Abstract
Background: Patients with rheumatic diseases (RDs) like DM are known to be vulnerable towards various types of infections due to aggressive disease activity mandating high dose immunosuppressive therapy. The severity of COVID-19 in RDs is limited in literature due to the heterogeneous nature of the condition. Therefore, specific details on mortality is essential to navigate any precautions required in the treatment.
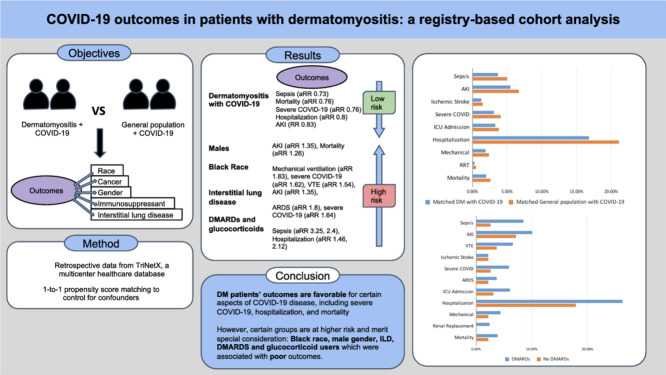
Objectives: To determine outcomes of COVID-19 in DM as compared to controls, and identify the risk association of gender, race, interstitial lung disease, neoplasms, and use of immunosuppressant.
Methods: Retrospective data of individuals with DM and COVID-19 and the general population with COVID-19 between January 2020 to August 2021 was retrieved from the TriNetX database. 1:1 Propensity Score matching was used to adjust for confounders. We assessed COVID-19 outcomes such as mortality, hospitalisation, ICU admission, severe COVID-19, mechanical ventilation (MV), acute kidney injury (AKI), venous thromboembolism (VTE), ischemic stroke, acute respiratory distress syndrome (ARDS), renal replacement therapy (RRT) and sepsis. Subgroup analyses included gender, race, ILD, cancer patients, disease-modifying rheumatic drugs (DMARDs) use, and glucocorticoids (GC) use.
Results: We identified 5,574 DM patients with COVID-19, and 5,574 general population with COVID-19 (controls). DM with COVID-19 had a lower risk of mortality in comparison to controls [RR 0.76], hospitalisation [RR 0.8], severe COVID-19 [RR 0.76], AKI [RR 0.83], and sepsis [RR 0.73]. Males and African Americans were more likely to develop AKI [RR 1.35, 1.65], while African Americans had higher odds for severe COVID-19 [RR 1.62] and VTE [RR 1.54]. DM with ILD group also experienced higher odds for severe COVID-19 infection [RR 1.64], and VTE [RR 2.06].
DM patients receiving DMARDs and glucocorticoids had higher odds for hospitalisation [RR 1.46, 2.12], and sepsis [RR 3.25, 2.4] Subgroup analysis of 5-year neoplasm history amongst DM patients with COVID-19 was inadequate for meaningful comparison.
Conclusion: Dermatomyositis patients without comorbities have reasonable COVID-19 outcomes including mortality and hospitalisation. Black race, male gender, ILD, DMARDS and glucocorticoid users, are associated with poor outcomes.
Keywords: Dermatomyositis, COVID-19, Immunosuppressant, Interstitial lung disease
Graphical abstract
Introduction
The severity of Coronavirus disease 2 (COVID-19) in rheumatic diseases (RDs) is dependent on several factors, such as underlying condition, treatment received, and general debility, apart from age and co-morbidities [1,2]. It is clear that there is a higher risk of COVID-19 on autoimmune RD patients, but the individual disease specific risks may vary considerably [3,4]. Furthermore, the outcomes of COVID-19 in those on immunosuppressive medication for severe RDs such as idiopathic inflammatory myopathies (IIM) are currently unknown [5], [6], [7]. Given that each chronic RD has unique clinical and pathogenic features, it is essential to study outcomes of COVID-19 infection in specific chronic RDs [8].
Idiopathic Inflammatory Myopathies (IIMs) are heterogenous and the specific vulnerability of subsets such as dermatomyositis (DM) is unclear [9], [10], [11], [12], [13]. The predominant symptoms of DM are muscle weakness and rash, with significant frequency of end organ damage such as interstitial lung disease (ILD) and cardiac involvement that can exacerbate the risk of severe COVID-19 outcomes. Furthermore, an emerging spectrum of vasculitis, and vasculopathy has been identified recently in COVID-19, identifying another convergence point with certain severe forms of DM, specifically patients with anti-MDA5 antibodies [14,15]. Moreover, some forms of DM may be predisposed to macrophage activation syndrome (MAS), a condition widely prevalent in severe COVID-19 [16], [17], [18]. DM is a form of interferonopathy, with possible heightened interferon response, which may culminate in accentuated viral clearance. Mutations or polymorphisms in anti-MDA5 have been proposed to be one of the possible pathogenic pathways, creating a gateway for virus driven autoimmunity in certain forms of IIM [19,20]. With these considerations, it is imperative to explore the risk and outcomes of COVID-19 in patients with IIM, specifically its most common subset, DM.
Currently, the data on outcomes of COVID-19 in DM is very limited, and specific details on the mortality of DM patients due to COVID-19 is essential to navigate any precautions required in the treatment [22]. DM is often chronic and debilitating, and this information can be crucial in devising guidelines for specific risk groups [22]. Hence, we investigated the outcomes of COVID-19 in patients with DM compared to the general population to understand whether they have a higher or lower risk for COVID-19 related complications.
