Introduction
The essential amino acid tryptophan is the unique substrate for the biosynthesis of neurotransmitter serotonin, sleep hormone melatonin and co-factor nicotinamide adenine dinucleotide (NAD+) through its metabolic pathways. 1 The catabolism of tryptophan by indoleamine 2,3-dioxygenases (IDO1/2) through the kynurenine pathway (KP) leads to the generation of multiple bioactive metabolites referred as kynurenines.2,3 Within the central nervous system (CNS), brain cells including glia and neurons produce different kynurenines. 4 During ageing a shift from the serotonergic to kynurenine pathway progressively occurs in the tryptophan catabolism resulting in an increased activity of the enzyme IDO-1 in turn increases the production of neuroactive KP metabolites. 5 The progressive alterations of the innate immune system linked to ageing trigger the development of chronic inflammatory cascades referred as ‘inflammaging’ which include astrogliosis, microgliosis and increased production of cytokines like IL-6, IL-1β, IFN-γ, TNF-α etc. . ..2,6 IFN-γ and TNF-α are both know to trigger a strong induction of IDO1 through positive feedback mechanism in older population. 7 This increased IDO1 activity with age not only results in higher production of kynurenines, but also stimulates inducible nitric oxide synthase (iNOS) activity and increases nitric oxide (NO) production. iNOS is mainly expressed by activated microglia, astrocytes, endothelial cells and infiltrating lymphocytesa.8-10
Could IDO-1 Activation be Associated With Vascular Dementia?
The activation of IDO-1 and KP could possibly elicit vascular dementia directly through vasoactive metabolites and/or indirectly by stimulating iNOS through multiple pathways. (1) Several KP metabolites can have a direct effect on blood vessel by eliciting vascular inflammation mediated arterial stiffness and atherosclerosis by binding to aryl hydrocarbon receptors. KYN is an endothelium-derived relaxing factor produced during inflammation.11-16 The basolateral increase of kynurenines especially quinolinic acid at BBB endothelial cells and pericytes also results in local neurotoxicity thus leading to BBB disruption. 17 The excess of quinolinic acid also contribute to the production of β-amyloid and neurofibrillary tangles,4,18,19 while amyloid precursor protein and β-amyloid are involved in eliciting endothelium dependent vasoconstriction, reduced cerebral blood flow thus abnormal vascular autoregulation 20 leading to neurodegeneration and cognitive impairment. 4 Alterations in tryptophan metabolism in aged population contribute to the occurrence of vascular impairments, progressive neurodegeneration and in turn cognitive impairment.
(2) As IDO-1 activation leads to iNOS induction, the generated NO reacts with reactive oxygen species (ROS) to produce peroxynitrite (ONOO−) which causes oxidative damage to biomacromolecules like lipids, proteins and deoxyribonucleic acid (DNA) leading to deposition of toxic substances. All those compounds disrupt mitochondrial functions resulting in the failure to meet the biological energy needs for the neuronal homoeostasis. The overall effects lead to neuronal apoptosis and blood brain barrier (BBB) disruption contributing to neurovascular dysfunctions. 21 Additionally, increased levels of NO inhibit long-term potentiation (LTP) formation by decreasing production of Brain derived neurotrophic factor (BDNF), shifting the neuronal stem cells to differentiate into astrocytes thus hindering neurorepair and neurovascular impairment. 22
(3) The excessive formation of NO triggers intracellular accumulation of p53 (pro-apoptotic factor), leading to necrosis of cerebrovascular endothelial cells and vascular smooth muscle cells (VSMCs) and ultimately to neurovascular dysfunctions. 23 (4) iNOS also inhibits proliferation of T cells (Th-17), and increases circulating levels of IL-17, which then activates the Rho A/RHO kinase pathway. The latter inhibits endothelial nitric oxide synthase (eNOS) activity leading to cerebrovascular endothelial cells dysfunction. 23
Is IDO-1 Linked to Vulnerability of Aged Population to COVID-19 Infection?
