Abstract
Liposomes are now considered the most commonly used nanocarriers for various potentially active hydrophobic and hydrophilic molecules due to their high biocompatibility, biodegradability, and low immunogenicity. Liposomes also proved to enhance drug solubility and controlled distribution, as well as their capacity for surface modifications for targeted, prolonged, and sustained release. Based on the composition, liposomes can be considered to have evolved from conventional, long-circulating, targeted, and immune-liposomes to stimuli-responsive and actively targeted liposomes. Many liposomal-based drug delivery systems are currently clinically approved to treat several diseases, such as cancer, fungal and viral infections; more liposomes have reached advanced phases in clinical trials. This review describes liposomes structure, composition, preparation methods, and clinical applications.
Keywords: Liposomes, Phospholipids, Lamellarity, Stealth liposomes, Vaccinations
Liposomes; Phospholipids; Lamellarity; Stealth liposomes; Vaccinations.
1. Introduction
Drug delivery systems (DDSs) offer the potential to enhance the therapeutic index of drugs by increasing the drug concentration, the residence time in target cells and minimizing the side effects [1]. DDSs involve delivering the potentially active drug to the site of action via a nano-vehicle to enhance the pharmacological properties of free drugs and cover their undesirable features through improving drug pharmacokinetics and biodistribution, as well as acting as drug reservoirs [2, 3]. These nanoparticles (NPs) usually ranged from a few nanometers to several hundred nanometers according to their intended application [4]. Different natural, organic and inorganic materials are used to create NPs including ceramic, polymers, metals [4], and lipids that generate nanoparticles like micelles and liposomes [5, 6, 7].
Therapeutic drugs are incorporated into the NPs mainly by physical interactions including, entrapment, surface attachment, or encapsulation [8]. These variations and unique properties of different NPs could be used to improve the characteristics of traditional therapeutics [8]. Nanomedicine facilitates designing novel therapeutic options in the nanoscale range to deliver a variety of active biomedical ingredients for the treatment, prevention, and diagnosis of many diseases [1, 9].
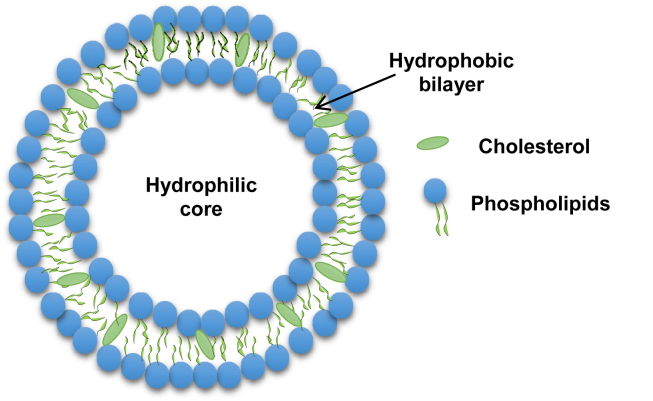
Despite the fast progress in this field, most nanoparticles-based drug delivery systems show improper loading capacity with a lack of specificity against their targets [10]. As a result, the promising advances in the drug delivery systems should involve designing high and regulated capacity nanocarriers functionalized by recognition ligands that target specifically unique or overexpressed biomarkers [11]. Liposomes are the most explored nanocarriers used in targeted drug delivery systems. Liposomes are spherical lipid vesicles (usually 50–500 nm in diameter particle size) composed of one or more lipid bilayers, as a result of emulsifying natural or synthetic lipids in an aqueous medium [12, 13] (Figure 1). Liposomes were firstly discovered in the 1960's by Bengham and later became among the most expansive drug delivery systems [14]. Liposomes nanoemulsions are widely used nanoparticles in nanomedicine mainly due to their biocompatibility, stability, ease to synthesize and high drug loading efficiency [15, 16], high bioavailability [17], and their safe excipients used in these formulations [18]. Due to their size, hydrophobic and hydrophilic characteristics and their ability to encapsulate drug molecules either in the aqueous interior of the vesicles or in the lipophilic membrane [19], liposomes are considered promising to be used effectively as drug delivery systems. Several liposomal-based drug delivery systems have been approved by Food and Drug Administration (FDA) for disease treatment in the market [20, 21]. Moreover, liposomes are suitable for diagnostic and therapeutic applications using several routes of administration, including ocular [22], oral [23], pulmonary [24], transdermal [25], and parenteral [26, 27, 28]. Liposomes are primarily created from phospholipids such as soybean phosphatidylcholine [29] or synthetic dialkyl or trialkyl lipids [30]. Incorporation of cholesterol into liposomes is indispensable since cholesterol modulates membrane permeability, changes fluidity, and improves the stability of bilayer membranes in the presence of biological fluids such as blood and plasma [31, 32]. Liposomal formulations may also contain polymers [33], and even membrane protein [34] to prolong their circulation half-life, improve the biodistribution profile and enhance the encapsulated drug effectiveness [35] Moreover, Stealth stabilized liposomes, incorporating phospholipids-attached polyethyleneglycol (PEG) into liposomes infrastructure, has been shown to be a useful method for modifying liposomes pharmacokinetic properties and biodistribution profiles [36]. The current review describes liposomes compositions, types, methods of preparation, and clinical applications.
Figure 1.
Schematic representation of liposomes.
2. Liposomes
According to the liposomes structures, they are classified into four categories based on size and number of bilayers: small unilamellar vesicles (SUV), large unilamellar vesicles (LUV), multilamellar vesicle (MLV), and multivesicular vesicles (MVV). Liposomes have mono phospholipid bilayer in a unilamellar structure, while they have an onion-like structure in a multilamellar structure. MVV form a multilamellar arrangement with concentric phospholipid spheres as many unilamellar vesicles are produced within larger liposomes [37]. Liposome encapsulation efficiency increases with liposome size and decreases with the number of bilayers for hydrophilic compounds only [38]. The size of the vesicles is an important factor that controls the circulation half-life of liposomes. Both the size and number of bilayers influence the amount of the encapsulated drug. When liposomes are employed for drug delivery, the desired vesicles usually extend from 50 nm to 150 nm. Liposomes interaction with the cell membrane is represented by various theories: specific (modified with receptor-mediated) or nonspecific endocytosis [39], local fusion (adhesion) [40], phagocytosis [41], and absorption into the cell membrane [42]. Liposome-cell interactions are influenced by a variety of factors, including composition [43], the diameters of liposomes, surface charge [44], targeting ligand on the liposome surface, and biological environment [45].
2.1. Liposomes compositions
2.1.1. Lipids and phospholipids used for liposomes
Structurally, liposomes are spherical or multilayered spherical vesicles made by the self-assembly of diacyl-chain phospholipids (lipid bilayer) in aqueous solutions [46]. The bilayer phospholipid membrane has a hydrophobic tail and a hydrophilic head [21, 47] that leads to the formation of an amphiphilic structure. Liposomes can be made from both natural and synthetic phospholipids [48]. Lipid composition strongly affects liposome characteristics that include: particle size, rigidity, fluidity, stability, and electrical charge [5, 49]. For example, liposomes formulated from natural unsaturated phosphatidylcholine, as egg or soybean phosphatidylcholine, provide highly permeable and low stable properties. Though, saturated-phospholipids-based liposomes such as dipalmitoyl phosphatidylcholine led to rigid and almost impermeable bilayer structures [21].
The hydrophilic group in the lipids may be negatively, positively charged, or zwitterionic (both negative and positive charge in the same molecule). The charge of the hydrophilic group provides stability through electrostatic repels. The hydrophobic group of lipids varies in the acyl chain length, symmetry, and saturation [50]. The lipids that used in liposomes preparation may be classified as:
2.1.2. Natural lipids
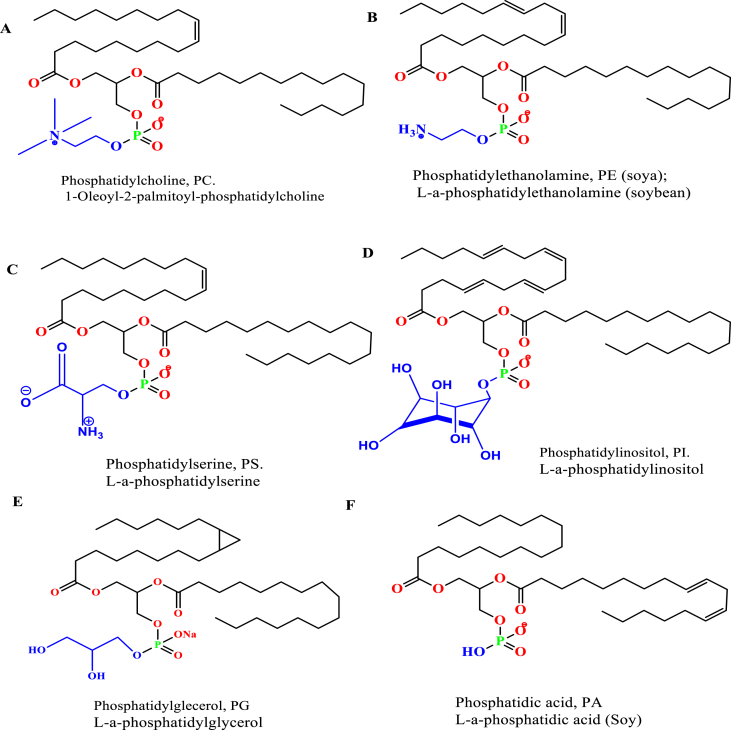
The membrane bilayer of normal cells are mainly composed of glycerophospholipids. Phospholipids are consist of a glycerol unit that is bonded to a phosphate group (PO42−) and to two fatty acid molecules. The phosphate group can be also bonded to small, essential choline organic molecule [21, 51] (Figure 2A). Natural phospholipids can be obtained from various sources such as soya bean, egg yolk [52]. Phospholipids are classified as phosphatidylcholine (PC), phosphatidylethanolamine (PE), phosphatidylserine (PS), phosphatidylinositol (PI), phosphatidylglycerol (PG), and phosphatidic acid (PA) regarding to the polar head groups. Natural phospholipids are less stable than synthetic phospholipids in liposomes preparation due to the unsaturated characteristics of the hydrocarbon chain [53, 54] (Figure 2). Natural phospholipids composed of a variety of fatty acids, one is a saturated fatty acid as palmitic acid (hexadecanoic acid, H3C-(CH2)14-COOH); margaric acid (heptadecanoic acid, H3C-(CH2)15-COOH) and the other is an unsaturated fatty acid (here oleic acid, or 9Z-octadecenoic acid that identified in egg yolk lecithin [55]. The egg derived phospholipids and PCs are made of these fatty acids patterns: palmitic acid (C16:0), stearic acid (C18:0), oleic acid (C18:1), linoleic acid (C18:2), and arachidonic acid (C20:4). These fatty acids are account for about 92% of the total fatty acid composition with a typical presence of the polyunsaturated fatty acids C 20:4 (n-6) and C22:6 (n-3) in egg phospholipids. Egg PC contains about 40% 1-palmitoyl-2-oleoylphosphatidylcholine. The principal saturated acid was stearic in PE and PS, and palmitic in the other lipids. Furthermore, the fatty acid pattern of the soybean derived account for about 95% palmitic acid (C16:0), stearic acid (C18:0), oleic acid (C18:1), linoleic acid (C18:2), and linolenic acid (C18:3) [56]. Since the unsaturated fatty acids of PE, PS, and PC amounted to over 50% of the total acids, they must occur at both the α- and β-positions of the glycerol moiety of these phospholipids [57].
Figure 2.
Natural phosphatides the most used to produce liposomes; A) Phosphatidylcholine, B) Phosphatidylethanolamine, C) Phosphatidylserine, D) Phosphatidylinositol, E) Phosphatidylglecerol, and F) Phosphatidic acid.
2.1.3. Synthetic lipids
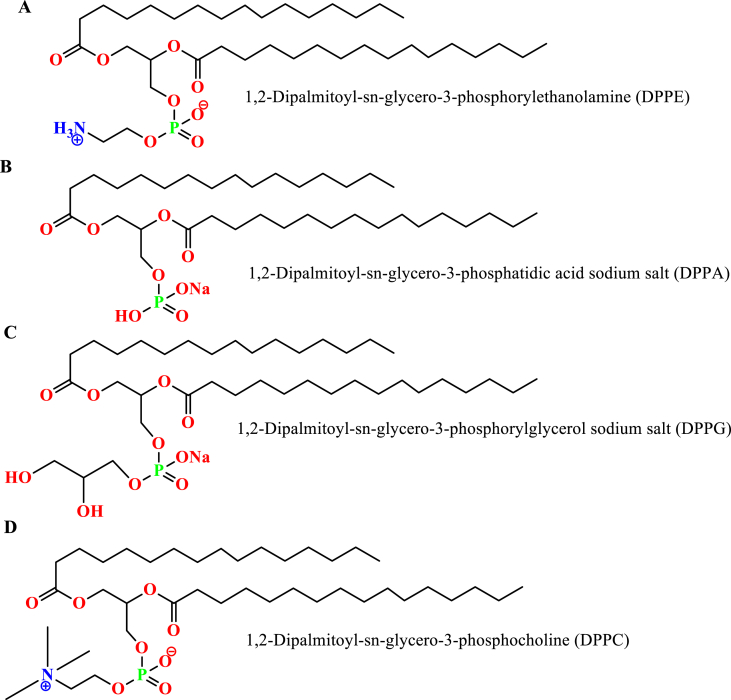
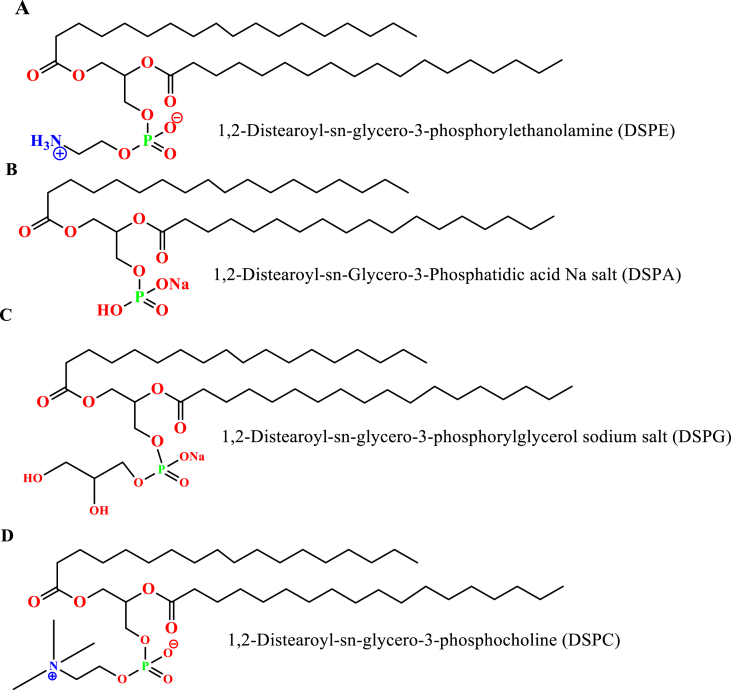
Synthetic phospholipids are made by specific chemical modifications to the non-polar and polar regions of the natural phospholipids. The modification enables an unlimited variety of well-defined and categorized phospholipids [56]. The major saturated synthetic phospholipids are based on either using stearic and/or palmitic fatty acid. Figures 3 and 4 represent different possible and commercial, synthetic, and saturated phospholipids usually used to prepare liposomes [53].
