Abstract
A female-term neonate showed a severe respiratory distress syndrome (RDS) at hour 3 of life requiring her transfer to intensive care. She was intubated and started on assist-control mechanical ventilation associated with inhaled nitric oxide then high-frequency oscillation ventilation at day 12. Chest X-ray was gradually deteriorating. Chest computed tomography (CT) scan revealed diffuse interstitial lung disease. Flexible bronchoscopy excluded pulmonary alveolar proteinosis. The genetics study confirmed surfactant protein-B (SP-B) deficiency caused by the novel homozygous c.770T>C, p.Leu257Pro mutation in the SFTPB gene (NM_000542.5). Methylprednisolone pulse therapy was administered from day 20. As the infant worsened, azithromycin, sildenafil, and inhaled steroids were added at the age of 6 months and azathioprine at the age of 10 months. At the age of 12 months, chest CT showed diffuse “crazy-paving.” The infant died of respiratory failure at the age of 13 months. Unexplained neonatal RDS should raise the suspicion of SP-B disease. This novel mutation could be part of the mutations allowing partial SP-B production result in prolonged survival. Lung transplant in infants, unavailable in numerous countries, remains the unique way to reverse the fatal outcome.
Keywords: Diffuse interstitial lung disease, infant, neonatal respiratory distress, newborn, surfactant protein-B deficiency
Respiratory distress syndrome (RDS) is the most frequent cause of morbidity in newborns. Conditions such as meconium aspiration, pneumonia, and persistent pulmonary hypertension (PH) are usually suspected in a full-term newborn. Severe RDS resistant to treatment should raise the suspicion of genetic causes particularly inherited surfactant protein deficiency. ATP-binding cassette transporter A3 (ABCA3) deficiency is the most common inherited surfactant disorder, however, surfactant protein-B (SP-B) deficiency should be considered since it is potentially lethal.[1] We report on a case of an SP-B deficient infant survivor.
Case Report
A female neonate, from first-degree cousins, was born at 38 weeks gestation with spontaneous breath on delivery, normal APGAR scores, and birth weight of 3200 g. At hour 3 of life, she showed severe RDS requiring her transfer to intensive care.
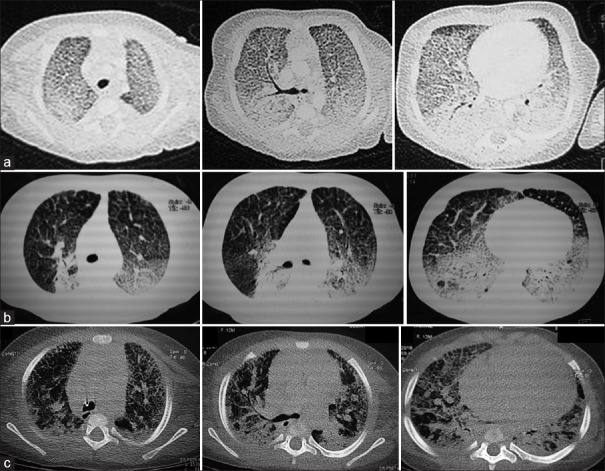
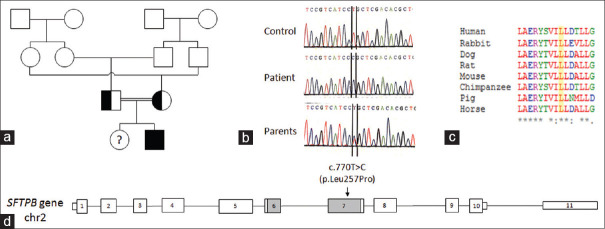
On examination, she had tachypnea with a respiratory rate of 60 rpm and a Silverman-Anderson score of 5. Initial chest X-ray (CXR) revealed hyperlucent lungs and normal cardiac silhouette. The newborn was intubated and started on assist-control mechanical ventilation (MV) associated with dobutamine infusion (10 μg/kg/min). Echocardiography at day 2 of life showed left ventricular ejection fraction at 53%, right ventricular enlargement and paradoxical septal motion suggestive of PH. Therefore, inhaled nitric oxide (iNO) was added during 2 days. Transient improvement in respiratory parameters allowed MV to switch to noninvasive positive pressure ventilation within 5 days. However, this improvement was not sustainable. The newborn continued to have high oxygen requirements, motivating reintubation at day 12 and starting high-frequency oscillation ventilation (HFOV). CXR was gradually deteriorating with bilateral infiltrations. Chest computed tomography (CT) revealed diffuse interstitial lung disease (DILD) [Figure 1a]. Flexible bronchoscopy excluded pulmonary alveolar proteinosis. In fact, the liquid of the bronchoalveolar lavage did not have a milky appearance and the cytological examination does not show foamy macrophages with extracellular globular hyaline material positive on periodic acid–Schiff. Lung biopsy was impossible to achieve. The genetics study, performed in the Department of Biochemistry