Abstract
This study reviewed the literature about the diagnosis, antepartum surveillance, and time of delivery of fetuses suspected to be small for gestational age or growth restricted. Several guidelines have been issued by major professional organizations, including the International Society of Ultrasound in Obstetrics and Gynecology and the Society for Maternal-Fetal Medicine. The differences in recommendations, in particular about Doppler velocimetry of the ductus venosus and middle cerebral artery, have created confusion among clinicians, and this review has intended to clarify and highlight the available evidence that is pertinent to clinical management. A fetus who is small for gestational age is frequently defined as one with an estimated fetal weight of <10th percentile. This condition has been considered syndromic and has been frequently attributed to fetal growth restriction, a constitutionally small fetus, congenital infections, chromosomal abnormalities, or genetic conditions. Small for gestational age is not synonymous with fetal growth restriction, which is defined by deceleration of fetal growth determined by a change in fetal growth velocity. An abnormal umbilical artery Doppler pulsatility index reflects an increased impedance to flow in the umbilical circulation and is considered to be an indicator of placental disease. The combined finding of an estimated fetal weight of <10th percentile and abnormal umbilical artery Doppler velocimetry has been widely accepted as indicative of fetal growth restriction. Clinical studies have shown that the gestational age at diagnosis can be used to subclassify suspected fetal growth restriction into early and late, depending on whether the condition is diagnosed before or after 32 weeks of gestation. The early type is associated with umbilical artery Doppler abnormalities, whereas the late type is often associated with a low pulsatility index in the middle cerebral artery. A large randomized clinical trial indicated that in the context of early suspected fetal growth restriction, the combination of computerized cardiotocography and fetal ductus venosus Doppler improves outcomes, such that 95% of surviving infants have a normal neurodevelopmental outcome at 2 years of age. A low middle cerebral artery pulsatility index is associated with an adverse perinatal outcome in late fetal growth restriction; however, there is no evidence supporting its use to determine the time of delivery. Nonetheless, an abnormality in middle cerebral artery Doppler could be valuable to increase the surveillance of the fetus at risk. We propose that fetal size, growth rate, uteroplacental Doppler indices, cardiotocography, and maternal conditions (ie, hypertension) according to gestational age are important factors in optimizing the outcome of suspected fetal growth restriction.
Keywords: abdominal circumference, cardiotocography, cesarean delivery, Disproportionate Intrauterine Growth Intervention Trial at Term, Doppler velocimetry, ductus venosus, fetal biometry, fetal death, fetal distress, fetal growth, longitudinal, middle cerebral artery, neurodevelopmental outcome, Prospective Observational Trial to Optimize Pediatric Health, randomized controlled trial, short-term variation, small for gestational age, systematic review, umbilical artery Doppler, umbilical artery pH, uterine artery, Trial of Umbilical and Fetal Flow in Europe
Introduction
The diagnosis, surveillance, and time of delivery of fetuses with suspected fetal growth restriction (FGR) are major issues in obstetrical practice.1,2 The Society for Maternal-Fetal Medicine (SMFM)3 and the International Society for Ultrasound in Obstetrics and Gynecology (ISUOG)4 have recently issued clinical guidelines concerning FGR management. Furthermore, several professional organizations have made recommendations, and the differences and similarities of the recommendations have been previously reviewed in the American Journal of Obstetrics & Gynecology.1 This clinical opinion reviewed the key concepts on the diagnosis, surveillance, and time of delivery of patients with suspected FGR.
Diagnosis of Suspected Fetal Growth Restriction
Fetal growth restriction is frequently associated with a fetus failing to reach its genetic and biologic growth potential and is a consequence of several causes, with placental dysfunction being one of the most common causes. Before the publication of the Delphi consensus criteria in 2016 (Table 1),5 the most common definition of FGR was based on the evaluation of fetal size (estimated fetal weight [EFW] or abdominal circumference [AC]) and its statistical deviation from specific reference ranges or standards. Fetal size <10th percentile has most frequently been used to define a small for gestational age (SGA) fetus1 and is most commonly defined using the classic Hadlock formula.6 This definition derives from that proposed in 1967 by Battaglia and Lubchenco,7 which classified newborns as SGA, appropriate for gestational age (AGA), and large for gestational age (LGA) based on the deviation of birthweight from the mean and falling <10th or >90th percentiles, SGA and LGA, respectively. Their definition identified a newborn below the absolute birthweight cutoff of 10th percentile as SGA but did not distinguish between the pathologically small and normally (or constitution-ally) small newborn. A birthweight that identifies SGA is undoubtedly associated with an increased risk of adverse outcome.8,9 However, basing the definition of abnormal growth on a weight threshold alone has limitations.
TABLE 1.
Delphi consensus criteria for the definition of early and late fetal growth restrictions5
| Early FGR: GA<32 wk, in the absence of congenital anomalies | Late FGR: GA>32 wk, in the absence of congenital anomalies |
|---|---|
|
| |
| AC or EFW of <third percentile or UA-AEDF Or 1. AC or EFW of <10th percentile combined with 2. Uta-PI of >95th percentile and/or 3. UA-PI of >95th percentile |
AC or EFW of <third percentile Or at least 2 of 3 of the following: 1. AC or EFW of <10th percentile 2. AC or EFW crossing percentiles of >2 quartiles on growth percentiles 3. CPR of <5th percentile or UA-PI of >95th percentile |
AC, abdominal circumference; AEDF, absent end-diastolic flow; CPR, cerebroplacental ratio; EFW, estimated fetal weight; FGR, fetal growth restriction; GA, gestational age; PI, pulsatility index; UA, umbilical artery; UtA, uterine artery.
Fetal Growth Restriction: Which Thresholds are Important?
The definition of SGA makes no distinction between a constitutionally small but healthy fetus and a fetus with growth restriction. In a given population of fetuses, 10% will have a birthweight <10th percentile, and this group is considered to be SGA. From a biologic point of view, it is implausible to assume that all fetuses whose size is <10th percentile suffer from growth restriction and that none with a weight >10th percentile do. In a population of just under 1.2 million pregnancies in the Netherlands from 2002 to 2008 with >5000 perinatal deaths, perinatal mortality progressively increased as birthweight reduced from the 80th birthweight percentile.10 Although mortality was the highest below the 2.3rd birthweight percentile (Figure 1), the inflection point of the curve increased progressively starting from the 20th to 50th percentile, suggesting increased perinatal risk exists even above the 10th percentile of fetal size. This finding was consistent with an earlier study of 82,361 infants born at term in 1 US institution during an 8-year period where infants with a birthweight of less than the third percentile were at the highest risk of morbidity and mortality.11 In the multicenter Irish Prospective Observational Trial to Optimize Pediatric Health (PORTO) study of fetal SGA, an adverse outcome was most closely related to an EFW of <3rd percentile; however, this was not true for other biometric cutoffs (EFW of <5th percentile and ACs of <3rd, 5th, and 10th percentile).12 Similarly, the risk of fetal death for each week of gestation is the highest at birthweights of <3rd percentile, which are 3 times greater than birthweights between the 3rd the 5th percentiles and 4 to 7 times greater than birthweights between the 5th and 10th percentiles.10,13 In the same study by Pilliod et al,13 the “number needed to deliver” to prevent 1 fetal death at 34 weeks of gestation was 3.7 and 6.1 times higher at the 3rd to 5th percentile and 5th to 10th percentile, respectively, than the 3rd percentile; by 39 weeks of gestation, the difference was 6.0 and 13.7 times higher. The risk of fetal death in SGA fetuses is incrementally cumulative for each week beyond 37 weeks: 21 per 10,000 ongoing pregnancies with SGA fetuses (at 37 weeks), 26 per 10,000 ongoing pregnancies with SGA fetuses (at 39 weeks), and 60 per 10,000 ongoing pregnancies with SGA fetuses (>40 weeks).14
FIGURE 1. Perinatal mortality according to birthweight percentile.
