Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) or coronavirus disease 2019 (Covid-19), has uncontrollable effects on many organs. A great number of previously published scientific reports have revealed that patients with diabetes mellitus face a more severe form of Covid-19 with a higher death rate. Here we present the case of a 13-year-old unvaccinated boy who was admitted to an intensive care unit (ICU) with a history of fever, cough, dyspnea, throat pain, nausea, and confusion that progressed to lethargy after 24 h. On clinical examination, he was in a coma with Kussmaul’s breathing, and was anuric. His blood biochemical analysis demonstrated hyperglycemia, severe metabolic acidosis, kidney failure, electrolyte disturbances, and inflammation. Chest x-ray showed pneumonia and a pleural effusion. The results of the SARS-CoV-2 real-time polymerase chain reaction were positive. The patient was diagnosed with Covid-19-induced acute respiratory distress syndrome associated with multisystem inflammatory syndrome in children secondary to his acute respiratory failure, acute kidney injury, and new-onset type 1 diabetes mellitus with diabetic ketoacidosis. He was intubated for invasive mechanical ventilation and received a normal saline infusion and continuous insulin infusion (0.1 IU/kg/h) for the treatment of his diabetic ketoacidosis. He was also treated with methylprednisolone, aspirin, and heparin, and underwent continuous renal replacement therapy for acute renal failure for 9 days. The patient was discharged from ICU on day 16 and was followed up regularly as an outpatient with daily treatment, including subcutaneous insulin injection (30 IU/day) and a calcium channel blocker for hypertension (nifedipine 20 mg/day).
Keywords: Acute respiratory distress syndrome, ARDS, SARS-CoV-2, Covid-19, Continuous renal replacement therapy, Ketoacidosis, Diabetes mellitus
Key Summary Points
| Evidence-based diagnosis and treatment of acute respiratory distress syndrome (ARDS) associated with multiple systemic inflammatory syndrome in children (MIS-C) related to Covid-19 is still limited. |
| Ketoacidosis coma might be at the onset of Covid-19-induced diabetes in children associated with ARDS. |
| Early diagnosis of ARDS and ketoacidosis in children with Covid-19 is very important for starting the appropriate treatment. |
| Invasive mechanical ventilation combined with conventional treatment for MIS-C is crucial for patient recovery. |
Introduction
The coronavirus disease 2019 (Covid-19) pandemic, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has triggered an unprecedented global emergency. Covid-19 has uncontrollable effects on many organs. Since the start of this pandemic, a great number of published scientific reports have revealed that patients with diabetes mellitus face a more severe form of Covid-19 with a higher death rate [1]. Interestingly, “new-onset” hyperglycemia and “new-onset” diabetes mellitus are now identified as possible consequences of Covid-19, particularly among hospitalized children and adults [2, 3]. Moreover, clinical manifestations of Covid-19 are significantly different between age groups [4]. Adults develop respiratory symptoms, which can proceed to the most severe form of acute respiratory distress syndrome (ARDS), while children are largely immune to respiratory illness, but can develop multisystem inflammatory syndrome-in-children (MIS-C) [5, 6] that may rapidly progress to multiorgan failure and be life-threatening [7]. Children with MIS-C often present with persistent fever (100%), gastrointestinal symptoms (84%), cardiac abnormalities (75%), neurological symptoms (26–56%), respiratory symptoms (12–34%), and acute kidney injury (18.2%) [8]. Most reported cases illustrate that patients with MIS-C were admitted to the hospital with cardiac and gastrointestinal problems [8]. In this case report, we describe the clinical course and follow-up of a 13-year-old child with severe MIS-C secondary to Covid-19 with acute kidney injury, confusion, pneumonia, diabetic ketoacidosis (DKA), and new-onset type 1 diabetes mellitus.
