Abstract
Treatment with the Toll-like receptor 7 (TLR7) agonist, resiquimod (R848), is effective in various types of cancer, such as breast, pancreatic and colorectal cancer. The reported antitumor effect of R848 in lung cancer is considered to be achieved by targeting macrophages. In the present study, it was demonstrated that TLR7 expression on various immune cell types initially rises, then declines in the late stage of lung cancer. Intraperitoneal injection of R848 resulted in a reduction in tumor burden and prolonged survival in both subcutaneous and metastatic lung cancer models in C57BL/6 mice. Initial treatment with R848 at an early stage was found to be the optimal choice. Systemic injection of R848 promoted the activation of innate and adaptive immune responses. Systemic administration of R848 upregulated TLR7 expression in dendritic cells (DCs) and enhanced the activation of DCs and natural killer (NK) cells. Moreover, this treatment also resulted in increased production of T helper cell-associated cytokines in serum, including IFN-γ, TNF-α and IL-2. In addition, continuous treatment with R848 increased the proportion of DCs, NK and CD8+ T cells, and reduced that of Foxp3+ regulatory T cells in the tumor microenvironment. These findings supported the use of R848 treatment for lung cancer via TLR7 targeting and provided insight into the underlying therapeutic mechanism.
Keywords: resiquimod, Toll-like receptor 7, lung cancer, tumor microenvironment, immune cells, immunity
Introduction
According to a status report on the global burden of cancer, lung cancer is the most commonly diagnosed cancer, representing 11.6% of the total cases, and the leading cause of cancer-related death (1). With recent advances in the field of cancer immunotherapy, various highly effective therapies, particularly immune checkpoint inhibitors (ICIs) have been developed to enhance anti-tumor immunity, which have demonstrated a survival benefit for a subset of patients with lung cancer (2,3). However, despite observable clinical improvement, the majority of patients fail to respond to ICI therapy due to drug resistance, resulting both from tumor cell-intrinsic mechanisms and other factors relating to the tumor microenvironment (TME) (4,5). Thus, new immunotherapeutic approaches are urgently needed for patients with lung cancer.
Toll-like receptor (TLR) agonists are immune adjuvants used in antiviral and anti-bacterial treatment that can promote T helper (Th) 1 immune response (6,7). Among the 13 known TLRs, TLR7 and TLR8, are mainly expressed on the endosomal membranes (8) of cells of the innate immune system, including dendritic cells (DCs), macrophages and natural killer (NK) cells (9). Resiquimod (R848), an antiviral imidazoquinoline derivative, stimulates immune cells through TLR7 and/or TLR8 in a MyD88-dependent manner. Nevertheless, R848 selectively acts on TLR7 in mice (10,11) and has been demonstrated to induce Th1 immune responses following activation of antigen-presenting cells, leading to the production of numerous pro-inflammatory cytokines including IL-12 and IFN-γ. R848 exhibits a robust antitumor effect in several tumor models, such as dermatological malignancies, cutaneous T-cell lymphoma and breast cancer (12-14). Moreover, recent studies focusing on macrophages have highlighted the therapeutic efficacy of R848, either alone or in combination with other drugs, in lung cancer (15,16). However, the mechanism of R848's efficacy in lung cancer is not completely clear.
In the present study, it was initially confirmed that R848 targets TLR7 expression in the immune cells of lung tumor-bearing mice and the effects of the aforementioned compound in subcutaneous (s.c.) and metastatic models of lung cancer were evaluated. The volumes of the s.c. solid tumors, the survival of tumor-bearing mice and tumor metastasis were examined. Furthermore, the ability of R848 to stimulate host immunity in tumor-bearing mice was assessed. It was also determined whether R848 would alter the composition of immune cells in the TME. The present findings may provide insight into the future use of R848-based immunotherapy for lung cancer.
Materials and methods
Mice and cell lines
A total of 151, male, 6-8 weeks-old C57BL/6 mice (~20 g) were purchased from Beijing Tengxin Company and housed in individually ventilated cages. OT1 mice (stock no. 003831) and TLR7 knock-out mice (stock no. 008380) were purchased from the Jackson Laboratory Company and kept in a specific-pathogen-free animal facility (temperature, 22±2 degrees; 12-h light cycle; air changed 16 times/h; humidity, 55±10%) with free access to water and food. Mice were anesthetized by intraperitoneal (i.p.) injection with 1% pentobarbital sodium (50 mg/kg), then sacrificed by cervical dislocation. Death was confirmed by cessation of the heartbeat and breathing. The animal study was approved (approval no. 2022-152) by the ethics committee of The First Affiliated Hospital of Chongqing Medical University (Chongqing, China). The Lewis lung carcinoma (LLC) cell line was provided by Professor Haixia Long (Department of Oncology, Xinqiao Hospital, The Army Medical University), and maintained in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% FBS and 1% penicillin-streptomycin (all from Gibco; Thermo Fisher Scientific, Inc.) in a humidified incubator at 37°C with 5% CO2/95% air. The B16 F10 cell line was provided by Professor Haixia Long and was cultured in RPMI-1640 (Hyclone; Cytiva) supplemented with 10% FBS and 1% penicillin-streptomycin at 37°C.
Reagents and antibodies
For in vivo administration, R848 (MedChemExpress) was dissolved in DMSO and frozen in −80°C. Live/Dead viability kits (cat. no. L34958) and anti-TLR7 antibodies (cat. no. MA5-16249) were purchased from Invitrogen; Thermo Fisher Scientific, Inc. and diluted at 1:200 for use. For flow cytometry, antibodies specific for CD45 (cat. no. 103108), CD3 (cat. no. 100218), CD8 (cat. no. 100714), CD4 (cat. no. 100422), CD11c (cat. no. 100422), CD11b (cat. no. 101230), CD49b (cat. no. 108910), CD19 (cat. no. 115520), Ly6G (cat. no. 127607), Ly6C (cat. no. 128016), CD80 (cat. no. 104705), CD86 (cat. no. 105014), MHC-II (cat. no. 116407), PD-1(cat. no. 135224), FoxP3 (cat. no. 126404), F4/80 (cat. no. 123116) and IFN-γ (cat. no. 505826) were purchased from Biolegend, Inc. and used for staining following the manufacturer's protocol. Fixation/Permeabilization kit (with BD Golgistop™ (cat. no. 554715; BD Biosciences) was used for intracellular staining. The Foxp3/Transcription Factor Staining Buffer Set (cat. no. 00-5523; Invitrogen; Thermo Fisher Scientific, Inc.) was used for intranuclear Foxp3 staining. The anti-asialo GM1 rabbit polyclonal antibody (cat. no. 986-10001) was purchased from FUJIFILM Wako Pure Chemical Corporation. Anti-mouse CD8 (clone 2.43; cat. no. BE0061) and anti-mouse CD4 (clone YTS191; cat. no. BE0119) antibodies were purchased from Bio X Cell and used following the indicated concentrations for cells depletion.