Adapted from Vasak et al.10
The Dutch Multicenter Disproportionate Intrauterine Growth Intervention Trial at Term (DIGITAT) study was a randomized controlled trial (RCT) of induced labor vs expectant management of fetuses with an EFW or AC of <10th percentile or a “flattening of the third-trimester growth curve” as judged by the clinician. A birthweight of <2.3rd percentile was the strongest predictor of an adverse 2-year neurodevelopmental outcome.15,16 Finally, in one of the largest prospective longitudinal studies on late suspected FGR, infants with a birthweight Z-score of <−2 (in other words, the 2.3rd percentile) had a significantly higher risk of composite adverse outcome (relative risk [RR], 2.9; 95% confidence interval [CI], 1.8–4.0).17 Taken together, these data showed that the 10th percentile, although a convenient cutoff from a population statistical standpoint, is not Adapted from Vasak et al.10 suited as a “stand-alone” to define suspected FGR as it potentially exposes a significant proportion of healthy fetuses to unnecessary intervention in late preterm period. If one were to use a single biometric “cutoff ” to define suspected FGR, the 3rd percentile would be more appropriate.18
Fetal Size VS Growth Velocity Rate
The second limitation of any definition based purely on a point estimate of fetal size is that it does not take into account that fetal growth velocity may slow while the absolute fetal size is still >10th percentile (Figure 2), particularly in late suspected FGR. Fetal growth and fetal size are not the same: as opposed to absolute fetal size, fetal growth occurs over time, is equivalent to velocity, and therefore requires longitudinal evaluation.21 In postnatal life, a flattening of the growth velocity curve might alert the physician to possible pathologic growth failure of the neonate or infant.22,23 It is entirely logical that this concept would apply during fetal life when the growth rate is particularly rapid.23 In a prospective single-center cohort study on universal third-trimester sonography, an EFW of <10th percentile was significantly associated with any neonatal morbidity, which included poor immediate birth outcome, severe morbidity, and death (RR, 1.6; 95% CI, 1.2–2.1).19 However, when AC growth velocity was considered, the risk of adverse outcomes only increased for fetuses that in addition to an EFW of <10th percentile were at the lowest decile of the AC growth velocity (RR, 2.5; 95% CI, 1.7–3.6); (Figure 2). Together with abnormally raised umbilical artery (UA) Doppler pulsatility index (PI), AC growth velocity had the strongest relationship in a stratified risk analysis of neonatal composite adverse outcome in SGA.
FIGURE 2. Patterns of fetal growth in SGA and FGR.
The figure illustrates 3 growth velocity pattern scenarios: (1) small for gestational age (blue circles), (2) suspected FGR with an EFW of <3rd percentile (red triangles), and (3) deceleration of growth velocity (orange diamonds). If clinical judgment is based only on time point evaluation of the fetal size, then the 2 last examinations (arrows: the blue circles and red triangles) would be both classified as FGR and the orange diamonds would be classified as appropriate for gestational age. However, the blue circles indicate a fetus with a maintained growth velocity, which indicates a constitutionally small fetus, whereas the orange diamonds indicate a fetus with a deceleration of growth velocity, although above the 10th percentile. The study by Sovio et al19 showed that in fetuses with a birthweight of <10th percentile (blue circles and red triangles), only fetuses with the AC growth velocity in lowest deciles (red triangles) have a significantly higher risk of adverse outcome than the fetuses in the control group; moreover, there was no difference between fetuses with a birthweight of <10th percentile and maintained growth velocity (blue circles) and fetuses in the control group in terms of adverse outcomes. To identify suspected FGR, Doppler evaluation of uteroplacental-fetal circulation is important (Figure 3).
AC, abdominal circumference; EFW, estimated fetal weight; FGR, fetal growth restriction; GA, gestational age.
Considering suspected FGR only in terms of fetal AC or EFW size of <10th percentile assumes that fetal size predicts fetal well-being in terms of oxygenation and acid-base status.24 Approximately 60% to 70% of fetal deaths, especially toward term, occur in AGA fetuses,10,25,26 and a recent case-control study showed that 88% of stillbirths had histologic features of hypoxia on postmortem or placental examination.27 Individualized growth assessment is a method for the evaluation of fetal growth and neonatal growth outcome in which each fetus is its own control, based on estimates of individual growth potentials.21 This type of longitudinal evaluation addresses the concept of possible fetal growth failure even if the EFW is >10th percentile28 (Figure 2), hence identifying potential “at-risk” fetuses that might otherwise be missed. Even within a population of fetuses whose EFW was <10th percentile recruited within the PORTO study, a recent reanalysis has shown that 38% showed a normal fetal growth pattern.29 Using individualized growth assessment, different growth trajectories were associated with fetal Doppler and cardiac function and delivery outcome.29
Although a reduction in fetal growth velocity may not be sufficient as a stand-alone criterion for suspected FGR,30 it should alert the clinician to consider additional monitoring parameters, such as Doppler velocimetry as an important adjunct to identify fetal compromise or uteroplacental impairment (Figure 3).
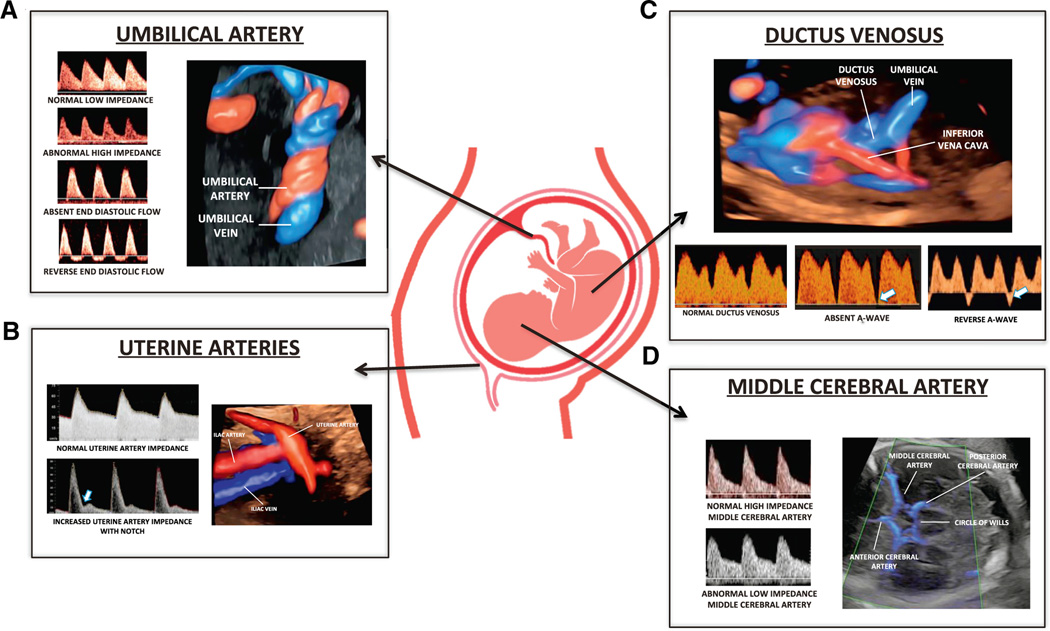
FIGURE 3. Uteroplacental-fetal vascular components evaluated with Doppler velocimetry.
The figure represents the main uteroplacental-fetal vascular components that can be evaluated with Doppler velocimetry and that play a role in the diagnosis, surveillance, and/or time of delivery of fetuses with suspected FGR. A, The umbilical arteries reflect the impedance to blood flow on the fetal side of the placenta. With increasing GA, umbilical impedance becomes progressively reduced. With progressive damage of the chorionic villi, the umbilical artery impedance will increase until absent end-diastolic flow and reversed end-diastolic flow supervene. The umbilical artery PI does not correlate with fetal hypoxemia. B, The uterine artery blood flow reflects the resistance to the blood flow at the maternal side of the placenta. Similarly, to the umbilical artery uterine impedance decreases with GA in physiological states, whereas the resistance to the blood flow remains elevated in abnormal placental invasion and is predictive of FGR especially when associated with preeclampsia. C, The ductus venosus represents a fetal shunt that carries oxygenated blood at high velocity from the umbilical vein, through the foramen ovale into the left atrium and then left ventricle of the heart; the crista dividens separates this oxygenated blood from the deoxygenated blood of the inferior vena cava that passes through the right ventricle and systemic circulation. Physiologically, it appears tri-phasic with low impedance during the a-wave. An absent or reversed ductus venosus a-wave is an expression of an increased end-diastolic intracardiac pressure because of increased resistance in afterload and/or an expression of progressive dilatation of the ductus venosus to increase the delivery of the oxygenated blood directly to the myocardium and fetal brain. Changes in the ductus venosus waveform are associated with increased risk of perinatal mortality and morbidity. D, The middle cerebral artery normally shows high impedance to blood flow. Cerebral blood flow redistribution is a fetal adaptive response to hypoxemia and/or hypercapnia and can be identified as low impedance in the middle cerebral artery. For further discussion on Doppler velocimetry and indices, please refer to the recently published International Society of Ultrasound in Obstetrics and Gynecology guideline by Bhide et al.20
FGR, fetal growth restriction; GA, gestational age; PI, pulsatility index.
