Abstract
Campylobacter jejuni is a major cause of food-borne human gastroenteritis worldwide and is designated as a high priority antimicrobial-resistant pathogen by the World Health Organization (WHO). In this study, a total of 26 C. jejuni isolates from broiler chickens were screened for the presence of virulence and antimicrobial resistance genes by PCR. As a result, the study detected 11/26 (42.3%), 9/26 (34.6%), 8/26 (30.8%), 7/26 (26.9%), 6/26 (23.1%), and 6/26 (23.1%) of cdtC, pldA, cdtB, cdtA, cadF, and ciaB virulence genes, respectively, with seven of the isolates carrying more than two virulence genes. The majority of the isolates n = 25 (96.1%) were resistant to nalidixic acid, followed by n = 21 (80.7%), n = 22 (84.6%), and n = 5 (19.2%) for tetracycline, erythromycin, and ciprofloxacin, respectively. Most isolates were harboring catI (n = 16; 84.2%), catII (n = 15; 78.9%), catIII (n = 10; 52.6%), catIV (n = 2; 10.5%), floR (n = 10; 52.6%), ermB (n = 14; 73.7%), tetO (n = 13; 68.4%), tetA (n = 9; 47.4%), mcr-4 (n = 8; 42.1%), and ampC (n = 2; 10.5%). Meanwhile, mcr-1, mcr-2, mcr-3, mcr-5, tet(X), tet(P), and tet(W) genes were not detected in all isolates. Class I and Class II integrons were detected in 92.3% (n = 24) and 65.4% (n = 17) isolates, respectively. About 31% (8 of the 26 isolates) isolates were carrying more than two resistance genes. According to our knowledge, this is the first study to detect class II integrons in Campylobacter spp. (C. jejuni). The high prevalence of cdtA, cdtB, cdtC, cadF, pldA, and ciaB genes and antibiotic resistance genes in C. jejuni in this study indicates the pathogenic potential of these isolates. Majority of the isolates demonstrated resistance to nalidixic acid, tetracycline (tet), and erythromycin (ermB), which are the drugs of choice for treating Campylobacter infections. Therefore, these findings highlight the importance of implementing an efficient strategy to control Campylobacter in chickens and to reduce antimicrobial use in the poultry industry, which will help to prevent the spread of infections to humans.
1. Introduction
Poultry meat is an important source of protein and one of the most consumed meat sources in South Africa [1]. To date, about 2.152 million tonnes of poultry meat are consumed in South Africa per year [2]. Despite chickens being considered the main source of protein, they are also responsible for about 80% of human cases of food-borne and zoonotic diseases [3]. Poultry is considered as the main reservoir of many bacterial pathogens including Campylobacter [4]. A number of chicken-borne Campylobacter species of zoonotic importance include C. ureolyticus, C. concisus, C. mucosalis, C. jejuni, C. hyointestinalis, C. insulaenigrae, C. sputorum, C. helveticus, C. lari, C. fetus, C. coli, C. upsaliensis, and C. rectus [5]. Of these, C. jejuni, C. lari, and C. coli are documented as the main contributors to food-borne diseases such as campylobacteriosis in humans [3, 6, 7].
In humans, Campylobacter infections are usually self-limiting, although bacteraemia is more common among the elderly, immunocompromised people, and children [8]. In comparison with other enteric bacteria, Campylobacter has multiple cell surface layers expressing virulent factors that are responsible for its high prevalence and pathogenicity [9]. Motility, toxin production, mucus colonization, attachment, and translocation are all virulence mechanisms used by Campylobacter to cause disease [10]. The antibiotics used for treating Campylobacter infections will usually target these mechanisms of virulence in order to be efficacious.
Different antibiotics such as erythromycin, amoxicillin, azithromycin, clarithromycin, tetracycline, and ciprofloxacin have been used for treating campylobacteriosis [11, 12]. In animals, some of these antibiotics are used as additives to improve the growth rate and feed intake ratio [13, 14]. As a result, the misuse of these antibiotics as additives can lead to antibiotic residues on animal products and the environment as well as the development of antibiotic resistance [1, 11]. Antibiotic resistance is a global health issue that involves the transfer of bacteria and genes between humans and animals [15]. A number of genes which confer resistance to antibiotics in Campylobacter have been determined by previous studies [2, 6, 11]
Tetracycline is exported from the cell via membrane-bound efflux proteins encoded by the efflux genes tetA and tetB [16, 17]. Tetracycline resistance is caused by the tetO gene, and it produces a ribosome-protective protein [16]. A ribosomal methylase encoded by ermB is one of the Campylobacter mechanisms that confer resistance to macrolides [16, 17]. The C257T mutation in the gyrase gene (gyrA) in Campylobacter is the most common mechanism creating quinolone and fluoroquinolone resistance [18]. In C. jejuni, the gyrA gene region contains Thr86Ala, which is responsible for high levels of nalidixic acid resistance and low levels of ciprofloxacin resistance [15, 19]. The Thr86Ile amino acid alteration in the QRDR of gyrA is seen in most ciprofloxacin-resistant Campylobacter spp., especially C. jejuni strains [20]. Antimicrobial resistance genes such as erm(B), aadE, blaOXA−61, and aphA-3 have also been linked to multidrug resistance in Campylobacter strains [3].
Integrons, in particular, play a key role in the acquisition and spread of antibiotic resistance [21, 22]. There are five classes, but only two classes, i.e., I and II are the most important [23]. Class I and II integrons are frequently associated with the Tn7 transposon family [24, 25]. Gram-negative bacteria have a wide range of class I integrons, which are transferred by Tn402 [21]. Dihydroflavonol-4-reductase (dfr), sulfonamide (sul1), broad-spectrum-lactamase, quaternary ammonium compound disinfectants (qacE1), and aminoglycoside-modifying enzymes (AMEs) are all encoded by antimicrobial resistance gene cassettes found in class I integrons [21]. However, the int gene in class II is less active, it can carry unusual cassettes that encode the lipoprotein signal peptidase [21, 26], and it has Dfr1, sul1, and aadA1 gene cassettes [21]. Even though integrons have been detected from class I in Campylobacter [27], neither class II nor III have been detected in Campylobacter spp. [27]. From the study conducted by van Essen-Zandbergen et al. [28] in the Netherlands in broilers, none of the Campylobacter isolates carried the integrons (class I, II, and III). Hence, the aim of this study was to investigate the presence of the virulence genes profile. Class I and II integrons and antimicrobial resistance genes in C. jejuni isolates recovered from the faeces of slaughter-age broiler chickens in the North West province, South Africa.
2. Materials and Methods
2.1. Identification of Campylobacter
26 Campylobacter jejuni strains from our previous study were used [4]. In brief, C. jejuni was isolated from faecal samples, and the genomic DNA was extracted following Zymo Research Fungal/Bacterial DNA kit instructions (Zymo Research Corp., CA, USA). The DNA concentration was quantified using a NanoDrop spectrophotometer [29]. Conventional PCR was used to detect the Campylobacter spp. in the chicken faeces using universal 16S rRNA Campylobacter spp. All the PCR products were sequenced at Inqaba Biotechnical Industries (Pty) Ltd., Pretoria, South Africa, and sequence identity was determined using the nucleotide Basic Local Alignment Search Tool (BLASTn) (https://blast.ncbi.nlm.nih.gov/Blast.cg). The nucleotide sequences were deposited in the GenBank database and assigned with accession numbers (MZ209102 − MZ209127) available at https://www.ncbi.nlm.nih.gov/nucleotide.
