Abstract
The fluvial deposition of mine tailings generated from historic mining operations near Butte, Montana, has resulted in substantial surface and shallow groundwater contamination along Silver Bow Creek. Biogeochemical processes in the sediment and underlying hyporheic zone were studied in an attempt to characterize interactions consequential to heavy-metal contamination of shallow groundwater. Sediment cores were extracted and fractionated based on sediment stratification. Subsamples of each fraction were assayed for culturable heterotrophic microbiota, specific microbial guilds involved in metal redox transformations, and both aqueous- and solid-phase geochemistry. Populations of cultivable Fe(III)-reducing bacteria were most prominent in the anoxic, circumneutral pH regions associated with a ferricrete layer or in an oxic zone high in organic carbon and soluble iron. Sulfur- and iron-oxidizing bacteria were distributed in discrete zones throughout the tailings and were often recovered from sections at and below the anoxic groundwater interface. Sulfate-reducing bacteria were also widely distributed in the cores and often occurred in zones overlapping iron and sulfur oxidizers. Sulfate-reducing bacteria were consistently recovered from oxic zones that contained high concentrations of metals in the oxidizable fraction. Altogether, these results suggest a highly varied and complex microbial ecology within a very heterogeneous geochemical environment. Such physical and biological heterogeneity has often been overlooked when remediation strategies for metal contaminated environments are formulated.
Metal sulfide mine wastes (tailings), generated during 125 years of mining and smelting activities, contaminate 200 km of riverine habitat in the Upper Clark Fork Valley of Western Montana (8, 26, 32, 36). Fluvial redeposition of these wastes along the floodplain has created vast wastelands, called slickens, void of vegetative cover. These tailings deposits are part of the largest U.S. Environmental Protection Agency Superfund site in the United States (32). The biogeochemical processes responsible for metal sulfide oxidation have contributed to extensive surface and groundwater contamination (5, 25, 28, 32). Consequently, several exploratory reclamation projects have been undertaken along the Clark Fork River, as well as along Silver Bow Creek, a heavily impacted tributary of the Clark Fork River. A fundamental problem in assessing the long-term effectiveness of various remediation options, particularly that of acid neutralization (liming) (10, 37), is the lack of information on the microbial ecology of this ecosystem.
Though few in number, previous investigations exploring the microbial ecology of mine waste-contaminated environments have demonstrated considerable metabolic diversity in bacterial populations (12, 35, 39, 40, 41). Many of these bacteria catalyze specific oxidation-reduction reactions that alter the mobility of heavy-metal contaminants. Previous studies have focused on bacterial distribution in heap leach piles or waste impoundments rather than on riverine environments. Tailings in the Upper Clark Fork Valley and in other historic mining areas were fluvially deposited over many decades (8, 32). Consequently, Silver Bow Creek floodplain sediments are a heterogeneous mixture of tailings, silts, clays, fine- to coarse-grained sands, and gravel mixed with layers high in organic content (23). The microbial ecology of this type of environment has yet to be explored.
The complex geochemical nature of this environment suggests that it might support an equally complex microbial community. In addition to sulfidic wastes that could provide substrates for autotrophic bacterial growth, weathered tailings contain large quantities of iron and manganese (hydro)oxides. In recent years it has become evident that some organisms utilize these compounds as terminal electron acceptors in their respiratory processes. These organisms are widespread in nature and play a major role in mineral cycling in both aquatic and sedimentary environments (20, 25, 30). With few exceptions (35, 39), little attention has been paid to these physiological groups in tailings environments, where their impact on surface and groundwater contamination may be significant. In anoxic zones, these reductive processes can release Fe(II), Mn(II), and other associated heavy metals to the local groundwater (9, 20, 21, 29). The reduction of sulfate to sulfide mediated by dissimilatory sulfate-reducing bacteria (SRB) is yet another anaerobic process that can affect metal transport. In contrast to the reactions described above that serve to mobilize heavy metals, the sulfide produced during sulfate reduction can rapidly react with soluble metals to immobilize them as insoluble metal sulfides.
In previous studies we have characterized the geochemistry (2) and microbiology (45) of stream bed and hyporheic zones along Silver Bow Creek. The present studies were initiated to explore simultaneously the geochemistry and microbiology in this fluvially deposited tailings ecosystem.
MATERIALS AND METHODS
General site description.
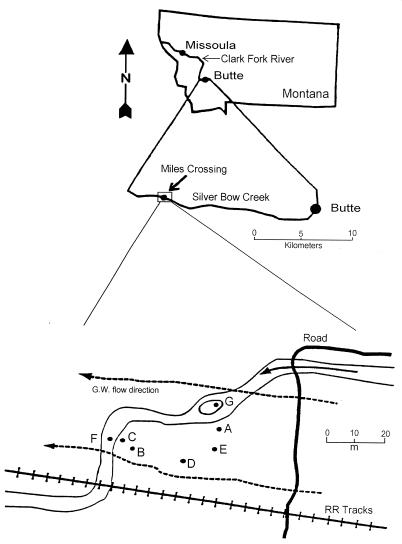
The research site is located at Miles Crossing, approximately 15 km west of Butte, Montana, in the Silver Bow Creek floodplain (Fig. 1). The pure tailings at this site are up to 1.75 m thick and are enriched in As, Cd, Cu, Fe, Mn, Pb, and Zn. The shallow groundwater (on average 1 to 2 m below the surface) flows to the west down the alluvial valley and is well connected to the surface water (2, 23).