Case Report
Here we present the case of a 13-year-old male who was admitted to an intensive care unit (ICU), referred to as Covid-19 Unit of Phu Chanh, Binh Duong General Hospital, Binh Duong Province, Vietnam, at the end of October 2021, with a 2-day medical history of fever, cough, dyspnea, throat pain, nausea, and confusion that progressed to lethargy after 24 h. He had no medical history of chronic diseases during his childhood. Before being ill, he did not have any medical history of diabetes or neurological disorders. He had not received the Covid-19 vaccine yet. There was no family history of diabetes; however, his father had been diagnosed with Covid-19 previously. In the ICU, the patient’s initial vital signs were a temperature of 38.5 °C, heart rate of 140 beats per minute, blood pressure of 90/40 mmHg, respiratory rate of 50 breaths per minute, and oxygen saturation of 99% via nasal cannula at 6 L/min. On clinical examination, he was in a coma, unresponsive to pain and stimulation, with Glasgow score of 7, positive pupillary reaction, Kussmaul’s breathing, and was anuric. He did not have any skin infection nor other source of infection. His weight was 50 kg, height 154 cm, and body mass index (BMI) 21.1 kg/m2 (50% percentile for age).
His blood biochemical analysis demonstrated hyperglycemia, severe metabolic acidosis, kidney failure, electrolyte disturbances, and inflammation (Table 1). Chest x-ray showed a mild pneumonia (Fig. 1). Abdominal ultrasound revealed free fluid in the abdomen and scrotum. Echocardiogram demonstrated normal cardiac muscle, valves, ejection fraction, and wall motion. Brain computed tomography (CT) scan was also normal. An electrocardiogram showed sinus tachycardia, with a rate of 150 beats per minute. The result of a SARS-CoV-2 real-time polymerase chain reaction (RT-PCR) was positive.
Table 1.
Laboratory data of patient before and after treatment MIS-C
| Variables | Reference range | Patient’s value Before treating MIS-C |
Patient’s value After treating MIS-C |
Difference (%) |
|---|---|---|---|---|
| White blood cells (109/L) | 4–11 | 23.7 | 8.7 | 92.59 ↓ |
| Neutrophil (%) | 45–75 | 75.3 | 65.7 | 13.62 ↓ |
| Lymphocyte (%) | 20–45 | 20.7 | 19.56 | 5.66 ↓ |
| Red blood cell (1012/L) | 3.8–6.3 | 3.49 | 3.84 | 9.55 ↑ |
| Hemoglobin (g/dL) | 12–18 | 10.2 | 15.3 | 40.00 ↑ |
