Abstract
Neutrophil extracellular traps (NETs) are extracellular webs composed of neutrophil granular and nuclear elements. Because of the potentially dangerous amplification circuit between inflammation and tissue damage, NETs are becoming one of the investigated components in the current Coronavirus Disease 2019 (COVID-19) pandemic. The purpose of this systematic review is to summarize studies on the role of NETs in determining the prognosis of COVID-19 patients. The study used six databases: PubMed, Science Direct, EBSCOHost, Europe PMC, ProQuest, and Scopus. This literature search was implemented until October 31, 2021. The search terms were determined specifically for each databases, generally included the Neutrophil Extracellular Traps, COVID-19, and prognosis. The Newcastle Ottawa Scale (NOS) was then used to assess the risk of bias. Ten studies with a total of 810 participants were chosen based on the attainment of the prerequisite. Two were of high quality, seven were of moderate quality, and the rest were of low quality. The majority of studies compared COVID-19 to healthy control. Thrombosis was observed in three studies, while four studies recorded the need for mechanical ventilation. In COVID-19 patients, the early NETs concentration or the evolving NETs degradations can predict patient mortality. Based on their interactions with inflammatory and organ dysfunction markers, it is concluded that NETs play a significant role in navigating the severity of COVID-19 patients and thus impacting their prognosis.
Supplementary Information
The online version contains supplementary material available at 10.1007/s12026-022-09293-w.
Keywords: COVID-19, Mechanical ventilation, Mortality, Neutrophil extracellular traps, Thrombosis
Introduction
Neutrophil extracellular traps (NETs) are extracellular webs made up of Deoxyribonucleic Acid (DNA), histones (H1, H2A, H2B, H3, and H4), oxidant enzymes, microbicidal proteins, and cytoplasmic proteins that originate from decondensed chromatin [1]. Other components include neutrophil elastase (NE), myeloperoxidase (MPO), cathepsin G, proteinase 3, calgranulin, α-defensin, hydrolase, lysozyme, azurocidin, alcaic phosphatase, lactoferrins, lysozyme, cathelicidins, collagenase, gelatinase, catalase, catalase, cytokeratin-10, actin, myosin, cytoplasmic proteins, glycolytic enzymes, pentraxin, matrix metalloproteinase-9 (MMP-9), and peptidoglycan [2–6]. NETs are one of the neutrophil tactics for restricting and removing external infections [7, 8]. Microbes such as bacteria, fungi, and viruses are susceptible to NETs in many instances, including being caught, killed, or having their growth hindered [9]. It applies to previous research on human immunodeficiency virus (HIV) infection (by preventing HIV invasion to CD4 cells and promoting HIV-1 elimination through myeloperoxidase and α-defensin involvement) [10, 11]. In contrast, NETs were associated with overall disease severity and a worse outcome during influenza cases, primarily due to acute lung injury [12].
During several thrombo-inflammatory conditions, such as sepsis, thrombosis, and respiratory failure, neutrophils produce NETs to confine pathogens [13, 14]. Lack of proper NET control, on the other hand, could result in immunothrombosis [15]. In short, the role of NETs is a double-edged sword, posing a different effect related to certain levels of the compound and underlying circumstances [16]. Thus, this molecule is highly appealing to researchers during the current COVID-19 pandemic, as the disease has been strongly associated with coagulation abnormalities and thrombotic events [17].
NETs are becoming one of the investigated immune-related components in the present Coronavirus Disease 2019 (COVID-19), a pandemic caused by the global expansion of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). It is because of the potentially dangerous amplification circuit between inflammation and tissue damage induced by NET dysregulation, mainly by the involvement of elastase [18]. Elastase demonstrated potency in speeding up virus entry and generating hypertension, thrombosis, and vasculitis [18]. Excessive NET production during extended duration of inflammation increases the risk of adverse effects such as thromboembolic problems and impairment to surrounding tissues and organs [19]. However, there is still a lack of consensus regarding the empirical literature on NETs, which needs to be addressed. Also, the needs for creating better evidence for the prognostic indicator of COVID-19 must be undertaken. Hence, the purpose of this systematic review is to synthesize studies referring to the role of NETs for the prognostication of COVID-19 patients.
Methods
The manuscript has been arranged using the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) 2020 guidelines [20]. The study procedure was approved by all members of the review panel before the conduction of the literature search.
Literature search
An extensive literature search across multiple databases was done to identify literature about neutrophil extracellular traps and their prognostic role in COVID-19 patients. This topic was searched electronically in six directories: Europe PMC (728 hits), ProQuest (426 hits), Science Direct (244 hits), Scopus (21 hits), PubMed (12 hits), and EBSCOHost (3 hits), in addition to one additional search. The keywords used in the investigation were listed in Supplementary Table 1. The studies revealed through this database search were evaluated based on the titles and abstracts, and only those that met the inclusion criteria were considered. The search was accomplished by October 31, 2021, without any time constraints on the published work. We did not register our protocol on International Prospective Register of Systematic Reviews (PROSPERO) to prevent any unwanted delay in manuscript publication process.
