Abstract
Propolis is a resinous natural product produced by honeybees using beeswax and plant exudates. The chemical composition of propolis is highly complex, and varies with region and season. This inherent chemical variability presents several challenges to its standardisation and quality control. The present review was aimed at highlighting marker compounds for different types of propolis, produced by the species Apis mellifera, from different geographical origins and that display different biological activities, and to discuss strategies for quality control. Over 800 compounds have been reported in the different propolises such as temperate, tropical, birch, Mediterranean, and Pacific propolis; these mainly include alcohols, acids and their esters, benzofuranes, benzopyranes, chalcones, flavonoids and their esters, glycosides (flavonoid and diterpene), glycerol and its esters, lignans, phenylpropanoids, steroids, terpenes and terpenoids. Among these, flavonoids (> 140), terpenes and terpenoids (> 160) were major components. A broad range of biological activities, such as anti-oxidant, antimicrobial, anti-inflammatory, immunomodulatory, and anticancer activities, have been ascribed to propolis constituents, as well as the potential of these compounds to be biomarkers. Several analytical techniques, including non-separation and separation methods have been described in the literature for the quality control assessment of propolis. Mass spectrometry coupled with separation methods, followed by chemometric analysis of the data, was found to be a valuable tool for the profiling and classification of propolis samples, including (bio)marker identification. Due to the rampant chemotypic variability, a multiple-marker assessment strategy considering geographical and biological activity marker(s) with chemometric analysis may be a promising approach for propolis quality assessment.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11101-022-09816-1.
Keywords: Propolis, Phytochemistry, Marker compounds, Quality control, Standardisation
Introduction
Folk medicine plays a vital role in the human health care systems of many countries around the globe, and has done so since antiquity. In traditional systems of medicine, thousands of plants and their derived products, such as propolis, are reported to have significant medical value, but most remain untested in new drug discovery approaches. Propolis (bee glue) is a resinous, sticky, coloured material with a characteristic smell that is prepared by honeybees, using beeswax and plant exudates (Sforcin 2016). The colour of propolis depends largely on the plant source and collection time (Chen et al. 2004), and varies from green to red to dark brown, with dark brown being the most common (Lotti et al. 2010; Kasote et al. 2017). Honeybees use propolis as a construction material for sealing openings and cracks in their beehive, also to avoid the entry of intruders, and to maintain aseptic conditions within the beehive (Bankova et al. 2002; Salatino et al. 2005). Commercial propolis is produced mainly by the honeybee, Apis mellifera, although small amounts are derived from stingless bee species (Kasote 2017; Kasote et al. 2019).
The chemical composition of propolis is highly complex and changeable. It generally comprises a vegetable resin and balsam (up to 70%), beeswax (< 10% to 87%), volatile organic compounds (< 1% to 3%), pollen, and other substances, representing compound classes such as aliphatic acids and their esters, aromatic acids and their esters, flavonoids and other plant phenolics, terpenoids, carbohydrates, amino acids, vitamins (B1, B2, B6, C, and E), and minerals (aluminum, antimony, calcium, cesium, copper, iron, lanthanum, manganese, mercury, nickel, silver, vanadium, and zinc) (Burdock 1998; De Almeida and Menezes 2002; Cunha et al. 2004; Anjum et al. 2019; Salatino and Salatino 2021).
Since approximately 300 bc, propolis has been used in traditional Egyptian medicine as a therapeutic agent (Banskota et al. 1998; Hegazi and Abd El Hady 2002). The use of propolis to accelerate wound-healing and for other conditions, were also recorded in archaic Greek, Roman, and Georgian folk medicine (Salatino et al. 2005; Sforcin and Bankova 2011). In modern time, the use of propolis in pharmaceutical and food preparations has been increasing, due to its wide range of therapeutic and health-promoting activities, which include anti-oxidant, antibacterial, antifungal, anti-inflammatory, antiviral, immunomodulatory, hepatoprotective, anti-allergic, septic wound healing, antitumour and antidiabetic activities (Banskota et al. 2002; Oršolić et al. 2006; Schnitzler et al. 2010; Adewumi and Ogunjinmi 2011; Shi et al. 2012b; Bhadauria 2012; Wu et al. 2012; Chan et al. 2013; Przybyłek and Karpiński 2019; Pobiega et al. 2019; Nandre et al. 2021) Recently, it was reported that Brazilian and Egyptian propolis can be regarded as a safe and effective adjunct therapy to treat severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (Silveira et al. 2021; H Elwakil et al. 2021), which caused a global pandemic referred to as coronavirus disease 2019 (COVID-19) that is still not under control.
Despite its promising therapeutic and health-promoting potential, the use of propolis as a drug or herbal supplement has not been officially accepted in most countries, due to its inconsistent composition. The phytochemistry of propolis varies according to the region and season, and this may be the biggest hurdle impeding the standardisation and quality control of this substance. The availability of comprehensive information regarding the possible phytochemical composition of propolis samples from different specific places of origin, including qualitative and quantitative biomarker data, will aid in the quality control of propolis. Several review articles have been published in the context of the phytochemistry of different types of propolis, the factors affecting the phytochemistry, related health benefits of propolis, and the problems associated with chemical standardisation (Salatino et al. 2011; Farooqui and Farooqui 2012; Bankova et al. 2014; Pobiega et al. 2019; Shahinozzaman et al. 2021). However, comprehensive information regarding the diverse range of phytochemicals, specific geographical and biological marker compounds, and the analytical techniques used for the standardisation and quality control of propolis, has not been collectively and comprehensively compiled. Hence, the approach taken in this review was to summarise the phytochemical content of propolis from all over the world, according to compound classes, and, where possible, link it to the botanical source and place of origin. Moreover, herein, marker compounds that characterise the geographic origin and biological activities are collated, the existing quality control strategies are discussed, and future research perspectives are proposed.
Phytochemical composition and chemo-geographical variation
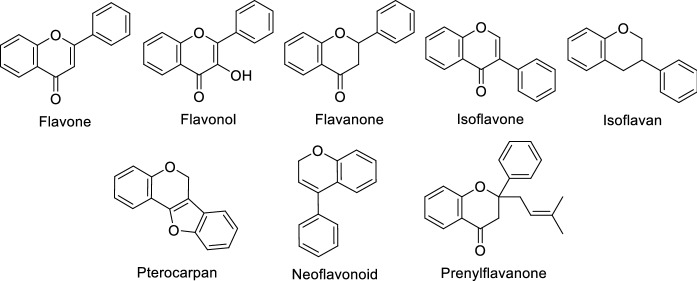
Propolis is a very rich source of a wide range of bioactive compounds. Numerous factors, such as the origin, time of collection, plant sources visited by the bees, and bee species, determine the chemical composition of propolis (Tagliacollo and Orsi 2011; Huang et al. 2014). Although it was reported earlier (Huang et al. 2014) that about 300 phytoconstituents, such as polyphenols, esters of phenolic acids, flavonoids, sesquiterpene, diterpenes, triterpenes, lignans, prenylated benzophenones, aldehydes, steroids, and coumarins, are present in propolis samples from various parts of the world, this number has increased to more than 800 (Šturm and Ulrih 2019). Polyphenols are the most abundant constituents of the majority of propolis samples (Bankova et al. 1995), and are represented mainly by phenolic acids and their esters, and flavonoids. Brazilian red and green propolis were reported to contain proanthocyanidins (condensed tannins), which is quite rare (Mayworm et al. 2014). By 2014, approximately 140 flavonoids had been reported in propolis, and these mainly include compounds from nine classes, namely; flavones, flavonols, flavanones including prenylflavanones, isoflavones, isoflavanones, isoflavans, neoflavonoids, pterocarpans (Table S1; Fig. 1). Until 2011, it was believed that flavonoid glycosides are uncommon in propolis obtained from A. mellifera (Salatino et al. 2011). However, since then, at least 55 flavonoid glycosides were reported in propolis samples from Serbia, the UK, Portugal and Brazil (Šturm and Ulrih 2019). Similarly, diterpene glycosides, such as diterpene rhamnosides, were reported in propolis from El Salvador (Popova et al. 2001a).
Fig. 1.
Structures of different classes and sub-classes of flavonoids reported in propolis
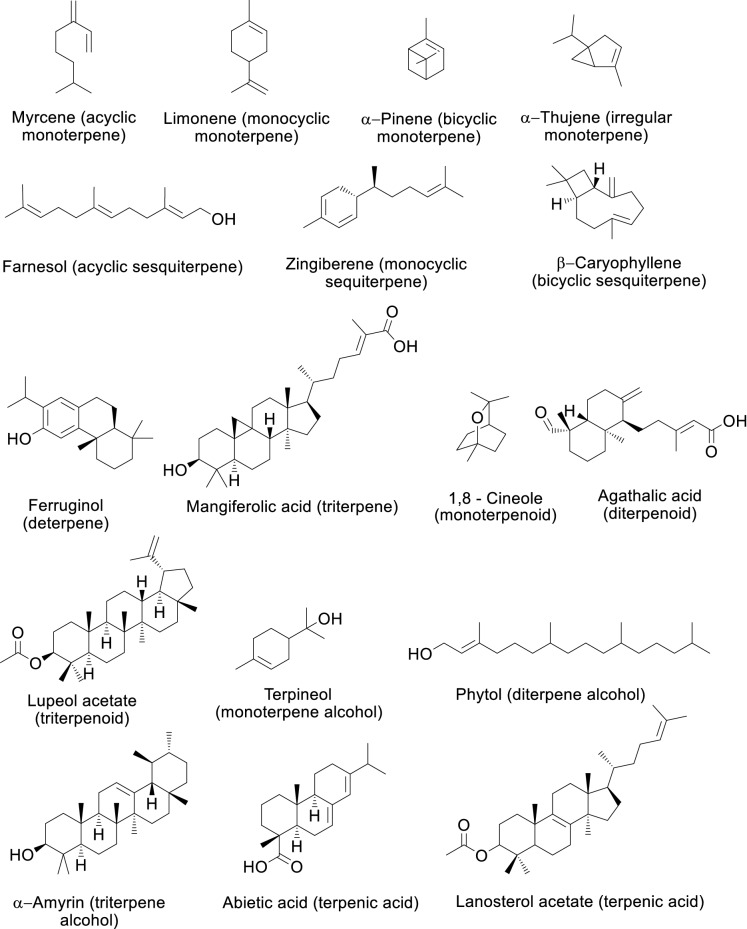
Nearly 160 terpenes, including terpenoids, have been reported in propolis. These primarily includes monoterpenes, sesquiterpenes, diterpenes, triterpenes, monoterpenoids, diterpenoids, and triterpenoids (Table S1; Fig. 2). The occurrence of nitrogenous compounds, such as alkaloids and cyanogens, was also considered to be unusual in propolis obtained from A. mellifera (Salatino et al. 2011). Nonetheless, several alkaloids and alkaloid derivatives, such as pagicerine, demecolcine, papaverine, aspidospermidine, morphinan-6-one-2-ol, thebaine, N,O-dimethyl stephine, morpholine, 5(4H)-thebenidinone, 4-(phenylthioxomethyl)morpholine, 4-methyl-2,6-bis(4-morpholylmethyl)phenol, 3-[(trimethylsilyl)oxy]4,5a-epoxy-14-hydroxy-17-(2-propenyl)morphinan-6-one and nicotinaldehyde semicarbazone were reported from Algerian propolis (Soltani et al. 2017). However, as this is the only communication of alkaloids in propolis and they have been detected by GC–MS, and there is no report on isolation of alkaloids from propolis so far, this information should be corroborated by further research.
Fig. 2.
Terpenes and terpenoids reported in propolis
The volatile oil composition of propolis is comparatively low (typically less than 1%, rarely 2–3% of the total weight of the propolis sample) and generally comprises mono- or sesquiterpenoids, prenylated acetophenones and other volatile organic compounds (Bankova et al. 1998, 2014; Sun et al. 2012). Despite the relatively low concentrations, their characteristic aroma and significant biological activity make them indispensable for propolis characterisation, and profiles of the volatiles can be useful to differentiate propolis samples from different geographical regions (Kaškonienė et al. 2014; Bankova et al. 2014). Samples from temperate zones can be classified as one of two types, based on the presence of representative amounts of β-eudesmol (40–60%) or benzyl benzoate (20–40%) in the volatile fraction (Petri et al. 1988; Pavlovic et al. 2020). Bankova et al. (2014) reviewed the chemical compositions of propolis volatiles from various regions and reported that, other than the usual chemical diversity, most of the European propolis samples are rich in sesquiterpenes, while samples from temperate zones contain mainly monoterpenes in their volatile fractions.
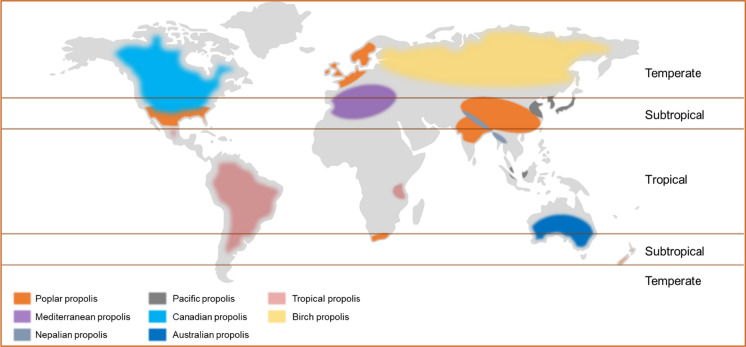
The chemical composition of propolis is largely dependent on the flora frequented by the bees in the vicinity of their hives (Lotti et al. 2010; Righi et al. 2011). The chemistry of propolis samples from different parts of the world has been extensively studied, and notably, was found to be unique to specific geographical zones, in many cases. In general, there have been attempts to classify samples from around the globe according to their chemical composition and floral origin (Fig. 3). Poplar (temperate), Birch, Tropical, Mediterranean and Pacific are the main recognised classes of propolis (Salatino et al. 2011).
Fig. 3.
Global classification of common propolis types based on chemical composition from different geographical zones.
