Abstract
Background
Cancer is considered as the second deadliest disease globally. Plants have continuously offered unique secondary metabolites with remarkable biological applications. Lignans have gained great importance due to their biological activity. Previous studies revealed that the most remarkable bioactivity of lignan class of molecules is anticancer. They are derived from the oxidative dimerization of two phenylpropanoid units. This review covers the isolated anticancer lignans and their mechanistic aspects.
Main body
A bibliographic investigation was performed by analyzing the information available on anticancer lignans in the internationally accepted scientific databases including Web of Science, SciFinder, PubMed, Scopus, and Google Scholar. In this review we have tried to sum up the isolated anticancerous lignan, its source, active plant part, extract and various cell lines used to establish different studies. Here we have included a total number of 113 natural lignans. Many studies that mainly performed in human cell lines have reported. Very few plants have been evaluated for their in vivo anticancer activity.
Conclusion
It can be concluded that in near future the lignans may be an effective pharmacon for the treatment of cancer. Fruitful areas of future research may be in modifying natural lignans or synthesizing new lignans with structural diversity and potent pharmacological activities. Extensive studies are needed to be done highlighting the mechanism of anticancer action of explored and unexplored plants. The data will definitely attract many researchers to start further experimentation that might lead to the drugs for the cancer treatment.
Graphical Abstract
Keywords: Lignan, Anticancer plants, Podophyllotoxin, Cytotoxicity
Background
There is a great burden of disease internationally and cancer is in the top priority due to its high incidence rate that causes disability and premature mortality among human populations [1].
Cancer is not a single disease but it is a group of 100 different and distinguishing disorders that affect the entire physiological balances [2]. It is an uncontrolled growth of cells that have damaged DNA expression [3]. If the spread of these abnormal cells is not managed with certain means, it can lead to worse situations or may be death. These abnormal cells are termed as cancer cells, malignant cells, or tumor cells. Many cancers that comprises of abnormal cells are further recognized by the name of the organ that the abnormal cells originated from (for example, breast cancer, lung cancer, prostate cancer, and colorectal cancer). There are various kinds of cancers depends upon the type of genes associated with specific cancer like sarcomas, carcinomas, leukemia, and lymphomas. Carcinogenesis is a multi-leveled process consists of three noticeable stages, i.e., initiation, promotion, and progression [4]. It is the prime result of disturbances that occurred in two types of genes, tumor suppressor genes (TSG) and oncogenes.
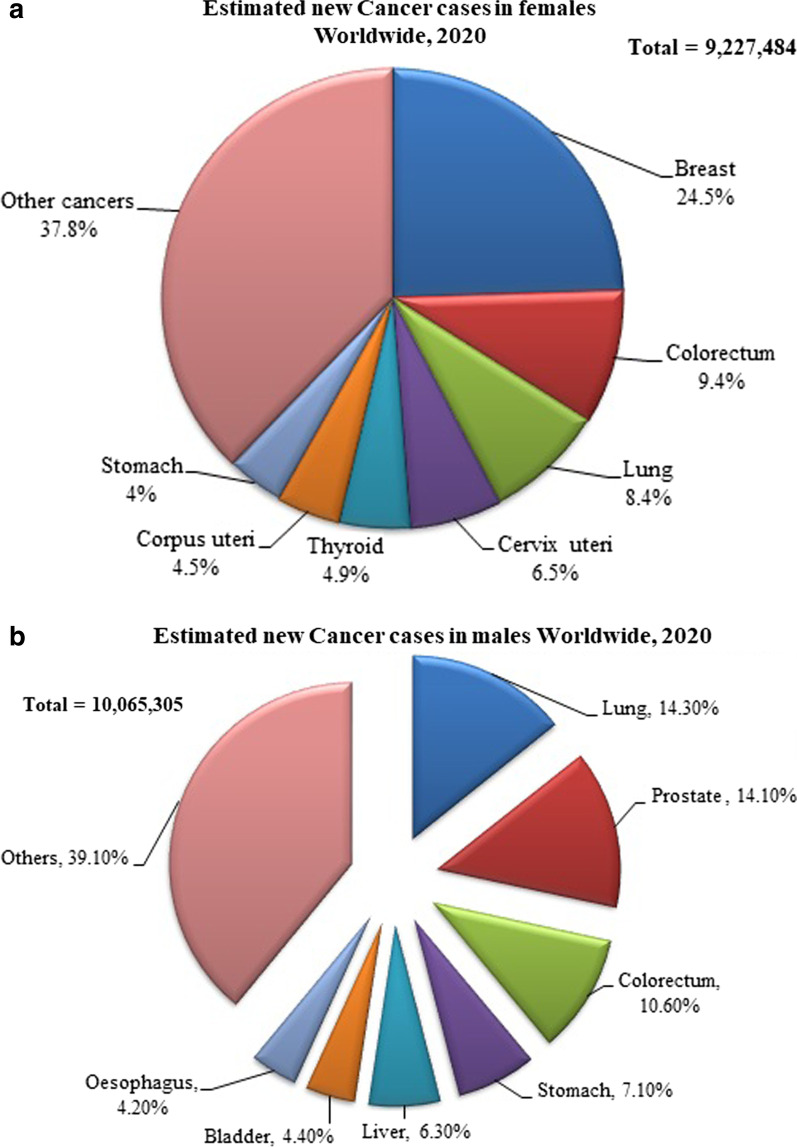
Deaths from cancer are rising continuously worldwide with an estimated 11.5 million deaths in 2030 [5]. The International Agency for Research on Cancer (IARC) estimated a shocking number of 19.3 million new cases including every possible distribution criteria (Fig. 1a, b) and approx 10 million of reported death worldwide [6].
Fig. 1.
a Global cancer cases distribution types including all age groups of females. Source: GLOBOCAN, 2020. b Global cancer cases distribution types including all age groups of males. Source: GLOBOCAN, 2020
Globally, non-communicable diseases (NCDs) accounted for 71% of total deaths. In India, NCDs were estimated to account for 63% of all deaths, and cancer was one of the leading causes (9%). The projected number of patients with cancer in India is 1,392,179 for the year 2020, and the common five leading origins are breast, lung, mouth, cervix uteri, and tongue [7]. Persons with any type of existing cancer are prone to get affected with coronavirus (SARS-CoV-2), and it is a deadly combination for individuals [8]. Studies revealed that prostate and breast cancer constitutes major types of cancer found, respectively, in men and women [9]. In children the blood cancer and the cancers related to the brain and lymph nodes are more frequent than other types of cancer [10, 11]. There are certain risk factors that increase the development of cancer in any person such as ageing, tobacco, ionizing radiation, some chemical compounds, some viruses and bacteria, alcohol consumption, family history of cancer, certain hormones, and overweight [12].
The treatment options of cancer involve surgery of tumor, radiotherapy and chemotherapy depends upon the stage and location of tumor [13]. But these treatments are very costly and require highly specialized health professionals [14]. Additionally, these chemotherapeutic agents are not free from side effects like myelosuppression, mucositis, alopecia, cardiotoxicity, neurotoxicity, immunosuppression, etc. An ideal anticancer drug would specifically be cytotoxic toward the cancer cells only and research findings suggests that phytochemicals and their derivatives are emerging alternatives for better and less toxic chemotherapeutic agents [13].
Various active compounds such as podophyllotoxin, vincristine, vinblastine, taxol, etc., have been isolated from plants, and these molecules acted as lead metabolites to modify and yield analogues better than the parent compound for activity with low toxicity and improved bioavailability [15–17].
There are diverse classes of secondary metabolites which are biosynthesized by plants and, among them, lignans are identified as the major group of natural products with a broad range of important bioactivities.
Main text
Lignans are the class of plant secondary metabolites derived from the phenylpropanoid pathway and was first introduced by Haworth [18]. They play an important role in plant protection and are also proved to be fruitful in human nutrition and medicine [19]. The chief sources of dietary lignans are various vegetables and fruits, legumes, whole grain cereals, and oilseeds [20, 21]. Sesame and flax seeds are the edible plant components which are the most concentrated sources of lignans [22].
Chemistry of lignans
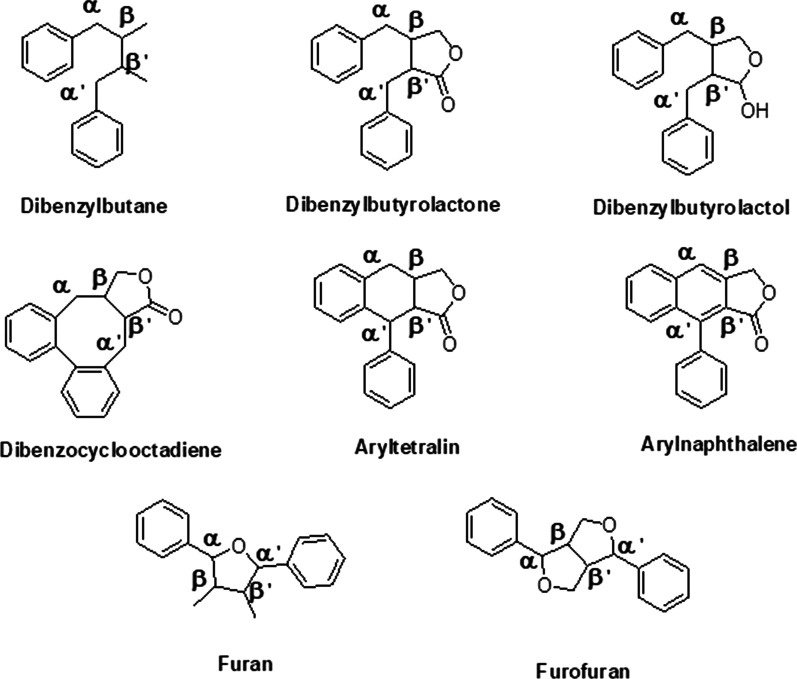
It is well-established that the supergroup of natural phenolics is biosynthesized through the shikimic acid pathway. The biodiversity of this lignan class of molecules is found in various parts of more than 60 families of plants and they are potential bioactive principles toward cancerous cells. Beside their cytotoxic property they are also useful to treat diabetes, oxidation of living cells as antioxidants, cardiovascular diseases, microbial infections, and other major or minor inflammatory responses [23, 24]. As per the earlier findings, the basic structure of lignan contains the nine carbon (in a C6-C3 fusion) phenylpropane unit (Fig. 2a) from cinnamyl structures [25] which was redefined by Haworth [18] as dimer of C6-C3 unit via β-β′ bonding (Fig. 2b). Besides this basic hydrocarbon skeleton they possess numerous additional side groups either in the form of aliphatic or aromatic origin and they are classified accordingly. There are eight subtypes of major lignans (Fig. 3) such as dibenzylbutane (e.g., Enterodiol), dibenzylbutyrolactone (e.g., Enterolactone), dibenzylbutyrolactol (e.g., Gnetucleistol F), dibenzocyclooctadiene (e.g., Gomisins), aryltetralin (e.g., Podophyllotoxins), arylnaphthalene (e.g., Justcidins), furan (e.g., Belischmins), and furofuran (e.g., Epimagnolin) derivatives. Except these eight subtypes, they are also diversified based on the presence or absence of oxygen [26, 27]. Hybrid lignans are molecules which have other secondary metabolites like flavonoids (flavolignans), coumarins (coumarinolignans), xanthones (xantholignans), stilbenes (stilbenolignans), etc., and possess lignan like biological and chemical properties.