2.2. Antimicrobial Resistance (AMR) Profile
Antibacterial susceptibility screening to ciprofloxacin (5 g), nalidixic acid (30 g), erythromycin (15 g), and tetracycline (30 g) (Davies Diagnostics, Johannesburg, South Africa) was conducted based on the World Health Organization (WHO) Advisory Group on Integrated Surveillance of Antimicrobial Resistance guidelines [30] on food-borne bacteria. The Kirby–Bauer disc diffusion method was used, and the results were interpreted according to the Clinical and Laboratory Standards Institute (CLSI) [4]. Following CLSI recommendations, antibacterial susceptibility testing was performed on Muller–Hinton (MH) agar (LAB M, Neogen Company) supplemented with 10% sheep blood. The zones of inhibition detected around each antibiotic disc in millimeters were used to calculate antibiotic susceptibility. Standard reference strains of Staphylococcus aureus (ATCC® 29213) and Campylobacter jejuni ATCC (33560) were used as controls.
2.3. Detection of Antibiotic Resistance Genes
The presence or absence of chloramphenicol (catI, catII, catIII, catIV, and floR), erythromycin (ermB), tetracycline (tet(A), tet(O), tet(X), tet(P), and tet(W)), colistin (mcr-1, mcr-2, mcr-3, mcr-4, and mcr-5), and ampicillin (AmpC) resistance genes, including two classes of integrons (Int) (class I and II), was determined in Campylobacter jejuni isolates using the qualitative PCR technique. All the primers were obtained from Inqaba Biotechnical Industries (Pty) Ltd., Pretoria, South Africa. Each PCR reaction included a total reaction of 25 μL containing 12.5 μL of a 2X DreamTag Green Master Mix (0.4 mM dATP, 0.4 mM dCTP, 0.4 mM dGTP, 0.4 mM dTTP, 4 mM MgCl2, and loading buffer) (ThermoFisher Scientific, South Africa), 8.5 μL of nuclease-free water, 2.0 μL of the template DNA, and 1.0 μL of each oligonucleotide primer. PCR reactions were performed using the ProFlex PCR System (Applied Biosystems, USA). Amplified PCR products were resolved on a 1.5% (w/v) agarose gel in a 40 mM Tris (Sigma Aldrich, US), 1X TAE buffer (20 mM acetic acid (Merck, US), and 1 mM EDTA (Merck, South Africa) at pH 8.0), stained with 0.001 μg/mL ethidium bromide, and visualized under ultraviolet (UV) light using the ENDURO GDS Gel Documentation System (Labnet International Inc., US). A molecular weight marker, 100 bp ladder (PROMEGA, Madison, WI, USA), was used to determine the size of the PCR product. A ChemiDoc Imaging System (Bio-Rad ChemiDocTM MP Imaging System, UK) was used to capture the images using Gene Snap software, version 6.0022. The primers and PCR cycling conditions used in the study are shown in Table 1.
Table 1.
Antibiotic resistance genes (ARGs), primers, and PCR conditions used in this study.
| Target gene | Primer | Primer sequence (5′ ⟶ 3′) | Conditions | Amplicon size (bp) | References | |
|---|---|---|---|---|---|---|
| Tetracycline | tet(A) | TETA-FTETA-R | GCGCTNTATGCGTTGATGCAACAGCCCGTCAGGAAATT | 94°C for 6 min (1x), 94°C for 30 s, 62°C for 30 s, 72°C for 60 s (30x), and 72°C for 6 min | 387 | [31] |
| tet(O) | TETO-FTETO-R | ACGGARAGTTTATTGTATACCTGGCGTATCTATAATGTTGAC | 94°C for 6 min (1x), 94°C for 30 s, 60°C for 30 s, 72°C for 60 s (30x), and 72°C for 6 min | 171 | [32] | |
| tet(X) | TETX-FTETX-R | CCGACACGGAAGTTGAAGAACCTTGGTGAGATGCCATTAGC | 94°C for 6 min (1x), 94°C for 30 s, 60°C for 30 s, 72°C for 60 s (30x), and 72°C for 6 min | 468 | [32] | |
| tet(P) | TETP-FTETP-R | CTTGGATTGCGGAAGAAGAGATATGCCCATTTAACCACGC | 94°C for 6 min (1x), 94°C for 30 s, 63°C for 30 s, 72°C for 60 s (30x), and 72°C for 6 min | 676 | [33] | |
| tet(W) | TETW-FTETW-R | GAGAGCCTGCTATATGCCAGCGGGCGTATCCACAATGTTAAC | 94°C for 6 min (1x), 94°C for 30 s, 64°C for 30 s, 72°C for 60 s (30x), and 72°C for 6 min | 168 | [33] | |
|
| ||||||
| Erythromycin | ermB | ERMB-FERMB-R | GCATTTAACGACGAAACTGGCTGACAATACTTGCTCATAAGTAATGGT | 95°C for 2 min (1x), 95°C for 30 s, 60°C for 45 s, 72°C for 1 min (35x), and 72°C for 7 min | 573 | [34] |
|
| ||||||
| Colistin | mcr-1 | mcr-1-Fmcr-1-R | TATCGCTATGTGCTAAAGCCTGCGTCTGCAGCCACTGGG | 94°C for 5 min and 25 cycles, 94°C for 30 s, 56°C for 1 min, 72°C for 1 min, and 72°C for 5 min | 1139 | [35] |
| mcr-2 | mcr-2-Fmcr-2-R | TATCGCTATGTGCTAAAGCCTGAAAATACTGCGTGGCAGGTAGC | 816 | [35] | ||
| mcr-3 | mcr-3-Fmcr-3-R | CAATCGTTAGTTACACAATGATGAAGAACACATCTAGCAGGCCCTC | 676 | [35] | ||
| mcr-4 | mcr-4-Fmcr-4-R | ATCCTGCTGAAGCATTGATGGCGCGCAGTTTCACC | 405 | [35] | ||
| mcr-5 | mcr-5-Fmcr-5-R | GGTTGAGCGGCTATGAACGAATGTTGACGTCACTACGG | 207 | [35] | ||
|
| ||||||
| Ampicillin | ampC | AmpC FAmpC R | GTGACCAGATACTGGCCACATTACTGTAGCGCCTCGAGGA | 95°C for 2 min, 35 cycles of 95°C for 30 s, 60°C for 45 s, 72°C for 1 min, and 72°C for 7 min | 822 | [36] |
|
| ||||||
| Chloramphenicol | catI | catI FcatI R | GGTGATATGGGATAGTGTTCCATCACATACTGCATGATG | 1 min at 95°C, followed by 40 cycles of 15 s at 95°C, 30 s at 60°C, and 30 s at 72°C | 349 | [37] |
| catII | catII FcatII R | GATTGACCTGAATACCTGGAACCATCACATACTGCATGATG | 567 | [37] | ||
| catIII | catIII FCatIII R | CCATACTCATCCGATATTGACCATCACATACTGCATGATG | 275 | [37] | ||
| catIV | CatIV F catIV R | CCGGTAAAGCGAAATTGTATCCATCACATACTGCATGATG | 451 | [37] | ||
| floR | FloR FFloR R | CGCCGTCATTCCTCACCTTCGATCACGGGCCACGCTGTGTC | 1 min at 95°C, followed by 40 cycles of 15 s at 95°C, 30 s at 50°C, and 30 s at 72°C | 215 | [37] | |
| Integrons | IntI1 | IntI1-FIntI1-R | GCCTTGCTGTTCTTCTACGGGATGCCTGCTTGTTCTACGG | 94°C for 5 min (1x); 30 s at 94°C, 30 s, 55–60°C, 2 min at 72°C (35x), and 5 min at 72°C | 558 | [35] |
| IntI2 | IntI2-FIntI2-R | CACGGATATGCGACAAAAAGGTGTAGCAAACGAGTGACGAAATG | 94°C for 5 min (1x); 94°C for 1 min, 60°C for 1 min, 72°C for 2 min (32x), and 72°C for 10 min | 740 | [35] | |
2.4. Determination of Virulence Genes
Six virulence genes; Cytolethal distending toxin subunits; cdtA, cdtB, and cdtC, Campylobacter adhesion to fibronectin protein (CadF), phospholipase A (pldA), and Campylobacter invasion antigen B (ciaB) were screened from Campylobacter jejuni isolates. The primers and PCR cycling conditions were obtained from the previously published articles [10, 38, 39] and are shown in Table 2. The 25 μL total reaction consisted of 12.5 μL of a 2X DreamTag Green Master Mix (0.4 mM dATP, 0.4 mM dCTP, 0.4 mM dGTP, and 0.4 mM dTTP, 4 Mm MgCl2, and loading buffer), 8.5 μL of nuclease-free water, 2.0 μL of template DNA, and 1.0 μL of each oligonucleotide primer. PCR reactions were performed using the ProFlex PCR System (Applied Biosystems, USA). Amplified PCR products were resolved on a 1% (w/v) agarose gel in a 40 mM Tris (Sigma Aldrich, US), 1X TAE buffer (20 mM acetic acid (Merck, US), and 1 mM EDTA (Merck, South Africa) at pH 8.0), stained with 0.001 μg/mL ethidium bromide and visualized under ultraviolet (UV) light using the ENDURO GDS Gel Documentation System (Labnet International Inc., US).