FIG. 1.
Miles Crossing research site map showing the location of cores A through F.
Aqueous-phase sampling.
Pore water samples were collected approximately every 2 weeks by using the suction lysimeters described by Fetter (11). Temperature, pH, and dissolved oxygen (DO) were measured in the field with Orion electrodes (Orion Research, Inc., Beverly, Mass.).
Groundwater was sampled via piezometers constructed of 3.2-cm polyvinyl chloride pipe with the bottom 6 in. slotted. To remove stagnant water in the piezometer, one and one-half well volumes were pumped and discarded before sampling. Temperature, pH, and DO were measured in a flowthrough sampling apparatus to minimize contact with the atmosphere. The groundwater was pumped into a 200-ml plastic reservoir through an inflow tube attached to the peristaltic pump. The pH-temperature and dissolved oxygen electrodes were inserted through a rubber stopper into the enclosed reservoir. A constant flow of water through the sampling apparatus was maintained while field parameters were measured. Cation and anion samples were pumped through an in-line 0.45-μm (pore size) filter (Gelman GN-6) into 500-ml plastic sample bottles. Cation samples (pore water and groundwater) were acidified with concentrated trace-metal grade nitric acid to approximately pH 2. All water samples were transported to the laboratory on ice and refrigerated at 4°C until analysis. Water samples were analyzed within 2 weeks for Al, As, Ca, Cd, Co, Cu, Fe, Mg, Mn, Mo, Na, Ni, Pb, Si, Sr, Ti, and Zn by using a Jarrell Ash inductively coupled plasma emission spectrophotometer (ICP). Sulfate concentrations were determined within 1 week of collection by using a Dionex 2000 ion chromatograph.
Solid-phase sampling.
Seven sediment cores were collected between 1 July 1994 and 17 August 1994 by mechanically driving sterile polycarbonate tubes (4.5 cm [internal diameter]) through the tailings. Coring location was based, in part, on the presence of previously installed groundwater monitoring wells. Cores were fractionated in the field based on visually determined sediment stratigraphy. The outer 0.5 cm of sediment was pared from the core to minimize vertical contamination. Fractions were placed in Whirl-pak bags and transported back to the lab, on ice, for processing. The anoxic, bottom sections of the cores were transferred to bags under a stream of nitrogen gas and transported in Gas-Pak anaerobic jars. Upon return to the lab, anoxic sections were immediately placed in an anaerobic glove box (Coy Laboratories, Grass Lake, Mich.) and processed in an atmosphere consisting of N2, CO2, and H2 in a ratio of 85:8:7.
Geochemical analysis.
Sediment was immediately transferred to acid-washed petri dishes and frozen. Subsequently, they were freeze-dried and ground to approximately clay size (∼2.0 μm) by using a Shatter-Box grinder. Samples were sequentially extracted to remove the water-soluble (sulfate salts), reducible (oxide coatings), oxidizable (primarily sulfides and a portion of the organic fraction), and residual (highly crystalline material) fractions of the sediment (3, 23). Each of these fractions was analyzed for Al, As, Ca, Cd, Co, Cu, Fe, Mg, Mn, Mo, Na, Ni, Pb, Si, Sr, Ti, and Zn by using an ICP. Total carbon was determined by using a Coulometrics CO2 coulometer system.
Microbial sampling and culturing.
Fractions were homogenized by stomaching, and approximately 2 cm3 of sediment was added to 10 ml of 0.1% sodium pyrophosphate (pH 6.8) to a final volume of 12 ml. Bacteria were dislodged from the sediment by vortexing (high speed, 15 s) followed by low-level sonication (10 min), a method modified from Schallenberg et al. (38).
Bacterial enumeration.
Aerobic chemoorganotrophic (COT) bacteria were enumerated by plating 10-fold serial dilutions (0.1% peptone) on R2A agar (Difco Laboratories, Detroit, Mich.). Anaerobic COT bacteria were counted in the same manner by using prereduced dilution blanks and anaerobically equilibrated R2A plates (stored in the glove box for 1 week prior to use). Aerobic and anaerobic plates were incubated at 20 and 25°C (±2.0), respectively. Both groups were counted after 7 days and again after 14 days.
A five-tube most-probable-number (MPN) method was used to enumerate cultivable iron- and sulfur-oxidizing bacteria, as well as sulfate-, iron-, and manganese-reducing bacteria. The computer program described by Klee (18) was used in MPN calculations. All MPN enrichments were performed in Bio-Blocks (Rainin, Inc., Emeryville, Calif.) except for those for the manganese-reducing bacteria described below.
(i) Iron- and sulfur-oxidizing bacteria.
Iron- and sulfur-oxidizing bacteria were enumerated in the basal mineral salts solution (MS) described by Harrison (15). MS supplemented with 4% (wt/vol) FeSO4 · 7H2O (pH 3.5, adjusted with 6 N H2SO4) was used for cultivation of acidophilic iron-oxidizing populations. Wells positive for Fe2+ oxidation formed an orange ferric iron precipitate. Neutrophilic sulfur-oxidizing species were cultured in MS containing 1% (wt/vol) powdered sulfur (pH 6.5, adjusted with 1 N NaOH). The sulfur was sterilized separately by heating to 90°C for 0.5 h on 3 successive days. Wells positive for sulfur oxidation became acidic and turned bright red on the addition of 75 μl of methyl orange (0.01% in distilled H2O). Enrichments were incubated at 20°C in the dark and counted after 30 and 45 days, respectively.
