Abstract
Even though type 2 diabetes mellitus (T2DM) represents a worldwide chronic health issue that affects about 462 million people, specific underlying determinants of insulin resistance (IR) and impaired insulin secretion are still unknown. There is growing evidence that chronic subclinical inflammation is a triggering factor in the origin of T2DM. Increased C-reactive protein (CRP) levels have been linked to excess body weight since adipocytes produce tumor necrosis factor α (TNF-α) and interleukin 6 (IL-6), which are pivotal factors for CRP stimulation. Furthermore, it is known that hepatocytes produce relatively low rates of CRP in physiological conditions compared to T2DM patients, in which elevated levels of inflammatory markers are reported, including CRP. CRP also participates in endothelial dysfunction, the production of vasodilators, and vascular remodeling, and increased CRP level is closely associated with vascular system pathology and metabolic syndrome. In addition, insulin-based therapies may alter CRP levels in T2DM. Therefore, determining and clarifying the underlying CRP mechanism of T2DM is imperative for novel preventive and diagnostic procedures. Overall, CRP is one of the possible targets for T2DM progression and understanding the connection between insulin and inflammation may be helpful in clinical treatment and prevention approaches.
1. Introduction
Type 2 diabetes mellitus (T2DM) is becoming a prime global health problem. The prevalence of T2DM quadrupled during the past 35 years and still has a constant growth projection [1, 2]. Moreover, we anticipate that T2DM incidence would dramatically increase with increased adolescent obesity [3, 4].
Diabetes is a long-term metabolic disease in which the impaired ability of the body produce and/or respond to the hormone insulin. T2DM is characterized by abnormally elevated blood glucose levels, which affect the kidneys, heart, and blood vessels. Most diagnosed diabetes cases are type 1 diabetes mellitus (T1DM) or T2DM. T1DM, also known as juvenile diabetes, is characterized by an absolute insulin deficiency. On the other hand, T2DM is a progressive disease sustained by insulin resistance (IR) and beta cell dysfunction [5]. The most common way to diagnose diabetes in patients is by measuring fasting plasma glucose (FPG). FPG levels less than 100 mg/dL (5.6 mmol/L) are normal glucose levels in the blood, while levels ranging from 100 to 125 mg/dL (5.6 to 6.9 mmol/L) are considered a prediabetes indicator. Patients are only diagnosed with diabetes after at least two separate tests show FPG levels higher than 126 mg/dL (7 mmol/L). Hemoglobin A1C (HbA1C) is also an indicator of T2DM, which indicates the quality of diabetes treatment since it provides healthcare professionals with information about the average blood glucose levels in the past two or three months.
Accumulating evidence corroborates the crucial role of inflammation in T2DM pathologies [1, 6]. Low-grade inflammation characterized by elevated inflammatory protein levels, including C-reactive protein (CRP), is linked with T2DM pathogenesis [1, 7–9]. CRP, the typical inflammatory biomarker produced in the liver, is regulated by adipocyte-derived proinflammatory cytokines, including interleukin 6 (IL-6) and tumor necrosis factor α (TNF-α) [10, 11]. The level of CRP is usually low in healthy individuals but can elevate 100- to 200-fold or higher in acute systemic inflammation [12] and is chronically elevated in patients with T2DM. In individuals with T2DM, CRP levels range between 4.49 and 16.48 mg/L [13, 14] and among individuals with acute systemic inflammatory response syndromes from 31.08 [15] to 226.1 mg/L [16].
The production of CRP may be triggered by many metabolic and inflammatory factors associated with the development of T2DM, such as increased blood glucose, adipokines, and free fatty acid levels. In addition, an increased level of CRP represents a reliable predictor of vascular complications and progression of cardiovascular disease in diabetic patients [17, 18]. Furthermore, numerous human [1, 19, 20] and animal studies [21–23] demonstrated the associations of elevated serum CRP levels with obesity and the progression of IR leading to T2DM. These findings add to the notion that the inflammatory state demonstrated by higher CRP levels is an essential factor in the pathogenesis of T2DM. Numerous studies report a significant positive association between elevated CRP levels and the risk of T2DM development [1, 7–9, 24]. On the other hand, some studies lack this association after adjusting for many factors contributing to T2DM, including adiposity and hyperinsulinemia [20, 25]. Since increased body fat and obesity are among the main factors in the development of T2DM, which are also associated with increased risk for progression of obesity-related IR and inflammation, we review the published literature to collate and provide a comprehensive summary of the relationship between T2DM and CRP.
2. CRP
CRP was first described as a serum protein capable of precipitating C-polysaccharide pneumococcal cell walls during the acute phase of infective conditions in the presence of calcium [26–28]. We know CRP belongs to a conserved protein family called pentraxins and has been identified in several organisms ranging from arthropods to humans [29, 30]. Structurally, it is a 206 amino acid cyclic pentameric protein with five identical subunits noncovalently connected, with a molecular weight of ~23 kDa (Figure 1) [31]. Each of the five subunits is similar to a discoid orientation toward a central pore folded into two antiparallel two-layered β-sheets [32]. Native CRP (nCRP) dissociates to monomeric/modified isoform of CRP (mCRP) across lysophosphatidylcholine in platelets, apoptotic monocytic THP-1, and Jurkat T cells [33, 34]. Moreover, a study by Ji et al. demonstrates that nCRP, when bound to the cell membrane, dissociates into subunits while retaining some native conformation before entirely dissociating into mCRP subunits, which detaches from the membrane [35]. This intermediate isoform, termed mCRPm, seems to have similar biological functions as mCRP, including enhancing the classical complement pathway activation and promoting proinflammatory activity [36]. Unlike these two isoforms, nCRP displays more anti-inflammatory activities, probably because it limits the production of the membrane attack complex (MAC) and C5a, thus inhibiting the alternative complement activation [37]. mCRP has strong angiogenic effects, both in vitro and in vivo, and likely leads to the neovascularization of tissues in which it is deposited or synthesized [38]. Thus, treating CRP-mediated pathological conditions could include a novel therapeutic strategy that inhibits mCRP activity [39] or prevents the dissociation of nCRP into mCRP.
Figure 1.
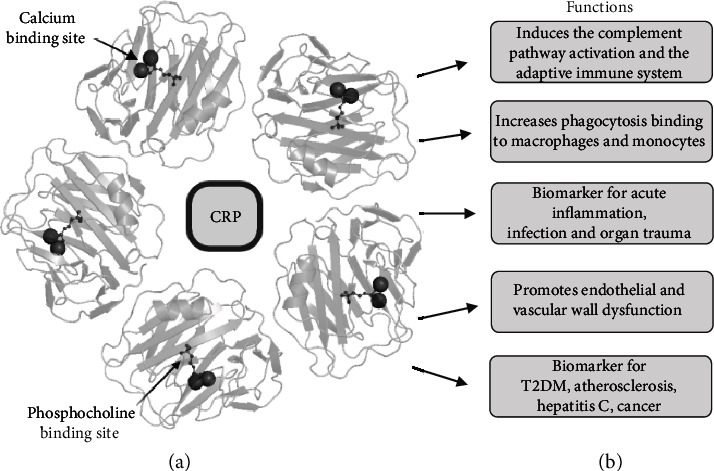
(a) C-reactive protein (CRP) crystal pentametric structure with calcium and phosphocholine binding sites. (b) Functions of CRP. CRP: C-reactive protein. Biorender.com and Protein Data Bank in Europe were used to generate the ribbon diagram of the CRP complex.
CRP can recognize and bind to endogenous damage-associated molecular patterns (DAMPs) and exogenous pathogen-associated molecular patterns (PAMPs). Thus, CRP initiates an immune response and contributes to eliminating various pathogens and damaged necrotic or apoptotic cells [40, 41]. After binding to a specific ligand, CRP manifests anti-inflammatory features by activating the C1q molecule in the classic complement pathway engaging C3, the main adhesion molecule of the complement system, and the terminal membrane attack complex, C5–C9. In this way, CRP leads to the opsonization of the pathogen [42, 43]. However, CRP also acts as a proinflammatory mediator that binds to the Fcγ receptors of IgG, leading to the release of proinflammatory cytokines [11, 44, 45].
2.1. CRP Levels and Detection
The Pentraxin 1 (PTX1) gene encodes CRP, and in humans, it is located on chromosome 1q21–q23 on the long arm and consists of a long 3′ untranslated region and 2 exons [46]. The first exon encodes two amino acids and a signal peptide, and the second exon encodes 204 amino acids [47]. Human CRP binds to phosphocholine (PCh) across five PCh-binding locations, and every individual subunit links two calcium ions [29]. Bacterial, eukaryotic, fungal, and endothelial cells induce PCh, and its binding to CRP is mediated mainly through Phe66 and Glu81 [32, 48, 49]. The C1q molecule binds and stimulates classical complement pathways on the CRP contrary site, particularly C1, C4, and C2 [43, 47]. Baseline CRP levels are affected substantially by polymorphisms in the noncoding regions in the promoter and the untranslated region. Polymorphisms of IL-6 and IL-1 genes, which stimulate CRP production, also affect the baseline CRP levels [50]. Groups of CRP single-nucleotide polymorphisms (SNPs) inherited together are known as five common main haplotypes, of which two are associated with higher baseline CRP levels, and two are associated with lower baseline CRP levels [51]. Moreover, these haplotypes influence the acute phase level of CRP and the development of several diseases [50, 52].
CRP participates in acute phase response to inflammation, infection, or organ trauma in humans, increasing up to 1000-fold within 24 to 72 hours [53, 54]. CRP is found in several cell types, including neurons, epithelial cells, monocytes, lymphocytes, and smooth muscle cells. However, the CRP gene is primarily induced in the hepatocytes due to elevated inflammatory cytokines, dominantly IL-6 [46]. Still, these extrahepatic sites do not impact CRP levels in plasma [47]. The CRP half-life is approximately 19 hours, and its average levels are generally presented as mg/L or mg/dL [55, 56]. Nonetheless, CRP levels differ among laboratories since there is no optimized standard. Levels below 0.3 mg/dL are considered physiological, while levels above 10 mg/dL indicate bacterial and virus infections and severe tissue damage [57].
Several methods are in use for CRP detection and measurement. Immunoturbidimetry is the most commonly used method for clinical CRP determination [58, 59]. Conventional enzyme-linked immunosorbent assay (ELISA) and fluorescence-linked immunosorbent assay (FLISA) have also been widely used for the quantification of CRP [33, 60]. The main limitations of these methods are the use of complex features and the lack of cost-effectiveness. Recently, CdSe/ZnS quantum dot-based FLISA and Histag-HRP functionalized nanoconjugate-based immunoassays were developed [61, 62]. The main advantages of these two assays are high sensitivity, reduced time for analysis, and expanded detection range for serum samples. In addition, the assays provide a lower detection limit (LOD) and, consequently, facilitate early detection of the CRP biomarker, which is imperative for prompt diagnosis. Novel electrochemical biosensor platforms and microfluxgate sensor systems contribute to these requirements [63–65].
Levels of CRP are directly correlated with the presence and elimination of inflammatory agents [57]. CRP's role as a diagnostic and prognostic biomarker has been established for acute infections [66–69], as well as for various chronic conditions, such as T2DM [1], atherosclerosis [70, 71], hepatitis C [72], and different types of cancer [73, 74]. Accumulating evidence suggests a connection between the activation of the complement system and the pathogenesis of T2DM [75–77].