Methods
Data source
This study used TriNetX database, a federated database of over 60 million electronic medical records of unique patients from around 48 healthcare organizations (HCOs) at the time of analysis. The TriNetX Research Network database includes variables on diagnoses, procedures, medications, and lab values when possible, using coding such as ICD-10, CPT, RxNorm, and LOINC from all contributing HCOs. All data is aggregated, deidentified, and Health Insurance Portability and Accountability Act-compliant from member HCOs contributing to the TriNetX research network in order to generate statistical summaries. Data from typical HCOs are mostly from large academic health centers in the USA and their affiliates (∼93%) while the rest is from international HCO's. Member HCOs remain anonymous due to a Business Associate Agreement contract signed with TriNetX where rights, consents, approvals, and authority to provide data is possible as long as sources are anonymous, and data is to be used for research purposes. This study was considered non-human subjects research and was waived from IRB review.
Study design
A retrospective comparative cohort study was assembled using previous literature [23], [24], [25], [26] and the TriNetX database to identify 5,578 and 859,166 COVID-19 patients with and without DM respectively. Subsequently propensity matching was undertaken to account for age, sex, race, body mass index, as well as a variety of comorbidities - essential hypertension, chronic lower respiratory diseases, diabetes mellitus, ischemic heart disease, nicotine dependence, cerebrovascular diseases, chronic kidney disease, heart failure and alcohol related disorders. A 1:1 propensity matched cohort of 5,574 COVID-19 patients with and without DM respectively was formed for analysis.
Participants
Adults (≥ 18 yrs) diagnosed with COVID-19 anytime from January 20, 2020 to August 31, 2021 were first identified. Any patient who was vaccinated for COVID-19 prior to infection was excluded. Then patients were divided into DM and non-DM cohorts. COVID-19 diagnosis was identified by validated International Classification of Diseases 10th revision (ICD-10) and positive serology codes as defined by the Center Disease Control and Prevention (CDC) and the World Health Organization (WHO). Identification of Dermatomyositis was based using a modified adaption of a previously published classification system by Leclair et al [23] to work within the confines of the TriNetX database. Inclusion for this study was when ≥ 2 visits with an DM diagnosis code were recorded and a follow up visit occurred within 1 year for patients with an DM code. All of this must have occurred prior to COVID-19 infection.
Outcomes
COVID-19 outcomes included mortality, hospitalization, intensive care unit (ICU) admission, mechanical ventilation, severe COVID-19 (composite of mechanical ventilation or mortality), acute kidney injury (AKI), renal replacement therapy (RRT), acute respiratory distress syndrome (ARDS), ischemic stroke, venous thromboembolism (VTE), and sepsis. All examined outcomes were assessed within 30 days after COVID-19 diagnosis using methods detailed in previous reports [24], [25], [26], [27], [28].
Other variables
Baseline characteristics included age, gender, race, body mass index (BMI), comorbidities (hypertension, chronic lower lung disease, diabetes mellitus, ischemic heart disease, chronic kidney disease, heart failure, cerebrovascular disease, nicotine dependence, alcohol-related disorders, and neoplasms). Based on established risk factors for worse COVID-19 outcomes, subgroup analysis among DM patients with COVID-19 were further evaluated. Examined subgroups included how differences in gender race, presence of interstitial lung disease, history of neoplasms within 5 years of COVID-19 diagnosis, and 1-year recorded use of DMARDs or glucocorticoids affected the risk for COVID-19 related complications
Statistical analysis
Descriptive statistics were used for baseline characteristics that included demographics and comorbidities in the cohorts. A comparative analysis of COVID-19 outcomes between DM with COVID-19 as compared to non-DM with COVID-19 cohorts was performed. To control for confounding variables, propensity score matching was conducted using the greedy nearest-neighbor algorithm with a caliper of 0.1 pooled standard deviations. Cohorts were considered well-matched if the standardized mean difference for confounders was <0.1. Unadjusted and adjusted Risk ratios were calculated for all assessed outcomes. The standard methodology used for this analysis have been described in detail elsewhere [23], [24], [25]. A two-sided p value <0.05 was considered statistically significant. All statistical analyses were performed on the TriNetX platform. Whenever cell counts were <11, TriNetX requires that results be obfuscated in order to protect patient privacy.
Results
Baseline characteristics of DM versus controls
A total of 864,744 adults with COVID-19 were identified during the assessed timeframe. 5,578 (0.6%) of these COVID-19 positive patients had DM. Prior to matching, DM patients with COVID-19 were older (58.45 years ±14.58 years) and there was a higher proportion of females (78.92%), and whites (74.45%) (Table 1 ). Most of the patients had one or more risk factors for COVID-19 like hypertension (66.79%), diabetes mellitus (33.82%), ischemic heart disease (27.18%), heart failure (16.1%), cerebrovascular disease (19.5%), nicotine dependence (23%), history of neoplasms (67%), alcohol related disorder (7.8%), and chronic lower respiratory disease (57%). After matching, the standardized mean difference for all baseline characteristics was <0.1, a sign that confounders were well-controlled.
Table 1.
