Abstract
Background
Perinatal stroke injures motor regions of the brain, compromising movement for life. Early, intensive, active interventions for the upper extremity are efficacious, but interventions for the lower extremity remain understudied.
Objective
To determine the feasibility and potential efficacy of ELEVATE—Engaging the Lower Extremity Via Active Therapy Early—on gross motor function.
Methods
We conducted a single-blind, two-arm, randomized controlled trial (RCT), with the Immediate Group receiving the intervention while the Delay Group served as a 3-month waitlist control. A separate cohort living beyond commuting distance was trained by their parents with guidance from physical therapists. Participants were 8 months to 3 years old, with MRI-confirmed perinatal ischemic stroke and early signs of hemiparesis. The intervention was play-based, focused on weight-bearing, balance and walking for 1 hour/day, 4 days/week for 12 weeks. The primary outcome was the Gross Motor Function Measure-66 (GMFM-66). Secondary outcomes included steps and gait analyses. Final follow-up occurred at age 4.
Results
Thirty-four children participated (25 RCT, 9 Parent-trained). The improvement in GMFM-66 over 12 weeks was greater for the Immediate than the Delay Group in the RCT (average change 3.4 units higher) and greater in younger children. Average step counts reached 1370-3750 steps/session in the last week of training for all children. Parent-trained children also improved but with greater variability.
Conclusions
Early, activity-intensive lower extremity therapy for young children with perinatal stroke is feasible and improves gross motor function in the short term. Longer term improvement may require additional bouts of intervention.
Clinical trial registration
This study was registered at ClinicalTrials.gov (NCT01773369).
Keywords: neuroplasticity, early intervention, cerebral palsy, motor skill training, intensive training, exercise
Introduction
Perinatal stroke is a cerebrovascular event that occurs between gestational age of 20 weeks and 28 days postnatal, and has an incidence between one in 1600 to 2300 live births.1,2 It is the leading cause of hemiparetic cerebral palsy (CP), which may involve weakness, spasticity and impaired coordination in the affected upper and lower extremity. 3 The life-long gross motor impairments contribute to long-term musculoskeletal complications, impaired gait, reduced physical activity and participation.4,5
Intensive, active approaches to rehabilitation have been effective for improving upper extremity function (e.g., constraint-induced movement therapy [CIMT] and bimanual training, reviewed in 6-8). In contrast, active treatment approaches for the lower extremity are limited for young children with CP, 7 although some studies combining upper and lower extremity training show promise9,10 or are underway. 11 Optimizing lower extremity function is especially important now because of the recent reduction in severity of CP among developed countries, resulting in more children with the potential to walk. 12
Current clinical practice for lower extremity function in young children with CP is often passive in nature, typically waiting until clinical signs appear, then focusing on static stretching, the traditional Neural Developmental Therapy (NDT), bracing with ankle-foot orthoses and botulinum toxin injection of spastic muscles,13-15 a “wait-and-see”approach. 16 Yet targeted walking training in school-aged children improves walking performance in children with CP.17,18 The passive, infrequent and delayed approach to treatment of the lower extremity for young children with hemiparetic CP is in contrast to evidence from animal models of early brain injury, which demonstrates the importance of early, intensive rehabilitation.
Inactivating the primary motor cortex in kittens during a critical period of development impairs the development of motor circuits and motor function.19,20 Initiating motor training of the affected limb while kittens are young improves motor function and the integrity of motor circuits, whereas training at an older age is less effective. 21 Critical periods of lower extremity motor development in the human may occur before the age of 2 years because post-mortem studies have shown mature myelin on the corticospinal tract at the lumbar neurological level around this age. 22 Since mature myelin is associated with reduced neuroplasticity, 23 we suggest that plasticity would be greatest prior to the emergence of mature myelin. Therapeutic approaches to enhance developmental neuroplasticity of children with perinatal stroke have been reviewed recently. 24 In a separate pilot study with no participant overlap with this study, we showed that intensive activity-based rehabilitation for the lower extremity in children with hemiparesis under the age of 2 resulted in large improvements in walking. 25
We hypothesized that early, intensive, child-initiated therapy for the lower extremity in children with perinatal stroke would result in greater improvements in motor function than usual care. Here, we focus on the changes in gross motor function.
Methods
The protocol of the study has been described 26 and will be recounted briefly here. The study was approved by the Health Research Ethics Board at the University of Alberta (Pro00032297). Written consent was provided by parents/guardians of all participants.
Participants
Inclusion Criteria
1. Hemiparesis with MRI-confirmed perinatal stroke, categorized as neonatal arterial ischemic stroke (NAIS), arterial presumed perinatal ischemic stroke (APPIS), or periventricular venous infarct (PVI). 27
2. Born at ≥32 weeks gestation, with age at entry to study between 8 months and 3.0 years old.
3. No other neurological disorders.
4. Parent/guardian able to attend all tests and training.
5. Written, informed consent from parent/guardian.
We focus on ischemic, perinatal stroke to reduce confounds associated with other causes of hemiparetic CP. The age range was based on our estimate that an 8-month-old would be able to participate in the intervention for an hour, and encompasses a time of rapid gross motor development.
Exclusion Criteria
1. Extensive brain injuries beyond unilateral ischemic stroke.
2. Musculoskeletal, cognitive or behavioral impairments that preclude participation in the protocol.
3. Unstable epileptic seizures within the past 6 months or taking anti-epileptic medication.
4. Any contraindications to transcranial magnetic stimulation (TMS) because TMS was an outcome measure.
5. Botulinum toxin injection or surgery in the lower extremity in the past 6 months.
Clinical partners identified participants and potentially suitable participants were screened in-person by a research physical therapist (PT). Evidence of perinatal stroke on MRI was confirmed by a pediatric neurologist (AK).
