Abstract
The daily removal of billions of apoptotic cells in the human body via the process of efferocytosis is essential for homeostasis. To allow for this continuous efferocytosis, rapid phenotypic changes occur in the phagocytes enabling them to engulf and digest the apoptotic cargo. In addition, efferocytosis is actively anti-inflammatory and promotes resolution. Owing to its ubiquitous nature and the sheer volume of cell turnover, efferocytosis is a point of vulnerability. Aberrations in efferocytosis are associated with numerous inflammatory pathologies, including atherosclerosis, cancer and infections. The recent exciting discoveries defining the molecular machinery involved in efferocytosis have opened many avenues for therapeutic intervention, with several agents now in clinical trials.
Subject terms: Biologics, Apoptosis
Aberrations in efferocytosis are associated with numerous inflammatory pathologies, including atherosclerosis, cancer and infections. Here, Mehrotra and Ravichandran discuss the mechanisms of efferocytosis and the role of this physiological process in disease, and assess strategies and agents for therapeutic intervention.
Introduction
Cell death and cellular turnover are fundamentally critical to our healthy living. Roughly 200–300 billion cells are turned over in the human body daily, primarily via the death process of caspase-dependent apoptosis. This apoptotic process has been established to be critical for routine tissue homeostasis, embryonic development, wound healing and inflammation resolution in various tissues1,2. For example, neutrophils, the major innate immune cells that are the first line of defence against invading pathogens, have a short lifespan (~24 h), and >100 billion neutrophils are turned over daily. In the thymus or bone marrow, millions of immature T lymphocytes or B lymphocytes are generated, of which only a small fraction matures with the rest being eliminated by apoptosis. During organ development and embryogenesis, a rapid turnover of cells via apoptosis supports tissue restructuring. In addition, ‘used’ cells such as aged red blood cells are eliminated in large numbers on a daily basis as part of our bodily ‘house cleaning’3. Furthermore, during inflammation resolution, infiltrating neutrophils, monocytes and lymphocytes undergo apoptosis after their job is ‘done’. There are also other types of cell death such as necroptosis, pyroptosis and ferroptosis that arise from the death cue and tissue context1,4. But the questions are, what happens to all of these dying cells and what comprises the clearance crew?
Despite the large numbers of apoptotic cells being generated routinely, they are hardly observable in tissues in vivo. This is because cell death is remarkably well coupled to the highly effective phagocytic removal of apoptotic cells, a process termed ‘efferocytosis’5. In most tissues, efferocytosis is performed by either tissue-resident professional phagocytes with high phagocytic capacity — such as macrophages and dendritic cells — or non-professional phagocytes with lower phagocytic capacity that can be neighbouring cells — such as epithelial cells and fibroblasts. In certain tissues there are also ‘specialized’ phagocytes, such as retinal pigmented epithelial cells (eye) or Sertoli cells (testes), that perform phagocytic clearance in addition to their primary role as nurse/support cells. Efferocytosis has two benefits6,7: the quick removal of the apoptotic cells prevents them from becoming secondarily necrotic, where they might release their potentially toxic intracellular contents that may induce inflammation; and, equally important, engulfing phagocytes switch to a more ‘pro-resolution’ phenotype by increasing the secretion of anti-inflammatory mediators and upregulating pro-repair transcriptional programmes. This allows billions of cells dying in the body to be removed in a manner that avoids inflammation and is non-immunogenic. In contrast to efferocytosis, other types of phagocytosis such as Fc receptor-mediated phagocytosis are geared towards initiating an immune response.
Failed or defective clearance of apoptotic cells can be causative in numerous pathological conditions such as atherosclerosis, ageing-associated inflammation, cancer and infections3. The relevance of efferocytosis to diverse physiological and pathological outcomes opens the possibility to therapeutically target the various steps in the process and to harness the beneficial anti-inflammatory aspects of efferocytosis. In contrast, anti-inflammatory features of efferocytic tumour-associated macrophages may be detrimental to an antitumour response, and in this context, converting efferocytosis to a more inflammatory nature could potentially be beneficial.
Numerous strategies to therapeutically harness efferocytosis are emerging. For example, small molecules targeting receptors for phosphatidylserine (PS; an ‘eat-me’ signal exposed on the plasma membrane of apoptotic cells) on phagocytes are under clinical trials for numerous cancers. The application of autologous apoptotic cells for transplant tolerance is already under use. Further, the metabolites released from apoptotic cells or the molecular secretions from phagocytes engulfing apoptotic cells are actively anti-inflammatory with a beneficial role in inflammatory diseases. In addition, recent research suggests that anti-inflammatory and pro-resolution effects of efferocytosis are influenced by the solute carrier (SLC) family of transporters, which are highly druggable. In this Review, we focus on the distinct steps of efferocytosis, their linkage to human disease and the therapeutic interventions targeting this exciting physiological process, with implications for many human pathologies and ageing.
The mechanics of efferocytosis
Although there are many forms of cell death, apoptosis is the primary cell death modality under homeostatic conditions3,8. Efficient uptake of apoptotic cargo by phagocytes requires that the dynamic efferocytosis machinery operates optimally. The factors governing optimal functionality include the ratio of phagocytes to apoptotic cells, the nature of phagocytes (professional or non-professional), the size of the dying cell, the rate of death in a given spatial location, as well as the presence of stimulatory and signalling molecules facilitating the process3. All of these factors can impact efferocytosis, which is a multistep process. First is the ‘smell phase’, during which phagocytes ‘sense’ the presence of apoptotic cells and locate them. Second is the ‘eating phase’, when ligands on apoptotic cells are engaged by phagocytic receptors on phagocytes to specifically recognize and ingest the dying cell. Third is the ‘digestion phase’, when the ingested corpse and its contents are processed. This digestion phase is critical, resulting in a multifold increase in the amounts of proteins, nucleotides, carbohydrates and lipids that the phagocyte must manage while maintaining its own metabolic homeostasis. Thus, enhancing any of these steps could improve efferocytosis, and, in turn, provide an opportunity to dampen tissue inflammation. The subsections below will discuss the key molecular details and strategies to therapeutically target each of the phases of efferocytosis.
The smell phase
The very first step towards successful efferocytosis is the ‘smell phase’. Apoptotic cells release soluble mediators which include multiple ‘find-me’ signals, such as the nucleotides ATP and UTP9,10, membrane lipids such as sphingosine-1-phosphate and lysophosphatidylcholine (LysoPC)11,12, and chemokines such as fractalkine (CX3CL1)13. These soluble factors play dual roles: first, acting as attraction signals for the phagocytes to migrate towards the apoptotic cells; and, second, preparing the phagocytes for engulfment by modulating their cytoskeleton, enhancing the expression of engulfment receptors and the digestion machinery14,15. The same signals also attribute anti-apoptotic, regenerative and anti-inflammatory properties to the infiltrating and resident phagocytes14,15. The relative importance of each of these signals is dependent on the physiological context, cell type and stage of apoptosis. The variation in the find-me signals might be a mechanism to ensure apoptotic cell clearance in every tissue context16; for example, whereas some find-me signals may help to attract motile phagocytes such as macrophages to carry out clearance, other find-me signals may improve the efferocytosis machinery in the non-professional phagocyte. With the rapid technological advancements in mass-spectrometric imaging, it is now feasible to decipher the relative importance and abundance of each of these find-me signals in different tissue contexts. Also, determining the local concentration and strength of the chemical gradient in vivo will help understand how these may be ‘drugged’ in the future.
Nucleotides
The regulated release of small amounts of the intracellular nucleotides (<1%) from apoptotic cells can act as a potent inducer of monocyte/macrophage migration towards the apoptotic cells and also promote an anti-inflammatory tone in the tissue neighbourhood (see below). This nucleotide release is mediated by pannexin channels on the plasma membrane that become activated by caspase 3/7-mediated cleavage during apoptosis. The released ATP can induce migration of phagocytes via the cognate purinergic receptor P2Y, and clearance of apoptotic cells is impaired in P2Y–/– mice9. Further, UDP released by damaged microglia can induce the upregulation of P6Y in the hippocampus and aid in clearance of damaged cells. Nucleotides also ‘prepare’ the phagocytes for engulfment by inducing the expression of binding and engulfment receptors such as CD11b and α5β3 integrin17. In addition, nucleotides can exert immunomodulatory effects on macrophages and are, hence, called ‘calm-down’ signals18. For example, the ATP or AMP released by apoptotic cells can be converted to adenosine via the sequential actions of membrane nucleotidases such as CD39 and CD73; adenosine, in turn, can dampen inflammation through adenosine receptors19. This also leads to the expression of anti-inflammatory and pro-resolution molecules, such as Nr4a1, Nr4a2 and thrombospondin18,20. It is important to note that the regulated nucleotide release in small amounts (0.1%) from early-stage apoptotic cells (when the cells are still ‘whole’) is different from complete cell lysis and the release of the intracellular ATP at high concentrations, as the latter is well known to be pro-inflammatory. Recently, it has been shown that pannexin channel-mediated ATP and AMP release from viable T cells, and their subsequent conversion to adenosine, is critical for dampening airway inflammation21. It is important to note that the pannexin channels in viable T cells in this context are not activated by the irreversible caspase-mediated cleavage but, rather, by other ‘reversible’ mechanisms21.
Metabolites as preparatory signals
In addition to attracting the neighbouring phagocytes, apoptotic cells also actively release >100 metabolites during early stages of apoptosis15 These metabolites, in turn, induce transcriptional changes in phagocytes and neighbouring cells. The ‘metabolite secretome’ of apoptotic cells has revealed numerous signalling metabolites such as polyamines and nucleotides released in a regulated fashion from dying cells after induction of apoptosis. A good fraction of these metabolites (>20%) are released via the large-pore pannexin 1 channels (which can conduct molecules up to 1 kDa). Three aspects of metabolite release from apoptotic cells are particularly interesting from a therapeutic standpoint. First, apoptotic cells are not simply emptying their contents but, rather, release select metabolites among the thousands normally present in a cell; even more intriguing, whereas many pathways are shut down during apoptosis, the polyamine pathway continues to remain active, and the polyamine spermidine (but not all metabolites in the polyamine pathway) is selectively released15. Second, groups of metabolites are much more active than individual metabolites in inducing responses in neighbouring cells, suggesting cooperativity among metabolites in tissue communication15. Last, metabolites released from apoptotic cells are not unique to a particular type of apoptotic trigger or cell type. A few ‘core’ metabolites released during apoptosis were shared among different cell types and different apoptosis triggers. The metabolites were identified to induce gene expression linked to inflammation resolution and tissue repair15. Further, administering a metabolite cocktail reduced symptoms in models of inflammatory arthritis, and minor histocompatibility mismatch in lung transplantation15.
Approaches to target the smell phase
The immunomodulation induced by secreted soluble messengers released by apoptotic cells has brought forth an interesting therapeutic opportunity. A small subset of these conserved, often actively synthesized metabolites could be harnessed as ‘drugs’ to induce pro-repair and anti-inflammatory gene transcription programmes. For many decades now, the approach of ‘extracorporeal photopheresis’ (ECP) has been widely used to dampen inflammation in systemic lupus erythematosus (SLE) and arthritis, and facilitate organ transplantation22,23. In ECP, the leukocytes from a patient are removed, treated with UVA light along with 8-methoxysorelin and, then, reintroduced into the patient, which has been highly beneficial in dampening overall inflammation in many organs. Further, the benefits of apoptotic cell infusion for transplantations have been widely accepted, and experimental transplant models ranging from murine bone marrow to the heart, pancreatic islets and aortic transplants have shown much improved graft acceptance when the recipient was exposed to donor apoptotic cells24,25. However, how the donor cells given during ECP function has been a mystery. Perhaps, the anti-inflammatory gene signature induced by metabolites released from apoptotic cells may be part of the answer. Thus, boosting the regulated release of AMP, other nucleotides or metabolites via pannexin channels is a potential drug targeting strategy. Recently, direct activation of pannexin 1 via deacetylation has been identified in vitro, suggesting drug-based channel activation is likely feasible26. In addition, the membranes of apoptotic cells contain PS, and their contact with neighbouring cells may also provide an anti-inflammatory tone to the tissue. ECP is a highly onerous procedure, has to be done repeatedly every few months and is expensive. The ability to modulate inflammatory diseases with soluble mediators released by apoptotic cells or apoptotic cell therapy could revolutionize the concept of personalized medicine for inflammatory diseases (Fig. 1).
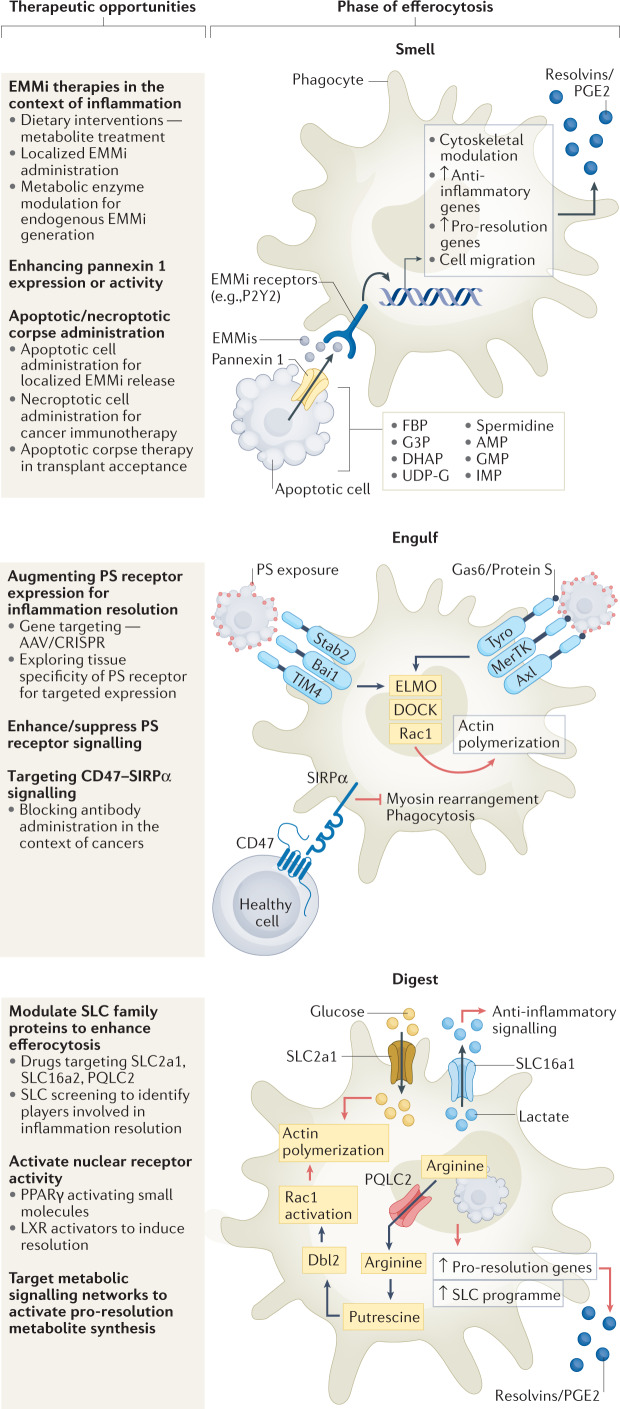
Fig. 1. Steps in efferocytosis and therapeutic opportunities.
Smell phase: clearance of apoptotic cells is a multistep process, initiated by secreted molecules released by the apoptotic corpse, inducing mobilization and migration of phagocytes towards dying cells. Pannexin 1 channel-mediated release of endogenous metabolic modulators (EMMis) from apoptotic cells induces pro-resolution and anti-inflammatory gene expression in phagocytes. Plausible methods for targeting this phase include administration of EMMis, altering activity of metabolic enzymes involved in generation of EMMi or enhancing pannexin 1 channel activity. Administration of donor apoptotic cells, for example via extracorporeal photopheresis (ECP), can reduce transplant rejection in patients and intravenous injections of apoptotic cells suppress anti-donor humoral immune response. In contrast, necroptotic cells elicit a strong immunogenic response and are used in targeting tumour cells. Engulfing phase: apoptotic cell phosphatidylserine (PS) recognition by phagocytes occurs either directly (for example, brain-specific angiogenesis inhibitor 1 (BAI1), T cell immunoglobulin mucin receptor TIM1) or by indirect PS recognition (for example, AXL, MerTK). Key downstream efferocytic signalling involves GTPase RAC1, which is activated via ELMO and DOCK proteins to enhance actin remodelling and formation of the phagocytic cup. Context-dependent modulation of PS receptor expression using gene editing technologies such as CRISPR and AAV-mediated gene delivery helps in disease models. Small-molecule inhibitors of PS receptor signalling are in advanced stages of clinical trials as cancer therapeutics (Table 1). CD47, the classical ‘do not eat-me’ signal, is often overexpressed in pathologies with defective apoptotic cell clearance and blocking CD47–SIRP1 interaction used in cancer treatments and atherosclerosis. Digestion phase: engulfment of multiple apoptotic cells leads to high metabolic burden on phagocytes. Complex signalling machinery enabling cargo digestion and successive cargo engulfment is being deciphered. Nuclear receptors such as PPARγ and liver-X receptor (LXR) enable cargo digestion. Metabolite transfer between intracellular compartments and from extracellular space during efferocytosis (via solute carriers (SLCs)) can improve anti-inflammatory gene expression and continuous efferocytosis66. Further, specialized pro-resolving mediator (SPM) synthesis and secretion by phagocytes is another step in modulating anti-inflammatory function of efferocytosis. AMP, adenosine-5′-monophosphate; Dbl2, guanine nucleotide exchange factor (gene name mcf2); DHAP, dihydroxyacetone phosphate; FBP, fructose-1,6-bisphosphate; G3P, glycerol-3-phosphate; GMP, guanosine-5′-monophosphate; IMP, inosine-5′-monophosphate; PQLC2 (also known as SLC66A1), PQ loop repeat-containing protein 2; UDP-G, UDP-glucose.
