Abstract
Several types of vaccines have been developed to prevent the coronavirus disease 2019 (COVID-19). It is important to understand whether demographic and clinical variables affect the effectiveness of various types of vaccines. This study analysed the association between demographic/clinical factors, antibody response and vaccine effectiveness in healthcare workers vaccinated with inactivated virus.
We enrolled 101 healthcare workers who received two doses of inactivated viral vaccine (CoronaVac). Blood samples were analysed at 1, 3, and 5 months after the second dose of vaccination. Data regarding demographic characteristics, medical histories, and clinical parameters were collected by interview and medical examination. In a separate retrospective study, we analysed the incidence of vaccine breakthrough infection on 2714 healthcare workers who received two doses of inactivated viral vaccine. Medical histories and demographic data were collected using a structured self-reported questionnaire.
We found that antibody titres markedly increased at 1 month after vaccination but gradually decreased at 3–5 months post-vaccination. We observed a significant association between age (≥40 years) and antibody level, whereas sex and body mass index (BMI) exhibited no effect on antibody titres. Amongst clinical variables analysed, high blood pressure and history of hypertension were significantly correlated with lower antibody titres. Consistently, we found a significant association in the retrospective study between hypertension and the incidence of breakthrough infection.
In conclusion, our results showed that hypertension is associated with lower antibody titres and breakthrough infection following COVID-19 vaccination. Thus, blood pressure control might be important to improve the efficacy of inactivated virus vaccine.
Keywords: COVID-19, Inactivated viral vaccine, Antibody response, Comorbidity, Hypertension, Breakthrough infection
Abbreviations: COVID-19, coronavirus disease 2019; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; BMI, body mass index; CVD, cardiovascular diseases; DM, diabetes mellitus; RBD, receptor binding domain
1. Introduction
The coronavirus disease 2019 (COVID-19) pandemic is a major health problem causing a heavy burden to health service systems, the economy, and civil societies worldwide. At the end of 2021, there were already more than 275 million COVID-19 confirmed cases reported globally, and COVID-19 was responsible for more than 5.3 million deaths [1]. Vaccines are believed to be one of the most effective ways to tackle the disease and to possibly end the pandemic [2]. There were substantial efforts to develop vaccines against COVID-19 during the first year of the pandemic, and at the beginning of 2021, there were at least three types of vaccine platforms that showed significant protection against COVID-19 based on phase 3 clinical trials. These include adenovirus-based vaccines [3], [4], [5], mRNA vaccines [6], [7] and inactivated viral vaccines [8], [9], [10], [11].
The real-life effectiveness of each vaccine has also been reported. Overall, the WHO-listed COVID-19 vaccines are highly protective against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)-related illness in real-world settings, which are comparable to those reported in the phase III clinical trials. However, it is important to note that more recent data indicate that the protective effect of the vaccine may wane over time, with an increasing number of breakthrough infections observed at 6 months after the second vaccination dose [12], [13].
A number of demographic characteristics, including age, sex, and body mass index (BMI), may play a significant role in immunity development following vaccination [14]. In addition, the presence of diseases such as diabetes mellitus and cardiovascular diseases (CVD) may also influence the immune response to vaccine [15]. For example, uncontrolled diabetes mellitus may lead to a decreased cell-mediated immunity compared to that in normal or well-regulated diabetes. This may contribute to the reduction in immune responses following vaccination [16], [17]. It is widely understood that age, obesity, and several comorbidities, including diabetes mellitus, CVD, and hypertension, are strongly associated in determining disease severity, likely through a dysregulated immune response [18], [19], [20]. Therefore, it is important to understand if the presence of these underlying diseases affects the immunogenicity and effectiveness of the COVID-19 vaccine.
As part of Indonesia’s effort to fight against the pandemic, a national COVID-19 vaccination programme was initiated on 13 January 2021, wherein health care workers were among the priority groups. The inactivated SARS-CoV-2 vaccine (CoronaVac) was the only type of vaccine administered to the health care workers during the first 3 months of the vaccination programme in Indonesia. To understand the presence of underlying diseases in vaccine immunogenicity and effectiveness, we assessed the antibody response following vaccination in healthcare workers in East Java, Indonesia with varying demographic characteristics and presence of underlying disease. We then evaluated the association between comorbidities and the occurrence of vaccine breakthrough infection in the first 6 months post-vaccination.
2. Methods
2.1. Antibody response study
2.1.1. Study participants
We conducted a prospective cohort study involving the health care staff of Dr. Soetomo General Hospital in Surabaya, Indonesia. We recruited 101 healthy, non-pregnant individuals aged 20 years or older. Based on the sample size calculation method used in a previous vaccine trial [21] the sample size of this study would achieve at least 80% statistical power with α = 0.05, assuming multiple endpoints between 2 and 5, a sample size ratio between 1 and 1.5, and a correlation coefficient varied between 0 and 0.9 [21]. All study participants underwent serological testing before receiving the first vaccine dose. They received two doses of inactivated SARS-CoV-2 vaccine (CoronaVac) with a 14–28-day interval between the first and second doses. Follow-up blood tests were conducted at 1, 3, and 5 months after the second dose of vaccination. Individuals infected with SARS-CoV-2 following vaccination, as confirmed using polymerase chain reaction (PCR) testing, were excluded from the study. We gathered demographic data and interviewed participants regarding smoking habits as well as the presence or history of comorbidities (i.e., diabetes mellitus, CVD, liver diseases, allergic disease, asthma, and kidney disease) before they received the first vaccine dose. Blood pressure was measured at the beginning of the study and during follow-up visits. The criteria for hypertension were systolic blood pressure ≥ 140 mmHg and/or diastolic blood pressure ≥ 90 mmHg according to the recommendation of the International Society of Hypertension [22]. This study was initiated when the first participant received the first dose of vaccine on 26 January 2021 and was completed on 29 June 2021, when the last participant underwent final assessment
2.1.2. Serological testing
Blood samples were collected and the level of IgG against the SARS-CoV-2 receptor-binding domain was detected in the serum samples using a commercially available kit (Elecsys Anti-SARS-CoV-2 S, Roche Diagnostics, Germany) according to the manufacturer’s protocol. White blood cell profile, including leucocyte, neutrophil, monocyte, and lymphocyte counts, were determined using an automatic blood analyser (ADVIA2120i Hematology System, Siemens, USA).
2.1.3. Statistical analysis
Data are presented as geometric mean titres and 95% confidence intervals (CI). To analyse post-vaccination changes in IgG level, we used the non-parametric Kruskal-Wallis test followed by post-hoc multiple comparisons. To analyse the effects of demographic characteristics (age, sex, BMI), and comorbidities on the antibody response we used a non-parametric multiple comparisons (Mann-Whitney U) test. To account for multiple comparisons, a P value < 0.0125 was considered significant. To control the possible confounding factors, multiple linear regression analysis was conducted with inclusion of age, sex, BMI, blood pressure, smoking habit, and histories of diabetes mellitus, cardiovascular diseases, allergic disease, asthma and lung diseases. Data on white blood cell profile were analysed using unpaired t-test. In this analysis, a P value less than 0.05 was considered statistically significant. We used GraphPad Prism ver. 9 (GraphPad Software, LLC) to analyse the data.
