ABSTRACT.
Crimean-Congo hemorrhagic fever (CCHF) is a highly fatal zoonotic disease endemic to Kazakhstan. Previous work estimated the seroprevalence of CCHF virus (CCHFV) among livestock owners in the Zhambyl region of southern Kazakhstan at 1.2%. To estimate CCHFV seroprevalence among cattle and sheep, we selected 15 villages with known history of CCHFV circulation (endemic) and 15 villages without known circulation (nonendemic) by cluster sampling with probability proportional to livestock population size. We collected whole blood samples from 521 sheep and 454 cattle from randomly selected households within each village and collected ticks found on the animals. We tested livestock blood for CCHFV-specific IgG antibodies by ELISA; ticks were screened for CCHFV RNA by real-time reverse transcription polymerase chain reaction and CCHFV antigen by antigen-capture ELISA. We administered questionnaires covering animal demographics and livestock herd characteristics to an adult in each selected household. Overall weighted seroprevalence was 5.7% (95% CI: 3.1, 10.3) among sheep and 22.5% (95% CI: 15.8, 31.2) among cattle. CCHFV-positive tick pools were found on two sheep (2.4%, 95% CI: 0.6, 9.5) and three cattle (3.8%, 95% CI: 1.2, 11.5); three CCHFV-positive tick pools were found in nonendemic villages. Endemic villages reported higher seroprevalence among sheep (15.5% versus 2.8%, P < 0.001) but not cattle (25.9% versus 20.1%, P = 0.42). Findings suggest that the current village classification scheme may not reflect the geographic distribution of CCHFV in Zhambyl and underscore that public health measures must address the risk of CCHF even in areas without a known history of circulation.
INTRODUCTION
Crimean-Congo hemorrhagic fever (CCHF) is a tick-associated arboviral disease endemic to regions of Asia, Europe, and Africa.1–4 It is clinically characterized by a mild febrile illness that can progress, in a subset of cases, to severe hemorrhagic disease, with case fatality ranging from 2% to 30%.1,5 The CCHF virus (CCHFV) circulates in nature through tick–vertebrate–tick cycles, causing little or no damage in its animal hosts.1,2 Although many tick species are capable of transmitting the virus, the primary vectors are Hyalomma spp. ticks, in which CCHFV can survive transstadially (from larvae to nymph to adult), interseasonally, and transovarially from parent to offspring.2,6
People are most commonly infected with CCHFV via bites from infected vectors, crushing infected ticks bare-handed, or through close contact with the blood or bodily fluids of viremic animals.1,7 Human cases of CCHF are commonly attributed to contact with large domestic animals, such as cattle and sheep, with livestock handlers and abattoir workers at increased risk of infection.1,4,8–10 Livestock can serve as a sentinel reservoir for CCHF among humans. However, as livestock typically do not exhibit symptoms when infected, seroprevalence surveys of CCHFV among livestock provide critical evidence of circulating virus.1,9
Seroprevalence among animals is largely dependent on the local distribution of competent tick vectors, the host preference of vectors, and the tick load on animals.11–13 Longitudinal studies have found CCHFV seroprevalence in animals varies seasonally, with the highest prevalence in years following periods of vector population growth due to optimal weather conditions, such as mild winters, and/or anthropogenic change.2,14 Both increasing animal age, which likely reflects the potential for repeated exposure, and high levels of tick infestation on animals have been associated with seropositivity among livestock.11–13,15 Regular vector control treatments can protect livestock from tick-borne infections; however, environmental resistance remains a concern.14
CCHF is a reportable disease in Kazakhstan and considered endemic to the southern regions of the country; however, few serologic surveys have been conducted and the burden of disease remains unknown.16–19 Zhambyl is an agricultural and pastoral southern region comprising > 55,000 km2, with 363 villages and four cities, and a population of roughly 1.2 million. Occupational exposure to livestock and close animal contact are common, increasing the risk of zoonotic infections.8 From 2000 to 2013, 73 cases of CCHF were reported in Zhambyl, most of which were attributed to tick bites or the crushing of ticks during sheep shearing.17 Villages that report at least one CCHFV-positive tick or human CCHF case in the past 5 years are classified as endemic, and as part of routine prevention, tick control measures in these endemic villages are funded through state and national budgets in the region.
This is the second publication generated from a One Health study examining human, animal, and tick prevalence of CCHFV, along with the knowledge, attitudes, and practices (KAP) of livestock owners in Zhambyl. Previous analyses estimated the weighted human seroprevalence among livestock owners in Zhambyl at 1.2% when combining villages with known and unknown history of CCHFV circulation, and 3.4% in villages with a known history of CCHFV circulation, and examined the risk factor knowledge and frequency of risk-related activities.8 In this article, we estimate the CCHFV seroprevalence among cattle and sheep in Zhambyl, as well as the prevalence of CCHFV RNA and antigen among ticks removed from these livestock in both endemic and nonendemic villages. We further identify risk factors, including animal and herd characteristics, husbandry practices, and environmental characteristics, associated with animal seropositivity.
