Abstract
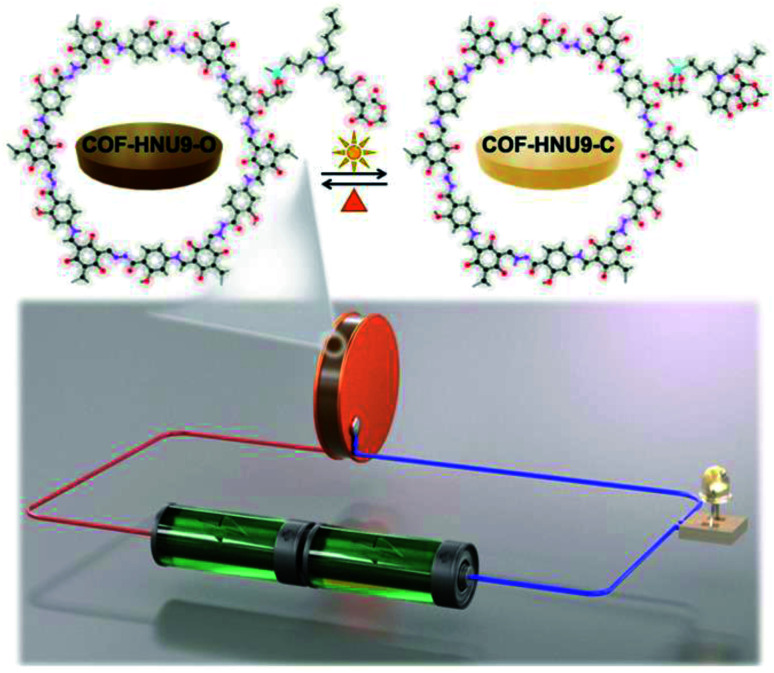
In recent years, covalent organic frameworks (COFs) have attracted enormous interest as a new generation of proton-exchange membranes, chemical sensors and electronic devices. However, to design high proton conductivity COFs, especially those with stimulus responsive performance remains a great challenge. Here, the first example of a light/heat switchable COF (COF-HNU9) has been synthesized by grafting a donor–acceptor Stenhouse adduct (DASA) within the channels of a β-ketoenamine-based COF. DASA groups in the nanopores of COF-HNU9 undergo a reversible open–closed photoisomerization upon visible light irradiation and are recovered by heating. Thus, COF-HNU9 exhibits not only a remarkably high proton conductivity, but also a highly effective switching performance. Under visible light irradiation at 98% RH, the proton conductivity of COF-HNU9 increases by three orders of magnitude at 25 °C and is up to 0.02 S cm−1 at 80 °C. Furthermore, the proton conductivity does not display any significant decrease even after 20 switching cycles. These results have been rationalized by a Grotthuss-type mechanism and verified by DFT calculations. The stimuli-responsive COF is conceptually confirmed by an optical control device with the light/heat switching proton conductive COF-HNU9 film, which is able to remote-control the illumination and switching off of an LED lamp without any current amplifier.
A novel visible light/heat responsive 2D COF is designed and synthesized to achieve high proton conductivity (0.02 S cm−1), remarkable conductivity modulation (three orders of magnitude), and excellent recyclability (more than 20 cycles).
Introduction
Proton exchange membrane (PEM) fuel cells1,2 and electronic devices3–5 have advantages such as high power density, zero pollution, and mild working conditions, therefore they have attracted extensive attention in recent years. The proton-conductive material for the PEM is the key component of these devices. From the viewpoint of practical application, a promising candidate for solid state proton conductors requires not only high proton conductivity but also high stability to make them viable for long-term performance.1c At present, many solid proton electrolytes are mainly based on polymeric materials, particularly perfluorosulfonic polymers due to their high proton conductivity.2b Nonetheless, the amorphous nature of these polymers makes it difficult to regulate the transport pathway of protons and then to improve the proton conductivity. Thus, great efforts have been devoted to developing new stable proton conductors with well-defined pore structures, high proton conductivity and tunability.6–9
Covalent organic frameworks (COFs) are a new class of crystalline polymers covalently constructed from organic building blocks.10 Due to the unique characteristics of periodic structure, designable regular open channels, and tunable functionality, COFs offer a useful platform for exploring new proton conductors11–14 and have shown great potential in PEM fuel cells and electronic devices.15–19 For example, two novel sulfonated COFs were designed and synthesized by Zhao and co-workers,12 and both of these COFs showed high intrinsic proton conductivity of up to 3.96 × 10−2 S cm−1 at 97% relative humidity (RH) under ambient conditions because of the denser content of sulfonic acid groups. Banerjee's group15a reported the first example of an extrinsic proton-conducting COF (Tp–Azo) synthesized from triformylphloroglucinol (Tp) and 4,4′-azodianiline (Azo), where H3PO4 was immobilized in Tp–Azo and applied as a proton conductor to obtain a conductivity of 9.9 × 10−4 S cm−1 at 59 °C and 98% RH. Recently, Zhu's group16a prepared a cationic COF and then the phosphotungstic anionic PW12O403− was introduced by ion exchange for proton conduction, and the charged COF exhibited a proton conductivity of 3.32 × 10−3 S cm−1 at room temperature and 97% RH. In another example, Jiang and co-workers17a reported an ultra-stable COF in which H3PO4 was confined for efficient proton conduction at high temperature. The anhydrous proton conductivity was high up to 1.91 × 10−1 S cm−1 at 160 °C upon continuous running over 20 h. However, literature survey reveals that stimuli-responsive proton conductive COFs have never been reported so far in spite of their great potential in remote-controllable sensors and electronic devices as shown by stimuli-responsive metal organic frameworks (MOFs).20,21
Among various external stimuli,22 visible light is one of the most appealing, widely available, and environmentally friendly triggers.23 Significantly, light signal can be controlled remotely and precisely in space and time and switched rapidly in a clean and noninvasive way.24 For the fabrication of light-responsive porous materials, an attractive strategy is to incorporate photo-switchable molecules, including azobenzene, spiropyran and diarylethene,25 into the modular framework structures. Recently, a new kind of light/heat switchable molecules, donor–acceptor Stenhouse adduct (DASA), has been reported, which is capable of a reversible switching between the open neutral state (DASA-O) and closed zwitterionic state (DASA-C) triggered by visible light irradiation and heating in non-polar aromatic solvents.26 Such a structural transformation results in a significant dipole moment change of the molecules. Thus, DASA can be considered to be a promising candidate of photo-switchable molecules for light-responsive porous materials.
Herein, for the first time, DASA groups were incorporated within the channels of a β-ketoenamine-based COF as dangling groups by chemical grafting to form a photoresponsive smart COF (denoted as COF-HNU9). We found that the pendent DASA groups in COF-HNU9 showed photoswitchable property from the open neutral state (COF-HNU9-O) to the closed zwitterionic state (COF-HNU9-C) under the irradiation of visible light and were recovered by heating, which is similar to the dissolved molecules in aromatic solvents. Importantly, the as-synthesized COF-HNU9 exhibits three orders of magnitude increase in proton conductivity compared with the DASA-free β-ketoenamine-based COF after visible light irradiation at 25 °C and 98% RH when reaching the photostationary state, and the proton conductivity is as high as 0.02 S cm−1 at 80 °C. It is also revealed that the proton conductance in the light-responsive COF follows a Grotthuss-type mechanism. As a proof of concept, a light-controlled proton conductive film was assembled into a photo-responsive device to realize the switchable lighting and turning off of a light-emitting diode (LED) lamp without any current amplifier. It was clearly demonstrated that the device was able to illuminate the LED lamp under irradiation with visible light, which could then be switched off in the dark as shown in Fig. 1.