Cell culture
Primary bone marrow-derived DCs isolated from the femur and tibia of C57BL/6 mice were cultured with 10 ng/ml IL-4 and GM-CSF (Novoprotein) at 37°C for 4 days. Immature DCs were then collected and purified with Histodenz (Sigma-Aldrich; Merck KGaA). DC suspensions with a purity >85% with CD11C and MHC-II double-staining were used for subsequent assays. NK cells were positively selected from the spleen (SPL) of C57BL/6 mice using the Mouse CD49b Positive Selection kit (cat. no. 18755, Stemcell Technologies, Inc.). CD8+ T cells were negatively isolated from SPL and lymph nodes (LN) from OT-1 mice according to the manufacturer's protocol (cat. no. 19835A; Stemcell Technologies, Inc.). The isolated CD8+ T cells pre-labeled with 5,6-carboxyfluorescein diacetate succinimidyl ester (eBioscience; Thermo Fisher Scientific, Inc.) were added after DCs co-cultured with NK cells in the presence of R848 for 24 h. Following 3 days of co-culture, IFN-γ+ CD8+ T and CD8+ T proliferation was analyzed by flow cytometry.
Proliferation and apoptosis assay
Proliferation was measured using the Cell Titer 96® AQueous One Solution Cell Proliferation Assay (Promega Corporation). 1×106 LLC cells were seeded in a 100-µl volume in a 96-well plate and stimulated with R848 at 1, 5 or 10 µg/ml for 24 or 48 h. Each well was plated in triplicate. A 20-µl volume of Cell Titer 96® AQueous One Solution Reagent was then added into each well. The plate was then incubated at 37°C for 1-4 h in a humidified 5% CO2 atmosphere. The absorbance at 490 nm was then measured using a 96-well plate reader.
To measure apoptosis, LLC cells were stimulated under the same conditions, then stained using a PE Annexin V Apoptosis Detection kit (cat. no. 559763; BD Biosciences) for 20 min at room temperature (RT) in the dark according to the manufacturer's protocol.
Preparation of single-cell suspensions and flow cytometry
Following sacrifice, tumors, SPL, LN and femur of tumor-bearing mice were isolated. Tumors were immersed in DMEM on ice, then minced and enzymatically digested in DMEM containing 1.0 mg/ml collagenase IV and 50 U/ml DNAse I for 1 h at 37°C. The digested tumors were then ground, and the cells were filtered through 70- and 40-µM cell strainers. The other samples, such as SPL and LN, were collected in cold PBS with 2% FBS, gently ground, then filtered through 70-µM cell strainers. The cells were then centrifuged at 500 × g for 5 min at 4°C, then resuspended and immediately stained for flow cytometry at 4°C for 30 min.
For surface staining, the cells were firstly blocked with CD16/32 antibody (1:200; cat. no. 101320; Biolegend, Inc.) for 20 min at 4°C, then stained with live/dead kits and streaming antibodies for 20 min. For intracellular staining, 1×106 cultured cells were added to GolgiStop™ buffer 4-6 h before staining. Intracellular Foxp3 was conducted using a Foxp3/Transcription Factor Staining Buffer Set according to the manufacturer's protocol. Lastly, the samples were tested using a Beckman Gallios flow cytometer (Beckman Coulter, Inc.) and analyzed with FlowJo version 7.6.1 (FlowJo LLC).
Systemic immune activation
In vivo, 3 mg/kg R848 (17) was intravenously injected into mice with a s.c. tumor on day 7; serum, LN and SPL were collected at 0, 6, 24 and 48 h. TLR7 expression on DCs, macrophages, myeloid-derived suppressor cells (MDSCs), NK cells, CD4+ T and CD8+ T cells, CD80 and CD86 expression on DCs, and CD69 expression on NK cells, CD4+ T cells and CD8+ T cells were analyzed by flow cytometry. Levels of cytokines in the serum of R848-treated mice at 0, 6, 24 and 48 h were determined using a Multiple mouse cytokine detection kit (cat. no. B299045; Biolegend, Inc.) following the manufacturer's protocol.
S.c. lung cancer model and treatment
A total of 1×106 LLC cells in 100 µl PBS were injected subcutaneously into the right flank of C57BL/6 mice. At the same day following inoculation, the mice were injected i.p. with 20 µg R848 every other day till endpoint. The control group received 100 µl PBS. After the formation of palpable tumors, tumor growth was evaluated by measuring the length and width using electronic calipers every 3 days. The results are expressed as volumes, which were calculated according to the following formula: 0.52 × (length) × (width)2. Immune cells in the TME were detected after continuous R848 treatment for 10 times.
Metastatic lung cancer model
A total of 1×106 B16/F10 melanoma cells were cultured in complete RPMI-1640 medium, then injected intravenously into C57BL/6 mice. On day 0 after inoculation, treatment with 20 µg R848 i.p. was initiated and continued every other day till endpoint. In addition, 80 µg R848 in 100 µl PBS therapy was started on day 0 and injected twice with a 4-day interval between the two injections. The numbers of metastatic tumor nodules in the lung were recorded from day 14. The left lung with metastatic tumor nodules was fixed in 4% paraformaldehyde at RT for 24 h, embedded, then sectioned at the maximum cross-section for further analysis. The 5 mm-thick slides were stained with hematoxylin and eosin. The tumor nodules were examined and enumerated under a fluorescence microscope. A total of 1×106 LLC cells in 100 µl PBS were injected intravenously into C57BL/6 mice. On day 21 after inoculation, tumor nodes in the lung were extracted for TLR7 gene quantification.
Depletion experiment
Anti-asialo GM1 rabbit polyclonal antibody (50 µl in 100 µl PBS) was administered by i.p. injection at day -1 every 3 days till the endpoint for NK cell depletion. Anti-CD4 and anti-CD8 in vivo depletion antibodies were used at 250 µg/injection, injected daily for the first 2 days, then twice-weekly until the end of the experiment as previously described (18). It was confirmed that >90% CD8+ T, NK and CD4+ T cells were depleted.
RNA extraction, cDNA synthesis and reverse transcriptionquantitative (RT-q) PCR
Freshly isolated tumors were immediately frozen and ground to powder. Total RNA from LLC powder was isolated using TRIzol® (Invitrogen; Thermo Fisher Scientific, Inc.) according to the manufacturer's protocol. The RNA was reverse-transcribed to cDNA using a Reverse Transcription kit (Promega Corporation) following the manufacturer's protocol. The following primer sequences were used: mouse TLR7 forward, 5′-GGT CCA AAG CCA ATG TGT GT-3′ and reverse, 5′-GGA TGG CAG ATC CTG TGG TA-3′; and β-actin forward, 5′-CTA GGC CAC AGA ATT GAA AGA TCT-3′ and reverse, 5′-GTA GGT GGA AAT TCT AGC ATC ATC C-3′. qPCR was performed on a Bio-Rad Fast RT-PCR system using GoTaq® Green Master Mix (Promega Corporation). The qPCR conditions were as follows: Initial denaturation at 95°C for 15 sec; 40 cycles of 95°C for 10 sec, 60°C annealing for 20 sec and extension at 72°C for 20 sec. All samples were set up in triplicate. The raw cycle threshold (Ct) values obtained for β-actin mRNA were deducted from those of TLR7 to obtain the ΔCt values. Analysis of relative gene expression data was performed using qPCR and the 2−ΔΔCq method (19).