(ii) SRB.
SRB were cultured in Sulfate API Broth (Difco) amended with 0.075% agar. The medium was reduced by boiling under a stream of N2 gas, dispensed to Bio-Blocks, and then allowed to equilibrate in an anaerobic glove box prior to inoculation. The blackening of the medium as a result of ferrous sulfide production indicated sulfate reduction. Enrichments were incubated at room temperature and counted after 30 days.
(iii) Iron-reducing bacteria.
Iron-reducing bacteria were cultivated in FWA medium as described by Lovley and Phillips (22). Amorphous ferric oxyhydroxide (22) was added to provide a final concentration of ca. 200 mM Fe(III). Production of Fe2+ was determined by combining 50 μl of culture with 50 μl of 1,10-phenanthroline (0.1% in distilled H2O), an orange color indicating the presence of Fe(II) (1).
(iv) Enrichments for manganese-reducing bacteria.
Enrichments for manganese-reducing bacteria were carried out in 1.5-ml microcentrifuge tubes following a protocol similar to that described by Myers and Nealson (29). A 200-μl aliquot from the serial dilutions was mixed with 200 μl of a 0.5% soft agar. The soft agar was allowed to gel, and 1 ml of FWA medium, supplemented with 0.75% agar and δMnO2 (100 mM) (4), was added. A clearing of the top agar indicated manganese reduction. Enrichments were incubated at room temperature and scored after 30 days.
Media for the enrichment of both the iron- and the manganese-reducing bacteria were also supplemented with a mixture of acetate and succinate (7.5 mM final concentration of each) and sodium molybdate (20 mM final concentration). Acetate and succinate were added to provide a nonfermentable carbon source, and molybdate was used to inhibit sulfate reduction (31).
Statistical analysis.
A Spearman rank order correlation analysis was performed by using Jandel SigmaStat software (Jandel Corp., San Rafael, Calif.) in order to determine whether an association existed between the distribution of specific microbial populations and the geochemical parameters measured.
RESULTS
Water chemistry.
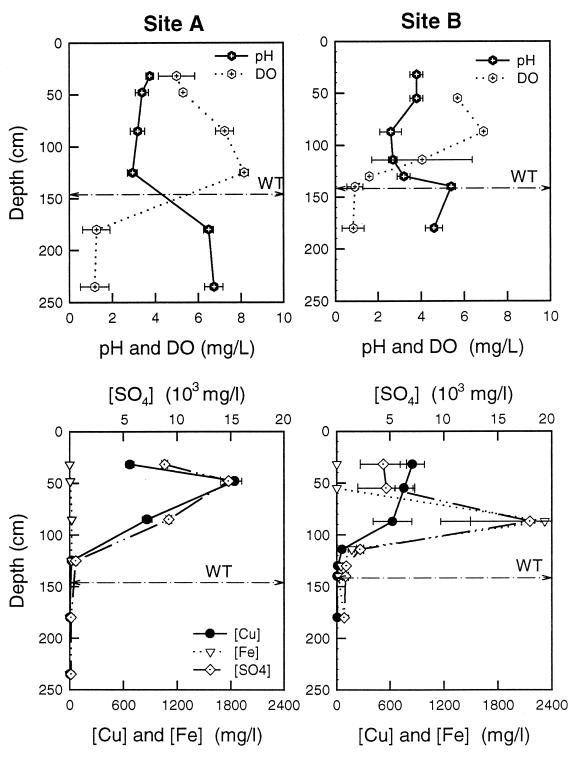
Sites A and B were fully instrumented for the study of pore water and groundwater chemistry (Fig. 1). Groundwater in the floodplain was characterized by zones with various levels of contamination (Fig. 2, sites A and B). Concentrations of the metals Cu, Fe, Mn, and Zn in the groundwater at site B were 9, 100, 8, and 7 times greater, respectively, than those measured at site A. Porewater pH values at site B were 1 to 2 log units lower, and SO42− concentrations were an order of magnitude higher than those found at site A. At both sites, there was a steep concentration gradient for DO near the groundwater interface, with the groundwater becoming anoxic.
FIG. 2.
Pore water chemistry at sites A (left) and B (right) as a function of depth. The upper panel shows field-measured parameters, pH, and DO content. Concentrations of sulfate, copper, and iron are presented in the lower panel. The depth of the water table (WT) is denoted by the dashed line.
Sediment geochemistry.
