Abstract
Purpose
Management of adult soft tissue sarcomas entails a multidisciplinary approach with surgery and radiation therapy with or without chemotherapy. The use of preoperative irradiation has been well established, and although conventional fractionation involves daily treatments over the course of 5 weeks, higher doses per fraction may be beneficial due to the radiobiologic profile of sarcoma. In this study we report long-term oncologic outcomes from a single-institution, phase II study evaluating a 5-fraction hypofractionated course of preoperative radiation.
Methods and materials
Preoperative hypofractionated radiation therapy was administered to 35 Gy in 5 fractions every other day followed by resection 4 to 6 weeks later. If given, chemotherapy consisted of a doxorubicin-ifosfamide-based regimen delivered neoadjuvantly. The primary endpoint was local control. Additional survival and pathologic outcomes, including overall and distant metastasis-free survival, tumor, and treatment-related pathology, as well as acute and late toxicity were examined.
Results
Thirty-two patients were enrolled in this prospective, single-arm phase II trial. At a median follow-up of 36.4 months (range, 3-56), no patient developed a local recurrence, and the 3-year overall and distant metastasis-free survival was 82.2% and 69%, respectively. Major acute postoperative wound complications occurred in 25% of patients. Grade 2 and 3 fibrosis occurred in 21.7% and 13% of patients, respectively. The 2-year median and mean Musculoskeletal Tumor Society score for all patients was 28 and 27.4, respectively.
Conclusions
A condensed course of preoperative hypofractionated radiation therapy leads to excellent rates of local control and survival with acceptable toxicity profiles. Potential studies ideally with phase II or III randomized trials would help corroborate these findings and other preoperative hypofractionated results in soft tissue sarcomas.
Soft tissue sarcomas (STS) of the extremity and superficial trunk are rare tumors that comprise <1% of all adult malignancies.1 The management of STS of has evolved to incorporate a multimodality approach of radiation therapy and surgery with or without systemic therapy. Although preoperative radiation therapy has been shown to lead to an increase in the incidence of postoperative wound complications, it does offer several advantages, such as decreased radiation dose, decreased field size and improved long-term toxicity.2, 3, 4 Standard preoperative radiation occurs over approximately 5 weeks, followed by an additional 4- to 6-week break, yielding an overall management time of 9 to 11 weeks. The addition of neoadjuvant chemotherapy increases the overall treatment time to 18 to 20 weeks. Due to the prolonged treatment time, many patients who travel long distances may defer management at high-volume sarcoma centers that have expertise in the treatment of this rare disease.
The use of hypofractionated radiation therapy has been reported in several malignancies, such as prostate cancer, breast cancer, and melanoma.5, 6, 7, 8, 9, 10, 11, 12 Like these cancer types, sarcoma holds a lower α/β ratio and thus may respond to higher doses of radiation per fraction.13, 14, 15, 16 Hypofractionated preoperative radiation therapy in STS is beneficial, as it reduces time to surgery and increases patient convenience; however, finding a balance between disease control and toxicity is imperative.
Although there may be concern of acute and long-term morbidity, emerging data on the use of preoperative hypofractionation in STS have demonstrated toxicity rates on par with protracted, standard preoperative irradiation regimens. Although promising phase II data have been presented, primary endpoints, length of follow-up, and timing of surgery have varied (Tables 1 and 2).7,8,17, 18, 19, 20, 21, 22
Table 1.
Compilation of publications in hypofractionated preoperative radiation therapy in STS
| Author | No. | Dose/Fraction | Median Follow-up (mo) | RT to Surgery Time | Chemotherapy* |
|---|---|---|---|---|---|
| Kosela-Paterczyk et al | 272 | 25 Gy/5 fx | 35 | 3-7 d | Yes |
| Kalbasi et al | 52 | 30 Gy/5 fx | 29 | 2-6 wk | No |
| Kubicek et al | 14 | 35-40 Gy/5 fx | 9.3 | 4-8 wk | Yes |
| Temple et al | 42 | 30 Gy/10 fx | 72 | 4-6 wk | Yes |
| Parasi et al | 16 | 30 Gy/5 fx | 10.7 | 0-7 d | Yes (1 with stage IV) |
| MacDermed et al | 34 | 28 Gy/8 fx | 33.5 | 4-8 wk | Yes |
| Meyer et al | 16 | 28 Gy/8 fx | 26 | Not reported | Yes |
| Ryan et al | 25 | 28 Gy/8 fx | 24 | 4-5 wk | Yes |
| Pennington et al | 116 | 28Gy/8 fx | 71 | 2-3 wk | Yes |
| Current study | 32 | 35 Gy/5 fx | 36.4 | 4-6 wk | Yes |
Chemotherapy administered preoperatively or concurrent.
Abbreviation: fx = fractions.
Table 2.
Compilation of publications in hypofractionated preoperative radiation therapy in STS
| Author | R0 Resection | Wound Complication* | ≥G2 Fibrosis | Local Control @ y | Overall Survival @ y |
|---|---|---|---|---|---|
| Kosela-Paterczyk et al | 78.7% | 32.4% | 3.7% | 81% @ 3 y | 72% @ 5 y |
| Kalbasi et al | 82% | 32.0% | 11% | 94.3% @ 2 y | Not reported |
| Kubicek et al | 100% | 28.6% | 0% | 92.3% @ 1 y | Not reported |
| Temple et al | Not reported | 14.2% | Not reported | 97% @ 5 y | 79% @ 5 y |
| Parasi et al | 62.5% | 31.2% | 0% | 100% @ 1 y | Not reported |
| MacDermed et al | 100% | 17% requiring operation | 13.8% | 89% @ 5 y | 42.3% @ 5 y |
| Meyer et al | 94% | 38% | Not reported | 100% @ 2 y | 86% @ 2 y |
| Ryan et al | 88% | 20% | Not reported | 88% @ 2 y | 84% @ 2 y |
| Pennington et al | 93% | 10% | Not reported | 89% @ 3 y | 82% @ 3 y |
| Current study | 91% | 25% | 25% | 100%@ 3 y | 94.4% @ 3 y |
Wound complications per the CAN-NCIC-SR2 study.2
Abbreviation: G = grade.