Baseline demographics and comorbidities of unmatched and matched cohort of myositis with COVID-19 as compared to controls.
| Characteristics | Unmatched DM with COVID-19 | Unmatched General population with COVID-19 | Matched DM with COVID-19 | Matched General population with COVID-19 | |||
|---|---|---|---|---|---|---|---|
| Total numbers | N = 5,578 | N = 859,166 | Standard Mean Difference | N = 5,574 | N =5,574 | Standard Mean Difference | |
| Age at Index | 58.46±14.58 | 47.5±18.62 | 0.656 | 58.45±14.58 | 58.95±15.15 | 0.034 | |
| Female | 4403 (78.94%) | 467889 (54.46%) | 0.538 | 4399 (78.92%) | 4368 (78.36%) | 0.014 | |
| BMI | 32.1±7.58 | 30.57±7.5 | 0.204 | 32.09±7.58 | 32.85±7.72 | 0.099 | |
| RACE | White | 4154 (74.47%) | 529948 (61.68%) | 0.277 | 4150 (74.45%) | 4228 (75.85%) | 0.032 |
| Black | 985 (17.66%) | 156260 (18.19%) | 0.014 | 985 (17.67%) | 917 (16.45%) | 0.032 | |
| COMORBIDITIES | Hypertension | 3727 (66.82%) | 215211 (25.05%) | 0.923 | 3723 (66.79%) | 3747 (67.22%) | 0.009 |
| CKD | 993 (17.8%) | 43365 (5.05%) | 0.409 | 990 (17.76%) | 968 (17.37%) | 0.01 | |
| Diabetes mellitus | 1889 (33.87%) | 104305 (12.14%) | 0.534 | 1885 (33.82%) | 1923 (34.5%) | 0.014 | |
| IHD | 1516 (27.18%) | 62753 (7.3%) | 0.545 | 1515 (27.18%) | 1454 (26.09%) | 0.025 | |
| HF | 898 (16.1%) | 35174 (4.09%) | 0.407 | 897 (16.09%) | 848 (15.21%) | 0.024 | |
| CVD | 1087 (19.49%) | 37116 (4.32%) | 0.482 | 1083 (19.43%) | 1008 (18.08%) | 0.034 | |
| Nicotine Dependence | 1285 (23.04%) | 60529 (7.05%) | 0.459 | 1281 (22.98%) | 1279 (22.95%) | 0.001 | |
| Neoplasms | 3756 (67.34%) | 144764 (16.85%) | 1.19 | 3752 (67.31%) | 3767 (67.58%) | 0.006 | |
| Alcohol Related Disorder | 378 (6.78%) | 19292 (2.25%) | 0.22 | 374 (6.71%) | 328 (5.88%) | 0.034 | |
| Chronic Lower Respiratory Diseases | 3180 (57.01%) | 117805 (13.71%) | 1.016 | 3176 (56.98%) | 3197 (57.36%) | 0.008 | |
| CKD- Chronic kidney disease, IHD- Ischemic heart disease, HF- Heart failure, CVD- Cerebrovascular disease. | |||||||
Comparison of unmatched and matched COVID-19 outcomes
In unmatched analysis, DM patients with COVID-19 had a higher risk of being hospitalized (16.8% vs 14.6%, RR 1.15[1.08,1.22]), ICU admission (3.3% vs 2.4%, RR 1.41[1.23,1.63]), ARDS (1.5% vs 1.1% RR 1.28[1.03,1.59]), Ischemic stroke (1.3% vs 0.6%, RR 2.18[1.73, 2.74]), VTE (2.9% vs 1.5%, RR 1.96[1.69, 2.29]), AKI (5.5% vs 3.9%, RR 1.43[1.28, 1.6]), and sepsis (3.7% vs 3.1%, RR 1.19[1.04, 1.37]) with no differences in other outcomes. In matched analysis, DM patients had decreased mortality in comparison to those without DM in the setting of COVID-19 (2%vs 2.6%, aRR 0.76 [0.6, 0.97]) (Table 2 ). Patients with DM also experienced fewer hospitalizations (16.8% vs. 21.1%, aRR 0.8[0.74,0.86]), acute kidney injury (5.5% vs 6.7%, aRR 0.83 [0.72,0.96]), sepsis (3.7% vs. 5%, aRR 0.73[0.62,0.88]), and overall less severe COVID-19 infections (3.1% vs. 4.1%, aRR 0.76 [0.63,0.92]) when compared to the control group (Table 2). No significant difference was observed in the number of RRT, mechanical ventilation, critical care/ICU admission, ARDS, ischemic stroke and VTE between the two matched cohorts.
Table 2.
Frequency and comparison of adverse outcomes related to COVID-19 infection between propensity-matched myositis cohort with COVID-19 as compared to control.
