Abstract
Three antifungal compounds, designated xanthobaccins A, B, and C, were isolated from the culture fluid of Stenotrophomonas sp. strain SB-K88, a rhizobacterium of sugar beet that suppresses damping-off disease. Production of xanthobaccin A in culture media was compared with the disease suppression activities of strain SB-K88 and less suppressive strains that were obtained by subculturing. Strain SB-K88 was applied to sugar beet seeds, and production of xanthobaccin A in the rhizosphere of seedlings was confirmed by using a test tube culture system under hydroponic culture conditions; 3 μg of xanthobaccin A was detected in the rhizosphere on a per-plant basis. Direct application of purified xanthobaccin A to seeds suppressed damping-off disease in soil naturally infested by Pythium spp. We suggest that xanthobaccin A produced by strain SB-K88 plays a key role in suppression of sugar beet damping-off disease.
Damping-off disease in sugar beet seedlings is caused by a number of soilborne plant pathogens, including Rhizoctonia solani, Pythium ultimum, and Aphanomyces cochlioides. It is one of the most serious diseases affecting sugar beet production in Japan. Biological control with antagonistic microorganisms has been investigated as a possible means of controlling the disease (17, 19, 20, 23, 25). Homma et al. (10) reported that when applied as a seed coating, a rhizobacterium of sugar beet, strain SB-K88, suppressed seedling damping-off disease almost as effectively as conventional chemical control. Based on certain bacterial characteristics (lack of an umbonate colony form, no growth on a medium containing 4% sodium chloride and 0.1% triphenyl tetrazolium chloride), strain SB-K88 was considered a member of a Stenotrophomonas species (formerly classified as a Xanthomonas species) but not Stenotrophomonas maltophilia (8). It became apparent in the course of investigating the mechanism of disease suppression by strain SB-K88 that this bacterium produces some antifungal substances that are effective in vitro against fungi that cause damping-off disease (18). Because antibiotics are thought to play a major role in disease suppression by rhizobacteria (14, 21), we focused on the relationship between the production of antifungal substances in the culture medium and in the rhizosphere of sugar beet by this strain and the suppression of damping-off disease.
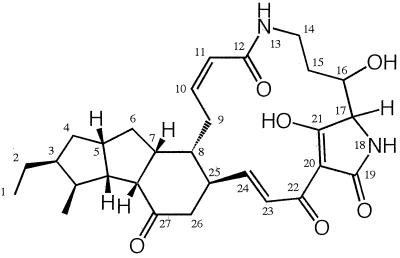
We first isolated three antifungal compounds, xanthobaccin A (XB-A), XB-B, and XB-C, from the culture fluid of strain SB-K88. The chemical structure of XB-A was reported recently and is shown in Fig. 1 (6). This compound is a macrocyclic lactam that has a unique 5,5,6-tricyclic skeleton (tricyclo[7.3.0.02,7]dodecane) and a tetramic acid moiety as a putative chromophore; the plane structure is the same as that of maltophilin, which is produced by S. maltophilia (12). In order to clarify the role of xanthobaccins in disease suppression, we investigated the relationship between the production of XB-A (the major antifungal compound) in vitro and the degree of disease suppression by using less suppressive (LS) strains derived from strain SB-K88. In this paper we also describe the detection and quantification of XB-A produced by strain SB-K88 in the rhizosphere of sugar beet seedlings by using a test tube culture system and disease suppression by XB-A applied directly to sugar beet seeds.
FIG. 1.
Chemical structure of XB-A (6).
MATERIALS AND METHODS
Strain SB-K88.
Stenotrophomonas sp. strain SB-K88 was isolated from the root of sugar beet (10) and was stored in sterilized dispersion medium (10% skim milk, 1% sodium glutamate) at −86°C until it was used.
LS strains.
The antibiosis of strain SB-K88 against R. solani and P. ultimum on potato dextrose agar (PDA) weakened spontaneously during repeated subculturing (5 to 10 subcultures) on 0.2× King’s B medium. The LS strains used in this study were isolated by dilution plating from the resulting subculture. A total of 144 randomly selected, single-colony strains were screened based on their ability to suppress growth of R. solani and P. ultimum on PDA, and 11 (7.6%) of the isolates, which exhibited apparently weaker activity than the wild-type strain, were selected for further study; 5 of these 11 isolates (3.4%) were then selected because they exhibited decreased suppression of damping-off disease in soil naturally infested by Pythium spp. compared to the parent strain (the testing method used is described below). These five LS isolates were designated strains SB-K88-a3, SB-K88-a56, SB-K88-a82, SB-K88-a108, and SB-K88-a120. All of the LS strains exhibited the same natural resistance to kanamycin, streptomycin, spectinomycin, and cycloheximide (each at 50 ppm in King’s B medium) as strain SB-K88. Their ability to grow on sugar beet seedlings was investigated by checking the change in the population of each strain; each strain was applied to sugar beet seeds, and the seeds were sown and cultivated for 14 days in a test tube amended with resin-sand substrate as described below. The population of each strain in the spermosphere or rhizosphere was determined by grinding either seeds or seedling roots with the adhered substrate, followed by dilution plating onto selective medium.
Monitoring chromatographic fractions and antifungal activities.
During purification of the xanthobaccins, each fraction was characterized by thin-layer chromatography (TLC), which was performed on Silica Gel 60 HPTLC plates (Merck) by using chloroform (CHCl3)-methanol (MeOH)-H2O (65:25:4) as the mobile phase. The fraction components were visualized by illuminating the plates with UV light (254 nm) or by spraying them with an orcinol-sulfuric acid reagent and then heating them at 120°C for 5 min. A paper disk method (see below) in which R. solani and P. ultimum were used as indicator fungi was used to check the antifungal activity of each fraction after every chromatographic step.
