Summary
CRC is a common cause of cancer mortality. Screening reduces incidence and mortality, but is underutilized. An understanding of the evidence to support screening, and promotion of policies and clinical strategies to promote uptake and complete follow up testing and treatment have high potential to save lives across the population.
Keywords: colorectal cancer, screening, early detection, prevention, incidence, mortality
Pathogenesis and Natural History
Colorectal cancer (CRC) is an invasive, malignant neoplasm which develops from the mucosa of the colon and rectum, with the vast majority classified as adenocarcinomas. The vast majority of CRCs develop from normal mucosa that gives rise to colorectal polyps, a fraction of which progress to invasive CRCs. The normal mucosa to polyp to CRC sequence occurs due to a range of epigenetic and genetic events which result in progressive silencing of tumor suppressor genes, activation of oncogenes, and chromosomal instability1 (Figure 1). Multiple subtypes of polyp initiation and cancer progression have been proposed, including an adenoma-carcinoma sequence, in which the cancer precursor is an adenoma, as well as a serrated polyp-carcinoma sequence, in which the cancer precursor is a sessile serrated lesion (also known as sessile serrated polyp and sessile serrated adenoma). Morbidity and mortality associated with colorectal cancer develop as a result of anemia caused by polyps and cancer, and bowel obstruction, weight loss, and metastases caused by CRCs.
Figure 1.
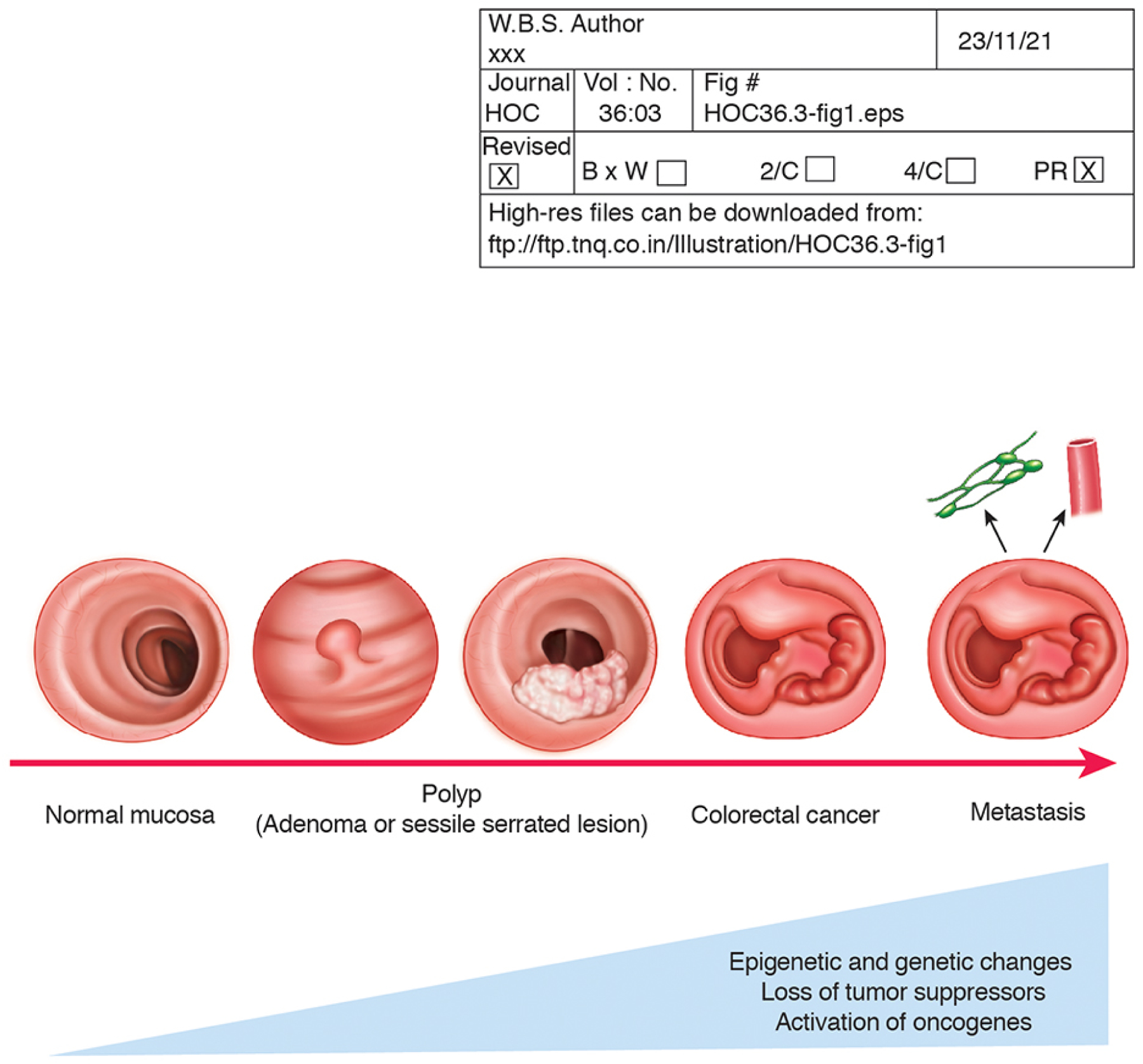
Pathogenesis and natural history of colorectal cancer.