MATERIALS AND METHODS
The study was performed in June 2017 in 30 rural villages in Zhambyl region. Five teams, each consisting of an epidemiologist, a nurse, a veterinary expert, and an entomologist, administered human and livestock serosurveys, a molecular survey in ticks, a livestock questionnaire, and a KAP and risk factor survey. The human serosurvey and KAP methodologies have been described previously.8 Before conducting fieldwork, the field teams were trained on household and participant/animal selection methods, electronic data entry, sample handling and collection, and biosafety practices. The US CDC coordinated implementation with the Committee for Public Health Protection of the Ministry of Health of the Republic of Kazakhstan (RK) and the Committee of Veterinary Control and Surveillance of the Ministry of Agriculture of the RK. Team leads, national experts from the Surveillance Service and Central Reference Laboratory, and US CDC experts ensured adherence to all biosafety protocols.
Ethics.
All participants provided written, informed consent, and no personal identifying information was collected. The study protocol was approved by the RK School of Public Health ethical committee and received a nonresearch determination (2017-073) and Institutional Animal Care and Use Committee approval (2860SALMULX-A1) from the US CDC.
Livestock and tick sampling and sample size calculations.
The study population comprised sheep, cattle, and healthy adult humans (≥ 18 years of age) who own or care for these animals residing within rural villages of the Zhambyl region. The 363 villages in Zhambyl were stratified by “endemic” or “nonendemic” status, where “endemic” villages were those that reported at least one CCHFV-positive tick or human CCHF case in the past 5 years (from 2012 to 2016), and “nonendemic” villages were those that did not report any CCHFV-positive tick or human cases in the same period. We selected 15 endemic and 15 nonendemic villages by cluster sampling with probability proportional to livestock population size. Sample size calculations and sampling protocols were described previously in more detail.8 Target sample sizes for sheep (N = 561) and cattle (N = 473) were based on an expected CCHFV seroprevalence of 24% in sheep and 19% in cattle, with 5% precision.4 We used registries of livestock-owning households provided by local veterinarians to randomly select 19 sheep-owning households and 16 cattle-owning households within each village, and randomly selected one cow and/or one sheep from each selected household herd and one adult human from the household for participation.20
Eligible animals were ≥ 1 year of age. District veterinarians collected 10 mL of whole blood from each selected animal using Vacutainer and needle following standard collection procedures,21 and examined all selected livestock for ticks on their body with attention paid to areas surrounding the ears, along the nape of the neck, perineum, scrotum or udder, and tail base. Any ticks found on the animal were collected (up to a maximum of 26 ticks per animal), washed in 70% alcohol followed by saline, and pooled by species in tubes for laboratory testing.
Livestock questionnaire.
The field team administered a livestock questionnaire with the owner or manager of the livestock in each selected household. Questions covered the characteristics of the household’s livestock herds (e.g., herd size and grazing patterns) and each selected animal (e.g., age, sex, signs of illness, and tick treatment history). Signs of illness included thin body condition, adverse symptoms, or having received medication in the 4 months before sampling. Herd movement was categorized as “stationary” if animals were kept in housings, “nomadic” if animals were kept at pasture, and “seminomadic” if the herd contained a mix of stationary and nomadic animals. Owners’ knowledge of transmission pathways and animal husbandry practices were collected during the KAP/risk factor survey.8
Sample processing and testing.
Laboratory testing was performed by the Central Reference Laboratory in Almaty. Animal sera were analyzed using a CDC-developed ELISA to detect anti-CCHFV-specific IgG antibodies for evidence of past CCHFV exposure. The ELISA format was adapted for CCHFV using a method originally described for Ebola virus.22 Tick pools were homogenized using a MiniBeadBeater-16, Model 607 EUR at 3,450 RPM, scrolled three times for 15 minutes, and analyzed by both reverse transcription polymerase chain reaction (RT-PCR) to detect CCHFV RNA with the commercial kit CCHFV-FL (AmpliSens®, Moscow, Russia) and antigen-capture ELISA VectoKrym-KGL-Antigen (Vector-Best, Novosibirsk, Russia) to detect CCHFV antigen. Tick pools were considered CCHFV-positive if they recorded a positive result from either method. Human serosurvey sample processing and testing procedures were reported previously.8
Data analysis.
The data were analyzed using RStudio (version 1.3.1093). Sheep and cattle population estimates were adjusted using village-level veterinary registries. Within each stratum, base sampling weights were calculated for each animal according to the survey design as the product of the inverse probability of selection at each sampling stage. Sampling weights were trimmed to the 80th percentile using the multiplicative effect as a measure of weighting variability.20 The final trimmed weights were calibrated via poststratification adjustment using the estimate of sheep and cattle within each stratum. Design-based standard errors were applied in all analyses using the R survey package (version 4.0). Animals that tested positive for anti-CCHFV IgG were compared against those that tested IgG negative using χ2 test for categorical variables and Student’s t test for continuous variables, with statistical significance defined at 95% probability (P < 0.05). Similarly, animals from endemic villages were compared against those from nonendemic villages.
