Abstract
Triple negative breast cancer (TNBC) lacks targeted treatment resulting in poor prognosis. Targeting overexpressing mesothelin (MSLN) using MSLN-specific T cells is an attractive treatment approach and the aim of the present study. The expression of MSLN in human TNBC paraffin sections was analyzed by immunohistochemistry. Lentiviral vector harbored granulocyte-macrophage colony stimulating factor (GM-CSF), interleukin-4 (IL-4) and MSLN cDNAs was constructed to generate self-differentiated myeloid-derived antigen-presenting-cells reactive against tumor expressing MSLN dendritic cell (MSLN-SmartDC) for MSLN-specific T cell activation. The results showed high MSLN in 32.8% of all breast cancer subtypes and 57% in TNBC. High MSLN was significantly associated with TNBC subtype and the absence of estrogen receptor, progesterone receptor and human epidermal growth factor receptor 2. MSLN-SmartDC exhibited comparable phenotype to DC generated by exogenous cytokine treatment and an addition of 40s ribosomal protein subunit 3 (RPS3), a toll-like receptor 4 ligand, enhanced DC maturation and function by upregulation of CD40, CD80 and CD83 expressions and IL-12p70 secretion. MSLN-specific CD8+CD69+ IFN-γ+ T cells were detected in T cells activated by both MSLN-SmartDC and RPS3-MSLN-SmartDC. MSLN-specific T cells activated by these DCs showed more specific killing capability against naturally expressed MSLN-HCC70 and artificially MSLN-overexpressing MDA-MB-231 compared with parental MDA-MB-231 in both two dimensional (2D)- and 3D-culture systems. In conclusion, the results demonstrated the efficacy of MSLN-SmartDC to promote MSLN-specific T cells response against TNBC and RPS3 can enhance the cytolytic activity of these T cells providing an alternative treatment approach for patients with TNBC.
Keywords: dendritic cell, self-differentiated myeloid-derived antigen-presenting-cell, mesothelin, triple negative breast cancer, 40S ribosomal protein subunit 3
Introduction
Triple negative breast cancer (TNBC) is a subtype of breast cancer accounting for 15–20% of all breast cancer and characterized by the absence of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor 2 (HER2) (1). TNBC is predominant in younger patients and demonstrates aggressive clinical features with poor prognosis (1). Due to lack of hormonal receptors and HER2 expression, TNBC is less sensitive to hormonal- and HER2-targeted treatment (1). The current treatment standard for TNBC is surgical resection and chemotherapy (1). Although, several studies have shown that TNBC is more sensitive to chemotherapy than other subtypes, a portion of patients experience recurrence at the visceral organs within five years after treatment (1,2). A number of biomarkers are identified as the theranostic biomarkers for breast cancer, such as microRNAs, checkpoint molecules, epigenetic profiles and aberrant signaling molecules, though the effective utilization of these markers in the management of patients with TNBC is limited (3–5). Therefore, developing a novel treatment approach to improve the treatment outcome for patients with TNBC is required.
Dendritic cells (DCs)-based immunotherapy aims to induce an antigen-specific immune response through DCs presenting antigens to stimulate cancer-specific T cells returned to the patients to eliminate cancer cells (6,7). Our group has previously demonstrated the ability of monocyte-derived DCs generated by lentiviral transduction of granulocyte-macrophage colony stimulating factor (GM-CSF), interleukin-4 (IL-4) and protein kinase cAMP-dependent type I-alpha regulatory subunit (PRKAR1A) to efficiently induce PRKAR1A-specific T cells against cholangiocarcinoma cells (8). These DCs are called self-differentiated myeloid derived antigen-presenting cells reactive against tumor (SmartDCs) and can produce the cytokines required for DCs differentiation in an autocrine manner and the tumor-associated antigen processing for cytotoxic T cells activation (8). The SmartDC platform has been demonstrated an efficient antigen-specific T cells in leukemia, melanoma and cholangiocarcinoma (8–12).
Mesothelin (MSLN) is a glycophosphatidylinositol-linked glycoprotein limitedly expressed in mesothelial cells but reported to be aberrantly expressed in various types of cancer, such as ovarian cancer, pancreatic cancer, mesothelioma and TNBC (13–16). Notably, it has been recognized as a potential target for cancer immunotherapy (15–17). In breast cancer, several studies have reported the overexpression of MSLN specifically in TNBC and its high expression is associated with poor prognosis (14,18–22). The overexpression of MSLN in TNBC but limited expression in normal tissue highlight the potential of MSLN as a target for T cell therapy.
The efficacy of T cells activated by DCs is greatly affected by the maturation status of DCs including the human leukocyte antigens (HLAs), co-stimulatory molecules and cytokines expressions (6). In a steady state, DCs express these molecules limitedly but on encountering factors derived from pathogen or cancer cells, DCs can be activated via the pattern recognition receptors signaling pathway such as toll-like receptors (TLRs) (23). TLR ligands are used as adjuvant to induce immune response in several cancers (23–25). A TLR4 ligand called 40s ribosomal protein subunit 3 (RPS3) can induce DCs maturation (26). Taken together, the present study aimed to investigate the expression of MSLN in breast cancer tissues and the potential of genetically manipulated MSLN-SmartDC to induce MSLN-specific T cells responding against TNBC cells. Moreover, the effect of RPS3 treatment on MSLN-SmartDC immunophenotype and T cells activity to enhance cytolytic activity of T cells were reported. These findings suggested the effect of MSLN as a potential TNBC antigen and MSLN-specific T cells produced from RPS3-MSLN-SmartDC presented effective capability to destroy MSLN-positive TNBC cells.
Materials and methods
Immunohistochemistry
A total of 351 cases of paraffin-embedded breast cancer tissues and clinicopathological data (Table I) were collected under Siriraj Institutional Review Board ethical approval (COA no. Si 580/2018). Following antigen retrieval in pH 6 citrate buffer, the samples were stained with anti-human MSLN antibody (cat. no. sc-271540; Santa Cruz Biotechnology, Inc.) at 4°C overnight. Then incubated with goat anti-mouse Envision (cat. no. K4001; Dako; Agilent Technologies, Inc.) for 30 min and HRP activity was detected by Dako-HRP detection kit (Dako; Agilent Technologies, Inc.) before counterstained with Mayer's hematoxylin. The samples were scanned at 400× by Scanscope slide scanner (Aperio Technologies, Inc.). MSLN expression was scored by multiplying the intensity [I; graded: 0 (negative), 1 (weak), 2 (moderate) and 3 (strong)], with the percentage of MSLN positive cells [P; graded: as 0 (no positive cells), 1 (1–25%), 2 (26–50%), 3 (51–75%) and 4 (76–100%)]. The MSLN expression scores calculated by multiplying I and P resulting in score between 0 to 12. The samples were classified into low if the score is less than median (which was 0) and rated as high if the score was equal or more than median.
Table I.
