal Metabolites of the Study Population
| Metabolites (ng/ml) | HC (M ± SD) | Ps (M ± SD) | MetS (M ± SD) | Ps + MetS (M ± SD) |
|---|---|---|---|---|
| TMAO | 1.98 ± 0.91 | 6.27 ± 2.92*** | 10.25 ± 5.49### | 3.10 ± 1.83### |
| Carnitine | 50.18 ± 11.32 | 44.20 ± 7.93*** | 42.52 ± 11.46 | 51.32 ± 9.45### |
| Betaine | 16.27 ± 6.78 | 55.94 ± 15.83*** | 40.64 ± 15.10### | 32.34 ± 7.50### |
| Choline | 8.99 ± 2.27 | 10.25 ± 2.14 | 13.20 ± 4.74### | 8.50 ± 2.79### |
Notes: ***P< 0.01, compared to HC. ###P< 0.01, compared to Ps.
Abbreviation: TMAO, trimethylamine N-oxide.
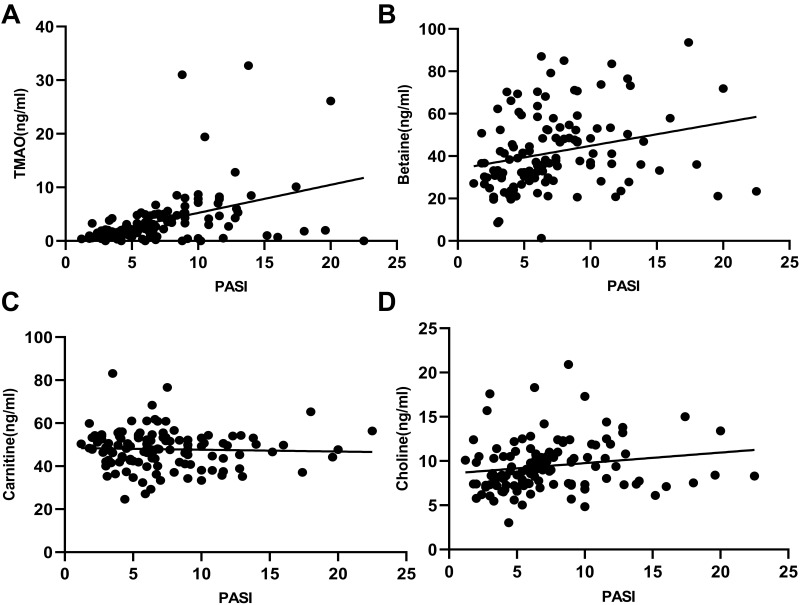
Further, the correlation between intestine metabolites and PASI score was analyzed in Figure 1. Based on the statistical results, it was found that TMAO (P< 0.0001), betaine (P< 0.01) and choline (P < 0.05) were positively correlated with PASI score. Moreover, there was no correlation between carnitine and PASI score (P> 0.05).
Figure 1.
Correlation between several intestinal metabolites and PASI score. (A) TMAO had a positive correlation with PASI score (r = 0.4237, P < 0.001); (B) Betaine had a positive correlation with PASI score (r = 0.2533, P = 0.0049); (C) Carnitine had no correlation with PASI score (r = −0.3852, P = 0.6736); (D) Choline had a negative correlation with PASI score (r = −0.1799, P = 0.0473).
Discussion
A strong relationship between psoriasis and MetS has been reported in the literature.25,26 Naturally, diets that have direct relationships with MetS became one of the hot topics. It was reported specific nutrients in food had an influence on inflammatory cell signaling transcriptional factors by affecting endogenous miRNA synthesis.27 Furthermore, it has been proved that intermittent fasting has beneficial effects on psoriatic arthritis, regardless of the implicated drug therapy.28,29 Meanwhile, different dietary structures do have impacts on psoriasis patients undergoing phototherapy.30 Therefore, reasonable diet planning is of great hope to controlling psoriasis and MetS.
Although the exact underlying mechanism of MetS in psoriasis has not been fully explained, it was suggested that overlap in the pathological mechanism between Ps and MetS. For instance, the psoriasis-associated inflammatory factors (IL-1β, IL6, TNF-α) could alter the lipoprotein composition, enhance adhesion molecule expression and aggravate cholesterol crystallization formation, finally leading to atherosclerosis.31 In return, a large number of adipokines produced by adipose tissue in MetS have a contributive role in the initiation and maintenance of psoriatic inflammatory. In clinical treatment, the psoriatic patients with MetS have a longer duration, increased costs and higher mortality rate.32 Therefore, if some metabolic risks could be detected in the early stages, it would provide an opportunity window for effective intervention.