Statistical analysis
Statistical analysis was performed using GraphPad Prism software version 6 (GraphPad Software, Inc.). Comparisons between groups were carried out using unpaired Student's-test. Tumor growth speed was evaluated with Linear regression test. Survival analysis was performed by log-rank test. For all comparisons, P<0.05 was considered to indicate a statistically significant difference.
Results
TLR7 is predominantly expressed in LLC s.c. tumorinfiltrating lymphocytes (TiLs)
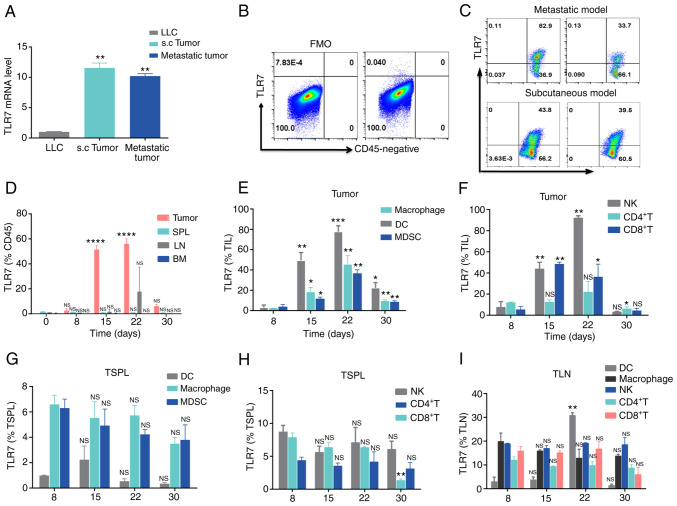
R848 selectively targets TLR7 in mice (20). Therefore, prior to the initiation of R848 treatment for murine lung cancer, TLR7 expression was examined in mice bearing lung tumors. qPCR results suggested that TLR7 mRNA was highly expressed both in LLC metastatic and s.c. tumors, compared with LLC cells (Fig. 1A). Moreover, TLR7 was expressed at high levels on CD45+ leukocytes in both LLC metastatic and s.c. tumors, but not in CD45− cells (Fig. 1B and C). The stimulation of TLR7 mainly located on innate cells initiates adaptive immunity by TLR7 agonists, which lay the basis of R848 for its antiviral and antitumor effect (8,21). Therefore, the dynamic change of TLR7 on leukocytes was investigated in the TME, LN, SPL and bone marrow (BM) from mice with LLC s.c. tumors. The proportion of TLR7+ TiL increased from day 0 to day 22, decreased in the later period from day 22 to day 30 of s.c. lung tumors (Fig. 1D). However, there was no significant difference in the frequency TLR7+ leukocytes in the BM, LN and SPL (Fig. 1D). Next, TLR7 distribution was compared among various immune cells from SPL, LN and tumor tissue samples from LLC s.c. tumor-bearing mice. The results demonstrated that TLR7 on various immune cells was dynamically expressed. Indeed, TLR7 expression increased from day 8 to 22, then declined during days 22-30 (Fig. 1E and F). However, similar to TLR7+ TiLs, TLR7 expression on various immune cells was stable from day 8 to day 30 in LN and SPL (Fig. 1G and H). The upregulation of TLR7+ on immune cells could result from tumor antigen stimulation in the TME. TLR7 expression levels on immune cells, including macrophages, NK, CD4+ T and CD8+ T cells were relatively stable in LN and SPL, with the exception of LN DCs, where TLR7 expression increased gradually from day 22 (Fig. 1I), which we hypothesize may be caused by tumor cell invasion in the LN. These findings on the dynamic patterns of expression of TLR7 may help in the design of R848 therapeutic regimens for lung cancer.
Figure 1.
Dynamic TLR7 expression in immune cells from LLC tumor-bearing mice. (A) TLR7 mRNA abundance in parental LLC, s.c. and metastatic tumors in C57BL/6 mice.**P<0.01 compared with LLC. (B and C) Representative flow cytometry plots of TLR7 expression in (B) CD45− and (C) CD45+ cells from LLC s.c. and metastatic tumors. (D) Histogram representing TLR7+CD45+ cells in bone marrow, SPL, LN and tumor samples at day 0, 8, 15, 22 and 30 in mice with LLC s.c. tumors (n=3). (E and F) TLR7 expression on (E) DCs, macrophages, MDSCs and (F) NK, CD4+ T and CD8+ T cells from LLC s.c. tumors at day 8, 15, 22 and 30 (n=3). (G-I) Dynamic changes in TLR7 expression on various immune cells from the SPL and LN of LLC tumor-bearing mice (n=3). The data are presented as the mean ± SEM. *P<0.05, **P<0.01, ***P<0.001 and ****P<0.0001 compared with day 0. TLR, Toll-like receptor; LLC, Lewis lung carcinoma; s.c., subcutaneous; SPL, spleen; LN, lymph nodes; DCs, dendritic cells; MDSCs, myeloid-derived suppressor cells; NK, natural killer; NS, not significant.
Administration of R848 initially targets DCs and induces systemic immune activation
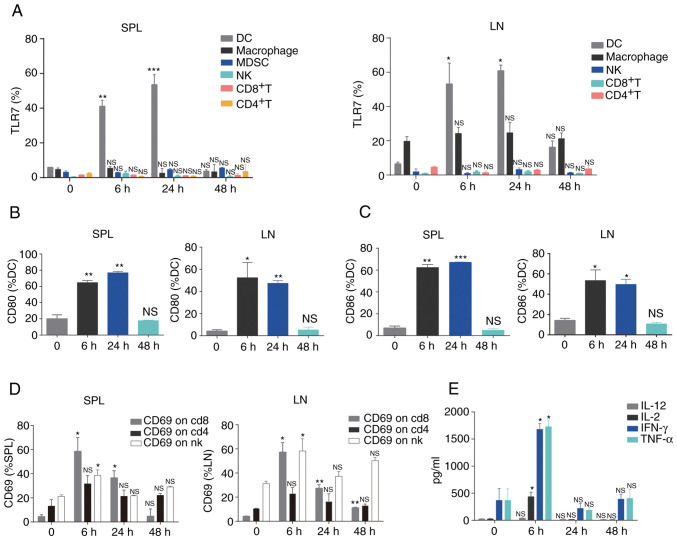
Previous studies have demonstrated that R848 promotes the maturation of DCs, including the upregulation of co-stimulatory molecules and production of IL-12 and enhances adaptive immunity (22,23). It was next sought to determine the ability of R848 to enhance systemic immune activation in the LLC s.c. tumor model. Notably, systemic administration of R848 significantly upregulated TLR7 in DCs, but not in macrophages, MDSCs, NK, CD4+ T and CD8+ T cells from SPL and LN (Fig. 2A). Compared with CD8+ T, CD4+ T, NK and MDSCs, TLR7 expression was relatively higher on macrophages, although no significant difference was observed following R848 injection (Fig. 2A). R848 administration significantly unregulated the co-stimulatory markers CD80 (Fig. 2B) and CD86 (Fig. 2C) of DCs in LN and SPL. TLR7 expression in MDSCs from LN was not evaluated, as very few of these cells were detected. Although no significant changes in TLR7 expression were observed in NK and CD8+ T cells, these cells displayed an activated phenotype (NK cells: CD69; CD8+ T cells: CD69+) in LN and SPL (Fig. 2D). Increased CD69 expression on CD4+ T cells was observed after R848 administration, although this was not statistically significant. Furthermore, increased serum levels of Th1-associated cytokines, including IL-2, IFN-γ and TNF-α (24,25), were observed following R848 injection (Fig. 2E). These findings suggested that R848 initially activates DCs and further induces activation of the immune system.
