Abstract
Background
Since the COVID-19 pandemic has started, Serbia has faced problems in implementing proper public health measures in the population, including non-pharmaceutical interventions, as well as protecting health care workers (HCWs) from disease, like all other countries. This study aimed to estimate COVID-19 seroprevalence and evaluate the risk perception of COVID-19 among HCWs in three different hospitals in Belgrade, Serbia: non-COVID hospital, Emergency Center (EC), and dedicated COVID hospital.
Methods
A cross-sectional study was conducted in three hospitals during the second wave of the outbreak in Serbia, from June to early October. All staff in these hospitals were invited to voluntarily participate in blood sampling for IgG antibodies against SARS-CoV-2 and questionnaire testing. The questionnaire included socio-demographic characteristics, known exposure to COVID-19 positive persons, previous signs and symptoms related to COVID-19 infection since the outbreak had started in our country, and SARS-CoV-2 PCR testing.
Results
The overall prevalence of SARS-CoV-2 antibody among 1580 HCWs was 18.3 % [95 % CI 16.4–20.3 %]. Significantly higher prevalence of HCWs with positive results for the serum IgG antibody test was observed in COVID hospital (28.6 %, 95 %CI: 24.0–33.6 %) vs. prevalence in the EC (12.6 %, 95 %CI: 10.1–15.4 %), and in the non-COVID hospital (18.3 %, 95 %CI: 15.2–26.7 %). The prevalence adjusted for declared test sensitivity and specificity would be 16.8 %; that is 27.4 % in COVID-19 hospital, 10.9 % in EC, and 16.8 % in non-COVID hospital. In multivariate logistic regression analysis, the independent predictors for seropositivity were working in COVID-hospital, the profession of physician, and the presence of the following symptoms: fever, shortness of breath, and anosmia/ageusia.
Conclusions
We found an overall seropositivity rate of 18.3 % and 16.0 % of the adjusted rate that is higher than seroprevalence obtained in similar studies conducted before vaccinations started. The possibility that patients in non-COVID dedicated hospitals might also be infectious, although PCR tested, imposes the need for the use of personal protective equipment also in non-COVID medical institutions.
Keywords: Healthcare workers, Serology, COVID-19, Hospital, Seroprevalence
Introduction
The first autochthonous case of COVID-19 was diagnosed on March 6, 2020, in Serbia [1]. During the first months following the pandemic, as all countries in the region [2], Serbia has faced problems in implementing proper public health measures in the population, including non-pharmaceutical interventions, as well as protecting health care workers (HCWs) from the disease. Soon after the outbreak was declared on March 19th, the capacities of hospital infectious wards became insufficient to treat numerous COVID-19 patients, and the entire hospitals had to be transformed into hospitals only for the treatment of COVID-19 patients. Even before the epidemic began, all HCWs were educated and trained on how to safely use personal protective equipment (PPE) according to the WHO and the ECDC recommendations. The complete equipment was reserved only for COVID dedicated hospitals. In non-COVID hospitals, in addition to the standard medical uniform, HCWs used a surgical mask. Later, in May 2020, when the number of patients increased enormously, it was recommended to wear a face shield according to the well-known fact that the virus is predominantly transmitted by droplets.
Infection prevention measures were implemented in all hospitals with special attention to patients triage at the hospital admission point in non-COVID hospitals [3]. Each patient was asked for potential COVID-19 contacts in the family and relatives, whether he traveled anywhere during 14 previous days and the temperature was measured. At that time, the number of PCR tests and laboratory capacities were still limited, while rapid antigen tests were not available at all. Therefore, triage at admission to a non-COVID hospital posed a major challenge for both clinicians and infection control physicians.
Patients with suspected infection underwent radiographic examination of the lungs or CT, and in case of characteristic signs of infection, they were referred to the COVID hospital. Patients with a COVID-19 diagnosis confirmed in the non-COVID triage hospital as well as patients with positive PCR test or clinically confirmed COVID-19 in the primary health care center were admitted to the COVID hospital, when necessary. Therefore, all HCWs in the COVID hospital were aware that they treated COVID-19 patients and sought to adhere strictly to infection prevention and control measures.
Unfortunately, some patients without any sign or symptom, during the incubation period, but who could be contagious, might be admitted to a non-COVID hospital [4], [5]. Those patients posed a threat of infection transmission to other patients and the hospital staff.
Despite implementing the strict triage of patients at admission, a special challenge was health care in emergency centers, because patients are contagious at least two days before the first symptoms of the disease. COVID-19 seroprevalence can estimate the proportion of people who had exposure to the SARS-CoV-2 virus, regardless of clinical manifestations and disease severity. Measurement of antibodies against SARS-CoV-2 could be used as an indicator of viral exposure and therefore might represent one of the approximations of proper wearing of PEE in the health care institutions. Therefore, the aims of this study were to (a) estimate COVID-19 seroprevalence (b) evaluate the risk perception of COVID-19 among HCWs in three different hospitals (COVID hospital, non-COVID hospital, and Emergency Center), from June to early October, during the second wave of COVID-19 outbreak in Serbia.
Material and methods
Study design and settings
Health care workers of the three University Hospitals in Belgrade, Serbia, were invited to participate on a voluntary basis in this cross-sectional multicentre study. The study was conducted in the Clinical Hospital Center “Bezanijska Kosa”, a non-COVID hospital at that time, the Emergency Centre (EC) of the University Clinical Center of Serbia, and the Clinical Hospital Center “Dr. Dragisa Misovic”, as a hospital which was transformed into the dedicated COVID hospital soon after COVID-19 outbreak in Serbia. All staff was invited to voluntarily participate in the study by the director of the hospital and the head of each service. Staff quarantined due to active symptoms or recent private contacts with ill persons without PPE were not included. All HCWs received an informative letter reporting the aim of the survey.
Staff working in the COVID-19 hospital were wearing complete personal protective equipment (PPE) in accordance with the national standards [6] prepared in line with the WHO and the ECDC recommendations. Staff in the EC and non-COVID hospital responsible for the care and treat patients without known or suspected COVID-19 patients, applied standard precautions, and were required to wear surgical masks and a visor since mid-May.