Given the diversity in data, we aimed to increase the wealth of knowledge pertaining to preoperative hypofractionated radiation therapy in STS. In this phase II prospective study, we report long-term clinical outcomes and toxicity in patients receiving 35 Gy in 5 fractions administered every other day.
Materials and Methods
Patients
The phase II prospective trial for this study was reviewed and approved by the institutional review board, and all investigators completed training in both human research and patient privacy (NCTXXXX). The study was conducted in accordance with the Declaration of Helsinki and the International Conference on Harmonization Guideline for Good Clinical Practice.
Eligible participants were patients ≥18 years of age with American Joint Committee on Cancer, 8th edition stage I-III histologically confirmed STS of the extremity or trunk. All patients were deemed to be medically operable with a Karnofsky performance status of ≥60. Key exclusion criteria comprised patients with stage IV disease or simultaneously treated secondary malignancy, pathology other than sarcoma subtypes, or patients who had prior radiation therapy in the proposed treatment area. Participants were discussed in a multidisciplinary tumor board involving radiation and medical oncology, surgical and orthopedic oncology, pathology, and radiology. All enrolled patents provided written and informed consent.
Radiation therapy
Patients enrolled in the study received preoperative radiation therapy with or without neoadjuvant chemotherapy, followed by resection ideally 4 to 6 weeks postradiation. Concurrent chemotherapy was not allowed per protocol. Planning for radiation therapy included a computed tomography (CT) scan and magnetic resonance imaging (MRI) simulation with immobilization. Tumor volumes were designed according to Radiation Therapy Oncology Group 0630.23 CT imaging obtained at simulation was fused with the T1- and T2-weighted MRI sequences acquired at the time of MRI simulation. The gross tumor volume (GTV) was defined by the MRI fused sequences, and the clinical target volume (CTV) for intermediate- to high-grade tumors that measured over 8 cm included the GTV with a 3 cm longitudinal and 1.5 cm radial margin. Anatomic boundaries such as uninvolved bone and muscle compartment that were not abutting the GTV were respected.
For all other tumors the CTV included the GTV and suspicious edema with a 2 cm longitudinal margin and 1 cm radial margin in which the same anatomic boundaries were respected. A planning target volume (PTV) was created by expanding the CTV by 5 mm as daily image guided radiation therapy was performed. The PTV was truncated by 3 to 5 mm from the skin surface in deep tumors and if there was no concern of skin involvement.
Per protocol, at least 90% of the PTV received a prescription dose of 35 Gy in 5 fractions, which was delivered every other day with a minimum of 48 hours between treatments to allow for tissue recovery. The dose chosen was one that emulates the biologic effective dose of the standard dose and fractionation of 50 Gy in 25 fractions and was calculated on a presumed α/β = 5 for sarcomas.
Three-dimensional conformal or intensity modulated radiation therapy was used for each subject. Dosimetric constraints adjusted for a hypofractionated regimen were used (Table E1). No patient enrolled violated any dosimetric constraints. In addition, per protocol, all patients underwent image guided radiation therapy with either a cone beam kV or MV CT or CT on rails for each fraction.
Chemotherapy
Chemotherapy was recommended and administered neoadjuvantly in patients who were typically <70 years of age, with large (>5 cm), deep, and high-grade lesions and chemotherapy-sensitive histologies. Chemotherapy was a doxorubicin-ifosfamide-based regimen given for 1 to 3 cycles based on clinical response and tolerance.
Surgery
All patients underwent surgery at a single institution. Wide local resection was performed by fellowship-trained musculoskeletal oncologists grossly through normal tissue planes. Preservation of neurovascular structures was performed when possible. The goal of surgery was to achieve negative margins (R0). Vascular or reconstructive plastic surgeons were involved in cases that required vascular reconstruction, difficult wound closures, and free flap reconstructions.
Follow-up
After resection, patients were seen by the multidisciplinary team, including radiation, orthopedic, and medical oncology (the latter if they received chemotherapy). Patients were evaluated clinically postoperatively and for surveillance every 3 to 4 months for the first 2 years while on study. Off study after 2 years, patients were followed every 6 months up until year 5, then yearly thereafter. Imaging occurred at the time of follow-up and included a CT scan of the chest and imaging of the primary tumor with MRI. In patients with myxoid liposarcoma, a yearly MRI of the entire spine was also acquired.
Endpoints
The primary endpoint of this study was to assess the local control. Local recurrence was determined by physical examination and imaging. Secondary endpoints included the incidence of major acute wound complications and pathologic change post neoadjuvant therapies as delineated by a musculoskeletal pathologist, which included percent fibrosis, necrosis, and viable cells. In addition, distant metastasis-free survival (DMFS) and overall survival (OS), physician-reported function outcomes using the Musculoskeletal Tumor Society (MSTS) scale (scale 0-30), and grade 3 or greater fibrosis per the Common Terminology Criteria for Adverse Events version 4.0 (National Cancer Institute) were evaluated. Postoperative wound complications were defined according to the Canadian National Cancer Institute of Canada SR2 (CAN-NCIC-SR2) Multicenter Trial and were recorded if they occurred within 6 months after limb-salvage surgery.2 In general, cases that required reoperation, prolonged wound care, or antibiotics after resection were considered wound complications. Local control, OS, and DMFS were defined from the date of biopsy to the date of calculated endpoint. Wound complications were assessed from the time of surgery.
