Abstract
Methionine restriction (MetR) can extend lifespan and delay the onset of aging‐associated pathologies in most model organisms. Previously, we showed that supplementation with the metabolite S‐adenosyl‐L‐homocysteine (SAH) extends lifespan and activates the energy sensor AMP‐activated protein kinase (AMPK) in the budding yeast Saccharomyces cerevisiae. However, the mechanism involved and whether SAH can extend metazoan lifespan have remained unknown. Here, we show that SAH supplementation reduces Met levels and recapitulates many physiological and molecular effects of MetR. In yeast, SAH supplementation leads to inhibition of the target of rapamycin complex 1 (TORC1) and activation of autophagy. Furthermore, in Caenorhabditis elegans SAH treatment extends lifespan by activating AMPK and providing benefits of MetR. Therefore, we propose that SAH can be used as an intervention to lower intracellular Met and confer benefits of MetR.
Keywords: Caenorhabditis elegans, methionine restriction (MetR), Saccharomyces cerevisiae, S‐adenosyl‐L‐homocysteine (SAH), S‐adenosyl‐L‐methionine (SAM)
A model depicting how SAH extends lifespan. The intake of SAH reduces intracellular Met and induces benefits associated with MetR in yeast and nematodes. SAH treatment extended lifespan via inhibition of mTORC1 and activation of autophagy and AMPK.

1.
Dietary restriction, including MetR, is an effective strategy for promoting longevity and counteracting age‐related morbidities (Ables & Johnson, 2017; Parkhitko et al., 2019). In addition, genetic manipulation or pharmacological inhibition of Met metabolic pathways (Annibal et al., 2021; Hepowit et al., 2021; Johnson & Johnson, 2014; Obata & Miura, 2015; Ogawa et al., 2016; Ruckenstuhl et al., 2014) and a Met‐restricted diet prolong lifespan (Orentreich et al., 1993; Wu et al., 2013). Several studies indicate that a MetR diet is possible for humans (Dong et al., 2018; Gao et al., 2019; McCarty et al., 2009; Olsen et al., 2020, 2021), but long‐term compliance to such a diet is considered problematic. Previously, we showed that a yeast mutant that accumulates S‐adenosyl‐L‐methionine (SAM) to high levels exhibited reduced intracellular Met and lifespan extension mediated through AMPK activation (Ogawa et al., 2016) (Figure 1a). We also showed that in a wild‐type (WT) strain, supplementation with SAH increased SAM levels, activating AMPK, and extending lifespan. However, we did not determine whether SAH supplementation might be sufficient to reduce Met levels or determine how SAH supplementation leads to SAM accumulation and lifespan extension.
FIGURE 1.
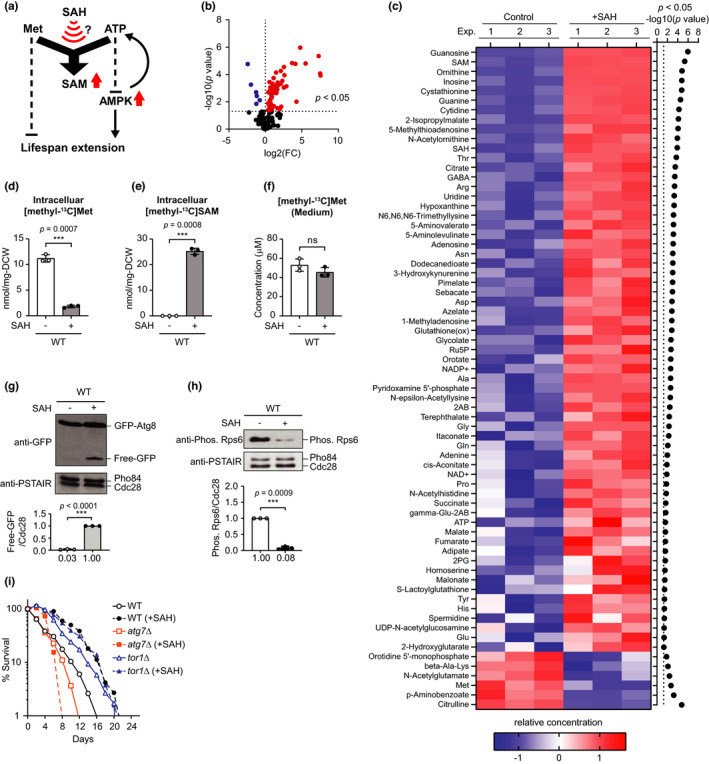