2.2. Breakthrough infection study
2.2.1. Study participants
We performed a retrospective study to evaluate the incidence of vaccine breakthrough infection among health care staff in Dr. Soetomo General Academic Hospital in Surabaya and Syarifah Ambami Rato Ebu Hospital in Bangkalan, Madura, East Java, Indonesia. A total of 2714 hospital staff (1796 from Dr. Soetomo Hospital and 918 from Syarifah Ambami Hospital), who were fully vaccinated during the study period, participated in this study. This sample size exceeded the minimum of 1537 samples required to estimate incidence rate with relative precision of 5% (ε = 0.05) and 95% confidence interval as described by Lwanga and Lemeshow [23]. A computer-based questionnaire to gather demographic characteristics and comorbidities was sent to the participants. After participants completed the questionnaire, we added the data from the hospital medical record regarding COVID-19 vaccination and COVID-19 PCR test results during the study period (1 February 2021 to 30 October 2021). Vaccine breakthrough infection was defined as COVID-19 infection at more than 14 days after the second dose of vaccination. All study participants were vaccinated with inactivated SARS-CoV-2 vaccine (CoronaVac).
2.2.2. Data analysis
Data were analysed and tabulated using IBM SPSS Statistics software version 25 (IBM Corp., Armonk, NY). The incidence of COVID-19 breakthrough infection was compared between groups of participants according to demographic characteristics and the presence of comorbidities. To determine if vaccine breakthrough infection was associated with age, sex, and BMI, we analysed the odds ratio of acquiring infection between categories in each factor. The odds ratios were determined using the crosstabs function in the SPSS software, while Fisher’s exact test was used to test the statistical significance. To adjust for the confounding effects of each variable, a multivariate logistic regression analysis was performed. A P value < 0.05 was considered statistically significant.
2.3. Ethics statement
This study has been approved and has received ethical approval from the Local Health Research Ethics Committee of Dr. Soetomo General Academic Hospital, Surabaya, Indonesia (No. 0145/KEPK/II/2021). All participants have agreed to participate and signed the informed consent.
3. Results
3.1. Antibody response study
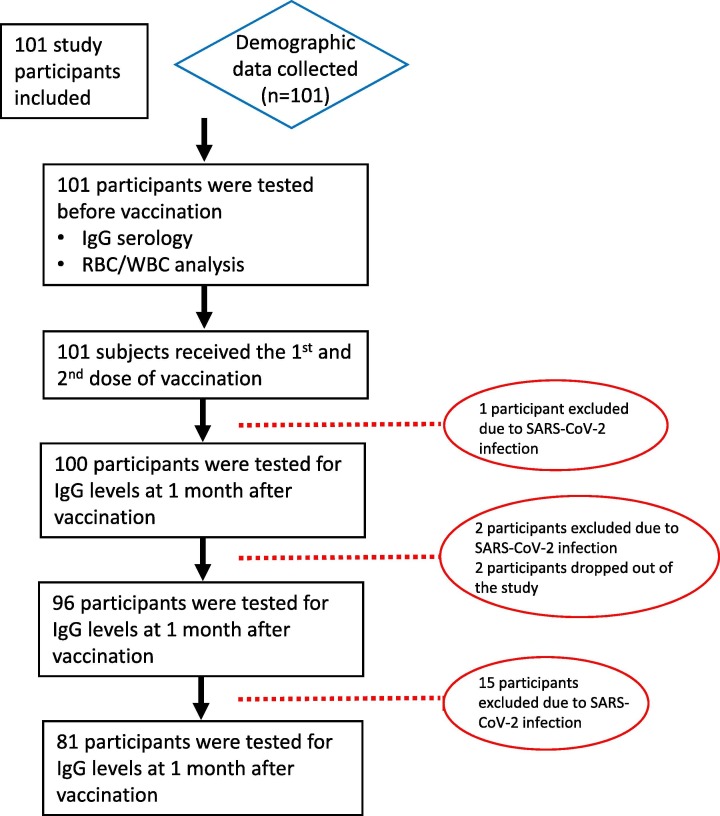
Table 1 shows the baseline demographic data of study participants. From this cohort, 59.5% were male, and the overall average age was 47.7 ± 18.86 years. Blood pressure was monitored at baseline and during the study period. Hypertension (blood pressure ≥ 140/90 mmHg) was detected in 23.7% of participants. Several other comorbidities, including diabetes mellitus (17.8%), CVD (11.9%), allergic diseases (39.6%), asthma (8.9%), and cancer (3%) were also reported. During the follow up, 18 participants were excluded due to infection with SARS-CoV-2, whereas two participants dropped out of the study (Fig. 1 ).
Table 1.
Demographic data of study participants in the antibody response study.
| Study participants (n = 101) | |
|---|---|
| Sex | |
| Male | 60 (59.4 %) |
| Female | 41 (40.6 %) |
| Age at vaccination (y) | 47.7 ± 18.86 |
| Age group | |
| <40 y | 46 (45.5 %) |
| ≥40 y | 55 (54.5 %) |
| BMI group | |
| <25 (non-obese) | 59 (58.5 %) |
| 25–29.99 (obesity class I) | 30 (29.7 %) |
| ≥30 (obesity class II) | 12 (11.9 %) |
| Smoking | |
| Non-smoker | 90 (89.1 %) |
| Active or previous smoker | 11 (10.9 %) |
| Blood pressure | |
| Non-hypertension | 77 (76.3 %) |
| Hypertension (≥140/90) | 24 (23.7 %) |
| Co-morbidities | |
| Diabetes mellitus | 18 (17.8 %) |
| Cardiovascular disease | 12 (11.9 %) |
| Allergic diseases | 40 (39.6 %) |
| Lung disease/asthma | 9 (8.9 %) |
| Chronic kidney disease | 0 |
| Cancer | 3 (3 %) |
| Liver diseases | 0 |
| Autoimmune disease | 0 |
BMI, body mass index.
Fig. 1.
Study profile. Prospective study cohort involving health care workers vaccinated with inactivated SARS-CoV-2 (CoronaVac). Serology assays were conducted at 1, 3, and 5 months following the second vaccination dose. A total of 20 participants were excluded or dropped out during the study, most of whom were excluded due to infection with SARS-CoV-2 during the course of the study. Of the two participants who dropped out, one died due to non-COVID-19 disease, whilst the other one objected to continue to participate due to reluctance to travel to the assessment centre.