Weighted logistic regression models were developed to test associations between risk factors and seropositivity. Risk factors included variables known to be associated with seropositivity and those identified to be associated with sheep or cattle seropositivity in the above-described bivariate analysis. QGIS version 3.10.5 (Open-Source Geospatial Foundation Project, http://qgis.osgeo.org) was used to map seroprevalence among livestock by village.
RESULTS
A total of 521 sheep (243 from endemic villages) and 454 cattle (219 from endemic villages) were sampled from 925 households, 50 of which provided both cattle and sheep. The mean sheep age was 3.1 years, and 69% (95% CI: 50, 83) were female. Among cattle, the mean age was 4.2 years and 93% (95% CI: 83, 97) were female. Of the animals sampled, 23% (95% CI: 13, 38) of sheep and 43% (95% CI: 26, 61) of cattle were from endemic villages. The average herd size per household was 51 sheep and 9 cattle. Ticks were present on 40% of sheep (95% CI: 27, 54%) and 47% of cattle (95% CI: 33, 62%) at the time of sampling (Table 1); 0.8% of sheep (7/521, 95% CI: 0.3, 2.3%) and 3.5% of cattle (20/454, 95% CI: 1.1, 10%) were estimated to have more than 10 ticks.
Table 1 .
Comparison of animal demographics, herd characteristics, and tick treatment history between CCHFV seropositive and seronegative livestock in Zhambyl oblast, Kazakhstan, 2017
| Sheep | Cattle | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| CCHFV positive (N = 65) | CCHFV negative (N = 456) | CCHFV positive (N = 132) | CCHFV negative (N = 322) | |||||||
| % Animals (N) | 95% CI | % Animals (N) | 95% CI | P value | % Animals (N) | 95% CI | % Animals (N) | 95% CI | P value | |
| Village classified as endemic | 62.9% (43) | 41.8–80.0% | 20.9% (200) | 10.9–36.1% | < 0.001 | 48.8% (78) | 28.0–70.0% | 40.6% (141) | 23.8–60.0% | 0.42 |
| Animal characteristics | ||||||||||
| Mean age (years) | 3.2 (65) | 2.9–3.6 | 3.1 (453) | 2.8–3.3 | 0.46 | 4.4 (131) | 4.0–4.8 | 4.2 (320) | 3.6–4.7 | 0.34 |
| Female sex | 73.2% (44) | 50.0–88.2% | 68.7% (307) | 48.7–83.6% | 0.70 | 91.7% (122) | 79.9–96.8% | 93.2% (304) | 81.1–97.8% | 0.72 |
| Born on farm | 70.4% (48) | 32.0–92.3% | 90.6% (384) | 74.8–96.9% | 0.046 | 85.5% (111) | 60.2–95.8% | 82.8% (255) | 58.3–94.3% | 0.56 |
| Animal health | ||||||||||
| Signs of illness* | 10.0% (5) | 2.6–32.0% | 1.3% (20) | 0.6–3.0% | < 0.001 | 3.4% (8) | 0.7–15.0% | 1.5% (4) | 0.2–9.2% | 0.49 |
| Presence of ticks on body | 50.3% (42) | 29.5–71.0% | 38.9% (210) | 25.8–53.8% | 0.20 | 52.8% (88) | 32.7–72.1% | 45.9% (168) | 32.3–60.0% | 0.36 |
| CCHFV positive tick pool collected† | 0.0% (0) | – | 2.37% (2) | 0.56–9.46% | – | 0.0% (0) | – | 5.16% (3) | 1.66–14.9% | – |
| Herd characteristics | ||||||||||
| Mean herd size | 44.3 (65) | 18.9–69.7 | 51.3 (451) | 39.2–63.4 | 0.62 | 7.3 (132) | 4.0–10.5 | 9.1 (321) | 4.4–13.8 | 0.27 |
| Herd movement | 0.04 | 0.84 | ||||||||
| Stationary | 29.8% (18) | 7.8–68.0% | 7.5% (67) | 1.9–25.6% | 12.1% (18) | 2.6–41.7% | 15.4% (58) | 4.4–42.1% | ||