Fig. 1. Illustration of the photo-controlled circuit to light the LED lamp due to the change in proton conductivity resulting from the open–closed–open isomerization of DASA groups in COF-HNU9 switched by visible light and heat.
Results and discussion
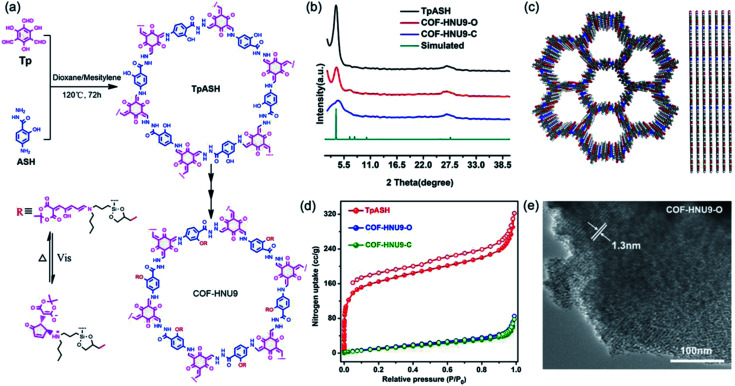
Due to the high chemical stability and easy derivative nature, in this work, we selected a β-ketoenamine-linked COF (TpASH) with free hydroxyl (−OH) groups as the parent framework for the synthesis of DASA functionalized COFs. TpASH was synthesized via a condensation reaction between 1,3,5-triformylphloroglucinol (Tp) and 4-aminosalicylhydrazide (ASH) by a traditional solvothermal approach (see details in the ESI†). Compared to the original synthetic route by solid grinding,27 the TpASH synthesized by the solvothermal approach showed much enhanced crystallinity. The chemical structure of the TpASH is shown in Fig. 2a and characterized by FT-IR (Fig. S1†), SEM and TEM (Fig. S2†), and PXRD (Fig. 2b), which suggest the preferable crystal structure of the AA eclipsed stacking mode (Fig. 2c). It was found that TpASH had a BET surface area of 614 m2 g−1 (Fig. S3†), which is obviously higher than that synthesized by solid grinding reported in the literature (500 m2 g−1).27 The total pore volume of TpASH was estimated (at P/P0 = 0.99) to be Vp = 0.50 cm3 g−1 (Fig. S3†). Meanwhile, high thermal stability (Fig. S4†) and chemical stability (Fig. S5†) of the TpASH were also confirmed.
Fig. 2. (a) Synthetic scheme of TpASH and COF-HNU9. (b) PXRD patterns of TpASH and COF-HNU9. The first peak in the PXRD pattern is retained after post-synthetic modification signifying its structural integrity. (c) Views of the layered 2D-crystal structure of TpASH resulting from an eclipsed AA stacking model. (d) Nitrogen absorption curves of TpASH and COF-HNU9 at 77 K. (e) TEM image of COF-HNU9 before visible light irradiation.
The visible light and heat responsive COF-HNU9 was synthesized by anchoring the photo-switchable DASA molecules onto the channel walls using post-synthetic modification steps. Due to the high reactivity, moderate greasy nature, and suitable photostationary/dark equilibrium,28 (3-butylaminopropyl)trimethoxysilane was chosen as the “donor” amine to react with activated furans to synthesize DASA units. It was noted that the FT-IR spectra of COF-HNU9 exhibited asymmetric and symmetric C–H stretching vibration peaks at 2920 and 2853 cm−1, respectively (Fig. S6†), confirming the existence of the methyl and methylene fragments of the linking groups. As shown by the PXRD patterns in Fig. 2b, COF-HNU9 showed a similar diffraction pattern to that of TpASH but with a broadened peak width at 2θ = ∼4.0°. This result can be ascribed to the existence of a large number of flexible chains in the cavities of COF-HNU9, thus slightly contorting the π–π stacking interactions between its layers. SEM analysis indicates that COF-HNU9 had similar morphology to that of TpASH (Fig. S2†), but its BET surface area was only 54 m2 g−1 (Fig. 2d). This low surface area compared to the DASA-free TpASH (614 m2 g−1, Fig. S3†) could be caused by the presence of the DASA groups within the pores. The total pore volume of COF-HNU9 was also estimated, which was about 0.13 cm3 g−1. Such a significant decrease after DASA modification indicates once again that the DASA functional group is distributed inside the pores of COF-HNU9 and occupies a certain pore volume. We also measured the CO2 adsorption isotherm of COF-HNU9 at 195 K, which showed a type I adsorption isotherm (Fig. S7†) and a BET surface area of 125 m2 g−1 (Fig. S7†). It is clear that the BET surface area was much higher than that (54 m2 g−1) determined by the N2 adsorption isotherm of COF-HNU9 at 77 K. This indicates that the flexible DASA units might block the pore opening at low temperatures like 77 K, whereas 195 K is a temperature that is high enough to give the DASA units sufficient flexibility and thus a higher value of BET surface area. The pore size distribution profiles revealed that COF-HNU9 exhibited only one type of micropore structure with the size distribution at 1.3 nm (Fig. S8†), which is smaller than the value of 1.5 nm before functionalization (Fig. S3†). All these results were well consistent with that derived from HR-TEM analysis (Fig. 2e). In addition, compared with TpASH, the solid state 13C NMR spectrum of COF-HNU9 exhibited additional characteristic signals of –CH3, –CH2 and –CH of the DASA groups at around 6, 19–62, and 70 ppm, respectively (Fig. 3a and b), verifying the successful incorporation of DASAs in TpASH as dangling groups. The elemental analysis of COF-HNU9 showed that the C, H, N, and O contents were 52.12, 6.33, 9.59 and 27.50%, respectively, which were close to the values of 57.69, 5.55, 7.92 and 24.87% estimated from an infinite 2D sheet (C34H39N4O11Si).