Study selection
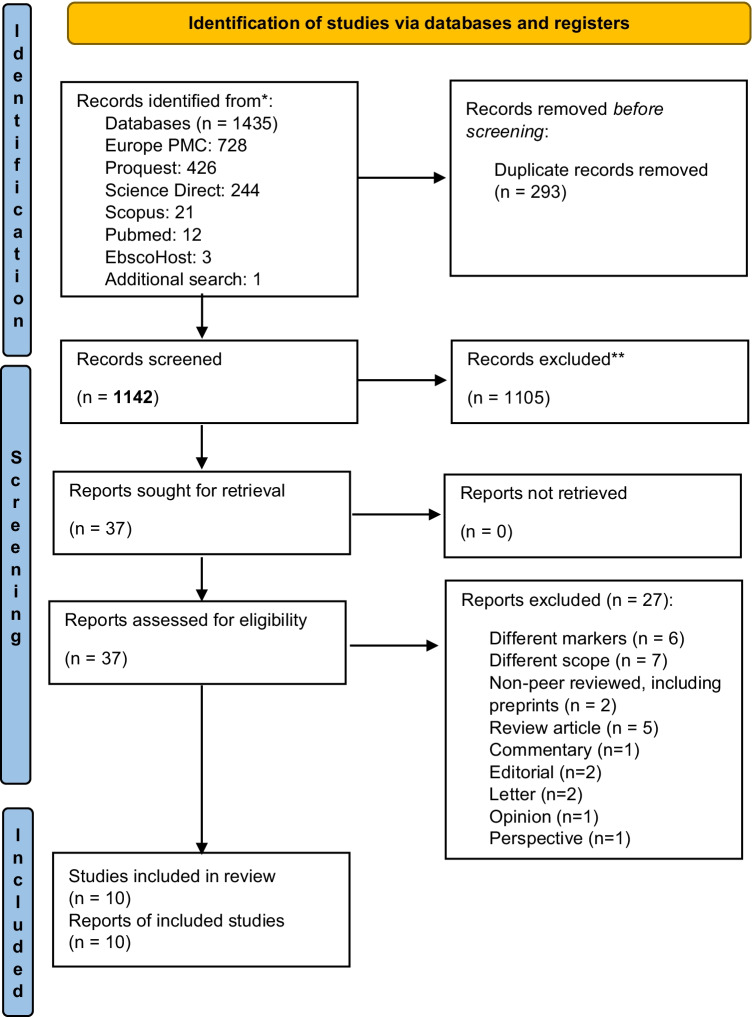
We examined the cross-sectional, case control, and cohort studies which assessed the capacity of NETs for delineating the prognosis of COVID-19 patients. We only utilized full-text manuscripts published in English to ensure data accuracy. Articles investigating other practicality of NETs (such as pathogenesis role (without clear clinical role) and interaction analysis) in COVID-19 patients, as well as literature reviews, case studies, clinical trials, protocols, conference abstracts, editorials, letters, correspondence, perspective, and posters, were all excluded. Duplicates were eliminated, and three independent reviewers (PL, TPU, and IAL) assessed titles and abstracts (primary screening) using Rayyan QCRI, a semi-automated abstract and title filtering software [21]. Any disagreements were discussed and resolved to reach an acquiescence. From the 1434 articles collected, we chose 37 manuscripts for full-text and reference appropriateness review (secondary screening). In total, ten studies met the criteria for data synthesis inclusion (Fig. 1).
Fig. 1.
PRISMA flow diagram of literature searching
Data extraction
Data were obtained from a series of studies. Three authors (ZH, PL, and TPU) extracted pertinent data from each study separately. The following information was extracted: authorship, years, country, study populations, study design, and main findings.
Risk of bias analysis
Following the systematic evaluation of each included study, a synthesis of the study findings related to the prognostic role of NETs in COVID-19 patients was performed. The Newcastle Ottawa Quality Assessment Scale (NOS) was applied to retrieve information from included research and then critically evaluate and analyze the methodologic quality of each study [22, 23], which is primarily used for case control or cohort research design. However, to assess the cross-sectional study, we were using a modified scale [24]. The NOS is divided into three sections: selection, comparability, and outcome. It is graded using the star method, with a maximum of 9 stars (or 10 stars for cross-sectional research) awarded across the three domains [25]. The method of evaluating the quality of the articles was as follows: Two independent investigators (TPU and PL) used the NOS to evaluate each of the included articles before comparing the scores assigned to each study. If the scores differed, they were discussed and resolved through mutual understanding. When the agreement could not be reached, a moderator (KM) was present to properly assess these articles for subsequent quality control to finalize the decision regarding manuscript inclusion. The included publications’ overall methodologic integrity was classified as having a high risk of bias (0–3 NOS points), a moderate risk of bias (4–6 NOS points), or a high risk of bias (7–9 NOS points) [26]. For cross-sectional studies, the modified NOS scale follows a different pattern, with low risk (7–10), moderate risk (5–6), and high risk (0–4) of bias [27]. Risk-of-bias VISualization (robvis) software was used to outline the risk of bias appraisal [28].
Results
Characteristics of included studies
Table 1 outlines the key characteristics of the included studies. The sample size ranged between 35 and 126 individuals. The total number of participants from all included research was 810, with 696 of them diagnosed with COVID-19 (confirmed by RT-PCR test), all of whom were adults. Four of those studies only examined COVID-19 patients [30, 34, 36, 37]. The most common markers used to determine the level of circulating NETs were MPO-DNA complexes [13, 31–33, 37], cfDNA [13, 29, 30, 37], and histones (either in H3Cit or histone-DNA complexes) [13, 29–31, 37]. The majority of the studies compared the surrogate marker of NET levels between COVID-19 patients and healthy donors, while only four studies contrasted it to the severity spectrum of COVID-19 [31, 32, 34, 35]. Thromboembolic events [29, 31–33, 37] and the need for mechanical ventilation [13, 29, 32] were the most frequently observed criterion for identifying increased disease severity. Eight studies were cohort [13, 29–34, 37], meanwhile the remaining publications were cross-sectional [35, 36] in design.
Table 1.