| Seminomadic | 46.2% (28) | 19.6–75.2% | 58.3% (253) | 31.6–80.8% | 46.9% (78) | 21.9–73.5% | 42.3% (164) | 20.4–67.7% | ||
| Nomadic | 24.0% (19) | 9.6–48.7% | 34.2% (134) | 13.9–62.7% | 41.1% (35) | 17.0–70.4% | 42.3% (99) | 19.5–69.0% | ||
| Graze extent beyond village | 17.8% (9) | 3.9–53.5% | 9.5% (68) | 2.3–31.4% | 0.13 | 16.7% (33) | 3.4–53.3% | 7.5% (35) | 1.5–30.3% | < 0.001 |
| Tick treatment history | ||||||||||
| Mean tick treatments over last year | 1.5 (44) | 1.0–2.0 | 1.2 (277) | 0.9–1.5 | 0.12 | 1.6 (129) | 1.1–2.0 | 1.4 (315) | 1.2–1.7 | 0.37 |
| Animal had received tick treatment in past year | 84.3% (61) | 56.1–95.7% | 91.8% (410) | 71.9–98.0% | 0.12 | 86.9% (120) | 50.8–97.7% | 93.6% (297) | 74.6–98.6% | 0.01 |
| If yes, specify treatment | 0.018 | 0.02 | ||||||||
| Ivermectin | 12.9% (8) | 2.9–42.1% | 48.5% (141) | 23.2–74.6% | 25.7% (29) | 8.6–55.9% | 39.5% (110) | 18.8–64.8% | ||
| Deltamethrin‡ | 4.4% (3) | 1.0–17.3% | 5.7% (31) | 1.2–22.6% | 16.2% (12) | 3.8–48.9% | 8.3% (22) | 1.8–30.4% | ||
| Diazinon§ | 79.7% (46) | 54.5–92.8% | 44.5% (214) | 20.7–71.1% | 53.9% (58) | 29.2–76.8% | 51.2% (152) | 28.3–73.7% | ||
| Other | 0.0% (0) | – | 0.2% (1) | 0.0–1.6% | 0.3% (1) | 0.0–2.3% | 0.0% (0) | – | ||
| Don’t know | 2.9% (4) | 0.6–13.2% | 1.2% (23) | 0.4–3.9% | 3.9% (20) | 0.8–16.5% | 1.0% (13) | 0.2–5.0% | ||
CCHFV = Crimean-Congo hemorrhagic fever virus; PCR = polymerase chain reaction. Boldface font indicates statistical significance at the P < 0.05 level.
Thin body condition or symptoms or medication in last 4 months.
Positive result on either reverse transcription PCR or antigen-capture ELISA. Animals that had no ticks on their bodies are excluded from this count; the reference group refers to animals that had ticks collected but those tick pools tested negative for CCHFV.
This category includes Butox.
This category includes Neochidol.
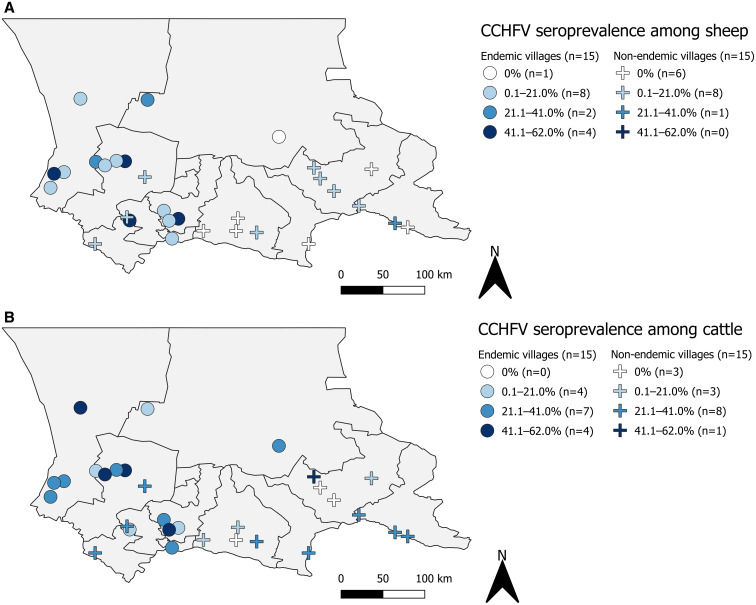
Accounting for weighting and combining villages with known and unknown history of CCHFV circulation, an estimated 5.7% (65/521, 95% CI: 3.1, 10.3) of sheep and 22.5% (132/454, 95% CI: 16, 31) of cattle in Zhambyl tested positive for anti-CCHFV IgG. Villages with a known history of CCHFV circulation reported a higher seroprevalence among sheep (16% versus 2.8%, P < 0.001) but not cattle (26% versus 20%, P = 0.42) (Supplemental Appendix S1). Seropositive livestock were detected in all 15 endemic villages (15/15 seropositive cattle; 14/15 seropositive sheep) and in 14 of the 15 nonendemic villages (12/15 seropositive cattle; 9/15 seropositive sheep). The estimated village-level cumulative CCHFV seroprevalence of sheep and cattle is presented in Figure 1. Seropositivity among livestock was not localized to any particular district, and village elevation was not associated with village-level seroprevalence among cattle (r[28] = −0.13, P = 0.48) or sheep (r[28] = −0.33, P = 0.07).
Figure 1.
Weighted seroprevalence of Crimean-Congo hemorrhagic fever virus (CCHFV) among (A) sheep and (B) cattle within selected endemic and nonendemic villages of Zhambyl, Kazakhstan, 2017. This figure appears in color at www.ajtmh.org.