Questionnaire
All participants filled in an anonymously questionnaire with the help of infection control staff if it was necessary to clarify a question. The first part of the questionnaire contained socio-demographic characteristics (age, sex, professional category, and length of employment), known exposure to COVID-19 positive persons, previous signs and symptoms related to COVID-19 infection since the outbreak had started in our country, and SARS-CoV-2 PCR testing. The second part of the questionnaire contained questions about risk perception of COVID-19 for HCWs, the availability and type of PPE used, and attitudes toward immunization when the vaccine will be available in the country. The study team prepared the draft version of the questionnaire. Then, it was pre-tested in the pilot survey that was undertaken with randomly selected 20 healthcare workers to ensure the clarity of the questions, their suitability to the participants, and possible problems when entering answers in the database. These filled questionnaires were not included in the main study (data not shown).
Sample collection and serology
The period of sampling was during the second wave of the outbreak in Serbia which lasted from June to early October and started much earlier than in other EU countries. All participants signed the informed consent form for participating in the study. The trained nurse collected about 5 ml of venous blood under aseptic precautions. Although the antibody levels in serum/plasma are not subject of circadian rhythm, some factors might analytically interfere with the determination of antibodies, like lipemia, hemolysis, and icterus. In order to avoid grossly lipemic sera samples and unnecessary resampling, blood samples were preferably taken in the morning, after overnight fasting, but if this was not possible, sampling was performed during the day, at least two hours after a meal. Blood was drawn into serum vacutainer with clot activator and serum gel separator, and after forming cloth, serum was separated by centrifugation at 3000 g for 10 min and kept at 4–8 oC. Within the same day, sera samples were transported to the Institute for Application for Nuclear Energy - INEP for further serology testing.
Determination of IgM and IgG antibodies against SARS-CoV-2 was performed using ELISA SARS-CoV-2 IgM and ELISA SARS-CoV-2 IgG (INEP, Belgrade, Serbia). Tests are created at the INEP during May 2020, the CE labeled and registered as the IVD at the Serbian Agency for Drugs and Medical Devices (ALIMS). The principle of the tests is indirect ELISA, and both Nucleocapsid and Spike protein (in equivalent mass ration) are used as capture antigens. Results are expressed semi-quantitatively, as an index, relative to the reference calibrator sample. Sensitivity and specificity for IgM were 97 % and 95 %, respectively, and for IgG were 99 % and 98 %, respectively. The sensitivity of ELISA SARS-CoV-2 IgG and ELISA SARS-CoV-2 IgM were tested using sera samples from COVID-19 confirmed cases and healthy subjects. The internal reference standard was made of pooled sera samples from COVID-19 patients, and signals of all samples were expressed according to the reference sample. For the purpose of this study, the presence of antibodies was confirmed if IgG was positive (antibody index higher than 20, determined on the basis of ROC curve analysis).
Statistical analysis
Descriptive and analytical statistical methods were used in the data processing. Data were presented as mean± SD and number (percentage) for categorical variables. A Chi-square test was used for the analysis of categorical data and an independent t-test was used for continuous variables. The crude prevalence with 95 % CI of seropositivity to IgG was calculated for all participants and for participants in each hospital. The adjusted seroprevalences against SARS-CoV-2 were calculated for total prevalence and across three hospitals, as the following: (observed prevalence + specificity–1) / (sensitivity + specificity–1) [7]. The professional categories were classified into three categories based on the degree of exposure to COVID-19: high, moderate, and low-risk exposure [8]. The sensitivity and specificity were obtained from the manufacturer of the ELISA test, e.g. Institute of nuclear science, Belgrade. Bivariate and multivariate logistic regression was performed using seropositivity as the outcome variable, and other variables with p value of 0.10 or lower in the bivariate analysis as independent variables. The serostatus was used as the dependent variable.
This study was approved by the Ethics Committee of all three University hospitals. Written informed consent was obtained from all participants. All study was carried out in accordance with the ethical standards of the Helsinki Declaration.
Results
Overall, 1608 HCWs participated in the study, 26 were excluded, as they were diagnosed with COVID-19 within the previous month. Thus, 1580 individuals were eligible for the final analysis, representing that was 51.9 % of total HCWs employed in three study hospitals: 62.8 % in non-COVID-19 hospital, 53.7 % in the EC, and 38.9 % in COVID-19 hospital.
Demographic and serology characteristics of HCWs in three hospitals are presented in Table 1. Out of all included HCWs, 289 had positive SARS-CoV-2 IgG antibodies, so the overall seroprevalence across the cohort of HCWs in three hospitals was 18.3 % [95 %CI 16.4–20.3 %]. Significantly higher prevalence of HCWs with positive results for the serum IgG antibody test was observed in COVID hospital (28.6 %, 95 % CI: 24.0–33.6 %) vs. prevalence in the Emergency center (12.6 %, 95 %CI: 10.1–15.4 %), and in the non-COVID hospital (18.3 %, 95 %CI: 15.2–26.7 %) (p < 0.001) (Table 1). The overall prevalence adjusted for declared test sensitivity and specificity would be 16.8 %; that is 27.4 % in COVID-19 hospital, 10.9 % in EC, and 16.8 % in non-COVID hospital.
Table 1.
