Abstract
Background
Duration of post‐vaccination protection against COVID‐19 in nursing home (NH) residents is a critical issue. The objective of this study was to estimate the duration of the IgG(S) response to the mRNA BNT162b2 vaccine in NH residents with (COV‐Yes) or without (COV‐No) history of SARS‐CoV‐2 infection.
Methods
A 574 COV‐Yes and COV‐No NH residents were included in 2 cohorts: Main (n = 115, median age 87 years) or Confirmatory (n = 459, median age 89 years). IgG(S) quantification was carried out at three different time points following the BNT162b2 vaccine: three (1st) and seven (2nd) months after the 2nd dose, and 1 month after the 3rd dose (3rd quantification) in the Main cohort, and twice (2nd and 3rd) in the Confirmatory cohort. The seroneutralization capacity according to COVID‐19 history was also measured in a subgroup of patients.
Results
Neutralization capacity was strongly correlated with IgG(S) levels (R 2:76%) without any difference between COV‐Yes and COV‐No groups for the same levels of IgG(S). After the 2nd dose, duration of the assumed robust protection (IgG(S) >264 BAU/ml) was two‐fold higher in the COV‐Yes vs. COV‐No group: 12.60 (10.69–14.44) versus 5.76 (3.91–8.64) months, with this advantage mainly due to the higher IgG(S) titers after the 2nd dose and secondary to a slower decay over time. After the 3rd dose, duration of robust protection was estimated at 11.87 (9.88–14.87) (COV‐Yes) and 8.95 (6.85–11.04) (COV‐No) months. These results were similar in both cohorts.
Conclusions and Relevance
In old subjects living in NH, history of SARS‐CoV‐2 infection provides a clear advantage in the magnitude and duration of high IgG(S) titers following the 2nd dose. Importantly, the 3rd dose induces a much more pronounced IgG(S) response than the 2nd dose in COV‐No subjects, the effect of which should be able to ensure a prolonged protection against severe forms of COVID‐19 in these subjects.
Keywords: nursing homes, SARS‐CoV‐2, vaccination
Key points
This study estimates the duration of the immune protection of the BNT162b2 vaccine in old subjects, with or without history of SARS‐Cov‐2 infection, by measuring the increase in IgG(S) antibodies following vaccination and their waning rates over time.
In this very old population, previous SARS‐Cov‐2 infection induces a more pronounced IgG(S) response to the 2nd dose of the vaccine and a less pronounced decay over time, both leading to a longer immune protection compared to the group without history of SARS‐Cov‐2 infection.
In subjects without history of SARS‐Cov‐2 infection, the 3rd dose, as compared to the 2nd dose of the Pfizer BioNtech vaccine, induces a much more pronounced IgG(S) response leading to a longer protection against severe forms of COVID‐19
Why does this paper matter?
This study provides an estimation of the duration of protection against severe forms of COVID‐19 following the 2nd and 3rd doses of the BNT162b2 (Pfizer BioNtech) vaccine in nursing homes residents, with or without history of natural SARS‐CoV‐2 infection.
INTRODUCTION
Mass vaccination of nursing home (NH) residents, the population most likely to develop severe forms of COVID‐19, has resulted in an impressive decrease in SARS‐CoV‐2 contaminations as well as a dramatic decrease in COVID‐19‐related mortality. 1 In France, NH residents received mRNA vaccines (mainly BNT162b2, Pfizer BioNtech), the vast majority of whom (81%) were fully vaccinated between January and June 2021, 2 while the 3rd dose was administered between October and December 2021. Clinical studies in NHs demonstrated that, after the second dose, this very old population was able to develop SARS‐CoV‐2 IgG(S) antibodies, thus obtaining protection against severe forms of COVID‐19, 3 , 4 and that previous infection by SARS‐CoV‐2 increased the immunogenicity of the vaccines. 4 This latter result has also been observed in younger adults. 5 , 6 , 7
One of the most critical issues is the duration of post‐vaccination protection in individuals with or without history of natural SARS‐CoV‐2 infection. 7 , 8 , 9 The waning in serum SARS‐CoV‐2 antibodies has raised pressing questions regarding long‐term immunity thus leading to the 3rd dose vaccination.
In the present study, we investigated the IgG(S) response to the BNT162b2 vaccine in NH residents with or without history of SARS‐CoV‐2 infection at three different time points: (a) approximately 3 months after the leading 2 doses of the vaccine, (b) 4 months later just before the booster (3rd) vaccine and (c) 1 month after the booster vaccine. In addition, a sero‐neutralization assay was performed for a subgroup of subjects and the results were analyzed according to their COVID‐19 status and IgG(S) levels.
The results of these analyses should enable modeling the decrease over time of IgG(S) SARS‐CoV‐2 antibodies and provide an estimate of the duration of protection of the 3rd dose of the mRNA vaccine against severe forms of COVID‐19 in this very old population.
