Abstract
Herein, we studied the variation in leaf and fruit morphological traits and antioxidant contents in 43 local and foreign cultivars (cvs) grown under the same experimental conditions in the widely cultivated plum species Prunus domestica and Prunus salicina. The peel contribution of fruit bioactive compounds in a serving portion, correlations among the examined parameters, and group patterns in each plum species were also studied. The species and cvs were sufficiently separated. Compared to Japanese cvs, European cvs had less elongated leaves and smaller and sweeter fruit with less total phenol and antioxidant capacities. The Japanese cvs ‘Red ace’ and the widely grown ‘Black Amber’, together with the European ‘Tuleu Dulce’, ‘BlueFre’, and the landrace ‘Asvestochoriou’ make up groups with rich dietary sources of phytochemicals. The peel tissue contained higher total phenols and antioxidant capacities compared to the flesh, while the peel/flesh ratios varied widely among the cvs (6.6-fold). The variation in the antioxidant contents was lower among the cvs calculated per serving portion (3.7-fold); yet the peel tissue contribution was equal to that of the flesh (48.6%), signifying its high nutritive value. We observed increased sweetness in the fruit in the later-harvested cultivars, while cvs with more blue- and red-colored peel generally contained higher antioxidant contents mainly in the European plums. Moreover, larger fruit sizes were positively correlated with larger and more elliptic leaf shapes. In conclusion, the significant role of the genotype and the peel tissue as a source of bioactive compounds in plums were outlined with prospects of utilization in future breeding programs.
Keywords: antioxidants, fruit color, fruit fresh weight, fruit shape, leaf characters, Prunus domestica, Prunus salicina, total soluble content
1. Introduction
Plums are of the most appreciated stone fruits worldwide, following peaches and nectarines in production (12.2 Mt of plums, 24.6 Mt of peach and nectarine; FAOSTAT 2020). Τhe most important commercial plum cultivars (cvs) belong to the hexaploid European plum (Prunus domestica L.) and the diploid Japanese plum (Prunus salicina Lindl.). Fresh fruit of P. salicina is mainly available during the summer and autumn months, while P. domestica is mostly processed into dried fruit and consumed year-round. Plum products such as jams and juice are produced from fruit that does not meet the fresh fruit marketable standards and is cherished year-round. Plums have a significant impact on human nutrition due to their health-promoting properties. They are also potentially valuable for the development and production of dietary supplements and functional food enriched with biologically active compounds [1]. The consumption of plum is related to osteoporosis prevention, improving memory, and has anti-inflammatory, antioxidant, and laxative effects [2].
The increased interest in research on plums is attributed to high levels of polyphenols that are known to be natural antioxidants; it is of interest to know the quantitative phenolics contained. More than 40 individual phenolics, mainly including chlorogenic acids (19), flavonols (9), flavan-3-ols (10), and anthocyanins (5), are accumulated in plum peel and flesh, with a remarkable diversity in their profiles depending on the variety and environmental conditions [1,3,4,5,6,7]. Moreover, the evaluation of the total antioxidant capacity, which takes into account the antioxidant activity of single compounds present in food as well as their potential synergistic interactions, is also of particular importance and has received much attention in recent years. In plums, the contribution of phenolic compounds to antioxidant activity was found to be more important than that of vitamin C and carotenoids [8].
Considering the significant impact of plum on human nutrition, it is of interest to consumers to define cvs with high polyphenolic content. There is high variability reported in the phytochemical and antioxidative compositions among different European [3,9,10,11,12,13,14,15] and Japanese plum cvs [16,17,18]. However, the above studies were conducted separately and little is known about the differences between those two widely cultivated species. The cultivation, climatic differences, and the ripening stage during harvest and postharvest treatments were also shown to affect the fruit’s phenolic and antioxidant contents [19,20].
Moreover, there is high inter-varietal phenotypic diversity in plum pomological traits, such as the ripening time, fruit shape and size, and peel and flesh color parameters [21]. Considering the above diversity, the European plum genotypes are classified into different pomological groups, including prunes, egg plums, greengages, damsons and bullaces, and mirabelle plums [21]. Peel color may be black, purple, red, green, or yellow, while flesh color may be yellow or red, with many shades of both colors; some cvs have a combination of both yellow and red flesh. The fruit color attributes are related to the anthocyanin contents [22,23]. Fruit color is also generally related to antioxidant content, but the use of peel color in predicting the fruit antioxidant content was reported to have limited application in fruit and vegetables with different colors in their inner and outer parts, such as in plums [23]. Relatively little is known about the potential contribution of the peel and flesh parts to the total antioxidant capacity in different plum cvs [8,11,18] or the relations between the fruit’s morphological traits and nutritional properties in plum [22,23,24,25,26].
European plum is native to Greece and local cultivars/accessions can be found abundantly. The local cvs with lower to higher commercial importance are: (a) ‘Skopelou’ (‘Agen Skopelou’ or ‘Glyka Skopelou’) on the island of Skopelos, Sporades regional unit, with significant production meant for drying, (b) ‘Mpardaki’ (in circular or elliptic shape) in the Fthiotida regional unit, and (c) ‘Praousti’ on the island of Samothrace, Evros regional unit. In the past, the local cv ‘Asvestochoriou’ also used to have a significant marketable share in central Macedonia. The pomological assignment and genetic relatedness of the local Greek cvs were previously characterized [21,27,28]. Due to the extensive use of foreign cvs with worldwide use, local germplasm is endangered. Therefore, the evaluation and exploitation of fruit quality and antioxidant attributes of Greek plum genetic material is of great importance and will assist in the promotion of interesting genotypes for breeding purposes to select cultivars with higher antioxidant content.
In the present study, the natural phenotypic variation in old and more recent commercial cultivars, as well as local cvs for the enhancement of plum antioxidant contents, were studied. Particularly, the objectives were to (a) determine the variations in the physical and chemical traits in fruit from 43 European and Japanese plum cvs grown under the same environmental and horticultural conditions, (b) examine the contribution to the fruit bioactive compounds in the peel and flesh tissue, and (c) dissect potential correlations among the examined physical and chemical traits.
2. Materials and Methods
2.1. Plant Material and Location
Fruits from 43 plum cvs; 19 European (Prunus domestica), 23 Japanese (Prunus salicina Lindl.), and 1 interspecific hybrid between plum and apricot (‘Pluot’) were analyzed (Table 1). From the European cvs, 12 were of international origin (1 from Bulgaria, 3 from France, 2 from Hungary, 2 from Romania, 1 from Russia, 1 from the United Kingdom, and 2 from the United States) and 7 were of local origin. The local cvs included ‘Skopelou’, ‘Asvestochoriou’, ‘Ksina Skopelou’ (also known as ‘Agiorgitiko’), ‘Avgato Skopelou’, ‘Mpardaki elliptic’, ‘Mpardaki circular’, and ‘Praousti’. Most of the studied Japanese plum cvs were of US origin (22), one was from Italy, and one from South Africa.
Table 1.
Cultivar names, origin, fruit fresh weight, and fruit shape in lateral view (UPOV characterization) of the European and Japanese plum cultivars studied.
| Origin | Fruit Weight (g) | Fruit Shape | |
|---|---|---|---|
| European cvs | |||
| Anna Spath Oradea | Hungary | 61.1 | Elliptic |
| Anna Spath Pitetsi | Hungary | 70.5 | Circular |
| Asvestochoriou | Greece | 65.3 | Circular |
| Avgata Skopelou | Greece | 40.0 | Drop shape |
| Bluefre | USA | 62.9 | Oblate |
| Giley | Bulgaria | 51.5 | Ovate |
| Ksina Skopelou 1 | Greece | 26.5 | Obovate |
| Mpardaki Circular | Greece | 41.3 | Circular |
| Mpardaki Elliptic | Greece | 18.4 | Elliptic |
| Praousti | Greece | 41.7 | Ovate |
| President | UK | 63.1 | Oblong |
| Prune d’ente 632 | France | 25.6 | Ovate |
| Prune d’ente 633 | France | 101.7 | Circular |
| Reine-Cl.di Violette | France | 57.5 | Circular |
| Russian | Russia | 20.7 | Circular |
| Scoldus SS | Romania | 45.5 | Elliptic |
| Skopelou 2 | Greece | 38.5 | Elliptic |
| Stanley | USA | 49.1 | Oblong |
| Tuleu Dulce | Romania | 37.3 | Ovate |
| Japanese cvs | |||
| Angeleno | USA | 120.9 | Oblate |
| Autumn Giant | USA | 151.0 | Circular |
| Beauty | USA | 53.8 | Cordate |
| Black Amber | USA | 68.2 | Oblate |
| Black Beauty | USA | 97.8 | Oblate |
| Black Gold | USA | 112.3 | Oblate |
| Black Star | USA | 116.2 | Circular |
| Calita | USA | 84.6 | Circular |
| Casselman | USA | 62.1 | Obovate |
| Florentia | Italy | 77.0 | Cordate |
| Fortune | USA | 78.1 | Obovate |
| Friar | USA | 92.4 | Circular |
| Frontier | USA | 87.7 | Oblate |
| John W | USA | 127.8 | Circular |
| Laroda | USA | 53.8 | Circular |
| October Sun | USA | 65.8 | Oblong |
| Ozark Premier | USA | 90.2 | Oblate |
| Pluot | USA | 69.8 | Cordate |
| Red Ace | USA | 75.5 | Oblate |
| Santa Rosa | USA | 57.5 | Oblong |
| Shiro | USA | 28.0 | Circular |
| Simka | USA | 64.7 | Cordate |
| Sun Gold | South Africa | 100.2 | Obovate |
| T.C. Sun | USA | 90.7 | Circular |
1 Also named ‘Agiorgitiko’; 2 also named ‘Agen Skopelou’ and Glyka Skopelou’.
Fruits from the studied cvs were harvested from an evaluation orchard maintained at the Department of Deciduous Fruit Trees in Naoussa (40°37′ 13.40″ N; 22°06′ 59.80″ E, at 119 masl). The annual mean temperature was 15.7 °C. Precipitation was 717 mm, with lower values in the summer and higher values in the winter. Cultivars were grafted on GF677 rootstock and planted with a spacing of 5 × 4 m in soil with a medium-heavy mechanical composition and a neutral pH (pH 7.4). The trees were planted in a randomized block design of four trees per cultivar, with two replicate trees per block.