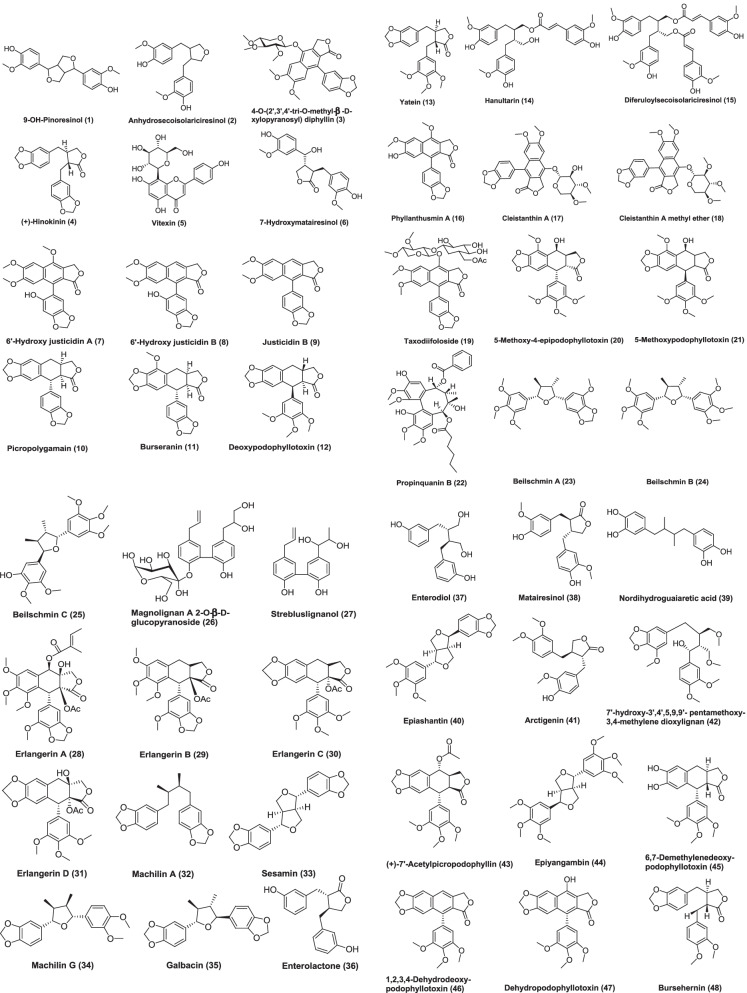
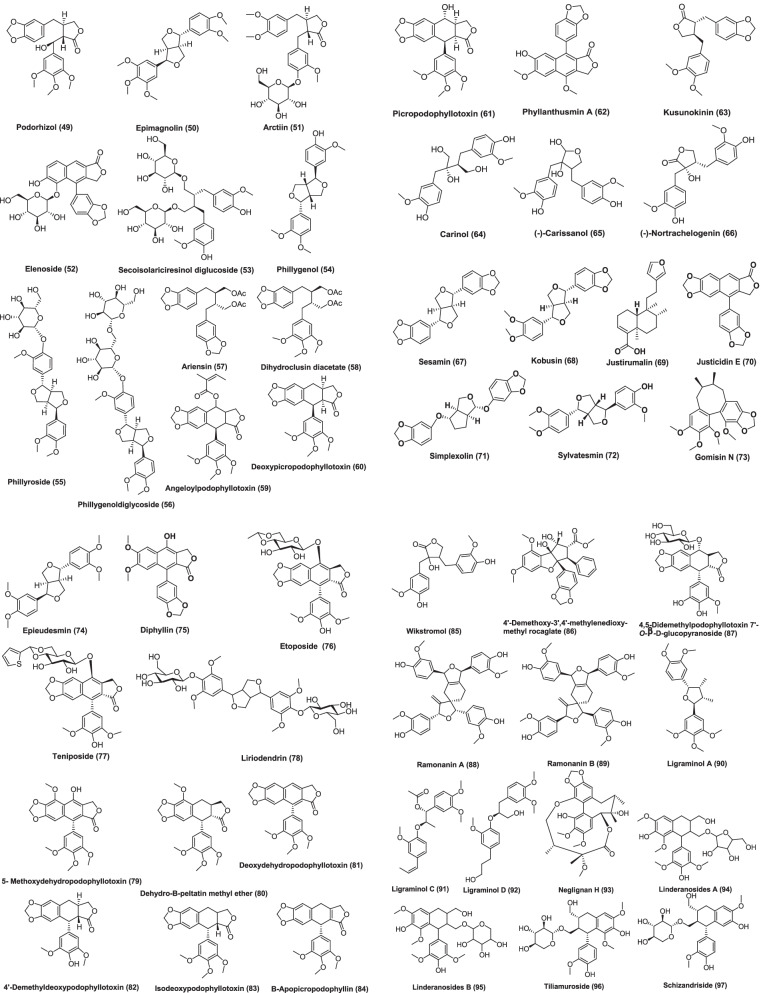
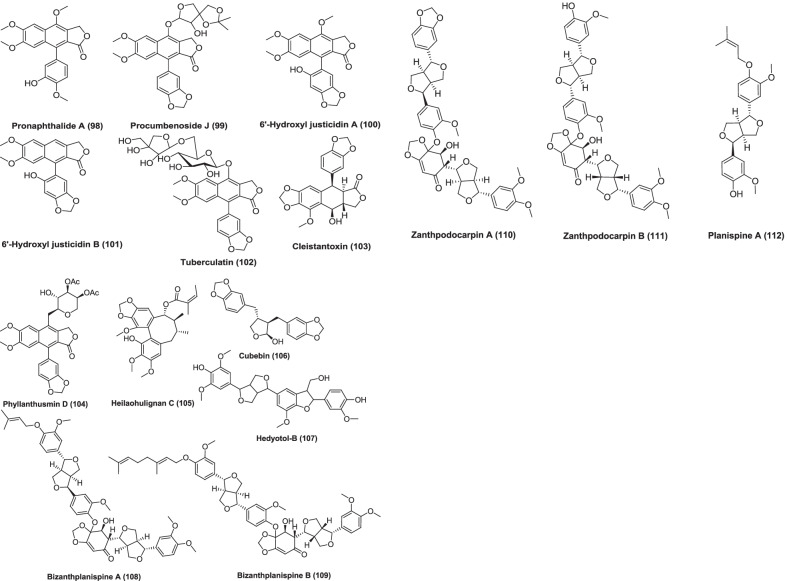
Fig. 4.
Chemical structures of anticancer isolated lignans from plants
Fig. 2.

a Phenylpropane unit. b Dimer of C6-C3 unit via β-β′ bonding
Fig. 3.
Eight chemical classes of lignan molecules
The discovery of Podophyllotoxins as gold standard in leading lignans along with establishing its cytotoxic property and topoisomerase-II inhibitory potentials helped the research community to develop other clinically important drugs like etoposide, teniposide, clinical candidates like Etopophos, NK611, GL331, etc. [28]. Ward reported a total number of 83 synthetic and transformational schemes including stereospecific and asymmetric consideration [29] to obtain them in laboratory.
There is persistent interest in the cancer-protective effects of lignans, which have been shown to have an advantageous anti-tumor effect throughout the early phases of carcinogenesis. The present review, summarizes the recent literature which deals with the lignans isolated from plants having anticancer potential with their reported mechanism of action which are listed in Table 1. Lignans has been considered as the promising anticancer agents.
Table 1.