MATERIAL AND METHODS
Participants
Two cohorts were formed (Main and Confirmatory) comprised of NH residents from the Nancy‐Lorraine region with (COV‐Yes) or without (COV‐No) history of prior SARS‐CoV‐2 infection. Residents belonging in the Main cohort were included between April 17, 2021 and September 21, 2021 in 5 NHs, whereas subjects belonging in the Confirmatory cohort were included between August 10, 2021 and December 15, 2021 in 12 NHs (Table S1). In the Main cohort, two IgG(S) quantifications were performed at mean 3 months (1st IgG(S) quantification) and 7 months (2nd IgG(S) quantification) following the 2nd vaccination. Indeed, to analyze the decrease in IgG(S) between the 1st and 2nd quantification, a minimum interval of 28 days was set between the 2nd vaccination and the 1st quantification, thus after the IgG(S) peak following the 2nd dose of the vaccine. In the Confirmatory cohort, a single IgG(S) quantification was performed 7 months after the 2nd vaccination (also defined as 2nd IgG(S) quantification). In both cohorts, a 3rd IgG(S) quantification was performed 1.5 months after the 3rd (booster) dose (Figure S1).
Age, sex, vaccination dates, SARS‐CoV‐2 infection dates, as well as IgG(S) quantification dates were recorded for all residents in both cohorts. In addition, for participants in the Main cohort, BMI, autonomy status, and number of medications were also collected.
This study was registered in ClinicalTrials.gov (NCT04964024) and received the approval of the Ethics Committee of the Nancy University Hospital (CHRU) (Comite d'Ethique CHRU de Nancy, decision no. 326, August 3rd, 2021).
Inclusion criteria for both cohorts
NH residents aged 65 and older, with or without prior SARS‐CoV‐2 infection. Previous infection was identified either by history of positive RT‐PCR or by anti‐nucleoprotein (N) IgG quantification (UNScience, COVID‐19 IgG/IgM Rapid Test Kit).
SARS‐CoV‐2 anti‐spike IgG(S) antibody quantification performed using the same quantification method 10 in the same laboratory.
Complete vaccination scheme, meaning two injections of the BNT162b2 mRNA vaccine with the second injection at least 28 days before the first antibody quantification.
Consent of the NH residents or their tutors for the use of the clinical and biological results for the aims of this study.
Method for IgG(S) quantification
Blood samples were centrifuged to collect serum, the latter of which was stored at −20°C. Anti‐spike IgGs were detected using the LIAISON® SARS‐CoV‐2 TrimericS IgG assay (Diasorin SA, France) on a Liaison XL Device (Diasorin SA, France), based on recombinant Trimeric Spike glycoprotein as capture antigen. 10 Quantitative results are expressed as Binding Antibody Units (BAU/ml) according to the WHO first International Standard (IS) for anti‐SARS‐CoV‐2 immunoglobulin (NIBSC code 20/136). The positive threshold was ≥33.8 BAU/ml. Samples with an antibody titer >2080 were diluted to 1:20 as recommended by the manufacturer in order to determine accurate IgG levels. For technical reasons, the samples of 14 patients of the Main cohort and 6 patients from the Confirmatory cohort were not further diluted and these subjects were considered to have the plateau value of 2080 BAU/ml.
Microneutralization assay
We aimed to compare the neutralization activity of COV‐No and COV‐Yes samples to equivalent levels of antibodies. Thus, a sub‐population of 39 NH residents was selected from the entire population according to IgG(S) levels following the 2nd vaccination (“low” < 650 BAU/ml vs. “high” > 2080 BAU/ml) and prior SARS‐CoV‐2 infection (COV‐Yes vs. COV‐No) to assess serum neutralization capacities. There was no difference in mean age or sex distribution between subjects who were included in the neutralization study and those who were not.
Four sub‐groups were studied: COV‐No/low IgG(S), n = 10, median 215 BAU/ml; COV‐Yes/low IgG(S), n = 10, median 217 BAU/ml; COV‐No/high IgG, n = 9, median 3270 BAU/ml; and COV‐Yes/high IgG, n = 10, median 4486 BAU/ml.
The SARS‐CoV‐2 B.1.617.2 (Delta) strain from a positive respiratory sample (Covi‐Lor collection, Nancy University Hospital, France) was cultured on Vero E6 cells. Sera positive for anti‐SARS‐CoV‐2 antibodies were diluted from 1/10 to 1/640 and incubated with live‐virus suspension for 2 h. Cells were inoculated with the final suspension. Each dilution was tested five times in each experiment and each sample was tested in two independent experiments. The cytopathic effect was read on day +6.
Negative controls consisted of uninfected cells while positive controls consisted of the virus incubated without sera and virus incubated with SARS‐CoV‐2–negative sera at a 1/10 ratio.