Trial Design
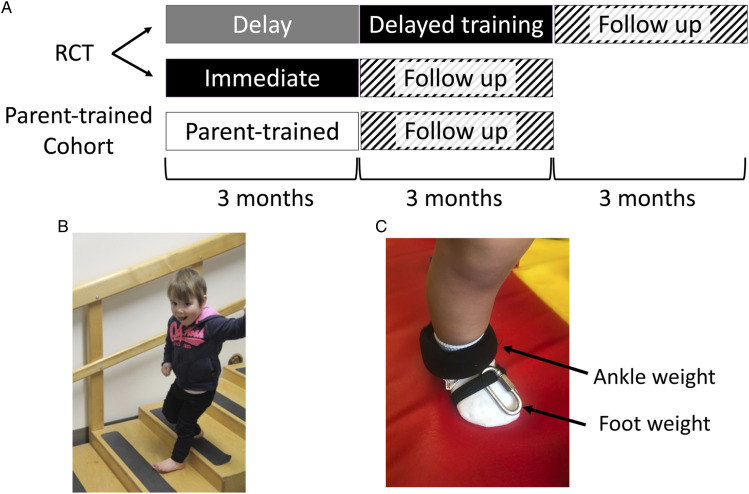
A waitlist control, single-blind, pilot randomized controlled trial (RCT) was conducted at 2 centers (Edmonton and Calgary, Canada). The experimental design is represented in Figure 1A. Children were stratified by city, then randomized to train immediately (Immediate Group) or delay training for 3 months (Delay Group). Children in the Delay group did not undergo an alternative intervention, but served as a waitlist control by continuing with their usual care for the 3 month delay period. Usual care in the participants in this study typically consisted of 1 hour sessions of occupational and/or physical therapy approximately 1–2 times/month. Children in an additional parallel cohort who lived beyond commuting distance were trained immediately by their parent or guardian (Parent-trained Group) with guidance from a PT. These children were not part of the RCT but were included to increase accessibility and to assess the feasibility of the intervention when delivered by a parent. Data from this cohort are descriptive and were assessed separately from the RCT. Final follow-up occurred within 3 months of each child’s fourth birthday.
Figure 1.
Experimental Design and MethodsA) A schematic diagram of the experimental design indicating the RCT component (top two rows) and the Parent-trained Group (third row). B) A child demonstrating an example of an activity performed during training. C) Ankle weights were fastened around the lower leg for added strength training, and ¼" chain links were used on the foot to strengthen dorsiflexors. Abbreviations: RCT, randomized controlled trial.
Randomization
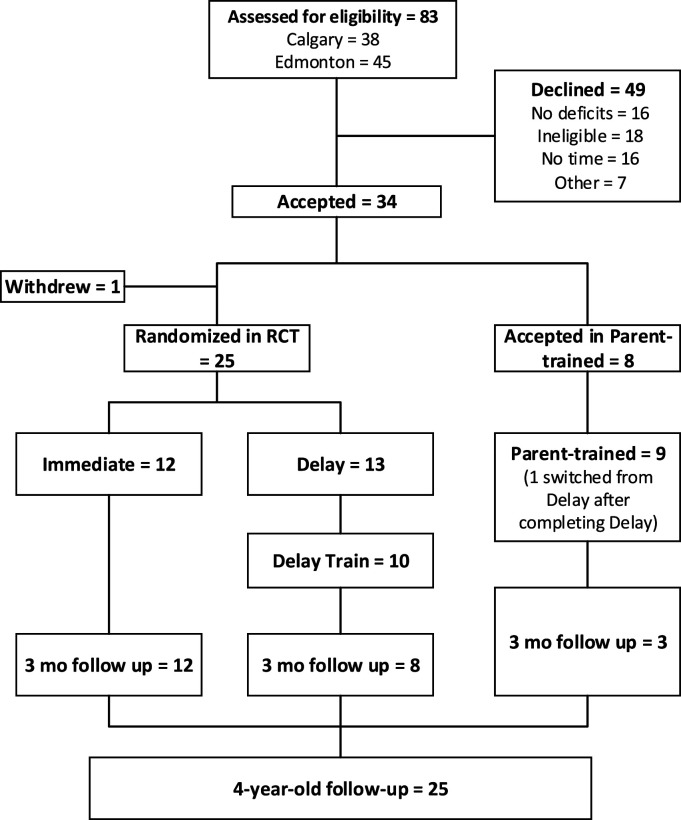
Participant flow is shown in a CONSORT diagram (Figure 2). Participants completed all baseline testing before being randomized into either Immediate or Delayed-training Groups in a 1:1 allocation ratio. A biostatistician generated the group allocation sequence using a computer-generated permuted block design with a block size of 2 to 8. Group assignment was concealed in sequentially numbered, sealed envelopes.
Figure 2.
CONSORT Flow DiagramEligible children entered the randomized controlled trial (RCT) or the Parent-trained cohort if they lived beyond commuting distance. Children in the RCT were allocated to either train immediately (Immediate Group) or delay training for 3 months (Delay Group). The Delay Group served as a usual care control and were trained after the delay period. Children were followed for 3 months after training and reassessed at 4 years old.
Intervention
The ELEVATE (Engaging the Lower Extremity Via Active Therapy Early) intervention was 1 hour/day, 4 days/week for 12 weeks. The intervention was delivered by 1 of 2 PTs (DL or KB) in a laboratory at the University of Alberta (Edmonton) or a satellite campus in Calgary. In the Parent-trained cohort, the intervention was delivered by the child’s parent in their home, guided by 1 of the 2 therapists. This included weekly support by phone and monthly in-person collaborative training session between the therapist and parent when children returned to the laboratory for assessments. If parents requested more frequent in-person training, it was accommodated.
The intervention was intensive, child-initiated, play-based movement of the lower extremities, as described in the protocol. 26 The intervention occurred primarily over ground on various surfaces, including linoleum, carpet, mats, grass, and sidewalks. Children occasionally walked on a treadmill for parts of the training sessions if that was their preference. Children wore soft-soled slippers and no orthotics while training to enhance the use of muscles in the feet and around the ankle. Activities included ascending and descending stairs and ramps, walking on stable and unstable surfaces, stepping over obstacles, balancing in standing, kicking, and squatting to pick up items (Figure 1B). Manual support from the therapist was provided as needed to facilitate 60 minutes of child-initiated activity. To increase exercise intensity and to augment movement error, 28 weights were placed on the dorsum of the foot and the ankle of the affected leg of children with sufficient endurance to stay active for 50 to 60 minutes. Weights were added to the affected foot and ankle, which makes a child’s gait more asymmetric and the amount of weight was increased when the child’s gait became more symmetrical. Children with cerebral palsy adapt to weights in a similar way to uninjured children. 28 Using weights allowed for specific strengthening of the musculature in the affected lower extremity. Commercially available weights in increments of 110 g were used for the ankle, and one-quarter-inch chain links, ∼20 g each, were affixed to the dorsum of the foot with elasticized fabric (Figure 1C). Parents of the Parent-trained children were provided with the weights.