and Molecular Biology at Henri Mondor hospital in Creteil, France, confirmed SP-B deficiency caused by the novel homozygous c. 770T> C, p.Leu257Pro mutation in the SFTPB gene (NM_000542.5). This substitution affects a well-conserved amino acid within the mature form of SP-B. It was predicted by bioinformatic tools to be probably damaging (CADD score: 23.9)[2] [Figure 2]. MV was continued for a total of 26 days, followed by high-flow nasal cannula (HFNC) oxygen therapy. Methylprednisolone pulse therapy (300 mg/m2/day, three doses monthly) was administered from day 20.
Figure 1.
Patient's chest computed tomography findings: (a) At day 12 of life: diffuse interstitial lung disease with markedly prominent interlobular septa; (b) At 6 months: diffuse ground-glass opacities associated with reticular infiltrates and posterior segmental consolidations in both upper and lower lobes; (c) At 12 months: radiologic worsening: “crazy-paving” pattern of ground-glass opacities associated with interlobular septal thickening and enlargement of the right heart
Figure 2.
Genetic analysis of the SFTPB c. 770T>C (p.Leu257Pro) variant. (a) Genetic tree of the family. The homozygous affected patient is in black, her healthy heterozygous parents are in white and black. His healthy sister was not tested. (b) Sanger sequencing of the patient and his parents. The normal base T (red) and the mutated base C (blue) are framed. (c) Partial amino acid alignment of surfactant protein-B sequences (codon 249–263) showed important conservation through mammalians. Multiple sequence alignment was performed with Clustal Omega (1.2.2). Asterisks indicate positions which have a fully conserved residue. The human Leu257 position is highlighted (yellow). (d) Schematic representation of the gene protein and predicted position of the p.Leu257Pro mutation. Surfactant protein-B protein undergoes a complex maturation process involving proteolytic cleavages. The gray parts encode the sequences conserved in the mature surfactant protein-B protein (271–279)
At the age of 2 months, slow weight growth (3300 g) motivated high-calorie diet. Echocardiography showed PH at 40 mmHg and 65 mmHg, respectively, at the age of 3 and 6 months. At the age of 6 months, chest CT showed DILD with less ground-glass opacities [Figure 1b]. Azithromycin, sildenafil, and inhaled steroids were added at this stage of the disease.
At 10 months, the patient had acceptable growth. She was 74 cm tall (+1SD above normal) and weighed 7.6 kg (−1SD below normal). However, she was more dyspneic and increased oxygen requirement. Azathioprine was added.
At the age of 12 months, chest CT showed diffuse “crazy-paving” [Figure 1c]. The infant kept worsening. Her peripheral oxygen saturation was around 89%–90% under 15 L/min of oxygen through HFNC. She died of respiratory failure at the age of 13 months.
One year later, the mother was pregnant again. Antenatal genetic testing showed the fetus to be heterozygous. Consequently, the newborn was healthy.
Discussion
To our knowledge, this is the first documented case of SP-B deficiency in Tunisia with genetic confirmation. SP-B deficiency was first described in 1993 in two full-term siblings with fatal RDS.[3] SP-B is a hydrophobic protein essential for surfactant homeostasis. SP-B deficiency is a rare autosomal recessive condition mostly fatal in homozygotes, whereas heterozygous carriers are asymptomatic. Almost 30 mutations have been identified. The c. 361delinsGAA mutation (legacy name: 121 ins 2), associated with approximately 60% of the reported cases, causes severe RDS shortly after birth, lethal within the first 6 months of life. Other less frequent mutations allowing partial SP-B production result in prolonged survival.[1,4] Our novel mutation could be part of them.