The samples were classified according to neutralization activity, which was considered positive if neutralization reached 50% (NT50) at the 1:40 dilution.
Statistical analyses
Descriptive IgG data are presented as medians (IQR) and as mean (SD) values or percentages for the other variables. Due to the absence of normal distribution of IgG(S) values, comparisons between the different groups were performed after logarithmic transformation. Comparisons were conducted with ANOVA tests. Age and time (Δ) between the last immune stimulation (COVID‐19 or vaccine) and antibody quantification were used for the adjusted models. Multiple regression analyses were also performed to test the role of clinical and demographic variables, time from immunization, and history of prior SARS‐CoV‐2 infection on IgG(S) levels. Pearson's correlation was used to study the association between IgG(S) values and time of immunization for each studied group. p < 0.05 was considered statistically significant.
In an additional analysis, NH residents were classified into four categories according to IgG(S) thresholds in BAU/ml: ≥34 (level of positivity), ≥264 (threshold associated with higher protection against severe COVID‐19 forms, 11 ≥2080 threshold highly predictive of strong neutralization response 12 Differences between COV‐No vs. COV‐Yes were compared using a χ 2 test.
Models used for the assessment of IgG decay with time: In both cohorts, the natural logarithm of the IgG rate follows a linear regression with steepness B, 1 meaning that the decay rate of the IgG is exponential. Hence, the proportion q30 of IgG that is lost during 1 month is q30 = 1 − exp(30 * B 1 ).
If the rate of IgG(S) decays exponentially by a proportion q30 each month, then if a quantity I(0) is measured at time t = 0, the immunity will be lost at a time t loss (in months) at which the IgG(S) rate equals I loss given by t loss = −log(I(0)/Iloss)/log(1 − q30).
A linear mixed model with random effect on subjects was used to examine IgG kinetics after the second dose of vaccine in the Main cohort. The dependent variable consisted of the IgG(S) level, which was log‐transformed. Fixed effect covariates included time after the second dose of vaccine in months as linear, SARS‐CoV‐2 status, and interaction between time and SARS‐CoV‐2 status. Given the low power of interaction tests, 13 , 14 a significance level of 0.10 was used for interaction p‐values. Similar analysis was performed in the Confirmatory cohort using a classical linear regression model.
RESULTS
Cohort characteristics
The Main cohort was comprised of 115 subjects (41 COV‐No and 75 COV‐Yes) whereas the Confirmatory cohort was comprised of 459 subjects (366 COV‐NO and 93 COV‐Yes) (Figure 1). Demographic characteristics and IgG(S) levels are presented in Table 1. BMI, autonomy status, and number of daily medications collected in the Main cohort were similar in the COV‐Yes and COV‐No groups.
FIGURE 1.
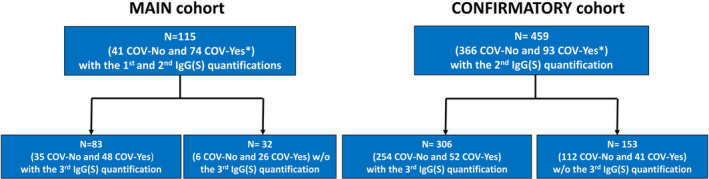
Study flowchart. *Among COV‐Yes subjects, 20 out of 74 in the Main cohort and 7 out of 93 in the Confirmatory cohort had SARS‐CoV‐2 infection a few days after the 2nd vaccine dose (February–March 2021). All other subjects had SARS‐CoV‐2 infection prior to the 1st vaccine dose.
TABLE 1.