Early and Late Fetal Growth Restriction
There are 2 widely described phenotypes of suspected FGR, early and late, commonly distinguished by time of diagnosis before and after 32 weeks of gestation, respectively.31 The main differences in etiology and Doppler findings that can assist in the identification of early and late suspected FGRs are shown schematically in Figure 4. Increased Doppler PI in the umbilical and uterine arteries, a sign of placental insufficiency, is much more frequent in early suspected FGR and comparatively rare in late suspected FGR.32,33 Doppler signs of cerebral blood flow redistribution reflect fetal hypoxemia and aid in the identification of late suspected FGR.31,34,35 Doppler velocimetry abnormalities of these vessels in suspected fetuses with FGR are associated with an increased risk of adverse perinatal outcomes.17,19,32,35–40 Therefore, where fetal size alone is not sufficient to identify suspected FGR, Doppler velocimetry of the uterine, umbilical, and middle cerebral arteries evaluates the full spectrum of fetal vascular compensatory abnormalities attributable to uteroplacental impairment across gestation and is crucial for a more accurate diagnosis of both early and late FGRs. The Trial of Umbilical and Fetal Flow in Europe (TRUFFLE) group expert consensus defined early FGR as fetal AC of <10th percentile and UA-PI of >95th percentile.41 A similar definition was described by the Irish consortium’s PORTO study based on the results of their study of suspected SGA and FGR fetuses.12 Moreover, it is important to recognize that a lack of evidence for middle cerebral artery Doppler in delivery timing for suspected FGR does not disqualify this modality as important for its identification. Furthermore, the Delphi consensus criteria (on which definitions the ISUOG guideline is based) were more predictive of adverse neonatal outcomes than an EFW of <10th percentile, though both performed poorly.42
FIGURE 4. Different clinical and biophysical characteristics of early and late suspected FGR.
The figure illustrates the different clinical and biophysical characteristics of early and late suspected FGR. Early suspected FGR (<32 weeks of gestation) is a rare condition and is characterized by placental insufficiency and reduced placental vascular perfusion on maternal (uterine arteries) and fetal interface (umbilical arteries). The fetus is usually very small, and profound Doppler changes might be present, with absent or reverse ductus venosus a-wave, which are associated with an increased risk of perinatal mortality and morbidity. The main challenge is the management because of prematurity and/or maternal hypertension (70% of cases). Late suspected FGR (>32 weeks of gestation) is a frequent condition caused by placental dysfunction. Vascular perfusion at the maternal and fetal interface is usually normal. The fetus is not necessarily very small, and signs of cerebral blood flow redistribution (low impedance in the middle cerebral artery and/or altered ratio to the umbilical artery) might be the only Doppler sign. The main challenge is the diagnosis.
AEDF, absent end-diastolic flow; BPP, biophysical profile; cCTG, computerized cardiotocography; FGR, fetal growth restriction; REDF, reversed end-diastolic flow; STV, short-term variation; UA, umbilical artery.
Monitoring of Fetal Growth Restriction
In early-onset suspected FGR, fetal deterioration is heralded by progressively increasing UA impedance, most commonly expressed as PI, resistance index or S-to-D (systolic-to-diastolic) ratio. This change may occur during many weeks before severe fetal cardiovascular and metabolic deterioration43,44 and may be less predictable if pre-eclampsia supervenes.45 The absence of umbilical end-diastolic velocities as a reflection of worsening uteroplacental insufficiency precede reversed end-diastolic velocity in the UA and eventually ductus venosus abnormalities46–48 (Figure 3).
Strong evidence supports the use of ductus venosus Doppler in early suspected FGR for both prognostic and monitoring purposes.49–54 The ductus venosus deviates oxygenated blood from the umbilical vein toward the heart at the confluence of the inferior vena cava with the right atrium (Figure 3), and the crista dividens preferentially streams this oxygenated blood toward the left atrium and cerebral vessels. The ductus venosus waveform commonly appears triphasic with the a-wave, a sensitive reflection of the atrial contraction. An augmented a-wave is provoked by increased preload, inotropic drive because of increased afterload, hypoxia, or a combination of these factors and/or as an expression of progressive dilatation of the ductus venosus to increase the delivery of the oxygenated blood directly to the systemic circulation.55–57 In extreme cases, the a-wave reaches the baseline or is reversed; these findings presage an increased risk of fetal death (40% –70%), neonatal mortality50,58,59; they represent late-stage acidemia independent of gestational age (GA) at delivery and frequently appear with the onset of spontaneous decelerations on cardiotocography.46,49,60 When these advanced cardiovascular changes are observed, spontaneous heart rate decelerations, an abnormal biophysical profile followed by fetal death may be anticipated within 1 week in 40% to 70% of fetuses.32,35,46,60
In late-onset suspected FGR, deterioration is far more subtle and is characterized by either a mild elevation of the UA Doppler PI, a decline in cerebral PI, or sometimes both.24,36,61 This is the reason why a ratio between the middle cerebral artery and UA-PI, the so-called cerebroplacental ratio, or its inverse, the so-called umbilical-cerebral ratio, has been proposed. Abnormal Doppler findings in the middle cerebral artery, cerebroplacental or umbilical-cerebral ratio, reflect the hemodynamic phenomenon of blood flow redistribution to the fetal brain. Moreover, this phenomenon has been called “brain sparing”62 and is triggered by decreased fetal oxygenation as shown in both animals63,64 and humans.62,64–66 Cerebral vasodilatation can easily be evaluated by measurement of middle cerebral artery PI (Figure 3), which becomes progressively lower with worsening hypoxemia.
It is incontrovertible that there is a link between low middle cerebral artery Doppler impedance and adverse perinatal outcome. Cerebral blood flow redistribution is widely reported to be associated with poorer perinatal outcomes,17 including fetal death,35 higher risk of cesarean delivery,67–69 increased risk of abnormal neurodevelopment at birth70 and at 2 or 3 years of age.37,71,72 Recent systematic review and meta-analysis evidence support these findings.39,40 The assessment of cerebral blood flow redistribution is particularly useful in identifying late suspected FGR, (Figure 4) and may improve the prediction of adverse outcomes.73 Indeed, the alteration of the UA Doppler waveform is uncommon in late preterm and term suspected FGR32,35 and does not predict adverse pregnancy outcomes accurately.61,74 Hence, assessment of cerebral blood flow redistribution plays an important role to identify and monitor fetuses exposed to placental dysfunction and hypoxemia. Although it remains to be elucidated whether cerebral redistribution is causative for adverse outcomes (as opposed to being a marker for it) assessment of cerebral Doppler velocimetry remains important for diagnosis and surveillance purposes.
The goals of fetal monitoring are the prevention of fetal death and delivery of the fetus in the best possible condition. These goals can only be successfully achieved when the disease-specific features of the fetal condition are taken into consideration.75 In early-onset suspected FGR, this requires Doppler evaluation of the UA and ductus venosus Doppler and to increase surveillance frequency when there is evidence of progressing cardiovascular dysfunction in informing decisions regarding in-patient vs outpatient monitoring.49–52 In late-onset suspected FGR, Doppler examination of the middle cerebral artery and consideration of its ratio to UA is required to make similar decisions.76–80 Accordingly, not evaluating these vessels in the specific clinical context of early- and late-onset suspected FGR is likely to result in suboptimal surveillance intervals and possibly increased risk of fetal death because features that indicate impending deterioration are not captured.