Table 2.
Primer sequences of virulence genes and PCR conditions used in this study.
| Target gene | Primer | Primer sequence (5′ ⟶ 3′) | Conditions | Cycles | Size (bp) | References |
|---|---|---|---|---|---|---|
| cdtA | CDTA-FCDTA-R | CCTTGTGATGCAAGCAATC ACACTCCATTTGCTTTCTG | 94°C for 15 min, 94°C for 1 min, 49°C for 1 min, and 72°C for 1 min, 72°C for 7 min | 45 | 370 | [39] |
|
| ||||||
| cdtB | CDTB-FCDTB-R | GTTAAAATCCCCTGCTATCAACCA GTTGGCACTTGGAATTTGCAAGGC | 94°C for 15 min, 94°C for 1 min, 51°C for 1 min, and 72°C for 1 min, 72°C for 7 min | 45 | 495 | [39] |
|
| ||||||
| cdtC | CDTCFCDTCR | CGATGAGTTAAAACAAAAAGATA TTGGCATTATAGAAAATACAGTT | 94°C for 15 min, 94°C for 1 min, 48°C for 1 min, and 72°C for 1 min, 72°C for 7 min | 45 | 182 | [39] |
|
| ||||||
| cadF | cadF-F2BcadF-R1B | TTGAAGGTAATTTAGATATGCTAATACCTAAAGTTGAAAC | 95°C for 3 min, 94°C for 30 s, for 30 s, 43°C and 72°C for 1 min, 72°C for 5 min | 45 | 400 | [10] |
|
| ||||||
| ciaB | CIAB-652CIAB R1159 | TGCGAGATTTTTCGAGAATGTGCCCGCCTTAGAACTTACA | 95°C for 3 min, 94°C for 30 s, for 30 s, 54°C and 72°C for 1 min, 72°C for 5 min | 45 | 527 | [38] |
|
| ||||||
| pldA | PLDA-FPLDA-R | AAGAGTGAGGCGAAATTCCAGCAAGATGGCAGGATTATCA | 95°C for 3 min, 94°C for 30 s, for 30 s, 46°C and 72°C for 1 min, 72°C for 5 min | 45 | 385 | [38] |
2.5. Data Analysis
Statistical analysis was carried out using Microsoft Excel 2016 (Microsoft Corporation, Redmond, DC, USA) and Statistical Package for the Social Sciences v. 26 (IBM Corporation, Armonk, NY, USA). The sequenced 16S rRNA gene of the 26 isolates was compared to nucleotide sequences available in GenBank. The closest representative sequences of Campylobacter spp. strains were retrieved and aligned with the sequenced isolates using ClustalW program for phylogenetic analysis. The phylogenetic tree was constructed in the MEGAX package using the maximum likelihood method and Kimura 2-parameter model [40] with a bootstrap analysis of 1000 replicates [41].
3. Results
3.1. Molecular Detection of Virulence Genes
Many isolates carried cdtC (11/26; 42.3%), followed by pldA (9/26; 34.6%), then cdtB (8/26; 30.8%), cdtA (7/26; 26.9%), cadF (6/26; 23.1%), and ciaB (6/26; 23.1%). Multiple virulence genes were observed in seven (n = 7) isolates. The distribution of virulence genes in C. jejuni is presented on the heatmap (Figure 1).
Figure 1.
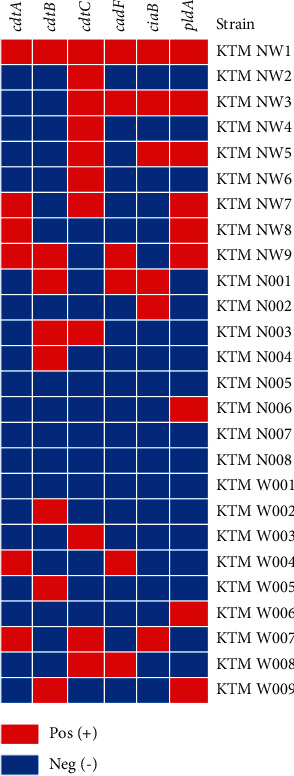
Distribution of the six virulence genes in C. jejuni isolates. Red colour represents the presence, and blue colour represents the absence of the virulence gene.
3.2. Phylogenetic Analysis of Campylobacter jejuni
Based on the 16S rRNA phylogenetic analysis, C. jejuni formed two well-supported monophyletic clades and were separated from other Campylobacter species. All the C. jejuni isolates from this study clustered in a single large monophyletic clade consisting of C. jejuni and C. coli. This clade represents a polytomy with both campylobacter species. Some of the isolates/strains from the current study formed poorly supported clades nested within this polytomy (Figure 2).
Figure 2.