Adapted from Salatino et al. (2011)
Propolis samples originating from temperate zones (West Asia, North Africa, Europe and North America, parts of Argentina, New Zealand) reportedly have similar phytochemical compositions and are predominantly rich in flavonoids and phenolic acid esters, which are almost absent in those originating from tropical regions (Cuesta-Rubio et al. 2007). The propolis obtained from of North-Russia, and mountainous regions of Switzerland and Italy were characterised by phenolic glycerides, namely dicoumaroyl acetyl-, diferuloyl acetyl-, feruloyl coumaroyl acetyl- and caffeoyl coumaroyl acetyl glycerol (Pavlovic et al. 2020) coming from Populus tremula which grows at colder climate and higher altitudes than P. nigra. In contrast, propolis obtained from tropical regions (particularly South America) are dominated by prenylated p-coumaric acid derivatives, flavonoids, benzophenones, lignans and terpenes (Popova et al. 2009). Polyprenylated benzophenones and lignans are characteristic of samples from tropical regions (Fig. 4). The observed chemical composition variation between propolis from temperate and tropical zones has been attributed to differences in vegetation sources (Burdock 1998; Banskota et al. 2000). Samples from Mediterranean countries, such as Croatia, Algeria, Greece (Island of Evia), and Cyprus, exhibit a somewhat mixed chemical profile, distinguished by high concentrations of diterpenoids and the absence (or very low concentrations) of flavonoids (Trusheva et al. 2007; Popova et al. 2010b). Pacific propolis samples obtained from Taiwan and Japan (Okinawa) were found to contain predominantly prenylated flavanones, which is probably linked to the botanical source, Macaranga tanarius (Shahinozzaman et al. 2021). The chemical composition of samples from New Zealand showed resemblance to samples originating from Europe and North America (Markham et al. 1996).
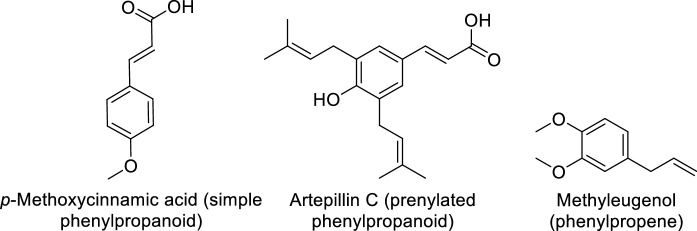
Fig. 4.
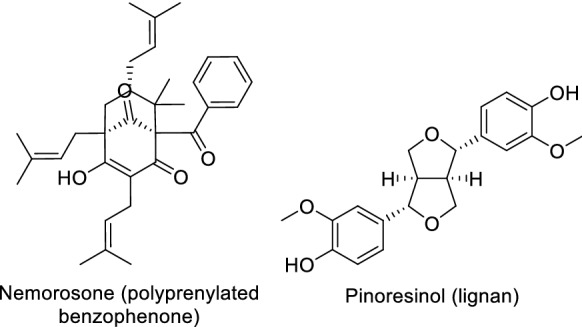
Polyprenylated benzophenones and lignans characteristic of tropical region propolis
Despite characteristic phytochemical similarities of propolis from within the same geographical zone, region-specific uniqueness and variation in chemical composition have also been observed. This phenomenon is most probably related to region-specific flora. An excellent example is Brazilian propolis, which has been classified into 12 different groups based on the geographical origin, plant source, and chemical composition (Park et al. 2000, 2002). The Brazilian propolis derived from bees frequenting Baccharis plant species contains mainly prenylated derivatives of p-coumaric acid, sesquiterpenes, acetophenones, diterpenes and lignans (Bankova 2005a, 2005b; Piccinelli et al. 2005). In addition, prenylated and straightforward p-coumaric acids, are unique to Brazilian green propolis (Fig. 5). Green and brown Brazilian propolis are the most common among the above-mentioned types of propolis. Green propolis, which is derived from Baccharis dracunculifolia, is found to be rich in prenylated phenylpropanoids, chlorogenic and benzoic acids, as well as triterpenoids (Righi et al. 2011). In contrast, Dalbergia ecastaphyllum is the source for red propolis; the pigments retusapurpurin A and B are responsible for this characteristic red colour (Piccinelli et al. 2011). This type contains isoflavonoids, prenylated benzophenones (from flowers of Clusia spp.) and naphthoquinone epoxide (Trusheva et al. 2006). Similar to Brazilian propolis, phytochemical variation has also been reported amongst Cuban propolis (Cuesta-Rubio et al. 2007). Based on their physicochemical characteristics, they are further divided into three types; brown, yellow, and red (Piccinelli et al. 2011). Polyisoprenylated benzophenones from the flowers of Clusia nemorosa and triterpenoids are the main components of brown and yellow propolis, respectively, whereas the red type contains mainly chalcones, pterocarpans, isoflavans, and isoflavones originating from the resin of Clusia and Dalbergia species (Piccinelli et al. 2005, 2011). In addition to Brazil and Cuba, samples from other countries on the South American continent, including Chile, Venezuela, Argentina, and Uruguay, have also been well studied. Chilean propolis reportedly contains several classes of compounds, such as phenylpropanes, benzaldehydes, dihydrobenzofurans, benzopyrans and lignans (Valcic et al. 1998, 1999). It has been reported that Chilean propolis originates from the species Eucalyptus and Ricinus and five native Chilean species, namely; Baccharis linearis, Buddleja globosa, Peumus boldus, Quillaja saponaria and Salix humboldtiana (Montenegro et al. 2001). Similar to samples from Brazil, Venezuelan propolis contains polyisoprenylated benzophenones, its plant sources are the flowers of Clusia major and Clusia minor (Tomás-Barberán et al. 1993; Cuesta-Rubio et al. 2007). Larrea nitida is the plant source for Andean Argentinian propolis, which contains characteristic epoxy lignans (Agüero et al. 2010).
Fig. 5.
Types of phenylpropanoids reported in Brazilian propolis
The bud exudates of poplar trees are the main floral source of propolis from temperate regions (North America, Europe, and West Asia); hence, propolis samples obtained from this region are generally termed Poplar propolis (Bankova et al. 2002). This propolis contains poplar bud phenolics, such as flavonoid aglycones (flavones and flavanones) and phenolic acids, and their esters (Bankova et al. 2002). Uruguayan propolis has a chemical composition similar to propolis of European and Chinese origin, which explains its classification into the Poplar class (Kumazawa et al. 2002). Populus nigra bud secretions are the major resin source for propolis from various European countries, such as France, Great Britain, Spain and Bulgaria (Bankova et al. 1992). However, some chemical variations have been observed among the same and different countries. This could be due to the other vegetal sources of resins for “Poplar-type” propolis, such as different Pinus spp., Prunus spp., Acacia spp. and also Betula pendula, Aesculus hippocastanum and Salix alba (Dezmirean et al. 2021). Variation in the chemical compositions of samples from various European countries has been reported, namely Bulgaria, Italy, and Switzerland; even though they displayed the characteristic typical chemical patterns of Poplar propolis (Bankova et al. 2000).
Portuguese propolis was found to contain a dimer of a p-coumaric ester derivative, together with four unique methylated, esterified, and/or hydroxylated derivatives of common poplar flavonoids, in addition to six unusual derivatives of pinocembrin/pinobanksin, including phenylpropanoic acid derivatives (Falcão et al. 2013). Propolis from the Canary Islands showed the presence of furofuran lignans, which are uncommon in European and Mediterranean propolis (Bankova et al. 1998). In addition to the typical compounds in Poplar-types, Australian and Canadian products contained chalcones, which are uncommon in propolis from temperate regions (Christov et al. 2006; Tran et al. 2012). The observed chemical composition of Russian propolis differ from that of Poplar propolis. This finding may be due to its different plant sources, namely Betula verrucosa and P. tremula (Christov et al. 2006). Consequently, phenylpropanoids, cinnamic acid esters, phenylpropanoid glycerides and sesquiterpenols, along with flavonoids and chalcones, were reported in Russian propolis samples (Isidorov et al. 2014).
Mediterranean propolis, obtained from counties on the Mediterranean Sea present a somewhat mixed and characteristic diterpene composition. Two chemotypes have been identified in the abovementioned Mediterranean countries; one rich in esters of phenolic acids (benzyl, phenylethyl and pentenyl caffeates) and flavonoids (pinocembrin, pinobanksin, pinobanksin-3-O-acetate, galangin)—e.g. the typical poplar type, and the second type abundant in diterpenes, including totarol, 13-epi-manool, iso-agatholal, agathadiol, communic acid, junicedric acid, 13-epi-cupressic acid, isocupressic acid (Popova et al. 2008; Piccinelli et al. 2013). The buds of poplars and Cupressus sempervirens resin are the main source of the phenolic and characteristic diterpene composition, respectively, of Mediterranean propolis (Velikova et al. 2000; Popova et al. 2011). The non‐volatile fraction of Mediterranean propolis was characterised by the presence of phenolic acids and their esters and flavonoids. However, diterpenes were also reported in propolis samples from countries including Italy, Croatia, Malta, Greece, Turkey, Cyprus, Egypt, Libya, Algeria and Morocco (El‐Guendouz et al. 2019).
Chinese propolis samples are reported to have a similar composition to that of European propolis. However, propolis samples obtained from Taiwan, Japan and the Solomon Islands reportedly contain some components that are not generally observed in propolis from other regions, such as Europe and Brazil (Kumazawa et al. 2008). These products are termed Pacific propolis and are characteristically rich in prenylated flavanones (Popova et al. 2010a). The fruit of M. tanarius are reportedly the resin source (Kumazawa et al. 2008; Popova et al. 2010b). Similar to Pacific propolis, Indonesian and Myanmar (Burmese) propolis were found to contain prenylflavanones and cycloartane-type triterpenes (Li et al. 2009; Trusheva et al. 2011). Macaranga tanarius and Mangifera indica were identified as the plant sources for Indonesian propolis (Trusheva et al. 2011).
Samples obtained from Nepal were rich in neoflavonoids, isoflavones, flavanones and pterocarpans (Awale et al. 2005; Shrestha et al. 2007). Five different coloured products (brown, green, green–brown, red, and red–brown) were reported among Indian samples; most displayed a chemical composition similar to those originating from temperate regions. Moreover, it was proposed that Indian samples be classified into two groups, northern and southern state propolis samples (Kasote et al. 2017).
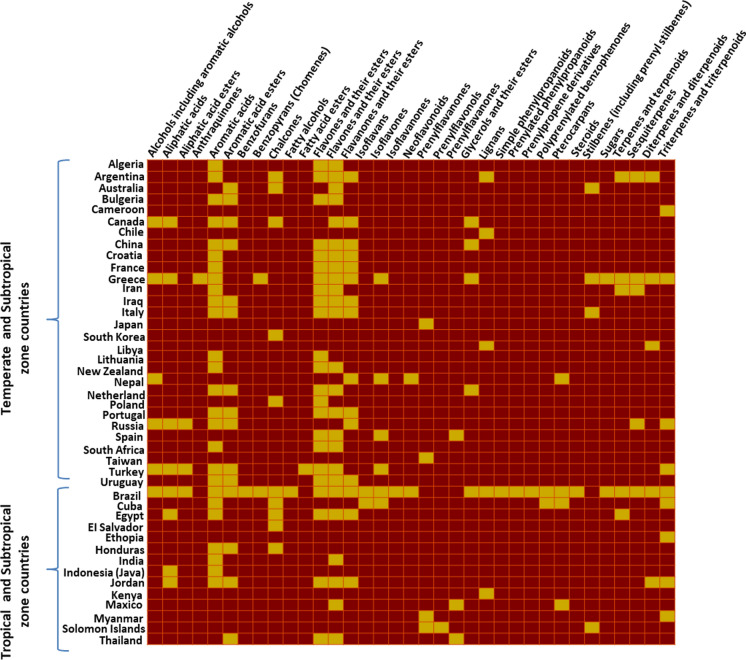
Available literature data was summarised in the form of a heat map to assist in understanding the chemical composition variation and uniqueness within propolis samples from different countries of the world (Fig. 6). Interestingly, it became evident that the chemical composition of propolis samples from temperate and subtropical climatic zones are very similar. However, no specific chemical composition similarity was revealed within samples from tropical or subtropical regions.
Fig. 6.
Heat map showing chemical composition variation within propolis samples studied from different countries of world. The yellow colouring indicates the presence, and the brown colouring, the absence, of specified compound classes. Temperate and subtropical climatic zone countries reflect a similar chemical composition. However, no distinct chemical composition is evident within tropical and subtropical climatic zone countries. (Color figure online)
Analytical methods employed in propolis quality control
Propolis is an important therapeutic and health-promoting agent that is usually consumed in the form of extracts or fractions. Thus, the setting of definite quality control parameters is a prerequisite for the marketing of propolis as a medicine and heath supplement. This step will help to ensure its uniform quality, stability and therapeutic efficacy. However, in reality, the qualitative and quantitative standardisation of propolis is difficult, due to its complex and varying chemical composition. Therefore, to guarantee the quality of propolis, it is necessary to have detailed information regarding the overall complex and varying chemistry of propolis, and the existing quality control strategies.
Propolis is not consumed in its natural form due to the presence of some inert material. Instead, it is generally purified with solvents to remove the inert material and to enrich the polyphenolic content (Pobiega et al. 2019). Numerous solvents, including water, ethanol, methanol, chloroform and hexane, have been used for the preparation of extracts and fractions; however, the use of diluted ethanol (70%) is most common (Pietta et al. 2002; Alencar et al. 2007; Cvek et al. 2007; Miguel and Antunes 2011; Sforcin and Bankova 2011; Elbaz and Elsayad 2012). Moreover, oils and natural deep eutectic solvents (NADES) have demonstrated promising potential (Bankova et al. 2021). Cvek et al. (2007) optimised the extraction conditions with respect to time, temperature, and concentration of extraction solvent, and found 80% ethanol and one hour at room temperature to yield the best results. In contrast, Pietta et al. (2002) reported that multi-step extraction with ethanol is a more suitable procedure to obtain dewaxed propolis that is rich in polyphenolics. The method of extraction was found to be a crucial step that defines not only the quality, but also the yield of the bioactive constituents of propolis. To date, various modern techniques, in addition to conventional maceration and Soxhlet extraction have been employed to increase the yield of the extract. Compared to the traditional maceration extraction method, MAE (microwave assisted extraction) and ultrasonic extraction (UE) provide high yields in a short timeframe and are less labour-intensive (Trusheva et al. 2007). Similarly, high-hydrostatic-pressure extraction (HHPE) has also been employed to concentrate flavonoids from propolis, and was found to be more effective than the conventional extraction methods in terms of time, efficiency and yield (Shouqin et al. 2005). Furthermore, bioactivity-guided selective fractionation of the hydroalcoholic extracts of propolis has also been conducted using both conventional and unconventional techniques, such as supercritical carbon dioxide extraction (Paviani et al. 2010). A recent review (Bankova et al. 2021) evaluated data generated from classical (maceration and Soxhlet) and modern (ultrasound-assisted and microwave-assisted extraction, supercritical CO2 extraction, high-pressure methods) propolis extraction methods, as well as potential large-scale applications.