Tick pools from three cattle and two sheep were positive for CCHFV at the time of collection; the estimated proportion of animals with CCHFV-positive tick pools was 3.8% (95% CI: 1.2, 12) among cattle and 2.4% (95% CI: 0.6, 9.5) among sheep. Seven species of ticks were observed among sheep and cattle (Hyalomma asiaticum, H. marginatum, H. turanicum, Rhipicephalus pumilio, R. turanicus, Ornithodorus, and Argas persicus). Most tick pools were of genus Hyalomma (138/246 among sheep, 174/247 among cattle) or genus Rhipicephalus (103/246 among sheep, 63/247 among cattle). Both CCHFV-positive tick pools collected from sheep were R. turanicus, and the three CCHFV-positive tick pools found on cattle were H. species (two spp. turanicum, one spp. asiaticum). Tick pool size (tick count per pool) ranged from 1 to 19 ticks for sheep, and 1 to 26 ticks for cattle. The presence of ticks was not significantly associated with CCHFV seropositivity or village endemicity status (Supplemental Appendix S1). Three CCHFV-positive tick pools were found in villages with an unknown history of CCHFV circulation.
Animal characteristics associated with seropositivity among sheep included residing in a village classified as endemic and having signs of recent illness (10.0% versus 1.3%, P < 0.001, see table note) (Table 1). Signs of illness were observed in 1.8% of all sheep (25/521, 95% CI: 0.8, 4.4%) and 2.0% of all cattle (12/454, 95% CI: 2.0, 6.5%). Being born on the farm was inversely associated with seropositivity among sheep (70% versus 91%, P = 0.046).
Herd movement was associated with seropositivity in sheep (P = 0.04), but not cattle (P = 0.84), with a greater proportion of seropositive sheep being part of a stationary herd relative to seronegative sheep (30% versus 7.5%) (Table 1). Among cattle, grazing beyond the village was associated with seropositivity (17% versus 7.5%, P < 0.001).
Among cattle, not having received any tick treatment in the last year was associated with seropositivity (86.9% versus 93.6%, P = 0.01). The distribution of tick treatments administered was statistically different between endemic and nonendemic villages (Supplemental Appendix S2), and between seropositive and seronegative sheep and cattle (Table 1). Tick treatments with diazinon were more common in endemic villages relative to nonendemic villages (sheep: 72% versus 37%; cattle: 82% versus 27%), whereas tick treatments with ivermectin were almost exclusively used in nonendemic villages (Supplemental Appendix S2).
As previously reported, among residents of sheep-owning households, five tested positive for anti-CCHFV IgG or IgM,8 all among people whose sheep tested IgG negative (Table 2). Among residents of cattle owning households, 11 people tested positive for anti-CCHFV IgG or IgM,8 4/127 (5.1%, 95% CI: 1.4, 16.8) among people whose cattle tested IgG positive, and 7/312 (1.2%, 95% CI: 0.4–4.0) among people whose cattle tested IgG negative. Among people whose animals were seropositive, 51% of sheep owners and 46% of cattle owners reported engaging in at least one high-risk activity with the potential for exposure to animal blood or infected ticks, such as slaughtering, birthing, or butchering livestock. There were no significant differences in animal seropositivity between households engaged and not engaged in high-risk activities, except that owners of seropositive sheep were more likely to report ever slaughtering an animal (43% versus 24%, P = 0.03).
Table 2.
Comparison of human–animal interactions between livestock owners in households with CCHFV seropositive and seronegative livestock in Zhambyl oblast, Kazakhstan, 2017
| Sheep | Cattle | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| CCHFV positive (N = 65) | CCHFV negative (N = 456) | CCHFV positive (N = 132) | CCHFV negative (N = 322) | |||||||
| Human—animal interactions | % Animals (N) | 95% CI | % Animals (N) | 95% CI | P value | % Animals (N) | 95% CI | % Animals (N) | 95% CI | P value |
| CCHFV positive human household resident* | 0.0% (0) | – | 1.1% (5) | 0.3–3.4% | – | 5.1% (4) | 1.4–16.8% | 1.2% (7) | 0.4–4.0% | 0.09 |
| Ever herded | 40.3% (31) | 20.0–64.5% | 24.0% (146) | 14.5–37.2% | 0.19 | 26.1% (40) | 10.3–52.1% | 37.5% (103) | 23.1–54.5% | 0.30 |
| Ever slaughtered | 42.5% (24) | 27.4–59.2% | 24.2% (149) | 15.7–35.5% | 0.03 | 22.9% (37) | 13.5–36.2% | 31.1% (96) | 20.3–44.4% | 0.13 |
| Ever birthed | 28.0% (22) | 13.2–49.8% | 13.9% (122) | 8.2–22.6% | 0.10 | 13.2% (34) | 5.5–28.4% | 14.9% (65) | 8.5–24.6% | 0.73 |
| Ever butchered | 39.0% (25) | 21.6––59.8% | 35.7% (170) | 27.4–44.8% | 0.73 | 41.5% (53) | 30.4–53.4% | 34.6% (115) | 23.5–47.6% | 0.16 |
| Any high-risk activity† | 51.3% (36) | 34.0–68.4% | 42.9% (233) | 34.2–52.1% | 0.37 | 46.2% (65) | 31.7–61.3% | 44.6% (156) | 35.3–54.2% | 0.80 |
| PPE used during high risk activities | 71.1% (16) | 36.6–91.3% | 69.5% (100) | 52.6–82.4% | 0.91 | 57.1% (25) | 25.1–84.1% | 60.0% (50) | 43.5–74.5% | 0.83 |
| Ever handled ticks with bare hands | 4.1% (7) | 1.5–11.2% | 1.6% (25) | 0.8–3.2% | 0.11 | 7.0% (9) | 1.2–32.8% | 4.0% (18) | 1.6–9.6% | 0.35 |
| Knowledge of transmission pathways | 0.25 | 0.95 | ||||||||
| Yes | 38.5% (15) | 20.3–60.6% | 38.8% (112) | 19.2–62.8% | 29.6% (26) | 13.3–53.4% | 28.5% (80) | 14.9–47.5% | ||
| No | 28.8% (21) | 12.4–53.5% | 15.5% (97) | 9.97–23.2% | 16.1% (26) | 6.09–36.1% | 14.4% (62) | 7.93–24.8% | ||
| Don’t know | 32.7% (21) | 17.5–52.8% | 45.8% (166) | 23.2–70.2% | 54.4% (54) | 30.6–76.3% | 57.1% (132) | 37.8–74.4% | ||
CCHFV = Crimean-Congo hemorrhagic fever virus. Boldface font indicates statistical significance at the P < 0.05 level.