Outcomes
Gross Motor Function: The primary outcome measure was the Gross Motor Function Measure-66 (GMFM-66). 29 GMFM-66 is a criterion-referenced observational measure to assess change in gross motor function in children with CP. Sixty-six tasks spanning 5 dimensions were scored, including lying and rolling, sitting, crawling and kneeling, standing, and walking/running/jumping. Reliability, validity, and responsiveness to change have been established for children ≥6 months of age.30,31 GMFM-66 assessments were performed by pediatric PTs who were blinded to the child’s group assignment. GMFM-66 assessments were performed twice at baseline, separated by a week and monthly thereafter (throughout the study period, see Figure 1A). The 2 baseline assessments were averaged for each participant.
Step Counts: Step counts were assessed using the StepWatch activity monitor (Orthocare Innovations, Seattle, WA). Full-day step counts were recorded at the beginning, middle, and end of each study period (delay, training, and follow-up) as a measure of activity outside training sessions. Parents placed step counters on the ankle of the child’s affected leg for waking hours (excluding water activities) for a target of 6 days and documented activities and time of day in a log. Counts from a minimum of 2 full days of recording were averaged. Step counts were also measured using the StepWatch during every training session to quantify the amount of activity on the affected lower extremity for both therapist-trained and parent-trained children.
Kinematics of Walking: Secondary outcomes included assessment of gait while walking on a treadmill. Experimenters for these outcomes were not blinded. Children walked with soft-soled slippers, played with toys on a table in front of them and received manual support on their lateral thorax, as needed. Gait assessments were performed once at baseline and monthly thereafter throughout the study.
A custom-built split-belt treadmill, with a force plate under each belt, was used in Edmonton. A commercial treadmill (TR1200B; LifeSpan, Salt Lake City, UT) without force plates was used in Calgary. Walking kinematics were captured either with the 3-D Investigator (NDI, Waterloo, Ontario, Canada) in Edmonton or the Motion Analysis System using 12 Eagle-4 cameras (Santa Rosa, CA, USA) in Calgary. Markers were placed at the top of the iliac crest, greater trochanter, knee joint line, lateral malleolus, and head of the fifth metatarsal, bilaterally. Customized MatLab script calculated toe clearance (maximum vertical height of the toe marker during the swing phase) and weight-bearing (mean vertical force during stance phase).
In each assessment, children walked for 2 trials of approximately 1 minute each at 3 different speeds, for a total of 6 trials per assessment. We focused on the speed that included at least 10 steps at all assessments throughout the study, typically the median speed of .4 m/s or .6 m/s.
We quantified the symmetry of toe clearance using the following formula
| (1) |
where is symmetry of toe clearance and and are the vertical toe clearance of the unaffected and affected foot, respectively. The percent of a child’s weight borne by the lower extremities in walking was calculated as
| (2) |
where is the percent of body weight supported on their feet, and are the average vertical forces during the stance phase from the right and left force plates, respectively, and BW is total body weight as measured on a scale.
Sample Size
Pilot data from 4 children suggested that the effect size of the primary outcome measure (GMFM-66) was 1.1. 25 This was based on a GMFM-66 change of 5.5 points, and the predicted change over 3 months without intervention of approximately 3.5, for children with a Gross Motor Function Classification System (GMFCS) level I or II at approximately 1.5 years old. The standard deviation of the change score was estimated to be 2, resulting in a sample size of approximately 16 per group. Formal stopping guidelines were not established because this study evaluated an intervention that did not present increased risk beyond usual care. Clinical decision-making was used to screen for potential overuse injuries and prevent injurious falls throughout training.
Blinding
Assessors of the primary outcome measure, GMFM-66, were blinded to group allocation but it was not possible to blind treating therapists or parents to group allocation.
Statistical Methods
Statistical analyses for primary and secondary outcome measures were performed using SAS Ver. 9.4 (SAS Institute Inc., Cary, NC). A multivariable linear regression model was used to examine the effect of age and group allocation on change in GMFM score and toe clearance from baseline to 3 months. All other analyses were conducted using SPSS 20.0 software, including descriptive statistics (mean, standard deviation [SD], and standard error of the mean [SEM]). For participant characteristics, one-way ANOVAs were used to analyze group differences for continuous variables, whereas chi squared tests were used to analyze group differences for categorical data.
Results
Recruitment
Recruitment began in 2012 and ended in March, 2018. The recruitment ended at the end of the funding period, at which point we were approaching but below the targeted sample size. The final 3-month follow-up occurred in July 2019, and the final four-year-old follow-up was completed in June 2021.
Participant Flow
The CONSORT diagram (Figure 2), shows 83 potential participants were referred (45 in Edmonton, 38 in Calgary) and screened by a research PT. Twenty-four did not meet inclusion criteria, the most common reasons being no deficits observed (n = 14), other neurological complications (n = 3) or not ischemic perinatal stroke (n = 3). A further 25 potential participants declined to enroll in the study, with the most common reason reported by parents being a prohibitive time commitment (n = 16). Thirty-four children (21 in Edmonton, 13 in Calgary) were accepted into the study. One participant in the Immediate Group withdrew before the intervention began due to the child’s unwillingness to participate in assessments. This participant was excluded from the analysis because no data were collected. The total number of participants included in the Delay, Immediate, and Parent-trained Groups in the first 3-month period was 13, 12, and 9, respectively. One child withdrew following the 3-month delay because the family moved out of the province. One child in the Immediate Group missed the assessments at the end of training due to a family issue unrelated to the study so their data were not included in the statistical analysis, but the child returned for 3-month follow-up assessments. Ten children in the Delay Group participated in training following the delay period. One child switched from the Delay Group to the Parent-trained Group after completing the delay period because the family moved out of town. No participants in the Immediate or Parent-trained groups withdrew during the intervention phase. All but 1 child in the Delay group completed the 3 months of training following the control period. All participants in the Immediate Group completed monthly follow-up for 3 months following training. The study design did not initially include 3-month follow-up for the Delay and Parent-trained Groups but was amended midway to monitor their function following the training. Hence, 3-month follow-up was conducted for only 8 participants in the Delay Group and 3 participants in the Parent-trained Group. Twenty-five children completed a 4-year-old follow-up GMFM-66 assessment. The most common reason for loss to follow-up was scheduling difficulties. Other reasons included refusal to subject child to further assessments, moving out of town following the study and inability to collect data due to COVID-19 restrictions.
Baseline Data
Baseline demographics are shown in Table 1. The average age (months) at baseline was significantly different between the groups (P = .03), whereas the proportion of females was not significantly different (Delay=.31, Immediate=.42, and Parent-trained=.56; P = .51). The proportion of children classified as Gross Motor Function Classification System (GMFCS) level I vs II when they entered the study was not significantly different between groups (Delay=.85, Immediate=1.00, and Parent-trained=.78; P = .26). All children were classified as GMFCS level I at their four-year-old follow-up.