Demographic and serology characteristics of health care workers in three hospitals.
| Total n (%) | SARS-CoV-2 antibody IgG negative n (%) | SARS-CoV-2 antibody IgG positive n (%) | p value | SARS-CoV-2 antibody IgG positive n (%) | ||||
|---|---|---|---|---|---|---|---|---|
| All | 1580 (100) | 1289 (81.7) | 289 (18.3) | |||||
| Hospital | Hospital | |||||||
| Non-COVID | 575 (36.3) | 468 (81.7) | 105 (18.3) | <0.001 | Non-COVID | Emergency | COVID | P value |
| Emergency | 645 (40.9) | 564 (87.4) | 81 (12.6) | |||||
| COVID | 360 (22.8) | 257 (71.4) | 103 (28.6) | |||||
| Gender | ||||||||
| Females | 1166 (75.4) | 963 (76.2) | 203 (72.0) | 0.138 | 77 (76.2) | 54 (66.7) | 72 (72.0) | 0.360 |
| Males | 380 (24.6) | 301 (23.8) | 79 (28.0) | 24 (23.8) | 27 (33.3) | 28 (28.0) | ||
| Age, years mean±SD | 43.1 ± 29.9 | 43.4 ± 32.6 | 42.1 ± 11.5 | 0.554 | 43.2 ± 11.0 | 41.2 ± 12.2 | 41.8 ± 12.2 | 0.429 |
| 18–29 | 208 (15.7) | 161 (14.8) | 47 (19.7) | 0.213 | 11 (15.5) | 19 (24.4) | 17 (18.9) | 0.613 |
| 30–39 | 350 (26.4) | 293 (27.0) | 57 (23.8) | 18 (25.4) | 21 (26.9) | 18 (20.0) | ||
| 40–49 | 360 (27.2) | 304 (28.0) | 56 (23.4) | 19 (26.8) | 13 (16.7) | 24 (26.7) | ||
| 50–59 | 334 (25.2) | 268 (24.7) | 66 (27.6) | 20 (28.2) | 19 (24.4) | 27 (30.0) | ||
| 60–65 | 73 (5.5) | 60 (5.5) | 13 (5.4) | 3 (4.2) | 6 (7.7) | 4 (4.4) | ||
| Occupation | ||||||||
| Doctors | 346 (21.9) | 271 (21.0) | 75 (26.0) | 0.185 | 27 (25.7) | 18 (22.2) | 30 (29.1) | 0.445 |
| Nurses | 782 (49.6) | 645 (50.0) | 137 (47.4) | 48 (45.7) | 37 (45.7) | 52 (50.5) | ||
| Other | 456 (28.9) | 373 (28.9) | 77 (26.6) | 30 (28.6) | 26 (32.1) | 21 (20.4) | ||
| Risk | ||||||||
| High risk | 450 (28.5) | 355 (27.5) | 95 (32.9) | 0.180 | 11 (10.5) | 12 (14.8) | 72 (69.9) | < 0.001 |
| Medium risk | 806 (51.0) | 666 (51.6) | 140 (48.4) | 78 (74.3) | 45 (55.6) | 17 (16.5) | ||
| Low risk | 324 (20.5) | 270 (20.9) | 54 (18.7) | 16 (15.2) | 24 (29.6) | 14 (13.6) | ||
| Years of service mean±SD | 17.3 ± 11.6 | 17.4 ± 11.6 | 16.7 ± 11.7 | 0.461 | 17.4 ± 11.9 | 16.8 ± 12.2 | 16.6 ± 11.5 | 0.878 |
Gender, occupation, and years of service were not significantly associated with SARS-CoV-2 seropositivity. IgG antibody positive HCWs in three included hospitals did not differ by age, sex, or occupation (p = 0.429; 0.360; and 0.445, respectively) (Table 1).
By comparing SARS-CoV-2 seropositivity in three hospitals with the level of exposure, no statistically significant difference was obtained (Tables 1 and 2). Only when we compared the exposure level among antibody IgG positive HCWs, we found that positive staff in COVID dedicated hospital had had a higher risk of exposure (p < 0.001) (Table 1).
Table 2.
Attitudes and risk perception of healthcare workers in three hospitals related to COVID-19.
| Question | Hospital |
||||
|---|---|---|---|---|---|
| Non-COVID n (%) | Emergency n (%) | COVID n (%) | p value | ||
| Concerns about getting COVID-19 in the workplace | Yes | 405 (76.1) | 427 (69.5) | 287 (84.4) | <0.001 |
| No | 71 (13.3) | 100 (16.3) | 24 (7.1) | ||
| Don't know | 56 (10.5) | 87 (14.2) | 29 (8.5) | ||
| Sufficient level of PPE in the hospital | Yes | 402 (76.0) | 365 (60.0) | 278 (80.6) | <0.001 |
| No | 55 (10.4) | 146 (24.0) | 28 (8.1) | ||
| Don't know | 72 (13.6) | 97 (16.0) | 39 (11.3) | ||
| Which type of mask provides adequate protection during work in the hospital | Medical | 107 (20.8) | 164 (27.7) | 25 (7.3) | <0.001 |
| FFP2N95/KN95 | 372 (72.2) | 374 (63.1) | 298 (87.1) | ||
| None | 36 (7.0) | 55 (9.3) | 19 (5.6) | ||
| Which type of mask provides adequate protection during work in population | Medical | 278 (54.8) | 237 (40.7) | 71 (21.1) | <0.001 |
| FFP2/N95/KN95 | 199 (39.3) | 297 (50.9) | 253 (75.3) | ||
| None | 30 (5.9) | 49 (8.4) | 12 (3.6) | ||
| Believe that has sufficient knowledge about COVID-19 | Yes | 243 (46.6) | 295 (48.7) | 199 (57.8) | 0.007 |
| No | 109 (20.9) | 139 (22.9) | 55 (16.0) | ||
| Don't know | 170 (32.6) | 172 (28.4) | 90 (26.2) | ||
| Will be vaccinated when the vaccine is available | Yes | 134 (25.0) | 157 (25.5) | 153 (44.3) | <0.001 |
| No | 127 (23.6) | 169 (27.5) | 55 (15.9) | ||
| Don't know | 276 (51.4) | 289 (47.0) | 137 (39.7) | ||
Among staff with seropositive test result for IgG against SARS-CoV-2, 30.1 % (87/289) reported no symptoms at all, while 69.9 % (202/289) had at least one COVID-19-compatible symptom.