The fruit characteristics were measured on a 30-fruit sample per cultivar harvested at the commercial stage based on size, color, and firmness.
2.2. Fruit Morphometric and Colorimetric Traits
Upon harvesting, the fruit fresh weight (FW) was measured and the fruit shape was evaluated according to the UPOV characterization. Differentiation among the European plum cultivars was assigned based on the descriptions presented in the study by Gaši et al. (2020) to one of six pomological groups; (1) egg plums sensu lato (E), (2) prunes of the French d’Agen type (P/A), (3) prunes of the Central-Southeast European Zwetschen type (P/Z), (4) greengages (G), (5) mirabelles (M), and (6) bullaces, damsons and var. pomariorum (D/B) [21]. Differentiation between the two prune types is not clear-cut, but the P/A were described as having more pointed ends on both the fruits and stones.
The CIE color parameters L* (brightness or lightness; 0 = black, 100 = white), a* (−a* = greenness, +a* = redness) and b* (−b* = blueness, +b* = yellowness), hue (h*) (calculated as tan−1b*/a*; 0° = red-purple, 90° = yellow, 180° = bluish-green, 270° = blue), and Chroma (C*) (calculated as (a*2 + b*2)½; degree of departure from grey to pure chromatic color) were measured in 12 fruit replicates, using a Minolta chromatometer (Minolta CR-400, Ramsey, NJ, USA). Readings were taken in the exocarp at both sides of each fruit and the mesocap was taken after cutting the fruit into slices.
The soluble solid content (SSC) and total acidity (TA) were determined from juice extracted using a food processor in three replicates of four plums. The SSC was determined using a digital refractometer (model PR-1, Atago, Japan) and expressed as °Brix, and the TA was analyzed in the juices by titration with 0.1 N NaOH to a pH endpoint of 8.2 and expressed as the citric acid equivalent (g/100 mL). The maturity index (MI) was calculated as the SSC/TA ratio.
2.3. Polyphenol Determinations and Antioxidant Capacities
Two wedge-shaped slices from the intact fruit were dissected, and the exocarp was separated and immediately frozen into liquid nitrogen and stored at −20 °C until needed. Frozen samples (about 0.5 g peel and 1.0 g flesh) were homogenized in 8 mL of 80% MeOH/H2O (v/v) using a mortar and pestle. The extract was centrifuged at 10,000× g for 10 min, and the supernatant was recovered.
2.3.1. Total Phenols (TPs)
The TP content was measured using the Folin–Ciocalteu colorimetric method [29]. The reaction mixture consisted of 0.3 mL of diluted extract, 0.2 mL of distilled water, and 2.5 mL of 10% Folin–Ciocalteu reagent. The tube was vortexed and then allowed to stand at room temperature for 3 min while 2 mL of saturated sodium carbonate solution was added. The solution was incubated for 5 min at 50 °C, and the absorbance was measured at 760 nm against a blank solution. Each measurement was repeated in duplicate. The total phenolic content was expressed as mg gallic acid equivalents (GAE)/100 g FW.
2.3.2. Total Antioxidant Capacity (TAC)
TAC was evaluated using 1,1- diphenyl-2-picrylhydrazyl (DPPH) (TACDPPH) and ferric reducing antioxidant power (FRAP) (TACFRAP) assays, and they were performed as described by Drogoudi et al. [30].
For the DPPH assay, the reaction mixtures containing 0 or 20 μL of diluted MeOH extract, 2.3 mL of 106.5 μM DPPH in MeOH, and 680 μL of H2O were vortexed and then kept at room temperature in the darkness for 4 h [31]. The absorbance of each reaction mixture was measured at 517 nm.
For the FRAP assay, a sample containing 3 mL of freshly prepared FRAP solution (0.3 Μ acetate buffer (pH 3.6) containing 10 mΜ 2,4,6-tripyridyl-s-triazine and 40 mΜ FeCl3 10H2O) and 20 μL of peel or 50 μL of flesh extract was incubated at 37 °C for 4 min, and the absorbance was measured at 593 nm.
A standard curve was obtained on each measurement day, using ascorbic acid standard solution, and accordingly, the results are expressed as milligram ascorbic acid equivalents (AAE)/100g FW in peel (TACDPPH–peel and TACFRAP–peel) and flesh (TACDPPH–flesh and TACFRAP–flesh) tissue.
The TPs and antioxidant contents were also calculated per serving portion, with 100 g FW consisting of 5.8 g peel and 89.5 g flesh. This was based on a significant positive correlation (r = 0.840) found between the TPs measured in the total fruit vs. the TPs estimated from peel and flesh measurements in 12 European plum cvs [11]. A similar peel and flesh fresh weight contribution was found in the Japanese plum cv ‘Angeleno’ in the present study (data not shown). However, a higher peel contribution was used in the study of Gil et al. (15 g peel + 80 g flesh) [8].
2.4. Leaf Characteristics
The leaf characteristics, such as leaf shape, leaf tip shape, and the shape of the base, were evaluated according to UPOV. The leaf blade length (LBL), leaf blade width (LBW), and stalk length (SL) were measured. The ratios LBL/LBW and LBL/SL were calculated. The measurements were made in 12 fully developed leaves collected from the middle part of the previous year’s grown shoots.
2.5. Statistical Analyses
The data were subject to one-way analysis of variance (ANOVA), with cultivars and species as the treatments. The least significant difference (LSD) values were calculated in cases where significance at p < 0.05 variance was found. A correlation analysis was performed. Statistical analyses were performed using SPSS 13.0 (SPSS Inc., Chicago, IL, USA).
Principal component analysis (PCA) was applied to the mean values of the measured traits and a heatmap was created using the ClustVis online software [32]. Both the rows and columns were clustered using Euclidean distance and the Ward method.
3. Results and Discussion
3.1. Fruit Morphological Traits
Fruit from the European cvs was considerably diversified in size, shape, and color, and the cvs belonged to different pomological groups (Table 1 and Table 2; Table S1; Figure S1). The landraces ‘Asvestohoriou’ and ‘Avgato Skopelou’ were classified as egg plums sensu lato, with a medium to large size and ovate or elliptic shape with rounded-end fruits and tender sweet flesh that often clings to the stone [21]. Anna Spath Pitesti’, ‘Mpardaki Circular’, ‘Prune d’ente 633’, and ‘Reine-Claude di Violette’ were classified as greengages, with a medium size, rounded and usually greenish fruits with tender and very sweet cling-stone flesh. ‘Praousti’ and ‘Russian’ were classified as mirabelles, with small, rounded fruits with yellow to orange peels and very sweet, free-stone flesh. ‘Ksina Skopelou’ and ‘Mpardaki Elliptic’ were classified as damsons, bullaces, or var. pomariorum. Finally, the Greek ‘Skopelou’ together with the remaining eight studied cvs were classified as prunes of the French d’Agen type or prunes of the Central-Southeast European Zwetschen type. ‘Skopelou’ is reported to be a sort of ‘d’Ente’; however, it does not have the characteristic pointed ends that refer to the French d’Agen type and is not considered to be self-fertile.
Table 2.
Mean (minimum–maximum), percentage coefficient variation (CV%), and p values of fruit quality and leaf traits when compared among 19 European and 24 Japanese plum cultivars grown in a cultivar evaluation orchard in Naoussa, Greece. Parameters measured were fruit fresh weight (g), peel and flesh color CIELAB parameters, soluble solid content (SSC, °Brix), titratable acidity (TA, grams citric acid equivalent/100 mL), maturity index (SSC/TA), total phenols (TPs) (mg GAE/100 g FW), total antioxidant capacity using the DPPH and FRAP methods (TACDPPH and TACFRAP, mg AAE/100 g FW) in peel and flesh tissue and serving portion (100 g FW), percentage TPs–peel/TPs–serving (% TPs–p/s), leaf shape (1, ovate; 2, elliptic; 3, obovate), leaf tip shape (1, acute; 2, right-angled; 3, obtuse), shape of base (1, acute; 2, obtuse; 3, truncate), leaf blade length (LBL, cm), leaf blade width (LBW, cm), stalk length (SL, cm) and ratios of LBL/LBW and LBL/SL. Different letters in the line indicate significant differences.