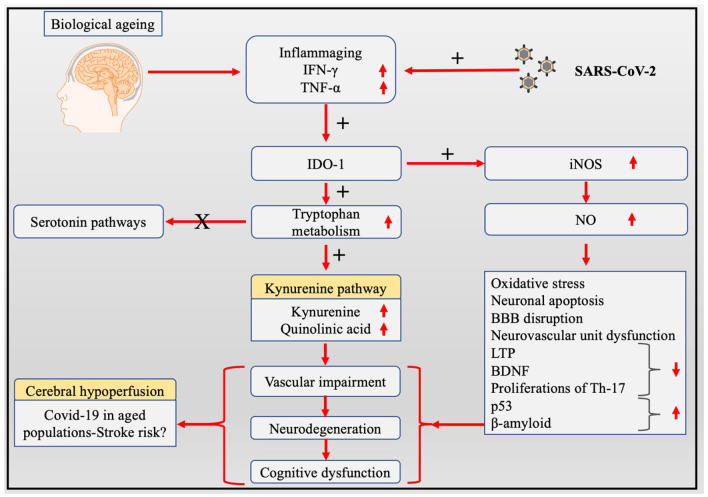
Severe acute respiratory syndrome coronavirus-2 (SARS Cov-2) infection initially begins with peripheral inflammation and causes activation of endothelial cells leading to BBB disruption associated with astrogliosis and microgliosis. 24 At later stage, this latter events triggers neuroinflammation with increased cytokine/chemokine production, oxidative stress, and the altered immune cells trafficking contribute to further damage of the BBB, generating a strong neuroinflammatory and neurotoxic loop.25-27 The dysregulated tryptophan metabolism, especially the high levels of quinolinic acid, in elderly population will amplify the COVID-19 associated risk factors by elevating neuroinflammation, 28 compromised the BBB, neurovascular impairments through cerebrovascular endothelial cells and VSMCs dysfunctions. 17 This chain of complex and multifactorial of detrimental events highlights the important effects resulting of the alteration of the tryptophan metabolism in older patients with Covid-19. The viral infection by itself triggers CNS pathologies including strong inflammatory responses, formation of amyloid plaques both leading to increased risk of intracerebral haemorrhage and ischaemic brain damage.29,30 The dysregulation of the KP in elderly population could be linked with an higher incidence of cerebrovascular diseases like acute stroke and increased fatality rate in COVID-19 affected elderly population.29,31,32 We hypothesise that it is likely that the activation of the kynurenine pathway in elderly COVID-19 patients and survivors (‘long-term COVID’) could be a major contributor for cerebrovascular damages and a likely therapeutic target (Figure 1).
Figure 1.
Potential mechanisms of altered tryptophan metabolism induced cerebrovascular and neurodegenerative diseases in Covid-19 aged population.
Abbreviations: BDNF, brain derived neurotrophic factor; IDO-1, indoleamine 2,3-dioxygenases; IFN-γ, interferon-gamma; iNOS, inducible nitric oxide synthase; LTP, long term potentiation; NO, Nitric oxide; TNF-α, tumour necrosis factor-alpha.
Acknowledgments
AMM, SP, BR and SBC thank JSS Academy of Higher Education and Research (JSSAHER), Mysuru, India for providing facilities for this editorial note preparation.
Footnotes
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs: Shasthara Paneyala  https://orcid.org/0000-0001-5095-8602
https://orcid.org/0000-0001-5095-8602
Benjamin Heng  https://orcid.org/0000-0002-0247-2920
https://orcid.org/0000-0002-0247-2920
Gilles J Guillemin  https://orcid.org/0000-0001-8105-4470
https://orcid.org/0000-0001-8105-4470
Saravana Babu Chidambaram  https://orcid.org/0000-0003-2357-056X
https://orcid.org/0000-0003-2357-056X
References
- 1. Bhat A, Pires AS, Tan V, Babu Chidambaram S, Guillemin GJ. Effects of sleep deprivation on the tryptophan metabolism. Int J Tryptophan Res. 2020;13:1178646920970902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Sorgdrager FJH, Naudé PJW, Kema IP, Nollen EA, Deyn PP. Tryptophan metabolism in inflammaging: from biomarker to therapeutic target. Front Immunol. 2019;10:2565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Kar S, Choudhury I. An empirical review on oxidative stress markers and their relevance in obsessive-compulsive disorder. Int J Nutr Pharmacol Neurol Dis. 2016;6:139. [Google Scholar]
- 4. Guillemin GJ, Brew BJ, Noonan CE, Takikawa O, Cullen KM. Indoleamine 2,3 dioxygenase and quinolinic acid immunoreactivity in Alzheimer’s disease hippocampus. Neuropathol Appl Neurobiol. 2005;31:395-404. [DOI] [PubMed] [Google Scholar]
- 5. van der Goot AT, Zhu W, Vázquez-Manrique RP, et al. Delaying aging and the aging-associated decline in protein homeostasis by inhibition of tryptophan degradation. Proc Natl Acad Sci USA. 2012;109:14912-14917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Mahalakshmi AM, Ray B, Tuladhar S, et al. Sleep, brain vascular health and ageing. GeroScience. 2020;42:1257-1283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Widner B, Ledochowski M, Fuchs D. Interferon-gamma-induced tryptophan degradation: neuropsychiatric and immunological consequences. Curr Drug Metab. 2000;1:193-204. [DOI] [PubMed] [Google Scholar]
- 8. de Bie J, Guest J, Guillemin GJ, Grant R. Central kynurenine pathway shift with age in women. J Neurochem. 2016;136:995-1003. [DOI] [PubMed] [Google Scholar]
- 9. Sorgdrager FJH, Vermeiren Y, Van Faassen M, et al. Age- and disease-specific changes of the kynurenine pathway in Parkinson’s and Alzheimer’s disease. J Neurochem. 2019;151:656-668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Lugo-Huitrón R, Ugalde Muñiz P, Pineda B, Pedraza-Chaverrí J, Ríos C, Pérez-de la Cruz V. Quinolinic acid: an endogenous neurotoxin with multiple targets. Oxid Med Cell Longev. 2013;2013:104024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Baumgartner R, Berg M, Matic L, et al. Evidence that a deviation in the kynurenine pathway aggravates atherosclerotic disease in humans. J Intern Med. 2021;289:53-68. [DOI] [PubMed] [Google Scholar]