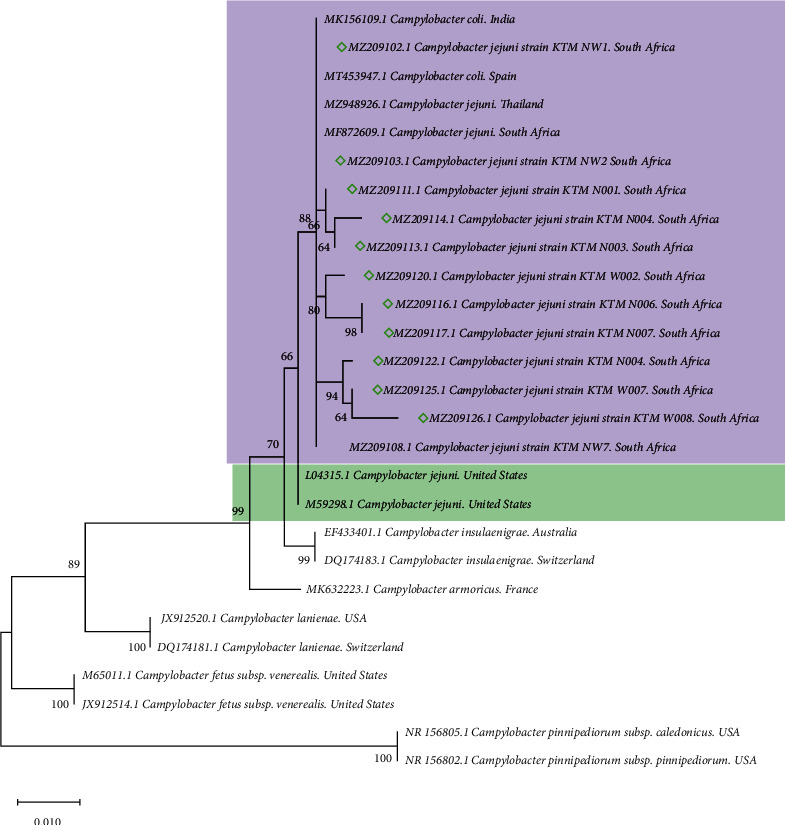
Phylogenetic tree of the 16S rRNA gene constructed by using the maximum likelihood method and Kimura 2-parameter model among Campylobacter species. The node numbers represent the levels of bootstrap support based on 1000 replicates. The scale bar represents 0.010 substitutions per nucleotide position. All positions containing gaps and missing data were eliminated from the dataset (complete deletion option). The diamonds indicate C. jejuni isolates of the current study.
3.3. Antimicrobial Resistance Profile
Out of 26 tested isolates for the occurrence of AMR, a majority (96.1%; n = 25) of the isolates in this study showed resistance to nalidixic acid, followed by erythromycin (84.6%; n = 22), tetracycline (80.7%; n = 21), and ciprofloxacin (19.2%; n = 5) (Table 3).
Table 3.
Distribution of integrons, phenotypic, and genotypic antibiotic resistance in C. jejuni strains.
| Samples ID | Strain | Accession number | Antibiotic class | Resistant genes pattern | Integrase | |
|---|---|---|---|---|---|---|
| IntI I | IntI II | |||||
| 1 | KTM NWI | MZ209102 | NAL, TET, and ERY | tetA, tetO, catI, catII, catIII, and floR | + | + |
| 2 | KTM NW2 | MZ209103 | TET and ERY | tetO, catI, and catII | + | + |
| 3 | KTM NW3 | MZ209104 | NAL, TET, and ERY | mcr-4 and ermB, | + | − |
| 4 | KTM NW4 | MZ209105 | NAL, TET, and CIP | tetA, catII, and catIII | + | − |
| 5 | KTM NW5 | MZ209106 | NAL, TET, and ERY | mcr-4, ampC, ermB, tetA, tetO, catI, catII, catIII, and floR | + | − |
| 6 | KTM NW6 | MZ209107 | NAL, ERY, and CIP | tetA, catI, and catIII | + | + |
| 7 | KTM NW7 | MZ209108 | NAL and TET | tetO and floR | + | − |
| 8 | KTM NW8 | MZ209109 | NAL, TET and ERY | tetO, catI, catIV, and floR | + | + |
| 9 | KTM NW9 | MZ209110 | NAL, TET, ERY, and CIP | ermB | + | + |
| 10 | KTM N001 | MZ209111 | NAL and TET | mcr-4, ermB, tetA, catII, and catIII | + | + |
| 11 | KTM N002 | MZ209112 | NAL, TET, and ERY | mcr-4 and tetO | + | + |
| 12 | KTM N003 | MZ209113 | NAL and ERY | ermB and catI | + | + |
| 13 | KTM N004 | MZ209114 | NAL, TET, and ERY | mcr-4, ampC, ermB, and catIII | + | + |
| 14 | KTM N005 | MZ209115 | NAL, TET, and ERY | mcr-4, ermB, tetO, and catII | + | + |
| 15 | KTM N006 | MZ209116 | NAL and ERY | ermB and floR | + | + |
| 16 | KTM N007 | MZ209117 | NAL, TET, and ERY | ermB | + | + |
| 17 | KTM N008 | MZ209118 | NAL, TET, and ERY | mcr-4, ermB, tetA, and catII | + | − |
| 18 | KTM W001 | MZ209119 | NAL, TET, and ERY | mcr-4, tetA, catIII, and floR | + | + |
| 19 | KTM W002 | MZ209120 | NAL, TET, and ERY | tetO, tetA, and catI | + | + |
| 20 | KTM W003 | MZ209121 | NAL, TET, and ERY | tetO | + | + |
| 21 | KTM W004 | MZ209122 | NAL and ERY | ermB, tetO, catIII, and catIV | + | − |
| 22 | KTM W005 | MZ209123 | NAL, TET, ERY, and CIP | ermB, catI, catIII, and floR | + | − |
| 23 | KTM W006 | MZ209124 | NAL and ERY | ermB and floR | + | + |
| 24 | KTM W007 | MZ209124 | NAL, TET, and ERY | tetO | − | + |
| 25 | KTM W008 | MZ209126 | NAL, TET, ERY, and CIP | tetA and tetO | − | − |
| 26 | KTM W009 | MZ209127 | NAL and TET | ermB and tetO | + | − |
3.4. Detection of Antibiotic Resistance Genes
Out of 19 genes investigated, only 63.2% (n = 12) were positively amplified (Table 3). Most of the isolates harbored chloramphenicol (catI (n = 16; 84.2%), catII (n = 15; 78.9%), catIII (n = 10; 52.6%), catIV (n = 2; 10.5%), floR (n = 10; 52.6%)), erythromycin (ermB (n = 14; 73.7%)), tetracycline (tetO (n = 13; 68.4%), tetA (n = 9; 47.4%)), colistin (mcr-4 (n = 8; 42.1%)), and ampicillin (ampC (n = 2; 10.5%)) resistance genes. Thirty-one percent (n = 8) of the isolates were carrying more than two resistance genes, whereby most isolates carried class I and II integrons. About 92.3% (n = 24) isolates and 65.4% (n = 17) harbored class I and II integrons, respectively. The mcr-1, mcr-2, mcr-3, mcr-5, tet(X), tet(P), and tet(W) genes were not detected. A heatmap was generated to analyze the antibiotic resistance genes of C. jejuni used in this study (Figure 3). Eighty-one percent (n = 21) of the isolates were tetracycline (TET) resistant and carried tetO gene, 33% carried tetA gene, whilst 14.2% carried both tetO and tetA genes. The ermB gene, which confers erythromycin resistance, was present in the same Salmonella isolates (n = 12) that demonstrated phenotypic resistance. The occurrences of mismatch related to erythromycin (ermB) and tetracycline (tetA and tetO) were observed in eight and two isolates, respectively.
Figure 3.
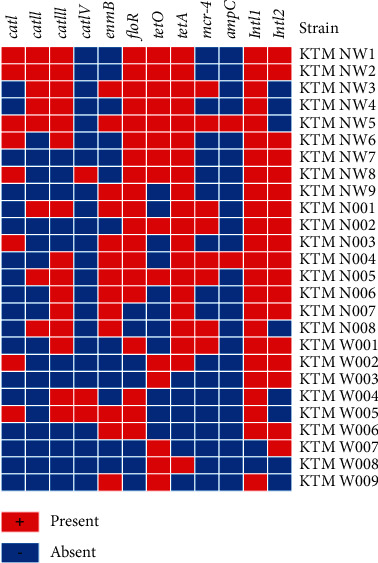
Antibiotic resistance profile of the C. jejuni strains using the 12 amplified antibiotic resistance genes in this study. The red colour represents the presence of antibiotic resistance genes, and blue represents the absence of antibiotic resistance genes.