Concepts regarding the pathogenesis and natural history of colorectal cancer are shown. Most CRCs arise from a normal mucosa to polyp to cancer sequence, which can ultimately result in metastatic disease.
Endoscopically, polyps and cancers are detectable, offering an opportunity for early CRC detection and even prevention. The progression from normal mucosa to cancer is accompanied by progressive accumulation of epigenetic and genetic changes in the mucosa, which may include loss of tumor suppressors, activation of oncogenes, and chromosomal instability.
Several features support CRC as an ideal target for screening. A large proportion of adenomas and sessile serrated lesion polyp precursors are endoscopically identifiable, allowing for polypectomy and cancer prevention. Adenomas, sessile serrated lesions, and CRCs shed abnormal cells with detectable molecular markers, and have a proclivity to bleed, allowing for stool-based detection. Emerging blood-based tests may allow for detection of genetic and epigenetic changes associated with polyps and cancer leaked into the circulation. The long time period required for development of polyps and cancer, as well as the tendency for early stage cancers to be asymptomatic in many individuals, allows for a window of opportunity for polypectomy and prevention of cancers, as well as early detection and delivery of highly effective treatment for early stage cancers. Overall, our current understanding of the pathogenesis and natural history of CRC supports the concept and demonstrated observations that screening can be effective.
Epidemiology
Current CRC Incidence and Mortality
