Abstract
Introduction:
Mixed phenotype acute leukemia (MPAL) is a rare type of acute leukemia with immunophenotypic features of both myeloid-derived and lymphoid-derived lineages.
Case Presentation:
We present an atypical case of a 32-year-old woman presenting with an anterior mediastinal mass and pericardial/pleural involvement that was initially diagnosed as primary mediastinal diffuse large B-cell lymphoma. However, flow cytometry on pleural fluid confirmed the diagnosis of MPAL of B-cell/myeloid lineage without peripheral blood/bone marrow involvement. The patient was treated with an acute lymphoblastic leukemia-type regimen and proceeded with myeloablative allogeneic hematopoietic cell transplantation in first complete remission.
Conclusion:
MPAL can rarely present with isolated extramedullary disease without leukemic involvement and can often be misdiagnosed as a non-Hodgkin lymphoma. Careful integration of all the clinical data, particularly flow cytometry results, can clarify the diagnosis and change the treatment plan.
Introduction
Accurate diagnosis of leukemia is imperative given that treatment and prognosis rely heavily on the lineage of the proliferative blast. Evaluation typically incorporates blast morphology, flow cytometry, karyotyping, and other genetic analyses to identify the leukemic cells as primarily myeloid or B- or T-lymphoblasts.1 Assignment of lineage typically relies on flow cytometry and immunohistochemical stains. There exists a rare situation in which the leukemic blasts are of both myeloid- and lymphoid-derived lineages, a condition known as mixed phenotype acute leukemia (MPAL). MPAL may be bilineal, in which there are 2 or more discrete components with lineage-specific markers, or biphenotypic, in which there is 1 population having coexpression of lineage-specific markers. MPAL comprises roughly 1%–4% of all leukemias.2 Exact data regarding the true incidence and survival of MPAL have been limited owing to the relatively sparse number of cases, but the prognosis is typically poorer compared with other leukemias.3 Like most leukemias, diagnosis relies on bone marrow biopsy, which will often demonstrate an increased blast population of multiple lineages. However, in rare circumstances, leukemias present as solid extramedullary tumors known as myeloid sarcomas (MS). One study suggested that these tumors occur in about 0.8% of patients with acute myeloid leukemia (AML).4 Therefore, MPAL without peripheral blood/bone marrow involvement is extremely rare with few documented cases in the literature.5,6 We present a rare case of MPAL of myeloid and B-cell lineage presenting with an anterior mediastinal mass and pericardial/pleural involvement without leukemic involvement.
Case Presentation
A 32-year-old woman with no substantial past medical history presented with 1 month of progressive dyspnea, cough, and intermittent fevers. Computed tomography (CT) of the chest demonstrated a 9.6-cm anterior mediastinal mass, moderate right pleural effusion, and large pericardial effusion with mass effect.
She was admitted for expedited workup and underwent pericardiocentesis with drainage of 650 mL of bloody fluid. She then underwent a CT-guided needle biopsy of the mediastinal mass, which showed extensive crush artifact and focal necrosis. In intact areas, there was an atypical lymphoid infiltrate composed of large cells that were positive for CD19, BCL-2, and c-MYC and negative for CD5, CD10, CD20, CD30, CD34, BCL-6, and MUM-1. A TdT stain was equivocal, and the Ki-67 proliferation index was 60%–70%. Fluorescence in situ hybridization tests were negative for MYC, BCL-2, and BCL-6 gene rearrangements. Overall, the findings were felt to be most consistent with CD20-negative diffuse large B-cell lymphoma (DLBCL), non-germinal center B-cell–like subtype.
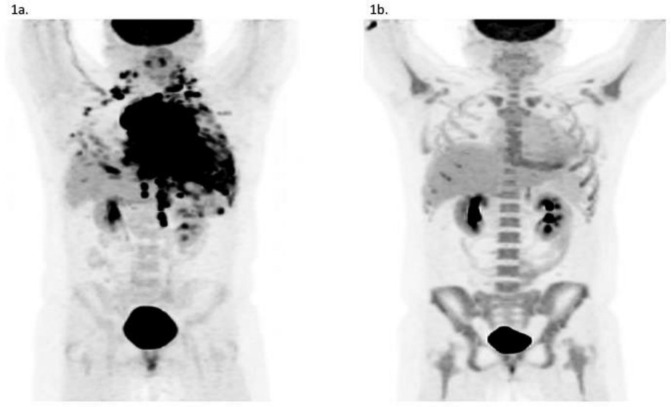
Positron emission tomography (PET)/CT scan demonstrated a large intensely hypermetabolic 9.8-cm anterior mediastinal mass with pericardial involvement; extensive hypermetabolic bilateral pleural masses; and numerous mediastinal, cervical, axillary, and upper retroperitoneal lymph nodes (Figure 1).