| European | Japanese | ||||||
|---|---|---|---|---|---|---|---|
| Mean | Min–Max | CV% | Mean | Min–Max | CV% | p | |
| Fruit weight | 48.3 b | 18.4–101.7 | 42 | 84.4 a | 28.0–151.0 | 33 | <0.001 |
| L–peel | 38.2 | 22.5–57.4 | 25 | 36.1 | 21.3–59.7 | 32 | 0.686 |
| a*–peel | 9.8 b | −23.7 | 73 | 13.5 a | −31.2 | 63 | 0.009 |
| b*–peel | 9.3 | −56.6 | 204 | 5.3 | −46.2 | 262 | 0.840 |
| h*–peel | 162.0 | 38.8–319 | 162 | 186.6 | 20.3–337.0 | 67 | 0.324 |
| Chroma–peel | 21.3 | 6.7–45.0 | 57 | 21.0 | 9.4–36.2 | 262 | 0.221 |
| L–flesh | 50.5 b | 37.4–59.0 | 13 | 55.3 a | 38.6–66.1 | 12 | <0.001 |
| a*–flesh | 4.8 | −37.3 | 175 | 5.0 | −25.2 | 132 | 0.188 |
| b*–flesh | 25.1 a | 7.7–41.2 | 41 | 20.8 b | 8.3–44.3 | 38 | 0.012 |
| h*–flesh | 77.9 | 19.2–97.5 | 27 | 77.1 | 20.5–95.3 | 22 | 0.197 |
| Chroma–flesh | 27.3 a | 12.7–41.3 | 33 | 21.9 b | 8.4–44.3 | 35 | <0.001 |
| SSC | 15.4 a | 11.5–19.6 | 17 | 14.4 b | 8.5–20.5 | 20 | 0.046 |
| TA | 1.2 | 0.5–1.9 | 33 | 1.3 | 0.8–1.9 | 25 | 0.077 |
| Maturity index | 15.4 a | 6.7–35.1 | 50 | 12.1 b | 6.1–21.7 | 34 | 0.001 |
| TPs–peel | 582.5 b | 152.1–984.6 | 43 | 916.7 a | 202.7–1797.3 | 42 | <0.001 |
| TACDPPH–peel | 594.1 b | 277.9–1135.7 | 42 | 852.7 a | 443.9–1535.8 | 41 | <0.001 |
| TACFRAP–peel | 361.4 b | 166.9–685.1 | 37 | 456.8 a | 206.1–1090.8 | 44 | 0.008 |
| TPs–flesh | 130.1 b | 55.9–220.9 | 33 | 164.5 a | 63.3–352.3 | 45 | 0.006 |
| TACDPPH–flesh | 121.9 b | 30.8–178.5 | 34 | 168.7 a | 66.9–343.9 | 42 | <0.001 |
| TACFRAP–flesh | 64.7 b | 47.2–86.3 | 17 | 76.8 a | 44.8–133.6 | 31 | 0.003 |
| TPs–serving | 150.2 b | 64.5–238.8 | 31 | 200.4 a | 88.1–409.2 | 41 | <0.001 |
| TACDPPH–ser. | 143.6 b | 58.4–215.0 | 29 | 199.3 a | 85.7–394.9 | 37 | <0.001 |
| TACFRAP–ser. | 78.4 b | 56.0–109.9 | 17 | 94.3 a | 56.9–154.6 | 29 | 0.001 |
| % TPs–p/s | 22.4 | 12.3–48.3 | 27.3 | 13.3–48.6 | |||
| Leaf shape | 2.1 | 1–3 | 40 | 2.1 | 1–3 | 20 | 0.932 |
| Leaf tip shape | 1.9 | 1–3 | 43 | 1.3 | 1–3 | 48 | 0.005 |
| Shape of base | 1.6 | 1–3 | 37 | 1.0 | 1–3 | 20 | <0.001 |
| LBL | 8.8 b | 5.6–12.0 | 17 | 9.7 a | 8.0–12.8 | 12 | <0.001 |
| LBW | 5.3 a | 3.5–6.9 | 18 | 4.2 b | 3.0–5.6 | 17 | <0.001 |
| SL | 1.7 a | 1.1–2.6 | 23 | 1.4 b | 1.0–2.2 | 23 | <0.001 |
| LBL/LBW | 1.7 | 1.4–2.2 | 14 | 2.4 | 1.8–3.2 | 14 | 0.107 |
| LBL/LPL | 5.6 b | 3.8–10.0 | 30 | 7.3 a | 4.8–10.7 | 18 | <0.001 |
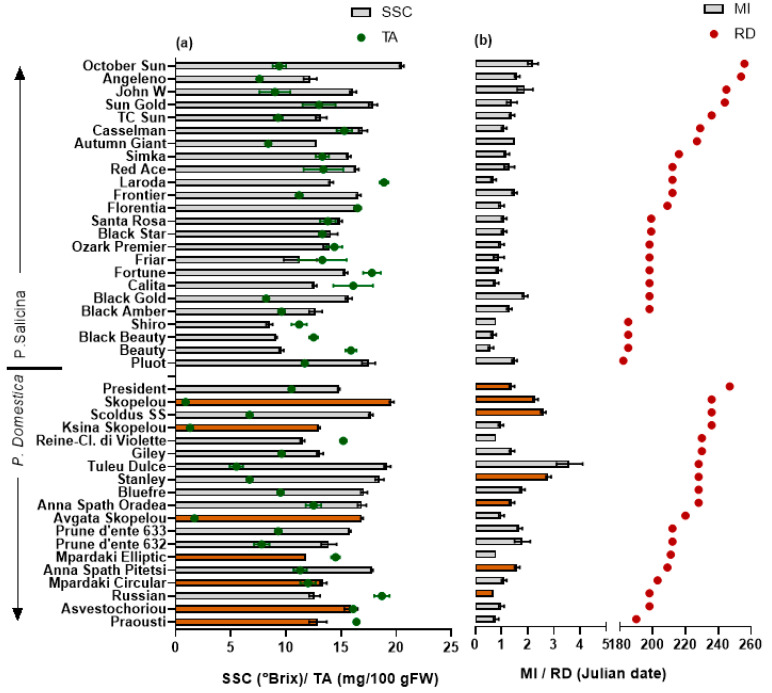
Among the Japanese plum cvs studied, ‘Pluot’, being an interspecific hybrid between Japanese plum and apricot, had the earliest ripening time (2 July), and after three days, ‘Beauty’, ‘Black Beauty’ and ‘Shiro’ ripened (Figure 1b). The ripening time of ‘Pluot’, in combination with its relatively high SSC and MI for the respective period (Figure 1a,b), provides an additional economic value for ‘Pluot’ cultivation. The earliest ripening European plum cvs were ‘Praousti’, ‘Asvestochoriou’, and ‘Russian’ with medium, large, and small fruit sizes, respectively, according to the classification described by Gaši et al. [21]. The superior features of ‘Asvestochoriou’ (large size and early ripening time) are the reasons it had a marketable value in Northern Greece in the past, but damages due to the spread of plum pox virus resulted in abandoning its cultivation.
Figure 1.
Mean (±SE) (a) soluble solid content (SSC, °Brix), titratable acidity (TA, mg citric acid/100 g FW), (b) maturity index (MI = SSC/TA), and ripening date (RD, Julian date) of 43 European and Japanese plums. Colored columns represent local cultivars. Least significant difference; SSC = 1.23, TA = 2.18, SSC/TA = 0.5.
There were no significant correlations between the ripening dates and the TPs or antioxidant capacities measured in the present study. Similar results were also found in the study by Arion et al. (2014), although they concluded that autumn compared to summer-harvested plum cvs contained higher phenolic content [33]. Nevertheless, in other fruit species, higher antioxidant contents were found in mid- to late-ripening cvs compared to earlier ripening cvs [30,34,35,36], which was related to higher temperatures and light intensity [37]. Moreover, precipitation during July–September was highly positively correlated with TPs in the fruit from Italian autochthonous plum cvs [15].
A large fruit size is preferred for both direct consumption and drying. Among the European plum cvs, extremely high fruit weights were found in ‘Prune d’ente 633’ and ‘Anna Spath Pitesti (70.5–101.7 g), and among the Japanese cvs, large or extremely large fruit sizes were found in ‘Autumn Giant’, ‘John W’, and ‘Angelino’ (120.9–151.0 g) (Table 1 and Table 2). The smallest FW was found in ‘Mpardaki elliptic’, ‘Russian’ (European cvs), ‘Shiro’, ’Beauty’, and ‘Laroda’ (Japanese cvs), being of a less favorable agronomic trait.
The greatest variations among all the studied morphological and chemical parameters were found in the peel blueness to yellowness (b*–peel) (CV% = 204–262) and the flesh greenness to redness attributes (a*–flesh) (CV% = 132–175) (Table 2; Figures S1 and S2). Among the European cvs, the most blue-colored peels (low b* values) were found in ‘Stanley’, ‘Anna Spath Pitesti’, and ‘Scoldus SS’, while the most yellow (high b* values) were in ‘Mpardaki elliptic’, ‘Praousti’, and ‘Mpardaki circular’ (Table S1; Figure S1). The flesh color varied from being the reddest in cv ‘Russian’, followed by ‘D’ente 633’, to the most green in ‘Skopelou’ and ‘Anna Spath Oradea’, and yellow-colored in ‘Ksina Skopelou’. The plum ‘Russian’ was the only one among the European ones with a red-colored peel and flesh, suggesting it contains high anthocyanin contents. Concerning the Japanese plum cvs, the peel colors varied from the most yellow (‘Shiro’, ‘Sun Gold’, and ‘T.C. Sun’, high b*–peel) to red (‘Fortune’, ‘Autumn Giant’, ‘Beauty’, ‘Ozark Premier’, and ‘Casselman’, highest a*–peel). The collection contained only one genotype with red flesh, cv ‘Frontier’ (highest a*–flesh = 23.8), while ‘Red ace’ and ‘Black Beauty’ also had reddish-colored flesh (Table S1; Figure S2).
Cvs with more blue- and red-colored peel generally contained higher antioxidant contents mainly in European plum cvs (peel–b* vs. TPs–peel and TPs–serving, r = −0.546–(−0.592), peel–a* vs. TPs–peel, r = 0.577; Chroma–peel vs. TPs–peel, r = −0.568) although the correlations were not strong. Less significant correlations between color parameters and fruit antioxidants were found in Japanese plums (L–peel vs. TPs–peel or TACFRAP–peel, r = −0.385–(−0.485)). Johnson et al. [26] and Yu et al. [25] observed the highest levels of phenolic compounds and antioxidant capacities in the black- or purple-flesh genotypes, although there were no significant correlations. In the study by Goldner et al. [24], conducted with the juice from 43 European plum varieties with yellow, blue, and dark blue fruit peels, weak red pigmentation co-occurred with low total phenol levels; however, there were exceptions, suggesting that the breeder can combine yellow fruit skin with a high level of health beneficial phenolic compounds by using the appropriate donor genotypes.