Colorectal cancer is the 2nd leading cause of cancer death in the United States2. In 2021, approximately 149,500 individuals were expected to be diagnosed with CRC, and nearly 53,000 were expected to die from the disease86. The dominant risk factor for CRC is age, with age-specific incidence and mortality rising dramatically over the lifespan (Figure 2). Incidence and mortality rise from 6 and 1 per 100,000 for individuals age 30 to 34 years to 228 and 105 per 100,000 for individuals age 80 to 84 years, respectively (Figure 2)3. Lifetime risk for CRC is similar among women and men, at 4.1% and 4.4% respectively, though increases in age-specific incidence and mortality occur later for women than men (Figure 3). CRC incidence varies substantially by family history and genetic risk, and both CRC incidence and mortality vary by race/ethnicity and structural factors such as socioeconomic position, as is reviewed in detail later in this issue.
Figure 2.
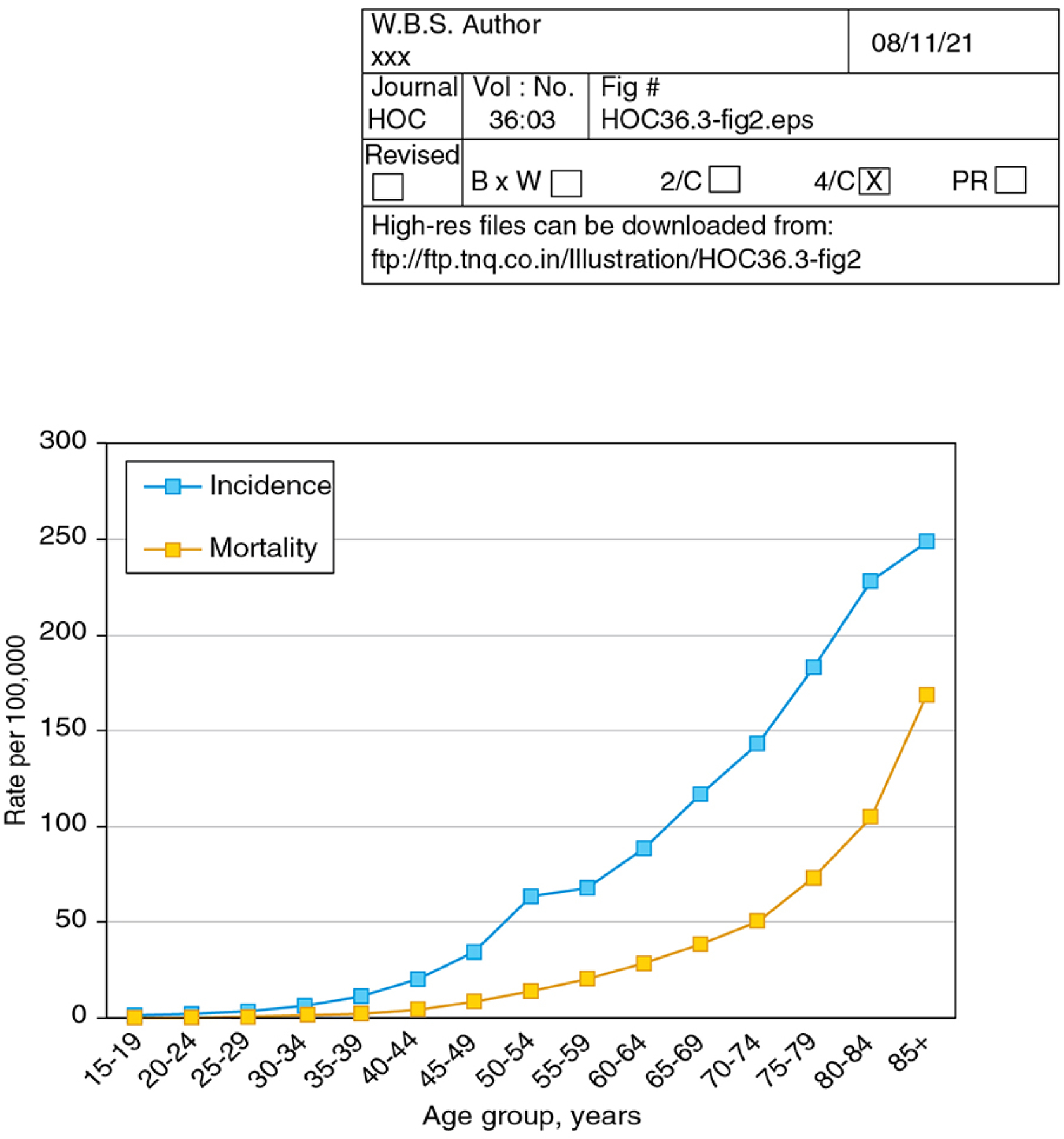
Age-Specific Colorectal Cancer Incidence and Mortality, 2014–2018, US Surveillance Epidemiology and End Results Program. Colorectal cancer incidence and mortality are highly age specific, with rates markedly increasing by age group. Source: Surveillance, Epidemiology, and End Results 21 program delay-adjusted incidence and colorectal cancer mortality data, accessed 8/27/21.
Figure 3.
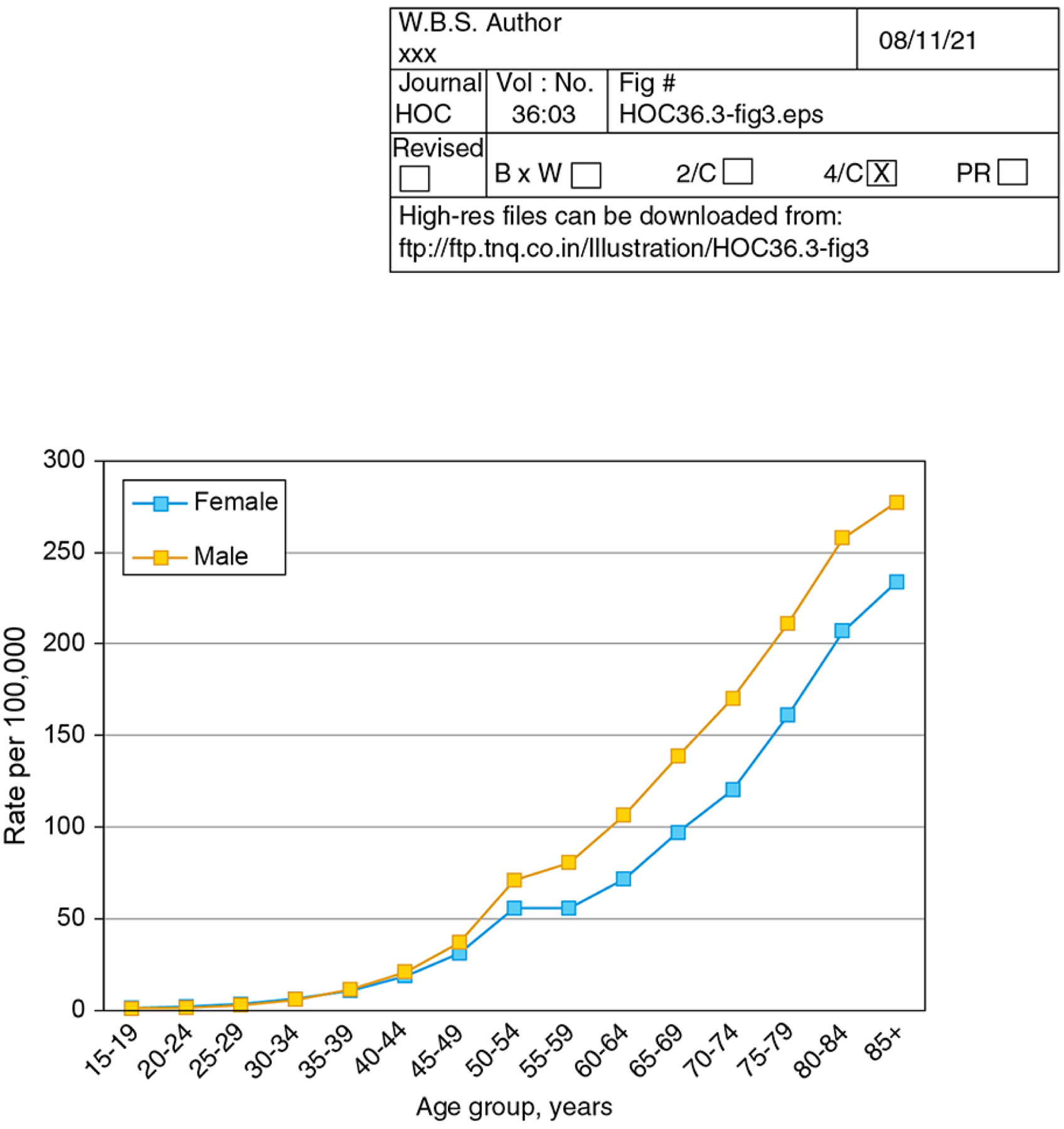
Age-Specific Colorectal Cancer Incidence for Males and Females, 2014–2018, US Surveillance, Epidemiology and End Results Program. Age-specific colorectal cancer incidences for males and females are shown, demonstrating that the rise in age-specific incidence is delayed for females compared to males. Source: Surveillance, Epidemiology, and End Results 21 program delay-adjusted incidence, accessed 8/27/21.
Risk Factors