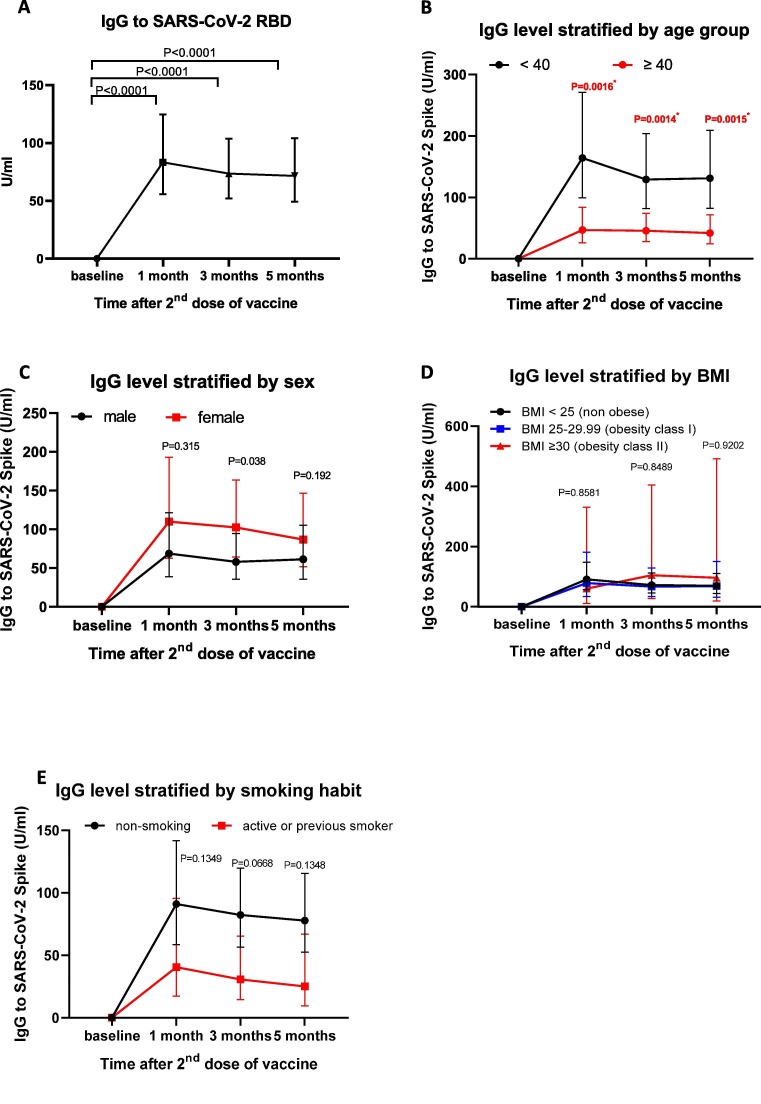
We examined the serum level of IgG antibody against the SARS-CoV-2 receptor binding domain before the first vaccine dose and at 1, 3, and 5 months following the second vaccine dose. As expected, the IgG level was significantly elevated at 1 month post-vaccination at a level that corroborates the findings previously reported in a clinical trial using inactivated SARS-CoV-2 vaccine [9], [11] (Fig. 2 A).
Fig. 2.
Levels of IgG against the SARS-CoV-2 receptor binding domain (RBD) in the health care worker cohort stratified by demographic parameters. A) Concentrations of serum IgG against the SARS-CoV-2 receptor binding domain (RBD) in all participants over 5 months following vaccination. B–E) The dynamics of IgG levels were assessed according to age groups (B), sex (C), body mass index (BMI) (D), and smoking habit (E). Symbols represent geometric mean titres (GMT) of the IgG. Error bars represent 95% CI. Statistical tests used: A) non-parametric Kruskal-Wallis followed by multiple comparisons; B–D) non-parametric multiple comparisons (Mann Whitney U) test. P value < 0.0125 was considered significant.
We then analysed whether age, sex, and BMI affected the vaccine-induced antibody level (Fig. 2B–D). Participants aged 40 years or older showed a significantly lower IgG level compared to those aged below 40 years at all time points (Fig. 2B). In contrast, we observed that antibody response against inactivated viral vaccine was not affected by sex and BMI (Fig. 2C–D). Further, a trend toward lower antibody titres was observed in participants with a smoking habit, although it did not reach statistical significance (Fig. 2E).
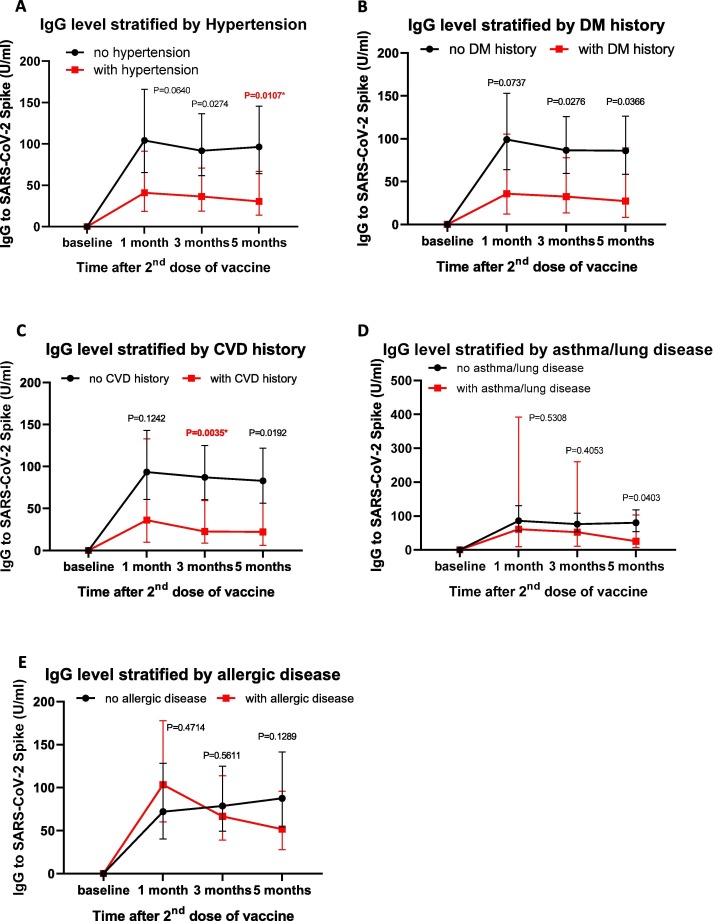
To determine whether co-morbidities were associated with the IgG response, we examined the induction of serum IgG level following vaccination in participants with co-morbidities. We observed a strong trend for a reduced antibody response in participants with hypertension, with the most significant effect noted at 5 months post-vaccination (Fig. 3 A). Despite the trend of a reduced IgG level in participants with a history of diabetes mellitus, the difference at each time point was not statistically significant (Fig. 3B). In addition, we observed a significant effect in IgG levels for participants with CVD history at 3 months, but there was no significant difference at 5 months post-vaccination (Fig. 3C). In contrast, asthma and history of allergic disease did not significantly affect the antibody levels (Fig. 3D–E). To test the confounding effect between variables, a multiple linear regression analysis was conducted. The results showed that blood pressure remained significantly associated with antibody response (P = 0.038, Table 2 ). Overall, our data indicated that among co-morbidities, hypertension significantly influence the antibody response following inoculation with inactivated virus.
