SUMMARY
A population-based study investigated the burden of illness, including the duration of illness associated with laboratory-confirmed cases of campylobacteriosis in two health unit areas. Questionnaire data were collected for 250 cases. The median duration of illness was 8 days and 66% of cases reported symptoms of moderate severity or greater. A Cox proportional hazards model identified antimicrobial use factors associated with a significantly increased rate of symptom resolution (shorter duration of illness): macrolides for less than the recommended number of days, ciprofloxacin for at least 3 days, and antimicrobials not recommended for campylobacteriosis. The impact of antimicrobial use was consistent regardless of when, during the course of illness, the antimicrobial use began. The effectiveness of ciprofloxacin in these results may be due to the low prevalence of resistance to ciprofloxacin in isolates from this study. The effect of antimicrobials not recommended for campylobacteriosis should be further investigated.
Key words: Antimicrobial drugs, Campylobacter, epidemiology, modelling
INTRODUCTION
Campylobacter is the one of the most commonly reported enteric bacterial pathogens in many countries, including Canada [1, 2]. The burden of illness of Campylobacter cases on society results from lost time at work or school for either the case or the caregiver, and the costs of utilization of healthcare providers, hospitalization, laboratory testing and treatment. This impact is influenced by the duration, severity and scope of the patient's symptoms. The case-fatality rate for campylobacteriosis has been reported as 1–2/1000 cases [2–4]. Potential sequelae include post-infectious irritable bowel syndrome, Gullain–Barré syndrome, and reactive arthritis [5–9]. When all of these factors are considered, the overall cost of campylobacteriosis has been estimated at about US$8000 per case [10].
Campylobacteriosis is usually a self-limiting infection and treatment with macrolides or fluoroquinolones is recommended only in vulnerable populations with severe or invasive disease [8, 11–13]. However, observational studies have shown that a substantial percentage of patients take antimicrobials for their campylobacteriosis [11, 14]. The reported effect of antimicrobial treatment on duration of illness has been variable [15]. It has been suggested that the effect may vary with the period between initiation of symptoms and initiation of antimicrobial treatment [15, 16].
A population-based study was conducted over a 2-year period in the Perth District (PD) and Wellington-Dufferin-Guelph (WDG) health units in Ontario, with an objective of documenting the burden of illness associated with clinical cases of campylobacteriosis. A second objective was to investigate the factors associated with the duration of illness in Campylobacter cases.
METHODS
This research project was approved by the University of Guelph Research Ethics Board. Agreements were put in place with hospital and private laboratories to have isolates obtained from clients living in two health unit areas forwarded to the Public Health Ontario Laboratory – Toronto (PHL). These agreements ensured the confidentiality of patient information.
Data collection and laboratory methods for this project have been described previously [17]. In brief, laboratory-confirmed cases of campylobacteriosis during the study period were eligible for inclusion in the study. Cases provided consent for participation in the study at the time of questionnaire administration. Isolates were forwarded to the PHL for confirmation, speciation, and antimicrobial susceptibility testing. Minimum inhibitory concentrations were determined by the E-test® (bioMérieux, Sweden) with the following breakpoints: amoxicillin/clavulanic acid (⩾32 μg/ml), ampicillin (⩾32 μg/ml), chloramphenicol (⩾32 μg/ml), ciprofloxacin (⩾4 μg/ml), clindamycin (⩾4 μg/ml), erythromycin (⩾8 μg/ml), gentamicin (⩾16 μg/ml), nalidixic acid (⩾32 μg/ml), and tetracycline (⩾16 μg/ml).
Questionnaire data
Data on the burden of illness associated with campylobacteriosis were collected as part of a comprehensive telephone questionnaire. The telephone interview was conducted by personnel from the local health unit when a laboratory-confirmed case of Campylobacter was identified. Diarrhoea was defined as ⩾2 loose stools or bowel movements in a 24-h period and was self-reported. Burden-of-illness indicators included the reported type, duration, and severity of clinical signs, activity limitations, and healthcare utilization. Severity of illness was reported by cases based on the following definitions:
Quite mild: feeling slightly unwell but able to do all normal activities.
Fairly mild: feeling quite unwell but able to do most normal activities.
Moderate: having to stay at home but able to get out of bed for limited activities.
Fairly severe: confined to bed at home and unable to do any normal activities.
Quite severe: hospitalized.
Antimicrobial use data captured through the questionnaire included: antimicrobial(s) used in the 30 days prior to illness, antimicrobial(s) used during illness, and the start and end dates of antimicrobial use during illness. Dichotomous variables were analysed using the χ2 and Fisher's exact tests as appropriate. Differences between groups of continuous variables were evaluated with the Student's t test.
Survival analysis
Stata Intercooled version 11 (Stata Corporation, USA) was used for the analysis. A Cox proportional hazards model was developed to identify factors associated with duration of illness. The date of failure was the date all symptoms ceased. The date of censor was the date of last contact when symptoms were ongoing. Covariates included in the univariable analysis are listed in Table 2. Age was analysed as both continuous and categorical variables, category breakpoints were determined based on historical age distribution data from the participating health units as described previously (5, 17, 41, 61 years) [17]. Season was categorized as winter (December–February), spring (March–May), summer (June–August) and autumn (September–November). Individual variables were created for the most common medications taken during the 4 weeks prior to illness including gastroesophageal reflux, pain, high blood pressure, immunosuppression, and thryroid medications. A general variable was also created which included any medications taken during the 4 weeks prior to illness.
Table 2.