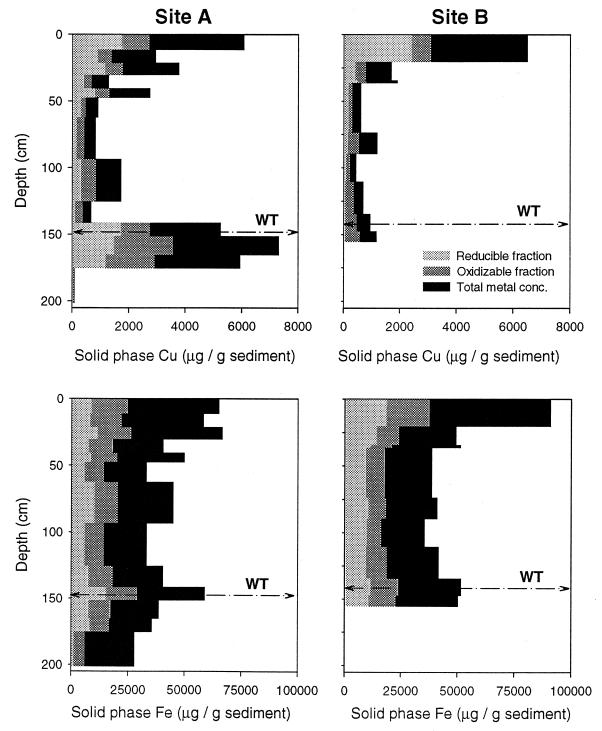
All tailings in the unsaturated zone above the water table (vadose) were coated with orange-red to orange-brown amorphous iron oxyhydroxides except in some small sand-silt layers or on the interior of clay chunks or layers. Metal content decreased with depth to the water table in cores located on the highest level portion of the floodplain (23). Total Cu and Fe abundance as well as Cu and Fe concentrations in the oxidizable and reducible fractions from sites A and B are shown in Fig. 3. Sediments at the water table in cores A (Fig. 3) and E (not shown) were encrusted in a ferricrete layer [soil cemented in a thick coating of amorphous metal (hydro)oxides]. There was a marked increase in reducible-phase zinc and manganese associated with the ferricrete layer, and iron was 30 to 50% higher in samples at the water table than in samples above or below the water table. Total carbon was generally highest in the top 50 cm of tailings (see Fig. 6) and was associated with the zones of fine grain silts and clays. Concentrations of most metals in the oxidizable fraction (sulfides) also reached their maxima in the top 40 cm. A more complete geochemical profile for all cores from this location is detailed by Lucy (23).
FIG. 3.
Solid-phase Cu (top) and Fe (bottom) concentrations at sites A (left) and B (right) as a function of depth. The various fractions are operationally defined based on selective extractions as reducible (amorphous and crystalline oxides), oxidizable (sulfides and organic), and total metals in all fractions. The depth of the water table (WT) is denoted by the dashed line.
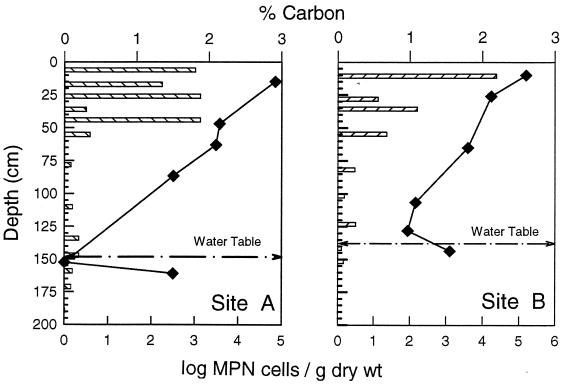
FIG. 6.
Vertical concentration profile showing the percent total carbon (bars) and MPN estimates for sulfate-reducing bacteria (⧫) observed at sites A and B.
COT bacteria.
Numbers of culturable aerobic and anaerobic COT bacteria ranged from 7.5 × 105 and 9.7 × 105 CFU/g (dry weight) of sediment, respectively, to <100 (46). Both groups were patchily distributed in the tailings. Surprisingly, concentrations of anaerobic COT bacteria recovered from oxic regions (8.2 × 104 ± 4.2 × 104, mean ± standard error) were often greater than those recovered from the anoxic zones (2.0 × 104 ± 6.1 × 103). In contrast, greater numbers of aerobic COT bacteria were recovered from anoxic zones than were observed in oxic regions (8.0 × 104 ± 6.2 × 104 and 4.9 × 104 ± 1.9 × 104, respectively).
Iron- and sulfur-oxidizing bacteria.
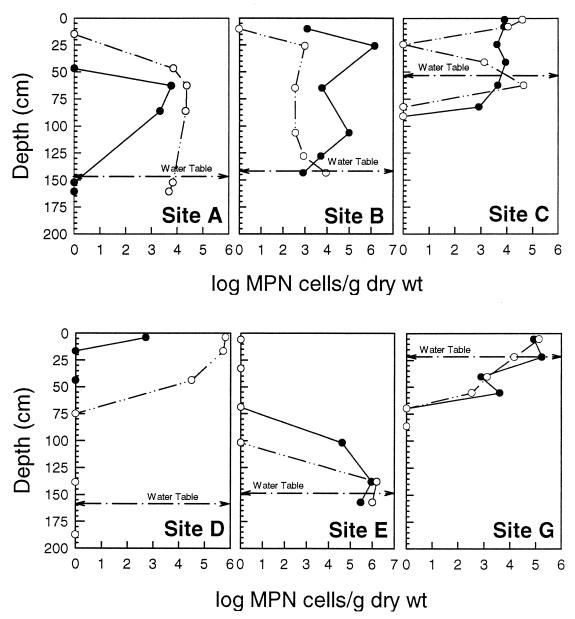
Iron-oxidizing and sulfur-oxidizing bacteria were distributed throughout the tailings to depths of 175 cm (Fig. 4). The abundance of iron-oxidizing bacteria at site B was generally 1 to 2 orders of magnitude greater than at site A (Fig. 4, sites A and B), and they were much more widely distributed at site B. In contrast, at site A, sulfur-oxidizing bacteria predominated over iron-oxidizing bacteria. Interestingly, large populations of iron-oxidizing bacteria (104 to 106) were present in anoxic zones at and below the groundwater table in four of the cores (Fig. 4, sites B, C, E, and G).