Figure 2.
Administration of R848 promotes systemic immune activation. (A) TLR7 expression on DCs, macrophages, MDSCs, NK, CD4+ T and CD8+ T cells was analyzed by flow cytometry (n=3). (B) CD80 and (C) CD86 expression on DCs was detected by flow cytometry (n=3). (D) CD69 expression was assessed in NK, CD4+ T and CD8+ T cells (n=3). (E) Levels of cytokines were evaluated in the serum (n=3). *P<0.05, **P<0.01 and ***P<0.001 compared with PBS. TLR, Toll-like receptor; DCs, dendritic cells; MDSCs, myeloid-derived suppressor cells; NK, natural killer; NS, not significant.
R848 inhibits tumor growth and prolongs survival in the s.c. lung cancer model
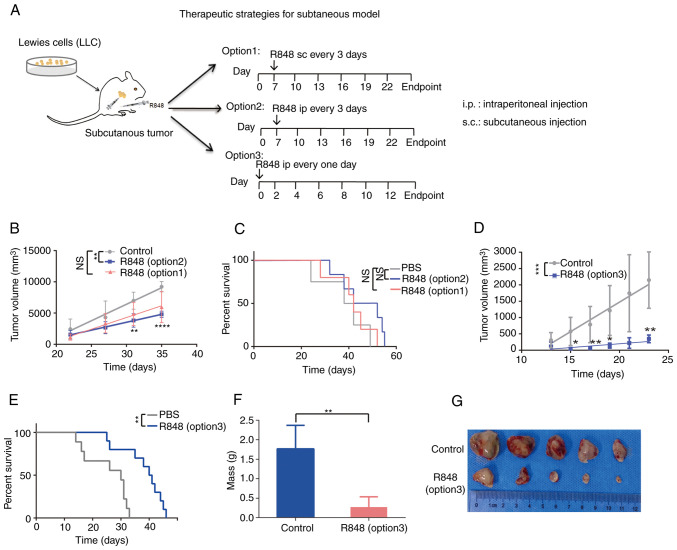
R848 has been used in the treatment of breast cancer, lymphoma and pancreatic ductal adenocarcinoma (17,26). Since TLR7 expression was demonstrated in a s.c. lung cancer model and it was confirmed that R848 could stimulate systemic immunity, it was hypothesized that R848 treatment could act on TLR7+ leukocytes, which would reduce tumor burden in mice. Firstly, 20 µg (27) R848 s.c. (option 1) or i.p. (option 2) administration in lung tumor-bearing mice bearing from day 7, twice a week until the experimental endpoint was reached, and tumors were measured every 3 days once palpable (Fig. 3A). The i.p. or s.c. administration of R848 reduced tumor growth compared with the control group; however, this was statistically significant only in the i.p. group (Fig. 3B). There were no differences in the survival rates of the R848 group (i.p. or s.c.) and the PBS control group (Fig. 3C). Since i.p. administration showed a significant inhibitory effect on tumor growth, whereas s.c. did not, this route of administration was used in subsequent experiments. A more intensive scheme (option 3), in which R848 (20 µg) treatment was initiated at day 0 and repeated every other day (Fig. 3D) also significantly prolonged the survival time (Fig. 3E) and effectively reduced tumor growth (Fig. 3F and G). These results demonstrated that R848 inhibits lung tumor growth and prolongs the survival of LLC tumor-bearing mice.
Figure 3.
R848 inhibits tumor growth and prolongs the survival of mice with s.c. lung tumors. (A) R848 treatment regimen. A total of 1×106 LLC cells were injected s.c. into 6-8 weeks-old C57BL/6 mice. Starting at day 7 post-inoculation, 20 µg R848 in 100 µl PBS was injected s.c. (option 1) or i.p. (option 2) every 3 days until the experimental endpoint. The control received 100 µl PBS. In a separate group, 20 µg R848 injected i.p. at day 0, then every other day (option 3). (B) Tumor growth curves were generated for the R848 (20 µg i.p. or s.c.) and the control groups. (C) The survival of tumor-bearing mice in the R848 (20 µg, i.p. or s.c.) and control groups was demonstrated. (D) Tumor growth curves were generated for the R848 (option 3, 20 µg, i.p.) and control groups. (E) Kaplan-Meier curves of the survival of tumor-bearing mice in the R848 (option 3, 20 µg, i.p.) and control groups. (F and G) Tumor weight (F) and picture of tumors (G) were demonstrated in the R848 (option 3, 20 µg, i.p.) and control groups (n=8-10). *P<0.05, **P<0.01, ***P<0.001 and ****P<0.0001 compared with the control group. R848, resiquimod; s.c., subcutaneous; LLC, Lewis lung carcinoma; i.p., intraperitoneal.
R848 prevents melanoma cell metastasis to the lung
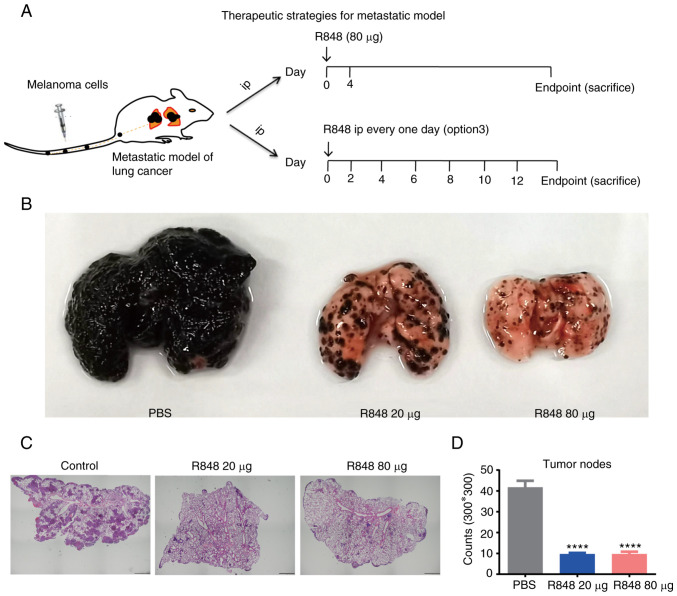
To evaluate whether R848 can prevent metastasis to the lung, the murine B16 melanoma metastasis model was used. A total of 1×106 B16 F10 cells were injected via the tail vein into C57BL/6 mice, and R848 therapy was initiated from day 0. As a high dose of R848 (80 µg) may induce toxicity when applied every other day, 20 µg R848 (i.p.) was administered every other day (option 3) until the experimental endpoint or 80 µg (i.p.) R848 twice with a 4-day interval (Fig. 4A). The results demonstrated that either 20 or 80 µg R848 treatment effectively inhibited the metastasis of melanoma cells to the lung (Fig. 4B-D). During the experiment, B16 F10 metastatic nodes were not observed on the liver, chest wall, or abdominal cavity without checking of brain or bones.