According to the previous study, we chose several metabolic indicators, such as blood glucose, TG, HDL, LDL and CHO, considered as MetS risk factors.33 Meanwhile, there is evidence that MetS has a close association with kidney malfunctions.34 So, we also detected the serum level of UA, Cr and BUN. Besides the mentioned above, TMAO and three of its precursors, intestinal metabolites, were chosen, for TMAO is the early predictor of MetS.20 This study was performed to find out the association between psoriasis and MetS risk factors and figure out some early predictors of psoriasis with concomitant MetS, or even serve as an efficacy predictor.
We found that three non-healthy groups had different degrees of dyslipidemia based on the statistical results The severity of dyslipidemia seemed to meet a gradually increasing trend, namely Ps+ MetS> Ps >MetS. CHO, TG, LDL and HDL are common indexes of blood lipid in the clinic. The increase of CHO, TG, LDL and the decrease of HDL component is a reflection of a lipid metabolism disturbance. Specifically, in comparison with the HC, the Ps had only one abnormal index, which was consistent with the study of Farshchian et al,35 while the Ps+MetS had three abnormal indexes (TG, LDL and CHO); Generally speaking, diabetic dyslipidemia is typically featured by high TG and low HDL levels.36 The MetS only had low HDL in the study due to TG is closely related to diet. Because the MetS patients we included strictly abide by the hospitalized diet. Therefore, we have observed dyslipidemia in patients with psoriasis, which is consistent with previous reports.37
As for the sugar metabolism, the MetS and Ps-MetS had abnormal BG levels. It was no doubt that the MetS had the highest level for the majority of the MetS were diagnosed with T2DM. The Ps-MetS had a relatively higher BG level in comparison with HC. However no significant difference was found between Ps and HC, the mean BG of Ps had an increasing trend.
As far as kidney function, although the high UA level of Ps and Ps-MetS indicated abnormalities in purine metabolism, there was no difference between Ps and Ps-MetS. While in the study of Yilmaz et al,38 they determined that UA levels were higher in patients with Ps and MetS than in patients with Ps alone. It is possible that the different results were due to the severity of psoriasis, because Zhang Ying’s meta-analysis presented that moderate to severe psoriasis were more likely to have high level UA.39
On the question of intestinal metabolites, the results indicated that Ps and Ps+MetS both had a significantly high level of metabolites except carnitine compared with HC, and the serum metabolites concentrations in Ps were higher than that in Ps+MetS. Meanwhile, the severity of psoriasis had a positive correlation with intestinal metabolites, namely TMAO, choline and betaine. It has been reported that TMAO is the early predictor of MetS, particularly in cardiovascular disease. Betaine and choline are the dietary precursors of TMAO. On the one hand, TMAO caused vascular aging and vascular cell senescence and through oxidative stress;40 On the other hand, TMAO increased thrombosis risk through multiple mechanisms, including changing lipid and hormone homeostasis, promoting platelet hyperreactivity, regulating cholesterol and sterol metabolism, and inducing endothelial dysfunction by activating NLRP3 inflammatory bodies.41,42 The treatment of MetS in psoriasis can improve the condition of psoriasis, and it has been proved that weight loss interventions were effective in reducing the psoriatic arthritis severity and improving skin lesions.43 Decreasing the TMAO level has become an effective means to prevent or treat atherosclerosis by adjusting dietary substrate, regulating microbial metabolism and inhibiting FMOs.44,45 Meanwhile, psoriatic patients are at higher risk of cardiovascular disease. Our study reveals that TMAO and its related dietary precursors had positive correlations with psoriasis. It suggests that TMAO could be used as a marker to prevent the occurrence of metabolic syndrome or worsening cardiovascular disease in psoriatic patients.
According to this study’s findings, lipid metabolism indexes were abnormal in psoriatic patients, and the higher levels of the lipoprotein, the higher the risk of accompanying MetS. The same applies to UA. In addition, TMAO, betaine and choline could serve as indexes reflecting the severity of psoriasis.
Conclusion
The intestinal metabolites (TMAO and betaine) in the patient’s serum are positively correlated with psoriasis, suggesting that psoriasis and metabolic syndrome have a common pathological mechanism, which can predict the risk of MetS in psoriasis. However, due to lack of sufficient human resources and good invest, our research sample was relatively small and the sampling range was narrow, so a large-scale clinical study may be performed. In addition, more efforts should be made to verify this conclusion by performing in vitro and vivo experiments.