Culture conditions and purification of antifungal compounds.
Stenotrophomonas sp. strain SB-K88 was cultured in a 500-ml flask containing 100 ml of potato semisynthetic (PS) medium (15) at 25°C for 7 days with shaking at 100 rpm. The culture fluid was centrifuged at 10,000 × g for 30 min, and the supernatant was collected and passed through an Amberlite XAD-2 resin column (Rohm and Haas Co., Ltd.) conditioned with H2O. After the column was rinsed with H2O, the adsorbates were eluted with MeOH and then concentrated under a vacuum.
Samples (5.5 g) extracted from the XAD-2 resin were loaded onto a silica gel column (Wakogel C-200; 50 by 280 mm; Wako Pure Chemical Industries, Ltd.) and fractionated by stepwise gradient elution with CHCl3-MeOH. Fractions (2.7 g) eluted with 20 to 40% MeOH in CHCl3 were collected and rechromatographed with a reverse-phase silica gel column (Cosmosil 75C18-OPN; 35 by 90 mm; Nacalai Tesque, Inc.) by using a stepwise H2O-MeOH gradient. The active fractions were obtained by elution with 80 to 100% aqueous MeOH. Fractions eluted with 90 to 100% MeOH (1.2 g) were subsequently chromatographed with a silica gel column (Wakogel FC-40; 40 by 110 mm) by using a stepwise gradient of dichloromethane (CH2Cl2)-MeOH containing H2O (3%, vol/vol). Fractions eluted with moistened CH2Cl2-MeOH (1:1) were collected and concentrated, and the residue obtained (514 mg) was further fractionated by silica gel column chromatography (Wakogel FC-40; 18 by 200 mm) by using CHCl3-MeOH-H2O as the mobile phase. After the charged column was washed with 350 ml of CHCl3-MeOH-H2O (85:10:1), fractions were eluted with stepwise gradients of CHCl3-MeOH-H2O (85:10:1, 150 ml), CHCl3-MeOH-H2O (83:13:1, 500 ml), CHCl3-MeOH-H2O (80:15:2, 400 ml), CHCl3-MeOH-H2O (77:18:2, 500 ml), and CHCl3-MeOH-H2O (75:20:2, 200 ml) and finally with MeOH (500 ml) and then were collected in order to obtain solute (457 mg). Preparative TLC in which Silica Gel 60 F254 TLC plates (thickness, 0.25 or 0.5 mm; Merck) were developed with CHCl3-MeOH-H2O (65:25:4) was used for the final purification step. Pure XB-A (Rf, 0.49; 133 mg), XB-B (Rf, 0.43; 35 mg), and XB-C (Rf, 0.67; 3 mg) were obtained as amorphous powders.
Antifungal and antibacterial assays.
The antimicrobial activities of the purified xanthobaccins against 11 fungi (A. cochlioides, Phytophthora vignae f. sp. adzukicola, P. ultimum, Gaeumannomyces graminis var. tritici, R. solani AG-4, Botrytis cinerea, Pyricularia oryzae, Verticillium dahliae, Cercospora beticola, Fusarium oxysporum f. sp. lycopersici, Pyrenochaeta lycopersici), one yeast (Saccharomyces cerevisiae), one actinomycete (Streptomyces scabies), and three bacteria (Pseudomonas fluorescens, Escherichia coli, Bacillus subtilis) were tested. The results are presented in Table 1. The fungi, yeast, and actinomycete were grown on PDA, while nutrient agar was used for B. subtilis and Pseudomonas agar F (Difco) was used for P. fluorescens. Assays involving R. solani, P. ultimum, A. cochlioides, P. vignae, G. graminis, and P. oryzae were performed by using a cork borer to cut mycelial disks (diameter, 5 mm) from the margins of mycelial mats grown in petri dishes (diameter, 9 mm) on potato sucrose agar (PSA) for 7 to 10 days. The disks were then inoculated onto the centers of new PSA plates (10 ml; diameter, 90 mm). Heat-sterilized paper disks (diameter, 8 mm) were charged with 25 μl of an XB-A or XB-B solution (0.8 mg/ml in CHCl3-MeOH [1:1, vol/vol]), and the solvent was evaporated under a vacuum. The paper disks charged with test compounds were then placed around the inocula at a distance of 30 mm, and the plates were incubated at 25°C for 2 to 7 days.
TABLE 1.
Antimicrobial activities of xanthobaccins
| Microorganism | Activitya
|
||
|---|---|---|---|
| XB-A | XB-B | XB-C | |
| Aphanomyces cochlioides | +++ (10)b | +++ (10) | ++ |
| Phytophthora vignae f. sp. adzukicola | +++ (0.1) | +++ (0.1) | +++ |
| Pythium ultimum | +++ (10) | ++ (10) | ++ |
| Gaeumannomyces graminis var. tritici | +++ (1.0) | +++ (1.0) | +++ |
| Rhizoctonia solani AG-4 | ++ (>20)c | ++ (>20)c | + |
| Botrytis cinerea | ++ (>20)c | ++ (>20)c | NTd |
| Pyricularia oryzae | +++ (>20)c | +++ (>20)c | ++ |
| Verticillium dahliae | +++ (10) | ++ (10) | + |
| Cercospora beticola | ++ (20) | + (20) | NT |
| Fusarium oxysporum f. sp. lycopersici | + (10) | + (10) | NT |
| Pyrenochaeta lycopersici | − (>20)c | − (>20)c | NT |
| Saccharomyces cerevisiae | + (>20)c | − (>20)c | NT |
| Streptomyces scabies | − | − | NT |
| Bacillus subtilis | − | − | − |
| Pseudomonas fluorescens | − | − | − |
Evaluated on the basis of the width of the inhibitory zone formed around a paper disk at a concentration of 20 μg/disk, as follows: +++, >10 mm; ++, 5 to ∼10 mm; +, ∼5 mm; −, no inhibition.