Summary of reported symptoms in laboratory-confirmed cases of campylobacterosis in Perth District and Wellington-Dufferin-Guelph health units
| Symptom | No. of cases | N* | (%) |
|---|---|---|---|
| Diarrhoea | 248 | 250 | (99·2) |
| Stomach cramps | 212 | 235 | (90·2) |
| Fatigue | 221 | 246 | (89·8) |
| Loss of appetite | 208 | 247 | (84·2) |
| Fever | 180 | 246 | (73·2) |
| Weight loss | 147 | 228 | (64·5) |
| Headache | 142 | 224 | (63·4) |
| Nausea | 149 | 235 | (63·4) |
| Blood in stool | 114 | 237 | (48·1) |
| Bloating | 86 | 235 | (36·6) |
| Vomit | 71 | 247 | (28·7) |
| Other† | 79 | 248 | (31·9) |
Number of cases with responses varied by symptom.
Symptoms most commonly reported under ‘Other’ included joint pain (11 cases), muscle pain (21 cases), dizziness (8 cases), and rash (4 cases).
In order to account for time from initiation of symptoms to initiation of antimicrobial therapy, a categorical time-varying variable was created. This variable was coded according to the antimicrobial utilized and if/when the antimicrobial was taken for the recommended duration. The antimicrobial categories evaluated were ciprofloxacin, macrolide or ‘other’ antimicrobial. These categories were selected based on previous research and clinical guidelines for the treatment of campylobacterosis [8, 12, 13, 16, 18–20]. For classification of completeness of therapy duration, the following recommended durations were used: 3 days for azithromycin [18]; 3 days for ciprofloxacin [18]; 5 days for erythromycin [12, 18]; and 7 days for clarithromycin [13]. Antimicrobials in the ‘other’ category were not expected to be clinically effective and a recommended duration was not considered [8, 12, 13, 18, 20]. Therefore, antimicrobial use during illness (AMU) was categorized as no antimicrobial use, incomplete macrolide, complete macrolide, incomplete ciprofloxacin, complete ciprofloxacin, or ‘other’ treatment. Cases were categorized as: no use prior to antimicrobials being taken, incomplete use once antimicrobials were initiated, and as complete use if/when the recommended duration was achieved. Although some cases continued to have symptoms after antimicrobial use had ended, the antimicrobial use variable was not returned to the value for ‘no use’ due to the expected impact of antimicrobials on the gastrointestinal flora and the relatively short overall duration of symptoms. In these instances, the antimicrobial use variable remained at its value when antimicrobial use ceased, i.e. incomplete or complete. Antimicrobial use that continued after the date of failure (symptom resolution) was not included in the analysis. Cases that took more than one antimicrobial during their illness were excluded from the analysis.
The AMU variable was assessed for a time-varying effect by first, testing the proportional hazards assumption on the basis of the Schoenfeld residuals on the log and time scales and second, adding interaction terms between AMU and analysis time to the univariable Cox proportional hazards model. Aalen's linear hazard model was used to parameterize the potential time-varying effect of the AMU variable [21].
Univariable analysis was performed with potential covariates utilizing log rank and Wilcoxon tests of equality as well as univariable Cox proportional hazards models. Variables significant at P < 0·2 for any of these tests were considered for inclusion into the multivariable analysis. Variables with a prevalence of < 2% in cases were omitted from further analysis. Variables considered for inclusion into the multivariable analysis were examined for collinearity using Pearson's or Spearman's correlation coefficients as appropriate. When the correlation coefficient for two predictors was significant at P < 0·05 after Bonferroni adjustment, one was selected for inclusion into the multivariable analysis based on level of significance, number of missing observations, and reliability.
The preliminary main-effects Cox proportional hazards model was developed utilizing manual backward selection. Variables were retained if the likelihood ratio test was significant (P ⩽ 0·05) or if the coefficient(s) of other covariates changed by >20%. When all remaining variables were significant at P ⩽ 0·05 or were confounders, variables that were not significant in the univariable analysis were introduced and evaluated for significance. Biologically plausible interaction terms for the resulting model were assessed. The assumption of proportional hazards was evaluated by the link test, scaled Schoenfeld residuals, interaction terms for covariates and time, Cox proportional hazards plots stratified by each level of a covariate after adjusting for other covariates, and Kaplan–Meier vs. predicted survival plots for each level of covariate. Outliers and influential observations were identified with Cox–Snell residuals and assessed for biological plausibility.
RESULTS
There were 317 laboratory-confirmed cases during the study period and 78·9% (n = 250) were successfully contacted and agreed to participate resulting in questionnaire data collected from 51 cases in PD and 199 cases in WDG.
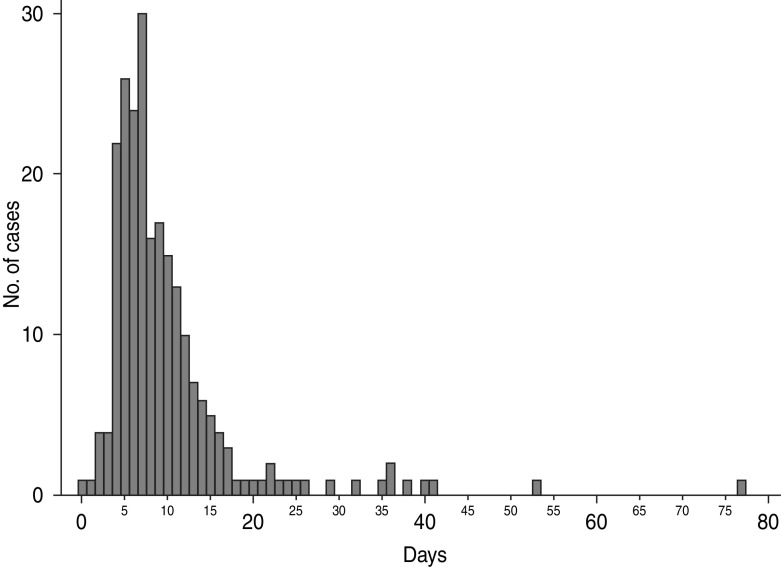
Demographic data from this study have been reported previously [17]. Briefly, 140 cases were male and 109 were female. Cases had a median age of 27·4 (mean 29·0) years. The mean duration of illness was 10 days (median 8 days, range 0·5–77 days) with a right-skewed distribution (Fig. 1). Fourteen (5·6%) cases had ongoing symptoms at the time of the telephone interview, which ranged from 4 to 29 days after symptoms began (median 7 days).