Beyond age, gender, and genetics, established risk factors for CRC which plausibly contribute to pathogenesis include diet and lifestyle factors such as increased red meat and alcohol intake, decreased fruit and vegetable intake, decreased calcium intake, decreased physical activity, and smoking, and comorbid conditions such as obesity, type 2 diabetes, and inflammatory bowel disease4,5. Exposures to aspirin, non-steroidal anti-inflammatory medications, hormone replacement therapy, and statins have been associated with reduced CRC risk4. Risk factors for CRC have also been tied to risk for adenomas, while increasing age, smoking, alcohol, and reduced non-steroidal anti-inflammatory medication exposure appear to be risk factors for sessile serrated lesions6.
Polyp Incidence and Prevalence
Because most CRCs arise from polyp precursors such as adenomas and sessile serrated lesions, incidence and prevalence of these lesions may inform screening strategies. Utilizing data from a national colonoscopy screening program in Germany, one study estimated the annual incidence of non-advanced adenoma to be 1.8 to 2.4% for men and 1.2 to 1.6% for women between the ages of 55 to 797. At screening colonoscopy among individuals age ≥50 years, a recent meta-analysis estimated the prevalence of any adenoma to be 25.9% and any advanced adenoma to be 5.2%8. Prevalence of sessile serrated lesions at colonoscopy has been estimated to be 5.1% in the US, with prevalence as high as 9.1% at higher quality colonoscopy9. Clinically significant serrated polyps, a definition including sessile serrated lesions, traditional serrated polyps, and proximal hyperplastic polyps, have an estimated prevalence of 12.3%9.
CRC Survival
Survival after CRC diagnosis is highly stage-specific, ranging from over 90% for localized disease, to less than 15% for stage 4 disease (Figure 4)10. As covered in detail elsewhere in this issue, within stage survival varies substantially by race, ethnicity, and other factors, most likely attributable to inequities in access rather than biologic factors.