SAH reduces intracellular Met and induces MetR‐like conditions in S. cerevisiae. (a) Model for yeast longevity mediated by the stimulation of SAM synthesis by SAH. Volcano plot (b) or heat map (c) showing metabolite levels in WT cells with or without SAH supplementation. n = 3. FDR < 0.05, two‐sided unpaired t‐test. See also in Table S1. Intracellular [methyl‐13C]Met (d), Intracellular [methyl‐13C]SAM levels (e), and [methyl‐13C]Met levels in the medium (f) were assessed using CE‐TOFMS. Mean ± S.D, n = 3, two‐sided unpaired t‐test. The relative intensity of free GFP (g) or phosphorylated Rps6 (h) normalized to Cdc28 is shown. Mean ± SD, n = 3, two‐sided unpaired t‐test. (i) The CLS curve is indicated. (d–f, g, h) ns, not significant; ***p < 0.001. (i) Statistical analyses are shown in Table S2
To investigate the basis for SAH‐mediated longevity, we performed metabolomics (CE‐TOFMS) analysis of a WT S. cerevisiae strain. In response to 1 mM SAH, which can extend lifespan (Ogawa et al., 2016), 148 metabolites were detected (Table S1). Of these metabolites, 63 and 6 were significantly up‐regulated and down‐regulated, respectively (Figure 1b,c). As previously reported, SAH administration increased levels of SAH and SAM, a methyl group donor (Ogawa et al., 2016) (Figure 1c and Figure S1a). SAH is a potent competitive inhibitor of SAM‐dependent methyltransferases, and SAH accumulation thereby impairs cell growth (Christopher et al., 2002). Previously, we showed that exogenous SAM improved the growth of the SAH hydrolase mutant sah1‐1, which accumulates high levels of SAH, suggesting that SAM is protective against SAH‐dependent growth inhibition (Mizunuma et al., 2004). Therefore, we speculate that SAH supplementation can increase SAM synthesis through an unknown mechanism. Since SAM synthesis requires Met (Figure 1a), stimulating SAM production can decrease the quantity of intracellular Met. Notably, among the amino acids, Met exhibited significantly reduced levels after SAH supplementation (Figure S1b).
To investigate whether the decrease in intracellular Met was due to accelerated consumption, we substituted L‐[methyl‐13C]Met in the culture medium for Met and followed its fate with or without addition of SAH. SAH supplementation significantly decreased [methyl‐13C]Met and increased [methyl‐13C]SAM intracellularly compared with the control (Figure 1d,e). Furthermore, extracellular metabolomic data showed that after SAH treatment [methyl‐13C]Met levels were comparable with that of the control (Figure 1f). These results suggest that SAH reduces Met levels by converting endogenous Met to SAM.