Fig. 1.
Duration of illness in laboratory-confirmed cases of campylobacterosis in Perth District and Wellington-Dufferin-Guelph health units (n = 249).
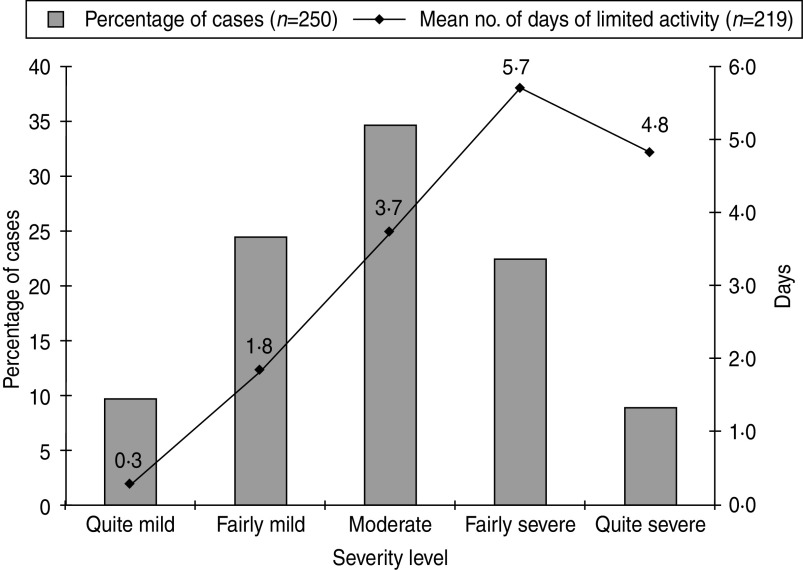
One hundred and ninety-one (76·0%) cases were unable to carry out regular activities during their illness. Of 150 cases attending school or working outside the home, 135 (90·0%) took time off due to their illness (mean and median 4 days, range 0·5–14 days). The mean number of days that cases were unable to perform their usual activities including going to work or school significantly increased as severity of illness increased (P = 0·001) (Fig. 2). Thirty-five (14·0%) cases had relatives/friends who missed a mean of 1·8 days of work (median 1, range 1–7 days) in order to care for them. One hundred and sixty-five (66·0%) cases reported symptoms of moderate severity or greater (Fig. 2).
Fig. 2.
Self-reported severity of illness according to a defined severity scale and mean number of days of limited activity in laboratory-confirmed cases of campylobacterosis in Perth District and Wellington-Dufferin-Guelph health units. For definitions of severity levels see the Questionnaire data section.
Symptoms reported by more than 70% of patients were diarrhoea, fatigue, stomach cramps, loss of appetite, and fever (Table 1). Forty-eight per cent of cases reported blood in the stool (Table 1) but as previously reported, the proportion of cases with blood in the stool was significantly higher for those aged <5 years (P = 0·001) [17]. In this study, three cases reported diarrhoea as the only symptom of their illness and 230 (92%) cases reported at least three symptoms. All cases in this study were laboratory-confirmed and therefore accessed the healthcare system. Of the 249 cases who reported the type of healthcare accessed during their illness, most (182, 73·1%) utilized one route, most commonly the family doctor (162, 65·1%), followed by emergency rooms (114, 45·8%) and walk-in clinics (38, 15·3%). In smaller communities, walk-in clinics may not have been available.
Table 1.
Potential covariates evaluated in univariable survival analysis in laboratory-confirmed cases of campylobacteriosis in Perth District and Wellington-Dufferin-Guelph health units
| Variable | Present/total | Percentage | Significant* |
|---|---|---|---|
| Year | |||
| Year 1 | 122/249 | 49·0 | No |
| Year 2 | 127/249 | 51·0 | |
| Season | |||
| Winter | 30/249 | 12·0 | No |
| Spring | 42/249 | 16·9 | |
| Summer | 101/249 | 40·6 | |
| Autumn | 76/249 | 30·5 | |
| Severity of illness | |||
| Quite mild | 23/249 | 9·2 | No |
| Fairly mild | 61/249 | 24·5 | |
| Moderate | 87/249 | 34·9 | |
| Fairly severe | 56/249 | 22·5 | |
| Quite severe | 22/249 | 8·8 | |
| Age (years) | |||
| <5 | 42/248 | 16·9 | No |
| 5 to <17 | 33/248 | 13·3 | |
| 17 to <41 | 99/248 | 39·9 | |
| 41 to <61 | 53/248 | 21·4 | |
| ⩾61 | 21/248 | 8·5 | |
| Gender | |||
| Male | 140/249 | 56·2 | No |
| Female | 109/249 | 43·8 | |
| Chronic health conditions† | 74/250 | 29·6 | Yes |
| Antimicrobial use prior to illness | 12/244 | 4·9 | No |
| Non-antimicrobial use prior to illness | 90/249 | 36·1 | Yes |
| Gastroesophageal reflux medication prior to illness | 28/249 | 11·2 | No |
| Pain medication prior to illness | 16/249 | 6·4 | Yes |
| Blood pressure medication prior to illness | 11/249 | 4·4 | No |
| Immunosuppressive medication prior to illness | 9/249 | 3·6 | No |
| Thyroid medication prior to illness | 7/249 | 2·8 | No |
| Antimicrobial use during illness‡ | Yes | ||
| Incomplete ciprofloxacin | 17/210 | 8·1 | |
| Complete ciprofloxacin | 17/210 | 8·1 | |
| Incomplete macrolide | 44/210 | 21·0 | |
| Complete macrolide | 7/210 | 3·3 | |
| Other antimicrobial | 11/210 | 5·2 | |
| No antimicrobials | 114/210 | 54·3 | |
| Anti-diarrhoeal medication during illness | 129/244 | 52·9 | Yes |
| Analgesic during illness | 180/248 | 72·6 | Yes |
| Anti-nausea medication during illness | 57/245 | 23·3 | No |
| Rehydration fluids during illness† | 59/244 | 24·2 | Yes |
| Campylobacter isolate resistant to ciprofloxacin | 6/124 | 4·8 | Yes |
| Campylobacter isolates resistant to erythromycin | 2/124 | 1·6 | Yes |
| Campylobacter isolate resistant to ⩾1 of the antimicrobials tested | 66/124 | 53·2 | No |
Significant at P < 0·2 on at least one of log rank or Wilcoxon tests, or in a univariable Cox proportional hazards model.