Fig. 3.
Quantification of antibody response stratified by the presence of comorbidities. The kinetics of IgG levels before vaccination (baseline) up to 5 months following vaccination are shown in participants with different comorbidities, such as A) hypertension, B) history of diabetes mellitus (DM), C) history of cardiovascular diseases (CVD), D) asthma/lung disease, and E) allergic disease. Data are presented as geometric mean titres (GMT) of IgG levels with 95% CI. Non-parametric multiple comparisons (Mann Whitney U) test was used to assess significance. P value < 0.0125 was considered significant.
Table 2.
Multivariable linear regression analysis of serum antibody level.
| Variable | Regression Coefficient | P value |
|---|---|---|
| Age (years) | 0.576 | 0.917 |
| Male sex (female as reference) | −121.867 | 0.470 |
| BMI | 22.277 | 0.290 |
| Blood pressure (mmHg) | −11.208 | 0.038 |
| Smoking (non-smoking as reference) | −459.120 | 0.069 |
| History of DM (no history of DM as reference) | −59.917 | 0.796 |
| History of CVD (no history of CVD as reference) | −10.040 | 0.969 |
| History of asthma and lung disease (no history of asthma/lung disease as reference) | 106.936 | 0.692 |
| History of allergic disease (no history of allergic disease as reference) | −91.969 | 0.590 |
BMI, body mass index; DM, diabetes mellitus; CVD, cardiovascular diseases.
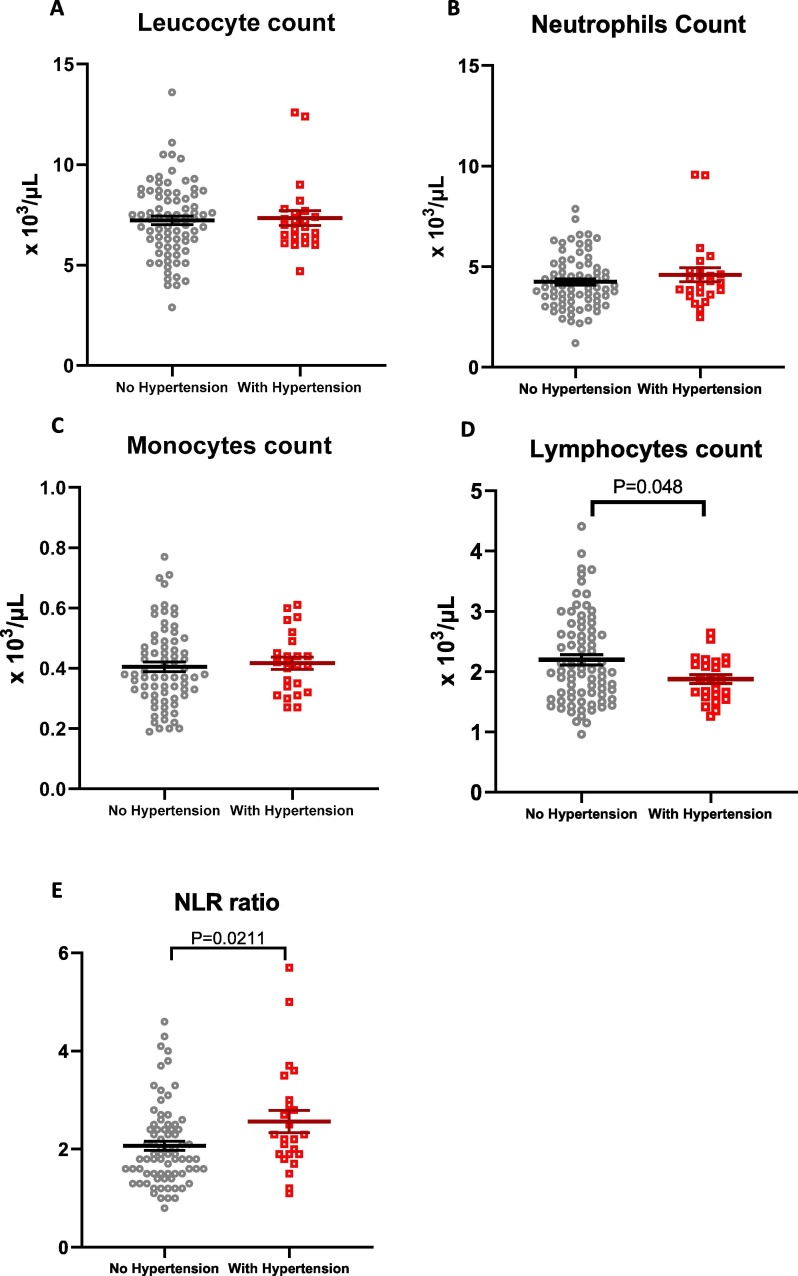
Hypertension is associated with systemic inflammation [24], [25], thus we examined the leucocyte profile of participants with hypertension at baseline (before vaccination). Despite the comparable level of total leucocyte, monocyte, and neutrophil counts (Fig. 4 A–C), we found that individuals with hypertension displayed a lower level of lymphocytes and hence a higher neutrophil-to-lymphocyte ratio compared to participants with normal blood pressure (Fig. 4D–E).
Fig. 4.