3. T2DM and Inflammation
T2DM is a systemic, noncommunicable disease with multiple metabolic disorders, characterized by defects in insulin secretion and/or insulin action leading to hyperglycemia [78]. Chronic hyperglycemia induces oxidative stress, inflammation, and local and whole-body failures [79]. Of all the types of diabetes, two forms are most common: T1DM and T2DM. T1DM occurs due to autoimmune destruction of a critical mass of pancreatic β cells, which causes a lack of insulin synthesis and secretion [80]. Therefore, T1DM is an autoimmune disease with the rate of β cell destruction varying from rapid to slow descent [80, 81]. T1DM patients are rarely obese and often develop other autoimmune disorders such as Hashimoto's thyroiditis, Graves' disease, and vitiligo [79]. This suggests that multiple genetic and environmental factors may induce the autoimmune destruction of β cells. Contrarily, T2DM is the most prevalent form, with a frequency of 90-95% of all cases in the population. T2DM characterizes a whole spectrum of events, from insulin secretion defects to the impaired action of different enzymes regulated by insulin [79, 82]. There are numerous causes of T2DM, but unlike T1DM, autoimmune destruction of β cells never occurs [79]. The disease at earlier stages frequently goes undiagnosed because classic symptoms develop gradually as hyperglycemia develops, and insulin levels in such patients are often normal or elevated [83]. Age, obesity, and lack of physical activity are probably the most crucial factors affecting the development of T2DM [79, 84]. Long before the clinical manifestation of T2DM, IR occurs, characterized by hyperinsulinemia often combined with obesity, hypertension, and dyslipidemia [85]. During IR, compensatory hyperinsulinemia helps maintain normal glucose levels [83], but β cells lose their ability to overcome IR through hypersecretion, which leads to hyperglycemia [83, 85]. Subclinical inflammation is an essential part of IR, and various inflammatory markers correlate with IR, including CRP [82, 86, 87]. The long-term activation of the innate immune system results in the development and progression of T2DM instead of reestablishing the normal physiological state (Figure 2). Besides CRP, the innate immune system produces acute phase response proteins, fibrinogen, and serum amyloid A, whose levels noticeably change in response to infection, tissue injury, or inflammation [88]. High levels of CRP, fibrinogen, sialic acid, serum amyloid A, and low albumin and transferrin levels have been linked with T2DM occurrence [89].
Figure 2.
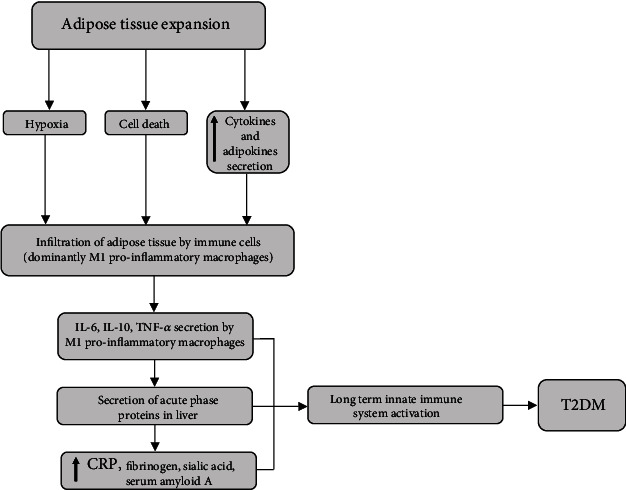
T2DM development as a result of chronic low-grade inflammation. CRP: C-reactive protein; IL-6: interleukin 6; IL-10: interleukin 10; i.p.: intraperitoneal; TNF-α: tumor necrosis factor α; T2DM: type 2 diabetes mellitus.
Adipose tissue, especially visceral white adipose tissue (WAT), plays a significant role in T2DM's inflammatory process and development. In addition, studies demonstrated that human adipocytes could produce CRP under the stimulation of several proinflammatory cytokines, suggesting a link between obesity and its comorbidities, including IR [90–92]. CRP mRNA levels in human adipose tissue also positively correlated with IL-6 mRNA levels. CRP expression in vitro was also increased by IL-6 and lipopolysaccharide stimulation, contributing to the elevated plasma CRP levels found in obese individuals [93]. Moreover, human CRP overexpression in transgenic mice fed a high-fat diet contributed to the development of IR, hepatosteatosis, adiponectin downregulation, and expression of proinflammatory cytokines in epididymal adipose tissue [94]. These data indicate the role of CRP in the pathogenesis of obesity-induced metabolic disorders [94]. A recent study reported a significant decrease in weight gain and food intake, and improved insulin sensitivity was observed in CRP knockout rats placed on a high-fat diet. These results suggest that CRP is not only a biomarker of inflammation but also has a crucial role in energy balance, body weight, insulin sensitivity, and glucose homeostasis [95]. In addition, numerous molecules such as cytokines and other bioactive substances involved in the inflammatory pathways are produced and secreted by WAT [96]. The abdominally distributed WAT plays an essential role in the inflammatory process [79].
Furthermore, macrophages and immune cells infiltrate as the adipose tissue expands, contributing to local and whole-body low-grade inflammation. Adipose tissue expansion does not follow simultaneous capillaries' development, which causes adipocytes to become too distant from the vasculature [97]. Thus, hypoxia, adipocyte cell death, and increased secretion of chemokines and adipokines may be part of the mechanisms that initiate adipose tissue inflammation [98]. Infiltration of adipose tissue is accompanied by a decrease in anti-inflammatory M2-type macrophages, while the number of proinflammatory M1-type macrophages increases [96]. Although T2DM does not imply autoimmune destruction of β cells, the inflammatory process in the pancreas islets does occur. It is still unclear what induces that process, but it appears to be highly dependent on IL-1 action [96].
Adipocyte-derived bioactive metabolites like leptin and adiponectin are also involved in T2DM pathogenesis [96]. The leptin levels in serum are directly proportional to the total fat mass; its production increases during inflammation. Also, leptin activates and modulates innate and adaptive immune responses and promotes proinflammatory pathways [96, 99]. Contrarily to leptin, adiponectin is involved in anti-inflammatory pathways. Thus, low adiponectin levels are associated with T2DM incidence, which shows an inverse relationship between plasma adiponectin levels and CRP levels [95, 100]. Additionally, TNF-α, IL-6, and IL-10 are essential cytokines produced by adipocytes and immune cells. TNF-α and IL-6 are known proinflammatory factors, whereas IL-10 exhibits anti-inflammatory properties [96], and IL-6 predominantly controls CRP production by hepatocytes [86]. Besides IL-6 and TNF-α, the liver produces IL-1, which stimulates the synthesis of acute-phase proteins [89]. Also, the correlation between fasting insulin and CRP levels implicates the link between IR and inflammatory processes [78, 101, 102].
Inflammation of adipose tissue and high production of TNF-α, IL-6, and IL-1β in obesity is vital for T2DM incidence and progression [103]. Some authors indicated that TNF-α contributes to the development of IR in skeletal muscle of human individuals by suppressing Akt substrate [104] and stimulating IL-18 expression [105]. Furthermore, TNF-α constrains insulin-stimulated glucose uptake and endothelium vasodilation [106]. In addition, TNF-α receptors mediate the upregulation of the NF-κB pathway and influence proteins that disturb insulin signaling and proinflammatory response [107]. Nevertheless, IL-6 affects insulin-degrading enzyme expression and activity in the liver and skeletal muscle tissues, and modulation of this enzyme may contribute to T2DM and obesity [108]. According to a new meta-analysis, IL-6 indeed mediates chronic inflammation in T2DM. However, IL-6 influence on the general population seems insufficient, and acting on the IL-6 pathway may not reduce the risk for T2DM occurrence [109]. Even though IL-6 is essential for liver homeostasis, only a few cell types express IL-6 receptors, including hepatocytes, and its persistent activation is associated with liver pathologies [110]. Studies demonstrate that IL-6-inducible protein SOSC-3 promotes IR by direct binding to insulin receptors and inhibiting its kinase activity [111]. However, the results of one study indicate anti-inflammatory properties and the homeostatic role of IL-6 in obesity-associated inflammation and IR [112]. Also, it was demonstrated that IL-6 increases the responsiveness of macrophages to IL-4 and thus balances its shifting toward proinflammatory M1 macrophages [112]. In addition, the results from many studies also confirmed the pleiotropic nature of IL-6 and indicate that the effects of IL-6 on inflammation differs depending on the duration of exposure, tissue type, and factors such as concentration and source of IL-6 [113–117].
Growing evidence indicates the role of gut microbiota in the immune system regulation and pathogenesis of T2DM [118–120]. The product of certain bacteria triggers an inflammatory cascade, including recruitment of interleukins and CRP [121, 122], which leads to impaired insulin action and T2DM development [123, 124]. The level of imidazole propionate (ImP), a microbial-produced histidine metabolite, is higher in subjects with T2DM [125]. Furthermore, the proposed mechanism by which microbial-derived ImP diminished glucose metabolism includes activating the p38γ-mTOR1-S6K1 signaling cascade that caused insulin receptor substrate degradation and inflammation [125, 126]. In addition, increased circulating levels of LPS have been recognized as an important marker that implies a link between variations in microbiota composition and inflammation in T2DM [122]. LPS promotes IR via stimulation of Toll-like receptors on adipocytes, upregulation of NFkB, and activation of cytokines TNF-α and IL-6 [127–129]. In addition, lower diversity in the gut microbiota is associated with increased white blood cell counts and high sensitivity CRP (hs-CRP) levels [121, 130].
Some evidence indicates the role of CRP in diabetes-induced microvascular complications, such as neuropathy, retinopathy, and nephropathy. Elevated glucose levels could trigger microvascular alterations and increased production of inflammatory factors, including CRP, IL-6, and TNF-α [131]. Serum hs-CRP is linked with diabetic neuropathy occurrence, one of the most prevalent diabetes complications [132–134]. Nevertheless, in male and female patients, symptomatic peripheral diabetic neuropathy and inflammation are related to endothelial dysfunction and elevated CRP serum levels [135]. It is assumed that in patients with T2DM peripheral neuropathy, increased CRP levels are positively correlated with inflammation grade [133]. Furthermore, according to data from a large cohort study where participants were monitored for one year, hs-CRP levels above 2.5 mg/L could predict neuropathy complications in T2DM [136]. Considering hs-CRP low-cost and accessibility features, it might be a useful predictable biomarker for neurovascular disorder in T2DM [131].
Diabetic retinopathy (DR) is the leading cause of visual loss worldwide. Several studies have investigated the relationship between CRP levels and DR with inconsistent results. Qiu et al. observed a positive correlation between elevated hs-CRP levels in the blood and DR onset and progression [137]. In contrast, Song et al. reported that CRP levels could be associated only with the severity of DR [138]. Recently, it was shown that CRP levels could be associated with the stage of DR in T2DM patients [139]. However, previous studies demonstrated that patients with higher CRP and BMI levels were less likely to develop DR [140]. Inconsistent results from various studies may be caused by ethnical differences in CRP and BMI levels and excluding some clinical parameters such as duration of DM and complex genetic and environmental variations.
Diabetic nephropathy (DN) is a leading cause of end-stage renal disease. Numerous studies confirmed higher CRP levels in T2DM patients with DN [141–145]. A study by Tang et al. demonstrated the pathogenic role of CRP in renal inflammation and fibrosis using diabetic animal models [141]. The meta-analysis also confirmed higher hs-CRP levels in T2DM patients with DN than in healthy subjects and T2DM patients without DN [142]. Hayashino et al. reported that serum hs-CRP levels could be a helpful factor for predicting the risk of DN developing in T2DM patients [143]. Additionally, it was shown that higher hs-CRP levels were associated with DN complications in T2DM patients [145].
Although these findings open new doors in understanding diabetes pathology, further research is needed to answer current ambiguities.
4. T2DM and CRP
4.1. Evidence from Animal Studies
Numerous animal studies have shown the essential role of CRP in infections and inflammatory processes (Table 1). CRP is synthesized in response to monocytic mediators such as IL-1 and IL-6 in the acute phase of infections [11]. CRP recognizes and binds to specific polysaccharides in the bacterial wall and induces further complement pathway activation, leading to the opsonization of pathogens [146]. Moreover, there is evidence of CRP involvement in proliferation and apoptotic processes through activation of Fc receptors and the consequent production of proinflammatory mediators and proapoptotic cytokines [147]. In addition, growing evidence show CRP is not only an inflammatory marker as its level is proven to be elevated in T2DM cases (Table 1).
Table 1.