Its region-specific chemical composition is one of the characteristic properties of propolis. Almost all types of reported propolis samples are unique and rich in a diversity of phytochemicals. The determination and identification of these unique and prominent constituents of propolis could be vital for its standardisation and chemical quality control (Fernandes-Silva et al. 2013). Several analytical techniques, including non-separation [ultraviolet spectroscopy (UV), near-infrared (NIR) and nuclear magnetic resonance (NMR) spectroscopy] and separation [gas chromatography (GC), high-performance liquid chromatography (HPLC), capillary electrophoresis (CE), and HPTLC (high-performance thin-layer chromatography] methods have been described in the literature for the analysis of propolis (Pellati et al. 2011). NMR spectroscopy is an important non-separation technique that has been used frequently to characterise propolis extracts. Several flavonoids from various fractions of Chinese propolis have been structurally elucidated using carbon-13 (13C) NMR after purification (Zhou et al. 1999). Proton (1H) NMR has been extensively applied to prepare chemical profiles of various propolis extracts, prepared from the Brazilian, Chinese, Greek and Indian-propolis. In combination with chemometric analysis, these datasets were used to classify propolis samples based on their chemical proximity (Maraschin et al. 2016; Kasote et al. 2017; Wang et al. 2020; Stavropoulou et al. 2021). A particular advantage of NMR in the chemical characterisation of propolis is that it helps to identify isomeric compounds (Pavlovic et al. 2020). Using similar chemometric techniques to that applied to the 1H NMR datasets, NIR spectroscopy data was used to classify Egyptian propolis into three types, namely the (O), (G) and (B) types (Shawky and Ibrahim 2018).
GC–MS is a powerful analytical technique that has been extensively used for the identification of both the volatile and the semi-volatile constituents of propolis for the last few decades (Bankova et al. 1995; Naik et al. 2013; Cruz et al. 2020). The use of GC–MS for the analysis of the volatile components of propolis is more common, and over 100 volatile constituents have been identified from propolis using GC–MS. Both volatile and semi-volatile compounds can be determined in propolis using this technique (Paviani et al. 2010; Nunes and Guerreiro 2012; Bankova et al. 2014; Balogun et al. 2020; Ribeiro et al. 2021). Spectroscopic data obtained from GC–MS analyses were processed with the help of multivariate analysis techniques and used to classify propolis samples according to regions and seasons. In addition, metabolic profiles were related to the biological activities (Nunes and Guerreiro 2012; Pavlovic et al. 2020; Ghallab et al. 2021).
Application of GC–MS to non-volatile propolis constituents is possible after derivatization (silylation). Non-volatile propolis constituents are mostly identified using liquid chromatography–mass spectrometry (LC–MS). This superior separating technique was used to identify 76 polyphenols in the ethanolic extracts of Portuguese samples from different geographical locations (Falcão et al. 2013). Chemical profiling is an important qualitative analysis parameter; LC–MS was used to demonstrate the chemical fingerprinting of propolis from different regions. The ethanolic extracts of propolis samples from Argentina, Italy, and Spain have approximately the same total ion chromatogram (TIC) profile due to the presence of the same molecular species, as determined using the online HPLC–ESI/MS technique (Volpi and Bergonzini 2006). In contrast, some researchers reported characteristic TIC profiles for samples from Azerbaijan, China, Ethiopia and Kenya (Volpi 2011). Chemometric analysis of the LC–MS data, involving the construction of unsupervised principal component analysis (PCA) and supervised orthogonal projection to latent structures-discriminant analysis (OPLS-DA) models, was found to be a valuable tool for the classification of propolis samples and for the identification of marker compounds in various types of propolis. Both the LC–MS chromatographic profiles and the 1H- and 13C NMR spectral data of the secondary metabolites of Cuban samples were used to classify them into different types, namely brown, red, and yellow Cuban propolis (Cuesta-Rubio et al. 2007). Chemometric analysis of ultra-performance liquid chromatography–mass spectrometry (UPLC–MS) data, generated from 39 South African propolis, revealed two distinct groups based on the sample chemistry. Moreover, the majority of the samples were phytochemically congruent with propolis from temperate regions (Kasote et al. 2014). Recently, 67 marker compounds, belonging to chemical classes, namely flavonoids, stilbenes, terpenoids, acid derivatives, steroid derivatives, and some miscellaneous components, were identified in propolis samples from Australia, China and Brazil using UPLC–MS in combination with chemometric analysis (Bhuyan et al. 2021). Capillary electrophoresis coupled to mass spectrometry (CE–MS), an important technique that provides selective information with regard to analytes, has also been used for the analysis of complex propolis extracts, but this technique is far less common (Gómez‐Romero et al. 2007).
Similar to LC–MS, TLC/HPTLC (thin layer chromatography/high-performance thin-layer chromatography) have also been used for fingerprinting, as well as for the qualitative and quantitative determination of the individual components of propolis. The floral and geographic origins of Romanian samples were identified using TLC fingerprinting and imaging (Sârbu and Moţ 2011). Similarly, reversed-phase high-performance thin-layer chromatography (RP-HPTLC), reversed-phase HPLC and GC–MS, were collectively used to identify the botanical origin and chemical composition of Brazilian samples (Daugsch et al. 2007). The HPTLC fingerprints of 35 Egyptian samples, obtained after derivatisation and fluorescence detection (FLD) at λ 366 nm, were analysed using multivariate data analysis (Shawky and Ibrahim 2018). The data were used to identify nine marker compounds for the different sample clusters. Using the HPTLC-bioautography-MS technique, pinobanksin, pinocembrin, and caffeic acid were identified in South African propolis, as antibacterial, antifungal and anti-quorum components, respectively (Kasote et al. 2015).
Due to the complex chemical compositions, the quantitative determination of the chemical constituents in propolis samples has not attracted much interest. Hence, spectroscopic and chromatographic analysis methods, in addition to the use of reference standards, are a more routine and valid method for the analysis of propolis samples. A single reference-standard-based chemical standardisation method is not appropriate for propolis; hence, the need to identify multiple standards for various propolis types, according to their corresponding chemical profiles, has been proposed (Popova et al. 2010a). A validated spectrophotometric method was developed to quantify total flavones, flavonols, flavanones, dihydroflavonols and total phenolics, based on aluminum chloride complexation (Popova et al. 2004). Similarly, a DNP (dinitrophenylhydrazine) spectrophotometric method was reported to quantify prenylated flavanones in Pacific-type propolis from Taiwan (Popova et al. 2004). For routine analyses of Macaranga-type propolis, a HPLC-based method for quantifying propolins C, D, F and G was recommended for samples from Taiwan (Popova et al. 2010a).
Challenges in propolis quality control: biomarker selection
Propolis has received increasing attention from researchers over the last few decades, due to its broad spectrum of therapeutic activities and potential commercial applications in food, cosmetic, and health care preparations. The therapeutic potential of various types of propolis with different chemical compositions has been assessed worldwide. However, these data have not resulted in the universal acceptance of propolis as an authenticated drug or health-promoting product in international food and healthcare markets. For this, the lack of common uniform quality control parameters for propolis is an important contributing factor. The missing link between the marker compound(s) for propolis and respective therapeutic potential is one of the major hurdles in quality control.
In general, marker compounds for herbal products are selected based on their uniqueness, abundance and/or specific biological activity. However, the complex and variable chemistry of propolis prevents the compilation of uniform propolis quality control parameters, although multiple-marker-compound-targeted approach that enables the characterising of propolis types would be appropriate to ensure quality products. Potential marker compounds related to geographical origin and bioactivity, according to the propolis type, are summarised in Table 1.
Table 1.
Propolis type, including geographical zone, country of origin, botanical source(s) and marker compounds (in bold) characterising origin and biological activity, reported for propolis
| Geographical zone | Major plant source(s) | Country of origin | Key compounds | Markers compounds and bioactivity | References |
|---|---|---|---|---|---|
| Temperate | Birch | Russia, Poland | Flavones and flavonols different from Poplar-type | Bioflavonoids (anti-oxidant activity) | Gromenko et al. (2008) and Sforcin and Bankova (2011) |
| Poplar-(Populus tremuloides, P. trichocarpa) | Canada | Aliphatic acids, aromatic acids, chalcones, flavonols and flavanones | Benzyl hydroxybenzoate (anti-oxidant); Cinnamic acid (anti-oxidant); Dihydrochalcones (anti-oxidant); Galangin (anti-inflammatory, antifungal); p-Hydroxyacetophenone (anti-oxidant) | Blonska et al. (2004) and Christov et al. (2006) | |
| Poplar (P. nigra, P. × euramercana, P. candaensis—China) | Britain, China, Germany, France, India, Italy, Netherlands, New Zealand, Portugal, South Africa, Spain, Switzerland, Uruguay, USA | Cinnamic acid, caffeic acid, p-coumaric acid, flavanones, flavones and flavonols | Benzyl caffeate (antiproliferative); Caffeic acid (immunomodulatory, anti-quorum sensing, anti-oxidant); Caffeic acid phenethyl ester (immunomodulatory antitumor, anti-oxidant, antidiabetic, anti-inflammatory; neuroprotective); Chrysin (anti-oxidant, anti-inflammatory, anticancer); Cinnamic acid (antifungal); p-Coumaric acid (antifungal); Cinnamyl caffeate (antiproliferative); 3,4-Dimethoxycinnamic acid (antifungal); Ferulic acid (antifungal); Galangin (antifungal); Kaempferol (anti-inflammatory, antifungal); Morin (antifungal); Naringenin (antifungal); Pinobanksin (antibacterial); Pinocembrin (anti-oxidant antifungal); Quercetin (immunomodulatory, antimetastatic, anti-inflammatory); Rhamnetin (cytotoxic) | Banskota et al. (2002), Blonska et al. (2004), Márquez et al. (2004), Oršolić et al. (2004), Park et al. (2004), Okutan et al. (2005), Oršolić et al. (2006), Quiroga et al. (2006), Sha et al. (2009), Guo et al. (2011), Sawicka et al. (2012), Silva et al. (2013), Kasote et al. (2015) and Kasote et al. (2017) | |
| Sub-tropical | Poplar (P. nigra, P. euphatica) | Turkey | Aliphatic and aromatic acids, alcohols, fatty acid esters, hydrocarbons, flavones, flavonols and ketones | Caffeic acid and its esters (antimicrobial); Galangin (anti-inflammatory); Chrysin (anti-oxidant, anticancer, anti-inflammatory); Quercetin (immunomodulatory, antimetastatic) | Kartal et al. (2003), Blonska et al. (2004), Oršolić et al. (2004), Uzel et al. (2005), Duran et al. (2011) and Sawicka et al. (2012) |
| Lepidosperma spp., Acacia paradoxa | Australia | Cinnamic acid esters, chalcones, flavonols and stilbenes | Prenylated cinnamate (anti-oxidant); Stilbenes (anti-oxidant) | Abu-Mellal et al. (2012) and Tran et al. (2012) | |
| Poplar, unknown source of triterpenes | Egypt | Aliphatic acid, aromatic acid, chalcones, flavones, flavonols and their esters, and triterpenes | Chrysin (anti-oxidant, anticancer, anti-inflammatory); Aromatic acid esters (anti-oxidant, antimicrobial, antiviral); Triterpenoids (anti-oxidant, antimicrobial, antiviral) | Abd El Hady and Hegazi (2002), Hegazi and Abd El Hady (2002) and Sawicka et al. (2012) | |
| Ferula spp., Poplar | Iran | Caffeic acid phenethyl ester, flavonols, mono- and sesquiterpene esters of benzoic acids, prenylated coumarin and suberosin | Kaempferol (anti-inflammatory); Mono- and sesquiterpene esters of benzoic acids (antibacterial) | Blonska et al. (2004) and Trusheva et al. (2010) | |
| Poplar, unknown source of triterpenes | Jordan | Aromatic acid, flavones, flavanones and sesquiterpenoids | Chrysin (anti-oxidant, anticancer, anti-inflammatory) | Shaheen et al. (2011) and Sawicka et al. (2012) | |
| Cupressus sempervirens, minor source—poplar (Mediterranean) | Greece, Algeria, Croatia, Cyprus, Malta | Diterpenes, aromatic acids and their esters, flavonols and flavanones | Diterpenes (antimicrobial) | Velikova et al. (2000), Sahinler and Kaftanoglu (2005), Popova et al. (2009), Popova et al. (2010b) and Popova et al. (2011) | |
| Dalbergia sissoo | Nepal | Flavanones, isoflavones, neoflavonoids and pterocarpans | Neoflavonoids (inhibition of nitric oxide production) | Awale et al. (2005) and; Shrestha et al. (2007) | |
| Pacific (Macaranga tanarius) | Japan (Okinawa), Taiwan | Prenylflavanones | Propolin A (cytotoxic and anti-oxidant); Propolin B (cytotoxic and anti-oxidant); Propolin D (antimicrobial); Propolin G (antimicrobial, cytotoxic and anti-oxidant); Propolin H (antimicrobial) | Chen et al. (2003), Chen et al. (2004), Kumazawa et al. (2004), Kumazawa et al. (2007) and Raghukumar et al. (2010) | |
| Tropical | Poplar | Argentina | Aromatic acids and their esters, chalcone, epoxy lignans, flavones and flavonols | Chrysin (anticancer, anti-inflammatory); 2′, 4′-Dihydroxychalcone (anti-oxidant and antibacterial); 2′,4′-Dihydroxy 3′-methoxychalcone (anti-oxidant and antibacterial); Kaempferol (anti-inflammatory); Pinocembrin (antifungal); Quercetin (immunomodulatory, antimetastatic) | Blonska et al. (2004), Oršolić et al. (2004), Isla et al. (2005), Quiroga et al. (2006) and Agüero et al. (2011) |