| Hematocrit (%) | 37–52 | 31.6 | 44.6 | 34.12 ↑ |
| Platelet (109/L) | 140–500 | 197 | 289 | 37.86 ↑ |
| CRP (mg/dL) | < 1 | 3.01 | 1.37 | 74.89 ↓ |
| Pro-calcitonin (ng/ml) | < 0.05 | 2.99 | 0.095 | 187.68 ↓ |
| Ferritin (ng/ml) | 23.9–336.2 | 523.8 | 246.1 | 72.14 ↓ |
| Albumin (g/L) | 35–52 | 25.1 | 28.9 | 14.07 ↑ |
| Lactate (mmol/L) | 0.5–22 | 0.4 | 1.28 | 104.76 ↑ |
| LDH (U/L, 37 °C) | < 247 | 299.71 | 225.3 | 28.35 ↓ |
| Urea (mmol/L) | 2.8–7.2 | 11.13 | 18.8 | 51.25 ↑ |
| Creatinine (µmol/L) | 72–127 | 187.91 | 275.06 | 37.65 ↑ |
| eGFR (mL/min/1.73 m2) | > 60 | 46.29 | 73.21 | 45.05 ↑ |
| Glucose (mg/dL) | 70–110 | 296 | 142 | 70.32 ↓ |
| HbA1c (%Hb) | < 5.7 | 13.0 | 9.7 | 29.07 ↓ |
| pH | 7.35–7.45 | 6.82 | 7.44 | 8.70 ↑ |
| PaCO2 (mmHg) | 35–45 | 24 | 33 | 31.58 ↑ |
| PaO2 (mmHg) | 80–100 |
169 (with FiO2 of 30%) |
90 (with FiO2 of 21%) |
61.00 ↓ |
| HCO3− (mmol/L) | 18–23 | 3.9 | 22.4 | 140.68 ↑ |
| BE− (mmol/L) | −2 to +3 | −29.8 | −1.1 | 185.76 ↑ |
| Serum ketone (mg/dL) | Negative | 40 | 0 | 200 ↓ |
| D-dimer (µg/mL) | < 0.5 | 6.42 | 3.91 | 48.60 ↓ |
| Fibrinogen (g/L) | 1.5–4 | 5.9 | 2.14 | 93.53 ↓ |
| PT (%) | > 70 | 74 | 96 | 25.88 ↑ |
| APTT (s) | 10–16 | 33.5 | 28.6 | 15.78 ↓ |
| Sodium (mmol/L) | 135–145 | 134 | 137 | 2.21 ↑ |
| Potassium (mmol/L) | 3.5–5 | 2.9 | 4.5 | 43.24 ↑ |
| Calcium (mmol/L) | 1.1–1.6 | 1.17 | 1.05 | 10.81 ↓ |
| Magnesium (mmol/L) | 0.77–1.33 | 0.82 | 0.84 | 2.41 ↑ |
| AST (U/L, 37 °C) | 0–50 | 38.18 | 23.14 | 49.05 ↓ |
| ALT (U/L, 37 °C) | 0–50 | 15.8 | 12.78 | 21.13 ↓ |
| FT3 (pg/mL) | 2.30–3.90 | 2.24 | – | – |
| FT4 (ng/mL) | 0.61–1.12 | 0.63 | – | – |
| TSH (µIU/mL) | 0.32–5.33 | 0.28 | – | – |
Data presented as number and %
APTT activated partial thromboplastin time; AST aspartate aminotransferase; ALT alanine aminotransferase; CRP C-reactive protein; eGFR estimated glomerular filtration rate; FiO2 fraction of inspired oxygen; FT3 free triiodothyronine; FT4 free thyroxine; HbA1C hemoglobin A1c; LDH lactate dehydrogenase; MIS-C multisystem inflammatory syndrome in children; PT prothrombin time; TSH thyroid stimulating hormone
*With FiO2
Fig. 1.
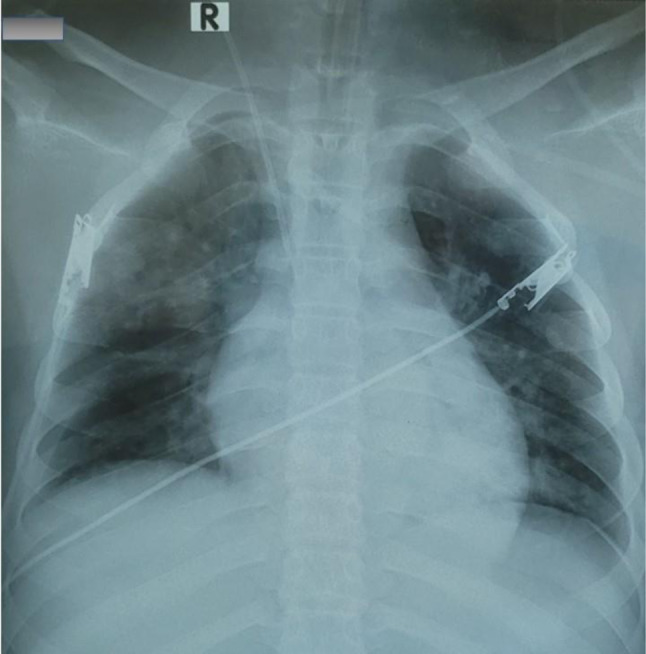
Chest x-ray imaging with pneumonia at admission. The presence of diffused and bilateral infiltration in lung parenchyma of reported patient with acute respiratory distress syndrome
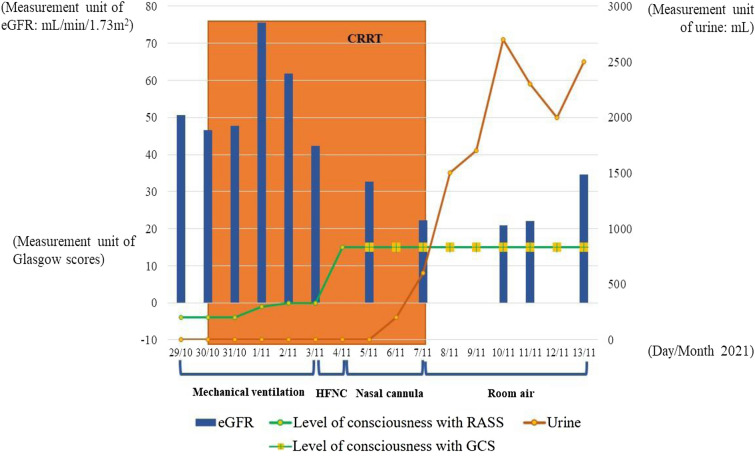