Figure 3.
Palmitic acid -based different synthetic phospholipids; A) 1,2-Dipalmitoyl-sn-glycero-3-phosphorylethanolamine, B) 1,2-Dipalmitoyl-sn-glycero-3-phosphatidic acid sodium salt, C) 1,2-Dipalmitoyl-sn-glycero-3-phosphorylglycerol sodium salt, and D) 1,2-Dipalmitoyl-sn-glycero-3-phosphocholine (DPPC).
Figure 4.
Stearic acid -based different synthetic phospholipids; A) 1,2-Distearoyl-sn-glycero-3-phosphorylethanolamine, B) 1,2-Distearoyl-sn-Glycero-3-Phosphatidic acid Na salt, C) 1,2-Distearoyl-sn-glycero-3-phosphorylglycerol sodium salt, and D) 1,2-Distearoyl-sn-glycero-3-phosphocholine.
Additionally, Synthetic phospholipids can be made from mixed fatty acids, unsaturated fatty acids in both hydrocarbons or only in one hydrocarbon chain [52] (Figure 5).
Figure 5.
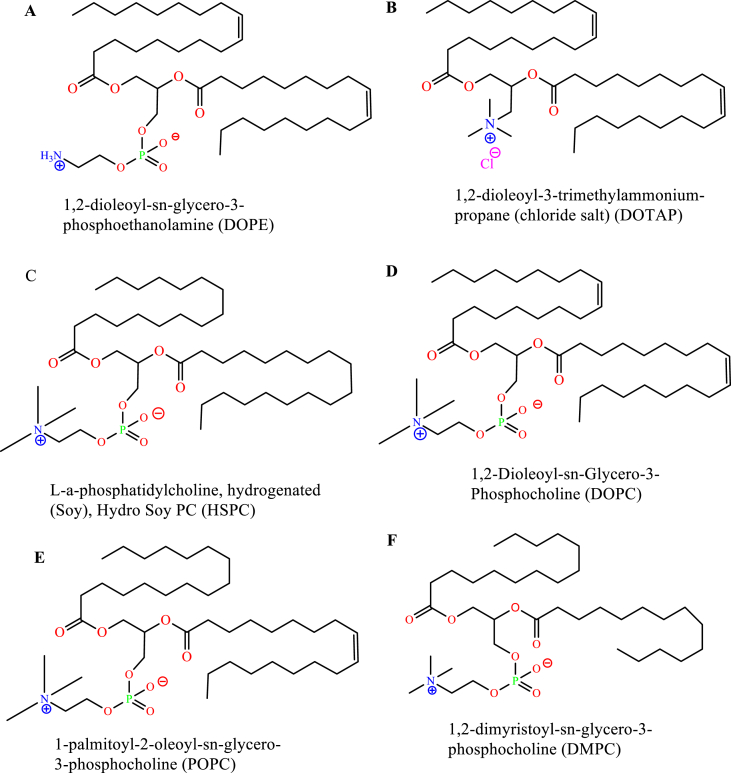
Mixed and different types of synthetic phospholipids; A) 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine, B) 1,2-dioleoyl-3-trimethylammonium-propane (chloride salt), C) L-a-phosphatidylcholine, D) 1,2-Dioleoyl-sn-Glycero-3-Phosphocholine, E) 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine, and F) 1,2-dimyristoyl-sn-glycero-3-phosphocholine.
2.1.4. Steroid
Steroid are hydrophobic lipids consists of four-ring structure as shown in Figure 6. Steroid's diversity comes from the various functional groups attached to those rings. Cholesterol is the major steroid usually used in liposomes preparation in a ratio less than 30 % of the total lipids to improve liposomes rigidity and stability since its incorporated in the liposomes lipid bilayer [47, 58]. In a comparative study for cholesterol and β-Sitosterol effect on the liposome membrane features, they found that both steroids reduce liposomes membrane fluidity, increase absolute zeta potential, cause significant changes in particle size, and decrease DPPC phase transition temperature (Tm) and enthalpy [59].
Figure 6.
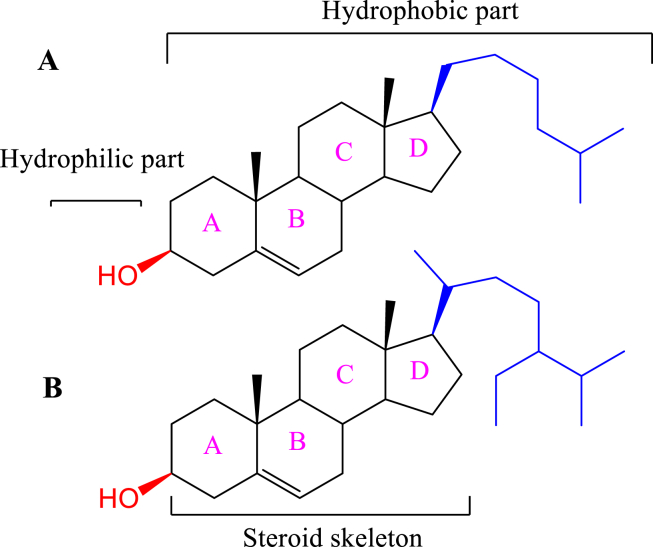
Chemical structure of A) cholesterol, B) β-sitosterol.
2.1.5. Surfactants
Surfactants were utilized in liposomes formulations to modify the encapsulation and release properties of liposomes through surface tension reduction between different immiscible phases [60]. Surfactants are single acyl-chain amphiphiles that destabilize the lipid bilayer of liposomal nanoparticles (Figure 7), thus increasing nano-vessel deformability [61, 62]. Commonly utilized surfactants in liposomes formulations are: sodium cholate, Span 60, Span 80, Tween 60, and Tween 80 [62, 63]. Various surfactants-containing liposomes have been widely used as a carrier in drug delivery to enhance skin penetration of encapsulated therapeutic agents [64]. Ultra deformable liposomes, also called transfersomes, are a Surfactants-based nanovesicles with positive findings in transdermal drug delivery [65, 66]. The key factor making liposomes deformable is edge activator (surfactant). The edge activator can alter the lipid bilayers of vesicles increasing the deformability of them [67]. These nanovesicles are different from conventional liposomes in which they can respond to osmotic pressure by rapid shape transformations only by low energy [67]. Moreover, ultra deformable liposomes showed an increased in the drug transepidermal flow made them more suitable nanovehicle for the topical administration of antihypertensives [68].
Figure 7.
Chemical structure of surfactants A) sodium cholate, B) Sodium dodecyl sulfate (SDS).
2.2. Liposomes types
Based on their compositions and applications, liposmes can be classified into conventional liposomes [69], charged liposomes [70], stealth stable liposomes [71], actively targeted liposomes [72], stimuli-responsive liposomes [73], and bubble liposomes [74].
2.2.1. Conventional liposomes
These liposomes were synthesized from natural or synthetic phospholipids with or without cholesterol as a liposomes first generation [75]. Cholesterol was added to improve liposomes fluidity, altering the bilayer rigidity and liposomes stability [31, 32]. Wu et al hat revealed that liposomal membrane rigidity decreased with the addition of cholesterol into a liposomes composed of hydrogenated soybean phospholipids (HSPC) and DSPE-PEG2000. Moreover, liposomes with some rigidity showed excellent tumor penetration and enhanced anti-tumor activity [76]. Kaddah et al investigated the cholesterol content on the permeability and fluidity of DPPC liposome membrane. High cholesterol concentration increased the average liposomes size accompanying with a shape transition from irregular to nanosized, regular and spherical vesicles. In addition, cholesterol induced a decrease in the bilayer fluidity and modulating the release of hydrophilic molecules from lipid vesicles [77]. Jovanović et al reported that increasing cholesterol content in the liposomes decreased the fluidity and enhanced the rigidity of liposomal membranes. They verified that a stable liposome should have an optimal 50 mol %. concentration of cholesterol to obtain and an appropriate membrane fluidity [78]. As a result, cholesterol plays a crucial role in liposomes bilayer fluidity and rigidity, but these properties affected by the cholesterol molar ratio with types of phospholipids used along with the nature of the encapsulated drug. Conventional liposomes showed a short blood circulation time due to their susceptability to elimination by the mononuclear phagocyte system [79] with rapid accumulation in liver and spleen. Hence, MPS obstructs the delivery of conventional liposomes to the target region and restricts their distribution to other tissues of the body [80]. Conventional liposomes also showed relatively limited stability in vitro [81]. As a result, stealth stable liposomes were invented to increase blood circulation and enhanced in vivo liposomes stability [82].
2.2.2. Charged liposomes
Oleic acid and N-[1(2,3-dioleoyloxy) propyl]-N,N,N-trimethylam-monium chloride (DOTAP) are usually used to prepare anionic and cationic liposomes, respectively. Charged liposomes showed higher liposomal stability during the storage, as charged particles repel each other and reduce aggregation abilities. Cationic liposomes are used in gene therapy due to their ability to successfully encapsulate nucleic acids by electrostatic attractions [83].
Cationic liposomes are suitable for delivering various negatively charged macromolecules such as DNA, RNA, and oligonucleotides because their negative charge and rather a large size restrict their passive diffusion into cells [84]. Cationic liposomes also selectively target angiogenic endothelial cells in tumors [85]. Cationic liposomes are considered a potential tool for delivering therapeutics to the brain [6, 86]. Cationic liposomes can cross the BBB by receptor-mediated transcytosis [87] or absorptive-mediated transcytosis [88]. The higher positive charge on the surface of cationic liposomes may affect their blood circulation and lead to due to electrostatic interactions with anionic species in the blood and increase liposomes aggregation that reduces their localization site of action [89, 90]. Decorating the surface of these liposomes with poly ethylene glycol (PEG) protects the them from the circulating proteins, improving the drug efficiency through improving systemic circulation time and decrease immunogenicity [91, 92].
Anionic liposomes are less stable in the bloodstream than neutral and cationic liposomes; they showed a higher clearance rate [44, 93]. Anionic liposomes are usually utilized for transdermal drug delivery because they improve penetration properties through the stratum corneum of the skin [94].
2.2.3. Stealth stabilized liposomes
These second-generation liposomes are characterized by surface decoration with synthetic polymers, glycoproteins, polysaccharides, or specific receptors ligands to achieve narrowed distribution, and accumulation at the intended site [95]. Huayluorinic acid [96], polyvinyl alcohol (PVA), and polyethylene glycol (PEG)) were considered the best model for liposome steric protection. PEGylated liposomes are denoted as stealth liposomes [97, 98]. Doxil® was the first successful pegylated liposome-based product [99]. Stealth stabilized liposomes showed longer circulation time, leading to a better target accumulation than conventional liposomal drugs [100].
2.2.4. Actively-targeted liposomes
Actively-targeted liposomes represent third-generation liposomes. Liposomes' active targeting increases the selectivity of liposome interaction with diseased cells and triggers receptor-mediated endocytosis of the liposome and its payload into the desired cellular target [101, 102].
Many liposomes nanocarriers have been approved for anti-tumor agents delivery by passive ways based on the enhanced permeability and retention (EPR) effect of cancerous cells [103]. Passive targeting does not discriminate between normal and diseased cells [104]; therefore, cell-specific targeting liposomes have been developed to increase the accumulation and localization of anti-tumor agents in diseased cells [104]. Liposomes targeting can be enhanced by incorporating molecular recognition moieties, which can lead to drug transport with better efficacy and low side effects [105]. For example, liposomal targeting strategies have utilized simple peptides [106], proteins (including antibodies) or protein fragments [107], carbohydrates, nucleic acids, or vitamins [108, 109, 110, 111, 112].
Both active (ligands-conjugated) and passive (‘non’-conjugated) targeted liposomes are distributed to target cells via the same passive distribution mechanism [113]. The field of ligand-targeted liposomes has expanded rapidly despite that several non-targeted liposomes have reached the clinic or in clinical trials [114, 115].
New efforts in targeted drug delivery systems utilize polyunsaturated fatty acids, folic acid, hyaluronic acid, or oligopeptides as tumor recognition moieties. These ligands encounter many discussion fields around their affinity and specificity with no detailed mechanism of tumor-targeting accompanied to limited success for certain small ligands [116], besides the enzymatic degradation in the systemic circulation, making them inappropriate for many in vivo studies [117]. Recently, aptamers and aptamer-functionalized nanoparticles high affinity and specificity have great attention in targeted drug delivery systems [118, 119, 120].
Active targeting of the nanocarriers can be achieved through non-covalent or covalent conjugation of targeting ligands to the drug molecule or to the surface of nanocarrier to bind the overexpressed targeting biomarkers selectively on the tumor cells [110, 121]. The direct conjugation of drugs to the targeting ligand can disrupt the receptor/ligand recognition [122] and may alter the drug efficacy [123]. Nano-carrier active targeting enables drugs to be localized within the action site with higher effectiveness [101] to reduce drug dose, minimize the drug side effects and reduced drug variation in blood concentration [124]. Stealth and conventional liposomes usually showed a slow release of the loaded drugs and failed fusion with the endosome after internalization. Consequently, stimuli-responsive liposomes have been introduced to overcome these challenges [125].
2.2.5. Stimuli-responsive liposomes
Stimuli-responsive liposomes are smart liposomal systems that display rapid release of their drug payload upon physicochemical or biochemical stimuli, such as pH, temperature, redox potentials, enzymes concentrations, ultrasound, electric or magnetic fields [126].
Stimuli-responsive liposomes should contain a certain constituent that controls the lipid bilayer's stability and permeability [73]. There are two basic kinds of inductions, remote and local. Remote inductions respond to outside stimuli including, heat, magnetic field, light, electric field, and ultrasounds [73, 125]. Local triggering releases respond to stimuli inside the target tissues, such as pH, redox potential [127, 128], and enzymes [129]. Table 1 represents the most common stimuli-responsive liposomes that respond to specific triggers that lead to a controlled release nanosystem, enhanced intracellular distribution [130].
Table 1.
The most common stimuli-responsive liposomes.
| Stimuli liposomes | Stimuli | Principle | Advantages | Reference |
|---|---|---|---|---|
| Light-sensitive liposomes | UV, near infrared or visible light irradiation, | Modification of fatty acyl chains of the phospholipids with light-sensitive functional groups and the resulting phospholipids have yielded photoactivable liposomes | Controlling time, exposure, wavelength, and intensity | [131, 132] |
| Thermosensitive (temperature-sensitive) liposomes | Radiofrequency or microwave ablation | Lipids with a transition temperature of 40–45 °C, such as DPPC, have been employed to make these liposomes | Drug release at high-temperature sites | [133, 134, 135, 136] |
| Redox-sensitive liposomes | Reactive oxygen species (ROS) peroxides, hydroxyl radicals, singlet oxygen | Depends on the redox potential difference between the intracellular reducing space and oxidizing extracellular space that occur during biological activities. | ROS leads to high concentration levels of glutathione (GSH) in tumor cells cleaving the liposomal formulations | [137, 138] |
| Enzyme-responsive liposomes | Protease, amidase, and esterase enzymes | Based on amides or esters hydrolysis by protease or esterase enzymes release loaded drugs. | Decreases the adverse side effects of toxic drugs and enable encapsulation of prodrugs | [139, 140, 141] |
| pH-sensitive liposomes | pH change | Cholesteryl hemisuccinate (CHEMS) and 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine (DOPE), were used to prepare pH-sensitive liposomes | Liposomes with pH-dependent release features | [142, 143, 144] |
2.2.6. Bubble liposomes
Bubble liposomes (gas-encapsulated liposomes) are expected to create new applications in the field of gene delivery and drug delivery systems [145]. Recently, liposomes have been used to encapsulate bioactive gases and/or drugs for ultrasound-controlled drug release with enhanced drug delivery [146]. Nitric oxide (NO) bubble liposomes offer a distinguishing NO intravenous therapeutics option overcome common microbubbles, in which liposomes shield NO from hemoglobin rummaging in vitro as usually occurred by free NO. Oxygen bubble liposome (OBL) enables high oxygen fixations with high pO2 conditions of the lungs. This separates OBL from great fluorocarbon and hemoglobin-based oxygen transporters and keeps their utilization as supported oxygen conveyance stages [147].