Relationship between MSLN expression and clinicopathological factors analyzed by Fisher's exact test. (Some clinical data were not available for some patient samples).
| Full cohort (n=351) | HER2 cohort (n=134) | TNBC cohort (n=165) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
||||||||
| Characteristic | Low | High | P-value | Low | High | P-value | Low | High | P-value | |
| Age (years) (n=351) | ≤50 | 83 | 46 | 0.410 | 28 | 5 | 1.000 | 24 | 41 | 0.260 |
| >50 | 153 | 69 | 86 | 15 | 47 | 53 | ||||
| pT (n=351) | 1-2 | 210 | 107 | 0.255 | 103 | 19 | 0.693 | 63 | 87 | 0.424 |
| 3-4 | 26 | 8 | 11 | 1 | 8 | 7 | ||||
| pN (n=350) | 0 | 121 | 71 | 0.086 | 56 | 9 | 0.810 | 41 | 62 | 0.331 |
| 1-3 | 114 | 44 | 57 | 11 | 30 | 32 | ||||
| pM (n=348) | 0 | 226 | 114 | 0.175 | 112 | 20 | 1.000 | 71 | 94 | NA |
| 1 | 9 | 1 | 1 | 0 | 0 | 0 | ||||
| Clinical staging (n=348) | 1-2 | 168 | 92 | 0.087 | 80 | 14 | 1.000 | 53 | 78 | 0.244 |
| 3-4 | 66 | 22 | 32 | 5 | 18 | 16 | ||||
| Subtype (n=351) | TNBC | 71 | 94 | <0.001 | - | - | NA | - | - | NA |
| HER2 | 114 | 20 | - | - | - | - | ||||
| Luminal | 51 | 1 | - | - | - | - | ||||
| ER (n=351) | Neg | 185 | 114 | <0.001 | - | - | NA | - | - | NA |
| Pos | 51 | 1 | - | - | - | - | ||||
| PR (n=351) | Neg | 190 | 114 | <0.001 | - | - | NA | - | - | NA |
| Pos | 46 | 1 | - | - | - | - | ||||
| HER2 (n=342) | Neg | 69 | 63 | <0.001 | - | - | NA | - | - | NA |
| Pos | 158 | 52 | - | - | - | - | ||||
| LN metastasis (n=350) | Neg | 129 | 73 | 0.136 | 55 | 10 | 1.000 | 44 | 63 | 0.515 |
| Pos | 106 | 42 | 58 | 10 | 27 | 31 | ||||
| Perineural metastasis (n=351) | Neg | 185 | 99 | 0.111 | 83 | 15 | 1.000 | 59 | 83 | 0.370 |
| Pos | 51 | 16 | 31 | 5 | 12 | 11 | ||||
MSLN, mesothelin; HER2, human epidermal growth factor receptor 2; TNBC, triple negative breast cancer; p, pathological stage; ER, estrogen receptor; PR, progesterone receptor; LN, lymph node; NA, Not available.
Cell culture
MDA-MB-231, HCC70 and T2 cell lines were from American Type Culture Collection. Lenti-X™ 293T cells were from Takara Bio (Takara Bio, San Jose, CA). The Lenti-X™ 293T cells, MDA-MB-231 and MSLN-MDA-MB-231, produced by lentiviral transduction, were maintained in DMEM with 10% fetal bovine serum (FBS; Invitrogen; Thermo Fisher Scientific, Inc.). HCC70 cells were cultured in RPMI-1640 with 10% FBS (Invitrogen; Thermo Fisher Scientific, Inc.). The T2 cell line was cultured in RPMI-1640 with 10% FBS and 2 mM L-glutamine (Invitrogen; Thermo Fisher Scientific, Inc.). All cell lines were cultured at 37°C with 5% CO2.
MSLN-SmartDC lentiviral vector construction and production
The construction of lentiviral vector was performed as previously described (8). Briefly, the full-length MSLN cDNA with restriction cut sites was amplified from HCC70 cells and cloned into pCDH1/GM-CSF/IL-4 plasmid. The sequence integrity of MSLN-SmartDC plasmid was confirmed by Sanger's sequencing. A mock control pCDH1/GM-CSF/IL-4 containing an irrelevant red fluorescence protein (IRFP), IRFP-SmartDC, was constructed (8). MSLN-SmartDC lentiviral particle was produced by transfecting the 30 µg of MSLN-SmartDC plasmid along with 21 µg of lentiviral envelop plasmid, 6 µg of pMD2.G and lentiviral packaging plasmid, psPAX2 into 8×106 Lenti-X™ 293T using calcium phosphate method at room temperature (RT). The lentiviral particle in the culture medium was collected 3 days after transfection and concentrated by 20,000 × g ultracentrifugation at 4°C before measured for viral titer using the qualitative (q)PCR Lentivirus Titration kit (Applied Biological Materials, Inc.).
Generation of MSLN-SmartDC and rhRPS3-treated MSLN-SmartDC
The peripheral blood mononuclear cells (PBMCs) were isolated from 30 ml of HLA-A2+ healthy donor blood with written consent under Siriraj Institutional Review Board ethical approval (COA no. Si 580/2018) by density centrifugation at 800 × g for 20 min at RT in lymphocyte separating medium (Corning Life Sciences). The monocytes were isolated and incubated for 1 h at 37°C. The non-adherence cells were collected and cryopreserved in human AB serum (MilliporeSigma) containing 10% dimethyl sulfoxide until use. The monocytes were transduced with IRFP-SmartDC or MSLN-SmartDC at 75 multiplicity of infection (MOI) together with 10 µg/ml protamine sulfate in AIM-V medium (Invitrogen; Thermo Fisher Scientific, Inc.). On day 5 post-transduction, 1 µg/ml of recombinant human RPS3 (cat. no. NBP2-90977; Novus Biologicals, LLC) was added. Monocytes were cultured in 100 ng/ml of rhGM-CSF (cat. no. 11343125; ImmunoTools GmbH) and 50 ng/ml of rhIL-4 (cat. no. 11340045; ImmunoTools GmbH) for five days and treated with 100 ng/ml of rhIFN-γ (cat. no. 11343536; ImmunoTools GmbH) and rhTNF-α (cat. no. 11343015; ImmunoTools GmbH) for additional 48 h served as positive control or conventional DC (conv. DC). All DCs were harvested on day 7 to check the activated characters.
Activation and expansion of T cells by MSLN-SmartDC
The cryopreserved T cells were thawed and co-cultured with DCs for 48 h at a 10:1 ratio to support T cell activation and proliferation (27) in AIM-V medium at 37°C with 5% CO2. T cells were expanded in AIM-V with 5% human AB serum (MilliporeSigma), 20 ng/ml of rhIL-2 (cat. no. 11340025, ImmunoTools GmbH), 10 ng/ml of rhIL-7 (cat. no. 11340075, ImmunoTools GmbH) and 20 ng/ml of rhIL-15 (cat. no. 11340155, ImmunoTools GmbH) for seven days at 37°C with 5% CO2.
Flow cytometry analysis
The immunophenotype of monocytes and DCs were assessed by anti-CD14 (cat. no. 21620143, ImmunoTools GmbH), anti-CD40 (cat. no. 21270403, ImmunoTools GmbH) and anti-human leukocyte antigen (HLA)-DR (cat. no. 21278993, ImmunoTools GmbH), anti-CD80 (cat. no. 12-0809-42, Thermo Fisher Scientific, Rockford, IL), anti-CD83 (cat. no. 12-0839-42, Thermo Fisher Scientific) and anti-CD86 antibodies (cat. no. 12-0869-42, Thermo Fisher Scientific). All antibodies were used at 1:50 dilution for 30 min at 4°C. Isotype antibodies were used as negative controls.
The memory T cell subsets were stained by anti-CD3 (cat. no. 21620033, ImmunoTools GmbH), anti-CD45RA (cat. no. 21819456, ImmunoTools GmbH) and anti-CD62L (cat. no. 21819624, ImmunoTools GmbH), anti-CD4 (cat. no. 56-0049-42, Thermo Fisher Scientific) and anti-CD8 (cat. no. A15448, Thermo Fisher Scientific). For intracellular cytokines, the activated T cells were re-stimulated with 10 µg of MSLN antigenic peptides (SLLFLLFSL and VLPLTVAEV) (GenScript, Jiangsu, China) (13,28) for 6 h in the presence of BD GolgiPlug (BD Biosciences, San Jose, CA) at 37°C with 5% CO2. The cells were stained with anti-CD3, anti-CD4, anti-CD8 and anti-CD69 (cat. no. MA1-10277, Thermo Fisher Scientific). Cells were fixed in BD Cytofix/Cytoperm (BD Biosciences) for 20 min at 4°C, stained with anti-IFN-γ antibody (cat. no. 17-7319-82, Thermo Fisher Scientific). All antibodies were stained at 1:100 dilution for 30 min at 4°C.