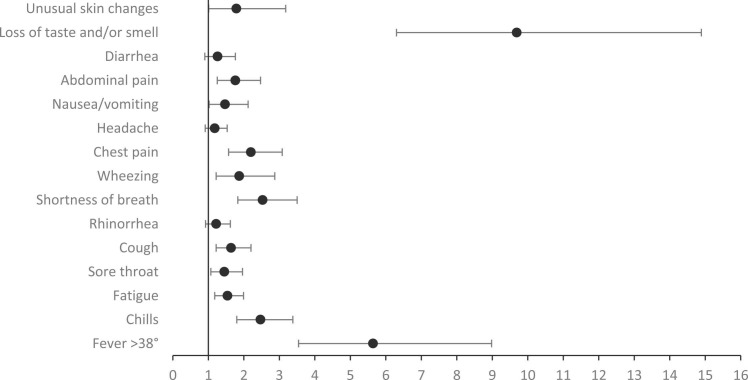
Symptoms strongly associated with seropositivity were anosmia/ageusia (OR = 9.69 95 %CI 6.30–14.89), fever> 380 C (OR = 5.64 95 %CI 3.54–8.98), shortness of breath (OR= 2.53 95 %CI 1.83–3.50), chills (OR = 2.47 95 %CI 1.80–3.38), abdominal pain (OR = 1.76 95 %CI 1.25–2.47), and dry cough (OR = 1.64 95 %CI 1.22–2.20). Rhinorrhea (OR = 1.22 95 %CI 0.92–1.62), headache (OR = 1.18 95 %CI 0.91–1.53), and diarrhea (OR= 1.26 95 %CI 0.90–1.76) did not differ in prevalence between seronegative and seropositive HCWs ( Fig. 1).
Fig. 1.
Symptoms in healthcare workers presented as Odds ratio with 95 % CI for prediction of positivity to SARS-CoV-2 IgG antibody.
Compared to HCWs with an IgG negative serological test, a higher percentage of seropositive HCWs considered that their symptoms were related to COVID-19 (OR = 4.44, 95 %CI 3.16–6.24), more often visited a doctor because of symptoms (OR = 6.14, 95 %CI 3.50–6.48), and were more often absent from work and hospitalized (OR = 12.71, 95 %CI 6.71–24.81).
One hundred and three (35.6 %) HCWs out of 289 with a positive IgG antibody stated that they had a positive PCR test in the past, so they had a history of infection. Seventy-one (10.7 %) of participants that had no prior RT-PCR test were positive for SARS-CoV-2 IgG, so they were asymptomatic cases or they had mild symptoms that did not indicate RT-PCR testing. In addition, Of the 770 HCWs previously tested negative for the RT-PCR test, 115 (14.9 %) were seropositive.
Attitudes and risk perception of HCWs in three hospitals related to COVID-19 are presented in Table 2.
HCWs working in the COVID hospital were most concerned about the possibility of being infected in the hospital (p < 0.001), although they considered in the significantly highest percentage that there have a sufficient quantity of the PPE in hospital. Besides using the respirators (N95 of Chinese equivalent KN95) in the hospital, they preferred to use these types of masks during contact in the population. About half of HCWs in the COVID hospital considered that they had sufficient knowledge of COVID-19, significantly more of those employed at non-COVID hospitals. A small percentage of HCWs in non-COVID hospital, EC, and COVID hospital expressed the statement that they would be vaccinated when the vaccine became available (25.0 %, 25.5 %, 44.3 %, respectively, p < 0.001).
The bivariate and multivariate logistic regression analyses were used to identify risk factors associated with seropositivity. In multivariate logistic regression analysis, the independent predictors for seropositivity were working in COVID-hospital, male gender, the profession of physician, and the presence of the following symptoms: fever, shortness of breath, and anosmia/ageusia ( Table 3).
Table 3.
Risk factors for COVID-19 in healthcare workers, according to bivariate and multivariate logistic regression.
| Variables | Bivariate logistic regression |
Multivariate logistic regression |
||
|---|---|---|---|---|
| OR (95 % CI) | P value | OR (95 % CI) | P value | |
| Non-COVID hospital | ref. | ref. | ||
| Emergency hospital | 0.64 (0.47–0.88) | 0.006 | 0.87 (0.56–1.35) | 0.540 |
| COVID-hospital | 1.79 (1.31–2.45) | <0.001 | 1.78 (1.16–2.78) | 0.009 |
| Gender | ||||
| Male Female |
ref. 1.246 (0.93–1.67) |
0.137 | ||
| Age | 1.00 (0.99–1.01) | 0.659 | ||
| Occupation | ||||
| Physicians | 1.34 (0.94–1.90) | 0.109 | 1.62 (1.05–2.62) | 0.048 |
| Nurses | 1.03 (0.76–1.40) | 0.863 | 0.85 (0.54–1.32) | 0.465 |
| Other | ref. | ref. | ||
| Risk | ||||
| High risk | 1.34 (0.92–1.94) | 0.123 | ||
| Medium risk | 1.05 (0.74–1.48) | 0.777 | ||
| Low risk | ref. | |||
| Years of service | 0.99 (0.98–1.01) | 0.461 | ||
| Contact with COVID-19 positive person | 1.79 (1.33–2.40) | <0.001 | ||
| Had symptoms | 1.15 (0.86–1.53) | 0.344 | ||
| Symptoms | ||||
| Fever >38° | 5.64 (3.54–8.98) | <0.001 | 2.95 (1.63–5.34) | 0.001 |
| Chills | 2.47 (1.80–3.38) | <0.001 | ||
| Fatigue | 1.54 (1.18–1.99) | 0.001 | ||
| Sore throat | 1.45 (1.07–1.96) | 0.017 | ||
| Cough | 1.64 (1.22–2.20) | 0.001 | ||
| Rhinorrhea | 1.22 (0.92–1.62) | 0.166 | ||
| Shortness of breath | 2.53 (1.83–3.50) | <0.001 | 1.91 (1.22–2.99) | 0.005 |
| Wheezing | 1.87 (1.22–2.87) | 0.004 | ||
| Chest pain | 2.20 (1.57–3.08) | <0.001 | ||
| Headache | 1.18 (0.91–1.53) | 0.209 | ||
| Nausea/vomiting | 1.47 (1.02–2.12) | 0.043 | ||
| Abdominal pain | 1.76 (1.25–2.47) | 0.001 | ||
| Diarrhea | 1.26 (0.90–1.76) | 0.172 | ||
| Anosmia and/or ageusia | 9.69 (6.30–14.89) | <0.001 | 4.88 (2.82–8.44) | <0.001 |
| Unusual skin changes | 1.79 (1.01–3.18) | 0.046 | ||
| Do you think that the hospital where you work has a sufficient level of protection against COVID-19? | ||||
| No | ref. | |||
| Yes | 1.54 (1.03–2.32) | 0.037 | ||
| I don’t know | 1.47 (0.88–2.46) | 0.139 | ||
| Which type of mask provides adequate protection during your work in the hospital? | ||||
| Medical | ref. | |||
| FFP2/N95/KN95 | 1.18 (0.84–1.66) | 0.345 | ||
| None | 0.96 (0.55–1.78) | 0.963 | ||
| Do you consider that you have had adequate on-the-job training on protection measures against COVID-19? | ||||
| No | ref. | |||
| Yes | 1.26 (0.91–1.75) | 0.163 | ||
| I don’t know | 1.00 (0.63–1.58) | 0.992 | ||
Discussion
We presented the results of the SARS-CoV-2 seroprevalence study conducted among three university hospitals in Belgrade, Serbia, that were under different COVID regimes Overall adjusted prevalence was 16.8 % with significant differences associated with the type of hospital, non-COVID, Emergency, and COVID-19 hospital. The highest seroprevalence was observed in COVID dedicated hospital. Further, a higher proportion of HCWs in this hospital had high-risk exposure.