The value in parentheses is the MIC (in micrograms per milliliter of agar medium).
Test microorganisms grew even at the highest concentration tested (20 μg/ml).
NT, not tested.
A 20-μg portion of xanthobaccin loaded onto a paper disk was deemed to be the appropriate amount for evaluating antimicrobial activities against each test microorganism, because the inhibitory zones for fungi causing damping-off disease were clearly apparent when this amount was used. A disk charged with xanthobaccin-free solvent was used as a control. Spores or cells of the other test microorganisms were suspended in sterilized H2O and mixed with 10 ml of molten agar at a temperature below 50°C; the mixture was then poured into petri dishes (diameter, 90 mm) and cooled. The final concentrations of spores or cells were not determined. The paper disks loaded with 20-μg portions of a test compound were placed onto these agar plates, and the plates were incubated at 25°C for 2 to 7 days. The average width of the inhibitory zones formed around the disks was used to evaluate antimicrobial activity. Two replicates were prepared for each test microorganism-xanthobaccin combination, and the results were averaged. The MIC of XB-A and XB-B were determined by thoroughly mixing specific amounts of XB-A or XB-B dissolved in 50 μl of CHCl3-MeOH (1:1, vol/vol) with 10-ml portions of PDA in order to obtain final concentrations of XB-A or XB-B of 0.01, 0.05, 0.1, 1.0, 10, and 20 μg/ml. Cultures were inoculated with test fungi by using spores obtained from PSA cultures or mycelial disks obtained as described above; they were then incubated at 25°C for 2 to 7 days. Test medium containing xanthobaccin-free solvent was also inoculated as a control. Two agar plates were used for each test fungus-xanthobaccin concentration combination, and the hyphal growth from the spores or mycelial disks of test fungi was examined. The lowest concentration at which no growth was observed was considered the MIC.
Damping-off disease suppression test. (i) Test for bacterial strains.
Strain SB-K88 or LS strains were cultured in 500-ml flasks containing 100 ml of King’s B liquid medium at 27°C for 3 days with shaking at 88 rpm with a reciprocal shaker. The bacterial cells were collected by centrifugation (10,000 × g, 15 min), and each pellet was washed three times with H2O. Water (0.4 ml) and sugar beet (Beta vulgaris cv. Monoace S) seeds (1 g) were then added to the pellet obtained from 100 ml of culture, and the preparation was mixed thoroughly. The treated seeds were spread on a petri dish and air dried overnight at room temperature. The amount of bacteria on the seed surfaces was approximately 108 CFU/seed.
A honeycomb-shaped paper pot (paper pot no. 1; Nippon Beet Sugar Manufacturing Co., Ltd.), which was an aggregate of hexagonal paper tubes (diameter, 19 mm) glued to each other, was filled with field soil naturally infested mainly by Pythium spp. (obtained from the Hokkaido National Agriculture Experiment Station, Sapporo, Japan). For each treatment we used blocks of paper pots (6 × 19 pots); 30 seeds coated with bacterial cells were sown in the inner 2 × 15 pots, which left nonsown pots on the outside. Four replicates were prepared for each treatment. The blocks of pots were placed in a greenhouse and watered every day. The numbers of emerged and damped-off seedlings were recorded for 3 weeks after planting. Damping-off disease severity was evaluated by determining the average damping-off index (D) for four replicates, which was calculated by using following equation: D={[(N−N0)×R×1+(N1×0.5)]÷N}×100, where N is the number of sown seeds, N0 is the number of emerged seedlings, N1 is the number of damped-off seedlings after emergence, and R is the germination ratio of sugar beet seeds. The damping-off index ranged from 0 for no damping-off to 100 for no emerged seedlings.
As a control, nontreated seeds were sown in the same way (Table 2). Seeds were also chemically treated by applying 250 ml of hymexazol diluted with water 103-fold (0.001× hymexazol) to paper pots in which untreated seeds were sown (Table 2).
TABLE 2.
Comparison of suppression of damping-off disease, in vitro production of XB-A, and presence of Stenotrophomonas sp. strain SB-K88 and LS strains in the rhizosphere
| Treatmenta | Suppression of damping-off diseaseb | In vitro production of XB-A (mg/liter of culture fluid)c | Concn in rhizosphere (108 CFU/seed or seedling)d
|
|
|---|---|---|---|---|
| Zero time | Day 14 | |||
| Chemicale | 12.1 ± 6.0 af | |||
| SB-K88 | 16.4 ± 5.2 a | 18.6 | 10.3 | 2.1 |
| SB-K88-a56 | 29.3 ± 7.3 bcde | 4.9 | 4.7 | 1.9 |
| SB-K88-a3 | 30.6 ± 5.3 cdef | 4.7 | 1.9 | 3.4 |
| SB-K88-a120 | 32.8 ± 7.6 def | 5.3 | 0.7 | 0.5 |
| SB-K88-a82 | 35.8 ± 6.7 ef | 4.1 | 16.7 | 4.4 |
| SB-K88-a108 | 42.7 ± 14.2 fg | 5.0 | 0.6 | 1.0 |
| Control | 50.0 ± 11.0 g | |||
Thirty seeds coated with each bacterial strain (ca. 108 CFU/seed) were sown in a paper pot containing field soil that was naturally infested by Pythium spp. The pots were incubated in a greenhouse for 3 weeks.