Figure 1.
PET/CT scan showing a large, FDG-avid mediastinal mass (A) before and (B) after induction chemotherapy. FDG = fluorine-18 fluorodeoxyglucose.
She was discharged but was readmitted soon after with worsening dyspnea. A repeat CT chest showed an interval increase in size of the mediastinal mass now measuring 10.3 cm, with pericardial invasion. There was also an interval increase in her left pleural effusion. Her labs showed a lactate dehydrogenase level of 273 U/L, white blood cell count of 11.7 × 103/μL, hemoglobin level of 12 g/dL, and platelet count of 370 × 103/μL. She underwent a left-sided thoracentesis and was immediately started on DA-R-EPOCH (dose-adjusted rituximab, etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin) for presumed primary mediastinal DLBCL.
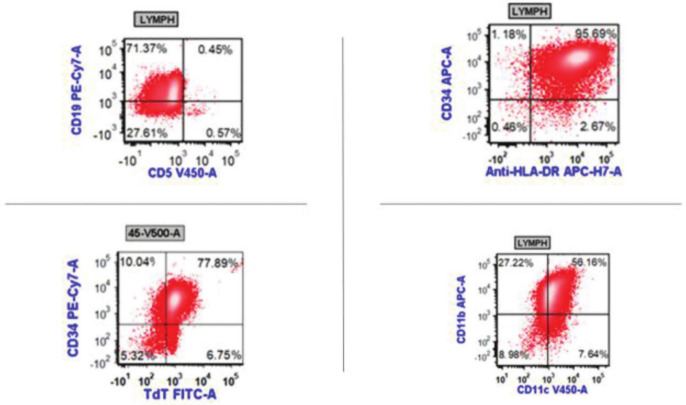
However, flow cytometry from the pleural fluid came back showing a population of cells that were positive for CD34, CD19 (dim), CD33, CD15 (partial), CD11b, CD11c, and TdT (dim partial) (Figure 2). The population also expressed partial cytoplasmic CD22 and partial cytoplasmic CD79a. CD64 expression was equivocal but favored to be positive. On the basis of these flow results, her diagnosis was felt to be more consistent with MPAL of B-cell and myeloid lineage.
Figure 2.
Flow cytometry results on pleural fluid showing a cell population that is positive for CD19, CD34, TdT, HLA-DR, CD11b, and CD11c.
DA-R-EPOCH was discontinued on day 2, and she underwent bone marrow biopsy and lumbar puncture for staging. Both bone marrow and cerebrospinal fluid were negative for leukemic involvement based on morphology and flow cytometry.
Molecular analysis of her pleural fluid was negative for BCR-ABL rearrangement, FLT3 gene mutation, MLL gene rearrangement, and Philadelphia chromosome-like genetic alterations.
She was treated with an acute lymphoblastic leukemia (ALL) pediatric regimen (CALGB 10403 protocol) and achieved complete remission on post-induction PET scan (Figure 1B). She was transitioned to consolidation therapy with hyperCVAD (hyperfractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone) for cycles 2 and 3. She then proceeded with a myeloablative matched related donor allogeneic hematopoietic cell transplantation and remains in remission at > 6 months from transplant. The timeline of her diagnostic and therapeutic events is summarized in Table 1.
Table 1.
Timeline of the major clinical events
| Date | Log type | Notes |
|---|---|---|
| 3/2020 | Symptoms |
|
| 5/11/20 | Symptoms, signs, workup |
|
| 5/12/20 | Procedure, workup |
|
| 6/8/20 | Symptoms, transfer |
|
| 6/11/20 | Treatment |
|
| 6/13/20 | Results |
|
| 6/19/20 | Treatment |
|
| 9/15/20 | Treatment |
|
Discussion
The diagnosis of MPAL was first reported in the 1980s, when leukemias stemming from multiple lineages were found to proliferate in the bone marrow.7 Although this disease is much rarer than typical leukemias, the true incidence may be underestimated because the use of restrictive immunophenotypic panels or combinations can lower the sensitivity of detection.8 Extramedullary disease, also known as MS, without concomitant peripheral blood/bone marrow involvement is a rare phenomenon in acute leukemia.9 Extramedullary MPAL without leukemic involvement suggests an ultrarare disease presentation, with a limited number of cases reported.