4. Discussion
This study was designed to determine the antibiotic resistance profiles and occurrence of virulence genes associated with pathogenesis mediated by many virulence factors [38] and the survival of Campylobacter spp. [10]. Six (cdtA, cdtB, cdtC, cadF, pldA, and ciaB) virulence genes were assessed in this study including Campylobacter invasion antigens A, B, and C (cdtA, cdtB, and cdtC), the Cytolethal distending toxin (cdt) gene which encodes for a protein that releases cytotoxins that promote DNA damage [2, 10] and the inhibition of the cell cycle in G2 or M phase [15].
The presence of the cdt gene is linked with the severity of human campylobacteriosis. The cdt, which is encoded by three linked genes, namely, cdtA, cdtB, and cdtC, is one of the most well-studied virulence factors in Campylobacter spp. [10, 42]. In this study, the cdtC gene was the most prevalent gene (42.3%) followed by cdtB and cdtA with 30.8% and 26.9%, respectively. The detection of cytotoxicity genes (cdtA, cdtB, and cdtC) raises food safety concerns. Our results are in line with previous studies where cdtA, cdtB, and cdtC genes were detected in isolates from pigs and chickens [2, 10, 38]. In this study, 23.1% of the isolates harbored Campylobacter adhesion to the fibronectin (cadF) gene, which facilitates adherence to fibronectin in contact regions [15]. This was lower than the results obtained from previous studies where the cadF gene was detected in Campylobacter isolated from communal chicken, patriotic stool and water, human and cattle, children, and raw meat in South Africa [10, 20], Canada [43], Brazil [44], and South Africa [45], with the prevalence of 18.4%, 85.7%, 100%, 37.3%, and 85.7%, respectively.
The prevalence of the pldA gene was 34.6% which is high as compared to other studies conducted in South Africa, whereby this gene was detected at 7.4% [38]. This gene is responsible for adhesion and invasion, whereas the ciaB gene which contributes to the invasion of epithelial cells [3, 46]. In this study, the ciaB gene was detected in 23.1% of the isolates. Multiple virulence genes (n = 7) were observed in 12 isolates, and one isolate harbored all six virulence genes investigated in this study. These results are in line with the reports of Igwaran et al. and Han et al. [6, 47], where the isolates carried more than three virulence genes. The detection of these genes in these isolates indicates the pathogenic potential of the isolates [10]. They attach and invade the host epithelial cells [20], thus leading to detrimental effects on human health [3].
In our phylogenetic analysis, Campylobacter species isolated from avian/chicken hosts formed a well-supported monophyly with other closely related species of Campylobacter from a diverse range of vertebrate hosts. The clustering of C. jejuni and C. coli strains in one clade is similar to the findings reported in previous studies [48, 49]. The sequencing of the 16S rRNA gene is commonly used to differentiate various bacterial species. However, given the highly conserved sequence similarity between the 16S rRNA gene of C. jejuni and C. coli, the phylogenetic analysis results may demonstrate a closer relatedness than what is truly represented in the case. Furthermore, this underlines the importance of using the multigene target approach in discriminating and explaining the evolutionary history of these species globally, as highlighted by numerous authors [50, 51]. This approach could result in well-supported clades, and this polytomy of C. jejuni and C. coli could be resolved. Our two isolates were clustered together and both carried Campylobacter invasion antigens A, B, and C (cdtA, cdtB, cdtC, cadF, and ciaB). Both isolates carried 70% of antibiotic resistance genes including class 1 and 2 integrons.
The use of antibiotics in the poultry industry has become a big concern globally due to the spread of antimicrobial resistance [52]. They are utilized in chicken farming companies for feed efficiency, growth promotion, and disease prevention [53–55]. Their continued use and misuse have resulted in the emergence of antibiotic-resistant Campylobacter [53, 54]. Between 1998 and 2011, ciprofloxacin resistance in clinical C. jejuni isolates from commercial chicken in South Africa increased from 1.4% to 79% [53].
Furthermore, between 1998 and 2011, tetracycline resistance in C. jejuni isolated from commercial poultry increased from 14.2% to 86% in South Africa, according to Basardien, [53]. Despite the fact that the therapeutic use of tetracycline in humans with campylobacteriosis has decreased in recent years [56], the high (80%) detection of the tetracycline resistance gene is not surprising in this study. Furthermore, the high resistance to nalidixic acid, erythromycin (macrolide that inhibits protein synthesis and translation) [57], and ciprofloxacin (fluoroquinolone that acts on bacterial DNA replication) [58] observed in this study raise more concerns to public health. This is because antibiotic resistance in Campylobacter spp., isolated from both humans and animals, has emerged as a major public health concern [11, 59].
The current study also investigated the occurrence of integrons (class I and II) and resistance gene patterns such as catI, catII, catIII, catIV, floR, ermB, tet(A), tet(O), tet(X), tet(P), tet(W), and AmpC of C. jejuni isolated from faeces of slaughter-age broiler chickens. Molecular detection of the antibiotic resistance gene disclosed the presence of 9 genes of which 2 were for tetracycline resistance (tetO (42.3%) and tetA (26.9%)). This finding is similar to previous findings of 92.3%, 83.1%, and 43.5% of tetO gene that was detected in pigs, poultry, and broiler chicken in South Africa, Iran, and China, respectively [6, 60, 61]. More specifically, about 81% of the isolates were tetracycline (TET) resistant and carried the tetO gene, 33% carried the tetA gene, and 14.2% carried both tetO and tetA genes. Tetracycline inhibits protein synthesis [58]. The tetO gene in C. jejuni can be found on the chromosome or, more commonly, on the plasmid pTet [18, 62, 63], and results in binding to the 30S subunit of ribosomes to inhibit protein synthesis [58, 64]. In C. jejuni, tetracycline resistance is encoded on a self-transmissible plasmid [65]. The alteration tetracycline ribosomal target and efflux are two known mechanisms of tetracycline resistance [18].
It is important to note that few (28%) isolates in this study carried the ampC gene encoding for ampicillin resistance which is a lower detection prevalence as compared to other reported studies with a 55% and 63% prevalence in South Africa from meat, milk, and water [6, 45]. Other ARGs detected included catI, catII, catIII, catIV, floR, and ampC and were 61.5%, 57.7%, 38.5%, 7.7%, 38.5%, and 8%, respectively. Colistin (polymyxin that acts in the destructuring bacterial cell membrane) has been screened by different studies phenotypically on Campylobacter spp. [64, 66–68]. However, the current study tested this antibiotic genotypically, resulting in 42.1% of the isolates carrying the mcr-4 gene that encodes for colistin resistance. Some of the isolates were carrying more than two resistance genes. Our findings are also similar to a previous study in South Africa [39] where 33% of the isolates were carrying more than two resistance genes. Multidrug resistance genes discovered in Campylobacter isolates may limit treatment options for campylobacteriosis patients.
The class I integrons have been reported to harbor aminoglycoside resistance genes in C. jejuni [69]. Out of the 26 confirmed isolates, 88% carried the integrase gene (IntI1), a gene-encoding class 1 integrons. Similar findings concerning the predominance of class 1 integrons were reported previously by Chang et al. [70] and El-Aziz et al. [71], whereby 86% and 97% of the Campylobacter isolates from animals and humans carried the intI1 gene in Taiwan and Egypt, respectively.