Our results are similar for those obtained in the studies conducted during almost the same period in the East of Scotland [9] where the seroprevalence was 14.5 %, then in two hospitals in the London area (UK) [10] (19.7 %) and two hospitals in North Italy (17.1 %) [11].
Contrary to our finding, the overall prevalence in three hospitals in Turkey was 2.7 %, the highest 7.2 % in one of these hospitals [12]. Low prevalence was also recorded in other counties such as Spain [13], Greece [14], and Portugal [15], [16]. However, the organization of work in these hospitals was different than in our three hospitals. In Turkey, doctors worked in COVID-19 clinics on a daily or monthly basis and then moved to non-COVID hospitals. In Belgrade, there was a strict separation of COVID and non-COVID hospitals. In those that were COVID hospitals, like one included in this study, all wards were used for the treatment of patients with COVID-19, and staff only worked there. Long-term contact exclusively with COVID-19 patients, as well as the possibility of losing attention and non-compliance with protection measures, may have caused the highest prevalence of positive staff in COVID hospital. Higher seroprevalence among personnel delivering direct care to COVID-19 patients than among those with lower exposure has been observed in other studies, [17], [18]. However, SARS-CoV-2 antibody prevalence among HCWs in the COVID dedicated hospital was lower than the prevalence in the Spanish referral hospital obtained at the beginning of the pandemic, during the peak of the first wave [19], but higher than overall seroprevalence among more than 6 thousand employees in 17 hospitals in this country [20].
In the recently published systematic review and meta-analysis with 49 articles finally included in meta-analysis out of 3632 included at the beginning, it was found that the overall seroprevalence of SARS-CoV-2 antibodies was 8.7 %, higher in studies conducted in North America (12.7 %) than in Europe (8.5 %) [21]. The study described here was conducted at the end of the second wave which started earlier than in EU counties. Higher community prevalence is reflected in a higher prevalence in HCWs [22]. Therefore, a higher prevalence of seropositivity in Serbian HCWs compared to the EU at the same period, might be due to the second wave started and peaked in Serbia earlier than in other European countries.
Longer hospitalization of patients who might be without any symptom at admission, but in the incubation period and later became RT-PCR test positive, might influence higher seroprevalence in non-COVID hospital than in the Emergency Center, which is a short stay center for persons suffering from an accident or re-aggravation of certain chronic diseases. Personnel in the non-COVID hospital were obligated to wear surgical masks and visors. However, the last recommendation has not always been respected.
We did not reveal the correlation between workplace exposure levels and seropositivity. Contrary to our findings, it was found that seroprevalence was higher in high- and moderate-risk exposure [20]. Unfortunately, we did not record the length of exposure, nor whether the PPE was worn properly. It was already found that exposure should be considered by possible exposure in the community settings, not only during patient care work activities [23]. Our primary goal was to compare risk in three types of hospitals since one of the hospitals was completely transformed into a hospital only for the care of COVID-19 patients. The process of converting the whole hospital into a dedicated COVID-19 specialty hospital has been practiced in other countries in response to unprecedented surges in COVID-19 patients [24], [25].
One-third of seropositive HCWs remained subclinical in our study, which is twice as high as in the study conducted in the one hospital in Belgium (15 %) [26]. According to the systematic review and meta-analysis, 40 % of HCWs had not any COVID-19 symptoms at the time of diagnosis [27].
Knowledge of the frequency of asymptomatic cases of COVID-19 among HCWs is important for obtaining information regarding the extent of exposure to SARS-CoV-2 virus and infections in the population. We found that 10.7 % of non-tested by PCR HCWs and 14.9 % of RT-PCR negative HCWs were serologically positive. A lower proportion of IgG positive staff with previous negative RT-PCR tests was found in studies conducted in the USA [28], [29]. Although PCR test is assumed as the gold standard in COVID-19 diagnostics, false-negative results are rarely seen. Those people could be false negative, which is particularly worrisome because they can continue to spread the infection. A similar situation is with asymptomatic individuals. In the large cross-sectional study that included 17 hospitals in different regions in Spain, it was revealed that 40 % of HCWs seropositive for IgG against SARS-CoV-2 were asymptomatic [20]. Both of these groups have great epidemiological significance not only in health care institutions but also in the population.
Anosmia and ageusia were the most common symptoms related to seropositivity, as was noted in other studies [30]. One year after the beginning of the pandemic, in the analysis of the multicenter international HOPE registry of COVID-19 patients, it was found that olfactory and gustatory dysfunction be inversely related to death [31] and can be one of the prognostic factors. A significant proportion of COVID-19 cases have these impairments 3–7 months after recovering [32] that can lead to the development of anxiety and depression [33]. In our study, among the symptoms analyzed by multivariate logistic regression, fever, shortness of breath, and anosmia/ageusia were the independent predictors of the COVID-19.