Clinical data and IgG(S) levels in the Main and Confirmatory cohorts in the two subgroups of NH residents with (COV‐Yes) or without (COV‐No) history of prior SARS‐CoV‐2 infection
| Main cohort | Confirmation cohort | |||||||
|---|---|---|---|---|---|---|---|---|
| COV‐No | N | COV‐Yes | N | COV‐No | N | COV‐Yes | N | |
| Women (%) | 63% | 41 | 73% | 74 | 78% | 366 | 73% | 93 |
| Age (years) | 84 ± 9 | 41 | 84 ± 10 | 74 | 88 ± 8 | 366 | 87 ± 8 | 93 |
| BMI | 25.0 ± 7.0 | 40 | 25.6 ± 5.2 | 68 | — | — | ||
| GIR | 1.90 ± 0.63 | 40 | 1.90 ± 0.92 | 68 | — | — | ||
| Number of treatments | 6.93 ± 2.56 | 40 | 7.38 ± 3.49 | 68 | — | — | ||
| IgG(S) titers (BAU/ml) | ||||||||
| 1st IgG(S) median (IQR) | 621 (189–1741) | 41 | 2901*** a (1873–7805) | 74 | ||||
| 2nd IgG(S) median (IQR) | 92 (46–415) | 41 | 1555*** (776–2080) | 74 | 114 (38–322) | 366 | 1740*** (1095–2900) | 93 |
| 3rd IgG(S) median (IQR) | 3040 (1880–7970) | 35 | 4965(NS) (3070–9393) | 48 | 3795 (1748–7470) | 254 | 6675** (2503–14,550) | 52 |
| D Time (days) | ||||||||
| 1st IgG(S) b | 84 ± 22 | 41 | 101 ± 68 | 74 | ||||
| 2nd IgG(S) b | 210 ± 31 | 41 | 215 ± 38 | 74 | 212 ± 38 | 366 | 173 ± 41*** | 93 |
| 3rd IgG(S) c | 44 ± 8 | 35 | 42 ± 10 | 48 | 39 ± 10 | 254 | 42 ± 13 | 52 |
Note: IgG(S) (BAU/ml) represent median (IQR) values. BMI, body mass index, GIR, Group Iso Resources: Dependence score from 1 (completely dependent for most daily life activities) to 6 (completely independent) by using the AGGIR grid (Autonomy, Gerontology Group Iso Resources). Assessment of this score is mandatory for all NH residents in France (https://www.capretraite.fr/aide‐a‐domicile/perte‐dautonomie/grille‐aggir/).
Among the 74 COV‐Yes subjects, 50 had SARS‐Cov‐2 infection before the first vaccination and 20 a few days after the 2nd dose. After adjusting for the time since the last immunization, the 1st IgG(S) quantification showed similar levels in these 2 subgroups (median (IQR)): 3133 (1896–7497) vs. 2791 (1483–7592) respectively (p = 0.54).
Delta time between 2nd vaccination or COVID‐19 (if occurred after the 2nd vaccination) and the 1st or 2nd IgG (S) quantification.
Delta time between 3rd vaccination and 3nd IgG(S) quantification. **p < 0.01 and ***p < 0.001 versus COV‐No of the same cohort.
SARS‐CoV‐2 IgG(S) antibody quantification: comparison between COV‐No versus COV‐Yes groups
In the Main cohort (Table 1, left), significantly lower IgG(S) titers were found in the COV‐No vs. COV‐Yes subjects for the 1st IgG(S) quantification (median: 621 vs. 2901 BAU/ml)) (p < 0.001). No differences in IgG(S) (time‐adjusted) levels were observed between the subjects who presented SARS‐CoV‐2 infection before or after the 2nd vaccination (Table 1 legend). The same results were observed for the 2nd IgG quantification for both cohorts (Table 1, right). After the booster dose (3rd IgG quantification), IgG(S) increased dramatically in all groups. A significant difference in favor of COV‐Yes was found only in the Confirmatory cohort (3795 vs. 6675 BAU/ml) (p < 0.01). No differences in IgG(S) levels were observed between the Main and Confirmatory cohorts in each COVID‐19 status group both for the 2nd and 3rd IgG quantifications. The COV‐Yes/COV‐No IgG(S) ratio after the 3rd dose dramatically decreased compared to the ratio after the 2nd dose (2nd quantification) from 16.9 to 1.6 (Main cohort) and from 15.3 to 1.8 (Confirmatory cohort).
The evolution of the individual IgG(S) levels in the two groups of the Main cohort in residents having all 3 IgG(S) measurements showed that, in the COV‐No group, the response to the 3rd vaccination was significantly higher than the response to the 2nd vaccination, whereas no such difference was observed in the COV‐Yes group (Figure S2).
Figure 2 shows the classification of residents according to IgG(S) levels during the different quantifications: 95.1% of the COV‐No and 100% of the COV‐Yes residents of the Main cohort showed a positive SARS‐CoV‐2 (>34 BAU/ml) serology during the first IgG(S) quantification. A more pronounced IgG(S) response in the COV‐Yes was clearly observed during the first and the second quantifications (p < 0.0001) but not in the 3rd especially in the Confirmatory cohort (p = 0.47). Of note, in the 2nd quantification performed 7 months after the second dose, the percentage of residents with levels >264 BAU/ml reached up to 93% in the COV‐Yes group but only 28% in the COV‐No group of the Main and 92.5% and 28.2% in the Confirmatory cohort, respectively.
FIGURE 2.