Table 1.
Participant Demographics
| Delay (n = 13) | Immediate (n = 12) | Parent-trained (n = 9) | |
|---|---|---|---|
| Age at baseline (months ± SD) | 23.2 ± 9.7 | 14.2 ± 5.1 | 21.1 ± 6.8 |
| Sex (proportion female vs male) | .31 | .42 | .56 |
| GMFCS level (proportion I vs II) | .85 | 1.00 | .78 |
| Type of stroke (proportion arterial vs venous) | .77 | .58 | .44 |
| Lower extremity affected (proportion right vs left) | .62 | .75 | .33 |
| Walking independently at baseline (proportion walking independently vs not) | .69 | .42 | .67 |
Gross Motor Function Measure-66 (GMFM-66)
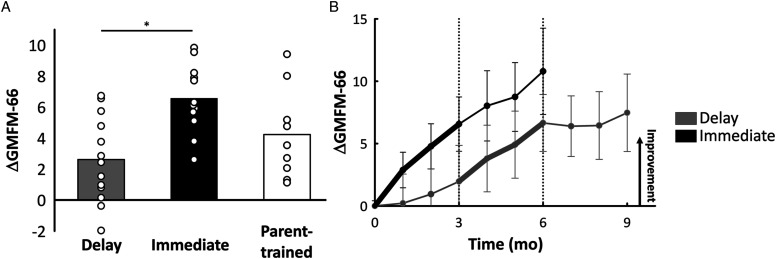
The average change in GMFM-66 score over the first 3 months was 2.69±2.65 for the Delay Group, 6.62±2.27, for the Immediate Group, and 4.58±3.02 for the Parent-trained Group (Figure 3A). Since there was a difference in age between the Delay and Immediate Groups, GMFM-66 results were analyzed in a multivariable model with both age and group as independent variables. Group allocation but not age was statistically significant. Children in the Immediate Group on average had a change in GMFM score 3.4 points higher (95% confidence interval 1.08–5.64, P = .006) than children in the Delay Group, adjusted for age. The change in GMFM-66 scores over time in the study for the Delay and Immediate Groups is shown in Figure 3B. Because of the low numbers of participants in each group, no additional statistical comparisons were made. Nevertheless, the descriptive data suggests a continued improvement in the Immediate Group over the follow-up period, while the Delay Group showed smaller improvements in the same period. Further, the changes (Δ) in standing and walking subscores mirrored the change in total GMFM-66 scores (standing: ΔDelay=7.20±7.98, ΔImmediate=24.24±15.88, and ΔParent=16.97±18.24; walking: ΔDelay=6.38±8.86, ΔImmediate=11.17±6.18, and ΔParent=8.49±6.96).
Figure 3.
Primary outcome measure: change in Gross Motor Function Measure-66 (GMFM-66)A) Average change scores (i.e., difference between measures at three months and baseline) for the total GMFM-66 score are shown by the bars for each group and change scores for each participant are shown by the white circles. Asterisk indicates p<0.05. B) Average change scores for the total GMFM-66 score are indicated for each month for the Delay Group (grey; n=8 for 3-month follow-up) and Immediate Group (black; n=12 for 3-month follow-up) for each month. Bold lines represent the three-month training period, error bars represent standard error of the mean. Vertical dotted lines segment the 3-month time periods in the study. Abbreviations: GMFM-66, Gross Motor Function Measure-66.
At the four-year-old follow-up, the average GMFM-66 score was 69.6±5.8 for children who had been in the Delay Group (n = 9), 74.5±7.6 for those in the Immediate Group (n = 9) and 73.3±6.5 for the Parent-trained Group (n = 7). These mean scores fall between the 40th and 65th percentiles for children classified as GMFCS level I at 4 years old in the reference curves. 32 The percentile scores for most children at the beginning of the study are unknown because there are no percentile scores for children younger than 2 years old.
Step Counts
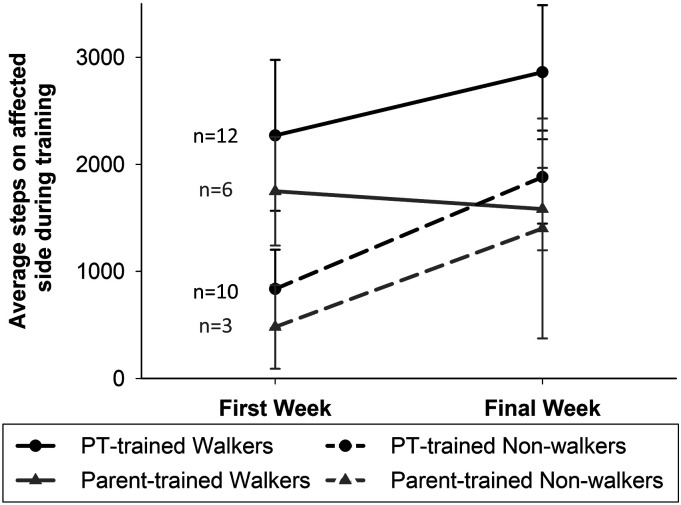
Step counts measured the training intensity. Average step counts on the affected lower extremity during the first and last week of training are illustrated in Figure 4. The average number of steps increased over the training period for all children in the RCT who were trained by a PT. The average step counts in the Parent-trained Group, however, increased over the training period for children who were not walking independently at the beginning of training but did not increase among children who were walking independently (details in Supplemental material, Table 1). The average age of independent walking, defined as the ability to take 10 steps without assistance, assessed with the GMFM-66 or reported by their parents, was 19.8±6.3, 15.3±2.5, and 19.0±4.3 (mean months ± SD) for the Delay, Immediate, and Parent-trained Groups, respectively.
Figure 4.
Step counts during trainingBlack circles represent the average step count on the affected lower extremity for PT-trained children during the first and final week of training. Gray triangles represent the average step counts for children in the Parent-trained Group. Solid lines represent children who were able to walk independently during the first week of training and dashed lines indicate children who required assistance to walk at the beginning of training. Error bars represent standard error of the mean. Abbreviations: PT, physical therapist.
Full-day step counts gauged the level of activity in children on non-training days. In general, children with perinatal stroke showed increases of about 1000 steps/day over a 3-month period with or without training, with considerable variation between children (Supplemental material, Figure 1).