Suppression was represented by the damping-off index (D), which was calculated as follows: D=[(N−N0)×R×1+(N1×0.5)]×N−1×100, where N is the number of seeds sown, N0 is the number of seeds that emerge, N1 is the number of damped-off seedlings after emergence, and R is the germination ratio. The values are means ± standard deviations based on five replicates.
Each strain was cultured in PS liquid medium. The culture fluid was sampled after 177 h, and the amount of XB-A was determined by HPLC.
Five seeds coated with each bacterial strain were sown in a test tube containing resin-sand substrate and were cultivated for 14 days. Each bacterial population was evaluated by the dilution plate method before sowing (zero time) and on day 14.
The fungicide hymexazol (0.001×) was added as a drench (3 liters/m2).
Values followed by the same letter(s) did not differ significantly, as determined by Duncan’s multiple-range test (P < 0.05).
(ii) Test for purified XB-A.
Sixty seeds of sugar beet (B. vulgaris cv. Monoace S) were soaked in 5-ml solutions of XB-A in CHCl3-MeOH (1:1, vol/vol) at concentrations of 6.25, 1.25, and 0.625 μg/ml for 30 s. The seeds were then spread on a petri dish and air dried overnight at room temperature. The amount of XB-A applied to the seeds was checked by high-performance liquid chromatography (HPLC) as described below. Ten XB-A-treated seeds were sown in each of five flowerpots (diameter, 10 cm) filled with approximately 300 ml of the soil used for bacterial testing (see above). As controls, untreated seeds and seeds treated with XB-A-free solvent were also sown in the same manner. A chemical treatment was also performed by applying a 25-ml solution of 0.001× hymexazol to pots in which untreated seeds were sown. The pots were placed in a growth chamber (24°C; 12 h of light and 12 h of darkness per day) and watered every day. The numbers of emerged and damped-off seedlings were recorded for 3 weeks after planting. Damping-off disease severity was evaluated by determining the damping-off index, and the results of five replicates of each treatment were averaged.
Quantification of XB-A. (i) General HPLC conditions.
An HPLC analysis was performed by using a Hitachi model L-6320 intelligent pump, a Wakosil-II 5C18HG column (4.6 by 250 mm; Wako Pure Chemical), a model L-4250 UV-visible light detector, and a model D-2500 chromatointegrator. A 20-μl sample was injected and eluted with tetrahydrofuran–25 mM KH2PO4 (40:60, vol/vol) at a flow rate of 0.8 ml/min. The eluates were monitored with a UV detector at 320 nm, and the peak areas were calculated by the integrator. XB-A was quantified by using a standard curve previously prepared with pure XB-A and 2-methoxynaphthalene (2-MN). The UV spectra of the constituents in rhizosphere samples were obtained under the same analytical conditions by using a Waters model 996 photodiode array detector and a model 600E HPLC pump supported by Millennium 2010J chromatography manager software (Waters).
(ii) Sample preparation for culture fluid.
Strain SB-K88 or LS strains were cultured in 500-ml flasks containing 100 ml of PS medium (15) with shaking at 25°C and 100 rpm for 7 days. One milliliter of culture fluid was passed through a Sep-Pak Plus C18 cartridge (Waters) conditioned with H2O. After the charged cartridge was washed with 5 ml of H2O and then sequentially with 5 ml of 50% aqueous MeOH, the adsorbates were eluted with 5 ml of MeOH. The eluate was dried under a vacuum and dissolved in 100 μl of MeOH containing 12.5 μg of 2-MN as the internal standard.
(iii) Quantification of XB-A in rhizosphere extract.
The amount of XB-A produced in the rhizosphere was determined by using a modified test tube culture system that was originally developed for studies of the inoculation and multiplication of Polymyxa betae and beet necrotic yellow vein virus (1). A test tube (24 by 120 mm) equipped with a drain was filled with approximately 40 ml of a mixture of Amberlite XAD-2 resin and quartz sand (1:1, vol/vol) and conditioned with H2O. Five sugar beet (B. vulgaris cv. Monoace S) seeds coated with cells of strain SB-K88 (approximately 108 CFU/seed) were sown in each tube and cultivated at either 15 or 22°C (14 h of light and 10 h of darkness per day) with daily watering. Untreated seeds were sown as a control. After 5, 7, 10, 15, 20, and 30 days, the portions of resin-sand substrate from five test tubes that received the same treatment were combined, and a mesh was used to separate the seedlings. The substrate mixture was washed with H2O, and the adsorbates were then eluted with 500 ml of MeOH. The eluate was concentrated under a vacuum to remove the MeOH. The residual aqueous solution (approximately 10 ml) was passed through a Sep-Pak Plus C18 cartridge and then treated in the same way as the culture fluid when the HPLC sample was prepared. Five test tubes comprised one replicate, and the estimated amounts of XB-A in two replicates were averaged.
(iv) Quantification of XB-A applied to seeds.