The b* color parameter (suggesting blue to yellow coloration) was positively correlated with its flesh counterpart (r = 0.678–0.736 in European and Japanese cvs) (Table 3 and Table 4). Only in European plum cvs, were the color parameters a*, hue, and Chroma also positively correlated in the peel and flesh (r= 0.464, −0.490, and 0.475, respectively). Although the color parameters appeared to correlate between the peel and flesh, there were no similar correlations in the TPs and antioxidant contents found in the peel and flesh (Table 3 and Table 4). Furthermore, the coloration and antioxidant contents were not previously correlated with their flesh counterparts in previous studies for apple [38] and pomegranate [39].
Table 3.
Pearson correlation analyses between phenotypic and chemical traits in 19 European plum cvs. RD, ripening date; FW, fruit fresh weight; SSC, soluble solid content; TA, titratable acidity; MI, maturity index; TPs, total phenols; DPPH and FRAP, total antioxidant capacity using the DPPH and FRAP radicals, respectively. ns. non significant; Absolute linear correlations ≥|0.60| are marked in bold.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. RD | 1 | |||||||||||||||||||||||
| 2. FW | ns | 1 | ||||||||||||||||||||||
| 3. L*−peel | −0.473 | ns | 1 | |||||||||||||||||||||
| 4. a*−peel | ns | ns | ns | 1 | ||||||||||||||||||||
| 5. b*−peel | ns | ns | 0.793 | −0.492 | 1 | |||||||||||||||||||
| 6. h*−peel | ns | ns | ns | ns | −0.678 | 1 | ||||||||||||||||||
| 7. Chroma−peel | −0.607 | ns | 0.844 | ns | 0.888 | ns | 1 | |||||||||||||||||
| 8. L*−flesh | ns | ns | ns | ns | ns | ns | ns | 1 | ||||||||||||||||
| 9. a*−flesh | ns | ns | ns | 0.464 | ns | ns | ns | ns | 1 | |||||||||||||||
| 10. b*−flesh | ns | ns | ns | −0.471 | 0.736 | −0.845 | ns | ns | ns | 1 | ||||||||||||||
| 11. h*−flesh | ns | ns | ns | −0.467 | ns | −0.490 | ns | ns | −0.950 | 0.583 | 1 | |||||||||||||
| 12. Chroma−flesh | ns | ns | ns | ns | 0.677 | −0.675 | 0.475 | ns | ns | 0.806 | ns | 1 | ||||||||||||
| 13. SSC | ns | ns | ns | ns | −0.546 | ns | −0.571 | ns | ns | ns | ns | −0.481 | 1 | |||||||||||
| 14. TA | −0.518 | ns | 0.482 | ns | 0.575 | ns | 0.595 | ns | ns | ns | ns | 0.523 | −0.598 | 1 | ||||||||||
| 15. MI | ns | ns | ns | ns | −0.625 | 0.553 | −0.578 | ns | ns | ns | ns | −0.548 | 0.784 | −0.894 | 1 | |||||||||
| 16. TPs−peel | ns | ns | ns | 0.577 | −0.592 | ns | −0.568 | ns | ns | ns | ns | ns | ns | ns | ns | 1 | ||||||||
| 17. DPPH−peel | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.797 | 1 | |||||||
| 18. FRAP−peel | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 1 | ||||||
| 19. TPs−flesh | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 1 | |||||
| 20. DPPH−flesh | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.469 | ns | ns | 0.738 | 1 | ||||
| 21. FRAP−flesh | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.567 | ns | ns | 0.872 | 0.702 | 1 | |||
| 22. TPs−serving | ns | ns | ns | 0.490 | −0.522 | ns | −0.546 | ns | ns | ns | ns | ns | ns | ns | ns | 0.684 | 0.457 | ns | 0.961 | 0.750 | 0.891 | 1 | ||
| 23. DPPH−serving | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.680 | 0.477 | ns | 0.719 | 0.942 | 0.754 | 0.801 | 1 | |
| 24. FRAP−serving | ns | ns | −0.484 | ns | ns | ns | −0.477 | ns | ns | ns | ns | ns | ns | ns | ns | 0.598 | 0.563 | 0.684 | 0.637 | 0.524 | 0.818 | 0.708 | 0.650 | 1 |
Table 4.
Pearson correlation analyses between phenotypic and chemical traits in 24 Japanese plum cvs. Abbreviations as in Table 3.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. RD | 1 | |||||||||||||||||||||||
| 2. FW | ns | 1 | ||||||||||||||||||||||
| 3. L*−peel | ns | ns | 1 | |||||||||||||||||||||
| 4. a*−peel | ns | ns | ns | 1 | ||||||||||||||||||||
| 5. b*−peel | ns | ns | 0.749 | ns | 1 | |||||||||||||||||||
| 6. h*−peel | ns | ns | −0.414 | ns | −0.804 | 1 | ||||||||||||||||||
| 7. Chroma−peel | ns | ns | 0.759 | ns | 0.773 | −0.606 | 1 | |||||||||||||||||
| 8. L*−flesh | ns | ns | ns | ns | ns | ns | ns | 1 | ||||||||||||||||
| 9. a*−flesh | ns | ns | ns | ns | ns | ns | ns | −0.612 | 1 | |||||||||||||||
| 10. b*−flesh | ns | ns | ns | ns | 0.678 | −0.721 | 0.552 | ns | ns | 1 | ||||||||||||||
| 11. h*−flesh | ns | ns | ns | ns | ns | ns | ns | 0.575 | −0.963 | ns | 1 | |||||||||||||
| 12. Chroma−flesh | ns | ns | ns | ns | 0.583 | −0.574 | ns | ns | ns | 0.791 | −0.192 | 1 | ||||||||||||
| 13. SSC | 0.488 | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 1 | |||||||||||
| 14. TA | −0.421 | −0.493 | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 1 | ||||||||||
| 15. MI | 0.643 | 0.438 | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.620 | −0.774 | 1 | |||||||||
| 16. TPs−peel | ns | ns | −0.385 | ns | ns | ns | −0.409 | ns | ns | ns | ns | ns | ns | ns | ns | 1 | ||||||||
| 17. DPPH−peel | ns | ns | −0.181 | ns | ns | ns | −0.132 | ns | ns | ns | ns | ns | ns | ns | ns | 0.828 | 1 | |||||||
| 18. FRAP−peel | ns | ns | −0.495 | ns | ns | ns | −0.334 | ns | ns | ns | ns | 0.411 | ns | ns | ns | 0.672 | 0.683 | 1 | ||||||
| 19. TPs−flesh | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.629 | 0.609 | 0.259 | 1 | |||||
| 20. DPPH−flesh | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.511 | 0.511 | 0.291 | 0.923 | 1 | ||||
| 21. FRAP−flesh | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.590 | 0.623 | 0.306 | 0.943 | 0.702 | 1 | |||
| 22. TPs−serving | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.777 | 0.716 | 0.39 | 0.978 | 0.750 | 0.891 | 1 | ||
| 23. DPPH−serving | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.660 | 0.705 | 0.411 | 0.941 | 0.942 | 0.754 | 0.801 | 1 | |
| 24. FRAP−serving | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.703 | 0.726 | 0.565 | 0.887 | 0.524 | 0.818 | 0.708 | 0.650 | 1 |
3.2. SSC and TA
European plums with SSC of more than 16 °Brix are considered sweet and suitable for fresh consumption [40]. In the present study, the SSC ranged from 11.5 to 19.6 °Brix in European plum cvs; only seven cvs had SSC higher than 16 °Brix, and ‘Skopelou’, ‘Tuley Dulce’, and ‘Stanley’ contained the highest values (mean of 19.1 °Brix) (Figure 1). Sahamishirazi et al. [13] found a wider range of SSC in a relatively high number of European plum cvs grown in Germany (178 cvs) (9.6–29.5%). In the present study, the range of SSC found in the Japanese was higher compared to the European plum cvs studied and ranged from 8.5 to 20.5 °Brix, with only four cvs having lower than 12 °Brix (‘Shiro’, ‘Black Beauty’, ‘Beauty’, and ‘Friar’).
The maturity index (MI) is the ratio between the SSC and TA (SSC/TA) contents that represents a reliable indicator of a cultivar’s suitability for acceptance by consumers [41]. The variation in the MI was higher than in the SSC (CV% = 50 and 17, respectively). The MI varied between 6.1 and 35.1, with the highest values in European plums ‘Stanley’, Tuley Dulce’, ‘Scoldus SS’, and ‘Skopelou’. Among the Japanese plum cvs ‘October Sun’, ‘Sun gold’, and ‘Pluot’ had the highest SSC and MI (means of 18.6 °Brix and 1.7, respectively). The earliest ripening cvs, ‘Shiro’, ‘Black Beauty’, and ‘Beauty’, had the lowest SSC and MI (means of 9.1 °Brix and 0.7, respectively) (Figure 1). Fruit from the cv ‘Shiro’ was also shown to have a low acceptance level in the study by Myracle et al. [42].
In summary, the European plums were smaller in size and had higher SSCs and MIs compared to the Japanese cultivars, whereas there was no difference in the TA. Higher SSCs in European plums were also found in the studies by Wolf et al. [18] and Liverani et al. [43], which further signifies the results obtained in the present study and suggests that European plums are more tasteful.