Not submitted to multivariable model due to collinearity.
Assessed using a time-varying variable. For the purposes of this table, classification was at the time symptoms ceased and includes cases that took an antimicrobial and provided a start date as well as cases that did not take an antimicrobial during illness.
Ninety cases took medication other than antimicrobials in the 4 weeks prior to their illness, most commonly for gastroesophageal reflux (28 cases), pain (16 cases), high blood pressure (11 cases), immunosuppression (nine cases), and hyper/hypo thyroid (seven cases). Three cases took a laxative in the 4 weeks prior to illness and one case took an antidiarrhoeal drug during that time period.
Twelve (4·9%) cases took antimicrobials in the 4 weeks prior to their illness; including amoxicillin (five cases), cefaclor (one case), clarithromycin (one case), norfloxacin (one case), tetracycline (one case), trimethoprim–sulfamethoxazole and penicillin (one case), and vancomycin (one case). The reason for antimicrobial use prior to illness was not known.
The PHL received isolates from 124 cases for speciation and susceptibility testing. Isolates from the remaining 126 cases were discarded in error at the primary laboratory or were unable to be matched with case data. Of the 124 isolates, 121 (97·6%) were Campylobacter jejuni and three (2·4%) C. coli. Antimicrobial resistance in these isolates has been previously reported [17]. Briefly, no resistance to amoxicillin/clavulanic acid, chloramphenicol, or gentamicin was found. Six (4·8%) isolates were resistant to nalidixic acid and ciprofloxacin, two (1·6%) were resistant to erythromycin and 58 (46·8%) isolates were susceptible to all antimicrobials tested.
Overall 135/249 (52%) cases took antimicrobials for their campylobacteriosis. The most common antimicrobials taken during illness were ciprofloxacin (43 cases, 17·6%), azithromycin (32 cases, 13·1%), erythromycin (29 cases, 11·8%), and clarithromycin (11 cases, 4·5%). Antimicrobials not expected to be clinically effective for campylobacteriosis that were taken by cases included amoxicillin, metronidazole, trimethoprim–sulfamethoxazole, sulfa drugs, and tetracycline. Ten cases took more than one antimicrobial for their illness. As previously reported, cases from this study that were treated with antimicrobials did not significantly differ from those that were not treated with antimicrobials with regards to age category, severity of illness, chronic medical condition or history of recent international travel (P > 0·05) [17].
In this study 22 cases began taking antimicrobials within 3 days of the onset of symptoms and 108 cases began taking antimicrobials >3 days from the onset of symptoms. The number of days between the onset of symptoms and the initiation of antimicrobial treatment ranged from 0 to 37 days (median 6 days). Thirty-two (24·4%) cases began taking antimicrobials after their symptoms had stopped.
In cases that used antimicrobials during their illness, 51 cases took a macrolide and provided a start date. Of these, seven (13·7%) took the recommended course and were classified as complete macrolide and 44 were incomplete macrolide. Thirty-nine cases were incomplete because their symptoms ended prior to the completion of the recommended course of treatment. Although antimicrobial use after symptoms ended was not included in the analysis, it is worth noting that of these 39 cases, 29 continued to take the macrolide after their symptoms had ended and until at least the recommended course of treatment was completed.
Thirty-four cases took ciprofloxacin and provided a start date. Seventeen were classified as complete ciprofloxacin and 17 were incomplete ciprofloxacin. Of these, 14 cases were incomplete when their symptoms ended prior to the completion of the recommended course of treatment. Eleven cases continued to take ciprofloxacin after their symptoms had ended for at least the recommended course of treatment.
Survival analysis
Variables that were assessed in the univariable analysis are given in Table 2. Resistance to erythromycin was not included in the analysis because the prevalence was less than 2% of cases. The use of rehydration fluids was significant in the univariable survival analysis but this variable was significantly collinear with the consumption of pain medications. Since more cases took the latter and it was more significantly associated with duration of illness, the rehydration fluid variable was not submitted to the multivariable model (Table 2). Having a chronic infection that could weaken the immune system and taking a non-antimicrobial medication in the 4 weeks prior to illness were both significant in the univariable survival analysis but were significantly collinear (Table 2). Taking a non-antimicrobial medication in the 4 weeks prior to illness was more significantly associated with duration of illness than chronic illness and was more reliable. Therefore taking a non-antimicrobial medication in the 4 weeks prior to illness was submitted to the multivariable model. Variables for gastroesophageal reflux, blood pressure, immunosuppression, and thyroid medications in the 4 weeks prior to illness were not significant by univariable analysis. There was no significant time-varying effect of AMU during illness. Therefore the impact of antimicrobial use on the resolution of symptoms was consistent regardless of when antimicrobial use began during the course of illness.