White blood cell counts in participants with hypertension. Total numbers of A) leucocytes, B) neutrophils, C) monocytes, and D) lymphocytes and E) the neutrophil-to-lymphocyte ratio (NLR) were analysed in study participants without hypertension (grey dots) and with hypertension (red dots). Hypertension was defined as systolic blood pressure ≥ 140 mmHg and/or diastolic blood pressure ≥ 90 mmHg. Lines and error bars represent mean ± SEM. Unpaired t-test was used to assess significance. P value < 0.05 was considered significant. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
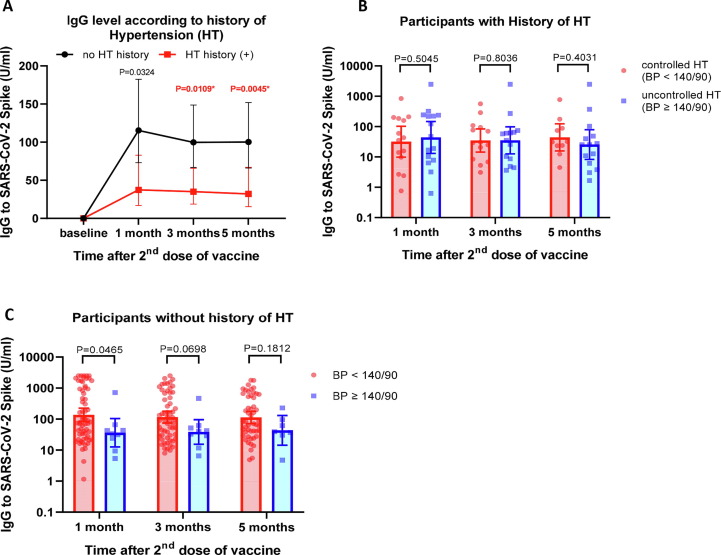
We then evaluated if history of hypertension, whether controlled or uncontrolled, was associated with the antibody response. As shown in Fig. 5 A participants with history of hypertension showed a significantly lower antibody response. We next assessed the effect of controlling blood pressure on antibody response among participants with a history of hypertension. Of the 29 participants who had history of hypertension, 14 individuals showed blood pressure ≤ 140/90 mmHg while 15 still displayed uncontrolled blood pressure (≥140/90) at the time of vaccination. The data in Fig. 5B suggested that a reduction of blood pressure at the normal level did not significantly influence the antibody response in subjects with history of hypertension. Interestingly, in the group of participants with no history of hypertension, there was a trend toward high blood pressure reducing the antibody response (Fig. 5C). Overall, our data suggest that both high blood pressure and history of hypertension are important determinants of lower antibody response following vaccination with inactivated SARS-CoV-2.
Fig. 5.
Effects of history of hypertension on antibody response. A) IgG level stratified by history of hypertension. B) IgG levels in participants with history of hypertension (HT) comparing between participants with controlled HT (blood pressure < 140/90) vs uncontrolled HT (blood pressure ≥ 140/90). C) IgG levels in participants with no history of hypertension (HT) comparing between participants with controlled HT (blood pressure < 140/90) vs uncontrolled HT (blood pressure ≥ 140/90). Data are presented as geometric mean titres (GMT) of IgG levels with 95% CI. Non-parametric multiple comparisons (Mann Whitney U) test was used to assess significance. P value < 0.0125 was considered significant.
3.2. Vaccine breakthrough infection study
Our findings from assessing the antibody response prompted us to evaluate whether there was an association between co-morbidities and the incidence of vaccine breakthrough infection. The demographic data of 2714 participants involved in our retrospective cohort study are shown in Table 3 . The majority of the participants were aged less than 40 years (68.5%), with an average age of 36.4 ± 9.86 years; 53.5% were male. The distribution of participants according to BMI and smoking habit was relatively similar to the cohort group in the antibody study. However, the presence of co-morbidities was generally lower in this cohort, which can be attributed to the younger age of the participants.
Table 3.
Demographic data of study participants of the vaccine breakthrough infection study.
| Study participants (n = 2714) | |
|---|---|
| Sex | |
| Male | 1452 (53.5 %) |
| Female | 1262 (46.5 %) |
| Age (y) | 36.4 ± 9.86 |
| Age group | |
| <40 y | 1859 (68.5 %) |
| ≥40 y | 855 (31.5 %) |
| BMI group | |
| <25 (non-obese) | 1388 (51.1 %) |
| 25–29.99 (obesity class I) | 961 (35.4 %) |
| ≥30 (obesity class II) | 359 (13.2 %) |
| Smoking | |
| Non-smoker | 2338 (86.2 %) |
| Active smoker | 278 (10.2 %) |
| Previous smoker | 98 (3.6%) |
| Co-morbidities (disease history) | |
| Hypertension | 282 (10.4%) |
| Diabetes mellitus | 88 (3.2 %) |
| Cardiovascular disease | 42 (1.5 %) |
| Lung disease/asthma | 233 (8.6 %) |
| Chronic kidney disease | 15 (0.6%) |
| Number of Covid-19 breakthrough infections (total) | 572 (21.1%) |
| Breakthrough infection between 0 and 1 month after 2nd vaccine | 16 (0.6 %) |
| Breakthrough infection between 1 and 3 months after 2nd vaccine | 55 (2 %) |
| Breakthrough infection after 3 months following 2nd vaccine | 501 (18.5%) |
BMI, body mass index.
During the study period, 572 vaccine breakthrough infections with a confirmed PCR test (21.1% of study participants) were reported. The majority of infections (87.6%) occurred more than 3 months after the second vaccination dose (Table 3). We did not find any significant association between age group, sex, and the incidence of breakthrough infection (Table 4 ).
Table 4.
Effects of demographic and clinical variables on vaccine breakthrough infection.
| Number of individuals | Number of COVID-19 infection at more than 14 days after the 2nd vaccination | Unadjusted Odds ratio for COVID-19 infection after vaccination (95% CI) | P value (Fisher’s exact test) | Adjusted odds ratio for COVID-19 infection after vaccination (95% CI) | P value (Multinomial logistic regression) | |
|---|---|---|---|---|---|---|
| Age | ||||||
| <40 y | 1859 | 388 (20.9%) | (reference) | |||
| ≥40 y | 855 | 184 (21.5%) | 1.04 (0.853–1.267) | 0.723 | 0.989 (0.808 – 1.21) | 0.916 |
| Sex | ||||||
| Male | 1452 | 291 (20.0%) | (reference) | |||
| Female | 1262 | 281 (22.3%) | 1.143 (0.950–1.375) | 0.157 | 1.127 (0.930 – 1.366) | 0.222 |
| BMI | ||||||
| <25 (non-obese) | 1388 | 270 (19.5%) | (reference) | |||
| 25–29.99 (obesity class I) | 961 | 222 (23.1%) | 1.244 (1.018–1.520) | 0.035 | 0.816 (0.666 – 1.000) | 0.050 |
| ≥30 (obesity class II) | 359 | 79 (22.0%) | 1.168 (0.880–1.550) | 0.300 | 0.904 (0.677 – 1.207) | 0.495 |
| Smoking | ||||||
| No | 2338 | 501 (21.4%) | (reference) | |||
| Yes | 278 | 55 (19.8%) | 0.904 (0.663–1.234) | 0.587 | 1.032 (0.748 – 1.424) | 0.847 |
| Previous smoker | 98 | 16 (16.3%) | 0.715 (0.415 – 1.233) | 0.257 | 1.340 (0.771 – 2.331) | 0.300 |
| Diabetes mellitus | ||||||
| No | 2626 | 549 (20.9%) | (reference) | |||
| Yes | 88 | 23 (26.1%) | 1.339 (0.824–2.174) | 0.233 | 1.269 (0.757 – 2.129) | 0.366 |
| Hypertension | ||||||
| No | 2432 | 498 (20.5%) | (reference) | |||
| Yes | 282 | 74 (26.2%) | 1.382 (1.041–1.834) | 0.031 | 1.369 (1.009 – 1.859) | 0.044 |
| CVD history | ||||||
| No | 2672 | 565 (21.1%) | (reference) | |||
| Yes | 42 | 7 (16.7%) | 0.746 (0.330–1.688) | 0.571 | 0.596 (0.253 – 1.402) | 0.236 |
| Lung disease/asthma history | ||||||
| No | 2481 | 526 (21.2%) | (reference) | |||
| Yes | 233 | 46 (19.7%) | 0.914 (0.653–1.280) | 0.674 | 0.890 (0.634 – 1.251) | 0.503 |
We then analysed the association of co-morbidities in fully vaccinated participants with the incidence of breakthrough infection. As described in Table 4, our analysis indicated that participants with hypertension showed a significantly higher risk of contracting breakthrough infection than individuals with normal blood pressure (odds ratio: 1.382 (95% CI: 1.041–1.834); P = 0.031, Fisher’s exact test). Multivariable analysis to adjust for the confounding effect of all variables tested consistently supported the finding of the significant effect of hypertension (adjusted odds ratio: 1.369 [95% CI: 1.009–1.859]; P = 0.044, multinomial logistic regression). We recorded 498 (20.5%) breakthrough infections in participants with normal blood pressure (n = 2432) during the study period. In contrast, there were 74 (26.2 %) cases of breakthrough infection among participants with hypertension (n = 282) during the same study period. There were no significant associations between other comorbidities (diabetes mellitus, CVD, asthma/lung disease, and allergic disease) and the incidence of breakthrough infection (Table 4).