Pathologic analysis
Resection specimens were reviewed by fellowship-trained and board-certified bone and soft tissue pathologists. Percent viable tumor, necrosis, and fibrosis/hyalinization were scored per slide, and the overall percentages were calculated for the entire specimen so that the sum of the 3 components totaled 100%. Viable cells were quantified based on residual cells with similarity to the diagnostic biopsy material and often with radiation-associated changes, including pleomorphic cells with enlarged, hyperchromatic nuclei and abnormal mitotic figures.
Necrosis was quantified as a confluent area of dead and degenerating cells with karyorrhectic nuclear debris and polymorphonuclear neutrophil infiltrate. Fibrosis/hyalinization was quantified as hypocellular areas with dense, collagenous matrix and associated with fibroblasts.
Statistical analysis
Descriptive statistics were used to describe local failure, survival, toxicity, and postoperative wound complications. DMFS and OS were estimated using the Kaplan-Meier estimate of the survival function. Fisher exact or χ2 analysis and McNemar test were used for unpaired and paired categorical variables, respectively. All intervals were calculated from the date of biopsy. All analyses were performed with MedCalc version 19.8 (MedCalc Software Ltd.). For all analyses, the type I error was maintained at 0.05 and 2-sided tests were used.
Results
Patient, tumor and treatment characteristics
Between March 2016 and February 2020, 35 patients with localized STS of the extremity or trunk were enrolled on this phase II protocol, of which 32 were evaluable. Median follow-up was 36.4 months (range, 3-56) One patient died of a myocardial infarction at 3 months, and 2 patients were lost to follow-up. Median age was 63.5 years (range, 28-86; Table 3).
Table 3.
Patient, tumor and treatment characteristics
| All Patients | No Chemotherapy | Chemotherapy | |
|---|---|---|---|
| No. | 32 | 22 | 10 |
| Median age | 63.5 (range, 28-86) | 67.5 (range, 28-86) | 53.5 (range, 32-74) |
| Tumor size (median) | 9.5 cm | 7.8 cm | 12.1 cm |
| American Joint Committee on Cancer 8th ed. Stage | |||
| IB | 0 | 0 | 0 |
| II | 6 | 6 | 0 |
| IIIA | 11 | 8 | 3 |
| IIIB | 15 | 8 | 7 |
| Histology | |||
| Undifferentiated pleomorphic sarcoma | 10 | 6 | 4 |
| Leiomyosarcoma | 6 | 5 | 1 |
| Liposarcoma | 7 | 3 | 4 |
| Myxofibrosarcoma | 7 | 6 | 1 |
| Synovial sarcoma | 2 | 2 | 0 |
| Location | |||
| Trunk | 2 | 2 | 0 |
| Upper extremity | 5 | 4 | 1 |
| Lower extremity | 25 | 16 | 9 |
Most patients (22 of 32) had lower extremity tumors and the median tumor size was 9.5 cm (range, 2.8-22). Neoadjuvant doxorubicin and ifosfamide were administered in 31.5% of subjects (10 of 32; Table 3). All but 1 patient received 3 cycles of chemotherapy, with that participant stopping short secondary to elevated liver function tests. No patients received adjuvant chemotherapy.
All enrolled patients received preoperative radiation to 35 Gy in 5 fractions delivered every other day. The median time from initiation to completion of radiation was 10 days (range, 9-13).
Surgical outcomes
Thirty-one patients underwent wide local excision after preoperative hypofractionated radiation therapy. One patient sustained a fall and subsequent fracture after radiation therapy with gross tumor contamination and local spread and thus elected amputation. The median time from the end of radiation to surgery was 41 days (range, 19-67). Gross total resections (R0) were achieved in 91% of patients (29 of 32), and 9% of patients (3 of 32) had microscopic positive margins (R1). Planned flap closure involving plastic and reconstructive surgery occurred in 41% of patients (13 of 32). Median tumor size was 9.5 cm. The most common histology was undifferentiated pleomorphic sarcoma (31.2%) and most tumors were located in the lower extremity.
Toxicity and oncologic outcomes
During radiation, there were no grade 3 or higher acute dermatitis reactions. Grade 2 acute dermatitis occurred in 15.6% of patients (5 of 32). Major acute postoperative wound complications occurred in 25% of patients (8 of 31). Of the 8 patients with a wound complication, 6 (75%) occurred in the lower extremity, all of which were in the proximal lower extremity. One patient ultimately elected to undergo a hip disarticulation due to persistent infection. Five of the 10 patients who received neoadjuvant chemotherapy developed a wound complication, whereas 13.6% (3 of 22) who did not receive chemotherapy developed a wound complication (P = .07). In addition to chemotherapy, age, tumor size, tumor location, and acute dermatitis did not significantly affect the incidence of postoperative wound complications (Table 4).
Table 4.