Delivery Timing in Fetal Growth Restriction
The timing of delivery in any condition that places the fetus at risk is indicated when the risk of remaining in utero exceeds those of delivery. In suspected FGR, this balances changes with advancing GA requiring adjustment of the delivery thresholds with advancing GA. The TRUFFLE study of early-onset suspected FGR was conducted in 20 European centers between 2005 and 2010,41,81 and all women received surveillance using UA Doppler and computerized cardiotocography (cCTG). Participants were randomized to delivery according to 3 management arms: “early” changes (ductus venosus PI of >95th percentile), “late” changes (ductus venosus with absent or reversed “a”-wave), and cCTG short-term variation (STV) thresholds (Figure 5).41,81 An absolute indication for delivery irrespective of allocated randomization arm, the so-called safety net, was represented by spontaneous repeated persistent unprovoked decelerations in all 3 arms and short-term variation <2.6 milliseconds (ms) at 26 0/7 to 28 6/7 weeks of gestation and <3 ms at 29 0/7 to 31 6/7 weeks of gestation in the ductus venosus arms, respectively. The protocol recommended delivery no later than 32 weeks of gestation with reversed umbilical end-diastolic flow and no later than 34 weeks of gestation with absent umbilical end-diastolic flow. The TRUFFLE study concluded that timing delivery based on ductus venosus Doppler measurement in conjunction with STV obtained from cCTG led to the best long-term (2-year neurodevelopmental) outcome in survivors.41,81 This constitutes the strongest evidence in favor of the use of ductus venosus Doppler combined with cCTG for monitoring and triggering delivery in early suspected FGR.
FIGURE 5. Schematic representation of the TRUFFLE randomization and the “safety net”.
The inclusion criteria were singleton fetus between 26 0/7 and 31 6/7 weeks of gestation with an estimated fetal weight >500 g, an abdominal circumference <10th percentile, and an umbilical artery >95th percentile, with a normal ductus venosus pulsatility index and short-term variation. Chromosomal and congenital anomalies constituted an exclusion criteria.
TRUFFLE, Trial of Umbilical and Fetal Flow in Europe.
The importance of cCTG concerning the TRUFFLE study findings is that it cannot be generalized to a simple visual interpretation of the fetal heart rate trace. cCTG has been used successfully in identifying acidemia in fetuses with growth restriction not only in Europe82–84 but also in the United States with prospective validation of its accuracy (Figure 6).85,86 Although it is true that visual interpretation of cardiotocography based on late decelerations is moderately powerful in detecting or ruling out fetal hypoxia and antepartum acidemia in the setting of suspected FGR,87–94 this results in large inter- and intraobserver variations. This prompted Dawes and Redman to develop a numeric analysis of antenatal fetal heart rate changes in the 1970s.95 With this algorithm, the STV is defined as the mean difference in fetal pulse at intervals between successive epochs of 3.75 sec, after exclusion of decelerations.96 Obvious advantages are greater reproducibility and the conversion of fetal heart rate variation into objective numeric data. STV increases with GA and so does the lower limit of normality.97 In suspected FGR, STV (and the similar long-term variation) is correlated to fetal oxygen partial pressure obtained from umbilical cord blood raphy.84,98 Very low STV is associated with fetal acidemia84,85,99–101 with a correlation coefficient of 0.72 between STV and UA pH in recordings made <4 hours before cesarean delivery.85 With an STV ≤3 ms, 54% of fetuses with early suspected FGR had metabolic acidemia, compared with 10% of fetuses with an STV >3 ms.100 In 1 large study, where STV was >3 ms, there was no case of fetal death.99 When assessed longitudinally in suspected FGR, late decelerations and reduced fetal heart rate variation appeared at more or less the same time, most likely because of the onset of fetal hypoxemia (and academia).82,94
FIGURE 6. Main characteristics of computerized cardiotocography and STV.
The figure summarizes the main characteristics of computerized cardiotocography and STV. An example in which STV is useful is represented.
fHRV, fetal heart rate variability; STV, short-term variation.
This relationship was further analyzed in the TRUFFLE study. Of 217 fetuses delivered before 32 weeks of gestation because of abnormal cardiotocography or ductus venosus, abnormal fetal heart rate was the indication for delivery in 165 fetuses. Of those fetuses, STV was reduced in 41%, late fetal heart rate decelerations were present in 48%, and both alterations were present in 11%,102 emphasizing the value of both parameters in giving a high degree of confidence in decision-making. Hence, given the favorable outcome data of the TRUFFLE trial, both STV and occurrence of decelerations should be taken into account and delivery undertaken if 1 of the 2 alterations becomes abnormal. Although thresholds denoting neonatal acidemia are described, the exact values of STV that mandate delivery in the context of clinical management of suspected FGR are still unknown.100
As far as UA Doppler is concerned, a Cochrane systematic review and meta-analysis explored Doppler ultrasound vs no Doppler or Doppler not revealed in high-risk pregnancy.103 The pooled data of the use of UA Doppler velocimetry in high-risk pregnancies showed fewer perinatal deaths (RR, 0.71 [95% CI, 0.52–0.98]; 1.2% vs 1.7%; number needed to treat, 203 [95% CI, 103–4352]; evidence graded moderate).103 Similarly, there were fewer fetal and neonatal deaths, although this did not reach statistical significance: fetal death (RR, 0.65; 95% CI, 0.41–1.04) and neonatal deaths (RR, 0.81; 95% CI, 0.53–1.24; evidence graded low).103 These findings underline the importance of UA Doppler in monitoring high-risk pregnancies, including suspected FGR, but importantly, no evidence exists from an RCT to inform the timing of delivery with different degrees of abnormality of the UA Doppler waveform.
Hence, the relationship between the findings from fetal Doppler monitoring and findings from cardiotocography monitoring are not directly consistent with one another,104 underlying the usefulness of utilizing both modalities to assess fetal condition.
Comparison Between Society for Maternal-Fetal Medicine and International Society of Ultrasound in Obstetrics and Gynecology Guidelines and Considerations on Suspected Fetal Growth Restriction
The key differences between the SMFM and ISUOG recommendations concerning the diagnosis, surveillance, and time of delivery of fetuses with suspected FGR are summarized in Table 2. ISUOG guidelines adopt the Delphi consensus diagnostic criteria for FGR, thus introducing the concept of growth velocity and Doppler evaluation to define and differentiate between early and late suspected FGR as different entities. The SMFM guideline adopts a definition of FGR as fetal size (EFW or AC) of <10th percentile.
TABLE 2.
Differences between Society for Maternal-Fetal Medicine and International Society of Ultrasound in Obstetrics and Gynecology recommendations in the diagnosis, surveillance, and time of delivery decision of fetuses with suspected fetal growth restriction
| Variable Diagnosis of suspected FGR | SMFM recommendations Estimated fetal weight or abdominal circumference <10th percentile | ISUOG recommendations Delphi consensus criteria |
|---|---|---|
| Surveillance | ||
| UA | Yes | Yes |
| Ductus venosus | No | Yes |
| Middle cerebral artery | No | Yes |
| Cardiotocography | Yes | Yes |
| Short-term variation | No | Yes |
| Delivery timing | ||
| Ductus venosus | No | ≥26 0/7 to 31 6/7 wk: ductus venosus a-wave absent or reverse |
| UA reverse end-diastolic flow | 30–32 wk | >30 0/7 to 32 0/7 wk |
| UA absent end-diastolic flow | 33–34 wk | >32 0/7 to 34 0/7 wk |
| UA pulsatility index >95th percentile | 37 wk | ≥36 0/7 to 37 6/7 wk |
| Middle cerebral artery | No | 38 0/7 to 39 0/7 wk |
| Short-term variation | No | ≥26 0/7 to 28 6/7 wk: <2.6 ms ≥29 0/7 to 31 6/7 wk: <3.0 ms ≥32 0/7 wk: <3.5 ms >34 0/7 wk: <4.5 ms |
FGR, fetal growth restriction; ISUOG, International Society of Ultrasound in Obstetrics and Gynecology; SMFM, Society for Maternal-Fetal Medicine; UA, umbilical artery.
In general, the SMFM guideline relies particularly on UA Doppler and conventional cardiotocography in monitoring and delivery planning of both early and late suspected FGRs, recommending against the use of middle cerebral artery and ductus venosus Doppler. Concerning surveillance of suspected FGR, the ISUOG guideline recommends different monitoring modalities (umbilical and middle cerebral artery, ductus venosus Doppler, and cCTG) depending on the context in early and late suspected FGR.