List of lignans isolated from plant with anticancer activity
| Name | Structure ID (Fig. 4) | Source | Extract | Part | Cancer cell line used (in vitro) | IC50 | In vivo | Dose | Comments | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| 9-OH-Pinoresinol | 1 | Saussurea salicifolia (L.) DC, Asteraceae | Chloroform fraction of ethanolic extract | Aerial parts | L5178Y | – | – | 10 μg/mL | Ethanolic extract of the plant reduced the growth of leukemia mouse lymphoma cells to 23.8%. It looks like lignan 9-OH-pinoresinol is responsible for the activity which is well known from other plant sources | [30] |
| Anhydrosecoisolariciresinol | 2 | Linum usitatissimum, Linaceae | – | Seeds | MCF-7 | 100 µM | The isolated lignan causes 30% inhibition of cell growth as compared to control | [31] | ||
| 4-O-(2′,3′,4′-tri-O-methyl-β-D-xylopyranosyl) diphyllin | 3 | Phyllanthus taxodiifolius, Phyllanthaceae | – | Aerial parts | HCT116 | 0.08 ± 0.03 µM | In vitro studies has been shown to inhibit the growth of a number of cancer cell. It shows strongest antiproliferative effect on HCT116 cells. The compound induces apoptosis in HCT116 cells by activating caspase-3 pathway and antiproliferative effect is due to promotion of microtubule depolymerization | [32] | ||
| ( +)-Hinokinin | 4 | Wikstroemia lanceolata, Thymelaeaceae | Methanol | Stems and roots | P-388 | 1.54 µg/mL (ED50) | Showed significant cytotoxic activity | [33] | ||
| Vitexin | 5 | Vitex negundo, Verbenaceae | Ethanol | Seeds | MCF-7, ZR-75-1, SK-BR-7, MDA-MB-231, MDA-MB-435s, PC-3, LNCaP and COC1 | 100 mg/kg | In vivo studies done using tumor xenograft models like MCF-7, MA782, MDA-MB-435s, and T47D xenografts for breast, PC-3 for prostate, HeLa cells for cervical, and HepG2 for liver xenograft | Vitexins (lignan mixture) has cytotoxic effects on MCF-7, ZR-75-1, SK-BR-7, MDA-MB-231, MDA-MB-435s, PC-3, LNCaP COC1 cancer cells. Vitexin induced antitumor effect and cytotoxic activity is exerted through proapoptotic process, which is mediated by a decreased Bcl-2/Bax ratio and activation of caspases | [34] | |
| 7-Hydroxymatairesinol (HMR) | 6 | Picea abies, Pinaceae | Acetone–water (9:1) | Heartwood | LNCaP human prostate cancer xenografts in athymic nude male mice | There is significant decrease in tumor volume. A control diet supplemented with 0.15% or 0.30% of HMR was administered to mice and the tumor take rate and growth was observed for 9 weeks. The diet supplemented with HMR has been shown to inhibit the growth of LNCaP tumors. Mice treated with HMR had smaller tumor volume, lower tumor take rate, increased proportion of non-growing tumors, and higher tumor cell apoptotic index compared with controls. Cell proliferation index was also decreased in mice receiving the 0.30% HMR diet when compared with mice receiving the control diet | [35] | |||
| 6′-Hydroxy justicidin A, 6′-hydroxy justicidin B, justicidin B | 7, 8, 9 | Justicia procumbens, Acanthaceae | Ethanolic | Whole plant | K562 | 20, 43.9 and 45.4 µM | All the compounds significantly inhibited the growth of K562 cells by decreasing both proliferation and SOD activity and inducing apoptosis in dose-dependent manner. Activation of caspase-3 pathway suggests that these compounds induce apoptosis through caspase intrinsic or extrinsic pathway | [36] | ||
| Picropolygamain, Burseranin | 10, 11 | Bursera graveolens, Burseraceae | Methanol | Stem | HT1080 | 1.9, 5.5 µg/mL (ED50) | Showed significant cytotoxic activity | [37] | ||
| (-)-Deoxypodophyllotoxin, (-)-yatein | 12, 13, | Hernandia nymphaeifolia, Hernandiaceae | Methanol | Bark | P-388, KB16, A549, HT-29 | < 1 µg/mI (ED50) | Showed significant cytotoxic activity | [38] | ||
| Hanultarin, 1,4-O-Diferuloylsecoisolariciresinol | 14, 15 | Trichosanthes kirilowii, Cucurbitaceae | 80% Aqueous methanol | Seeds | A549, SK-Mel-2, B16F1 | 3–13 µg/mL | Inhibitory effect on the polymerization of the actin cytoskeleton in normal epidermal keratinocyte (HaCaT cells) has been shown by compound Hanultarin as compared to those of the other isolates | [39] | ||
| Phyllanthusmin A | 16 | Phyllanthus oligospermus, Phyllanthaceae | Chloroform fraction of methanolic extract | Stems and roots | KB and P-388 | 2.24 µg/mL and 0.13 µg/mL | Showed significant cytotoxic activity | [40] | ||
| Cleistanthin A, Cleistanthin A methyl ether, Taxodiifoloside | 17, 18, 19 | Phyllanthus taxodiifolius, Euphorbiaceae | Ethanol fraction of methanol extract | Aerial parts | Five cultured mammalian cell lines. P-388, KB, Col-2, MCF-7 and Lu-1 | Compounds showed GI50 value in the range 10–6–10–9 M | Cleistanthin A, Cleistanthin A methyl ether has shown potent cytotoxic activity and Taxodiifoloside showed moderate activity | [41] | ||
| 5-Methoxy-4-epipodophyllotoxin, 5-methoxypodophyllotoxin | 20, 21 | Libocedrus chevalier, Cupressaceae | Ethyl acetate | Bark | KB | 45 µM and 11 µM | Both isolated lignans were also evaluated for their tubulin assembly inhibitory activity. 5-methoxy-4-epipodophyllotoxin inhibited the assembly of tubulin into microtubules with an IC50 of 9 µM, whereas the IC50 of 5-methoxypodophyllotoxin was 5 µM | [42] | ||
| Propinquanin B | 22 | Schisandra propinqua (Wall.), Schisandraceae | Chloroform | Stems | HL-60, Hep-G2, R- Hep-G2, KB, Bel-7402 | 7.15, 9.81, 14.00, 11.70, 18.81 µM | Cell cycle study and Hoechst 33,258 staining assay suggests that cytotoxic activity of compound may be due to induction of apoptosis | [43] | ||
|
Beilschmin A, Beilschmin B, Beilschmin C |
23, 24, 25 | Beilschmiedia tsangii, Lauraceae | - | Stems | P-388 and HT-29 |
1.2 and 5.0 µg/mL 2.2 and 5.1 µg/mL 3.6 and 10.5 µg/mL |
Showed significant cytotoxic activity | [44] | ||
| Magnolignan A 2-O-β-D-glucopyranoside, Strebluslignanol | 26, 27 | Streblus asper, Moraceae | Chloroform fraction of 75% ethanol | Heartwood | Hep-2 and Hep-G2 | 13.3 μM, 46.4 μM and 10.1 μM, 21.7 μM | Both lignans showed medium cytotoxic activity | [45] | ||
| Erlangerin A to D | 2, 29, 30, 31 | Commiphora erlangeriana, Burseraceae | Resin | EAhy926 and HeLa, L929 and RAW 264.7 |
68 ± 6, 40 ± 5, 90 ± 5 and 44 ± 9 µg/mL (EC50) 23 ± 1.4, 4.0 ± 1.4, 68 ± 6 and 28 ± 0.3 (EC50) 0.16 ± 0.09, 0.55 ± 0.007, 5.6 ± 1.5 (EC25), and 0.97 ± 0.21 µg/mL (EC50) 0.026 ± 0.007, 0.026 ± 0.009, 3.5 ± 1 µg/mL(EC25), 0.11 ± 0.017 µg/mL (EC50) |
Erlangerins C and D were similar to podophyllotoxin on the basis of their structure and biological activity so may have same mechanism of action. They induced a concentration-dependent cytotoxicity in RAW 264.7 and cytostatic effect in HeLa, EAhy926, and L929 cells. But Erlangerins A and B suppressed cell viability at relatively higher concentrations when compared with Erlangerin C and D | [46] | |||
|
Machilin A, (-)-Sesamin, Machilin G, ( +)-Galbacin |
32, 33, 34, 35 | Machilus thunbergii, Lauraceae | Dichloromethane | Bark | HCT-15, MCF-7 and A549 |
12.4, 12.4 and 7.9 µM 4.4, 3.4 and 11.0 µM 1.4, 2.7 and 8.3 µM 6.2, 7.9 and 7.9 µM |
PLCγ1 plays a key role in proliferation and progression of human cancer. These compounds inhibit PLCγ1 and showed strong antiproliferative activity | [47] | ||
| Enterolactone, Enterodiol | 36, 37 | Mammalian lignans | LNCaP | 57 mM and 100 mM | 10–100 microM | Growth of prostate cancer cells were suppressed may be by hormonally dependent and independent mechanisms | [48] | |||
| Matairesinol | 38 | Carthamus tinctorius, Asteraceae | – | Seeds | HL-60 | 60 μM | DNA content histogram was analyzed by flow cytometry and it showed rapid increase in subdiploid cells and a concomitant decrease in diploid cells exposed to 100 μM matairesinol. It was concluded that cell death was due to the DNA damage and apoptosis | [49] | ||
| Nordihydroguaiaretic acid | 39 | Larrea tridentata DC. Coville, Zygophyllaceae | Resinous exudate | Bush | SW480 | 1.9 ± 0.5 µg | It caused time and dose-dependent loss of mitochondrial membrane potential (MMP), down regulation of the anti-apoptotic protein bclxl and an increase of the apoptotic index. It also induced a shift of the culture population to the G2/M phase of the cell cycle | [50] | ||
| Epiashantin | 40 | Artemisia absinthium L., Asteraceae | – | Warmwood | SW480 | 9.8 ± 4.5 µM | The compound caused a time and dose-dependent loss of mitochondrial membrane potential (MMP), down regulation of the anti-apoptotic protein bclxl and an increase of the apoptotic index | [50] | ||
| Arctigenin | 41 | Arctium lappa L., Asteraceae | – | Root | SW480 | 16.5 ± 8.5 µM | The compound caused a time and dose-dependent loss of mitochondrial membrane potential (MMP), down regulation of the anti-apoptotic protein bclxl and an increase of the apoptotic index | [50] | ||
| 7′-Hydroxy-3′,4′,5,9,9′-pentamethoxy-3,4-methylene dioxylignan | 42 | Phyllanthus urinaria, Phyllanthaceae | Ethyl acetate | Whole plant | HEp-2 | 4.46 µM | 7′-hydroxy-3′,4′,5,9,9′-pentamethoxy-3,4-methylene dioxylignan was capable of inhibiting telomerase activity and also could inhibit bcl2 and activate caspase 3 and caspase 8 whose significance in the induction of apoptosis is well known | [51] | ||