The lower Met content in SAH‐treated cells suggests that longevity from SAH supplementation can induce a MetR state. Hence, since MetR extends chronological lifespan (CLS) (Fabrizio & Longo, 2007) in an autophagy‐dependent manner (Plummer & Johnson, 2019; Ruckenstuhl et al., 2014), we investigated the effect of SAH on autophagy by monitoring the GFP‐Atg8 cleavage assay (Nair et al., 2011). SAH treatment increased degradation of the autophagy marker GFP‐Atg8 to yield free GFP (Figure 1g), suggesting that SAH administration promotes autophagy. Furthermore, since TORC1 negatively regulates autophagy (Shimobayashi & Hall, 2014), we tested whether SAH inhibits TORC1. In WT cells, treatment with SAH reduced the phosphorylation of Rps6, a homolog of ribosomal protein S6 (Figure 1h), which is phosphorylated by TORC1 (Wullschleger & Hall, 2006), suggesting that SAH reduces levels of TORC1 activity. Additionally, the CLS of tor1Δ and atg7Δ (deletion mutant of an essential autophagic machinery component) cells was not prolonged compared with WT cells treated with SAH (Figure 1i). Thus, consistent with the induction of MetR, SAH extends lifespan through the inhibition of TORC1 and activation of autophagy.
Subsequently, to determine whether SAH acts as an anti‐aging metabolite in a metazoan, we investigated its effects on the nematode C. elegans. SAH treatment extended the lifespan of WT animals in a concentration‐dependent manner, with 50 μM SAH inducing the most significant increase (Figure 2a). We also obtained similar results with 50 μM SAH in the absence of the reproduction blocker fluorodeoxyuridine (FUdR), or when SAH was supplemented only during adulthood. The latter finding ruled out a role for possible developmental effects (Figures S2a,b). Notably, SAH did not affect food consumption, brood size, or viability (Figure S2c,d,e). Additionally, the longevity‐extending effects of SAH were independent of bacterial metabolism (Figure S2f). SAH also partially prevented the aging‐associated decrease in physical capacity (Figure S2g). Altogether, these results suggest that SAH mediates phylogenetically conserved anti‐aging effects.
FIGURE 2.
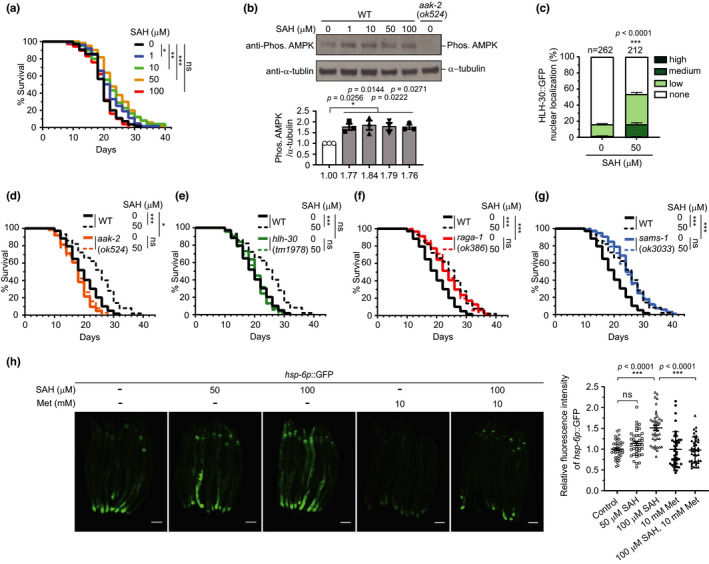
SAH extends lifespan via activation of AMPK, inhibition of mTORC1, and activation of autophagy in C. elegans. Representative survival curve (a) or relative AMPK phosphorylation level (b) of N2 WT animals in C. elegans, either untreated or treated with 1, 10, 50, 100 µM SAH. Mean ± SEM, n = 3, one‐way ANOVA with Tukey's correction. (c) Quantification of HLH‐30::GFP nuclear localization. n = number of worms. Mean ± SEM, chi‐square test. (d–g) Representative survival curves. (h) Representative images of hsp‐6p::GFP. Scale bar = 100 µm. Quantification of relative GFP intensity in the intestine is shown. Mean ± SEM, n = 40, one‐way ANOVA with Tukey's correction. (a–h) ns, not significant; *p < 0.05; **p < 0.01; ***p < 0.001. (a, d–g) Statistical analyses are shown in Table S3
Similar to findings in yeast (Ogawa et al., 2016), SAH supplementation increased the phosphorylation of AAK‐2, a C. elegans homolog of the catalytic AMPK subunit (Figure 2b). HLH‐30, an orthologue of the human transcription factor TFEB, is a master regulator that promotes autophagy (Settembre et al., 2011). Thus, we examined the autophagy activity by monitoring a GFP‐tagged HLH‐30 that translocates to the nucleus upon mechanistic TORC1 (mTORC1) inhibition (Settembre et al., 2012). SAH induced HLH‐30 nuclear accumulation (Figure 2c), suggesting that it can reduce mTORC1 activity and promote autophagy. Subsequently, to investigate how the SAH extends lifespan, we used the loss‐of‐function mutant strains aak‐2(ok524) (Apfeld et al., 2004), hlh‐30(tm1978) (Visvikis et al., 2014), and raga‐1(ok386) (Schreiber et al., 2010). No effect of SAH on lifespan extension in these mutants was observed, suggesting that SAH extends lifespan through a mechanism dependent on AMPK, mTORC1, and autophagy (Figure 2d,e,f).