Factors associated with post-operative wound complication & grade ≥2 fibrosis
| Post-operative WC (%) | P - value | ≥ Grade 2 fibrosis | P - value | |
|---|---|---|---|---|
| Median age | ||||
| ≤65 | 4/16(66.7) | 0.99 | 3/16 (18.8) | 0.66 |
| >65 | 4/16 (66.7) | 4/13 (30.8) | ||
| Median size | ||||
| ≤9.5 cm | 3/16 (18.8) | 0.68 | 3/16 (18.8) | 0.66 |
| >9.5 cm | 5/16 (31.3) | 4/13 (30.8) | ||
| Location | ||||
| Upper Extremity/Trunk | 2/7 (28.6) | 0.99 | 1/6 (16.7) | 0.99 |
| Lower Extremity | 6/25 (24) | 6/23 (26.1) | ||
| Acute dermatitis | ||||
| No | 4/19 (21.) | 0.68 | 2/17 (11.8) | 0.09 |
| Yes | 4/13 (30.8) | 5/12 (41.7) | ||
| Chemotherapy | ||||
| No | 3/22 (13.7) | 0.07 | 4/19 (21.1) | 0.66 |
| Yes | 5/10 (50) | 3/10 (30%) | ||
| Wound complication | ||||
| No | N/A | 5/22 (22.7) | 0.99 | |
| Yes | 2/7 (28.6) |
There were 23 patients who had evaluable fibrosis at 2 years. In this subset, grade 3 fibrosis occurred in 13.0% (3 of 23), grade 2 occurred in 21.7% (5 of 23), and grade 1 occurred in 34.7% (8 of 23) of patients. Age, tumor size, tumor location, administration of chemotherapy, acute dermatitis, and development of postoperative wound complications were not significantly associated with the development of grade ≥2 fibrosis (Table 4). The median and mean MSTS score for all patients as well as those with 2-year follow-up was 28 and 27.4, respectively.
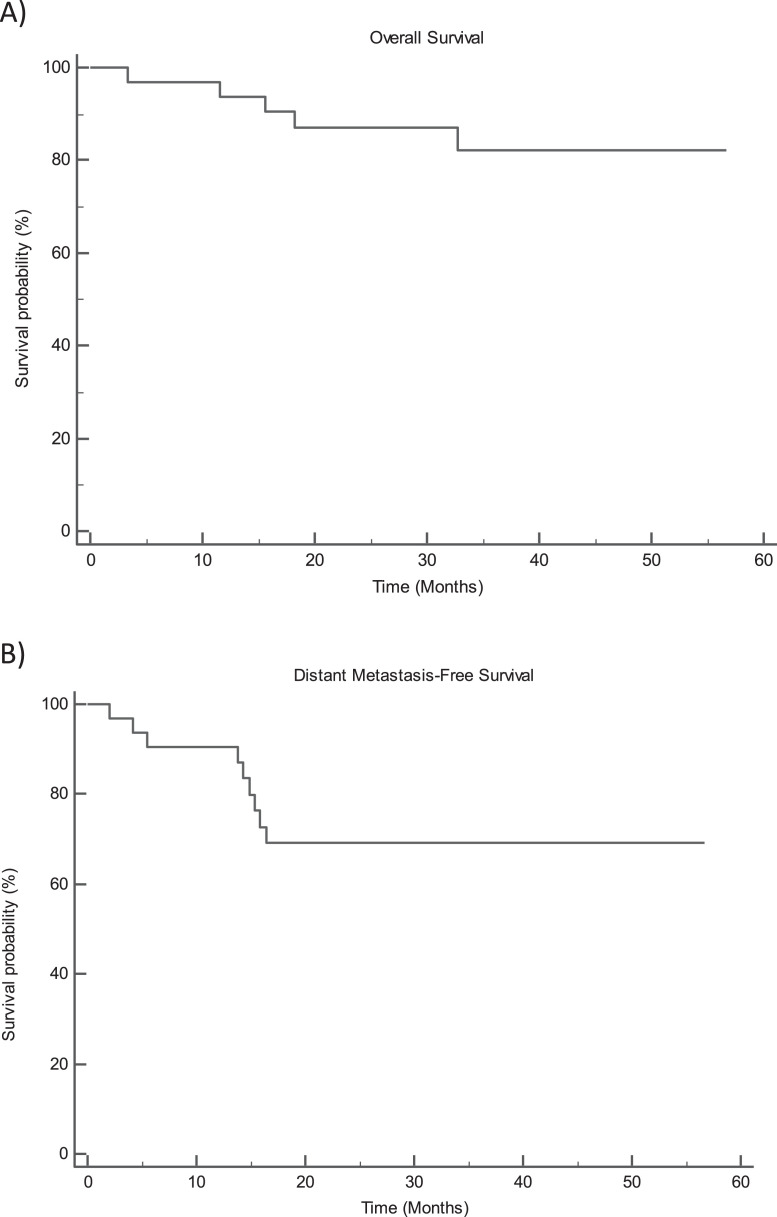
No patient developed a local recurrence, and the 3-year local control was 100%. The 3-year OS was 82.2%. Distant metastasis developed in 28% of patients (9 of 32) of patients, and the 3-year DMFS was 69% (Fig. 1). The median time to the development of distant metastasis was 14.6 months (range, 2.1-38.8).
Fig. 1.
Kaplan-Meier curves for overall survival (A) and distant metastasis-free survival (B).
Of the 32 patients enrolled, 24 have had a minimum 2-year follow-up. Of these, none has had a local recurrence, 1 patient (4.1%) died, and 25% of patients (6 of 24) developed distant metastasis. The 3-year local control in this subset remains at 100%. The 3-year OS and DMFS are 94.4% and 75.0%, respectively.
Pathologic outcomes
Median necrosis, fibrosis, and viable cells from all 32 patients who underwent resection were 17.5%, 20%, and 50% respectively. No pathologic variable was associated with DMFS. Treatment-related necrosis and fibrosis ≥90% occurred in 4 patients. Although not statistically significant, therapy-related changes of necrosis and fibrosis ≥90% were associated with a 3-year OS of 100% versus 77.9% (P = .31). R0 resection was achieved in 100% of patients who had ≥90% treatment-related necrosis and fibrosis versus 88.3% of patients who had <90% treatment-related necrosis and fibrosis.