The patient was diagnosed with Covid-19-induced acute respiratory distress syndrome associated with MIS-C secondary to his acute respiratory failure, acute kidney injury (AKI), suspected brain injury, and new-onset type 1 diabetes mellitus with DKA secondary to β-cell injury. He was intubated for invasive mechanical ventilation to assure respiratory function with the following parameters: ventilatory mode, pressure control; FiO2, 55%; positive end expiratory pressure (PEEP) 14 cm H2O; respiratory rate, 20 times per minute. Midazolam and rocuronium were prescribed for preventing mechanical ventilatory desynchrony. The patient received a normal saline infusion (3000 mL/24 h) combined with potassium chloride 10% (40 mL/24 h) for 2 days and continuous insulin infusion (0.1 IU/kg/h) for the treatment of DKA. He was also treated with pulse doses of methylprednisolone therapy for 3 days, and then changed to methylprednisolone 2 mg/kg/day for 5 days as recommended locally for treating Covid-19. In addition, he received aspirin for his MIS-C, heparin for preventing coagulation, and an antibiotic for preventing ventilator-associated pneumonia. He also underwent continuous renal replacement therapy (CRRT) for acute renal failure for 9 days. On day 6, the patient was woken and extubated from invasive mechanical ventilation, transferred to high-flow nasal cannula, followed by conventional oxygen therapy over the following days. His urine increased from 50 ml per day from day 8 to over 2000 ml per day on day 12. By day 10, his vital signs normalized with a Glasgow score of 14, body temperature of 37 °C, SpO2 of 97% on room air; his hemodynamic parameters stabilized; and his biochemical blood testing was nearly normal, except for his renal functional parameters (Table 1 and Fig. 2). The patient was discharged from the ICU on day 16 and was followed up regularly as an outpatient with daily treatment, including subcutaneous insulin injection (30 IU/day) and a calcium channel blocker for hypertension (nifedipine 20 mg/day). Currently, the patient has been in a good health status and seen regularly in Outpatients Department of Binh Duong General Hospital every month for his new onset of type 1 diabetes during post-Covid-19 period. Written informed consent was obtained from the patient’s parents for the publication of clinical details.
Fig. 2.