sams‐1 encodes an evolutionarily conserved SAM synthetase, knockdown of which extends lifespan (Hansen et al., 2005). SAH does not increase median lifespan further in sams‐1(ok3033), a loss‐of‐function mutant allele of sams‐1 (Walker et al., 2011) (Figure 2g), consistent with the idea that SAH extends lifespan through SAM synthesis. These results suggest that lifespan extension in the sams‐1 strain, which is unable to produce SAM, is likely to occur through a mechanism entirely different from MetR.
Additionally, the expression of HSP‐6, an orthologue of the mitochondrial chaperone mitochondrial Hsp70, is induced by MetR through its induction of the mitochondrial unfolded protein response (UPRmt) (Amin et al., 2020). Supplementation with 100 μM SAH significantly increased the expression level of hsp‐6p::GFP (Figure 2h). Furthermore, this increase was suppressed upon Met supplementation, consistent with a model of MetR in C. elegans.
In conclusion, our results suggest that SAH extends lifespan by inducing MetR or mimicking its downstream effects. Since the lifespan‐extending effects of SAH are conserved in yeast and nematodes, and MetR extends the lifespan of many species, exposure to SAH is expected to have multiple benefits across evolutionary boundaries. Our findings offer the enticing possibility that in humans the benefits of a MetR diet can be achieved by promoting Met reduction with SAH. The use of endogenous metabolites, such as SAH, is considered safer than drugs and other substances, suggesting that it may be one of the most feasible ways to prevent age‐related diseases.
EXPERIMENTAL PROCEDURES
Full detailed methods and experimental procedures are available in Appendix S1.
CONFLICT OF INTEREST
None declared.
AUTHOR CONTRIBUTIONS
M.M. conceived the study and designed the experiments. T.O. and M.M. carried out C. elegans experiments. T.O., K.M., and Y.K. performed yeast experiments. M.K. performed [methyl‐13C]Met analyses. T.S. performed metabolome analyses. T.O. and Y.O. discussed the results and contributed to the improvement of the manuscript. T.K.B. and M.M. wrote the manuscript. T.K.B. and M.M. supervised the work.
Supporting information
Supplementary Material
Fig S1‐S3
Table S1
Table S2
Table S3
ACKNOWLEDGMENTS
We thank Takeshi Noda and the National BioResource Project/Yeast Genetic Resource Center, Japan, for plasmid and yeast strain. We are grateful to Kaori Igarashi, Ayano Ueno, Maki Ohishi, Ikuko Fukuba, Eri Sanda, Maiko Baba, Feimei Zhu, Juliane Campos, and Laura Paulette Fernandez Cardenas for helpful technical assistance and advice. We also thank Dai Hirata, Kazunori Kume and Makoto Horikawa for helpful discussion. M.M. is supported by the PRIME from Japan Agency for Medical Research and Development JP21gm6110029, JSPS KAKENHI Scientific Research (B) 16H04898, a Challenging Exploratory Research grant 19K22285, Ohsumi Frontier Science Foundation, and Takeda Science Foundation, T.O. and M.M. by the Program for Advancing Strategic International Networks to Accelerate the Circulation of Talented Researchers (S2902), and T.K.B by NIH grants GM122610 and AG54215. T.O. is the recipient of a JSPS Overseas Research Fellowships. K.M. is the recipient of the Research Fellowship of the JSPS for Young Scientists (DC1). Generous support from the "Distinguished Researchers" program of Hiroshima University and HiHA are gratefully acknowledged.