Seven seeds treated with XB-A (see above) were combined and rinsed three times with 1 ml of a CHCl3-MeOH solution (1:1, vol/vol) in order to recover the XB-A. The rinsing solutions were combined, filtered with a 0.45-μm-pore-size cellulose acetate filter cartridge (Toyo Roshi Kaisha), and evaporated to dryness. The residue was redissolved in 100 μl of MeOH containing 12.5 μg of 2-MN, and the XB-A recovered was quantified by HPLC. The estimated amounts of XB-A obtained from two replicates (seven seeds constituted one replicate) were averaged.
RESULTS
Purification of the antifungal compounds.
The poor solubility of the antifungal compounds in organic solvents, such as ethyl acetate, meant that attempts to extract them from culture fluids with such solvents were not successful. On the other hand, solid-phase extraction with Amberlite XAD-2 resin by using MeOH as the eluant did result in extraction of the active compounds from culture fluids. Reversed-phase silica gel column chromatography following rough fractionation by the first silica gel column chromatography step allowed us to remove a yellow fluorescent component that overlapped and interfered with the detection of XB-A by TLC. Further purification was attained by silica gel chromatography in which CHCl3-MeOH-H2O was used as the main solvent system. All three xanthobaccins emitted a characteristic whitish blue fluorescence on TLC plates under illumination with UV light at 365 nm, which was a useful indicator during fractionation. Consequently, 133 mg of XB-A, 35 mg of XB-B, and 3 mg of XB-C were obtained from 15 liters of culture fluid.
Antimicrobial activities of xanthobaccins.
The results of antimicrobial tests in which xanthobaccins were tested with various plant pathogens are summarized in Table 1. The xanthobaccins inhibited the growth of three representative pathogens that cause sugar beet damping-off disease, A. cochlioides, P. ultimum, and R. solani, although growth inhibition of R. solani was rather weak compared to growth inhibition of the other two organisms. The xanthobaccins also inhibited the growth of seven other plant pathogens, and P. vignae exhibited the greatest sensitivity. No inhibition of the growth of an actinomycete or of bacteria was observed. Although the growth of P. ultimum or A. cochlioides was not completely inhibited on agar containing 1.0 μg of XB-A per ml or 1.0 μg of XB-B per ml, the growth of these organisms was markedly suppressed (data not shown). Either XB-A or XB-B at a concentration of 10 μg/ml completely inhibited the growth of both P. ultimun and A. cochlioides, whereas the growth of R. solani was not completely inhibited by either compound at a concentration of 20 μg/ml. However, serious deformations of mycelia were observed at concentrations greater than 10 μg/ml.
Relationship between production of XB-A and disease suppression activity.
Strain SB-K88 produced up to 18 mg of XB-A per liter in PS liquid cultures, whereas the amounts of XB-A in the culture fluids of five LS strains grown under the same conditions were about one-fourth the amount in the strain SB-K88 culture fluid. These five LS strains exhibited significantly less suppression of damping-off disease. The populations of the LS strains in the rhizosphere were almost equal before and after cultivation in test tubes for 14 days (Table 2).
Production of xanthobaccins in the rhizosphere of sugar beet seedlings.
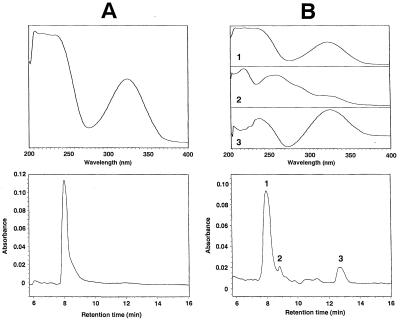
Production of XB-A by strain SB-K88 in the rhizosphere of sugar beet seedlings was investigated by using a test tube culture system. A peak that coincided with the peak of authentic XB-A (retention time, approximately 8.0 min) was detected on the HPLC chromatograms obtained for all of the rhizosphere extracts of bacterium-treated seedlings (Fig. 2B). Although XB-A and XB-B were not separated by HPLC under the analytical conditions used, it seemed that the peak corresponding to XB-A in Fig. 2B consisted mainly of XB-A and contained very small amounts of XB-B, since the retention time and UV spectrum of the XB-A equivalent peak in Fig. 2B were identical to the retention time and UV spectrum of authentic XB-A (Fig. 2A). Consequently, the subsequent XB-A quantitative analyses were conducted by assuming that the peak was composed of XB-A alone.
FIG. 2.
HPLC chromatograms (bottom) and UV spectra (top) of XB-A and rhizosphere extract obtained with a photodiode array detector. (A) Authentic XB-A. (B) Rhizosphere extract of sugar beet seedlings bacterized with Stenotrophomonas sp. strain SB-K88. The eluates were detected at UV 320 nm. The UV spectrum of peak 1 (maximum absorbance at 321 nm) corresponds to the UV spectrum of XB-A. The other two peaks in the chromatogram of the rhizosphere extract were not identified.
Five days after sowing (Table 3), approximately 1.2 and 1.4 μg of XB-A per seedling were isolated from the rhizosphere extracts of the bacterized seedlings grown at 15 and 22°C, respectively. The amount of detectable XB-A at 15°C gradually increased and reached about 3.1 μg/seedling after 30 days, whereas at 22°C the maximum amount was observed after 10 days, after which the amount of XB-A tended to decline. No XB-A was detected in the extracts obtained from the rhizospheres of the nonbacterized seedlings. XB-C (retention time, approximately 20 min under our analytical conditions) was not identified in the rhizosphere extracts of either bacterized or nonbacterized seedlings.
TABLE 3.