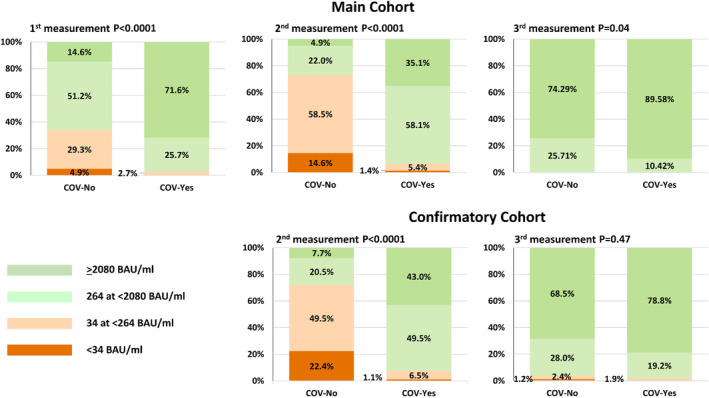
Classification of the subjects in four IgG(S) level categories (BAU/ml) during the 1st (left), 2nd (middle) and 3rd (right) quantification according to history of SARS‐CoV‐2 contamination in the Main (upper) and the Confirmatory (lower) cohorts
Multivariate analyses in the Main cohort revealed that IgG(S) levels during the first and second quantifications were dependent on both SarS‐CoV‐2 status (p < 0.00001) and time since last immunization (p < 0.001). Age, sex, and clinical parameters did not influence IgG(S) levels.
Microneutralization assay
A strong positive relationship was found between IgG(S) levels and neutralization activity (NT50) (R 2 = 0.77, p < 0.001) (Figure S3). No difference was observed when comparing neutralization activity of post‐vaccination serum in residents with or without history of prior COVID‐19 (COV‐Yes vs. COV‐No: p = 0.102 for low IgG(S) titers and p = 0.567 for high IgG titers). Multivariate analysis showed that neutralization activity was determined by IgG(S) levels (p < 0.0001) but not by SARS‐CoV‐2 status (p = 0.98).
Serum from NH residents with low‐level IgG titers (<264 BAU/ml) neutralized the virus in vitro (NT50 ≥40) in 2/17 (11.8%) cases, whereas serum from NH residents with high‐level IgG titers (≥264 BAU/ml) neutralized the virus in vitro (NT50 ≥40) in all 22/22 (100%) cases (p < 0.0001 vs. low IgG titers).
IgG waning rates in the Main and Confirmatory cohorts
In both the Main (longitudinal) and Confirmatory (cross‐sectional) cohorts, the exponential model was significantly better than the linear model for assessing the decay in IgG (S) levels over time following the 2nd dose. Thus, the exponential pattern allowed establishing a linear model between the log of IgG and the time since the 2nd vaccination (Table 2). In the Main cohort, the beta coefficients of the IgG/time relationship were highly significant for both groups (p < 0.001) and the interaction between time and SARS‐CoV‐2 status group was significant (p = 0.051). The proportion of IgG(S) lost during 1 month, estimated from the linear mixed model, was 29.5% (95% CI: 26.4–32.5) in the COV‐No group and 25.4% (95% CI: 22.6–28.0) in the COV‐Yes group. Figure 3 shows the distribution of IgG(S) levels according to time after the second dose and SARS‐CoV‐2 status in the Main and the Confirmatory cohorts.
TABLE 2.
Linear regression models for the association with log‐IgG(S) after the second dose of BNT162b2 vaccine
| Main cohort (linear mixed effect model) | Confirmatory cohort (linear model) | |||
|---|---|---|---|---|
| Beta (SE) | p‐value | Beta (SE) | p‐value | |
| History of SARS‐Cov‐2 infection | ||||
| COV‐No | 7.31 (0.21) | <0.0001 | 8.30 (0.47) | <0.0001 |
| COV‐Yes | 9.21 (0.17) | <0.0001 | 9.20 (0.72) | <0.0001 |
| Time since second dose of vaccine (months) | ||||
| Among COV‐No subjects | −0.35 (0.02) | <0.0001 | −0.49 (0.07) | <0.0001 |
| Among COV‐Yes subjects | −0.29 (0.02) | <0.0001 | −0.31 (0.12) | 0.011 |
| Time since second dose of vaccine × Covid‐19 status | Interaction p‐value = 0.051 | Interaction p‐value = 0.20 | ||
FIGURE 3.
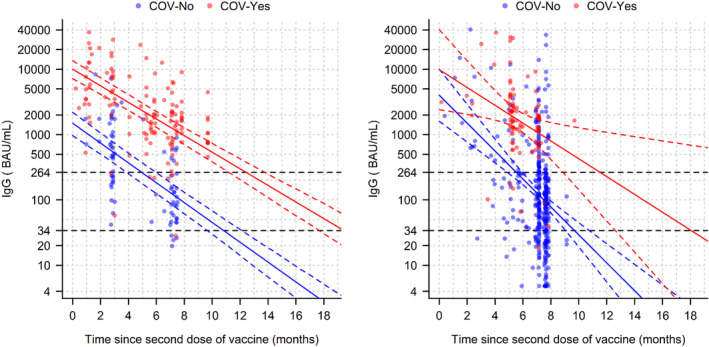
Distribution of IgG(S) levels according to time after the second dose of vaccine and COVID‐19 status in the Main (left) and Confirmatory (right) cohorts. IgG is represented in logarithmic scale. Solid lines represent the predictions from the model and dotted lines the 95% confidence interval. The horizontal black dotted lines correspond to the IgG(S) titers of negative serology 34 BAU/ml) and of “robust” protection (264 BAU/ml) In the Main cohort, each individual is represented twice corresponding to the 1st and the 2nd IgG(S) quantifications in accordance with the linear mixed model described in the Methods section.