2.3. Methods of preparation
Liposomes can be formulated using different approaches. The process of liposome manufacture and the phospholipids type critically affects the final liposomes characteristics [148]. Liposome's fabrication procedures can be classified into:
2.3.1. Thin film hydration method (Bangham method)
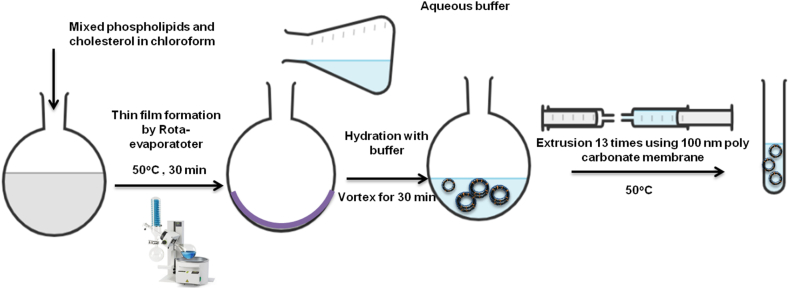
In this method, all lipids and the hydrophobic drug are dissolved in suitable organic solvent using a round-bottom flask [50]. The organic solvent then evaporated gently under reduced pressure to create a thin film layer [21]. The obtained thin film is then hydrated, at above the transition temperature (Tm) of the used lipid, with an aqueous buffer solution. The hydration solution may contain a hydrophilic drug/s to be loaded into the liposomes aqueous core. The rate of hydration determines the efficiency of drug encapsulation [148], which the slower the rate of hydration, the higher the encapsulation efficiency [50]. Liposomes resizing, lamellarity types and particles distributions may be controlled by either extrusion through a polycarbonate membranes of specific pore sizes or the use of bath or probe sonicators. Extrusion method ensures stable liposomes with more encapsulation efficiency over sonication. Sonication usually produce SUVs liposomes and may also degrade or hydrolyze encapsulated drugs and/or lipids. Probe sonication may subject liposomes suspensions to potential metal contamination (Figure 8) [21].
Figure 8.
Liposomes preparation via thin-film hydration extrusion technique.
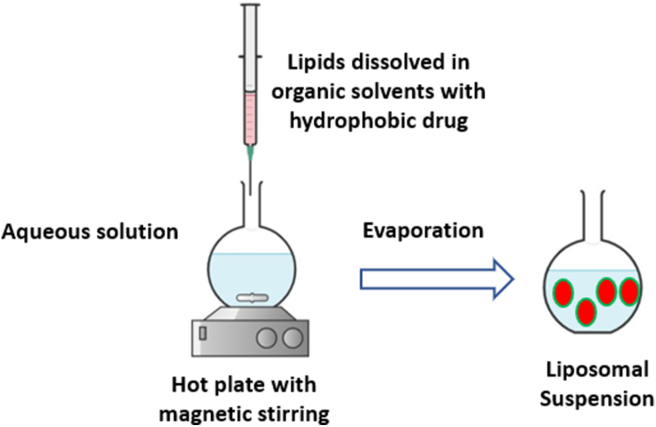
2.3.2. Reverse-phase evaporation method
The reverse-phase evaporation method is usually used as an alternative to thin-film hydration by forming a water-in-oil emulsion [148]. First, the lipids are dissolved in an organic solvent which is then directly mixed with an aqueous buffer containing the hydrophilic drug. The organic solvent then evaporated under a reduced pressure rotary evaporator leading to form lipid vesicles dispersed in the aqueous solution. The average size and polydispersity of the preformed vesicles can be reduced by extrusion [149]. This method is suitable for high molecular weight molecules, but therapeutic peptides may be denatured due to organic solvents and to sonication conditions [150].
2.3.3. Solvent injection methods
The injection methods were classified according to the type of organic solvent used (Figure 9) [151]. An organic solvent dissolving the lipids and the hydrophobic active agents were rapidly injected into an aqueous phase. Diethyl ether enable direct solvent evaporation during mixing process at a temperature above to the boiling point of the used solvent [152]. Utilizing ethanol for injection required a 10-to-20-fold aqueous solution and ethanol can be evaporated under vacuum using a rotary evaporator, dialysis, or filtering. This method mostly prepared liposomal formulations with higher polydispersity indexes (PDI) [153]. In addition, continuous exposure to high temperature and organic solvent might reduce drug and lipids stability [154].
Figure 9.
Schematic representation of injection methods method.
2.3.4. Detergent removal method
In this method, lipids and a high critical micelle concentration (CMC) surfactant were dissolved in a suitable organic solvent using a round bottom flask. A thin film was obtained at the bottom of the flask after solvent gentle evaporation [155]. A mixed micelles solution then obtained by hydrating the lipid film in an aqueous solution containing the drug molecules [156]. The surfactant is then removed by dialysis, size-exclusion chromatography, adsorption onto hydrophobic beads or dilution [157, 158, 159, 160]. Finally, a LUVs liposomes vesicle will be formulated after solution concentration [161]. A main drawback of this method is that most hydrophilic drugs are separated from the liposomes during detergent removal step [162].
2.3.5. Dehydration-rehydration method
It is an organic solvent free method to produce LUVs using sonication. This method based on direct dispersing of the lipids at low concentrations into an aqueous solution containing the drug molecules followed by sonication [163]. First, the dehydration step to evaporate water under nitrogen to create multilayered film entrapping the drug molecules. Then, a hydration step to form large vesicles encapsulating the drug molecules [50, 163]. This method is simple but with high heterogeneity of the liposomes sizes [164].
2.3.6. Heating method
It is also an organic solvent free technique. In this method, lipids are hydrated directly with aqueous solution, and heated for not less than one hour above the Tm of the used phospholipids in the presence of a 3–5 % hydrating agent as glycerin or propylene glycol. The suspension can be heated up to 100 °C when adding cholesterol to the formulation [165]. The hydrating agents act as a stabilizer and isotonizing additives that prevent nanoparticle coagulation and sedimentation. Moreover. The hydration agents provide a cryoprotective effect that makes the heating method an efficient method for the formulation of powder inhalable liposomes [166].
2.3.7. pH jumping method
Another solvent-free method for liposomes preparation is the pH jumping method. In this method, the aqueous solution of phosphatidic acid and phosphatidylcholine are exposed to almost four-fold increase in pH over a short time to break down MLVs into SUVs [167, 168]. The ratio of phosphatidic acid: phosphatidyl choline determine the percentage of SUVs versus LUVs produced [169].
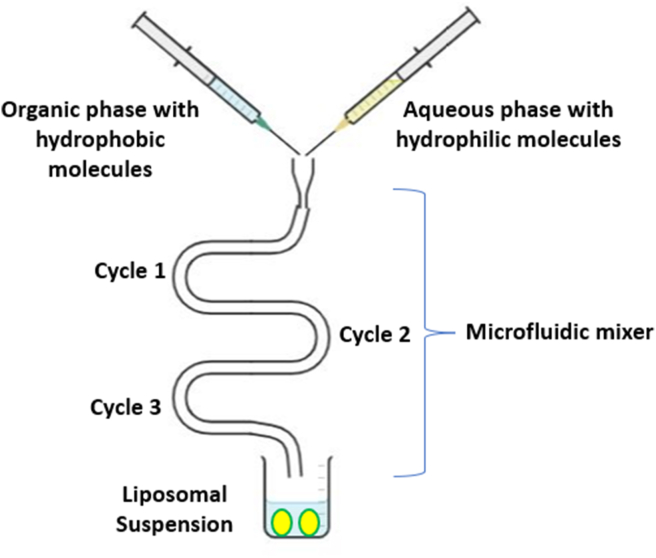
2.3.8. Microfluidic channel method
The microfluidic channel method (Figure 10) has been recently proposed as a novel method for liposomes preparation. Microfluidics provides a tool to employ liquids within microscopic channels [170]. In this method, lipids are dissolved in ethanol or isopropanol, and the resultant solution is injected upright or in the opposite direction to the aqueous medium within the micro-channels. This method involves continuous axial mixing of the organic and aqueous solutions leads to liposomes formation. Liposomes are stabilized using surfactants to avoid coagulation and separation [171]. Microfluidic channel methods control the mixing process of organic and aqueous phases to achieve reproducible liposomes with proper average size, polydispersity, morphology, and lamellarity [172].
Figure 10.
Schematic representation of injection methods method.
2.3.9. Supercritical fluidic method
This method utilized a supercritical fluid, carbon dioxide (CO2), to dissolve lipids instead of using organic solvents. A high-performance liquid pump provides a continuous flow of the aqueous phase into a cell that contains the supercritical lipid solution, allowing phase transition of the dissolved phospholipids [173]. Upon abrupt decrease in pressure, liposomes will formed after completely removing of CO2. 5-fold higher encapsulation efficiencies were obtained by this method. This method suffers from high cost, low yield, and special infrastructures even with using the environmentally safe and cheap carbon dioxide [174].
2.4. Post preparation handlings
2.4.1. Freeze-thaw cycles
This technique is usually used during liposomes preparations to increase the encapsulation efficiency and to enhance liposome lamellarity. This approach utilized a freeze-thaw cycles between -196 °C in liquid nitrogen and below the transition temperature of the used phospholipids lipids [175, 176].
2.4.2. Freeze-drying (lyophilization)
This treatment is applied to preserve the liposomal products and improve their shelf stability. Freeze-drying involves deep freezing of the liposomes suspension after mixing with a cryoprotective, mainly 5–10 % sucrose or trehalose [177]. Then, a sublimation step at very low temperature and a reduced vacuum was applied to convert the liquid samples to fluffy solid particulates. Lyophilization becomes essential treatment for liposomes encapsulating thermo-sensitive biomolecules [178].
2.5. Liposomes characterization
Liposome physiochemical characterization include average size and size distribution (or polydispersity index (PDI)), surface charge (or Zeta potential), shape and morphology, lamellarity, encapsulation efficiency, phase behavior (or polymorphism) and in vitro release profile (Table 2).
Table 2.
Represent different techniques used for the assessment of liposome parameters.
| Liposomes characteristics | Characterization technique | References |
|---|---|---|
| Average particle size | Dynamic light scattering (DLS) and microscope technology: Scanning and transmission electron microscopy (SEM/TEM), cryogenic-TEM (Cryo-TEM), and atomic force microscopy (AFM) | [179, 180] |
| Zeta potential/Surface charge | Electrophoretic mobility, DLS | [181] |
| Particle shape/morphology | TEM, Cryo-TEM, and AFM | [182] |
| Lamellarity | Cryo-TEM and 31P-NMR | [182] |
| Phase behavior | X-ray diffraction (XRD), differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA) | [183, 184] |
| Encapsulation efficiency/Drug release | Centrifugation, dialysis followed by drug content determination using chromatographic and/or spectrophotometric methods | [185, 186] |
2.6. Liposomes drug loading
Liposomes drug loading can be attained by passive or active approaches [103]. Passive loading entraps hydrophilic drug in the liposomes aqueous core during lipid bilayer formation, while hydrophobic drugs accumulate in the small-sized hydrophobic lipid bilayer [103, 187, 188, 189]. Passive loading suffers from bilayer destabilization, high drug/lipid ratio, and rapid drug release [101]. Therefore, improving the aqueous solubility of these hydrophobic drugs by cyclodextrin host-guest complexation were successfully applied and permitliposomes aqueous core loading by forming drug-in-cyclodextrins-in-liposomes delivery system [190].
Active or remote loading has been developed to ensure high encapsulation efficiency of precious chemotherapeutic agents [191]. Remote loading can be achieved into preformed liposomes by pH gradient and/or potential ionic differences across liposomal bilayer membranes [101,187, 192]. The success of intraliposomal remote loading are govern by to main parameters, (i) drug aqueous solubility (ii) presence of an ionizable functional group in drug chemical [192, 193, 194].
Intraliposomal active loading of hydrophobic drugs in response to ionic and/or pH gradients across the liposomes bilayer was developed [194, 195]. This procedure enables hydrophobic drugs to accumulate inside the liposomes core after the vesicles are created. The advantage of this method is that the loading of the drug can be performed independently of liposomes preparation conditions [101]. Most potentially active drugs are weak bases possessing primary, secondary, or tertiary amine functional groups that can be loaded in response to pH gradients [196]. Drugs that are not weak bases, or do not have an ionizable functional group, can be converted to weak base prodrugs or encapsulated with amino-modified carriers as cyclodextrins, therefore allowing encapsulation and intraliposomal retention [193, 197, 198].
3. Protein corona fingerprints of liposomes
Liposomes have been used to overcome many problems associated with low efficiency of anticancer drugs [199]. Recently, a concept is emerging that the limited success of liposomal drugs in clinical practice due to poor knowledge of liposomes behavior in vivo. Lipid vesicles are usually covered by plasma proteins in vivo forming a biomolecular coating, referred to as the protein corona (PrC) [200]. Recent studies verified that PrC fingerprints (PrCFs) enhanced liposome attachment with cancer cells, triggering efficient particle localization and internalization [201].
Accordingly, enrichment in PrCFs was utilized to predict the targeting ability of synthesized liposomal formulations. Palchetti et al reported that the targeting capability of liposome–protein complexes clearly relate with cellular uptake in pancreatic adenocarcinoma (PANC-1) and insulinoma (INS-1) cells as quantified by flow-assisted cell sorting (FACS). The results showed that cellular uptake of the liposomal formulation with the highest abundance of PrCFs was much larger than that of Onivyde®, an Irinotecan liposomal drug approved by the Food and Drug Administration in 2015 for the treatment of metastatic Pancreatic ductal adenocarcinoma (PDAC) [201]. Furthermore, Digiacomo et al identified a potential protein biomarker for pancreatic ductal adenocarcinoma (PDAC) by utilizing liposomes to accumulate PrC coating layer from human plasma proteins. These targeting liposomes may be used for the early diagnosis of PDAC [202]. This approach could open the interesting possibility to identify novel biomarkers for liposomes formulations in the context of personalized medicine.
4. Liposomes in clinical applications
Various liposomal-based formulations were successfully implemented in clinical fields as antitumor, anti-fungal therapies, analgesics [203]. Doxil® was the first approved clinical anticancer liposome drug in the USA (1995). It opened the way to several other liposomal formulations to get to the clinical application fields by innovating the pH gradient active loading and usage of PEGylation for stealth liposomes [203, 204]. Conventional liposome without PEGylation, can be attractive when circulation half-life is not the goal [205]. DepoFoam™ is mostly used for gradual drug release, thus maintaining a continuous drug supply for long-lasting effect [206].