Production of XB-A in the rhizospheres of sugar beet seedlings bacterized with Stenotrophomonas sp. strain SB-K88
| Days after sowing | Amt of XB-A in rhizosphere (μg/seedling)a
|
|
|---|---|---|
| 15°C | 22°C | |
| 5 | 1.18 (8.0)b | 1.43 (4.0) |
| 7 | 1.66 (17.5) | NTc |
| 10 | 2.19 (17.5) | 2.05 (11.5) |
| 15 | NT | 1.84 (16.5) |
| 20 | NT | 1.59 (17.5) |
| 30 | 3.05 (17.5) | 1.41 (19.5) |
Five seeds coated with bacterial cells (ca. 108 CFU/seed) were sown in a test tube containing resin-sand substrate and cultivated at 15 or 22°C. Each replicate consisted of five tubes. The amount of XB-A adsorbed by the substrate was determined by HPLC. The results are the averages of two replicates.
The values in parentheses are the average numbers of germinated seeds obtained with two replications.
NT, not tested.
Disease suppression activity of XB-A applied directly to sugar beet seeds.
The amounts of XB-A applied to the seeds used in the test are shown in Table 4. Because the number of emerged seeds was increased by treating seeds with XB-A, the damping-off index decreased significantly compared to no treatment (control) or to the solvent treatment. Thirty micrograms of XB-A applied to a seed suppressed damping-off disease as effectively as the chemical control. However, there was no significant difference between the results obtained for treatments that differed in the amount of XB-A applied.
TABLE 4.
Suppression of damping-off disease by direct seed application of XB-A
| Treatmenta | No. of seedsb | No. of emerged seedlingsc | No. of damped-off seedlingsc | Damping-off indexd |
|---|---|---|---|---|
| Chemicale | 10 | 9.8 ± 0.4 | 0.6 ± 0.9 | 5.0 ± 5.0 af |
| XB-A (30.2 μg/seed)g | 10 | 10.0 ± 0.0 | 1.0 ± 1.0 | 5.0 ± 5.0 a |
| XB-A (2.6 μg/seed) | 10 | 9.2 ± 1.3 | 4.6 ± 4.0 | 30.8 ± 20.6 a |
| XB-A (0.9 μg/seed) | 10 | 8.6 ± 2.1 | 2.8 ± 2.5 | 27.7 ± 26.0 a |
| Solventh | 10 | 5.6 ± 1.7 | 4.2 ± 1.1 | 64.1 ± 15.5 b |
| Control | 10 | 4.2 ± 2.6 | 3.0 ± 1.4 | 71.8 ± 18.7 b |
Ten seeds coated with each bacterial strain (ca. 108 CFU/seed) were sown in a pot containing field soil that was naturally infested by Pythium spp. The pots were incubated in a greenhouse for 3 weeks. Suppressiveness was determined by determining the damping-off index.
Number of seeds per pot.
Values are averages ± standard deviations based on five replications.
See Table 2, footnote b.
The fungicide hymexazol (0.001×) was applied (25 ml/pot).
Values followed by the same letter did not differ significantly, as determined by Duncan’s multiple-range test (P < 0.05).
The amount of XB-A applied was determined by HPLC.
Seeds were treated with XB-A-free solvent.
DISCUSSION
S. maltophilia (Xanthomonas maltophilia) has been reported to be a biocontrol agent for both soilborne diseases (3–5, 16) and foliar diseases (26). However, there have been no previous reports that a bioactive compound that is involved in disease control is actually produced by this species. Kwok et al. (16) reported that strain 76 of X. maltophilia, which suppressed Rhizoctonia-mediated damping-off disease in radish, apparently produced an antifungal substance on King’s B agar. However, the antifungal substance was not identified, and antibiosis was not considered the principal mechanism responsible for disease suppression (16).
During our experiments, it became apparent that Stenotrophomonas sp. strain SB-K88 produced at least three macrocyclic lactam antibiotics, XB-A, XB-B, and XB-C, which have a characteristic 5,5,6-tricyclic skeleton and a tetramic acid chromophore (6). The plane structure of XB-A is the same as that of maltophilin, which is produced by a rhizobacterium of rape, S. maltophilia R3089 (12). The structures of XB-B and XB-C have not been fully elucidated; however, the results of spectroscopic analyses suggest that XB-B has a hydroxyl group substitution at position 27 in the 6-member ring of XB-A and XB-C has a dehydroxylated structure at position 16 of XB-A (data not shown). Elucidation of the structures of XB-B and XB-C will be reported elsewhere. The structures of xanthobaccins and maltophilin are closely related to the structure of ikarugamycin, an antibiotic produced by Streptomyces phaeochromogenes subsp. ikaruganensis (11). Interestingly, the xanthobaccins and maltophilin all exhibited antifungal activity in vitro but no antibacterial activity (Table 1) (12), while ikarugamycin has been reported to exhibit antibacterial activity against gram-positive bacteria (11). The differences in the antimicrobial spectra of these antibiotics may be a consequence of the structural differences in their tricyclic skeletons.
Previous discussions of the role of antibiotics in the biological control of soilborne plant diseases have been based on chemical and microbiological studies (2, 9, 13, 21, 22). In this study, we tried to understand the role of xanthobaccins in damping-off disease suppression by (i) clarifying the relationship between disease suppression and the production of the major antifungal compound XB-A by using strain SB-K88 and LS strains, (ii) detecting and quantifying XB-A in the rhizosphere of bacterized seedlings, and (iii) estimating the ability of XB-A to suppress damping-off disease when it is applied directly to seeds.