Finally, important differences exist regarding the timing of delivery decision in early suspected FGR. Ductus venosus Doppler alteration and low STV on cCTG are recommended in the ISUOG guideline, but explicitly not in SMFM guideline. These additional tools may finesse the delivery guidance based on UA Doppler after 30 weeks of gestation on which the guidelines otherwise largely agree.
Conclusion
A substantial body of evidence has been gathered in the past 4 decades that has helped to clarify the clinical features of suspected FGR across GA and the role of fetal surveillance parameters to predict the clinical course and fetal status at the time of testing. Nevertheless, important differences remained in the recommended management of suspected FGR. Management is a composite process consisting of diagnosis, surveillance, and time of delivery: Doppler evaluation of uteroplacental and fetal vessels all play a separate and defined role in each of these. The evidence underlying fetal placental Doppler as essential components in the management of FGR formed a cornerstone of the ISUOG guideline,4 but not in the SMFM guideline,3 and we consider this to be an important omission. The major areas of contention were the definition of suspected FGR; the use of Doppler parameters for surveillance, quite separately, for decision for delivery; and the value of cCTG vs conventional cardiotocography assessment. These differences might have a profound effect on clinical management, namely, frequency of follow-up, time of delivery, and consequent fetal death rate, and short-and long-term outcomes for these fetuses and infants.
Evaluation of a single biometric fetal measurement defines the size and not fetal growth. Fetoplacental Doppler and evaluation of fetal growth velocity are important tools to identify uteroplacental insufficiency associated with suspected FGR, allowing its differentiation from a constitutionally small fetus.
Level 1 evidence supports the application of UA and ductus venosus Doppler in the monitoring of early FGR. High-quality observational studies support the application of the middle cerebral artery and/or its ratios to UA in the surveillance of late suspected FGR. Collectively, these Doppler parameters can predict fetal deterioration and guide optimal surveillance intervals.
cCTG analysis of fetal heart rate variation and, in particular, the STV provided an objective basis on which to evaluate the fetal condition. Low STV was associated with fetal acidemia and fetal death, and there was strong evidence that monitoring early suspected FGR with ductus venosus Doppler and STV from cardiotocography leads to optimal perinatal outcome.
Acknowledgments
W.L. received limited ultrasound equipment support for research of fetal growth restriction from GE Healthcare. K.H. received honoraria for lectures from GE Healthcare. The unit of E.F. received limited support for research of fetal growth restriction from Samsung, GE, and Esoate. T.F. is President of the Society of Ultrasound in Obs & Gyn of Italy and as a Society they received donations from GE Philips and Samsung for member webinars during 2020–2021. T.F. is also Head of the Obs & Gyn Department of the University of Parma, where Samsung has supported clinical research projects from 2014–2021 providing an ultrasound equipment free of charge to the Unit of Ultrasound and Prenatal Diagnosis. A.D. declares an unrestricted research collaboration with Samsung Medison (Seoul, South Korea) in respect of the evaluation of ultrasound imaging technologies and speaking honoraria from Samsung Medison (Seoul, South Korea) not in relation to this work. C.C.L. has received honoraria and research support from GE, Canon and Samsung.
CCL is supported by the National Institute for Health Research (NIHR) Biomedical Research Centre based at Imperial College Healthcare NHS Trust and Imperial College London. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
Glossary
- AC
abdominal circumference
- AGA
appropriate for gestational age
- CI
confidence interval
- cCTG
computerised Cardiotocography
- CPR
cerebro-placental ratio
- DIGITAT
Disproportionate Intrauterine Growth Intervention Trial at Term
- (A)(R)EDF
(Absent)(Reversed) End-diastolic flow
- EFW
estimated fetal weight
- FGR
fetal growth restriction
- GA
gestational age
- ISUOG
International Society of Ultrasound in Obstetrics and Gynecology
- LGA
large for gestational age
- MCA
middle cerebral artery
- msec
milliseconds
- pH
per hydrion
- PI
pulsatility index
- PORTO
Prospective Observational Trial to Optimize Pediatric Health
- RR
relative risk
- SGA
small for gestational age
- SMFM
Society of Maternal-Fetal Medicine
- STV
short-term variation
- TRUFFLE
Trial of Umbilical and Fetal Flow in Europe
- UA
Umbilical artery
- UCR
Umbilical-cerebral ratio
Footnotes
The other authors report no conflict of interest.
REFERENCES
- 1.McCowan LM, Figueras F, Anderson NH. Evidence-based national guidelines for the management of suspected fetal growth restriction: comparison, consensus, and controversy. Am J Obstet Gynecol 2018;218:S855–68. [DOI] [PubMed] [Google Scholar]
- 2.Chauhan SP, Gupta LM, Hendrix NW, Berghella V; American College of Obstetricians and Gynecologists. Intrauterine growth restriction: comparison of American College of Obstetricians and Gynecologists practice bulletin with other national guidelines. Am J Obstet Gynecol 2009;200:409.e1–6. [DOI] [PubMed] [Google Scholar]
- 3.Society for Maternal-Fetal Medicine, Martins JG, Biggio JR, Abuhamad A. Society for Maternal-Fetal Medicine Consult Series #52: diagnosis and management of fetal growth restriction: (replaces clinical guideline number 3, April 2012). Am J Obstet Gynecol 2020;223: B2–17. [DOI] [PubMed] [Google Scholar]
- 4.Lees CC, Stampalija T, Baschat A, et al. ISUOG Practice Guidelines: diagnosis and management of small-for-gestational-age fetus and fetal growth restriction. Ultrasound Obstet Gynecol 2020;56:298–312. [DOI] [PubMed] [Google Scholar]
- 5.Gordijn SJ, Beune IM, Thilaganathan B, et al. Consensus definition of fetal growth restriction: a Delphi procedure. Ultrasound Obstet Gynecol 2016;48:333–9. [DOI] [PubMed] [Google Scholar]
- 6.Hadlock FP, Harrist RB, Martinez-Poyer J. In utero analysis of fetal growth: a sonographic weight standard. Radiology 1991;181:129–33. [DOI] [PubMed] [Google Scholar]
- 7.Battaglia FC, Lubchenco LO. A practical classification of newborn infants by weight and gestational age. J Pediatr 1967;71:159–63. [DOI] [PubMed] [Google Scholar]
- 8.Malin GL, Morris RK, Riley RD, Teune MJ, Khan KS. When is birthweight at term (≥37 weeks’ gestation) abnormally low? A systematic review and meta-analysis of the prognostic and predictive ability of current birthweight standards for childhood and adult outcomes. BJOG 2015;122:634–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Arcangeli T, Thilaganathan B, Hooper R, Khan KS, Bhide A. Neurodevelopmental delay in small babies at term: a systematic review. Ultrasound Obstet Gynecol 2012;40:267–75. [DOI] [PubMed] [Google Scholar]
- 10.Vasak B, Koenen SV, Koster MP, et al. Human fetal growth is constrained below optimal for perinatal survival. Ultrasound Obstet Gynecol 2015;45:162–7. [DOI] [PubMed] [Google Scholar]