The eating phase
The ability of phagocytes to specifically recognize apoptotic cells is based on the exposure of ‘eat-me’ signals on the surface of apoptotic cells, in contrast to ‘do not eat-me’ signals that are exposed on the surface of live cells27. The ‘eat-me’ signals are recognized by a cohort of phagocytic receptors as detailed below. The most potent, evolutionarily conserved and well-studied ‘eat-me’ signal is PS exposed on the outer leaflet of the plasma membrane of apoptotic cells28. Under normal conditions, PS is actively kept on the inner leaflet of the plasma membrane in a regulated and energy-dependent fashion by specific enzymes called flippases and scramblases that help maintain this PS asymmetry29–34. Upon induction of apoptosis, caspase 3 causes the cleavage and inactivation of flippases while simultaneously activating scramblases33. Other ‘eat-me’ signals exposed on apoptotic cells include calreticulin, annexin 1, oxidized low-density lipoprotein, thrombospondin 1 binding sites and complement proteins C1q or C3b binding sites6,7. Although PS exposure is clearly central, the collection of other ‘eat-me’ signals dictates the optimal recognition and eating by phagocytes. It is important to note that not all apoptotic cells expose the same set of ‘eat-me’ signals, and a subset of them together with PS seem to be sufficient for recognition by specific receptors on phagocytes.
Phagocytic receptors
PS exposed on the surface of apoptotic cells is recognized via receptors that can bind PS either directly or indirectly. Receptors such as the brain-specific angiogenesis inhibitor 1 (BAI1), T cell immunoglobulin mucin receptors TIM1–TIM4 (TIM family) and Stabilin 2 bind PS directly, whereas receptors of the Tyro3/Axl/Mer (TAM) family of tyrosine kinases engage PS via soluble bridging molecules such as Gas6 and Protein S7,35,36. Dendritic cells, macrophages and endothelial cells are also capable of recognizing and binding to apoptotic cells via the scavenger receptor class F member 1 (SCARF1). SCARF1 binds to the complement component C1q that is bound to the PS on apoptotic cells. This complement-mediated uptake plays an important role in the aversion of autoimmunity (as will be discussed later in the Review). Among these PS recognition receptors, there are considerable differences in how they trigger intracellular signalling. BAI1, upon binding to PS via its thrombospondin repeats (TSR), initiates intracellular signalling via the ELMO1–DOCK complex to induce Rac1-mediated actin cytoskeletal rearrangements to facilitate efferocytosis35. BAI1 and downstream signalling via the ELMO pathway have also been linked to suppression of inflammation in models of gut inflammation37. TIM4, on the other hand, can interact with PS but is incapable of independently transducing cytoplasmic signalling, and signals via other integrins, BAI1 or other receptors36,38–40. Numerous reports in the past years have shed light on the fact that the different cell types express different combinations of phagocytic receptors, and this is influenced by the stage of efferocytosis, tissue context and developmental and immune states. Thus, the existence of a wide variety of engulfment receptors appears prudent to allow tissue-specific engulfment of apoptotic cargo. The binding of apoptotic cells to receptors on phagocytes triggers a complex array of cytoskeletal rearrangements, facilitating cargo engulfment. The signalling mechanism, the proteins involved and the kinetics of the process are highly variable and depend on the nature of the phagocytes1,2,41. The fusion of the apoptotic cell-containing phagosome with lysosomes, where many pH-sensitive degradative enzymes reside, aids in the breakdown of the biomolecular content of the apoptotic cargo. How the phagosomes traffic inside phagocytes has been reviewed extensively elsewhere1,2.
Interestingly, the TAM family of receptors are implicated in multiple physiological processes, distinct from, but closely related to, their primary role in efferocytosis. MerTK and AXL have direct roles in suppressing inflammatory responses and building tolerance towards ‘self’ antigens42. During efferocytosis, MerTK blocks NF-κB signalling in macrophages to suppress inflammatory cytokine production and macrophage M1-like polarization43. Similar observations were made in monocyte-derived dendritic cells44. In dendritic cells, a strong feedback loop is activated upon TLR stimulation that causes the expression of AXL45. Also, AXL was found to interact with the type I interferon receptor to trigger the expression of suppressor of cytokine signalling 1 (SOCS1) and SOCS3, which directly acts in inhibiting inflammatory cytokine production and inflammation. Further, MerTK expression and activation are associated with suppression of dendritic cell maturation and effector T cell expansion, promoting T cell tolerance46. Besides suppressing an M1-like phenotype, MerTK also helps polarize macrophages to the alternative or reparative M2-like phenotype47,48.
Approaches to target the eating phase
Owing to the diversity of PS receptors, their ubiquitous nature and their involvement in multiple pathologies (Fig. 2), PS receptors may serve as attractive therapeutic targets for multifactorial benefits. There are multiple methods by which the activity/effectiveness of PS receptors can be altered, and many therapeutics are already in clinical trials (Table 1). First, simply increasing the numbers of individual phagocytic receptors can enhance clearance. For example, whereas the levels of Bai1 mRNA progressively decreased in the gut epithelial cells and colonic tissue after DSS-induced colitis in wild-type mice, transgenic overexpression of Bai1 could attenuate the pathology of DSS-induced colitis in vivo. Consistent with this beneficial effect, BAI1 transgenic mice showed fewer apoptotic cells in the colonic epithelium and reduced colonic inflammation compared with wild-type mice37. Along similar lines, Stab2 expression was found to be critical for myoblast fusion during the process of muscle differentiation and regeneration, and overexpression of Stab2 was found to enhance myoblast fusion and myotube formation, both in vitro and in vivo49. Reduction in the expression of ‘anti-inflammatory’ phagocytic receptors may also be useful in cancer therapies. Targeting the downregulation or blocking the activity of MerTK has shown benefit in non-small lung cell carcinoma, gastric cancer cells, head and neck cancers, and glioblastoma50,51. Thus, modifying the expression levels/activity of PS receptors to optimal levels, in the context of individual pathologies, could be therapeutically beneficial. Second, the fact that there are so many PS recognition receptors and that they are not all equally expressed in all phagocytes strongly suggests specificity, both in signalling and in outcome, after corpse ingestion52. Therefore, defining context-specific PS receptors would need to be a priority for drug targeting of the eating phase. A third method could be to engineer a ‘universal’ booster of efferocytosis that can increase the expression of multiple PS recognition receptors in an inflammatory tissue context. An alternative possibility might be to engineer efferocytic receptors to increase their efficiency, that can then be expressed in a tissue context during ongoing inflammation to improve apoptotic cell clearance and, in turn, dampen the source of inflammation (Fig. 1).
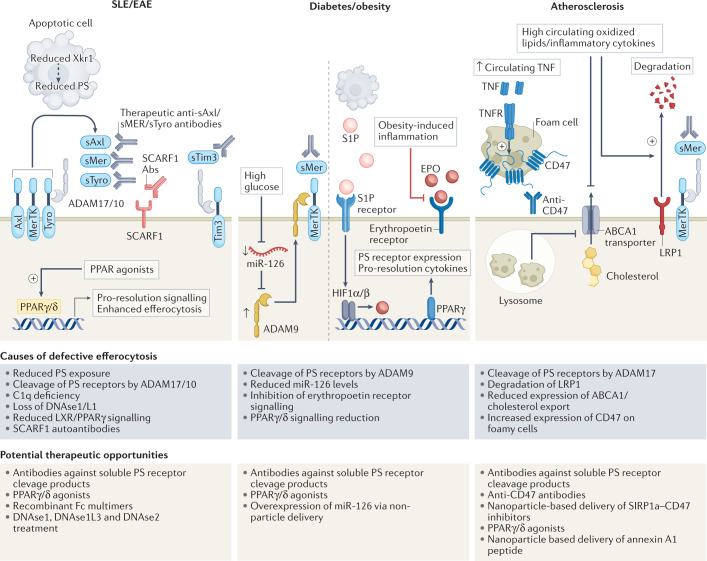
Fig. 2. Opportunities to target efferocytosis steps in specific pathologies.
Systemic lupus erythmatosus (SLE): defects in multiple steps of efferocytosis have been recognized in SLE. High levels of autoantibodies against scavenger receptor class F member 1 (SCARF1) (a phosphatidylserine (PS) receptor acting in conjunction with C1q) are found in patients with SLE, and correlated with accumulation of apoptotic cells. Enhanced cleavage of PS receptors MerTK, Axl, Tyro and Tim by ADAM17/10 proteases, and reduced activity and bioavailability of DNASE1L3, further suppress efferocytosis, exacerbating inflammation. Reduction in scramblase Xkr1, responsible for PS exposure, is also seen in mouse models of SLE. Plausible therapeutic opportunities in SLE could include: targeting PS receptors to improve efferocytosis; targeting PPAR/liver-X receptor (LXR) to enhance efferocytosis and LC3-associated phagocytosis (US Patent US20190145961); and extending administration of nanosphere-coupled DNAse1 to SLE. Diabetes and obesity: in diabetes, defective efferocytosis has been recognized. Further, downregulation of miR-126 that suppresses ADAM9 leads to MerTK cleavage and reduced efferocytosis. Overexpression of miR-126 was shown to restore efferocytosis. Erythropoietin (EPO) and PPARγ-mediated expression of phagocytosis receptors/associated molecules (Mfge8, MerTK, CD-36, Gas6) is defective in obese mice and a potential point of intervention in diabetes. Further, administration of endogenous metabolic modulators (EMMis) may also be effective strategies for enhancing efferocytosis in diabetes and obesity. Atherosclerosis: defective efferocytosis contributes to accumulation of lipid-laden apoptotic cells, which eventually add to necrotic lesions. Causes for poor efferocytosis include ADAM17/10-dependent cleavage of MerTK and decrease in ABCA1 upregulation. Reduced cholesterol export from phagocytes further diminishes engulfment capacity of cells. Enhanced expression of CD47 on apoptotic cells further suppresses apoptotic cell engulfment in atherosclerotic plaques. Numerous clinical trials are underway, targeting CD47–SIRP1α interaction in cancers, and repurposing these for atherosclerosis can be of value. Turning specialized pro-resolving mediator (SPM) ratios in favour of resolution and efferocytosis may help delay atherogenesis. Induction of foam cell apoptosis and localized administration of EMMis in plaques may be a supplementary therapeutic strategy. EAE, experimental autoimmune encephalomyelitis; LRP1, low-density lipoprotein receptor-related protein 1; TIM, T cell immunoglobulin mucin receptor.
Table 1.
Ongoing or completed clinical trials for therapeutics targeting efferocytosis
| Target | Drug/company (trial details) | Pathologies | Trial phase | Clinical trial ID/refs |
|---|---|---|---|---|
| Axl | Bemcentinib; also known as BGB324 and R428/trial by BerGenBio ASA | COVID-19 | II | NCT04890509 (refs206,207) |
| Bemcentinib/Rigel Pharmaceutical/BerGenBio (alone or in combination with other chemotherapeutics) | Recurrent glioblastoma, advanced solid tumours, metastatic breast cancer, AML, non-small cell lung cancer, malignant mesothelioma |
II II II I+II I |
NCT03654833 (refs130,208–211) |
|
| Bemcentinib/trial by BerGenBio | Non-small cell lung cancer | I | NCT02488408 | |
| Amuvatinib/trial by Astex Pharmaceuticals (in combination with platinum–etoposide) | Small cell lung cancer | II | NCT01357395 (refs212–214) | |
| Cabozantini/trial by Memorial Sloan Kettering Cancer Centre and Exelixis | Non-small cell lung cancer | II | NCT01639508 | |
| TP-0903/trial by Sumitomo Dainippon Pharma Oncology | Advanced solid tumours | I | NCT02729298 (refs215–218) | |
| MerTK | ONO-7475/Ono Pharmaceuticals (alone or in combination with venetoclax) | Cancers219: acute leukaemia and myelodysplastic syndromes | I+II | NCT03176277 |
| MRX-2843/trials by Meryx, Inc. (NCT04872478 and NCT03510104), Emory University (NCT04762199) and Betta Pharmaceuticals Co., Ltd (NCT04946890) (alone or in combination with osimertinib) | Solid tumours, non-small cell lung cancer, refractory AML |
I+II I I I |
||
| PF-07265807/trial by Pfizer | Metastatic solid tumours | I | NCT04458259 (ref.134) | |
| Gene therapy (AAV-mediated delivery) | Retinitis pigmentosa | I | NCT01482195 (refs220–222) | |
| CD47 | Magrolimab/trials by Gilead Sciences (alone or in combination with pembrolizumab/docetaxel/azacitidine/neopaclitazel/venetoclax/mitoxantrone/etoposide/cytarabine/CC-486/cetuximab) |
AML Refractory classic Hodgkin lymphoma/solid tumour/myelodysplastic syndrome/metastatic triple negative breast cancer/refractory multiple myeloma/myeloid leukaemia, head and neck squamous cell carcinoma/myeloid malignancies/T cell lymphoma/AML/solid tumours and advanced colorectal cancer, recurrent brain tumour, neuroblastoma |
III III III II II II II II II I+II I+II I+II I+II I I |
NCT02953509 (ref.223) |
| ALX148/most trials by ALX Oncology (alone or in combination with venetoclax/azacitidine/rituximab/lenalidomide/zantidamab) | B cell non-Hodgkin lymphoma, myelodysplastic syndromes, AML, advanced head and neck squamous carcinoma, advanced solid tumours, Her2+ cancers, microsatellite stable metastatic colorectal cancer |
II+III II II II I+II I+II I+II I+II I |
||
| AO-176/trial by Arch Oncology and Merck Sharp & Dohme Corp. | Malignancies/multiple myeloma |
I+II I+II |
||
| AK117/Akesobio Pharmaceuticals | AML/myelodysplastic syndrome, malignant neoplasm |
I+II I+II I I |
||
| AO-176/trial by Arch Oncology (alone or in combination with paclitaxel/pembrolizumab/magrolimab or with bortezomib/dexamethasone) | Advanced solid tumours, relapsed/refractory multiple myeloma |
I+II I+II |
NCT04445701 (ref.146) |
|
| HX009/trial by Waterstone Han X Bio Pty Ltd | Advanced solid tumours | I | NCT04097769 | |
| Tim4/3 | Sabatolimab; also known as MBG453/trials by Novartis (alone or in combination with PDR001/azacitidine/decitabine/venetoclax) | Cancers: advanced malignancies/lower risk myelodysplastic syndrome/AML/myelodysplastic syndromes |
III II II II II II I+II I+II |
|
| TSR-022/trials by Tersaro, Inc. (alone or in combination with TSR-042) | Advanced solid tumours/liver cancer/melanoma |
II I I |
NCT0281763 |
|
| LY3321367/Eli Lilly and Company (alone or in combination with LY3300054) | Advanced/refractory solid tumours | I | NCT03099109 (ref.138) |
AML, acute myeloid leukaemia; TIM, T cell immunoglobulin mucin receptor.
Anti-phagocytic receptors and ‘do not eat-me’ signals
The exposure of ‘do not eat-me’ signals on live cells helps avoid their efferocytosis by phagocytes53. Numerous such ‘do not eat-me’ molecules have now been discovered, including CD47 and CD24. These ‘do not eat-me’ signals, in turn, engage ‘anti-phagocytic receptors’ on phagocytes, and thereby help evade efferocytosis. For example, CD47 on live cells binds to SIRPα on macrophages, and this interaction causes tyrosine phosphorylation of the cytoplasmic domain of SIRPα, leading to the recruitment and activation of the phosphatases SHP1/2. SHP1/2 then inhibit phagocytosis by inhibiting non-muscle myosin IIA54. Thus, targeting the ‘do not eat-me’ signals to promote active engulfment of ‘live’ tumour cells is currently being pursued as part of anticancer immunotherapies (discussed in greater detail in the following sections). Another ‘do not eat-me’ signal, CD24 was recently reported to be highly expressed on tumour cells and the interaction of CD24 with sialic acid binding immunoglobulin-like lection 10 (Siglec10) on tumour-associated macrophages prevented the efferocytosis of cancer cells55. Blocking the CD24–Siglec10 interaction via monoclonal antibodies allowed for improved clearance of tumour cells. CD31, expressed on viable cells, via homotypic interactions, is thought to mediate the detachment of live cells from the surface of phagocytes56. Although targeting anti-phagocytic receptors to promote efferocytosis of apoptotic cells is less studied, this could also be a powerful approach in the context of ongoing inflammation27.
The digestion phase
Very often, the number of apoptotic cells far exceeds that of phagocytes, causing each phagocyte to successively ingest multiple apoptotic corpses, some of which can be very large in size53. The processing and degradation of the apoptotic cargo is carried out either by the canonical degradation programme or in other ways that may work in parallel. The canonical phagocytosis pathway involves the maturation of the early phagosome to late phagosome with eventual fusion with the lysosomal compartment. The additional pathway entails microtubule-associated protein 1A/1B light chain 3 (LC3)-associated phagocytosis and has been covered in other reviews1,2,41. Phagocytosis of apoptotic corpses brings with it a multilayered complexity: upon ingesting multiple corpses, the phagocyte needs to manage its volume and surface area; the ingested cargo is a metabolic burden, bearing membranes, cholesterol, proteins, nucleic acids and so on, which need to be metabolized and either rerouted into the metabolic flux cycles of the phagocyte or excreted; and the ingested cargo might bring with it some harmful content, and managing digestion and degradation of the apoptotic cargo while shifting gears towards an anti-inflammatory, pro-repair phenotype after efferocytosis is a unique challenge for the phagocytes.
Lipids and associated receptors
Degradation of apoptotic cargo causes phagocyte cholesterol levels to increase, a phenomenon exacerbated in atherosclerosis when apoptotic ‘foamy’ macrophages (foam cells) undergo efferocytosis and have to be taken up by other macrophages. A complex cascade of signalling events aid phagocytes in handling the excessive lipid overload. One of the major mechanisms involved is in the efflux of cholesterol to apolipoprotein A-1 (ApoA-1) via upregulation of the cholesterol transporter protein ABCA1, which is activated by the engagement of PS receptors such as BAI1 by apoptotic cargo57. Further, sterols derived from the breakdown of the apoptotic cargo cause the activation of liver-X receptors (LXRs) α/β58, which induce the activation of ABCA1. Low density lipoprotein receptor 1 (LRP1), a cell surface receptor, was also demonstrated to be essential for efferocytosis. The receptor causes PPARγ activation, which induces the expression of LXRα/β, leading to ABCA1 expression and cholesterol efflux59. Under conditions where apoptotic cargo is cholesterol heavy, macrophages may subvert the toxic effects of the high cholesterol cargo via activation of anti-apoptotic and pro-survival pathways (for example, Akt-NF-κB signalling in vitro)60.