The following variables were included in the initial multivariable model: gender, antimicrobial use during campylobacteriosis, taking an antidiarrhoeal medication during campylobacteriosis, taking an analgesic during campylobacteriosis, taking any non-antimicrobial medication in the 4 weeks prior to illness, and whether the Campylobacter isolate was resistant to ciprofloxacin. The general variable for any non-antimicrobial medications and the variable for pain medications in the 4 weeks prior to illness were significant by univariable analysis. Each of these variables was submitted to separate model-building processes. The multivariable model that included the pain medications variable had a smaller number of observations but the same final outcome as the model which included the general variable. Therefore, the model with the general variable for the use of any non-antimicrobial medications prior to illness was utilized.
None of the non-significant variables in the univariable analysis were significant when added to the preliminary model. The interaction term for the use of non-antimicrobial medications prior to illness and antimicrobial use during illness was not significant. The evaluation of the model indicated that the proportional hazards assumption was met. One case identified as an outlier was omitted from the analysis due to a very prolonged duration of illness of quite mild severity and a chronic illness where diarrhoea was a symptom. All other outliers and influential observations were biologically plausible and therefore were retained in the analysis.
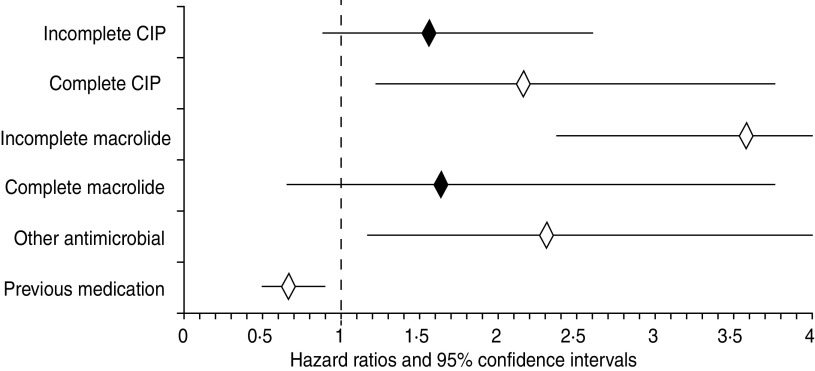
The final model included antimicrobial use during illness and the use of non-antimicrobial medication in the 4 weeks prior to illness (Fig. 3). When compared to cases who did not take antimicrobials, the rate of symptom resolution was increased for cases who took a macrolide for less than the recommended number of days (350% greater rate of symptom resolution), ciprofloxacin for at least the recommended number of days (214%) and cases who took an antimicrobial not recommended for campylobacteriosis (225%) (Fig. 4). Since the increased rate of symptom resolution is consistent regardless of when antimicrobial use began during the illness, it would result in a shorter duration of illness at the population level. Taking ciprofloxacin for less than the recommended number of days and taking a macrolide for at least the recommended number of days was not significantly associated with the rate of symptom resolution and therefore did not have a significant impact on duration of illness. When cases had taken non-antimicrobial medication in the 4 weeks prior to illness, the rate of symptom resolution was decreased by 33% compared to cases who did not take medication in the 4 weeks prior to illness. Therefore the duration of illness at the population level will be longer for cases taking non-antimicrobial medication in the 4 weeks prior to illness.
Fig. 3.
Cox proportional hazards model for duration of illness in laboratory-confirmed cases of campylobacterosis in Perth District and Wellington-Dufferin-Guelph health units (n = 227) showing hazard ratios and 95% confidence intervals. A hazard ratio >1 indicates an increased rate of symptom resolution and therefore a decreased duration of illness compared to no antimicrobial use. A hazard ratio <1 indicates a decreased rate of symptom resolution and therefore an increased duration of illness compared to no antimicrobial use. ◊, Significant effect on duration of illness; ⧫, non-significant effect on duration of illness; CIP, ciprofloxacin. * Previous medication includes only non-antimicrobial medication.
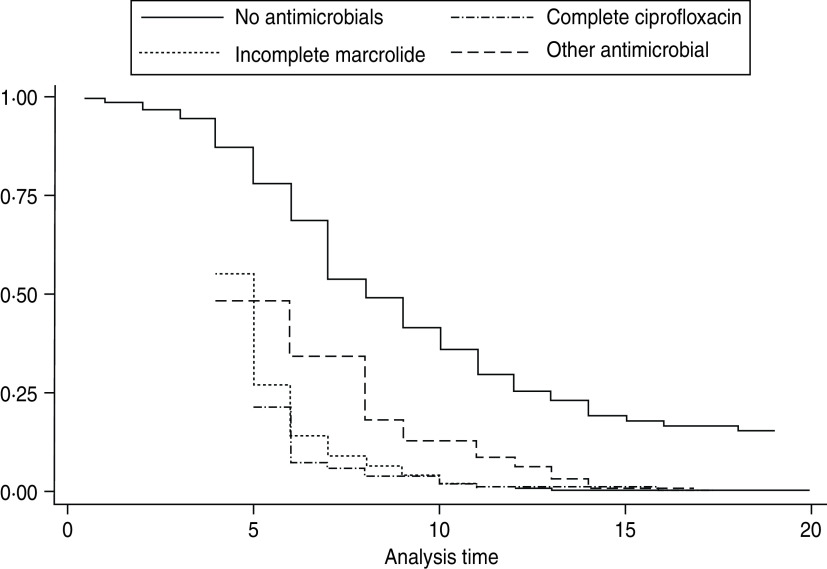
Fig. 4.
Predicted survival curves of significant antimicrobial use variables from Cox proportional hazards model for duration of illness in laboratory-confirmed cases of campylobacterosis in Perth District and Wellington-Dufferin-Guelph health units (n = 227) with adjustment for prior use of non-antimicrobials.