At the time of the study, vaccines against COVID-19 were in the final stages of clinical trials in the world. HCWs in our COVID dedicated hospital were more aware of vaccine uptake when the vaccine would be available. At that time, the national vaccination campaign aimed at raising awareness of the importance of vaccines in the prevention of COVID-19 had not yet started in our country. An effective vaccine campaign is a necessary tool to reduce rapid SARS-CoV-2 virus transmission [34]. Healthcare professionals have a special and important role in achieving high vaccine coverage in the population [35], [36], especially in the new vaccine introduction, such as the mRNA vaccines. Adverse events following the use of mRNK vaccine by healthcare workers have been demonstrated to be benign and similar to those in the general population [37]. Based on their own example, health care providers have even greater opportunities to promote the safety of new types of vaccine.
Multivariate logistic regression also revealed a significantly higher risk of seropositivity among HCWs working in COVID-hospital, physicians, and males.
The cohort study conducted in the UK revealed that HCWs have seven times higher risk for COVID-19 than those with other types of jobs [38]. Working in a COVID dedicated hospital increased the risk of seropositivity by 75 % in our study. HCWs are at increased risk of COVID-19 due to the close and long contact with patients during their daily activities. In the recently published systematic review and meta-analysis which included articles about seroprevalence in HCWs before the immunization has started, it was found inconsistent evidence about a higher risk of COVID-19 for frontline HCWs [39].
Nurses, particularly those in the intensive care units, are at the highest risk for COVID-19 infections among all HCWs [40], [41]. Contrary to these findings, we found a higher estimated risk for infections among physicians. In the study which includes 13,500 physicians, it was shown that all male physicians aged 60 or older have a 3.8 hazard ratio for death of COVID-19 with exacerbating by ethnicity and personal comorbidities [42].
The limitations of our study were well-known limitations of cross-sectional study design, i.e., observation in one point of time. Enrolment in the study was not random but voluntary. Consequently, there is a possibility for selection bias. Concerns regarding own serological status due to the known close contact with a COVID-19 patient either in a household without PPE or inappropriate use of PPE in the workplace could motivate participation in the survey. Furthermore, we did not specifically ask whether family members had COVID-19. Further, we conducted this study at the end of the second wave when there was a high number of cases in the population and a risk of infection for HCWs. At the time of conducting the study, Serbia was not in total lockdown, while the patients' flow was intense and exposure of HCW correspond to exposure in the general population. All of this could cause an overestimate of prevalence.
The main strength of our study is that it was conducted in three hospitals with different groups of patients in relation to COVID-19 and different levels of protection for HCWs. We were able to assess the risk to HCWs not only in COVID-19 hospital but also in hospitals where patients potentially might be infectious when admitted as asymptomatic or during the incubation period but may transmit the disease. For this reason, the mandatory wearing of face shields with a medical mask has been introduced in non-COVID hospitals. In the third wave, which was more dangerous than the previous two, staff was wearing respirators not only in COVID-19 dedicated hospitals but also in other hospitals if they were working in high-risk departments. Fortunately, enough PPE was available in Serbia, mainly KN95 and FFP2 respirators.
Conclusions
Working in COVID-hospital, being a physician, and males, and having fever, shortness of breath, and anosmia/ageusia were associated with increased probability of SARS-CoV-2 infection. The possibility that patients in non-COVID hospitals, and those admitted to the Emergency room might also be infectious, although PCR tested, imposes the need for the use of PPE also in non-COVID medical institutions.
Acknowledgements
This work was supported by the Ministry of Education, Science and Technological development of Serbia and the Ministry of Health of Republic of Serbia.
Contributor Information
Members of SRBSEHCW Study group:
Vuk Marusic, Viseslav Popadic, Bogdan Crnokrak, Borislav Toskovic, Slobodan Klasnj, Andrea Manojlovic, Marija Brankovic, Vesna Mioljevic, Zlatko Perisic, E, Maja Djordjevic, Stevana Vukasinovic, Sladjana Mihajlovic, and Olivera Ostojic
References
- 1.Ministry of Health or Republic of Serbia. First case of COVID-19, 〈https://www.zdravlje.gov.rs/vest/346180/registrovan-prvi-oboleli-od-covid-19.php〉; 2021[accessed 8 March 2021].
- 2.Forte A., Orri M., Brandizzi M., Iannaco C., Venturini P., Liberato D., et al. “My Life during the Lockdown”: emotional experiences of european adolescents during the COVID-19 crisis. Int J Environ Res Public Health. 2021;18(14):7638. doi: 10.3390/ijerph18147638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Institute of Public Health of Serbia. Belgrade, Republic Expert Committee for healthcare-associated infections. Guideline of prevention and control of SARS-CoV-2 transmission in the health care facilities [in Serbian], 〈https://www.batut.org.rs/download/aktuelno/MereprevencijeSARS-CoV-2zdravstvenim%20ustanovama-dopunjeno.pdf〉; 2021[accessed 15 September 2021].
- 4.Elias C., Sekri A., Leblanc P., Cucherat M., Vanhems P. The incubation period of COVID-19: a meta-analysis. Int J Infect Dis. 2021;104:708–710. doi: 10.1016/j.ijid.2021.01.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rai B., Shukla A., Dwivedi L.K. Incubation period for COVID-19: a systematic review and meta-analysis. Z Gesund Wiss. 2021;23:1–8. doi: 10.1007/s10389-021-01478-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Institute of public health of Serbia, Republic Expert Committee for Healthcare-associated Infections. Personal protective equipment. Protection of health workers from infection caused by a SARS-CoV-2 virus, 〈https://www.batut.org.rs/download/aktuelno/LZO%20za%20COVID-19_RSK%20za_BI_15.3.2020.pdf〉; 2021 [accessed 15 September 2021].