CRP values in animal model studies.
| Animal model | T2DM induction | CRP values | Diabetes-associated disorders | Ref. |
|---|---|---|---|---|
| Sprague-Dawley male rats | High-glucose/high-fat diets (15 weeks), and streptozotocin (single dose 30 mg/kg i.p.) | Increased | Irregular cardiac muscle fibers and degenerative necrosis | [148] |
| Sprague-Dawley male rats | High-fat diet (10 weeks), and streptozotocin (single dose 25 mg/kg, i.p.) | Increased | Altered oxidative stress parameters in the pancreas and serum | [196] |
| Sprague-Dawley male rats | High-fat and high-sugar (5 weeks), and streptozotocin (7 days, dose 30 mg/kg, i.p.) | Increased | Injury of the intestinal mucosa reduced antioxidant capacity | [197] |
| Wistar male rats | High-fat diet (10 weeks), and streptozotocin (single dose 40 mg/kg i.p.) | Increased | Elevated systolic blood pressure, congested blood vessels, necrosis, and inflammation of the heart, pancreas, liver, and kidney. | [198] |
| Wistar male rats | High-fat diet (10 weeks) and alloxan (single dose 50 mg/kg, i.p.) | Similar to the control values | Physiological pancreas cytoarchitecture, altered lipid profile | [151] |
| Wistar male rats | Streptozotocin (60 mg/kg i.p.) | Increased | Aorta smooth muscle cell proliferation, altered lipid profile | [21] |
| Wistar male rats | Streptozotocin (single dose 55 mg/kg i.p.) | Increased | Increased cytokine levels | [22] |
| Sprague-Dawley male rats | Streptozotocin (two-dose within two days 40 mg/kg i.p.) | Increased | Endothelial dysfunction, modified protein profile | [149] |
| Wistar male rats | Alloxan monohydrate (single dose 120 mg/kg i.p.) | Increased | Hypercholesterolemia | [150] |
| Wistar male rats | High-fat, high-fructose, high-casein diet (3 months) and streptozotocin (60 mg/kg in 2 equally doses with a 12 h interval i.p. and 20–30 mg/kg after 2 weeks for some animals) | Increased | Systolic blood pressure deviations | [155] |
| Wistar male rats | Streptozotocin (single dose 55 mg/kg i.p.) | Increased | Altered lipid profile increased glycosylated hemoglobin and liver enzymes, decreased levels of antioxidant enzymes | [156] |
| Wistar male rats | Alloxan monohydrate (single dose 120 mg/kg i.p.) | Increased | Altered oxidative stress parameters in serum | [153] |
| Albino rats | Alloxan monohydrate (single dose 120 mg/kg i.p.) | Increased | Altered oxidative stress parameters in serum, pathological pancreas cytoarchitecture | [154] |
| Wistar male rats | Streptozotocin three doses (for 3 continuous days 45 mg/kg i.p.) | Increased | Altered oxidative stress parameters in the kidneys | [199] |
CRP: C-reactive protein; T2DM: type 2 diabetes mellitus.
Zou et al. showed elevated serum CRP levels in male streptozotocin-induced Sprague-Dawley rats compared to untreated rats [148]. The same authors noticed degenerative changes in the heart of streptozotocin-induced rats, such as irregular cardiac muscle fibers and degenerative necrosis [148]. Furthermore, Shirpoor et al. investigated the effect of vitamin E on oxidized low-density lipoprotein, lipid profile, CRP, and VSMC proliferation of rat aorta in streptozotocin-induced Wistar rats. They found elevated CRP levels in the diabetic rats compared to the untreated rats [21]. Additionally, the same authors reported that the antioxidative effects of vitamin E attenuated the level of CRP and arterial complications in diabetic rats [21]. Using the same model, Talebi-Garakani and Safarzade showed that four weeks of resistance training decreases serum inflammatory markers in diabetic rats, including CRP levels [22]. These authors also reported increased CRP levels in the diabetic rats compared to the untreated ones.
We have previously shown that rats with high-fat diet-induced obesity and IR develop cardiac hypertrophy and elevated CRP levels in serum [23]. We also showed that estradiol treatment leads to CRP level reduction and significantly reduces heart mass in treated rats compared to untreated obese rats [23]. In another study, Cho and colleagues reported the differentially expressed proteins in the kidney, eye, aorta, and serum of diabetic rats compared to the controls [149]. They reported elevated serum CRP levels in diabetic rats compared to untreated rats. Beyond the high CRP level, they studied protein alterations in the vasculature and found three potential biomarkers in aorta lysate samples that could present early signs of diabetic vascular complications development [149]. The Asgary et al. study also indicated CRP increases in alloxan-induced diabetic rats compared to untreated. The same authors concluded that four weeks of pumpkin powder administration decreased glucose, cholesterol, triglyceride, LDL, and CRP levels in diabetic rats compared to untreated diabetic rats [150]. Interestingly, Ige et al. reported similar CRP values, and also, histological pancreas findings were entirely physiological, with small amounts of secretions in diabetic and control group rats [151].
The variance in study design may explain the differences in CRP levels in diabetic animals. Although alloxan induces diabetes through a different mechanism (oxidative stress through Fenton reaction) than streptozotocin (alkylation of DNA) [152], higher doses of alloxan applied to male and female rats induce the same effects on diabetes development and alterations of CRP levels in circulation, as streptozotocin [150, 153, 154]. Some authors suggest that infections in T2DM should be treated with antibiotics and indicate that medication with fluoroquinolone antibiotics may decrease elevated CRP values in diabetic rats [155]. Nevertheless, Almatroodi et al. showed that Thymoquinone, the key active component of the medicinal plant Nigella sativa, improved CRP, antioxidant enzymes, liver enzymes, and inflammatory markers levels in diabetic rats [156].
4.2. Evidence from Human Studies
Accumulating evidence from human-based studies suggests that inflammation contributes to the pathogenesis of T2DM (Table 2). Several human studies show elevated CRP levels correlate with the development of T2DM, even without adjustment of other parameters, such as adiposity, hyperinsulinemia, hypertriglyceridemia, and low HDL cholesterol [1, 7–9, 24].
Table 2.
CRP values in human studies.
| Subjects | CRP values | Association of CRP and T2DM incidence | Ref |
|---|---|---|---|
| Men | Increased | Positive | [10] |
| Women | Increased | Positive | [19] |
| Man and women | Increased | Positive | [9] |
| Man and women | Non-significant | Positive | [158] |
| Man and women | Increased | Positive | [163] |
| Man and women | Increased | Positive | [164] |
| Man and women | Not measured | Positive | [165] |
| Men and women | Increased | Not measured | [170] |
| Men and women | Increased | Non-significant | [1] |
| Men and women | Increased | Positive | [173] |
| Men and women | Increased | Positive | [176] |
CRP: C-reactive protein; T2DM: type 2 diabetes mellitus.
The West of Scotland Coronary Prevention Study showed that elevated CRP indicates T2DM development in men (middle-aged), independent of established risk factors, such as fasting plasma triglyceride, body mass index (BMI), and glucose [10]. Pradhan et al. [19] also observed elevated CRP levels in diabetic middle-aged women compared to the healthy control, supporting a possible role of inflammation in the pathogenesis of T2DM. Furthermore, Han et al. reported sex differences in the association of elevated CRP levels with the incidence of T2DM [9]. This strong association in women may be explained by the hormone differences and the higher adiposity percentage [157]. A study by Nakanishi et al. [158] showed the influence of CRP on T2DM development in Japanese Americans and not in the original Japanese population, which probably indicates the impact of different lifestyles on T2DM manifestation. The possible explanation considering the link between CRP and T2DM development may be the role of oxidative stress in inducing hyperglycemia [159], which further promotes inflammatory response and elevation of CRP [160]. Furthermore, it was found that oxidative stress might impair insulin endocytosis in endothelial cells [161] and thus could lead to endothelial dysfunction and IR [158, 162]. Doi et al. [163] reported a clear link between elevated CRP levels and obesity-induced hyperglycemia and T2DM in relatively lean Asian populations (Japanese population) in both sexes even after adjustment for comprehensive risk factors related to IR. Also, in the study by Marques-Vidal et al. [164], higher hs-CRP levels were associated with all T2DM and IR markers, and these associations persevered after multivariate adjustment. Similarly, the study showed that participants with impaired glucose tolerance had higher hs-CRP levels than euglycemic subjects, although this difference became nonsignificant after BMI adjustment. Likewise, Akbarzadeh et al. [165] reported a significant positive association between the hs-CRP level and IR markers (HOMA-IR and FIRI) and a negative association between hs-CRP level and insulin sensitivity markers (QUICKI, McAuley. and Bennett indexes). Some studies showed an association between elevated levels of CRP and leptin in diabetic, obese, and CVD subjects [166–169], suggesting the possible role of CRP in modulating leptin action [170]. In addition, variations in leptin levels were independently associated with CRP [171], indicating that a regulatory loop interrelates CRP and leptin levels. Furthermore, exploring the mechanisms of leptin resistance adds to the notion that CRP, particularly the smaller mCRP, may change the action of leptin by binding to the extracellular domain of the leptin receptor [170]. In this way, CRP contributes to the pathogenesis of obesity-related diseases, including T2DM and CVD [166–169, 172]. A study by Kanmani et al. [1] showed a positive association between CRP levels and the incidence of T2DM in a large Korean population. In addition, the association was more noticeable among the older group (≥50 years), and the combination of CRP levels with obesity and hypertension led to increased incidence of T2DM. Lainampetch et al. [173] reported that patients with increased baseline levels of CRP were at increased risk of developing T2DM. These findings support the premise that CRP may indirectly impair insulin sensitivity and production due to increased systemic inflammation through innate immune response alteration [174]. Also, elevated CRP levels influence the production of adhesion molecules, including E-selectin, intercellular adhesion molecule-1 (ICAM-1), and vascular cell adhesion molecule-1 (VCAM-1) that are directly involved in the regulation of insulin action and local IR [175]. A recent retrospective case-control study [176] imparted that elevated hs-CRP and FPG, insulin, HbA1c, HOMA-IR, and IL-6 were found in T2DM patients. The same authors concluded that obesity-induced dyslipidemia (also demonstrated in the study) causes IR and the subsequent increase in levels of inflammatory markers [176]. Chronic inflammation in obesity disrupts glucose homeostasis causing a persistent increase in blood glucose levels [87, 177]. Also, IL-6 produced by the adipose tissues may stimulate CRP secretion [178], which further increases IR and initiates low-grade inflammation leading to the development of T2DM [176]. On the other hand, some studies suggest that higher CRP levels and T2DM development are attenuated or missing after adjustment to a wide range of confounding factors, including adiposity and insulin sensitivity [20, 25]. The degree of adiposity and baseline glycemia adjustment could explain the heterogeneity between studies and suggests that CRP might not be an independent risk factor for T2DM development [6].
Regarding testing CRP levels, there is a standard CRP assay measuring lower (baseline) levels of this biomarker, contributing to the diagnosis of acute inflammation. Also, a frequently used and a more sensitive hs-CRP assay reflects low-grade chronic inflammatory processes having a predictive value of future CVD risk [179]. In addition, emerging evidence supports the use of hs-CRP levels for CVD risk assessment in IR-diabetic and nondiabetic subjects [180, 181]. The limitation of the abovementioned human studies, which used CRP and not hs-CRP, might be apparent. However, treatment with pioglitazone, which has insulin-sensitizing and anti-inflammatory properties (combined with antilipidemic statin therapy) decreases hs-CRP levels independently of the glucose-lowering in T2DM and nondiabetic patients with increased hs-CRP levels [182, 183]. This suggests that hs-CRP levels in T2DM might not necessarily establish a cause-effect pathophysiological association.
Although the clinical relationship between diabetes and increased level of CRP is well established, the molecular mechanisms by which CRP potentially induces diabetes are yet to be clarified. Particular progress has been made in investigating multiple therapeutic approaches targeting different inflammatory factors [177]. Treatment of T2DM patients with IL-1 receptor blocker [184, 185] or IL-1β antibodies [186–189] reduced levels of IL-6 and CRP as markers of systemic inflammation, with simultaneous improvement of glycemia and insulin secretion. Treatment of obese and diabetic subjects with IκB kinase complex β (IKKβ)/nuclear factor κB (NF-κB) inhibitor, as a central proinflammatory player, decreased the level of CRP and improved insulin sensitivity and glycemia [190, 191]. Also, some studies showed beneficial effects of TNF antagonism therapy on CRP level reduction, with a tendency to improve β cell function, but without impact on insulin sensitivity [192, 193].