| Baccharis dracunculifolia | Brazil (Brazilian Green) | Benzoic and chlorogenic acids, prenylated phenylpropanoids (e.g. artepillin C) | Artepillin C (anti-tumor and anticancer, vaccine adjuvants); Baccharin (antitumor); Caffeic acid (anti-ulcer); Chrysin (anticancer, anti-inflammatory); Cinnamic acids (anti-ulcer); p-Coumaric acid (anti-ulcer); Coniferyl aldehyde (cytotoxic); Drupanin (antitumor); Ferulic acid (anti-ulcer); Kaempferide (cytotoxic); Quercetin (immunomodulatory, antimetastatic); 3,5,7-Trihydroxy-4’-methoxyflavanol (cytotoxic); | Bankova et al. (1996), Banskota et al. (1998), Blonska et al. (2004), Oršolić et al. (2004), Bankova (2005a), Mishima et al. (2005), Piccinelli et al. (2005), Trusheva et al. (2006), Awale et al. (2008), Li et al. (2008), Castro et al. (2009), Messerli et al. (2009), Fischer et al. (2010), Teixeira et al. (2010), Righi et al. (2011), Endo et al. (2012), Sawicka et al. (2012), Silva et al. (2013), Bueno-Silva et al. (2013) and Olegário et al. (2019) | |
| Dalbergia ecastaphyllum, Clusia spp. | Brazil (Brazilian red) | Isoflavonoids, phenolics, prenylated benzophenones and triterpenoids | Hyperibone A (cytotoxic and antimicrobial); (6aR,11aR)-3,8-Dihydroxy-9-methoxypterocarpan (cytotoxic); Moronic acid (anti-HIV); Mucronulatol (cytotoxic); Neovestitol (anti-inflammatory and antimicrobial); Vestitol (anti-inflammatory and antimicrobial) | ||
| Araucaria angistifolia, Eucalyptus spp. | Brazil (Brazilian brown) | Ferulic acid, caffeic acid, 4-methoxycinnamic acid, 3,4-dimethoxycinnamic acid, 3-hydroxy-4-methoxybenzaldehyde, 3-methoxy-4-hydroxypropiophenone,19-acetoxy-13-hydroxyabda-8(17),14-diene, totarol, 7-oxodehydroabietic acid, dehydroabietic acid, communic acid, isopimaric acid, | Communic acid (antimicrobial, antiplasmodial), isopimaric acid (cytotoxic, antimicrobial), imbricataloic acid (antimicrobial) | ||
| Mangifera indica, Macaranga tanarius | Indonesia | Alk(en)ylresorcinols, cycloartane-type triterpenes and prenylflavanones | Prenylflavanones (anti-oxidant, antimicrobial) | Trusheva et al. (2010) | |
| Mangifera indica | Thailand | alk(en)ylresorcinols, anacardic acids, cycloartane-type triterpenes | Alk(en)ylresorcinols, anacardic acids (antibacterial) | Sanpa et al. (2017) | |
| Mangifera indica | Myanmar | Cycloartane-type triterpenes | (22Z,24E)-3-Oxocycloart-22,24-dien-26-oic acid (cytotoxic); (2S)-5,7-dihydroxy-4′-methoxy-8,3′-diprenylflavanone (cytotoxic) | Li et al. (2009) | |
| Unknown | Chile | Benzaldehyde, benzopyran or dihydrobenzofuran and phenylpropane | Benzaldehyde, benzopyran or dihydrobenzofuran and phenylpropane (antimicrobial) | Valcic et al. (1999) | |
| Dalbergia ecastaphyllum | Cuba (Cuban red) | Isoflavones, isoflavanes, pterocarpans | Isoflavonoids (antibacterial and antifungal, anticancer) | Rubio et al. (1999), Hernández et al. (2005), Piccinelli et al. (2005), Monzote et al. (2012) and Díaz-Carballo et al. (2013) | |
| Clusia rosea, Clusia nemorosa | Cuba (Cuban brown) | Polyprenylated benzophenones | Polyprenylated benzophenones (antimicrobial, anticancer) | ||
| Unknown | El Salvador | Chalcones and diterpene glycosides | 2′,3′-Dihydroxy-4,4′-dimethoxychalcone (antibacterial and antifungal); ent-8(17)-Labden-15-O-(3′-O-acetyl)-alpha-l-rhamnoside (antibacterial); ent-8(17)-Labden-15-O-alpha-l-rhamnoside (antibacterial); 2′,3′,4-Trihydroxy-4′-methoxy-chalcone (antibacterial and antifungal) | Popova et al. (2001b) | |
| Macaranga schweinfurthii | Kenya | Arylnaphthalene, geranylstilbenes, geranylflavone and lignans | Geranylstilbenes (antibacterial); Geranylflavon (anti-oxidant) | Petrova et al. (2010) | |
|
Dalbergia unknown source |
Mexico | Diphenyl propanes, flavanones, isoflavans and pterocarpans | (2R,3R)-3,5-Dihydroxy-7-methoxyflavanone 3-(2-methyl)butyrate(cytotoxic); (2R,3R)-6-[1-(4′-Hydroxy-3'-methoxyphenyl)prop-2-en-1-yl]pinobanksin (cytotoxic); (2R,3R)-6-[1-(4′-Hydroxy-3′-methoxyphenyl)prop-2-en-1-yl]pinobanksin 3-acetate (cytotoxic); (7″R)-8-[1-(4'-Hydroxy-3′-methoxyphenyl)prop-2-en-1-yl]pinobanksin (cytotoxic); (7″R)-8-[1-(4'-Hydroxy-3′-methoxyphenyl)prop-2-en-1-yl]galangin (cytotoxic); Chrysin (cytotoxic); (2R,3S)-8-[4-Phenylprop-2-en-1-one]-4′,7-dihydroxy-3′,5-dimethoxyflavan-3-ol (cytotoxic) | Lotti et al. (2010) and Li et al. (2011) | |
| Macaranga spp. | Solomon Islands | Prenylated flavonoids and prenylated stilbene | Bonannione A (anti-oxidant); (2S)-5,7-Dihydroxy-4′-methoxy-8-prenylflavanone (anti-oxidant); Solomonin (antibacterial); ++ Solophenol A (anti-oxidant; antibacterial); Solophenol B (antibacterial); Solophenol C (antibacterial); Solophenol D (antibacterial); Sophoraflavanone A (anti-oxidant) | Inui et al. (2012) | |
| Clusia spp. | Venezuela | Flavonoids and polyprenylated benzophenones | Acetyl triterpenes (antimicrobial); Scrobiculatones A (antibacterial) Scrobiculatones B (antibacterial) | Righi et al. (2011), Monzote et al. (2012) and Tomás-Barberán et al. (1993) |
The phytochemical uniqueness and variation in propolis samples from different places of world have been reported, and which provide an opportunity to propose geographical markers for their standardisation. Such marker(s) could be a single compound or a group of compounds that are supposedly unique to, and abundant in, certain types of propolis. However, there is a need to consider the physicochemical properties and phytochemical variations between different propolis types during the establishment of geographical markers. For example, Brazilian and Cuban propolis exhibit different physicochemical properties within their sub-types. Similarly, Canadian propolis showed the presence of chalcones, in addition to the typical Poplar-type chemical composition.
A broad range of biological activities, such as anti-oxidant, antimicrobial, anti-inflammatory, immunomodulatory, and anticancer activities, have been ascribed to propolis and its isolated chemical constituents (Oršolić et al. 2004; Okutan et al. 2005; Monzote et al. 2012; Bueno-Silva et al. 2013). Both propolis solvent extracts and their isolated components have been found to exert promising bioactivities; however, the correlation between propolis quality and biological activity has not been investigated in enough depth.
In general, it is expected that propolis with different chemical compositions would have different bioactivities. However, existing reports on the chemistry-dependent biological activity of propolis are controversial. Propolis samples from temperate and tropical zones showed similar antibacterial, antifungal, and antiviral activities, despite their diverse chemical compositions (Kujumgiev et al. 1999). Conversely, Brazilian propolis observed stronger bactericidal activity than that from Bulgaria, and these authors attributed this region-wise dissimilar bactericidal potential to their different chemical compositions (Orsi et al. 2005). The probable reason for these conflicting results could be differences in propolis quality-defining parameters, such as the available plant source, climatic conditions and extraction conditions. The antibacterial activities of propolis samples from different geographical climatic zones, such as tropical, subtropical and temperate, against a range of Gram-positive and Gram-negative bacteria were comparatively investigated and found that propolis samples from tropical regions had varying activity profiles, which may be due to the climatic characteristics and the available resin plant sources of the collection sites (Seidel et al. 2008). In addition, variations in bioactivity have also been observed within similar geographical climatic zone countries (Seidel et al. 2008). Samples from Austria, Germany, and France display diverse antimicrobial activities, despite some similarities in their qualitative composition. These researchers attributed this variation in antimicrobial activity to the predominant chemical compounds (Hegazi et al. 2000). The findings of this study further showed that phenylethyl-trans-caffeate, benzyl ferulate, and galangin are predominant in German propolis, whereas benzyl caffeate and pinocembrin are predominant in French and Austrian samples.
It is important to note that these differences could be due to a large extent to the difference in extraction procedures and protocols for performing biological tests.
In addition to crude propolis extracts, the chemical compounds isolated from propolis samples have been found to be encouraging with respect to their biological potential. In fact, most of the chemical compounds isolated were reported to have specific biological activity (Table 1). Caffeic acid phenethyl ester (CAPE) and chrysin from temperate propolis, and artepillin C from Brazilian propolis, have been extensively studied and show promising therapeutic potential in the treatment of cancer, degenerative diseases, and other pathologies (Okutan et al. 2005; Messerli et al. 2009; Li et al. 2011). These compounds could have significance in the biological-activity-marker-based standardisation of temperate and Brazilian propolis, respectively. Moreover, other types of propolis also exhibit the presence of specific bioactive compounds that could be used as biological marker compounds for the standardisation of the respective propolis types (Table 1).
Conclusions and future perspectives
In most cases, one or more analytical markers based on its uniqueness, abundance and specific bioactivity, are assigned for the quality assessment of a herbal product. However, in the case of propolis, more than 800 different phytoconstituents have been reported in samples from around the world, and these are found to vary with geography and season. Furthermore, available literature reports have not demonstrated whether the specific therapeutic potential of propolis is associated with a certain chemical entity. Therefore, there is a need to adapt a quality control strategy for propolis standardisation which will include quantitative botanical, geographical, and biological activity marker(s). Chromatographic fingerprinting methods, such as LC and HPTLC, are other tools that can provide valuable information regarding the origin of samples. Considering this, we propose that these techniques be emphasised as primary quality control parameters for propolis standardisation.
To date, researchers have only considered two major geographical zones, temperate and tropical, for the classification of propolis. We propose that propolis obtained from subtropical zones, including the Mediterranean and Pacific regions, exhibit a somewhat mixed chemical composition, and should be studied in greater detail.
Various techniques, such as HPTLC, GC–MS, LC–MS, CE–MS, and NMR, have been found to be useful for establishing the chemical profiles of propolis samples. However, due to the complex and variable chemical composition of propolis, there is a need to strengthen the propolis quality assessment techniques and use defined solvent extraction methods to improve overall efficacy of methods. Chemometric techniques have been successfully used in herbal drug quality control; and have been found to be promising for the quality assessment of propolis. These techniques can also be valuable for unravelling the link between the composition and the bioactivity, including to understand synergetic interactions. Multivariate statistical tools, such as cluster heat maps, PCA, and linear discriminant analysis, were used to classify different Portuguese samples according to their botanical/geographical origin (Dias et al. 2012). According to their geographical origin and the local flora, the fuzzy divisive hierarchical clustering approach was employed for the discrimination and fingerprinting of Romanian samples (Sârbu and Moţ 2011). Vibrational spectroscopic techniques (infrared, NIR Raman spectroscopy and hyperspectral imaging), combined with chemometric techniques, may also be powerful techniques for the quality assessment of large numbers of samples in a short time (Sandasi et al. 2011; Shikanga et al. 2013).
However, these studies were performed on propolis samples from a relatively small territory and remain of limited regional importance. Their results can hardly be used with respect to propolis from other geographic regions without analysis of a new sample set. Chemometric approaches have a promising potential but the results should be discussed and used with caution.
In conclusion, propolis contains more than 800 different chemical components with complex and varying chemistry, and hence, the maintenance of its quality at one global or regional platform remains challenging. A multiple-marker assessment strategy with chemometric analysis may be a promising approach for propolis quality assessment.
Supplementary Information
Below is the link to the electronic supplementary material.
Abbreviations
- COVID-19
Coronavirus disease
- DNP
Dinitrophenylhydrazine
- EC-MS
Electrophoresis coupled to mass spectrometry
- ESI–MS
Mass spectrometry with electrospray ionization
- GC–MS
Gas chromatography–mass spectrometry
- HHPE
High-hydrostatic-pressure extraction
- HPTLC
High-performance thin layer chromatography
- LC-DAD-ESI–MS
Diode-array detection coupled to electrospray ionisation tandem mass spectrometry
- LC–MS
Liquid chromatography–mass spectrometry
- MAE
Microwave-assisted extraction
- NIR
Near-infrared
- NMR
Nuclear magnetic resonance
- OPLS-DA
Orthogonal projections to latent structures discriminant analysis
- PCA
Principal component analysis
- RPHPLC
Reversed-phase high-performance liquid chromatography
- RPHPTLC
Reversed-phase high-performance thin-layer chromatography
- SARS-CoV-2
Severe acute respiratory syndrome coronavirus 2
- TLC
Thin layer chromatography
- UV
Ultraviolet spectroscopy
- UE
Ultrasonic extraction
- UPLC–MS
Ultra-performance liquid chromatography–mass spectrometry
Author contributions
DK: Developed review concept, wrote the manuscript and revised according to comments and suggestions; AV: Contributed to conceptualisation, acted as supervisor, reviewed and edited the document; VB: reviewed and edited the manuscript.
Funding
DMK is thankful to Tshwane University of Technology for providing a Post-doctoral Fellowship.