In the context of efferocytosis, nuclear receptors LXR, PPARγ and Nr4a2 have multiple roles and are seen as the nodal points between inflammation and metabolic signalling. Upon binding to ligands, nuclear receptors repress the transcription of inflammatory genes such as TNF, IL-1β, IL-6, COX2 and iNOS, and chemoattractants such as MMP9, MCP3 and MCP1, while increasing the expression of the efferocytic receptor MerTK. A major mechanism of nuclear receptor action is called ‘trans-repression’, which entails the inhibition of NF-κB and other inflammatory pathway target gene transcription61. Along with the repression of inflammatory gene transcription, LXRα/β receptor signalling has also been demonstrated to induce the expression of MerTK, along with switching the macrophage to a tolerogenic programme by increasing the expression of IL-10 and TGFβ. The secretion of these cytokines also suppresses the recruitment of additional inflammatory cells. ABCA1–ApoA-1 interaction during cholesterol efflux was demonstrated to be anti-inflammatory, suppressing the release of inflammatory cytokines by engaging STAT3 (ref.62). Further, it is interesting to note that oxysterols derived from apoptotic cargo are active inhibitors of inflammasome NLRP3 activation59,63.
Sugars, nucleotides and transporters
In the quest to uncover the utilization of apoptotic cell-derived metabolic load, multiple interesting facts about metabolic flux occurring in phagocytes have emerged. Continuous efferocytosis is an essential mechanism in resolution and repair. Macrophages have been shown to utilize the arginine and ornithine derived from the first round of engulfed apoptotic cells to facilitate subsequent rounds of efferocytosis64. Arginine and ornithine were shown to be converted to putrescine via the action of macrophage-expressed ornithine decarboxylate and arginase1. Interestingly, putrescine could increase the activity of the small GTPase Rac1, which then facilitates actin rearrangement and subsequent rounds of efferocytosis. The additional observations from this study gain importance in tissues with high rates of cell turnover, where multiple cargoes need to be engulfed rather rapidly64 (Figs 1 and 3). An interesting recent finding also highlights how efferocytosis of apoptotic cargo can enable proliferation of the pro-resolution macrophages in vitro and in vivo. The nucleotides released by the hydrolysis of apoptotic cell-derived DNA could enhance Myc activity via the mTPRC2/Rictor pathway, leading to the proliferative phenotype and inflammation resolution65.
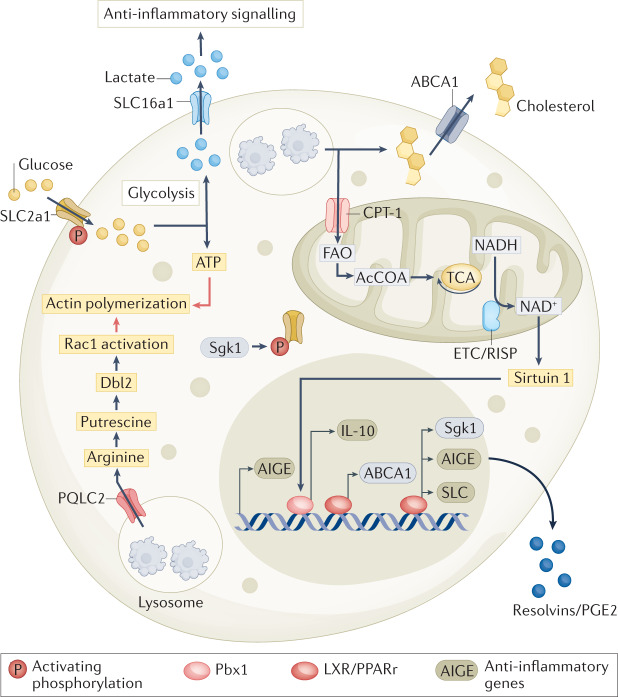
Fig. 3. The metabolic dynamics of efferocytosis.
The immense metabolic burden that apoptotic cells carry into a phagocyte requires phagocytes to rapidly change their immuno-metabolic landscape to enable immunologically silent clearance of apoptotic cells. Metabolites secreted by apoptotic cells before engulfment, apart from being potent chemokines, also drive transcriptional changes in phagocytes, including expression of anti-inflammatory and pro-resolution genes. Lysosomal degradation of apoptotic cargo releases free fatty acids, cholesterol, amino acids, sugars and nucleic acids into metabolic flux cycles of the phagocyte. There are multiple modes by which apoptotic cells alter the immunometabolism of the phagocyte. First, apoptotic cargo as signal transducers — apoptotic cell binding to phosphatidylserine (PS) receptors leads to activation of liver-X receptor (LXR) signalling and upregulation of ABCA1 transporters, leading to plasma membrane export of cholesterol. Second, degradation of apoptotic cells for continuous efferocytosis — mitochondrial oxidation of apoptotic cell-derived fatty acids alter NAD+/NADH balance, activating sirtuin 1 signalling, which then triggers expression of pro-resolving cytokine IL-10; apoptotic cargo digestion enhances mitochondrial fission, enabling successive cargo engulfment allowing phagolysosomal sealing and membrane recycling. Third, direct utilization of apoptotic cell metabolites — arginine derived from lysosomal degradation of apoptotic cargo is transported via PQLC2 lysosomal transporter into phagocyte cytosol; ornithine decarboxylase converts arginine into putrescine, which aids in Rac1 activation, driving successive rounds of apoptotic cell uptake. Fourth, reprogramming phagocyte solute balance via solute carrier (SLC) family reprogramming — binding of apoptotic cells to receptors that recognize PS leads to upregulation of glucose transporter 1 (GLUT1; also known as SLC2A1) and serum/glucocorticoid regulated kinase 1 (SGK1), which is required for GLUT1 transport to the plasma membrane. The increased glycolysis supported by enhanced glucose import is necessary for ATP generation and actin polymerization. The lactate, generated as a result of increased aerobic glycolysis, acts as a paracrine anti-inflammatory mediator after secretion through the upregulated lactate transporter SLC16a1. Along with adjusting metabolite compartmentalization and transport, efferocytosis also equilibrates ionic balance in engulfing cells. Plasma membrane chloride import via SLC12A2 channels was upregulated and activated after efferocytosis. Inhibiting transporter activity caused an increase in efferocytosis but the response to this corpse uptake was pro-inflammatory rather than anti-inflammatory.
The subcellular compartmentalization of metabolites has emerged as an important aspect governing cellular metabolic fluxes. Metabolite transport is mediated via a specialized family of transmembrane proteins called the SLC family. SLC family members (numbering >400) are localized in the plasma membrane as well as in intracellular membranes. The different phases of efferocytosis rewire the SLC expression network in both macrophages and fibroblasts66. The mere presence of the ‘secretome’ of apoptotic cells was shown to induce the expression of the mRNA for the enzyme Sgk1, which phosphorylates SLC2a1 (a glucose transporter) and increases SLC2a1 abundance in the plasma membrane66. Further, the engagement of PS with cognate phagocytic receptors was shown to increase the expression of SLC2a1 itself66. The enhancement of glucose uptake and glycolysis, owing to the increased activity and expression of SLC2a1, was found to be essential for initial and continuous efferocytosis both in vitro and in vivo. Later stages of efferocytosis, possibly tending towards ‘digestion’ of the apoptotic cargo, were found to increase gene expression of the lactate transporter SLC16a1. The lactate released via SLC16a1 was biologically active — extracellular lactate derived via the upregulation of SLC16a1 was capable of triggering the expression of pro-resolution genes such as il10 (ref.66). Recent studies also suggest that mitochondrial dynamics within the phagocytes may play a significant role in regulating their homeostasis, and successive corpse uptake67,68. The fusion of the phagosome containing the apoptotic cargo with the lysosomes leads to the release of a large number of lipid metabolites within the macrophage. The lipid substrate abundance and oxidative phosphorylation were linked to an increase in the NAD+/NADH ratio, in turn triggering a member of the sirtuin family of proteins, SIRT1, ultimately inducing the release of IL-10 via Pbx1 (Fig. 3). Further, efferocytosis by mouse bone marrow derived macrophages (BMDMs) led to increased cytosolic calcium and mitochondrial fission mediated by Drp1, and mitochondrial morphology could influence high-burden efferocytosis67.
Approaches to target the digestion phase
The intricate protein interaction networks that operationally link immunological behaviour to metabolic fluxes have recently become high-priority drug targets4. Efferocytosis provides a unique phenomenon in this regard, wherein the ingestion of an apoptotic cargo triggers metabolic reprogramming that aids the phagocyte to suppress inflammatory signalling (Figs 1 and 3). Aberrations in this signalling network contribute to multiple pathologies, as detailed below. Pioglitazone, a drug approved for type 2 diabetes, is a PPARγ agonist. In recent years, PPARγ has been demonstrated to boost efferocytosis (via increasing PS receptor expression) and induce anti-inflammatory gene expression signatures when tested on monocytes derived from patients with human chronic granulomatous disease69. Repurposing of PPARγ/LXRα agonists to boost efferocytosis in the context of inflammatory diseases and infections is a promising therapeutic opportunity. The regulation of the SLC family of proteins by efferocytosis implies that metabolite localization, subcellular compartmentalization and temporal fluctuations during the process of cargo engulfment have a major immunomodulatory role. Boosting efferocytosis by altering the activity or expression of selected solute carrier proteins such as SLC2a1, while simultaneously modulating the activity of ABCA1 and SLC16a1, could prove powerful in pathologies where poor efferocytosis is observed and is pro-inflammatory (for example, atherosclerosis). Interestingly, β-hydroxybutyrate increases SLC2a1 expression levels (epigenetically)70. β-Hydroxybutyrate, as a dietary supplement, is already in clinical trials for type1 diabetes (NCT03999853). In conditions where successive efferocytosis by phagocytes is hampered (for example, during inflammation), the ability to enhance the activity of unique SLC family members, or the lysosomal arginine transporter Pqlc2 (ref.64), which can then drive continued efferocytosis in vitro and in vivo, poses a novel window of therapeutic opportunity. The research on how phagocytes utilize apoptotic cargo to drive their immunological programmes is bound to provide more leads into novel therapeutic avenues. A large ongoing body of research on SLCs, which represent the second largest family of proteins in the human genome (after GPCRs), has identified numerous small-molecule inhibitors/activators, and targeting specific SLCs relevant during efferocytosis could be an approach to improve outcomes in inflammatory diseases71 (Fig. 3).
Efferocytosis in pathologies and therapeutic opportunities
A large number of human disorders, including atherosclerosis, cancer, SLE, diabetes, obesity, rheumatoid arthritis and ageing, are now linked to defects or alterations in the efferocytosis machinery. Efferocytic machinery is highly sophisticated, involving a network of receptors, cytosolic signalling molecules, rapid cytoskeletal rearrangements, cargo digestion and subsequent immunosuppressive signalling. Owing to these distinguishable and well-defined steps during efferocytosis and the accessible molecular pathways, the steps in efferocytosis are emerging as highly attractive for therapeutic intervention in a broad range of diseases, some of which will be discussed below.
Atherosclerosis
Atherosclerosis is a condition that develops due to the accumulation of oxidized lipids and lipoproteins in the sub-endothelial layer of arteries in the heart. The heart vasculature contains phagocytes such as dendritic cells, macrophages and monocytes, and the further accumulation of lipids and lipoproteins during atherogenesis also causes infiltration of leukocytes into the vasculature. Initially, the resident and infiltrating macrophages ingest these sub-endothelial lipid deposits, but when these macrophages are overwhelmed with intracellular lipid and cholesterol (‘foamy macrophages’), this triggers their apoptosis. Defective efferocytosis and accumulation of apoptotic foam cells have been demonstrated in the necrotic core of atherosclerotic plaques and large necrotic cores are correlated with higher risk of myocardial infarction. Multiple causes for reduced efferocytosis in lesions have emerged including defective ABCA1 expression72–74, as well as increased degradation of efferocytic receptors such as MerTK and LRP1. MerTK is liable to cleavage by a member of the ADAM family proteases, ADAM17, and a cleavage-resistant mutant of MerTK accelerated resolution in the model of zymosan-stimulated peritonitis75. MerTK cleavage might be a phenomenon in atherosclerosis, as macrophages in the necrotic cores of atherosclerotic plaques exhibit decreased expression of MerTK on the cell surface and have heightened expression of ADAM17 (refs76,77).
A well-understood phenomenon associated with macrophage-mediated efferocytosis is the generation of a class of lipids called ‘specialized pro-resolvin mediators’ (SPMs), which include Resolvins D1, D2 and E4, lipoxin A4 (LXA4) and Maresin. The precursors of SPMs are often carried by apoptotic cargo and efferocytosis leads to the activation of a critical enzyme, 12/15-lipoxygenase, needed for SPM biosynthesis from these precursors78,79. Molecules such as LXA4 and Resolvin D1 have been demonstrated to boost efferocytosis in macrophages and promote an overall anti-inflammatory, pro-resolution phenotype80,81. However, in advanced atherosclerotic plaques, the ratio of Resolvin D1 to the pro-inflammatory lipid mediator, leukotriene B4, was skewed towards inflammation. Treatment of atherosclerotic mice with Resolvin D1 suppressed reactive oxygen species (ROS) levels and necrosis in the lesions, while simultaneously inducing efferocytosis82. Further, the levels of 12(S)-hydroxyeicosatetraenoic acid (12(S)-HETE) are increased in patients with acute coronary syndrome83 The levels of 12(S)-HETE were inversely correlated with the efferocytosis capacity of macrophages as these compounds activate RhoA, which inhibits apoptotic cargo internalization83.
Lastly, the expression of CD47 as a ‘do not eat-me’ signal prevents efferocytosis within the plaque. The inflammatory milieu of atherosclerotic lesions is linked to the aberrant expression of CD47 on apoptotic cells, causing them to accumulate and, in turn, become secondarily necrotic, further exacerbating lesional inflammation1. Inflammation-induced TLR signalling can further suppress the expression of ligands such as MFGE8 (ref.84). Thus, owing to an increasingly inflammatory environment and greatly dysregulated metabolic machinery, efferocytosis in atherosclerotic plaques is suppressed, furthering disease pathology.
Opportunities for targeting the efferocytic machinery in atherosclerosis
The most common therapeutic intervention for atherosclerosis and coronary heart diseases is statin administration. Along with the direct metabolic consequences, statin treatment also aids in enhancing efferocytosis by inhibiting RhoA isoprenylation, as RhoA is a negative regulator of efferocytosis85. Inhibiting ROCK1, the downstream target of RhoA, also improves efferocytosis, and has shown promise in atherosclerosis, reducing the lesion size and inflammatory macrophage accumulation in the plaques86. ROCK activity has been implicated in numerous inflammatory diseases and ROCK1 inhibition (along with statin treatment) is used in multiple models of inflammatory diseases including inflammatory arthritis, acute lung injury, systemic sclerosis87, LPS-induced lung injury and Crohn’s disease24,88,89. Although most of the beneficial effects of ROCK inhibition (for example, fasudil treatment) are attributed to the anti-inflammatory phenotype that emerges owing to the suppression of NF-κB activation24, fasudil treatment also increased neutrophil apoptosis and efferocytosis in a model of intraperitoneal LPS challenge90. CD47 blocking antibodies have also shown promise by enhancing efferocytosis in atherosclerotic lesions in numerous mouse models91. The expression of CD47 and SIRP1α was increased in human atherosclerotic arteries and the loss of SIRP1α signalling protected mice from atherosclerosis92. In addition, enhanced CD47–SIRP1α signalling in atherosclerotic lesions has been observed to lead to a high burden of apoptotic and secondary necrotic cells. Delivery to atherosclerotic plaques of a small-molecule inhibitor of SHP1, a downstream effector of SIRPα receptor on macrophages that recognizes CD47, increased lesional phagocytosis, reduced the inflammatory gene expression and allowed plaque regression in apoE–/– mice93. An added complication in atherosclerosis was identified to be the accumulation and reduced efferocytosis of necroptotic cells along with an associated reduction in SPM secretion94. The administration of RevD1 or CD47 blocking antibodies aided in enhancing necroptotic cell efferocytosis94.
As ABCA1 and MerTK expression are influenced by LXR and PPAR, agonists of these nuclear receptors seem to have beneficial effects on efferocytosis and anti-inflammatory signalling during efferocytosis in vivo95. Similarly, LXR agonists also suppressed inflammation in vivo96. Although targeting these multifunctional nuclear receptors is complicated, a selected and localized delivery approach may hold promise in promoting efferocytosis to remove the lesional apoptotic burden. Further, efferocytosis-associated factors seem to improve plaque stability, and boosting efferocytosis may again provide additional benefits. Overall, great progress has been made in unravelling the mechanistic underpinnings of efferocytosis in atherosclerosis4. With the advent of nanomaterials for localized and targeted delivery of pro-efferocytic compounds and Resolvin-like lipid mediators, their combination with classical drugs such as statins may provide new therapeutic strategies (Fig. 2). In another approach, mesenchymal stem cells induced M2-like behaviour in macrophages in vitro97,98, and administering mesenchymal stem cells in mouse models of cardiac inflammation led to enhance M2 macrophage infiltration and efferocytosis, aiding clearance of apoptotic neutrophils and improving cardiac function99.
Ageing
Ageing has been associated with sterile, chronic low-grade inflammation, called ‘inflammaging’100. In recent years, numerous studies have noted the increasing presence of cellular debris with age contributing to this inflammaging. The presence of cellular debris and misplaced ‘self-antigens’ such as cellular and mitochondrial DNA increase in circulation in aged human subjects, and this has been shown to be positively correlated with inflammatory mediators such as TNF and IL-6 (ref.101). Aged mice show impaired capacity to clear either apoptotic cells injected into the peritoneum or apoptotic keratinocytes generated by UV irradiation of the skin102. Similarly, in a self-resolving model of peritonitis, aged mice have dramatically reduced SPMs, reduced capacity of inflammation resolution and heightened circulating inflammatory cytokines103. Further, ex vivo differentiated BMDMs from older mice have reduced efferocytic capacity that could be enhanced by SPMs such as Resolvin D3 (ref.103).