5. Conclusions
Numerous prospective studies reported the association between serum CRP level and risk of incident T2DM. However, there is heterogeneity between studies, with some showing an independent positive association of CRP with T2DM incidence [1, 19, 20], while others demonstrate no association after adjustment for adiposity and IR [20, 25]. Since it is unequivocally associated with the development of prediabetes and diabetes-induced vascular complications, the elevated CRP might be an indirect risk factor for T2DM progression. Additionally, elevated CRP levels should be considered one more parameter in the overall assessment of T2DM risk, besides elevated FPG and HbA1C levels, abnormal OGTT, hyperinsulinemia, etc. Thus, elevated CRP levels in patients with higher T2DM risk must warn the clinician to perform available diagnostic procedures to confirm diabetes [194, 195]. Since the incidence of T2DM is expected to increase in the following years dramatically [2], further analysis of the CRP and diabetes association is needed to provide an adjunctive method for early detection of risk for this disease.
Acknowledgments
This work was funded by the Ministry of Education, Science and Technological Development of the Republic of Serbia (Contract No. #451-03-9/2021-14/200017) and KAUST grant OSR#4129 (awarded to E.R.I. and V.B.B.), which also supported M.O. M.E. has been supported by the KAUST Office of Sponsored Research (OSR) Award no. FCC/1/1976-20-01, and TG by the King Abdullah University of Science and Technology (KAUST) Base Research Fund (BAS/1/1059-01-01).
Disclosure
This work is part of the collaboration between the Department of Radiobiology and Molecular Genetics, “VINČA” Institute of Nuclear Sciences-National Institute of the Republic of Serbia, University of Belgrade, Belgrade, Serbia, and Computational Bioscience Research Center (CBRC) at King Abdullah University of Science and Technology (KAUST).
Conflicts of Interest
The authors confirm that this article's content has no conflict of interest.
Authors' Contributions
Julijana Stanimirovic and Jelena Radovanovic share the first authorship.
References
- 1.Kanmani S., Kwon M., Shin M. K., Kim M. K. Association of C-reactive protein with risk of developing type 2 Diabetes mellitus, and role of obesity and hypertension: a large population-based Korean cohort study. Scientific Reports . 2019;9(1):p. 4573. doi: 10.1038/s41598-019-40987-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Who. Global Report on Diabetes . World Health Organization; 2016. [Google Scholar]
- 3.Chen L., Magliano D. J., Zimmet P. Z. The worldwide epidemiology of type 2 diabetes mellitus--present and future perspectives. Nature Reviews Endocrinology . 2011;8(4):228–236. doi: 10.1038/nrendo.2011.183. [DOI] [PubMed] [Google Scholar]
- 4.Pulgaron E. R., Delamater A. M. Obesity and type 2 diabetes in children: epidemiology and treatment. Current Diabetes Reports . 2014;14(8):508–508. doi: 10.1007/s11892-014-0508-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Russo G. T., Giorda C. B., Cercone S., Nicolucci A., Cucinotta D., on behalf of BetaDecline Study Group Factors associated with beta-cell dysfunction in type 2 diabetes: the BETADECLINE study. PLoS One . 2014;9(10, article e109702) doi: 10.1371/journal.pone.0109702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lee C. C., Adler A. I., Sandhu M. S., et al. Association of C-reactive protein with type 2 diabetes: prospective analysis and meta-analysis. Diabetologia . 2009;52(6):1040–1047. doi: 10.1007/s00125-009-1338-3. [DOI] [PubMed] [Google Scholar]
- 7.Wang X., Bao W., Liu J., et al. Inflammatory markers and risk of type 2 diabetes: a systematic review and meta-analysis. Diabetes Care . 2013;36(1):166–175. doi: 10.2337/dc12-0702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bertoni A. G., Burke G. L., Owusu J. A., et al. Inflammation and the incidence of type 2 diabetes: the Multi-Ethnic Study of Atherosclerosis (MESA) Diabetes Care . 2010;33(4):804–810. doi: 10.2337/dc09-1679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Han T. S., Sattar N., Williams K., Gonzalez-Villalpando C., Lean M. E. J., Haffner S. M. Prospective study of C-reactive protein in relation to the development of diabetes and metabolic syndrome in the Mexico City Diabetes Study. Diabetes Care . 2002;25(11):2016–2021. doi: 10.2337/diacare.25.11.2016. [DOI] [PubMed] [Google Scholar]
- 10.Freeman D. J., Norrie J., Caslake M. J., et al. C-reactive protein is an independent predictor of risk for the development of diabetes in the West of Scotland Coronary Prevention Study. Diabetes . 2002;51(5):1596–1600. doi: 10.2337/diabetes.51.5.1596. [DOI] [PubMed] [Google Scholar]
- 11.Sproston N. R., Ashworth J. J. Role of C-reactive protein at sites of inflammation and infection. Frontiers in Immunology . 2018;9:754–754. doi: 10.3389/fimmu.2018.00754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gabay C., Kushner I. Acute-phase proteins and other systemic responses to inflammation. New England Journal of Medicine . 1999;340(6):448–454. doi: 10.1056/NEJM199902113400607. [DOI] [PubMed] [Google Scholar]
- 13.Vinagre I., Sánchez-Quesada J., Sánchez-Hernández J., et al. Inflammatory biomarkers in type 2 diabetic patients: effect of glycemic control and impact of LDL subfraction phenotype. Cardiovascular Diabetology . 2014;13(1):p. 34. doi: 10.1186/1475-2840-13-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tabassum R., Mia A. R., Reza-Ul-Haq K. M., Yesmin M., Faruqui J. M. C-reactive protein level in type-2 diabetic patients attending Mymensingh Medical College Hospital, Mymensingh. Mymensingh Medical Journal . 2017;26(1):56–60. [PubMed] [Google Scholar]
- 15.Yachnyk I. M. C-reactive protein for differential diagnosis of sepsis and systemic inflammatory response syndrome. Galician Medical Journal . 2015;22(4):53–56. [Google Scholar]
- 16.Goldberg I., Shalmon D., Shteinvil R., et al. A second C-reactive protein (CRP) test to detect inflammatory burst in patients with acute bacterial infections presenting with a first relatively low CRP. Medicine . 2020;99(42, article e22551) doi: 10.1097/MD.0000000000022551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mugabo Y., Li L., Renier G. The connection between C-reactive protein (CRP) and diabetic vasculopathy. Focus on preclinical findings. Current Diabetes Reviews . 2010;6(1):27–34. doi: 10.2174/157339910790442628. [DOI] [PubMed] [Google Scholar]
- 18.Esser N., Paquot N., Scheen A. J. Inflammatory markers and cardiometabolic diseases. Acta Clinica Belgica . 2015;70(3):193–199. doi: 10.1179/2295333715Y.0000000004. [DOI] [PubMed] [Google Scholar]
- 19.Pradhan A. D., Manson J. E., Rifai N., Buring J. E., Ridker P. M. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. JAMA . 2001;286(3):327–334. doi: 10.1001/jama.286.3.327. [DOI] [PubMed] [Google Scholar]
- 20.Festa A., D'Agostino R Jr, Tracy R. P., Haffner S. M., Insulin Resistance Atherosclerosis Study Elevated Levels of Acute-Phase Proteins and Plasminogen Activator Inhibitor-1 Predict the Development of Type 2 Diabetes: The Insulin Resistance Atherosclerosis Study. Diabetes . 2002;51(4):1131–1137. doi: 10.2337/diabetes.51.4.1131. [DOI] [PubMed] [Google Scholar]
- 21.Shirpoor A., Norouzi L., Nemati S., Khadem Ansari M. H. Protective effect of vitamin E against diabetes-induced oxidized LDL and aorta cell wall proliferation in rat. Iranian Biomedical Journal . 2015;19(2):117–123. doi: 10.6091/ibj.1449.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Talebi-Garakani E., Safarzade A. Resistance training decreases serum inflammatory markers in diabetic rats. Endocrine . 2013;43(3):564–570. doi: 10.1007/s12020-012-9786-9. [DOI] [PubMed] [Google Scholar]
- 23.Obradovic M., Sudar E., Zafirovic S., Stanimirovic J., Labudovic-Borovic M., Isenovic E. R. Estradiol in vivo induces changes in cardiomyocytes size in obese rats. Angiology . 2015;66(1):25–35. doi: 10.1177/0003319713514477. [DOI] [PubMed] [Google Scholar]
- 24.van Woudenbergh G. J., Kuijsten A., Sijbrands E. J. G., Hofman A., Witteman J. C. M., Feskens E. J. M. Glycemic index and glycemic load and their association with C-reactive protein and incident type 2 diabetes. Journal of Nutrition and Metabolism . 2011;2011:623077. doi: 10.1155/2011/623076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Thorand B., Löwel H., Schneider A., et al. C-reactive protein as a predictor for incident diabetes mellitus among middle-aged men: results from the MONICA Augsburg cohort study, 1984-1998. Archives of Internal Medicine . 2003;163(1):93–99. doi: 10.1001/archinte.163.1.93. [DOI] [PubMed] [Google Scholar]
- 26.Tillett W. S., Francis T. Serological reactions in pneumonia with a non-protein somatic fraction of pneumococcus. The Journal of Experimental Medicine . 1930;52(4):561–571. doi: 10.1084/jem.52.4.561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Abernethy T. J., Avery O. T. The occurrence during acute infections of a protein not normally present in the blood : i. distribution of the reactive protein in patients' sera and the effect of calcium on the flocculation reaction with c polysaccharide of pneumococcus. The Journal of Experimental Medicine . 1941;73(2):173–182. doi: 10.1084/jem.73.2.173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Abernethy T. J., Francis T. Studies on the somatic c polysaccharide of pneumococcus : i. cutaneous and serological reactions in pneumonia. The Journal of Experimental Medicine . 1937;65(1):59–73. doi: 10.1084/jem.65.1.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pathak A., Agrawal A. Evolution of C-reactive protein. Frontiers in Immunology . 2019;10(943) doi: 10.3389/fimmu.2019.00943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Trpkovic A., Obradovic M., Petrovic N., Davidovic R., Sudar-Milovanovic E., Isenovic E. R. C-reactive protein. In: Choi S., editor. Encyclopedia of Signaling Molecules, 2nd Edition, (edited by Sangdun Choi) Springer, New York: Springer International Publishing; 2016. pp. 1–5. [Google Scholar]
- 31.Shrive A. K., Gheetham G. M. T., Holden D., et al. Three dimensional structure of human C-reactive protein. Nature Structural Biology . 1996;3(4):346–354. doi: 10.1038/nsb0496-346. [DOI] [PubMed] [Google Scholar]
- 32.Eisenhardt S. U., Thiele J. R., Bannasch H., Stark G. B., Peter K. C-reactive protein: how conformational changes influence inflammatory properties. Cell Cycle . 2009;8(23):3885–3892. doi: 10.4161/cc.8.23.10068. [DOI] [PubMed] [Google Scholar]
- 33.Morioka K., Sato H., Kuboyama M., Yanagida A., Shoji A. Quantification of CRP in human serum using a handheld fluorescence detection system for capillary-based ELISA. Talanta . 2021;224, article 121725 doi: 10.1016/j.talanta.2020.121725. [DOI] [PubMed] [Google Scholar]
- 34.Wu Y., Potempa L. A., el Kebir D., Filep J. G. C-reactive protein and inflammation: conformational changes affect function. Biological Chemistry . 2015;396(11):1181–1197. doi: 10.1515/hsz-2015-0149. [DOI] [PubMed] [Google Scholar]