Declarations
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Abd El Hady FK, Hegazi AG. Egyptian propolis: 2. Chemical composition, antiviral and antimicrobial activities of East Nile Delta propolis. Z Nat C. 2002;57(3–4):386–394. doi: 10.1515/znc-2002-3-431. [DOI] [PubMed] [Google Scholar]
- Abu-Mellal A, Koolaji N, Duke RK, Tran VH, Duke CC. Prenylated cinnamate and stilbenes from Kangaroo Island propolis and their antioxidant activity. Phytochemistry. 2012;77:251–259. doi: 10.1016/j.phytochem.2012.01.012. [DOI] [PubMed] [Google Scholar]
- Adewumi A, Ogunjinmi A. The healing potential of honey and propolis lotion on septic wounds. Asian Pac J Trop Biomed. 2011;1(1):S55–S57. doi: 10.1016/S2221-1691(11)60123-8. [DOI] [Google Scholar]
- Agüero MaBn, Gonzalez M, Lima B, Svetaz L, Sanchez M, Zacchino S, et al. Argentinean propolis from Zuccagnia punctata Cav. (Caesalpinieae) exudates: phytochemical characterization and antifungal activity. J Agric Food Chem. 2010;58(1):194–201. doi: 10.1021/jf902991t. [DOI] [PubMed] [Google Scholar]
- Agüero MB, Svetaz L, Sánchez M, Luna L, Lima B, López ML, et al. Argentinean Andean propolis associated with the medicinal plant Larrea nitida Cav. (Zygophyllaceae). HPLC–MS and GC–MS characterization and antifungal activity. Food Chem Toxicol. 2011;49(9):1970–1978. doi: 10.1016/j.fct.2011.05.008. [DOI] [PubMed] [Google Scholar]
- Ahn M-R, Kumazawa S, Usui Y, Nakamura J, Matsuka M, Zhu F, et al. Antioxidant activity and constituents of propolis collected in various areas of China. Food Chem. 2007;101(4):1383–1392. doi: 10.1016/j.foodchem.2006.03.045. [DOI] [Google Scholar]
- Alencar S, Oldoni TC, Castro M, Cabral I, Costa-Neto C, Cury J, et al. Chemical composition and biological activity of a new type of Brazilian propolis: red propolis. J Ethnopharmacol. 2007;113(2):278–283. doi: 10.1016/j.jep.2007.06.005. [DOI] [PubMed] [Google Scholar]
- Aliboni A, D’Andrea A, Massanisso P. Propolis specimens from different locations of central Italy: chemical profiling and gas chromatography–mass spectrometry (GC–MS) quantitative analysis of the allergenic esters benzyl cinnamate and benzyl salicylate. J Agric Food Chem. 2011;59(1):282–288. doi: 10.1021/jf1034866. [DOI] [PubMed] [Google Scholar]
- Anjum SI, Ullah A, Khan KA, Attaullah M, Khan H, Ali H, et al. Composition and functional properties of propolis (bee glue): a review. Saudi J Biol Sci. 2019;26(7):1695–1703. doi: 10.1016/j.sjbs.2018.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Athikomkulchai S, Awale S, Ruangrungsi N, Ruchirawat S, Kadota S. Chemical constituents of Thai propolis. Fitoterapia. 2013;88:96–100. doi: 10.1016/j.fitote.2013.04.008. [DOI] [PubMed] [Google Scholar]
- Awale S, Li F, Onozuka H, Esumi H, Tezuka Y, Kadota S. Constituents of Brazilian red propolis and their preferential cytotoxic activity against human pancreatic PANC-1 cancer cell line in nutrient-deprived condition. Bioorg Med Chem. 2008;16(1):181–189. doi: 10.1016/j.bmc.2007.10.004. [DOI] [PubMed] [Google Scholar]
- Awale S, Shrestha SP, Tezuka Y, Ueda J-y, Matsushige K, Kadota S. Neoflavonoids and related constituents from Nepalese propolis and their nitric oxide production inhibitory activity. J Nat Prod. 2005;68(6):858–864. doi: 10.1021/np050009k. [DOI] [PubMed] [Google Scholar]
- Balogun OS, Oluduro AO, Liu Z. Chemical composition and in vitro biological studies of volatile oils from Nigerian bee propolis. J Apic Res. 2020 doi: 10.1080/00218839.2020.1862392. [DOI] [Google Scholar]
- Bankova V. Chemical diversity of propolis and the problem of standardization. J Ethnopharmacol. 2005;100(1–2):114–117. doi: 10.1016/j.jep.2005.05.004. [DOI] [PubMed] [Google Scholar]
- Bankova V. Recent trends and important developments in propolis research. Evid Based Complement Altern Med. 2005;2(1):29–32. doi: 10.1093/ecam/neh059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bankova V, Boudourova-Krasteva G, Popov S, Sforcin J, Funari SC. Seasonal variations in essential oil from Brazilian propolis. J Essent Oil Res. 1998;10(6):693–696. doi: 10.1080/10412905.1998.9701012. [DOI] [Google Scholar]
- Bankova V, Christov R, Kujumgiev A, Marcucci M, Popov S. Chemical composition and antibacterial activity of Brazilian propolis. Z Nat C. 1995;50(3–4):167–172. doi: 10.1515/znc-1995-3-402. [DOI] [PubMed] [Google Scholar]
- Bankova V, Dyulgerov A, Popov S, Evstatieva L, Kuleva L, Pureb O, et al. Propolis produced in Bulgaria and Mongolia: phenolic compounds and plant origin. Apidologie. 1992;23(1):79–85. doi: 10.1051/apido:19920109. [DOI] [Google Scholar]
- Bankova V, Marcucci M, Simova S, Nikolova N, Kujumgiev A, Popov S. Antibacterial diterpenic acids from Brazilian propolis. Z Nat C. 1996;51(5–6):277–280. doi: 10.1515/znc-1996-5-602. [DOI] [PubMed] [Google Scholar]
- Bankova V, Popova M, Bogdanov S, Sabatini A-G. Chemical composition of European propolis: expected and unexpected results. Z Nat C. 2002;57(5–6):530–533. doi: 10.1515/znc-2002-5-622. [DOI] [PubMed] [Google Scholar]
- Bankova V, Popova M, Trusheva B. Propolis volatile compounds: chemical diversity and biological activity: a review. Chem Cent J. 2014;8(1):1–8. doi: 10.1186/1752-153X-8-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bankova V, Trusheva B, Popova M. Propolis extraction methods: a review. J Apic Res. 2021 doi: 10.1080/00218839.2021.1901426. [DOI] [Google Scholar]
- Bankova VS, de Castro SL, Marcucci MC. Propolis: recent advances in chemistry and plant origin. Apidologie. 2000;31(1):3–15. doi: 10.1051/apido:2000102. [DOI] [Google Scholar]
- Banskota A, Tezuka Y, Adnyana I, Midorikawa K, Matsushige K, Massage D, et al. Biological evaluation of propolis from Brazil, Peru, The Netherlands and China. J Ethnopharmacol. 2000;72(239):e246. doi: 10.1016/S0378-8741(00)00252-X. [DOI] [PubMed] [Google Scholar]
- Banskota AH, Nagaoka T, Sumioka LY, Tezuka Y, Awale S, Midorikawa K, et al. Antiproliferative activity of the Netherlands propolis and its active principles in cancer cell lines. J Ethnopharmacol. 2002;80(1):67–73. doi: 10.1016/S0378-8741(02)00022-3. [DOI] [PubMed] [Google Scholar]
- Banskota AH, Tezuka Y, Prasain JK, Matsushige K, Saiki I, Kadota S. Chemical constituents of Brazilian propolis and their cytotoxic activities. J Nat Prod. 1998;61(7):896–900. doi: 10.1021/np980028c. [DOI] [PubMed] [Google Scholar]
- Barbarić M, Mišković K, Bojić M, Lončar MB, Smolčić-Bubalo A, Debeljak Ž, et al. Chemical composition of the ethanolic propolis extracts and its effect on HeLa cells. J Ethnopharmacol. 2011;135(3):772–778. doi: 10.1016/j.jep.2011.04.015. [DOI] [PubMed] [Google Scholar]
- Basnet P, Matsushige K, Hase K, Kadota S, Namba T. Four di-O-caffeoyl quinic acid derivatives from propolis. Potent hepatoprotective activity in experimental liver injury models. Biol Pharm Bull. 1996;19(11):1479–1484. doi: 10.1248/bpb.19.1479. [DOI] [PubMed] [Google Scholar]
- Bertelli D, Papotti G, Bortolotti L, Marcazzan GL, Plessi M. 1H NMR simultaneous identification of health-relevant compounds in Propolis extracts. Phytochem Anal. 2012;23(3):260–266. doi: 10.1002/pca.1352. [DOI] [PubMed] [Google Scholar]
- Bhadauria M. Propolis prevents hepatorenal injury induced by chronic exposure to carbon tetrachloride. Evid Based Complement Altern Med. 2012 doi: 10.1155/2012/235358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhuyan DJ, Alsherbiny MA, Low MN, Zhou X, Kaur K, Li G, et al. Broad-spectrum pharmacological activity of Australian propolis and metabolomic-driven identification of marker metabolites of propolis samples from three continents. Food Funct. 2021;12(6):2498–2519. doi: 10.1039/D1FO00127B. [DOI] [PubMed] [Google Scholar]
- Blonska M, Bronikowska J, Pietsz G, Czuba Z, Scheller S, Krol W. Effects of ethanol extract of propolis (EEP) and its flavones on inducible gene expression in J774A. 1. Macrophages. J Ethnopharmacol. 2004;91(1):25–30. doi: 10.1016/j.jep.2003.11.011. [DOI] [PubMed] [Google Scholar]
- Bueno-Silva B, Alencar SM, Koo H, Ikegaki M, Silva GV, Napimoga MH, et al. Anti-inflammatory and antimicrobial evaluation of neovestitol and vestitol isolated from Brazilian red propolis. J Agric Food Chem. 2013;61(19):4546–4550. doi: 10.1021/jf305468f. [DOI] [PubMed] [Google Scholar]
- Burdock G. Review of the biological properties and toxicity of bee propolis (propolis) Food Chem Toxicol. 1998;36(4):347–363. doi: 10.1016/S0278-6915(97)00145-2. [DOI] [PubMed] [Google Scholar]
- Cao Y, Wang Y, Yuan Q. Analysis of flavonoids and phenolic acid in propolis by capillary electrophoresis. Chromatographia. 2004;59(1):135–140. doi: 10.1365/s10337-003-0138-z. [DOI] [Google Scholar]
- Castro ML, do Nascimento, AM, Ikegaki, M, Costa-Neto, CM, Alencar, SM, Rosalen, PL, Identification of a bioactive compound isolated from Brazilian propolis type 6. Bioorg Med Chem. 2009;17(14):5332–5335. doi: 10.1016/j.bmc.2009.04.066. [DOI] [PubMed] [Google Scholar]
- Chan GC-F, Cheung K-W, Sze DM-Y. The immunomodulatory and anticancer properties of propolis. Clin Rev Allergy Immunol. 2013;44(3):262–273. doi: 10.1007/s12016-012-8322-2. [DOI] [PubMed] [Google Scholar]
- Chen C-N, Hsiao C-J, Lee S-S, Guh J-H, Chiang P-C, Huang C-C, et al. Chemical modification and anticancer effect of prenylated flavanones from Taiwanese propolis. Nat Prod Res. 2012;26(2):116–124. doi: 10.1080/14786419.2010.535146. [DOI] [PubMed] [Google Scholar]
- Chen C-N, Weng M-S, Wu C-L, Lin J-K. Comparison of radical scavenging activity, cytotoxic effects and apoptosis induction in human melanoma cells by Taiwanese propolis from different sources. Evid Based Complement Altern Med. 2004;1(2):175–185. doi: 10.1093/ecam/neh034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C-N, Wu C-L, Shy H-S, Lin J-K. Cytotoxic prenylflavanones from Taiwanese propolis. J Nat Prod. 2003;66(4):503–506. doi: 10.1021/np0203180. [DOI] [PubMed] [Google Scholar]
- Christov R, Trusheva B, Popova M, Bankova V, Bertrand M. Chemical composition of propolis from Canada, its antiradical activity and plant origin. Nat Prod Res. 2006;20(06):531–536. doi: 10.1080/14786410500056918. [DOI] [PubMed] [Google Scholar]
- Cruz JN, da Silva AG, da Costa WA, Gurgel ESC, Campos WEO, Silva RC et al (2020) Volatile compounds, chemical composition and biological activities of Apis mellifera bee propolis. In: Essential oils—bioactive compounds, new perspectives and applications. InTechOpen, Rijeka
- Cuesta-Rubio O, Frontana-Uribe BA, Ramírez-Apan T, Cárdenas J. Polyisoprenylated benzophenones in Cuban propolis; biological activity of nemorosone §. Z Nat C. 2002;57(3–4):372–378. doi: 10.1515/znc-2002-3-429. [DOI] [PubMed] [Google Scholar]
- Cuesta-Rubio O, Piccinelli AL, Campo Fernandez M, Marquez Hernandez I, Rosado A, Rastrelli L. Chemical characterization of Cuban propolis by HPLC–PDA, HPLC–MS, and NMR: the brown, red, and yellow Cuban varieties of propolis. J Agric Food Chem. 2007;55(18):7502–7509. doi: 10.1021/jf071296w. [DOI] [PubMed] [Google Scholar]
- Cunha I, Sawaya AC, Caetano FM, Shimizu MT, Marcucci MC, Drezza FT, et al. Factors that influence the yield and composition of Brazilian propolis extracts. J Braz Chem Soc. 2004;15(6):964–970. doi: 10.1590/S0103-50532004000600026. [DOI] [Google Scholar]
- Cvek J, Medić-Šarić M, Jasprica I, Zubčić S, Vitali D, Mornar A, et al. Optimisation of an extraction procedure and chemical characterisation of Croatian propolis tinctures. Phytochem Anal. 2007;18(5):451–459. doi: 10.1002/pca.1001. [DOI] [PubMed] [Google Scholar]
- da Silva Frozza CO, Garcia CSC, Gambato G, de Souza MDO, Salvador M, Moura S, et al. Chemical characterization, antioxidant and cytotoxic activities of Brazilian red propolis. Food Chem Toxicol. 2013;52:137–142. doi: 10.1016/j.fct.2012.11.013. [DOI] [PubMed] [Google Scholar]
- Daugsch A, Moraes C, Fort P, Park Y (2007) Botanical origin of Brazilian reddish propolis and its major chemical constituents. Honeybee Science-Tamagawa University (Japan)
- De Almeida E, Menezes H. Anti-inflammatory activity of propolis extracts: a review. J Venom Anim Toxins. 2002;8(2):191–212. doi: 10.1590/S0104-79302002000200002. [DOI] [Google Scholar]