| Parameters | Unmatched DM with COVID-19 | Unmatched General population with COVID-19 | Matched DM with COVID-19 | Matched General population with COVID-19 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | N = 5,578 | N = 859,166 | Risk Ratio | Risk Difference | p-value | N = 5,574 | N =5,574 | Adjusted Risk Ratio | Risk Difference | p-value |
| Mortality | 2% (112/5578) | 1.8% (15527/859166) | 1.11 (0.92,1.34) | 0.2% (-0.17%,0.57%) | 0.262 | 2% (112/5574) | 2.6% (147/5574) | 0.76 (0.6,0.97) | -0.63% (-1.19%,-0.07%) | 0.028 |
| Renal Replacement Therapy | 0.3% (19/5485) | 0.3% (2630/852378) | 1.12 (0.72,1.76) | 0.04% (-0.12%,0.19%) | 0.615 | 0.3% (19/5482) | 0.5% (26/5457) | 0.73 (0.4,1.31) | -0.13% (-0.37%,0.11%) | 0.289 |
| Mechanical Ventilation | 1.9% (107/5578) | 1.8% (15791/859166) | 1.04 (0.86,1.26) | 0.08% (-0.28%,0.44%) | 0.656 | 1.9% (107/5574) | 2.4% (136/5574) | 0.79 (0.61,1.01) | -0.52% (-1.06%,0.02%) | 0.060 |
| Hospitalization | 16.8% (939/5578) | 14.6% (125783/859166) | 1.15 (1.08,1.22) | 2.19% (1.21%,3.18%) | <0.001 | 16.8% (938/5574) | 21.1% (1174/5574) | 0.8 (0.74,0.86) | -4.23% (-5.69%,-2.78%) | 0.000 |
| Critical Care/ICU Admission | 3.3% (186/5578) | 2.4% (20268/859166) | 1.41 (1.23,1.63) | 0.98% (0.5%,1.45%) | <0.001 | 3.3% (186/5574) | 3.8% (213/5574) | 0.87 (0.72,1.06) | -0.48% (-1.17%,0.21%) | 0.169 |
| ARDS | 1.5% (82/5578) | 1.1% (9866/859166) | 1.28 (1.03,1.59) | 0.32% (0.01%,0.64%) | 0.025 | 1.5% (82/5574) | 1.5% (85/5574) | 0.96 (0.71,1.3) | -0.05% (-0.5%,0.4%) | 0.815 |
| Severe COVID (Mortality+Ventilation) | 3.1% (174/5578) | 2.8% (24329/859166) | 1.1 (0.95,1.28) | 0.29% (-0.17%,0.75%) | 0.197 | 3.1% (174/5574) | 4.1% (229/5574) | 0.76 (0.63,0.92) | -0.99% (-1.68%,-0.29%) | 0.005 |
| Ischemic Stroke | 1.3% (74/5578) | 0.6% (5231/859166) | 2.18 (1.73,2.74) | 0.72% (0.42%,1.02%) | <0.001 | 1.3% (74/5574) | 1.5% (85/5574) | 0.87 (0.64,1.19) | -0.2% (-0.64%,0.24%) | 0.380 |
| VTE | 2.9% (163/5578) | 1.5% (12779/859166) | 1.96 (1.69,2.29) | 1.43% (0.99%,1.88%) | <0.001 | 2.9% (163/5574) | 2.7% (153/5574) | 1.07 (0.86,1.32) | 0.18% (-0.44%,0.8%) | 0.568 |
| AKI | 5.5% (308/5578) | 3.9% (33170/859166) | 1.43 (1.28,1.6) | 1.66% (1.06%,2.26%) | <0.001 | 5.5% (308/5574) | 6.7% (371/5574) | 0.83 (0.72,0.96) | -1.13% (-2.02%,-0.24%) | 0.013 |
| Sepsis | 3.7% (205/5578) | 3.1% (26447/859166) | 1.19 (1.04,1.37) | 0.6% (0.1%,1.09%) | 0.010 | 3.7% (205/5574) | 5% (279/5574) | 0.73 (0.62,0.88) | -1.33% (-2.08%,-0.57%) | 0.001 |
| ICU- Intensive care unit, AKI- Acute kidney injury, ARDS- Acute respiratory distress syndrome, VTE- Venous thromboembolism | ||||||||||
Matched Subgroup analysis of gender
Among the DM patients with COVID-19, males (n=1141) more likely to develop the major complication of AKI with COVID-19 in comparison to females (8.5% vs 6.3%, aRR 1.35 [1.01,1.81]) (Supplementary Table 2). No statistical difference was found in other outcomes between the genders.
Matched Subgroup analysis of race
Among the DM patients with COVID-19, Blacks (n=970) experienced more hospitalizations (20.4% vs. 16.8%, aRR 1.21[1.01,1.46]), mechanical ventilation (3.4% vs 1.9%, aRR 1.83[1.04,3.23]), AKI (8.7% vs. 5.3%, aRR 1.65[1.18,2.31]), VTE (5.6% vs. 3.6%, aRR 1.54[1.02, 2.34]) and overall severe COVID-19 infection (4.3% vs 2.7%, aRR 1.62[1.01, 2.61]) in comparison to whites (Supplementary Table 3). However, the mortality among black patients were similar to white patients.
Matched Subgroup analysis of Interstitial Lung Disease
Among the DM patients with COVID-19, those with ILD (n=382) experienced a higher number of hospitalizations (37.2% vs. 28.5%, aRR 1.3[1.06,1.6]) and severe COVID-19 infection (10.7% vs. 6.5%, aRR 1.64[1.02,2.64]) in comparison to those with DM COVID-19 patients without ILD (Supplementary Table 4). An increased likelihood of VTE was also observed in those with ILD as opposed to those without (8.6% vs. 4.2%, aRR 2.06 [1.15,3.68]) (Supplementary Table 4).
Matched Subgroup analysis of DMARDs
DM patients with COVID-19 and a 1-year history of DMARDs use (n=457) compared to those without DMARD use (n=457) had higher hospitalizations (26.3% vs. 17.9, aRR 1.46[1.14,1.88]), ICU admissions (6.1% vs 3.1%, aRR 2.00[1.07,3.75]), severe COVID-19 (5.9% vs. 2.6%, aRR 2.25 [1.15,4.39]) and sepsis (8.5% vs. 2.6%, aRR 3.25 [1.72,6.13]) (Supplementary Table 5).
Matched Subgroup analysis of neoplasms
The data for the presence and absence of neoplasms in DM patients within 5 years prior to infection (n=969) was inadequate for meaningful comparison, however, intergroup comparison did not yield any difference in outcomes between the two groups (Supplementary Table 6).