- 35.Ji S. R., Wu Y., Zhu L., et al. Cell membranes and liposomes dissociate C-reactive protein (CRP) to form a new, biologically active structural intermediate: mCRP(m) The FASEB Journal . 2007;21(1):284–294. doi: 10.1096/fj.06-6722com. [DOI] [PubMed] [Google Scholar]
- 36.Khreiss T., József L., Potempa L. A., Filep J.´. G. Opposing effects of C-reactive protein isoforms on shear-induced neutrophil-platelet adhesion and neutrophil aggregation in whole blood. Circulation . 2004;110(17):2713–2720. doi: 10.1161/01.CIR.0000146846.00816.DD. [DOI] [PubMed] [Google Scholar]
- 37.Thiele J. R., Habersberger J., Braig D., et al. Dissociation of pentameric to monomeric C-reactive protein localizes and aggravates inflammation: in vivo proof of a powerful proinflammatory mechanism and a new anti-inflammatory strategy. Circulation . 2014;130(1):35–50. doi: 10.1161/CIRCULATIONAHA.113.007124. [DOI] [PubMed] [Google Scholar]
- 38.Turu M. M., Slevin M., Matou S., et al. C-reactive protein exerts angiogenic effects on vascular endothelial cells and modulates associated signalling pathways and gene expression. BMC Cell Biology . 2008;9(1):p. 47. doi: 10.1186/1471-2121-9-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Slevin M., Matou S., Zeinolabediny Y., et al. Monomeric C-reactive protein-a key molecule driving development of Alzheimer's disease associated with brain ischaemia? Scientific Reports . 2015;5(1):p. 13281. doi: 10.1038/srep13281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Shrivastava A. K., Singh H. V., Raizada A., Singh S. K. C-reactive protein, inflammation and coronary heart disease. The Egyptian Heart Journal . 2015;67(2):89–97. doi: 10.1016/j.ehj.2014.11.005. [DOI] [Google Scholar]
- 41.Shim K., Begum R., Yang C., Wang H. Complement activation in obesity, insulin resistance, and type 2 diabetes mellitus. World Journal of Diabetes . 2020;11(1):1–12. doi: 10.4239/wjd.v11.i1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Volanakis J. E. Complement activation by C-reactive protein complexes. Annals of the New York Academy of Sciences . 1982;389(1 C-Reactive Pr):235–250. doi: 10.1111/j.1749-6632.1982.tb22140.x. [DOI] [PubMed] [Google Scholar]
- 43.Mold C., Gewurz H., Du Clos T. W. Regulation of complement activation by C-reactive protein. Immunopharmacology . 1999;42(1-3):23–30. doi: 10.1016/S0162-3109(99)00007-7. [DOI] [PubMed] [Google Scholar]
- 44.Du Clos T. W. Function of C-reactive protein. Annals of Medicine . 2000;32(4):274–278. doi: 10.3109/07853890009011772. [DOI] [PubMed] [Google Scholar]
- 45.Nehring S. M., Goyal A., Patel B. C. C Reactive Protein . Treasure Island (FL): StatPearls Publishing; 2020. [PubMed] [Google Scholar]
- 46.Boras E., Slevin M., Alexander M. Y., et al. Monomeric C-reactive protein and Notch-3 co-operatively increase angiogenesis through PI3K signalling pathway. Cytokine . 2014;69(2):165–179. doi: 10.1016/j.cyto.2014.05.027. [DOI] [PubMed] [Google Scholar]
- 47.Srikantiah C. C - reactive protein: an inflammatory marker with specific role in physiology, pathology, and diagnosis. Internet Journal of Rheumatology and Clinical Immunology . 2014;2(S1) doi: 10.15305/ijrci/v2iS1/117. [DOI] [Google Scholar]
- 48.Thompson D., Pepys M. B., Wood S. P. The physiological structure of human C-reactive protein and its complex with phosphocholine. Structure . 1999;7(2):169–177. doi: 10.1016/S0969-2126(99)80023-9. [DOI] [PubMed] [Google Scholar]
- 49.Kaplan M. H., Volanakis J. E. Interaction of C-reactive protein complexes with the complement system. I. Consumption of human complement associated with the reaction of C-reactive protein with pneumococcal C-polysaccharide and with the choline phosphatides, lecithin and sphingomyelin. Journal of Immunology . 1974;112(6):2135–2147. [PubMed] [Google Scholar]
- 50.Du Clos T. W. Pentraxins: structure, function, and role in inflammation. ISRN Inflammation . 2013;2013 doi: 10.1155/2013/379040.379040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Dehghan A., Dupuis J., Barbalic M., et al. Meta-analysis of genome-wide association studies in >80 000 subjects identifies multiple loci for C-reactive protein levels. Circulation . 2011;123(7):731–738. doi: 10.1161/CIRCULATIONAHA.110.948570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Rhodes B., Fürnrohr B. G., Vyse T. J. C-reactive protein in rheumatology: biology and genetics. Nature Reviews Rheumatology . 2011;7(5):282–289. doi: 10.1038/nrrheum.2011.37. [DOI] [PubMed] [Google Scholar]
- 53.Goldberger G., Bing D. H., Sipe J. D., Rits M., Colten H. R. Transcriptional regulation of genes encoding the acute-phase proteins CRP, SAA, and C3. Journal of Immunology . 1987;138(11):3967–3971. [PubMed] [Google Scholar]
- 54.Pepys M. B., Baltz M. L. Acute phase proteins with special reference to C-reactive protein and related proteins (pentaxins) and serum amyloid A protein. Advances in Immunology . 1983;34:141–212. doi: 10.1016/S0065-2776(08)60379-X. [DOI] [PubMed] [Google Scholar]
- 55.Kilpatrick J. M., Volanakis J. E. Molecular genetics, structure, and function of C-reactive protein. Immunologic Research . 1991;10(1):43–53. doi: 10.1007/BF02918166. [DOI] [PubMed] [Google Scholar]
- 56.Shine B., de Beer F. C., Pepys M. B. Solid phase radioimmunoassays for human C-reactive protein. Clinica Chimica Acta . 1981;117(1):13–23. doi: 10.1016/0009-8981(81)90005-X. [DOI] [PubMed] [Google Scholar]
- 57.Nehring S. M., Goyal A., Patel B. C. StatPearls . Treasure Island (FL): StatPearls Publishing LLC; 2021. C Reactive Protein. [PubMed] [Google Scholar]
- 58.Dupuy A. M., Badiou S., Descomps B., Cristol J. P. Immunoturbidimetric determination of C-reactive protein (CRP) and high-sensitivity CRP on heparin plasma. Comparison with serum determination. Clinical Chemistry and Laboratory Medicine . 2003;41(7):948–949. doi: 10.1515/CCLM.2003.144. [DOI] [PubMed] [Google Scholar]
- 59.Şişman A. R., Küme T., Taş G., Akan P. N., Tuncel P. N. Comparison and evaluation of two C-reactive protein assays based on particle-enhanced immunoturbidimetry. Journal of Clinical Laboratory Analysis . 2007;21(2):71–76. doi: 10.1002/jcla.20141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wu T. L., Tsao K. C., Chang C. P. Y., Li C. N., Sun C. F., Wu J. T. Development of ELISA on microplate for serum C-reactive protein and establishment of age-dependent normal reference range. Clinica Chimica Acta . 2002;322(1-2):163–168. doi: 10.1016/S0009-8981(02)00172-9. [DOI] [PubMed] [Google Scholar]
- 61.Lv Y., Wu R., Feng K., et al. Highly sensitive and accurate detection of C-reactive protein by CdSe/ZnS quantum dot-based fluorescence-linked immunosorbent assay. Journal of Nanobiotechnology . 2017;15(1):p. 35. doi: 10.1186/s12951-017-0267-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Siddiqui M. F., Khan Z. A., Park S. Detection of C-reactive protein using Histag-HRP functionalized nanoconjugate with signal amplified immunoassay. Nanomaterials . 2020;10(6):p. 1240. doi: 10.3390/nano10061240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Sardesai A. U., Dhamu V. N., Paul A., Muthukumar S., Prasad S. Design and electrochemical characterization of spiral electrochemical notification coupled electrode (SENCE) Platform for Biosensing Application. Platform for Biosensing Application . 2020;11(3):p. 333. doi: 10.3390/mi11030333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ma Y., Yang J., Yang T., et al. Electrochemical detection of C-reactive protein using functionalized iridium nanoparticles/graphene oxide as a tag. RSC Advances . 2020;10(16):9723–9729. doi: 10.1039/C9RA10386D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Guo L., Yang Z., Zhi S., Feng Z., Lei C., Zhou Y. A sensitive and innovative detection method for rapid C-reactive proteins analysis based on a micro-fluxgate sensor system. PLoS One . 2018;13(3, article e0194631) doi: 10.1371/journal.pone.0194631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Vanderschueren S., Deeren D., Knockaert D. C., Bobbaers H., Bossuyt X., Peetermans W. Extremely elevated C-reactive protein. European Journal of Internal Medicine . 2006;17(6):430–433. doi: 10.1016/j.ejim.2006.02.025. [DOI] [PubMed] [Google Scholar]
- 67.Tian T., Wei B., Wang J. Study of C-reactive protein, procalcitonin, and immunocyte ratios in 194 patients with sepsis. BMC Emergency Medicine . 2021;21(1):p. 81. doi: 10.1186/s12873-021-00477-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sayit A. T., Elmali M., Deveci A., Gedikli O. Relationship between acute phase reactants and prognosis in patients with or without COVID-19 pneumonia. Revista do Instituto de Medicina Tropical de São Paulo . 2021;63, article e51 doi: 10.1590/s1678-9946202163051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ali A., Noman M., Guo Y., et al. Myoglobin and C-reactive protein are efficient and reliable early predictors of COVID-19 associated mortality. Scientific Reports . 2021;11(1):p. 5975. doi: 10.1038/s41598-021-85426-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zhou H.-T., Zhao D. L., Wang G. K., Wang T. Z., Liang H. W., Zhang J. L. Assessment of high sensitivity C-reactive protein and coronary plaque characteristics by computed tomography in patients with and without diabetes mellitus. BMC Cardiovascular Disorders . 2020;20(1):p. 435. doi: 10.1186/s12872-020-01704-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Badimon L., Peña E., Arderiu G., et al. C-reactive protein in Atherothrombosis and angiogenesis. Frontiers in Immunology . 2018;9(430) doi: 10.3389/fimmu.2018.00430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.de Souza Pires-Neto O., da Silva Graça Amoras E., Queiroz M. A. F., et al. Hepatic TLR4, MBL and CRP gene expression levels are associated with chronic hepatitis C. Infection, Genetics and Evolution . 2020;80, article 104200 doi: 10.1016/j.meegid.2020.104200. [DOI] [PubMed] [Google Scholar]
- 73.Socha M. W., Malinowski B., Puk O., et al. C-reactive protein as a diagnostic and prognostic factor of endometrial cancer. Critical Reviews in Oncology/Hematology . 2021;164, article 103419 doi: 10.1016/j.critrevonc.2021.103419. [DOI] [PubMed] [Google Scholar]
- 74.Deng Y., Zhao Y., Qin J., et al. Prognostic value of the C-reactive protein/albumin ratio and systemic immune-inflammation index for patients with colorectal liver metastasis undergoing curative resection. Pathology Oncology Research . 2021;27, article 633480 doi: 10.3389/pore.2021.633480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Engstrom G., Hedblad B., Eriksson K. F., Janzon L., Lindgarde F. ComplementC3 is a risk factor for the development of Diabetes. Diabetes . 2005;54(2):570–575. doi: 10.2337/diabetes.54.2.570. [DOI] [PubMed] [Google Scholar]
- 76.Engström G., Hedblad B., Janzon L., Lindgärde F. Weight gain in relation to plasma levels of complement factor 3: results from a population-based cohort study. Diabetologia . 2005;48(12):2525–2531. doi: 10.1007/s00125-005-0021-6. [DOI] [PubMed] [Google Scholar]