Summary and key findings of included studies
| Author (year) | Country | Methods | Study populations | Primary aim | Key findings |
|---|---|---|---|---|---|
| Huckriede, et.al. (2021) [29] | Netherlands | Cohort (prospective) | 126 participants (100 ICU-COVID-19 patients, 11 ICU-control patients, 15 healthy donors) | Study the levels and evolution of the DAMPS and NETs marker in relation to clinical parameters, ICU scoring systems and mortality in patients with severe COVID-19 |
• Surrogate markers of NETosis, cfDNA, differ significantly between ICU patients with COVID-19 and both types of control (p < 0.001), as do NE, H3Cit-DNA, and Gas6, but not sAXL • Different markers of NETs were correlated significantly with organ dysfunction markers dan events, including LDH, troponin I, the needs for dialysis, thromboembolic events, delirium, and ICUAW • No correlation between cfDNA, H3, NE, sAXL and GAS6 with SAPS-III or SOFA scores at the ICU admission • Dynamics of cfDNA (p = 0.042) and GAS6 (p = 0.031) was related with ventilator-free days, with lower decrease associated with longer use of ventilator. The same applies for GAS6 • The change in cfDNA and GAS6 concentration from early to late samples is able to significantly predict mortality |
| Ng, et. al. (2021) [30] | Sweden | Cohort (prospective) | 106 patients with COVID-19 | Evaluating the association of NETs markers with COVID-19 disease severity and clinical outcome |
• Plasma levels of H3Cit-DNA, cfDNA, and NE were all elevated in patients with COVID-19 compared with healthy individuals (p < 0.0001) • H3Cit-DNA, cfDNA, and NE all increased with increasing respiratory support •High levels of H3Cit-DNA, cfDNA, and NE at the time of admission were associated with poor clinical outcome (ICU admission and/or short-term mortality) (p = 0.0002, p < 0.0001, p = 0.0058, respectively) • NE, H3Cit-DNA, and cfDNA all was correlated with different markers of inflammation, impaired hemostasis, and endothelial injury. Out of 11 studied markers, NE significantly correlated with all parameters, H3Cit-DNA only has one insignificant correlation (with TAT), meanwhile for cfDNA, IL-6 and ADAMTS13 did not significantly correlated. It is linked with COVID-19-associated NET formation and immunothrombosis |
| Ouwendijk, et. al. (2021) [31] | Netherlands | Cohort (prospective) |
84 patients (77 patients with PCR-confirmed COVID-19 and ARDS, 7 healthy controls) |
Investigate the presence of NETs and its correlation with the pathogenesis of COVID-19 and ARDS |
• Higher surrogate markers (his-DNA complexes and MPO-DNA) for NETs in COVID-19 patients than healthy control was found • Baseline NETs abundance was negatively correlated with the number of days after hospital admission, figuring early accumulation of NETs • NETs were significantly correlated with CRP and IL-6 levels in patients requiring prolonged ICU admission • Plasma NETs levels in patients with fatal disease were correlated with overall disease severity (SOFA score) and severity of lung disease (Pao2/Fio2 ratio), but not the thromboembolism |
| Petito, et. al. (2021) [32] | Italy | Cohort (prospective) | 67 patients (36 with positive RT-PCR, 31 healthy subjects) | Analyze platelet, neutrophil activation markers, and MMP-9 role in COVID-19-related thrombosis |
• NET biomarkers (MPO-DNA) were found to be associated with disease severity (SOFA score), with higher levels in COVID-19 patients admitted to the ICU and requiring mechanical ventilation compared to patients with milder COVID-19 • Male COVID-19 patients had significantly higher MPO-DNA complexes than females |
| Skendros, et. al. (2020) [33] | Greece | Cohort (prospective) | 35 patients (25 COVID-19 patients and 10 control) | Investigate the role of neutrophils in COVID-19 thromboinflammation through complement activation |
• Significant increase (p < 0.05) in MPO-DNA complex in COVID-19 patients vs. healthy control was positively correlated with TAT activity • NETs are associated with activation of the TF/thrombin axis in COVID-19 patients, thus potentially contributing to immunothrombosis |
| Torres-Ruiz (2021)a [34] | Mexico | Cohort (prospective) | 121 patients with COVID-19 |
Create and validating a compound explanatory model to classify COVID-19 patients according to their disease severity and to predict adverse outcomes |
• Circulating NETs complexes were higher in severe and critical patients in comparison to mild/moderate subjects |
| Torres-Ruiz, et.al. (2021)b [35] | Mexico | Cross-sectional | 92 patients (82 with positive RT-PCR, 10 healthy donors) | Investigate the production of NETs and their role as inducers of macrophage and autoimmune responses in COVID-19 patients |
• COVID-19 patients produced a higher level of NETs vs. healthy donors • COVID-19 patients in severe states have a significant deficient degradation of NETs in comparison to mild/moderate COVID-19 (p = 0.0297) • Patient with COVID-19 (either mild/moderate, severe and critical) all have an increase in NETs-recognizing antibodies, and it is sequential according to disease conditions |
| Wang, et. al. (2020) [36] | China | Cross-sectional | 55 COVID-19 patients | Clarifying the pathogenesis of neutrophils leading to severe COVID-19 pneumonia | • In patients with COVID-19, NETs may adversely inhibit T and NK cell immune function via LGAS9 and CEACAM1, resulting in reduced antiviral immunity and direct lung tissue injury |
| Zuo, et al. (2020)a [13] | United States | Cohort (prospective) | 80 patients (50 COVID-19 patients and 30 control) | Measure various markers of NETs in sera of hospitalized COVID-19 patients and their relationship to the severity of illness |
• When compared to healthy controls, COVID-19 samples had greater levels of cfDNA, MPO-DNA complexes, and H3cit. As oxygenation diminished (the need for mechanical ventilation), all three NET markers increased • While compared to MPO-DNA and H3Cit, cfDNA has a strong correlation with several laboratory parameters, including CRP, D-dimer, LDH, and ANC |
| Zuo, et al. (2020)b [37] | United States | Cohort (prospective) | 44 COVID-19 patients (11 with thrombosis, 33 as control) | Describing thrombosis cases in patients hospitalized with COVID-19 and its association with neutrophil hyperactivity and NET release | • Three different markers of NETs (cfDNA (p < 0.001), MPO-DNA (p < 0.01) complexes, and H3Cit (p < 0.05)) were markedly elevated in the thrombosis group as compared with the matched controls |
ADAMTS13 = a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13; ANC = Absolute neutrophil count; CEACAM1 = Carcinoembryonic antigen-related cell adhesion molecule 1; cfDNA = cell-free DNA; COVID-19 = Coronavirus Disease 2019; CRP: C-reactive protein; DAMPS = damage associated molecular patterns; DNA = deoxyribonucleic acid; FiO2 = Fraction of inspired oxygen; Gas6 = Gasdermin 6; H3Cit = Citrullinated histone H3; ICU = Intensive Care Unit; ICUAW = intensive care unit acquired weakness; LDG = low-density granulocytes; LDH = lactate dehydrogenase; LGAS9 = Galectin 9; MPO = Myeloperoxidase; NDG = normal-density granulocytes; NE = Neutrophil elastase; NETs = Neutrophil Extracellular Traps; NK cells = Natural Killer Cells; PaO2 = partial pressure of oxygen, SAPS-III = Simplified Acute Physiology Score III; sAXL = soluble AXL receptor tyrosine kinase; SOFA = Sequential Organ Failure Assessment.; TAT = thrombin-antithrombin
Study quality
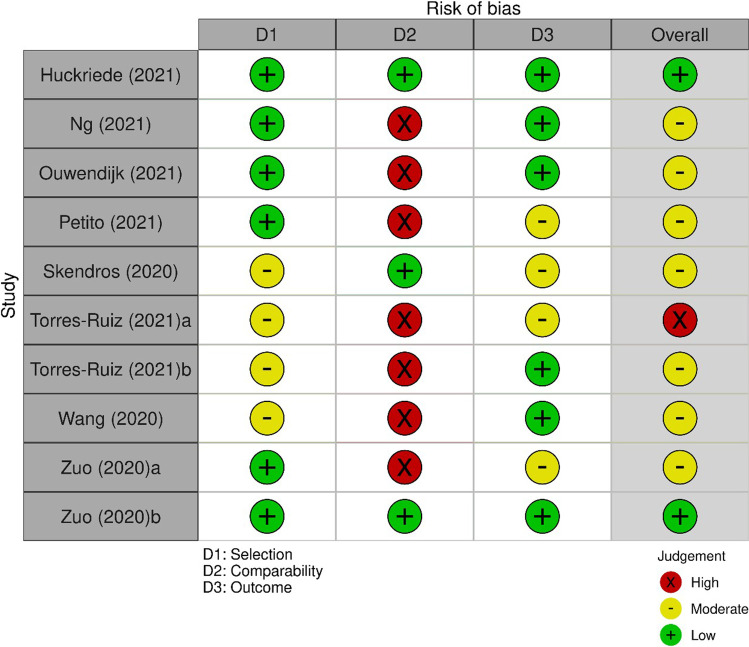
According to the risk-of-bias analysis, most studies were of moderate quality (Table 2). It is concluded from the NOS scoring, with seven studies obtaining 4–6 scores, consistent with the previously mentioned threshold for moderate risk of bias [26]. Some trends were identified in the 10 studies regarding the three separate categories within the scoring instrument, all explained in the following sections. The risk-of-bias examination [38] was summarized in Fig. 2.