| ( +)-7′-Acetylpicropodophyllin, Epiyangambin | 43, 44 | Hernandia ovigera L., Hernandiaceae | Ethyl acetate | Twigs | JB6 | 0.15 and 0.4.2 µg/mL | Significant inhibition of the transformation of murine epidermal JB6 cells, | [52] | ||
|
Deoxypodophyllotoxin, 6,7 Demethylenedeoxypodophyllotoxin, 1,2,3,4-Dehydrodeoxypodophyllotoxin, Dehydropodophyllotoxin, Bursehernin, Podorhizol, Epimagnolin |
12, 45, 46, 47, 48, 49, 50 | Hernandia ovigera L., Hernandiaceae | Seeds | Epstein-Barr virus early antigen activation (EBV-EA) induced by 12-O tetradecanoylphorbol 13-acetate (TPA) in Raji cells |
550 mol ratio/32 pmol TPA, 510 520 470 470 480 590 |
Inhibitory effects on EBV activation has been shown by all isolated compounds | [53] | |||
|
Arctiin, Arctigenin |
51, 41 | Saussurea medusa, Composite | Methanol | Aerial parts | Two stage skin carcinogenesis model using DMBA (7,12-dimethylbenz[a]anthracene) and TPA (12-O-tetradecanoyl phorbol-13-acetate) | Both lignans arctiin and arctigenin exhibited a significant inhibitory effect on the tumor promotion induced by DMBA and TPA by both topical application and oral administration. When both compounds were administered orally reduction in papillomas per mouse at 15 weeks of promotion in case of arctigenin was 4.2 ± 0.1 and Arctiin 4.0 ± 0.2, and at 20 weeks of promotion arctigenin was 6.1 ± 0.1 and Arctiin was 6.1 ± 0.2 | [54] | |||
| Elenoside | 52 | Justicia hyssopifolia L., Acanthaceae | Ethanolic | Leaves | CCRFCEM, K-526, MOLT-4, RPMI-8226 | 79–97% growth inhibition | 10–4 M | Elenoside was cytotoxic to leukemic cell lines (CCRFCEM, K-526, MOLT-4, RPMI-8226) at a concentration of 10–4 M (79–97% growth inhibition). Elenoside does not show significant activity at concentration less than 10–4 | [55] | |
| Secoisolariciresinol diglycoside | 53 | Linum usitatissimum, Linaceae | Ethanolic | Seeds | Female Sprague–Dawley rats | 2.93 mmoles/g |
Increased plasma insulin-like growth factor I (IGF-I) concentrations are associated with increased breast cancer risk. Secoisolariciresinol diglycoside reduced plasma IGF-I levels. It inhibit Mammary tumor development in rats |
[56] | ||
|
Phillygenol Phillyroside Phillygenoldiglycoside |
54, 55, 56 | Lancea tibetica, Mazaceae | SMMC-7721, HeLa, V79, B16 | Phillygenol has shown strong cytotoxic activity on the tested cell lines whereas Phillyroside and Phillygenoldiglycoside had little effect on the proliferation of the tested cell lines | [57] | |||||
| Podophyllotoxins | 45, 46, 47… | Podophyllum peltatum, Podophyllum emodi, Podophyllum versipelle, Linum Juniperus | small-cell lung cancer (SCLC) dose: > 1 µg/mL (etoposide) | Disrupt the organization of the karyokinetic spindle single-strand and double-strand breaks in DNA through their interactions with DNA topoisomerase II induce cell cycle arrest in the G2-phase of the cell cycle | [17] | |||||
|
Ariensin Burseran Dihydroclusin diacetate |
57, 11, 58 | Bursera microphylla A. Gray, Burseraceae | Methanol | Resin obtained from the bark of the plant | RAW264.7, M12.C3.F6 murine cancer cell line (macrophages transformed by virus Abelson leukemia) | 9.8, 0.4, 0.2 μM for all three isolated compounds in RAW264.7 and 2.5 μM for Dihydroclusin diacetate in M12.C3.F6 | Dihydroclusin diacetate was shown to be active against both murine cancer cell lines while ariensin, burseran, were active against only RAW246.7 murine cell line only | [58] | ||
| (-)-Hinokinin | 4 | Zanthoxylum pistaciiflorum Hayata, Rutaceae | Methanol | Stem Bark | HT-29 cell line | 3.52 µg/mL (ED50 value) | Showed significant cytotoxic activity against HT-29 cell line | [59] | ||
|
(-)-Deoxypodophyllotoxin, Angeloylpodophyllotoxin, Deoxypicropodophyllotoxin, Picropodophyllotoxin |
12, 59, 60, 61 | Anthriscus sylvestris Hoffm., Umbelliferae | Methanol | Roots | HL-60 | Compounds have an apoptosis-inducing effect in HL-60 cells and it was determined by caspase-3 activation and DNA fragmentation. Typical ladders of DNA fragmentation were observed when treated with compound angeloylpodophyllotoxin, picropodophyllotoxin at 1 mM and (-)-Deoxypodophyllotoxin at 0.01 mM | [60] | |||
| Phyllanthusmin A | 62 | Phyllanthus oligospermus, Phyllanthaceae | Chloroform fraction of methanol extract | Stems and roots | KB and P-388 | 2.24 and 0.13 µg/mL | Phyllanthusmin A showed significant cytotoxicity | [61] | ||
| (-)-Kusunokinin | 63 | Piper nigrum, Piperaceae | Dichloromethane | Fruits | MCF-7 and MDA-MB-468 | 1.18 and 1.62 µg/mL | This compound induced cell apoptosis and drove cells toward the G2/M phase which is determined by cell studies. It also decreases topoisomerase II and Bcl-2. There is increase in p53, p21, bax, cytochrome c, and caspase-8, -7, and -3 activities, except caspase-9. This shows that kusunokinin has potent anticancer activity through the extrinsic pathway and G2/M phase arrest | [62] | ||
| Yatein | 13 | Austrocedrus chilensis, Cupressaceae | Methanol | Heartwood | P3X63-Ag8.653 | Yatein exhibited potent cytotoxicity, inducing 75% cell death at 25 mg/mL after 24 h of treatment | Yatein showed toxicity in P3X cells in a dose-dependently. In cells that survived to yatein treatment, the microtubular apparatus was altered, as determined by immunofluorescence techniques, and SEM and TEM analyses displayed changes in morphological and ultrastructural level. There was alteration in cell shape and membrane system was damaged | [63] | ||
|
(-)-Carinol, (-)-Carissanol, and (-)-Nortrachelogenin |
64, 65, 66 | Carissa spinarum L., Apocynaceae | Methanol | Stem | MCF7 and A549 |
< 1 µg/mL 11.0 and 17.4 µg/mL 29.0 and 88.3 µg/mL |
The most active lignan was (-)-carinol and (-)-carissanol was more potent than (-)-nortrachelogenin | [64] | ||
|
Sesamin, Kobusin, 4′O Demethyl magnolin |
67, 68 | Zanthoxylum alatum, Rutaceae | Petroleum ether | Stem bark | A549 and MIA-PaCa |
37.46 ± 1.097 and 34.04 ± 1.7621 34.71 ± 2.331 and 32.86 ± 2.0271 26.47 ± 1.871 and 26.47 ± 1.871 mg/mL |
Cytotoxic activity has been shown by all three isolated lignans in different ranges. 4′O dimethyl magnolin was the novel bioactive compound from a plant source and found to be most active. In apoptosis study, treatment caused typical apoptotic morphological changes. It enhances the apoptosis at IC50 dose (21.72 mg/mL) on MIA-PaCa cell line. This compound induce apoptosis as the mechanism of cell death | [65] | ||
| Justirumalin | 69 | Justicia neesii, Acanthaceae | MCF-7, AGS | 42.8 and 42.1%, inhibition, respectively | 25 μg/mL | Justirumalin inhibited human stomach and breast cancer cells | [66] | |||
|
Justicidin E, Simplexolin |
70 | Justi caorbiculata, Acanthaceae | MCF-7, SF-268, CNS, NCI-H460, HCT-116 and AGS | 25 μg/mL | Justicidin E inhibited the proliferation of lung, breast and colon cancer cell lines with inhibition values ranged between 40 and 53% and simplexolin gave 40–50% inhibition against lung, breast, colon, and CNS cancer cell lines when tested at 25 µg/mL | [66] | ||||
| Sylvatesmin | 72 | Lancea tibetica Hook. f. et Thoms, Scrophulariaceae | Methanol | Whole plant | B16, SMMC-7721, Hela | 40.4 ± 1.4 mg/mL, 113.4 ± 2.16 mg/mL, 127.9 ± 3.20 mg/mL | 25 µg/mL | Sylvatesmin exhibited the effective antitumor activity, especially on B16 cells | [67] | |
| Gomisin N | 73 |
Schisandra chinensis (Turcz.) Baill., Schisandraceae or Magnoliaceae |
Dichloromethane | Ripe berries | HT-29 | 43 µM | Effective against colorectal proliferative processes | [68] | ||
| Epieudesmin | 74 | Hernandia nymphaeifolia (Presl) Kubitzki, Hernandiaceae | CH3OH/CH2Cl2 (1:1) extract | Fruits | A549, MCF-7 and HER2, MDA-MB-231 | 5.7 µM, 8.1 µM, 231 8.2 µM | Compounds displayed significant anti proliferative activity | [69] | ||
| Podophyllotoxins, Diphyllin, Etoposide (VP–16), teniposide | 12, 75, 76, 77 | Podophyllum peltatum, Berberidaceae | Whole plant | P-388, HT-29, A-549 and MEL-28 |
This bioactive lignan is very effective on small cell lung cancer, malignant lymphoma, and testicular carcinoma It is also potent on Wilms tumors, ovarian cancer, brain tumors, urinary tract cancer, etc. |
[70] | ||||
| Liriodendrin | 78 | Plumeria rubra, Apocynaceae | Water soluble fraction of methanolic extract | Stem bark | P-388 murine lymphocytic leukemia and human cancer cell types (fibrosarcoma, melanoma, breast, lung, colon and KB) |
P-388—2.4 µg/mL Fibrosarcoma—98.9 µg/mL Melanoma—19 µg/mL Breast cancer—30 µg/mL Lung cancer—6.0 µg/mL Colon cancer—16 µg/mL KB—6.0 µg/mL (ED50 values) |
Exhibit cytotoxic activity | [71] | ||
|
5- Methoxydehydro podophyllotoxin, dehydro-β-peltatin methyl ether, Dehydropodophyllotoxin, Deoxydehydropodophyllotoxin, Yatein, 4′-Demethyldeoxypodophyllotoxin, Isodeoxypodophyllotoxin, Deoxypicropodophyllin, β-apopicropodophyllin |
79, 80, 47, 81, 13, 82, 83, 60, 84 | Hyptis verticillata, Lamiaceae | Chloroform | Aerial parts |
P-388, HT-low, KB, A43l, ZR-75-1, LNCaP and U373 |