- 77.Wlazlo N., van Greevenbroek M. M. J., Ferreira I., et al. Complement factor 3 is associated with insulin resistance and with incident type 2 diabetes over a 7-year follow-up period: the CODAM study. Diabetes Care . 2014;37(7):1900–1909. doi: 10.2337/dc13-2804. [DOI] [PubMed] [Google Scholar]
- 78.Oguntibeju O. O. Type 2 diabetes mellitus, oxidative stress and inflammation: examining the links. International Journal of Physiology, Pathophysiology and Pharmacology . 2019;11(3):45–63. [PMC free article] [PubMed] [Google Scholar]
- 79.American Diabetes, A. Diagnosis and classification of diabetes mellitus. Diabetes Care . 2010;33 Suppl 1(Suppl 1):S62–S69. doi: 10.2337/dc10-S062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ramalingam L., Menikdiwela K., LeMieux M., et al. The renin angiotensin system, oxidative stress and mitochondrial function in obesity and insulin resistance. Biochimica et Biophysica Acta - Molecular Basis of Disease . 2017;1863(5):1106–1114. doi: 10.1016/j.bbadis.2016.07.019. [DOI] [PubMed] [Google Scholar]
- 81.Atkinson M. A., Eisenbarth G. S., Michels A. W. Type 1 diabetes. The Lancet . 2014;383(9911):69–82. doi: 10.1016/S0140-6736(13)60591-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Festa A., D’Agostino R., Jr., Howard G., Mykkänen L., Tracy R. P., Haffner S. M. Chronic subclinical inflammation as part of the insulin resistance syndrome: the Insulin Resistance Atherosclerosis Study (IRAS) Circulation . 2000;102(1):42–47. doi: 10.1161/01.CIR.102.1.42. [DOI] [PubMed] [Google Scholar]
- 83.Fujimoto W. Y. The importance of insulin resistance in the pathogenesis of type 2 diabetes mellitus. The American Journal of Medicine . 2000;108(6):9–14. doi: 10.1016/S0002-9343(00)00337-5. [DOI] [PubMed] [Google Scholar]
- 84.Obradovic M., Sudar-Milovanovic E., Gluvic Z., Gojobori T., Essack M., Isenovic E. R. Diabetes and treatments. In: Faintuch J., Faintuch S., editors. Obesity and Diabetes: Scientific Advances and Best Practice . Cham: Springer International Publishing; 2020. pp. 705–717. [Google Scholar]
- 85.Rao G. Insulin resistance syndrome. American Family Physician . 2001;63(6):1159–1163. [PubMed] [Google Scholar]
- 86.Trpkovic A., Stanimirovic J., Resanovic I., et al. High sensitivity C-reactive protein and cardiovascular risk prediction. Current Pharmaceutical Analysis . 2014;11(1):60–65. doi: 10.2174/1573412910666140822003911. [DOI] [Google Scholar]
- 87.Obradovic M. M., Trpkovic A., Bajic V., et al. Interrelatedness between C-reactive protein and oxidized low-density lipoprotein. Clinical Chemistry and Laboratory Medicine . 2015;53(1):29–34. doi: 10.1515/cclm-2014-0590. [DOI] [PubMed] [Google Scholar]
- 88.Jain S., Gautam V., Naseem S. Acute-phase proteins: as diagnostic tool. Journal of Pharmacy & Bioallied Sciences . 2011;3(1):118–127. doi: 10.4103/0975-7406.76489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Crook M. Type 2 diabetes mellitus: a disease of the innate immune system? An update. Diabetic Medicine . 2004;21(3):203–207. doi: 10.1046/j.1464-5491.2003.01030.x. [DOI] [PubMed] [Google Scholar]
- 90.Calabro P., Chang D. W., Willerson J. T., Yeh E. T. H. Release of C-reactive protein in response to inflammatory cytokines by human adipocytes: linking obesity to vascular inflammation. Journal of the American College of Cardiology . 2005;46(6):1112–1113. doi: 10.1016/j.jacc.2005.06.017. [DOI] [PubMed] [Google Scholar]
- 91.Ouchi N., Kihara S., Funahashi T., et al. Reciprocal association of C-reactive protein with adiponectin in blood stream and adipose tissue. Circulation . 2003;107(5):671–674. doi: 10.1161/01.CIR.0000055188.83694.B3. [DOI] [PubMed] [Google Scholar]
- 92.Uemura H., Katsuura-Kamano S., Yamaguchi M., et al. Relationships of serum high-sensitivity C-reactive protein and body size with insulin resistance in a Japanese cohort. PLoS One . 2017;12(6, article e0178672) doi: 10.1371/journal.pone.0178672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Anty R., Bekri S., Luciani N., et al. The inflammatory C-reactive protein is increased in both liver and adipose tissue in severely obese patients independently from metabolic syndrome, type 2 diabetes, and NASH. The American Journal of Gastroenterology . 2006;101(8):1824–1833. doi: 10.1111/j.1572-0241.2006.00724.x. [DOI] [PubMed] [Google Scholar]
- 94.Kaneko H., Anzai T., Nagai T., et al. Human C-reactive protein exacerbates metabolic disorders in association with adipose tissue remodelling. Cardiovascular Research . 2011;91(3):546–555. doi: 10.1093/cvr/cvr088. [DOI] [PubMed] [Google Scholar]
- 95.Yang M., Qiu S., He Y., et al. Genetic ablation of C-reactive protein gene confers resistance to obesity and insulin resistance in rats. Diabetologia . 2021;64(5):1169–1183. doi: 10.1007/s00125-021-05384-9. [DOI] [PubMed] [Google Scholar]
- 96.Lontchi-Yimagou E., Sobngwi E., Matsha T. E., Kengne A. P. Diabetes mellitus and inflammation. Current Diabetes Reports . 2013;13(3):435–444. doi: 10.1007/s11892-013-0375-y. [DOI] [PubMed] [Google Scholar]
- 97.Trayhurn P. Adipose tissue in obesity—an inflammatory issue. Endocrinology . 2005;146(3):1003–1005. doi: 10.1210/en.2004-1597. [DOI] [PubMed] [Google Scholar]
- 98.Wellen K. E., Hotamisligil G. S. Obesity-induced inflammatory changes in adipose tissue. The Journal of Clinical Investigation . 2003;112(12):1785–1788. doi: 10.1172/JCI20514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Iikuni N., Kwan Lam Q., Lu L., Matarese G., Cava A. Leptin and inflammation. Current Immunology Reviews . 2008;4(2):70–79. doi: 10.2174/157339508784325046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Ouchi N., Walsh K. Adiponectin as an anti-inflammatory factor. Clinica Chimica Acta . 2007;380(1-2):24–30. doi: 10.1016/j.cca.2007.01.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Wellen K. E., Hotamisligil G. S. Inflammation, stress, and diabetes. The Journal of Clinical Investigation . 2005;115(5):1111–1119. doi: 10.1172/JCI25102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Borst S. E. The role of TNF-α in insulin resistance. Endocrine . 2004;23(2-3):177–182. doi: 10.1385/ENDO:23:2-3:177. [DOI] [PubMed] [Google Scholar]
- 103.Burhans M. S., Hagman D. K., Kuzma J. N., Schmidt K. A., Kratz M. Contribution of adipose tissue inflammation to the development of type 2 diabetes mellitus. Comprehensive Physiology . 2018;9(1):1–58. doi: 10.1002/cphy.c170040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Plomgaard P., Bouzakri K., Krogh-Madsen R., Mittendorfer B., Zierath J. R., Pedersen B. K. Tumor necrosis factor-alpha induces skeletal muscle insulin resistance in healthy human subjects via inhibition of Akt substrate 160 phosphorylation. Diabetes . 2005;54(10):2939–2945. doi: 10.2337/diabetes.54.10.2939. [DOI] [PubMed] [Google Scholar]
- 105.Krogh-Madsen R., Plomgaard P., Møller K., Mittendorfer B., Pedersen B. K. Influence of TNF-α and IL-6 infusions on insulin sensitivity and expression of IL-18 in humans. American Journal of Physiology. Endocrinology and Metabolism . 2006;291(1):E108–E114. doi: 10.1152/ajpendo.00471.2005. [DOI] [PubMed] [Google Scholar]
- 106.Rask-Madsen C., Domínguez H., Ihlemann N., Hermann T., Køber L., Torp-Pedersen C. Tumor Necrosis Factor-α Inhibits Insulin’s Stimulating Effect on Glucose Uptake and Endothelium-Dependent Vasodilation in Humans. Circulation . 2003;108(15):1815–1821. doi: 10.1161/01.CIR.0000091406.72832.11. [DOI] [PubMed] [Google Scholar]
- 107.Mohallem R., Aryal U. K. Regulators of TNFα mediated insulin resistance elucidated by quantitative proteomics. Scientific Reports . 2020;10(1):p. 20878. doi: 10.1038/s41598-020-77914-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Kurauti M. A., Costa-Júnior J. M., Ferreira S. M., et al. Interleukin-6 increases the expression and activity of insulin-degrading enzyme. Scientific Reports . 2017;7(1):p. 46750. doi: 10.1038/srep46750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Bowker N., Shah R. L., Sharp S. J., et al. Meta-analysis investigating the role of interleukin-6 mediated inflammation in type 2 diabetes. eBioMedicine . 2020;61, article 103062 doi: 10.1016/j.ebiom.2020.103062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Schmidt-Arras D., Rose-John S. IL-6 pathway in the liver: from physiopathology to therapy. Journal of Hepatology . 2016;64(6):1403–1415. doi: 10.1016/j.jhep.2016.02.004. [DOI] [PubMed] [Google Scholar]
- 111.Klover P. J., Mooney R. A. Hepatocytes: critical for glucose homeostasis. The International Journal of Biochemistry & Cell Biology . 2004;36(5):753–758. doi: 10.1016/j.biocel.2003.10.002. [DOI] [PubMed] [Google Scholar]
- 112.Mauer J., Chaurasia B., Goldau J., et al. Signaling by IL-6 promotes alternative activation of macrophages to limit endotoxemia and obesity-associated resistance to insulin. Nature Immunology . 2014;15(5):423–430. doi: 10.1038/ni.2865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Akbari M., Hassan-Zadeh V. IL-6 signalling pathways and the development of type 2 diabetes. Inflammopharmacology . 2018;26(3):685–698. doi: 10.1007/s10787-018-0458-0. [DOI] [PubMed] [Google Scholar]
- 114.Goldman J. L., Sammani S., Kempf C., et al. Pleiotropic effects of interleukin-6 in a “two-hit” murine model of acute respiratory distress syndrome. Pulmonary Circulation . 2014;4(2):280–288. doi: 10.1086/675991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Kishimoto T. Interleukin-6: discovery of a pleiotropic cytokine. Arthritis Research & Therapy . 2006;8(2):p. S2. doi: 10.1186/ar1916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Knüpfer H., Preiß R. Significance of interleukin-6 (IL-6) in breast cancer (review) Breast Cancer Research and Treatment . 2007;102(2):129–135. doi: 10.1007/s10549-006-9328-3. [DOI] [PubMed] [Google Scholar]
- 117.Rohleder N., Aringer M., Boentert M. Role of interleukin-6 in stress, sleep, and fatigue. Annals of the New York Academy of Sciences . 2012;1261(1):88–96. doi: 10.1111/j.1749-6632.2012.06634.x. [DOI] [PubMed] [Google Scholar]
- 118.Amoroso C., Perillo F., Strati F., Fantini M., Caprioli F., Facciotti F. The role of gut microbiota biomodulators on mucosal immunity and intestinal inflammation. Cell . 2020;9(5):p. 1234. doi: 10.3390/cells9051234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Scheithauer T. P. M., Rampanelli E., Nieuwdorp M., et al. Gut microbiota as a trigger for metabolic inflammation in obesity and type 2 diabetes. Frontiers in Immunology . 2020;11(2546) doi: 10.3389/fimmu.2020.571731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Molinaro A., Consortium T. M. C., Lassen P. B., et al. Imidazole propionate is increased in diabetes and associated with dietary patterns and altered microbial ecology. Nature Communications . 2020;11(1):5881–5881. doi: 10.1038/s41467-020-19589-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.van den Munckhof I. C. L., Kurilshikov A., ter Horst R., et al. Role of gut microbiota in chronic low-grade inflammation as potential driver for atherosclerotic cardiovascular disease: a systematic review of human studies. Obesity Reviews . 2018;19(12):1719–1734. doi: 10.1111/obr.12750. [DOI] [PubMed] [Google Scholar]