Figure 4.
R848 inhibits lung metastasis of melanoma tumors. (A) R848 treatment regimen. R848 was administrated at 20 µg every other day, or at 80 µg twice, with a 4-day interval between the two doses. n=6-8 mice/group. (B) Representative images of metastatic tumor nodules in the left lung of R848-treated and control mice. (C) Representative H&E staining images at ×40 magnification of left lung sections. (D) Histogram showing the average numbers of tumor nodules within a 300×300 mm area. ****P<0.0001, compared with control group. R848, resiquimod.
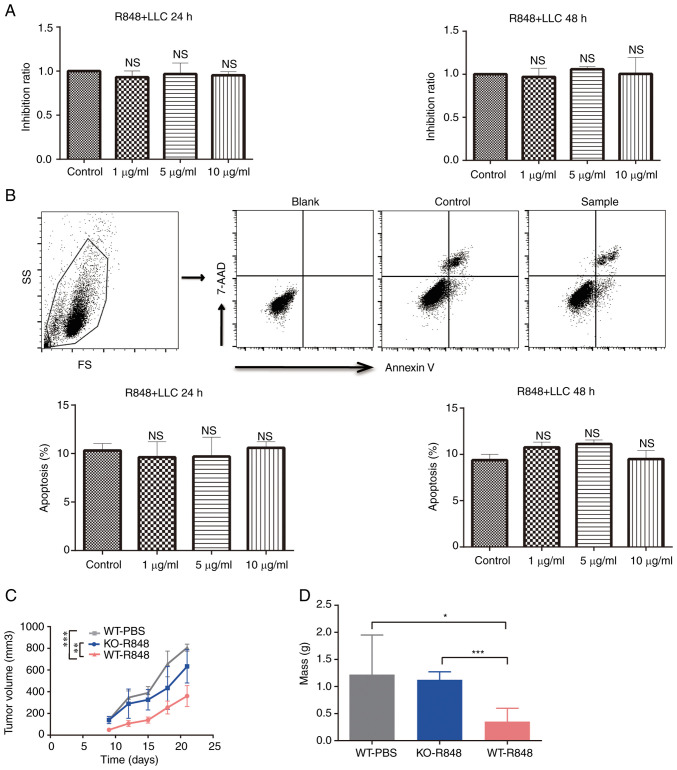
R848 exerts no direct effect on the proliferation and apoptosis of LLC cells
To investigate the mechanism underlying the antitumor effects of R848, LLC cells were treated with R848 in vitro at different concentrations. Cell proliferation was not affected by R848 treatment (Fig. 5A). Similarly, no significant effect was observed on LLC cell apoptosis (Fig. 5B). Moreover, R848 inhibited tumor burden in wild-type but not TLR7 knock-out (TLR7-KO) mice (Fig. 5C). Indeed, the tumor mass and volume at the endpoint confirmed that R848 did not significantly inhibit the growth of s.c. LLC tumors in TLR7-KO mice (Fig. 5D). These results indicated that R848 served an antitumor role that is dependent on TLR7 expression.
Figure 5.
R848 does not affect the proliferation and apoptosis of LLC cells. (A and B) LLC cells were incubated in presence of R848 with concentrations ranging from 1 to 10 µg/ml. (A) The proliferation and (B) apoptosis of LLC cells were evaluated. The total rate of apoptosis includes early and late apoptosis. (C) Growth curves of LLC s.c. tumors implanted in TLR7-KO and WT mice treated with R848 or PBS (n=8-10). (D) Tumor mass of s.c. tumors implanted in TLR7-KO and WT mice treated with R848 or PBS (n=8-10). *P<0.05, **P<0.01 and ***P<0.001 compared with the control group. R848, resiquimod; LLC, Lewis lung carcinoma; s.c., subcutaneous; TLR, Toll-like receptor; TLR-KO, TLR knockout; WT, wild-type; NS, not significant.
R848 modulates the TME
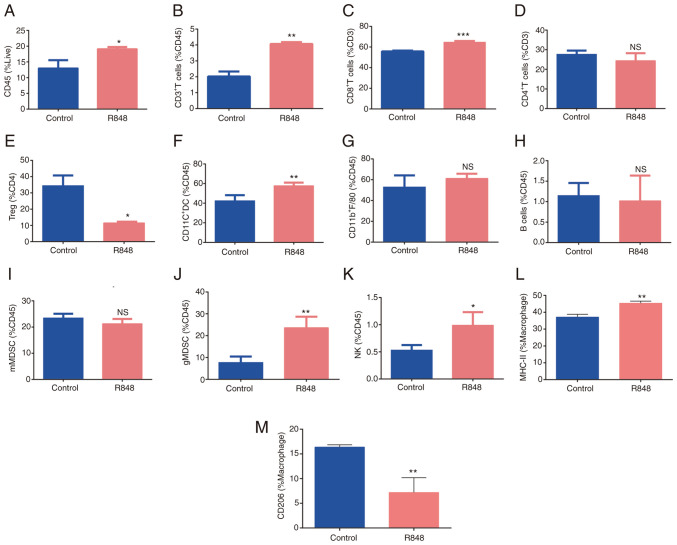
Since R848 did not affect LLC cells directly, but induced the activation of DCs, CD8+ T cells and NK cells, it was sought to determine whether R848 could modulate the TME, as another underlying mechanism for its antitumor effect. Tumor-infiltrating immune cell populations were detected in R848- and PBS-treated s.c. LLC tumors. The gating strategy is shown in Fig. S1. The total frequency of CD45+ leukocytes was increased in R848-treated tumors compared with the controls (Fig. 6A; 19.1±0.3480 vs. 13±1.8% of live cells; P<0.05). CD3+ T cell (4.06±0.068 vs. 2.02±0.22% of CD45+ cells; P<0.01) and CD8+ T cell (64.5±0.74 vs. 55.8±0.45% of CD8+ T cells; P<0.01) frequency increased following R848 treatment (Fig. 6B and C), while no significant difference was observed in CD4+ T cells (Fig. 6D). The frequency of FoxP3+ regulatory T (Treg) cells decreased following R848 treated (11.5±0.45 vs. 34.36±3.66% of CD4+ T cells; P<0.05) (Fig. 6E). However, a significant downregulation of PD-1 on CD8+ T cells was not observed. The proportion of NK cells significantly increased following R848 treatment (0.98±0.12 vs. 0.53±0.05% of CD45+ cells, P<0.05) (Fig. 6K). No significant difference was observed in the B cell population (Fig. 6H). Macrophages (CD11b+F4/80+) appeared enriched after treatment with R848, although this was not statistically significant (Fig. 6G). However, the R848 treatment promoted M2 polarization towards the M1 subtype, as evidenced by increased MHC-II expression (Fig. 6L) and lower CD206 expression (Fig. 6M). The frequency of tumor-infiltrating DCs (CD11b+CD11C+) significantly increased (57.72±1.45 vs. 42.3±3.33% of CD45+ cells; P<0.01) following treatment with R848 (Fig. 6F). In addition, the frequency of neutrophils (CD11b+Ly6G+Ly6C−, granulocytic -MDSC) increased following treatment with R848 (23.6±2.52 vs. 7.63±1.62% of CD45+ cells; P<0.05) (Fig. 6J). There was a trend towards reduced monocyte frequency (CD11b+Ly6G−Ly6C+, monocytic-MDSC) following R848 treatment, although this was not statistically significant (Fig. 6I). These findings suggested that R848 modulates the distribution of immune cells in the TME of s.c. LLC tumors.