- 12. Zapolski T, Kamińska A, Kocki T, Wysokiński A, Urbanska EM. Aortic stiffness-is kynurenic acid a novel marker? Cross-sectional study in patients with persistent atrial fibrillation. PLoS One. 2020;15:e0236413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Onida S, Tan MKH, Kafeza M, et al. Metabolic phenotyping in venous disease: the need for standardization. J Proteome Res. 2019;18:3809-3820. [DOI] [PubMed] [Google Scholar]
- 14. Ketelhuth DFJ. The immunometabolic role of indoleamine 2,3-dioxygenase in atherosclerotic cardiovascular disease: immune homeostatic mechanisms in the artery wall. Cardiovasc Res. 2019;115:1408-1415. [DOI] [PubMed] [Google Scholar]
- 15. Rudzite V, Sileniece G, Liepina D, Dalmane A, Zirne R. Impairment of kynurenine metabolism in cardiovascular disease. In: Schwarcz R, Young SN, Brown RR, eds. Kynurenine and Serotonin Pathways: Progress in Tryptophan Research. Springer; 1991:663-667. [DOI] [PubMed] [Google Scholar]
- 16. Wang Y, Liu H, McKenzie G, et al. Kynurenine is an endothelium-derived relaxing factor produced during inflammation. Nat Med. 2010;16:279-285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Owe-Young R, Webster NL, Mukhtar M, et al. Kynurenine pathway metabolism in human blood-brain-barrier cells: implications for immune tolerance and neurotoxicity. J Neurochem. 2008;105:1346-1357. [DOI] [PubMed] [Google Scholar]
- 18. Wu W, Nicolazzo JA, Wen L, et al. Expression of tryptophan 2,3-dioxygenase and production of kynurenine pathway metabolites in triple transgenic mice and human Alzheimer’s disease brain. PLoS One. 2013;8:e59749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Guillemin GJ, Brew BJ. Implications of the kynurenine pathway and quinolinic acid in Alzheimer’s disease. Redox Rep. 2002;7:199-206. [DOI] [PubMed] [Google Scholar]
- 20. Smith EE, Greenberg SM. β-amyloid, blood vessels, and brain function. Stroke. 2009;40:2601-2606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Song K, Li Y, Zhang H, et al. Oxidative stress-mediated blood-brain barrier (BBB) disruption in neurological diseases. Oxid Med Cell Longev. 2020;2020:1-27. [Google Scholar]
- 22. Canossa M, Giordano E, Cappello S, Guarnieri C, Ferri S. Nitric oxide down-regulates brain-derived neurotrophic factor secretion in cultured hippocampal neurons. Proc Natl Acad Sci USA. 2002;99:3282-3287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Zhu H-Y, Hong F-F, Yang S-L. The roles of nitric oxide synthase/nitric oxide pathway in the pathology of vascular dementia and related therapeutic approaches. Int J Mol Sci. 2021;22:4540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Tremblay M-E, Madore C, Bordeleau M, Tian L, Verkhratsky A. Neuropathobiology of COVID-19: the role for Glia. Front Cell Neurosci. 2020;14:592214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Pensato U, Muccioli L, Cani I, et al. Brain dysfunction in COVID-19 and CAR-T therapy: cytokine storm-associated encephalopathy. Ann Clin Transl Neurol. 2021;8:968-979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Ye Q, Wang B, Mao J. The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19. J Infect. 2020;80:607-613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Bouças AP, Rheinheimer J, Lagopoulos J. Why severe COVID-19 patients are at greater risk of developing depression: a molecular perspective. Neuroscientist. Published online November 2, 2020. doi: 10.1177/1073858420967892 [DOI] [PubMed] [Google Scholar]
- 28. Guillemin GJ, Croitoru-Lamoury J, Dormont D, Armati PJ, Brew BJ. Quinolinic acid upregulates chemokine production and chemokine receptor expression in astrocytes. Glia. 2003;41:371-381. [DOI] [PubMed] [Google Scholar]
- 29. Fan H, Tang X, Song Y, Liu P, Chen Y. Influence of COVID-19 on cerebrovascular disease and its possible mechanism. Neuropsychiatr Dis Treat. 2020;16:1359-1367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Mahalakshmi AM, Ray B, Tuladhar S, et al. Does COVID-19 contribute to development of neurological disease? Immun Inflamm Dis. 2021;9:48-58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Lou M, Yuan D, Liao S, Tong L, Li J. Potential mechanisms of cerebrovascular diseases in COVID-19 patients. J Neurovirol. 2021;27:35-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Turski WA, Wnorowski A, Turski GN, Turski CA, Turski L. AhR and IDO1 in pathogenesis of covid-19 and the “Systemic AhR Activation Syndrome” translational review and therapeutic perspectives. Restor Neurol Neurosci. 2020;38:343-354. [DOI] [PMC free article] [PubMed] [Google Scholar]