- de Castro Ishida VF, Negri G, Salatino A, Bandeira MFC. A new type of Brazilian propolis: prenylated benzophenones in propolis from Amazon and effects against cariogenic bacteria. Food Chem. 2011;125(3):966–972. doi: 10.1016/j.foodchem.2010.09.089. [DOI] [Google Scholar]
- Dezmirean DS, Paşca C, Moise AR, Bobiş O. Plant sources responsible for the chemical composition and main bioactive properties of Poplar-type propolis. Plants. 2021;10(1):22. doi: 10.3390/plants10010022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dias LG, Pereira AP, Estevinho LM. Comparative study of different Portuguese samples of propolis: pollinic, sensorial, physicochemical, microbiological characterization and antibacterial activity. Food Chem Toxicol. 2012;50(12):4246–4253. doi: 10.1016/j.fct.2012.08.056. [DOI] [PubMed] [Google Scholar]
- Díaz-Carballo D, Gustmann S, Strumberg D, Acikelli AH, Bardenheuer W. Multi-targeted polycyclic polyprenylated acylphloroglucinols are major constituents of Cuban propolis and contributors to its anticancer activity. Int J Clin Pharmacol Ther. 2013;51(1):54–55. doi: 10.5414/cpp51054. [DOI] [PubMed] [Google Scholar]
- Duran N, Muz M, Culha G, Duran G, Ozer B. GC–MS analysis and antileishmanial activities of two Turkish propolis types. Parasitol Res. 2011;108(1):95–105. doi: 10.1007/s00436-010-2039-z. [DOI] [PubMed] [Google Scholar]
- El-Guendouz S, Lyoussi B, Miguel MG. Insight on propolis from Mediterranean countries: chemical composition, biological activities and application fields. Chem Biodivers. 2019;16(7):e1900094. doi: 10.1002/cbdv.201900094. [DOI] [PubMed] [Google Scholar]
- Elbaz GA, Elsayad II. Comparison of the antimicrobial effect of Egyptian propolis vs New Zealand propolis on Streptococcus mutans and lactobacilli in saliva. Oral Health Prev Dent. 2012;10(2):155–160. [PubMed] [Google Scholar]
- Endo S, Matsunaga T, Kanamori A, Otsuji Y, Nagai H, Sundaram K, et al. Selective inhibition of human type-5 17β-hydroxysteroid dehydrogenase (AKR1C3) by baccharin, a component of Brazilian propolis. J Nat Prod. 2012;75(4):716–721. doi: 10.1021/np201002x. [DOI] [PubMed] [Google Scholar]
- Erdogan S, Ates B, Durmaz G, Yilmaz I, Seckin T. Pressurized liquid extraction of phenolic compounds from Anatolia propolis and their radical scavenging capacities. Food Chem Toxicol. 2011;49(7):1592–1597. doi: 10.1016/j.fct.2011.04.006. [DOI] [PubMed] [Google Scholar]
- Falcão SI, Vale N, Gomes P, Domingues MR, Freire C, Cardoso SM, et al. Phenolic profiling of Portuguese propolis by LC–MS spectrometry: Uncommon propolis rich in flavonoid glycosides. Phytochem Anal. 2013;24(4):309–318. doi: 10.1002/pca.2412. [DOI] [PubMed] [Google Scholar]
- Farooqui T, Farooqui AA. Beneficial effects of propolis on human health and neurological diseases. Front Biosci (elite Ed) 2012;4:779–793. doi: 10.2741/418. [DOI] [PubMed] [Google Scholar]
- Fernandes-Silva CC, Salatino A, Salatino MLF, Breyer ED, Negri G. Chemical profiling of six samples of Brazilian propolis. Quím Nova. 2013;36(2):237–240. doi: 10.1590/S0100-40422013000200006. [DOI] [Google Scholar]
- Fischer G, Paulino N, Marcucci MC, Siedler BS, Munhoz LS, Finger PF, et al. Green propolis phenolic compounds act as vaccine adjuvants, improving humoral and cellular responses in mice inoculated with inactivated vaccines. Mem Inst Oswaldo Cruz. 2010;105(7):908–913. doi: 10.1590/S0074-02762010000700012. [DOI] [PubMed] [Google Scholar]
- Furukawa S, Takagi N, Ikeda T, Ono M, Nafady AM, Nohara T, et al. Two novel long-chain alkanoic acid esters of lupeol from alecrim-propolis. Chem Pharm Bull. 2002;50(3):439–440. doi: 10.1248/cpb.50.439. [DOI] [PubMed] [Google Scholar]
- Ghallab DS, Mohyeldin MM, Shawky E, Metwally AM, Ibrahim RS. Chemical profiling of Egyptian propolis and determination of its xanthine oxidase inhibitory properties using UPLC–MS/MS and chemometrics. LWT. 2021;136:110298. doi: 10.1016/j.lwt.2020.110298. [DOI] [Google Scholar]
- Gómez-Romero M, Arráez-Román D, Moreno-Torres R, García-Salas P, Segura-Carretero A, Fernández-Gutiérrez A. Antioxidant compounds of propolis determined by capillary electrophoresis–mass spectrometry. J Sep Sci. 2007;30(4):595–603. doi: 10.1002/jssc.200600354. [DOI] [PubMed] [Google Scholar]
- Gromenko D, IuIu G, ShN G, Farkhutdinov R, Mukhamedzianov R. Effect of propolis bioflavonoids on lipoperoxidation processes in the gonads of rats intoxicated by polychlorinated biphenyls. Vopr Pitan. 2008;77(6):9–13. [PubMed] [Google Scholar]
- Guo X, Chen B, Luo L, Zhang X, Dai X, Gong S. Chemical compositions and antioxidant activities of water extracts of Chinese propolis. J Agric Food Chem. 2011;59(23):12610–12616. doi: 10.1021/jf202818p. [DOI] [PubMed] [Google Scholar]
- Elwakil H, B, Shaaban, MM, Bekhit, AA, El-Naggar, MY, Olama, ZA, Potential anti-COVID-19 activity of Egyptian propolis using computational modeling. Future Virol. 2021;16(2):107–116. doi: 10.2217/fvl-2020-0329. [DOI] [Google Scholar]
- Hayashi K, Komura S, Isaji N, Ohishi N, Yagi K. Isolation of antioxidative compounds from Brazilian propolis: 3, 4-dihydroxy-5-prenylcinnamic acid, a novel potent antioxidant. Chem Pharm Bull. 1999;47(11):1521–1524. doi: 10.1248/cpb.47.1521. [DOI] [Google Scholar]
- Hegazi AG, Abd El Hady FK. Egyptian propolis: 3. Antioxidant, antimicrobial activities and chemical composition of propolis from reclaimed lands. Z Nat C. 2002;57(3–4):395–402. doi: 10.1515/znc-2002-3-432. [DOI] [PubMed] [Google Scholar]
- Hegazi AG, Abd El Hady FK, Abd Allah FA. Chemical composition and antimicrobial activity of European propolis. Z Nat C. 2000;55(1–2):70–75. doi: 10.1515/znc-2000-1-214. [DOI] [PubMed] [Google Scholar]
- Hernández IM, Fernandez MC, Cuesta-Rubio O, Piccinelli AL, Rastrelli L. Polyprenylated benzophenone derivatives from Cuban propolis. J Nat Prod. 2005;68(6):931–934. doi: 10.1021/np0495884. [DOI] [PubMed] [Google Scholar]
- Huang S, Zhang C-P, Wang K, Li GQ, Hu F-L. Recent advances in the chemical composition of propolis. Molecules. 2014;19(12):19610–19632. doi: 10.3390/molecules191219610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inui S, Shimamura Y, Masuda S, Shirafuji K, Moli RT, Kumazawa S. A new prenylflavonoid isolated from propolis collected in the Solomon Islands. Biosci Biotechnol Biochem. 2012;76(5):1038–1040. doi: 10.1271/bbb.120021. [DOI] [PubMed] [Google Scholar]
- Isidorov VA, Szczepaniak L, Bakier S. Rapid GC/MS determination of botanical precursors of Eurasian propolis. Food Chem. 2014;142:101–106. doi: 10.1016/j.foodchem.2013.07.032. [DOI] [PubMed] [Google Scholar]
- Isla MI, Paredes-Guzman JF, Nieva-Moreno MI, Koo H, Park YK. Some chemical composition and biological activity of northern Argentine propolis. J Agric Food Chem. 2005;53(4):1166–1172. doi: 10.1021/jf040130h. [DOI] [PubMed] [Google Scholar]
- Jasprica I, Bojic M, Mornar A, Besic E, Bucan K, Medic-Saric M. Evaluation of antioxidative activity of Croatian propolis samples using DPPH and ABTS+ stable free radical assays. Molecules. 2007;12(5):1006–1021. doi: 10.3390/12051006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalogeropoulos N, Konteles SJ, Troullidou E, Mourtzinos I, Karathanos VT. Chemical composition, antioxidant activity and antimicrobial properties of propolis extracts from Greece and Cyprus. Food Chem. 2009;116(2):452–461. doi: 10.1016/j.foodchem.2009.02.060. [DOI] [Google Scholar]
- Kartal M, Yıldız S, Kaya S, Kurucu S, Topçu G. Antimicrobial activity of propolis samples from two different regions of Anatolia. J Ethnopharmacol. 2003;86(1):69–73. doi: 10.1016/S0378-8741(03)00042-4. [DOI] [PubMed] [Google Scholar]
- Kaškonienė V, Kaškonas P, Maruška A, Kubilienė L. Chemometric analysis of volatiles of propolis from different regions using static headspace GC–MS. Cent Eur J Chem. 2014;12(6):736–746. doi: 10.2478/s11532-014-0521-7. [DOI] [Google Scholar]
- Kasote D, Ahmad A, Chen W, Combrinck S, Viljoen A. HPTLC–MS as an efficient hyphenated technique for the rapid identification of antimicrobial compounds from propolis. Phytochem Lett. 2015;11:326–331. doi: 10.1016/j.phytol.2014.08.017. [DOI] [Google Scholar]
- Kasote D, Suleman T, Chen W, Sandasi M, Viljoen A, van Vuuren S. Chemical profiling and chemometric analysis of South African propolis. Biochem Syst Ecol. 2014;55:156–163. doi: 10.1016/j.bse.2014.03.012. [DOI] [Google Scholar]
- Kasote DM. Propolis: a neglected product of value in the Indian beekeeping sector. Bee World. 2017;94(3):80–83. doi: 10.1080/0005772X.2017.1345223. [DOI] [Google Scholar]
- Kasote DM, Pawar MV, Bhatia RS, Nandre VS, Gundu SS, Jagtap SD, et al. HPLC, NMR based chemical profiling and biological characterisation of Indian propolis. Fitoterapia. 2017;122:52–60. doi: 10.1016/j.fitote.2017.08.011. [DOI] [PubMed] [Google Scholar]
- Kasote DM, Pawar MV, Gundu SS, Bhatia R, Nandre VS, Jagtap SD, et al. Chemical profiling, antioxidant, and antimicrobial activities of Indian stingless bees propolis samples. J Apic Res. 2019;58(4):617–625. doi: 10.1080/00218839.2019.1584960. [DOI] [Google Scholar]
- Kujumgiev A, Tsvetkova I, Serkedjieva Y, Bankova V, Christov R, Popov S. Antibacterial, antifungal and antiviral activity of propolis of different geographic origin. J Ethnopharmacol. 1999;64(3):235–240. doi: 10.1016/S0378-8741(98)00131-7. [DOI] [PubMed] [Google Scholar]
- Kumazawa S, Hamasaka T, Nakayama T. Antioxidant activity of propolis of various geographic origins. Food Chem. 2004;84(3):329–339. doi: 10.1016/S0308-8146(03)00216-4. [DOI] [Google Scholar]
- Kumazawa S, Hayashi K, Kajiya K, Ishii T, Hamasaka T, Nakayama T. Studies of the constituents of Uruguayan propolis. J Agric Food Chem. 2002;50(17):4777–4782. doi: 10.1021/jf020279y. [DOI] [PubMed] [Google Scholar]
- Kumazawa S, Nakamura J, Murase M, Miyagawa M, Ahn M-R, Fukumoto S. Plant origin of Okinawan propolis: honeybee behavior observation and phytochemical analysis. Naturwissenschaften. 2008;95(8):781. doi: 10.1007/s00114-008-0383-y. [DOI] [PubMed] [Google Scholar]
- Kumazawa S, Ueda R, Hamasaka T, Fukumoto S, Fujimoto T, Nakayama T. Antioxidant prenylated flavonoids from propolis collected in Okinawa, Japan. J Agric Food Chem. 2007;55(19):7722–7725. doi: 10.1021/jf071187h. [DOI] [PubMed] [Google Scholar]
- Li F, Awale S, Tezuka Y, Kadota S. Cytotoxic constituents from Brazilian red propolis and their structure–activity relationship. Bioorg Med Chem. 2008;16(10):5434–5440. doi: 10.1016/j.bmc.2008.04.016. [DOI] [PubMed] [Google Scholar]
- Li F, Awale S, Zhang H, Tezuka Y, Esumi H, Kadota S. Chemical constituents of propolis from Myanmar and their preferential cytotoxicity against a human pancreatic cancer cell line. J Nat Prod. 2009;72(7):1283–1287. doi: 10.1021/np9002433. [DOI] [PubMed] [Google Scholar]
- Li F, He Y-M, Awale S, Kadota S, Tezuka Y. Two new cytotoxic phenylallylflavanones from Mexican propolis. Chem Pharm Bull. 2011;59(9):1194–1196. doi: 10.1248/cpb.59.1194. [DOI] [PubMed] [Google Scholar]
- Lotti C, Campo Fernandez M, Piccinelli AL, Cuesta-Rubio O, Marquez Hernandez I, Rastrelli L. Chemical constituents of red Mexican propolis. J Agric Food Chem. 2010;58(4):2209–2213. doi: 10.1021/jf100070w. [DOI] [PubMed] [Google Scholar]
- Lotti C, Piccinelli AL, Arevalo C, Ruiz I, Migliani De Castro GM, Sá FRD, L,, et al. Constituents of Hondurian propolis with inhibitory effects on Saccharomyces cerevisiae multidrug resistance protein Pdr5p. J Agric Food Chem. 2012;60(42):10540–10545. doi: 10.1021/jf302578r. [DOI] [PubMed] [Google Scholar]
- Maciejewicz W, Daniewski M, Bal K, Markowski W. GC–MS identification of the flavonoid aglycones isolated from propolis. Chromatographia. 2001;53(5–6):343–346. doi: 10.1007/BF02490438. [DOI] [Google Scholar]
- Maraschin M, Somensi-Zeggio Al, Oliveira SK, Kuhnen S, Tomazzoli MM, Raguzzoni JC, et al. Metabolic profiling and classification of propolis samples from Southern Brazil: an NMR-based platform coupled with machine learning. J Nat Prod. 2016;79(1):13–23. doi: 10.1021/acs.jnatprod.5b00315. [DOI] [PubMed] [Google Scholar]
- Marcucci MC, Ferreres F, Garcıa-Viguera C, Bankova V, De Castro S, Dantas A, et al. Phenolic compounds from Brazilian propolis with pharmacological activities. J Ethnopharmacol. 2001;74(2):105–112. doi: 10.1016/S0378-8741(00)00326-3. [DOI] [PubMed] [Google Scholar]