Positive result on either IgM or IgG.
High-risk activities are those with potential exposure to animal blood or infected ticks, including butchering, slaughtering, and birthing; does not include herding animals.
Results from the multivariable logistic regression analysis of risk factors for CCHFV seropositivity and animal seropositivity are presented in Table 3. Relative to sheep belonging to stationary herds, the odds of seropositivity were 7.69 times lower among sheep belonging to nomadic herds (P = 0.035) and 6.25 times lower among sheep belonging to seminomadic herds (P = 0.055). In addition, the odds of seropositivity were 9.16 times higher among sheep with signs of illness (P = 0.006) relative to sheep without signs of illness.
Table 3.
Weighted logistic regression results of risk factors and CCHFV seropositivity among sheep and cattle in Zhambyl oblast, Kazakhstan, 2017
| Sheep | Cattle | |||||
|---|---|---|---|---|---|---|
| Animal risk factor | Adjusted OR | 95% CI | P value | Adjusted OR | 95% CI | P value |
| Sex (male) | 1.11 | 0.38–3.28 | 0.848 | 1.08 | 0.27–4.39 | 0.909 |
| Age (years) | 1.01 | 0.81–1.26 | 0.926 | 1.13 | 0.91–1.39 | 0.275 |
| Signs of illness present | 9.16 | 2.21–37.91 | 0.006 | 1.68 | 0.12–23.76 | 0.705 |
| Presence of ticks on body | 1.89 | 0.84–4.23 | 0.138 | 1.38 | 0.76–2.49 | 0.298 |
| Herd movement | ||||||
| Stationary* | – | – | – | – | – | – |
| Seminomadic | 0.16 | 0.03–0.94 | 0.055 | 1.45 | 0.58–3.64 | 0.435 |
| Nomadic | 0.13 | 0.02–0.77 | 0.035 | 1.38 | 0.76–2.49 | 0.861 |
CCHFV = Crimean-Congo hemorrhagic fever virus; OR = odds ratio. Boldface font indicates statistical significance at the P < 0.05 level. *Stationary herd movement used as reference group.
DISCUSSION
The broad distribution of CCHFV-positive livestock and ticks residing in villages classified as both endemic and nonendemic suggests that the entire Zhambyl region may be considered CCHFV endemic. Existing regional surveillance is passive and based on reporting of human cases and positive ticks, and may be insufficient to characterize the full geographic distribution of CCHFV. It is possible the virus is expanding into new previously nonendemic areas,14 as supported by the growing number of villages classified as endemic by regional surveillance in recent years, from 49 in 2014 to 68 in 2016. However, seroprevalence among animals is a more sensitive indicator of CCHFV endemicity. Implementing active routine animal surveillance is critical to monitoring the true geographic range of CCHFV in the region over time.
The estimated CCHFV seroprevalence among cattle in Zhambyl was 23%, comparable to findings from a study in Iran reporting a seroprevalence of 25% among cattle, but lower than estimates from other countries in the region, such as Pakistan (44%) and Turkey (36%).13,23,24 The estimated CCHFV seroprevalence among sheep in Zhambyl was 5.7%, with similar results from Turkey (6.3%).23 Studies from Pakistan and Iran reported a substantially higher CCHFV seroprevalence among sheep (33–59%).13,24 Seroprevalence estimates may be influenced by a variety of factors, including animal age, grazing habits, housings, and tick treatment; further work is needed to examine differences between regions and livestock species to identify drivers of infection.