Table 2.
NOS risk of bias assessment
| Study | Selection | Comparability | Outcome | Total stars |
|---|---|---|---|---|
| Cohort | ||||
| Huckriede et al. (2021) [29] | 4 | 2 | 2 | 8 |
| Ng (2021) [30] | 3 | 0 | 3 | 6 |
| Ouwendijk, et. al. (2021) [31] | 3 | 0 | 3 | 6 |
| Petito et. al. (2021) [32] | 3 | 2 | 1 | 6 |
| Skendros et. al. (2020) [33] | 2 | 2 | 1 | 5 |
| Torres-Ruiz (2021)b [35] | 2 | 0 | 2 | 4 |
| Zuo et al. (2020)a [13] | 3 | 0 | 1 | 4 |
| Zuo et al. (2020)b [37] | 4 | 2 | 2 | 8 |
| Cross-sectional | ||||
| Torres-Ruiz et.al. (2021)a [34] | 2 | 0 | 1 | 3 |
| Wang et al. (2020) [36] | 2 | 0 | 3 | 5 |
Fig. 2.
Risk of bias assessment using Newcastle,Ottawa score. Plots created using risk-of-bias visualization (robvis) tool [28]
Selection
Two research [29, 37] got four stars in the selection segment, indicating that the cases and controls were well defined and carefully chosen using appropriate inclusion and exclusion criteria, with no noticeable possible bias, a crucial parameter in cohort studies. There are four studies [13, 30–32] which did not obtain a maximum score since they did not indicate the states regarding the outcome of interest at the commencement of the study.
Comparability
Four studies [29, 32, 33, 37] obtained the highest possible number of stars in the comparability category after controlling for crucial confounding factors such as age and gender. However, the majority of studies (six) received zero stars in this section, implying that attempts to limit at least some of the obvious confounding factors in terms of enhancing methodologic quality were not implemented properly.
Exposure
Scores for the exposure aspect ranged from one to three stars. The studies that received three stars [30, 31, 36] used rigorous methods of patient recruitment and adequate patient follow-up. In juxtaposition, the inferior reports in the exposure section that received 1 or 2 stars used different procedures for retaining patients and controls or did not provide satisfactory justification for the follow-up approach.
Prognostic role of NETs in COVID-19 patients
As pointed out previously, thromboembolism and the need for mechanical ventilation are two clinical situations that are used to present the role of NETs in determining the prognosis of COVID-19 patients. Thromboembolism was found to be strongly linked to the NET marker [29, 33, 37]. Ouwendijk et al. [31], on the other hand, found no correlation among NET abundance and thromboembolic events.
Mechanical ventilation requirements (or the use of further non-invasive ventilation support) were linked to NET marker accumulation (especially cfDNA and MPO-DNA complexes) [13, 30, 32]. Huckriede et al. [29] discovered that the dynamics of NET level can be associated with ventilator-free days, with a greater significant reduction in NET levels over time being associated with shorter ventilator use.
Sequential Organ Failure Assessment (SOFA) score was used as one indicator for severe COVID-19 scenarios. Plasma NET concentrations in those patients were correlated with overall disease severity as measured by the SOFA score [31, 32]. This association becomes more recognizable in male patients [32]. Huckriede et al. [29], on the other hand, did not reach the same results, owing to the failure to obtain a significant correlation with Simplified Acute Physiology Score III (SAPS-III). However, they found that another parameter, intensive care unit acquired weakness (ICUAW), was found to be significantly correlated with NETs [29].
Other laboratories and clinical parameters associated with a poor prognosis in COVID-19 patients have also been studied with NETs. Those include the lactate dehydrogenase (LDH), troponin I, leukocyte count, neutrophil count, neutrophil–lymphocyte ratio (NLR), C-reactive protein (CRP), Tumor Necrosis Factor-α (TNF-α), interleukin-6 (IL-6), D-Dimer, a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS13),), thrombin-antithrombin (TAT) complex, plasmin-antiplasmin complex (PAPand von Willebrand factor (vWF) [29, 30]. Dialysis and delirium clinical events were also demonstrated as prognostication parameters [29].
Ultimately, the abundance of NETs has been linked to overall disease severity [31, 32, 34, 35]. The abundance of NET density can be attributed to a lack of NET degradation and an increase in NET-recognizing antibodies, as well as increased production [35]. It is also in charge of ICU admission and extended ICU stays for critically ill patients [29–32]. Negative regulation of T cells and NK cells as a result of NET disruption was tied to insufficient antiviral immunity and lung injury [36]. According to Huckriede et al. [29] and Ng et al. [30], either increased NET concentrations during patient admission or insignificant diminishment (as measured by dynamic of NET concentrations during early and late disease stages) of NETs could predict mortality in COVID-19 patients.