4.0, 15.6, 6.0, 6.2, > 20, 11.6 and 16.3 µg/mL 1.8, 3.4, 2.2, > 20, > 20, 3.2, and 5.9 µg/mL > 5, 9.7, 5.0, > 20, > 20, 11.7 and > 20 µg/mL > 5, > 20, 11.4, 6.2, > 20, 11.6 and > 20 µg/mL 0.4, 0.07, 0.08, > 20, 0.5, 0.16, and 0.3 µg/mL 0.005, 0.01, 0.01, 0.08, 2.1, 0.02 and 0.1 µg/mL > 20, 10.7, 6.7, 6.2, 13.2, 12.0 and 2.9 µg/mL 0.1, 0.2, 0.1, > 20, 0.6, 0.2 and 0.1 µg/mL 0.002, 0.003, 0.05, 4.3, 2.0, 0.01 and 0.001 µg/mL. (ED50 values) |
[72] | |||
| Wikstromol | 85 | Wikstroemia foetida var.oahuensis and Wikslroemia uwa-ursi Gray Thymelaeaceae | Chloroform fraction of ethanolic extract | Whole plant | P-388 Iympho cyticleukemia (3PS) test system | 16, 10, 4, 2, and 1 mg/kg | Wikstromol demonstrate activities of 154, 146, 137, 141, and 130% test/control at dose of 16, 10, 4, 2, and 1 mg/kg, respectively | [73] | ||
| 4′-Demethoxy-3′,4′-methylenedioxy-methyl rocaglate | 86 | Aglaia elliptica Bl., Meliaceae | Chloroform | Stem | HT-1080, KB, A431, LNCaP, ZR-75-1, and U373, BCl | 10.0, 6.0, 10.0, 2.0, 2.0, 0.8, 0.9 ng/mL | Antitumor potential of compound was performed with female Balb/c athymic nude mice. Compound significantly inhibited the growth of BC1 cells in culture. The growth of tumor was retarded by treatment with isolated compound during the first 23 days of the study, but after that tumor growth paralleled to the control group | This compound acts by cytostatic mechanism, rather than inducing necrosis or apoptosis. Cells were transiently blocked in the G1/G0 phases of the cycle, and this may be due to inhibition of protein biosynthesis | [74] | |
| 4,5-Didemethylpodophyllotoxin 7′-O-b-D-glucopyranoside | 87 | Sinopodophyllum emodi, Berberidaceae | n-butanol | Roots and rhizomes | Hela, K562, SH-SY5Y and CNE | Compound showed cytotoxicity against four human cancer cell lines | [75] | |||
|
Ramonanin A Ramonanin B |
88, 89 | Guaiacum officinale, Zygophyllaceae | Chloroform | Heartwood | MD-MBA 231 | 18 μM | The ramonanins exhibit cytotoxic activity against human breast cancer cell lines with an IC50 value of 18 μM and induce cell death via apoptotic mechanisms | Ramonanin A-treated MD-MBA 231 cells showed characteristic features of apoptotic cell death, which appeared in a time and dose-dependent manner and cell cycle distribution was monitored via flow cytometry using fluorescence-activated cell sorting. It was noted that the ramonanins strongly disrupt cell cycle progression at the G1/S phase transition | [76] | |
|
Ligraminol A, Ligraminol C, Ligraminol D |
90, 91, 92 | Acorus gramineus, Araceae | Methanol | Rhizomes | A549, SK-OV-3, SK-MEL-2 | 6.92, 9.44, and 4.53 μM | Compounds showed weak inhibitory activity against various cancerous cell lines. Study has also been performed to check whether the cytotoxicity was selective between tumor and normal cells. For this compounds were evaluated for normal human cell line, HUVEC. This was noted that cytotoxicity of isolated compounds was higher against tumor cells than normal cells. Ligraminol A showed the highest selective cytotoxicity against the SK-MEL-2 cell | [77] | ||
| Neglignan H | 93 | Schisandra neglecta, Schisandraceae | Ethyl acetate layer of 70% aqueous acetone | Stem | NB4, A549 and MCF7 | 8.1, 7.4 and 6.7 µM | [78] | |||
| Linderanosides A and B | 94, 95 | Lindera glauca, Lauraceae | Methanolic | Twigs | A549 | 20.86 ± 0.94, 21.85 ± 0.61 µM | [79] | |||
|
Tiliamuroside, Schizandriside |
96, 97 | Tilia amurensis Rupr., Tiliaceae | Methanolic | Trunk | A549, SK-OV-3, SK-MEL-2, and HCT-15 |
7.32, 8.89, 7.84, and 6.18 μM 6.90, 5.88, 3.26, and 6.65 μM |
cytotoxic activity of compounds against the tested cell lines were due to absence of a methoxy group at C-3 in the aryl-tetralin type lignan as indicated by the results | [80] | ||
| Pronaphthalide A, Procumbenoside J, 6′-hydroxyl justicidin A, 6′-hydroxyl justicidin B, Tuberculatin | 98, 99, 100, 101, 102 | Justica procumbens, Acanthaceae | Ethanol | Whole plants | Human LoVo and BGC-823 | 0.03–10.0 μM, | [81] | |||
| Cleistantoxin | 103 | Cleistanthus indochinensis, Euphorbiaceae | Dichloromethane | Fruits | KB, MCF-7, MCF-7R | 0.022, 0.036, 0.014 μM | Cleistantoxin had strong activity against KB cells also showed significant activity against MCF-7 and MCF-7R | [82] | ||
| Phyllanthusmin D | 104 | Phyllanthus poilanei, Phyllanthaceae | Chloroform fraction of methanol extract | Air-dried leaves, twigs, flowers, and fruits | HT-29 | 170 nM | Compound showed activity when tested in an in vivo hollow fiber assay using HT-29 cells implanted in immunodeficient NCr nu/nu mice | 5 μM |
Cytotoxic effects of phyllanthusmin D were by inducing tumor cell apoptosis through activation of caspase-3. DNA topoisomerase IIα activity was not inhibited Treatment of HT-29 cells with phyllanthusmin D for 72 h resulted in 28.2% or 30.3% of HT-29 cells undergoing early apoptosis, respectively, |
[83] |
| Heilaohulignan C | 105 | Kadsura coccinea, Schisandraceae | 80% ethanol | Roots | HepG-2, BGC-823 and HCT-116 | 9.92, 16.75 and16.59 µM | heilaohulignan C showed good cytotoxicity in HepG-2 cancer cells and weak cytotoxicity against BGC-823 and HCT-116 cancer cells | [84] | ||
| (-)-Cubebin | 106 | Piper cubeba, Piperaceae | Acetone | Seeds | A549, K562, SiHa, KB | 8.30 ± 0.16, 8.66 ± 0.43, 8.16 ± 0.41 µM | [85] | |||
| Hedyotol-B | 107 | Herpetospermum pedunculosum, Cucurbitaceae | Ethyl acetate | Stems | SGC7901, A549 | 1.7 ± 0.1 and 6.1 ± 0.5 μM | Hedyotol-B displayed potent inhibitory effect against gastric and lung carcinoma | [86] | ||
| Bizanthplanispine A and B, Zanthpodocarpin A and B, Planispine A | 108, 109, 110, 111, 112 | Zanthoxylum planispinum Sieb., Rutaceae | 95% aqueous MeOH | Roots | Hela, HL-60, PC-3 | Bizanthplanispine A and B, zanthpodocarpin A and B showed significant reduction in the proliferation of Hela with IC50 values ranging from 15.00 to 26.44 µg/mL. Planispine A showed the strongest inhibition on the growth of HL-60 and PC-3 with IC50 values of 4.90 and 23.45 µg/mL | All isolated compounds showed inhibitory effect on different cancer cell lines | [87] |
L5178Y, leukemia mouse lymphoma cells; MCF-7, breast cancer cell lines; HCT116, human colon carcinoma cell lines; P-388, leukemia cancer cells; SK-BR-7, breast cancer cells; MDA-MB-231, breast cancer cells; MDA-MB-435s, breast cancer cells; LNCaP, prostate cancer cells; COC1, ovarian cancer cells; K562, human chronic myeloid leukemia; HT1080, human fibrosarcoma cells; KB16, human epidermoid carcinoma cells; HT-29 human colorectal adenocarcinoma cell line; A549, human lung cancer cell line; SK-Mel-2, human skin melanoma cell lines; B16F1, mouse melanoma cell lines; Col-2, human colon cancer cell lines; Lu-1, lung adenocarcinoma cell line; HL-60, human acute promyelocytic leukemia cell line; Hep-G2, human hepatocellular carcinoma; R-Hep-G2, human resistant hepatoma; Bel-7402, hepatocellular carcinoma; Hep-2 alveolar epithelial carcinoma cell line; EAhy926, human umbilical vein cell line; HeLa, human uterine cervix carcinoma cell lines; L929, murine fibroblast cell line; RAW 264.7, murine macrophage cell line; HCT-15, human colorectal carcinoma cell line; SW480, colon carcinoma cells; JB6, murine epidermal cells; CCRFCEM, leukemia cell lines; K-526, leukemia cell lines; MOLT-4, leukemia cell lines; RPMI-8226, leukemia cell lines; SMMC-7721, human hepatoma cell line; V79, hamster lung fibroblast cell; B16, mouse melanoma cell; M12.C3.F6, murine cancer cell line (macrophages transformed by virus Abelson leukemia); MDA-MB-468, breast cancer cell lines; P3X63-Ag8.653, murine myeloma cell line; AGS, gastric cancer cell lines; MIA-PaCa, pancreatic carcinoma cell line; SMMC-7721, human hepatoma cells; HER2, negative breast cancer cell line; MDA-MB-231, triple negative breast cancer cell line; MEL-28, melanoma cell lines; HT-low, Human fibrosarcoma cells; KB, human oral epidermoid carcinoma; A43l, human epidermoid carcinoma; ZR-75–1, human hormone-dependent breast cancer; U373, human glioblastoma cell lines; BCl, human breast cancer; SH-SY5Y, neuroblastoma cell line; CNE, nasopharyngeal carcinoma; SK-OV-3, ovary malignant ascites; NB4, human acute promyelocytic leukemia cell line; MCF-7R, human breast cancer cell line; SiHa, human cervical carcinoma; SGC7901, human gastric carcinoma; PC-3, human prostate carcinoma cells; SCLC, small-cell lung cancer
Material and methods
The bibliography was crucially analyzed from worldwide established scientific databases like SCOPUS, PubMed, ScienceDirect, Springerlink, Web of Science, Wiley, SciFinder, and Google Scholar. The botanical names of these selected plant species were verified from the plant list. The inclusion criteria for the selection of data are lignans isolated from Medicinal plants with reported anticancer activity. Both the reviews and the research articles on medicinal plants are considered. The search terms were lignans, anticancer plants containing lignans, chemistry of lignans without narrowing or limiting search items.