Kinematics of Walking
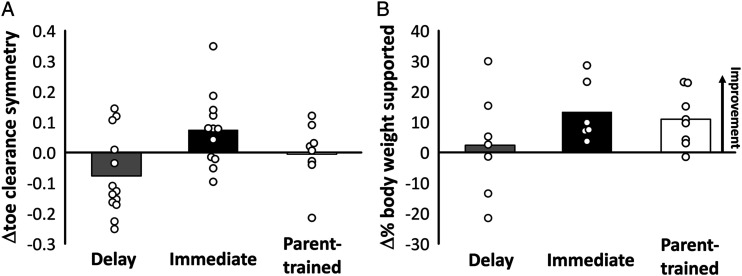
Perfect symmetry in toe clearance of the left and right foot is represented by zero (equation (1)). Since asymmetry could be positive or negative depending on whether the unaffected or affected toe is lifted higher, the change in the symmetry of toe clearance (ΔTS) over 3 months was calculated as: , where is the symmetry at baseline and is the symmetry at the end of the 3-month period (i.e., delay or training). Thus, a positive means they became more symmetric, and a negative means the opposite. The average change in symmetry of toe clearance (Figure 5A) worsened for the Delay Group (−.08±.13), improved for the Immediate Group (.07±.12), and changed minimally for the Parent-trained Group (−.01±.10). Since there was a significant difference in age between the Delay and Immediate groups, statistical analysis was performed using a multivariable model. In the multivariable model with both age and group as independent variables, group was not statistically significant (P = .10).
Figure 5.
Treadmill Walking ResultsA) Average change scores (i.e., difference between measures at three months and baseline) for the toe clearance symmetry score are shown by the bars for each group and change scores for each participant are shown by the white circles. Delay n=13, Immediate n=12, Parent-trained n=9. B) Average change scores for the percent of their body weight a child could independently support are shown by the bars for each group and change scores for each participant are shown by the white circles. Delay n=7, Immediate n=6, Parent-trained n=8.
Weight-bearing during walking was expressed as a percentage of the child’s body weight. Independent weight-bearing will approach or exceed 100%, so increases in weight-bearing indicated more proficient walkers. Weight-bearing increased by 2.4%±7.2 for children in the Delay Group, 13.3%±10.1 for the Immediate Group and 10.9%±9.0 for the Parent-trained Group (Figure 5B). Only descriptive statistics were performed since a treadmill with force plates was only available to children in Edmonton (n = 21).
Harms
No unintended effects were observed in any participants.
Discussion
The intensive ELEVATE rehabilitation program for young children with perinatal stroke resulted in significant improvement in gross motor function as compared to usual care. Children in the Immediate Group on average had a change in GMFM-66 score 3.4 units higher (95% confidence interval 1.08–5.64) than children in the Delay Group adjusted for age. This change is higher than previously reported minimum clinically important differences (MCIDs) of 1.7 (medium) and 2.7 (large), 33 although these MCIDs were established in an older population of children and included children with unilateral and bilateral CP. By the end of the training, children who were walking independently were taking an average of approximately 3000 steps (2862) with their affected lower extremity in 1 hour of training, and children who were walking with assistance were taking an average of approximately 2000 steps (1881). There was also a trend for improved symmetry in walking with training, but this was not statistically significant. These results demonstrate that it is possible to engage young children in intensive training of the lower extremity resulting in significant improvement in gross motor function in the short term.
Limitations
Despite randomization there was a difference in age between the control and intervention groups. Since intervening at an earlier age could be more efficacious, a multivariable statistical modeling was used to account for the age difference. Another limitation is the relatively small number of participants (n = 25 in RCT), and equipment limitations that only allowed weight-bearing in walking to be assessed at 1 site. Further, step counts were used to measure dosage of the intervention, but the training also included other active while stationary skills such as standing balance and squatting, which are not reflected in step counts. Usual care for these children typically consists of 1 hour sessions of occupational and/or physical therapy approximately 1–2 times/month; however, this was not systematically tracked throughout the delay period in this study. Finally, 1 3-month bout of intensive training may not be sufficient to have a significant impact on long-term motor function. Determining the ideal time for additional training is needed.
Nature of Motor Intervention
The premise of this study is based on enhancing neuroplasticity in the developing nervous system. The development of the nervous system is driven by activity, so a child-initiated, activity-based approach was used. This approach incorporates principles such as specificity, repetition, and intensity to promote experience-dependent plasticity. 34 Novak and colleagues 35 indicated that there is clear evidence for rehabilitation approaches that improve function and those that do not; interventions that use volitional active movements at high intensity to practice task-specific training of real-life activities enhance motor outcomes. Intensive rehabilitation has been shown to result in greater improvements in gross motor function than non-intensive treatment and the effect tends to be stronger for children under 2 years of age. 36 We observed this trend for larger improvements in GMFM-66 scores when training was initiated before 2 years old (Supplemental Material Figure 2). However, other reasons besides the greater neuroplasticity of a young nervous system should not be discounted, including the natural reduction in improvement of the GMFM-66 with age, 32 the greater challenge in directing children around the age of 2 years old, and the difficulty of enhancing lower extremity function in independently walking children. Differentiating between these factors is difficult.
ELEVATE was tailored to each child to continually challenge their functional ability. For example, children walked on different surfaces with as little support as needed and tasks were made progressively difficult (e.g., walking in different direction or at different speeds, ascending stairs and ramps). Improvements in function only continue when difficulty of the training is increased. 37 The challenge in ELEVATE included increasing the number of repetitions (steps) and the presumed activity of the muscles (e.g., use of weights and progressively increasing task difficulty to challenge balance and gross motor skills).
We provided ample opportunities for children to develop motor skills through a variety of sensorimotor experiences, analogous to approaches used by the Neuronal Group Selection Theory. 38 For example, children in our study trained on many walking surfaces. Motivation was increased and reinforced throughout the training using play and positive feedback. Motivation and child-initiated movement are essential to enhance neuroplasticity, so task-specific training that is rewarding and enjoyable may promote higher intensity of training and spontaneous, child-initiated practice of skills outside of training sessions. We speculate that the continued improvement during the follow-up period suggests the children continued to use their newly learned skills. Indeed, the full-day step counts (Supplemental Material, Figure 1) suggest that children in the Immediate and Parent-trained Groups continued to increase in daily steps and GMFM scores (Figure 3B), while the Delay Group showed less improvement in both. The differences may be related to the personal characteristics of the children.