Fruit from the later-harvested plum cvs tended to be sweeter; the ripening date was positively correlated with the MI (r = 0.643) in the Japanese cvs and negatively correlated with the TA (r= −0.518) in the European plums (Figure 1), probably resulting from a greater fruit developmental time and enabling the fruit to become sweeter over a longer period. Previous studies have also reported that early-season cultivars had lower SSCs than late-season cultivars, while the TA was not related to the time of the season in plums [41] and other stone fruits [34,44,45,46,47].
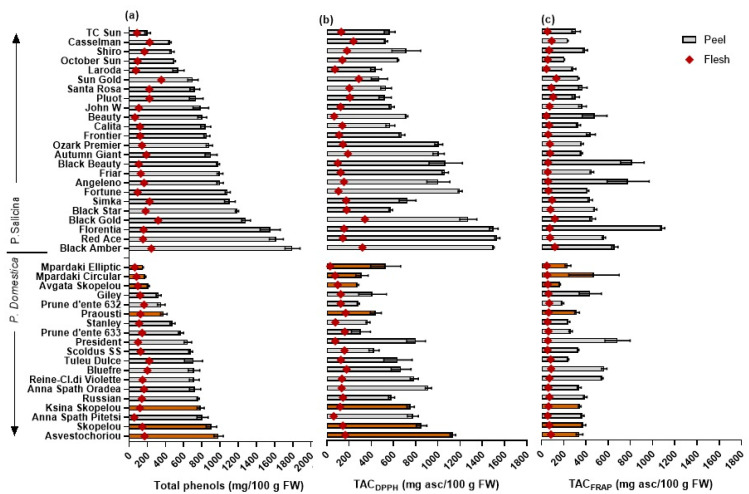
3.3. TP and Radical Scavenging Activities
The TPs–peel content was lower in the European plum cvs (ranging from 152.1 to 984.6 mg/100 g FW) compared to the Japanese plums (202.7 to 1797.3 mg/100 g FW) (Table 2; Figure 2). The highest TPs–peel values were found in the European ‘Asvestochoriou’ and Skopelou’ and in the Japanese plums ‘Black Amber’, ‘Red Ace’, and ‘Florentia’. Relatively few studies have conducted separate measurements of TPs in the peel and flesh tissue, and similar or lower values have been reported for European (250–773 mg GAE/100 g FW in Cosmulescu et al. [11]) and Japanese plum cvs (72–656 in Wolf et al. [18]; 163.3–332.3 in Gil et al. [8]).
Figure 2.
Mean (±SE) (a) total phenol content (mg gallic acid equivalent/100 g FW), (b) total antioxidant capacity using the DPPH (TACDPPH), and (c) the FRAP radical (TACFRAP) (mg ascorbic acid equivalent/100 g FW), in fruit peel (solid bars) and flesh (rhombus) tissue of 43 European and Japanese plum cvs. Colored columns represent local cultivars. LSD: TPs–peel, 143.5; TPs–flesh, 40.5; TACDPPH–peel, 181.1; TACDPPH–flesh, 38.3; TACFRAP–peel, 172.4; TACFRAP–flesh, 15.9.
The TPs–flesh content was much lower compared to that in the peels of the European (55.9–220.9 mg GAE/100 g FW) and Japanese plum cvs (63.3–352.3 mg GAE/100 g FW). The highest values of TPs–flesh content were measured in the European ‘Tuley Dulce’, ‘Bluefre’, and ‘Asvestochoriou’ and the Japanese cvs ‘Red Ace’ and ‘Black Amber’. Similar values of TPs–flesh content have been found in other European (61–181 in Cosmulescu et al. [11]) and Japanese plum cvs (38–313 in Wolf et al. [18]; 22.0–76.9 in Gil et al. [8]).
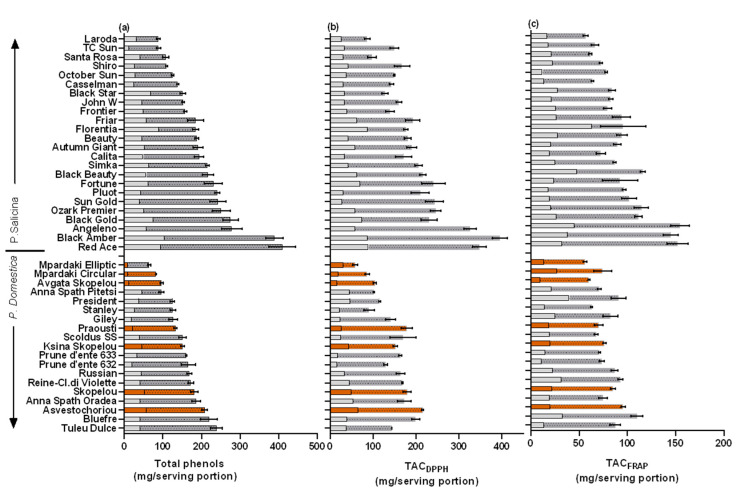
To evaluate the impact of plum fruit consumption on the dietary intake of TPs and the ingested antioxidant equivalents, the antioxidants supplied by a plum fruit serving were determined. Among the 19 European plum cvs studied, the TPs–serving varied 3.7-fold (64.5–238.8 mg/100 g FW), and higher values were found in ‘Tuleu Dulce’, ‘BlueFre’, and ‘Asvestochoriou’, with the lowest in ‘Mpardaki Elliptic’, ‘Mpardaki circular’, and ‘Avgato’ Skopelou’ (Table 2; Figure 3). The local cvs ‘Skopelou’ and ‘Ksina Skopelou’ were also in the highest range of TPs–serving, and similar mean values were previously recorded [12].
Figure 3.
Mean (±SE) (a) total phenols (TPs, mg gallic acid equivalent) and total antioxidant capacity using the (b) DPPH (TACDPPH) and (c) FRAP radicals (TACFRAP) (mg ascorbic acid equivalent), expressed as per serving portion (100 g FW). The percentage (%) contributions of peel and flesh are shown as empty and hatched superimposed columns, respectively. Colored columns represent local cultivars. LSD; total phenols = 35.9, TACDPPH = 33.2, TACFRAP = 19.1.
Fruits from the Japanese cvs contained higher TPs–serving compared to European plums (an increase by 1.3-fold) and varied among the studied cvs by 4.6-fold (88.1–409.2 mg/100 g FW) (Table 2, Figure 3). Higher values of TPs–serving were found in ‘Red ace’, ‘Black Amber’, and ‘Angeleno’. Similarly, fruit from the cv ‘Black Amber’ was previously separated for having a high TP and antioxidant capacity when compared to other Japanese plum cvs (5 cvs, Lozano et al. [48]; 17 cvs, Wolf et al. [18]). Moreover, Gil et al. [8] similarly found high TPs and antioxidant content in ‘Angeleno’ fruit compared to four other cvs. In the present study, the lowest values of TPs–serving were found in ‘Laroda’, ‘T.C. Sun’, and ‘Santa Rosa’. The range of TPs found was similar to those reported for other Japanese plum cvs (227–383 mg GAE/100 g FW in 10 cvs, Venter et al. [16]; 317–761 in 5 yellow flesh genotypes, DiNardo et al. [17]; 51–430 in 15 cvs, Wolf et al. [18]).
The total antioxidant capacities were measured using the DPPH and FRAP methods, which determine the ability of antioxidants to reduce the DPPH or ferric complex (Fe3+) to the ferrous complex (Fe2+) via color and absorbance change, respectively. The TACDPPH–peel and –flesh were similarly higher in the Japanese compared to the European plum cvs when measured in the peel, flesh, or per serving portion (Table 2). In the European plums, the mean values of the TACDPPH–peel and –flesh were 594.1 and 121.9 mg AAE/100 g FW, respectively, while in the Japanese plums, they were 852.7 and 168.7 mg AAE/100 g FW, respectively (Figure 2). The above values are similar to those reported in the study by Gil et al. [8]. The fruit TACFRAP–serving portion ranged from 56.0 to 154.6 mg AAE/100 g FW, which is similar to the ranges reported in the studies by Vasantha Rupasinghe et al. [10] and Dowling et al. [49] (105–424 and 121 to 229 mg, respectively), but significantly lower than the values reported by DiNardo et al. [17] (15,605 to 28,413 mg AAE/100 g FW).
The TP content was highly positively correlated with TACDPPH or TACFRAP in different plant tissues in the European and Japanese cvs (Table 3 and Table 4, respectively), with correlation r values usually being higher in the TACDPPH vs. TPs (ranging from 0.797 to 0.923) than in the TACFRAP vs. TPs (0.672–0.943, while it was non-significant in the peel tissue of the European cvs). Similarly, highly significant correlations have been found between the TPs and TACDPPH or TACFRAP in other studies [10,11,13,18]. The results from the present study suggest that measurements of TACDPPH could be better used than those of TACFRAP as a proxy for estimating the total phenolic content in plums, or vice versa, in screening programs.
3.4. Contribution of Peel to TPs and TAC in a Serving Portion
A large variation in the peel/flesh TP ratio in a cultivar (varied 6.6-fold, from 2.2 to 14.6 times) and peel/flesh TACDPPH and TACFRAP ratios (varied 9.6-fold, from 1.9 to 18.3 times) were found among the studied cvs. The peel tissue had a much higher content compared to its flesh counterpart in ‘Anna Spath Pitesti’ (European), ‘Florentia’, and ‘Black Star’ (Japanese cvs) (Figure 3). Nevertheless, lower ranges of peel/flesh TP ratios have been found in previous studies (4.5-fold in 12 European plums, Cosmulescu et al. [11]; 2.0-fold in Japanese plums, apart from ‘Shiro’, in which it was higher in the flesh, Wolf et al. [18]; 4.0–8.2-fold in 5 cvs, Gil et al. [8]), which may be related to the lower number of cvs studied.