Discussion
Many viruses can stimulate the production of NETs. Virus-induced NETs (constituted of double-stranded DNA complexes, histones, and granular proteins) can circulate uncontrollably, triggering a radical systemic response in the body, resulting in the production of immune complexes, cytokines, and chemokines and ultimately favoring inflammation [39]. In the new era of COVID-19, disease progression has shown a correlation with NETs level. The correlation mainly have a negative impact, with higher NETs level was associated with the decrease in overall COVID-19 patients survivability [30, 40, 41].
In COVID-19 patients, neutrophilia is prevalent in severe cases, and is showing a progressive rise, especially in non-survivors [42]. In COVID-19 patients, transcriptome analysis showed a link between neutrophil authentication and 16 NET-associated genes, which include metabolic enzymes, structural proteins, anti-microbial related peptides, peroxisomal peptide, and others. All of this interacted with T/natural killer (NK)/B cells and was related to innate immunity (via IFN signaling). However, the effects were associated with negative regulation of T cell and NK cell immune function via LGAS9 and CEACAM1, respectively, resulting in reduced antiviral immunity and direct lung injury (due to toxicity in the alveolar-capillary barrier) [36]. It is also related to some other NETs components, including NE, MPO, and histone proteins which are causing the direct cytotoxic effect on the alveolar epithelium and disrupting the endothelium. The damage-associated molecular patterns (DAMPs)-like the role of these components also able to induce thrombotic and inflammatory responses in various acute infections, including COVID-19 [43]. Overall process of SARS-CoV-2–induced NETs formation requires angiotensin-converting enzyme/ACE2 (through the ACE2–TMPRSS2 axis) receptor and serine protease [44].
The surrogate marker of NETs, histone-DNA (his-DNA) and NETs-specific MPO-DNA complexes were significantly raised in plasma samples from critically ill COVID-19 patient populations. In paired sputum samples from COVID-19 patients, MPO-DNA levels were significantly correlated with viral RNA load. Inflammatory markers (such as C-reactive protein (CRP) and interleukin (IL-6)) were also linked to the findings, in addition to white blood cells and absolute neutrophil count [13, 30, 31]. Increased NETs level was also observed in tracheal aspirate of the COVID-19 patients who undergone mechanical ventilation [44].
On the chest computed tomography (CT) examination, numerous consolidations with air bronchograms across all pulmonary fields, with peripheral and peribronchovascular spread (particularly prominent in the lower lobes), accompanied with ground-glass opacification, compliant with diffuse alveolar injury were identified [44]. Meanwhile, the autopsy of critical COVID-19 patient’s lung (distributed in the airway, interstitial, and vascular environments) also showed an increase in MPO+H3Cit+ neutrophils and filamentous NETs [31, 45]. According to histopathological evaluation, COVID-19 causes significant alveolar and small airway epithelial alterations with viral cytopathic effects, as well as squamous metaplasia, lymphocytic infiltration, endothelial edema, and tiny fibrinous thrombus formation in pulmonary arterioles [44]. It is concluded that during SARS-CoV-2–induced ARDS, neutrophils invade and perform NETosis in the lungs [31]. It is believed that the markers of NETs formation (citrullinated histone H3, cfDNA, and NE) could be used to assess the requirement for respiratory support and short-term mortality in COVID-19 patients [30].
The level of identified NETs-related biomarkers was shown to be correlated with increasing severity of illness, as evidenced by a higher value in ICU patients compared to non-ICU admissions and outpatients [46]. According to a cohort study, the level of NETs-related biomarkers peaks at the time of patient admission to the ICU and then gradually decline over time as the disease progresses (either discharge from ICU or death). NETs have been found to have a negative correlation with the length of stay in the hospital [31]. According to the statistics, ICU admission began on average 9.5 days (range: 7–12.5 days) after the onset of the COVID-19, indicating the development of pulmonary and hyperinflammation phase, and in average, ICU admission mortality rate was around 35%, which can escalate to 65% in subjects receiving extracorporeal membrane oxygenation [47, 48]. In addition, for patients receiving mechanical ventilation related to COVID-19, the maximum possibility for mortality can reach 84.5–96.7%, which indicated the respiratory failure (may be accompanied with sepsis and multi-organ failure) [49, 50].
The increase in NETs indicators corresponds to changes in the phenotype of activated neutrophils which is prone to cellular aggregation in the acute phase of COVID-19 [51]. NETs were also linked to at least seven acute-phase protein genes, as per interaction analyses (IL-6, TNF-α, CRP, CXCL8, IL -1ß, IL17A, and IL-1) which have been linked to short-term complications of ARDS [52]. NETs are also linked to the complement system (C3 and C5); thus its inhibition can reduce NETs levels. In severe COVID-19 instances, complement is one of the key mechanisms promoting hyperinflammation, immunothrombosis, and microvascular endothelial damage [53].
Thrombosis (usually referred to as immunothrombosis) is a deleterious event which is significantly worsening COVID-19 patient prognosis (in the case of mortality and morbidity) [54]. Some of the observed thrombotic-related conditions in the COVID-19 patients are pulmonary embolism, ischemic stroke, cerebral venous thrombosis, lower limb ischemia, aortic thrombosis, and deep vein thrombosis [37, 55–57]. In the patients who are developing thrombotic events, an increase in calprotectin, a marker of neutrophil activation, as well as NETs markers (cfDNA, MPO-DNA complexes, and H3cit) was observed. An increase in clinical biomarkers of the thrombotic phenomenon, D-dimer, CRP, ferritin, and von Willebrand Factor (vWF) antigen was also higher in the thrombosis group [15, 37]. Enhanced coagulation and NETs turnover in COVID-19 patients were found, in accordance to the previously described findings [51]. Vascular occlusion by neutrophil extracellular traps in COVID-19 is promoted by neutrophil aggregation which increases NETs density and occluding tubular structures [58] and evidenced by cell-rich intra-vascular clots contained vast amounts of aggregated neutrophils that express NE and citrullinated histone H3, in addition to CD31. However, smaller vessels often showed preserved endothelium [51]. The findings were not only limited to lungs, it can be observed in the kidney and liver tissues from COVID-19 patients, potentially leading to multi-organ failure [51]. In a study involving COVID-19 patients with ST segment elevation myocardial infarction (STEMI), the thrombus were predominantly composed of fibrin with some degree of polymorphonuclear cell infiltration and about 61% of NET density [59].