- 122.Al Bander Z., Nitert M. D. The gut microbiota and inflammation: an overview. International Journal of Environmental Research and Public Health . 2020;17(20) doi: 10.3390/ijerph17207618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Allin K. H., Tremaroli V., Caesar R., et al. Aberrant intestinal microbiota in individuals with prediabetes. Diabetologia . 2018;61(4):810–820. doi: 10.1007/s00125-018-4550-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Wu H., Tremaroli V., Schmidt C., et al. The gut microbiota in prediabetes and diabetes: a population-based cross-sectional study. Cell Metabolism . 2020;32(3):379–390.e3. doi: 10.1016/j.cmet.2020.06.011. [DOI] [PubMed] [Google Scholar]
- 125.Koh A., Molinaro A., Ståhlman M., et al. Microbially produced imidazole propionate impairs insulin signaling through mTORC1. Cell . 2018;175(4):947–961.e17. doi: 10.1016/j.cell.2018.09.055. [DOI] [PubMed] [Google Scholar]
- 126.Koh A., Mannerås-Holm L., Yunn N. O., et al. Microbial imidazole propionate affects responses to metformin through p38γ-dependent inhibitory AMPK phosphorylation. Cell Metabolism . 2020;32(4):643–653.e4. doi: 10.1016/j.cmet.2020.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Creely S. J., McTernan P. G., Kusminski C. M., et al. Lipopolysaccharide activates an innate immune system response in human adipose tissue in obesity and type 2 diabetes. American Journal of Physiology. Endocrinology and Metabolism . 2007;292(3):E740–E747. doi: 10.1152/ajpendo.00302.2006. [DOI] [PubMed] [Google Scholar]
- 128.Ghanim H., Abuaysheh S., Sia C. L., et al. Increase in plasma endotoxin concentrations and the expression of toll-like receptors and suppressor of cytokine signaling-3 in mononuclear cells after a high-fat, high-carbohydrate meal: implications for insulin resistance. Diabetes Care . 2009;32(12):2281–2287. doi: 10.2337/dc09-0979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Chung S., LaPoint K., Martinez K., Kennedy A., Boysen Sandberg M., McIntosh M. K. Preadipocytes mediate lipopolysaccharide-induced inflammation and insulin resistance in primary cultures of newly differentiated human adipocytes. Endocrinology . 2006;147(11):5340–5351. doi: 10.1210/en.2006-0536. [DOI] [PubMed] [Google Scholar]
- 130.Claesson M. J., Jeffery I. B., Conde S., et al. Gut microbiota composition correlates with diet and health in the elderly. Nature . 2012;488(7410):178–184. doi: 10.1038/nature11319. [DOI] [PubMed] [Google Scholar]
- 131.Gasecka A., Siwik D., Gajewska M., et al. Early biomarkers of neurodegenerative and neurovascular disorders in diabetes. Journal of Clinical Medicine . 2020;9(9):p. 9. doi: 10.3390/jcm9092807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Papanas N., Katsiki N., Papatheodorou K., et al. Peripheral neuropathy is associated with increased serum levels of uric acid in type 2 diabetes mellitus. Angiology . 2011;62(4):291–295. doi: 10.1177/0003319710394164. [DOI] [PubMed] [Google Scholar]
- 133.Zhang H. H., Han X., Wang M., et al. The association between genomic DNA methylation and diabetic peripheral neuropathy in patients with type 2 diabetes mellitus. Journal of Diabetes Research . 2019;2019 doi: 10.1155/2019/2494057.2494057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Zhang G., Wei W., Tan B., Liu J. Correlations between hormone levels and cardiovascular autonomic neuropathy in menopausal patients with diabetes mellitus. Pakistan Journal of Medical Sciences . 2020;36(6):1382–1386. doi: 10.12669/pjms.36.6.2088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Doupis J., Lyons T. E., Wu S., Gnardellis C., Dinh T., Veves A. Microvascular reactivity and inflammatory cytokines in painful and painless peripheral diabetic neuropathy. The Journal of Clinical Endocrinology and Metabolism . 2009;94(6):2157–2163. doi: 10.1210/jc.2008-2385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Aryan Z., Ghajar A., Faghihi-Kashani S., Afarideh M., Nakhjavani M., Esteghamati A. Baseline high-sensitivity C-reactive protein predicts macrovascular and microvascular complications of type 2 diabetes: a population-based study. Annals of Nutrition & Metabolism . 2018;72(4):287–295. doi: 10.1159/000488537. [DOI] [PubMed] [Google Scholar]
- 137.Qiu F., Ma X., Shin Y. H., et al. Pathogenic role of human C-reactive protein in diabetic retinopathy. Clinical Science . 2020;134(13):1613–1629. doi: 10.1042/CS20200085. [DOI] [PubMed] [Google Scholar]
- 138.Song J., Chen S., Liu X., Duan H., Kong J., Li Z. Relationship between C-reactive protein level and diabetic retinopathy: a systematic review and meta-analysis. PLoS One . 2015;10(12, article e0144406) doi: 10.1371/journal.pone.0144406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Naveen Nishal G., Kaliaperumal R. D., Nallamuthu P., Subhashini M. Correlation of C-reactive protein and BMI with severity of diabetic retinopathy. Asian Journal of Medical Sciences . 2022;13(4):51–55. doi: 10.3126/ajms.v13i4.41460. [DOI] [Google Scholar]
- 140.Lim L. S., Tai E. S., Mitchell P., et al. C-reactive protein, body mass index, and diabetic retinopathy. Investigative Ophthalmology & Visual Science . 2010;51(9):4458–4463. doi: 10.1167/iovs.09-4939. [DOI] [PubMed] [Google Scholar]
- 141.Tang P. M.-K., Zhang Y. Y., Hung J. S. C., et al. DPP4/CD32b/NF-κB circuit: a novel druggable target for inhibiting CRP-driven diabetic nephropathy. Molecular Therapy . 2021;29(1):365–375. doi: 10.1016/j.ymthe.2020.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Liu Q., Jiang C. Y., Chen B. X., Zhao W., Meng D. The association between high-sensitivity C-reactive protein concentration and diabetic nephropathy: a meta-analysis. European Review for Medical and Pharmacological Sciences . 2015;19(23):4558–4568. [PubMed] [Google Scholar]
- 143.Hayashino Y., Mashitani T., Tsujii S., Ishii H., Diabetes Distress and Care Registry at Tenri Study Group Serum high-sensitivity C-reactive protein levels are associated with high risk of development, not progression, of diabetic nephropathy among Japanese type 2 diabetic patients: a prospective cohort study (Diabetes Distress and Care Registry at Tenri [DDCRT7]) Diabetes Care . 2014;37(11):2947–2952. doi: 10.2337/dc14-1357. [DOI] [PubMed] [Google Scholar]
- 144.Fu C. C., Wu D. A., Wang J. H., Yang W. C., Tseng C. H. Association of C-reactive protein and hyperuricemia with diabetic nephropathy in Chinese type 2 diabetic patients. Acta Diabetologica . 2009;46(2):127–134. doi: 10.1007/s00592-008-0069-0. [DOI] [PubMed] [Google Scholar]
- 145.Varma V., Varma M., Varma A., Kumar R., Bharosay A., Vyas S. Serum Total sialic acid and highly sensitive C-reactive protein: prognostic markers for the diabetic nephropathy. Journal of Laboratory Physicians . 2016;8(1):025–029. doi: 10.4103/0974-2727.176230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Marnell L., Mold C., Du Clos T. W. C-reactive protein: ligands, receptors and role in inflammation. Clinical Immunology . 2005;117(2):104–111. doi: 10.1016/j.clim.2005.08.004. [DOI] [PubMed] [Google Scholar]
- 147.Ryu J., Lee C., Shin J., et al. FcγRIIa mediates C-reactive protein-induced inflammatory responses of human vascular smooth muscle cells by activating NADPH oxidase 4. Cardiovascular Research . 2007;75(3):555–565. doi: 10.1016/j.cardiores.2007.04.027. [DOI] [PubMed] [Google Scholar]
- 148.Zou X. L., Yang J., Yang J. M., et al. Brief communication (original). Immune injury in rat models of type 2 diabetes mellitus. Asian Biomedicine . 2017;6(6):903–908. [Google Scholar]
- 149.Cho W. C., Yip T. T., Chung W. S., Leung A. W., Cheng C. H., Yue K. K. Differential expression of proteins in kidney, eye, aorta, and serum of diabetic and non-diabetic rats. Journal of Cellular Biochemistry . 2006;99(1):256–268. doi: 10.1002/jcb.20923. [DOI] [PubMed] [Google Scholar]
- 150.Asgary S. Hypoglycaemic and hypolipidemic effects of pumpkin (Cucurbita pepo L.) on alloxan-induced diabetic rats. African Journal of Pharmacy and Pharmacology . 2011;5(23) doi: 10.5897/AJPP11.635. [DOI] [Google Scholar]
- 151.Ige A. O., Ajayi O. A., Adewoye E. O. Anti-inflammatory and insulin secretory activity in experimental type-2 diabetic rats treated orally with magnesium. Journal of Basic and Clinical Physiology and Pharmacology . 2018;29(5):507–514. doi: 10.1515/jbcpp-2017-0104. [DOI] [PubMed] [Google Scholar]
- 152.Szkudelski T. The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiological Research . 2001;50(6):537–546. [PubMed] [Google Scholar]
- 153.Obasi D. C., Ogugua V. N. GC-MS analysis, pH and antioxidant effect of Ruzu herbal bitters on alloxan-induced diabetic rats. Biochemistry and Biophysics Reports . 2021;27, article 101057 doi: 10.1016/j.bbrep.2021.101057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Yusuf B. O., Yakubu M. T., Akanji M. A. Chromatographic fractions from _Chrysophyllum albidum_ stem bark boost antioxidant enzyme activity and ameliorate some markers of diabetes complications. Journal of Traditional and Complementary Medicine . 2021;11(4):336–342. doi: 10.1016/j.jtcme.2021.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Gharib H. M., Abajy M. Y., Omaren A. Investigating the effect of some fluoroquinolones on C-reactive protein levels and ACh-induced blood pressure reduction deviations after aging of diabetes in STZ-induced diabetic Wistar rats. Heliyon . 2020;6(4, article e03812) doi: 10.1016/j.heliyon.2020.e03812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Almatroodi S. A., Alnuqaydan A. M., Alsahli M. A., Khan A. A., Rahmani A. H. Thymoquinone, the Most prominent constituent of nigella sativa, attenuates liver damage in streptozotocin-induced diabetic rats via regulation of oxidative stress, inflammation and cyclooxygenase-2 protein expression. Applied Sciences . 2021;11(7):p. 3223. doi: 10.3390/app11073223. [DOI] [Google Scholar]
- 157.Thorand B., Baumert J., Kolb H., et al. Sex differences in the prediction of type 2 diabetes by inflammatory Markers. Diabetes Care . 2007;30(4):854–860. doi: 10.2337/dc06-1693. [DOI] [PubMed] [Google Scholar]
- 158.Nakanishi S., Yamane K., Kamei N., Okubo M., Kohno N. Elevated C-reactive protein is a risk factor for the development of type 2 diabetes in Japanese Americans. Diabetes Care . 2003;26(10):2754–2757. doi: 10.2337/diacare.26.10.2754. [DOI] [PubMed] [Google Scholar]
- 159.Nishikawa T., Edelstein D., du X. L., et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature . 2000;404(6779):787–790. doi: 10.1038/35008121. [DOI] [PubMed] [Google Scholar]
- 160.Arnalich F., Hernanz A., López-Maderuelo D., et al. Enhanced acute-phase response and oxidative stress in older adults with type II diabetes. Hormone and Metabolic Research . 2000;32(10):407–412. doi: 10.1055/s-2007-978662. [DOI] [PubMed] [Google Scholar]
- 161.Bertelsen M., Anggård E. E., Carrier M. J. Oxidative stress impairs insulin internalization in endothelial cells in vitro. Diabetologia . 2001;44(5):605–613. doi: 10.1007/s001250051667. [DOI] [PubMed] [Google Scholar]