Conclusions
Lignans are secondary metabolites are also phenolic in nature and have diversity in biological activities. Previous studies revealed that the most remarkable bioactivity of lignan class of molecules are antioxidant and anticancer. This review covers a considerable number of naturally obtained lignans that are reported to have anticancer potential. In this review we have tried to sum up the isolated anti-cancerous lignan, its source, active plant part, extract and various cell lines used to establish different studies. Here we have included a total 113 numbers of natural lignans. Many studies that mainly performed in human cell lines have reported inhibition of enzymes that retards tumor growth. Very few plants have been evaluated for their in vivo anticancer activity.
It can be concluded that in near future the lignans may be an effective pharmacon for the treatment of cancer. Fruitful areas of future research may be in modifying natural lignans or synthesizing new lignans with structural diversity and potent pharmacological activities. However, among the vast numbers of existing plants on this planet, only a few species have been studied so far for their anticancer principles. Extensive studies are needed to be done highlighting the mechanism of anticancer action of explored and unexplored plants.
Potent anticancer lignans reported in this review needed to be further explored in clinical trials on different models for their effectiveness, toxicological studies, and also targeting particular genotoxic profile against a wide range of cancer in both in vitro and in vivo. These compounds are obtained from plants in very minute quantities so this is one of the main challenges to be addressed in the future and their total synthesis in order to allow further bioactivity studies. The data will definitely attract many researchers to start further experimentation that might lead to the drugs for the cancer treatment and to manufacture new herbal drugs which have significant anticancer potential.
Acknowledgements
We express our sincere thanks to Management of the institute for providing facilities.
Author contributions
MM participated in conceptualization and writing of the manuscript. BCJ, PSB contributes in editing. AB, ANS review the final version of article. All authors read and approved the final manuscript.
Funding
Not applicable.
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Shih AH, Abdel-Wahab O, Patel JP, Levine RL. The role of mutations in epigenetic regulators in myeloid malignancies. Nat Rev Cancer. 2012;12(9):599–612. doi: 10.1038/nrc3343. [DOI] [PubMed] [Google Scholar]
- 2.Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100(1):57–70. doi: 10.1016/s0092-8674(00)81683-9. [DOI] [PubMed] [Google Scholar]
- 3.Gilbert S (2011) From the Toxipedia website in original form. Last updated by Toxipedia in 2011. https://www.healthandenvironment.org/docs/ToxipediaCancerPageArchive.pdf
- 4.Kaur S, Pandit K, Chandel M, Kaur S. Antiproliferative and apoptogenic effects of Cassia fistula L. n-hexane fraction against human cervical cancer (HeLa) cells. Environ Sci Pollut Res Int. 2020;27(25):32017–32033. doi: 10.1007/s11356-020-08916-9. [DOI] [PubMed] [Google Scholar]
- 5.World health organization (2009) NMH Fact sheet January 2010. https://www.who.int/nmh/publications/fact_sheet_cancers_en.pdf
- 6.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J Clin. 2021;71(3):209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 7.Mathur P, Sathishkumar K, Chaturvedi M, Das P, Sudarshan KL, Santhappan S, Nallasamy V, John A, Narasimhan S, Roselind FS. Cancer statistics, 2020: report from National Cancer Registry Programme, India. JCO Glob Oncol. 2020;6:1063–1075. doi: 10.1200/go.20.00122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang Q, Berger NA, Xu R. Analyses of risk, racial disparity, and outcomes among US patients with cancer and COVID-19 infection. JAMA Oncol. 2021;7(2):220. doi: 10.1001/jamaoncol.2020.6178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Siegel RL, Miller KD, Jemal A. Cancer statistics 2016. CA Cancer J Clin. 2016;66:7–30. doi: 10.3322/caac.21332. [DOI] [PubMed] [Google Scholar]
- 10.Schottenfeld D, Fraumeni JF. Cancer epidemiology and prevention. England: Oxford University Press; 2006. [Google Scholar]
- 11.Yoo KY, Shin HR. Cancer epidemiology and prevention. Korean J Epidemiol. 2003;25:1–15. [Google Scholar]
- 12.Cunha JP (2021) Medicine Net. Cancer risk factors and causes. Medically Reviewed on 5/17/2021. https://www.medicinenet.com/cancer_causes/article.htm
- 13.Singh S, Sharma B, Kanwar SS, Kumar A. Lead phytochemicals for anticancer drug development. Front Plant Sci. 2016 doi: 10.3389/fpls.2016.01667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tariq A, Mussarat S, Adnan M. Review on ethnomedicinal, phytochemical and pharmacological evidence of Himalayan anticancer plants. J Ethnopharmacol. 2015;164:96–119. doi: 10.1016/j.jep.2015.02.003. [DOI] [PubMed] [Google Scholar]
- 15.Ambrosio SR, Tirapelli CR, da Costa FB, de Oliveira AM. Kaurane and pimarane-type diterpenes from the Viguiera species inhibit vascular smooth muscle contractility. Life Sci. 2006;79(10):925–933. doi: 10.1016/j.lfs.2006.05.015. [DOI] [PubMed] [Google Scholar]
- 16.Choudhari AS, Mandave PC, Deshpande M, Ranjekar P, Prakash O. Phytochemicals in cancer treatment: from preclinical studies to clinical practice. Front Pharmacol. 2020;10:1614. doi: 10.3389/fphar.2019.01614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lichota A, Gwozdzinski K. Anticancer activity of natural compounds from plant and marine environment. Int J Mol Sci. 2018;19(11):3533. doi: 10.3390/ijms19113533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Haworth RD. The chemistry of the lignan group of natural products. J Chem Soc. 1942 doi: 10.1039/JR9420000448. [DOI] [Google Scholar]
- 19.Adlercreutz H. Lignans and human health. Crit Rev Clin Lab Sci. 2007;44(5–6):483–525. doi: 10.1080/10408360701612942. [DOI] [PubMed] [Google Scholar]
- 20.Durazzo A, Zaccaria M, Polito A, Maiani G, Carcea M. Lignan content in cereals, buckwheat, derived foods. Foods. 2013;2:53–63. doi: 10.3390/foods2010053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Durazzo A, Turfani V, Azzini E, Maiani G, Carcea M. Phenols, lignans and antioxidant properties of legume and sweet chestnut flours. Food Chem. 2013;140(4):666–671. doi: 10.1016/j.foodchem.2012.09.062. [DOI] [PubMed] [Google Scholar]
- 22.Landete J. Plant and mammalian lignans: a review of source, intake, metabolism, intestinal bacteria and health. Food Res Int. 2012;46(1):410–424. doi: 10.1016/j.foodres.2011.12.023. [DOI] [Google Scholar]
- 23.Rodríguez-García C, Sánchez-Quesada C, Toledo E, Delgado-Rodríguez M, Gaforio J. Naturally lignan-rich foods: a dietary tool for health promotion? Molecules. 2019;24(5):917. doi: 10.3390/molecules24050917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sok DE, Cui H, Kim M. Isolation and boactivities of furfuran type lignan compounds from edible plants. Recent Pat Food Nutr Agric. 2009;1(1):87–95. doi: 10.2174/2212798410901010087. [DOI] [PubMed] [Google Scholar]
- 25.Robinson R. The relationship of some complex natural products to the simple and amino acids. Proc Univ Durh Philos Soc. 1928;8:14–59. [Google Scholar]
- 26.Calvo-Flores FG, Dobado JA, Isac-García J, Martín-Martínez FJ. Lignin and lignans as renewable raw materials: chemistry, technology and applications. New York: Wiley; 2015. [Google Scholar]
- 27.Cunha WR, Silva MLA, Veneziani RCS, Ambrósio SR, Bastos JK (2012) Lignans: chemical and biological properties. www.intechopen.com.