Early Intervention
The children entered this study as young as 8 months old, when typically developing children increase their lower extremity strength as they begin to pull to stand and increase weight-bearing. Lower extremity muscle volume is comparable between typically developing children and those with CP until 15 months of age. 39 The change after 15 months is likely related in part to reduced use as typically developing children begin to walk independently around 12±3 months and involves thousands of steps per day, while those with CP are less active and have fewer opportunities to develop lower extremity strength.40,41 Although it is unclear whether physical activity prevents contractures, early rehabilitation focusing on using the full range of motion may help, since decreased muscle extensibility has been observed as early as 25 to 30 months of age, with contractures developing before the age of 3.39,42
Several training-based protocols have been reported or are underway for young children (e.g., GAME, HABIT-ILE, small steps, baby-CIMT, baby-bimanual, reviewed in 15,43). While most of these early interventions target the upper extremities, some have included the lower extremity.10,11 Here, we show that early, activity-based intervention focused on the lower extremity is feasible and can improve gross motor function.
As the efficacy of early interventions is established, early diagnosis of perinatal stroke, and CP in general, is increasingly important (reviewed in 44–46). Facilitating early detection and diagnosis is especially important for perinatal strokes where approximately half of cases are not identified at the time of birth and children often have few risk factors for CP. 47 The importance of early diagnosis and rehabilitation is reflected in recent recommendations. 48
Parent Involvement
Children trained by their parents demonstrated variable consistency and efficacy. Home programs have been recommended to increase hand function and self-care for children with CP10,35,49 and are well-received by parents. 10 In addition to increasing the intensity of intervention, home programs offer practice in the natural environment, flexibility with scheduling, and cost-effectiveness. 50 A major barrier to intensive therapy is the time commitment required from families (i.e., 42% of eligible potential participants in this study declined, with 27% indicating that the time required was prohibitive). Some of the difficulties parents encountered in the past included compliance pressure and feeling a lack of confidence.51,52 Here, it was especially challenging for parents with children already independently walking, as seen from the step counts during training (Figure 4). Training families to administer early interventions at home could empower them with the skills to facilitate their child’s rehabilitation while also enhancing their accessibility to a PT. Parents of 2 participants in the Parent-trained Group requested closer contact with the PT. This included more frequent visits than the monthly in-person training. Interestingly, the same 2 children showed the greatest improvement in gross motor function among children in the Parent-trained Group. Thus, more frequent in-person parental support from the therapist may be beneficial.
Future Directions
An intervention that requires PT training one hour/day, four days per week, is costly and not always feasible for families. Exploring different delivery models, including a hybrid approach of both therapist and parental delivery of intervention may be a good compromise, which we are currently investigating. We are also pursuing the generalizability of ELEVATE for children at risk of bilateral CP from encephalopathy of prematurity.
Supplemental Material
Supplemental Material for Early, Intensive, Lower Extremity Rehabilitation Shows Preliminary Efficacy After Perinatal Stroke: Results of a Pilot Randomized Controlled Trial by Caitlin Hurd, Donna Livingstone, Kelly Brunton, Allison Smith, Monica Gorassini, Man-Joe Watt, John Andersen, Adam Kirton, and Jaynie F. Yang in Neurorehabilitation and Neural Repair
Acknowledgments
The authors thank Dr Elizabeth Condliffe for assistance reviewing potential participants for eligibility, Dr Jerome Yager for assistance with recruitment, Michelle Teves for assistance with data collection and analysis, and all the PT assessors in Edmonton and Calgary. We thank Dr Maryna Yaskina for statistical analysis, which was facilitated by the Women and Children’s Health Research Institute through the generous support of the Stollery Children’s Hospital Foundation. We thank the C.H.Riddell Family Movement Assessment Centre at the Alberta Children’s Hospital for recording the kinematics of treadmill walking in Calgary. We are tremendously grateful to the parents and guardians who dedicated large amounts of time to our study. Without their dedication, this study would not have been possible.
Footnotes
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research [MOP 126107], Alberta Innovates Health Solutions [Collaborative Research and Innovation Opportunities Project Funding #201200830], and CH was awarded a Women and Children’s Health Research Institute (WCHRI) graduate studentship that has been funded through the generous support of the Stollery Children’s Hospital Foundation.
ORCID iDs
John Andersen https://orcid.org/0000-0001-7138-3414
Jaynie F. Yang https://orcid.org/0000-0002-7507-8924
References
- 1.Chabrier S, Husson B, Dinomais M, Landrieu P, Nguyen The Tich S. New insights (and new interrogations) in perinatal arterial ischemic stroke. Thromb Res. 2011;127(1):13-22. doi: 10.1016/j.thromres.2010.10.003. [DOI] [PubMed] [Google Scholar]
- 2.Nelson KB, Lynch JK. Stroke in newborn infants. Lancet Neurol. 2004;3:150-158. http://neurology.thelancet.com. [DOI] [PubMed] [Google Scholar]
- 3.Dunbar M, Kirton A. Perinatal stroke: mechanisms, management, and outcomes of early cerebrovascular brain injury. Lancet Child Adolesc Heal. 2018;2(9):666-676. doi: 10.1016/S2352-4642(18)30173-1. [DOI] [PubMed] [Google Scholar]
- 4.Murphy KP. Cerebral palsy lifetime care - four musculoskeletal conditions. Dev Med Child Neurol. 2009;51(suppl 4):30-37. doi: 10.1111/j.1469-8749.2009.03431.x. [DOI] [PubMed] [Google Scholar]