4.1. Marketed clinical liposomes
4.1.1. Cancer treatment
Doxil® or Caelyx® was presented in 1995 by Sequus Pharmaceuticals. Doxil was designed as a polyethylene glycol coated doxorubicin (DOX) liposome intended for the treatment of Kaposi's sarcoma [204]. LipoDox® is another FDA approved PEGylated liposomal formulation encapsulating DOX manufactured by Sun Pharma in 2012 [207]. Daunorubicin was the second anthracycline antineoplastic drug loaded in liposomes to treat acute myeloid leukemia (AML) under the generic name DaunoXome® [208]. Myocet® is a non-PEGylated liposomes encapsulating DOX that showed a shorter circulation half-life with less cardiac side effects [205,209].
Depocyt® consists of Citarabine, a cell-cycle cytotoxic drug, enclosed in the DepoFoam™ multivesicular enclosure, which allows a sustained two-week release [210]. A new liposomes formulation called Mepact® was globally approved for the treatment of osteosarcoma [211]. Vincristine also incorporated into sphingomyelin/cholesterol-based liposome under the name of Marqibo®. This approved formula offered longer circulation time without surface-modified, resulting in a higher accumulation in target tissues in which vincristine is gradually released [212]. Onivyde® is another PEGylated liposome carrying irinotecan and exhibits a long-acting, antitumor effect [213]. In addition, Vyxeos® also known as CPX-351, is composed of a combination of Cytarabine and daunorubicin, encapsulated in a liposome in a ratio of 5:1. This formulation reduced adverse effects with enhanced effectiveness [213, 214]. Finally, Paclitaxel, an anticancer drug, was also incorporated into Lipusu® liposomes to treat gastric carcinoma efficiently with much less adverse effects [215].
4.1.2. Fungal treatment
A two major approved anti-fungal liposomes formulation were Ambisome® and Fungisome®. They encapsulate Amphotericin B anti-fungal drug with many advantages compared free drug [216, 217]. These Amphotericin B liposomes were stabilized in saline and have longer bioavailability and less toxicity and side effects [218, 219].
4.1.3. Photodynamic therapy
Visudyne®: is the only liposomal drug delivery agent approved for age-related macular degeneration therapy by inhibiting the generation of blood vessels in the eye [220].
4.1.4. Pain management
DepoDur™ is a morphine formulation using DepoFoam™ Technology that resulted in a sustained release formula with prolonging the clinical effect time [206]. Exparel® also uses the DepoFoam™ technology to release Bupivacaine for sustained pain relief [197] gradually.
Table 3 summarizes the different clinically approved liposomes formulations in terms of their purpose, lipid constituents, active ingredients, and administration route.
Table 3.
Clinically used liposomes grouped by therapeutic usage.
| Usage | Trade name | Active ingredient(s) | Liposome platform (Molar Ratio) | Manufacturer | Year Approved | Administration Route | References |
|---|---|---|---|---|---|---|---|
| Anti-Cancer | Doxil® | Doxorubicin | HSPC:Cholesterol:PEG 2000-DSPE (56:38:5) | Sequus Pharmaceuticals | 1995 | I.V | [204] |
| DaunoXome® | Daunorubicin | DSPC:Cholesterol (2:1) | NeXstar Pharmaceuticals | 1996 | I.V | [208] | |
| Depocyt® | Cytarabine | DepoFoam™ | SkyPharma Inc. | 1999 | Spinal | [210] | |
| Myocet® | Doxorubicin | Cholesterol:EPC (45:55) | Elan Pharmaceuticals | 2000 | I.V | [209] | |
| Mepact® | Mefamurtide | DOPS:POPC (3:7) Multilamellar liposome | Takeda Pharmaceutical Limited | 2004 | I.V | [211] | |
| Lipodox® | Doxorubicin | DSPC:Cholesterol:PEG 2000-DSPE (56:39:5) | Sun Pharma | 2012 | I.V | [205, 207] | |
| Marqibo® | Vincristine | SM:Cholesterol (60:40) | Talon Therapeutics | 2012 | I.V | [212] | |
| Onivyde™ | Irinotecan | DSPC:Cholesterol:MPEG-2000-DSPE (3:2:0.015) | Merrimack Pharmaceuticals | 2015 | I.V | [213] | |
| Lipusu® | Paclitaxel | NA | Luye Pharma Group | 2006 | I.V | [215] | |
| Vyxeos® | Cytarabine:Daunorubicin 5:1 | DSPC:DSPG:Cholesterol (7:2:1) | Jazz Pharmaceuticals | 2017 | I.V | [214, 221] | |
| Anti-Fungal | Ambisome® | Amphotericin B | HSPC:Cholesterol:DSPG (2:1:0.8) | Astellas Pharma | 1997 | I.V | [216] |
| Fungisome® | Amphotericin B | PC:Cholesterol (7:3) | Lifecare Innovations | 2003 | I.V | [219] | |
| Photodynamic therapy | Visudyne® | Verteporphin | Verteporphin:DMPC&EPG (1:8) | Novartis AG | 2000 | I.V | [220] |
| Analgesic | DepoDur™ | Morphine sulfate | DepoFoam™ | SkyPharma | 2004 | Epidural | [206] |
| Exparel® | Bupivacaine | DepoFoam™ | Pacira pharmaceuticals | 2011 | I.V | [222] |
4.2. Liposomes in clinical trials
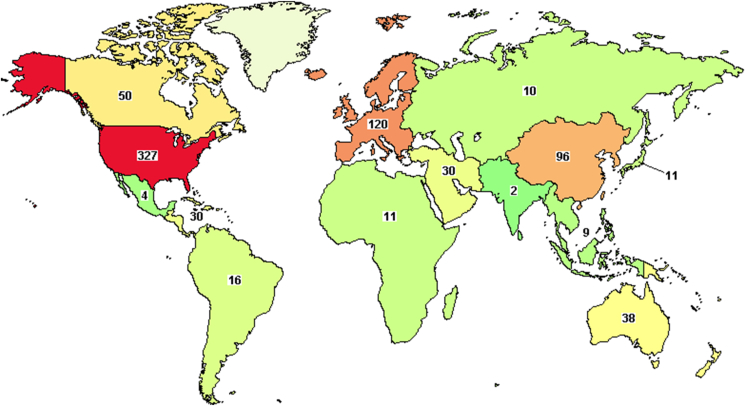
From the 83316 active clinical trials registered, 511 liposomal clinical trials investigating liposomal products which are distributed worldwide as shown in Figure 11. The drugs being examined belong to anticancer drugs, analgesics, immune-modulators, anti-fungal, etc. Among these drugs, 121 of the 511 are in phase III testing, 236 are in phase II, 120 are in phase I, and 6 in early phase I [20].
Figure 11.
Active clinical trials as per 28/dec/2021, source: https://ClinicalTrials.gov.
4.3. Liposomes in vaccinations
Liposome formulations could protect DNA/RNA and proteins payload from biodegradation. Furthermore, their transfection efficiency could be enhanced by modifying surface charge, size, and lipid structure. Two commercial vaccines based on virosome technology are currently on the market, Epaxal® and Inflexal® V (Berna Biotech Ltd, Bern, Switzerland), a hepatitis A vaccine. Virosomes are liposomal formulations that have viral envelope proteins anchored to their lipid membrane [223].
Recently, COVID-19 mRNA based-vaccines utilized liposomes protection to increase their in vitro and in vivo stability [224]. Liposome-based mRNA anti-COVID-19 vaccine has been designed by Pfizer/BioNTech and Moderna, and already administered worldwide. These vaccines were made to maintain the stability of liposomes in blood and to promote immune responses. Their components include distearoyl phosphatidyl choline and cholesterol that considered the main constituents of conventional liposomes [225]. Four major ingredients were used in COVID-19 vaccines: Cationic lipids, for instance 1,2-dioleoyl-3 (trimethylammonium) propane (DOTAP), to binds to the negatively charged mRNA, pegylated lipids stabilize the particle, phospholipids and cholesterol molecules that form the required structure [226]. These formulas encapsulate mRNA, protect it from nucleases, and deliver it into cells, where the mRNA is released and used to generate proteins. During COVID-19 pandemic, many liposome-based vaccines have been developed with great success. Accordingly, mRNA coding for the protein spike of the Coronavirus, would be encapsulated into liposomes that are designed to be stable in the circulating blood until they are taken up by phagocytic cells in the body by endocytosis. The mRNA will then be expressed as the spike protein in turn promoting an immune response to it that will kill or inactivate the invading virus [224].
5. Conclusion
Liposomes were successfully utilized as an efficient drug delivery system for various diseases ranging from cancer treatment to pain managing. The biocompatible, biodegradable, and low immunogenicity liposomes formulation enhanced the pharmacokinetics and pharmacodynamics properties of water insoluble, poor bioavailable and highly toxic drug. Liposomes undergone numerous evolutions in terms of their constituents and manufacturing process to overcome their early limitations. Several liposomes formulation is currently approved in the market to treat various diseases and more than five hundred liposomal formulations are now in different phases of clinical investigation. Nevertheless, liposomes critical challenges are their physical and chemical stability. As a result, there are a n essential need to develop liposomes with high stability significantly impacts their clinical application. Thus, in silico simulation and computational investigations may enable approximate estimation for the best liposomal formulation in their constituents and 3-D structure morphology.
Declarations
Author contribution statement
All authors listed have significantly contributed to the development and the writing of this article.
Funding statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data availability statement
No data was used for the research described in the article.
Declaration of interests statement
The authors declare no conflict of interest.
Additional information
No additional information is available for this paper.
Footnotes
This article is a part of the "Lipid-Based Nanoparticles in Diagnosis and Treatment" Special issue.
References
- 1.Kim B.Y., Rutka J.T., Chan W.C. Nanomedicine. N. Engl. J. Med. 2010;363:2434–2443. doi: 10.1056/NEJMra0912273. [DOI] [PubMed] [Google Scholar]
- 2.Allen T.M., Cullis P.R. Drug delivery systems: entering the mainstream. Science. 2004;303:1818–1822. doi: 10.1126/science.1095833. [DOI] [PubMed] [Google Scholar]
- 3.Khater D., Nsairat H., Odeh F., Saleh M., Jaber A., Alshaer W., Al Bawab A., Mubarak M.S. Design, preparation, and characterization of effective dermal and transdermal lipid nanoparticles: a review. Cosmetics. 2021;8 [Google Scholar]
- 4.Yezhelyev M.V., Gao X., Xing Y., Al-Hajj A., Nie S., O'Regan R.M. Emerging use of nanoparticles in diagnosis and treatment of breast cancer, the Lancet. Oncology. 2006;7:657–667. doi: 10.1016/S1470-2045(06)70793-8. [DOI] [PubMed] [Google Scholar]
- 5.Sahoo S.K., Labhasetwar V. Nanotech approaches to drug delivery and imaging. Drug Discov. Today. 2003;8:1112–1120. doi: 10.1016/s1359-6446(03)02903-9. [DOI] [PubMed] [Google Scholar]
- 6.Nsairat H., Khater D., Odeh F., Al-Adaileh F., Al-Taher S., Jaber A.M., Alshaer W., Al Bawab A., Mubarak M.S. Lipid nanostructures for targeting brain cancer. Heliyon. 2021;7 doi: 10.1016/j.heliyon.2021.e07994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Celia C., Paolino D., Santos H.A. Advanced nanosystems for clinical translation. Adv. Ther. 2021;4:2000215. [Google Scholar]
- 8.Haley B., Frenkel E. Nanoparticles for drug delivery in cancer treatment. Urol. Oncol. 2008;26:57–64. doi: 10.1016/j.urolonc.2007.03.015. [DOI] [PubMed] [Google Scholar]
- 9.Doane T.L., Burda C. The unique role of nanoparticles in nanomedicine: imaging, drug delivery and therapy. Chem. Soc. Rev. 2012;41:2885–2911. doi: 10.1039/c2cs15260f. [DOI] [PubMed] [Google Scholar]
- 10.Peer D., Karp J.M., Hong S., Farokhzad O.C., Margalit R., Langer R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007;2:751–760. doi: 10.1038/nnano.2007.387. [DOI] [PubMed] [Google Scholar]
- 11.Alshaer W., Hillaireau H., Fattal E. Aptamer-guided nanomedicines for anticancer drug delivery. Adv. Drug Deliv. Rev. 2018;134:122–137. doi: 10.1016/j.addr.2018.09.011. [DOI] [PubMed] [Google Scholar]
- 12.Sun T., Zhang Y.S., Pang B., Hyun D.C., Yang M., Xia Y. Engineered nanoparticles for drug delivery in cancer therapy. Angew. Chem. 2014;53:12320–12364. doi: 10.1002/anie.201403036. [DOI] [PubMed] [Google Scholar]
- 13.Jha S., Sharma P.K., Malviya R. Liposomal drug delivery system for cancer therapy: advancement and patents. Recent Pat. Drug Deliv. Formulation. 2016;10:177–183. doi: 10.2174/1872211310666161004155757. [DOI] [PubMed] [Google Scholar]
- 14.Sercombe L., Veerati T., Moheimani F., Wu S.Y., Sood A.K., Hua S. Advances and challenges of liposome assisted drug delivery. Front. Pharmacol. 2015;6:286. doi: 10.3389/fphar.2015.00286. 286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Noble G.T., Stefanick J.F., Ashley J.D., Kiziltepe T., Bilgicer B. Ligand-targeted liposome design: challenges and fundamental considerations. Trends Biotechnol. 2014;32:32–45. doi: 10.1016/j.tibtech.2013.09.007. [DOI] [PubMed] [Google Scholar]
- 16.Hafner A., Lovric J., Lakos G.P., Pepic I. Nanotherapeutics in the EU: an overview on current state and future directions. Int. J. Nanomed. 2014;9:1005–1023. doi: 10.2147/IJN.S55359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.McClements D.J., Rao J. Food-grade nanoemulsions: formulation, fabrication, properties, performance, biological fate, and potential toxicity. Crit. Rev. Food Sci. Nutr. 2011;51:285–330. doi: 10.1080/10408398.2011.559558. [DOI] [PubMed] [Google Scholar]
- 18.Chen T., Gong T., Zhao T., Fu Y., Zhang Z., Gong T. A comparison study between lycobetaine-loaded nanoemulsion and liposome using nRGD as therapeutic adjuvant for lung cancer therapy. Eur. J. Pharmaceut. Sci. 2018;111:293–302. doi: 10.1016/j.ejps.2017.09.041. [DOI] [PubMed] [Google Scholar]
- 19.Fathi S., Oyelere A.K. Liposomal drug delivery systems for targeted cancer therapy: is active targeting the best choice? Future Med. Chem. 2016;8:2091–2112. doi: 10.4155/fmc-2016-0135. [DOI] [PubMed] [Google Scholar]