Figure 4.

Colorectal Cancer Stage Specific 5-year Survival, US Surveillance, Epidemiology, and End Results Program, 2011–2017. 5-year stage specific colorectal cancer survival is shown, demonstrating over 90% survival for localized disease, and less than 15% survival for distant disease. Source: Surveillance, Epidemiology, and End Results Program 18 data, accessed 8/27/21.
CRC Incidence and Mortality Trends
Across all age groups, CRC incidence and mortality have been decreasing over time (Figure 5). These decreases have been attributed to improvements in risk factor exposures in the population, treatment for established CRC, and particularly to widespread uptake of screening11. Observed trends in CRC incidence and mortality appear to correlate with increases in the proportion of age-eligible individuals who are up-to-date with screening, though direct causality cannot be established2. From 2000 to 2018, CRC incidence and mortality decreased from 56 and 20 per 100,000 to 37 and 13 per 100,000, respectively (Figure 5)12, while proportion of individuals age 50 to 75 years up-to-date with screening increased from 34.6%13 to 67.0%14. A notable exception to overall decreasing trends in incidence and mortality include increasing incidence and mortality among individuals under age 50, which is attributable to a birth cohort effect for individuals born after 1950; this birth cohort effect may also be reversing previous decreasing trends among individuals age 50 to 54 years (covered in detail elsewhere in this issue2).
Figure 5.
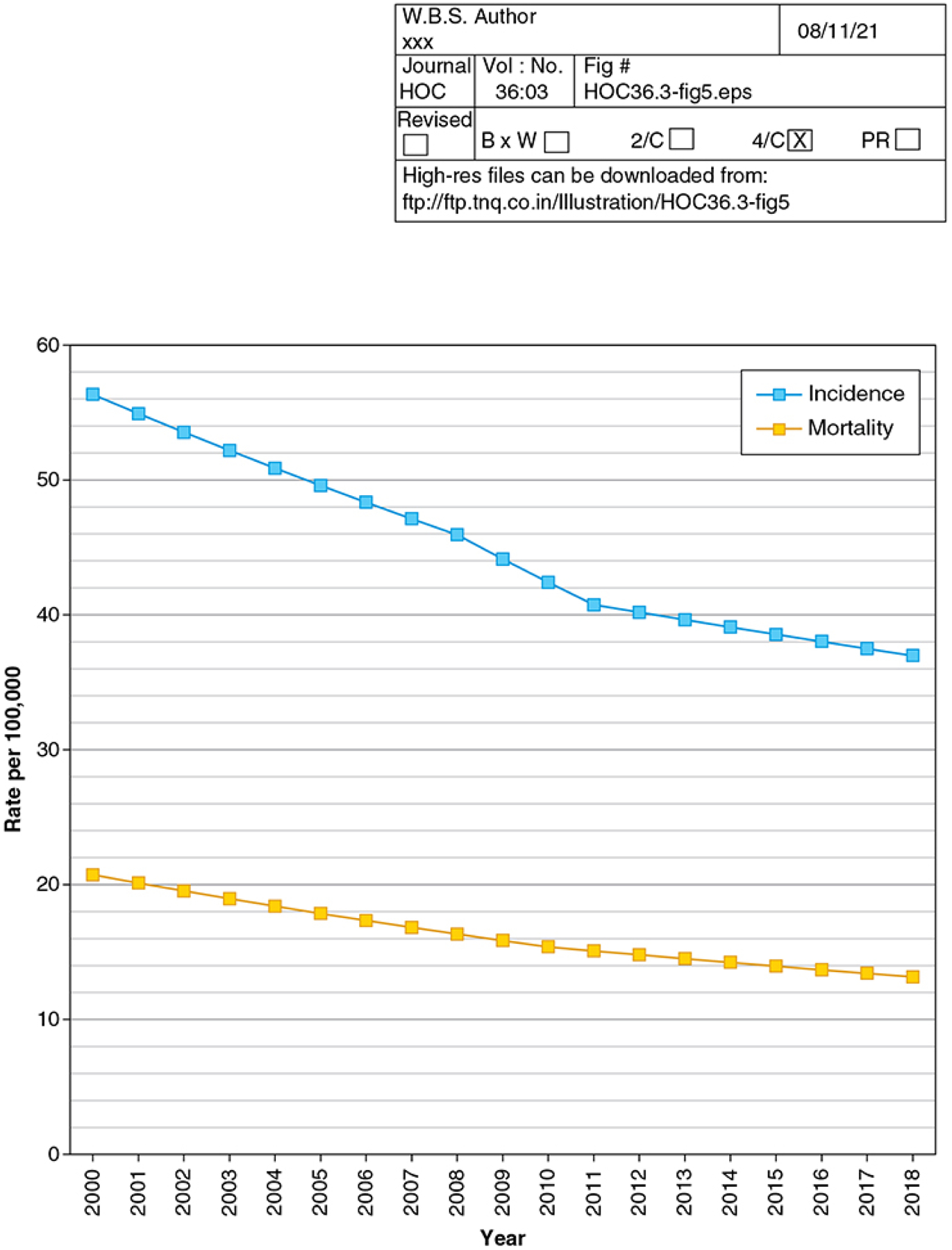
Colorectal Cancer Incidence and Mortality Rates, 2000–2018, US Surveillance Epidemiology and End Results Program. Trends in delay-adjusted colorectal cancer incidence, and colorectal cancer mortality from the Surveillance Epidemiology and End Results 21 program are shown, demonstrating a trend towards reduced incidence and mortality that appears to be flattening over time. Source: Surveillance, Epidemiology, and End Results Program 21 data, accessed 8/27/21.