The flow cytometry data of DCs and T cell immunophenotypes were acquired by CytoFLEX (Beckman Coulter, Inc.); the intracellular cytokine staining was acquired by BD LSRFortessa (BD Biosciences). The data were analyzed by FlowJo version 10.7 (FlowJo LLC) and shown as geometric mean fluorescence intensity (MFI) of each marker normalized by isotype control.
Enzyme-linked immunosorbent assay (ELISA)
GM-CSF, IL-4 and IL-12p70 were measured using Human GM-CSF (cat. no. DGM00), IL-4 (cat. no. D4050) and IL-12p70 (cat. no. D1200) Quantikine ELISA kits (R&D systems, Inc.). IFN-γ was measured in medium collected from activated T cells co-cultured with target cancer cells using Human IFN-γ Quantikine ELISA kit (cat. no. DIF50, R&D systems, Inc.).
Enzyme-linked immunosorbent spot assay (ELISpot)
The IFN-γ ELISpot assay was performed using a Human IFN-γ ELISpotBASIC kit (Mabtech AB). Briefly, 15 µg/ml of IFN-γ capture antibody was coated for overnight at 4°C. The 2×105 activated T cells were then re-stimulated with 1×104 MSLN peptides-pulsed HLA-A2+ T2 cells. T cells treated with 20 ng/ml of phorbol 12-myristate 13-acetate and 1 µg/ml of ionomycin (MilliporeSigma) served as positive controls. After removing these T cells, 1 µg/ml of biotinylated-IFN-γ antibody was incubated for 2 h and ALP-conjugated streptavidin for 1 h at RT. The IFN-γ spots were evaluated by 5-bromo-4-chloro-3-indolyl phosphate/nitro blue tetrazolium plus substrate (Mabtech AB) and captured by ELISpot plate reader (BIOREADER® 5000 Fγ, BIOSyS). The numbers of spots were counted by CellCounter software (https://github.com/nghiaho12/CellCounter) version 0.2.1.
Western blot analysis
Lenti-X™ 293T and cancer cell lysates were prepared in RIPA Lysis Buffer System (Santa Cruz Biotechnology). The protein concentration was determined by Bradford assay and 30 µg of protein lysates were separated in 12% SDS-PAGE before transferred to PVDF membrane (GE Healthcare, Buckinghamshire, UK). The membrane was blocked in 5% skimmed milk (MilliporeSigma) for 30 min at RT. The membrane was incubated with 1:500 anti-MSLN and 1:5,000 anti-β-actin antibodies (cat. no. sc-47778; Santa Cruz Biotechnology, Inc.) at 4°C overnight. The 1:1,000 HRP-conjugated goat anti-mouse antibody (cat. no. 7076; Cell Signaling Technology, Inc.) was added and incubated for 1 h at RT. The signal was detected by Clarity™ western ECL substrate (Bio-Rad Laboratories, Inc.) using a Gel Doc instrument (G:Box Chemi XR5; Syngene Europe). The band intensity was analyzed by ImageJ software version 1.53 (NIH).
2D cancer killing by luciferase assay
T cells were co-cultured with 5,000 cells of luciferase expressing HCC70, MDA-MB-231 and MSLN-MDA-MB-231 cells at Effector: Target ratio of 1:1, 5:1 and 10:1 for 24 h at 37°C with 5% CO2. The luciferase activity was determined using Pierce Firefly Luciferase Glow Assay kit (Pierce; Thermo Fisher Scientific, Inc.) and Lumat LB 9507 Ultra-sensitive Luminometer (Berthold Technologies GmbH & Co. KG). After normalizing the luciferase activity of every condition with target cell alone condition, the percentage of cancer cell lysis was calculated. The luciferase activity of target cell alone was served as an internal control.
3D-spheroid cancer killing assay
The 1×103 target mWasabi-transduced cancer cells were formed into spheroid in 96-well ultra-low attachment plates (Corning Life Sciences) in 200 µl culture medium containing 2.5% cold Matrigel (BD Biosciences) by centrifugation at 300 × g for 3 min at 4°C. The spheroid was incubated for 4 days at 37°C with 5% CO2 with CellTracker™ Orange CMRA Dye (Invitrogen; Thermo Fisher Scientific, Inc.)-labelled activated T cells at Effector: Target ratios of 1:1, 5:1, 10:1 and 20:1 for 48 h at 37°C with 5% CO2. The mWasabi and CMRA fluorescence signals were detected by inverted fluorescence microscope and cellSens standard program version 1.15 (Olympus Corporation).
Statistical analysis
The association between MSLN score and clinicopathological data were accessed by Fisher's exact test. One-way ANOVA and Tukey's post-hoc test was used for all experiments except in the intracellular cytokine staining for comparison of control- and peptide-challenged T cells from the same condition in which Student's t-test was used. The Fisher's exact test was performed in SPSS 17.0 (SPSS, Inc.), whereas GraphPad prism V (GraphPad Software, Inc.) was used for one-way ANOVA and Student's t-test. All results were shown as mean ± standard deviation from at least three independent experiments. P<0.05 was considered to indicate a statistically significant difference.
Results
MSLN expression in breast cancer tissues
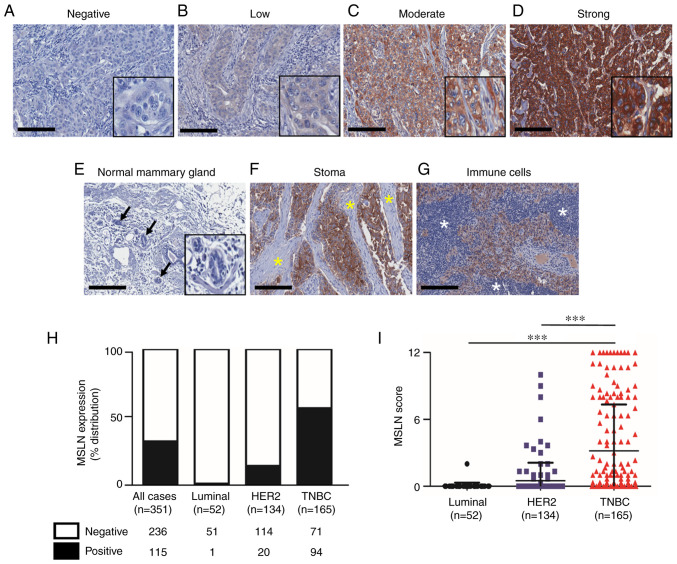
All patient cases were female with mean age of 54±11.5 years diagnosed as luminal, HER2-positive and TNBC subtypes for 52 (15%), 154 (38%) and 165 (47%) cases, respectively (Table I). The cytoplasmic and membranous patterns of MSLN positive were detected in cancer cells with various levels (Fig. 1A-D). No MSLN expression was observed in the adjacent normal mammary cells (Fig. 1E), stromal cells (Fig. 1F) and immune cells (Fig. 1G). Among 351 total cases, 115 cases (32.8%) were positive for MSLN. When stratifying the patients according to the subtypes, MSLN expression was found in 94 of 165 cases (57%) for TNBC subtype, 20 of 154 cases (14.9%) in HER2-positive and 1 of 52 cases (1.9%) in luminal subtypes (Fig. 1H). The mean MSLN expression score was significantly higher in TNBC subtype compared with that in HER2-positive and luminal subtype (Fig. 1I). MSLN was significantly correlated with the absence of ER, PR and HER2 and TNBC subtype (Table 1). There was no association between MSLN and overall or disease-free survival in all samples, HER2-positive and TNBC subtypes (data not shown).
Figure 1.