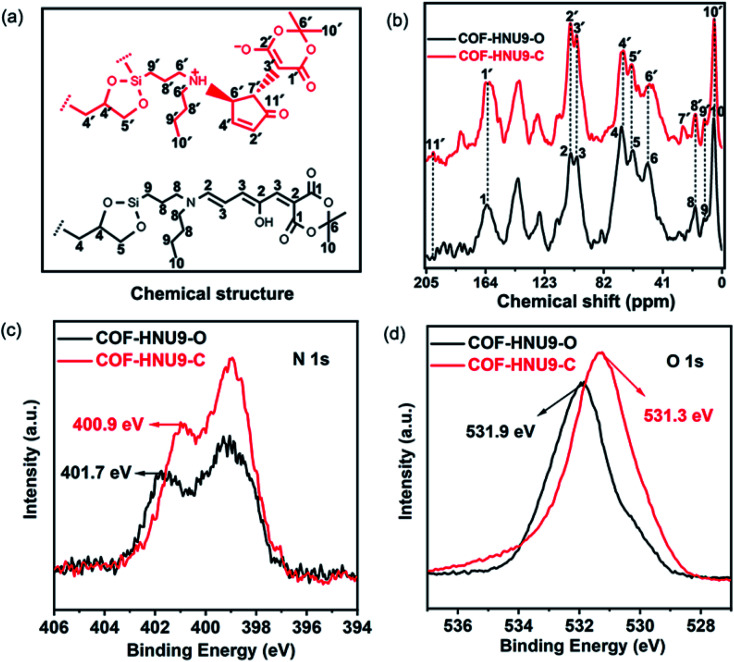
Fig. 3. (a) The structure and carbon atom numbering of DASA groups in the as-prepared COF-HNU9-O and COF-HNU9-C. (b) The 13C CP MAS solid state NMR spectra of COF-HNU9-O and COF-HNU9-C. (c and d) Comparison of the XPS survey of N 1s spectra (c) and O 1s spectra (d) of COF-HNU9 before and after visible light irradiation.
The isomerization of the DASA groups in COF-HNU9 was investigated by various characterization techniques. As shown in Fig. 3b, two new peaks were observed at about 27 ppm and 203 ppm in the solid state 13C NMR spectra of COF-HNU9-O compared to that of COF-HNU9-C, which correspond to C7′ and C11′ of the five-membered ring of the closed DASA groups. Meanwhile, the peak at about 50 ppm corresponding to the broadening of the C6′ peak was also observed because of the influence of the NH+ cation. The appearance of these new peaks and peak broadening indicates that structural transformation happened from COF-HNU9-O into COF-HNU9-C after visible light irradiation (Fig. 3a). In the XPS spectrum before and after visible light irradiation (Fig. 3c and d), two N spectra and two O spectra were used directly without fitting to compare the structural changes. It is clearly noted that after visible light irradiation on the COF-HNU9, the binding energy of N 1s and O 1s significantly shifted from 401.7 and 531.9 eV to 400.9 and 531.3 eV, respectively (Fig. 3c and d). This result indicates that DASA groups in COF-HNU9 were isomerized after visible light irradiation. In order to explore the changes in atom types before and after visible light irradiation, the core-level C 1s, N 1s and O 1s XPS spectra were deconvoluted into different peaks, respectively (Fig. S9†). The peaks at 400.3 eV (N 1s) and 532.5 eV (O 1s) can be assigned to C–N and C C–OH (Fig. S9c and e†) in the structure of COF-HNU9-O.29 After irradiation with visible light, they shifted to higher binding energies of 401.8 eV (N 1s) and 533.0 eV (O 1s), which correspond to C–N+ and C–O− (Fig. S9d and f†), respectively.30 These results were attributed to the photoisomerization of the DASA groups in the COF-HNU9-C pores. Furthermore, COF-HNU9 showed different thermal decomposition temperatures of 200 and 120 °C before and after visible light irradiation (Fig. S4†). This result suggested that the open form was more thermally stable than the closed state, which probably resulted from the large conjugation and strong π–π interactions in the open structure of DASA groups. In addition, the loading value of the DASA moieties on COF-HNU9 was calculated to be 0.44 ± 0.04 mmol g−1 from the thermogravimetric analysis results (Fig. S4†).
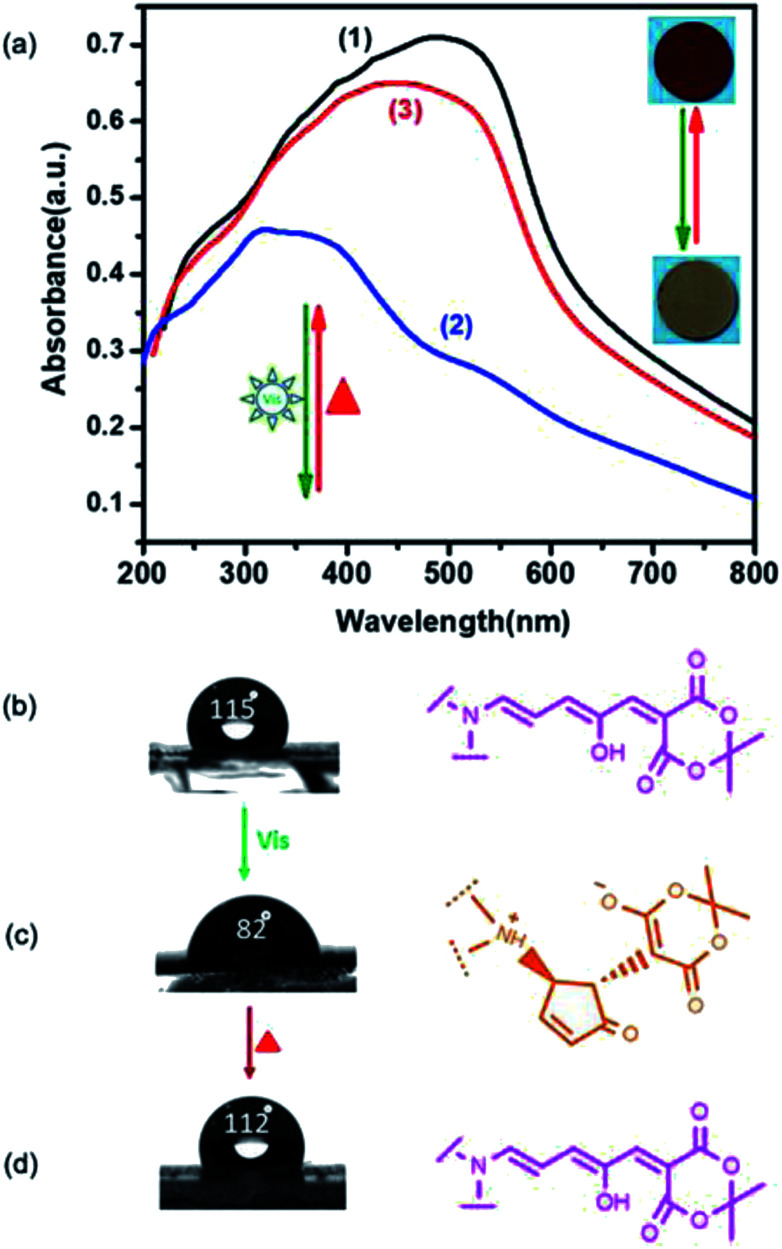
The isomerization was also studied by using UV-vis spectroscopy31 and the results are presented in Fig. 4a. The intensity of the characteristic absorption band of COF-HNU9-O at 350–600 nm significantly decreased (see curves 1 and 2) under irradiation with 515 nm visible light, but recovered almost to the original intensity by heating (see curve 3). The conversion yield was found to be 65.3% (Fig. S10†) and 85.1% (Fig. S11†), respectively, after the isomerization equilibrium was achieved. This open–closed–open isomerization was switchable for many consecutive cycles without obvious photodegradation (Fig. S12†). In order to better illustrate the reversible isomerization of the DASA groups in COF-HNU9, we synthesized a small molecule DASA from (3-butylaminopropyl) trimethoxysilane and activated furans according to the procedures reported in the literature26a (see details in the ESI†). The photoisomerization of the molecules was monitored by UV/vis spectroscopy in ethyl acetate (Fig. S13†). It was found that the intensity of the absorption band centered at 538 nm decreased continuously upon irradiation with visible light. This change indicated that the small molecule DASA was significantly transformed into its colourless cyclopentenone isomer. The resulting solution was subsequently kept in the dark at room temperature. The absorption band almost went back to the original intensity due to the thermal reversion to the colored triene.32 These results were well consistent with that of COF-HNU9.