4. Discussion
This research consisted of two independent studies: the first was a cohort analysis of 101 participants focusing on antibody response following inoculation of inactivated SARS-CoV-2, and the second was a retrospective observation of 2714 participants who received inactivated SARS-CoV-2, with the goal to assess the incidence of vaccine breakthrough infection with respect to co-morbid conditions. The main finding was consistent between the two studies, in that both hypertension and previous history of hypertension played a major role in determining antibody response and was associated with COVID-19 breakthrough infections among participants who received two vaccine doses.
Following two doses of inactivated viral vaccine, serum IgG levels increased significantly at 1 month after the second dose. However, IgG serum titres began to decrease at 3 and 5 months after the second dose. Vaccine breakthrough infections occurred in the cohort and the incidence increased sharply at 3 months after the second dose. The timing coincided with the second surge of COVID-19 cases in Indonesia, which was likely caused by the delta variant (B.1.617.2) as indicated by the pattern of variant sequence from Indonesian samples submitted to the GISAID database during the study period [26]. When we analysed the associations between demographic/clinical variables and the serum IgG level, we found a significant association between hypertension, previous history of hypertension and a lower antibody response, suggesting that hypertension might play an important role in determining antibody levels following vaccination with inactivated virus.
In the current study, we examined the associations between the serum antibody level and demographic parameters such as age, sex, BMI, and smoking habit. We found that antibody titres in older adults (≥40 years) were significantly lower compared to younger adults (<40 years), which is consistent with emerging reports [12], [27], [28], [29]. This finding can be explained by the presence of immunosenescence with aging [30], [31]. However, since we did not observe any significant difference in the incidence of breakthrough infection between the age group, the reduction in antibody response in relation to age did not translate to the incidence of breakthrough infection amongst health care workers (Table 3). A younger age group participated in our study cohort to evaluate breakthrough infection, relative to the group in the antibody response study. In addition, during the second surge of COVID-19 in Indonesia (June to Sep 2021), the government imposed a strict restriction, including a work-from-home recommendation for older people and those with underlying diseases. Consequently, younger and older groups in the health care staff might have experienced varying levels of viral exposure, which may explain our findings.
The most important finding of this study was the association between hypertension and lower antibody response against inactivated viral vaccine. Indeed, several previous studies reported that antibody titres and vaccine effectiveness were lower in individuals with hypertension [32], [33], however, other studies did not show any association between hypertension and the antibody response to COVID-19 vaccine [14], [34]. Recent evidence suggests that certain types of hypertension may be associated with the immune system. For instance, a significant change in T cell immunometabolism can modulate the metabolic processes and eventually lead to aberrant T cell activation, differentiation, and proliferation, which contribute to the pathogenesis of hypertension [35], [36], [37]. Interestingly, we found that participants with hypertension displayed lower lymphocytes count and a higher neutrophil/lymphocyte ratio compared to participants with normal blood pressure (Fig. 4D–E). Since lymphocytes play an important role in the response against vaccination, this finding may provide a link between hypertension and the antibody response. Our data also suggested that participants with previous history of hypertension, regardless whether the blood pressure was controlled or not, displayed lower antibody levels; this suggests that successful treatment of high blood pressure might not affect the immune response to vaccination.
Our retrospective study evaluating the incidence of breakthrough infection in a larger cohort corroborates with the findings of our antibody response study. Amongst several co-morbidities, history of hypertension appeared to be the most significant factor associated with vaccine breakthrough infection. The lower antibody level found in participants with hypertension and history of hypertension may explain the higher incidence of breakthrough infection in this group.
Studies have shown that hypertension is associated with systemic inflammation [24], [25], [38], [39]. In conditions with chronic systemic inflammation, there are considerable alterations in all tissues and organs that may affect normal cell functions, including the immune response to vaccines [40], [41]. Analysis of cellular subsets and inflammatory cytokine profiles in this setting confirmed that the increased frequency of activated innate immune cells and the elevated levels of pro-inflammatory cytokines correlated with hypo-responsiveness to a vaccine [40]. In addition, recent studies have suggested that metaflammation [42] and perivascular inflammation in hypertension [43] may lead to impaired immune function, consequently influencing the response to vaccination.
Despite the interesting findings, this study also has some limitations. First, in the breakthrough infection study some variables, such as body weight, height, and history of diseases, were self-reported by the participants via the online questionnaire. This might lead to some degree of bias. However, because all of the participants were healthcare workers, it is probably safe to assume that they have a good understanding of health and diseases. Second, in the antibody response study, we did not assess the level of neutralizing antibodies, cellular immunity, and protection against different virus variants, which might be important to be evaluated in future studies.
Overall, our data provide evidence on the effects of demographic factors and comorbidities on the kinetics of antibody response against inactivated SARS-CoV-2 vaccine. This will add beneficial insights on the effectiveness of this type of vaccine, since similar studies focused on evaluating other types of COVID-19 vaccine [12], [13], [14], [15] (e.g. mRNA and adenovirus vaccines). In addition, our data may also provide possible link between hypertension, antibody kinetics, and the incidence of breakthrough infection following inactivated SARS-Cov-2 vaccination. This finding may be useful in establishing strategies to maximise vaccine protection amongst individuals with different demographic characteristics and in the presence of underlying diseases.