Matched Subgroup analysis of glucocorticoids
The occurrence of hospitalization amongst DM patients with COVID-19 with a 1-year history of gluccorcoticoid use (n=690) was found to be higher than those that were not taking glucocorticoids within the past year (17.8% vs. 8.4%, aRR 2.12[1.58,2.85]) (Supplementary Table 7). Patients with glucocorticoid history for one year also had a higher risk of developing AKI (5.4% vs. 2.5%, aRR 2.18), and sepsis (3.5% vs 1.4%, aRR 2.4) (Supplementary Table 7).
Discussion
This was a retrospective cohort analysis of 5,574 patients with DM and COVID-19 versus 859,166 non-DM patients with COVID-19 followed by a 1:1 propensity matched analysis. The COVID-19 outcomes of matched DM patients exhibited lower mortality, hospitalization, severe COVID-19, AKI and sepsis. Therefore, DM could be assumed as a protective factor against COVID-19. However, conventional risk factors such as the male gender and the black race predisposed individuals with DM to worse outcomes. Additionally, matched patients with ILD and those on DMARDs and glucocorticoids fared worse, with higher occurrences of severe COVID-19 infection.
These observations are surprising and almost counter intuitive, as patients with chronic RDs, particularly those with debilitating illnesses such as DM are expected to be more vulnerable to severe COVID-19 [21]. In fact, most patients with IIM have been anxiety ridden and shielding during the entirety of the pandemic [22]. While outcomes of COVID-19 in individual RDs such as RA suggest worse outcomes, it is possible that DM patients were shielding better due to wider awareness of risks incurred by the sheer severity of underlying IIM. The incidence of COVID-19 on IIM patients is relatively low as reported by many studies, however, they are also strongly recommended to adhere to prevention measures and their former treatment plans [27,28].
While favorable outcomes of this study are noteworthy, it seems imperative for physicians to still account for biases while triaging patients with RDs, particularly during waves in the pandemic when bed availability may be limited. The hospitalization and mortality rates in matched DM patients are lower in comparison to the general population in our study according to Table 2. The use of particular immunosuppression medications in DM patients may be a protective factor in this case, as suggested by a previous study done on systemic lupus erythematosus [29]. On the other hand, our results on patients taking glucocorticoids show a higher risk of a multitude of outcomes such as hospitalization, AKI and sepsis. Our analysis on matched DM patients with COVID-19 taking DMARDs also show higher risk in hospitalization, severe COVID-19 outcomes, ICU admissions, and sepsis. Generally, the use of biological DMARDs, such as Ritixumab, are known to be associated with a higher immunosuppressed state which increases the risk of opportunistic infections [39,40]. This is in alignment with trends of higher COVID-19 mortality among other research cohorts with autoimmune diseases like myasthenia gravis and inflammatory RDs on Ritixumab, in contrast to those on conventional DMARDs who survive fairly better [39,41,42]. Ritixumab had also shown severe COVID-19 outcomes in a study done on rheumatoid arthritis patients with COVID-19 [43]. Thus the identification on what kind of immunosuppressants can be protective against COVID-19 should be further investigated. It could also be plausible that pathogenic pathways specific to IIM provide them a distinctive advantage in survival. The pathophysiology being that COVID-19 associated ARDS is caused by the cytokine storm, which is the hyperactivation of T-cells, however, in patients with autoimmune diseases that are being treated with immunosuppressants such as DMARDS, the cytokine storm is inactivated thus preventing the advancement to ARDS. Additionally, interferons which are key antiviral cytokines are elevated in majority of the individuals with autoimmune disease, which may have some protective effect against the SARS-CoV-2 virus [30]. A head-to-head comparison of various RDs with sizable numbers may provide further insights into this hypothesis and pave the way for further in vitro studies to understand this aspect.
One potential area of concern in the setting of COVID-19 seems to be VTE as observed to be higher in matched patients with DM and COVID-19. The risk of VTE is already high in the COVID-19 infection alone, but with the added disease of DM, the risk is greater than in the general population [31,32]. We found an accentuated risk of VTE in the matched subset with ILD, which may be contributed by immobility due to the lung condition. The contribution of VTE in most autoimmune rheumatic diseases is because of chronic inflammation and/or the presence of the antiphospholipid syndrome (APLs) [33]. However, APLs are rare in IIM, and transient in COVID-19 [34]. NETosis is known to contribute to inflammation and thrombosis in both COVID-19 and IIM, and the possibility of its interplay with transient aPLs merits further exploration.
Thrombosis is also an area of overlap for several forms of IIM, such as anti-MDA5 positive disease, which may at times be confused with COVID-19 [35]. However, the current data set does not include details on myositis autoantibodies, limiting further analysis. Whether similar pathogenic pathways in both diseases (COVID-19 and anti-MDA5 positive disease) lead to protective effect in viral clearance is a hypothesis worth exploring. Activated IFN pathways may also accentuate viral clearance, potentially limiting viral load and reducing disease severity. Studies in other IFN driven diseases such as systemic lupus erythematosus may render a clearer picture in this regard.
The current study identified conventional risk factors such as the male gender, which faced an increased risk of AKI in the matched COVID-19 with myositis cohort, and the black ethnicity, which experienced a higher risk of mechanical ventilation, hospitalization, severe COVID-19, VTE and AKI in the matched COVID-19 with myositis cohort. This could be due to a genetic predisposition which is known in this population however absence of individual data is a limitation from TrinetX database [36]. Many studies show that cancer patients with COVID-19 in comparison to cancer patients without COVID-19 have a higher probability of mortality and are strongly recommended to adhere to preventive measures [37,38]. Therefore, more data is required to identify the severe outcomes between DM cancer patients with COVID-19 and those without the infection.