Fig. 4. (a) UV-vis spectrum change for the isomerization of COF-HNU9-O (1) to COF-HNU9-C (2) upon irradiation with a 515 nm light and for the recovery of the UV-vis spectra of COF-HNU9-O upon mild heating at 40 °C in the dark under 32% RH (3); (b–d) contact angle of a water droplet on a COF-HNU9-coated glass surface before and after visible light-irradiation and reversed by heating (corresponding to (1)–(3) in (a)).
The visible light and heat triggered reversible isomerization of the DASA groups in COF-HNU9 was further supported by water contact angle measurements on a glass surface coated initially with the freshly prepared COF-HNU9. The surface coated with the open COF-HNU9-O was found to be hydrophobic in nature with a contact angle of 115 ± 1.0° (Fig. 4b). Then the open COF-HNU9-O was isomerized to the closed COF-HNU9-C after visible light irradiation, and the contact angle decreased to 82 ± 1.0° (Fig. 4c). Then, the contact angle was restored to 112 ± 1.0° by mild heating at 40 °C in the dark under 32% RH (Fig. 4d and S14†), which was in good agreement with UV-vis results. The water vapor adsorption isotherm for COF-HNU9 was also measured at 25 °C before and after irradiation with visible light. It was found that water adsorption capacity increased from 7.83 to 10.55 mmol g−1 under 97% RH, confirming that the hydrophilicity of COF-HNU9 pores was enhanced after visible light irradiation (Fig. S15†).
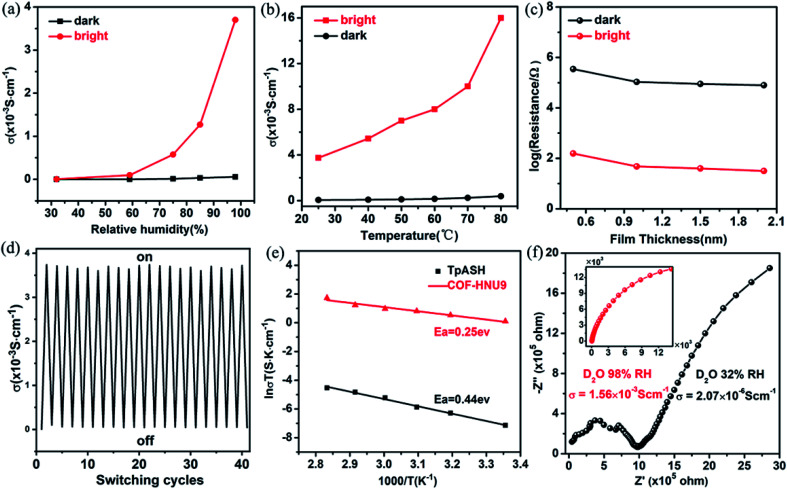
We evaluated the proton conductivity of COF-HNU9 by AC impedance spectroscopy using the compacted films with a thickness of 0.5 to 2 mm after saturating with water vapor for more than 3 h at different RH. As shown by our Nyquist plots (Fig. S16†), the proton conductivity of the COF-HNU9 film in the dark (COF-HNU9-O) was 1.86 × 10−6 under 32% RH and 5.62 × 10−5 S cm−1 under 98% RH at 25 °C, which shows that increasing RH facilitates proton conductivity, but the hydrophobic framework of COF-HNU9-O cannot provide high conductivity even at a high RH (Fig. 5a). However, after visible light irradiation, the proton conductivity of COF-HNU9-C exhibited a significant increase with increasing RH and reached 3.74 × 10−3 S cm−1 at 98% RH and 25 °C (Fig. S16†), which is about 1000 times conductivity increment compared to the value (3.50 × 10−6 S cm−1) of DASA-free TpASH under the same conditions. Therefore, both the isomerization of the grafted DASA groups and the high RH are of great importance in the significant enhancement of proton conduction.
Fig. 5. (a) Proton conductivity of COF-HNU9 film with increasing RH at 25 °C. (b) Proton conductivity of COF-HNU9 film at 98% RH with the increase of temperature. (c) Resistance of the open and closed states of COF-HNU9 with different film thicknesses (0.5, 1, 1.5 and 2 mm) before and after irradiation with visible light. (d) Reversible proton conductivity between 3.74 × 10−3 S cm−1 and 5.62 × 10−5 S cm−1 of COF-HNU9 film by turning on/off light illumination at 25 °C and 98% RH. (e) Arrhenius plots of the TpASH and COF-HNU9 films under visible light irradiation at 98% RH. (f) Nyquist plots of COF-HNU9 film at 25 °C under 32% and 98% RH using D2O.
In Fig. 5b, we studied the proton conductivity of COF-HNU9 film before and after irradiation with visible light at 98% RH in the temperature range from 25 °C to 80 °C. It can be seen that the proton conductivity under visible light significantly increased from 3.74 × 10−3 to 0.02 S cm−1 when the temperature rose to 80 °C, showing that in addition to visible light irradiation and high RH, temperature was also an important factor that affected proton conductivity. Conventionally, AC impedance is composed of ohmic resistance, proton-transfer resistance and proton-diffusion resistance under ideal conditions (Scheme S4†).33 Theoretically, visible light irradiation and high RH are greatly beneficial to the proton transport and diffusion, but to a different extent. This has been confirmed by our experimental Nyquist plots (Fig. S16†). Upon irradiation of visible light or increase of RH from 32% to 98%, the ohmic resistance and proton-transfer resistance became small and even disappeared at 25 °C (Fig. S16†). However, the proton-diffusion resistance still remained although its value was also obviously decreased. This indicates that the proton-diffusion resistance was mainly responsible for proton conduction in this case. On the other hand, the preferable crystal structure of the AA eclipsed stacking mode of COF-HNU9 resulted in regular open channels; as a result, proton conduction happened conveniently not only in the regular interlayers of parallel crystal planes, but also in the channels of COF-HNU9. Thus, the effect of thicknesses (0.5–2 mm) of the films on the diffusion resistance of samples was greatly reduced and even negligible, as shown in Fig. 5c. This type of behavior was similarly observed by Yu and co-workers.25b
We determined the equilibrium time of conductivity for the transformation of DASA units from the open to the closed state in COF-HNU9 upon visible light irradiation at 80 °C and 98% RH. It was found that the proton conductivity rapidly increased in the first 5 hours of irradiation, and then became almost constant when the transformation reached the photostationary state (Fig. S17†). We also evaluated proton transport number in the temperature range of 25 to 80 °C (see details in the ESI†),34 and found that it was in the range of 0.82 to 0.98 (Fig. S18†), indicating that COF-HNU9 is an excellent proton conductor. In addition, the reversible switching of proton conductivity was examined at 25 °C and 98% RH. Unless otherwise specified, it takes about 120 minutes from the open to closed isomers, and 180 minutes for its reverse, which is close to that of some azobenzene decorated COFs reported in the literature by using ultraviolet light as a trigger.25d It was shown that no significant decrease of the proton conductivity was observed even after 20 cycles (Fig. 5d), exhibiting excellent reversibility and recyclability.