Ogawa, T. , Masumura, K. , Kohara, Y. , Kanai, M. , Soga, T. , Ohya, Y. , Blackwell, T. K. , & Mizunuma, M. (2022). S‐adenosyl‐L‐homocysteine extends lifespan through methionine restriction effects. Aging Cell, 21, e13604. 10.1111/acel.13604
Takafumi Ogawa and Koji Masumura contributed equally to this work.
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- Ables, G. P. , & Johnson, J. E. (2017). Pleiotropic responses to methionine restriction. Experimental Gerontology, 94, 83–88. 10.1016/j.exger.2017.01.012 [DOI] [PubMed] [Google Scholar]
- Amin, M. R. , Mahmud, S. A. , Dowgielewicz, J. L. , Sapkota, M. , & Pellegrino, M. W. (2020). A novel gene‐diet interaction promotes organismal lifespan and host protection during infection via the mitochondrial UPR. PLoS Genetics, 16, e1009234. 10.1371/journal.pgen.1009234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Annibal, A. , Tharyan, R. G. , Schonewolff, M. F. , Tam, H. , Latza, C. , Auler, M. M. K. , Gronke, S. , Partridge, L. , & Antebi, A. (2021). Regulation of the one carbon folate cycle as a shared metabolic signature of longevity. Nature Communications, 12, 3486. 10.1038/s41467-021-23856-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Apfeld, J. , O'Connor, G. , McDonagh, T. , DiStefano, P. S. , & Curtis, R. (2004). The AMP‐activated protein kinase AAK‐2 links energy levels and insulin‐like signals to lifespan in C. elegans . Genes and Development, 18, 3004–3009. 10.1101/gad.1255404 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christopher, S. A. , Melnyk, S. , James, S. J. , & Kruger, W. D. (2002). S‐adenosylhomocysteine, but not homocysteine, is toxic to yeast lacking cystathionine beta‐synthase. Molecular Genetics and Metabolism, 75, 335–343. 10.1016/S1096-7192(02)00003-3 [DOI] [PubMed] [Google Scholar]
- Dong, Z. , Sinha, R. , & Richie, J. P. Jr (2018). Disease prevention and delayed aging by dietary sulfur amino acid restriction: translational implications. Annals of the New York Academy of Sciences, 1418, 44–55. 10.1111/nyas.13584 [DOI] [PubMed] [Google Scholar]
- Fabrizio, P. , & Longo, V. D. (2007). The chronological life span of Saccharomyces cerevisiae. Methods in Molecular Biology, 371, 89–95. 10.1007/978-1-59745-361-5_8 [DOI] [PubMed] [Google Scholar]
- Gao, X. , Sanderson, S. M. , Dai, Z. , Reid, M. A. , Cooper, D. E. , Lu, M. , Richie, J. P. Jr , Ciccarella, A. , Calcagnotto, A. , Mikhael, P. G. , Mentch, S. J. , Liu, J. , Ables, G. , Kirsch, D. G. , Hsu, D. S. , Nichenametla, S. N. , & Locasale, J. W. (2019). Dietary methionine influences therapy in mouse cancer models and alters human metabolism. Nature, 572, 397–401. 10.1038/s41586-019-1437-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen, M. , Hsu, A. L. , Dillin, A. , & Kenyon, C. (2005). New genes tied to endocrine, metabolic, and dietary regulation of lifespan from a Caenorhabditis elegans genomic RNAi screen. PLoS Genetics, 1, 119–128. 10.1371/journal.pgen.0010017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hepowit, N. L. , Macedo, J. K. A. , Young, L. E. A. , Liu, K. , Sun, R. C. , MacGurn, J. A. , & Dickson, R. C. (2021). Enhancing lifespan of budding yeast by pharmacological lowering of amino acid pools. Aging (Albany NY), 13, 7846–7871. 