Laboratory, clinical findings, and treatment trends during hospitalization. Modification of urine volume, glomerular filtration, conscious status, and treatment of acute respiratory distress syndrome during patients’ hospitalization. eGFR estimated glomerular filtration rate; RASS Richmond agitation sedation scale; GCS Glasgow coma scale; CRRT continuous renal replacement therapy; HFNC high-flow nasal cannula
Discussion
The Covid-19 pandemic poses a significant risk to both adults and children. Although children were initially considered less likely to be affected by SARS-CoV-2, MIS-C is a recently recognized manifestation in children associated with coronavirus SARS-CoV-2 infection with a proven higher mortality rate [9]. The US Centers for Disease Control and Prevention (CDC) has provided a case definition of MIS-C, which includes patients under 21 years of age with fever (> 38.0 °C for ≥ 24 h, or report of subjective fever lasting ≥ 24 h), laboratory evidence of inflammation (with one or more of the following: elevated C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), fibrinogen, procalcitonin, D-dimer, ferritin, lactic acid dehydrogenase (LDH), or interleukin 6 (IL- 6), elevated neutrophils, reduced lymphocytes, and low albumin), severe illness needing hospitalization, and involvement of two or more organ systems (cardiac, renal, respiratory, hematologic, gastrointestinal, dermatologic, or neurological), with a positive test for SARS-CoV-2 indicating current or recent infection or Covid-19 exposure; and no other alternative plausible diagnoses [10]. Our patient had a 24 h fever and multisystem illness (lung, kidney, suspected brain injury, and injured β-cells-induced diabetes), as well as elevated inflammatory markers such as increased neutrophils and CRP, and decreased albumin, consistent with the US CDC case definition of MIS-C. In addition, RT-PCR testing to detect SARS-CoV-2 infection was positive.
Published data have illustrated that children with Covid-19 can develop a life-threatening multisystem inflammatory syndrome, but rarely exhibit severe respiratory symptoms, and that most reported patients were admitted to the hospital for symptoms including diarrhea, vomiting, abdominal pain, ventricular dysfunction, or coronary artery dilation [8, 11]. However, in our case, the patient was admitted to the hospital with coma, DKA, and clinical signs of acute kidney failure, including anuria, and severe edema of the legs, hands, eyes, abdomen, and a pleural effusion. Previous studies demonstrated that most patients developed AKI on the first day of hospitalization and the pathophysiology of Covid-19-induced AKI might be related to local and systemic inflammatory and immune responses, endothelial injury, activation of coagulation pathway, and the renin–angiotensin system, or alternatively direct viral infection of kidney tissue [12, 13]. Furthermore, severe disease, acute lung injury, obesity, lower median left ventricular ejection fraction, systolic dysfunction, inflammation, and exposure to nephrotoxins also contribute to Covid-19-related AKI [14]. In our patient with acute Covid-19 and MIS-C, AKI appeared to be related to inflammation, as evidenced by a high neutrophil/lymphocyte rate, and increased inflammatory biomarker levels (Table 1).
Obviously, DKA is a serious complication of diabetes that can be life-threatening, and unfortunately we suspect delays in the diagnosis and care of DKA has not been uncommon during the Covid-19 pandemic. In our report, the patient was diagnosed with DKA on the basis of the typical clinical symptoms, such as Kussmaul’s breathing, loss of consciousness, biochemical features [blood glucose higher than ≥ 200 mg/dL, arterial pH of < 7.3 and bicarbonate (HCO3) level < 15 mmol/L] and ketonemia (Table 1). DKA is serious complication due to a lack of total or partial insulin deficiency that is most commonly encountered in patients with type 1 diabetes mellitus [13]. Type 1 diabetes may develop secondary to autoimmune destruction of insulin-producing β-cells in the pancreas [15, 16]. In his medical history, our patient did not have any health problems before being infected by Covid-19. We attributed his new-onset DKA and type 1 diabetes to Covid-19-induced autoimmune pancreatitis, leading to the destruction of β-cells. Although genetic predisposition is important in the development of type 1 diabetes mellitus, infections are also important in the pathogenesis of autoimmune type 1 diabetes mellitus. It is speculated that antibodies against SARS-CoV-2 may trigger an inflammatory process that impacts β-cell function and potentially leads to hastened β-cell death and the onset of diabetes [17].