Discussion
Conventional preoperative radiation therapy for STS of the extremity and trunk involves daily treatments over the course of 5 weeks. With the addition of surgery, the overall treatment “package” time may extend to 8 to 10 weeks with preoperative radiation alone or up to 22 weeks if neoadjuvant systemic therapy is administered. Patients undergoing radiation therapy with or without systemic therapy often prefer shorter courses of irradiation to facilitate earlier tumor resection.
In addition to decreased treatment time, hypofractionated radiation therapy offers other advantages. STS are known to be more resistant to irradiation due to its enhanced ability to repair DNA damage between fractions compared with other malignancies.24,25 Thus, a regimen that offers a higher dose per fraction, or biologic equivalent dose, leads to more tumor cells killed per fraction and decreased ability for sarcoma cells to repair themselves between fractions. Moreover, the global COVID-19 pandemic has led many specialties to examine mitigating risk of hospital-acquired infections in this high-risk population.26 Due to past and recent data demonstrating good rates of local control and acceptable incidence of short- and long-term toxicity, hypofractionated preoperative radiation therapy in STS offers an effective option to abate prolonged hospital exposure, treatment delays, and if performed at a sarcoma center, delivery of high quality therapy in appropriate patient populations.26 Lastly, mitigation of the implicit costs of travel, decreased hours worked, and other social factors make hypofractionated radiation therapy an attractive option.
There is a growing body of literature investigating short-course dose-intensified radiation therapy in STS, including ongoing studies evaluating moderately hypofractionated radiation therapy at large sarcoma centers within the United States.27,28 One of the initial and largest prospective studies examined preoperative hypofractionated radiation therapy administered to 25 Gy in 5 fractions. After a median follow-up of 35 months, the 3-year OS was 72% and local control was 81%. Although OS is comparable to historical data, local control is lower than reported in the preoperative radiation therapy literature for STS.7 The investigators of the study calculated an equivalent dose in 2 Gy fractions (EQD2) of 40 Gy with an α/β ratio of 3. In the current study, we chose to treat patients with an α/β ratio of 5 to 35 Gy in 5 fractions (EQD2 = 60 Gy) every other day to allow for tissue recovery, which is based on a regimen that has been reported in the literature with excellent outcomes.8
More recently, Kalbasi et al prospectively enrolled 52 patients in a phase II preoperative hypofractionation trial.17 In this study, radiation was delivered over 5 consecutive days to 30 Gy at 6 Gy per fraction with an EQD2 of 50 Gy. Major acute wound complications occurred in 32% of patients, and grade ≥2 fibrosis and joint stiffness were present in 11% of patients each. After a minimum 2-year follow-up, local control was 94.3% and 21.2% of patients developed distant disease. In the current study, the primary endpoint was local control, which was 100% in all patients and in those who had a minimum 2-year follow-up. In addition, neoadjuvant chemotherapy was administered in 31% of patients, and the addition of chemotherapy did not influence local control, DMFS (P = .66), or OS (P = .48).
Although excellent outcomes have been demonstrated (Tables 1 and 2), more condensed preoperative radiation regimens in STS may not be used due to concerns of acute and long-term toxicity. Although not powered for toxicity, in this study the incidence of major acute postoperative wound complications was 25%, which is lower than the incidence in the CAN-NCIC-SR2 study (35%), in which patients were administered standard preoperative dose to 50 Gy at 2 Gy per fraction.2,4 As demonstrated in other studies,2 the incidence of postoperative complications was primarily located in the proximal lower extremity in the current study. Furthermore, it is on par with the incidence of wound complications in other studies that have investigated preoperative hypofractionated radiation therapy with varying dose regimens (Tables 1 and 2). The rate of grade ≥2 fibrosis in the present study was 34.8%, which is comparable with the grade ≥2 fibrosis in the CAN-NCIC-SR2 study (31.5%) but had higher rates of fibrosis than in other hypofractionated preoperative studies (3.7%-13.8%).4,7,8,17, 18, 19, 20, 21, 22,29 Doses greater than 60 to 65 Gy in 2 to 3 Gy per fraction have been shown to lead to a 50% risk of developing fibrosis.30 In this study, the biologic equivalent dose of 35 Gy in 5 fractions is at or greater than the dose reported in these studies, which could explain the incidence of grade ≥2 fibrosis. It is also plausible that as the data continue to mature, the rate of grade ≥2 fibrosis may decrease; however, as the denominator is small, any change in either the numerator or denominator affects the incidence to a large degree. In addition, the impact of late toxicity in this trial did not translate to decreased MSTS scores, for which the median and mean scores were 28 and 27.4, respectively. This correlates with the study of Davis et al, where the 2-year mean MSTS score was 28 in patients who received preoperative standard fraction irradiation.4
Preoperative hypofractioned irradiation has been used in combination with chemotherapy. Spencer et al enrolled 48 patients with high-grade extremity STS who received 3 cycles of doxorubicin and ifosfamide in conjunction with hypofractionated radiation to 25 Gy in 5 fractions.31 Radiation was delivered during cycle 2, given concurrently with doxorubicin alone. The 3-year OS was 86.3% and the addition of systemic therapy did not impact the development of wound complications, which occurred in 19.5% of subjects. In the present study, the use of neoadjuvant chemotherapy was recommended in a multidisciplinary setting and contingent on histology, tumor size and grade, and patient factors such as age, performance status, and comorbidities. Although a small number of patients received neoadjuvant systemic therapy, an increased incidence of postoperative wound complications was seen in this group compared with those who did not receive chemotherapy (50% vs 13.6%), which trended toward significance (P = .07). Other studies that have assessed the impact of neoadjuvant chemotherapy in patients treated with conventionally fractioned irradiation have, for the most part, not been associated with an increased incidence of postoperative wound complications.32, 33, 34
Treatment-related pathologic outcomes such as necrosis have been investigated as prognostic factors in STS.19,21,35, 36, 37 Necrosis is strongly prognostic in osteosarcoma, with improved survival in tumors that have >90% necrosis.38 Pathologic response to neoadjuvant therapy had been investigated in Radiation Therapy Oncology Group 9514, where 27% of patients who received interdigitated chemotherapy and radiation therapy had a pathologic complete response (pCR).39 Recent data have shown pCR such as necrosis and/or fibrosis has led to improved OS and distant recurrence-free survival.19,37,39, 40, 41 Seldon et al retrospectively reviewed 79 patients who received neoadjuvant irradiation, chemotherapy, or chemoradiation, and showed that chemoradiation was associated with higher rates of pCR, as defined by ≥90% necrosis.40 Pathologic complete response was also associated with improved OS (100% vs 81.2%, P = .18). Similarly in this study, therapy-related pathologic changes ≥90% led to improved OS (100% vs 77.9%, P = .31).