- Markham KR, Mitchell KA, Wilkins AL, Daldy JA, Lu Y. HPLC and GC–MS identification of the major organic constituents in New Zealand propolis. Phytochemistry. 1996;42(1):205–211. doi: 10.1016/0031-9422(96)83286-9. [DOI] [Google Scholar]
- Maróstica Junior MR, Daugsch A, Moraes CS, Queiroga CL, Pastore GM, Parki YK. Comparison of volatile and polyphenolic compounds in Brazilian green propolis and its botanical origin Baccharis dracunculifolia. Food Sci Technol. 2008;28(1):178–181. doi: 10.1590/S0101-20612008000100026. [DOI] [Google Scholar]
- Márquez N, Sancho R, Macho A, Calzado MA, Fiebich BL, Muñoz E. Caffeic acid phenethyl ester inhibits T-cell activation by targeting both nuclear factor of activated T-cells and NF-κB transcription factors. J Pharmacol Exp Ther. 2004;308(3):993–1001. doi: 10.1124/jpet.103.060673. [DOI] [PubMed] [Google Scholar]
- Mayworm MA, Lima CA, Tomba AC, Fernandes-Silva CC, Salatino ML, Salatino A. Does propolis contain tannins? Evid Based Complement Altern Med. 2014 doi: 10.1155/2014/613647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medana C, Carbone F, Aigotti R, Appendino G, Baiocchi C. Selective analysis of phenolic compounds in propolis by HPLC–MS/MS. Phytochem Anal. 2008;19(1):32–39. doi: 10.1002/pca.1010. [DOI] [PubMed] [Google Scholar]
- Melliou E, Chinou I. Chemical analysis and antimicrobial activity of Greek propolis. Planta Med. 2004;70(06):515–519. doi: 10.1055/s-2004-827150. [DOI] [PubMed] [Google Scholar]
- Melliou E, Stratis E, Chinou I. Volatile constituents of propolis from various regions of Greece—antimicrobial activity. Food Chem. 2007;103(2):375–380. doi: 10.1016/j.foodchem.2006.07.033. [DOI] [Google Scholar]
- Messerli SM, Ahn MR, Kunimasa K, Yanagihara M, Tatefuji T, Hashimoto K, et al. Artepillin C (ARC) in Brazilian green propolis selectively blocks oncogenic PAK1 signaling and suppresses the growth of NF tumors in mice. Phytother Res. 2009;23(3):423–427. doi: 10.1002/ptr.2658. [DOI] [PubMed] [Google Scholar]
- Miguel MG, Antunes MD. Is propolis safe as an alternative medicine? J Pharm Bioallied Sci. 2011;3(4):479. doi: 10.4103/0975-7406.90101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mishima S, Ono Y, Araki Y, Akao Y, Nozawa Y. Two related cinnamic acid derivatives from Brazilian honey bee propolis, baccharin and drupanin, induce growth inhibition in allografted sarcoma S-180 in mice. Biol Pharm Bull. 2005;28(6):1025–1030. doi: 10.1248/bpb.28.1025. [DOI] [PubMed] [Google Scholar]
- Montenegro, G, Peña, R, Avila, G, Timmermann, B (2001) Botanical origin and seasonal production of propolis in hives of Central Chile. Boletim de Botânica da Universidade de São Paulo, pp 1–6
- Monzote L, Cuesta-Rubio O, Campo Fernandez M, Márquez Hernandez I, Fraga J, Pérez K, et al. In vitro antimicrobial assessment of Cuban propolis extracts. Mem Inst Oswaldo Cruz. 2012;107(8):978–984. doi: 10.1590/S0074-02762012000800003. [DOI] [PubMed] [Google Scholar]
- Naik DG, Vaidya HS, Namjoshi TP. Essential oil of Indian propolis: chemical composition and repellency against the honeybee Apis florea. Chem Biodivers. 2013;10(4):649–657. doi: 10.1002/cbdv.201200165. [DOI] [PubMed] [Google Scholar]
- Nandre VS, Bagade AV, Kasote DM, Lee JH, Kodam KM, Kulkarni MV, et al. Antibacterial activity of Indian propolis and its lead compounds against multi-drug resistant clinical isolates. J Herb Med. 2021;29:100479. doi: 10.1016/j.hermed.2021.100479. [DOI] [Google Scholar]
- Nunes CA, Guerreiro MC. Characterization of Brazilian green propolis throughout the seasons by headspace GC/MS and ESI-MS. J Sci Food Agric. 2012;92(2):433–438. doi: 10.1002/jsfa.4596. [DOI] [PubMed] [Google Scholar]
- Okutan H, Ozcelik N, Yilmaz HR, Uz E. Effects of caffeic acid phenethyl ester on lipid peroxidation and antioxidant enzymes in diabetic rat heart. Clin Biochem. 2005;38(2):191–196. doi: 10.1016/j.clinbiochem.2004.10.003. [DOI] [PubMed] [Google Scholar]
- Olegário LS, Andrade JKS, Andrade GRS, Denadai M, Cavalcanti RL, da Silva MAAP, et al. Chemical characterization of four Brazilian brown propolis: an insight in tracking of its geographical location of production and quality control. Food Res Int. 2019;123:481–502. doi: 10.1016/j.foodres.2019.04.004. [DOI] [PubMed] [Google Scholar]
- Orsi RO, Sforcin JM, Funari SR, Bankova V. Effects of Brazilian and Bulgarian propolis on bactericidal activity of macrophages against Salmonella typhimurium. Int Immunopharmacol. 2005;5(2):359–368. doi: 10.1016/j.intimp.2004.10.003. [DOI] [PubMed] [Google Scholar]
- Oršolić N, Knežević AH, Šver L, Terzić S, Bašić I. Immunomodulatory and antimetastatic action of propolis and related polyphenolic compounds. J Ethnopharmacol. 2004;94(2–3):307–315. doi: 10.1016/j.jep.2004.06.006. [DOI] [PubMed] [Google Scholar]
- Oršolić N, Šaranović AB, Bašić I. Direct and indirect mechanism(s) of antitumour activity of propolis and its polyphenolic compounds. Planta Med. 2006;72(01):20–27. doi: 10.1055/s-2005-873167. [DOI] [PubMed] [Google Scholar]
- Park Y, Ikegaki M, de Alencar S, de Moura F. Evaluation of Brazilian propolis by both physicochemical methods and biological activity. Honeybee Sci. 2000;21(2):85–90. [Google Scholar]
- Park YK, Alencar SM, Aguiar CL. Botanical origin and chemical composition of Brazilian propolis. J Agric Food Chem. 2002;50(9):2502–2506. doi: 10.1021/jf011432b. [DOI] [PubMed] [Google Scholar]
- Park YK, Paredes-Guzman JF, Aguiar CL, Alencar SM, Fujiwara FY. Chemical constituents in Baccharis dracunculifolia as the main botanical origin of southeastern Brazilian propolis. J Agric Food Chem. 2004;52(5):1100–1103. doi: 10.1021/jf021060m. [DOI] [PubMed] [Google Scholar]
- Paviani LC, Dariva C, Marcucci MC, Cabral FA. Supercritical carbon dioxide selectivity to fractionate phenolic compounds from the dry ethanolic extract of propolis. J Food Process Eng. 2010;33(1):15–27. doi: 10.1111/j.1745-4530.2008.00256.x. [DOI] [Google Scholar]
- Pavlovic R, Borgonovo G, Leoni V, Giupponi L, Ceciliani G, Sala S, et al. Effectiveness of different analytical methods for the characterization of propolis: a case of study in Northern Italy. Molecules. 2020;25(3):504. doi: 10.3390/molecules25030504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pellati F, Orlandini G, Pinetti D, Benvenuti S. HPLC-DAD and HPLC-ESI-MS/MS methods for metabolite profiling of propolis extracts. J Pharm Biomed Anal. 2011;55(5):934–948. doi: 10.1016/j.jpba.2011.03.024. [DOI] [PubMed] [Google Scholar]
- Pereira A, Norsell M, Cardoso J, Aquino Neto F, Ramos M. Rapid screening of polar compounds in Brazilian propolis by high-temperature high-resolution gas chromatography–mass spectrometry. J Agric Food Chem. 2000;48(11):5226–5230. doi: 10.1021/jf000594p. [DOI] [PubMed] [Google Scholar]
- Petri G, Lemberkovics E, Foldvari M. Examination of differences between propolis (bee glue) produced from different floral environments. Dev Food Sci. 1988;18:439–446. [Google Scholar]
- Petrova A, Popova M, Kuzmanova C, Tsvetkova I, Naydenski H, Muli E, et al. New biologically active compounds from Kenyan propolis. Fitoterapia. 2010;81(6):509–514. doi: 10.1016/j.fitote.2010.01.007. [DOI] [PubMed] [Google Scholar]
- Piccinelli AL, Campo Fernandez M, Cuesta-Rubio O, Márquez Hernández I, De Simone F, Rastrelli L. Isoflavonoids isolated from Cuban propolis. J Agric Food Chem. 2005;53(23):9010–9016. doi: 10.1021/jf0518756. [DOI] [PubMed] [Google Scholar]
- Piccinelli AL, Lotti C, Campone L, Cuesta-Rubio O, Campo Fernandez M, Rastrelli L. Cuban and Brazilian red propolis: botanical origin and comparative analysis by high-performance liquid chromatography–photodiode array detection/electrospray ionization tandem mass spectrometry. J Agric Food Chem. 2011;59(12):6484–6491. doi: 10.1021/jf201280z. [DOI] [PubMed] [Google Scholar]
- Piccinelli AL, Mencherini T, Celano R, Mouhoubi Z, Tamendjari A, Aquino RP, et al. Chemical composition and antioxidant activity of Algerian propolis. J Agric Food Chem. 2013;61(21):5080–5088. doi: 10.1021/jf400779w. [DOI] [PubMed] [Google Scholar]
- Pietta P, Gardana C, Pietta A. Analytical methods for quality control of propolis. Fitoterapia. 2002;73:S7–S20. doi: 10.1016/S0367-326X(02)00186-7. [DOI] [PubMed] [Google Scholar]
- Pobiega K, Kraśniewska K, Gniewosz M. Application of propolis in antimicrobial and antioxidative protection of food quality—a review. Trends Food Sci Technol. 2019;83:53–62. doi: 10.1016/j.tifs.2018.11.007. [DOI] [Google Scholar]
- Popova M, Bankova V, Butovska D, Petkov V, Nikolova-Damyanova B, Sabatini AG, et al. Validated methods for the quantification of biologically active constituents of poplar-type propolis. Phytochem Anal. 2004;15(4):235–240. doi: 10.1002/pca.777. [DOI] [PubMed] [Google Scholar]
- Popova M, Bankova V, Spassov S, Tsvetkova I, Silva MV, Tsartsarova M, et al. New bioactive chalcones in propolis from El Salvador. Z Nat C. 2001;56(7–8):593–596. doi: 10.1515/znc-2001-7-819. [DOI] [PubMed] [Google Scholar]
- Popova M, Bankova V, Tsvetkova I, Naydenski C, Silva MV. The first glycosides isolated from propolis: diterpene rhamnosides. Z Nat C. 2001;56(11–12):1108–1111. doi: 10.1515/znc-2001-11-1230. [DOI] [PubMed] [Google Scholar]
- Popova M, Chen CN, Chen PY, Huang CY, Bankova V. A validated spectrophotometric method for quantification of prenylated flavanones in pacific propolis from Taiwan. Phytochem Anal. 2010;21(2):186–191. doi: 10.1002/pca.1176. [DOI] [PubMed] [Google Scholar]
- Popova M, Graikou K, Bankova V, Chinou I. Chemical composition of 10 selected samples of Mediterranean propolis. Planta Med. 2008;74(09):PC60. doi: 10.1055/s-0028-1084578. [DOI] [Google Scholar]
- Popova M, Trusheva B, Antonova D, Cutajar S, Mifsud D, Farrugia C, et al. The specific chemical profile of Mediterranean propolis from Malta. Food Chem. 2011;126(3):1431–1435. doi: 10.1016/j.foodchem.2010.11.130Get. [DOI] [Google Scholar]
- Popova MP, Chinou IB, Marekov IN, Bankova VS. Terpenes with antimicrobial activity from Cretan propolis. Phytochemistry. 2009;70(10):1262–1271. doi: 10.1016/j.phytochem.2009.07.025. [DOI] [PubMed] [Google Scholar]
- Popova MP, Graikou K, Chinou I, Bankova VS. GC-MS profiling of diterpene compounds in Mediterranean propolis from Greece. J Agric Food Chem. 2010;58(5):3167–3176. doi: 10.1021/jf903841k. [DOI] [PubMed] [Google Scholar]
- Prytzyk E, Dantas AP, Salomão K, Pereira AS, Bankova VS, De Castro SL, et al. Flavonoids and trypanocidal activity of Bulgarian propolis. J Ethnopharmacol. 2003;88(2–3):189–193. doi: 10.1016/S0378-8741(03)00210-1. [DOI] [PubMed] [Google Scholar]
- Przybyłek I, Karpiński TM. Antibacterial properties of propolis. Molecules. 2019;24(11):2047. doi: 10.3390/molecules24112047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quiroga EN, Sampietro DA, Soberon JR, Sgariglia MA, Vattuone MA. Propolis from the northwest of Argentina as a source of antifungal principles. J Appl Microbiol. 2006;101(1):103–110. doi: 10.1111/j.1365-2672.2006.02904.x. [DOI] [PubMed] [Google Scholar]
- Raghukumar R, Vali L, Watson D, Fearnley J, Seidel V. Antimethicillin-resistant Staphylococcus aureus (MRSA) activity of ‘pacific propolis’ and isolated prenylflavanones. Phytother Res. 2010;24(8):1181–1187. doi: 10.1002/ptr.3096. [DOI] [PubMed] [Google Scholar]
- Ribeiro VP, Arruda C, Mejía JAA, Candido ACBB, Dos Santos RA, Magalhães LG, et al. Brazilian southeast brown propolis: gas chromatography method development for its volatile oil analysis, its antimicrobial and leishmanicidal activities evaluation. Phytochem Anal. 2021;32(3):404–411. doi: 10.1002/pca.2988. [DOI] [PubMed] [Google Scholar]
- Righi AA, Alves TR, Negri G, Marques LM, Breyer H, Salatino A. Brazilian red propolis: unreported substances, antioxidant and antimicrobial activities. J Sci Food Agric. 2011;91(13):2363–2370. doi: 10.1002/jsfa.4468. [DOI] [PubMed] [Google Scholar]
- Rubio OC, Cuellar AC, Rojas N, Castro HV, Rastrelli L, Aquino R. A polyisoprenylated benzophenone from Cuban propolis. J Nat Prod. 1999;62(7):1013–1015. doi: 10.1021/np980339n. [DOI] [PubMed] [Google Scholar]
- Russo A, Longo R, Vanella A. Antioxidant activity of propolis: role of caffeic acid phenethyl ester and galangin. Fitoterapia. 2002;73:S21–S29. doi: 10.1016/S0367-326X(02)00187-9. [DOI] [PubMed] [Google Scholar]