Expression of MSLN in human breast cancer tissue samples. Representative images of MSLN expression levels in breast cancer tissues ranging from (A) negative, (B) low, (C) moderate and (D) strong staining. (E) Normal lobules (black arrow) showed no MSLN expression. Negative staining of MSLN in (F) stromal cells (yellow asterisk) and (G) immune cells (white asterisk). (H) Proportion of MSLN expressing samples in full cohort and stratified subtypes. (I) Mean MSLN score stratified by subtypes of breast cancer. Original magnification, 200×; scale bars=200 µm. ***P<0.001. MSLN, mesothelin.
Generation and immunophenotyping of MSLN-SmartDC and RPS3-MSLN-SmartDC
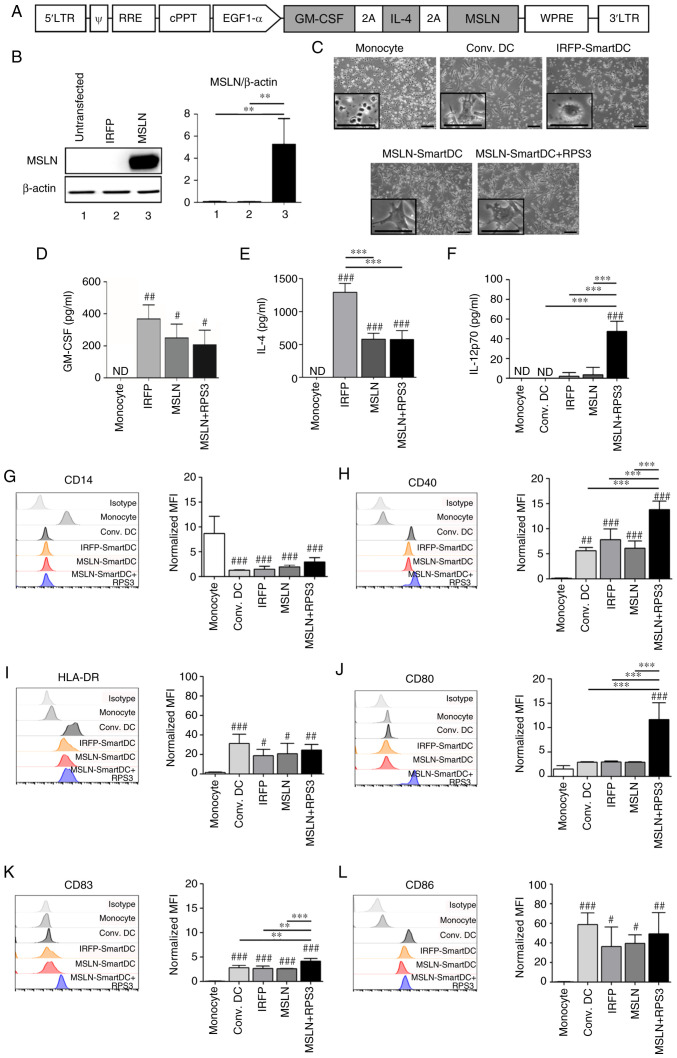
MSLN-SmartDC was generated by transducing the lentiviral vector containing GM-CSF, IL-4 and MSLN (Fig. 2A) into PBMCs-derived monocytes from four HLA-A2 positive healthy donors. The expression of MSLN protein from MSLN-SmartDC lentiviral vector was confirmed in Lenti-X™ 293T (Fig. 2B). All DCs demonstrated dendritic-like morphology (Fig. 2C). The GM-CSF was significantly detected higher levels in both MSLN-SmartDC and RPS3-MSLN-SmartDC compared with monocytes (Fig. 2D). MSLN-SmartDC and RPS3-MSLN-SmartDC also secreted IL-4 significantly higher than the monocytes but significantly lower compared with IRFP-SmartDC (Fig. 2E). Production of IL-12p70 was detected in significantly higher levels in RPS3-MSLN-SmartDC compared with other conditions (Fig. 2F). The significant reduction of CD14 monocyte marker in all DCs conditions (Fig. 2G) and mature DC markers including CD40, HLA-DR, CD80, CD83 and CD86 (Fig. 2H-L) were significantly increased compared with monocytes. The addition of RPS3 to MSLN-SmartDC could significantly increase CD40, CD80 and CD83 (Fig. 2H and J-K) compared with those of MSLN-SmartDC.
Figure 2.
The lentiviral vector schematic maps and immunophenotype of MSLN-SmartDC and RPS3-MSLN-SmartDC. (A) Construction of MSLN-SmartDC lentiviral vector. (B) Expression of MSLN protein in Lenti-X™ 293T transfected with IRFP-SmartDC and MSLN-SmartDC lentiviral vectors. (C) Representative images of monocytes at day 0 and DCs at day 7 of the experiment. Production of (D) GM-CSF, (E) IL-4 and (F) IL-12p70 by monocytes and different DCs. The geometric mean fluorescence intensity of surface markers of DCs in different treatment conditions including (G) CD14, (H) CD40, (I) HLA-DR, (J) CD80, (K) CD83 and (L) CD86 in monocytes and DCs. Original magnification, ×100 and scale bar=50 µm. The results were collected from four independent experiments. ND, not detected. #P<0.05, ##P<0.01 and ###P<0.001 vs. monocytes. **P<0.01 and ***P<0.001. MSLN, mesothelin; MSLN-SmartDC, MSLN dendritic cells; RPS3, ribosomal protein subunit 3; GM-CSF, granulocyte-macrophage colony stimulating factor; DCs, dendritic cells; HLA, human leukocyte antigen.
Immunophenotype of T cells activated by MSLN-SmartDC and RPS3-MSLN-SmartDC
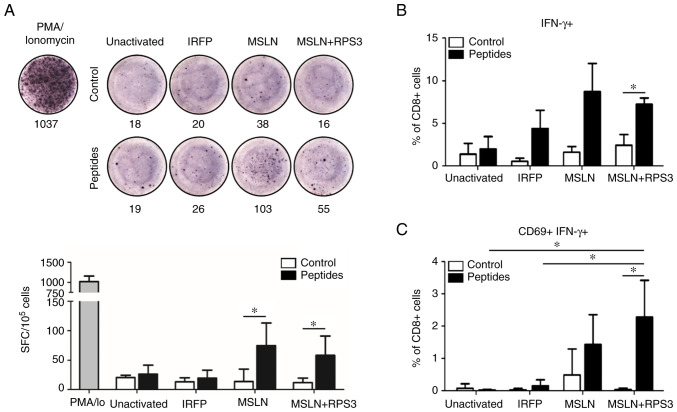
The results showed slight changes in CD4+ and CD8+ T cells frequencies and memory T cells subsets compared with those in PBMCs at day 0 (Fig. S1A and B). Re-stimulation with MSLN antigenic peptides in T cells activated by MSLN-SmartDC and RPS3-MSLN-SmartDC showed significantly higher number of IFN-γ than those without peptide treatment (Fig. 3A). The frequency of IFN-γ+ CD8+ T cells after MSLN peptides challenging were found at higher levels in MSLN-SmartDC- and RPS3-MSLN-activated T cells than unactivated and IRFP-activated T cells (Fig. 3B). However, only the RPS3-MSLN-activated T cells revealed significant increased IFN-γ+ CD8+ T cells (Fig. 3B). The MSLN-SmartDC-activated T cells result did not achieved a statistically significant level. The dual expressions of CD69 and IFN-γ were observed in MSLN- and RPS3-MSLN-activated T cells, but very low level in T cells treated with unactivated- and IRFP-activated T cells (Fig. 3C). The frequency of MSLN-specific CD69+ IFN-γ+ CD8+ T cells activated by RPS3-MSLN-SmartDC was clearly higher than unactivated and IRFP-activated T cells. Moreover, CD69+ IFN-γ+ CD8+ T cells in RPS3-MSLN-activated T cells was significantly higher compared with those without peptides re-stimulation.