FIG. 4.
MPN-determined cell densities of iron- and sulfur-oxidizing bacteria as a function of depth. Symbols: ●, acidophilic iron-oxidizing bacteria; ○, neutrophilic sulfur-oxidizing bacteria. Capital letters denote the core site location.
Iron- and manganese-reducing bacteria.
The distribution of cultivable Fe(III)-reducing bacteria at site A differs markedly from that observed at site B (Fig. 5, closed circles). At site A, all of the organisms from these groups were recovered from the chemocline or below, in the anoxic groundwater. The greatest concentrations of iron-reducing bacteria from site B were located at 25 cm, the same depth at which SRB and iron-oxidizing bacteria showed concentration peaks. Mn(IV)-reducing bacteria were more consistently distributed and generally less abundant (Fig. 5, open circles).
FIG. 5.
Abundance of iron-reducing bacteria (●) and manganese-reducing bacteria (○) in Silver Bow Creek tailings as a function of depth at coring locations A and B.
SRB.
SRB were recovered from all core locations and were not limited to the anoxic sub-groundwater zones (Fig. 6). SRB density tended to be highest in the uppermost 40 cm and was associated with a zone of fine grain silt and/or clay high in organic carbon. Their numbers declined with depth through the vadose and increased near the groundwater interface. With the exception of core C, which had very low total carbon throughout the core, this distribution pattern was observed in all cores (46).
Correlation analyses.
There was a moderately strong positive correlation between local porewater concentrations of iron and lead and the distribution of iron-oxidizing bacteria (0.6, P = 0.03). As expected, a negative correlation was observed between porewater pH and these acidophiles (−0.62, P = 0.03). We also observed a negative association between water-extractable molybdenum and SRB abundance (−0.50, P = 0.002). The distribution of Mn(IV)-reducing bacteria correlated negatively with As, Cd, Cu, Mn, and Pb in the reducible fraction, with Mn in the oxidizable fraction, and with solution phase Cu (C.C. ≤ −0.75, P ≤ 0.01).
DISCUSSION
Fluvial deposition of mine wastes along Silver Bow Creek, Montana, over a period of several decades has resulted in a highly complex biogeochemical environment. The vadose sediment is characterized by fine-grained, metal-sulfide-rich, organic-material-rich layers randomly interstratified with coarser-grained sediment. The tailing thickness and vertical distribution of metals is variable across the site and the reworking of floodplain sediment has dramatically altered metal concentrations in some areas (23). This complex geochemical mosaic is mirrored by equally complex and variable patterns of microbial abundance.
Historical oxidation of metal sulfides has been the principal source of surface and groundwater contamination in the Upper Clark Fork River Valley (5, 25, 32). Data presented here indicate that iron-oxidizing bacteria are abundant and widely distributed in these highly weathered tailing deposits. The concentration of iron-oxidizing bacteria observed in Silver Bow Creek tailings deposits is equal to or greater than that reported in other mine waste deposits (39, 40), where their presence was also correlated to zones of active sulfide leaching.
The activity of dissimilatory metal-reducing bacteria (DMRB) may provide a secondary mechanism for trace metal mobilization in these tailings. It is well documented that the microbial reduction of Fe(III) and Mn(IV) oxides can result in the release of a wide variety of toxic trace metals that have been sorbed to or coprecipitated with the oxides (9, 20, 30). Ribet et al. (34) have suggested that, given the large concentrations of ferric-bearing secondary minerals common to tailing deposits, the microbial reduction of Fe(III) will be thermodynamically favored even at very low concentrations of dissolved organic carbon. In mineralogical studies of the Nickel Rim tailings (Ontario) Ribet et al. (34) found that >80% of the Fe, Ni, Cu, Cr, and Co in the oxidized zone were potentially available for release by reductive-dissolution reactions.
The geochemical heterogeneity in the Silver Bow Creek tailings appears to influence the distribution of iron-reducing bacteria. At site A large numbers of Fe(III)-reducing bacteria are found only at the anoxic groundwater interface associated with a ferricrete layer (compare Fig. 3 and 5). This layer is formed when acidic, metal-rich pore water contacts the circumneutral pH groundwater, and the metals precipitate as amorphous (hydro)oxides. These poorly crystalline iron and manganese oxides are considerably more amenable to microbial reduction (20). In contrast, at site B iron-reducing bacterial numbers are highest in the top 75 cm, where concentrations of reducible, solid-phase iron, as well as solution-phase iron, are elevated. Given the low pH (2.7 to 3.9) and high dissolved oxygen content (6 to 8 mg/liter) in this region, the iron species most likely present was Fe3+. This Fe3+ would therefore be available as a terminal electron acceptor. The abundance of iron-reducing bacteria (∼106 MPN/g [dry weight]) at site A in association with high concentrations of Fe, Cu, Mn, and Pb (7,800, 1,140, 660, and 530 μg/g of sediment, respectively) in the reducible fraction (23) suggests a possible role for microbial iron reduction in groundwater contamination.