NETs have been found to affect the tissue factor (TF)/thrombin axis in COVID-19 patients as one of the hypothesized thrombotic mechanisms. It is based on the positive correlations between thrombin-antithrombin (TAT) activity and MPO-DNA complex. Thrombin inhibition with dabigatran may be beneficial to mitigate TF expression and activity, as well as NET release in COVID-19 patients. Significantly elevated concentrations of soluble platelet-derived factors that trigger NETosis, such as Platelet factor 4 (PF4) and RANTES, have been found in COVID-19 patients, suggesting that the interaction of NETs and platelets may contribute to a thrombo-inflammatory cluster that leads to COVID-19 hypercoagulability and thrombosis [15].
The lack of studies determining satisfactory matched control for the cohort is one of our study’s limitations. The included studies were also primarily retrospective. Furthermore, while the prognostic role of NETs in COVID-19 appears promising in the current context, more evidence across the disease spectrum is required. We encourage additional research into developing prognostic models that include the NETs marker. We also intended not to undertake a meta-analysis due to the clinically vast diversity of the included studies (such as different metrics, measurement methods, and heterogeneity of observed NETs markers), the discrepancy of study participants’ characteristics, and the small study size.
In terms of future prospect, more multicenter randomized controlled trials (RCT), primarily using targeted therapy (e.g., DNase) [16], are required to determine the precise role of NETs in influencing COVID-19 severity. The multicenter trial involving participants with similar demographic characteristics may also aid in addressing host response issues related to the therapeutic approach, which is primarily multifactorial. Also, as COVID-19 is progressing to be an endemic, the role of NETs formation in providing protection and vaccination response can be investigated (since the association between cases and deaths appears to have been eroded in highly vaccinated nations) [60]. Another cohort can be performed to investigate the common phenomenon known as COVID-19-associated coagulopathy [61, 62], and NETs can be utilized as the predictive factor.
Conclusion
Neutrophil extracellular traps (NETs) are a type of compound produced by the neutrophils in response to various infections or inflammatory conditions. This molecule is also crucial in affecting disease severity in COVID-19 patients and thus affecting their prognosis. It is based on several complications, such as thrombosis, endothelial dysfunction, and the need for mechanical ventilation, in addition to its interaction with inflammatory and organ dysfunction markers (both extrapulmonary and pulmonary involvement). Future research could focus on this marker as a potential approach to improving patient outcomes. The multicenter RCT can be used to assess the functionality of this molecule. It may, however, take a significant amount of time and pose ethical issues, especially for patients in severe or critical conditions, and must be carefully planned. Furthermore, the relationship between NETs and commonly used laboratory parameters can be investigated to improve the usability of this biomarker.
Supplementary Information
Below is the link to the electronic supplementary material.
Author contribution
The authors confirm contribution to the paper as follows: study conception and design, TPU and PL; data collection, TPU and IAL; analysis and interpretation of results, TPU, PL, and ZH; draft manuscript preparation, TPU and PL. All authors reviewed the results and approved the final version of the manuscript. All authors were involved in the revision of the draft manuscript and have agreed to the final content.
Declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Phey Liana and Tungki Pratama Umar contributed equally to this work.
Contributor Information
Phey Liana, Email: pheyliana@fk.unsri.ac.id.
Iche Andriyani Liberty, Email: icheandriyaniliberty@fk.unsri.ac.id.
Krisna Murti, Email: krisna.arinafril@unsri.ac.id.
Zen Hafy, Email: zenhafy@gmail.com.
Eddy Mart Salim, Email: eddymsalim@gmail.com.
Mohammad Zulkarnain, Email: septi_2003@yahoo.com.
Tungki Pratama Umar, Email: tungkipratama@gmail.com.
References
- 1.Chapman EA, Lyon M, Simpson D, Mason D, Beynon RJ, Moots RJ, et al. Caught in a trap? proteomic analysis of neutrophil extracellular traps in rheumatoid arthritis and systemic lupus erythematosus. Front Immunol. 2019;10:423. doi: 10.3389/fimmu.2019.00423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lauková L, Konečná B. NETosis − Dr. Jekyll and Mr. Hyde in inflammation. J Appl Biomed. 2018;16:1–9. 10.1016/j.jab.2017.10.002.
- 3.Arazna M, Pruchniak MP, Zycinska K, Demkow U. Neutrophil extracellular trap in human diseases. Adv Exp Med Biol. 2013;756:1–8. doi: 10.1007/978-94-007-4549-0_1. [DOI] [PubMed] [Google Scholar]
- 4.Dąbrowska D, Jabłońska E, Garley M, Ratajczak-Wrona W, Iwaniuk A. New aspects of the biology of neutrophil extracellular traps. Scand J Immunol. 2016;84:317–322. doi: 10.1111/sji.12494. [DOI] [PubMed] [Google Scholar]
- 5.Meegan JE, Yang X, Coleman DC, Jannaway M, Yuan SY. Neutrophil-mediated vascular barrier injury: role of neutrophil extracellular traps. Microcirculation. 2017;24:e12352. doi: 10.1111/micc.12352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Branzk N, Papayannopoulos V. Molecular mechanisms regulating NETosis in infection and disease. Semin Immunopathol. 2013;35:513–530. doi: 10.1007/s00281-013-0384-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yang H, Biermann MH, Brauner JM, Liu Y, Zhao Y, Herrmann M. New insights into neutrophil extracellular traps: mechanisms of formation and role in inflammation. Front Immunol. 2016;7:302. doi: 10.3389/fimmu.2016.00302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Papayannopoulos V, Zychlinsky A. NETs: a new strategy for using old weapons. Trends Immunol. 2009;30:513–521. doi: 10.1016/j.it.2009.07.011. [DOI] [PubMed] [Google Scholar]