An age-related decline in inflammation resolution is well documented103,104. During acute inflammation in a dermal model of sterile inflammation, efferocytic clearance of the infiltrating polymorphonuclear neutrophils was reduced in older human subjects compared with younger subjects105. This phenotype was, in part, mediated by p38MAPK-mediated inhibition of p300, which caused a reduction in the expression of Tim4, and correlated with reduced expression of pro-resolution signalling and TGFβ release. Blocking p38MAPK with the inhibitor losmapimod enhanced Tim4 expression and efferocytosis in mice. These findings indicate that the resolution phase of inflammation is impacted with age and can be therapeutically targeted.
The low-grade chronic inflammation associated with ageing is accompanied by the presence of senescent cells. Senescence is defined as an irreversible and long-term arrest of the cell cycle, and is caused by shortened telomeres, DNA damage106, defects in mitochondrial function and alteration of the epigenomic status107,108. Accumulation of senescent cells is seen at the sites of age-related pathologies such as atherosclerosis, Alzheimer disease, osteoarthritis, chronic obstructive pulmonary disease and Parkinson disease109–111. Bioactive molecules (SASP) secreted by senescent cells such as IL-1, IL-6, IL-18, TNF, IL-8, GM-CSF and MCP1–MCP4 induce paracrine signalling in the neighbouring cells to activate senescence112. It is plausible that the presence of senescent cells and associated SASP could feed into the general ‘polarization’ of macrophages and other phagocytes towards a non-efferocytic phenotype with age. Conditioned media from senescent cells induced MerTK cleavage and decreased macrophage efferocytosis in vitro, whereas cleavage-resistant mutants of MerTK were non-responsive to the presence of the conditioned medium from senescent cells113. This decreased efferocytosis could be countered by the administration of Resolvin D1 (ref.113). Thus, a decrease in efferocytosis of apoptotic cargo, and accumulation of apoptosis-resistant senescent cells along with the influence of SASP on inflammation resolution and efferocytosis, could be major players in inflammaging.
Opportunities for targeting the efferocytic machinery in ageing
Current ‘senotherapies’ are directed towards induction of apoptosis in senescent cells and suppression of the damaging effects of SASP. Following the elegant work by Baker et al.109,114, efforts are underway to develop pharmacological agents that could potentially trigger cell death in senescent cells via senolytic agents. Owing to a decline in efferocytosis and an increase in the accumulation of apoptotic and senescent cells with age, combinatorial therapies increasing efferocytosis and inducing apoptosis in senescent cells could be a powerful intervention in increasing healthspan. In this context, compounds such as resolvins could be administered in combination with senolytic compounds. The co-treatment with potentially localized delivery of senolytic compounds could be useful for conditions such as osteoarthritis115. Localized delivery could be further enhanced by pro-resolving approaches, as demonstrated in the zymosan model of inflammation116. Spermidine is a bioactive polyamine, which has been proven beyond doubt to have beneficial effects in ageing and increasing lifespan in human subjects. Apoptotic cells synthesize and secrete spermidine, and administration of spermidine in combination with purine nucleotides (GMP and IMP) was demonstrated to improve the pathology in an arthritis model as well as in a lung transplant rejection model15. This reveals an exciting possibility of combining apoptotic metabolite-based or oral therapies with senolytic compounds and SPMs to affect suppression of inflammation to improve efferocytosis.
Cancer
Cancer is often referred to as a disease with inflammation as one of its hallmarks, and prolonged, unresolved inflammation as a contributing factor to oncogenesis117,118. On the contrary, the tumour microenvironment is increasingly recognized as immunosuppressive, allowing escape of tumour cells from immunosurveillance. Macrophages exist in large numbers in the tumour microenvironment (referred to as ‘tumour-associated macrophages’) and play significant immunomodulatory roles at progressive stages of cancer from initiation to progression, epithelial–mesenchymal transition, metastasis, angiogenesis, tumour cell motility and matrix degradation119,120. Macrophages are also linked to suppression of tumour immunosurveillance, leading to poor disease prognosis. Efferocytosis machinery promotes naïve macrophages to adopt an ‘M2’-like phenotype, which is proven beyond doubt to be a major contributor to poor cancer prognosis by suppressing antitumoural activity121, aiding in tissue repair-type responses and angiogenesis (via production of VEGF)119.
The reason for the existence of the M2 polarizing milieu in the tumour mass is an area of active research. Cell death is a common occurrence in solid tumours as they progress towards malignancy, with chemotherapies increasing the number of dying cells. It is plausible that anti-inflammatory mediators such as TGFβ and IL-10, released by tumour macrophages engulfing apoptotic cells within the tumour mass, could be the drivers of the observed alternative M2 polarization. Further, as noted in the previous sections, PS receptor activation actively interferes with NF-κB signalling and type I interferon production, in turn, limiting antitumour immune responses. Thus, a combination of pro-survival function of TAM receptors on tumour cells and their ability to suppress inflammation in the tumour microenvironment (via the macrophages) may promote immunosuppression and tumour progression. The TIM family of PS receptors are also implicated in poor prognosis of malignant tumours including colorectal cancer, non-small cell lung cancer122 and gastric cancer123. A potential deleterious effect of anti-inflammatory signalling-associated efferocytosis within the tumours is the link to myeloid-derived suppressor cells (MDSCs) within the tumour microenvironment. MDSCs can protect the tumour from assaults on the patient’s immune system via regulatory T cells, promoting angiogenesis (by secreting VEGF) and production of type I interferons124. Cytokines released during efferocytosis such as IL-10, PGE2 and VEGF promote MDSC development and function. MDSCs in tumour-bearing mice have dramatically upregulated TAM receptors and ligands, and knocking down the TAM receptors in MDSCs led to increased T cell proliferation and tumour immunosurveillance125. Transplantation of MerTK–/– bone marrow in a mouse model of metastatic breast cancer led to a higher number of cytotoxic CD8+ T cells and increasing antitumour immune responses126. The roles of TAM receptor activation in tumours have been reviewed extensively elsewhere127–129.
Opportunities for targeting the efferocytic machinery in cancer
Owing to their role in aiding tumour growth, angiogenesis and metastasis, the TIM and TAM families of PS receptors are hot topics of research for antitumour therapy, along with antibodies aimed at PS blockade. R428 (bemcentinib) is a selective Axl inhibitor with a half-maximal inhibitory concentration of 14 nM130, which could suppress breast cancer metastasis, angiogenesis and inflammatory cytokine production by inhibiting Axl and downstream Akt phosphorylation130. Axl inhibition by bemcentinib could also sensitize cells that have undergone epithelial–mesenchymal transition to a large number of anti-mitotic drugs — particularly docetaxel in vivo131. Bemcentinib is currently in phase II clinical trials (NCT03824080) for acute myeloid leukaemia (AML) and low/high-risk myelodysplastic syndrome (Table 1). Cabozantinib, a broad receptor tyrosine kinase inhibitor also suppressing Axl activity, is under phase II clinical trials (NCT01639508) (Table 1). Multiple other compounds targeting Axl are under clinical trials for different types of cancers. TP-0903, a novel oral Axl inhibitor, and amuvatinib are under phase I and phase II clinical trials respectively, for small cell lung cancer.
Apart from Axl, small molecules targeting MerTK and TIM4/3 are in advanced stages of clinical trials. ONO-7475 is a dual Axl–MerTK inhibitor tested in acute leukaemia and myelodysplastic syndromes, either alone or in combination with the chemotherapeutic venetoclax (NCT03176277; Table 1), which has been demonstrated to overcome venetoclax resistance in AML models132. MRX-2843 is a MerTK/FLT3 inhibitor that aids in treating FLT3 mutations causing AML133 and is currently undergoing phase I clinical trials (NCT03510104/ NCT04946890; Table 1). PF-07265807, a selective inhibitor of MerTK and Axl which has shown antitumour activity when combined with anti-PD1 therapy134, is also undergoing phase I clinical trial (NCT04458259; Table 1). The TIM3 inhibitor sabatolimab (MBG453) was recently granted fast-track designation by the US Food and Drug Administration (FDA) for myelodysplastic syndrome and is being tested in numerous clinical studies for myelodysplastic syndromes, either alone or in combination with other chemotherapeutics135–137 (Table 1). In addition, the novel anti-TIM3 antibody TSR-02 is currently undergoing phase I clinical trials against solid tumours (NCT02817633; Table 1) and is being tested in combination with anti-PD1 antibody for liver cancer (in phase II clinical trials, NCT03680508; Table 1). Finally, LY3321367, an anti-TIM3 monoclonal antibody, is undergoing phase I clinical trials alone or in combination with anti-PD1 antibody for the treatment of solid tumours (NCT03099109; Table 1). Thus far, no dose-limiting toxicity has been observed in either the monotherapy or combinatorial therapy138.
A surprising phenomenon in patients with myeloproliferative disease is the ability of tumour cells to upregulate the expression of the ‘do not eat-me’ signal molecule CD47, to limit engulfment by phagocytes. CD47 classically interacts with the macrophage receptor SIRPα, suppressing efferocytosis of live cells. Increased CD47 expression has been observed in patients with ovarian cancer, glioma, myeloid leukaemia, non-Hodgkin lymphoma and glioblastoma, which correlated with poor prognosis and tumour growth with successive stages of malignancy139,140. Anti-CD47 monoclonal antibody-mediated engulfment of tumour cells by macrophages facilitated the priming of CD8+ T cells to mount a cytotoxic response141. Although anti-CD47 therapies have shown promise in animal models, due to CD47 expression also on red blood cells, specific targeting of tumours without interfering with other ‘normal’ roles of CD47 is an ongoing challenge142–144. So far, the observations point towards a beneficial role for CD47 blockade in cancer. It is worth noting that the phagocytosis mediated under conditions of CD47 antibody blockade is not typical efferocytosis, and is more akin to FcR-mediated phagocytosis, and may induce a more pro-inflammatory response within the tumours145. A large number of clinical trials are underway, testing the efficacy of anti-CD47 therapy in combination with pre-existing chemotherapeutics in various cancer types. AO-176, a humanized IgG2 antibody that specifically binds and blocks CD47, is being tested in multiple myeloma (NCT04445701; Table 1). Ao-176 has been shown to promote efferocytosis along with inducing antitumour T cell responses, as well as T cell-mediated killing of CD47-expressing tumour cells146. HX009 is a humanized bispecific antibody targeting CD47 and PD1, which is undergoing clinical trials for patients with advanced tumours and malignancies (NCT04886271, NCT03834948)147. In addition, AK117, a humanized IgG4 monoclonal antibody against CD47, has shown promise in phase Ia trials (NCT04980885, NCT04900350), being tolerated up to 20 mg kg–1 and achieving sustained peripheral receptor occupancy at 10 mg kg–1 and higher148. Notably, ALX14 is a high affinity anti-CD47 antibody with lower molecular weight, allowing for greater dosage149, and is under clinical trials for a large number of cancers (Table 1). Magrolimab is an anti-CD47 monoclonal antibody that was recently given FDA breakthrough therapy designation for interventions in myelodysplastic syndrome due to promising clinical trial data150. The drug, in combination with either azacitidine or venetoclax + azacitidine, is in phase III trials for TP53-mutated AML (NCT04778397). About 20 clinical trials are currently underway using magrolimab in combination with other available chemotherapeutic drugs (Table 1). Recently, a few other inhibitory receptors and cognate molecules have been identified on tumour-associated macrophages and/or the tumour cells, such as the LILRB1/MHC class I and PD1/PDL1, and these appear to impact clearance of tumour cells and, in turn, disease prognosis151.
Another approach to promote the development of antitumour immunity involves induction of non-apoptotic and immunogenic forms of cell death, such as necroptosis. Immunogenic cell death is characterized by chronic exposure of intracellular damage-associated molecular patterns — particularly calreticulin, HMGB1, HSP70/90 and ATP — to the tumour microenvironment, stimulating the otherwise suppressed antitumour immune niche152. A cohort of drugs repurposed for inducing immunogenic cell death are in various stages of clinical trials. These include anthracyclines, glycosides, oxaliplatin, bleomycin and cyclophosphamide, among others, and have been extensively reviewed153,154.
Although efferocytosis is generally anti-inflammatory, this can be modified. For example, whether macrophages ‘interpret’ a dying cell as anti-inflammatory versus pro-inflammatory depends on the expression of SLC12a2 and the associated chloride efflux during efferocytosis155. Blocking SLC12a2 promoted enhanced apoptotic corpse uptake but the response of the efferocytic macrophages was pro-inflammatory. As SLC12a2 can be targeted via small molecules, targeting SLC12a2 within the tumour microenvironment may help antitumour responses; an approach which remains to be tested. In summary, efferocytosis, efferocytic receptors and subsequent responses of efferocytic phagocytes could play important roles in suppressing antitumour immunity. Finding the correct balance between dead cell clearance and development of antitumour immunity is a challenging aspect of drug development, and combinatorial therapies coupled to localized delivery of these drugs might be a way forward.
Infectious diseases
Host cell death is a well-defined characteristic of pathogenic insults. The modality of host cell death, such as necroptosis, pyroptosis or apoptosis, is largely dependent on the strain of the infecting pathogen. Whether host cell death triggered by infection benefits or harms the host is governed by numerous factors such as the nature of cell death, kinetics of death post infection, degree of death and extent of the removal of dead cell debris. Efferocytosis of dying infected cargo plays a crucial role in clearing infection, or, as it happens in some cases, provides a new host niche for the pathogen upon engulfment.
Beneficial role of efferocytosis
Mycobacterium tuberculosis uses macrophages as its primary host. Virulent strains of the bacteria are known to suppress apoptosis of infected macrophages while pushing necrotic death. Upon efferocytosis of infected apoptotic macrophages by healthy macrophages, the bacterial load has been demonstrated to be significantly reduced both in vitro and in the lungs of infected mice156. Further, blocking TIM4 in resident peritoneal macrophages significantly increased the incidence of secondarily infected cells, and this was attributed to secondary necrosis of the primary infected cells156. Along with aiding bacterial clearance, efferocytosis promotes antigen presentation. Dendritic cells taking up M. tuberculosis-infected apoptotic vesicles was shown to increase antigen presentation to CD8+ T cells157. Pulmonary tuberculosis is often defined by the presence of caseous granulomas with a lipid-rich necrotic core, formed as a result of rapid infection-induced death of lipid-laden macrophages. It is possible that those treatments activating efferocytosis in the early stages of granuloma development, before fibrotic cuff formation, might aid in reducing the bacterial burden. Other bacterial infections also show efferocytosis-mediated clearance of pathogens, albeit by different processes. Pyroptotic cell death, triggered by Klebsiella pneumoniae infection of macrophages, aided efficient clearance of the pathogen via efferocytosis of the pyroptotic debris158. Salmonella typhimurium infection could trigger pyroptotic cell death in host macrophages leading to the formation of pore-induced intracellular traps. Pore-induced intracellular traps expose numerous ‘find-me’ and ‘eat-me’ signals, and these structures could aid in efferocytosis and clearance of bacilli.
Efferocytosis also plays a critical role in viral infections. Influenza virus H1N1-infected mouse lungs show a rapid influx of macrophages and neutrophils. These phagocytes then engulf influenza virus-infected apoptotic cells159. Severe cases of influenza often lead to ARDS (acute respiratory distress syndrome) and pneumonia-like symptoms owing to virus-induced alveolar epithelium damage and the ensuing immune cell infiltration and cytokine storm. How efferocytosis of the dying neutrophils aids in infection clearance was recently demonstrated159,160. Influenza strain H3N2 infection in mouse airway elicited a massive, yet temporary, infiltration of neutrophils and monocytic cells into the trachea. The infiltrating neutrophils were engulfed by tissue-resident phagocytes and the infiltrating monocytes. Monocyte depletion in chemokine receptor 2 (CCR2) knockout mice led to a significant accumulation of apoptotic neutrophils in the lungs. Apoptotic neutrophil-educated Ly6C+/CD64+ inflammatory monocytes were critical antigen-presenting cells, responsible for CD8+ T cell activation. Further, EGF secreted from apoptotic neutrophils promoted the differentiation of infiltrating monocytes into antigen-presenting cells, and also facilitated CD8+ T cell effector roles160. Efferocytosis may also play a role in human immunodeficiency virus (HIV-1), which primarily infects CD4+ T cells, causing a steady decline in the numbers of these cells161. Similar to other viral infections, HIV-1 infection induces PS exposure on infected cells by triggering either apoptosis or pyroptosis162,163. The efferocytosis of HIV-infected T cells was facilitated by MerTK-mediated uptake by macrophages and this was found to be aided by Gas6 and Protein S164. Efferocytosis was shown to play a positive role in HIV infections, as the engulfment of apoptotic HIV-1-infected cells by dendritic cells aided the expansion of viral antigen-specific CD4+ and CD8+ T cells165.
Detrimental role of efferocytosis
Although efferocytosis aids in suppressing infections in several scenarios, this is not always the case. Listeria monocytogenes has evolved to utilize efferocytosis machinery as a means of cell to cell spread. The bacteria were shown to induce membrane damage and localized PS exposure on the host cell membrane166. Eventually, these areas of high-pathogen density were found to form bacteria-carrying vesicles, which could infect surrounding macrophages by means of PS binding to TIM4 (ref.166). In M. tuberculosis infections, the outcome of efferocytosis is often heavily dependent on the nature of the infecting strain, as the virulent strains of M. tuberculosis triggered necrosis of infected host neutrophils. The efferocytosis of these infected and necrotic neutrophils by macrophages, instead of infection clearance, has been shown to lead to further replication of the bacilli inside the efferocytic macrophages and the spread of infection167. Parasites such as Leishmania major and Trypanosoma cruzi have also evolved strategies to manipulate the efferocytosis machinery for their advantage. In addition to interfering with phagolysosomal fusion, efferocytosis of L. major-infected neutrophils by dendritic cells led to a reduction in the presentation of the parasite-derived antigens, and was dependent on MerTK signalling168. Engulfment of L. major-infected neutrophils by macrophages also led to parasite replication and suppression of inflammatory cytokine production by macrophages169. T. cruzi-infected apoptotic cells, when engulfed by macrophages, acted as Trojan horses for the parasite, allowing parasite growth in the engulfing cells. This was linked to TGFβ, which not only suppressed inflammatory cytokines but also triggered the activity of ornithine decarboxylase, leading to increases in putrescine, which aided intracellular parasite replication170.