- 20.Bulbake U., Doppalapudi S., Kommineni N., Khan W. Liposomal formulations in clinical use: an updated review. Pharmaceutics. 2017;9 doi: 10.3390/pharmaceutics9020012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Akbarzadeh A., Rezaei-Sadabady R., Davaran S., Joo S.W., Zarghami N., Hanifehpour Y., Samiei M., Kouhi M., Nejati-Koshki K. Liposome: classification, preparation, and applications. Nanoscale Res. Lett. 2013;8:102. doi: 10.1186/1556-276X-8-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ebrahim S., Peyman G.A., Lee P.J. Applications of liposomes in ophthalmology. Surv. Ophthalmol. 2005;50:167–182. doi: 10.1016/j.survophthal.2004.12.006. [DOI] [PubMed] [Google Scholar]
- 23.He H., Lu Y., Qi J., Zhu Q., Chen Z., Wu W. Adapting liposomes for oral drug delivery. Acta Pharm. Sin. B. 2019;9:36–48. doi: 10.1016/j.apsb.2018.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mehta P.P., Ghoshal D., Pawar A.P., Kadam S.S., Dhapte-Pawar V.S. Technology, Recent advances in inhalable liposomes for treatment of pulmonary diseases: concept to clinical stance. J. Drug Deliv. Sci. Technol. 2020;56:101509. [Google Scholar]
- 25.Pierre M.B.R., Costa I.d.S.M. Liposomal systems as drug delivery vehicles for dermal and transdermal applications. Arch. Dermatol. Res. 2011;303:607–621. doi: 10.1007/s00403-011-1166-4. [DOI] [PubMed] [Google Scholar]
- 26.Barenholz Y.C.J. Doxil®—the First FDA-Approved Nano-Drug: Lessons Learned. J. Controll. Release. 2012;160:117–134. doi: 10.1016/j.jconrel.2012.03.020. [DOI] [PubMed] [Google Scholar]
- 27.Cevenini A., Celia C., Orrù S., Sarnataro D., Raia M., Mollo V., Locatelli M., Imperlini E., Peluso N., Peltrini R., De Rosa E., Parodi A., Del Vecchio L., Di Marzio L., Fresta M., Netti P.A., Shen H., Liu X., Tasciotti E., Salvatore F. Liposome-embedding silicon microparticle for oxaliplatin delivery in tumor chemotherapy. Pharmaceutics. 2020;12 doi: 10.3390/pharmaceutics12060559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kirui D.K., Celia C., Molinaro R., Bansal S.S., Cosco D., Fresta M., Shen H., Ferrari M. Mild hyperthermia enhances transport of liposomal gemcitabine and improves in vivo therapeutic response. Adv. Healthc. Mater. 2015;4:1092–1103. doi: 10.1002/adhm.201400738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sahoo S.K., Labhasetwar V. Nanotech approaches to drug delivery and imaging. Drug Discov. Today. 2003;8:1112–1120. doi: 10.1016/s1359-6446(03)02903-9. [DOI] [PubMed] [Google Scholar]
- 30.Hattori Y., Hu S., Onishi H.J. Effects of cationic lipids in cationic liposomes and disaccharides in the freeze-drying of siRNA lipoplexes on gene silencing in cells by reverse transfection. J. Liposome Res. 2020;30:235–245. doi: 10.1080/08982104.2019.1630643. [DOI] [PubMed] [Google Scholar]
- 31.Laouini A., Jaafar-Maalej C., Limayem-Blouza I., Sfar S., Charcosset C., Fessi H.J. Biotechnology, Preparation, characterization and applications of liposomes: state of the art. J. Coll. Sci. Biotechnol. 2012;1:147–168. [Google Scholar]
- 32.Bulbake U., Doppalapudi S., Kommineni N., Khan W. Liposomal formulations in clinical use: an updated review. Pharmaceutics. 2017;9:12. doi: 10.3390/pharmaceutics9020012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mori A., Klibanov A.L., Torchilin V.P., Huang L. Influence of the steric barrier activity of amphipathic poly (ethyleneglycol) and ganglioside GM1 on the circulation time of liposomes and on the target binding of immunoliposomes in vivo. FEBS Lett. 1991;284:263–266. doi: 10.1016/0014-5793(91)80699-4. [DOI] [PubMed] [Google Scholar]
- 34.Tadokoro S., Hirashima N., Utsunomiya-Tate N.J.B., Bulletin P. Vol. 39. 2016. Effect of Complexin II on membrane fusion between liposomes containing mast cell SNARE proteins; pp. 446–449. [DOI] [PubMed] [Google Scholar]
- 35.Pasut G., Paolino D., Celia C., Mero A., Joseph A.S., Wolfram J., Cosco D., Schiavon O., Shen H., Fresta M. Polyethylene glycol (PEG)-dendron phospholipids as innovative constructs for the preparation of super stealth liposomes for anticancer therapy. J. Contr. Release : Off. J. Controll. Release Soc. 2015;199:106–113. doi: 10.1016/j.jconrel.2014.12.008. [DOI] [PubMed] [Google Scholar]
- 36.Mare R., Paolino D., Celia C., Molinaro R., Fresta M., Cosco D. Post-insertion parameters of PEG-derivatives in phosphocholine-liposomes. Int. J. Pharm. 2018;552:414–421. doi: 10.1016/j.ijpharm.2018.10.028. [DOI] [PubMed] [Google Scholar]
- 37.Leitgeb M., Knez Ž., Primožič M. Sustainable technologies for liposome preparation. J. Supercrit. Fluids. 2020;165:104984. [Google Scholar]
- 38.Ong S.G.M., Ming L.C., Lee K.S., Yuen K.H. Influence of the encapsulation efficiency and size of liposome on the oral bioavailability of griseofulvin-loaded liposomes. Pharmaceutics. 2016;8:25. doi: 10.3390/pharmaceutics8030025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kelly C., Jefferies C., Cryan S.-A.J. Targeted Liposomal Drug Delivery to Monocytes and Macrophages. J. Drug Deliv. 2011:2011. doi: 10.1155/2011/727241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Caracciolo G., Pozzi D., Caminiti R., Amenitsch H. Lipid mixing upon deoxyribonucleic acid-induced liposomes fusion investigated by synchrotron small-angle x-ray scattering. Appl. Phys. Lett. 2005;87:133901. [Google Scholar]
- 41.Storm G., Belliot S.O., Daemen T., Lasic D.D. Surface modification of nanoparticles to oppose uptake by the mononuclear phagocyte system. Adv. Drug Deliv. Rev. 1995;17:31–48. [Google Scholar]
- 42.Bozzuto G., Molinari A. Liposomes as nanomedical devices. Int. J. Nanomed. 2015;10:975. doi: 10.2147/IJN.S68861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Düzgüneş N., Nir S. Mechanisms and kinetics of liposome–cell interactions. Adv. Drug Deliv. Rev. 1999;40:3–18. doi: 10.1016/s0169-409x(99)00037-x. [DOI] [PubMed] [Google Scholar]
- 44.Lee K.D., Nir S., Papahadjopoulos D. Quantitative analysis of liposome-cell interactions in vitro: rate constants of binding and endocytosis with suspension and adherent J774 cells and human monocytes. Biochemistry. 1993;32:889–899. doi: 10.1021/bi00054a021. [DOI] [PubMed] [Google Scholar]
- 45.Kono K., Nakai R., Morimoto K., Takagishi T. Temperature-dependent interaction of thermo-sensitive polymer-modified liposomes with CV1 cells. FEBS Lett. 1999;456:306–310. doi: 10.1016/s0014-5793(99)00975-8. [DOI] [PubMed] [Google Scholar]
- 46.Wu X., Dai X., Liao Y., Sheng M., Shi X. Investigation on drug entrapment location in liposomes and transfersomes based on molecular dynamics simulation. J. Mol. Model. 2021;27:111. doi: 10.1007/s00894-021-04722-3. [DOI] [PubMed] [Google Scholar]
- 47.Nakhaei P., Margiana R., Bokov D.O., Abdelbasset W.K., Jadidi Kouhbanani M.A., Varma R.S., Marofi F., Jarahian M., Beheshtkhoo N. Liposomes: structure, biomedical applications, and stability parameters with emphasis on cholesterol. Front. Bioeng. Biotechnol. 2021;9:705886. doi: 10.3389/fbioe.2021.705886. 705886. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 48.Pavelić Z., Skalko-Basnet N., Jalsenjak I. Characterisation and in vitro evaluation of bioadhesive liposome gels for local therapy of vaginitis. Int. J. Pharm. 2005;301:140–148. doi: 10.1016/j.ijpharm.2005.05.022. [DOI] [PubMed] [Google Scholar]
- 49.Calvagno M.G., Celia C., Paolino D., Cosco D., Iannone M., Castelli F., Doldo P., Frest M. Effects of lipid composition and preparation conditions on physical-chemical properties, technological parameters and in vitro biological activity of gemcitabine-loaded liposomes. Curr. Drug Deliv. 2007;4:89–101. doi: 10.2174/156720107779314749. [DOI] [PubMed] [Google Scholar]
- 50.Large D.E., Abdelmessih R.G., Fink E.A., Auguste D.T. Liposome composition in drug delivery design, synthesis, characterization, and clinical application. Adv. Drug Deliv. Rev. 2021;176:113851. doi: 10.1016/j.addr.2021.113851. [DOI] [PubMed] [Google Scholar]
- 51.Daraee H., Etemadi A., Kouhi M., Alimirzalu S., Akbarzadeh A. Application of liposomes in medicine and drug delivery. Artif. Cell Nanomed. Biotechnol. 2016;44:381–391. doi: 10.3109/21691401.2014.953633. [DOI] [PubMed] [Google Scholar]
- 52.Monteiro N., Martins A., Reis R.L., Neves N.M. Liposomes in tissue engineering and regenerative medicine. J. R. Soc. Interface. 2014;11:20140459. doi: 10.1098/rsif.2014.0459. 20140459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Li J., Wang X., Zhang T., Wang C., Huang Z., Luo X., Deng Y. A review on phospholipids and their main applications in drug delivery systems. Asian J. Pharm. Sci. 2015;10:81–98. [Google Scholar]
- 54.Ramrakhiani L., Chand S. Assay Methods, Industrial Potential and Pathogenicity, Applied Biochemistry and Biotechnology. Vol. 164. 2011. Recent progress on phospholipases: different sources; pp. 991–1022. [DOI] [PubMed] [Google Scholar]
- 55.van der Veen J.N., Kennelly J.P., Wan S., Vance J.E., Vance D.E., Jacobs R.L. The critical role of phosphatidylcholine and phosphatidylethanolamine metabolism in health and disease. Biochim. Biophys. Acta Biomembr. 2017;1859:1558–1572. doi: 10.1016/j.bbamem.2017.04.006. [DOI] [PubMed] [Google Scholar]
- 56.van Hoogevest P., Wendel A. The use of natural and synthetic phospholipids as pharmaceutical excipients. Eur. J. Lipid Sci. Technol. 2014;116:1088–1107. doi: 10.1002/ejlt.201400219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lordan R., Tsoupras A., Zabetakis I. Phospholipids of animal and marine origin: structure, function, and anti-inflammatory properties. Molecules. 2017;22:1964. doi: 10.3390/molecules22111964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Briuglia M.L., Rotella C., McFarlane A., Lamprou D.A. Influence of cholesterol on liposome stability and on in vitro drug release. Drug Deliv. Transl. Res. 2015;5:231–242. doi: 10.1007/s13346-015-0220-8. [DOI] [PubMed] [Google Scholar]
- 59.Jovanović A.A., Balanč B.D., Ota A., Ahlin Grabnar P., Djordjević V.B., Šavikin K.P., Bugarski B.M., Nedović V.A., Poklar Ulrih N. Comparative effects of cholesterol and β-sitosterol on the liposome membrane characteristics. Eur. J. Lipid Sci. Technol. 2018;120:1800039. [Google Scholar]
- 60.Cipolla D., Wu H., Gonda I., Eastman S., Redelmeier T., Chan H.-K. Modifying the release properties of liposomes toward personalized medicine. J. Pharmaceut. Sci. 2014;103:1851–1862. doi: 10.1002/jps.23969. [DOI] [PubMed] [Google Scholar]
- 61.Bozzuto G., Molinari A. Liposomes as nanomedical devices. Int. J. Nanomed. 2015;10:975–999. doi: 10.2147/IJN.S68861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lee E., Kim A., Oh Y.-K., Kim C.-K. Effect of edge activators on the formation and transfection efficiency of ultradeformable liposomes. Biomaterials. 2005;26:205–210. doi: 10.1016/j.biomaterials.2004.02.020. [DOI] [PubMed] [Google Scholar]
- 63.Tian Y., Chen L., Zhang W. Influence of ionic surfactants on the properties of nanoemulsions emulsified by nonionic surfactants span 80/tween 80. J. Dispersion Sci. Technol. 2016;37:1511–1517. [Google Scholar]
- 64.de la Maza A., Coderch L., Lopez O., Baucells J., Parra J.L. Permeability changes caused by surfactants in liposomes that model the stratum corneum lipid composition. J. Am. Oil Chem. Soc. 1997;74:1–8. [Google Scholar]
- 65.Gangwar M., Singh R., Goel R.K., Nath G. Recent advances in various emerging vescicular systems: an overview. Asian Pac. J. Trop. Biomed. 2012;2:S1176–S1188. [Google Scholar]
- 66.Paolino D., Cosco D., Cilurzo F., Trapasso E., Morittu V.M., Celia C., Fresta M. Improved in vitro and in vivo collagen biosynthesis by asiaticoside-loaded ultradeformable vesicles. J. Contr. Release : Off. J. Controll. Release Soc. 2012;162:143–151. doi: 10.1016/j.jconrel.2012.05.050. [DOI] [PubMed] [Google Scholar]
- 67.Chen R., Li R., Liu Q., Bai C., Qin B., Ma Y. J. Han, ultradeformable liposomes: a novel vesicular carrier for enhanced transdermal delivery of procyanidins: effect of surfactants on the formation, stability, and transdermal delivery. AAPS PharmSciTech. 2017;18:1823–1832. doi: 10.1208/s12249-016-0661-5. [DOI] [PubMed] [Google Scholar]
- 68.Souto E.B., Macedo A.S., Dias-Ferreira J., Cano A., Zielińska A., Matos C.M. Elastic and ultradeformable liposomes for transdermal delivery of active pharmaceutical ingredients (APIs) Int. J. Mol. Sci. 2021;22 doi: 10.3390/ijms22189743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.A.D. Bangham, M.W. Hill, N. Miller, Preparation and use of liposomes as models of biological membranes, Methods Member. Biol., Springer1974, pp. 1-68.
- 70.Gonzalez-Rodriguez M., Rabasco A. Charged liposomes as carriers to enhance the permeation through the skin. Expet Opin. Drug Deliv. 2011;8:857–871. doi: 10.1517/17425247.2011.574610. [DOI] [PubMed] [Google Scholar]
- 71.V. Torchilin, PEGylated Pharmaceutical Nanocarriers, Long Acting Injections and Implants, Springer2012, pp. 263-293.