Our study also showed a higher risk of severe COVID-19 outcomes and hospitalization, amongst matched ILD patients versus non-ILD patients. COVID-19 attacks the respiratory system by terminating its host with ARDS. ILD and other structural lung diseases such as COPD are known to contribute to severe COVID-19 [44], [45], [46]. A study done on ILD patients with COVID-19 versus non ILD patients with COVID-19 showed that the former was of a higher risk and the susceptibility of COVID-19 was not different by the type of ILD including that associated with CTD [47].
The limitations in our study stem from the TriNetX database itself, such as the nondisclosure of the health care organizations from which the data is obtained and the dependency of this study data quality of the contributing networks. The cohort was classified through ICD codes and therefore would not necessarily meet the current classification for IIM. This was evident in the approach our study used to identify DM patients when utilizing a modified adaption of the algorithm proposed by Leclair et al. Since identification was based on ICD 10 codes without any indication on whether DM was diagnosed by a specialist (dermatologist, internist, neurologist, or rheumatologist) as in the Leclair et al algorithm, it is possible that some patients may have been misclassified as DM and included in this study [45], [46]. Additionally the proportion of cancer was overrepresented in our study population as 5-year history of both benign and malignant neoplasms prior to COVID-19 infection were included in the analysis. Therefore it was not possible to discern current cancer status or whether patients had myositis associated cancer due to the aggregate nature of the dataset.. Further limitations include not being able to analyze individualized data, the lack of information about disease activity at time of infection, and severity of DM in patients. Our results are on a large-scale database including multiple healthcare centers with deidentified data, therefore, our sample is representative and generalizable. The results were compared against propensity matching which allowed us to identify additional factors such as sex, age, and comorbidities in determining poor COVID-19 outcomes.
Conclusion
Dermatomyositis patients without comorbities have reasonable COVID-19 outcomes, including a lower risk of severe COVID-19, hospitalization, and even mortality, among others. The intake of DMARDs and glucocorticoids is associated with higher sepsis and AKI. The black race, male gender, and ILD, are also associated with poor outcomes.
Contributors
LG, HP and RR designed the study with inputs from SA and SK. HK, HP and RR performed statistical analyses. HK, HP, SA, CK, LG, and MA drafted the manuscript. All authors reviewed the study design and contributed to data interpretation and critical revision of the article. All authors approved the version of the article to be published. HP, and RR verified the underlying data.
Data availability
Data used cannot be made publicly available due to data-use agreement. Data request can be made to TriNetX.
Key messages
Dermatomyositis (DM) patients have fewer risks of COVID infection compared to the public, however, certain subgroups with DM have worse outcomes. Patients who were older, black, or had a history of interstitial lung disease experienced higher risks of severe COVID-19. DMARD and glucocorticoid use was associated with frequent hospitalisations and severe sepsis.
Declaration of Competing Interest
SA has received honorarium as speaker for Pfizer, DrReddy's, Cipla and Novartis, has received an APLAR research grant, all unrelated to the current study, and has no other potential conflicts of interest. SK has received congress travel, accommodation, and participation fee support (12th Anatolian Rheumatology Days) from Abbvie. All other authors declare no competing interests relevant to the manuscript.
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.semarthrit.2022.152034.
Appendix. Supplementary materials
References
- 1.Biswas M, Rahaman S, Biswas TK, Haque Z, Ibrahim B. Association of Sex, Age, and Comorbidities with Mortality in COVID-19 Patients: A Systematic Review and Meta-Analysis. Intervirology. 2021;64(1):36–47. doi: 10.1159/000512592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bhaskaran K, Bacon S, Evans SJ, et al. Factors associated with deaths due to COVID-19 versus other causes: population-based cohort analysis of UK primary care data and linked national death registrations within the OpenSAFELY platform. Lancet Reg Heal - Eur. 2021;6 doi: 10.1016/j.lanepe.2021.100109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Liu Y, Sawalha AH, Lu Q. COVID-19 and autoimmune diseases. Curr Opin Rheumatol. 2021;33(2):155–162. doi: 10.1097/BOR.0000000000000776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Belsky JA, Tullius BP, Lamb MG, Sayegh R, Stanek JR, Auletta JJ. COVID-19 in immunocompromised patients: A systematic review of cancer, hematopoietic cell and solid organ transplant patients. J Infect. 2021;82(3):329–338. doi: 10.1016/j.jinf.2021.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Thng ZX, De Smet MD, Lee CS, et al. COVID-19 and immunosuppression: A review of current clinical experiences and implications for ophthalmology patients taking immunosuppressive drugs. Br J Ophthalmol. 2021;105(3):306–310. doi: 10.1136/bjophthalmol-2020-316586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fung M, Babik JM. COVID-19 in Immunocompromised Hosts: What We Know so Far. Clin Infect Dis. 2021;72(2):340–350. doi: 10.1093/cid/ciaa863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Monti S, Balduzzi S, Delvino P, Bellis E, Quadrelli VS, Montecucco C. Clinical course of COVID-19 in a series of patients with chronic arthritis treated with immunosuppressive targeted therapies. Ann Rheum Dis. 2020;79(5):667–668. doi: 10.1136/annrheumdis-2020-217424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Certain Medical Conditions and Risk for Severe COVID-19 Illness | CDC. Accessed June 25, 2021. https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html.