The association between seropositivity and belonging to a stationary herd among sheep in the logistic regression model suggests that tick vectors responsible for CCHFV infection are likely present in housing commonly used by stationary herds. Combined with evidence from prior studies in which grazing outside of the village in regions where CCHFV is known to circulate in local tick populations is a known risk factor of CCHFV infection in animals, these findings suggest that vector populations are likely to be present in both environments.11 Further investigations are needed to examine CCHF control strategies within vector-infested animal housing and more generally on animal seropositivity. In addition, routine surveillance of ticks, both within animal housings and in nomadic grazing lands, will help improve disease detection.
While tick treatments were common among animals examined, the relatively high seroprevalence in both sheep and cattle and the high prevalence of tick infestation suggests that current tick treatment practices in the region may not be sufficient to prevent CCHFV infection. Ticks remain the primary mode of infection for livestock, and tick infestation continues to be an inherent risk factor, particularly in areas with active CCHFV circulation. The observed association between having received any tick treatment in the past year and an IgG negative result suggests that regular tick treatments continue to play an important role in overall prevention efficacy. Further work is needed to examine factors that influence the efficacy of specific treatment types and modes of application; furthermore, the existing tick prevention programs that provide livestock owners in endemic villages in Zhambyl with tick treatments should be expanded to include nonendemic villages.
As reported previously, high-risk human–animal interactions are common among livestock owners in Zhambyl, in both endemic and nonendemic villages.8 Despite the lack of an observed statistical association between seropositivity among livestock owners and their livestock, close contact with livestock, particularly high- risk activities, remains a critical exposure pathway to CCHFV for humans. The combination of high seroprevalence among livestock in both endemic and nonendemic villages, the frequency of high-risk activities among livestock owners, and the limited use of personal protective equipment (PPE) further emphasizes the need for ongoing targeted education and awareness efforts in Zhambyl.
This study is subject to several limitations. First, the large variability in herd sizes and the selection of only one individual animal from each household herd resulted in a wide distribution of sample weights. Weights were trimmed to the 80th percentile to reduce the effect on estimates; however, 95% confidence intervals remained wide, which may have obscured associations between risk factors and seropositivity. Second, this investigation was not designed to examine the efficacy of any individual tick preventive treatments, and the strong associations between tick treatment and village endemicity status made it difficult to examine the true associations between these variables and CCHFV seropositivity. Third, herds were occasionally composed of animals from multiple owners. In these cases, herd characteristics, as reported by a single owner, may not have been accurate representations of the composite herd. In addition, information on travel history of selected animals was not included. Fourth, serological testing identifies animals with past infection and may miss animals with current infections that have not yet seroconverted as IgG antibodies are not typically observed until after infection. Fifth, the small number of CCHFV-positive ticks observed limited the ability to make statistically meaningful associations with tick infection. Last, the geospatial analysis used only village locations, and did not include grazing locations for herds.
CCHFV continues to be a globally relevant high-consequence pathogen with high economic and human health implications. As climate change and globalized trade accelerate the expansion of tick-borne diseases from endemic areas to new regions, modern CCHFV surveillance systems that include both animal and vector surveillance are necessary for anticipating and documenting changes in the geographic distribution over time. Comprehensive veterinary care that incorporates validated tick treatments, accounting for the local degree of environmental resistance, and regular animal housing and vector control assessment components, remain the only reliable strategy available for CCHF prevention. Finally, tailored education and awareness campaigns that highlight the role of high-risk activities, such as slaughtering, butchering, and birthing livestock, in CCHFV transmission and the proper use of PPE to reduce these risks are needed for livestock owners, healthcare providers, and the general public in Kazakhstan.
Supplemental Material
ACKNOWLEDGMENTS
We are grateful to the Akimat of Zhambyl Oblast, the Health Protection Committee of the Ministry of Health and the Committee of Veterinary Control and Surveillance of the Ministry of Agriculture of Kazakhstan, Public Health Protection Department, Health Department, Sanitary Epidemiology Expertise Center, and the Veterinary Inspection unit of Zhambyl Oblast for their help in arranging and performing the investigation.
Note: Supplemental appendix appears at www.ajtmh.org.