Rationale and Evidence Base to Support Screening
Appraisal Criteria
Multiple criteria for appraisal of screening strategies exist, most based foundationally on the seminal work of Wilson and Jungner15. The evidence base to support CRC screening when assessed through the lens of these criteria is strong (Table 1). Our understanding of the pathogenesis and epidemiology of CRC demonstrate that CRC is an important health problem, and that that the pathogenesis of CRC includes a latent, asymptomatic stage. As covered elsewhere in this issue, treatments for CRC exist and are highly effective. Available evidence, presented herein, demonstrates that suitable tests exist, that many available tests are acceptable to the US population, and that a strategy of screening improves CRC incidence and outcomes. Overall, available evidence has led to widespread endorsement of CRC screening across a wide range of stakeholders, from advocacy organizations such as the American Cancer Society16, to specialty groups such as the US Multi-Society Task Force on Colorectal Cancer17 and the National Comprehensive Cancer Network18, and through guideline policy makers, such as the US Preventive Services Task Force19. CRC screening has been endorsed through country-wide programs across the world, including in the United Kingdom, the Netherlands, Germany, Poland, Taiwan, and many others20. In this article, the focus will be on tests endorsed by the US Preventive Services Task Force (USPSTF) 2021 colorectal cancer screening guideline, as this guideline has the most influential impact on policy and practice in the United States.
Table 1.
Criteria and Evidence to Support Colorectal Cancer Screening
| Wilson and Jungner Principles for Early Disease Detection | Evidence of Meeting Criteria |
|---|---|
| The condition sought should be an important health problem | CRC is the 2nd leading cause of cancer death. |
| The natural history of the condition, including development from latent to declared disease, should be adequately understood. | Pathogenesis of CRC, including the polyp to cancer sequence, is well understood. |
| There should be a recognizable latent or early symptomatic stage. | Many CRCs are associated with an asymptomatic period. Polyps generally have a long growth period that allows for early detection and CRC prevention. |
| There should be an accepted treatment for patients with recognized disease | Colonoscopy is accepted as both the primary reference test for early detection of CRC and identification and removal of polyps after an abnormal non-colonoscopy test, and also as an option for primary screening. Consensus guidelines for CRC treatment exist. |
| Facilities for diagnosis and treatment should be available. | For insured individuals, access to screening, colonoscopy follow up for abnormal tests, and CRC treatment is widely available, though some challenges to access continue to exist. |
| There should be a suitable test or examination. | Multiple options, including invasive and non-invasive tests, are available for CRC screening. |
| The test should be acceptable to the population. | Currently, 67% of the US population is up-to-date with screening, and evidence suggests that tests are highly acceptable, though test preferences differ across individuals. |
| There should be an agreed policy on whom to treat as patients. | USPSTF and others have consistent guidelines for screening. The National Comprehensive Cancer Network and others have consistent guidelines for CRC treatment. |
| The cost of case-finding (including diagnosis and treatment of patients diagnosed) should be economically balanced in relation to possible expenditure on medical care as a whole. | CRC screening has been shown to be cost-effective, and even potentially cost-saving. |
| Case-finding should be a continuing process and not a “once and for all” project. | USPSTF and others recommend strategies for screening, such as annual FIT or every 10 year colonoscopy. |
CRC, colorectal cancer; USPSTF, US Preventive Services Task Force; FIT, fecal immunochemical test.