Opportunities for targeting the efferocytic machinery in infections
The possibility of targeting efferocytosis for therapeutic benefit during infections is context-dependent. Some PS receptors may aid virus binding and entry into host cells. TIM family members are involved in viral infections including Marburg virus, HIV (in vitro)171, Ebola virus (in vivo)172, Japanese encephalitis virus173, Dengue virus174,175 and so on. Similarly, Gas6 was found to facilitate the entry of Ebola virus by bridging the viral envelope-expressed PS to Axl on target cells176. In the context of viral infections, there are two plausible mechanisms of therapeutically exploiting efferocytosis. First, antibodies or small peptides that block different PS receptors from interacting with vital PS molecules could be administered during the replicative phases of viral infections to inhibit cellular entry; as proof of concept, a reagent capable of blocking PS and PE mediated viral entry into host cells was highly effective in blocking Zika virus replication in vivo177. Second, perhaps enhancing the expression of PS receptors during later stages of enveloped virus infection may help inhibit viral shedding. During the late stages of HIV-1 replication, viral budding and release is associated with incorporation of PS receptors on HIV-1 virions178, and these HIV-1 particles are retained on the plasma membrane, inhibiting viral release. Targeting efferocytosis is also emerging as an interesting opportunity in COVID-19 infections (Box 1).
In bacterial infections, the plasticity with which bacterial pathogens engage the efferocytic machinery dictates the choice of intervention. The cytoplasmic efferocytosis protein ELMO1 was found to be essential for Salmonella enterica subsp. enterica serovar Typhimurium infection of enteric macrophages, bactericidal ROS production and LC3-mediated degradation of the engulfed bacteria179–181. M. tuberculosis infection induces the expression of miR-143 and miR-365, which suppress ELMO1 expression, aiding intracellular bacterial replication182. Small molecules that enhance intracellular signalling by PS receptors, while not increasing their membrane expression, might be an extremely successful host-directed therapy. The strategy will retain the antibacterial benefits while keeping the levels of bacterial invasion low. Owing to the complexity of host–pathogen interactions, the highly variable role of efferocytosis during infections and the ever-evolving nature of such interfaces, designing therapeutic interventions for directly inhibiting pathogenic infections is likely to be challenging.
Box 1 COVID-19 and its linkage to the efferocytic machinery.
Severe COVID-19 is characterized by an infiltration of monocytes, macrophages and T cells into the lungs, causing the build-up of a cytokine storm. This phenomenon, if unchecked, ultimately leads to pulmonary oedema, pneumonia and ARDS (acute respiratory distress syndrome), resulting in lung and multiorgan damage224. Patients with severe COVID-19 also display macrophage activation syndrome225. The causes for the development of ARDS in patients with COVID-19 are decreasing as the world has seen mass vaccination programmes. A detailed single-cell transcriptomics and proteomics study highlighted a highly dysregulated myeloid cell behaviour in patients with severe COVID-19 with myelopoiesis and expansion of dysfunctional neutrophils expressing PDL1 (ref.226). Further, an elevated neutrophil to lymphocyte ratio in the blood has been described as a potential indicator of disease severity and ARDS development227. Classically, patients with ARDS have been reported to show reduced neutrophil clearance of neutrophil extracellular traps (NETs), largely due to the reduced efferocytosis capacity of macrophages228, and a study of 33 patients with COVID-19 demonstrated evidence for NET-mediated immunothrombosis229. It is tempting to speculate that reduced efferocytosis in the lungs of patients with COVID-19, with exacerbated NET formation and death of infiltrating cells, contributes to the development of ARDS. It has recently been demonstrated that the efferocytosis of SARS-CoV-2-infected apoptotic cells augmented the production of inflammatory cytokines and diminished successive cargo uptake by macrophages in vitro. Furthermore, patient-derived lung monocytes and macrophages showed reduced expression of efferocytic receptors230. Thus, it is tempting to speculate that improving efferocytosis may facilitate clearance of dying cells (in specific affected tissues), and thereby help avoid chronic inflammation associated with ‘long COVID’.
Phosphatidylserine (PS) on membranes of viruses has long been shown to be relevant in infection. Interestingly, a recent finding highlighted PS exposure and the presence of multinucleated syncytia in the pneumocytes of patients with COVID-19 to the SARS-CoV-2 Spike protein. Through candidate drug screens, compounds inhibiting syncytia formation in vitro were identified and a prime candidate was found to be niclosamide. This compound is a strong inhibitor of the Ca2+-activated TMEM16 family of calcium-activated lipid scramblases and channels. Out of the ten members of this family, the expression of TMEM16F was found to be enhanced by the Spike protein. Inhibition of TMEM16F by niclosamide or benzbromarone reduced the PS exposure on cells expressing the Spike protein, and, in turn, dramatically reduced syncytia231. Proof of principle for the concept of PS receptor blocking as a potential therapeutic intervention for COVID-19 treatment has been suggested207. These observations offer a unique opportunity to further explore whether inhibition of PS exposure could be an approach to reducing the viral load. The role of efferocytosis in SARS-CoV-2 infections has been reviewed232.
Systemic lupus erythematosus
SLE is an autoimmune disease in which patients classically carry antibodies against self-antigens such as DNA, phospholipids and nuclei. Out of the numerous triggers associated with SLE induction, defective efferocytosis is considered a major contributor and patients with SLE often demonstrate accumulation of apoptotic cells183, which eventually turn secondarily necrotic, releasing damage-associated molecular patterns. Multiple molecules associated with efferocytosis have now been identified as defective or malfunctioning in patients with SLE. ADAM10 and ADAM17 metalloproteases exhibit higher cleavage activity in SLE models, leading to the cleavage of cell surface PS receptors, reducing efferocytosis efficiency184,185. Genetic deletion of MerTK induced lupus-like symptoms in older mice. The heightened amounts of soluble PS receptors (generated by ADAM10/17 activity) compete for PS binding, further reducing efferocytosis, a phenomenon observed in juvenile SLE onset186. Complement plays an important role in the clearance of apoptotic cells and immune complexes, and defects in early complement components such as C1q/s/r, C4 and C2 lead to SLE development187,188. Further, mice deficient in the phagocytic receptor SCARF1 develop SLE-like systems with large pools of uncleared apoptotic cells. A significant proportion of patients with SLE carry anti-SCARF autoantibodies189. Along with suppressing apoptotic cell engulfment, autoantibody accumulation in patients with SLE diminished DNAse1L3 activity, further exacerbating pathology190. Variations in the behaviour of apoptotic cells themselves can also contribute to the development of SLE. Further, mice lacking phospholipid scramblase Xcr1 also exhibit SLE-like symptoms191 (Fig. 2).
Opportunities for targeting the efferocytic machinery in SLE
Efferocytosis of apoptotic cells, as discussed earlier, is a potent inducer of nuclear receptor signalling (PPAR/LXR), which leads to an anti-inflammatory and pro-efferocytosis gene expression programme192. Owing to the pro-efferocytosis and resolution properties, treatment with the PPARγ agonist pioglitazone could improve vascular defects in patients with SLE (NCT02338999)193. A parallel study pointed to benefits of PPARγ/LXR pathway agonists in aiding LC3-associated phagocytosis (US20190145961A1). With the accumulation of DNA released by secondary necrotic cells, another complementary approach towards SLE treatment could be the administration of DNAse1L3 via nanoparticle-mediated delivery methods194 (Fig. 2). As discussed above, the cleavage of PS receptors such as MerTK is a contributor to poor prognosis in SLE. However, patients with SLE often develop glomerular nephritis owing to heightened proliferation signalling-mediated by Gas6–Axl. The Axl-specific inhibitor R428 suppressed disease progression in the mouse model of immune glomerulonephritis195 (Table 1). It can be appreciated that organ-specific roles of PS receptors in SLE provide a unique window of opportunity for targeted and personalized therapy. Combinatorial therapy, where classical treatment lines are augmented with agents such as Resolvins196 or PPARγ/LXR agonists, offer additional opportunities.
Diabetes and obesity
One of the major manifestations of defective efferocytosis in diabetes or obesity-induced diabetes is in the form of delayed wound healing in animal models197,198. In multiple diabetic animal models, apoptotic cells and increased inflammatory cytokines accumulate at the wound site197. Although the contributions of other efferocytic cells, such as dendritic cells/epithelial cells/fibroblasts at wound sites are still under study, the mechanisms mediating defective efferocytosis have started to emerge. Macrophages in vitro and from diabetic db/db mice exposed to high glucose had reduced efferocytosis capacity and reduced levels of miR-126 — responsible for suppressing the expression of ADAM9. Greater ADAM9-mediated MerTK cleavage was linked to a reduction in efferocytosis199 (Fig. 2). Aberrant signalling events are also responsible for reduced efferocytosis, and lower inflammation resolution in high-fat diet-induced obesity mouse models200. Under normal conditions, S1P released by apoptotic cells engaged HIF1α in macrophages leading to expression and release of erythropoietin (EPO). EPO was a strong inducer of PPARγ expression via the Jak2/ERK pathway, leading to the expression of pro-efferocytosis and anti-inflammatory genes201. Interestingly, the levels of secreted EPO, macrophage EPO receptor and p-Jak2 were significantly lower in obese mice with zymosan-induced peritonitis, compared with lean mice. The obese mice in the peritonitis model also exhibited significantly lower efferocytosis and increased accumulation of apoptotic cells (Fig. 2). Although detailed mechanistic understanding of the influence of high glucose or circulating TAGs on the efferocytic capacity of phagocytes is still incomplete, the studies above may point to routes for targeting efferocytic steps in obesity.
Opportunities for targeting the efferocytic machinery in diabetes and obesity
COVID mRNA vaccines and their wide acceptance has paved the way for exploring mRNA-based therapeutics. In diabetic mice, miR-126 levels were suppressed. A plausible intervention in such mice could be the delivery of miR-126 via nanoparticles or REDV peptide-modified chitosan202 to sites of heavy apoptotic cargo burden such as diabetic wounds. Another interesting approach could be neutralization of the cleaved soluble PS receptors. Obese mice in the zymosan peritonitis model201, when treated with recombinant EPO, were shown to exhibit significantly reduced apoptotic cell burden and improved inflammation resolution. Recombinant EPO is already used as treatment for kidney disorders and can be potentially repurposed as a treatment for obesity to decrease the apoptotic cell burden and associated inflammation. Another interesting finding in diabetes and obesity suggests that an increase in saturated fatty acids with a concomitant decrease in ω3-fatty acids leads to a dramatic reduction in macrophage efferocytic capacity. The efferocytosis capacity could be restored in obese mice by altering the diet balance to ω-fatty acids, hinting towards dietary interventions to improve efferocytic capacity198. Administration of anti-inflammatory metabolites/diet-based therapy may play out to be an effective treatment option depending on the patient’s medical history, opening new avenues for personalized therapeutics.
Challenges and future perspectives
The multistep process of efferocytosis has come to the forefront in the context of numerous pathologies, either as an aetiological factor or a contributor to poor prognosis when this process is disturbed or impaired. Increased understanding of the roles and molecular mechanisms of efferocytosis has revealed many avenues for therapeutic intervention, but has also highlighted numerous challenges.
Efferocytosis is a ubiquitous process involving highly tissue-specific PS receptors and apoptotic cell burden. One evident challenge in targeting efferocytosis therefore lies in targeting the correct receptor type in the organ involved in disease aetiology. In addition, although the SLC family of proteins are highly druggable, and they heavily influence the immunological and efferocytic ability of phagocytes, targeting this family of proteins is challenged by the redundancy in some transporters. Multiple solutes may be transported via a single SLC, which must also be considered.
In pathologies such as cancers, mechanisms have ‘evolved’ to undermine the beneficial effects of efferocytosis. Whereas inhibiting efferocytosis in cancer can help the development of inflammatory responses, it can also further reduce efferocytosis of cancerous cells. Although pathogenic infections, such as M. tuberculosis infections, often manifest a dependence on efferocytosis for bacterial spread, this is highly dependent on the infecting strain and prognosis167,203. Despite these challenges, efferocytosis — with its highly sophisticated multistage signalling — offers unique windows of therapeutic opportunities in both cancers and pathogenic infections. Depending on the nature and stage of cancer, combinatorial drugs targeting efferocytosis along with standard chemotherapy could prove valuable. Further, in pathogenic insults, when the infecting strain is known, personalized treatment targeting efferocytosis can be moulded to prevent bacterial spread and aid clearance of infected cells.
In recent years, PS receptors on phagocytes have been thoroughly explored in the context of cancer therapy, and drugs targeting these receptors have shown promising results (Table 1). Steps to redirect some of these therapeutics for atherosclerosis and diabetes are already underway and might open novel therapeutic avenues. The ability of efferocytosis to aid senescent cell removal and reduce chronic inflammation associated with inflammaging holds great potential. Thus, novel therapeutics capable of enhancing efferocytosis (even moderately) could be useful in improving quality of life for ageing individuals. A plethora of exciting findings linking the metabolic changes in efferocytic cells to their immunological behaviour have added a new dimension to how this process can be viewed and therapeutically targeted. Oral or topical administration of endogenous metabolic mediators is already being explored as a therapeutic intervention in numerous other pathologies via mechanisms distinct from efferocytosis204. Exploring metabolic combinations that can improve efferocytosis and, thus, disease prognosis can be a novel therapeutic tool (for example, administration of ω3-fatty acids198). The development of methodologies for directed metabolite delivery into sites of inflammation may improve disease prognosis (for example, arthritis)15 and methods for safe administration of apoptotic and necroptotic cells have the potential to revolutionize transplant biology and cancer therapy, respectively.
Immune and metabolic profiles of patients with the same pathology often vary widely205. Efferocytosis can engage directly with immune and metabolic pathways and provides opportunities to fine-tune immunological behaviour via metabolic interventions15. The developments in the field of pharmaco-metabolomics, coupled to the highly drug targetable nodes in efferocytosis, make this combination a unique avenue for personalized medicine. The future of therapeutics for lifestyle disorders and inflammatory diseases rests on multipronged treatments, at the centre of which lies the dynamic process of efferocytosis, connecting immunological outcomes to metabolic cues and cell death.
Acknowledgements
The authors thank the members of the Ravichandran laboratory for numerous discussions. This work was supported by Fonds Wetenschappelijk Onderzoek (FWO) Odysseus grant G0F5716N, EOS DECODE 30837538, Special Research Fund UGent (iBOF BOF20/IBF/037), European Research Council (ERC) (grant agreement no. 835243) and grants from the National Heart, Lung, and Blood Institute (NHLBI) (P01HL120840), National Institute of Allergy and Infectious Diseases (NIAID) (R01AI159551) and National Institute of General Medical Sciences (NIGMS) (R35GM122542). P.M. is supported by a FWO Postdoctoral Fellowship (1227220N).
Glossary
- Apoptosis
A process of cell death triggered by either extrinsic or intrisic cues that ultimately result in activation of executioner caspases and caspase-dependent cleavage of cellular substrates, leading to an ordered cellular demise.
- Necroptosis
A regulated form of lytic cell death, requiring the function of receptor interacting protein 1/3 and their substrate, MLKL, oligomerization-induced plasma membrane pore formation and eventual loss of membrane integrity.
- Pyroptosis
Inflammatory caspase-driven lytic cell death, usually triggered by pathogenic insults. This involves inflammasome assembly, which via caspase 1 induces Gasdermin D activation. Gasdermin D proteins form pores in the plasma membrane, leading to membrane rupture and lytic cell death accompanied by the release of IL-1β and IL-18.
- Ferroptosis
A form of caspase-independent lytic cell death induced by excess free intracellular iron inducing lipid peroxidation, causing plasma membrane pore formation and osmotic cell rupture.
- Fc receptor-mediated phagocytosis
The process in which a cell-surface immune cell receptor recognizes and binds to the Fc fragment of antibodies, allowing for phagocytosis of the antibody-coated particles.
- Solute carrier (SLC) family
A superfamily of membrane transporters that carry a wide number of substrates across the plasma membrane, and membranes of intracellular organelles.
- Chemokines
Secreted proteins that act as chemoattractants and induce immune cell migration.
- Pannexin channels
Heptameric plasma membrane channels that are activated in apoptotic cells by caspase 3 cleavage (or other modes in live cells) allowing the release of metabolites, including ATP.
- Purinergic receptor P2Y
A G-protein-coupled receptor that is activated by extracellular adenine and uridine nucleotides, and UDP-glucose. The P2Y family consists of eight receptors that can engage in intracellular signalling via adenylate cyclase or phospholipase C.
- Liver-X receptors (LXRs) α/β
Transcription factors belonging to the nuclear receptor family. The receptors are activated by the presence of oxysterols and form heterodimers with retinoic acid receptors to initiate transcriptional changes.
- PPARγ
A transcription factor belonging to the members of the nuclear receptor family, involved in regulating cellular metabolism and immune cell behaviour, and anti-inflammatory responses.
- Foam cells
Macrophages carrying high lipid and cholesterol burden seen in atherosclerotic plaques, due to enhanced lipoprotein uptake.
- ADAM family proteases
(A disintegrin and metalloprotease (ADAM) family of proteins). A family of transmembrane proteins with strong proteolytic activity. The substrates for ADAM proteases are very often the extracellular domains of transmembrane proteins.
- Specialized pro-resolvin mediators
(SPMs). A class of immunologically active lipid derivates that limit acute inflammatory responses and aid in inflammation resolution. The family includes lipoxins, E-series and D-series resolvins, protectins and maresins.
- Senotherapies
Therapies aimed at killing senescent cells and diminishing the effects of the senescence-associated secretory phenotype that is a key driver of sterile inflammation during ageing.
- Pore-induced intracellular traps
The cellular debris emanating from a pyroptotic cell that entrap the intracellular bacteria as they are released during pyroptosis of the host cell.
- ARDS
(Acute respiratory distress syndrome). A form of severe respiratory trauma and failure characterized by inflammation and oedema in the alveolar spaces.