- 9.Mehta B, Pedro S, Ozen G, et al. Serious infection risk in rheumatoid arthritis compared with non-inflammatory rheumatic and musculoskeletal diseases: A US national cohort study. RMD Open. 2019;5(1) doi: 10.1136/rmdopen-2019-000935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chatterjee R, Mehta P, Agarwal V, Gupta L. High burden of infections in Indian patients with Idiopathic Inflammatory Myopathy: validation of observations from the MyoCite dataset. Rheumatology. 2021 doi: 10.1093/rheumatology/keab015. Published online January 26. [DOI] [PubMed] [Google Scholar]
- 11.Mehta P, Agarwal V, Gupta L. High early mortality in idiopathic inflammatory myopathies: results from the inception cohort at a tertiary care centre in northern India. Rheumatology. 2021 doi: 10.1093/rheumatology/keab001. Published online January 25. [DOI] [PubMed] [Google Scholar]
- 12.Muhammed H, Gupta L, Zanwar AA, et al. Infections Are Leading Cause of In-Hospital Mortality in Indian Patients With Inflammatory Myopathy. J ClinRheumatol. 2021;27(3):114–119. doi: 10.1097/RHU.0000000000001214. [DOI] [PubMed] [Google Scholar]
- 13.Baig S, Paik JJ. Inflammatory muscle disease – An update. Best Pract Res Clin Rheumatol. 2020;34(1) doi: 10.1016/j.berh.2019.101484. [DOI] [PubMed] [Google Scholar]
- 14.Pauling JD, Skeoch S, Paik JJ. The clinicoserological spectrum of inflammatory myopathy in the context of systemic sclerosis and systemic lupus erythematosus. Indian J Rheumatol. 2020;15(6):S81–S90. doi: 10.4103/injr.injr_136_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pauling JD, Christopher-Stine L. The aetiopathogenic significance, clinical relevance and therapeutic implications of vasculopathy in idiopathic inflammatory myopathy. Revmatol. 2021;60(4):1593–1607. doi: 10.1093/rheumatology/keaa816. [DOI] [PubMed] [Google Scholar]
- 16.Poddighe D, Dauyey K. Macrophage activation syndrome in juvenile dermatomyositis: a systematic review. Rheumatol Int. 2020;40(5):695–702. doi: 10.1007/s00296-019-04442-1. [DOI] [PubMed] [Google Scholar]
- 17.Schulert GS. The storm beneath the storm: MAS-HLH in inflammatory myopathies. J Rheumatol. 2020;47(10):1461–1463. doi: 10.3899/jrheum.191274. [DOI] [PubMed] [Google Scholar]
- 18.Kishida D, Sakaguchi N, ichi Ueno K, et al. Macrophage activation syndrome in adult dermatomyositis: a case-based review. Rheumatol Int. 2020;40(7):1151–1162. doi: 10.1007/s00296-020-04590-9. [DOI] [PubMed] [Google Scholar]
- 19.Gupta P, Kharbanda R, Lawrence A, Gupta L. Systemic flare and cutaneous ulceration following cytomegalovirus infection in a patient with anti-melanoma differentiation-associated protein 5 (MDA5) associated myositis: Diagnostic challenge during the time of coronavirus disease (COVID-19) pandemic. Egypt Rheumatol. 2021;43(4):271–274. doi: 10.1016/j.ejr.2021.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rathore U, Haldule S, Gupta L. Psoriasiform rashes as the first manifestation of anti-MDA5 associated myositis. Rheumatology. 2020;7 doi: 10.1093/rheumatology/keaa821. Published online December. [DOI] [PubMed] [Google Scholar]
- 21.Saud A, Naveen R, Aggarwal R, Gupta L. COVID-19 and Myositis: What We Know So Far. CurrRheumatol Rep. 2021;23(8):63. doi: 10.1007/s11926-021-01023-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gupta L, Lilleker JB, Agarwal V, Chinoy H, Aggarwal R. COVID-19 and myositis-unique challenges for patients. Rheumatol (United Kingdom) 2021;60(2):907–910. doi: 10.1093/rheumatology/keaa610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Leclair Valérie, Svensson John, Lundberg Ingrid E., Holmqvist Marie. Acute Coronary Syndrome in Idiopathic Inflammatory Myopathies: A Population-based Study. J Rheumatol. 2019;46(11):1509–1514. doi: 10.3899/JRHEUM.181248. [DOI] [PubMed] [Google Scholar]
- 24.Raiker R, Pakhchanian H, Hussain A, Deng M. Outcomes of COVID-19 in patients with skin cancer. Br J Dermatol. 2021 doi: 10.1111/bjd.20386. Published online June 17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Singh S, Bilal M, Pakhchanian H, Raiker R, Kochhar GS, Thompson CC. Impact of Obesity on Outcomes of Patients With Coronavirus Disease 2019 in the United States: A Multicenter Electronic Health Records Network Study. Gastroenterology. 2020;159(6):2221–2225. doi: 10.1053/j.gastro.2020.08.028. e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pakhchanian H, Raiker R, Mukherjee A, Khan A, Singh S, Chatterjee A. Outcomes of COVID-19 in CKD Patients: A Multicenter Electronic Medical Record Cohort Study. Clin J Am SocNephrol. 2021;16(5):785–786. doi: 10.2215/CJN.13820820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Raiker Rahul, et al. Vol. 51. WB Saunders; 2021. Outcomes of COVID-19 in patients with rheumatoid arthritis: a multicenter research network study in the United States. (Seminars in arthritis and rheumatism). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zen M, Fuzzi E, Astorri D, et al. SARS-CoV-2 infection in patients with autoimmune rheumatic diseases in northeast Italy: A cross-sectional study on 916 patients. J Autoimmun. 