- 5.Horstmann HM, Hosalkar H, Keenan MA. Orthopaedic issues in the musculoskeletal care of adults with cerebral palsy. Dev Med Child Neurol. 2009;51(suppl 4):99-105. doi: 10.1111/j.1469-8749.2009.03417.x. [DOI] [PubMed] [Google Scholar]
- 6.Sakzewski L, Gordon A, Eliasson AC. The state of the evidence for intensive upper limb therapy approaches for children with unilateral cerebral palsy. J Child Neurol. 2014;29(8):1077-1090. doi: 10.1177/0883073814533150. [DOI] [PubMed] [Google Scholar]
- 7.Myrhaug HT, ØstensjØ S, Larun L, Odgaard-Jensen J, Jahnsen R. Intensive training of motor function and functional skills among young children with cerebral palsy: a systematic review and meta-analysis. BMC Pediatr. 2014;14(1):292. doi: 10.1186/s12887-014-0292-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hoare BJ, Wallen MA, Thorley MN, Jackman ML, Carey LM, Imms C. Constraint-induced movement therapy in children with unilateral cerebral palsy. Cochrane Database Syst Rev. 2019;2019(4):CD004149. doi: 10.1002/14651858.CD004149.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bleyenheuft Y, Arnould C, Brandao MB, Bleyenheuft C, Gordon AM. Hand and Arm Bimanual Intensive Therapy Including Lower Extremity (HABIT-ILE) in children with unilateral spastic cerebral palsy: a randomized trial. Neurorehabil Neural Repair. 2015;29(7):645-657. doi: 10.1177/1545968314562109. [DOI] [PubMed] [Google Scholar]
- 10.Holmström L, Eliasson A-C, Almeida R, et al. Efficacy of the small step program in a randomized controlled trial for infants under 12 months old at risk of Cerebral Palsy (CP) and other neurological disorders. J Clin Med. 2019;8(7):1016. doi: 10.3390/jcm8071016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Araneda R, Sizonenko SV, Newman CJ, et al. Functional, neuroplastic and biomechanical changes induced by early Hand-Arm Bimanual Intensive Therapy Including Lower Extremities (e-HABIT-ILE) in pre-school children with unilateral cerebral palsy: study protocol of a randomized control trial. BMC Neurol. 2020;20(1):133. doi: 10.1186/s12883-020-01705-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.ACPR Register . Report of the Australian Cerebral Palsy Register: Birth Years 1995-2012. North Adelaide, Australia: Australian Cerebral Palsy Register; 2018. [Google Scholar]
- 13.Tinderholt Myrhaug H, Østensjø S, Larun L, Odgaard-Jensen J, Jahnsen R. Intensive training of motor function and functional skills among young children with cerebral palsy: a systematic review and meta-analysis. BMC Pediatr. 2014;14:292. http://www.biomedcentral.com/1471-2431/14/292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Goldstein M, Harper D. Management of cerebral palsy: equinus Gait. Dev Med Child Neurol. 2001;43(8):563-569. [DOI] [PubMed] [Google Scholar]
- 15.Morgan C, Darrah J, Gordon AM, et al. Effectiveness of motor interventions in infants with cerebral palsy: a systematic review. Dev Med Child Neurol. 2016;58(9):900-909. doi: 10.1111/dmcn.13105. [DOI] [PubMed] [Google Scholar]
- 16.McIntyre S, Morgan C, Walker K, Novak I. Cerebral palsy-don’t delay. Dev Disabil Res Rev. 2011;17(2):114-129. doi: 10.1002/ddrr.1106. [DOI] [PubMed] [Google Scholar]
- 17.Booth ATC, Buizer AI, Meyns P, Oude Lansink ILB, Steenbrink F, van der Krogt MM. The efficacy of functional gait training in children and young adults with cerebral palsy: a systematic review and meta-analysis. Dev Med Child Neurol. 2018;60(9):866-883. doi: 10.1111/dmcn.13708. [DOI] [PubMed] [Google Scholar]
- 18.Moreau NG, Bodkin AW, Bjornson K, Hobbs A, Soileau M, Lahasky K. Effectiveness of rehabilitation interventions to improve gait speed in children with cerebral palsy: systematic review and meta-analysis. Phys Ther. 2016;96(12):1938-1954. https://academic.oup.com/ptj/article-abstract/96/12/1938/2866295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Martin JH, Donarummo L, Hacking A. Impairments in prehension produced by early postnatal sensory motor cortex activity blockade. J Neurophysiol. 2000;83(2):895-906. [DOI] [PubMed] [Google Scholar]
- 20.Martin JH, Friel KM, Salimi I, Chakrabarty S. Activity- and use-dependent plasticity of the developing corticospinal system. Neurosci Biobehav Rev. 2007;31(8):1125-1135. doi: 10.1016/j.neubiorev.2007.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Friel K, Chakrabarty S, Kuo HC, Martin J. Using motor behavior during an early critical period to restore skilled limb movement after damage to the corticospinal system during development. J Neurosci. 2012;32(27):9265-9276. doi: 10.1523/JNEUROSCI.1198-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Brody BA, Kinney C, Kloman AS, Gilles FH. Sequence of central nervous system myelination in human infancy. J Neuropathol Exp Neurol. 1987;47(3):217-234. https://academic.oup.com/jnen/article/46/3/283/2611410. [DOI] [PubMed] [Google Scholar]
- 23.Kempf A, Schwab ME. Nogo-a represses anatomical and synaptic plasticity in the central nervous system. Physiology. 2013;28(3):151-163. doi: 10.1152/physiol.00052.2012. [DOI] [PubMed] [Google Scholar]
- 24.Kirton A, Metzler MJ, Craig BT, et al. Perinatal stroke: mapping and modulating developmental plasticity. Nat Rev Neurol. 2021;17(7):415-432. doi: 10.1038/s41582-021-00503-x. [DOI] [PubMed] [Google Scholar]
- 25.Yang JF, Livingstone D, Brunton K, et al. Training to enhance walking in children with cerebral palsy: are we missing the window of opportunity? Semin Pediatr Neurol. 2013;20(2):106-115. doi: 10.1016/j.spen.2013.06.011. [DOI] [PubMed] [Google Scholar]
- 26.Hurd C, Livingstone D, Brunton K, et al. Early intensive leg training to enhance walking in children with perinatal stroke: protocol for a randomized controlled trial. Phys Ther. 2017;97(8):818-825. https://academic.oup.com/ptj. [DOI] [PubMed] [Google Scholar]
- 27.Dunbar M, Kirton A. Perinatal stroke. Semin Pediatr Neurol. 2019;32:100767. doi: 10.1016/j.spen.2019.08.003. [DOI] [PubMed] [Google Scholar]
- 28.Damiano DL, Stanley CJ, Bulea TC, Park HS. Motor learning abilities are similar in hemiplegic cerebral palsy compared to controls as assessed by adaptation to unilateral leg-weighting during gait: part I. Front Hum Neurosci. 2017;11. doi: 10.3389/fnhum.2017.00049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Russell D, Rosenbaum P, Avery L, Lane M. Gross Motor Function Measure (GMFM - 66 and GMFM - 88) User’s Manual. London, UK: Mac Keith Press; 2002. [Google Scholar]