Unexplained neonatal RDS should raise the suspicion of SP-B disease when associated with infiltrates at CXR and parental consanguinity. Chest CT typically reveals a diffuse “crazy-paving” pattern of ground-glass opacities and interlobular septal thickening.[4,5] This should prompt consideration of genetic testing for SP-B abnormalities which might guide treatment options, prognosis debate with the parents, and adapted genetic counseling.
Recurrent surfactant replacement therapy is ineffective. The patients are often managed with high-dose steroids and maximum supportive therapy including MV, HFOV, and iNO. Extracorporeal membrane oxygenation is a bridge to lung transplant which remains the unique way to reverse the fatal outcome. Additional treatments including hydroxychloroquine and azithromycin are options usually used for other inherited surfactant protein deficiencies, such as ABCA3 and SP-C deficiencies, since cases of SP-B deficiency with prolonged survival are rare.[5,6,7]
The five-year posttransplant survival rate is around 50%, similar to that of lung transplant recipients for other causes.[5,8] However, lung transplant in neonates remains unavailable in numerous countries, as in Tunisia, which made it impossible to save our patient's life. Gene therapy could be the hoped-for solution after promising results in animal models (viral-mediated SP-B gene delivery).[9]
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient's parents have given consent for images and other clinical information to be reported in the journal. The patient's parents understand that her name and initials will not be published and due efforts will be made to conceal identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.Magnani JE, Donn SM. Persistent respiratory distress in the term neonate: Genetic surfactant deficiency diseases. Curr Pediatr Rev. 2020;16:17–25. doi: 10.2174/1573396315666190723112916. [DOI] [PubMed] [Google Scholar]
- 2.Sievers F, Wilm A, Dineen D, Gibson TJ, Karplus K, Li W, et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol Syst Biol. 2011;7:539. doi: 10.1038/msb.2011.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nogee LM, de Mello DE, Dehner LP, Colten HR. Brief report: Deficiency of pulmonary surfactant protein B in congenital alveolar proteinosis. N Engl J Med. 1993;328:406–10. doi: 10.1056/NEJM199302113280606. [DOI] [PubMed] [Google Scholar]
- 4.López-Andreu JA, Hidalgo-Santos AD, Fuentes-Castelló MA, Mancheño-Franch N, Cerón-Pérez JA, Esteban-Ricós MJ, et al. Delayed presentation and prolonged survival of a child with surfactant protein B deficiency. J Pediatr. 2017;190:268–70. doi: 10.1016/j.jpeds.2017.07.009. [DOI] [PubMed] [Google Scholar]
- 5.Kurath-Koller S, Resch B, Kraschl R, Windpassinger C, Eber E. Surfactant protein B deficiency caused by homozygous C248X mutation – A case report and review of the literature. AJP Rep. 2015;5:e53–9. doi: 10.1055/s-0035-1545668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bush A, Gilbert C, Gregory J, Nicholson AG, Semple T, Zampoli M, et al. Pediatric interstitial lung disease. JPATS. 2021;2:18–32. [Google Scholar]
- 7.Klay D, Hoffman TW, Harmsze AM, Grutters JC, van Moorsel CH. Systematic review of drug effects in humans and models with surfactant-processing disease. Eur Respir Rev. 2018;27:170135. doi: 10.1183/16000617.0135-2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Palomar LM, Nogee LM, Sweet SC, Huddleston CB, Cole FS, Hamvas A. Long-term outcomes after infant lung transplantation for surfactant protein B deficiency related to other causes of respiratory failure. J Pediatr. 2006;149:548–53. doi: 10.1016/j.jpeds.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 9.Kang MH, van Lieshout LP, Xu L, Domm JM, Vadivel A, Renesme L, et al. A lung tropic AAV vector improves survival in a mouse model of surfactant B deficiency. Nat Commun. 2020;11:3929. doi: 10.1038/s41467-020-17577-8. [DOI] [PMC free article] [PubMed] [Google Scholar]