Figure 6.
R848 alters the components of immune cells in tumor microenvironment. (A) Distribution of CD45+ cells, (B) total T cells or CD3+ T cells, (C) CD8+ T cells, (D) CD4+ T cells, (E) Tregs, (F) DCs, (G) macrophages, (H) B cells, (I) mMDSCs, (J) gMDSCs and (K) NK cells in R848- and PBS-treated subcutaneous Lewis lung carcinoma tumors (n=3-5). (L) MHC-II (M1) and (M) CD206 (M2) expression on macrophages (n=3-5). *P<0.05, **P<0.01 and ***P<0.001 compared with the PBS control group. R848, resiquimod; Tregs, T regulatory cells; DCs, dendritic cells; MDSCs, myeloid-derived suppressor cells; NK, natural killer.
NK cells and CD8+T cells takes greatly part in R848-mediated therapeutic efficacy in lung cancer
Based on the activation and alteration of immune cells in host and TME of mice with s.c. LLC tumors following R848 treatment, the key immune cells involved in R848-mediated therapeutic efficacy in murine lung cancer were further explored. It has been documented that R848 enhances antitumor efficacy in murine lymphoma models via NK and CD4+ T cells (17). In the present study, the reduction in tumor burden by R848 was partly abrogated following NK cell depletion (Fig. S2A). On one hand, NK cells have been reported to be important immune cells for cancer immunotherapy due to their cytotoxicity against tumor cells; on the other hand, NK cells have been revealed to help TLR7 agonist-activated DCs to induce CD8+ T cell immunity (28,29). Consistently, R848-stimulated DCs induced CD8+ T cell proliferation and increased IFN-γ+CD8+ T cell frequencies in the presence of NK cells in the present study (Fig. S2B and C). R848 treatment with CD8+ T cell depletion reduced mouse survival and weight, which indicated that CD8+ T cells are likely to be a significant factor in R848-mediated lung cancer resistance (Fig. S2D). However, it was demonstrated in the present that the antitumor role of R848 in lung cancer is negatively associated with CD4+ T cells including tumor volume, mice weight and survival, which is consistent with the low frequency of CD4+ T cells in the TME after R848 treatment (Fig. S2A, D and E). Collectively, these results indicated that NK cells and CD8+ T cells are closely involved in therapeutic effecacy of R848 muring lung cancer.
Discussion
A growing number of immunotherapeutic drugs such as cytokines, tumor vaccines, immune-stimulatory small molecules and checkpoint inhibitors have exhibited considerable benefits in patients with lung cancer (30,31). Several studies have also highlighted the potential benefit of TLR7 agonists as antitumor agents (7,32). In the present study, it was demonstrated that the TLR7 agonist, R848, effectively inhibited murine LLC s.c. tumor progression and metastasis and improved the survival of LLC tumor-bearing mice. The antitumor effect elicited by R848 could be attributed to TLR7-induced immunity, which was characterized by activation of DCs, NK cells and CD8+ T cells in lymphoid organs and immunomodulation of the TME.
TLR7 is a receptor of the innate immune system that recognizes highly conserved molecules expressed by pathogens (33). Upon binding single stranded RNA or other agonists, TLR7 signaling results in the production of pro-inflammatory cytokines, T cell proliferation, and induction of adaptive immunity (34). In the present study, dynamic TLR7 expression was observed in lymphocytes from LLC tumors. In addition, increased frequency of TLR7+ DCs was also observed following R848 injection, which suggests that DCs are the primary target cells of R848 in murine s.c. LLC model. In addition, various immune cells play collaborating roles in antitumor responses to lung cancer. Although the use of R848 for the treatment of lung cancer has already been reported in certain studies (15,35), the present findings provided detailed, novel insight into the dynamics of TLR7 expression in lung tumor-bearing mice. Furthermore, these findings are necessary, as they provide a rationale for the use of R848 for treatment and may guide decisions on therapy duration. Indeed, the findings of the present study suggested that treatment with R848 at the early/mid stage of lung cancer may be more beneficial than at later stages.
Recent studies have reported the use of R848 in combination with chemotherapy or nanoparticles, which had therapeutic benefits in lung cancer (15,36). Consistent with these findings, a reduction in tumor burden and improvements in survival were observed in s.c. LLC model following i.p. injection of R848 in the present study. Furthermore, it was revealed that R848 treatment effectively prevented melanoma cells from metastasizing to the lung. In the present study, a comparison of therapeutic strategies was also made, including route of administration, dose and duration choice. The results indicated that i.p. injection was superior to s.c. administration. Abdominal blood flow is more abundant than in subcutaneous tissue, thus, it was hypothesized that the absorption of R848 i.p. may be more effective than the s.c. route. Since TLR7 was highly expressed in TiLs, R848 was initially used intratumorally. However, frequent injection of R848 directly into the tumor mass reduced natural tumor progression (data not shown). R848 encapsulated with nanoparticles (37) or combined with materials targeting TiLs may be worth exploring in future studies, as the present study provided evidence that TLR7 is highly expressed in TiLs.
An important factor to consider for cancer treatment is the complex immunity in the TME. Immune cells within the TME can have dual roles, either promoting or inhibiting tumor growth. Immune cells that promote tumor progression include MDSCs, Tregs and M2-like macrophages. By contrast, DCs, neutrophils, NK, B and effector T cells in the TME have antitumor effects (38,39). Changes were observed in the distribution of these immune populations within s.c. LLC tumors from mice treated with R848, including increased DCs, NK cells, neutrophils (g-MDSC) and CD8+ T-cells, and decreased Foxp3+ Tregs. These findings provided a rationale for the use of R848 to improve therapeutic efficacy in lung tumors. Although significant changes were not observed in the total frequency of macrophages, the proportion of M2-like macrophages was reduced and that of M1-like macrophages was increased, which is consistent with a previous study demonstrating that R848-loaded nanoparticles target the polarization of tumor-associated macrophages to enhance cancer immunotherapy (40). CD8+ T lymphocytes play a leading role in tumor immunity via their capacity to eliminate malignant cells upon recognition by the T cell receptor of specific antigenic peptides presented on the surface of cancer cells (41). The expression of immune checkpoint molecules, notably PD-1, is associated with poor prognosis and reduced therapeutic effect. Furthermore, CD8+ T cells with reduced PD-1 expression mediated greater antitumor results in vivo (42). Continuous R848 treatment-induced CD8+ T cell infiltration and showed PD-1 downregulation on CD8+ T cells, although this was not statistically significant. These findings, if confirmed, may provide a rationale for the use R848 in combination with PD-1 inhibitors.