- 30.Wang HY, Yang YH. Evaluating the responsiveness of 2 versions of the gross motor function measure for children with cerebral palsy. Arch Phys Med Rehabil. 2006;87(1):51-56. doi: 10.1016/j.apmr.2005.08.117. [DOI] [PubMed] [Google Scholar]
- 31.Wei S, Wang SJ, Liao YG, Hong Y, Xu XJ, Shao XM. Reliability and validity of the GMFM-66 in 0- to 3-year-old children with cerebral palsy. Am J Phys Med Rehabil. 2006;85(2):141-147. doi: 10.1097/01.phm.0000197585.68302.25. [DOI] [PubMed] [Google Scholar]
- 32.Hanna SE, Bartlett DJ, Rivard LM, Russell DJ. Reference curves for the gross motor function measure: percentiles for clinical description and tracking over time among children with cerebral palsy. Phys Ther. 2008;88(5):596-607. www.ptjournal.org. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Oeffinger D, Bagley A, Rogers S, et al. Outcome tools used for ambulatory children with cerebral palsy: responsiveness and minimum clinically important differences. Dev Med Child Neurol. 2008;50(12):918-925. doi: 10.1111/j.1469-8749.2008.03150.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kleim J, Jones T. Principles of experience-dependent neural plasticity: implications for rehabilitation after brain damage. J Speech Lang Hear Res. 2008;51:S225-S239. [DOI] [PubMed] [Google Scholar]
- 35.Novak I, Morgan C, Fahey M, et al. State of the evidence traffic lights 2019: systematic review of interventions for preventing and treating children with cerebral palsy. Curr Neurol Neurosci Rep. 2020;20(2):3. doi: 10.1007/s11910-020-1022-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Arpino C, Vescio MF, De Luca A, Curatolo P. Efficacy of intensive versus nonintensive physiotherapy in children with cerebral palsy: a meta-analysis. Int J Rehabil Res. 2010;33(2):165-171. doi: 10.1097/MRR.0b013e328332f617. [DOI] [PubMed] [Google Scholar]
- 37.Nielsen JB, Willerslev-Olsen M, Christiansen L, Lundbye-Jensen J, Lorentzen J. Science-based neurorehabilitation: recommendations for neurorehabilitation from basic science. J Mot Behav. 2015;47(1):7-17. doi: 10.1080/00222895.2014.931273. [DOI] [PubMed] [Google Scholar]
- 38.Hadders-Algra M. The neuronal group selection theory: promising principles for understanding and treating developmental motor disorders. Dev Med Child Neurol. 2000;42:707-715. [DOI] [PubMed] [Google Scholar]
- 39.Herskind A, Ritterband-Rosenbaum A, Willerslev-Olsen M, et al. Muscle growth is reduced in 15-month-old children with cerebral palsy. Dev Med Child Neurol. 2015;58(5):485-491. doi: 10.1111/dmcn.12950. [DOI] [PubMed] [Google Scholar]
- 40.WHO Multicentre Growth Reference Study Group . WHO motor development study: windows of achievement for six gross motor development milestones. Acta Paediatr Int J Paediatr. 2006;95:86-95. doi: 10.1080/08035320500495563. [DOI] [PubMed] [Google Scholar]
- 41.Adolph KE, Cole WG, Komati M, et al. How do you learn to walk? Thousands of steps and dozens of falls per day. Psychol Sci. 2012;23(11):1387-1394. doi: 10.1177/0956797612446346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Willerslev-Olsen M, Lorentzen J, Sinkjær T, Nielsen JB. Passive muscle properties are altered in children with cerebral palsy before the age of 3 years and are difficult to distinguish clinically from spasticity. Dev Med Child Neurol. 2013;55(7):617-623. doi: 10.1111/dmcn.12124. [DOI] [PubMed] [Google Scholar]
- 43.Hadders-Algra M, Boxum AG, Hielkema T, Hamer EG. Effect of early intervention in infants at very high risk of cerebral palsy: a systematic review. Dev Med Child Neurol. 2017;59(3):246-258. doi: 10.1111/dmcn.13331. [DOI] [PubMed] [Google Scholar]
- 44.Novak I, Morgan C, Adde L, et al. Early, accurate diagnosis and early intervention in cerebral palsy: advances in diagnosis and treatment. JAMA Pediatr. 2017;171(9):897-907. doi: 10.1001/jamapediatrics.2017.1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Novak I, Morgan C. High-risk follow-up: early intervention and rehabilitation. In: Vinken P, Bruyn G, eds. Handbook of Clinical Neurology, Vol 162. Amsterdam, Netherlands: Elsevier B.V.; 2019:483-510. doi: 10.1016/B978-0-444-64029-1.00023-0. [DOI] [PubMed] [Google Scholar]
- 46.Hadders-Algra M. Early diagnostics and early intervention in neurodevelopmental disorders-age-dependent challenges and opportunities. J Clin Med. 2021;10(4). doi: 10.3390/jcm10040861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lee J, Lisa Croen MA, Backstrand KH, et al. Maternal and infant characteristics associated perinatal arterial stroke in the infant. JAMA. 2005;293(6):723-729. https://jamanetwork.com/. [DOI] [PubMed] [Google Scholar]
- 48.Boychuck Z, Andersen J, Bussières A, et al. International expert recommendations of clinical features to prompt referral for diagnostic assessment of cerebral palsy. Dev Med Child Neurol. 2020;62(1):89-96. doi: 10.1111/dmcn.14252. [DOI] [PubMed] [Google Scholar]
- 49.Eliasson AC, Nordstrand L, Ek L, et al. The effectiveness of baby-CIMT in infants younger than 12 months with clinical signs of unilateral-cerebral palsy; an explorative study with randomized design. Res Dev Disabil. 2018;72:191-201. doi: 10.1016/j.ridd.2017.11.006. [DOI] [PubMed] [Google Scholar]
- 50.Chamudot R, Parush S, Rigbi A, Horovitz R. Effectiveness of modified constraint-induced movement therapy compared with bimanual therapy home programs for infants with hemiplegia: a randomized controlled trial varda gross-tsur. Am J Occup Ther. 2018;72(6):7206205010p1-7206205010p9. doi: 10.5014/ajot.2018. [DOI] [PubMed] [Google Scholar]
- 51.Novak I. Parent experience of implementing effective home programs. Phys Occup Ther Pediatr. 2011;31(2):198-213. doi: 10.3109/01942638.2010.533746. [DOI] [PubMed] [Google Scholar]
- 52.Kruijsen-Terpstra AJA, Ketelaar M, Boeije H, et al. Parents’ experiences with physical and occupational therapy for their young child with cerebral palsy: a mixed studies review. Child Care Health Dev. 2014;40(6):787-796. doi: 10.1111/cch.12097. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Material for Early, Intensive, Lower Extremity Rehabilitation Shows Preliminary Efficacy After Perinatal Stroke: Results of a Pilot Randomized Controlled Trial by Caitlin Hurd, Donna Livingstone, Kelly Brunton, Allison Smith, Monica Gorassini, Man-Joe Watt, John Andersen, Adam Kirton, and Jaynie F. Yang in Neurorehabilitation and Neural Repair