- 7.Sempos C.T., Tian L. Adjusting coronavirus prevalence estimates for laboratory test kit error. Am J Epidemiol. 2021;190:109–115. doi: 10.1093/aje/kwaa174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.OSHA. Guidance on Preparing Workplaces for COVID-19. U.S. Department of Labor. Occupational Safety and Health Administration, OSHA, 〈https://www.preventionweb.net/publication/guidance-preparing-workplaces-covid-19〉; 2020 [accessed 18. January 2022].
- 9.Abo-Leyah H., Gallant S., Cassidy D., Giam Y.H., Killick J., Marshall B., et al. The protective effect of SARS-CoV-2 antibodies in Scottish healthcare workers. ERJ Open Res. 2021;7(2):00080–02021. doi: 10.1183/23120541.00080-2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ken-Dror G., Wade C., Sharma S.S., Irvin-Sellers M., Robin J., Fluck D., et al. SARS-CoV-2 antibody seroprevalence in NHS healthcare workers in a large double-sited UK hospital. Clin Med (Lond) 2021;21(3):e290–e294. doi: 10.7861/clinmed.2020-1096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Airoldi C., Patrucco F., Milano F., Alessi D., Sarro A., Rossi M.A., et al. High Seroprevalence of SARS-CoV-2 among Healthcare Workers in a North Italy Hospital. Int J Environ Res Public Health. 2021;18(7):3343. doi: 10.3390/ijerph18073343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alkurt G., Murt A., Aydin Z., Tatli O., Agaoglu N.B., Irvem A., et al. Seroprevalence of coronavirus disease 2019 (COVID-19) among health care workers from three pandemic hospitals of Turkey. PLoS One. 2021;16(3) doi: 10.1371/journal.pone.0247865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Barallat J., Fernández-Rivas G., Quirant-Sánchez B., González V., Doladé M., Martinez-Caceres E., et al. Seroprevalence of SARS-CoV-2 IgG specific antibodies among healthcare workers in the Northern Metropolitan Area of Barcelona, Spain, after the first pandemic wave. PLoS One. 2020;15(12) doi: 10.1371/journal.pone.0244348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Psichogiou M., Karabinis A., Pavlopoulou I.D., Basoulis D., Petsios K., Roussos S., et al. Antibodies against SARS-CoV-2 among health care workers in a country with low burden of COVID-19. PLoS One. 2020;15(12) doi: 10.1371/journal.pone.0243025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Silva D.F., Silva P.D., Torgal A., Braga S., Rocha D., Ochoa C., et al. Seroprevalence of Anti-SARS-CoV-2 antibodies three months post infection in healthcare professionals at an oncology hospital in Northern Portugal. Acta Med Port. 2021;34(6):484–485. doi: 10.20344/amp.16336. [DOI] [PubMed] [Google Scholar]
- 16.Kislaya I., Gonçalves P., Barreto M., Sousa R., Garcia A.C., Matos R., et al. ISNCOVID-19 Group. Seroprevalence of SARS-CoV-2 infection in Portugal in May–July 2020: results of the First National Serological Survey (ISNCOVID-19) Acta Med Port. 2021;34(2):87–94. doi: 10.20344/amp.15122. [DOI] [PubMed] [Google Scholar]
- 17.Eyre D.W., Lumley S.F., O'Donnell D., Campbell M., Sims E., Lawson E., et al. Differential occupational risks to healthcare workers from SARS-CoV-2 observed during a prospective observational study. Elife. 2020;9 doi: 10.7554/eLife.60675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.van der Plaat D.A., Madan I., Coggon D., van Tongeren M., Edge R., Muiry R., et al. Risks of COVID-19 by occupation in NHS workers in England. Occup Environ Med. 2021 doi: 10.1136/oemed-2021-107628. oemed-2021-107628. [DOI] [PubMed] [Google Scholar]
- 19.Galán M.I., Velasco M., Casas M.L., Goyanes M.J., Rodríguez-Caravaca G., Losa-García J.E., et al. Working Group Alcorcón COVID-19 investigators; Working Group Alcorcón COVID-19 investigators. Hospital-Wide SARS-CoV-2 seroprevalence in health care workers in a Spanish teaching hospital. Enferm Infecc Microbiol Clin (Engl Ed) 2020;S0213–005X(20) doi: 10.1016/j.eimc.2020.11.015. 30418-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Varona J.F., Madurga R., Peñalver F., Abarca E., Almirall C., Cruz M., et al. Seroprevalence of SARS-CoV-2 antibodies in over 6000 healthcare workers in Spain. Int J Epidemiol. 2021;50(2):400–409. doi: 10.1093/ije/dyaa277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Galanis P., Vraka I., Fragkou D., Bilali A., Kaitelidou D. Seroprevalence of SARS-CoV-2 antibodies and associated factors in healthcare workers: a systematic review and meta-analysis. J Hosp Infect. 2021;108:120–134. doi: 10.1016/j.jhin.2020.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gupta K., Bellino P., Samano J.G., Strymish J., O’Brien W., Sprague R.S., et al. Minimal population prevalence and mortality of coronavirus disease 2019 in healthcare personnel. Open Forum Infect Dis. 2020;8(2):ofaa618. doi: 10.1093/ofid/ofaa618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gohil S.K., Quan K.A., Madey K.M., King-Adelsohn S., Tjoa T., Tifrea D., et al. Infection prevention strategies are highly protective in COVID-19 units while main risks to healthcare professionals come from coworkers and the community. Antimicrob Resist Infect Control. 2021;10(1):163. doi: 10.1186/s13756-021-01031-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Król Z., Szymański P., Bochnia A., Abramowicz E., Płachta A., Rzepliński R., et al. Transformation of a large multi-speciality hospital into a dedicated COVID-19 centre during the coronavirus pandemic. Ann Agric Environ Med. 2020;27(2):201–206. doi: 10.26444/aaem/123801. [DOI] [PubMed] [Google Scholar]