2020;112 doi: 10.1016/j.jaut.2020.102502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Favalli E.G., Monti S., Ingegnoli F., Balduzzi S., Caporali R., Montecucco C. Incidence of COVID-19 in Patients With Rheumatic Diseases Treated With Targeted Immunosuppressive Drugs: What Can We Learn From Observational Data? Arthritis Rheumatol. 2020;72:1600–1606. doi: 10.1002/art.41388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Spihlman AP, Gadi N, Wu SC, Moulton VR. COVID-19 and Systemic Lupus Erythematosus: Focus on Immune Response and Therapeutics. Front Immunol. 2020;11:2861. doi: 10.3389/fimmu.2020.589474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Goel RR, Kotenko SV., Kaplan MJ. Interferon lambda in inflammation and autoimmune rheumatic diseases. Nat Rev Rheumatol. 2021;17(6):349–362. doi: 10.1038/s41584-021-00606-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.D'Silva KM, Jorge A, Cohen A, et al. COVID-19 Outcomes in Patients With Systemic Autoimmune Rheumatic Diseases Compared to the General Population: A US Multicenter, Comparative Cohort Study. Arthritis Rheumatol. 2021;73(6):914–920. doi: 10.1002/art.41619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Malas MB, Naazie IN, Elsayed N, Mathlouthi A, Marmor R, Clary B. Thromboembolism risk of COVID-19 is high and associated with a higher risk of mortality: A systematic review and meta-analysis. EClinicalMedicine. 2020:29–30. doi: 10.1016/j.eclinm.2020.100639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kim SC, Schneeweiss S, Liu J, Solomon DH. Risk of venous thromboembolism in patients with rheumatoid arthritis. Arthritis Care Res. 2013;65(10):1600–1607. doi: 10.1002/acr.22039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gupta L, Zanwar A, Ahmed S, Aggarwal A. Outcomes of Pregnancy in Women With Inflammatory Myositis: A Cohort Study From India. J ClinRheumatol. 2020;26(5):165–168. doi: 10.1097/RHU.0000000000000996. [DOI] [PubMed] [Google Scholar]
- 36.Mehta P, Machado PM, Gupta L. Understanding and managing anti-MDA 5 dermatomyositis, including potential COVID-19 mimicry. RheumatolInt 2021 416. 2021;41(6):1021–1036. doi: 10.1007/S00296-021-04819-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Charoenngam N, Ilori TO, Holick MF, Hochberg NS, Apovian CM. Self-identified Race and COVID-19-Associated Acute Kidney Injury and Inflammation: a Retrospective Cohort Study of Hospitalized Inner-City COVID-19 Patients. J Gen Intern Med. 2021:1. doi: 10.1007/S11606-021-06931-1. Published online. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Saini KS, Tagliamento M, Lambertini M, et al. Mortality in patients with cancer and coronavirus disease 2019: A systematic review and pooled analysis of 52 studies. Eur J Cancer. 2020;139:43. doi: 10.1016/J.EJCA.2020.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Erdal GS, Polat O, Erdem GU, et al. The mortality rate of COVID-19 was high in cancer patients: a retrospective single-center study. Int J ClinOncol 2021 265. 2021;26(5):826–834. doi: 10.1007/S10147-021-01863-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bachiller-Corral J, Boteanu A, Garcia-Villanueva MJ, et al. Risk of Severe COVID-19 Infection in Patients With Inflammatory Rheumatic Diseases. J Rheumatol. 2021;48(7) doi: 10.3899/jrheum.200755. jrheum.200755. [DOI] [PubMed] [Google Scholar]
- 41.Sparks JA, Wallace ZS, Seet AM, et al. Associations of baseline use of biologic or targeted synthetic DMARDs with COVID-19 severity in rheumatoid arthritis: Results from the COVID-19 Global Rheumatology Alliance physician registry. Ann Rheum Dis. 2021;0:62. doi: 10.1136/annrheumdis-2021-220418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jakubíková M, Týblová M, Tesař A, et al. Predictive factors for a severe course of COVID-19 infection in myasthenia gravis patients with an overall impact on myasthenic outcome status and survival. Eur J Neurol. 2021 doi: 10.1111/ene.14951. Published online June 25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Gupta L, Misra DP, Agarwal V, Balan S, Agarwal V. Management of rheumatic diseases in the time of covid-19 pandemic: Perspectives of rheumatology practitioners from India. Ann Rheum Dis. 2021;80(1):E1. doi: 10.1136/annrheumdis-2020-217509. [DOI] [PubMed] [Google Scholar]
- 44.DMARD Effects on COVID-19 Outcomes | RheumNow. Accessed August 2, 2021. https://rheumnow.com/news/dmard-effects-covid-19-outcomes.
- 45.Lee H, Choi H, Yang B, et al. Interstitial lung disease increases susceptibility to and severity of COVID-19. Eur Respir J. 2021 doi: 10.1183/13993003.04125-2020. Published online April 22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Findlay AR, Goyal NA, Mozaffar T. An overview of polymyositis and dermatomyositis. Muscle Nerve. 2015;51(5):638–656. doi: 10.1002/MUS.24566. [DOI] [PubMed] [Google Scholar]
- 47.Briemberg HR, Amato AA. Dermatomyositis and Polymyositis. Curr Treat Options Neurol. 2003;5(5):2936–2943. doi: 10.1007/S11940-003-0025-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data used cannot be made publicly available due to data-use agreement. Data request can be made to TriNetX.