- 11.McIntire DD, Bloom SL, Casey BM, Leveno KJ. Birth weight in relation to morbidity and mortality among newborn infants. N Engl J Med 1999;340:1234–8. [DOI] [PubMed] [Google Scholar]
- 12.Unterscheider J, Daly S, Geary MP, et al. Optimizing the definition of intrauterine growth restriction: the multicenter prospective PORTO study. Am J Obstet Gynecol 2013;208:290. e1–6. [DOI] [PubMed] [Google Scholar]
- 13.Pilliod RA, Cheng YW, Snowden JM, Doss AE, Caughey AB. The risk of intrauterine fetal death in the small-for-gestational-age fetus. Am J Obstet Gynecol 2012;207:318.e1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Trudell AS, Cahill AG, Tuuli MG, Macones GA, Odibo AO. Risk of stillbirth after 37 weeks in pregnancies complicated by small-for-gestational-age fetuses. Am J Obstet Gynecol 2013;208:376.e1–7. [DOI] [PubMed] [Google Scholar]
- 15.van Wyk L, Boers KE, van der Post JA, et al. Effects on (neuro) developmental and behavioral outcome at 2 years of age of induced labor compared with expectant management in intrauterine growth-restricted infants: long-term outcomes of the DIGITAT trial. Am J Obstet Gynecol 2012;206:406.e1–7. [DOI] [PubMed] [Google Scholar]
- 16.Boers KE, Vijgen SM, Bijlenga D, et al. Induction versus expectant monitoring for intrauterine growth restriction at term: randomised equivalence trial (DIGITAT). BMJ 2010;341: c7087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stampalija T, Thornton J, Marlow N, et al. Fetal cerebral Doppler changes and outcome in late preterm fetal growth restriction: prospective cohort study. Ultrasound Obstet Gynecol 2020;56:173–81. [DOI] [PubMed] [Google Scholar]
- 18.Savchev S, Figueras F, Cruz-Martinez R, Illa M, Botet F, Gratacos E. Estimated weight centile as a predictor of perinatal outcome in small-for-gestational-age pregnancies with normal fetal and maternal Doppler indices. Ultrasound Obstet Gynecol 2012;39:299–303. [DOI] [PubMed] [Google Scholar]
- 19.Sovio U, White IR, Dacey A, Pasupathy D, Smith GCS. Screening for fetal growth restriction with universal third trimester ultrasonography in nulliparous women in the Pregnancy Outcome Prediction (POP) study: a prospective cohort study. Lancet 2015;386:2089–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bhide A, Acharya G, Baschat A, et al. ISUOG Practice Guidelines (updated): use of Doppler velocimetry in obstetrics. Ultrasound Obstet Gynecol 2021;58:331–9. [DOI] [PubMed] [Google Scholar]
- 21.Deter RL, Lee W, Yeo L, et al. Individualized growth assessment: conceptual framework and practical implementation for the evaluation of fetal growth and neonatal growth outcome. Am J Obstet Gynecol 2018;218:S656–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Black RE, Victora CG, Walker SP, et al. Maternal and child undernutrition and over-weight in low-income and middle-income countries. Lancet 2013;382:427–51. [DOI] [PubMed] [Google Scholar]
- 23.Lampl M, Jeanty P. Timing is everything: a reconsideration of fetal growth velocity patterns identifies the importance of individual and sex differences. Am J Hum Biol 2003;15:667–80. [DOI] [PubMed] [Google Scholar]
- 24.Thilaganathan B. Ultrasound fetal weight estimation at term may do more harm than good. Ultrasound Obstet Gynecol 2018;52:5–8. [DOI] [PubMed] [Google Scholar]
- 25.Poon LC, Tan MY, Yerlikaya G, Syngelaki A, Nicolaides KH. Birth weight in live births and stillbirths. Ultrasound Obstet Gynecol 2016;48: 602–6. [DOI] [PubMed] [Google Scholar]
- 26.Pacora P, Romero R, Jung E, et al. Reduced fetal growth velocity precedes antepartum fetal death. Ultrasound Obstet Gynecol 2021;57: 942–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jaiman S, Romero R, Pacora P, et al. Disorders of placental villous maturation in fetal death. J Perinat Med. 2020. Apr 1:/j/jpme.ahead-of-print/jpm-2020–0030/jpm-2020–00 30.xml. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Deter RL, Lee W, Sangi-Haghpeykar H, Kingdom J, Romero R. Third trimester growth restriction patterns: individualized assessment using a fetal growth pathology score. J Matern Fetal Neonatal Med 2018;31:2155–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Deter RL, Lee W, Dicker P, et al. Growth patterns and cardiovascular abnormalities in small for gestational age fetuses: 1. Pattern characteristics. J Matern Fetal Neonatal Med 2021;34:3029–38. [DOI] [PubMed] [Google Scholar]
- 30.Ciobanu A, Rouvali A, Syngelaki A, Akolekar R, Nicolaides KH. Prediction of small for gestational age neonates: screening by maternal factors, fetal biometry, and biomarkers at 35–37 weeks’ gestation. Am J Obstet Gynecol 2019;220:486.e1–11. [DOI] [PubMed] [Google Scholar]
- 31.Figueras F, Gratacós E. Update on the diagnosis and classification of fetal growth restriction and proposal of a stage-based management protocol. Fetal Diagn Ther 2014;36: 86–98. [DOI] [PubMed] [Google Scholar]
- 32.Savchev S, Figueras F, Sanz-Cortes M, et al. Evaluation of an optimal gestational age cut-off for the definition of early- and late-onset fetal growth restriction. Fetal Diagn Ther 2014;36: 99–105. [DOI] [PubMed] [Google Scholar]
- 33.Levytska K, Higgins M, Keating S, et al. Placental pathology in relation to uterine artery Doppler findings in pregnancies with severe intrauterine growth restriction and abnormal umbilical artery Doppler changes. Am J Perinatol 2017;34:451–7. [DOI] [PubMed] [Google Scholar]
- 34.Bilardo CM, Nicolaides KH, Campbell S. Doppler measurements of fetal and uteroplacental circulations: relationship with umbilical venous blood gases measured at cordocentesis. Am J Obstet Gynecol 1990;162:115–20. [DOI] [PubMed] [Google Scholar]
- 35.Crimmins S, Desai A, Block-Abraham D, Berg C, Gembruch U, Baschat AA. A comparison of Doppler and biophysical findings between liveborn and stillborn growth-restricted fetuses. Am J Obstet Gynecol 2014;211:669.e1–10. [DOI] [PubMed] [Google Scholar]
- 36.Mari G, Deter RL. Middle cerebral artery flow velocity waveforms in normal and small-for-gestational-age fetuses. Am J Obstet Gynecol 1992;166:1262–70. [DOI] [PubMed] [Google Scholar]
- 37.Stampalija T, Arabin B, Wolf H, Bilardo CM, Lees C, TRUFFLE Investigators. Is middle cerebral artery Doppler related to neonatal and 2-year infant outcome in early fetal growth restriction? Am J Obstet Gynecol 2017;216:521. e1–13. [DOI] [PubMed] [Google Scholar]
- 38.Khalil A, Morales-Roselló J, Townsend R, et al. Value of third-trimester cerebroplacental ratio and uterine artery Doppler indices as predictors of stillbirth and perinatal loss. Ultrasound Obstet Gynecol 2016;47:74–80. [DOI] [PubMed] [Google Scholar]
- 39.Vollgraff Heidweiller-Schreurs CA, De Boer MA, Heymans MW, et al. Prognostic accuracy of cerebroplacental ratio and middle cerebral artery Doppler for adverse perinatal outcome: systematic review and meta-analysis. Ultrasound Obstet Gynecol 2018;51: 313–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Conde-Agudelo A, Villar J, Kennedy SH, Papageorghiou AT. Predictive accuracy of cerebroplacental ratio for adverse perinatal and neurodevelopmental outcomes in suspected fetal growth restriction: systematic review and meta-analysis. Ultrasound Obstet Gynecol 2018;52:430–41. [DOI] [PubMed] [Google Scholar]