5. Conclusion
This study revealed the occurrence of virulence and antibiotic resistance genes from C. jejuni isolated from faecal samples obtained from slaughter-age broiler chickens. The antimicrobial resistance tests indicated that C. jejuni isolates used in this study were resistant toward tetracycline, nalidixic acid, ciprofloxacin, and erythromycin and are further harboring antibiotic resistance genes (catI, catII, catIII, catIV, floR, ermB, tetO, tetA, mcr-4, and ampC) from different classes. The class I and II integrons were also detected in this study. According to our knowledge, this is the first study in South Africa to detect integrons II in Campylobacter spp. (C. jejuni). Due to the negative impact on human health of these findings on Campylobacter, cautious use of antibiotics in farming practices must be scaled up. Furthermore, we need to develop appropriate control measures to reduce the emergence of multidrug-resistant strains and to prevent the spread of strains carrying virulent genes.
Data Availability
The datasets generated and analyzed will be available upon request to the corresponding author.
Ethical Approval
The animal and human experimentation and animal care procedures were approved by the ethical committee of NWU (ethics number: NWU-00511-18-A5).
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Ramatla T., Ngoma L., Adetunji M., Mwanza M. Evaluation of antibiotic residues in raw meat using different analytical methods. Antibiotics . 2017;6(4):p. 34. doi: 10.3390/antibiotics6040034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pillay S., Amoako D. G., Abia A. L. K., et al. Characterisation of Campylobacter spp. isolated from poultry in KwaZulu-Natal, South Africa. Antibiotics . 2020;9(2):p. 42. doi: 10.3390/antibiotics9020042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gharbi M., Béjaoui A., Hamda C. B., Ghedira K., Ghram A., Maaroufi A. Distribution of virulence and antibiotic resistance genes in Campylobacter jejuni and Campylobacter coli isolated from broiler chickens in Tunisia. Journal of Microbiology, Immunology, and Infection . 2021 doi: 10.1016/j.jmii.2021.07.001. In press. [DOI] [PubMed] [Google Scholar]
- 4.Mileng K., Ramatla T. A., Ndou R. V., Thekisoe O. M. M., Syakalima M. Isolation and antibiotic sensitivity of Campylobacter species from fecal samples of broiler chickens in North West Province, South Africa. Veterinary World . 2021;14(11):2929–2935. doi: 10.14202/vetworld.2021.2929-2935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hagos Y., Gugsa G., Awol N., et al. Isolation, identification, and antimicrobial susceptibility pattern of Campylobacter jejuni and Campylobacter coli from cattle, goat, and chicken meats in Mekelle, Ethiopia. PLoS One . 2021;16(2) doi: 10.1371/journal.pone.0246755.e0246755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Igwaran A., Okoh A. I. Occurrence, virulence and antimicrobial resistance-associated markers in Campylobacter species isolated from retail fresh milk and water samples in two district municipalities in the Eastern Cape Province, South Africa. Antibiotics . 2020;9(7):p. 426. doi: 10.3390/antibiotics9070426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kouglenou S. D., Agbankpe A. J., Dougnon V., et al. Prevalence and susceptibility to antibiotics from Campylobacter jejuni and Campylobacter coli isolated from chicken meat in southern Benin, West Africa. BMC Research Notes . 2020;13(1):305–306. doi: 10.1186/s13104-020-05150-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Thomas K. M., de Glanville W. A., Barker G. C., et al. Prevalence of Campylobacter and Salmonella in African food animals and meat: a systematic review and meta-analysis. International Journal of Food Microbiology . 2020;315 doi: 10.1016/j.ijfoodmicro.2019.108382.108382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Otigbu A., Clarke A., Fri J., Akanbi E., Njom H. Antibiotic sensitivity profiling and virulence potential of Campylobacter jejuni isolates from estuarine water in the Eastern Cape Province, South Africa. International Journal of Environmental Research and Public Health . 2018;15(5):p. 925. doi: 10.3390/ijerph15050925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bissong M. E. A., Ateba C. N. Detection of virulent thermophilic Campylobacter species in communal chickens. South African Journal of Science . 2019;115(10):1–5. doi: 10.17159/sajs.2019/4771. [DOI] [Google Scholar]
- 11.Shobo C. O., Bester L. A., Baijnath S., Somboro A. M., Peer A. K. C., Essack S. Y. Antibiotic resistance profiles of Campylobacter species in the South Africa private health care sector. The Journal of Infection in Developing Countries . 2016;10(11):1214–1221. doi: 10.3855/jidc.8165. [DOI] [PubMed] [Google Scholar]
- 12.Szczepanska B., Andrzejewska M., Spica D., Klawe J. J. Prevalence and antimicrobial resistance of Campylobacter jejuni and Campylobacter coli isolated from children and environmental sources in urban and suburban areas. BMC Microbiology . 2017;17(1):p. 80. doi: 10.1186/s12866-017-0991-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shin S. Y., Kim K. Y., Park J. H. Survival of Campylobacter jejuni under aerobic condition. Korean Journal of Food Science and Technology . 1998;30(4):916–923. [Google Scholar]
- 14.Pavelquesi S. L. S., de Oliveira Ferreira A. C. A., Rodrigues A. R. M., de Souza Silva C. M., Orsi D. C., da Silva I. C. R. Presence of tetracycline and sulfonamide resistance genes in Salmonella spp: literature review. Antibiotics . 2021;10:p. 1314. doi: 10.3390/antibiotics10111314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Reddy S., Zishiri O. T. Detection and prevalence of antimicrobial resistance genes in Campylobacter spp. isolated from chickens and humans. Onderstepoort Journal of Veterinary Research . 2017;84(1):1–6. doi: 10.4102/ojvr.v84i1.1411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tang M., Zhou Q., Zhang X., et al. Antibiotic resistance profiles and molecular mechanisms of Campylobacter from chicken and pig in China. Frontiers in Microbiology . 2020;11:p. 2722. doi: 10.3389/fmicb.2020.592496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gibreel A., Tracz D. M., Nonaka L., Ngo T. M., Connell S. R., Taylor D. E. Incidence of antibiotic resistance in Campylobacter jejuni isolated in Alberta, Canada, from 1999 to 2002, with special reference to tet (O)-mediated tetracycline resistance. Antimicrobial Agents and Chemotherapy . 2004;48(9):3442–3450. doi: 10.1128/aac.48.9.3442-3450.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Iovine N. M. Resistance mechanisms in Campylobacter jejuni. Virulence . 2013;4(3):230–240. doi: 10.4161/viru.23753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Colles F. M., Cain R. J., Nickson T., et al. Monitoring chicken flock behaviour provides early warning of infection by human pathogen Campylobacter. Proceedings of the Royal Society B: Biological Sciences . 2016;283(1822) doi: 10.1098/rspb.2015.2323.20152323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chukwu M. O., Luther King Abia A. L. K., Ubomba-Jaswa E., Obi L., Dewar J. B. Characterization and phylogenetic analysis of Campylobacter species isolated from paediatric stool and water samples in the Northwest Province, South Africa. International Journal of Environmental Research and Public Health . 2019;16(12):p. 2205. doi: 10.3390/ijerph16122205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Akrami F., Rajabnia M., Pournajaf A. Resistance integrons; a mini review. Caspian Journal of Internal Medicine . 2019;10(4):p. 370. doi: 10.22088/cjim.10.4.370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sin M., Yoon S., Kim Y. B., Noh E. B., Seo K. W., Lee Y. J. Molecular characteristics of antimicrobial resistance determinants and integrons in Salmonella isolated from chicken meat in Korea. The Journal of Applied Poultry Research . 2020;29(2):502–514. doi: 10.1016/j.japr.2019.12.010. [DOI] [Google Scholar]
- 23.Cavicchio L., Dotto G., Giacomelli M., et al. Class 1 and class 2 integrons in avian pathogenic Escherichia coli from poultry in Italy. Poultry Science . 2015;94(6):1202–1208. doi: 10.3382/ps/pev095. [DOI] [PubMed] [Google Scholar]