Kalbasi et al reported one of the few studies that examined pathologic effect by percentage of hyalinization and necrosis relative to biopsy specimens after 30 Gy in 5 fractions and found the average pathologic treatment effect was 44%.17 Although pathologic outcomes are currently being implemented as endpoints in prospective clinical trials, consistency in the definition of pCR and variables contributing to treatment-related response is imperative to guide future sarcoma trials.42
Although the study presented is a prospective trial, there are inherent biases. A selection bias was present as patients were enrolled whose tumors were <20 cm and thought to be more amenable to hypofractionated preoperative radiation therapy without leading to added toxicity. In addition, the modest sample size for a heterogeneous malignancy may impact various outcomes, which is one of the primary reasons prospective sarcoma trials are difficult to translate to all histologies. Ideally, phase II or III randomized controlled trials may be warranted to corroborate these findings, leading to routine implementation by sarcoma providers. Nonetheless, this study adds to the emerging data supporting preoperative hypofractionated radiation therapy in localized STS of the extremity and trunk.
Conclusions
The present study illustrates the safety, efficacy, and convenience of a 5-fraction course of preoperative hypofractionated radiation therapy in extremity and truncal STS. High rates of 3-year local control and overall survival as well as acceptable rates of acute and long-term toxicity were demonstrated. Therefore, if feasible, phase II or III multi-institutional studies with a larger sample size are warranted to shift the paradigm of preoperative radiation in STS.
Footnotes
Sources of support: Kurtis R. Froedtert Clinical Trials Office Seed Grant 3307337.
Disclosures: Dr Hackbarth is on the Musculoskeletal Transplant Foundation Board of Directors. Dr Neilson is on the Musculoskeletal Transplant Foundation Board of Trustees. Dr Charlson is on the Deciphera Advisory Board. All other authors have no disclosures to declare.
Research data are stored in an institutional repository and will be shared upon request to the corresponding author.
Supplementary material associated with this article can be found in the online version at doi:10.1016/j.adro.2021.100850.
Appendix. Supplementary materials
References
- 1.Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300. doi: 10.3322/caac.20073. [DOI] [PubMed] [Google Scholar]
- 2.O'Sullivan B, Davis AM, Turcotte R, et al. Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: A randomised trial. Lancet. 2002;359:2235–2241. doi: 10.1016/S0140-6736(02)09292-9. [DOI] [PubMed] [Google Scholar]
- 3.O'Sullivan B, Griffin AM, Dickie CI, et al. Phase 2 study of preoperative image-guided intensity-modulated radiation therapy to reduce wound and combined modality morbidities in lower extremity soft tissue sarcoma. Cancer. 2013;119:1878–1884. doi: 10.1002/cncr.27951. [DOI] [PubMed] [Google Scholar]
- 4.Davis AM, O'Sullivan B, Turcotte R, et al. Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol. 2005;75:48–53. doi: 10.1016/j.radonc.2004.12.020. [DOI] [PubMed] [Google Scholar]
- 5.Whelan T, MacKenzie R, Julian J, et al. Randomized trial of breast irradiation schedules after lumpectomy for women with lymph node-negative breast cancer. J Natl Cancer Inst. 2002;94:1143–1150. doi: 10.1093/jnci/94.15.1143. [DOI] [PubMed] [Google Scholar]
- 6.Arcangeli G, Saracino B, Arcangeli S, et al. Moderate hypofractionation in high-risk, organ-confined prostate cancer: Final results of a phase III randomized trial. J Clin Oncol. 2017;35:1891–1897. doi: 10.1200/JCO.2016.70.4189. [DOI] [PubMed] [Google Scholar]
- 7.Kosela-Paterczyk H, Szacht M, Morysinski T, et al. Preoperative hypofractionated radiotherapy in the treatment of localized soft tissue sarcomas. Eur J Surg Oncol. 2014;40:1641–1647. doi: 10.1016/j.ejso.2014.05.016. [DOI] [PubMed] [Google Scholar]
- 8.Kubicek GJ, LaCouture T, Kaden M, et al. Preoperative radiosurgery for soft tissue sarcoma. Am J Clin Oncol. 2018;41:86–89. doi: 10.1097/COC.0000000000000236. [DOI] [PubMed] [Google Scholar]
- 9.Norkus D, Miller A, Kurtinaitis J, et al. A randomized trial comparing hypofractionated and conventionally fractionated three-dimensional external-beam radiotherapy for localized prostate adenocarcinoma: A report on acute toxicity. Strahlenther Onkol. 2009;185:715–721. doi: 10.1007/s00066-009-1982-z. [DOI] [PubMed] [Google Scholar]
- 10.Haviland JS, Owen JR, Dewar JA, et al. The UK Standardisation of Breast Radiotherapy (START) trials of radiotherapy hypofractionation for treatment of early breast cancer: 10-Year follow-up results of two randomised controlled trials. Lancet Oncol. 2013;14:1086–1094. doi: 10.1016/S1470-2045(13)70386-3. [DOI] [PubMed] [Google Scholar]