Author contributions
The authors contributed equally to the discussion, writing, preparation, and editing of this manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Boada-Romero E, Martinez J, Heckmann BL, Green DR. The clearance of dead cells by efferocytosis. Nat. Rev. Mol. Cell Biol. 2020;21:398–414. doi: 10.1038/s41580-020-0232-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Green D, Oguin T, Martinez J. The clearance of dying cells: table for two. Cell Death Differ. 2016;23:915–926. doi: 10.1038/cdd.2015.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Morioka S, Maueröder C, Ravichandran KS. Living on the edge: efferocytosis at the interface of homeostasis and pathology. Immunity. 2019;50:1149–1162. doi: 10.1016/j.immuni.2019.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Doran AC, Yurdagul A, Tabas I. Efferocytosis in health and disease. Nat. Rev. Immunol. 2019;20:254–267. doi: 10.1038/s41577-019-0240-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Henson PM. Cell removal: efferocytosis. Annu. Rev. Cell Devel. Biol. 2017;33:127–144. doi: 10.1146/annurev-cellbio-111315-125315. [DOI] [PubMed] [Google Scholar]
- 6.Lauber K, Blumenthal SG, Waibel M, Wesselborg S. Clearance of apoptotic cells: getting rid of the corpses. Mol. Cell. 2004;14:277–287. doi: 10.1016/S1097-2765(04)00237-0. [DOI] [PubMed] [Google Scholar]
- 7.Arandjelovic S, Ravichandran KS. Phagocytosis of apoptotic cells in homeostasis. Nat. Immunol. 2015;16:907. doi: 10.1038/ni.3253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pasparakis M, Vandenabeele P. Necroptosis and its role in inflammation. Nature. 2015;517:311–320. doi: 10.1038/nature14191. [DOI] [PubMed] [Google Scholar]
- 9.Elliott MR, et al. Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature. 2009;461:282–286. doi: 10.1038/nature08296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chekeni FB, et al. Pannexin 1 channels mediate ‘find-me’ signal release and membrane permeability during apoptosis. Nature. 2010;467:863–867. doi: 10.1038/nature09413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lauber K, et al. Apoptotic cells induce migration of phagocytes via caspase-3-mediated release of a lipid attraction signal. Cell. 2003;113:717–730. doi: 10.1016/S0092-8674(03)00422-7. [DOI] [PubMed] [Google Scholar]
- 12.Gude DR, et al. Apoptosis induces expression of sphingosine kinase 1 to release sphingosine-1-phosphate as a “come-and-get-me” signal. FASEB J. 2008;22:2629–2638. doi: 10.1096/fj.08-107169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Truman LA, et al. CX3CL1/fractalkine is released from apoptotic lymphocytes to stimulate macrophage chemotaxis. Blood. 2008;112:5026–5036. doi: 10.1182/blood-2008-06-162404. [DOI] [PubMed] [Google Scholar]
- 14.Medina CB, Ravichandran KS. Do not let death do us part: ‘find-me’ signals in communication between dying cells and the phagocytes. Cell Death Differ. 2016;23:979–989. doi: 10.1038/cdd.2016.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Medina CB, et al. Metabolites released from apoptotic cells act as tissue messengers. Nature. 2020;580:130–135. doi: 10.1038/s41586-020-2121-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Elliott MR, Ravichandran KS. The dynamics of apoptotic cell clearance. Dev. Cell. 2016;38:147–160. doi: 10.1016/j.devcel.2016.06.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Marques-da-Silva C, Burnstock G, Ojcius DM, Coutinho-Silva R. Purinergic receptor agonists modulate phagocytosis and clearance of apoptotic cells in macrophages. Immunobiology. 2011;216:1–11. doi: 10.1016/j.imbio.2010.03.010. [DOI] [PubMed] [Google Scholar]
- 18.Yamaguchi H, Maruyama T, Urade Y, Nagata S. Immunosuppression via adenosine receptor activation by adenosine monophosphate released from apoptotic cells. eLife. 2014;3:e02172. doi: 10.7554/eLife.02172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Elliott MR, Koster KM, Murphy PS. Efferocytosis signaling in the regulation of macrophage inflammatory responses. J. Immunol. 2017;198:1387–1394. doi: 10.4049/jimmunol.1601520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Köröskényi K, et al. Involvement of adenosine A2A receptors in engulfment-dependent apoptotic cell suppression of inflammation. J. Immunol. 2011;186:7144–7155. doi: 10.4049/jimmunol.1002284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Medina CB, et al. Pannexin 1 channels facilitate communication between T cells to restrict the severity of airway inflammation. Immunity. 2021;54:1715–1727.e7. doi: 10.1016/j.immuni.2021.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Benden C, et al. Extracorporeal photopheresis after lung transplantation: a 10-year single-center experience. Transplantation. 2008;86:1625–1627. doi: 10.1097/TP.0b013e31818bc024. [DOI] [PubMed] [Google Scholar]
- 23.Urbani, L. et al. in Transplantation Proceedings 1175–1178 (Elsevier, 2004).
- 24.He Y, et al. Antiinflammatory effect of Rho kinase blockade via inhibition of NF-κB activation in rheumatoid arthritis. Arthritis Rheum. 2008;58:3366–3376. doi: 10.1002/art.23986. [DOI] [PubMed] [Google Scholar]
- 25.Dangi A, Luo X. Harnessing apoptotic cells for transplantation tolerance: current status and future perspectives. Curr. Transplant. Rep. 2017;4:270–279. doi: 10.1007/s40472-017-0167-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chiu Y-H, et al. Deacetylation as a receptor-regulated direct activation switch for pannexin channels. Nat. Commun. 2021;12:1–14. doi: 10.1038/s41467-021-24825-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kelley SM, Ravichandran KS. Putting the brakes on phagocytosis: “don’t-eat-me” signaling in physiology and disease. EMBO Rep. 2021;22:e52564. doi: 10.15252/embr.202152564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fadok VA, et al. Exposure of phosphatidylserine on the surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages. J. Immunol. 1992;148:2207–2216. [PubMed] [Google Scholar]
- 29.Arashiki N, et al. ATP11C is a major flippase in human erythrocytes and its defect causes congenital hemolytic anemia. Haematologica. 2016;101:559–565. doi: 10.3324/haematol.2016.142273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Segawa K, Nagata S. An apoptotic ‘eat me’ signal: phosphatidylserine exposure. Trends Cell Biol. 2015;25:639–650. doi: 10.1016/j.tcb.2015.08.003. [DOI] [PubMed] [Google Scholar]
- 31.Suzuki J, Imanishi E, Nagata S. Exposure of phosphatidylserine by Xk-related protein family members during apoptosis. J. Biol. Chem. 2014;289:30257–30267. doi: 10.1074/jbc.M114.583419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Suzuki J, et al. Calcium-dependent phospholipid scramblase activity of TMEM16 protein family members. J. Biol. Chem. 2013;288:13305–13316. doi: 10.1074/jbc.M113.457937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Suzuki J, Denning DP, Imanishi E, Horvitz HR, Nagata S. Xk-related protein 8 and CED-8 promote phosphatidylserine exposure in apoptotic cells. Science. 2013;341:403–406. doi: 10.1126/science.1236758. [DOI] [PubMed] [Google Scholar]
- 34.Fujii T, Sakata A, Nishimura S, Eto K, Nagata S. TMEM16F is required for phosphatidylserine exposure and microparticle release in activated mouse platelets. Proc. Natl Acad. Sci. USA. 2015;112:12800–12805. doi: 10.1073/pnas.1516594112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Park D, et al. BAI1 is an engulfment receptor for apoptotic cells upstream of the ELMO/Dock180/Rac module. Nature. 2007;450:430–434. doi: 10.1038/nature06329. [DOI] [PubMed] [Google Scholar]
- 36.Park D, Hochreiter-Hufford A, Ravichandran KS. The phosphatidylserine receptor TIM-4 does not mediate direct signaling. Curr. Biol. 2009;19:346–351. doi: 10.1016/j.cub.2009.01.042. [DOI] [PubMed] [Google Scholar]
- 37.Lee CS, et al. Boosting apoptotic cell clearance by colonic epithelial cells attenuates inflammation in vivo. Immunity. 2016;44:807–820. doi: 10.1016/j.immuni.2016.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kobayashi N, et al. TIM-1 and TIM-4 glycoproteins bind phosphatidylserine and mediate uptake of apoptotic cells. Immunity. 2007;27:927–940. doi: 10.1016/j.immuni.2007.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Moon B, et al. Mertk interacts with Tim-4 to enhance Tim-4-mediated efferocytosis. Cells. 2020;9:1625. doi: 10.3390/cells9071625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nishi C, Yanagihashi Y, Segawa K, Nagata S. MERTK tyrosine kinase receptor together with TIM4 phosphatidylserine receptor mediates distinct signal transduction pathways for efferocytosis and cell proliferation. J. Biol. Chem. 2019;294:7221–7230. doi: 10.1074/jbc.RA118.006628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Heckmann BL, Green DR. LC3-associated phagocytosis at a glance. J. Cell Sci. 2019;132:jcs222984. doi: 10.1242/jcs.222984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lemke G, Rothlin CV. Immunobiology of the TAM receptors. Nat. Rev. Immunol. 2008;8:327–336. doi: 10.1038/nri2303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tibrewal N, et al. Autophosphorylation docking site Tyr-867 in Mer receptor tyrosine kinase allows for dissociation of multiple signaling pathways for phagocytosis of apoptotic cells and down-modulation of lipopolysaccharide-inducible NF-κB transcriptional activation. J. Biol. Chem. 2008;283:3618–3627. doi: 10.1074/jbc.M706906200. [DOI] [PubMed] [Google Scholar]
- 44.Sen P, et al. Apoptotic cells induce Mer tyrosine kinase-dependent blockade of NF-κB activation in dendritic cells. Blood. 2007;109:653–660. doi: 10.1182/blood-2006-04-017368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rothlin CV, Ghosh S, Zuniga EI, Oldstone MB, Lemke G. TAM receptors are pleiotropic inhibitors of the innate immune response. Cell. 2007;131:1124–1136. doi: 10.1016/j.cell.2007.10.034. [DOI] [PubMed] [Google Scholar]
- 46.Wallet MA, et al. MerTK is required for apoptotic cell-induced T cell tolerance. J. Exp. Med. 2008;205:219–232. doi: 10.1084/jem.20062293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Filardy AA, et al. Proinflammatory clearance of apoptotic neutrophils induces an IL-12lowIL-10high regulatory phenotype in macrophages. J. Immunol. 2010;185:2044–2050. doi: 10.4049/jimmunol.1000017. [DOI] [PubMed] [Google Scholar]
- 48.Zizzo G, Hilliard BA, Monestier M, Cohen PL. Efficient clearance of early apoptotic cells by human macrophages requires M2c polarization and MerTK induction. J. Immunol. 2012;189:3508–3520. doi: 10.4049/jimmunol.1200662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Park S-Y, et al. Stabilin-2 modulates the efficiency of myoblast fusion during myogenic differentiation and muscle regeneration. Nat. Commun. 2016;7:1–15. doi: 10.1038/ncomms10871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cummings CT, DeRyckere D, Earp HS, Graham DK. Molecular pathways: MERTK signaling in cancer. Clin. Cancer Res. 2013;19:5275–5280. doi: 10.1158/1078-0432.CCR-12-1451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Park M, Kang KW. Phosphatidylserine receptor-targeting therapies for the treatment of cancer. Arch. Pharmacal Res. 2019;42:617–628. doi: 10.1007/s12272-019-01167-4. [DOI] [PubMed] [Google Scholar]
- 52.Penberthy KK, et al. Context-dependent compensation among phosphatidylserine-recognition receptors. Sci. Rep. 2017;7:14623. doi: 10.1038/s41598-017-15191-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Segawa K, Suzuki J, Nagata S. Constitutive exposure of phosphatidylserine on viable cells. Proc. Natl Acad. Sci. USA. 2011;108:19246–19251. doi: 10.1073/pnas.1114799108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Tsai RK, Discher DE. Inhibition of “self” engulfment through deactivation of myosin-II at the phagocytic synapse between human cells. J. Cell Biol. 2008;180:989–1003. doi: 10.1083/jcb.200708043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Barkal AA, et al. CD24 signalling through macrophage Siglec-10 is a target for cancer immunotherapy. Nature. 2019;572:392–396. doi: 10.1038/s41586-019-1456-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Brown S, et al. Apoptosis disables CD31-mediated cell detachment from phagocytes promoting binding and engulfment. Nature. 2002;418:200–203. doi: 10.1038/nature00811. [DOI] [PubMed] [Google Scholar]
- 57.Fond AM, Lee CS, Schulman IG, Kiss RS, Ravichandran KS. Apoptotic cells trigger a membrane-initiated pathway to increase ABCA1. J. Clin. Invest. 2015;125:2748–2758. doi: 10.1172/JCI80300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Viaud M, et al. Lysosomal cholesterol hydrolysis couples efferocytosis to anti-inflammatory oxysterol production. Circ. Res. 2018;122:1369–1384. doi: 10.1161/CIRCRESAHA.117.312333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Xian X, et al. LRP1 integrates murine macrophage cholesterol homeostasis and inflammatory responses in atherosclerosis. eLife. 2017;6:e29292. doi: 10.7554/eLife.29292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Cui D, et al. Pivotal advance: macrophages become resistant to cholesterol-induced death after phagocytosis of apoptotic cells. J. Leukoc. Biol. 2007;82:1040–1050. doi: 10.1189/jlb.0307192. [DOI] [PubMed] [Google Scholar]
- 61.Glass CK, Saijo K. Nuclear receptor transrepression pathways that regulate inflammation in macrophages and T cell. Nat. Rev. Imuunol. 2010;10:365–376. doi: 10.1038/nri2748. [DOI] [PubMed] [Google Scholar]
- 62.Gonzalez NA-, et al. Apoptotic cells promote their own clearance and immune tolerance through activation of the nuclear receptor LXR. Immunity. 2009;31:245–258. doi: 10.1016/j.immuni.2009.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ivanov S, et al. Lysosomal cholesterol hydrolysis couples efferocytosis to anti-inflammatory oxysterol production. Atherosclerosis. 2019;287:e77. doi: 10.1016/j.atherosclerosis.2019.06.223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Yurdagul A, Jr, et al. Macrophage metabolism of apoptotic cell-derived arginine promotes continual efferocytosis and resolution of injury. Cell Metab. 2020;31:518–533.e10. doi: 10.1016/j.cmet.2020.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gerlach BD, et al. Efferocytosis induces macrophage proliferation to help resolve tissue injury. Cell Metab. 2021;33:2445–2463.e8. doi: 10.1016/j.cmet.2021.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Morioka S, et al. Efferocytosis induces a novel SLC program to promote glucose uptake and lactate release. Nature. 2018;563:714–718. doi: 10.1038/s41586-018-0735-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zhang S, et al. Efferocytosis fuels requirements of fatty acid oxidation and the electron transport chain to polarize macrophages for tissue repair. Cell Met. 2019;29:443–456. doi: 10.1016/j.cmet.2018.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wang Y, et al. Mitochondrial fission promotes the continued clearance of apoptotic cells by macrophages. Cell. 2017;171:331–345.e22. doi: 10.1016/j.cell.2017.08.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Fernandez-Boyanapalli RF, et al. Impaired efferocytosis in human chronic granulomatous disease is reversed by pioglitazone treatment. J. Allergy Clin. Immunol. 2015;136:1399. doi: 10.1016/j.jaci.2015.07.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tanegashima K, et al. Epigenetic regulation of the glucose transporter gene Slc2a1 by β-hydroxybutyrate underlies preferential glucose supply to the brain of fasted mice. Genes. Cell. 2017;22:71–83. doi: 10.1111/gtc.12456. [DOI] [PubMed] [Google Scholar]
- 71.Lin L, Yee SW, Kim RB, Giacomini KM. SLC transporters as therapeutic targets: emerging opportunities. Nat. Rev. Drug Discov. 2015;14:543–560. doi: 10.1038/nrd4626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wang Y, Oram JF. Unsaturated fatty acids phosphorylate and destabilize ABCA1 through a phospholipase D2 pathway. J. Biol. Chem. 2005;280:35896–35903. doi: 10.1074/jbc.M506210200. [DOI] [PubMed] [Google Scholar]
- 73.Wang Y, Oram JF. Unsaturated fatty acids phosphorylate and destabilize ABCA1 through a protein kinase Cδ pathway. J. Lipid Res. 2007;48:1062–1068. doi: 10.1194/jlr.M600437-JLR200. [DOI] [PubMed] [Google Scholar]
- 74.Tang C, Oram JF. The cell cholesterol exporter ABCA1 as a protector from cardiovascular disease and diabetes. Biochim. Biophys. Acta. 2009;1791:563–572. doi: 10.1016/j.bbalip.2009.03.011. [DOI] [PubMed] [Google Scholar]