In our case report, the patient was in a comatose state that appears to be secondary to DKA-induced cerebral edema, rather than brain injury from the virus. Because of his mental status, he required invasive mechanical ventilation for airway protection. Although the possibility of SARS-CoV-2 infecting the central nervous system directly is still disputed, owing to the very low level of angiotensin-converting enzyme 2 (ACE-2) expression in the brain, human brain gene expression and immunohistochemistry have revealed that SARS-CoV-2 could infiltrate into the hypothalamus [18]. Interestingly, neuroimaging in children with neurologic manifestations has demonstrated only a low prevalence of acute encephalitis in MIS-C associated with Covid-19 [19, 20]. Our patient had a normal brain CT scan, and a total recovery from his coma without any neurologic sequela, suggesting his mental status changes were from DKA. Furthermore, low levels of serum albumin, calcium, and potassium were also found in our patient with Covid-19-induced MIS-C. It has been suggested that increased capillary permeability, secondary to SARS-CoV-2-induced MIS-C, might be responsible for the reduction of serum albumin, and thus calcium [14]. In addition, the consequence of Covid-19’s protein S binding to ACE-2 receptor might reduce angiotensin-2 degradation, leading to an increase of aldosterone, causing renal potassium loss, and finally leading to hypokalemia [17].
Although the first cases of multisystem inflammatory syndrome in children (MIS-C) and adults (MIS-A) have been reported previously [21, 22], the prevalence of MIS-C and MIC-A related to Covid-19 has not been well established [23]. These reports showed that there was a diversity of clinical manifestation of MIS across age groups: MIS-C with mucocutaneous lesions was more frequent in young children and myocarditis and gastrointestinal symptoms were more common in older children, while MIS-A usually had gastrointestinal complications [21, 22]. Until now, ARDS due to Covid-19 associated with MIS-C or MIS-A and diabetes has still been rare.
The treatment of MIS-C is often difficult and has required combination therapies. In our case report, the patient underwent continuous renal replacement therapy, owing to ARI with anuria, metabolic acidosis, and uremia. In addition, the patient was given a pulse dose of methylprednisolone in association with aspirin for his MIS-C. Although intravenous immune globulin has been recommended in some studies, we did not use it in our patient since the beneficial effect of intravenous immune globulin in Covid-19-induced MIS-C has not been clearly demonstrated [24]. In our case report, the treatment of DKA with fluid resuscitation using 0.9% saline, 0.45% saline, infused insulin, and correction of the electrolyte disturbances was performed as per recommendations [25]. As noted above, Covid-19-induced MIS-C with ARI, DKA, and new onset of type 1 diabetes has rarely been reported in children; however, making the diagnosis and instituting aggressive and individualized therapy can prevent loss of life.
The patient was discharged from hospital after 16 days in the ICU fully conscious and clinically stable, with full recovery from his respiratory failure, and with improved inflammatory biomarkers. His renal function has not fully recovered, but improved, and he has continued to require treatment of his new-onset diabetes with insulin, and also treatment of his hypertension with a calcium channel blocker. Six months after his discharge, the patient has returned to normal life and been followed in the long-Covid-19 clinic for his diabetes and hypertension.
Finally, the main limitation of this case report is related to a lack of clarity to effectively estimate a lag following initial Covid-19 infection with the presumably autoimmune MIS-C syndrome as demonstrated previously [26]. Although the diagnosis of MIS-C in our case report was based significantly on clinical manifestations and inflammatory biomarkers, the alternative diagnosis in favor of acute Covid-19 is still debatable.
Conclusion
MIS-C related to Covid-19 with ARDS remains an uncommon clinical manifestation, especially when it is complicated with COVID-19 new-onset type 1 diabetes. Our case report is the first case in Vietnam with the constellation of Covid-19-induced multisystemic inflammatory syndrome, respiratory failure, acute kidney injury, and new-onset diabetes in an unvaccinated child, despite more than 1.8 million people (0.8% children) having contracted Covid-19 in Vietnam. Fortunately, the accurate diagnosis and aggressive treatment saved the life of our patient. Finally, in addition to lung damage from Covid-19, the mechanism by which SARS-CoV-2 (Covid-19) can damage the kidney and pancreatic β-cells causing the long-term consequence of renal failure and diabetes needs to be further explored.