- 24.Deng Y., Liu X., Bao J., et al. Resistance integrons: class 1, 2 and 3 integrons. Annals of Clinical Microbiology and Antimicrobials . 2015;14(1):45–11. doi: 10.1186/s12941-015-0100-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Xu Z., Li L., Shirtliff M. E., Alam M. J., Yamasaki S., Shi L. Occurrence and characteristics of class 1 and 2 integrons in Pseudomonas aeruginosa isolates from patients in Southern China. Journal of Clinical Microbiology . 2009;47(1):230–234. doi: 10.1128/jcm.02027-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Márquez C., Labbate M., Ingold A. J., et al. Recovery of a functional class 2 integron from an Escherichia coli strain mediating a urinary tract infection. Antimicrobial Agents and Chemotherapy . 2008;52(11):4153–4154. doi: 10.1128/AAC.00710-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Piccirillo A., Dotto G., Salata C., Giacomelli M. Absence of class 1 and class 2 integrons among Campylobacter jejuni and Campylobacter coli isolated from poultry in Italy. Journal of Antimicrobial Chemotherapy . 2013;68(11):2683–2685. doi: 10.1093/jac/dkt242. [DOI] [PubMed] [Google Scholar]
- 28.van Essen-Zandbergen A., Smith H., Veldman K., Mevius D. Occurrence and characteristics of class 1, 2 and 3 integrons in Escherichia coli, Salmonella and Campylobacter spp. in Netherlands. Journal of Antimicrobial Chemotherapy . 2007;59(4):746–750. doi: 10.1093/jac/dkl549. [DOI] [PubMed] [Google Scholar]
- 29.Vizzini P., Manzano M., Farre C., et al. Highly sensitive detection of Campylobacter spp. in chicken meat using a silica nanoparticle enhanced dot blot DNA biosensor. Biosensors and Bioelectronics . 2021;171 doi: 10.1016/j.bios.2020.112689.112689 [DOI] [PubMed] [Google Scholar]
- 30.World Health Organization. Integrated surveillance of antimicrobial resistance in foodborne bacteria: application of a one health approach. 2017. https://www.who.int/foodsafety/publications/agisar_guidanc .
- 31.Jin J. L., Bum J. J., Huh M. D., et al. Detection of tetracycline-resistance determinants by multiplex polymerase chain reaction in Edwardsiella tarda isolated from fish farms in Korea. Aquaculture . 2004;240(1–4):89–100. doi: 10.1016/j.aquaculture.2004.07.025. [DOI] [Google Scholar]
- 32.Aminov R. I., Garrigues-Jeanjean N., Mackie R. I. Molecular ecology of tetracycline resistance: development and validation of primers for detection of tetracycline resistance genes encoding ribosomal protection proteins. Applied and Environmental Microbiology . 2001;67(1):22–32. doi: 10.1128/aem.67.1.22-32.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jiang L., Hu X., Xu T., Zhang H., Sheng D., Yin D. Prevalence of antibiotic resistance genes and their relationship with antibiotics in the Huangpu River and the drinking water sources, Shanghai, China. The Science of the Total Environment . 2013;458–460:267–272. doi: 10.1016/j.scitotenv.2013.04.038. [DOI] [PubMed] [Google Scholar]
- 34.Böckelmann U., Dörries H. H., Ayuso-Gabella M. N., et al. Quantitative PCR monitoring of antibiotic resistance genes and bacterial pathogens in three European artificial groundwater recharge systems. Applied and Environmental Microbiology . 2009;75(1):154–163. doi: 10.1128/AEM.01649-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ramatla T., Mileng K., Ndou R., et al. Molecular detection of integrons, colistin and β-lactamase resistant genes in Salmonella enterica serovars enteritidis and typhimurium isolated from chickens and rats inhabiting poultry farms. Microorganisms . 2022;10(2):p. 313. doi: 10.3390/microorganisms10020313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu D., Liu W., Lv Z., et al. Emerging erm(B)-mediated macrolide resistance associated with novel multidrug resistance genomic islands in Campylobacter. Antimicrobial Agents and Chemotherapy . 2019;63(7) doi: 10.1128/AAC.00153-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hong B., Ba Y., Niu L., et al. A comprehensive research on antibiotic resistance genes in microbiota of aquatic animals. Frontiers in Microbiology . 2018;9:p. 1617. doi: 10.3389/fmicb.2018.01617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sithole V., Amoako D. G., Abia A. L. K., Perrett K., Bester L. A., Essack S. Y. Occurrence, antimicrobial resistance, and molecular characterization of Campylobacter spp. in intensive pig production in South Africa. Pathogens . 2021;10(4):p. 439. doi: 10.3390/pathogens10040439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ngobese B., Zishiri O. T., El Zowalaty M. E. Molecular detection of virulence genes in Campylobacter species isolated from livestock production systems in South Africa. Journal of Integrative Agriculture . 2020;19(6):1656–1670. doi: 10.1016/s2095-3119(19)62844-3. [DOI] [Google Scholar]
- 40.Kimura M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. Journal of Molecular Evolution . 1980;16(2):111–120. doi: 10.1007/bf01731581. [DOI] [PubMed] [Google Scholar]
- 41.Felsenstein J. Phylogenies and the comparative method. The American Naturalist . 1985;125(1):1–15. doi: 10.1086/284325. [DOI] [PubMed] [Google Scholar]
- 42.Kabir S. M. L., Kikuchi K., Asakura M., et al. Evaluation of a cytolethal distending toxin (cdt) gene-based species-specific multiplex PCR assay for the identification of Campylobacter strains isolated from diarrheal patients in Japan. Japanese Journal of Infectious Diseases . 2011;64(1):19–27. doi: 10.7883/yoken.64.19. [DOI] [PubMed] [Google Scholar]
- 43.Biswas D., Hannon S. J., Townsend H. G., Potter A., Allan B. J. Genes coding for virulence determinants of Campylobacter jejuni in human clinical and cattle isolates from Alberta, Canada, and their potential role in colonization of poultry. International Microbiology: The Official Journal of the Spanish Society for Microbiology . 2011;14(1):25–32. doi: 10.2436/20.1501.01.132. [DOI] [PubMed] [Google Scholar]
- 44.Do Nascimento Veras H., Medeiros P. H. Q. S., Ribeiro S. A., et al. Campylobacter jejuni virulence genes and immune-inflammatory biomarkers association with growth impairment in children from North Eastern Brazil. European Journal of Clinical Microbiology & Infectious Diseases . 2018;37(10):2011–2020. doi: 10.1007/s10096-018-3337-0. [DOI] [PubMed] [Google Scholar]
- 45.Igwaran A., Okoh A. I. Campylobacteriosis agents in meat carcasses Collected from two district municipalities in the Eastern Cape Province, South Africa. Foods . 2020;9(2):p. 203. doi: 10.3390/foods9020203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Krause-Gruszczynska M., van Alphen L. B., Oyarzabal O. A., et al. Expression patterns and role of the CadF protein in Campylobacter jejuni and Campylobacter coli. FEMS Microbiology Letters . 2007;274(1):9–16. doi: 10.1111/j.1574-6968.2007.00802.x. [DOI] [PubMed] [Google Scholar]
- 47.Han X., Guan H., Zeng J., et al. Prevalence, antimicrobial resistance profiles and virulence-associated genes of thermophilic Campylobacter spp. isolated from ducks in a Chinese slaughterhouse. Food Control . 2019;1(104):157–166. [Google Scholar]
- 48.Ioannidou V., Ioannidis A., Magiorkinis E., et al. Multilocus sequence typing (and phylogenetic analysis) of Campylobacter jejuni and Campylobacter coli strains isolated from clinical cases in Greece. BMC Research Notes . 2013;6(1):359–410. doi: 10.1186/1756-0500-6-359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gunther N. W., IV, Almond J., Yan X., Needleman D. S. GyrB versus 16S rRNA sequencing for the identification of Campylobacter jejuni, Campylobacter coli, and Campylobacter lari. Journal of Nucleic Acids Investigation . 2011;2(1):p. e7. doi: 10.4081/jnai.2011.2303. [DOI] [Google Scholar]
- 50.Yamamoto S., Harayama S. PCR amplification and direct sequencing of gyrB genes with universal primers and their application to the detection and taxonomic analysis of pseudomonas putida strains. Applied and Environmental Microbiology . 1995;61(3):1104–1109. doi: 10.1128/aem.61.3.1104-1109.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kawasaki S., Fratamico P. M., Wesley I. V., Kawamoto S. Species-specific identification of campylobacters by PCR-restriction fragment length polymorphism and PCR targeting of the gyrase B gene. Applied and Environmental Microbiology . 2008;74(8):2529–2533. doi: 10.1128/aem.00975-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Redondo N., Carroll A., McNamara E. Molecular characterization of Campylobacter causing human clinical infection using whole-genome sequencing: virulence, antimicrobial resistance and phylogeny in Ireland. PLoS One . 2019;14(7) doi: 10.1371/journal.pone.0219088.e0219088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Basardien L. Molecular characterization of Campylobacter isolates from free range and commercial chicken in South Africa. 2012. https://etd.uwc.ac.za/handle/11394/5068 .