- 9.Lu T, Kobayashi SD, Quinn MT, Deleo FR. A NET Outcome Front Immunol. 2012;3:365. doi: 10.3389/fimmu.2012.00365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Barr FD, Ochsenbauer C, Wira CR, Rodriguez-Garcia M. Neutrophil extracellular traps prevent HIV infection in the female genital tract. Mucosal Immunol. 2018;11:1420–1428. doi: 10.1038/s41385-018-0045-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Saitoh T, Komano J, Saitoh Y, Misawa T, Takahama M, Kozaki T, et al. Neutrophil extracellular traps mediate a host defense response to human immunodeficiency virus-1. Cell Host Microbe. 2012;12:109–16. doi: 10.1016/j.chom.2012.05.015. [DOI] [PubMed] [Google Scholar]
- 12.Zhu L, Liu L, Zhang Y, Pu L, Liu J, Li X, et al. High level of neutrophil extracellular traps correlates with poor prognosis of severe influenza A infection. J Infect Dis. 2018;217:428–437. doi: 10.1093/infdis/jix475. [DOI] [PubMed] [Google Scholar]
- 13.Zuo Y, Yalavarthi S, Shi H, Gockman K, Zuo M, Madison JA, et al. Neutrophil extracellular traps in COVID-19. JCI Insight. 2020;5:e138999. doi: 10.1172/jci.insight.138999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ramos-Martínez E, Hernández-González L, Ramos-Martínez I, Mayoral LP-C, López-Cortés GI, Pérez-Campos E, et al. Multiple origins of extracellular DNA traps. Front Immunol. 2021;12:621311. 10.3389/fimmu.2021.621311 [DOI] [PMC free article] [PubMed]
- 15.Middleton EA, He X-Y, Denorme F, Campbell RA, Ng D, Salvatore SP, et al. Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood. 2020;136:1169–1179. doi: 10.1182/blood.2020007008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Liana P, Murti K, Hafy Z, Liberty IA, Umar TP. Neutrophil extracellular traps and its correlation with several pathological conditions: prosperities and deleterious implications. Mol Cell Biomed Sci. 2022;6:1–11. 10.21705/mcbs.v6i1.224
- 17.Xiong X, Chi J, Gao Q. Prevalence and risk factors of thrombotic events on patients with COVID-19: a systematic review and meta-analysis. Thromb J. 2021;19:32. doi: 10.1186/s12959-021-00284-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Thierry AR, Roch B. Neutrophil extracellular traps and by-products play a key role in COVID-19: pathogenesis, risk factors, and therapy. J Clin Med. 2020;9:2942. doi: 10.3390/jcm9092942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Janiuk K, Jabłońska E, Garley M. Significance of NETs formation in COVID-19. Cells. 2021;10:151. doi: 10.3390/cells10010151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi: 10.1136/bmj.n71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan—a web and mobile app for systematic reviews. Syst Rev. 2016;5:210. doi: 10.1186/s13643-016-0384-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603–605. doi: 10.1007/s10654-010-9491-z. [DOI] [PubMed] [Google Scholar]
- 23.Wells G, Shea B, O’Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa Hospital Research Institute. 2014. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 5 Nov 2021.
- 24.Modesti PA, Reboldi G, Cappuccio FP, Agyemang C, Remuzzi G, Rapi S, et al. Panethnic differences in blood pressure in Europe: a systematic review and meta-analysis. PLoS ONE. 2016;11:e0147601. doi: 10.1371/journal.pone.0147601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Luchini C, Stubbs B, Solmi M, Veronese N. Assessing the quality of studies in meta-analyses: advantages and limitations of the Newcastle Ottawa Scale. World J Meta-Anal. 2017;5:80–4. 10.13105/wjma.v5.i4.80
- 26.Lo CK-L, Mertz D, Loeb M. Newcastle-Ottawa Scale: comparing reviewers’ to authors’ assessments. BMC Med Res Methodol. 2014;14:1–5. 10.1186/1471-2288-14-45 [DOI] [PMC free article] [PubMed]
- 27.Yeh T-L, Lei W-T, Liu S-J, Chien K-L. A modest protective association between pet ownership and cardiovascular diseases: A systematic review and meta-analysis. PLoS ONE. 2019;14:e0216231. doi: 10.1371/journal.pone.0216231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McGuinness LA, Higgins JPT. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res Synth Methods. 2021;12:55–61. doi: 10.1002/jrsm.1411. [DOI] [PubMed] [Google Scholar]
- 29.Huckriede J, Anderberg SB, Morales A, de Vries F, Hultström M, Bergqvist A, et al. Evolution of NETosis markers and DAMPs have prognostic value in critically ill COVID-19 patients. Sci Rep. 2021;11:15701. doi: 10.1038/s41598-021-95209-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ng H, Havervall S, Rosell A, Aguilera K, Parv K, Von Meijenfeldt FA, et al. Circulating markers of neutrophil extracellular traps are of prognostic value in patients with COVID-19. Arterioscler Thromb Vasc Biol. 2021;41:988–994. doi: 10.1161/atvbaha.120.315267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ouwendijk WJD, Raadsen MP, van Kampen JJA, Verdijk RM, von der Thusen JH, Guo L, et al. High levels of neutrophil extracellular traps persist in the lower respiratory tract of critically ill patients with coronavirus disease 2019. J Infect Dis. 2021;223:1512–1521. doi: 10.1093/infdis/jiab050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Petito E, Falcinelli E, Paliani U, Cesari E, Vaudo G, Sebastiano M, et al. Association of neutrophil activation, more than platelet activation, with thrombotic complications in coronavirus disease 2019. J Infect Dis. 2021;223:933–944. doi: 10.1093/infdis/jiaa756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Skendros P, Mitsios A, Chrysanthopoulou A, Mastellos DC, Metallidis S, Rafailidis P, et al. Complement and tissue factor-enriched neutrophil extracellular traps are key drivers in COVID-19 immunothrombosis. J Clin Invest. 2020;130:6151–6157. doi: 10.1172/JCI141374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Torres-Ruiz J, Pérez-Fragoso A, Maravillas-Montero JL, Llorente L, Mejía-Domínguez NR, Páez-Franco JC, et al. Redefining COVID-19 severity and prognosis: the role of clinical and immunobiotypes. Front Immunol. 2021;12:689966. doi: 10.3389/fimmu.2021.689966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Torres-Ruiz J, Absalón-Aguilar A, Nuñez-Aguirre M, Pérez-Fragoso A, Carrillo-Vázquez DA, Maravillas-Montero JL, et al. Neutrophil extracellular traps contribute to COVID-19 hyperinflammation and humoral autoimmunity. Cells. 2021;10:2545. doi: 10.3390/cells10102545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wang J, Li Q, Yin Y, Zhang Y, Cao Y, Lin X, et al. Excessive neutrophils and neutrophil extracellular traps in COVID-19. Front Immunol. 