- 162.Gopaul N. K., Manraj M. D., Hébé A., et al. Oxidative stress could precede endothelial dysfunction and insulin resistance in Indian Mauritians with impaired glucose metabolism. Diabetologia . 2001;44(6):706–712. doi: 10.1007/s001250051679. [DOI] [PubMed] [Google Scholar]
- 163.Doi Y., Kiyohara Y., Kubo M., et al. Elevated C-reactive protein is a predictor of the development of diabetes in a general Japanese population: the Hisayama Study. Diabetes Care . 2005;28(10):2497–2500. doi: 10.2337/diacare.28.10.2497. [DOI] [PubMed] [Google Scholar]
- 164.Marques-Vidal P., Bastardot F., von Känel R., et al. Association between circulating cytokine levels, diabetes and insulin resistance in a population-based sample (CoLaus study) Clinical Endocrinology . 2013;78(2):232–241. doi: 10.1111/j.1365-2265.2012.04384.x. [DOI] [PubMed] [Google Scholar]
- 165.Akbarzadeh M., Eftekhari M. H., Dabbaghmanesh M. H., Hasanzadeh J., Bakhshayeshkaram M. Serum IL-18 and hsCRP correlate with insulin resistance without effect of calcitriol treatment on type 2 diabetes. Iranian Journal of Immunology . 2013;10(3):167–176. [PubMed] [Google Scholar]
- 166.Yanagawa T., Taniguchi A., Fukushima M., et al. Leptin, triglycerides, and interleukin 6 are independently associated with C-reactive protein in Japanese type 2 diabetic patients. Diabetes Research and Clinical Practice . 2007;75(1):2–6. doi: 10.1016/j.diabres.2006.04.019. [DOI] [PubMed] [Google Scholar]
- 167.van Dielen F. M., van't Veer C., Schols A. M., Soeters P. B., Buurman W. A., Greve J. W. M. Increased leptin concentrations correlate with increased concentrations of inflammatory markers in morbidly obese individuals. International Journal of Obesity and Related Metabolic Disorders . 2001;25(12):1759–1766. doi: 10.1038/sj.ijo.0801825. [DOI] [PubMed] [Google Scholar]
- 168.Gómez-Ambrosi J., Salvador J., Páramo J. A., et al. Involvement of leptin in the association between percentage of body fat and cardiovascular risk factors. Clinical Biochemistry . 2002;35(4):315–320. doi: 10.1016/S0009-9120(02)00320-X. [DOI] [PubMed] [Google Scholar]
- 169.Park J. S., Cho M. H., Nam J. S., et al. Visceral adiposity and leptin are independently associated with C-reactive protein in Korean type 2 diabetic patients. Acta Diabetologica . 2010;47(2):113–118. doi: 10.1007/s00592-009-0125-4. [DOI] [PubMed] [Google Scholar]
- 170.Sudhakar M., Silambanan S., Chandran A. S., Prabhakaran A. A., Ramakrishnan R. C-reactive protein (CRP) and leptin receptor in obesity: binding of monomeric CRP to leptin receptor. Frontiers in Immunology . 2018;9:1167–1167. doi: 10.3389/fimmu.2018.01167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Shamsuzzaman A. S., Winnicki M., Wolk R., et al. Independent association between plasma leptin and C-reactive protein in healthy humans. Circulation . 2004;109(18):2181–2185. doi: 10.1161/01.CIR.0000127960.28627.75. [DOI] [PubMed] [Google Scholar]
- 172.Obradovic M., Sudar-Milovanovic E., Soskic S., et al. Leptin and obesity: role and clinical implication. Frontiers in Endocrinology . 2021;12:585887–585887. doi: 10.3389/fendo.2021.585887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Lainampetch J., Panprathip P., Phosat C., et al. Association of tumor necrosis factor alpha, interleukin 6, and C-reactive protein with the risk of developing type 2 diabetes: a retrospective cohort study of rural Thais. Journal of Diabetes Research . 2019;2019 doi: 10.1155/2019/9051929.9051929 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Pickup J. C., Crook M. A. Is type II diabetes mellitus a disease of the innate immune system? Diabetologia . 1998;41(10):1241–1248. doi: 10.1007/s001250051058. [DOI] [PubMed] [Google Scholar]
- 175.Calle M. C., Fernandez M. L. Inflammation et diabete de type 2. Diabetes & Metabolism . 2012;38(3):183–191. doi: 10.1016/j.diabet.2011.11.006. [DOI] [PubMed] [Google Scholar]
- 176.Nisar M., Iqbal M. U. Influence of hs-CRP, IL-6 and TNF-α and it's role in dyslipidemia and type 2 diabetes in population of Karachi, Pakistan. Pakistan Journal of Pharmaceutical Sciences . 2021;34(3(Special)):1217–1225. [PubMed] [Google Scholar]
- 177.Tsalamandris S., Antonopoulos A. S., Oikonomou E., et al. The role of inflammation in Diabetes: current concepts and future perspectives. European Cardiology . 2019;14(1):50–59. doi: 10.15420/ecr.2018.33.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Mtintsilana A., Micklesfield L. K., Chorell E., et al. Adiposity mediates the association between the dietary inflammatory index and markers of type 2 diabetes risk in middle-aged Black South African women. Nutrients . 2019;11(6):p. 1246. doi: 10.3390/nu11061246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Camhi S. M., Stefanick M. L., Ridker P. M., Young D. R. Changes in C-reactive protein from low-fat diet and/or physical activity in men and women with and without metabolic syndrome. Metabolism . 2010;59(1):54–61. doi: 10.1016/j.metabol.2009.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Forst T., Karagiannis E., Lübben G., et al. Pleiotrophic and anti-inflammatory effects of pioglitazone precede the metabolic activity in type 2 diabetic patients with coronary artery disease. Atherosclerosis . 2008;197(1):311–317. doi: 10.1016/j.atherosclerosis.2007.05.006. [DOI] [PubMed] [Google Scholar]
- 181.Hanefeld M., Marx N., Pfützner A., et al. Anti-inflammatory effects of pioglitazone and/or simvastatin in high cardiovascular risk patients with elevated high sensitivity C-reactive protein: the PIOSTAT Study. Journal of the American College of Cardiology . 2007;49(3):290–297. doi: 10.1016/j.jacc.2006.08.054. [DOI] [PubMed] [Google Scholar]
- 182.Satoh N., Ogawa Y., Usui T., et al. Antiatherogenic effect of pioglitazone in type 2 diabetic patients irrespective of the responsiveness to its antidiabetic effect. Diabetes Care . 2003;26(9):2493–2499. doi: 10.2337/diacare.26.9.2493. [DOI] [PubMed] [Google Scholar]
- 183.Pfützner A., Marx N., Lübben G., et al. Improvement of cardiovascular risk markers by pioglitazone is independent from glycemic control: results from the pioneer study. Journal of the American College of Cardiology . 2005;45(12):1925–1931. doi: 10.1016/j.jacc.2005.03.041. [DOI] [PubMed] [Google Scholar]
- 184.Larsen C. M., Faulenbach M., Vaag A., et al. Interleukin-1–receptor antagonist in type 2 diabetes mellitus. New England Journal of Medicine . 2007;356(15):1517–1526. doi: 10.1056/NEJMoa065213. [DOI] [PubMed] [Google Scholar]
- 185.Larsen C. M., Faulenbach M., Vaag A., Ehses J. A., Donath M. Y., Mandrup-Poulsen T. Sustained effects of interleukin-1 receptor antagonist treatment in type 2 diabetes. Diabetes Care . 2009;32(9):1663–1668. doi: 10.2337/dc09-0533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Sloan-Lancaster J., Abu-Raddad E., Polzer J., et al. Double-blind, randomized study evaluating the glycemic and anti-inflammatory effects of subcutaneous LY2189102, a neutralizing IL-1β antibody, in patients with type 2 diabetes. Diabetes Care . 2013;36(8):2239–2246. doi: 10.2337/dc12-1835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Hensen J., Howard C. P., Walter V., Thuren T. Incidence de l'anticorps anti-interleukine-1β (canakinumab) sur les indicateurs de glycemie chez les patients atteints de diabete de type 2 : resultats des criteres d'evaluation secondaires d'un essai a repartition aleatoire et controle par placebo. Diabetes & Metabolism . 2013;39(6):524–531. doi: 10.1016/j.diabet.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 188.Ridker P. M., Howard C. P., Walter V., et al. Effects of interleukin-1β inhibition with canakinumab on hemoglobin A1c, lipids, C-reactive protein, interleukin-6, and fibrinogen: a phase IIb randomized, placebo-controlled trial. Circulation . 2012;126(23):2739–2748. doi: 10.1161/CIRCULATIONAHA.112.122556. [DOI] [PubMed] [Google Scholar]
- 189.Everett B. M., Donath M. Y., Pradhan A. D., et al. Anti-inflammatory therapy with canakinumab for the prevention and management of diabetes. Journal of the American College of Cardiology . 2018;71(21):2392–2401. doi: 10.1016/j.jacc.2018.03.002. [DOI] [PubMed] [Google Scholar]
- 190.Fleischman A., Shoelson S. E., Bernier R., Goldfine A. B. Salsalate improves glycemia and inflammatory parameters in obese young adults. Diabetes Care . 2008;31(2):289–294. doi: 10.2337/dc07-1338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Goldfine A. B., Silver R., Aldhahi W., et al. Use of salsalate to target inflammation in the treatment of insulin resistance and type 2 diabetes. Clinical and Translational Science . 2008;1(1):36–43. doi: 10.1111/j.1752-8062.2008.00026.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Dominguez H., Storgaard H., Rask-Madsen C., et al. Metabolic and vascular effects of tumor necrosis factor-alpha blockade with etanercept in obese patients with type 2 diabetes. Journal of Vascular Research . 2005;42(6):517–525. doi: 10.1159/000088261. [DOI] [PubMed] [Google Scholar]
- 193.Bernstein L. E., Berry J., Kim S., Canavan B., Grinspoon S. K. Effects of etanercept in patients with the metabolic syndrome. Archives of Internal Medicine . 2006;166(8):902–908. doi: 10.1001/archinte.166.8.902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Kato K., Otsuka T., Saiki Y., et al. Association between elevated C-reactive protein levels and prediabetes in adults, particularly impaired glucose tolerance. Canadian Journal of Diabetes . 2019;43(1):40–45.e2. doi: 10.1016/j.jcjd.2018.03.007. [DOI] [PubMed] [Google Scholar]
- 195.Kato K., Otsuka T., Saiki Y., et al. Elevated C-reactive protein levels independently predict the development of prediabetes markers in subjects with normal glucose regulation. Experimental and Clinical Endocrinology & Diabetes . 2021;129(4):289–295. doi: 10.1055/a-0869-7584. [DOI] [PubMed] [Google Scholar]
- 196.Zheng Z., Yin Y., Lu R. Lycopene ameliorated oxidative stress and inflammation in Type 2 diabetic rats. Journal of Food Science . 2019;84(5):1194–1200. doi: 10.1111/1750-3841.14505. [DOI] [PubMed] [Google Scholar]
- 197.Li J., Zhang H., Wang G. Correlations between inflammatory response, oxidative stress, intestinal pathological damage and intestinal flora variation in rats with type 2 diabetes mellitus. European Review for Medical and Pharmacological Sciences . 2020;24(19):10162–10168. doi: 10.26355/eurrev_202010_23236. [DOI] [PubMed] [Google Scholar]
- 198.Suman R. K., Ray Mohanty I., Borde M. K., Maheshwari U., Deshmukh Y. A. Development of an experimental model of diabetes co-existing with metabolic syndrome in rats. Advances in Pharmacological Sciences . 2016;2016 doi: 10.1155/2016/9463476.9463476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Chan K. C., Kok K. E., Huang K. F., Weng Y. L., Chung Y. C. Effects of fermented red bean extract on nephropathy in streptozocin-induced diabetic rats. Food & Nutrition Research . 2020;64 doi: 10.29219/fnr.v64.4272. [DOI] [PMC free article] [PubMed] [Google Scholar]