- Sahinler N, Kaftanoglu O. Natural product propolis: chemical composition. Nat Prod Res. 2005;19(2):183–188. doi: 10.1080/14786410410001704877. [DOI] [PubMed] [Google Scholar]
- Salatino A, Fernandes-Silva CC, Righi AA, Salatino MLF. Propolis research and the chemistry of plant products. Nat Prod Res. 2011;28(5):925–936. doi: 10.1039/C0NP00072H. [DOI] [PubMed] [Google Scholar]
- Salatino A, Salatino MLF. Scientific note: often quoted, but not factual data about propolis composition. Apidologie. 2021;52(2):312–314. doi: 10.1007/s13592-020-00821-x. [DOI] [Google Scholar]
- Salatino A, Teixeira ÉW, Negri G. Origin and chemical variation of Brazilian propolis. Evid Based Complement Altern Med. 2005;2(1):33–38. doi: 10.1093/ecam/neh060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salomão K, Dantas AP, Borba CM, Campos L, Machado D, Aquino Neto F, et al. Chemical composition and microbicidal activity of extracts from Brazilian and Bulgarian propolis. Lett Appl Microbiol. 2004;38(2):87–92. doi: 10.1111/j.1472-765X.2003.01458.x. [DOI] [PubMed] [Google Scholar]
- Sandasi M, Kamatou GP, Gavaghan C, Baranska M, Viljoen AM. A quality control method for geranium oil based on vibrational spectroscopy and chemometric data analysis. Vib Spectrosc. 2011;57(2):242–247. doi: 10.1016/j.vibspec.2011.08.002. [DOI] [Google Scholar]
- Sârbu C, Moţ AC. Ecosystem discrimination and fingerprinting of Romanian propolis by hierarchical fuzzy clustering and image analysis of TLC patterns. Talanta. 2011;85(2):1112–1117. doi: 10.1016/j.talanta.2011.05.030. [DOI] [PubMed] [Google Scholar]
- Savickas A, Ramanauskienė K, Pukalskas A, Kasparavičienė G, Briedis V, Inkėnienė AM et al (2010) The influence of solvent on the quantity and antioxidant activity of ethanolic extracts of Lithuanian propolis. Chemija. Vilnius: Lietuvos mokslų akademija, 2010, vol. 21(2–4)
- Sawicka D, Car H, Borawska MH, Nikliński J. The anticancer activity of propolis. Folia Histochem Cytobiol. 2012;50(1):25–37. doi: 10.2478/18693. [DOI] [PubMed] [Google Scholar]
- Schnitzler P, Neuner A, Nolkemper S, Zundel C, Nowack H, Sensch KH, et al. Antiviral activity and mode of action of propolis extracts and selected compounds. Phytother Res. 2010;24(S1):S20–S28. doi: 10.1002/ptr.2868. [DOI] [PubMed] [Google Scholar]
- Seidel V, Peyfoon E, Watson DG, Fearnley J. Comparative study of the antibacterial activity of propolis from different geographical and climatic zones. Phytother Res. 2008;22(9):1256–1263. doi: 10.1002/ptr.2480. [DOI] [PubMed] [Google Scholar]
- Sforcin JM. Biological properties and therapeutic applications of propolis. Phytother Res. 2016;30(6):894–905. doi: 10.1002/ptr.5605. [DOI] [PubMed] [Google Scholar]
- Sforcin JM, Bankova V. Propolis: is there a potential for the development of new drugs? J Ethnopharmacol. 2011;133(2):253–260. doi: 10.1016/j.jep.2010.10.032. [DOI] [PubMed] [Google Scholar]
- Sha N, Guan S-H, Lu Z-Q, Chen G-T, Huang H-L, Xie F-B, et al. Cytotoxic constituents of Chinese propolis. J Nat Prod. 2009;72(4):799–801. doi: 10.1021/np900118z. [DOI] [PubMed] [Google Scholar]
- Shaheen SA, Zarga MHA, Nazer IK, Darwish RM, Al-Jaber HI. Chemical constituents of Jordanian propolis. Nat Prod Res. 2011;25(14):1312–1318. doi: 10.1080/14786419.2010.509060. [DOI] [PubMed] [Google Scholar]
- Shahinozzaman M, Obanda DN, Tawata S. Chemical composition and pharmacological properties of Macaranga-type Pacific propolis: a review. Phytother Res. 2021;35(1):207–222. doi: 10.1002/ptr.6819. [DOI] [PubMed] [Google Scholar]
- Shawky E, Ibrahim RS. Bioprofiling for the quality control of Egyptian propolis using an integrated NIR-HPTLC-image analysis strategy. J Chromatogr B. 2018;1095:75–86. doi: 10.1016/j.jchromb.2018.07.029. [DOI] [PubMed] [Google Scholar]
- Shi H, Yang H, Zhang X, Sheng Y, Huang H, Yu L. Isolation and characterization of five glycerol esters from Wuhan propolis and their potential anti-inflammatory properties. J Agric Food Chem. 2012;60(40):10041–10047. doi: 10.1021/jf302601m. [DOI] [PubMed] [Google Scholar]
- Shi H, Yang H, Zhang X, Yu L. Identification and quantification of phytochemical composition and anti-inflammatory and radical scavenging properties of methanolic extracts of Chinese propolis. J Agric Food Chem. 2012;60(50):12403–12410. doi: 10.1021/jf3042775. [DOI] [PubMed] [Google Scholar]
- Shikanga EA, Viljoen AM, Vermaak I, Combrinck S. A novel approach in herbal quality control using hyperspectral imaging: discriminating between Sceletium tortuosum and Sceletium crassicaule. Phytochem Anal. 2013;24(6):550–555. doi: 10.1002/pca.2431. [DOI] [PubMed] [Google Scholar]
- Shimomura K, Sugiyama Y, Nakamura J, Ahn M-R, Kumazawa S. Component analysis of propolis collected on Jeju Island, Korea. Phytochemistry. 2013;93:222–229. doi: 10.1016/j.phytochem.2012.02.018. [DOI] [PubMed] [Google Scholar]
- Shouqin Z, Jun X, Changzheng W. High hydrostatic pressure extraction of flavonoids from propolis. J Chem Technol Biotechnol. 2005;80(1):50–54. doi: 10.1002/jctb.1153. [DOI] [Google Scholar]
- Shrestha SP, Narukawa Y, Takeda T. Chemical constituents of Nepalese propolis (II) Chem Pharm Bull. 2007;55(6):926–929. doi: 10.1248/cpb.55.926. [DOI] [PubMed] [Google Scholar]
- Silva RB, Santos N, Martins N, Ferreira D, Barbosa F, Jr, Souza VO, et al. Caffeic acid phenethyl ester protects against the dopaminergic neuronal loss induced by 6-hydroxydopamine in rats. Neuroscience. 2013;233:86–94. doi: 10.1016/j.neuroscience.2012.12.041. [DOI] [PubMed] [Google Scholar]
- Silveira MAD, De Jong D, Berretta AA, dos Santos Galvão EB, Ribeiro JC, Cerqueira-Silva T, et al. Efficacy of Brazilian green propolis (EPP-AF®) as an adjunct treatment for hospitalized COVID-19 patients: a randomized, controlled clinical trial. Biomed Pharmacother. 2021;138:111526. doi: 10.1016/j.biopha.2021.111526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soltani E-K, Cerezuela R, Charef N, Mezaache-Aichour S, Esteban MA, Zerroug MM. Algerian propolis extracts: chemical composition, bactericidal activity and in vitro effects on gilthead seabream innate immune responses. Fish Shellfish Immunol. 2017;62:57–67. doi: 10.1016/j.fsi.2017.01.009. [DOI] [PubMed] [Google Scholar]
- Stavropoulou M-I, Stathopoulou K, Cheilari A, Benaki D, Gardikis K, Chinou I, et al. NMR metabolic profiling of Greek propolis samples: comparative evaluation of their phytochemical compositions and investigation of their anti-ageing and antioxidant properties. J Pharm Biomed Anal. 2021;194:113814. doi: 10.1016/j.jpba.2020.113814. [DOI] [PubMed] [Google Scholar]
- Šturm L, Ulrih NP. Advances in the propolis chemical composition between 2013 and 2018: a review. eFood. 2019;1(1):24–37. doi: 10.2991/efood.k.191029.001. [DOI] [Google Scholar]
- Sulaiman GM, Al Sammarrae KW, Ad’hiah, AH, Zucchetti, M, Frapolli, R, Bello, E,, et al. Chemical characterization of Iraqi propolis samples and assessing their antioxidant potentials. Food Chem Toxicol. 2011;49(9):2415–2421. doi: 10.1016/j.fct.2011.06.060. [DOI] [PubMed] [Google Scholar]
- Sun L-P, Chen A-L, Hung H-C, Chien Y-H, Huang J-S, Huang C-Y, et al. Chrysin: a histone deacetylase 8 inhibitor with anticancer activity and a suitable candidate for the standardization of Chinese propolis. J Agric Food Chem. 2012;60(47):11748–11758. doi: 10.1021/jf303261r. [DOI] [PubMed] [Google Scholar]
- Tagliacollo VA, Orsi RdO. Quality of propolis commercialized in the informal market. Food Sci Technol. 2011;31(3):752–757. doi: 10.1590/S0101-20612011000300031. [DOI] [Google Scholar]
- Teixeira EW, Negri G, Salatino A, Stringheta PC. Seasonal variation, chemical composition and antioxidant activity of Brazilian propolis samples. Evid Based Complement Altern Med. 2010;7(3):307–315. doi: 10.1093/ecam/nem177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomás-Barberán FA, García-Viguera C, Vit-Olivier P, Ferreres F, Tomás-Lorente F. Phytochemical evidence for the botanical origin of tropical propolis from Venezuela. Phytochemistry. 1993;34(1):191–196. doi: 10.1016/S0031-9422(00)90804-5. [DOI] [Google Scholar]
- Tran VH, Duke RK, Abu-Mellal A, Duke CC. Propolis with high flavonoid content collected by honey bees from Acacia paradoxa. Phytochemistry. 2012;81:126–132. doi: 10.1016/j.phytochem.2012.06.002. [DOI] [PubMed] [Google Scholar]
- Trusheva B, Popova M, Bankova V, Simova S, Marcucci MC, Miorin PL, et al. Bioactive constituents of Brazilian red propolis. Evid Based Complement Altern Med. 2006;3(2):249–254. doi: 10.1093/ecam/nel006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trusheva B, Popova M, Koendhori EB, Tsvetkova I, Naydenski C, Bankova V. Indonesian propolis: chemical composition, biological activity and botanical origin. Nat Prod Res. 2011;25(6):606–613. doi: 10.1080/14786419.2010.488235. [DOI] [PubMed] [Google Scholar]
- Trusheva B, Todorov I, Ninova M, Najdenski H, Daneshmand A, Bankova V. Antibacterial mono-and sesquiterpene esters of benzoic acids from Iranian propolis. Chem Cent J. 2010;4(1):1–4. doi: 10.1186/1752-153X-4-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trusheva B, Trunkova D, Bankova V. Different extraction methods of biologically active components from propolis: a preliminary study. Chem Cent J. 2007;1(1):1–4. doi: 10.1186/1752-153X-1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uzel A, Önçağ Ö, Çoğulu D, Gençay Ö. Chemical compositions and antimicrobial activities of four different Anatolian propolis samples. Microbiol Res. 2005;160(2):189–195. doi: 10.1016/j.micres.2005.01.002. [DOI] [PubMed] [Google Scholar]
- Valcic S, Montenegro G, Mujica A-M, Avila G, Franzblau S, Singh MP, et al. Phytochemical, morphological, and biological investigations of propolis from Central Chile. Z Nat C. 1999;54(5–6):406–416. doi: 10.1515/znc-1999-5-617. [DOI] [PubMed] [Google Scholar]
- Valcic S, Montenegro G, Timmermann BN. Lignans from Chilean propolis. J Nat Prod. 1998;61(6):771–775. doi: 10.1021/np980017j. [DOI] [PubMed] [Google Scholar]
- Velikova M, Bankova V, Sorkun K, Houcine S, Tsvetkova I, Kujumgiev A. Propolis from the Mediterranean region: chemical composition and antimicrobial activity. Z Nat C. 2000;55(9–10):790–793. doi: 10.1515/znc-2000-9-1019. [DOI] [PubMed] [Google Scholar]
- Vera N, Solorzano E, Ordoñez R, Maldonado L, Bedascarrasbure E, Isla MI. Chemical composition of Argentinean propolis collected in extreme regions and its relation with antimicrobial and antioxidant activities. Nat Prod Commun. 2011 doi: 10.1177/1934578X1100600618. [DOI] [PubMed] [Google Scholar]
- Volpi N. Separation of flavonoids and phenolic acids from propolis by capillary zone electrophoresis. Electrophoresis. 2004;25(12):1872–1878. doi: 10.1002/elps.200405949. [DOI] [PubMed] [Google Scholar]
- Volpi N. Polyphenolic fingerprint of propolis extracts. Nutrafoods. 2011;10(2):55–60. doi: 10.1007/BF03223389. [DOI] [Google Scholar]
- Volpi N, Bergonzini G. Analysis of flavonoids from propolis by on-line HPLC–electrospray mass spectrometry. J Pharm Biomed Anal. 2006;42(3):354–361. doi: 10.1016/j.jpba.2006.04.017. [DOI] [PubMed] [Google Scholar]
- Wang T, Liu Q, Wang M, Zhang L. Metabolomics reveals discrimination of Chinese propolis from different climatic regions. Foods. 2020;9(4):491. doi: 10.3390/foods9040491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiryowidagdo S, Simanjuntak P, Heffen WL. Chemical composition of propolis from different regions in Java and their cytotoxic activity. Am J Biochem Biotechnol. 2009;5(4):180–183. doi: 10.3844/ajbbsp.2009.180.183. [DOI] [Google Scholar]
- Wu X-Y, Li F, Zhao T, Mao G-H, Li J, Qu H-Y, et al. Enhanced anti-diabetic activity of a combination of chromium(III) malate complex and propolis and its acute oral toxicity evaluation. Biol Trace Elem Res. 2012;148(1):91–101. doi: 10.1007/s12011-012-9347-3. [DOI] [PubMed] [Google Scholar]
- Yaghoubi M, Gh G, Satari R. Antimicrobial activity of Iranian propolis and its chemical composition. DARU J Pharm Sci. 2007;15(1):45–48. [Google Scholar]
- Zhou, L, Guo, J, Yu, J (1999) Flavonoids from Beijing propolis. Zhongguo Zhong Yao Za Zhi 24(3):162–164, 191 [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.