Figure 3.
Presence of MSLN-specific T cells after SmartDCs activation. (A) Representative ELISpot well images and the quantification of IFN-γ secreting cells after restimulation with MSLN antigenic peptides. Frequency of (B) IFN-γ+ and (C) MSLN-specific T cells (CD69+ IFN-γ+) gated from CD8+ T cells after restimulation with MSLN antigenic peptides. The results were collected from thee independent experiments. *P<0.05. MSLN, mesothelin.
2D cytolytic activity of MSLN-SmartDC- and RPS3-MSLN-SmartDC-activated T cells
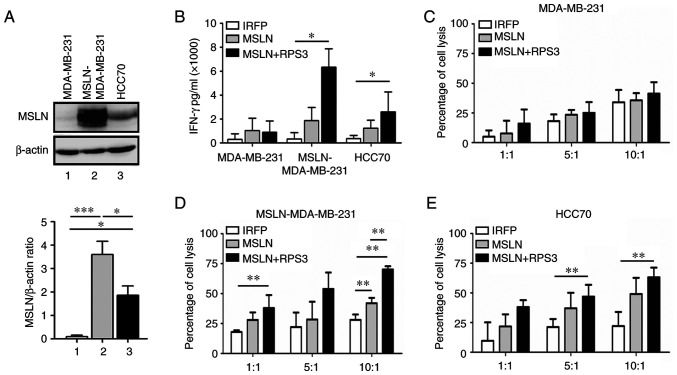
The MSLN showed the highest level in MSLN-MDA-MB-231, followed by HCC70, whereas MDA-MB-231 had no MSLN (Fig. 4A). The IFN-γ releasing from RPS3-MSLN-activated T cells after co-culturing with MSLN-MDA-MB-231 and HCC70 cells was higher than MSLN-activated T cells and significantly higher than those of IRFP-activated T cells (Fig. 4B). No changes of IFN-γ secreted from T cells cocultured with MDA-MB-231 were observed.
Figure 4.
MSLN expression in TNBC cells and 2D killing assay by T cells activated by MSLN-SmartDC and RPS3-MSLN-SmartDC. (A) Western blot analysis of MSLN in TNBC cell lines. (B) IFN-γ concentration in culture medium after 24 h of co-culturing with MDA-MB-231, MSLN-MDA-MB-231 and HCC70 at E:T ratio of 10:1. Percentage of cell lysis of (C) MDA-MB-231, (D) MSLN-overexpressing MDA-MB-231 (MSLN-MDA-MB-231) and (E) HCC70 co-cultured with activated T cells at various ratios of E:T. The results were collected from four independent experiments. *P<0.05, **P<0.01 and ***P<0.001. MSLN, mesothelin; TNBC, triple negative breast cancer; 2D, two dimensional; MSLN-SmartDC, MSLN dendritic cells; RPS3, ribosomal protein subunit 3; E:T, Effector cell: Target cell.
The results exhibited no difference in MDA-MB-231 cell lysis co-cultured with both MSLN-activated T cells and RPS3-MSLN-activated T cells at Effector: Target ratios of 1:1, 5:1 and 10:1 (Fig. 4C). T cells activated by MSLN-SmartDC demonstrated significantly higher MSLN-MDA-MB-231 cell cytotoxicity compared with IRFP-activated T cells at 10:1 ratio (Fig. 4D). At 10:1, T cells activated by RPS3-MSLN-SmartDC could significantly induce MSLN-MDA-MB-231 cell lysis more than MSLN-activated T cells (Fig. 4D). In HCC70, T cells activated by MSLN-SmartDC and RPS3-MSLN-SmartDC demonstrated higher killing activity than IRFP-activated T cells (Fig. 4E). However, only RPS3-MSLN-activated T cells achieved statistical significance of cancer cell killing compared with that of IRFP-activated T cells at ratios of 5:1 and 10:1 (Fig. 4E).
3D cytolytic activity of T cells activated by MSLN-SmartDC and RPS3-MSLN-SmartDC
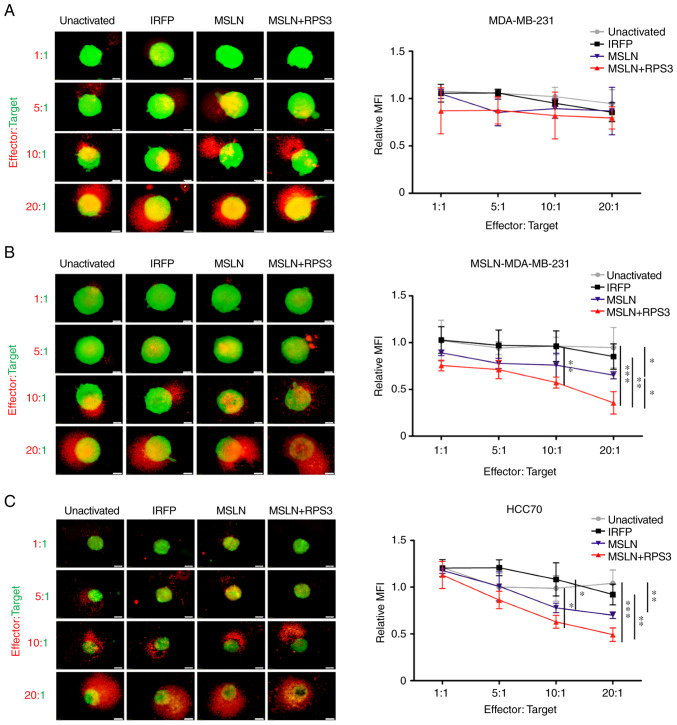
MDA-MB-231 spheroids co-cultured with activated T cells or unactivated T cells for 48 h revealed no differences in mWasabi green fluorescence signals representing viable cancer cells (Fig. 5A). MSLN-MDA-MB-231 spheroid co-cultured with T cells activated by MSLN-SmartDC significantly exhibited lower viable cells compared with those of unactivated T cells at 20:1 (Fig. 5B). T cells activated by RPS3-MSLN-SmartDC had significant decrease viable cells compared with unactivated T cells (P<0.001), IRFP-activated and MSLN-activated T cells at 20:1 (Fig. 5B). RPS3 could enhance DCs activation that subsequently increase T cells capability to recognize MSLN-MDA-MB-231 cells. For HCC70 spheroid, the significant decreased fluorescence signal was observed in MSLN-SmartDC-activated T cells at ratio 20:1 compared with unactivated T cells. Moreover, T cells activated by RPS3-MSLN-SmartDC demonstrated significant reduction of mWasabi fluorescence signal compared with both unactivated and IRFP-activated T cells at 10:1 and 20:1 (Fig. 5C).
Figure 5.
Cytotoxicity of cancer cells mediated by MSLN-SmartDC- and RPS3-MSLN-activated T cells in 3D cancer spheroid assay. Representative cancer spheroid (green) co-cultured with T cells (orange) for 48 h and the changes in MFI of mWasabi signal in (A) MDA-MB-231, (B) MSLN-MDA-MB-231 and (C) HCC70 representing the remaining viable cancer cells after exposure to T cells. The results were collected from four independent experiments. Original magnification, ×100 and scale bar=50 µm. *P<0.05, **P<0.01 and ***P<0.001. MSLN, mesothelin; MSLN-SmartDC, MSLN dendritic cells; RPS3, ribosomal protein subunit 3; 3D, three dimensional; MFI, mean fluorescence intensity.