Acknowledgements
Funding
No funding or sponsorship was received for this study or publication of this article.
Authors Contributions
Sy Duong-Quy, Duc Huynh-Truong-Anh, Nhung Le-Thi-Hong, Tap Le-Van, Sa Le-Thi-Kim, Tien Nguyen-Quang, Thanh Nguyen-Thi-Kim, Ngan Nguyen-Phuong, Thanh Nguyen-Chi, Tinh Nguyen-Van: data collection. Sy Duong-Quy, Van Duong-Thi-Thanh, Dung Nguyen-Tien, Carine Ngo, Timothy Craig: manuscript drafting. Sy Duong-Quy, Duc Huynh-Truong-Anh, Carine Ngo, and Timothy Craig: manuscript approving.
Disclosures
Sy Duong-Quy, Duc Huynh-Truong-Anh, Nhung Le-Thi-Hong, Tap Le-Van, Sa Le-Thi-Kim, Tien Nguyen-Quang, Thanh Nguyen-Thi-Kim, Ngan Nguyen-Phuong, Thanh Nguyen-Chi, Tinh Nguyen-Van, Van Duong-Thi-Thanh, Dung Nguyen-Tien, Carine Ngo, and Timothy Craig have nothing to disclose.
Compliance with Ethics Guidelines
Written informed consent was obtained from the patient’s parents for the publication of clinical details.
Data Availability
Patient data and the clinical course were retrieved from the hospital medical records. They are available from the corresponding author upon reasonable request.
References
- 1.Caballero AE, Ceriello A, Misra A, et al. COVID-19 in people living with diabetes: an international consensus. J Diabetes Complications. 2020;34(9):107671. doi: 10.1016/j.jdiacomp.2020.107671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Khunti K, Prato SD, Mathieu C, et al. COVID-19, hyperglycemia, and new-onset diabetes. Diabetes Care. 2021;44(12):2645–2655. doi: 10.2337/dc21-1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rubino F, Amiel SA, Zimmet P, et al. New-onset diabetes in covid-19. N Engl J Med. 2020;383(8):789–790. doi: 10.1056/NEJMc2018688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease control and prevention. JAMA. 2020;323(13):1239–1242. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 5.Cheung EW, Zachariah P, Gorelik M, et al. Multisystem inflammatory syndrome related to COVID-19 in previously healthy children and adolescents in New York City. JAMA. 2020;324(3):294–296. doi: 10.1001/jama.2020.10374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Feldstein LR, Rose EB, Horwitz SM, et al. Multisystem inflammatory syndrome in U.S. children and adolescents. N Engl J Med. 2020;383:334–346. doi: 10.1056/NEJMoa2021680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Weisberg SP, Connors TJ, Zhu Y, et al. Distinct antibody responses to SARS-CoV-2 in children and adults across the COVID-19 clinical spectrum. Nat Immunol. 2021;22:25–31. doi: 10.1038/s41590-020-00826-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tang Y, Li W, Baskota M, et al. Multisystem inflammatory syndrome in children during the coronavirus disease 2019 (COVID-19) pandemic: a systematic review of published case studies. Transl Pediatr. 2021;10(1):121–135. doi: 10.21037/tp-20-188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Acevedo L, Piñeres-Olave BE, Niño-Serna LF, et al. Mortality and clinical characteristics of multisystem inflammatory syndrome in children (MIS-C) associated with covid-19 in critically ill patients: an observational multicenter study (MISCO study) BMC Pediatr. 2021;21:1–12. doi: 10.1186/s12887-021-02974-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Centers for Disease Control and Prevention. MIS-A case definition (2021). https://www.cdc.gov/mis/mis-a/hcp.html. [Accessed 6 Dec 2021]