Our review of the literature revealed only 18 documented cases of MPAL without bone marrow infiltration. One report consisted of 11 pediatric cases: 7 B/myeloid, 2 T/myeloid, and 2 B/T-cell.10 All 11 cases achieved complete remission after standard ALL therapy. Only 1 case of adult B/myeloid MPAL was documented, who remained in remission 19 months after transplant.5 A common theme among these cases is the difficulty in obtaining an accurate diagnosis, which played a prominent role in our case as well. Pure medullary sarcoma (ie, without leukemic involvement) is often misdiagnosed as other lymphomas, Ewing’s sarcoma, or carcinoma up to 47% of the time.11,12 Indeed, after the initial biopsy, our patient was believed to have primary mediastinal DLBCL and treated with DA-R-EPOCH. This was likely confounded by a suboptimal biopsy sample of the mediastinal mass and lack of peripheral blood/bone marrow involvement. Remarkably, flow cytometry of the pleural effusion was able to elucidate and confirm the diagnosis of MPAL and, ultimately, changed her treatment plan.
Based on the 2016 World Health Organization classification, MPAL can be subdivided into the following categories: 1) acute undifferentiated leukemia, 2) MPAL with (9;22)(q34.1;q11.2);BCR-ABL1, 3) MPAL with t(v;11q23.3); KMT2A rearranged, 4) B/myeloid not otherwise specified, and 5) T/myeloid not otherwise specified.13 Although prognosis has been difficult to track owing to the relative paucity of cases, the presence of BCR-ABL (Philadelphia chromosome) gene rearrangement, the presence of MLL gene (KMT2A) rearrangement, and the T/myeloid phenotype have been associated with worse outcomes.14,15
Retrospective studies suggest that patients with MPAL do better with an ALL regimen rather than an AML regimen.13,16 One study of 100 patients with MPAL showed that an ALL regimen was associated with a higher response rate (85% vs 41%) and higher median overall survival (139 vs 11 months) compared with an AML regimen.3 As with Ph-positive ALL, addition of a tyrosine kinase inhibitor is recommended for MPAL with the Philadelphia chromosome. Regarding the role of allogeneic hematopoietic cell transplant (allo-HCT) in MPAL, it is generally recommended in first remission for adult MPAL patients based on cohort studies showing favorable outcomes with chemotherapy followed by allo-HCT compared with chemotherapy alone.17–19 It is possible that there are select patients (eg, early minimal residual disease negativity, patients < 40 years of age treated with a pediatric ALL regimen) who would do well with chemotherapy alone and be spared the toxicities of allo-HCT.20 In our patient’s case, she was treated with an ALL regimen followed by allo-HCT and remains in remission.
Conclusion
Here, we presented a rare case of MPAL presenting as a pure MS without leukemic involvement that was initially misdiagnosed as primary mediastinal DLBCL. What is also unique about this case is how flow cytometry on pleural fluid was able to confirm the diagnosis of MPAL. This shows that flow cytometry data can be crucial in making the diagnosis when the tissue sample is limited or suboptimal.
Acknowledgments
We thank Don Udall, MD, for providing the radiology images.
Footnotes
Author Contributions: Jeffrey Means, DO, participated in the collection of data, critical review, drafting, and submission of the final manuscript. Allison Shaw, MD, participated in the critical review and drafting of the final manuscript and assisted in obtaining images and figures for the final manuscript. David Feldman, MD, participated in the review of data and drafting of the final manuscript. Khoan Vu, MD, participated in the critical review, drafting, and submission of the final manuscript. All authors have given final approval to the manuscript.
Conflicts of Interest: None declared
Funding: None declared
Consent: Informed consent was received from all case patients.