Next, we analyzed the quantitative relationship between conductivity and temperature under visible light irradiation at 98% RH. As shown in Fig. 5e, the proton conduction of DASA-free TpASH and COF-HNU9 films follows an Arrhenius-like behavior. The activation energy (Ea) of TpASH film was 0.44 eV, which was close to the value for the Grotthuss mechanism (≤0.4 eV), suggesting that the proton conduction in TpASH film follows the proton hopping mechanism.35 For the COF-HNU9 film, the activation energy decreased to 0.25 eV and was much lower than 0.4 eV, which was also certainly in the range of a Grotthuss-type mechanism. Considering the hydrophilic property of COF-HNU9-C, many water molecules may be involved in the formation of abundant hydrogen-bond networks within the COF-HNU9 framework after visible light irradiation. This feature explains the unusually high proton conductivity observed above at visible light irradiation and high RH. We also studied the proton conductivity of the COF-HNU9 film in the environment of D2O vapor (Fig. 5f). In this case, the proton conductivity of COF-HNU9 increased from 2.07 × 10−6 to 1.56 × 10−3 S cm−1 at 25 °C when the RH was changed from 32% to 98%, which was nearly half smaller than that in a water vapor atmosphere. This result was ascribed to the heavier isotopic effect of D2O, where the mass of D is twice that of the H atom. As a result, the transport speed of the charge carrier was decreased upon replacing the proton with a deuteron.
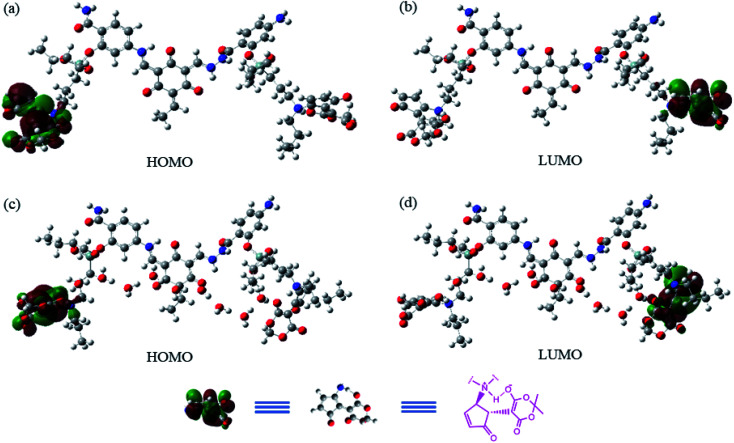
In order to investigate the charge transport through the framework, the models with/without water-assisted hydrogen bonds were used to calculate the energy of molecular orbitals (see details in the ESI†). To reduce the computational expense, a dimer structure fragment was built to describe the framework of COF-HNU9 (Fig. 6). It was shown that in the non-water system (Fig. 6a and b), the HOMO and LUMO were distributed in the two DASA groups, and the charge transport was very difficult due to the large energy difference between the HOMO and LUMO orbitals (about 2.58 eV). In the water-assisted system (Fig. 6c and d), although the HOMO and LUMO distribution was similar with the non-water system, the energy difference of the orbitals was only 1.75 eV, which was significantly less than that in the non-water system. For better illustration of the intermolecular hydrogen bonding shown in Fig. 6, we also modelled the intermolecular hydrogen bonding between water molecules and the small DASA groups without the COF-HNU9 framework, and calculated the energy of molecular orbitals (Fig. S19†). It was found that the energy difference between the HOMO and LUMO orbitals was 2.69 in the non-water and 2.49 eV in water environment, which was well consistent with the result shown in Fig. 6. It is known that water molecules can form hydrogen bonds and are conducive to the proton or electron transport, so the energy difference was declined in the presence of water vapor. Combined with the experimental results discussed above, these calculation findings suggest that the proton passage in the COF-HNU9 pores was in the way of proton hopping through the abundant hydrogen-bond networks between water vapor and the closed zwitterionic form of DASA groups inside the COF-HNU9 framework. Thus, protons can hop through the cavities easily under high RH, while it is almost impossible in a non-water environment.
Fig. 6. Frontier molecular orbitals of the structural fragments of COF-HNU9 in non-water (a and b) and water environments (c and d) (C, gray; N, blue; O, red; H, white). A dimer structure fragment is built to describe the COF-HNU9 framework. All geometric structure optimizations were performed at the B3LYP/6-311+G (d, p) level. The calculated energy for HOMO in non-water and water environment was −5.62 and −5.32 eV, and that for the corresponding LUMO was −3.04 and −3.57 eV, respectively.
As a proof of concept, a common electric circuit with LED was assembled to demonstrate the proton conduction change (Fig. S20 and S21†). Different from the results reported in the recent literature,20 we built the electric circuit with LED without any amplifier because the current flowing through the circuit was high enough to illuminate the LED lamp under consecutive visible light irradiation. It was found that under 98% RH, the LED lamp was illuminated under the irradiation of visible light at 80 °C, while it was switched off in the dark at 40 °C and 32% RH. This switch on and off process could be operated many times which makes it possible to apply this device under convenient conditions. The key to this photo-controlled process is the remarkable variation in the proton conduction under visible light and in the dark. Before visible light illumination, the film was stored in the dark and existed in its open neutral state (COF-HNU9-O), and the resistance was as high as 1.06 × 107 Ω. The current through the lamp was much smaller than the minimum current required to light up the lamp (6 mA). However, after the film was illuminated by visible light at 98% RH and 80 °C, the current through the LED lamp could reach 8 mA without any current amplifier, which was large enough to light up the lamp.
Experimental methods
Synthetic procedures
Synthesis of COF-Glc
COF-Glc was synthesized by the procedures reported in the literature.36 Briefly, 30 mg of the purified TpASH and 1.5 mL of triethylamine were dispersed in 10 mL of absolute ethanol, then sonicated for about 5 min in a water bath. Then 400 μL of glycidol (Glc) was added to the mixture and reacted for 6 h at 80 °C. After the mixture was cooled down, the precipitate was separated, washed with EtOH and dried at 60 °C for 12 h to afford the yellow target COF-Glc.