Bacteria that couple the oxidation of organic substrates to the dissimilatory reduction of sulfate are likely to play an important role in the biogeochemical cycling of iron and trace elements in these historic mine tailings. SRB were recovered from 23 of the 38 samples. In general, SRB were most abundant in the top 30 to 40 cm of the tailings. We also observed that carbon and metal sulfide concentrations were generally highest in the top 50 cm of the tailings. At sites A and B a slight increase in pH and a decrease in the SO42− concentration were also observed above 50 cm, where SRB abundance peaked. The presence of SRB in tailing impoundments or associated with waters affected by acid mine drainage has been observed by several investigators (12, 24, 39, 40, 42). Contaminant immobilization by SRB activity has likewise been demonstrated (7, 14, 19, 33, 44). The presence of SRB in the surface layers of these tailings in conjunction with our geochemical data suggests that sulfate reduction, when active, may decrease the concentration of metals that might otherwise reach the tailing surface by capillary wicking. The potential for utilizing autochthonous SRB in this system for metal stabilization warrants further investigation.
The culture-based enumeration methods employed in these studies are likely to underestimate the abundance of both SRB and iron-reducing bacteria and, thus, the impact these organisms have on their environment. Since core sections above the groundwater table were determined to be oxic based on the red-orange oxide coating of sediment particles and the high DO content, no attempt was made to prevent sample exposure to oxygen. It is therefore possible that strictly anaerobic members of these guilds were lost. In addition, a limited set of substrates was supplied as electron donors. Teske et al. (43) have demonstrated the importance of using multiple carbon sources when determining SRB diversity and abundance. These investigators also found that SRB numbers obtained by MPN counts were generally 3 orders of magnitude lower than those obtained by using molecular techniques (e.g., fluorescent in situ hybridization). Despite the potential limitations of the culture-based techniques employed here, they clearly demonstrate the presence of these important guilds in the Silver Bow Creek tailings. These estimates of microbial abundance also compare favorably to those reported by other investigators (24, 39, 40, 43).
The Silver Bow Creek environment is geochemically heterogeneous. As a result, distribution of major bacterial guilds with respect to oxic-anoxic boundaries and the relative abundance of thermodynamically favored electron acceptors does not conform readily to current models (12, 21, 30). In particular, we observed considerable overlap of aerobic sulfur- and iron-oxidizing bacteria with anaerobic sulfate-, iron-, and manganese-reducing bacteria. Similar overlap has also been demonstrated in other tailing deposits (24, 39). Traditionally, it has been thought that processes such as Fe3+ and SO42− reduction become important only after other, more energetically favorable electron acceptors such as O2, NO3−, and Mn(IV) are depleted. However, it has been shown recently that sulfate reduction can occur in oxic environments (6, 13, 16, 27, 43). In addition, Miller et al. (24) and Jorgensen (17) have observed sulfur-oxidizing bacteria and SRB in the same zones and have suggested the existence of microniches to explain this phenomenon.
Within any one zone in Silver Bow Creek tailings, high concentrations of several potential electron donors and electron acceptors can be found. Statistical analysis indicated a negative correlation between molybdenum in the water-extractable fraction and the presence of SRB, as well as a negative relation between the concentration of various metals and the distribution of Mn(IV)-reducing bacteria. Such confounding factors may play important roles in determining the order of terminal-electron-accepting processes in this unique environment. Further investigation is needed to determine the relative activity and importance of each of these groups in zones where different processes are likely to overlap.
The historic mine tailings along Silver Bow Creek, Montana, support abundant and diverse microbial populations. Bacterial groups whose activities can affect the mobility of heavy metals, either directly or indirectly, are widely but patchily distributed in this environment. Typical remedial technologies that work for small areas or low volumes are not considered feasible for the Silver Bow Creek site. Due in part to cost considerations, an in situ approach has been favored that calls for the incorporation of lime [CaCo3 and Ca(OH)2] into the top 30 to 40 cm, followed by fertilization and revegetation (36). These observations suggest that liming may not effectively control the leaching of metals into groundwater. At six of the seven sites evaluated here, we observed that iron-oxidizing bacteria were most numerous below 50 cm. In addition, environmental perturbations that tend to enhance the reductive dissolution of metal (hydro)oxides would promote metal destabilization. Tailing neutralization along with the addition of macronutrients and organic material could stimulate microbial iron reduction. While the density of COT bacteria observed was considerably lower than those typically found in soils, the amendments proposed would likely stimulate this important group. An increase in heterotrophic activity would tend to decrease DO levels and provide the small organic acids and alcohols typically required by DMRB (20, 30). Preliminary experiments in our lab indicate that such amendments can increase the rate of iron reduction by more than sixfold (46). On the other hand, these same perturbations might also enhance sulfate reduction, an environmentally beneficial process. The effect of reclamation measures on each of these microbial processes must be evaluated in order to make reasonable predictions about their chances for long-term success.
ACKNOWLEDGMENTS
This work was supported by grants from The Montana Water Center, The Mined Lands Reclamation Unit, and The Murdock Charitable Trust.
We thank William Holben and R. Frank Rosenzweig for their review of and suggestions for improving the manuscript. We also thank Shawn Rowland and Patrick Ball for their help with sample collection and processing and Lynn Biegelsen for her help with geochemical analysis.