10.18632/aging.202849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson, J. E. , & Johnson, F. B. (2014). Methionine restriction activates the retrograde response and confers both stress tolerance and lifespan extension to yeast, mouse and human cells. PLoS One, 9, e97729. 10.1371/journal.pone.0097729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCarty, M. F. , Barroso‐Aranda, J. , & Contreras, F. (2009). The low‐methionine content of vegan diets may make methionine restriction feasible as a life extension strategy. Medical Hypothesis, 72, 125–128. 10.1016/j.mehy.2008.07.044 [DOI] [PubMed] [Google Scholar]
- Mizunuma, M. , Miyamura, K. , Hirata, D. , Yokoyama, H. , & Miyakawa, T. (2004). Involvement of S‐adenosylmethionine in G1 cell‐cycle regulation in Saccharomyces cerevisiae. Proceedings of the National Academy of Sciences of the United States of America, 101, 6086–6091. 10.1073/pnas.0308314101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nair, U. , Thumm, M. , Klionsky, D. J. , & Krick, R. (2011). GFP‐Atg8 protease protection as a tool to monitor autophagosome biogenesis. Autophagy, 7, 1546–1550. 10.4161/auto.7.12.18424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obata, F. , & Miura, M. (2015). Enhancing S‐adenosyl‐methionine catabolism extends Drosophila lifespan. Nature Communications, 6, 8332. 10.1038/ncomms9332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogawa, T. , Tsubakiyama, R. , Kanai, M. , Koyama, T. , Fujii, T. , Iefuji, H. , Soga, T. , Kume, K. , Miyakawa, T. , Hirata, D. , & Mizunuma, M. (2016). Stimulating S‐adenosyl‐l‐methionine synthesis extends lifespan via activation of AMPK. Proceedings of the National Academy of Sciences of the United States of America, 113, 11913–11918. 10.1073/pnas.1604047113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen, T. , Øvrebø, B. , Haj‐Yasein, N. , Lee, S. , Svendsen, K. , Hjorth, M. , Bastani, N. E. , Norheim, F. , Drevon, C. A. , Refsum, H. , & Vinknes, K. J. (2020). Effects of dietary methionine and cysteine restriction on plasma biomarkers, serum fibroblast growth factor 21, and adipose tissue gene expression in women with overweight or obesity: A double‐blind randomized controlled pilot study. Journal of Translational Medicine, 18, 122. 10.1186/s12967-020-02288-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen, T. , Øvrebø, B. , Turner, C. , Bastani, N. E. , Refsum, H. , & Vinknes, K. J. (2021). Effects of short‐term methionine and cysteine restriction and enrichment with polyunsaturated fatty acids on oral glucose tolerance, plasma amino acids, fatty acids, lactate and pyruvate: results from a pilot study. BMC Research Note, 14, 43. 10.1186/s13104-021-05463-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orentreich, N. , Matias, J. R. , DeFelice, A. , & Zimmerman, J. A. (1993). Low methionine ingestion by rats extends life span. The Journal of Nutrition, 123, 269–274. 10.1093/jn/123.2.269 [DOI] [PubMed] [Google Scholar]