Funding source
This study was funded by Mandate Research Grant No: 1043/UN3.15/PT/2021 from Universitas Airlangga, Surabaya. The funder did not have any role in designing the study protocol, data collection, data analysis, interpretation of results, or manuscript writing.
Data sharing statement
The datasets created and analysed during this study are available from the corresponding author upon reasonable request.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Acknowledgement
We would like to thank Antonius Erbano and Eko Praptiningsih (Batara Prodia Laboratory) for the support during the laboratory work. We thank Erlita Puspitasari for blood sampling support, Anisa Octaviani and Astri N Amalia for administrative support.
Authors’ Contributions
GS conceived the original idea, designed the study, supervised biological sample collection, collected and analysed all research data, wrote and edited manuscript; LW designed the study, managed funding, supervised biological sample collection, collected and analysed research data; DP designed the study, supervised data analysis, edited manuscript; KDF, HIG, STH, MEP arranged participant visits, collected clinical data, and supervised blood sample collection; BAM arranged participant visits, collected clinical data and blood samples, and performed additional data analysis; NA, DP, PPN arranged participant visits and collected clinical data and blood samples; CRSP supervised, directed, and coordinated the project; AE supervised the project; DGAS supervised the vaccination programme and the project; DT supervised the ethical clearance; EBR supervised the vaccination programme and the project; EAT supervised the project; JW authorised the vaccination programme at Dr. Soetomo Hospital and supervised the project; CBK, FEW, FM collected clinical and vaccination data at Bangkalan Hospital; NK authorised the vaccination programme at Bangkalan Hospital and supervised the project; AB, DF supervised the project and provided suggestions during manuscript writing; WKN supervised the project; DO designed the study, performed data analysis, and wrote and edited the manuscript. All authors have read and approved the final manuscript.
References
- 1.Dong E., Du H., Gardner L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect Dis. 2020;20(5):533–534. doi: 10.1016/S1473-3099(20)30120-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.COVID-19 vaccines: acting on the evidence. Nat Med 2021;27(2):183. [DOI] [PMC free article] [PubMed]
- 3.Falsey A.R., Sobieszczyk M.E., Hirsch I., Sproule S., Robb M.L., Corey L., et al. Phase 3 Safety and Efficacy of AZD1222 (ChAdOx1 nCoV-19) Covid-19 Vaccine. N Engl J Med. 2021;385(25):2348–2360. doi: 10.1056/NEJMoa2105290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ramasamy M.N., Minassian A.M., Ewer K.J., Flaxman A.L., Folegatti P.M., Owens D.R., et al. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young and old adults (COV002): a single-blind, randomised, controlled, phase 2/3 trial. Lancet. 2020;396(10267):1979–1993. doi: 10.1016/S0140-6736(20)32466-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sadoff J., Gray G., Vandebosch A.n., Cárdenas V., Shukarev G., Grinsztejn B., et al. Safety and Efficacy of Single-Dose Ad26.COV2.S Vaccine against Covid-19. N Engl J Med. 2021;384(23):2187–2201. doi: 10.1056/NEJMoa2101544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Baden L.R., El Sahly H.M., Essink B., Kotloff K., Frey S., Novak R., et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med. 2021;384(5):403–416. doi: 10.1056/NEJMoa2035389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Polack F.P., Thomas S.J., Kitchin N., Absalon J., Gurtman A., Lockhart S., et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med. 2020;383(27):2603–2615. doi: 10.1056/NEJMoa2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Al Kaabi N., Zhang Y., Xia S., Yang Y., Al Qahtani M.M., Abdulrazzaq N., et al. Effect of 2 Inactivated SARS-CoV-2 Vaccines on Symptomatic COVID-19 Infection in Adults: A Randomized Clinical Trial. JAMA. 2021;326(1):35. doi: 10.1001/jama.2021.8565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fadlyana E., Rusmil K., Tarigan R., et al. A phase III, observer-blind, randomized, placebo-controlled study of the efficacy, safety, and immunogenicity of SARS-CoV-2 inactivated vaccine in healthy adults aged 18–59 years: An interim analysis in Indonesia. Vaccine. 2021;39(44):6520–6528. doi: 10.1016/j.vaccine.2021.09.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Han B., Song Y., Li C., Yang W., Ma Q., Jiang Z., et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy children and adolescents: a double-blind, randomised, controlled, phase 1/2 clinical trial. Lancet Infect Dis. 2021;21(12):1645–1653. doi: 10.1016/S1473-3099(21)00319-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang Y., Zeng G., Pan H., Li C., Hu Y., Chu K., et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18–59 years: a randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect Dis. 2021;21(2):181–192. doi: 10.1016/S1473-3099(20)30843-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Levin E.G., Lustig Y., Cohen C., Fluss R., Indenbaum V., Amit S., et al. Waning Immune Humoral Response to BNT162b2 Covid-19 Vaccine over 6 Months. N Engl J Med. 2021;385(24):e84. doi: 10.1056/NEJMoa2114583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Widge A.T., Rouphael N.G., Jackson L.A., Anderson E.J., Roberts P.C., Makhene M., et al. Durability of Responses after SARS-CoV-2 mRNA-1273 Vaccination. N Engl J Med. 2021;384(1):80–82. doi: 10.1056/NEJMc2032195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pellini R., Venuti A., Pimpinelli F., Abril E., Blandino G., Campo F., et al. Initial observations on age, gender, BMI and hypertension in antibody responses to SARS-CoV-2 BNT162b2 vaccine. EClinicalMedicine. 2021;36:100928. doi: 10.1016/j.eclinm.2021.100928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mitsunaga T., Ohtaki Y., Seki Y., Yoshioka M., Mori H., Suzuka M., et al. The evaluation of factors affecting antibody response after administration of the BNT162b2 vaccine: a prospective study in Japan. PeerJ. 2021;9:e12316. doi: 10.7717/peerj.12316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Marfella R., D'Onofrio N., Sardu C., et al. Does poor glycaemic control affect the immunogenicity of the COVID-19 vaccination in patients with type 2 diabetes: the CAVEAT study. Diabetes Obes Metab. 2022;24(1):160–165. doi: 10.1111/dom.14547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Qiao Y.-C., Shen J., He L., Hong X.-Z., Tian F., Pan Y.-H., et al. Changes of regulatory T Cells and of proinflammatory and immunosuppressive cytokines in patients with Type 2 diabetes mellitus: a systematic review and meta-analysis. J Diabetes Res. 2016;2016:1–19. doi: 10.1155/2016/3694957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bae S., Kim S.R., Kim M.-N., Shim W.J., Park S.-M. Impact of cardiovascular disease and risk factors on fatal outcomes in patients with COVID-19 according to age: a systematic review and meta-analysis. Heart. 2021;107(5):373–380. doi: 10.1136/heartjnl-2020-317901. [DOI] [PubMed] [Google Scholar]