Estimation of immunization duration and robust protection following the 3rd dose
For this estimation, the results of the IgG(S) decay following the 2nd dose in the Main (longitudinal) cohort were used with the assumption that the waning rates of IgG(S) over time in each COVID‐19 status group were similar after the 3rd dose as compared to the observed decay after the 2nd dose. Following this analysis in the totality of the subjects who had received the 3rd dose (n = 389), the time (months) to return under the threshold of 264 BAU/ml in the COVID‐No group was estimated at 8.95 (6.85–11.04) (median (IQR)) after the 3rd dose versus 5.76 (3.91–8.64) (p < 0.00001) after the 2nd dose and 11.87 (9.88–14.87) versus 12.60 (10.69–14.44) (NS) in the COV‐Yes group, respectively.
The corresponding times to return under the positivity levels (<34 BAU/ml) in COVID‐No residents were estimated at 14.73 (12.63–16.83) versus 11.54 (9.69–14.42) months (p < 0.00001) and 18.77 (16.78–21.76) versus 19.50 (17.58–21.34) months (NS) in the COVID‐Yes residents, respectively.
For both the 2nd and 3rd doses, these times were longer in the COV‐Yes than the COV‐No group (p < 0.0001), while the difference in absolute values related to COVID status were also attenuated after the 3rd dose.
DISCUSSION
In the present study, we analyzed the IgG(S) response to the 2nd and 3rd doses of the Pfizer BioNtech mRNA vaccine in 2 different cohorts (Main and Confirmatory) comprised of a total of 574 NH residents. This design enabled us to show that the totality of the results observed in the Main Cohort were also observed in the Confirmatory cohort which, in our opinion, provides robustness to the data and conclusions presented herein.
The study's focus was aimed at assessing the IgG(S) response after the 2nd and the 3rd vaccination since there is currently sufficient evidence regarding the importance of IgG(S) titers for the robust protection against severe forms of the disease 8 , 15 , 16 which is a major concern in this very old and highly frail population. In addition, in a subgroup of this population, we analyzed the relationship between IgG(S) and neutralizing capacity in residents with or without prior SARS‐CoV‐2 infection.
Two methodological aspects of the present study are particularly noteworthy:
The method used for antibody quantification involved a whole recombinant trimeric protein for the detection of IgG. 10 This method therefore allows detecting all antibodies directed against the S protein, and not only those linked to the RBD, the ACE2 binding site. The quantification range of this method is linked to the WHO standard, thus allow determining the IgG(S) titer in BAU/ml. 17
Similarly, the conventional microneutralization method used herein was based on the original live SARS‐CoV‐2 virus to better approximate in vivo humoral immunity.
Due to their characteristics, the results of these two methods are expected to be correlated and allow a relevant approach to the correlate of protection of anti‐SARS‐CoV‐2 IgG(S). Thus, as shown in previous reports, 3 , 8 the neutralization capacity was strongly correlated with IgG(S) levels in both COV‐Yes and COV‐No subjects. Of particular interest is that for similar levels of IgG(S), no difference between COV‐No versus COV‐Yes was observed.
A number of clinical studies conducted in NHs have shown that residents with a history of SARS‐Cov‐2 infection showed a more pronounced post‐vaccination IgG(S) level than residents without prior infection. 3 , 4 Our study confirms this significant difference and further adds key information showing that 7 months after the 2nd dose, this difference was even more pronounced, that is, the COV‐Yes group showed median IgG(S) titers 15‐fold higher than the COV‐No group, in both the Main and Confirmatory cohorts. In addition, the percentage of subjects with IgG(S) titers >264 BAU/ml, was only 28% in COV‐No residents in both cohorts, whereas among COV‐Yes residents, approximately 93% in both cohorts featured IgG(S) titers >264 BAU/ml which seems associated to robust protection. 11 These results indicate that among NH residents, the risk of loss of immunity over a relatively short period following the initial 2‐dose vaccination is much higher in subjects without prior SARS‐CoV‐2 infection.
As also shown previously, 18 our analyses revealed that in both COVID status groups, the decay in IgG(S) followed an exponential pattern, which allowed obtaining a linear relationship between the log‐IgG(S) and time interval after the second vaccination. These analyses showed that COV‐Yes residents presented a duration of immunization more than twice that of COV‐No residents, primarily due to the much higher IgG(S) levels after the second vaccination in the COV‐Yes group and secondarily due to the lower rates of IgG(S) waning in COV‐Yes individuals.