- 28.Yousefzadi M, Sharifi M, Behmanesh M, Moyano E, Bonfill M, Cusido RM, Palazon J. Podophyllotoxin: current approaches to its biotechnological production and future challenges. Eng Life Sci. 2010;10(4):281–292. doi: 10.1002/elsc.201000027. [DOI] [Google Scholar]
- 29.Ward RS. Recent advances in the chemistry of lignans. Stud Nat Prod Chem. 2000;1(24):739–798. doi: 10.1016/S1572-5995(00)80054-X. [DOI] [Google Scholar]
- 30.Chunsriimyatav G, Hoza I, Valášek P, Skrovanková S, Banzragch D, Tsevegsuren N. Anticancer activity of lignan from the aerial parts of Saussurea salicifolia (L.) DC. Czech J Food Sci. 2009;27(1):S256–S258. doi: 10.17221/1097-cjfs. [DOI] [Google Scholar]
- 31.Lehraiki A, Attoumbré J, Bienaimé C, Matifat F, Bensaddek L, Nava-Saucedo E, Fliniaux MA, Ouadid-Ahidouch H, Baltora-Rosset S. Extraction of lignans from flaxseed and evaluation of their biological effects on breast cancer MCF-7 and MDA-MB-231 cell lines. J Med Food. 2010;13(4):834–841. doi: 10.1089/jmf.2009.0172. [DOI] [PubMed] [Google Scholar]
- 32.Shen W, Zhao Y, Chen H, Zhang T, Wu S, Liu P. M3, a natural lignan xyloside, exhibits potent anticancer activity in HCT116 cells. Oncol Lett. 2018 doi: 10.3892/ol.2018.9823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lin RW, Tsai IL, Duh CY, Lee KH, Chen IS. New lignans and cytotoxic constituents from Wikstroemia lanceolata. Planta Med. 2004;70:234–238. doi: 10.1055/s-2004-815540. [DOI] [PubMed] [Google Scholar]
- 34.Zhou Y, Liu YE, Cao J, Zeng G, Shen C, Li Y, Zhou M, Chen Y, Pu W, Potters L, Shi YE. Vitexins, nature-derived lignan compounds, induce apoptosis and suppress tumor growth. Clin Cancer Res. 2009;15(16):5161–5169. doi: 10.1158/1078-0432.ccr-09-0661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bylund A, Saarinen N, Zhang JX, Bergh A, Widmark A, Johansson A, Lundin E, Adlercreutz H, Hallmans G, Stattin P, Makela S. Anticancer effects of a plant lignan 7-hydroxymatairesinol on a prostate cancer model in vivo. Exp Biol Med. 2005;230(3):217–223. doi: 10.1177/153537020523000308. [DOI] [PubMed] [Google Scholar]
- 36.Luo J, Hu Y, Kong W, Yang M. Evaluation and structure-activity relationship analysis of a new series of arylnaphthalene lignans as potential anti-tumor agents. PLoS ONE. 2014;9(3):e93516. doi: 10.1371/journal.pone.0093516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Nakanishi T, Inatomi Y, Murata H, Shigeta K, Iida N, Inada A, Murata J, Farrera MAP, Iinuma M, Tanaka T, Tajima S, Oku N. A new and known cytotoxic aryltetralin-type lignans from stems of Bursera graveolens. Chem Pharm Bull. 2005 doi: 10.1002/chin.200533271. [DOI] [PubMed] [Google Scholar]
- 38.Chen JJ, Ishikawa T, Duh CY, Tsai IL, Chen IS. New dimeric aporphine alkaloids and cytotoxic constituents of Hernandia nymphaeifolia. Planta Med. 1996;62(06):528–533. doi: 10.1055/s-2006-957963. [DOI] [PubMed] [Google Scholar]
- 39.Moon SS, Rahman AA, Kim JY, Kee SH. Hanultarin, a cytotoxic lignan as an inhibitor of actin cytoskeleton polymerization from the seeds of Trichosanthes kirilowii. Bioorg Med Chem. 2008;16(15):7264–7269. doi: 10.1016/j.bmc.2008.06.032. [DOI] [PubMed] [Google Scholar]
- 40.Wu SJ, Wu TS. Cytotoxic arylnaphthalene lignans from Phyllanthus oligospermus. ChemInform. 2007 doi: 10.1002/chin.200704203. [DOI] [PubMed] [Google Scholar]
- 41.Tuchinda P, Kumkao A, Pohmakotr M, Sophasan S, Santisuk T, Reutrakul V. Cytotoxic Arylnaphthalide lignan glycosides from the aerial parts of Phyllanthus taxodiifolius. Planta Med. 2006;72(01):60–62. doi: 10.1055/s-2005-873141. [DOI] [PubMed] [Google Scholar]
- 42.Zhang YJ, Litaudon M, Bousserouel H, Martin MT, Thoison O, Léonce S, Dumontet V, Sévenet T, Guéritte F. Sesquiterpenoids and cytotoxic lignans from the bark of Libocedrus chevalieri. J Nat Prod. 2007;70(8):1368–1370. doi: 10.1021/np070124q. [DOI] [PubMed] [Google Scholar]
- 43.Xu LJ, Huang F, Chen SB, Zhang QX, Li LN, Chen SL, Xiao PG. New lignans and cytotoxic constituents from Schisandra propinqua. Planta Med. 2006;72(02):169–174. doi: 10.1055/s-2005-873199. [DOI] [PubMed] [Google Scholar]
- 44.Chen JJ, Chou ET, Duh CY, Yang SZ, Chen IS. New cytotoxic tetrahydrofuran- and dihydrofuran-type lignans from the stem of Beilschmiedia tsangii. Planta Med. 2006;72(04):351–357. doi: 10.1055/s-2005-916220. [DOI] [PubMed] [Google Scholar]
- 45.Li J, Zhang YJ, Jin BF, Su XZ, Tao YW, She ZG, Lin YC. 1H and 13C NMR assignments for two lignans from the heartwood of Streblus asper. Magn Reson Chem. 2008;46:497–500. doi: 10.1002/mrc.2186. [DOI] [PubMed] [Google Scholar]
- 46.Habtemariam S. Cytotoxic and cytostatic activity of erlangerins from Commiphora erlangeriana. Toxicon. 2003;41(6):723–727. doi: 10.1016/s0041-0101(03)00048-5. [DOI] [PubMed] [Google Scholar]
- 47.Lee JS, Kim J, Yu YU, Kim YC. Inhibition of phospholipase Cγ1 and cancer cell proliferation by lignans and flavans from Machilus thunbergii. Arch Pharm Res. 2004;27(10):1043–1047. doi: 10.1007/bf02975429. [DOI] [PubMed] [Google Scholar]
- 48.Lin X, Switzer BR, Demark W. Effect of mammalian lignans on the growth of prostate cancer cell lines. Anticancer Res. 2001;21(6A):3995–3999. [PubMed] [Google Scholar]
- 49.Lee WJ, Park YH, Park SW, Yang EK, Kim JH. Lignan from safflower seeds induces apoptosis in human promyelocytic leukemia cells. Prev Nutr Food Sci. 2003;8(2):113–118. doi: 10.3746/jfn.2003.8.2.113. [DOI] [Google Scholar]
- 50.Hausott B, Greger H, Marian B. Naturally occurring lignans efficiently induce apoptosis in colorectal tumor cells. J Cancer Res Clin Oncol. 2003;129(10):569–576. doi: 10.1007/s00432-003-0461-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Giridharan P, Somasundaram ST, Perumal K, Vishwakarma RA, Karthikeyan NP, Velmurugan R, Balakrishnan A. Novel substituted methylenedioxy lignan suppresses proliferation of cancer cells by inhibiting telomerase and activation of c-myc and caspases leading to apoptosis. Br J Cancer. 2002;87(1):98–105. doi: 10.1038/sj.bjc.6600422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gu JQ, Park EJ, Totura S, Riswan S, Fong HHS, Pezzuto JM, Kinghorn AD. Constituents of the twigs of Hernandia ovigera that inhibit the transformation of JB6 murine epidermal cells. J Nat Prod. 2002;65(7):1065–1068. doi: 10.1021/np020042w. [DOI] [PubMed] [Google Scholar]
- 53.Ito C, Itoigawa M, Ogata M, Mou XY, Tokuda H, Nishino H, Furukawa H. Lignans as anti-tumor-promoter from the seeds of Hernandia ovigera. Planta Med. 2001;67(2):166–168. doi: 10.1055/s-2001-11501. [DOI] [PubMed] [Google Scholar]
- 54.Takasaki M, Konoshima T, Komatsu K, Tokuda H, Nishino H. Anti-tumor-promoting activity of lignans from the aerial part of Saussurea medusa. Cancer Lett. 2000;158(1):53–59. doi: 10.1016/s0304-3835(00)00499-7. [DOI] [PubMed] [Google Scholar]
- 55.Navarro E, Alonso SJ, Trujillo J, Jorge E, Pérez C. General behavior, toxicity, and cytotoxic activity of elenoside, a lignan from Justicia hyssopifolia. J Nat Prod. 2000;64(1):134–135. doi: 10.1021/np9904861. [DOI] [PubMed] [Google Scholar]
- 56.Rickard SE, Yuan YV, Thompson LU. Plasma insulin-like growth factor I levels in rats are reduced by dietary supplementation of flaxseed or its lignan secoisolariciresinol diglycoside. Cancer Lett. 2000;161(1):47–55. doi: 10.1016/s0304-3835(00)00592-9. [DOI] [PubMed] [Google Scholar]
- 57.Saleem M, Kim HJ, Ali MS, Lee YS. An update on bioactive plant lignans. Nat Prod Rep. 2005;22(6):696. doi: 10.1039/b514045p. [DOI] [PubMed] [Google Scholar]
- 58.Messina F, Curini M, di Sano C, Zadra C, Gigliarelli G, Rascón-Valenzuela LA, Robles Zepeda RE, Marcotullio MC. Diterpenoids and triterpenoids from the resin of Bursera microphylla and their cytotoxic activity. J Nat Prod. 2015;78(5):1184–1188. doi: 10.1021/acs.jnatprod.5b00112. [DOI] [PubMed] [Google Scholar]