- 41.Lees C, Marlow N, Arabin B, et al. Perinatal morbidity and mortality in early-onset fetal growth restriction: cohort outcomes of the trial of randomized umbilical and fetal flow in Europe (TRUFFLE). Ultrasound Obstet Gynecol 2013;42:400–8. [DOI] [PubMed] [Google Scholar]
- 42.Molina LCG, Odibo L, Zientara S, et al. Validation of Delphi procedure consensus criteria for defining fetal growth restriction. Ultrasound Obstet Gynecol 2020;56:61–6. [DOI] [PubMed] [Google Scholar]
- 43.Thompson RS, Trudinger BJ. Doppler waveform pulsatility index and resistance, pressure and flow in the umbilical placental circulation: an investigation using a mathematical model. Ultrasound Med Biol 1990;16:449–58. [DOI] [PubMed] [Google Scholar]
- 44.Trudinger B. Doppler: more or less? Ultrasound Obstet Gynecol 2007;29:243–6. [DOI] [PubMed] [Google Scholar]
- 45.Mari G, Hanif F, Kruger M. Sequence of cardiovascular changes in IUGR in pregnancies with and without preeclampsia. Prenat Diagn 2008;28:377–83. [DOI] [PubMed] [Google Scholar]
- 46.Turan OM, Turan S, Berg C, et al. Duration of persistent abnormal ductus venosus flow and its impact on perinatal outcome in fetal growth restriction. Ultrasound Obstet Gynecol 2011;38: 295–302. [DOI] [PubMed] [Google Scholar]
- 47.Baschat AA, Kush M, Berg C, et al. Hematologic profile of neonates with growth restriction is associated with rate and degree of prenatal Doppler deterioration. Ultrasound Obstet Gynecol 2013;41:66–72. [DOI] [PubMed] [Google Scholar]
- 48.Ferrazzi E, Bozzo M, Rigano S, et al. Temporal sequence of abnormal Doppler changes in the peripheral and central circulatory systems of the severely growth-restricted fetus. Ultrasound Obstet Gynecol 2002;19: 140–6. [DOI] [PubMed] [Google Scholar]
- 49.Hecher K, Bilardo CM, Stigter RH, et al. Monitoring of fetuses with intrauterine growth restriction: a longitudinal study. Ultrasound Obstet Gynecol 2001;18:564–70. [DOI] [PubMed] [Google Scholar]
- 50.Schwarze A, Gembruch U, Krapp M, Katalinic A, Germer U, Axt-Fliedner R. Qualitative venous Doppler flow waveform analysis in preterm intrauterine growth-restricted fetuses with ARED flow in the umbilical artery–correlation with short-term outcome. Ultrasound Obstet Gynecol 2005;25:573–9. [DOI] [PubMed] [Google Scholar]
- 51.Baschat AA, Gembruch U, Weiner CP, Harman CR. Qualitative venous Doppler waveform analysis improves prediction of critical perinatal outcomes in premature growth-restricted fetuses. Ultrasound Obstet Gynecol 2003;22: 240–5. [DOI] [PubMed] [Google Scholar]
- 52.Baschat AA, Gembruch U, Reiss I, Gortner L, Weiner CP, Harman CR. Relationship between arterial and venous Doppler and perinatal outcome in fetal growth restriction. Ultrasound Obstet Gynecol 2000;16:407–13. [DOI] [PubMed] [Google Scholar]
- 53.Kiserud T, Eik-Nes SH, Blaas HG, Hellevik LR, Simensen B. Ductus venosus blood velocity and the umbilical circulation in the seriously growth-retarded fetus. Ultrasound Obstet Gynecol 1994;4:109–14. [DOI] [PubMed] [Google Scholar]
- 54.Kiserud T, Eik-Nes SH, Blaas HG, Hellevik LR. Ultrasonographic velocimetry of the fetal ductus venosus. Lancet 1991;338: 1412–4. [DOI] [PubMed] [Google Scholar]
- 55.Ferrazzi E, Lees C, Acharya G. The controversial role of the ductus venosus in hypoxic human fetuses. Acta Obstet Gynecol Scand 2019;98:823–9. [DOI] [PubMed] [Google Scholar]
- 56.Kiserud T, Kessler J, Ebbing C, Rasmussen S. Ductus venosus shunting in growth-restricted fetuses and the effect of umbilical circulatory compromise. Ultrasound Obstet Gynecol 2006;28:143–9. [DOI] [PubMed] [Google Scholar]
- 57.Kiserud T, Acharya G. The fetal circulation. Prenat Diagn 2004;24:1049–59. [DOI] [PubMed] [Google Scholar]
- 58.Baschat AA, Hecher K. Fetal growth restriction due to placental disease. Semin Perinatol 2004;28:67–80. [DOI] [PubMed] [Google Scholar]
- 59.Baschat AA, Güclü S, Kush ML, Gembruch U, Weiner CP, Harman CR. Venous Doppler in the prediction of acid-base status of growth-restricted fetuses with elevated placental blood flow resistance. Am J Obstet Gynecol 2004;191:277–84. [DOI] [PubMed] [Google Scholar]
- 60.Bilardo CM, Wolf H, Stigter RH, et al. Relationship between monitoring parameters and perinatal outcome in severe, early intrauterine growth restriction. Ultrasound Obstet Gynecol 2004;23:119–25. [DOI] [PubMed] [Google Scholar]
- 61.Oros D, Figueras F, Cruz-Martinez R, Meler E, Munmany M, Gratacos E. Longitudinal changes in uterine, umbilical and fetal cerebral Doppler indices in late-onset small-for-gestational age fetuses. Ultrasound Obstet Gynecol 2011;37:191–5. [DOI] [PubMed] [Google Scholar]
- 62.Wladimiroff JW, Tonge HM, Stewart PA. Doppler ultrasound assessment of cerebral blood flow in the human fetus. Br J Obstet Gynaecol 1986;93:471–5. [PubMed] [Google Scholar]
- 63.Richardson BS, Bocking AD. Metabolic and circulatory adaptations to chronic hypoxia in the fetus. Comp Biochem Physiol A Mol Integr Physiol 1998;119:717–23. [DOI] [PubMed] [Google Scholar]
- 64.Giussani DA. The fetal brain sparing response to hypoxia: physiological mechanisms. J Physiol 2016;594:1215–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hecher K, Snijders R, Campbell S, Nicolaides K. Fetal venous, intracardiac, and arterial blood flow measurements in intrauterine growth retardation: relationship with fetal blood gases. Am J Obstet Gynecol 1995;173:10–5. [DOI] [PubMed] [Google Scholar]
- 66.Akalin-Sel T, Nicolaides KH, Peacock J, Campbell S. Doppler dynamics and their complex interrelation with fetal oxygen pressure, carbon dioxide pressure, and pH in growth-retarded fetuses. Obstet Gynecol 1994;84: 439–44. [PubMed] [Google Scholar]
- 67.Cruz-Martínez R, Figueras F, Hernandez-Andrade E, Oros D, Gratacos E. Fetal brain Doppler to predict cesarean delivery for non-reassuring fetal status in term small-for-gestational-age fetuses. Obstet Gynecol 2011;117:618–26. [DOI] [PubMed] [Google Scholar]
- 68.Severi FM, Bocchi C, Visentin A, et al. Uterine and fetal cerebral Doppler predict the outcome of third-trimester small-for-gestational age fetuses with normal umbilical artery Doppler. Ultrasound Obstet Gynecol 2002;19: 225–8. [DOI] [PubMed] [Google Scholar]
- 69.Hershkovitz R, Kingdom JC, Geary M, Rodeck CH. Fetal cerebral blood flow redistribution in late gestation: identification of compromise in small fetuses with normal umbilical artery Doppler. Ultrasound Obstet Gynecol 2000;15:209–12. [DOI] [PubMed] [Google Scholar]
- 70.Oros D, Figueras F, Cruz-Martinez R, et al. Middle versus anterior cerebral artery Doppler for the prediction of perinatal outcome and neonatal neurobehavior in term small-for-gestational-age fetuses with normal umbilical artery Doppler. Ultrasound Obstet Gynecol 2010;35:456–61. [DOI] [PubMed] [Google Scholar]
- 71.Eixarch E, Meler E, Iraola A, et al. Neurodevelopmental outcome in 2-year-old infants who were small-for-gestational age term foetuses with cerebral blood flow redistribution. Ultrasound Obstet Gynecol 2008;32:894–9. [DOI] [PubMed] [Google Scholar]
- 72.Monteith C, Flood K, Pinnamaneni R, et al. An abnormal cerebroplacental ratio (CPR) is predictive of early childhood delayed neurodevelopment in the setting of fetal growth restriction. Am J Obstet Gynecol 2019;221:273.e1–9. [DOI] [PubMed] [Google Scholar]