- 11.Erlandsson J, Holm T, Pettersson D, et al. Optimal fractionation of preoperative radiotherapy and timing to surgery for rectal cancer (Stockholm III): A multicentre, randomised, non-blinded, phase 3, non-inferiority trial. Lancet Oncol. 2017;18:336–346. doi: 10.1016/S1470-2045(17)30086-4. [DOI] [PubMed] [Google Scholar]
- 12.Lee WR, Dignam JJ, Amin MB, et al. Randomized phase III noninferiority study comparing two radiotherapy fractionation schedules in patients with low-risk prostate cancer. J Clin Oncol. 2016;34:2325–2332. doi: 10.1200/JCO.2016.67.0448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jacob R, Gilligan D, Robinson M, Harmer C. Hyper-fractionated radiotherapy for soft tissue sarcoma: Results of the second study of hyper-fractionated radiotherapy. Sarcoma. 1999;3:157–165. doi: 10.1080/13577149977587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Soyfer V, Corn BW, Kollender Y, et al. Hypofractionated adjuvant radiation therapy of soft-tissue sarcoma achieves excellent results in elderly patients. Br J Radiol. 2013;86 doi: 10.1259/bjr.20130258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Soyfer V, Corn BW, Kollender Y, Tempelhoff H, Meller I, Merimsky O. Radiation therapy for palliation of sarcoma metastases: a unique and uniform hypofractionation experience. Sarcoma. 2010;2010 doi: 10.1155/2010/927972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Engel CJ, Eilber FR, Rosen G, Selch MT, Fu YS. Preoperative chemotherapy for soft tissue sarcomas of the extremities: The experience at the University of California, Los Angeles. Cancer Treat Res. 1993;67:135–141. doi: 10.1007/978-1-4615-3082-4_9. [DOI] [PubMed] [Google Scholar]
- 17.Kalbasi A, Kamrava M, Chu FI, et al. A phase II trial of 5-day neoadjuvant radiotherapy for patients with high-risk primary soft tissue sarcoma. Clin Cancer Res. 2020;26:1829–1836. doi: 10.1158/1078-0432.CCR-19-3524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Temple WJ, Temple CL, Arthur K, Schachar NS, Paterson AH, Crabtree TS. Prospective cohort study of neoadjuvant treatment in conservative surgery of soft tissue sarcomas. Ann Surg Oncol. 1997;4:586–590. doi: 10.1007/BF02305541. [DOI] [PubMed] [Google Scholar]
- 19.MacDermed DM, Miller LL, Peabody TD, et al. Primary tumor necrosis predicts distant control in locally advanced soft-tissue sarcomas after preoperative concurrent chemoradiotherapy. Int J Radiat Oncol Biol Phys. 2010;76:1147–1153. doi: 10.1016/j.ijrobp.2009.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Meyer JM, Perlewitz KS, Hayden JB, et al. Phase I trial of preoperative chemoradiation plus sorafenib for high-risk extremity soft tissue sarcomas with dynamic contrast-enhanced MRI correlates. Clin Cancer Res. 2013;19:6902–6911. doi: 10.1158/1078-0432.CCR-13-1594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ryan CW, Montag AG, Hosenpud JR, et al. Histologic response of dose-intense chemotherapy with preoperative hypofractionated radiotherapy for patients with high-risk soft tissue sarcomas. Cancer. 2008;112:2432–2439. doi: 10.1002/cncr.23478. [DOI] [PubMed] [Google Scholar]
- 22.Pennington JD, Eilber FC, Eilber FR, et al. Long-term outcomes with ifosfamide-based hypofractionated preoperative chemoradiotherapy for extremity soft tissue sarcomas. Am J Clin Oncol. 2018;41:1154–1161. doi: 10.1097/COC.0000000000000443. [DOI] [PubMed] [Google Scholar]
- 23.Wang D, Zhang Q, Eisenberg BL, et al. Significant reduction of late toxicities in patients with extremity sarcoma treated with image-guided radiation therapy to a reduced target volume: Results of Radiation Therapy Oncology Group RTOG-0630 trial. J Clin Oncol. 2015;33:2231–2238. doi: 10.1200/JCO.2014.58.5828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Merimsky O, Kollender Y, Bokstein F, et al. Radiotherapy for spinal cord compression in patients with soft-tissue sarcoma. Int J Radiat Oncol Biol Phys. 2004;58:1468–1473. doi: 10.1016/j.ijrobp.2003.09.026. [DOI] [PubMed] [Google Scholar]
- 25.Yuce Sari S, Cengiz M, Dauletkazin A, et al. Hypofractionated radiotherapy for non-metastatic bone and soft tissue sarcomas. Cancer Radiother. 2019;23:853–859. doi: 10.1016/j.canrad.2019.06.011. [DOI] [PubMed] [Google Scholar]
- 26.Spalek MJ, Rutkowski P. Coronavirus disease (COVID-19) outbreak: Hypofractionated radiotherapy in soft tissue sarcomas as a valuable option in the environment of limited medical resources and demands for increased protection of patients. Front Oncol. 2020;10:993. doi: 10.3389/fonc.2020.00993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Guadagnolo BA. Shorter Course, Hypofractionated Pre-Surgery Radiation Therapy in Treating Patients With Localized, Resectable Soft Tissue Sarcoma of the Extremity and Superficial Trunk. NCT03819985. Available at: https://clinicaltrials.gov/ct2/show/study/NCT03819985. Accessed March 3, 2022.