Discussion
The lack of ER/PR and HER2 in TNBC limits the available treatment to systemic chemotherapy and surgical resection (1). Targeting the protein overexpressed in TNBC such as MSLN is currently an active area of immunotherapy (14,29). The high expression of MSLN in TNBC reported in the present study was consistent with previous studies, with the range of MSLN ~37–67% (14,18–22). The DCs-based immunotherapy is another potential approach for the TNBC treatment that could promote the antigen-specific immune response which leading to clinical response as observed in various types of cancer including melanoma, leukemia, cholangiocarcinoma and pancreatic cancer (6,8–10,13,24). In the present study, self-differentiated DCs presenting MSLN antigen, termed MSLN-SmartDC, was developed and shown to promote the MSLN-specific immune response against TNBC. The effect of RPS3, a TLR4 ligand, on MSLN-SmartDC immunophenotype and T cells activation capability was significantly enhanced. As high MSLN expression was confirmed in most TNBC cases, the MSLN-specific T cell production by MSLN-SmartDC platform is suggested as an alternative T cell treatment in patients with TNBC.
Several studies have reported the utility of DCs generated by lentiviral transduction of cytokine genes for DCs differentiation and by tumor-associated antigen gene to induce antigen-specific immune response leading to the tumor growth inhibition (8–12). The MSLN-SmartDC of the present study demonstrated consistent immunophenotypes with DCs generated by recombinant GM-CSF and IL-4 observed by the significant downregulation of CD14 monocyte marker, while the markers of DCs such as CD40, CD83, CD86 and HLA-DR were significantly upregulated. Moreover, the increased MSLN-SmartDC maturation profile by upregulation of the costimulatory molecules; CD40, CD80, CD83 and IL-12p70 production were observed. Although the secretion of GM-CSF and IL-4 were found to be higher in IRFP-SmartDC compared with MSLN-SmartDC which may be explained by the smaller tri-cistronic mRNA size produced by IRFP-SmartDC (30). Nevertheless, these higher GM-CSF and IL-4 levels secreted by IRFP-SmartDC did not confer the immunophenotype differences as seen by a comparable mature DCs markers expression compared with MSLN-SmartDC. CD40, CD80 and CD83 increment in RPS3-MSLN-SmartDC compared with MSLN-SmartDC supported previous findings that RPS3 could activate DC maturation (26). These characteristics suggested the potential of higher T cell activation of RPS3-MSLN-SmartDC compared with MSLN-SmartDC. The upregulation of CD40 in matured DCs is required for the DCs licensing by CD40L-expressing T cells which further augment the costimulatory molecules and cytokine production initiated by RPS3 treatment (31–33). This is the limitation of the present study, but it may be explained that RPS3 induces MSLN-SmartDC maturation and the upregulation of CD40 can further augment the maturation initiated by TLR4 signaling pathway (33).
The T cells characteristics following co-culturing with MSLN-SmartDC with or without RPS3 showed slightly changed in frequencies of CD4+, CD8+ and the memory T cells subsets. This may be due to the effect of cytokines used during T cells expansion process which can non-specifically promote T cell proliferation (34,35). However, the presence of MSLN-specific CD8+ T cells recognizes HLA-A2 restricted MSLN antigenic peptides (13,28) in cells activated by MSLN-SmartDC and RPS3-MSLN-SmartDC compared with the negative control conditions. Although there was a trend toward increased MSLN-specific T cells in RPS3-MSLN-SmartDC compared with other conditions, it did not achieve the statistically significant levels. The addition of RPS3 to MSLN-SmartDC could not affect the frequency of MSLN-specific T cells. It is possible that the upregulated factors found in RPS3-MSLN-SmartDC, including CD40, CD80 and IL-12p70, exert their effect on the quality of antigen-specific T cells; in particular the cytotoxicity function rather than the quantity or the frequency of T cells (31,36,37). This may be supported by the significant increase of IFN-γ+ and CD69+ IFN-γ+ T cells driven by RPS3-MSLN-SmartDC. T cells activated by MSLN-SmartDC or RPS3-MSLN-SmartDC promoted TNBC cell killing in effector cells and antigen-dependent manners. These findings are in agreement with previous studies in SmartDC system in different antigens and cancer models (8–11). Moreover, T cells activated by RPS3-MSLN-SmartDC demonstrated enhanced cytolytic activity against MSLN expressing cancer cells. This was associated with the increased IFN-γ production and may explain effective target cells lysis by RPS3-MSLN-activated T cells.
The cytolytic activity of T cells activated by MSLN-SmartDC in 3D-cancer spheroid was consistent with that observed in 2D culture system. To minimize the effect of non-specific T cells killing mediated by HLA-mismatched between the target cells and T cells, healthy donors with HLA-A2 partially matched with MDA-MB-231, but not HCC70 (HLA-A3), were selected. Using MSLN-MDA-MB-231 in comparison with parental MDA-MB-231 could eliminate the intrinsic factors of target cells that may interfere with the T cells cytolytic activity, except the presence of MSLN. Future study using the ex vivo generated SmartDC and T cells to kill patient-derived TNBC cells can eliminate this limitation. The addition of RPS3 in MSLN-SmartDC trend toward increased cancer cell cytotoxicity in HCC70. Collectively, the obtained findings of the present study highlighted the efficacy of T cells activated by MSLN-SmartDC to eliminate MSLN-expressing TNBC cells and the addition of RPS3 to MSLN-SmartDC prior co-cultured with T cells enhances T cells cytolytic activity against the TNBC cells.
The MSLN-specific T cells in cancer patients have been reported (28,38,39). Different approaches of MSLN targeted treatment demonstrate safety and efficacy in several types of cancer (16,29,40). Targeting MSLN in TNBC using T cells activated by MSLN-SmartDC and RPS3-MSLN-SmartDC may provide a potential safe and effective treatment for patients with TNBC. It is known that the immune response against the cancer cells gradually declines with age (41,42). The use of RPS3-MSLN-SmartDC treatment which utilizes patient's immune cells in younger patients might be both compatible with more aggressive nature of TNBC and also the more competent immune response in young patients with TNBC. The response of MSLN-SmartDC or MSLN-specific T cell treatment in luminal and HER2+ patients should be the same as patients with TNBC, if they expressed high level of MSLN in the cancer cells. The current trend in breast cancer treatment approach has now shifted from the monotherapy to the combinational treatment involving several interventions such as surgery, radiation, chemotherapy and immunotherapy (1). Combination of treatments targeting the bulk tumor mass or the stromal cells and reducing the immunosuppressive signal in tumor microenvironment via surgery, chemotherapy and checkpoint inhibitor together with the DCs-based immunotherapy that promote the antigen-specific T cells could result in significant improved clinical outcome (43–46). Therefore, further investigation of MSLN-SmartDC and RPS3-MSLN-SmartDC in combination with other treatment approaches is a great promise for the novel treatment modality in patients with TNBC.
In conclusion, the efficacy of MSLN-SmartDC promoting MSLN-specific immune response killing MSLN-expressing TNBC cells was successfully developed. The MSLN-SmartDC maturation enhancement by RPS3 treatment can improve the cytolytic activity of T cells against high MSLN TNBC cells. Though the clinical use of MSLN-SmartDC and RPS3-MSLN-SmartDC needs more convincing data in the in vivo where the effect of tumor microenvironment on this treatment could be assessed and in the ex vivo system where the fully-matched HLAs type could be done, the present findings demonstrated the potential of these DCs to activate MSLN-specific T cells as an alternative treatment for patients with TNBC.
Supplementary Material
Acknowledgements
The authors gratefully thank Miss Surat Phumphuang (Department of Immunology, Faculty of Medicine Siriraj Hospital, Mahidol University, Thailand) for her effort in clinicopathological data collection. The authors would also like to thank Emeritus Professor James A. Will (senior editor for the Faculty of Medicine, Khon Kaen University) for the English version of the present manuscript.