Strain SB-K88 and LS strains were considered equally able to grow in the rhizosphere of sugar beet seedlings, while disease suppression and the production of XB-A by the LS strains were found to be reduced compared to disease suppression and the production of XB-A by strain SB-K88 (Table 2). It is well recognized that root colonization by an introduced bacterial agent is an essential event for effective biocontrol (7, 24). Our results suggest that the reduced suppression by LS strains is mainly due to reduced production of XB-A and not to the inability of these organisms to establish themselves on roots. Our results also suggest that XB-A contributes to disease suppression. As far as strain SB-K88 is concerned, there does not appear to be a relationship between antibiotic production and the ability to colonize roots, as previously reported for P. fluorescens and the control of take-all disease in wheat (13, 22).
Applying XB-A directly to seeds remarkably lowered the damping-off index, mainly because of the greater number of emerged seedlings compared to nontreated controls (Table 4). The data indicate that 0.9 μg of XB-A in the rhizosphere (spermosphere) was sufficient to inhibit a pathogen and prevent the occurrence of preemergence damping-off (i.e., the death of a seed or seedling before emergence from the soil as a result of infection by a pathogen). Although detection and quantification of XB-A from rhizosphere soil have not been successful yet, under hydroponic culture conditions strain SB-K88 actually produced more than 1 μg of XB-A, which accumulated in the rhizosphere of one bacterized seed by 5 days after sowing (Table 3). Suppression of preemergence damping-off occurred when either 3 or 30 μg of XB-A was applied to a seed, but suppression after emergence was not satisfactory when only 3 μg was applied. This result implies that the concentration of XB-A in the rhizosphere gradually decreased, either due to diffusion or due to degradation, after the 3-μg treatment. In contrast, the level of XB-A in the rhizosphere of a bacterized seed (seedling) reached 3 μg/seedling on the 10th day, and this level was maintained throughout the experimental period. This indicates that when strain SB-K88 is present on a seed, it produces XB-A continuously, not transiently; maintenance of the XB-A levels is considered important in reducing the incidence of damping-off. The concentration of XB-A may be high enough to inhibit pathogen growth (more than 1 μg/ml for Pythium sp. [Table 1]) in the spermosphere, where the population of strain SB-K88 is considered high compared with other parts of the rhizosphere. This could be deduced from the fact that increased production of XB-A paralleled the growth of strain SB-K88 in culture (18), but further investigation is needed to clarify the relationship between distribution of the bacterium and XB-A levels. It is very possible that XB-A produced in the rhizosphere is able to suppress other fungi that cause damping-off disease, such as R. solani and A. cochlioides, because strain SB-K88 suppressed Rhizoctonia-mediated damping-off disease (8) and xanthobaccins produced by this strain suppressed the growth of both of these fungi (Table 1).
Nutrient competition and induction of plant defense systems (24), as opposed to antibiosis, are other possible mechanisms of disease suppression that remain to be investigated. Nutrient competition mediated by a fluorescent siderophore is unlikely, because strain SB-K88 produces no fluorescent substance on agar medium (10). The effects of XB-A on a host plant when it is produced in the rhizosphere by strain SB-K88 are unclear, but no effect on germination or seedling growth was observed when purified XB-A was applied at a concentration of 30 μg/seed. This finding contrasts with data obtained with the antibiotic 2,4-diacetylphloroglucinol produced by P. fluorescens CHA0, which was toxic to plants at a high concentration (13). We are particularly interested in knowing whether strain SB-K88 produces and excretes extracellular lytic enzymes, because one strain of S. maltophilia excretes such enzymes and they are considered central components in protecting sugar beet from Pythium-mediated damping-off disease (4). The importance of extracellular chitinase in the suppression of Bipolaris leaf spot of tall fescue by S. maltophilia C3 has also been reported (27).
In conclusion, XB-A produced in the rhizosphere by strain SB-K88 is considered to be crucial in suppressing Pythium-mediated damping-off disease in sugar beet, especially in reducing preemergence damping-off. In order to clarify the role of xanthobaccins in disease suppression, further investigation in which mutants that are not capable of producing XB-A are used is needed, in addition to an evaluation of xanthobaccins in actual rhizospheres. Strain SB-K88 has promise as a biocontrol agent against damping-off disease in sugar beet as it produces antifungal compounds; however, a seed-coating formula suitable for practical use has yet to be established. It will also be necessary to evaluate the potential for improved performance through use of this organism in combination with other biocontrol agents that have different suppression mechanisms, as described by Dunne et al. (4).