- 75.Cai B, et al. MerTK cleavage limits proresolving mediator biosynthesis and exacerbates tissue inflammation. Proc. Natl Acad. Sci. USA. 2016;113:6526–6531. doi: 10.1073/pnas.1524292113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Garbin U, et al. Expansion of necrotic core and shedding of Mertk receptor in human carotid plaques: a role for oxidized polyunsaturated fatty acids? Cardiovasc. Res. 2013;97:125–133. doi: 10.1093/cvr/cvs301. [DOI] [PubMed] [Google Scholar]
- 77.Cai B, et al. MerTK receptor cleavage promotes plaque necrosis and defective resolution in atherosclerosis. J. Clin. Invest. 2017;127:564–568. doi: 10.1172/JCI90520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Doran AC, et al. CAMKIIγ suppresses an efferocytosis pathway in macrophages and promotes atherosclerotic plaque necrosis. J. Clin. Invest. 2017;127:4075–4089. doi: 10.1172/JCI94735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Cai B, et al. MerTK signaling in macrophages promotes the synthesis of inflammation resolution mediators by suppressing CaMKII activity. Sci. Signal. 2018;11:eaar3721. doi: 10.1126/scisignal.aar3721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Godson C, et al. Cutting edge: lipoxins rapidly stimulate nonphlogistic phagocytosis of apoptotic neutrophils by monocyte-derived macrophages. J. Immunol. 2000;164:1663–1667. doi: 10.4049/jimmunol.164.4.1663. [DOI] [PubMed] [Google Scholar]
- 81.Dalli J, Serhan CN. Pro-resolving mediators in regulating and conferring macrophage function. Front. Immunol. 2017;8:1400. doi: 10.3389/fimmu.2017.01400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Fredman G, et al. An imbalance between specialized pro-resolving lipid mediators and pro-inflammatory leukotrienes promotes instability of atherosclerotic plaques. Nat. Commun. 2016;7:12859. doi: 10.1038/ncomms12859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Manega CM, et al. 12(S)-Hydroxyeicosatetraenoic acid downregulates monocyte-derived macrophage efferocytosis: new insights in atherosclerosis. Pharmacol. Res. 2019;144:336–342. doi: 10.1016/j.phrs.2019.03.012. [DOI] [PubMed] [Google Scholar]
- 84.Komura H, Miksa M, Wu R, Goyert SM, Wang P. Milk fat globule epidermal growth factor–factor VIII is down-regulated in sepsis via the lipopolysaccharide–CD14 pathway. J. Immunol. 2009;182:581–587. doi: 10.4049/jimmunol.182.1.581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Tosello-Trampont A-C, Nakada-Tsukui K, Ravichandran KS. Engulfment of apoptotic cells is negatively regulated by Rho-mediated signaling. J. Biol. Chem. 2003;278:49911–49919. doi: 10.1074/jbc.M306079200. [DOI] [PubMed] [Google Scholar]
- 86.Wu D-J, et al. Effects of fasudil on early atherosclerotic plaque formation and established lesion progression in apolipoprotein E-knockout mice. Atherosclerosis. 2009;207:68–73. doi: 10.1016/j.atherosclerosis.2009.04.025. [DOI] [PubMed] [Google Scholar]
- 87.Toyama T, et al. Fasudil ameliorates fibrosis, vasculopathy, and immune abnormalities in animal models of systemic sclerosis. J. Dermatol. Sci. 2016;84:e11. doi: 10.1016/j.jdermsci.2016.08.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Li Y, Wu Y, Wang Z, Zhang X-H, Wu W-K. Fasudil attenuates lipopolysaccharide-induced acute lung injury in mice through the Rho/Rho kinase pathway. Med. Sci. Monit. 2010;16:BR112–BR118. [PubMed] [Google Scholar]
- 89.Segain J-P, et al. Rho kinase blockade prevents inflammation via nuclear factor κB inhibition: evidence in Crohn’s disease and experimental colitis. Gastroenterology. 2003;124:1180–1187. doi: 10.1016/S0016-5085(03)00283-X. [DOI] [PubMed] [Google Scholar]
- 90.Galvão I, et al. ROCK inhibition drives resolution of acute inflammation by enhancing neutrophil apoptosis. Cells. 2019;8:964. doi: 10.3390/cells8090964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Kojima Y, et al. CD47-blocking antibodies restore phagocytosis and prevent atherosclerosis. Nature. 2016;536:86–90. doi: 10.1038/nature18935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Singla B, et al. Loss of myeloid cell-specific SIRPα, but not CD47, attenuates inflammation and suppresses atherosclerosis. Cardiovasc. Res. 2021 doi: 10.1093/cvr/cvab369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Flores AM, et al. Pro-efferocytic nanoparticles are specifically taken up by lesional macrophages and prevent atherosclerosis. Nat. Nanotechnol. 2020;15:154–161. doi: 10.1038/s41565-019-0619-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Gerlach BD, et al. Resolvin D1 promotes the targeting and clearance of necroptotic cells. Cell Death Differ. 2020;27:525–539. doi: 10.1038/s41418-019-0370-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Joseph SB, et al. Synthetic LXR ligand inhibits the development of atherosclerosis in mice. Proc. Natl Acad. Sci. USA. 2002;99:7604–7609. doi: 10.1073/pnas.112059299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Vucic E, et al. Regression of inflammation in atherosclerosis by the LXR agonist R211945: a noninvasive assessment and comparison with atorvastatin. JACC. 2012;5:819–828. doi: 10.1016/j.jcmg.2011.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Arabpour M, Saghazadeh A, Rezaei N. Anti-inflammatory and M2 macrophage polarization-promoting effect of mesenchymal stem cell-derived exosomes. Int. Immunopharmacol. 2021;97:107823. doi: 10.1016/j.intimp.2021.107823. [DOI] [PubMed] [Google Scholar]
- 98.Jiang W, Xu J. Immune modulation by mesenchymal stem cells. Cell Prolif. 2020;53:e12712. doi: 10.1111/cpr.12712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zhang Z, et al. Mesenchymal stem cells promote the resolution of cardiac inflammation after ischemia reperfusion via enhancing efferocytosis of neutrophils. J. Am. Heart Assoc. 2020;9:e014397. doi: 10.1161/JAHA.119.014397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Kennedy BK, et al. Geroscience: linking aging to chronic disease. Cell. 2014;159:709–713. doi: 10.1016/j.cell.2014.10.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Pinti M, et al. Circulating mitochondrial DNA increases with age and is a familiar trait: implications for “inflamm-aging”. Eur. J. Immunol. 2014;44:1552–1562. doi: 10.1002/eji.201343921. [DOI] [PubMed] [Google Scholar]
- 102.Aprahamian T, Takemura Y, Goukassian D, Walsh K. Ageing is associated with diminished apoptotic cell clearance in vivo. Clin. Exp. Immunol. 2008;152:448–455. doi: 10.1111/j.1365-2249.2008.03658.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Arnardottir HH, Dalli J, Colas RA, Shinohara M, Serhan CN. Aging delays resolution of acute inflammation in mice: reprogramming the host response with novel nano-proresolving medicines. J. Immunol. 2014;193:4235–4244. doi: 10.4049/jimmunol.1401313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Tourki B, et al. Lack of resolution sensor drives age-related cardiometabolic and cardiorenal defects and impedes inflammation-resolution in heart failure. Mol. Metab. 2020;31:138–149. doi: 10.1016/j.molmet.2019.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.De Maeyer RP, et al. Blocking elevated p38 MAPK restores efferocytosis and inflammatory resolution in the elderly. Nat. Immunol. 2020;21:615–625. doi: 10.1038/s41590-020-0646-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.di Fagagna FdA. Living on a break: cellular senescence as a DNA-damage response. Nat. Rev. Cancer. 2008;8:512–522. doi: 10.1038/nrc2440. [DOI] [PubMed] [Google Scholar]
- 107.Shay JW, Wright WE. Hayflick, his limit, and cellular ageing. Nat. Rev. Mol. Cell Biol. 2000;1:72–76. doi: 10.1038/35036093. [DOI] [PubMed] [Google Scholar]
- 108.Wiley CD, et al. Mitochondrial dysfunction induces senescence with a distinct secretory phenotype. Cell Metab. 2016;23:303–314. doi: 10.1016/j.cmet.2015.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Childs BG, et al. Senescent cells: an emerging target for diseases of ageing. Nat. Rev. Drug Discov. 2017;16:718. doi: 10.1038/nrd.2017.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Campisi J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell. 2005;120:513–522. doi: 10.1016/j.cell.2005.02.003. [DOI] [PubMed] [Google Scholar]
- 111.Hall BM, et al. Aging of mice is associated with p16(Ink4a)-and β-galactosidase-positive macrophage accumulation that can be induced in young mice by senescent cells. Aging. 2016;8:1294. doi: 10.18632/aging.100991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Rodier F, Campisi J. Four faces of cellular senescence. J. Cell Biol. 2011;192:547–556. doi: 10.1083/jcb.201009094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Rymut N, et al. Resolvin D1 promotes efferocytosis in aging by limiting senescent cell-induced MerTK cleavage. FASEB J. 2020;34:597–609. doi: 10.1096/fj.201902126R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Baker DJ, et al. Clearance of p16 Ink4a-positive senescent cells delays ageing-associated disorders. Nature. 2011;479:232–236. doi: 10.1038/nature10600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Martin JA, Brown T, Heiner A, Buckwalter JA. Post-traumatic osteoarthritis: the role of accelerated chondrocyte senescence. Biorheology. 2004;41:479–491. [PubMed] [Google Scholar]
- 116.Dalli J, Colas R, Shinohara M, Serhan CN. Aging delays resolution of acute inflammation in mice: reprogramming the host response with novel nano-proresolving. Medicines. 2014;193:4235–4244. doi: 10.4049/jimmunol.1401313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100:57–70. doi: 10.1016/S0092-8674(00)81683-9. [DOI] [PubMed] [Google Scholar]
- 118.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 119.Pollard JW. Tumour-educated macrophages promote tumour progression and metastasis. Nat. Rev. Cancer. 2004;4:71–78. doi: 10.1038/nrc1256. [DOI] [PubMed] [Google Scholar]
- 120.Murdoch C, Muthana M, Coffelt SB, Lewis CE. The role of myeloid cells in the promotion of tumour angiogenesis. Nat. Rev. Cancer. 2008;8:618–631. doi: 10.1038/nrc2444. [DOI] [PubMed] [Google Scholar]
- 121.Sica A, Schioppa T, Mantovani A, Allavena P. Tumour-associated macrophages are a distinct M2 polarised population promoting tumour progression: potential targets of anti-cancer therapy. Eur. J. Cancer. 2006;42:717–727. doi: 10.1016/j.ejca.2006.01.003. [DOI] [PubMed] [Google Scholar]
- 122.Zhang Q, et al. TIM-4 promotes the growth of non-small-cell lung cancer in a RGD motif-dependent manner. Br. J. Cancer. 2015;113:1484–1492. doi: 10.1038/bjc.2015.323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Tan X, Zhang Z, Yao H, Shen L. Tim-4 promotes the growth of colorectal cancer by activating angiogenesis and recruiting tumor-associated macrophages via the PI3K/AKT/mTOR signaling pathway. Cancer Lett. 2018;436:119–128. doi: 10.1016/j.canlet.2018.08.012. [DOI] [PubMed] [Google Scholar]
- 124.Tesi R. MDSC; the most important cell you have never heard of. Trends Pharmacol. Sci. 2019;40:4–7. doi: 10.1016/j.tips.2018.10.008. [DOI] [PubMed] [Google Scholar]
- 125.Holtzhausen A, et al. TAM family receptor kinase inhibition reverses MDSC-mediated suppression and augments anti-PD-1 therapy in melanoma. Cancer Immunol. Res. 2019;7:1672. doi: 10.1158/2326-6066.CIR-19-0008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Cook RS, et al. MerTK inhibition in tumor leukocytes decreases tumor growth and metastasis. J. Clin. Invest. 2013;123:3231–3242. doi: 10.1172/JCI67655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Graham DK, DeRyckere D, Davies KD, Earp HS. The TAM family: phosphatidylserine-sensing receptor tyrosine kinases gone awry in cancer. Nat. Rev. Cancer. 2014;14:769–785. doi: 10.1038/nrc3847. [DOI] [PubMed] [Google Scholar]
- 128.Kumar S, Calianese D, Birge RB. Efferocytosis of dying cells differentially modulate immunological outcomes in tumor microenvironment. Immunol. Rev. 2017;280:149–164. doi: 10.1111/imr.12587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Vaught DB, Stanford JC, Cook RS. Efferocytosis creates a tumor microenvironment supportive of tumor survival and metastasis. Cancer Cell Microenviron. 2015;2:e666. doi: 10.14800/ccm.666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Holland SJ, et al. R428, a selective small molecule inhibitor of Axl kinase, blocks tumor spread and prolongs survival in models of metastatic breast cancer. Cancer Res. 2010;70:1544–1554. doi: 10.1158/0008-5472.CAN-09-2997. [DOI] [PubMed] [Google Scholar]
- 131.Wilson C, et al. AXL inhibition sensitizes mesenchymal cancer cells to antimitotic drugs. Cancer Res. 2014;74:5878–5890. doi: 10.1158/0008-5472.CAN-14-1009. [DOI] [PubMed] [Google Scholar]
- 132.Post SM, et al. AXL/MERTK inhibitor ONO-7475 potently synergizes with venetoclax and overcomes venetoclax resistance to kill FLT3-ITD acute myeloid leukemia. Haematologica. 2020 doi: 10.3324/haematol.2021.278369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Minson KA, et al. The MERTK/FLT3 inhibitor MRX-2843 overcomes resistance-conferring FLT3 mutations in acute myeloid leukemia. JCI Insight. 2016;1:e85630. doi: 10.1172/jci.insight.85630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Subbiah, V. et al. Trials in progress: A phase 1, open-label, dose-escalation, pharmacokinetic, safety and tolerability study of the selective TAM kinase inhibitor PF-07265807 in patients with advanced or metastatic solid tumors [abstract TPS2671]. J. Clin. Oncol.10.1200/JCO.2021.39.15_suppl.TPS2671 (2021).
- 135.Zeidan AM, et al. The STIMULUS Program: clinical trials evaluating sabatolimab (MBG453) combination therapy in patients (Pts) with higher-risk myelodysplastic syndromes (HR-MDS) or acute mMyeloid leukemia (AML) Blood. 2020;136:45–46. doi: 10.1182/blood-2020-134718. [DOI] [Google Scholar]
- 136.Brunner AM, et al. Efficacy and safety of sabatolimab (MBG453) in combination with hypomethylating agents (HMAs) in patients (Pts) with very high/high-risk myelodysplastic syndrome (vHR/HR-MDS) and acute myeloid leukemia (AML): final analysis from a phase Ib study. Blood. 2021;138:244. doi: 10.1182/blood-2021-146039. [DOI] [Google Scholar]
- 137.Brunner AM, et al. Efficacy and safety of sabatolimab (MBG453) in combination with hypomethylating agents (HMAs) in patients with acute myeloid leukemia (AML) and high-risk myelodysplastic syndrome (HR-MDS): updated results from a phase 1b study. Blood. 2020;136:1–2. doi: 10.1182/blood-2020-136855. [DOI] [Google Scholar]
- 138.Harding JJ, et al. Blocking TIM-3 in treatment-refractory advanced solid tumors: a phase Ia/b study of LY3321367 with or without an anti-PD-L1 antibody. Clin. Cancer Res. 2021;27:2168–2178. doi: 10.1158/1078-0432.CCR-20-4405. [DOI] [PubMed] [Google Scholar]
- 139.Majeti R, et al. CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells. Cell. 2009;138:286–299. doi: 10.1016/j.cell.2009.05.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Jaiswal S, et al. CD47 is upregulated on circulating hematopoietic stem cells and leukemia cells to avoid phagocytosis. Cell. 2009;138:271–285. doi: 10.1016/j.cell.2009.05.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Tseng D, et al. Anti-CD47 antibody-mediated phagocytosis of cancer by macrophages primes an effective antitumor T-cell response. Proc. Natl Acad. Sci. USA. 2013;110:11103–11108. doi: 10.1073/pnas.1305569110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Chao MP, et al. Anti-CD47 antibody synergizes with rituximab to promote phagocytosis and eradicate non-Hodgkin lymphoma. Cell. 2010;142:699–713. doi: 10.1016/j.cell.2010.07.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Weiskopf K, et al. Engineered SIRPα variants as immunotherapeutic adjuvants to anticancer antibodies. Science. 2013;341:88–91. doi: 10.1126/science.1238856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Veillette A, Chen J. SIRPα–CD47 immune checkpoint blockade in anticancer therapy. Trends Immunol. 2018;39:173–184. doi: 10.1016/j.it.2017.12.005. [DOI] [PubMed] [Google Scholar]
- 145.Gordon SR, et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature. 2017;545:495–499. doi: 10.1038/nature22396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Burris III, H. A. et al. A first-in-human study of AO-176, a highly differentiated anti-CD47 antibody, in patients with advanced solid tumors [abstract 2516]. J. Clin. Oncol.39, no. 15_suppl 2516–2516 (2021).
- 147.Roohullah, A. et al. First-in-human phase 1 dose escalation study of HX009, a novel recombinant humanized anti-PD-1 and CD47 bispecific antibody, in patients with advanced malignancies [abstract 2517]. J. Clin. Oncol.39, no. 15_suppl 2517–2517 (2021).
- 148.Gan, H. K. et al. Safety of AK117, an anti-CD47 monoclonal antibody, in patients with advanced or metastatic solid tumors in a phase I study [abstract 2630]. J. Clin. Oncol.39, no. 15_suppl 2630–2630 (2021).