Funding Statement
This work has been funded by Midcareer Research Grant (grant no. RSA6280091), National Research Council of Thailand to CT; Graduate Grant, National Research Council of Thailand (grant no. N41D640036) and Siriraj Graduate Scholarship to NJ and TRF-IRN Scholarship (Scholarship number IRN5801PHDW05) to WC.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Authors' contributions
CT and PY conceived the study. NJ, TC, PT, PY and CT designed the experiments. NJ, ST and WC performed the experiments. DS and MW performed data resource and analysis. NJ, ST and CT analyzed and interpreted the data. CT supervised the overall research. NJ and CT confirm the authenticity of all raw data. NJ, ST and CT wrote the manuscript. CT reviewed and/or edited the manuscript. All authors reviewed and approved the final manuscript.
Ethics approval and consent to participate
All experiments in this study were approved by Siriraj Institutional Review Board (COA no. Si580/2018) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All blood donors in this study provided written informed consent for the use of blood samples for research.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Waks AG, Winer EP. Breast cancer treatment: A review. JAMA. 2019;321:288–300. doi: 10.1001/jama.2018.19323. [DOI] [PubMed] [Google Scholar]
- 2.Reddy SM, Barcenas CH, Sinha AK, Hsu L, Moulder SL, Tripathy D, Hortobagyi GN, Valero V. Long-term survival outcomes of triple-receptor negative breast cancer survivors who are disease free at 5 years and relationship with low hormone receptor positivity. Br J Cancer. 2018;118:17–23. doi: 10.1038/bjc.2017.379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cheung KL. Treatment strategies and survival outcomes in breast cancer. Cancers (Basel) 2020;12:735. doi: 10.3390/cancers12030735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Arnedos M, Roulleaux Dugage M, Perez-Garcia J, Cortes J. Window of opportunity trials for biomarker discovery in breast cancer. Curr Opin Oncol. 2019;31:486–492. doi: 10.1097/CCO.0000000000000583. [DOI] [PubMed] [Google Scholar]
- 5.Falzone L, Grimaldi M, Celentano E, Augustin LSA, Libra M. Identification of modulated micrornas associated with breast cancer, diet, and physical activity. Cancers (Basel) 2020;12:2555. doi: 10.3390/cancers12092555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Saxena M, Bhardwaj N. Re-emergence of dendritic cell vaccines for cancer treatment. Trends Cancer. 2018;4:119–137. doi: 10.1016/j.trecan.2017.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bernhard H, Neudorfer J, Gebhard K, Conrad H, Hermann C, Nährig J, Fend F, Weber W, Busch DH, Peschel C. Adoptive transfer of autologous, HER2-specific, cytotoxic T lymphocytes for the treatment of HER2-overexpressing breast cancer. Cancer Immunol Immunother. 2008;57:271–280. doi: 10.1007/s00262-007-0355-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Panya A, Thepmalee C, Sawasdee N, Sujjitjoon J, Phanthaphol N, Junking M, Wongkham S, Yenchitsomanus PT. Cytotoxic activity of effector T cells against cholangiocarcinoma is enhanced by self-differentiated monocyte-derived dendritic cells. Cancer Immunol Immunother. 2018;67:1579–1588. doi: 10.1007/s00262-018-2212-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sundarasetty BS, Chan L, Darling D, Giunti G, Farzaneh F, Schenck F, Naundorf S, Kuehlcke K, Ruggiero E, Schmidt M, et al. Lentivirus-induced ‘smart’ dendritic cells: Pharmacodynamics and GMP-compliant production for immunotherapy against TRP2-positive melanoma. Gene Ther. 2015;22:707–720. doi: 10.1038/gt.2015.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sundarasetty BS, Singh VK, Salguero G, Geffers R, Rickmann M, Macke L, Borchers S, Figueiredo C, Schambach A, Gullberg U, et al. Lentivirus-induced dendritic cells for immunization against high-risk WT1(+) acute myeloid leukemia. Hum Gene Ther. 2013;24:220–237. doi: 10.1089/hum.2012.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bialek-Waldmann JK, Domning S, Esser R, Glienke W, Mertens M, Aleksandrova K, Arseniev L, Kumar S, Schneider A, Koenig J, et al. Induced dendritic cells co-expressing GM-CSF/IFN-α/tWT1 priming T and B cells and automated manufacturing to boost GvL. Mol Ther Methods Clin Dev. 2021;21:621–641. doi: 10.1016/j.omtm.2021.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pincha M, Sundarasetty BS, Salguero G, Gutzmer R, Garritsen H, Macke L, Schneider A, Lenz D, Figueiredo C, Blasczyk R, et al. Identity, potency, in vivo viability, and scaling up production of lentiviral vector-induced dendritic cells for melanoma immunotherapy. Hum Gene Ther Methods. 2012;23:38–55. doi: 10.1089/hgtb.2011.170. [DOI] [PubMed] [Google Scholar]
- 13.de Goeje PL, Klaver Y, Kaijen-Lambers MEH, Langerak AW, Vroman H, Kunert A, Lamers CHJ, Aerts JGJV, Debets R, Hendriks RW. Autologous dendritic cell therapy in mesothelioma patients enhances frequencies of peripheral CD4 T cells expressing HLA-DR, PD-1, or ICOS. Front Immunol. 2018;9:2034. doi: 10.3389/fimmu.2018.02034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tchou J, Wang LC, Selven B, Zhang H, Conejo-Garcia J, Borghaei H, Kalos M, Vondeheide RH, Albelda SM, June CH, Zhang PJ. Mesothelin, a novel immunotherapy target for triple negative breast cancer. Breast Cancer Res Treat. 2012;133:799–804. doi: 10.1007/s10549-012-2018-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Le K, Wang J, Zhang T, Guo Y, Chang H, Wang S, Zhu B. Overexpression of mesothelin in pancreatic ductal adenocarcinoma (PDAC) Int J Med Sci. 2020;17:422–427. doi: 10.7150/ijms.39012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Haas AR, Tanyi JL, O'Hara MH, Gladney WL, Lacey SF, Torigian DA, Soulen MC, Tian L, McGarvey M, Nelson AM, et al. Phase I study of lentiviral-transduced chimeric antigen receptor-modified T cells recognizing mesothelin in advanced solid cancers. Mol Ther. 2019;27:1919–1929. doi: 10.1016/j.ymthe.2019.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hassan R, Ho M. Mesothelin targeted cancer immunotherapy. Eur J Cancer. 2008;44:46–53. doi: 10.1016/j.ejca.2007.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Parinyanitikul N, Blumenschein GR, Wu Y, Lei X, Chavez-Macgregor M, Smart M, Gonzalez-Angulo AM. Mesothelin expression and survival outcomes in triple receptor negative breast cancer. Clin Breast Cancer. 2013;13:378–384. doi: 10.1016/j.clbc.2013.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tozbikian G, Brogi E, Kadota K, Catalano J, Akram M, Patil S, Ho AY, Reis-Filho JS, Weigelt B, Norton L, et al. Mesothelin expression in triple negative breast carcinomas correlates significantly with basal-like phenotype, distant metastases and decreased survival. PLoS One. 2014;9:e114900. doi: 10.1371/journal.pone.0114900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bayoglu IV, Kucukzeybek BB, Kucukzeybek Y, Varol U, Yildiz I, Alacacioglu A, Akyol M, Demir L, Dirican A, Yildiz Y, et al. Prognostic value of mesothelin expression in patients with triple negative and HER2-positive breast cancers. Biomed Pharmacother. 2015;70:190–195. doi: 10.1016/j.biopha.2015.01.019. [DOI] [PubMed] [Google Scholar]