- 59.Chen JJ, Huang HY, Duh CY, Chen IS. Cytotoxic constituents from the stem bark of Zanthoxylum Pistaciiflorum. J Chin Chem Soc. 2004;51(3):659–663. doi: 10.1002/jccs.200400099. [DOI] [Google Scholar]
- 60.Jeong GS, Kwon OK, Park BY, Oh SR, Ahn KS, Chang MJ, Oh WK, Kim JC, Min BS, Kim YC, Lee HK. Lignans and coumarins from the roots of Anthriscus sylvestris and their increase of caspase-3 activity in HL-60 cells. Biol Pharm Bull. 2007;30(7):1340–1343. doi: 10.1248/bpb.30.1340. [DOI] [PubMed] [Google Scholar]
- 61.Wu SJ, Wu TS. Cytotoxic arylnaphthalene lignans from Phyllanthus oligospermus. Chem Pharmaceut Bull. 2006;54(8):1223–1225. doi: 10.1248/cpb.54.1223. [DOI] [PubMed] [Google Scholar]
- 62.Sriwiriyajan S, Sukpondma Y, Srisawat T, Madla S, Graidist P. (−)-Kusunokinin and piperloguminine from Piper nigrum: an alternative option to treat breast cancer. Biomed Pharmacother. 2017;92:732–743. doi: 10.1016/j.biopha.2017.05.130. [DOI] [PubMed] [Google Scholar]
- 63.Donoso-Fierro C, Tiezzi A, Ovidi E, Ceccarelli D, Triggiani D, Mastrogiovanni F, Taddei AR, Pérez C, Becerra J, Silva M, Passarella D. Antiproliferative activity of yatein isolated from Austrocedrus chilensis against murine myeloma cells: cytological studies and chemical investigations. Pharm Biol. 2014;53(3):378–385. doi: 10.3109/13880209.2014.922588. [DOI] [PubMed] [Google Scholar]
- 64.Wangteeraprasert R, Lipipun V, Gunaratnam M, Neidle S, Gibbons S, Likhitwitayawuid K. Bioactive compounds from Carissa spinarum. Phytother Res. 2012 doi: 10.1002/ptr.4607. [DOI] [PubMed] [Google Scholar]
- 65.Mukhija M, Dhar KL, Kalia AN. Bioactive lignans from Zanthoxylum alatum Roxb. stem bark with cytotoxic potential. J Ethnopharmacol. 2014;152(1):106–112. doi: 10.1016/j.jep.2013.12.039. [DOI] [PubMed] [Google Scholar]
- 66.Mulabagala V, Subbaraju GV, Ramani MV, DeWitt DL, Nair MG. Lipid peroxidation, cyclooxygenase enzyme and tumor cell proliferation inhibitory lignans from Justicia species. Nat Prod Commun. 2008;3(11):1934578X0800301. doi: 10.1177/1934578x0800301109. [DOI] [Google Scholar]
- 67.Su B, Zhu Q, Gao K, Yuan C, Jia Z. Lignan and phenylpropanoid glycosides from Lancea tibetica and their antitumor activity. Planta Med. 1999;65(6):558–561. doi: 10.1055/s-1999-14026. [DOI] [PubMed] [Google Scholar]
- 68.Gnabre J, Unlu I, Chang TC, Lisseck P, Bourne B, Scolnik R, Jacobsen NE, Bates R, Huang RC. Isolation of lignans from Schisandra chinensis with anti-proliferative activity in human colorectal carcinoma: structure–activity relationships. J Chromatogr B. 2010;878(28):2693–2700. doi: 10.1016/j.jchromb.2010.08.005. [DOI] [PubMed] [Google Scholar]
- 69.Aimaiti S, Saito Y, Fukuyoshi S, Goto M, Miyake K, Newman DJ, O’Keefe BR, Lee KH, Nakagawa-Goto K. Isolation, structure elucidation, and antiproliferative activity of butanolides and lignan glycosides from the fruit of Hernandia nymphaeifolia. Molecules. 2019;24(21):4005. doi: 10.3390/molecules24214005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kintzios SE. Terrestrial plant-derived anticancer agents and plant species used in anticancer research. Crit Rev Plant Sci. 2006;25(2):79–113. doi: 10.1080/07352680500348824. [DOI] [Google Scholar]
- 71.Kardono LBS, Tsauri S, Padmawinata K, Pezzuto JM, Kinghorn AD. Cytotoxic constituents of the bark of Plumeria rubra collected in Indonesia. J Nat Prod. 1990;53(6):1447–1455. doi: 10.1021/np50072a008. [DOI] [PubMed] [Google Scholar]
- 72.Novelo M, Cruz JG, Hernández L, Pereda-Miranda R, Chai H, Mar W, Pezzuto JM. Cytotoxic constituents from Hyptis verticillata. J Nat Prod. 1993;56(10):1728–1736. doi: 10.1021/np50100a011. [DOI] [PubMed] [Google Scholar]
- 73.Torrance SJ, Hoffmann JJ, Cole JR. Wikstromol, antitumor lignan from Wikstroemia foetida var. oahuensis gray and Wikstroemia uva-ursi gray (thymelaeaceae) J Pharmaceut Sci. 1979;68(5):664–665. doi: 10.1002/jps.2600680545. [DOI] [PubMed] [Google Scholar]
- 74.Lee SK, Cui B, Mehta RR, Kinghorn A, Pezzuto JM. Cytostatic mechanism and antitumor potential of novel 1H-cyclopenta[b]benzofuran lignans isolated from Aglaia elliptica. Chem Biol Interact. 1998;115(3):215–228. doi: 10.1016/s0009-2797(98)00073-8. [DOI] [PubMed] [Google Scholar]
- 75.Zhao CQ, Zhu YY, Chen SY, Ogihara Y. Lignan glucoside from Sinopodophyllum emodi and its cytotoxic activity. Chin Chem Lett. 2011;22:181–184. doi: 10.1016/j.cclet.2010.10.013. [DOI] [Google Scholar]
- 76.Chavez KJ, Feng X, Flanders JA, Rodriguez E, Schroeder FC. Spirocyclic lignans from Guaiacum (Zygophyllaceae) induce apoptosis in human breast cancer cell lines. J Nat Prod. 2011;74(5):1293–1297. doi: 10.1021/np100891y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kim KH, Kim HK, Choi SU, Moon E, Kim SY, Lee KR. Bioactive lignans from the rhizomes of Acorus gramineus. J Nat Prod. 2011;74(10):2187–2192. doi: 10.1021/np200541m. [DOI] [PubMed] [Google Scholar]
- 78.Ye YQ, Xia CF, Li YK, Wu XX, Du G, Hu QF, Gao XM. A New dibenzocyclooctadiene lignan from the stems of Schisandra neglecta and its cytotoxicities. Asian J Chem. 2014;26(7):1948–1950. doi: 10.14233/ajchem.2014.15583. [DOI] [Google Scholar]
- 79.Suh WS, Kim KH, Kim HK, Choi SU, Lee KR. Three new lignan derivatives from Lindera glauca (Sieboldet Zucc.) Blume. Helv Chim Acta. 2015;98(8):1087–1094. doi: 10.1002/hlca.201500002. [DOI] [Google Scholar]
- 80.Kim KH, Moon E, Kim SY, Choi SU, Lee KR. Lignan constituents of Tilia amurensis and their biological evaluation on antitumor and anti-inflammatory activities. Food Chem Toxicol. 2012;50(10):3680–3686. doi: 10.1016/j.fct.2012.07.014. [DOI] [PubMed] [Google Scholar]
- 81.Jin H, Yin HL, Liu SJ, Chen L, Tian Y, Li B, Wang Q, Dong JX. Cytotoxic activity of lignans from Justicia procumbens. Fitoterapia. 2014;94:70–76. doi: 10.1016/j.fitote.2014.01.025. [DOI] [PubMed] [Google Scholar]
- 82.Trinh Thi Thanh V, Cuong Pham V, Doan Thi Mai H, Litaudon M, Guéritte F, Retailleau P, Nguyen VH, Chau VM. Cytotoxic lignans from fruits of Cleistanthus indochinensis: synthesis of cleistantoxin derivatives. J Nat Prod. 2012;75(9):1578–1583. doi: 10.1021/np3003832. [DOI] [PubMed] [Google Scholar]
- 83.Ren Y, Lantvit D, Deng Y, Kanagasabai R, Gallucci J, Ninh T, Chai H, Soejarto D, Fuchs J, Yalowich J, Yu J, Swanson S, Kinghorn A. Potent cytotoxic arylnaphthalene lignan lactones from Phyllanthus poilanei. Planta Med. 2014 doi: 10.1055/s-0034-1382432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Liu Y, Yang Y, Tasneem S, Hussain N, Daniyal M, Yuan H, Xie Q, Liu B, Sun J, Jian Y, Li B, Chen S, Wang W. Lignans from Tujia ethnomedicine Heilaohu: chemical characterization and evaluation of their cytotoxicity and antioxidant activities. Molecules. 2018;23(9):2147. doi: 10.3390/molecules23092147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Rajalekshmi DS, Kabeer FA, Madhusoodhanan AR, Bahulayan AK, Prathapan R, Prakasan N, Varughese S, Nair MS. Anticancer activity studies of cubebin isolated from Piper cubeba and its synthetic derivatives. Bioorg Med Chem Lett. 2016;26(7):1767–1771. doi: 10.1016/j.bmcl.2016.02.041. [DOI] [PubMed] [Google Scholar]
- 86.Maa Y, Wang H, Wang R, Meng F, Dong Z, Wang G, Lan X, Quan H, Liao Z, Chen M. Cytotoxic lignans from the stems of Herpetospermum pedunculosum. Phytochemistry. 2019;164:102–110. doi: 10.1016/j.phytochem.2019.05.004. [DOI] [PubMed] [Google Scholar]
- 87.Su GY, Wang KW, Wang XY, Wu B. Bioactive lignans from Zanthoxylum planispinum with cytotoxic potential. Phytochem Lett. 2015;11:120–126. doi: 10.1016/j.phytol.2014.12.004. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.