Due to the low fruit weight contribution of the peel in a plum, the above large variation in the peel/flesh antioxidant contents found was diminished to a much lower variation when calculating the peel/flesh TPs and TACDPPH and TACFRAP ratios in a serving portion. This varied 3.7-fold from 12.3% to 48.6% (Figure 3). The percentage of the peel TP contribution was lower in the European (22.4%) compared to the Japanese (27.3%) plum cvs (Table 2) and coincided with having lower values of TP and antioxidant capacity per serving portion. The present study signifies that the peel tissue can have up to an equal contribution (48.6%) of the antioxidant contents contained in a serving portion as that of the flesh, and there is a relatively low variability among cvs.
3.5. Leaf Morphological Traits
In most of the studied cvs, the leaf shape was elliptic. The European and Japanese plum cvs differed in the leaf tip shape and shape of the leaf base, both being mainly acute in the Japanese cvs (Table 2; Table S2). The eaves from the studied Japanese cvs were more elongated with shorter stalks (longer LBL, shorter LBW and SL, and there was a trend of having a higher LBL/LBW ratio, (p = 0.107)), compared to the European plum cvs. More elongated leaves (elliptic shape and higher LBL/LBW ratio) and higher LBL/SL ratios were found in ‘Asvestochoriou’ and ‘Anna Spath Pitesti’, whereas the smallest leaves and petioles were found in ‘Praousti’ (Table S2). Significant positive correlations were found between the antioxidant contents in the fruit tissue and leaf shape (LBL/LBW) (r = 0.347–0.547). LBL and LBL/SL were positively correlated with FW (r = 0.376 and 0.550, respectively), suggesting that cvs with larger and more elliptic leaf shapes also produced larger fruit sizes. This was also reported in the study by Mirheidari et al. [50] that was conducted on autochthonous European plum accession in Iran; however, the r values were even lower (r < 0.300) compared to the present study.
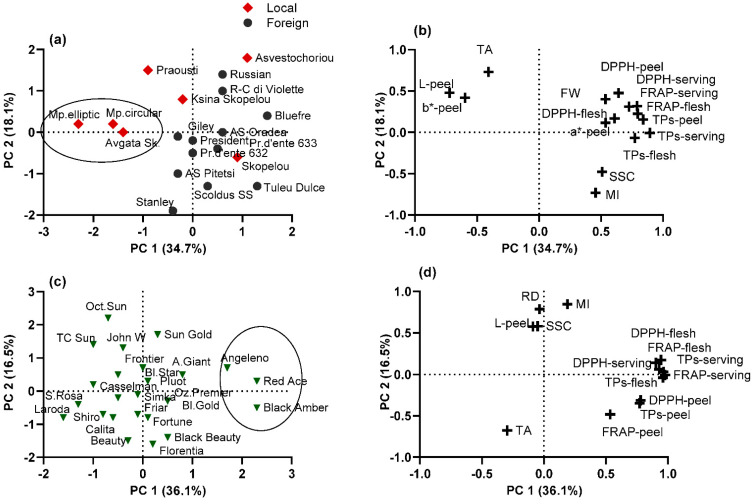
3.6. Principal Component Analyses
In the present study, PCA was applied to a fruit physical and chemical dataset to determine the most important variables that explain the correlations between cultivars and to identify group patterns in each plum species separately. Two principal components explained a similar cumulative variation of 52.3–52.8% in both the European and Japanese plum cvs, presented as a scatter diagram in Figure 4b,d. In European plum cvs, the parameters with significant correlation to PC1 (values > 0.50) (34.7% of variance) were TPs and TAC (in the peel, flesh, and serving portion), SSC, and fruit physical traits, such as FW and color parameters (a*–peel, L–peel, and b*–peel); the local cvs ‘Avgato Skopelou’, ‘Mpardaki circular’, and ‘Mpardaki elliptic’ were separated from the rest of the studied cvs (Figure 4a).
Figure 4.
(a,c) Segregation and (b,d) factor loadings, of (a,b) 19 European and (c,d) 24 Japanese plum cultivars, on the basis of fruit physical and chemical characters, determined by principal component analysis. Variable annotations are presented in Table 3.
In the Japanese plum cvs, the most important parameters incorporated into PC1 were the total phenol and antioxidant contents (in the peel, flesh, and serving portion) (36.1% of variance); ‘Black Amber’, ‘Red Ace’, and ‘Angeleno’ were separated with high values. PC2 (18.1% of variance) was positively correlated with the ripening date, L–peel, SSC, and MI and negatively correlated with the TA; the cvs ‘October Sun’, ‘Sun Gold’, ‘John W’ and T.C. Sun’ had high values (Figure 4c).
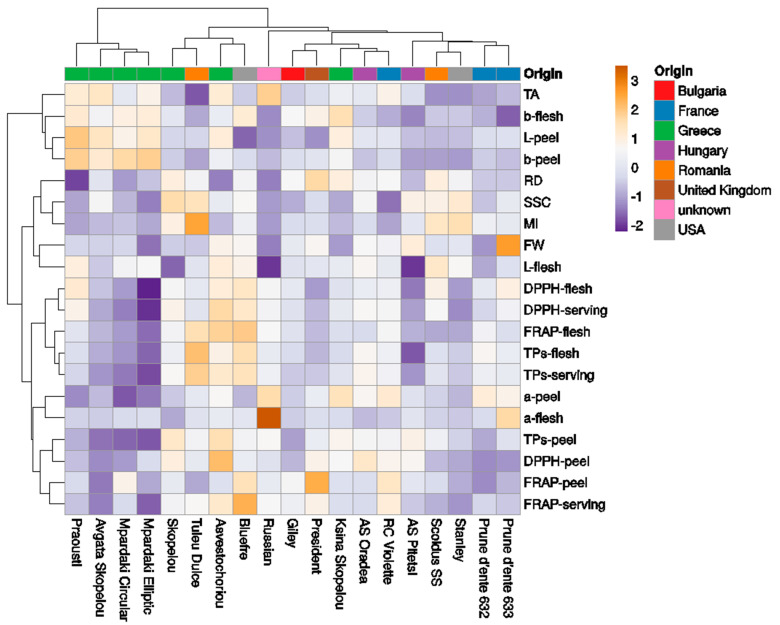
The collection of 19 European plum cultivars was classified into five clades (Figure 5). The first cluster included cvs Praousti’, ‘Avgato Skopelou’, ‘Mpardaki circular’, and ‘Mpardaki elliptic’, with more light and yellow-colored peel and flesh, high TA (high values in b*–flesh, L*–, b*–peel; TA), and low antioxidant contents in the peel, flesh, and serving portion. Previously, results from a Bayesian structure analysis conducted using SSR–HRM markers on the same cvs showed that ‘Avgato Skopelou’ differed from the rest of the Greek plum cultivars since it was not grouped into the same cluster (Merkouropoulos et al., 2016) [27]. However, in the present study, ‘Avgato Skopelou’ was closely grouped with the other Greek cvs. The second cluster included the cvs ‘Skopelou’, ‘Tuley dulce’, ‘Asvestochoriou’, and ‘Bluefre’, with high fruit antioxidant contents. The third cluster included only the cv ‘Russian’, separated for red coloration in the peel and flesh, high TA, and low SSC and FW. The fourth cluster included the cvs ‘Giley’, ‘President’, ‘Ksina Skopelou’, ‘Anna Spath Oradea’, and ‘Reine-Claude di Violette’. Finally, the fifth cluster included the cvs ‘Anna Spath Pitesti’ (low L*–flesh), ‘Scoldus SS’, ‘Stanley’, ‘D’ente 632’, and ‘D’ente 633’, having low TA and darker-colored peel.
Figure 5.
Heatmap showing the clustering of fruit phenotyping traits in 19 European plum cultivars using the ClustVis software. The columns correspond to the cultivars and the rows correspond to the fruit phenotypic traits studied. Both the rows and columns were clustered using Euclidean distance and the Ward method.
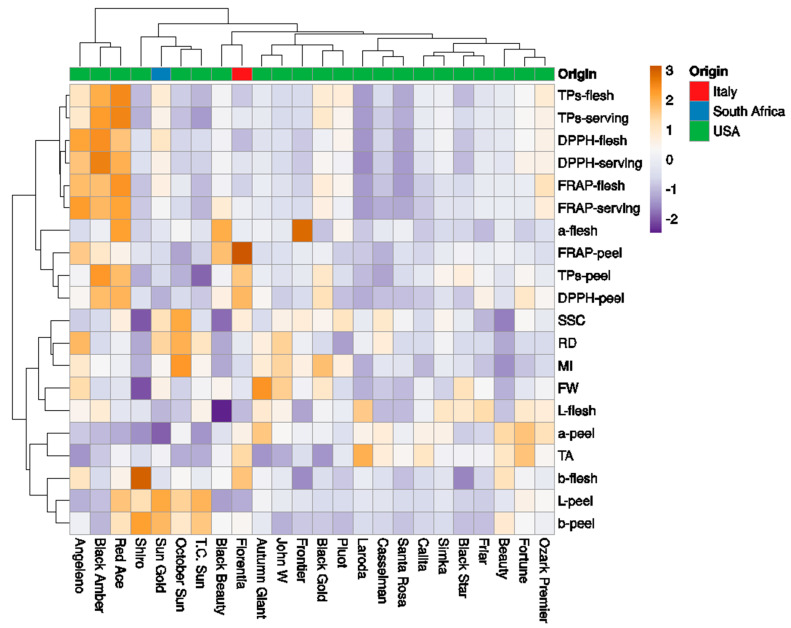
The collection of 24 Japanese plum cultivars was classified into seven clades (Figure 6). The first clade included ‘Angelino’, ‘Black amber’, and ‘Red Ace’, with high antioxidant contents. The second clade included the cvs ‘Shiro’, ‘Sun Gold’, ‘October Sun’, and ‘T.C. Sun’, with low antioxidant contents and high L– and b–peel. The third cluster included the cvs ‘Black Beauty’ and ‘Florentia’, with low L– peel and L–flesh. The fourth cluster included the cvs ‘Autumn Giant’, ‘John W’, ‘Frontier’, ‘Black Gold’, and ‘Pluot’. The fifth clade included the cvs ‘Laroda’, ‘Casselman’, and ‘Santa ‘Rosa’ (low antioxidant contents). The sixth cluster included the cvs ‘Calita’, ‘Simka’, ‘Black Star’, and ‘Friar’. Finally, the last cluster included the cvs ‘Beauty’, ‘Fortune’, and ‘Ozark Premier’.