Synthesis of COF-BAPTMS
COF-BAPTMS was synthesized by following a reported procedure,37,38 where 50 mg of the as-prepared COF-Glc and 500 μL of (3-butylaminopropyl) trimethoxysilane (BAPTMS) were dispersed in 25 mL of toluene in a N2 atmosphere. The mixture was refluxed overnight in a N2 atmosphere, and the product was separated and washed with toluene and EtOH, respectively. Finally, a yellow product was obtained after being dried at 60 °C overnight.
Synthesis of COF-HNU9
50 mg of the resulting COF-BAPTMS was dispersed in 7 mL of tetrahydrofuran and sonicated for about 5 min in a water bath. Then, to this solution was added 0.6 gram of 5-(furan-2-ylmethylene)-2,2-dimethyl-1,3-dioxane-4,6-dione. The mixture was stirred at room temperature for 20 min and then in ice-water for 10 min. The product was filtered, washed with cold diethyl ether and EtOH, and the brown-colored DASA-modified COF-HNU9 was obtained after being dried at 80 °C under vacuum.
AC impedance measurements
The conductivity data were determined by impedance spectroscopy using an Autolab CHI600E system. The as-synthesized powder was mechanically pressed into solid pellets, and the measurements were performed by using stainless steel sheets as electrodes. The amplitude of the AC impedance spectrum was 10 mV and the frequency change was in the range of 1 Hz to 1 MHz.
Conclusions
In summary, we grafted photo-switchable DASA groups into the channels of the TpASH framework to create the first light/heat responsive COF-HNU9 film for highly efficient and switchable proton conductivity. It was found that the DASA groups in the nanopores of COF-HNU9 underwent reversible isomerization from the open neutral isomer to the closed zwitterionic form under the irradiation of visible light at room temperature and reversed back with heating at 40 °C in the dark. The closed zwitterionic isomers significantly improve the proton conductivity through plenty of hydrogen bonds with the water vapor inside the nanochannels. Under visible light illumination, the proton conductivity of COF-HNU9 film was up to 0.02 S cm−1 at 80 °C and 98% RH. Furthermore, this photo-responsive COF allows remarkable photo-modulation of the proton conductivity, and a three orders of magnitude increase was observed. The studies from experimental activation energies and DFT calculations suggest that the proton conduction follows the proton hopping mechanism through the abundant hydrogen bonds between water vapor and the closed zwitterionic form of DASA groups inside the COF-HNU9 cavities. Furthermore, as an example of the applications of the above findings, an optical control device was assembled by using a light-switching proton conductive COF-HNU9 film to remotely illuminate an LED lamp and then switch it off without any current amplifier. The findings reported here pave the way toward the development of novel photo-switchable conducting materials based on smart COFs in remote-controllable fuel cells, chemical sensors and other advanced switchable devices. Finally, it should be pointed out that the COF used in this work is asymmetric. In future designs, we will examine whether the asymmetry affects the proton conductivity before and after visible light irradiation.
Data availability
All the data have been included in the ESI.†
Author contributions
J. W. and G. Z. designed the research and proposed the idea. Y. C. conducted the synthesis of the COF films and the performance tests. J. Q. participated in the synthesis. X. Z. performed the DFT calculations. H. W., W. Y., Z. L. and Q. X. participated in the characterizations of BET, FT-IR and XRD of the COF films and the discussion of the manuscript. Y. C. and J. Q. wrote the manuscript. J. W. and G. Z. revised and finalized the manuscript.
Conflicts of interest
There are no conflicts to declare.
Supplementary Material
Acknowledgments
This work is supported by the National Natural Science Foundation of China (U1704251, 22073024, 21803048), the National Key Research and Development Program of China (2017YFA0403101), the 111 Project (D17007), and the Program for Universities of Henan Province Science & Technology Innovation Talents (21HASTIT003).
Electronic supplementary information (ESI) available: Materials and instrumentation methods, synthetic procedures, Nyquist plots under ideal conditions, FT-IR spectra, SEM and TEM images, BET plots, TGA analysis, PXRD patterns, XPS spectra, UV-vis spectra, stability test, water vapor sorption isotherms and proton conductivity measurement. See https://doi.org/10.1039/d2sc02100e
Notes and references
- (a) Wang Y. Chen K. S. Mishler J. Cho S. C. Adroher X. C. Appl. Energy. 2011;88:981–1007. doi: 10.1016/j.apenergy.2010.09.030. [DOI] [Google Scholar]; (b) Hickner M. A. Ghassemi H. Kim Y. S. Einsla B. R. McGrath J. E. Chem. Rev. 2004;104:4587–4612. doi: 10.1021/cr020711a. [DOI] [PubMed] [Google Scholar]; (c) Garland N. L. Kopasz J. P. J. Power Sources. 2007;172:94–99. doi: 10.1016/j.jpowsour.2007.01.025. [DOI] [Google Scholar]
- (a) Nataraj S. K. Wang C.-H. Huang H.-C. Du H.-Y. Wang S.-F. Chen Y.-C. Chen L.-C. Chen K.-H. ChemSusChem. 2012;5:392–395. doi: 10.1002/cssc.201100366. [DOI] [PubMed] [Google Scholar]; (b) Trigg E. B. Gaines T. W. Maréchal M. Moed D. E. Rannou P. Wagener K. B. Stevens M. J. Winey K. I. Nat. Mater. 2018;17:725–731. doi: 10.1038/s41563-018-0097-2. [DOI] [PubMed] [Google Scholar]