- 19.Ciarambino T., Para O., Giordano M. Immune system and COVID-19 by sex differences and age. Womens Health (Lond) 2021;17 doi: 10.1177/17455065211022262. 17455065211022262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yates T., Zaccardi F., Islam N., Razieh C., Gillies C.L., Lawson C.A., et al. Obesity, chronic disease, age, and in-hospital mortality in patients with covid-19: analysis of ISARIC clinical characterisation protocol UK cohort. BMC Infect Dis. 2021;21(1) doi: 10.1186/s12879-021-06466-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yang J., Li J., Wang S., Luo L.i., Liu P. Comparison of three sample size calculation methods for non-inferiority vaccine trials with multiple continuous co-primary endpoints. Hum Vaccin Immunother. 2019;15(1):256–263. doi: 10.1080/21645515.2018.1514221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Unger T., Borghi C., Charchar F., Khan N.A., Poulter N.R., Prabhakaran D., et al. 2020 International Society of Hypertension global hypertension practice guidelines. J Hypertens. 2020;38(6):982–1004. doi: 10.1097/HJH.0000000000002453. [DOI] [PubMed] [Google Scholar]
- 23.Lwanga S., Lemeshow S. World Health Organization; Geneva: 1991. Sample size determination in health studies: a practical manual. [Google Scholar]
- 24.De Miguel C., Rudemiller N.P., Abais J.M., Mattson D.L. Inflammation and hypertension: new understandings and potential therapeutic targets. Curr Hypertens Rep. 2015;17(1):507. doi: 10.1007/s11906-014-0507-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Savoia C., Schiffrin E.L. Inflammation in hypertension. Curr Opin Nephrol Hypertens. 2006;5(3):245–251. doi: 10.1097/01.mnh.0000203189.57513.76. [DOI] [PubMed] [Google Scholar]
- 26.Khare S., Gurry C., Freitas L., et al. GISAID's Role in Pandemic Response. China CDC Wkly. 2021;3(49):1049–1051. doi: 10.46234/ccdcw2021.255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Collier D.A., Ferreira I.A.T.M., Kotagiri P., Datir R.P., Lim E.Y., Touizer E., et al. Age-related immune response heterogeneity to SARS-CoV-2 vaccine BNT162b2. Nature. 2021;596(7872):417–422. doi: 10.1038/s41586-021-03739-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Müller L., Andrée M., Moskorz W., Drexler I., Walotka L., Grothmann R., et al. Age-dependent immune response to the Biontech/Pfizer BNT162b2 coronavirus disease 2019 vaccination. Clin Infect Dis. 2021;73(11):2065–2072. doi: 10.1093/cid/ciab381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Richards N.E., Keshavarz B., Workman L.J., Nelson M.R., Platts-Mills T.A.E., Wilson J.M. Comparison of SARS-CoV-2 antibody response by age among recipients of the BNT162b2 vs the mRNA-1273 vaccine. JAMA Netw Open. 2021;4(9):e2124331. doi: 10.1001/jamanetworkopen.2021.24331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bartleson J.M., Radenkovic D., Covarrubias A.J., Furman D., Winer D.A., Verdin E. SARS-CoV-2, COVID-19 and the Ageing Immune System. Nat Aging. 2021;1(9):769–782. doi: 10.1038/s43587-021-00114-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Crooke S.N., Ovsyannikova I.G., Poland G.A., Kennedy R.B. Immunosenescence and human vaccine immune responses. Immun Ageing. 2019;16:25. doi: 10.1186/s12979-019-0164-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Watanabe M., Balena A., Tuccinardi D., Tozzi R., Risi R., Masi D., et al. Central obesity, smoking habit, and hypertension are associated with lower antibody titres in response to COVID-19 mRNA vaccine. Diabetes Metab Res Rev. 2022;38(1) doi: 10.1002/dmrr.v38.110.1002/dmrr.3465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yelin I, Katz R, Herzel E, et al. Associations of the BNT162b2 COVID-19 vaccine effectiveness with patient age and comorbidities. medRxiv 2021: 2021.03.16.21253686.
- 34.Nomura Y., Sawahata M., Nakamura Y., Kurihara M., Koike R., Katsube O., et al. Age and smoking predict antibody titres at 3 months after the second dose of the BNT162b2 COVID-19 vaccine. Vaccines (Basel) 2021;9(9):1042. doi: 10.3390/vaccines9091042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mattson D.L., Abais-Battad J.M. T Cell immunometabolism and redox signaling in hypertension. Curr Hypertens Rep. 2021;23(12):45. doi: 10.1007/s11906-021-01162-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Moshfegh C.M., Case A.J. The redox-metabolic couple of T lymphocytes: potential consequences for hypertension. Antioxid Redox Signal. 2021;34(12):915–935. doi: 10.1089/ars.2020.8042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rai A., Narisawa M., Li P., Piao L., Li Y., Yang G., et al. Adaptive immune disorders in hypertension and heart failure: focusing on T-cell subset activation and clinical implications. J Hypertens. 2020;38(10):1878–1889. doi: 10.1097/HJH.0000000000002456. [DOI] [PubMed] [Google Scholar]
- 38.Balan R., BĂlĂŞescu E., Ion D.A. Inflammation and arterial hypertension-pathophysiological links and clinical aspects. Curr Health Sci J. 2020;46(4):383–389. doi: 10.12865/CHSJ.46.04.09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Barrows I.R., Ramezani A., Raj D.S. Inflammation, immunity, and oxidative stress in hypertension-partners in crime? Adv Chronic Kidney Dis. 2019;26(2):122–130. doi: 10.1053/j.ackd.2019.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fourati S., Cristescu R., Loboda A., Talla A., Filali A., Railkar R., et al. Pre-vaccination inflammation and B-cell signalling predict age-related hyporesponse to hepatitis B vaccination. Nat Commun. 2016;7(1) doi: 10.1038/ncomms10369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Thomas A.L., Alarcon P.C., Divanovic S., Chougnet C.A., Hildeman D.A., Moreno-Fernandez M.E. Implications of inflammatory states on dysfunctional immune responses in aging and obesity. Front Aging. 2021;2(42) doi: 10.3389/fragi.2021.732414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Liu D., Guan L.i., Zhao Y., Liu Y.u., Sun X., Li H., et al. Association of triglycerides to high-density lipoprotein-cholesterol ratio with risk of incident hypertension. Hypertens Res. 2020;43(9):948–955. doi: 10.1038/s41440-020-0439-8. [DOI] [PubMed] [Google Scholar]
- 43.Iyonaga T., Shinohara K., Mastuura T., Hirooka Y., Tsutsui H. Brain perivascular macrophages contribute to the development of hypertension in stroke-prone spontaneously hypertensive rats via sympathetic activation. Hypertens Res. 2020;43(2):99–110. doi: 10.1038/s41440-019-0333-4. [DOI] [PubMed] [Google Scholar]