Figure 6.
Heatmap showing the clustering of fruit phenotyping traits in 24 Japanese plum cultivars using the ClustVis software. The columns correspond to the cultivars and the rows correspond to the fruit phenotypic traits studied. Both rows and columns were clustered using Euclidean distance and the Ward method.
4. Conclusions
The results from the present study give insight into the variability in leaf and fruit quality traits of 43 plum cvs from the most widely cultivated species P. domestica (European) and P. salicina (Japanese). The fruit was harvested under the same experimental conditions, and thus, the differences observed relate to genetic variability. To our knowledge, the above two species were not previously compared under the same experimental conditions. There were large genotype-dependent differences in the fruit qualitative traits and concentrations of the bioactive compounds studied; fruits from European plums were grouped as smaller and sweeter, with less phenol content and antioxidant capacity (1.3–1.6-fold decrease) compared to Japanese cvs.
The Japanese plums ‘Red ace’ and ‘Black Amber’ and the European plums ‘Tuleu Dulce’, ‘BlueFre’, and ‘Asvestochoriou’ were characterized by particularly rich dietary sources of phytochemicals. The highest TP content per serving portion was 409.2 mg in the cvs ‘Red ace’ and ‘Black Amber’, the latter being worldwide cultivated, and a value that is similar or higher to those reported for Rubus idaeus, Ribes nigrum, and Sambucus nigra, which are considered functional fruit [18]. This also contributes to the finding that plums are a rich source of phenolic compounds, being ranked superior to various other fruit, including those commonly consumed such as apple, banana, peach, pear, and watermelon [23,51]. Therefore, it is concluded that plums should be considered as having a significant impact on human nutrition, especially since they are widely consumed.
There was a large variation in the peel/flesh antioxidant content ratios found among the studied cvs, which was diminished to a much lower variation when expressed per serving portion. However, the peel contribution to the antioxidant content in a serving portion was almost equal (48.6%) in a cultivar, signifying that the peel can have significance equal to that of flesh in providing nutritive value in a plum fruit.
Acknowledgments
The authors thank K. Ziakou for helping with the laboratory analyses, and C. Bazakos and I. Ganopoulos for the critical reading and suggestions.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/plants11101338/s1.
Author Contributions
P.D.: Experiment design, data curation, formal analysis, writing, and funding acquisition. G.P.: Experiment design, laboratory work, and review. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This work was funded by the Operational Program Competitiveness, Entrepreneurship, and Innovation, under the name RESEARCH–CREATE–INNOVATE (project code: T1EDK−05438).
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Liaudanskas M., Okulevičiūtė R., Lanauskas J., Kviklys D., Zymonė K., Rendyuk T., Žvikas V., Uselis N., Janulis V. Variability in the Content of Phenolic Compounds in Plum Fruit. Plants. 2020;9:1611. doi: 10.3390/plants9111611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liu W., Nan G., Nisar M.F., Wan C. Chemical Constituents and Health Benefits of Four Chinese Plum Species. J. Food Qual. 2020;2020:8842506. doi: 10.1155/2020/8842506. [DOI] [Google Scholar]
- 3.Kim D.-O., Jeong S.W., Lee C.Y. Antioxidant capacity of phenolic phytochemicals from various cultivars of plums. Food Chem. 2003;81:321–326. doi: 10.1016/S0308-8146(02)00423-5. [DOI] [Google Scholar]
- 4.Cevallos-Casals B.A., Byrne D., Okie W.R., Cisneros-Zevallos L. Selecting new peach and plum genotypes rich in phenolic compounds and enhanced functional properties. Food Chem. 2006;96:273–280. doi: 10.1016/j.foodchem.2005.02.032. [DOI] [Google Scholar]
- 5.Usenik V., Kastelec D., Veberic R., Štampar F. Quality changes during ripening of plums (Prunus domestica L.) Food Chem. 2008;111:830–836. doi: 10.1016/j.foodchem.2008.04.057. [DOI] [Google Scholar]
- 6.Treutter D., Wang D., Farag M.A., Baires G.D.A., Rühmann S., Neumüller M. Diversity of Phenolic Profiles in the Fruit Skin of Prunus domestica Plums and Related Species. J. Agric. Food Chem. 2012;60:12011–12019. doi: 10.1021/jf303644f. [DOI] [PubMed] [Google Scholar]
- 7.Jaiswal R., Karaköse H., Rühmann S., Goldner K., Neumüller M., Treutter D., Kuhnert N. Identification of Phenolic Compounds in Plum Fruits (Prunus salicina L. and Prunus domestica L.) by High-Performance Liquid Chromatography/Tandem Mass Spectrometry and Characterization of Varieties by Quantitative Phenolic Fingerprints. J. Agric. Food Chem. 2013;61:12020–12031. doi: 10.1021/jf402288j. [DOI] [PubMed] [Google Scholar]
- 8.Gil M.I., Tomás-Barberán F.A., Hess-Pierce B., Kader A.A. Antioxidant Capacities, Phenolic Compounds, Carotenoids, and Vitamin C Contents of Nectarine, Peach, and Plum Cultivars from California. J. Agric. Food Chem. 2002;50:4976–4982. doi: 10.1021/jf020136b. [DOI] [PubMed] [Google Scholar]
- 9.Chun O.K., Kim D.-O., Moon H.Y., Kang H.G., Lee C.Y. Contribution of individual polyphenolics to total antioxidant capacity of plums. J. Agric. Food Chem. 2003;51:7240–7245. doi: 10.1021/jf0343579. [DOI] [PubMed] [Google Scholar]
- 10.Vasantha Rupasinghe H.P., Jayasankar S., Lay W. Variation in total phenolics and antioxidant capacity among European plum genotypes. Sci. Hort. 2006;108:243–246. doi: 10.1016/j.scienta.2006.01.020. [DOI] [Google Scholar]
- 11.Cosmulescu S., Trandafir I., Nour V., Botu M. Total phenolic, flavonoid distribution and antioxidant capacity in skin, pulp and fruit extracts of plum cultivars. J. Food Biochem. 2015;39:64–66. doi: 10.1111/jfbc.12112. [DOI] [Google Scholar]
- 12.Papachatzis A., Gougoulias N., Kalorizou H., Kalfountzos D., Wogiatzi E., Vyrlas P., Manthos I., Koutinas N., Pateras D. Comparative study on polyphenols content and antioxidant effect of plum cultivars from the island Skopelos, Greece. Acta Hortic. 2017;1175:77–82. doi: 10.17660/ActaHortic.2017.1175.15. [DOI] [Google Scholar]
- 13.Sahamishirazi S., Moehring J., Claupein W., Graeff-Hoenninger S. Quality assessment of 178 cultivars of plum regarding phenolic, anthocyanin and sugar content. Food Chem. 2017;214:694–701. doi: 10.1016/j.foodchem.2016.07.070. [DOI] [PubMed] [Google Scholar]
- 14.Murathan Z.T., Arslan M., Erbil N. Analyzing Biological Properties of Some Plum Genotypes Grown in Turkey. Int. J. Fruit Sci. 2020;20:S1729–S1740. doi: 10.1080/15538362.2020.1830917. [DOI] [Google Scholar]
- 15.Ceccarelli D., Antonucci F., Talento C., Ciccoritti R. Chemical characterization in the selection of Italian autochthonous genotypes of plum. Sci. Hortic. 2021;281:109922. doi: 10.1016/j.scienta.2021.109922. [DOI] [Google Scholar]
- 16.Venter A., Joubert E., De Beer D. Characterisation of Phenolic Compounds in South African Plum Fruits (Prunus salicina Lindl.) using HPLC Coupled with Diode-Array, Fluorescence, Mass Spectrometry and On-Line Antioxidant Detection. Molecules. 2013;18:5072–5090. doi: 10.3390/molecules18055072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dinardo A., Subramanian J., Singh A. Investigation of Antioxidant Content and Capacity in Yellow European Plums. Int. J. Fruit Sci. 2017;18:99–116. doi: 10.1080/15538362.2017.1381873. [DOI] [Google Scholar]
- 18.Wolf J., Ondrášek I., Nečas T. Evaluation of pomologi¬cal and qualitative traits in plum cultivars delivered from Prunus domestica, P. salicina, P. cerasifera and their hybrids. Acta Hortic. 2019;1260:171–179. doi: 10.17660/ActaHortic.2019.1260.27. [DOI] [Google Scholar]
- 19.Kristl J., Slekovec M., Tojnko S., Unuk T. Extractable antioxidants and non-extractable phenolics in the total antioxidant activity of selected plum cultivars (Prunus domestica L.): Evolution during on-tree ripening. Food Chem. 2011;125:29–34. doi: 10.1016/j.foodchem.2010.08.027. [DOI] [Google Scholar]