- 25.Fadaak R., Davies J.M., Blaak M.J., Conly J., Haslock J., Kenny A., et al. Rapid conversion of an in-patient hospital unit to accommodate COVID-19: An interdisciplinary human factors, ethnography, and infection prevention and control approach. PLoS One. 2021;16(1) doi: 10.1371/journal.pone.0245212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Steensels D., Oris E., Coninx L., Nuyens D., Delforge M.L., Vermeersch P., et al. Hospital-Wide SARS-CoV-2 antibody screening in 3056 staff in a tertiary center in Belgium. JAMA. 2020;324(2):195–197. doi: 10.1001/jama.2020.11160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gómez-Ochoa S.A., Franco O.H., Rojas L.Z., Raguindin P.F., Roa-Díaz Z.M., Wyssmann B.M., et al. COVID-19 in Health-care workers: a living systematic review and meta-analysis of prevalence, risk factors, clinical characteristics, and outcomes. Am J Epidemiol. 2021;190(1):161–175. doi: 10.1093/aje/kwaa191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Papasavas P., Olugbile S., Wu U., Robinson K., Roberts A.L., O’Sullivan D.M., et al. Seroprevalence of SARS-CoV-2 antibodies, associated epidemiological factors and antibody kinetics among healthcare workers in Connecticut. J Hosp Infect. 2021;114:117–125. doi: 10.1016/j.jhin.2021.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kataria Y., Cole M., Duffy E., de la Cena K., Schechter-Perkins E.M., Bouton T.C., et al. Seroprevalence of SARS-CoV-2 IgG antibodies and risk factors in health care workers at an academic medical center in Boston, Massachusetts. Sci Rep. 2021;11(1):9694. doi: 10.1038/s41598-021-89107-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rudberg A.S., Havervall S., Månberg A., Jernbom Falk A., Aguilera K., Ng H., et al. SARS-CoV-2 exposure, symptoms and seroprevalence in healthcare workers in Sweden. Nat Commun. 2020;11(1):5064. doi: 10.1038/s41467-020-18848-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Porta-Etessam J., Núñez-Gil I.J., González García N., Fernandez-Perez C., Viana-Llamas M.C., Eid C.M., et al. COVID-19 anosmia and gustatory symptoms as a prognosis factor: a subanalysis of the HOPE COVID-19 (Health Outcome Predictive Evaluation for COVID-19) registry. Infection. 2021;49(4):677–684. doi: 10.1007/s15010-021-01587-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bussière N., Mei J., Lévesque-Boissonneault C., Blais M., Carazo S., Gros-Louis F., et al. Chemosensory dysfunctions induced by COVID-19 can persist up to 7 Months: a study of over 700 healthcare workers. Chem Senses. 2021 doi: 10.1093/chemse/bjab038. 46:bjab038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Boscolo-Rizzo P., Hummel T., Hopkins C., Dibattista M., Menini A., Spinato G., et al. High prevalence of long-term olfactory, gustatory, and chemesthesis dysfunction in post-COVID-19 patients: a matched case-control study with one-year follow-up using a comprehensive psychophysical evaluation. Rhinology. 2021 doi: 10.4193/Rhin21.249. [DOI] [PubMed] [Google Scholar]
- 34.Rossman H., Shilo S., Meir T., Gorfine M., Shalit U., Segal E. COVID-19 dynamics after a national immunization program in Israel. Nat Med. 2021;27(6):1055–1061. doi: 10.1038/s41591-021-01337-2. [DOI] [PubMed] [Google Scholar]
- 35.Wang C., Wang Y., Han B., Zhao T.S., Liu B., Liu H., et al. Willingness and SARS-CoV-2 vaccination coverage among healthcare workers in China: a nationwide study. Vaccin (Basel) 2021;9(9):993. doi: 10.3390/vaccines9090993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Li M., Luo Y., Watson R., Zheng Y., Ren J., Tang J., et al. Healthcare workers’ (HCWs) attitudes and related factors towards COVID-19 vaccination: a rapid systematic review. Post Med J. 2021:2021–140195. doi: 10.1136/postgradmedj-2021-140195. (postgradmedj) [DOI] [PubMed] [Google Scholar]
- 37.Ripabelli G., Tamburro M., Buccieri N., Adesso C., Caggiano V., Cannizzaro F., et al. Active surveillance of adverse events in healthcare workers recipients after vaccination with COVID-19 BNT162b2 vaccine (Pfizer-BioNTech, Comirnaty): a cross-sectional study. J Community Health. 2022;47(2):211–225. doi: 10.1007/s10900-021-01039-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mutambudzi M., Niedwiedz C., Macdonald E.B., Leyland A., Mair F., Anderson J., et al. Occupation and risk of severe COVID-19: prospective cohort study of 120 075 UK Biobank participants. Occup Environ Med. 2020;78(5) doi: 10.1136/oemed-2020-106731. 307–1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kayı İ., Madran B., Keske Ş., Karanfil Ö., Arribas J.R., Psheniсhnaya N., et al. The seroprevalence of SARS-CoV-2 antibodies among health care workers before the era of vaccination: a systematic review and meta-analysis. Clin Microbiol Infect. 2021;27(9):1242–1249. doi: 10.1016/j.cmi.2021.05.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wratil P.R., Schmacke N.A., Osterman A., Weinberger T., Rech J., Karakoc B., et al. In-depth profiling of COVID-19 risk factors and preventive measures in healthcare workers. Infection. 2021;11:1–14. doi: 10.1007/s15010-021-01672-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Al Youha S., Alowaish O., Ibrahim I.K., Alghounaim M., Abu-Sheasha G.A., Fakhra Z., et al. Factors associated with SARS-CoV-2 infection amongst healthcare workers in a COVID-19 designated hospital. J Infect Public Health. 2021;14(9):1226–1232. doi: 10.1016/j.jiph.2021.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Verma A.M., Goddard A.F., O'Donoghue D., Newbery N., Phillips C., Trudgill N. The UK physician workforce: one-third at increased risk of death from COVID-19. Clin Med (Lond) 2021;21(2):e161–e165. doi: 10.7861/clinmed.2020-0756. [DOI] [PMC free article] [PubMed] [Google Scholar]