REFERENCES
- 1.American Public Health Association. Phenanthroline method. In: Greenberg A E, Clesceri L S, Eaton A D, editors. Standard methods for the examination of water and wastewater. Washington, D.C: American Public Health Association; 1994. pp. 3-66–3-68. [Google Scholar]
- 2.Benner S G, Smart E W, Moore J N. Metal behavior during surface-groundwater interaction, Silver Bow Creek, Montana. Environ Sci Technol. 1995;29:1789–1795. doi: 10.1021/es00007a015. [DOI] [PubMed] [Google Scholar]
- 3.Brook E J. Particle-size and chemical control of metals in Clark Fork River sediment. Masters thesis. Missoula, Mont: University of Montana; 1988. [Google Scholar]
- 4.Burdige D J, Nealson K H. Microbial manganese reduction by enrichment cultures from coastal marine sediments. Appl Environ Microbiol. 1985;50:491–497. doi: 10.1128/aem.50.2.491-497.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Camp, Dresser, and Mckee, Inc. Milltown Reservoir Sediments, Work Plan Scoping Document. Doc. No. 8469-WP-F-RO-010989. Helena, Mont: Montana Department of Health and Environmental Sciences; 1989. [Google Scholar]
- 6.Canfield D E, Des Marais D J. Aerobic sulfate reduction in microbial mats. Science. 1991;251:1471–1473. doi: 10.1126/science.11538266. [DOI] [PubMed] [Google Scholar]
- 7.Castro, J. M., B. W. Wielinga, J. E. Gannon, and J. N. Moore. Stimulation of sulfate-reducing bacteria in pit lake water through addition of organic wastes, Water Environ. Res., in press.
- 8.CH2M Hill, Inc. 1989. Field operations plan for Silver Bow Creek flood modeling study: Silver Bow Creek RI/FS, EPA region VIII. CH2M Hill, Helena, Mont.
- 9.Cummings, D. E., F. Caccavo, Jr., S. Fendorf, and R. F. Rozenzweig. Arsenic mobilization by the dissimilatory Fe(III)-reducing bacterium Shewanella alga BrY. Environ. Sci. Technol., in press.
- 10.Dollhopf D J. Montana State University, Reclamation Research Unit Report No. RRU 9201. Bozeman, Mont: Montana State University; 1992. Deep lime incorporation methods for neutralization of acidic minesoils; pp. 1–88. [Google Scholar]
- 11.Fetter C W. Groundwater and soil monitoring. In: McConnin R A, editor. Contaminant hydrogeology. New York, N.Y: MacMillan Publishing Co.; 1993. pp. 338–391. [Google Scholar]
- 12.Fortin D, Davis B, Southham G, Beveridge T J. Biogeochemical phenomena induced by bacteria within sulfidic mine tailings. J Ind Microbiol. 1995;14:178–185. [Google Scholar]
- 13.Fründ C, Cohen Y. Diurnal cycles of sulfate reduction under oxic conditions in cyanobacterial mats. Appl Environ Microbiol. 1992;58:70–77. doi: 10.1128/aem.58.1.70-77.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gould W D, Bechard G, Lortie L. The nature and role of microorganisms in the tailings environment. In: Jambor J L, Blowes D W, editors. Environmental geochemistry of sulfide mine-wastes. Ontario, Canada: Mineralogical Association of Canada, Waterloo; 1994. pp. 185–199. [Google Scholar]
- 15.Harrison A P. The acidophilic thiobacilli and other acidophilic bacteria that share their habitat. Annu Rev Microbiol. 1984;38:265–292. doi: 10.1146/annurev.mi.38.100184.001405. [DOI] [PubMed] [Google Scholar]
- 16.Hastings D, Emerson S. Sulfate reduction in the presence of low oxygen levels in the water column of the Cariaco Trench. Limnol Oceanogr. 1988;33:391–396. [Google Scholar]
- 17.Jorgensen B B. Bacterial sulfate reduction within reduced microniches of oxidized marine sediments. Mar Biol. 1977;41:7–17. [Google Scholar]
- 18.Klee A J. A computer program for the determination of most probable number and its confidence limits. J Microbiol Methods. 1993;18:91–98. [Google Scholar]
- 19.Kleinmann R L P, Hedin R S, Edenborn H M. Proceedings of the 2nd International Conference on Abatement and Acidic Drainage. Vol. 4. MEND Secretariat, Ottawa, Ontario, Canada; 1991. Biological treatment of mine water—an overview; pp. 27–43. [Google Scholar]
- 20.Lovley D R. Dissimilatory Fe(III) and Mn(IV) reduction. Microbiol Rev. 1991;55:259–287. doi: 10.1128/mr.55.2.259-287.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lovley D R. Dissimilatory metal reduction. Annu Rev Microbiol. 1993;47:263–290. doi: 10.1146/annurev.mi.47.100193.001403. [DOI] [PubMed] [Google Scholar]
- 22.Lovley D R, Phillips E J P. Organic matter mineralization with reduction of ferric iron in anaerobic sediments. Appl Environ Microbiol. 1986;51:683–689. doi: 10.1128/aem.51.4.683-689.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lucy J K. Hydrologic and geochemical controls on metals partitioning in the vadose zone and shallow groundwater zone of a highly-contaminated floodplain near Butte, Montana. Masters thesis. Missoula, Mont: University of Montana; 1995. [Google Scholar]
- 24.Miller C L, Landa E R, Updegraff D M. Ecological aspects of microorganisms inhabiting uranium mill tailings. Microb Ecol. 1987;14:141–155. doi: 10.1007/BF02013019. [DOI] [PubMed] [Google Scholar]
- 25.Moore J N, Ficklin W A, Johns C. Partitioning of arsenic and metals in reducing sulfidic sediments. Environ Sci Technol. 1988;22:432–437. [Google Scholar]
- 26.Moore J N, Luoma S N. Hazardous wastes from large-scale metal extraction. Environ Sci Technol. 1990;24:1278–1285. [Google Scholar]
- 27.Moser D P, Nealson K H. Growth of the facultative anaerobe Shewanella putrefaciens by elemental sulfur reduction. Appl Environ Microbiol. 1996;62:2100–2105. doi: 10.1128/aem.62.6.2100-2105.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Multitech (ed.). 1987. Silver Bow Creek remedial investigation final report/ground water and tailings investigation. Multitech, Helena, Mont.