The role of TLR expression in cancer cells is not fully understood and has been associated with either good or poor outcomes (43). Previous studies reported that TLR7 activation on immune cells results in therapeutic benefits, whereas TLR7 expression on lung tumor cells promotes tumor progression (44,45). Based on the absence of tumor reduction in TLR7-deficient hosts, it is proposed that the beneficial effects of R848 require a host, rather than tumor TLR7 expression. Future studies investigating the efficacy of R848 on tumors expressing TLR7 are warranted.
Supplementary Data
Acknowledgments
Not applicable.
Funding Statement
The present study was supported by The Fifth funding for young and middle-aged medical high talents of Chongqing (grant no. 2019GDRC028).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
JCZ, GSW and YX conceived and designed the study. JCZ and HY performed the experiments with the guidence of YX. GSW, THM, YX and YLL performed development of writing, review and revision of the manuscript. YLL and THM supervised the study. YLL and THM acquired funding. All authors made contributions to the article. JCZ and YX confirm the authenticity of all the raw data. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The animal study was approved (approval no. 2022-152) by the ethics committee of The First Affiliated Hospital of Chongqing Medical University (Chongqing, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7–33. doi: 10.3322/caac.21654. [DOI] [PubMed] [Google Scholar]
- 3.Chen R, Manochakian R, James L, Azzouqa AG, Shi H, Zhang Y, Zhao Y, Zhou K, Lou Y. Emerging therapeutic agents for advanced non-small cell lung cancer. J Hematol Oncol. 2020;13:58. doi: 10.1186/s13045-020-00881-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sharma P, Wagner K, Wolchok JD, Allison JP. Novel cancer immunotherapy agents with survival benefit: Recent successes and next steps. Nat Rev Cancer. 2011;11:805–812. doi: 10.1038/nrc3153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Horvath L, Thienpont B, Zhao L, Wolf D, Pircher A. Overcoming immunotherapy resistance in non-small cell lung cancer (NSCLC)-novel approaches and future outlook. Mol Cancer. 2020;19:141. doi: 10.1186/s12943-020-01260-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Vasilakos JP, Tomai MA. The use of toll-like receptor 7/8 agonists as vaccine adjuvants. Expert Rev Vaccines. 2013;12:809–819. doi: 10.1586/14760584.2013.811208. [DOI] [PubMed] [Google Scholar]
- 7.Adams S. Toll-like receptor agonists in cancer therapy. Immunotherapy. 2009;1:949–964. doi: 10.2217/imt.09.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Urban-Wojciuk Z, Khan MM, Oyler BL, Fahraeus R, Marek-Trzonkowska N, Nita-Lazar A, Hupp TR, Goodlett DR. The role of TLRs in anti-cancer immunity and tumor rejection. Front Immunol. 2019;10:2388. doi: 10.3389/fimmu.2019.02388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lim KH. TLR7 and TLR8, Resiquimod, and 852A. In: Marshall J, editor. Cancer Therapeutic Targets. Springer; New York, NY: 2013. pp. 1–8. [Google Scholar]
- 10.Schön MP, Schön M. TLR7 and TLR8 as targets in cancer therapy. Oncogene. 2008;27:190–199. doi: 10.1038/sj.onc.1210913. [DOI] [PubMed] [Google Scholar]
- 11.Zhou Z, Yu X, Zhang J, Tian Z, Zhang C. TLR7/8 agonists promote NK-DC cross-talk to enhance NK cell anti-tumor effects in hepatocellular carcinoma. Cancer Lett. 2015;369:298–306. doi: 10.1016/j.canlet.2015.09.017. [DOI] [PubMed] [Google Scholar]
- 12.Yin T, He S, Wang Y. Toll-like receptor 7/8 agonist, R848, exhibits antitumoral effects in a breast cancer model. Mol Med Rep. 2015;12:3515–3520. doi: 10.3892/mmr.2015.3885. [DOI] [PubMed] [Google Scholar]
- 13.Rook AH, Gelfand JM, Wysocka M, Troxel AB, Benoit B, Surber C, Elenitsas R, Buchanan MA, Leahy DS, Watanabe R, et al. Topical resiquimod can induce disease regression and enhance T-cell effector functions in cutaneous T-cell lymphoma. Blood. 2015;126:1452–1461. doi: 10.1182/blood-2015-02-630335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Killock D. Haematological cancer: Resiquimod-a topical CTCL therapy. Nat Rev Clin Oncol. 2015;12:563. doi: 10.1038/nrclinonc.2015.142. [DOI] [PubMed] [Google Scholar]
- 15.Koh J, Kim S, Lee SN, Kim SY, Kim JE, Lee KY, Kim MS, Heo JY, Park YM, Ku BM, et al. Therapeutic efficacy of cancer vaccine adjuvanted with nanoemulsion loaded with TLR7/8 agonist in lung cancer model. Nanomedicine. 2021;37:102415. doi: 10.1016/j.nano.2021.102415. [DOI] [PubMed] [Google Scholar]
- 16.Li F, Zheng X, Wang X, Xu J, Zhang Q. Macrophage polarization synergizes with oxaliplatin in lung cancer immunotherapy via enhanced tumor cell phagocytosis. Transl Oncol. 2021;14:101202. doi: 10.1016/j.tranon.2021.101202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cheadle EJ, Lipowska-Bhalla G, Dovedi SJ, Fagnano E, Klein C, Honeychurch J, Illidge TM. A TLR7 agonist enhances the antitumor efficacy of obinutuzumab in murine lymphoma models via NK cells and CD4 T cells. Leukemia. 2017;31:1611–1621. doi: 10.1038/leu.2016.352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gallotta M, Assi H, Degagne E, Kannan SK, Coffman RL, Guiducci C. Inhaled TLR9 agonist renders lung tumors permissive to PD-1 blockade by promoting optimal CD4+ and CD8+ T cell interplay. Cancer Res. 2018;78:4943–4956. doi: 10.1158/0008-5472.CAN-18-0729. [DOI] [PubMed] [Google Scholar]
- 19.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Method. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 20.Alam MM, Yang D, Trivett A, Meyer TJ, Oppenheim JJ. HMGN1 and R848 synergistically activate dendritic cells using multiple signaling pathways. Front Immunol. 2018;9:2982. doi: 10.3389/fimmu.2018.02982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Frega G, Wu Q, Le Naour J, Vacchelli E, Galluzzi L, Kroemer G, Kepp O. Trial watch: Experimental TLR7/TLR8 agonists for oncological indications. Oncoimmunology. 2020;9:1796002. doi: 10.1080/2162402X.2020.1796002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lu R, Groer C, Kleindl PA, Moulder KR, Huang A, Hunt JR, Cai S, Aires DJ, Berkland C, Forrest ML. Formulation and preclinical evaluation of a toll-like receptor 7/8 agonist as an anti-tumoral immunomodulator. J Control Release. 2019;306:165–176. doi: 10.1016/j.jconrel.2019.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cen X, Liu S, Cheng K. The role of toll-like receptor in inflammation and tumor immunity. Front Pharmacol. 2018;9:878. doi: 10.3389/fphar.2018.00878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Akira S, Takeda K, Kaisho T. Toll-like receptors: Critical proteins linking innate and acquired immunity. Nat Immunol. 2001;2:675–680. doi: 10.1038/90609. [DOI] [PubMed] [Google Scholar]