DISCUSSION
The median and range of duration of illness in this study is comparable with previously reported research [14, 22]. Duration of diarrhoea was not specifically recorded in this study. Although duration of diarrhoea is commonly reported in both observational studies and clinical trials, the overall duration of illness is less frequently reported [15, 22–25]. It is important to note that the overall duration of illness is not necessarily equivalent to the duration of diarrhoea. Overall duration better represents the total burden of illness due to campylobacteriosis on both the individual and society [14, 24, 26].
In this study the distribution of the duration of illness was right-skewed with a median duration of illness of 8 days but a range of 0·5–77 days. It is difficult to determine the factors impacting the duration of illness when the natural course of the illness is relatively short in a substantial proportion of cases. Moreover, 6% of cases still had symptoms at the time of the telephone interview. The Cox proportional hazards model was used to investigate the factors associated with duration of illness since it is a semi-parametric method and therefore does not include assumptions on the distribution of failure times. Since Cox proportional hazards models are essentially a series of conditional logistic regression models for each day where at least one case resolves, only cases that still have symptoms on a specific day are compared. For example, the effect of complete or incomplete antimicrobial treatment on the odds of symptoms ending 7 days after symptoms began is based only on cases that still had symptoms at the beginning of the seventh day. A limitation of this study is the small number of observations in these data once the duration of illness exceeded 17 days.
For cases taking a non-antimicrobial medication in the 4 weeks prior to their illness the rate of symptom resolution was significantly lower, resulting in an overall longer duration of illness. This may be due at least in part to effects on the underlying conditions that necessitated the medication, particularly in cases of chronic illness. These medications were taken orally and therefore may also have affected the gastrointestinal flora [27]. Reporting of medication use in our study appeared to be more reliable than reporting of chronic illness. A substantial number of cases replied ‘No’ when asked if they had a chronic condition yet reported the use of ongoing medications for chronic illness (e.g. anti-hypertensives) in a separate question. This may be due to the successful clinical management of the chronic illness with the ongoing medication. Since these variables were collinear, the previous use of the non-antimicrobial medications variable was retained for modelling. Although medications taken for non-chronic conditions were also included in the previous medications variable, the direction and magnitude of the effect was similar regardless of whether these cases were included. Therefore the more inclusive version of the variable was used.
Antimicrobial use prior to illness was not significantly associated with duration of illness in this analysis, although it was associated with an increased risk of Campylobacter infection in previous research [28]. Our study may not have had sufficient power to detect a significant effect, since <5% of cases took an antimicrobial in the 4 weeks prior to illness in this study. The potential role of prior antimicrobial use on risk or duration of illness needs to be further investigated.
Previous observational and clinical studies have evaluated the impact of antimicrobial use on the duration of campylobacteriosis by comparing the mean duration of diarrhoea or illness in cases that did or did not take antimicrobials, with variable results [14, 15]. A meta-analysis that examined 11 clinical trials on the effects of antimicrobial treatment of campylobacteriosis on duration of diarrhoea found that results from individual studies were variable, but overall antimicrobial treatment significantly decreased the mean duration of diarrhoea compared to a placebo using a random-effects model [15]. This is consistent with the effect on duration of illness of some antimicrobial treatment in the Cox proportional hazards model from this study. It should be noted, however, that there may have been an over-representation of some erythromycin treatment results in the meta-analysis due to the possible inclusion of some of the same cases in more than one paper [23, 26]. Some of the variability in effect that has been seen in previous work may be due to the use of mean duration of diarrhoea/illness as an outcome since the natural short course of illness in many cases, and the non-normal and right-censored nature of the data may have impacted the results. The increased rate of symptom resolution and therefore shorter duration of illness in cases with incomplete macrolide treatment was largely due to symptoms ending before the recommended course of treatment was complete. This may be the result of the specific macrolide and dosage used. The dosages of each antimicrobial were not recorded in this study. The specific macrolide used was captured in the questionnaire; however, there were insufficient cases to analyse these separately. Although a complete course of treatment with azithromycin was considered as 3 days for this analysis, some research has demonstrated that a single treatment is effective with higher dosages [29]. It appears that the effect of macrolides is quite rapid, regardless of when they are initiated in the course of the disease. Therefore if symptoms have not ceased by the end of the complete course of treatment, there is no treatment effect.
Ciprofloxacin is frequently recommended for the treatment of campylobacteriosis when antimicrobials are indicated [16, 18]; however, increasing prevalence of resistance to ciprofloxacin is an international concern [16, 19]. The apparent effectiveness of ciprofloxacin in this study is probably due to the relatively low prevalence of resistance to ciprofloxacin seen in isolates from this study and in previously reported Canadian data [30–32]. Cases with Campylobacter isolates resistant to ciprofloxacin had longer durations of illness in several studies [33–35] but not in others [22, 36]. Although infections with a ciprofloxacin-resistant isolate were identified as significant in the univariable Cox proportional hazards analysis (P < 0·2), this variable was not significant in the multivariable model and did not act as a confounder. The results of this study also indicate that a complete course of ciprofloxacin is required in order to have a significant impact on the rate of symptom resolution and therefore duration of illness.
Further research is necessary to investigate the effect of antimicrobials not expected to be clinically effective against Camplyobacter on the rate of symptom resolution and therefore duration of illness. However, it has been previously reported that duration of illness is decreased by antimicrobial therapy in cases of diarrhoea, even when no bacterial pathogen was isolated [23, 37], perhaps due to suppression of undetected pathogens or other effects on the intestinal flora.
Although it has been frequently reported that antimicrobial treatment is most effective when given early in the course of campylobacteriosis, there was no time-varying effect of antimicrobial use in this study [15, 16]. Therefore the impact of antimicrobial use on the resolution of symptoms was consistent regardless of when antimicrobial use began during the course of illness.