- 20.Drogoudi P., Pantelidis G., Papachatzis A. Fruit Physical and Chemical Characters in Twelve European and Japanese Plum Genotypes during Two Harvesting Years. Acta Hortic. 2012;968:193–196. doi: 10.17660/ActaHortic.2012.968.27. [DOI] [Google Scholar]
- 21.Gaši F., Sehic J., Grahic J., Hjeltnes S.H., Ordidge M., Benedikova D., Blouin-Delmas M., Drogoudi P., Giovannini D., Höfer M., et al. Genetic assessment of the pomological classification of plum Prunus domestica L. accessions sampled across Europe. Genet. Resour. Crop Evol. 2020;67:1137–1161. doi: 10.1007/s10722-020-00901-y. [DOI] [Google Scholar]
- 22.Rampáčková E., Göttingerová M., Kiss T., Ondrášek I., Venuta R., Wolf J., Nečas T., Ercisli S. CIELAB analysis and quantitative correlation of total anthocyanin content in European and Asian plums. Eur. J. Hortic. Sci. 2021;86:453–460. doi: 10.17660/eJHS.2021/86.5.1. [DOI] [Google Scholar]
- 23.Cömert E.D., Mogol B.A., Gökmen V. Relationship between color and antioxidant capacity of fruits and vegetables. Curr. Res. Food Sci. 2020;2:1–10. doi: 10.1016/j.crfs.2019.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Goldner K., Michaelis S.V., Neumüller M., Treutter D. Phenolic contents in fruit juices of plums with different skin colors. J. Appl. Bot. Food Qual. 2015;88:322–326. doi: 10.5073/jabfq.2015.088.046. [DOI] [Google Scholar]
- 25.Yu X.M., Rizwan H.M., Li P., Luo S.X., Sheramet I., Wu W.F., Lin J., Zheng S.X., Oelmüller R., Chen F.X. Comparative studies on the physiochemical properties, phenolic compounds and antioxidant activities in 13 Japanese plum cultivar grown in the subtropical region of China. Appl. Ecol. Envir. Res. 2020;18:3147–3159. doi: 10.15666/aeer/1802_31473159. [DOI] [Google Scholar]
- 26.Johnson J.B., Collins T., Mani J.S., Naiker M. Nutritional Quality and Bioactive Constituents of Six Australian Plum Varieties. Int. J. Fruit Sci. 2020;21:115–132. doi: 10.1080/15538362.2020.1860863. [DOI] [Google Scholar]
- 27.Merkouropoulos G., Ganopoulos I., Tsaftaris A., Papadopoulos I., Drogoudi P. Combination of high resolution melting (HRM) analysis and SSR molecular markers speeds up plum genotyping: Case study genotyping the Greek plum GeneBank collection. Plant Genet. Res. 2016;15:366–375. doi: 10.1017/S1479262116000022. [DOI] [Google Scholar]
- 28.Sehic J., Gaši F., Benedikova D., Blouin M., Drogoudi P., Giovannini D., Höfer M., Lācis G., Lateur M., Ognjanov V., et al. Genetic diversity of Prunus domestica selected from ten countries across Europe. Acta Hortic. 2019;1260:159–162. doi: 10.17660/ActaHortic.2019.1260.25. [DOI] [Google Scholar]
- 29.Singleton V.L., Orthofer R., Lamuela-Raventos R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 1999;299:152–178. [Google Scholar]
- 30.Drogoudi P., Gerasopoulos D., Kafkaletou M., Tsantili E. Phenotypic characterization of qualitative parameters and antioxidant contents in peach and nectarine fruit and changes after jam preparation. J. Sci. Food Agric. 2017;97:3374–3383. doi: 10.1002/jsfa.8188. [DOI] [PubMed] [Google Scholar]
- 31.Blois M.S. Antioxidant Determinations by the Use of a Stable Free Radical. Nature. 1958;181:1199–1200. doi: 10.1038/1811199a0. [DOI] [Google Scholar]
- 32.Metsalu T., Vilo J. ClustVis: A web tool for visualizing clustering of multivariate data using Principal Component Analysis and heatmap. Nucl. Acids Res. 2015;43:W566–W570. doi: 10.1093/nar/gkv468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Arion C.M., Tabart J., Kevers C., Niculaua M., Filimon R., Beceanu D., Dommes J. Antioxidant potential of different plum cultivars during storage. Food Chem. 2014;146:485–491. doi: 10.1016/j.foodchem.2013.09.072. [DOI] [PubMed] [Google Scholar]
- 34.Drogoudi P., Pantelidis G., Goulas V., Manganaris G., Ziogas V. The appraisal of qualitative parameters and antioxidant contents during postharvest peach fruit ripening underlines the genotype significance. Postharvest Biol. Technol. 2016;115:142–150. doi: 10.1016/j.postharvbio.2015.12.002. [DOI] [Google Scholar]
- 35.Sarıdaş M.A., Ağçam E., Akbaş F.C., Akyıldiz A., Kargı S.P. Comparison of superior bred strawberry genotypes with popular cultivars in terms of fruit bioactive compounds during the wide harvest dates. S. Afr. J. Bot. 2022;147:142–152. doi: 10.1016/j.sajb.2022.01.010. [DOI] [Google Scholar]
- 36.Ariza M., Martínez-Ferri E., Domínguez P., Medina J., Miranda L., Soria C. Effects of harvest time on functional compounds and fruit antioxidant capacity in ten strawberry cultivars. J. Berry Res. 2015;5:71–80. doi: 10.3233/JBR-150090. [DOI] [Google Scholar]
- 37.Wang S.Y., Zheng W. Effect of Plant Growth Temperature on Antioxidant Capacity in Strawberry. J. Agric. Food Chem. 2001;49:4977–4982. doi: 10.1021/jf0106244. [DOI] [PubMed] [Google Scholar]
- 38.Drogoudi P.D., Michailidis Z., Pantelidis G. Peel and flesh antioxidant content and harvest quality characteristics of seven apple cultivars. Sci. Hortic. 2008;115:149–153. doi: 10.1016/j.scienta.2007.08.010. [DOI] [Google Scholar]
- 39.Drogoudi P.D., Tsipouridis C., Michailidis Z. Physical and chemical characters in pomegranate. Hortscience. 2005;40:1200–1203. doi: 10.21273/HORTSCI.40.5.1200. [DOI] [Google Scholar]
- 40.Butac M., Militaru M., Budan S. Criteria for harvesting plums at the optimum maturity for Romanian varieties of the fresh fruit market. Fruit Grow. Res. 2012;28:1–6. [Google Scholar]
- 41.Crisosto C.H., Crisosto G.M., Echeverria G., Puy J. Segregation of plum and pluot cultivars according to their organoleptic characteristics. Postharvest Biol. Technol. 2007;44:271–276. doi: 10.1016/j.postharvbio.2006.12.005. [DOI] [Google Scholar]
- 42.Myracle A.D., Castonguay Z.J., Elwell A., Moran R.E. Fruit Quality and Consumer Acceptability of Three Plum Types and 14 Plum Cultivars Grown in Maine for a Local Market. Hort. Technol. 2018;28:230–238. doi: 10.21273/HORTTECH03958-18. [DOI] [Google Scholar]
- 43.Liverani A., Giovannini D., Versari N., Sirri S., Brandi F. Japanese and European Plum Cultivar Evaluation in the Po Valley of Italy: Yield And Climate Influence. Acta Hortic. 2010;874:327–336. doi: 10.17660/ActaHortic.2010.874.47. [DOI] [Google Scholar]
- 44.Dirlewanger E., Moing A., Rothan C., Svanella L., Pronier V., Guye A., Plomion C., Monet R. Mapping QTLs controlling fruit quality in peach (Prunus persica (L.) Batsch) Theor. Appl. Genet. 1999;98:18–31. doi: 10.1007/s001220051035. [DOI] [Google Scholar]
- 45.Cantín C.M., Gogorcena Y., Moreno M. Phenotypic diversity and relationships of fruit quality traits in peach and nectarine [Prunus persica (L.) Batsch] breeding progenies. Euphytica. 2010;171:211. doi: 10.1007/s10681-009-0023-4. [DOI] [Google Scholar]
- 46.Drogoudi P.D., Vemmos S., Pantelidis G., Petri E., Tzoutzoukou C., Karayiannis I. Physical characters and antioxidant, sugar and mineral nutrient contents in fruit from 29 apricot (Prunus armeniaca L.) cultivars and hybrids. J. Agric. Food Chem. 2008;56:10754–10760. doi: 10.1021/jf801995x. [DOI] [PubMed] [Google Scholar]
- 47.Ruiz D., Egea J. Phenotypic diversity and relationships of fruit quality traits in apricot (Prunus armeniaca L.) germplasm. Euphytica. 2008;163:143–158. doi: 10.1007/s10681-007-9640-y. [DOI] [Google Scholar]
- 48.Lozano M., Vidal-Aragón M.C., Hernández M.T., Ayuso M.C., Bernalte M.J., García J., Velardo B. Physicochemical and nutritional properties and volatile constituents of six Japanese plum (Prunus salicina Lindl.) cultivars. Eur. Food Res. Technol. 2008;228:403–410. doi: 10.1007/s00217-008-0946-3. [DOI] [Google Scholar]
- 49.Dowling C. Master Thesis. University of Guelph; Guelph, ON, Canada: 2014. The Polyphenolic Composition and Antioxidant Capacity of Yellow European plums (Prunus domestica L.) and Novel Golden Prunes. [Google Scholar]
- 50.Mirheidari F., Khadivi A., Moradi Y., Paryan S. The selection of superior plum (Prunus domestica L.) accessions based on morphological and pomological characterizations. Euphytica. 2020;216:87. doi: 10.1007/s10681-020-02617-7. [DOI] [Google Scholar]
- 51.Fu L., Bo-Tao X., Xiang-Rong X., Ren-You G., Yuan Z., En-Qin X., Hua-Bin L. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011;129:345–350. doi: 10.1016/j.foodchem.2011.04.079. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.