- (a) Liu R.-L. Liu Y.-R. Yu S.-H. Yang C.-L. Li Z.-F. Li G. ACS Appl. Mater. Interfaces. 2019;11:1713–1722. doi: 10.1021/acsami.8b18891. [DOI] [PubMed] [Google Scholar]; (b) Dou W. Zhu L. Q. Jiang J. Wan Q. Appl. Phys. Lett. 2013;102:093509. doi: 10.1063/1.4794905. [DOI] [Google Scholar]; (c) Musset B. Smith S. M. E. Rajan S. Morgan D. Cherny V. V. DeCoursey T. E. Nature. 2011;480:273–277. doi: 10.1038/nature10557. [DOI] [PMC free article] [PubMed] [Google Scholar]; (d) Yoon M. Suh K. Natarajan S. Kim K. Angew. Chem., Int. Ed. 2013;52:2688–2700. doi: 10.1002/anie.201206410. [DOI] [PubMed] [Google Scholar]
- (a) Sarango-Ramírez M. K. Lim D.-W. Kolokolov D. I. Khudozhitkov A. E. Stepanov A. G. Kitagawa H. J. Am. Chem. Soc. 2020;142:6861–6865. doi: 10.1021/jacs.0c00303. [DOI] [PubMed] [Google Scholar]; (b) Guo Z.-C. Shi Z.-Q. Wang X.-Y. Li Z.-F. Li G. Coord. Chem. Rev. 2020;422:213465. doi: 10.1016/j.ccr.2020.213465. [DOI] [Google Scholar]
- (a) Chand S. Elahi S. M. Pal A. Das M. C. Chem.–Eur. J. 2019;25:6259–6269. doi: 10.1002/chem.201806126. [DOI] [PubMed] [Google Scholar]; (b) Jiang Y. Heinke L. Langmuir. 2021;37:2–15. doi: 10.1021/acs.langmuir.0c02859. [DOI] [PubMed] [Google Scholar]
- Meng X. Wang H.-N. Song S.-Y. Zhang H.-J. Chem. Soc. Rev. 2017;46:464–480. doi: 10.1039/C6CS00528D. [DOI] [PubMed] [Google Scholar]
- Lim D.-W. Kitagawa H. Chem. Rev. 2020;120:8416–8467. doi: 10.1021/acs.chemrev.9b00842. [DOI] [PubMed] [Google Scholar]
- Liu R.-l. Wang D.-Y. Shi J.-R. Li G. Coord. Chem. Rev. 2021;431:213747. doi: 10.1016/j.ccr.2020.213747. [DOI] [Google Scholar]
- (a) Bian S. Zhang K. Wang Y. Liu Z. Wang G. Jiang X. Pan Y. Xu B. Huang G. Zhang G. ACS Appl. Energy Mater. 2022;5:1298–1304. doi: 10.1021/acsaem.1c03721. [DOI] [Google Scholar]; (b) Yang J. Xie C. Yang Q. Wang S. Gao Y. Ji J. Du Z. Kang Z. Wang R. Sun D. Chem. Commun. 2022;58:1131–1134. doi: 10.1039/D1CC06010D. [DOI] [PubMed] [Google Scholar]
- (a) Zhai Y. Liu G. Jin F. Zhang Y. Gong X. Miao Z. Li J. Zhang M. Cui Y. Zhang L. Liu Y. Zhang H. Zhao Y. Zeng Y. Angew. Chem., Int. Ed. 2019;58:17679–17683. doi: 10.1002/anie.201911231. [DOI] [PubMed] [Google Scholar]; (b) Liu Y. Diercks C. S. Ma Y. Lyu H. Zhu C. Alshmimri S. A. Alshihri S. Yaghi O. M. J. Am. Chem. Soc. 2019;141:677–683. doi: 10.1021/jacs.8b12177. [DOI] [PubMed] [Google Scholar]; (c) Geng K. He T. Liu R. Dalapati S. Tan K. T. Li Z. Tao S. Gong Y. Jiang Q. Jiang D. Chem. Rev. 2020;120:8814–8933. doi: 10.1021/acs.chemrev.9b00550. [DOI] [PubMed] [Google Scholar]; (d) Dai L. Huang K. Xia Y. Xu Z. Green Energy Environ. 2021;6:193–211. doi: 10.1016/j.gee.2020.09.015. [DOI] [Google Scholar]
- Ranjeesh K. C. Illathvalappil R. Veer S. D. Peter J. Wakchaure V. C. Goudappagouda Raj K. V. Kurungot S. Babu S. S. J. Am. Chem. Soc. 2019;141:14950–14954. doi: 10.1021/jacs.9b06080. [DOI] [PubMed] [Google Scholar]
- Peng Y. Xu G. Hu Z. Cheng Y. Chi C. Yuan D. Cheng H. Zhao D. ACS Appl. Mater. Interfaces. 2016;8:18505–18512. doi: 10.1021/acsami.6b06189. [DOI] [PubMed] [Google Scholar]
- Chandra S. Kundu T. Dey K. Addicoat M. Heine T. Banerjee R. Chem. Mater. 2016;28:1489–1494. doi: 10.1021/acs.chemmater.5b04947. [DOI] [Google Scholar]
- Cao L. Wu H. Cao Y. Fan C. Zhao R. He X. Yang P. Shi B. You X. Jiang Z. Adv. Mater. 2020;32:2005565. doi: 10.1002/adma.202005565. [DOI] [PubMed] [Google Scholar]
- (a) Chandra S. Kundu T. Kandambeth S. BabaRao R. Marathe Y. Kunjir S. M. Banerjee R. J. Am. Chem. Soc. 2014;136:6570–6573. doi: 10.1021/ja502212v. [DOI] [PubMed] [Google Scholar]; (b) Meng Z. Aykanat A. Mirica K. A. Chem. Mater. 2019;31:819–825. doi: 10.1021/acs.chemmater.8b03897. [DOI] [Google Scholar]; (c) Yang Y. He X. Zhang P. Andaloussi Y. H. Zhang H. Jiang Z. Chen Y. Ma S. Cheng P. Zhang Z. Angew. Chem., Int. Ed. 2020;59:3678–3684. doi: 10.1002/anie.201913802. [DOI] [PubMed] [Google Scholar]
- (a) Ma H. Liu B. Li B. Zhang L. Li Y.-G. Tan H.-Q. Zang H.-Y. Zhu G. J. Am. Chem. Soc. 2016;138:5897–5903. doi: 10.1021/jacs.5b13490. [DOI] [PubMed] [Google Scholar]; (b) Liu L. Yin L. Cheng D. Zhao S. Zang H.-Y. Zhang N. Zhu G. Angew. Chem., Int. Ed. 2021;60:14875–14880. doi: 10.1002/anie.202104106. [DOI] [PubMed] [Google Scholar]
- (a) Tao S. Zhai L. Wonanke A. D. D. Addicoat M. A. Jiang Q. Jiang D. Nat. Commun. 2020;11:1981. doi: 10.1038/s41467-020-15918-1. [DOI] [PMC free article] [PubMed] [Google Scholar]; (b) Li J. Wang J. Wu Z. Tao S. Jiang D. Angew. Chem., Int. Ed. 2021;60:12918–12923. doi: 10.1002/anie.202101400. [DOI] [PubMed] [Google Scholar]; (c) Wu X. Hong Y.-L. Xu B. Nishiyama Y. Jiang W. Zhu J. Zhang G. Kitagawa S. Horike S. J. Am. Chem. Soc. 2020;142:14357–14364. doi: 10.1021/jacs.0c06474. [DOI] [PubMed] [Google Scholar]
- (a) Sasmal H. S. Aiyappa H. B. Bhange S. N. Karak S. Halder A. Kurungot S. Banerjee R. Angew. Chem., Int. Ed. 2018;57:10894–10898. doi: 10.1002/anie.201804753. [DOI] [PubMed] [Google Scholar]; (b) Li Y. Wu H. Yin Y. Cao L. He X. Shi B. Li J. Xu M. Jiang Z. J. Membr. Sci. 2018;568:1–9. doi: 10.1016/j.memsci.2018.09.050. [DOI] [Google Scholar]