References
- 1.Weinberg OK, Arber DA. Mixed-phenotype acute leukemia: Historical overview and a new definition. Leukemia 2010;24(11):1844–1851. DOI: 10.1038/leu.2010.202 [DOI] [PubMed] [Google Scholar]
- 2.van den Ancker W, Terwijn M, Westers TM, et al. Acute leukemias of ambiguous lineage: Diagnostic consequences of the WHO2008 classification. Leukemia 2010;24(7):1392–1396. DOI: 10.1038/leu.2010.119 [DOI] [PubMed] [Google Scholar]
- 3.Matutes E, Pickl WF, van’t Veer M, et al. Mixed-phenotype acute leukemia: Clinical and laboratory features and outcome in 100 patients defined according to the WHO 2008 classification. Blood 2011;117(11):3163–3171. DOI: 10.1182/blood-2010-10-314682 [DOI] [PubMed] [Google Scholar]
- 4.Ward E, DeSantis C, Robbins A, Kohler B, Jemal A. Childhood and adolescent cancer statistics, 2014. CA Cancer J Clin 2014;64(2):83–103. DOI: 10.3322/caac.21219 [DOI] [PubMed] [Google Scholar]
- 5.Takakuwa T, Nakane T, Ohsawa M, et al. The first case of non-leukemic sarcoma composed of mixed-phenotype acute leukemia, B/myeloid, not otherwise specified. Intern Med 2018;57(8):1155–1158. DOI: 10.2169/internalmedicine.9144-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ghodke K, Tembhare P, Patkar N, Subramanian PG, Arora B, Gujral S. A rare extramedullary and extralymphoid presentation of mixed phenotypic blastic hematolymphoid neoplasm: A study of two cases. Indian J Med Paediatr Oncol 2017;38(3):394–397. DOI: 10.4103/ijmpo.ijmpo_94_16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Prentice AG, Smith AG, Bradstock KF. Mixed lymphoblastic-myelomonoblastic leukemia in treated Hodgkin’s disease. Blood 1980;56(1):129–133. DOI: 10.1182/blood.V56.1.129.129 [DOI] [PubMed] [Google Scholar]
- 8.Yu J, Li Y, Xing H, et al. Clinical characteristics and outcome of biphenotypic acute leukemia: 10 case reports and literature review. Cancer Manag Res 2019;11:9297–9306. DOI: 10.2147/CMAR.S226058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dores GM, Devesa SS, Curtis RE, Linet MS, Morton LM. Acute leukemia incidence and patient survival among children and adults in the United States, 2001-2007. Blood 2012;119(1):34–43. DOI: 10.1182/blood-2011-04-347872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Goyal G, Bartley AC, Patnaik MM, Litzow MR, Al-Kali A, Go RS. Clinical features and outcomes of extramedullary myeloid sarcoma in the United States: Analysis using a national data set. Blood Cancer J 2017;7(8):e592. DOI: 10.1038/bcj.2017.79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yamauchi K, Yasuda M. Comparison in treatments of nonleukemic granulocytic sarcoma: Report of two cases and a review of 72 cases in the literature. Cancer 2002;94(6):1739–1746. DOI: 10.1002/cncr.10399 [DOI] [PubMed] [Google Scholar]
- 12.Wilson CS, Medeiros LJ. Extramedullary manifestations of myeloid neoplasms . Am J Clin Pathol 2015;144(2):219–239. DOI: 10.1309/AJCPO58YWIBUBESX [DOI] [PubMed] [Google Scholar]
- 13.Orgel E, Alexander TB, Wood BL, et al. Mixed-phenotype acute leukemia: A cohort and consensus research strategy from the Children’s Oncology Group Acute Leukemia of Ambiguous Lineage Task Force. Cancer 2020;126(3):593–601. DOI: 10.1002/cncr.32552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Getta BM, Roshal M, Zheng J, et al. Allogeneic hematopoietic stem cell transplantation with myeloablative conditioning is associated with favorable outcomes in mixed phenotype acute leukemia. Biol Blood Marrow Transplant 2017;23(11):1879–1886. DOI: 10.1016/j.bbmt.2017.06.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee JH, Min YH, Chung CW, et al. Prognostic implications of the immunophenotype in biphenotypic acute leukemia. Leuk Lymphoma 2008;49(4):700–709. DOI: 10.1080/10428190701843247 [DOI] [PubMed] [Google Scholar]
- 16.Maruffi M, Sposto R, Oberley MJ, Kysh L, Orgel E. Therapy for children and adults with mixed phenotype acute leukemia: A systematic review and meta-analysis. Leukemia 2018;32(7):1515–1528. DOI: 10.1038/s41375-018-0058-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Munker R, Labopin M, Esteve J, Schmid C, Mohty M, Nagler A. Mixed phenotype acute leukemia: Outcomes with allogeneic stem cell transplantation. A retrospective study from the Acute Leukemia Working Party of the EBMT. Haematologica 2017;102(12):2134–2140. DOI: 10.3324/haematol.2017.174441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tian H, Xu Y, Liu L, et al. Comparison of outcomes in mixed phenotype acute leukemia patients treated with chemotherapy and stem cell transplantation versus chemotherapy alone. Leuk Res 2016;45:40–46. DOI: 10.1016/j.leukres.2016.04.002 [DOI] [PubMed] [Google Scholar]
- 19.Heesch S, Neumann M, Schwartz S, et al. Acute leukemias of ambiguous lineage in adults: Molecular and clinical characterization. Ann Hematol 2013;92(6):747–758. DOI: 10.1007/s00277-013-1694-4 [DOI] [PubMed] [Google Scholar]
- 20.Wolach O, Stone RM. How I treat mixed-phenotype acute leukemia. Blood 2015;125(16):2477–2485. DOI: 10.1182/blood-2014-10-551465 [DOI] [PubMed] [Google Scholar]