- Parkhitko, A. A. , Jouandin, P. , Mohr, S. E. , & Perrimon, N. (2019). Methionine metabolism and methyltransferases in the regulation of aging and lifespan extension across species. Aging Cell, 18, e13034. 10.1111/acel.13034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plummer, J. D. , & Johnson, J. E. (2019). Extension of cellular lifespan by methionine restriction involves alterations in central carbon metabolism and is mitophagy‐dependent. Frontiers in Cell and Developmental Biology, 7, 301. 10.3389/fcell.2019.00301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruckenstuhl, C. , Netzberger, C. , Entfellner, I. , Carmona‐Gutierrez, D. , Kickenweiz, T. , Stekovic, S. , Gleixner, C. , Schmid, C. , Klug, L. , Sorgo, A. G. , Eisenberg, T. , Büttner, S. , Mariño, G. , Koziel, R. , Jansen‐Dürr, P. , Fröhlich, K. , Kroemer, G. , & Madeo, F. (2014). Lifespan extension by methionine restriction requires autophagy‐dependent vacuolar acidification. PLoS Genetics, 10, e1004347. 10.1371/journal.pgen.1004347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiber, M. A. , Pierce‐Shimomura, J. T. , Chan, S. , Parry, D. , & McIntire, S. L. (2010). Manipulation of behavioral decline in Caenorhabditis elegans with the rag GTPase raga‐1. PLoS Genetics, 6, e1000972. 10.1371/journal.pgen.1000972 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Settembre, C. , Di Malta, C. , Polito, V. A. , Arencibia, M. G. , Vetrini, F. , Erdin, S. , Erdin, S. U. , Huynh, T. , Medina, D. , Colella, P. , Sardiello, M. , Rubinsztein, D. C. , & Ballabio, A. (2011). TFEB links autophagy to lysosomal biogenesis. Science, 332, 1429–1433. 10.1126/science.1204592 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Settembre, C. , Zoncu, R. , Medina, D. L. , Vetrini, F. , Erdin, S. , Erdin, S. , Erdin, S. , Huynh, T. , Ferron, M. , Karsenty, G. , Vellard, M. C. , Facchinetti, V. , Sabatini, D. M. , & Ballabio, A. (2012). A lysosome‐to‐nucleus signalling mechanism senses and regulates the lysosome via mTOR and TFEB. Embo Journal, 31, 1095–1108. 10.1038/emboj.2012.32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimobayashi, M. , & Hall, M. N. (2014). Making new contacts: The mTOR network in metabolism and signalling crosstalk. Nature Reviews Molecular Cell Biology, 15, 155–162. 10.1038/nrm3757 [DOI] [PubMed] [Google Scholar]
- Visvikis, O. , Ihuegbu, N. , Labed, S. A. , Luhachack, L. G. , Alves, A. F. , Wollenberg, A. C. , Stuart, L. M. , Stormo, G. D. , & Irazoqui, J. E. (2014). Innate host defense requires TFEB‐mediated transcription of cytoprotective and antimicrobial genes. Immunity, 40, 896–909. 10.1016/j.immuni.2014.05.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker, A. K. , Jacobs, R. L. , Watts, J. L. , Rottiers, V. , Jiang, K. , Finnegan, D. M. , Shioda, T. , Hansen, M. , Yang, F. , Niebergall, L. J. , Vance, D. E. , Tzoneva, M. , Hart, A. C. , & Näär, A. M. (2011). A conserved SREBP‐1/phosphatidylcholine feedback circuit regulates lipogenesis in metazoans. Cell, 147, 840–852. 10.1016/j.cell.2011.09.045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu, Z. , Song, L. , Liu, S. Q. , & Huang, D. (2013). Independent and additive effects of glutamic acid and methionine on yeast longevity. PLoS One, 8, e79319. 10.1371/journal.pone.0079319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wullschleger, S. , Loewith, R. , & Hall, M. N. (2006). TOR signaling in growth and metabolism. Cell, 124, 471–484. 10.1016/j.cell.2006.01.016 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material
Fig S1‐S3
Table S1
Table S2
Table S3
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