2020;11:2063. doi: 10.3389/fimmu.2020.02063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zuo Y, Zuo M, Yalavarthi S, Gockman K, Madison JA, Shi H, et al. Neutrophil extracellular traps and thrombosis in COVID-19. J Thromb Thrombolysis. 2021;51:446–453. doi: 10.1007/s11239-020-02324-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sanyaolu L, Scholz AFM, Mayo I, Coode-Bate J, Oldroyd C, Carter B, et al. Risk factors for incident delirium among urological patients: a systematic review and meta-analysis with GRADE summary of findings. BMC Urol. 2020;20:1–13. doi: 10.1186/s12894-020-00743-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mozzini C, Girelli D. The role of Neutrophil Extracellular Traps in Covid-19: Only an hypothesis or a potential new field of research? Thromb Res. 2020;191:26–27. doi: 10.1016/j.thromres.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Schönrich G, Raftery MJ, Samstag Y. Devilishly radical NETwork in COVID-19: oxidative stress, neutrophil extracellular traps (NETs), and T cell suppression. Adv Biol Regul. 2020;77:100741. doi: 10.1016/j.jbior.2020.100741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fernández‐Pérez MP, Águila S, Reguilón‐Gallego L, de los Reyes‐García AM, Miñano A, Bravo‐Pérez C, et al. Neutrophil extracellular traps and von Willebrand factor are allies that negatively influence COVID‐19 outcomes. Clin Transl Med. 2021;11:e268. 10.1002/ctm2.268 [DOI] [PMC free article] [PubMed]
- 42.Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Teluguakula N. Neutrophils set extracellular traps to injure lungs in coronavirus disease 2019. J Infect Dis. 2021;223:1503–1505. doi: 10.1093/infdis/jiab053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Veras FP, Pontelli MC, Silva CM, Toller-Kawahisa JE, de Lima M, Nascimento DC, et al. SARS-CoV-2-triggered neutrophil extracellular traps mediate COVID-19 pathology. J Exp Med. 2020;217:e20201129. doi: 10.1084/jem.20201129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Radermecker C, Detrembleur N, Guiot J, Cavalier E, Henket M, D’Emal C, et al. Neutrophil extracellular traps infiltrate the lung airway, interstitial, and vascular compartments in severe COVID-19. J Exp Med. 2020;217:e20201012. doi: 10.1084/jem.20201012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cavalier E, Guiot J, Lechner K, Dutsch A, Eccleston M, Herzog M, et al. Circulating nucleosomes as potential markers to monitor COVID-19 disease progression. Front Mol Biosci. 2021;8:600881. doi: 10.3389/fmolb.2021.600881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8:475–481. doi: 10.1016/S2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Roedl K, Jarczak D, Thasler L, Bachmann M, Schulte F, Bein B, et al. Mechanical ventilation and mortality among 223 critically ill patients with coronavirus disease 2019: A multicentric study in Germany. Aust Crit Care. 2021;34:167–175. doi: 10.1016/j.aucc.2020.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, Davidson KW, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323:2052–2059. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nicholson CJ, Wooster L, Sigurslid HH, Li RH, Jiang W, Tian W, et al. Estimating risk of mechanical ventilation and in-hospital mortality among adult COVID-19 patients admitted to Mass General Brigham: The VICE and DICE scores. EclinicalMedicine. 2021;33:100765. doi: 10.1016/j.eclinm.2021.100765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Leppkes M, Knopf J, Naschberger E, Lindemann A, Singh J, Herrmann I, et al. Vascular occlusion by neutrophil extracellular traps in COVID-19. EBioMedicine. 2020;58:102925. doi: 10.1016/j.ebiom.2020.102925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bouzid A, Uthman AT, Al-Rawi NN, Al-Rawi NH. Neutrophil extracellular traps in coronavirus infection: interaction network analysis. Syst Rev Pharm. 2020;11:1091–1101. [Google Scholar]
- 53.Mastellos DC, da Silva BGPP, Fonseca BAL, Fonseca NP, Auxiliadora-Martins M, Mastaglio S, et al. Complement C3 vs C5 inhibition in severe COVID-19: early clinical findings reveal differential biological efficacy. Clin Immunol. 2020;220:108598. doi: 10.1016/j.clim.2020.108598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hanff TC, Mohareb AM, Giri J, Cohen JB, Chirinos JA. Thrombosis in COVID-19. Am J Hematol. 2020;95:1578–1589. doi: 10.1002/ajh.25982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Price LC, McCabe C, Garfield B, Wort SJ. Thrombosis and COVID-19 pneumonia: the clot thickens! Eur Respir J. 2020;56:2001608. doi: 10.1183/13993003.01608-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kashi M, Jacquin A, Dakhil B, Zaimi R, Mahé E, Tella E, et al. Severe arterial thrombosis associated with Covid-19 infection. Thromb Res. 2020;192:75–77. doi: 10.1016/j.thromres.2020.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Cavalcanti DD, Raz E, Shapiro M, Dehkharghani S, Yaghi S, Lillemoe K, et al. Cerebral venous thrombosis associated with COVID-19. AJNR Am J Neuroradiol. 2020;41:1370–1376. doi: 10.3174/ajnr.A6644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Jiménez-Alcázar M, Rangaswamy C, Panda R, Bitterling J, Simsek YJ, Long AT, et al. Host DNases prevent vascular occlusion by neutrophil extracellular traps. Science. 2017;358:1202–1206. doi: 10.1126/science.aam8897. [DOI] [PubMed] [Google Scholar]
- 59.Blasco A, Coronado M-J, Hernández-Terciado F, Martín P, Royuela A, Ramil E, et al. Assessment of neutrophil extracellular traps in coronary thrombus of a case series of patients With COVID-19 and myocardial infarction. JAMA Cardiol. 2020;6:1–6. doi: 10.1001/jamacardio.2020.7308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Diseases TLI. Transitioning to endemicity with COVID-19 research. Lancet Infect Dis. 2022;22:297. doi: 10.1016/S1473-3099(22)00070-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gómez-Mesa JE, Galindo-Coral S, Montes MC, Muñoz Martin AJ. Thrombosis and coagulopathy in COVID-19. Curr Probl Cardiol. 2021;46:100742. doi: 10.1016/j.cpcardiol.2020.100742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Becker RC. COVID-19 update: Covid-19-associated coagulopathy. J Thromb Thrombolysis. 2020;50:54–67. doi: 10.1007/s11239-020-02134-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.