- 72.Sapra P., Tyagi P., Allen T.M. Ligand-targeted liposomes for cancer treatment. Curr. Drug Deliv. 2005;2:369–381. doi: 10.2174/156720105774370159. [DOI] [PubMed] [Google Scholar]
- 73.Bibi S., Lattmann E., Mohammed A.R., Perrie Y. Trigger release liposome systems: local and remote controlled delivery? J. Microencapsul. 2012;29:262–276. doi: 10.3109/02652048.2011.646330. [DOI] [PubMed] [Google Scholar]
- 74.Negishi Y., Ishii Y., Shiono H., Akiyama S., Sekine S., Kojima T., Mayama S., Kikuchi T., Hamano N., Endo-Takahashi Y., Suzuki R., Maruyama K., Aramaki Y. Bubble liposomes and ultrasound exposure improve localized morpholino oligomer delivery into the skeletal muscles of dystrophic mdx mice. Mol. Pharm. 2014;11:1053–1061. doi: 10.1021/mp4004755. [DOI] [PubMed] [Google Scholar]
- 75.Bangham A. A correlation between surface charge and coagulant action of phospholipids. Nature. 1961;192:1197–1198. doi: 10.1038/1921197a0. [DOI] [PubMed] [Google Scholar]
- 76.Wu H., Yu M., Miao Y., He S., Dai Z., Song W., Liu Y., Song S., Ahmad E., Wang D., Gan Y. Cholesterol-tuned liposomal membrane rigidity directs tumor penetration and anti-tumor effect. Acta Pharm. Sin. B. 2019;9:858–870. doi: 10.1016/j.apsb.2019.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kaddah S., Khreich N., Kaddah F., Charcosset C., Greige-Gerges H. Cholesterol modulates the liposome membrane fluidity and permeability for a hydrophilic molecule. Food Chem. Toxicol. 2018;113:40–48. doi: 10.1016/j.fct.2018.01.017. [DOI] [PubMed] [Google Scholar]
- 78.Jovanović A., Balanč B., Pravilović R., Ota A., Ulrih N., Nedović V., Branko B. 2017. Influence of Cholesterol on Liposomal Membrane Fluidity, Liposome Size and Zeta Potential. [Google Scholar]
- 79.Ceh B., Winterhalter M., Frederik P.M., Vallner J.J., Lasic D.D. Stealth® liposomes: from theory to product. Adv. Drug Deliv. Rev. 1997;24:165–177. [Google Scholar]
- 80.Senior J., Gregoriadis G. Stability of small unilamellar liposomes in serum and clearance from the circulation: the effect of the phospholipid and cholesterol components. Life Sci. 1982;30:2123–2136. doi: 10.1016/0024-3205(82)90455-6. [DOI] [PubMed] [Google Scholar]
- 81.Zhang J.-X., Wang K., Mao Z.-F., Fan X., Jiang D.-L., Chen M., Cui L., Sun K., Dang S.-C. Application of liposomes in drug development—focus on gastroenterological targets. Int. J. Nanomed. 2013;8:1325. doi: 10.2147/IJN.S42153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Klibanov A.L., Maruyama K., Beckerleg A.M., Torchilin V.P., Huang L. Activity of amphipathic poly (ethylene glycol) 5000 to prolong the circulation time of liposomes depends on the liposome size and is unfavorable for immunoliposome binding to target. Biochim. Biophys. Acta Biomembr. 1991;1062:142–148. doi: 10.1016/0005-2736(91)90385-l. [DOI] [PubMed] [Google Scholar]
- 83.San H., Yang Z.-Y., Pompili V.J., Jaffe M.L., Plautz G.E., Xu L., Felgner J.H., Wheeler C.J., Felgner P.L., Gao X. Safety and short-term toxicity of a novel cationic lipid formulation for human gene therapy. Hum. Gene Ther. 1993;4:781–788. doi: 10.1089/hum.1993.4.6-781. [DOI] [PubMed] [Google Scholar]
- 84.Majzoub R.N., Ewert K.K., Safinya C.R. Physical, E. Sciences, Cationic liposome–nucleic acid nanoparticle assemblies with applications in gene delivery and gene silencing. Math. Phys. Eng. Sci. 2016;374:20150129. doi: 10.1098/rsta.2015.0129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Dass C.R. Improving anti-angiogenic therapy via selective delivery of cationic liposomes to tumour vasculature. Int. J. Pharm. 2003;267:1–12. doi: 10.1016/j.ijpharm.2003.08.010. [DOI] [PubMed] [Google Scholar]
- 86.F. Lai, M. Schlich, C. Sinico, A.M. Fadda, Liposomes as Brain Targeted Delivery Systems, Nanomedicines for Brain Drug Delivery, Springer2021, pp. 29-59.
- 87.Youn P., Chen Y., Furgeson D.Y. A myristoylated cell-penetrating peptide bearing a transferrin receptor-targeting sequence for neuro-targeted siRNA delivery. Mol. Pharm. 2014;11:486–495. doi: 10.1021/mp400446v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Hervé F., Ghinea N., Scherrmann J.-M. CNS delivery via adsorptive transcytosis. AAPS J. 2008;10:455–472. doi: 10.1208/s12248-008-9055-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Mc Carthy D.J., Malhotra M., O’Mahony A.M., Cryan J.F., O’Driscoll C.M. Nanoparticles and the blood-brain barrier: advancing from in-vitro models towards therapeutic significance. Pharmaceut. Res. 2015;32:1161–1185. doi: 10.1007/s11095-014-1545-6. [DOI] [PubMed] [Google Scholar]
- 90.Knudsen K.B., Northeved H., Ek P.K., Permin A., Gjetting T., Andresen T.L., Larsen S., Wegener K.M., Lykkesfeldt J., Jantzen K.J.N.N. In vivo toxicity of cationic micelles and liposomes. Vol. 11. 2015. Biology, medicine; pp. 467–477. [DOI] [PubMed] [Google Scholar]
- 91.Semple S.C., Chonn A., Cullis P.R. Interactions of liposomes and lipid-based carrier systems with blood proteins: relation to clearance behaviour in vivo. Adv. Drug Deliv. Rev. 1998;32:3–17. doi: 10.1016/s0169-409x(97)00128-2. [DOI] [PubMed] [Google Scholar]
- 92.Suk J.S., Xu Q., Kim N., Hanes J., Ensign L.M. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv. Drug Deliv. Rev. 2016;99:28–51. doi: 10.1016/j.addr.2015.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Gregoriadis G., Neerunjun D.E. Control of the rate of hepatic uptake and catabolism of liposome-entrapped proteins injected into rats. Possible therapeutic applications. Eur. J. Biochem. 1974;47:179–185. doi: 10.1111/j.1432-1033.1974.tb03681.x. [DOI] [PubMed] [Google Scholar]
- 94.Gonzalez-Rodriguez M., Rabasco A. Charged liposomes as carriers to enhance the permeation through the skin. Expet Opin. Drug Deliv. 2011;8:857–871. doi: 10.1517/17425247.2011.574610. [DOI] [PubMed] [Google Scholar]
- 95.Rangger C., Helbok A., von Guggenberg E., Sosabowski J., Radolf T., Prassl R., Andreae F., Thurner G.C., Haubner R., Decristoforo C. Influence of PEGylation and RGD loading on the targeting properties of radiolabeled liposomal nanoparticles. Int. J. Nanomed. 2012;7:5889. doi: 10.2147/IJN.S36847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Sunamoto J., Iwamoto K. Protein-coated and polysaccharide-coated liposomes as drug carriers. J. Crit. Rev. Therapeut. Drug Carr. Syst. 1986;2:117–136. [PubMed] [Google Scholar]
- 97.Allen T. The use of glycolipids and hydrophilic polymers in avoiding rapid uptake of liposomes by the mononuclear phagocyte system. J. Adv. Drug Deliv. Rev. 1994;13:285–309. [Google Scholar]
- 98.Paolino D., Cosco D., Racanicchi L., Trapasso E., Celia C., Iannone M., Puxeddu E., Costante G., Filetti S., Russo D., Fresta M. Gemcitabine-loaded PEGylated unilamellar liposomes vs GEMZAR: biodistribution, pharmacokinetic features and in vivo antitumor activity. J. Contr. Release : Off. J. Controll. Release Soc. 2010;144:144–150. doi: 10.1016/j.jconrel.2010.02.021. [DOI] [PubMed] [Google Scholar]
- 99.He K., Tang M.J.C.-b.i. Safety of novel liposomal drugs for cancer treatment. Adv. Prosp. 2018;295:13–19. doi: 10.1016/j.cbi.2017.09.006. [DOI] [PubMed] [Google Scholar]
- 100.Vaage J., Donovan D., Mayhew E., Abra R., Huang A. Therapy of human ovarian carcinoma xenografts using doxorubicin encapsulated in sterically stabilized liposomes. Cancer. 1993;72:3671–3675. doi: 10.1002/1097-0142(19931215)72:12<3671::aid-cncr2820721219>3.0.co;2-u. [DOI] [PubMed] [Google Scholar]
- 101.Allen T.M., Cullis P.R. Liposomal drug delivery systems: from concept to clinical applications. Adv. Drug Deliv. Rev. 2013;65:36–48. doi: 10.1016/j.addr.2012.09.037. [DOI] [PubMed] [Google Scholar]
- 102.Leserman L.D., Weinstein J.N., Blumenthal R., Terry W.D. Receptor-mediated endocytosis of antibody-opsonized liposomes by tumor cells. Proc. Natl. Acad. Sci. U. S. A. 1980;77:4089–4093. doi: 10.1073/pnas.77.7.4089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Gubernator J. Active methods of drug loading into liposomes: recent strategies for stable drug entrapment and increased in vivo activity. Expet Opin. Drug Deliv. 2011;8:565–580. doi: 10.1517/17425247.2011.566552. [DOI] [PubMed] [Google Scholar]
- 104.Alshaer W., Hillaireau H., Vergnaud J., Ismail S., Fattal E. Functionalizing liposomes with anti-CD44 aptamer for selective targeting of cancer cells. Bioconjugate Chem. 2015;26:1307–1313. doi: 10.1021/bc5004313. [DOI] [PubMed] [Google Scholar]
- 105.Kaneda Y. Virosomes: evolution of the liposome as a targeted drug delivery system. Adv. Drug Deliv. Rev. 2000;43:197–205. doi: 10.1016/s0169-409x(00)00069-7. [DOI] [PubMed] [Google Scholar]
- 106.d'Avanzo N., Torrieri G., Figueiredo P., Celia C., Paolino D., Correia A., Moslova K., Teesalu T., Fresta M., Santos H.A. LinTT1 peptide-functionalized liposomes for targeted breast cancer therapy. Int. J. Pharm. 2021;597:120346. doi: 10.1016/j.ijpharm.2021.120346. [DOI] [PubMed] [Google Scholar]
- 107.Paolino D., Cosco D., Gaspari M., Celano M., Wolfram J., Voce P., Puxeddu E., Filetti S., Celia C., Ferrari M., Russo D., Fresta M. Targeting the thyroid gland with thyroid-stimulating hormone (TSH)-nanoliposomes. Biomaterials. 2014;35:7101–7109. doi: 10.1016/j.biomaterials.2014.04.088. [DOI] [PubMed] [Google Scholar]
- 108.Torchilin V.P. Recent advances with liposomes as pharmaceutical carriers, Nature reviews. Drug Discov. 2005;4:145–160. doi: 10.1038/nrd1632. [DOI] [PubMed] [Google Scholar]
- 109.Gabizon A.A., Shmeeda H., Zalipsky S. Pros and cons of the liposome platform in cancer drug targeting. J. Liposome Res. 2006;16:175–183. doi: 10.1080/08982100600848769. [DOI] [PubMed] [Google Scholar]
- 110.Rezler E.M., Khan D.R., Lauer-Fields J., Cudic M., Baronas-Lowell D., Fields G.B. Targeted drug delivery utilizing protein-like molecular architecture. J. Am. Chem. Soc. 2007;129:4961–4972. doi: 10.1021/ja066929m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Koren E., Apte A., Jani A., Torchilin V.P. Multifunctional PEGylated 2C5-immunoliposomes containing pH-sensitive bonds and TAT peptide for enhanced tumor cell internalization and cytotoxicity. J. Contr. Release : Off. J. Controll. Release Soc. 2012;160:264–273. doi: 10.1016/j.jconrel.2011.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Zhou X., Zhang M., Yung B., Li H., Zhou C., Lee L.J., Lee R.J. Lactosylated liposomes for targeted delivery of doxorubicin to hepatocellular carcinoma. Int. J. Nanomed. 2012;7:5465–5474. doi: 10.2147/IJN.S33965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Kirpotin D.B., Drummond D.C., Shao Y., Shalaby M.R., Hong K., Nielsen U.B., Marks J.D., Benz C.C., Park J.W. Antibody targeting of long-circulating lipidic nanoparticles does not increase tumor localization but does increase internalization in animal models. Cancer Res. 2006;66:6732–6740. doi: 10.1158/0008-5472.CAN-05-4199. [DOI] [PubMed] [Google Scholar]
- 114.Nellis D.F., Giardina S.L., Janini G.M., Shenoy S.R., Marks J.D., Tsai R., Drummond D.C., Hong K., Park J.W., Ouellette T.F., Perkins S.C., Kirpotin D.B. Preclinical manufacture of anti-HER2 liposome-inserting, scFv-PEG-lipid conjugate. 2. Conjugate micelle identity, purity, stability, and potency analysis. Biotechnol. Prog. 2005;21:221–232. doi: 10.1021/bp049839z. [DOI] [PubMed] [Google Scholar]
- 115.Suzuki R., Takizawa T., Kuwata Y., Mutoh M., Ishiguro N., Utoguchi N., Shinohara A., Eriguchi M., Yanagie H., Maruyama K. Effective anti-tumor activity of oxaliplatin encapsulated in transferrin-PEG-liposome. Int. J. Pharm. 2008;346:143–150. doi: 10.1016/j.ijpharm.2007.06.010. [DOI] [PubMed] [Google Scholar]
- 116.Jaracz S., Chen J., Kuznetsova L.V., Ojima I. Recent advances in tumor-targeting anticancer drug conjugates. Bioorg. Med. Chem. 2005;13:5043–5054. doi: 10.1016/j.bmc.2005.04.084. [DOI] [PubMed] [Google Scholar]
- 117.Jin E., Zhang B., Sun X., Zhou Z., Ma X., Sun Q., Tang J., Shen Y., Van Kirk E., Murdoch W.J., Radosz M. Acid-active cell-penetrating peptides for in vivo tumor-targeted drug delivery. J. Am. Chem. Soc. 2013;135:933–940. doi: 10.1021/ja311180x. [DOI] [PubMed] [Google Scholar]
- 118.Jiang F., Liu B., Lu J., Li F., Li D., Liang C., Dang L., Liu J., He B., Badshah S.A., Lu C., He X., Guo B., Zhang X.B., Tan W., Lu A., Zhang G. Progress and challenges in developing aptamer-functionalized targeted drug delivery systems. Int. J. Mol. Sci. 2015;16:23784–23822. doi: 10.3390/ijms161023784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Nsairat H., Mahmoud I.S., Odeh F., Abuarqoub D., Al-Azzawi H., Zaza R., Qadri M.I., Ismail S., Al Bawab A., Awidi A., Alshaer W. Grafting of anti-nucleolin aptamer into preformed and remotely loaded liposomes through aptamer-cholesterol post-insertion. RSC Adv. 2020;10:36219–36229. doi: 10.1039/d0ra07325c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Odeh F., Nsairat H., Alshaer W., Ismail M.A., Esawi E., Qaqish B., Bawab A.A., Ismail S.I. Aptamers chemistry: chemical modifications and conjugation strategies. Molecules. 2020:25. doi: 10.3390/molecules25010003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Gupta S., Gupta M.K. Possible role of nanocarriers in drug delivery against cervical cancer. Nano Rev. Exp. 2017;8:1335567. doi: 10.1080/20022727.2017.1335567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Backer M.V., Gaynutdinov T.I., Patel V., Jehning B.T., Myshkin E., Backer J.M. Adapter protein for site-specific conjugation of payloads for targeted drug delivery. Bioconjugate Chem. 2004;15:1021–1029. doi: 10.1021/bc0499477. [DOI] [PubMed] [Google Scholar]