Acknowledgments
We would like to extend my heartful thanks to the patients and researchers involved in this study.
Funding Statement
This clinical trial is part of the projects funded and supported by the National Natural Science Foundation of China (grant numbers 81603630 and 81873119), 2019 City of Millions project (Xuying Xu).
Disclosure
The authors declare that they have no conflicts for this work.
References
- 1.Sewerin P, Brinks R, Schneider M, Haase I, Vordenbäumen S. Prevalence and incidence of psoriasis and psoriatic arthritis. Ann Rheum Dis. 2019;78(2):286–287. doi: 10.1136/annrheumdis-2018-214065 [DOI] [PubMed] [Google Scholar]
- 2.Seth D, Ehlert A, Golden J, et al. Interaction of resistin and systolic blood pressure in psoriasis severity. J Invest Dermatol. 2020;140(6):1279–1282. doi: 10.1016/j.jid.2019.07.727 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Conic R, Damiani G, Schrom K, et al. Psoriasis and psoriatic arthritis cardiovascular disease endotypes identified by red blood cell distribution width and mean platelet volume. J clin med. 2020;9(1):186. doi: 10.3390/jcm9010186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Damiani G, Pacifico A, Rizzi M, et al. Patients with psoriatic arthritis have higher levels of FeNO than those with only psoriasis, which may reflect a higher prevalence of a subclinical respiratory involvement. Clin Rheumatol. 2020;39(10):2981–2988. doi: 10.1007/s10067-020-05050-2 [DOI] [PubMed] [Google Scholar]
- 5.Santus P, Rizzi M, Radovanovic D, et al. Psoriasis and respiratory comorbidities: the added value of fraction of exhaled nitric oxide as a new method to detect, evaluate, and monitor psoriatic systemic involvement and therapeutic efficacy. Biomed Res Int. 2018;2018:3140682. doi: 10.1155/2018/3140682 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Damiani G, Gironi L, Pacifico A, et al. Cutaneous and oral comorbidities in patients with geographic tongue: a multicenter multidisciplinary cross-sectional observational study. J Biol Regul Homeost Agents. 2021;35:339–344. doi: 10.23812/21-2supp1-33 [DOI] [PubMed] [Google Scholar]
- 7.Damiani G, Pacifico A, Malagoli P, et al. Geographic tongue clinical response in moderate-to-severe psoriatic patients undergoing secukinumab: a real-life, multicenter retrospective observational study. J Biol Regul Homeost Agents. 2021;35:331–337. doi: 10.23812/21-2supp1-32 [DOI] [PubMed] [Google Scholar]
- 8.Damiani G, Franchi C, Pigatto P, et al. Outcomes assessment of hepatitis C virus-positive psoriatic patients treated using pegylated interferon in combination with ribavirin compared to new direct-acting antiviral agents. World J Hepatol. 2018;10(2):329–336. doi: 10.4254/wjh.v10.i2.329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhou J, Ge H, Zhu M, et al. Neck circumference as an independent predictive contributor to cardio-metabolic syndrome. Cardiovasc Diabetol. 2013;12:76. doi: 10.1186/1475-2840-12-76 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Huang K, Yeh C, Wu S, et al.. Risk of dementia among individuals with psoriasis: a nationwide population-based cohort study in Taiwan. J Clin Psychiatry. 2019;80(3). doi: 10.4088/JCP.18m12462 [DOI] [PubMed] [Google Scholar]
- 11.Hu S, Chen G, Tu H. Epidemiology of depression in patients with psoriasis: a nationwide population-based cross-sectional study. Acta Derm Venereol. 2019;99(6):530–538. doi: 10.2340/00015555-3145 [DOI] [PubMed] [Google Scholar]
- 12.Lønnberg A, Skov L, Skytthe A, Kyvik K, Pedersen O, Thomsen S. Association of psoriasis with the risk for Type 2 diabetes mellitus and obesity. JAMA dermatol. 2016;152(7):761–767. doi: 10.1001/jamadermatol.2015.6262 [DOI] [PubMed] [Google Scholar]
- 13.Singh S, Young P, Armstrong A, Taheri S. An update on psoriasis and metabolic syndrome: a meta-analysis of observational studies. PloS one. 2017;12(7):e0181039. doi: 10.1371/journal.pone.0181039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Snekvik I, Nilsen T, Romundstad P, Saunes M. Psoriasis and cardiovascular disease risk factors: the HUNT Study, Norway. JEADV. 2018;32(5):776–782. doi: 10.1111/jdv.14835 [DOI] [PubMed] [Google Scholar]