- 73.Karlsen HO, Ebbing C, Rasmussen S, Kiserud T, Johnsen SL. Use of conditional centiles of middle cerebral artery pulsatility index and cerebroplacental ratio in the prediction of adverse perinatal outcomes. Acta Obstet Gynecol Scand 2016;95:690–6. [DOI] [PubMed] [Google Scholar]
- 74.Vergani P, Andreotti C, Roncaglia N, et al. Doppler predictors of adverse neonatal outcome in the growth restricted fetus at 34 weeks’ gestation or beyond. Am J Obstet Gynecol 2003;189:1007–11. [DOI] [PubMed] [Google Scholar]
- 75.Kontopoulos EV, Vintzileos AM. Condition-specific antepartum fetal testing. Am J Obstet Gynecol 2004;191:1546–51. [DOI] [PubMed] [Google Scholar]
- 76.Wolf H, Arabin B, Lees CC, et al. Longitudinal study of computerized cardiotocography in early fetal growth restriction. Ultrasound Obstet Gynecol 2017;50:71–8. [DOI] [PubMed] [Google Scholar]
- 77.Divon MY, Girz BA, Lieblich R, Langer O. Clinical management of the fetus with markedly diminished umbilical artery end-diastolic flow. Am J Obstet Gynecol 1989;161:1523–7. [DOI] [PubMed] [Google Scholar]
- 78.Baschat AA. Considering evidence in the management of fetal growth restriction. Ultrasound Obstet Gynecol 2021;57:25–8. [DOI] [PubMed] [Google Scholar]
- 79.Gramellini D, Folli MC, Raboni S, Vadora E, Merialdi A. Cerebral-umbilical Doppler ratio as a predictor of adverse perinatal outcome. Obstet Gynecol 1992;79:416–20. [DOI] [PubMed] [Google Scholar]
- 80.Harrington K, Carpenter RG, Nguyen M, Campbell S. Changes observed in Doppler studies of the fetal circulation in pregnancies complicated by pre-eclampsia or the delivery of a small-for-gestational-age baby. I. Cross-sectional analysis. Ultrasound Obstet Gynecol 1995;6:19–28. [DOI] [PubMed] [Google Scholar]
- 81.Lees CC, Marlow N, van Wassenaer-Leemhuis A, et al. 2 year neurodevelopmental and intermediate perinatal outcomes in infants with very preterm fetal growth restriction (TRUFFLE): a randomised trial. Lancet 2015;385:2162–72. [DOI] [PubMed] [Google Scholar]
- 82.Snijders RJ, Ribbert LS, Visser GH, Mulder EJ. Numeric analysis of heart rate variation in intrauterine growth-retarded fetuses: a longitudinal study. Am J Obstet Gynecol 1992;166:22–7. [DOI] [PubMed] [Google Scholar]
- 83.Ribbert LS, Visser GH, Mulder EJ, Zonneveld MF, Morssink LP. Changes with time in fetal heart rate variation, movement incidences and haemodynamics in intrauterine growth retarded fetuses: a longitudinal approach to the assessment of fetal well being. Early Hum Dev 1993;31:195–208. [DOI] [PubMed] [Google Scholar]
- 84.Ribbert LS, Snijders RJ, Nicolaides KH, Visser GH. Relation of fetal blood gases and data from computer-assisted analysis of fetal heart rate patterns in small for gestation fetuses. Br J Obstet Gynaecol 1991;98:820–3. [DOI] [PubMed] [Google Scholar]
- 85.Guzman ER, Vintzileos AM, Martins M, Benito C, Houlihan C, Hanley M. The efficacy of individual computer heart rate indices in detecting acidemia at birth in growth-restricted fetuses. Obstet Gynecol 1996;87:969–74. [DOI] [PubMed] [Google Scholar]
- 86.Guzman ER, Vintzileos A, Egan JF, Benito C, Lake M, Lai YL. Antenatal prediction of fetal pH in growth restricted fetuses using computer analysis of the fetal heart rate. J Matern Fetal Med 1998;7:43–7. [DOI] [PubMed] [Google Scholar]
- 87.Vintzileos AM, Campbell WA, Bors-Koefoed R, Rodis JF, Gaffney SE, Montgomery JT. Relationship between cyclic variation of fetal heart rate patterns and cord pH in preterm gestations. Am J Perinatol 1989;6:310–3. [DOI] [PubMed] [Google Scholar]
- 88.Vintzileos AM, Fleming AD, Scorza WE, et al. Relationship between fetal biophysical activities and umbilical cord blood gas values. Am J Obstet Gynecol 1991;165:707–13. [DOI] [PubMed] [Google Scholar]
- 89.Vintzileos AM, Gaffney SE, Salinger LM, Kontopoulos VG, Campbell WA, Nochimson DJ. The relationships among the fetal biophysical profile, umbilical cord pH, and Apgar scores. Am J Obstet Gynecol 1987;157:627–31. [DOI] [PubMed] [Google Scholar]
- 90.Manning FA, Snijders R, Harman CR, Nicolaides K, Menticoglou S, Morrison I. Fetal biophysical profile score. VI. Correlation with antepartum umbilical venous fetal pH. Am J Obstet Gynecol 1993;169:755–63. [DOI] [PubMed] [Google Scholar]
- 91.Visser GH, Sadovsky G, Nicolaides KH. Antepartum heart rate patterns in small-for-gestational-age third-trimester fetuses: correlations with blood gas values obtained at cordocentesis. Am J Obstet Gynecol 1990;162: 698–703. [DOI] [PubMed] [Google Scholar]
- 92.Visser GH, Redman CW, Huisjes HJ, Turnbull AC. Nonstressed antepartum heart rate monitoring: implications of decelerations after spontaneous contractions. Am J Obstet Gynecol 1980;138:429–35. [DOI] [PubMed] [Google Scholar]
- 93.Bekedam DJ, Visser GH, Mulder EJ, Poel-mann-Weesjes G. Heart rate variation and movement incidence in growth-retarded fetuses: the significance of antenatal late heart rate decelerations. Am J Obstet Gynecol 1987;157: 126–33. [DOI] [PubMed] [Google Scholar]
- 94.Pardi G, Cetin I, Marconi AM, et al. Diagnostic value of blood sampling in fetuses with growth retardation. N Engl J Med 1993;328: 692–6. [DOI] [PubMed] [Google Scholar]
- 95.Romero R. Giants in Obstetrics and Gynecology Series: a profile of Christopher Redman, MB, BChir, MRCP, FRCP. Am J Obstet Gynecol 2019;220:420–7.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Pardey J, Moulden M, Redman CW. A computer system for the numerical analysis of nonstress tests. Am J Obstet Gynecol 2002;186:1095–103. [DOI] [PubMed] [Google Scholar]
- 97.Nijhuis IJ, ten Hof J. Development of fetal heart rate and behavior: indirect measures to assess the fetal nervous system. Eur J Obstet Gynecol Reprod Biol 1999;87:1–2. [DOI] [PubMed] [Google Scholar]
- 98.Smith JH, Anand KJ, Cotes PM, et al. Antenatal fetal heart rate variation in relation to the respiratory and metabolic status of the compromised human fetus. Br J Obstet Gynaecol 1988;95:980–9. [DOI] [PubMed] [Google Scholar]
- 99.Dawes GS, Moulden M, Redman CW. Short-term fetal heart rate variation, decelerations, and umbilical flow velocity waveforms before labor. Obstet Gynecol 1992;80:673–8. [PubMed] [Google Scholar]
- 100.Serra V, Moulden M, Bellver J, Redman CW. The value of the short-term fetal heart rate variation for timing the delivery of growth-retarded fetuses. BJOG 2008;115:1101–7. [DOI] [PubMed] [Google Scholar]
- 101.Anceschi MM, Ruozi-Berretta A, Piazze JJ, et al. Computerized cardiotocography in the management of intrauterine growth restriction associated with Doppler velocimetry alterations. Int J Gynaecol Obstet 2004;86: 365–70. [DOI] [PubMed] [Google Scholar]
- 102.Visser GHA, Bilardo CM, Derks JB, et al. Fetal monitoring indications for delivery and 2-year outcome in 310 infants with fetal growth restriction delivered before 32 weeks’ gestation in the TRUFFLE study. Ultrasound Obstet Gynecol 2017;50:347–52. [DOI] [PubMed] [Google Scholar]
- 103.Alfirevic Z, Stampalija T, Dowswell T. Fetal and umbilical Doppler ultrasound in high-risk pregnancies. Cochrane Database Syst Rev 2017;6:CD007529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Romero R, Kalache KD, Kadar N. Timing the delivery of the preterm severely growth-restricted fetus: venous Doppler, cardiotocography or the biophysical profile? Ultrasound Obstet Gynecol 2002;19:118–21. [DOI] [PubMed] [Google Scholar]