- 54.Konkel M. E., Kim B. J., Rivera-Amill V., Garvis S. G. Bacterial secreted proteins are required for the internalization of Campylobacter jejuni into cultured mammalian cells. Molecular Microbiology . 1999;32(4):691–701. doi: 10.1046/j.1365-2958.1999.01376.x. [DOI] [PubMed] [Google Scholar]
- 55.Hafez H. M., Attia Y. A. Challenges to the poultry industry: current perspectives and strategic future after the COVID-19 outbreak. Frontiers in Veterinary Science . 2020;7:p. 516. doi: 10.3389/fvets.2020.00516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Moore J. E., Barton M. D., Blair I. S., et al. The epidemiology of antibiotic resistance in Campylobacter. Microbes and Infection . 2006;8(7):1955–1966. doi: 10.1016/j.micinf.2005.12.030. [DOI] [PubMed] [Google Scholar]
- 57.Vázquez-Laslop N., Mankin A. S. How macrolide antibiotics work. Trends in Biochemical Sciences . 2018;43(9):668–684. doi: 10.1016/j.tibs.2018.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Abushaheen M. A., Muzaheed A. J., Fatani A. J., et al. Antimicrobial resistance, mechanisms and its clinical significance. Disease-a-Month . 2020;66(6) doi: 10.1016/j.disamonth.2020.100971.100971 [DOI] [PubMed] [Google Scholar]
- 59.Said M. M., El-Mohamady H., El-Beih F. M., et al. Detection of gyrA mutation among clinical isolates of Campylobacter jejuni isolated in Egypt by MAMA-PCR. The Journal of Infection in Developing Countries . 2010;4(09):546–554. doi: 10.3855/jidc.963. [DOI] [PubMed] [Google Scholar]
- 60.Papadopoulos D., Petridou E., Papageorgiou K., et al. Phenotypic and molecular patterns of resistance among Campylobacter coli and Campylobacter jejuni isolates, from pig farms. Animals . 2021;11(8):p. 2394. doi: 10.3390/ani11082394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Abdi-Hachesoo B., Khoshbakht R., Sharifiyazdi H., Tabatabaei M., Hosseinzadeh S., Asasi K. Tetracycline resistance genes in Campylobacter jejuni and C. coli isolated from poultry carcasses. Jundishapur Journal of Microbiology . 2014;7(9) doi: 10.5812/jjm.12129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Dasti J. I., Grob U., Pohl S., Lugert R., Weig M., Schmidt-Ott R. Role of the plasmid-encoded tet(O) gene in tetracycline-resistant clinical isolates of Campylobacter jejuni and Campylobacter coli. Journal of Medical Microbiology . 2007;56(6):833–837. doi: 10.1099/jmm.0.47103-0. [DOI] [PubMed] [Google Scholar]
- 63.Taylor D. E. Plasmid-mediated tetracycline resistance in Campylobacter jejuni: expression in Escherichia coli and identification of homology with streptococcal class M determinant. Journal of Bacteriology . 1986;165(3):1037–1039. doi: 10.1128/jb.165.3.1037-1039.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Rossi M., Olkkola S., Roasto M., Kivistö R., Hänninen M.-L. Antimicrobial Resistance and Food Safety . Cambridge, MA, USA: Academic Press; 2015. Antimicrobial resistance and Campylobacter jejuni and C. coli. [DOI] [Google Scholar]
- 65.Page W. J., Huyer G., Huyer M., Worobec E. A. Characterization of the porins of Campylobacter jejuni and Campylobacter coli and implications for antibiotic susceptibility. Antimicrobial Agents and Chemotherapy . 1989;33(3):297–303. doi: 10.1128/aac.33.3.297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Feizabadi M. M., Dolatabadi S., Zali M. R. Isolation and drug-resistant patterns of Campylobacter strains cultured from diarrheic children in Tehran. Japanese Journal of Infectious Diseases . 2007;60(4) [PubMed] [Google Scholar]
- 67.El Baaboua A., El Maadoudi M., Bouyahya A., et al. Prevalence and antimicrobial profiling of Campylobacter spp. isolated from meats, animal, and human feces in Northern of Morocco. International Journal of Food Microbiology . 2021;349 doi: 10.1016/j.ijfoodmicro.2021.109202.109202 [DOI] [PubMed] [Google Scholar]
- 68.Martín-Maldonado B., Montoro-Dasi L., Pérez-Gracia M. T., et al. Wild Bonelli’s eagles (Aquila fasciata) as carrier of antimicrobial resistant Salmonella and Campylobacter in Eastern Spain. Comparative Immunology, Microbiology and Infectious Diseases . 2019;67 doi: 10.1016/j.cimid.2019.101372.101372 [DOI] [PubMed] [Google Scholar]
- 69.Wieczorek K., Osek J. Antimicrobial resistance mechanisms among Campylobacter. BioMed Research International . 2013;2013:12. doi: 10.1155/2013/340605.340605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Chang Y. C., Tien N., Yang J. S., et al. Class 1 integrons and plasmid-mediated multiple resistance genes of the Campylobacter species from pediatric patient of a university hospital in Taiwan. Gut Pathogens . 2017;9(1):50–58. doi: 10.1186/s13099-017-0199-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Abd El-Aziz N. K., Ammar A. M., Hamdy M. M., Gobouri A. A., Azab E., Sewid A. H. First report of aacC5-aadA7Δ4 gene cassette array and phage tail tape measure protein on class 1 integrons of Campylobacter species isolated from animal and human sources in Egypt. Animals: An Open Access Journal from MDPI . 2020;10(11):p. 2067. doi: 10.3390/ani10112067. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and analyzed will be available upon request to the corresponding author.