- 21.Li YR, Xian RR, Ziober A, Conejo-Garcia J, Perales-Puchalt A, June CH, Zhang PJ, Tchou J. Mesothelin expression is associated with poor outcomes in breast cancer. Breast Cancer Res Treat. 2014;147:675–684. doi: 10.1007/s10549-014-3077-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Suzuki T, Yamagishi Y, Einama T, Koiwai T, Yamasaki T, Fukumura-Koga M, Ishibashi Y, Takihata Y, Shiraishi T, Miyata Y, et al. Membrane mesothelin expression positivity is associated with poor clinical outcome of luminal-type breast cancer. Oncol Lett. 2020;20:193. doi: 10.3892/ol.2020.12055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vermaelen K. Vaccine strategies to improve anti-cancer cellular immune responses. Front Immunol. 2019;10:8. doi: 10.3389/fimmu.2019.00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mehrotra S, Britten CD, Chin S, Garrett-Mayer E, Cloud CA, Li M, Scurti G, Salem ML, Nelson MH, Thomas MB, et al. Vaccination with poly(IC:LC) and peptide-pulsed autologous dendritic cells in patients with pancreatic cancer. J Hematol Oncol. 2017;10:82. doi: 10.1186/s13045-017-0459-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chow LQM, Morishima C, Eaton KD, Baik CS, Goulart BH, Anderson LN, Manjarrez KL, Dietsch GN, Bryan JK, Hershberg RM, et al. Phase Ib trial of the Toll-like receptor 8 agonist, motolimod (VTX-2337), combined with cetuximab in patients with recurrent or metastatic SCCHN. Clin Cancer Res. 2017;23:2442–2450. doi: 10.1158/1078-0432.CCR-16-1934. [DOI] [PubMed] [Google Scholar]
- 26.Park HJ, Jang GY, Kim YS, Park JH, Lee SE, Vo MC, Lee JJ, Han HD, Jung ID, Kang TH, Park YM. A novel TLR4 binding protein, 40S ribosomal protein S3, has potential utility as an adjuvant in a dendritic cell-based vaccine. J Immunother Cancer. 2019;7:60. doi: 10.1186/s40425-019-0539-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Höpken UE, Lehmann I, Droese J, Lipp M, Schüler T, Rehm A. The ratio between dendritic cells and T cells determines the outcome of their encounter: Proliferation versus deletion. Eur J Immunol. 2005;35:2851–2863. doi: 10.1002/eji.200526298. [DOI] [PubMed] [Google Scholar]
- 28.Thomas AM, Santarsiero LM, Lutz ER, Armstrong TD, Chen YC, Huang LQ, Laheru DA, Goggins M, Hruban RH, Jaffee EM. Mesothelin-specific CD8(+) T cell responses provide evidence of in vivo cross-priming by antigen-presenting cells in vaccinated pancreatic cancer patients. J Exp Med. 2004;200:297–306. doi: 10.1084/jem.20031435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Del Bano J, Florès-Florès R, Josselin E, Goubard A, Ganier L, Castellano R, Chames P, Baty D, Kerfelec B. A bispecific antibody-based approach for targeting mesothelin in triple negative breast cancer. Front Immunol. 2019;10:1593. doi: 10.3389/fimmu.2019.01593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fernandes LD, Moura APS, Ciandrini L. Gene length as a regulator for ribosome recruitment and protein synthesis: Theoretical insights. Sci Rep. 2017;7:17409. doi: 10.1038/s41598-017-17618-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ara A, Ahmed KA, Xiang J. Multiple effects of CD40-CD40L axis in immunity against infection and cancer. Immunotargets Ther. 2018;7:55–61. doi: 10.2147/ITT.S163614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tay NQ, Lee DCP, Chua YL, Prabhu N, Gascoigne NRJ, Kemeny DM. CD40L expression allows CD8+ T cells to promote their own expansion and differentiation through dendritic cells. Front Immunol. 2017;8:1484. doi: 10.3389/fimmu.2017.01484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Michael Dohnal A, Luger R, Paul P, Fuchs D, Felzmann T. CD40 ligation restores type 1 polarizing capacity in TLR4-activated dendritic cells that have ceased interleukin-12 expression. J Cell Mol Med. 2009;13((8B)):1741–1750. doi: 10.1111/j.1582-4934.2008.00584.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ross SH, Cantrell DA. Signaling and function of interleukin-2 in T lymphocytes. Annu Rev Immunol. 2018;36:411–433. doi: 10.1146/annurev-immunol-042617-053352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Drake A, Kaur M, Iliopoulou BP, Phennicie R, Hanson A, Chen J. Interleukins 7 and 15 maintain human T cell proliferative capacity through STAT5 signaling. PLoS One. 2016;11:e0166280. doi: 10.1371/journal.pone.0166280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Agarwal P, Raghavan A, Nandiwada SL, Curtsinger JM, Bohjanen PR, Mueller DL, Mescher MF. Gene regulation and chromatin remodeling by IL-12 and type I IFN in programming for CD8 T cell effector function and memory. J Immunol. 2009;183:1695–1704. doi: 10.4049/jimmunol.0900592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen L, Flies DB. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nat Rev Immunol. 2013;13:227–242. doi: 10.1038/nri3484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen Y, Ayaru L, Mathew S, Morris E, Pereira SP, Behboudi S. Expansion of anti-mesothelin specific CD4+ and CD8+ T cell responses in patients with pancreatic carcinoma. PLoS One. 2014;9:e88133. doi: 10.1371/journal.pone.0088133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhenjiang L, Rao M, Luo X, Sandberg E, Bartek J, Jr, Schoutrop E, von Landenberg A, Meng Q, Valentini D, Poiret T, et al. Mesothelin-specific immune responses predict survival of patients with brain metastasis. EBioMedicine. 2017;23:20–24. doi: 10.1016/j.ebiom.2017.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang J, Khanna S, Jiang Q, Alewine C, Miettinen M, Pastan I, Hassan R. Efficacy of anti-mesothelin immunotoxin RG7787 plus nab-paclitaxel against mesothelioma patient-derived xenografts and mesothelin as a biomarker of tumor response. Clin Cancer Res. 2017;23:1564–1574. doi: 10.1158/1078-0432.CCR-16-1667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zirbes A, Joseph J, Lopez JC, Sayaman RW, Basam M, Seewaldt VL, LaBarge MA. Changes in immune cell types with age in breast are consistent with a decline in immune surveillance and increased immunosuppression. J Mammary Gland Biol Neoplasia. 2021;26:247–261. doi: 10.1007/s10911-021-09495-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hamilton JAG, Henry CJ. Aging and immunotherapies: New horizons for the golden ages. Aging Cancer. 2020;1:30–44. doi: 10.1002/aac2.12014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Waldman AD, Fritz JM, Lenardo MJ. A guide to cancer immunotherapy: From T cell basic science to clinical practice. Nat Rev Immunol. 2020;20:651–668. doi: 10.1038/s41577-020-0306-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bulgarelli J, Tazzari M, Granato AM, Ridolfi L, Maiocchi S, de Rosa F, Petrini M, Pancisi E, Gentili G, Vergani B, et al. Dendritic cell vaccination in metastatic melanoma turns ‘non-T cell inflamed’ into ‘T-cell inflamed’ tumors. Front Immunol. 2019;10:2353. doi: 10.3389/fimmu.2019.02353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sawasdee N, Thepmalee C, Sujjitjoon J, Yongpitakwattana P, Junking M, Poungvarin N, Yenchitsomanus PT, Panya A. Gemcitabine enhances cytotoxic activity of effector T-lymphocytes against chemo-resistant cholangiocarcinoma cells. Int Immunopharmacol. 2020;78:106006. doi: 10.1016/j.intimp.2019.106006. [DOI] [PubMed] [Google Scholar]
- 46.Kodumudi KN, Ramamoorthi G, Snyder C, Basu A, Jia Y, Awshah S, Beyer AP, Wiener D, Lam L, Zhang H, et al. Sequential anti-PD1 therapy following dendritic cell vaccination improves survival in a HER2 mammary carcinoma model and identifies a critical role for CD4 T cells in mediating the response. Front Immunol. 2019;10:1939. doi: 10.3389/fimmu.2019.01939. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.