- Li S. Liu Y. Li L. Liu C. Li J. Ashraf S. Li P. Wang B. ACS Appl. Mater. Interfaces. 2020;12:22910–22916. doi: 10.1021/acsami.0c04002. [DOI] [PubMed] [Google Scholar]
- Liang H.-Q. Guo Y. Shi Y. Peng X. Liang B. Chen B. Angew. Chem., Int. Ed. 2020;59:7732–7737. doi: 10.1002/anie.202002389. [DOI] [PubMed] [Google Scholar]
- (a) Nagarkar S. S. Horike S. Itakura T. Ouay B. L. Demessence A. Tsujimoto M. Kitagawa S. Angew. Chem., Int. Ed. 2017;56:4976–4981. doi: 10.1002/anie.201700962. [DOI] [PubMed] [Google Scholar]; (b) Müller K. Helfferich J. Zhao F. Verma R. Kanj A. B. Meded V. Bléger D. Wenzel W. Heinke L. Adv. Mater. 2018;30:1706551. doi: 10.1002/adma.201706551. [DOI] [PubMed] [Google Scholar]; (c) Kanj A. B. Chandresh A. Gerwien A. Grosjean S. Bräse S. Wang Y. Dube H. Heinke L. Chem. Sci. 2020;11:1404–1410. doi: 10.1039/C9SC04926F. [DOI] [Google Scholar]
- (a) Huang N. Ding X. Kim J. Ihee H. Jiang D. Angew. Chem., Int. Ed. 2015;54:8704–8707. doi: 10.1002/anie.201503902. [DOI] [PMC free article] [PubMed] [Google Scholar]; (b) She P. Qin Y. Wang X. Zhang Q. Adv. Mater. 2021:2101175. doi: 10.1002/adma.202101175. [DOI] [PubMed] [Google Scholar]; (c) Jhulki S. Evans A. M. Hao X.-L. Cooper M. W. Feriante C. H. Leisen J. Li H. Lam D. Hersam M. C. Barlow S. Brédas J.-L. Dichtel W. R. Marder S. R. J. Am. Chem. Soc. 2020;142:783–791. doi: 10.1021/jacs.9b08628. [DOI] [PubMed] [Google Scholar]
- Ou R. Zhang H. Truong V. X. Zhang L. Hegab H. M. Han L. Hou J. Zhang X. Deletic A. Jiang L. Simon G. P. Wang H. Nat. Sustain. 2020;3:1052–1058. doi: 10.1038/s41893-020-0590-x. [DOI] [Google Scholar]
- (a) Chen Y. Li Z. Wang H. Pei Y. Shi Y. Wang J. Langmuir. 2018;34:2784–2790. doi: 10.1021/acs.langmuir.7b03822. [DOI] [PubMed] [Google Scholar]; (b) Li Z. Feng Y. Liu X. Wang H. Pei Y. Nimal Gunaratne H. Q. Wang J. ACS Sustainable Chem. Eng. 2020;8:15327–15335. doi: 10.1021/acssuschemeng.0c05703. [DOI] [Google Scholar]
- (a) Dolgopolova E. A. Galitskiy V. A. Martin C. R. Gregory H. N. Yarbrough B. J. Rice A. M. Berseneva A. A. Ejegbavwo O. A. Stephenson K. S. Kittikhunnatham P. Karakalos S. G. Smith M. D. Greytak A. B. Garashchuk S. Shustova N. B. J. Am. Chem. Soc. 2019;141:5350–5358. doi: 10.1021/jacs.8b13853. [DOI] [PubMed] [Google Scholar]; (b) Yu F. Liu W. Li B. Tian D. Zuo J.-L. Zhang Q. Angew. Chem., Int. Ed. 2019;58:16101–16104. doi: 10.1002/anie.201909613. [DOI] [PubMed] [Google Scholar]; (c) Griffiths K. Halcovitch N. R. Griffin J. M. Chem. Sci. 2022;13:3014–3019. doi: 10.1039/D2SC00632D. [DOI] [PMC free article] [PubMed] [Google Scholar]; (d) Liu C. Zhang W. Zeng Q. Lei S. Chem.–Eur. J. 2016;22:6768–6773. doi: 10.1002/chem.201601199. [DOI] [PubMed] [Google Scholar]
- (a) Helmy S. Leibfarth F. A. Oh S. Poelma J. E. Hawker C. J. Read de Alaniz J. J. Am. Chem. Soc. 2014;136:8169–8172. doi: 10.1021/ja503016b. [DOI] [PubMed] [Google Scholar]; (b) Senthilkumar T. Zhou L. Gu Q. Liu L. Lv F. Wang S. Angew. Chem., Int. Ed. 2018;57:13114–13119. doi: 10.1002/anie.201807158. [DOI] [PubMed] [Google Scholar]
- Mitra S. Sasmal H. S. Kundu T. Kandambeth S. Illath K. Díaz Díaz D. Banerjee R. J. Am. Chem. Soc. 2017;139:4513–4520. doi: 10.1021/jacs.7b00925. [DOI] [PubMed] [Google Scholar]
- Mallo N. Foley E. D. Iranmanesh H. Kennedy A. D. W. Luis E. T. Ho J. Harper J. B. Beves J. E. Chem. Sci. 2018;9:8242–8252. doi: 10.1039/C8SC03218A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (a) Cui W.-R. Zhang C.-R. Jiang W. Liang R.-P. Qiu J.-D. ACS Appl. Nano Mater. 2019;2:5342–5349. doi: 10.1021/acsanm.9b01366. [DOI] [Google Scholar]; (b) Khatuna R. Biswasa S. Biswasa I. H. Riyajuddinb S. Haquea N. Ghoshb K. Islama S. M. J. CO2 Util. 2020;40:101180. doi: 10.1016/j.jcou.2020.101180. [DOI] [Google Scholar]
- Liu T.-T. Xu R. Yi J.-D. Liang J. Wang X.-S. Shi P.-C. Huang Y.-B. Cao R. ChemCatChem. 2018;10:2036–2040. doi: 10.1002/cctc.201800023. [DOI] [Google Scholar]
- Das G. Prakasam T. Addicoat M. A. Sharma S. K. Ravaux F. Mathew R. Baias M. Jagannathan R. Olson M. A. Trabolsi A. J. Am. Chem. Soc. 2019;141:19078–19087. doi: 10.1021/jacs.9b09643. [DOI] [PubMed] [Google Scholar]
- Helmy S. Oh S. Leibfarth F. A. Hawker C. J. Read de Alaniz J. J. Org. Chem. 2014;79:11316–11329. doi: 10.1021/jo502206g. [DOI] [PubMed] [Google Scholar]
- Farahani H. Wagiran R. Urban G. A. IEEE Sens. J. 2021;21:9657–9666. [Google Scholar]
- Zhang G. Smyth D. M. Solid State Ionics. 1995;82:153–160. doi: 10.1016/0167-2738(95)00199-8. [DOI] [Google Scholar]
- (a) Agmon N. Chem. Phys. Lett. 1995;244:456–462. doi: 10.1016/0009-2614(95)00905-J. [DOI] [Google Scholar]; (b) Kreuer K.-D. Chem. Mater. 1996;8:610–641. doi: 10.1021/cm950192a. [DOI] [Google Scholar]
- Chen J. Shum W. Tetrahedron Lett. 1995;36:2379–2380. doi: 10.1016/0040-4039(95)00282-H. [DOI] [Google Scholar]
- Mitra S. Subia B. Patra P. Chandra S. Debnath N. Das S. Banerjee R. Kundu S. C. Pramanik P. Goswami A. J. Mater. Chem. 2012;22:24145–24154. doi: 10.1039/C2JM35013K. [DOI] [Google Scholar]
- Guo Y. Wang H. He C. Qiu L. Cao X. Langmuir. 2009;25:4678–4684. doi: 10.1021/la803530h. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All the data have been included in the ESI.†