The consumption of antidiarrhoeal medications and analgesics during illness were significant in the univariable survival analysis, but they were not retained in the multivariable model. Antidiarrhoeal drugs are recommended in some cases of infectious diarrhoea and have been reported to decrease the duration of diarrhoea [38–40]. However, a large American study actually found a significantly increased duration of diarrhoea in cases taking an antidiarrhoeal [34]. The persistence of symptoms other than diarrhoea may also prevent these medications from having an impact on duration of overall illness. Although analgesics would be expected to have an impact on the rate of resolution of fever and headache symptoms, they should not impact other clinical symptoms of Campylobacter infections. The majority of cases in this study reported at least three symptoms for their illness.
Although rehydration fluids are very commonly recommended for acute gastroenteritis in general and campylobacteriosis in particular, there is no published information on the impact of this intervention on the duration of illness [12, 38].
Unexpectedly, there was no significant association between severity and duration of illness in this study population. Although severity was assessed through specific questions on activity limitations, this assessment was self-reported and did not include descriptors such as the number of stools per day. However, the number of days that cases were unable to perform normal activities and unable to attend work or school in this study was similar to previous research [14, 34].
Treatment with antimicrobials significantly accelerated the resolution of symptoms when analysed using a Cox proportional hazards model. This effect was not altered by the timing of the antimicrobial treatment. Although decreasing the duration of illness has a positive impact on the burden of illness for both the individual and society, the use of antimicrobials in the treatment of campylobacteriosis should be considered in the context of the self-limiting nature of this illness, the potential impact on antimicrobial resistance, and the impact of previous medication usage. In particular, the low prevalence of ciprofloxacin resistance in Canada is advantageous and should be protected through the judicious use of fluoroquinolones. The use of macrolides may provide the best opportunity to affect duration of illness without a substantial risk of antimicrobial resistance development. Further investigation into the effect on duration of illness of antimicrobials not expected to be clinically effective against Campylobacter is necessary.
ACKNOWLEDGEMENTS
The authors acknowledge C. Clarke and J. de Grosbois for questionnaire administration, the participating hospital and private laboratories for providing isolates, S. Brown and K. Harris for speciation and antimicrobial susceptibility testing, and Z. Poljak for statistical advice. Funding was received from the Public Health Agency of Canada and the Ontario Ministry of Agriculture, Food and Rural Affairs.
DECLARATION OF INTEREST
None.
REFERENCES
- 1.Canadian Notifiable Disease Program (http://dsol-smed.phac-aspc.gc.ca/dsol-smed/ndis/index-eng.php). Accessed 11 January 2012.
- 2.Olson CK, et al. Epidemiology of Campylobacter jejuni infections in industrialized nations. In: Nachamkin I, Szymanski CM and Blaser MJ, eds. Campylobacter, 3rd edn. Washington DC: ASM Press, 2008, pp. 163–189. [Google Scholar]
- 3.Swartz MN. Human diseases caused by foodborne pathogens of animal origin. Clinical Infectious Diseases 2002; 34 (Suppl. 3): S111–S122. [DOI] [PubMed] [Google Scholar]
- 4.European Food Safety Authority, European Centre for Disease Prevention and Control. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2010. EFSA Journal 2012; 10: 2597. [Google Scholar]
- 5.Haagsma JA, et al. Disease burden of post-infectious irritable bowel syndrome in The Netherlands. Epidemiology and Infection 2010; 138: 1–7. [DOI] [PubMed] [Google Scholar]
- 6.Thabane M, Marshall JK. Post-infectious irritable bowel syndrome. World Journal of Gastroenterology 2009; 15: 3591–3596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Allos BM. Campylobacter jejuni infections: update on emerging issues and trends. Clinical Infectious Diseases 2001; 32: 1201–1206. [DOI] [PubMed] [Google Scholar]
- 8.Blaser MJ, Engberg J. Clinical aspects of Campylobacter jejuni and Campylobacter coli infections. In: Nachamkin I, Szymanski CM and Blaser MJ, eds. Campylobacter, 3rd edn. Washington DC: ASM Press, 2008, pp. 99–121. [Google Scholar]
- 9.Smith JL, Fratamico PM. Fluoroquinolone resistance in Campylobacter. Journal of Food Protection 2010; 73: 1141–1152. [DOI] [PubMed] [Google Scholar]
- 10.Scharff RL. Economic burden from health losses due to foodborne illness in the United States. Journal of Food Protection 2012; 75: 123–131. [DOI] [PubMed] [Google Scholar]
- 11.Gupta A, et al. Antimicrobial resistance among Campylobacter strains, United States, 1997–2001. Emerging Infectious Diseases 2004; 10: 1102–1109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Guerrant RL, et al. Practice guidelines for the management of infectious diarrhea. Clinical Infectious Diseases 2001; 32: 331–351. [DOI] [PubMed] [Google Scholar]
- 13.Aberg JA, et al. Infectious Diseases Handbook. Canada: Lexi-Comp, 2006. [Google Scholar]
- 14.Kapperud G, et al. Clinical features of sporadic Campylobacter infections in Norway. Scandinavian Journal of Infectious Diseases 1992; 24: 741–749. [DOI] [PubMed] [Google Scholar]