- 123.Chau Y., Tan F.E., Langer R. Synthesis and characterization of dextran-peptide-methotrexate conjugates for tumor targeting via mediation by matrix metalloproteinase II and matrix metalloproteinase IX. Bioconjugate Chem. 2004;15:931–941. doi: 10.1021/bc0499174. [DOI] [PubMed] [Google Scholar]
- 124.Bertrand N., Leroux J.C. The journey of a drug-carrier in the body: an anatomo-physiological perspective. J. Contr. Release : Off. J. Controll. Release Soc. 2012;161:152–163. doi: 10.1016/j.jconrel.2011.09.098. [DOI] [PubMed] [Google Scholar]
- 125.Mura S., Nicolas J., Couvreur P. Stimuli-responsive nanocarriers for drug delivery. Nat. Mater. 2013;12:991–1003. doi: 10.1038/nmat3776. [DOI] [PubMed] [Google Scholar]
- 126.Lee Y., Thompson D.H. Vol. 9. Wiley Interdiscip Rev Nanomed Nanobiotechnol; 2017. (Stimuli-responsive Liposomes for Drug Delivery). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Pham S.H., Choi Y., Choi J. Stimuli-responsive nanomaterials for application in antitumor therapy and drug delivery. Pharmaceutics. 2020;12:630. doi: 10.3390/pharmaceutics12070630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Rayamajhi S., Marchitto J., Nguyen T.D.T., Marasini R., Celia C., Aryal S. pH-responsive cationic liposome for endosomal escape mediated drug delivery. Colloids Surf. B Biointerfaces. 2020;188:110804. doi: 10.1016/j.colsurfb.2020.110804. [DOI] [PubMed] [Google Scholar]
- 129.Paliwal S.R., Paliwal R., Vyas S.P. A review of mechanistic insight and application of pH-sensitive liposomes in drug delivery. Drug Deliv. 2015;22:231–242. doi: 10.3109/10717544.2014.882469. [DOI] [PubMed] [Google Scholar]
- 130.Wang Y., Shim M.S., Levinson N.S., Sung H.W., Xia Y. Stimuli-responsive materials for controlled release of theranostic agents. Adv. Funct. Mater. 2014;24:4206–4220. doi: 10.1002/adfm.201400279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Yavlovich A., Smith B., Gupta K., Blumenthal R., Puri A. Light-sensitive lipid-based nanoparticles for drug delivery: design principles and future considerations for biological applications. Mol. Membr. Biol. 2010;27:364–381. doi: 10.3109/09687688.2010.507788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Gai S., Yang G., Yang P., He F., Lin J., Jin D., Xing B. Recent advances in functional nanomaterials for light–triggered cancer therapy. Nano Today. 2018;19:146–187. [Google Scholar]
- 133.Mills J.K., Needham D. Lysolipid incorporation in dipalmitoylphosphatidylcholine bilayer membranes enhances the ion permeability and drug release rates at the membrane phase transition. Biochim. Biophys. Acta Biomembr. 2005;1716:77–96. doi: 10.1016/j.bbamem.2005.08.007. [DOI] [PubMed] [Google Scholar]
- 134.Ta T., Porter T.M. Thermosensitive liposomes for localized delivery and triggered release of chemotherapy. J. Contr. Release. 2013;169:112–125. doi: 10.1016/j.jconrel.2013.03.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Chu K.F., Dupuy D.E. Thermal ablation of tumours: biological mechanisms and advances in therapy. Nat. Rev. Cancer. 2014;14:199–208. doi: 10.1038/nrc3672. [DOI] [PubMed] [Google Scholar]
- 136.Nardecchia S., Sánchez-Moreno P., Vicente J.d., Marchal J.A., Boulaiz H. Clinical trials of thermosensitive nanomaterials: an overview. Nanomaterials. 2019;9:191. doi: 10.3390/nano9020191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Pezzoli D., Tallarita E., Rosini E., Candiani G. Characterization and investigation of redox-sensitive liposomes for gene delivery. Methods Mol. Biol. 2016;1445:217–233. doi: 10.1007/978-1-4939-3718-9_14. [DOI] [PubMed] [Google Scholar]
- 138.Ong W., Yang Y., Cruciano A.C., McCarley R.L. Redox-Triggered contents release from liposomes. J. Am. Chem. Soc. 2008;130:14739–14744. doi: 10.1021/ja8050469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Antoniou A.I., Giofrè S., Seneci P., Passarella D., Pellegrino S. Stimulus-responsive liposomes for biomedical applications. Drug Discov. Today. 2021;26:1794–1824. doi: 10.1016/j.drudis.2021.05.010. [DOI] [PubMed] [Google Scholar]
- 140.Li M., Zhao G., Su W.-K., Shuai Q. Vol. 8. 2020. Enzyme-Responsive Nanoparticles for Anti-tumor Drug Delivery. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Lou J., Best M.D. A general approach to enzyme-responsive liposomes. Chemistry. 2020;26:8597–8607. doi: 10.1002/chem.202000529. [DOI] [PubMed] [Google Scholar]
- 142.Chu C.-J., Szoka F.C. pH-Sensitive Liposomes. J. Liposome Res. 1994;4:361–395. [Google Scholar]
- 143.Karanth H., Murthy R.S. pH-sensitive liposomes--principle and application in cancer therapy. J. Pharm. Pharmacol. 2007;59:469–483. doi: 10.1211/jpp.59.4.0001. [DOI] [PubMed] [Google Scholar]
- 144.Lafi Z., Alshaer W., Hatmal M.m.M., Zihlif M., Alqudah D.A., Nsairat H., Azzam H., Aburjai T., Bustanji Y., Awidi A. Aptamer-functionalized pH-sensitive liposomes for a selective delivery of echinomycin into cancer cells. RSC Adv. 2021;11:29164–29177. doi: 10.1039/d1ra05138e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Katsuji S., Nobuki K., Katsuyuki Y., Ryo S., Kazuo M. IEEE Ultrasonics Symposium; 2008. Characterization of Bubble Liposomes by Measurements of Ultrasound Attenuation: Effects of Shell Materials, 2008; pp. 1675–1678. [Google Scholar]
- 146.Sun C., sciences p. 2017. BUBBLE LIPOSOME: A MODERN THERANOSTIC APPROACH OF NEW DRUG DELIVERY; pp. 1290–1314. [Google Scholar]
- 147.Mahanty A., Li Y., Yu Y., Banerjee P., Chaurasiya B., Tu J., Sun C. World Journal of Pharmacy and Pharmaceutical Sciences; 2017. BUBBLE LIPOSOME: A MODERN THERANOSTIC APPROACH OF NEW DRUG DELIVERY; pp. 1290–1314. [Google Scholar]
- 148.Bhupendra P., Narendra K., Suman S., Amit R. Liposome: method of preparation, advantages, evaluation and its application. J. Appl. Pharmaceut. Res. 2015;3:1–8. [Google Scholar]
- 149.Shi N.-Q., Qi X.-R. In: Liposome-Based Drug Delivery Systems. Lu W.-L., Qi X.-R., editors. Springer Berlin Heidelberg; Berlin, Heidelberg: 2017. Preparation of drug liposomes by reverse-phase evaporation; pp. 1–10. [Google Scholar]
- 150.Szoka F., Jr., Papahadjopoulos D. Comparative properties and methods of preparation of lipid vesicles (liposomes) Annu. Rev. Biophys. Bioeng. 1980;9:467–508. doi: 10.1146/annurev.bb.09.060180.002343. [DOI] [PubMed] [Google Scholar]
- 151.Has C., Sunthar P. A comprehensive review on recent preparation techniques of liposomes. J. Liposome Res. 2020;30:336–365. doi: 10.1080/08982104.2019.1668010. [DOI] [PubMed] [Google Scholar]
- 152.Szoka F., Jr., Papahadjopoulos D. Procedure for preparation of liposomes with large internal aqueous space and high capture by reverse-phase evaporation. Proc. Natl. Acad. Sci. U. S. A. 1978;75:4194–4198. doi: 10.1073/pnas.75.9.4194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Jaafar-Maalej C., Charcosset C., Fessi H. A new method for liposome preparation using a membrane contactor. J. Liposome Res. 2011;21:213–220. doi: 10.3109/08982104.2010.517537. [DOI] [PubMed] [Google Scholar]
- 154.Maherani B., Arab-Tehrany E., Mozafari M., Gaiani C., M L. Liposomes: a review of manufacturing techniques and targeting strategies. Curr. Nanosci. 2011;7:436–452. [Google Scholar]
- 155.Vemuri S., Rhodes C.T. Preparation and characterization of liposomes as therapeutic delivery systems: a review. Pharm. Acta Helv. 1995;70:95–111. doi: 10.1016/0031-6865(95)00010-7. [DOI] [PubMed] [Google Scholar]
- 156.Jiskoot W., Teerlink T., Beuvery E.C., Crommelin D.J. Preparation of liposomes via detergent removal from mixed micelles by dilution. The effect of bilayer composition and process parameters on liposome characteristics. Pharm. Weekbl. - Sci. Ed. 1986;8:259–265. doi: 10.1007/BF01960070. [DOI] [PubMed] [Google Scholar]
- 157.Milsmann M.H.W., Schwendener R.A., Weder H.-G. The preparation of large single bilayer liposomes by a fast and controlled dialysis. Biochim. Biophys. Acta Biomembr. 1978;512:147–155. doi: 10.1016/0005-2736(78)90225-0. [DOI] [PubMed] [Google Scholar]
- 158.Schwendener R.A., Asanger M., Weder H.G. n-alkyl-glucosides as detergents for the preparation of highly homogeneous bilayer liposomes of variable sizes (60–240 nm φ) applying defined rates of detergent removal by dialysis. Biochem. Biophys. Res. Commun. 1981;100:1055–1062. doi: 10.1016/0006-291x(81)91930-6. [DOI] [PubMed] [Google Scholar]
- 159.Enoch H.G., Strittmatter P. Formation and properties of 1000-A-diameter, single-bilayer phospholipid vesicles. Proc. Natl. Acad. Sci. Unit. States Am. 1979;76:145. doi: 10.1073/pnas.76.1.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Ueno M., Tanford C., Reynolds J.A. Phospholipid vesicle formation using nonionic detergents with low monomer solubility. Kinetic factors determine vesicle size and permeability. Biochemistry. 1984;23:3070–3076. doi: 10.1021/bi00308a034. [DOI] [PubMed] [Google Scholar]
- 161.Schurtenberger P., Mazer N., Waldvogel S., Känzig W. Preparation of monodisperse vesicles with variable size by dilution of mixed micellar solutions of bile salt and phosphatidylcholine. Biochim. Biophys. Acta Biomembr. 1984;775:111–114. doi: 10.1016/0005-2736(84)90241-4. [DOI] [PubMed] [Google Scholar]
- 162.Schubert R. Academic Press; 2003. Liposome Preparation by Detergent Removal, Methods Enzymol; pp. 46–70. [DOI] [PubMed] [Google Scholar]
- 163.Shew R.L., Deamer D.W. A novel method for encapsulation of macromolecules in liposomes. Biochim. Biophys. Acta Biomembr. 1985;816:1–8. doi: 10.1016/0005-2736(85)90386-4. [DOI] [PubMed] [Google Scholar]
- 164.Visht S., Awasthi R., Rai R., Srivastav P. Development of dehydration-rehydration liposomal system using film hydration technique followed by sonication. Curr. Drug Deliv. 2014;11:763–770. doi: 10.2174/1567201811666140910122945. [DOI] [PubMed] [Google Scholar]
- 165.Mozafari M.R. Liposomes: an overview of manufacturing techniques. Cell. Mol. Biol. Lett. 2005;10:711–719. [PubMed] [Google Scholar]
- 166.Mozafari M. 2005. A New Technique for the Preparation of Non-toxic Liposomes and Nanoliposomes: the Heating Method; pp. 91–98. [Google Scholar]
- 167.Gutman M. The pH jump: probing of macromolecules and solutions by a laser-induced, ultrashort proton pulse--theory and applications in biochemistry. Methods Biochem. Anal. 1984;30:1–103. doi: 10.1002/9780470110515.ch1. [DOI] [PubMed] [Google Scholar]
- 168.Genç R., Ortiz M., O′Sullivan C.K. Curvature-Tuned preparation of nanoliposomes. Langmuir. 2009;25:12604–12613. doi: 10.1021/la901789h. [DOI] [PubMed] [Google Scholar]
- 169.Hauser H., Gains N. Spontaneous vesiculation of phospholipids: a simple and quick method of forming unilamellar vesicles. Proc. Natl. Acad. Sci. Unit. States Am. 1982;79:1683. doi: 10.1073/pnas.79.6.1683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Yu B., Lee R.J., Lee L.J. Microfluidic methods for production of liposomes. Methods Enzymol. 2009;465:129–141. doi: 10.1016/S0076-6879(09)65007-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Carugo D., Bottaro E., Owen J., Stride E., Nastruzzi C. Liposome production by microfluidics: potential and limiting factors. Sci. Rep. 2016;6:25876. doi: 10.1038/srep25876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Gkionis L., Aojula H., Harris L.K., Tirella A. Microfluidic-assisted fabrication of phosphatidylcholine-based liposomes for controlled drug delivery of chemotherapeutics. Int. J. Pharm. 2021;604:120711. doi: 10.1016/j.ijpharm.2021.120711. [DOI] [PubMed] [Google Scholar]
- 173.William B., Noémie P., Brigitte E., Géraldine P. Supercritical fluid methods: an alternative to conventional methods to prepare liposomes. Chem. Eng. J. 2020;383:123106. [Google Scholar]
- 174.Bigazzi W., Penoy N., Evrard B., Piel G. Supercritical fluid methods: an alternative to conventional methods to prepare liposomes. Chem. Eng. J. 2019;383:123106. [Google Scholar]
- 175.Pick U. Liposomes with a large trapping capacity prepared by freezing and thawing of sonicated phospholipid mixtures. Arch. Biochem. Biophys. 1981;212:186–194. doi: 10.1016/0003-9861(81)90358-1. [DOI] [PubMed] [Google Scholar]
- 176.Liu L., Yonetani T. Preparation and characterization of liposome-encapsulated haemoglobin by a freeze-thaw method. J. Microencapsul. 1994;11:409–421. doi: 10.3109/02652049409034258. [DOI] [PubMed] [Google Scholar]
- 177.El-Nesr O.H., Yahiya S.A., El-Gazayerly O.N. Effect of formulation design and freeze-drying on properties of fluconazole multilamellar liposomes. Saudi Pharmaceut. J. 2010;18:217–224. doi: 10.1016/j.jsps.2010.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Franzé S., Selmin F., Samaritani E., Minghetti P., Cilurzo F. Lyophilization of liposomal formulations: still necessary, still challenging. Pharmaceutics. 2018;10:139. doi: 10.3390/pharmaceutics10030139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Mahmood S., Mandal U.K., Chatterjee B., Taher M. Advanced characterizations of nanoparticles for drug delivery: investigating their properties through the techniques used in their evaluations. Nanotechnol. Rev. 2017;6:355–372. [Google Scholar]