Screening Tests
Screening tests currently recommended by the USPSTF include high-sensitivity guaiac fecal occult blood testing, the fecal immunochemical test (FIT), stool DNA-FIT (sDNA-FIT, Cologuard, Exact Sciences), sigmoidoscopy, computed tomographic colonography (CT colonography), and colonoscopy. Additional tests which have been studied for screening include the colon capsule (PillCam COLON 2, Medtronic), as well as methylated serum septin 9 (Epi proColon, Epigenomics). A summary of test characteristics, as well as available evidence of impact on CRC and mortality, is provided in Table 2.
Table 2.
Test characteristics and outcomes of CRC screening tests currently available in the United States
| Test | Sensitivity for Advanced Colorectal Neoplasia | Sensitivity for CRC | Specificity | Impact on Incidence and Mortality (Compared to No Screening) |
|---|---|---|---|---|
|
gFOBT (Hemoccult Sensa, Beckman Coulter) |
6 to 17% | 50 to 75% | 96 to 99% | RCT evidence of 20% relative incidence reduction, 18% relative mortality reduction. |
|
FIT (OC Sensor and OC-Light, Polymedco) |
25 to 27% | 74 to 81% | 95 to 96% | No RCT evidence. Observational studies suggest 21% relative incidence reduction, 59% relative mortality reduction. |
|
sDNA-FIT (Cologuard, Exact Sciences) |
47% | 93% | 89% | No RCT or observational study evidence. |
| Sigmoidoscopy | 95%* | 95%* | 87%** | RCT evidence of 22% relative incidence reduction, 28% relative mortality reduction |
| Colonoscopy | 95% | 95% | 86 to 89%** | No RCT evidence. Observational studies suggest 69% relative incidence reduction, 68% relative mortality reduction. |
|
Colon Capsule (PillCam COLON 2, Medtronic) |
85 to 88%*** | Limited data | 97 to 99% | No RCT or observational study evidence. |
|
Methylated serum septin 9 (Epi proColon, Epigenomics AG) |
25% | 68% | 79% | No RCT or observational study evidence. |
See text for data references. gFOBT, guaiac fecal occult blood test; FIT, fecal immunochemical test; sDNA-FIT, muti-target stool DNA FIT test; RCT, randomized controlled trial; CRC, colorectal cancer
within reach of the sigmoidoscope;
false positives are defined by polyps biopsied or removed which were not adenomas;
reported sensitivity is for polyps ≥ 1 cm
gFOBT
gFOBT is a test in which a guaiac developer is applied to a spontaneous bowel movement to evaluate for a peroxidase reaction with heme. For one gFOBT (Hemoccult Sensa, Beckman Coulter), reported sensitivity for advanced colorectal neoplasia and colorectal cancer ranges from 6 to 17%, and 50 to 75%, respectively21. Specificity for advanced neoplasia ranges from 96 to 99%. Though testing can be conveniently done at home, because the test requires multiple samples, avoidance of red meat and medications such as aspirin which can cause false positives, and because a positive test could be due to bleeding from anywhere in the gastrointestinal tract, gFOBT has largely been replaced by FIT. USPSTF recommends annual testing, though biannual testing, which may be less effective, has also been endorsed by some groups. Tests can be distributed and returned in person or by mail.
FIT
A FIT utilizes an antibody against the globin moiety of heme to evaluate for the presence of occult blood in a spontaneous bowel movement. Notably, for FITs available in the US, performance differs widely by manufacturer and specific products. For two commonly available FITs with consistently good performance characteristics (OC-Sensor and OC-Light, Polymedco), sensitivity for advanced colorectal neoplasia and colorectal cancer range from 25 to 27%, and 74 to 81%, respectively21. Specificity for advanced neoplasia for these products varies from 95 to 96%. In contrast, a much wider range of sensitivities and specificities have been reported across other FIT products21. Dietary and medication modification is not required, and data are mixed on whether sensitivity, specificity, or positive predictive value are impacted by concurrent use of aspirin or antithrombotic medications22–28, though positive predictive value is likely decreased when testing is done in patients taking these medications. USPSTF recommends annual testing, though biannual testing has also been endorsed by some groups. Tests can be distributed and returned in person or by mail.
sDNA-FIT
The sDNA-FIT (Cologuard, Exact Sciences) utilizes an algorithm based on results of testing for 7 DNA markers and occult blood utilizing a FIT within a spontaneously evacuated bowel movement. Sensitivities for advanced colorectal neoplasia and colorectal cancer are reported to be 47% and 93%, respectively, with reported specificity for advanced neoplasia of 89%21. sDNA-FIT has superior sensitivity for large sessile serrated lesions compared to FIT29. The test is mailed directly to patients by the manufacturer upon provider order, and returned by mail as well.