- 15.Ternhag A, et al. A meta-analysis on the effects of antibiotic treatment on duration of symptoms caused by infection with Campylobacter species. Clinical Infectious Diseases 2007; 44: 696–700. [DOI] [PubMed] [Google Scholar]
- 16.Nachamkin I, Szymanski CM, Blaser MJ (eds). Campylobacter. Washington DC, USA: ASM Press, 2008. [Google Scholar]
- 17.Deckert AE, et al. Antimicrobial resistance and antimicrobial use associated with laboratory-confirmed cases of Campylobacter in two health unit areas. Canadian Journal of Infectious Diseases and Medical Microbiology (in press). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gantz NM, et al. Manual of Clinical Problems in Infectious Disease. Philadelphia: Lippincott Williams and Wilkins, 2006. [Google Scholar]
- 19.Engberg J, et al. Quinolone and macrolide resistance in Campylobacter jejuni and C. coli: resistance mechanisms and trends in human isolates. Emerging Infectious Diseases 2001; 7: 24–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.King A, Phillips I. A comparison of the in-vitro activity of clarithromycin, a new macrolide antibiotic, with erythromycin and other oral agents. Journal of Hospital Infection 1991; 19 (Suppl. A): 3–9. [DOI] [PubMed] [Google Scholar]
- 21.Hosmer DW, Royston P. Using Aalen's linear hazards model to investigate time-varying effects in the proportional hazards regression model. Stata Journal 2002; 2: 331. [Google Scholar]
- 22.The Campylobacter Sentinel Surveillance Scheme Collaborators. Ciprofloxacin resistance in Campylobacter jejuni: case-case analysis as a tool for elucidating risks at home and abroad. Journal of Antimicrobial Chemotherapy 2002; 50: 561–568. [DOI] [PubMed] [Google Scholar]
- 23.Robins-Browne RM, et al. Treatment of acute nonspecific gastroenteritis of infants and young children with erythromycin. American Journal of Tropical Medicine and Hygiene 1983; 32: 886–890. [DOI] [PubMed] [Google Scholar]
- 24.Mandal BK, et al. Double-blind placebo-controlled trial of erythromycin in the treatment of clinical Campylobacter infection. Journal of Antimicrobial Chemotherapy 1984; 13: 619–623. [DOI] [PubMed] [Google Scholar]
- 25.Anders BJ, et al. Double-blind placebo controlled trial of erythromycin for treatment of Campylobacter enteritis. Lancet 1982; 1: 131–132. [DOI] [PubMed] [Google Scholar]
- 26.Robins-Browne RM, et al. Treatment of Campylobacter-associated enteritis with erythromycin. American Journal of Diseases of Children 1983; 137: 282–285. [DOI] [PubMed] [Google Scholar]
- 27.PubMed Health (http://www.ncbi.nlm.nih.gov/pubmedhealth/s/drugs_and_supplements/d/). Accessed 20 December 2011.
- 28.Effler P, et al. Sporadic Campylobacter jejuni infections in Hawaii: associations with prior antibiotic use and commercially prepared chicken. Journal of Infectious Diseases 2001; 183: 1152–1155. [DOI] [PubMed] [Google Scholar]
- 29.Vukelic D, Trkulja V, Salkovic-Petrisic M. Single oral dose of azithromycin versus 5 days of oral erythromycin or no antibiotic in treatment of Campylobacter enterocolitis in children: a prospective randomized assessor-blind study. Journal of Pediatric Gastroenterology and Nutrition 2010; 50: 404–410. [DOI] [PubMed] [Google Scholar]
- 30.Levesque S, Frost E, Michaud S. Comparison of antimicrobial resistance of Campylobacter jejuni isolated from humans, chickens, raw milk, and environmental water in Quebec. Journal of Food Protection 2007; 70: 729–735. [DOI] [PubMed] [Google Scholar]
- 31.Guevremont E, et al. Antimicrobial susceptibilities of thermophilic Campylobacter from humans, swine, and chicken broilers. Canadian Journal of Veterinary Research 2006; 70: 81–86. [PMC free article] [PubMed] [Google Scholar]
- 32.Gaudreau C, Gilbert H. Antimicrobial resistance of clinical strains of Campylobacter jejuni subsp. jejuni isolates from 1985 to 1997 in Quebec, Canada. Antimicrobial Agents and Chemotherapy 1998; 42: 2106–2108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Smith KE, et al. Quinolone-resistant Campylobacter jejuni infections in Minnesota, 1992–1998. New England Journal of Medicine 1999; 340: 1525–1532. [DOI] [PubMed] [Google Scholar]
- 34.Nelson JM, et al. Prolonged diarrhea due to ciprofloxacin-resistant Campylobacter infection. Journal of Infectious Diseases 2004; 190: 1150–1157. [DOI] [PubMed] [Google Scholar]
- 35.Engberg J, et al. Quinolone-resistant Campylobacter infections in Denmark: risk factors and clinical consequences. Emerging Infectious Diseases 2004; 10: 1056–1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Evans MR, et al. Short-term and medium-term clinical outcomes of quinolone-resistant Campylobacter infection. Clinical Infectious Diseases 2009; 48: 1500–1506. [DOI] [PubMed] [Google Scholar]
- 37.Dryden MS, Gabb RJ, Wright SK. Empirical treatment of severe acute community-acquired gastroenteritis with ciprofloxacin. Clinical Infectious Diseases 1996; 22: 1019–1025. [DOI] [PubMed] [Google Scholar]
- 38.Kirkpatrick BD. Infectious diarrhea. In: Grace C, ed. Medical Management of Infectious Disease. New York: Marcel Dekker Inc., 2003, pp. 437. [Google Scholar]
- 39.Kaplan MA, et al. Loperamide-simethicone vs loperamide alone, simethicone alone, and placebo in the treatment of acute diarrhea with gas-related abdominal discomfort. A randomized controlled trial. Archives of Family Medicine 1999; 8: 243–248. [DOI] [PubMed] [Google Scholar]
- 40.Li ST, Grossman DC, Cummings P. Loperamide therapy for acute diarrhea in children: systematic review and meta-analysis. PLoS Medicine 2007; 4: e98. [DOI] [PMC free article] [PubMed] [Google Scholar]