- 180.Mourdikoudis S., Pallares R.M., Thanh N.T.K. Characterization techniques for nanoparticles: comparison and complementarity upon studying nanoparticle properties. Nanoscale. 2018;10:12871–12934. doi: 10.1039/c8nr02278j. [DOI] [PubMed] [Google Scholar]
- 181.Senn A., Pilet P.-E. Electrophoretic mobility, zeta potential and surface charges of maize root protoplasts. Z. Pflanzenphysiol. 1981;102:19–32. [Google Scholar]
- 182.Zhao Z., Katai H., Higashi K., Ueda K., Kawakami K., Moribe K. Cryo-TEM and AFM observation of the time-dependent evolution of amorphous probucol nanoparticles formed by the aqueous dispersion of ternary solid dispersions. Mol. Pharm. 2019;16:2184–2198. doi: 10.1021/acs.molpharmaceut.9b00158. [DOI] [PubMed] [Google Scholar]
- 183.Byrn S., Zografi G., Chen X. 2017. Differential Scanning Calorimetry and Thermogravimetric Analysis; pp. 124–141. [Google Scholar]
- 184.Clout A., Buanz A.B.M., Prior T.J., Reinhard C., Wu Y., O’Hare D., Williams G.R., Gaisford S. Simultaneous differential scanning calorimetry-synchrotron X-ray powder diffraction: a powerful technique for physical form characterization in pharmaceutical materials. Anal. Chem. 2016;88:10111–10117. doi: 10.1021/acs.analchem.6b02549. [DOI] [PubMed] [Google Scholar]
- 185.Panwar P., Pandey B., Lakhera P.C., Singh K.P. Preparation, characterization, and in vitro release study of albendazole-encapsulated nanosize liposomes. Int. J. Nanomed. 2010;5:101–108. doi: 10.2147/ijn.s8030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Wallace S.J., Li J., Nation R.L., Boyd B.J. Drug release from nanomedicines: selection of appropriate encapsulation and release methodology. Drug Deliv. Transl. Res. 2012;2:284–292. doi: 10.1007/s13346-012-0064-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Hwang S.H., Maitani Y., Qi X.R., Takayama K., Nagai T. Remote loading of diclofenac, insulin and fluorescein isothiocyanate labeled insulin into liposomes by pH and acetate gradient methods. Int. J. Pharm. 1999;179:85–95. doi: 10.1016/s0378-5173(98)00392-5. [DOI] [PubMed] [Google Scholar]
- 188.Ascenso A., Cruz M., Euleterio C., Carvalho F.A., Santos N.C., Marques H.C., Simoes S. Novel tretinoin formulations: a drug-in-cyclodextrin-in-liposome approach. J. Liposome Res. 2013;23:211–219. doi: 10.3109/08982104.2013.788026. [DOI] [PubMed] [Google Scholar]
- 189.Deng Y., Ling J., Li M.H. Physical stimuli-responsive liposomes and polymersomes as drug delivery vehicles based on phase transitions in the membrane. Nanoscale. 2018;10:6781–6800. doi: 10.1039/c8nr00923f. [DOI] [PubMed] [Google Scholar]
- 190.Zhang L., Zhang Q., Wang X., Zhang W., Lin C., Chen F., Yang X., Pan W. Drug-in-cyclodextrin-in-liposomes: a novel drug delivery system for flurbiprofen. Int. J. Pharm. 2015;492:40–45. doi: 10.1016/j.ijpharm.2015.07.011. [DOI] [PubMed] [Google Scholar]
- 191.Fenske D.B., Cullis P.R. Liposomal nanomedicines. Expet Opin. Drug Deliv. 2008;5:25–44. doi: 10.1517/17425247.5.1.25. [DOI] [PubMed] [Google Scholar]
- 192.Odeh F., Nsairat H., Alshaer W., Alsotari S., Buqaien R., Ismail S., Awidi A., Al Bawab A. Remote loading of curcumin-in-modified β-cyclodextrins into liposomes using a transmembrane pH gradient. RSC Adv. 2019;9:37148–37161. doi: 10.1039/c9ra07560g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Sur S., Fries A.C., Kinzler K.W., Zhou S., Vogelstein B. Remote loading of preencapsulated drugs into stealth liposomes. Proc. Natl. Acad. Sci. U. S. A. 2014;111:2283–2288. doi: 10.1073/pnas.1324135111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Drummond D.C., Noble C.O., Guo Z., Hong K., Park J.W., Kirpotin D.B. Development of a highly active nanoliposomal irinotecan using a novel intraliposomal stabilization strategy. Cancer Res. 2006;66:3271–3277. doi: 10.1158/0008-5472.CAN-05-4007. [DOI] [PubMed] [Google Scholar]
- 195.Bolotin E.M., Cohen R., Bar L.K., Emanuel N., Ninio S., Barenholz Y., Lasic D.D. Ammonium sulfate gradients for efficient and stable remote loading of amphipathic weak bases into liposomes and ligandoliposomes. J. Liposome Res. 1994;4:455–479. [Google Scholar]
- 196.Madden T.D., Harrigan P.R., Tai L.C., Bally M.B., Mayer L.D., Redelmeier T.E., Loughrey H.C., Tilcock C.P., Reinish L.W., Cullis P.R. The accumulation of drugs within large unilamellar vesicles exhibiting a proton gradient: a survey. Chem. Phys. Lipids. 1990;53:37–46. doi: 10.1016/0009-3084(90)90131-a. [DOI] [PubMed] [Google Scholar]
- 197.V Zhigaltsev I., Winters G., Srinivasulu M., Crawford J., Wong M., Amankwa L., Waterhouse D., Masin D., Webb M., Harasym N., Heller L., Bally M.B., Ciufolini M.A., Cullis P.R., Maurer N. Development of a weak-base docetaxel derivative that can be loaded into lipid nanoparticles. J. Contr. Release : Off. J. Controll. Release Soc. 2010;144:332–340. doi: 10.1016/j.jconrel.2010.02.029. [DOI] [PubMed] [Google Scholar]
- 198.Alshaer W., Zraikat M., Amer A., Nsairat H., Lafi Z., Alqudah D.A., Al Qadi E., Alsheleh T., Odeh F., Alkaraki A., Zihlif M., Bustanji Y., Fattal E., Awidi A. Encapsulation of echinomycin in cyclodextrin inclusion complexes into liposomes: in vitro anti-proliferative and anti-invasive activity in glioblastoma. RSC Adv. 2019;9:30976–30988. doi: 10.1039/c9ra05636j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Olusanya T.O.B., Haj Ahmad R.R., Ibegbu D.M., Smith J.R., Elkordy A.A. Liposomal drug delivery systems and anticancer drugs. Molecules. 2018;23:907. doi: 10.3390/molecules23040907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Giulimondi F., Digiacomo L., Pozzi D., Palchetti S., Vulpis E., Capriotti A.L., Chiozzi R.Z., Laganà A., Amenitsch H., Masuelli L., Peruzzi G., Mahmoudi M., Screpanti I., Zingoni A., Caracciolo G. Interplay of protein corona and immune cells controls blood residency of liposomes. Nat. Commun. 2019;10:3686. doi: 10.1038/s41467-019-11642-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Palchetti S., Caputo D., Digiacomo L., Capriotti A.L., Coppola R., Pozzi D., Caracciolo G. Protein corona fingerprints of liposomes: new opportunities for targeted drug delivery and early detection in pancreatic cancer. Pharmaceutics. 2019;11:31. doi: 10.3390/pharmaceutics11010031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Digiacomo L., Giulimondi F., Pozzi D., Coppola A., La Vaccara V., Caputo D., Caracciolo G. A proteomic study on the personalized protein corona of liposomes. Relevance for early diagnosis of pancreatic DUCTAL adenocarcinoma and biomarker detection. J. Nanotheran. 2021;2:82–93. [Google Scholar]
- 203.Lamichhane N., Udayakumar T.S., D'Souza W.D., Simone C.B., 2nd, Raghavan S.R., Polf J., Mahmood J. Liposomes: clinical applications and potential for image-guided drug delivery. Molecules. 2018;23 doi: 10.3390/molecules23020288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Cabanes A., Even-Chen S., Zimberoff J., Barenholz Y., Kedar E., Gabizon A. Enhancement of antitumor activity of polyethylene glycol-coated liposomal doxorubicin with soluble and liposomal interleukin 2, Clinical cancer research. Off. J. Am. Assoc. Canc. Res. 1999;5:687–693. [PubMed] [Google Scholar]
- 205.Fukuda A., Tahara K., Hane Y., Matsui T., Sasaoka S., Hatahira H., Motooka Y., Hasegawa S., Naganuma M., Abe J., Nakao S., Takeuchi H., Nakamura M. Comparison of the adverse event profiles of conventional and liposomal formulations of doxorubicin using the FDA adverse event reporting system. PLoS One. 2017;12 doi: 10.1371/journal.pone.0185654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Angst M.S., Drover D.R. Pharmacology of drugs formulated with DepoFoam: a sustained release drug delivery system for parenteral administration using multivesicular liposome technology. Clin. Pharmacokinet. 2006;45:1153–1176. doi: 10.2165/00003088-200645120-00002. [DOI] [PubMed] [Google Scholar]
- 207.Chou H., Lin H., Liu J.M. A tale of the two PEGylated liposomal doxorubicins. OncoTargets Ther. 2015;8:1719–1720. doi: 10.2147/OTT.S79089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Fassas A., Anagnostopoulos A. The use of liposomal daunorubicin (DaunoXome) in acute myeloid leukemia. Leuk. Lymphoma. 2005;46:795–802. doi: 10.1080/10428190500052438. [DOI] [PubMed] [Google Scholar]
- 209.Batist G., Barton J., Chaikin P., Swenson C., Welles L. Myocet (liposome-encapsulated doxorubicin citrate): a new approach in breast cancer therapy. Expet Opin. Pharmacother. 2002;3:1739–1751. doi: 10.1517/14656566.3.12.1739. [DOI] [PubMed] [Google Scholar]
- 210.Murry D.J., Blaney S.M. Clinical pharmacology of encapsulated sustained-release cytarabine. Ann. Pharmacother. 2000;34:1173–1178. doi: 10.1345/aph.19347. [DOI] [PubMed] [Google Scholar]
- 211.Meyers P.A., Chou A.J. Muramyl tripeptide-phosphatidyl ethanolamine encapsulated in liposomes (L-MTP-PE) in the treatment of osteosarcoma. Adv. Exp. Med. Biol. 2014;804:307–321. doi: 10.1007/978-3-319-04843-7_17. [DOI] [PubMed] [Google Scholar]
- 212.Silverman J.A., Deitcher S.R. Marqibo® (vincristine sulfate liposome injection) improves the pharmacokinetics and pharmacodynamics of vincristine. Cancer Chemother. Pharmacol. 2013;71:555–564. doi: 10.1007/s00280-012-2042-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Passero F.C., Jr., Grapsa D., Syrigos K.N., Saif M.W. The safety and efficacy of Onivyde (irinotecan liposome injection) for the treatment of metastatic pancreatic cancer following gemcitabine-based therapy. Expet Rev. Anticancer Ther. 2016;16:697–703. doi: 10.1080/14737140.2016.1192471. [DOI] [PubMed] [Google Scholar]
- 214.Krauss A.C., Gao X., Li L., Manning M.L., Patel P., Fu W., Janoria K.G., Gieser G., Bateman D.A., Przepiorka D., Shen Y.L., Shord S.S., Sheth C.M., Banerjee A., Liu J., Goldberg K.B., Farrell A.T., Blumenthal G.M., Pazdur R. FDA approval summary: (daunorubicin and cytarabine) liposome for injection for the treatment of adults with high-risk acute myeloid leukemia. Clin. Cancer Res. : Off. J. Am. Assoc. Canc. Res. 2019;25:2685–2690. doi: 10.1158/1078-0432.CCR-18-2990. [DOI] [PubMed] [Google Scholar]
- 215.Koudelka S., Turánek J. Liposomal paclitaxel formulations. J. Contr. Release : Off. J. Controll. Release Soc. 2012;163:322–334. doi: 10.1016/j.jconrel.2012.09.006. [DOI] [PubMed] [Google Scholar]
- 216.Azanza J.R., Sádada B., Reis J. Liposomal formulations of amphotericin B: differences according to the scientific evidence. Rev. Española Quimioter. : Pub. Ofic. Soc. Espan. Quimioter. 2015;28:275–281. [PubMed] [Google Scholar]
- 217.Ostrosky-Zeichner L., Marr K.A., Rex J.H., Cohen S.H. Clinical Infectious Diseases : an Official Publication of the Infectious Diseases Society of America. Vol. 37. 2003. Amphotericin B: time for a new "gold standard"; pp. 415–425. [DOI] [PubMed] [Google Scholar]
- 218.Romero E.L., Morilla M.J. Drug delivery systems against leishmaniasis? Still an open question. Expet Opin. Drug Deliv. 2008;5:805–823. doi: 10.1517/17425247.5.7.805. [DOI] [PubMed] [Google Scholar]
- 219.Rudramurthy S.M., Jatana M., Singh R., Chakrabarti A. In vitro antifungal activity of Indian liposomal amphotericin B against clinical isolates of emerging species of yeast and moulds, and its comparison with amphotericin B deoxycholate, voriconazole, itraconazole and fluconazole. Mycoses. 2013;56:39–46. doi: 10.1111/j.1439-0507.2012.02197.x. [DOI] [PubMed] [Google Scholar]
- 220.Chang H.I., Yeh M.K. Clinical development of liposome-based drugs: formulation, characterization, and therapeutic efficacy. Int. J. Nanomed. 2012;7:49–60. doi: 10.2147/IJN.S26766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Alfayez M., Kantarjian H., Kadia T., Ravandi-Kashani F., Daver N. CPX-351 (vyxeos) in AML. Leuk. Lymphoma. 2020;61:288–297. doi: 10.1080/10428194.2019.1660970. [DOI] [PubMed] [Google Scholar]
- 222.McAlvin J.B., Padera R.F., Shankarappa S.A., Reznor G., Kwon A.H., Chiang H.H., Yang J., Kohane D.S. Multivesicular liposomal bupivacaine at the sciatic nerve. Biomaterials. 2014;35:4557–4564. doi: 10.1016/j.biomaterials.2014.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Immordino M.L., Dosio F., Cattel L. Stealth liposomes: review of the basic science, rationale, and clinical applications, existing and potential. Int. J. Nanomed. 2006;1:297–315. [PMC free article] [PubMed] [Google Scholar]
- 224.Gregoriadis G. Liposomes and mRNA: two technologies together create a COVID-19 vaccine. Med. Drug Discov. 2021;12:100104. [Google Scholar]
- 225.Tai T.T., Wu T.J., Wu H.D., Tsai Y.C., Wang H.T., Wang A.M., Shih S.F., Chen Y.C. A strategy to treat COVID-19 disease with targeted delivery of inhalable liposomal hydroxychloroquine: a preclinical pharmacokinetic study. Clin. Transl. Sci. 2021;14:132–136. doi: 10.1111/cts.12923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Constantin C., Pisani A., Bardi G., Neagu M. Nano-carriers of COVID-19 vaccines: the main pillars of efficacy. Nanomedicine. 2021;16:2377–2387. doi: 10.2217/nnm-2021-0250. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data was used for the research described in the article.