- 25.Moingeon P, Haensler J, Lindberg A. Towards the rational design of Th1 adjuvants. Vaccine. 2001;19:4363–4372. doi: 10.1016/S0264-410X(01)00193-1. [DOI] [PubMed] [Google Scholar]
- 26.Michaelis KA, Norgard MA, Zhu X, Levasseur PR, Sivagnanam S, Liudahl SM, Burfeind KG, Olson B, Pelz KR, Ramos DMA, et al. The TLR7/8 agonist R848 remodels tumor and host responses to promote survival in pancreatic cancer. Nat Commun. 2019;10:4682. doi: 10.1038/s41467-019-12657-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hosoya T, Sato-Kaneko F, Ahmadi A, Yao S, Lao F, Kitaura K, Matsutani T, Carson DA, Hayashi T. Induction of oligoclonal CD8 T cell responses against pulmonary metastatic cancer by a phospholipid-conjugated TLR7 agonist. Proc Natl Acad Sci USA. 2018;115:E6836–E6844. doi: 10.1073/pnas.1803281115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Karimi K, Boudreau J, Fraser K, Liu H, Delanghe J, Gauldie J, Xing Z, Bramson JL, Wan Y. Enhanced antitumor immunity elicited by dendritic cell vaccines is a result of their ability to engage both CTL and IFN gamma-producing NK cells. Mol Ther. 2008;16:411–418. doi: 10.1038/sj.mt.6300347. [DOI] [PubMed] [Google Scholar]
- 29.Stojanovic A, Cerwenka A. Natural killer cells and solid tumors. J Innate Immun. 2011;3:355–364. doi: 10.1159/000325465. [DOI] [PubMed] [Google Scholar]
- 30.Gotwals P, Cameron S, Cipolletta D, Cremasco V, Crystal A, Hewes B, Mueller B, Quaratino S, Sabatos-Peyton C, Petruzzelli L, et al. Prospects for combining targeted and conventional cancer therapy with immunotherapy. Nat Rev Cancer. 2017;17:286–301. doi: 10.1038/nrc.2017.17. [DOI] [PubMed] [Google Scholar]
- 31.Meador CB, Hata AN. Acquired resistance to targeted therapies in NSCLC: Updates and evolving insights. Pharmacol Ther. 2020;210:107522. doi: 10.1016/j.pharmthera.2020.107522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang D, Precopio M, Lan T, Yu D, Tang JX, Kandimalla ER, Agrawal S. Antitumor activity and immune response induction of a dual agonist of toll-like receptors 7-8. Mol Cancer Ther. 2010;9:1788–1797. doi: 10.1158/1535-7163.MCT-09-1198. [DOI] [PubMed] [Google Scholar]
- 33.Pandey S, Kawai T, Akira S. Microbial sensing by toll-like receptors and intracellular nucleic acid sensors. Cold Spring Harb Perspect Biol. 2014;7:a016246. doi: 10.1101/cshperspect.a016246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Petes C, Odoardi N, Gee K. The toll for trafficking: Toll-like receptor 7 Delivery to the endosome. Front Immunol. 2017;8:1075. doi: 10.3389/fimmu.2017.01075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Smith AA, Gale EC, Roth GA, Maikawa CL, Correa S, Yu AC, Appel EA. Nanoparticles presenting potent TLR7/8 agonists enhance Anti-PD-L1 immunotherapy in cancer treatment. Biomacromolecules. 2020;21:3704–3712. doi: 10.1021/acs.biomac.0c00812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bahmani B, Gong H, Luk BT, Haushalter KJ, DeTeresa E, Previti M, Zhou J, Gao W, Bui JD, Zhang L, et al. Intratumoral immunotherapy using platelet-cloaked nanoparticles enhances antitumor immunity in solid tumors. Nat Commun. 2021;12:1999. doi: 10.1038/s41467-021-22311-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rodell CB, Ahmed MS, Garris CS, Pittet MJ, Weissleder R. Development of adamantane-conjugated TLR7/8 agonists for supramolecular delivery and cancer immunotherapy. Theranostics. 2019;9:8426–8436. doi: 10.7150/thno.35434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Goliwas K, Deshane J, Elmets C, Athar M. Moving immune therapy forward targeting TME. Physiol Rev. 2021;101:417–425. doi: 10.1152/physrev.00008.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Taube JM, Galon J, Sholl LM, Rodig SJ, Cottrell TR, Giraldo NA, Baras AS, Patel SS, Anders RA, Rimm DL, Cimino-Mathews A. Implications of the tumor immune microenvironment for staging and therapeutics. Mod Pathol. 2018;31:214–234. doi: 10.1038/modpathol.2017.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rodell CB, Arlauckas SP, Cuccarese MF, Garris CS, Li R, Ahmed MS, Kohler RH, Pittet MJ, Weissleder R. TLR7/8-agonist-loaded nanoparticles promote the polarization of tumour-associated macrophages to enhance cancer immunotherapy. Nat Biomed Eng. 2018;2:578–588. doi: 10.1038/s41551-018-0236-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Durgeau A, Virk Y, Corgnac S, Mami-Chouaib F. Recent advances in targeting CD8 T-cell immunity for more effective cancer immunotherapy. Front Immunol. 2018;9:14. doi: 10.3389/fimmu.2018.00014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zahm CD, Colluru VT, McIlwain SJ, Ong IM, McNeel DG. TLR stimulation during T-cell activation lowers PD-1 expression on CD8(+) T cells. Cancer Immunol Res. 2018;6:1364–1374. doi: 10.1158/2326-6066.CIR-18-0243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dajon M, Iribarren K, Cremer I. Dual roles of TLR7 in the lung cancer microenvironment. Oncoimmunology. 2015;4:e991615. doi: 10.4161/2162402X.2014.991615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chatterjee S, Crozet L, Damotte D, Iribarren K, Schramm C, Alifano M, Lupo A, Cherfils-Vicini J, Goc J, Katsahian S, et al. TLR7 promotes tumor progression, chemotherapy resistance, and poor clinical outcomes in non-small cell lung cancer. Cancer Res. 2014;74:5008–5018. doi: 10.1158/0008-5472.CAN-13-2698. [DOI] [PubMed] [Google Scholar]
- 45.Cherfils-Vicini J, Platonova S, Gillard M, Laurans L, Validire P, Caliandro R, Magdeleinat P, Mami-Chouaib F, Dieu-Nosjean MC, Fridman WH, et al. Triggering of TLR7 and TLR8 expressed by human lung cancer cells induces cell survival and chemoresistance. J Clin Invest. 2010;120:1285–1297. doi: 10.1172/JCI36551. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.