REFERENCES
- 1.Abe H, Tamada T. A test tube culture system for multiplication of Polymyxa betae and necrotic yellow vein virus in rootlets of sugar beet. Proc Sugar Beet Res Assoc. 1987;29:34–38. [Google Scholar]
- 2.Ahl P, Voisard C, Défago G. Iron-bound siderophores, cyanic acid, and antibiotics involved in suppression of Thielaviopsis basicola by a Pseudomonas fluorescens strain. J Phytopathol. 1986;116:121–134. [Google Scholar]
- 3.Chen W, Hoitink H A J, Schmittenner A F. Factors affecting suppression of Pythium damping-off in container media amended with compost. Phytopathology. 1987;77:755–760. [Google Scholar]
- 4.Dunne C, Moënne-Loccoz Y, McCarthy J, Higgins P, Powell J, Dowling D N, O’Gara F. Combining proteolytic and phloroglucinol-producing bacteria for improved biocontrol of Pythium-mediated damping-off of sugar beet. Plant Pathol. 1998;47:299–307. [Google Scholar]
- 5.Giesler J L, Yuen Y G. Evaluation of Stenotrophomonas maltophilia strain C3 for biocontrol of brown patch disease. Crop Prot. 1998;17:509–513. [Google Scholar]
- 6.Hashidoko Y, Nakayama T, Homma Y, Tahara S. Structure elucidation of xanthobaccin A, a new antibiotic produced from Stenotrophomonas sp. strain SB-K88. Tetrahedron Lett. 1999;40:2957–2960. [Google Scholar]
- 7.Homma Y. Biocontrol trait versus competitive ability as the basis for selection. Am J Altern Agric. 1995;10:61–62. [Google Scholar]
- 8.Homma Y, Katoh K, Uchino H, Kanzawa K. Suppression of sugar beet damping-off by seed bacterization with Stenotrophomonas sp. SB-K88 in a paper-pot system. In: Ogoshi A, Kobayashi K, Homma Y, Kodama F, Kondo N, Akino S, editors. Plant growth-promoting rhizobacteria—present status and future prospects. Sapporo, Japan: The 4th PGPR International Workshop Organizing Committee; 1997. pp. 205–208. [Google Scholar]
- 9.Homma Y, Sato Z, Hirayama F, Konno K, Shirahama H, Suzui T. Production of antibiotics by Pseudomonas cepacia as an agent for biological control of soilborne plant pathogens. Soil Biol Biochem. 1989;21:723–728. [Google Scholar]
- 10.Homma Y, Uchino H, Kanzawa K, Nakayama T, Sayama M. Suppression of sugar beet damping-off and production of antagonistic substances by strains of rhizobacteria. Ann Phytopathol Soc Jpn. 1993;59:282. [Google Scholar]
- 11.Ito S, Hirata Y. The structure of ikarugamycin, an acyltetramic acid antibiotic possessing a unique as-hydrindacene skelton. Bull Chem Soc Jpn. 1977;50:1813–1820. [Google Scholar]
- 12.Jakobi M, Winkelmann G, Kaiser D, Kempter C, Jung G, Berg G, Bahl H. Maltophilin, a new antifungal compound produced by Stenotrophomonas maltophilia R3089. J Antibiot. 1996;49:1101–1104. doi: 10.7164/antibiotics.49.1101. [DOI] [PubMed] [Google Scholar]
- 13.Keel C, Schnider U, Maurhofer M, Vousard C, Laville J, Burger U, Wirthner P, Haas D, Défago G. Suppression of root disease by Pseudomonas fluorescens CHA0: importance of the bacterial secondary metabolite 2,4-diacetylphloroglucinol. Mol Plant Microbe Interact. 1992;5:4–13. [Google Scholar]
- 14.Kerr A. Biological control of crown gall through production of agrocin 84. Plant Dis. 1980;64:25–30. [Google Scholar]
- 15.Kunitake S, Matsuyama N, Wakimoto S. Production of proteinous anti-fungal substance(s) by Pseudomonas avenae Manns. Ann Phytopathol Soc Jpn. 1988;54:640–642. [Google Scholar]
- 16.Kwok O C H, Fahy P C, Hoitink H A J, Kuter G A. Interactions between bacteria and Trichoderma hamatum in suppression of damping-off in bark compost media. Phytopathology. 1987;77:1206–1212. [Google Scholar]
- 17.Lee W H, Ogoshi A. Bacterization of sugar beets. II. Antibiosis to damping-off pathogens and growth stimulation of sugar beets by rhizobacteria. Ann Phytopathol Soc Jpn. 1986;52:175–183. [Google Scholar]
- 18.Nakayama T. Ph.D. thesis. Sapporo, Japan: Hokkaido University; 1996. [Google Scholar]
- 19.Pietro A D, Küng R, Gut-Rella M, Schwinn F J. Parameters influencing the efficacy of Chaetomium globosum in controlling Pythium ultimum damping-off of sugar beet. J Plant Dis Prot. 1991;98:565–573. [Google Scholar]
- 20.Suslow T V, Schloth M N. Rhizobacteria of sugar beet: effects of seed application and root colonization. Phytopathology. 1982;72:199–206. [Google Scholar]
- 21.Thomashow L S, Weller D M. Role of phenazine antibiotic in disease suppression by Pseudomonas fluorescens 2-79. Phytopathology. 1987;77:1724. [Google Scholar]
- 22.Thomashow L S, Weller D M. Role of phenazine antibiotic from Pseudomonas fluorescens in biological control of Gaeumannomyces graminis var. tritici. J Bacteriol. 1988;170:3499–3508. doi: 10.1128/jb.170.8.3499-3508.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Walther D, Gindrat D. Biological control of damping-off of sugar-beet and cotton with Chaetomium globosum or a fluorescent Pseudomonas sp. Can J Microbiol. 1988;34:631–637. [Google Scholar]
- 24.Weller D M. Biological control of soilborne plant pathogens in the rhizosphere with bacteria. Annu Rev Phytopathol. 1988;26:379–407. [Google Scholar]
- 25.Williams G E, Asher M J C. Selection of rhizobacteria for the control of Pythium ultimum and Aphanomyces cochlioides on sugar-beet seedlings. Crop Prot. 1996;15:479–486. [Google Scholar]
- 26.Zhang Z, Yuen G Y. Biological control of Bipolaris leaf spot of tall fescue by Stenotrophomonas maltophilia strain C3. Phytopathology. 1997;87:S108–S109. doi: 10.1094/PHYTO.1999.89.9.817. [DOI] [PubMed] [Google Scholar]
- 27.Zhang Z, Yuen G Y. Chitinolytic properties of Stenotrophomonas maltophilia strain C3, an antagonist of fungal turfgrass pathogens. Phytopathology. 1997;87:S109. [Google Scholar]