- 15.Shih C, Huang C, Wang K, et al. Oxidized low-density lipoprotein-deteriorated psoriasis is associated with the upregulation of Lox-1 receptor and Il-23 expression in vivo and in vitro. Int J Mol Sci. 2018;19(9):2610. doi: 10.3390/ijms19092610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Takeshita J, Grewal S, Langan S, et al. Psoriasis and comorbid diseases: epidemiology. J Am Acad Dermatol. 2017;76(3):377–390. doi: 10.1016/j.jaad.2016.07.064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Furue M, Tsuji G, Chiba T, Kadono T. Cardiovascular and metabolic diseases comorbid with psoriasis: beyond the skin. Internal Med. 2017;56(13):1613–1619. doi: 10.2169/internalmedicine.56.8209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Arnold K, Treister A, Lio P, Alenghat F. Association of atherosclerosis prevalence with age, race, and traditional risk factors in patients with psoriasis. JAMA dermatol. 2019;155(5):622–623. doi: 10.1001/jamadermatol.2018.5462 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Burton K, Krüger R, Scherz V, et al. NTrimethylamine–oxide postprandial response in plasma and urine is lower after fermented compared to non-fermented dairy consumption in healthy adults. Nutrients. 2020;12(1):234. doi: 10.3390/nu12010234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Barrea L, Annunziata G, Muscogiuri G, et al. Trimethylamine-N-oxide (TMAO) as novel potential biomarker of early predictors of metabolic syndrome. Nutrients. 2018;10(12):1971. doi: 10.3390/nu10121971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Al-Rubaye H, Perfetti G, Kaski J. The role of microbiota in cardiovascular risk: focus on trimethylamine oxide. Curr Probl Cardiol. 2019;44(6):182–916. doi: 10.1016/j.cpcardiol.2018.06.005 [DOI] [PubMed] [Google Scholar]
- 22.Burden-Teh E, Phillips R, Thomas K, Ratib S, Grindlay D, Murphy R. A systematicreview of diagnostic criteria for psoriasis in adults and children: evidence from studies with a primary aim to develop or validate diagnostic criteria. Br J Dermatol. 2018;178(5):1035–1043. doi: 10.1111/bjd.16104 [DOI] [PubMed] [Google Scholar]
- 23.Grundy S. Metabolic syndrome scientific statement by the American Heart Association and the national heart, lung, and blood institute. Arterioscler Thromb Vasc Biol. 2005;25(11):2243–2244. doi: 10.1161/01.ATV.0000189155.75833.c7 [DOI] [PubMed] [Google Scholar]
- 24.Dong W, Nie X, Wang J, et al. Randomised, double-blind, multicentre, phase I/II dose escalation and expansion trial of GR1501 in patients with plaque psoriasis: study protocol. BMJ open. 2020;10(11):e039067. doi: 10.1136/bmjopen-2020-039067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee E, Han J, Bang C, et al. Risk of end-stage renal disease in psoriatic patients: real-world data from a nationwide population-based cohort study. Sci Rep. 2019;9(1):16581. doi: 10.1038/s41598-019-53017-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fan J, Solomon D, López G, et al. Catastrophic stroke burden in a patient with uncontrolled psoriasis and psoriatic arthritis: a case report. BMC Neurol. 2020;20(1):106. doi: 10.1186/s12883-020-01681-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kocic H, Damiani G, Stamenkovic B, et al. Dietary compounds as potential modulators of microRNA expression in psoriasis. Ther Adv Chronic Dis. 2019;10:2040622319864805. doi: 10.1177/2040622319864805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Adawi M, Damiani G, Bragazzi N, et al. The impact of intermittent fasting (Ramadan Fasting) on psoriatic arthritis disease activity, enthesitis, and dactylitis: a multicentre study. Nutrients. 2019;11(3):601. doi: 10.3390/nu11030601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Damiani G, Watad A, Bridgewood C, et al. The impact of ramadan fasting on the reduction of PASI score, in moderate-to-severe psoriatic patients: a real-life multicenter study. Nutrients. 2019;11(2):277. doi: 10.3390/nu11020277 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pacifico A, Conic R, Cristaudo A, et al. Diet-related phototoxic reactions in psoriatic patients undergoing phototherapy: results from a multicenter prospective study. Nutrients. 