Of importance, following the 3rd vaccination, the IgG(S) levels were higher than those observed after the 2nd dose in COV‐No residents, thereby leading to an approximate 50% increase in the duration of robust protection in this group after the 3rd vaccination, although this effect was still less pronounced than the duration of robust protection of the 3rd dose in the COV‐Yes group. These results suggest that in NH residents without prior history of SARS‐CoV‐2 infection, protection against the severe forms of COVID‐19 with the 3rd dose will be considerably longer compared to the 2nd dose and similar to the protection obtained in COV‐Yes subjects after the 2nd vaccination. The present findings are in agreement with recent data showing the interest of the 3rd vaccination in the reduction of hospitalizations linked to the Delta and Omicron variants. 19
This study has some limitations: first, the definition of COV‐Yes by PCR and/or IgG(N) + cannot eliminate the possibility that some subjects considered as being COV‐No had indeed a history of previous asymptomatic SARS‐Cov‐2 infection. 7 Second, only exploration of humoral but not cellular immunity was performed, which probably has an important role in the prevention of serious disease forms. Third, only two IgG measurements were collected to assess IgG decay over time after the 2nd dose in the Main (longitudinal) cohort. Additional IgG measurements would have allowed exploring in greater detail the kinetics of IgG decay after the 2nd dose. Fourth, the emergence of omicron or other variants in the future may modify the duration of protection. Finally, we analyzed the IgG(S) response of only homologous mRNA boosters which may not be the same compared to the use of heterologous vaccines. 20
In conclusion, in this very old population, previous SARS‐Cov‐2 infection induces a more pronounced IgG(S) response to the 2nd dose of the vaccine and a longer protection compared to the group without history of SARS‐Cov‐2 infection. This difference between the two COVID‐19 groups is almost abolished after the 3rd dose. We anticipate than in NH residents without history of SARS‐CoV‐2, the 3rd dose of the mRNA vaccine, compared to the 2nd dose, will induce a more prolonged protection against severe forms of COVID‐19.
AUTHOR CONTRIBUTIONS
Helene Jeulin: conception and design of the study, acquisition of biological data, interpretation of the results, drafting of the article. Carlos Labat: conception and design of the study, preparation of the database, acquisition of clinical data, statistical analyses, critical revision of the article for important intellectual content. Kevin Duarte: acquisition of data, statistical analysis, critical revision of the article for important intellectual content. Simon Toupance: acquisition of biological data, critical revision of the article for important intellectual content. Gregoire Nadin: conception and design of the study, mathematic models and statistical analyses, critical revision of the article for important intellectual content. Denis Craus: conception and design of the study, organization of the nursing homes network participating in the study, critical revision of the article for important intellectual content. Ioannis Georgiopoulos: acquisition of clinical data, critical revision of the article for important intellectual content. Isabelle Gantois: conception and design of the study, organization of the NH network participating in this study, critical revision of the article for important intellectual content. François Goehringer: conception and design of the study, interpretation of the results, drafting of the article. Athanase Benetos: coordination of the study, conception and design of the study, interpretation of the results, drafting the article.
FUNDING INFORMATION
This study has been supported by the PIA project “Lorraine Université d'Excellence” reference ANR‐15‐IDEX‐04‐LUE, and the FHU project CARTAGE‐PROFILES (Aviesan, 2021‐2025).
CONFLICT OF INTEREST
The authors have declared no competing interest.
SPONSOR'S ROLE
The sponsor did not have any role in the design, methods, subject recruitment, data collection, analysis and interpretation of the results, or preparation of the article.
Supporting information
Data S1 Supporting information.
ACKNOWLEDGMENTS
The authors thank the following medical coordinators of the participating NHs: Marc Berr, Thierry Collin, Caroline Ferry‐Bert, Marie‐Chrisine Godard, Patrick Lucquin, Lydie Osnowycz, and Henri Rozenfarb. We thank Ms Anne Fréminet, Alice Metz and Cécile Lakomy for their valuable contribution in the acquisition of clinical and biological data. We thank all the directors and the staff of the 14 nursing homes for contributing to the realization of this study. We thank Mr Pierre Pothier for language review and stimulating discussions.
Jeulin H, Labat C, Duarte K, et al. Anti‐spike IgG antibody kinetics following the second and third doses of BNT162b2 vaccine in nursing home residents. J Am Geriatr Soc. 2022;1‐9. doi: 10.1111/jgs.17837
The paper has been published as a preprint on medRxiv: https://medrxiv.org/cgi/content/short/2022.02.07.22270557v1.