- 28.Ahmed SK. Hypofractionated Radiation Therapy Before Surgery for the Treatment of Localized, Resectable Soft Tissue Sarcoma of the Extremity and Superficial Trunk. NCT04562480. Available at: https://clinicaltrials.gov/ct2/show/NCT04562480?cond=NCT04562480&draw=2&rank=1. Accessed March 3, 2022.
- 29.Parsai S, Lawrenz J, Kilpatrick S, et al. Early outcomes of preoperative 5-fraction radiation therapy for soft tissue sarcoma followed by immediate surgical resection. Adv Radiat Oncol. 2020;5:1274–1279. doi: 10.1016/j.adro.2020.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Joiner MC, van de Kogel A. Basic Clinical Radiobiology. 4th ed. London: Taylor and Francis; 2009.
- 31.Spencer RM, Aguiar Junior S, Ferreira FO, et al. Neoadjuvant hypofractionated radiotherapy and chemotherapy in high-grade extremity soft tissue sarcomas: Phase 2 clinical trial protocol. JMIR Res Protoc. 2017;6:e97. doi: 10.2196/resprot.6806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Slump J, Bastiaannet E, Halka A, et al. Risk factors for postoperative wound complications after extremity soft tissue sarcoma resection: A systematic review and meta-analyses. J Plast Reconstr Aesthet Surg. 2019;72:1449–1464. doi: 10.1016/j.bjps.2019.05.041. [DOI] [PubMed] [Google Scholar]
- 33.Stoeckle E, Michot A, Rigal L, et al. The riskof postoperative complications and functional impairment after multimodality treatment for limb and trunk walk soft-tissue sarcoma: Long term results from a monocentric series. Eur J Surg Oncol. 2017;43:1117–1125. doi: 10.1016/j.ejso.2017.01.018. [DOI] [PubMed] [Google Scholar]
- 34.Sanniec KJ, Swanson S, Casey WJ, Schwartz A, Bryant L, Rebecca AM. Predictive factors after sarcoma resection requiring plastic surgeon involvement. Ann Plast Surg. 2013;71:283–285. doi: 10.1097/SAP.0b013e31827c7973. [DOI] [PubMed] [Google Scholar]
- 35.Eilber FC, Rosen G, Eckardt J, et al. Treatment-induced pathologic necrosis: A predictor of local recurrence and survival in patients receiving neoadjuvant therapy for high-grade extremity soft tissue sarcomas. J Clin Oncol. 2001;19:3203–3209. doi: 10.1200/JCO.2001.19.13.3203. [DOI] [PubMed] [Google Scholar]
- 36.Mack LA, Crowe PJ, Yang JL, et al. Preoperative chemoradiotherapy (modified Eilber protocol) provides maximum local control and minimal morbidity in patients with soft tissue sarcoma. Ann Surg Oncol. 2005;12:646–653. doi: 10.1245/ASO.2005.03.064. [DOI] [PubMed] [Google Scholar]
- 37.Shah D, Borys D, Martinez SR, et al. Complete pathologic response to neoadjuvant radiotherapy is predctive of oncologic outcome in patients with soft tissue sarcoma. Anticancer Res. 2012;32:3911–3916. [PMC free article] [PubMed] [Google Scholar]
- 38.Lindner NJ, Scarborough MT, Spanier SS, Enneking WF. Local host response in osteosarcoma after chemotherapy referred to radiographs, CT, tumour necrosis and patient survival. J Cancer Res Clin Oncol. 1998;124:575–580. doi: 10.1007/s004320050217. [DOI] [PubMed] [Google Scholar]
- 39.Kraybill WG, Harris J, Spirot IJ, et al. Phase II study of neoadjuvant chemotherapy and radiation therapy in the management of high-risk, high-grade, soft tissue sarcoma of the extremities and body wall: Radiation Therapy Oncology Group trial 9514. J Clin Oncol. 2006;24:619–625. doi: 10.1200/JCO.2005.02.5577. [DOI] [PubMed] [Google Scholar]
- 40.Seldon C, Shrivastava G, Fernandez M, et al. Pathologic response rates after neoadjuvant therapy for sarcoma: A single institution study. Cancers (Basel) 2021;13:1074. doi: 10.3390/cancers13051074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bonvolot S, Wunder J, Gronchi A, et al. Complete pathological response is associated with better survival outcomes in patients with soft tissue sarcoma: Results of a retrospective multicenter study. Eur J Surg Oncol. 2021;47:2166–2172. doi: 10.1016/j.ejso.2021.02.024. [DOI] [PubMed] [Google Scholar]
- 42.Bonvolot S, Rutkowski P, Thariat J, et al. NBTXR3, a first-in-class radioenhancer hafnium oxide nanoparticle, plus radiotherapy versus radiotherapy alone in patients with locally advanced soft-tisse sarcoma (Act.In.Sarc): A multicentre, phase 2-3, randomized, controlled trial. Lancet Oncol. 2019;20:1148–1159. doi: 10.1016/S1470-2045(19)30326-2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.