- 149.Lakhani N, et al. A phase 1 study of ALX148, a CD47 blocker, alone and in combination with established anti-cancer antibodies in patients with advanced malignancy and non Hodgkin lymphoma. Breast. 2018;1:1. [Google Scholar]
- 150.Haddad F, Daver N. Targeting CD47/SIRPα in acute myeloid leukemia and myelodysplastic syndrome: preclinical and clinical developments of magrolimab. J. Immunother. Precis. Oncol. 2021;4:67–71. doi: 10.36401/JIPO-21-X2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Barkal AA, et al. Engagement of MHC class I by the inhibitory receptor LILRB1 suppresses macrophages and is a target of cancer immunotherapy. Nat. Immunol. 2018;19:76–84. doi: 10.1038/s41590-017-0004-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Galluzzi L, Buqué A, Kepp O, Zitvogel L, Kroemer G. Immunogenic cell death in cancer and infectious disease. Nat. Rev. Immunol. 2017;17:97. doi: 10.1038/nri.2016.107. [DOI] [PubMed] [Google Scholar]
- 153.Kroemer G, Galluzzi L, Kepp O, Zitvogel L. Immunogenic cell death in cancer therapy. Annu. Rev. Immunol. 2013;31:51–72. doi: 10.1146/annurev-immunol-032712-100008. [DOI] [PubMed] [Google Scholar]
- 154.Zhou J, et al. Immunogenic cell death in cancer therapy: present and emerging inducers. J. Cell. Mol. Med. 2019;23:4854–4865. doi: 10.1111/jcmm.14356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Perry JSA, et al. Interpreting an apoptotic corpse as anti-inflammatory involves a chloride sensing pathway. Nat. Cell Biol. 2019;21:1532–1543. doi: 10.1038/s41556-019-0431-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Behar SM, Briken V. Apoptosis inhibition by intracellular bacteria and its consequence on host immunity. Curr. Opin. Immunol. 2019;60:103–110. doi: 10.1016/j.coi.2019.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Tzelepis F, et al. Annexin1 regulates DC efferocytosis and cross-presentation during Mycobacterium tuberculosis infection. J. Clin. Invest. 2015;125:752–768. doi: 10.1172/JCI77014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Codo AC, et al. Inhibition of inflammasome activation by a clinical strain of Klebsiella pneumoniae impairs efferocytosis and leads to bacterial dissemination. Cell Death Dis. 2018;9:1–14. doi: 10.1038/s41419-018-1214-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Hashimoto Y, Moki T, Takizawa T, Shiratsuchi A, Nakanishi Y. Evidence for phagocytosis of influenza virus-infected, apoptotic cells by neutrophils and macrophages in mice. J. Immunol. 2007;178:2448–2457. doi: 10.4049/jimmunol.178.4.2448. [DOI] [PubMed] [Google Scholar]
- 160.Lim K, et al. In situ neutrophil efferocytosis shapes T cell immunity to influenza infection. Nat. Immunol. 2020;21:1046–1057. doi: 10.1038/s41590-020-0746-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Douek DC, et al. HIV preferentially infects HIV-specific CD4+ T cells. Nature. 2002;417:95–98. doi: 10.1038/417095a. [DOI] [PubMed] [Google Scholar]
- 162.Doitsh G, et al. Cell death by pyroptosis drives CD4 T-cell depletion in HIV-1 infection. Nature. 2014;505:509–514. doi: 10.1038/nature12940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Terai C, Kornbluth R, Pauza CD, Richman DD, Carson DA. Apoptosis as a mechanism of cell death in cultured T lymphoblasts acutely infected with HIV-1. J. Clin. Invest. 1991;87:1710–1715. doi: 10.1172/JCI115188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Chua BA, et al. Protein S and Gas6 induce efferocytosis of HIV-1-infected cells. Virology. 2018;515:176–190. doi: 10.1016/j.virol.2017.12.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Larsson M, et al. Activation of HIV-1 specific CD4 and CD8 T cells by human dendritic cells: roles for cross-presentation and non-infectious HIV-1 virus. Aids. 2002;16:1319–1329. doi: 10.1097/00002030-200207050-00003. [DOI] [PubMed] [Google Scholar]
- 166.Czuczman MA, et al. Listeria monocytogenes exploits efferocytosis to promote cell-to-cell spread. Nature. 2014;509:230–234. doi: 10.1038/nature13168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Dallenga T, et al. M. tuberculosis-induced necrosis of infected neutrophils promotes bacterial growth following phagocytosis by macrophages. Cell Host Microbe. 2017;22:519–530.e3. doi: 10.1016/j.chom.2017.09.003. [DOI] [PubMed] [Google Scholar]
- 168.Ribeiro-Gomes F, et al. Apoptotic cell clearance of Leishmania major-infected neutrophils by dendritic cells inhibits CD8+ T-cell priming in vitro by Mer tyrosine kinase-dependent signaling. Cell Death Dis. 2015;6:e2018–e2018. doi: 10.1038/cddis.2015.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.van Zandbergen G, et al. Cutting edge: neutrophil granulocyte serves as a vector for Leishmania entry into macrophages. J. Immunol. 2004;173:6521–6525. doi: 10.4049/jimmunol.173.11.6521. [DOI] [PubMed] [Google Scholar]
- 170.Freire-de-Lima CG, et al. Uptake of apoptotic cells drives the growth of a pathogenic trypanosome in macrophages. Nature. 2000;403:199–203. doi: 10.1038/35003208. [DOI] [PubMed] [Google Scholar]
- 171.Sims B, et al. Role of TIM-4 in exosome-dependent entry of HIV-1 into human immune cells. Int. J. Nanomed. 2017;12:4823. doi: 10.2147/IJN.S132762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Brunton B, et al. TIM-1 serves as a receptor for Ebola virus in vivo, enhancing viremia and pathogenesis. PLoS Negl. Trop. Dis. 2019;13:e0006983. doi: 10.1371/journal.pntd.0006983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Kondratowicz AS, et al. T-cell immunoglobulin and mucin domain 1 (TIM-1) is a receptor for Zaire Ebolavirus and Lake Victoria Marburgvirus. Proc. Natl Acad. Sci. USA. 2011;108:8426–8431. doi: 10.1073/pnas.1019030108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Meertens L, et al. The TIM and TAM families of phosphatidylserine receptors mediate dengue virus entry. Cell Host Microbe. 2012;12:544–557. doi: 10.1016/j.chom.2012.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Evans JP, Liu S-L. Multifaceted roles of TIM-family proteins in virus–host interactions. Trends Microbiol. 2020;28:224–235. doi: 10.1016/j.tim.2019.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Morizono K, et al. The soluble serum protein Gas6 bridges virion envelope phosphatidylserine to the TAM receptor tyrosine kinase Axl to mediate viral entry. Cell Host Microbe. 2011;9:286–298. doi: 10.1016/j.chom.2011.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Song D-H, et al. Development of a blocker of the universal phosphatidylserine- and phosphatidylethanolamine-dependent viral entry pathways. Virology. 2021;560:17–33. doi: 10.1016/j.virol.2021.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Li M, et al. TIM-family proteins inhibit HIV-1 release. Proc. Natl Acad. Sci. USA. 2014;111:E3699–E3707. doi: 10.1073/pnas.1404851111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Das S, et al. ELMO1 has an essential role in the internalization of Salmonella Typhimurium into enteric macrophages that impacts disease outcome. Cell. Mol. Gastroenterol. Hepatol. 2015;1:311–324. doi: 10.1016/j.jcmgh.2015.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Sarkar A, et al. ELMO1 regulates autophagy induction and bacterial clearance during enteric infection. J. Infect. Dis. 2017;216:1655–1666. doi: 10.1093/infdis/jix528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Billings EA, et al. The adhesion GPCR BAI1 mediates macrophage ROS production and microbicidal activity against Gram-negative bacteria. Sci. Signal. 2016;9:ra14. doi: 10.1126/scisignal.aac6250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Tamgue O, et al. Differential targeting of c-Maf, Bach-1, and Elmo-1 by microRNA-143 and microRNA-365 promotes the intracellular growth of Mycobacterium tuberculosis in alternatively IL-4/IL-13 activated macrophages. Front. Immunol. 2019;10:421. doi: 10.3389/fimmu.2019.00421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Baumann I, et al. Impaired uptake of apoptotic cells into tingible body macrophages in germinal centers of patients with systemic lupus erythematosus. Arthritis Rheum. 2002;46:191–201. doi: 10.1002/1529-0131(200201)46:1<191::AID-ART10027>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 184.Thorp E, et al. Shedding of the Mer tyrosine kinase receptor is mediated by ADAM17 protein through a pathway involving reactive oxygen species, protein kinase Cδ, and p38 mitogen-activated protein kinase (MAPK) J. Biol. Chem. 2011;286:33335–33344. doi: 10.1074/jbc.M111.263020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Orme JJ, et al. Heightened cleavage of Axl receptor tyrosine kinase by ADAM metalloproteases may contribute to disease pathogenesis in SLE. Clin. Immunol. 2016;169:58–68. doi: 10.1016/j.clim.2016.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Ballantine L, et al. Increased soluble phagocytic receptors sMer, sTyro3 and sAxl and reduced phagocytosis in juvenile-onset systemic lupus erythematosus. Pediatr. Rheumatol. 2015;13:1–11. doi: 10.1186/s12969-015-0007-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Roumenina LT, et al. Functional complement C1q abnormality leads to impaired immune complexes and apoptotic cell clearance. J. Immunol. 2011;187:4369–4373. doi: 10.4049/jimmunol.1101749. [DOI] [PubMed] [Google Scholar]
- 188.Sullivan K, Petri M, Schmeckpeper B, McLean R, Winkelstein J. Prevalence of a mutation causing C2 deficiency in systemic lupus erythematosus. J. Rheumatol. 1994;21:1128–1133. [PubMed] [Google Scholar]
- 189.Jorge AM, et al. SCARF1-induced efferocytosis plays an immunomodulatory role in humans, and autoantibodies targeting SCARF1 are produced in patients with systemic lupus erythematosus. J. Immunol. 2021;208:955–967. doi: 10.4049/jimmunol.2100532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Hartl J, et al. Autoantibody-mediated impairment of DNASE1L3 activity in sporadic systemic lupus erythematosus. J. Exp. Med. 2021;218:e20201138. doi: 10.1084/jem.20201138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Kawano M, Nagata S. Lupus-like autoimmune disease caused by a lack of Xkr8, a caspase-dependent phospholipid scramblase. Proc. Natl Acad. Sci. USA. 2018;115:2132–2137. doi: 10.1073/pnas.1720732115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Noelia A, et al. Apoptotic cells promote their own clearance and immune tolerance through activation of the nuclear receptor LXR. Immunity. 2009;31:245–258. doi: 10.1016/j.immuni.2009.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Zhao W, et al. The peroxisome proliferator-activated receptor γ agonist pioglitazone improves cardiometabolic risk and renal inflammation in murine lupus. J. Immunol. 2009;183:2729–2740. doi: 10.4049/jimmunol.0804341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Lee YY, et al. Long-acting nanoparticulate DNase-1 for effective suppression of SARS-CoV-2-mediated neutrophil activities and cytokine storm. Biomaterials. 2021;267:120389. doi: 10.1016/j.biomaterials.2020.120389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Zhen Y, Lee IJ, Finkelman FD, Shao W-H. Targeted inhibition of Axl receptor tyrosine kinase ameliorates anti-GBM-induced lupus-like nephritis. J. Autoimmun. 2018;93:37–44. doi: 10.1016/j.jaut.2018.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Navarini L, et al. Role of the specialized proresolving mediator resolvin D1 in systemic lupus erythematosus: preliminary results. J. Immunol. Res. 2018 doi: 10.1155/2018/5264195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Khanna S, et al. Macrophage dysfunction impairs resolution of inflammation in the wounds of diabetic mice. PLoS ONE. 2010;5:e9539. doi: 10.1371/journal.pone.0009539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Li S, et al. Defective phagocytosis of apoptotic cells by macrophages in atherosclerotic lesions of ob/ob mice and reversal by a fish oil diet. Circ. Res. 2009;105:1072–1082. doi: 10.1161/CIRCRESAHA.109.199570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Babu SS, et al. microRNA-126 overexpression rescues diabetes-induced impairment in efferocytosis of apoptotic cardiomyocytes. Sci. Rep. 2016;6:1–12. doi: 10.1038/srep36207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Luo B, Wang Z, Zhang Z, Shen Z, Zhang Z. The deficiency of macrophage erythropoietin signaling contributes to delayed acute inflammation resolution in diet-induced obese mice. Biochim. Biophys. Acta. 2019;1865:339–349. doi: 10.1016/j.bbadis.2018.10.005. [DOI] [PubMed] [Google Scholar]
- 201.Luo B, et al. Erythropoeitin signaling in macrophages promotes dying cell clearance and immune tolerance. Immunity. 2016;44:287–302. doi: 10.1016/j.immuni.2016.01.002. [DOI] [PubMed] [Google Scholar]
- 202.Zhou F, et al. Targeted delivery of microRNA-126 to vascular endothelial cells via REDV peptide modified PEG-trimethyl chitosan. Biomater. Sci. 2016;4:849–856. doi: 10.1039/C5BM00629E. [DOI] [PubMed] [Google Scholar]
- 203.Martin CJ, et al. Efferocytosis is an innate antibacterial mechanism. Cell Host Microbe. 2012;12:289–300. doi: 10.1016/j.chom.2012.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Harrison SA, et al. Safety, tolerability, and biologic activity of AXA1125 and AXA1957 in subjects with nonalcoholic fatty liver disease. ACG. 2021;10:14309. doi: 10.14309/ajg.0000000000001375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Phapale P. Pharmaco-metabolomics opportunities in drug development and clinical research. Anal. Sci. Adv. 2021;2:611–616. doi: 10.1002/ansa.202000178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Bohan D, et al. Targeting the receptor AXL by bemcentinib prevents SARS-CoV-2 infection. Top. Antivir. Med. 2021;29:137–138. [Google Scholar]
- 207.Bohan D, et al. Phosphatidylserine receptors enhance SARS-CoV-2 infection. PLoS Pathog. 2021;17:e1009743. doi: 10.1371/journal.ppat.1009743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Vouri M, An Q, Birt M, Pilkington GJ, Hafizi S. Small molecule inhibition of Axl receptor tyrosine kinase potently suppresses multiple malignant properties of glioma cells. Oncotarget. 2015;6:16183. doi: 10.18632/oncotarget.3952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Chen F, Song Q, Yu Q. Axl inhibitor R428 induces apoptosis of cancer cells by blocking lysosomal acidification and recycling independent of Axl inhibition. Am. J. Cancer Res. 2018;8:1466. [PMC free article] [PubMed] [Google Scholar]
- 210.Woo SM, et al. Axl inhibitor R428 enhances TRAIL-mediated apoptosis through downregulation of c-FLIP and survivin expression in renal carcinoma. Int. J. Mol. Sci. 2019;20:3253. doi: 10.3390/ijms20133253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Felip, E. et al. A phase II study of bemcentinib (BGB324), a first-in-class highly selective AXL inhibitor, with pembrolizumab in pts with advanced NSCLC: OS for stage I and preliminary stage II efficacy [abstract 9098]. J. Clin. Oncol.37, no. 15_suppl 2630–2630 (2019).
- 212.Mita M, et al. Phase 1B study of amuvatinib in combination with five standard cancer therapies in adults with advanced solid tumors. Cancer Chemother. Pharmacol. 2014;74:195–204. doi: 10.1007/s00280-014-2481-1. [DOI] [PubMed] [Google Scholar]
- 213.Tibes R, et al. A phase I, first-in-human dose-escalation study of amuvatinib, a multi-targeted tyrosine kinase inhibitor, in patients with advanced solid tumors. Cancer Chemother. Pharmacol. 2013;71:463–471. doi: 10.1007/s00280-012-2019-3. [DOI] [PubMed] [Google Scholar]
- 214.Asiedu MK, et al. AXL induces epithelial-to-mesenchymal transition and regulates the function of breast cancer stem cells. Oncogene. 2014;33:1316–1324. doi: 10.1038/onc.2013.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Aveic S, et al. TP-0903 inhibits neuroblastoma cell growth and enhances the sensitivity to conventional chemotherapy. Eur. J. Pharmacol. 2018;818:435–448. doi: 10.1016/j.ejphar.2017.11.016. [DOI] [PubMed] [Google Scholar]
- 216.Sarantopoulos, J. et al. A phase 1a / 1b first-in-human, open-label, dose-escalation, safety, pharmacokinetic, and pharmacodynamic study of oral TP-0903, a potent inhibitor of AXL kinase, administered daily for 21 days to patients with advanced solid tumors [abstract TPS2612]. J. Clin. Oncol.10.1200/JCO.2018.36.15_suppl.TPS2612 (2018).
- 217.Jeon JY, et al. TP-0903 is active in models of drug-resistant acute myeloid leukemia. JCI Insight. 2020;5:e140169. doi: 10.1172/jci.insight.140169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Patel V, Keating MJ, Wierda WG, Gandhi V. Preclinical combination of TP-0903, an AXL inhibitor and B-PAC-1, a procaspase-activating compound with ibrutinib in chronic lymphocytic leukemia. Leuk. Lymphoma. 2016;57:1494–1497. doi: 10.3109/10428194.2015.1102243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Huelse JM, Fridlyand DM, Earp S, DeRyckere D, Graham DK. MERTK in cancer therapy: targeting the receptor tyrosine kinase in tumor cells and the immune system. Pharmacol. Ther. 2020;213:107577. doi: 10.1016/j.pharmthera.2020.107577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Ghazi NG, et al. Treatment of retinitis pigmentosa due to MERTK mutations by ocular subretinal injection of adeno-associated virus gene vector: results of a phase I trial. Hum. Genet. 2016;135:327–343. doi: 10.1007/s00439-016-1637-y. [DOI] [PubMed] [Google Scholar]
- 221.Conlon TJ, et al. Preclinical potency and safety studies of an AAV2-mediated gene therapy vector for the treatment of MERTK associated retinitis pigmentosa. Hum. Gene. Ther. Clin. Dev. 2013;24:23–28. doi: 10.1089/humc.2013.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Alexander JJ, Hauswirth WW. Adeno-associated viral vectors and the retina. Adv. Exp. Med. Biol. 2008;613:121–128. doi: 10.1007/978-0-387-74904-4_13. [DOI] [PubMed] [Google Scholar]
- 223.Fisher, G. A. et al. A phase Ib/II study of the anti-CD47 antibody magrolimab with cetuximab in solid tumor and colorectal cancer patients [abstract 114]. J. Clin. Oncol. 38, no. 4_suppl 114–114 (2020).
- 224.Tay MZ, Poh CM, Rénia L, MacAry PA, Ng LF. The trinity of COVID-19: immunity, inflammation and intervention. Nat. Rev. Immunol. 2020;20:363–374. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Giamarellos-Bourboulis EJ, et al. Complex immune dysregulation in COVID-19 patients with severe respiratory failure. Cell Host Microbe. 2020;27:992–1000.e3. doi: 10.1016/j.chom.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Schulte-Schrepping J, et al. Severe COVID-19 is marked by a dysregulated myeloid cell compartment. Cell. 2020;182:1419–1440.e23. doi: 10.1016/j.cell.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Lagunas‐Rangel FA. Neutrophil-to-lymphocyte ratio and lymphocyte-to-C-reactive protein ratio in patients with severe coronavirus disease 2019 (COVID-19): a meta-analysis. J. Med. Virol. 2020;92:1733–1734. doi: 10.1002/jmv.25819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Grégoire M, et al. Impaired efferocytosis and neutrophil extracellular trap clearance by macrophages in ARDS. Eur. Respir. J. 2018;52:1702590. doi: 10.1183/13993003.02590-2017. [DOI] [PubMed] [Google Scholar]
- 229.Middleton EA, et al. Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood. 2020;136:1169–1179. doi: 10.1182/blood.2020007008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.dos-Santos D, et al. Efferocytosis of SARS-CoV-2-infected dying cells impairs macrophage anti-inflammatory programming and continual clearance of apoptotic cells. medRxiv. 2021 doi: 10.1101/2021.02.18.21251504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Braga L, et al. Drugs that inhibit TMEM16 proteins block SARS-CoV-2 Spike-induced syncytia. Nature. 2021;594:88–93. doi: 10.1038/s41586-021-03491-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Dutta S, Mukherjee A, Nongthomba U. Before the “cytokine storm”: boosting efferocytosis as an effective strategy against SARS-CoV-2 infection and associated complications. Cytokine Growth Factor Rev. 2022;63:108–118. doi: 10.1016/j.cytogfr.2022.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]