- 11.Wu EY, Campbell MJ. Cardiac manifestations of multisystem inflammatory syndrome in children (MIS-C) following COVID-19. Curr Cardiol Rep. 2021;23(11):168. doi: 10.1007/s11886-021-01602-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ahmadian E, Khatibi SMH, Soofiyani SR, et al. COVID-19 and kidney injury: pathophysiology and molecular mechanisms. Rev Med Virol. 2020;31(3):e2176. doi: 10.1002/rmv.2176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lipton M, Mahajan R, Kavanagh C, et al. AKI in COVID-19–associated multisystem inflammatory syndrome in children (MIS-C) Kidney. 2021;2(4):611–618. doi: 10.34067/KID.0005372020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Basalely A, Gurusinghe S, Schneider J, et al. Acute kidney injury in pediatric patients hospitalized with acute COVID-19 and multisystem inflammatory syndrome in children associated with COVID-19. Kidney Int. 2021;100(1):138–145. doi: 10.1016/j.kint.2021.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lizzo JM, Goyal A, Gupta V. Adult diabetic ketoacidosis. https://www.ncbi.nlm.nih.gov/books/NBK560723/. [Accessed 30 Dec 2021] [PubMed]
- 16.Yang J-K, Lin S-S, Ji X-J, et al. Binding of SARS coronavirus to its receptor damages islets and causes acute diabetes. Acta Diabetol. 2010;47(3):193–199. doi: 10.1007/s00592-009-0109-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Unsworth R, Wallace S, Oliver NS, et al. New-onset type 1 diabetes in children during COVID-19: multicenter regional findings in the U.K. Diabetes Care. 2020;43(11):170–171. doi: 10.2337/dc20-1551. [DOI] [PubMed] [Google Scholar]
- 18.Nampoothiri S, Sauve F, Sauve F, et al. The hypothalamus as a hub for SARS-CoV-2 brain infection and pathogenesis. BioRxiv. 2021 doi: 10.1101/2020.06.08.139329. [DOI] [Google Scholar]
- 19.Olivotto S, Basso L, Lavatelli R, et al. Acute encephalitis in pediatric multisystem inflammatory syndrome associated with COVID-19. Eur J Paediatr Neurol. 2021;34:84–90. doi: 10.1016/j.ejpn.2021.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Orman G, Desai NK, Kralik SF, et al. Neuroimaging offers low yield in children positive for SARS-CoV-2. Am J Neuroradiol. 2021;42(5):951–954. doi: 10.3174/ajnr.A7022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dufort EM, Koumans EH, Chow EJ, New York State and Centers for Disease Control and Prevention Multisystem Inflammatory Syndrome in Children Investigation Team et al. Multisystem inflammatory syndrome in children in New York State. N Engl J Med. 2020;383(4):347–358. doi: 10.1056/NEJMoa2021756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Davogustto GE, Clark DE, Hardison E, et al. Characteristics associated with multisystem inflammatory syndrome among adults with SARS-CoV-2 infection. JAMA Netw Open. 2021;4(5):e2110323. doi: 10.1001/jamanetworkopen.2021.10323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Patel P, DeCuir J, Abrams J, Campbell AP, Godfred-Cato S, Belay ED. Clinical characteristics of multisystem inflammatory syndrome in adults: a systematic review. JAMA Netw Open. 2021;4(9):e2126456. doi: 10.1001/jamanetworkopen.2021.26456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Licciardi F, Baldini L, Dellepiane M, et al. MIS-C treatment: is IVIG always necessary? Front Pediatr. 2021;9:753123. doi: 10.3389/fped.2021.753123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lawrence SE. Diagnosis and treatment of diabetic ketoacidosis in children and adolescents. Paediatr Child Health. 2005;10(1):21–24. [PMC free article] [PubMed] [Google Scholar]
- 26.Kundu A, Maji S, Kumar S, Bhattacharya S, Chakraborty P, Sarkar J. Clinical aspects and presumed etiology of multisystem inflammatory syndrome in children (MIS-C): a review. Clin Epidemiol Glob Health. 2022;14:100966. doi: 10.1016/j.cegh.2022.100966. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Patient data and the clinical course were retrieved from the hospital medical records. They are available from the corresponding author upon reasonable request.