Funding information Agence Nationale de la Recherche, Grant/Award Number: ANR‐15‐IDEX‐04‐LUE
REFERENCES
- 1. Teran RA, Walblay KA, Shane EL, et al. Postvaccination SARS‐CoV‐2 infections among skilled nursing facility residents and staff members ‐ Chicago, Illinois, December 2020–March 2021. MMWR Morb Mortal Wkly Rep. 2021;30(70):632‐638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. https://www.santepubliquefrance.fr/content/download/354025/3069653 Assessed February 14, 2022
- 3. Helle F, Moyet J, Demey B, et al. Humoral anti‐SARS‐CoV‐2 immune response after two doses of Comirnaty vaccine in nursing home residents by previous infection status. Vaccine. 2022;40:531‐535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Salmerón Ríos S, Cortés Zamora EB, Avendaño Céspedes A, et al. Immunogenicity of the BNT162b2 vaccine in frail or disabled nursing home residents: COVID‐A study. J Am Geriatr Soc. 2021;69:1441‐1447. doi: 10.1111/jgs.17153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Gaebler C, Wang Z, Lorenzi JCC, et al. Evolution of antibody immunity to SARS‐CoV‐2. Nature. 2021;591:639‐644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Stamatatos L, Czartoski J, Wan YH, et al. mRNA vaccination boosts cross‐variant neutralizing antibodies elicited by SARS‐CoV‐ 2 infection. Science. 2021;372:1413‐1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Gallais F, Gantner P, Bruel T, et al. Evolution of antibody responses up to 13 months after SARS‐CoV‐2 infection and risk of reinfection. EBioMedicine. 2021;71:103561. doi: 10.1016/j.ebiom.2021.103561 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Levin EG, Lustig Y, Cohen C, et al. Waning immune humoral responseto BNT162b2 Covid‐19 vaccine over six months. N Engl J Med. 2021;385:e84. doi: 10.1056/NEJMoa2114583 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Zhong D, Xiao S, Debes AK, et al. Durability of antibody levels after vaccination with mRNA SARS‐CoV‐2 vaccine in individuals with or without prior infection. JAMA. 2021;326:2524‐2526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Bonelli F, Blocki FA, Bunnell T, et al. Evaluation of the automated LIAISON® SARS‐CoV‐2 TrimericS IgG assay for the detection of circulating antibodies. Clin Chem Lab Med. 2021;59:1463‐1467. [DOI] [PubMed] [Google Scholar]
- 11. Feng S, Phillips DJ, White T, et al. Correlates of protection against symptomatic and asymptomatic SARS‐CoV‐2 infection. Nat Med. 2021;27:2032‐2040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Meschi S, Matusali G, Colavita F, et al. Predicting the protective humoral response to a SARS‐CoV‐2 mRNA vaccine. Clin Chem Lab Med. 2021;59:2010‐2018. [DOI] [PubMed] [Google Scholar]
- 13. Greenland S. Tests for interaction in epidemiologic studies: a review and a study of power. Stat Med. 1983;2:243‐251. [DOI] [PubMed] [Google Scholar]
- 14. Brookes ST, Whitely E, Egger M, Smith GD, Mulheran PA, Peters TJ. Sub‐group analyses in randomized trials: risks of subgroup‐specific analyses; power and sample size for the interaction test. J Clin Epidemiol. 2004;57:229‐236. [DOI] [PubMed] [Google Scholar]
- 15. Khoury DS, Cromer D, Reynaldi A, et al. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS‐CoV‐2 infection. Nat Med. 2021;27:1205‐1211. [DOI] [PubMed] [Google Scholar]
- 16. Bergwerk M, Gonen T, Lustig Y, et al. Covid‐19 breakthrough infections in vaccinated health care workers. N Engl J Med. 2021;385:1474‐1484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Scheiblauer H, Nübling CM, Wolf T, et al. Antibody response to SARS‐CoV‐2 for more than one year kinetics and persistence of detection are predominantly determined by avidity progression and test design. J Clin Virol. 2022;146:105052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Garner‐Spitzer E, Wagner A, Kundi M, et al. SARS‐CoV‐2‐specific antibody (ab)levels and the kinetic of ab decline determine ab persistence over 1 year. Front Med. 2022;9:822316. doi: 10.3389/fmed.2022.822316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Ferdinands JM, Rao S, Dixon BE, et al. Waning 2‐dose and 3‐dose effectiveness of mrna vaccines against covid‐19‐associated emergency department and urgent care encounters and hospitalizations among adults during periods of delta and omicron variant predominance ‐ VISION network, 10 states, August 2021‐January 2022. MMWR Morb Mortal Wkly Rep. 2022;7:255‐263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Mayr FB, Talisa VB, Shaikh O, Yende S, Butt AA. Effectiveness of homologous or heterologous Covid‐19 boosters in veterans. N Engl J Med. 2022;386(14):1375‐1377. doi: 10.1056/NEJMc2200415 Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data S1 Supporting information.