- 29.Myers C R, Nealson K H. Microbial reduction of manganese oxides: interactions with iron and sulfur. Geochim Cosmochim Acta. 1988;52:2727–2732. [Google Scholar]
- 30.Nealson K H, Saffarini D. Iron and manganese in anaerobic respiration: environmental significance, physiology, and regulation. Annu Rev Microbiol. 1994;48:311–343. doi: 10.1146/annurev.mi.48.100194.001523. [DOI] [PubMed] [Google Scholar]
- 31.Newport P J, Nedwell D B. The mechanism of inhibition of Desulfovibrio and Desulfotomaculum species by selenate and molybdate. J Appl Bacteriol. 1988;65:419–423. [Google Scholar]
- 32.Nimick D A, Moore J N. Prediction of water-soluble metal concentrations in fluvially deposited tailings sediment, Upper Clark Fork Valley, Montana, U.S.A. Appl Geochem. 1991;6:635–646. [Google Scholar]
- 33.Perry K A. Sulfate-reducing bacteria and immobilization of metals. Mar Georesources Geotechnol. 1995;13:33–39. [Google Scholar]
- 34.Ribet I, Ptacek C J, Blowes D W, Jambor J L. The potential for metal release by reductive dissolution of weathered mine tailings. J Contam Hydrol. 1995;17:239–273. [Google Scholar]
- 35.Rusin P A, Quintana L, Sinclair N A, Arnold R G, Oden K L. Physiology and kinetics of manganese-reducing Bacillus polymyxa strain D1 isolated from manganiferous silver ore. Geomicrobiol J. 1991;9:13–25. [Google Scholar]
- 36.Schafer and Associates. Streambank tailings and revegetation studies. STARS Phase III final report. SBC-STARS-III--D-RO-013193. Bozeman, Mont: Montana State University; 1993. [Google Scholar]
- 37.Schafer and Associates, Reclamation Research Unit (MSU), and CH2M Hill. Final summary report. STARS Phase II: field scale treatability study plot construction. SBC-STARS-PHASE I-F-RI-051789. Bozeman, Mont: Montana State University; 1989. [Google Scholar]
- 38.Schallenberg M, Kalff J, Rasmussen J B. Solutions to problems in enumerating sediment bacteria by direct counts. Appl Environ Microbiol. 1989;55:1214–1219. doi: 10.1128/aem.55.5.1214-1219.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schippers A, Hallman R, Wentzien S, Sand W. Microbial diversity in uranium mine waste heaps. Appl Environ Microbiol. 1995;61:2930–2935. doi: 10.1128/aem.61.8.2930-2935.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Silver M. Distribution of iron-oxidizing bacteria in the Nordic Uranium tailings deposit, Elliot Lake, Ontario, Canada. Appl Environ Microbiol. 1987;53:846–852. doi: 10.1128/aem.53.4.846-852.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Southham G, Beveridge T J. Enumeration of thiobacilli within pH-neutral and acidic mine tailings and their role in the development of secondary mineral soil. Appl Environ Microbiol. 1992;58:1904–1912. doi: 10.1128/aem.58.6.1904-1912.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Telang A J, Voordouw G, Ebert S, Sifeldeen N, Foght J M, Fedorak P M, Westlake D W S. Characterization of the diversity of sulfate-reducing bacteria in soil and mining waste water environments by nucleic acid hybridization techniques. Can J Microbiol. 1994;40:955–964. doi: 10.1139/m94-152. [DOI] [PubMed] [Google Scholar]
- 43.Teske A, Wawer C, Muyzer G, Ramsing N B. Distribution of sulfate-reducing bacteria in a stratified fjord (Mariager Fjord, Denmark) as evaluated by most-probable-number counts and denaturing gradient gel electrophoresis of PCR-amplified ribosomal DNA fragments. Appl Environ Microbiol. 1996;62:1405–1415. doi: 10.1128/aem.62.4.1405-1415.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tuttle J H, Dugan P R. Inhibition of growth, iron, and sulfur oxidation in Thiobacillus ferrooxidans by simple organic compounds. Can J Microbiol. 1976;22:719–730. doi: 10.1139/m76-105. [DOI] [PubMed] [Google Scholar]
- 45.Wielinga B W, Benner S G, Brick C M, Moore J N, Gannon J E. Geomicrobiological profile through the hyporheic zone in a historic mining floodplain. In: Stanford J A, Valett H M, editors. Proceedings of the Second International Conference on Ground Water Ecology. Herndon, Va: American Water Resources Association; 1994. pp. 267–276. [Google Scholar]
- 46.Wielinga B W. Ph.D. thesis. Missoula, Mont: University of Montana; 1996. [Google Scholar]