2021;13(9):2934. doi: 10.3390/nu13092934 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Baumer Y, Ng Q, Sanda G, et al.. Chronic skin inflammation accelerates macrophage cholesterol crystal formation and atherosclerosis. JCI Insight. 2018;3(1). doi: 10.1172/jci.insight.97179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gui X, Yu X, Jin H, Zuo Y, Wu C. Prevalence of metabolic syndrome in Chinese psoriasis patients: a hospital-based cross-sectional study. J Diabetes Investig. 2018;9(1):39–43. doi: 10.1111/jdi.12663 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Carrillo-Venzor M, Erives-Anchondo N, Moreno-González J, et al. Pro12Ala PPAR-γ2 and +294T/C PPAR-δ polymorphisms and association with metabolic traits in teenagers from Northern Mexico. Genes. 2020;11(7):776. doi: 10.3390/genes11070776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Townsend R, Anderson A, Chen J, et al. Metabolic syndrome, components, and cardiovascular disease prevalence in chronic kidney disease: findings from the Chronic Renal Insufficiency Cohort (CRIC) Study. Am J Nephrol. 2011;33(6):477–484. doi: 10.1159/000327618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Farshchian M, Ansar A, Sobhan M. Associations between cardiovascular risk factors and psoriasis in Iran. Clin CosmetInvestig Dermatol. 2015;8:437–442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hu Z, Ren L, Wang C, Liu B, Song G. Associations between apolipoprotein CIII concentrations and microalbuminuria in type 2 diabetes. Exp Ther Med. 2014;8(3):951–956. doi: 10.3892/etm.2014.1830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Nowowiejska J, Baran A, Flisiak I. Aberrations in lipid expression and metabolism in psoriasis. Int J Mol Sci. 2021;22(12):e45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yilmaz E, Tamer E, Artüz F, Külcüçakmak S, Köktürk F. Evaluation of serum uric acid levels in psoriasis vulgaris. Turkish j Med Sci. 2017;47(2):531–534. doi: 10.3906/sag-1512-5 [DOI] [PubMed] [Google Scholar]
- 39.Zhang Y, Liu L, Sun X, et al. Updated evidence of the association between elevated serum uric acid level and psoriasis. Front med. 2021;8:645550. doi: 10.3389/fmed.2021.645550 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ke Y, Li D, Zhao M, et al. Gut flora-dependent metabolite Trimethylamine-N-oxide accelerates endothelial cell senescence and vascular aging through oxidative stress. Free Radic Biol Med. 2018;116:88–100. doi: 10.1016/j.freeradbiomed.2018.01.007 [DOI] [PubMed] [Google Scholar]
- 41.Chen M, Zhu X, Ran L, Lang H, Yi L, Mi M. Trimethylamine-N-Oxide induces vascular inflammation by activating the NLRP3 inflammasome through the SIRT3-SOD2-mtROS signaling pathway. J Am Heart Assoc. 2017;6(9). doi: 10.1161/JAHA.117.006347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhu W, Gregory J, Org E, et al. Gut microbial metabolite TMAO enhances platelet hyperreactivity and thrombosis risk. Cell. 2016;165(1):111–124. doi: 10.1016/j.cell.2016.02.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Klingberg E, Bilberg A, Björkman S, et al. Weight loss improves disease activity in patients with psoriatic arthritis and obesity: an interventional study. Arthritis Res Ther. 2019;21(1):17. doi: 10.1186/s13075-019-1810-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Mohania D, Kansal V, Shah D, et al. Therapeutic effect of probiotic dahi on plasma, aortic, and hepatic lipid profile of hypercholesterolemic rats. J Cardiovasc Pharmacol Ther. 2013;18(5):490–497. doi: 10.1177/1074248413487431 [DOI] [PubMed] [Google Scholar]
- 45.Trøseid M, Ueland T, Hov J, et al. Microbiota-dependent metabolite trimethylamine-N-oxide is associated with disease severity and survival of patients with chronic heart failure. J Intern Med. 2015;277(6):717–726. doi: 10.1111/joim.12328 [DOI] [PubMed] [Google Scholar]
- 46.Fredriksson T, Pettersson U. Severe psoriasis – oral therapy with a new retinoid. Dermatologica. 1978;157(4):238–244. [DOI] [PubMed] [Google Scholar]