REFERENCES
- 1. Bente DA Forrester NL Watts DM McAuley AJ Whitehouse CA Bray M , 2013. Crimean-Congo hemorrhagic fever: history, epidemiology, pathogenesis, clinical syndrome and genetic diversity. Antiviral Res 100: 159–189. [DOI] [PubMed] [Google Scholar]
- 2. Gargili A Estrada-Pena A Spengler JR Lukashev A Nuttall PA Bente DA , 2017. The role of ticks in the maintenance and transmission of Crimean-Congo hemorrhagic fever virus: a review of published field and laboratory studies. Antiviral Res 144: 93–119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Blair PW Kuhn JH Pecor DB Apanaskevich DA Kortepeter MG Cardile AP Polanco Ramos A Keshtkar-Jahromi M , 2019. An emerging biothreat: Crimean-Congo hemorrhagic fever virus in southern and western Asia. Am J Trop Med Hyg 100: 16–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Spengler JR Bergeron E Rollin PE , 2016. Seroepidemiological studies of Crimean-Congo hemorrhagic fever virus in domestic and wild animals. PLoS Negl Trop Dis 10: e0004210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Papa A Mirazimi A Koksal I Estrada-Pena A Feldmann H , 2015. Recent advances in research on Crimean-Congo hemorrhagic fever. J Clin Virol 64: 137–143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Hoogstraal H , 1979. The epidemiology of tick-borne Crimean-Congo hemorrhagic fever in Asia, Europe, and Africa. J Med Entomol 15: 307–417. [DOI] [PubMed] [Google Scholar]
- 7. Greiner AL Mamuchishvili N Kakutia N Stauffer K Geleishvili M Chitadze N Chikviladze T Zakhashvili K Morgan J Salyer SJ , 2016. Crimean-Congo hemorrhagic fever knowledge, attitudes, practices, risk factors, and seroprevalence in rural Georgian villages with known transmission in 2014. PLoS One 11: e0158049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Head JR et al. 2020. Risk factors for and seroprevalence of tickborne zoonotic diseases among livestock owners, Kazakhstan. Emerg Infect Dis 26: 70–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Spengler JR Estrada-Pena A Garrison AR Schmaljohn C Spiropoulou CF Bergeron E Bente DA , 2016. A chronological review of experimental infection studies of the role of wild animals and livestock in the maintenance and transmission of Crimean-Congo hemorrhagic fever virus. Antiviral Res 135: 31–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Christova I Panayotova E Trifonova I Taseva E Hristova T Ivanova V , 2017. Country-wide seroprevalence studies on Crimean-Congo hemorrhagic fever and hantavirus infections in general population of Bulgaria. J Med Virol 89: 1720–1725. [DOI] [PubMed] [Google Scholar]
- 11. Ibrahim AM Adam IA Osman BT Aradaib IE , 2015. Epidemiological survey of Crimean Congo hemorrhagic fever virus in cattle in East Darfur State, Sudan. Ticks Tick Borne Dis 6: 439–444. [DOI] [PubMed] [Google Scholar]
- 12. Mourya DT et al. 2015. Cross-sectional serosurvey of Crimean-Congo hemorrhagic fever virus IgG in livestock, India, 2013–2014. Emerg Infect Dis 21: 1837–1839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Zohaib A et al. 2020. Crimean-Congo hemorrhagic fever virus in humans and livestock, Pakistan, 2015–2017. Emerg Infect Dis 26: 773–777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Spengler JR Bergeron E Spiropoulou CF , 2019. Crimean-Congo hemorrhagic fever and expansion from endemic regions. Curr Opin Virol 34: 70–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Suliman HM Adam IA Saeed SI Abdelaziz SA Haroun EM Aradaib IE , 2017. Crimean Congo hemorrhagic fever among the one-humped camel (Camelus dromedaries) in Central Sudan. Virol J 14: 147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Knust B Medetov ZB Kyraubayev KB Bumburidi Y Erickson BR MacNeil A Nichol ST Bayserkin BS Ospanov KS , 2012. Crimean-Congo hemorrhagic fever, Kazakhstan, 2009–2010. Emerg Infect Dis 18: 643–645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Nurmakhanov T et al. 2015. Crimean-Congo haemorrhagic fever virus in Kazakhstan (1948–2013). Int J Infect Dis 38: 19–23. [DOI] [PubMed] [Google Scholar]
- 18. Abdiyeva K et al. 2019. Seroepidemiological and molecular investigations of infections with Crimean-Congo haemorrhagic fever virus in Kazakhstan. Int J Infect Dis 78: 121–127. [DOI] [PubMed] [Google Scholar]
- 19. Perfilyeva YV et al. 2020. Tick-borne pathogens and their vectors in Kazakhstan - a review. Ticks Tick Borne Dis 11: 101498. [DOI] [PubMed] [Google Scholar]
- 20. Kish L , 1965. Survey Sampling. New York, NY: John Wiley & Sons. [Google Scholar]
- 21. Texas A&M University CoVMBS , 2017. Foreign Animal Disease Investigation Manual (FAD PReP MANUAL 4-0). United States Department of Agriculture AaPHIS, Veterinary Services, ed: United States Department of Agriculture.
- 22. Ksiazek TG et al. 1999. Clinical virology of Ebola hemorrhagic fever (EHF): virus, virus antigen, and IgG and IgM antibody findings among EHF patients in Kikwit, Democratic Republic of the Congo, 1995. J Infect Dis 179 (Suppl 1): S177–S187. [DOI] [PubMed] [Google Scholar]
- 23. Ozan E Ozkul A , 2020. Investigation of Crimean-Congo hemorrhagic fever virus in ruminant species slaughtered in several endemic provinces in Turkey. Arch Virol 165: 1759–1767. [DOI] [PubMed] [Google Scholar]
- 24. Mostafavi E Haghdoost A Khakifirouz S Chinikar S , 2013. Spatial analysis of Crimean Congo hemorrhagic fever in Iran. Am J Trop Med Hyg 89: 1135–1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.