Sigmoidoscopy
Sigmoidoscopy most commonly utilizes a flexible endoscope to evaluate the rectum and distal colon for polyps after bowel preparation. The test is often performed without sedation. Sensitivities for large adenomas and CRC within reach of the sigmoidoscope have been estimated to be 95%30. Specificity has been reported to be 87%30, with false positives defined by polyps removed which are not adenomas or clinically significant serrated lesions. Sensitivity of a program of sigmoidoscopy is complemented by the colonoscopy evaluation that is triggered by the finding of distal polyps, though patients with isolated proximal colon polyps or CRC will not be detected. USPSTF recommends testing every 5 years, or every 10 years if accompanied by an annual FIT.
CT Colonography
CT colonography evaluates for polyps and cancer with a CT scan. Current protocols generally require both bowel preparation and oral contrast, as well as colon inflation, but not sedation. Sensitivity for lesions ≥1 cm ranges from 67 to 94%21. Specificity ranges from 86 to 98%. Estimates for potentially important extracolonic findings that require follow up range from 3.4–26.9%, and 1.3–11.4% for those likely unimportant or incompletely characterized and possibly requiring follow up31. Some studies have demonstrated superior CRC sensitivity compared to colonoscopy in which the endoscopist was initially blind to CT colonography results. USPSTF recommends testing every 5 years.
Colonoscopy
Colonoscopy utilizes a flexible endoscope to evaluate the entire colon and rectum after bowel preparation. In the US, colonoscopy is almost always performed with some type of sedation. Sensitivity for adenomas ≥1 cm and CRC are estimated to be 95%30, though 3 studies comparing colonoscopy to CT colonography estimated sensitivity for lesions ≥1 cm to be 89%21. Specificity is 86 to 89% with false positives defined by polyps biopsied or removed which are not adenomas21,30. USPSTF recommends a testing interval of 10 years after a normal colonoscopy, and the US Multi-Society Task Force on CRC has specified time intervals for surveillance for persons with abnormal colonoscopy, defined by having one or more adenomas or sessile serrated lesions32.
Colon Capsule
Colon capsule involves administration of a small pill camera to evaluate the entire colon and rectum after bowel preparation. A recording device captures the pictures taken, which are reviewed after a patient returns the device to the administering provider. Three studies using colonoscopy as a reference standard for evaluation of sensitivity and specificity in a screening population are available, utilizing the current generation colon capsule (PillCam COLON 2, Medtronic). Among individuals with an adequate exam, defined as having adequate bowel preparation and full transit time through the colon, sensitivity for polyps ≥1 cm ranged from 85 to 88%, and specificity from 97 to 99%33–35. These studies likely overestimate capsule performance in a usual care setting because subjects with inadequate bowel prep and inadequate transit time were excluded from analyses, and because centralized, experienced readers were assigned to read all capsules. Currently, the colon capsule has Food and Drug Administration (FDA) approval to market the test for use in individuals with incomplete colonoscopy or evidence of lower GI bleeding, but not for CRC screening. USPSTF has not endorsed colon capsule for average risk CRC screening.
Methylated serum septin 9
Methylated serum septin 9 (Epi proColon, Epigenomics AG) utilizes a blood sample to test for evidence of methylation of the promoter region of the septin 9 gene36. Sensitivities for advanced neoplasia and CRC for the currently available version of the test are 25% and 68%, respectively, with specificity of 79%21,36. A single manufacturer has FDA approval to market the test for average risk screening in individuals who have been offered but refused other usual care screening tests, with annual testing and recommendation for colonoscopy after an abnormal result. USPSTF has not endorsed methylated serum septin 9 for average risk screening.
Screening Efficacy and Effectiveness
A variety of strategies have been utilized to evaluate the potential efficacy and effectiveness of available screening tests, including randomized controlled trials, observational studies, and modeling studies. Because large sample size trials covering timespans of 5 to 10 or even greater duration are required to evaluate the impact of a screening strategy on CRC incidence and mortality, the potential impact of screening for the strategies currently recommend by the USPSTF is based on observational studies, modeling studies, and the inference that tests with at least as good sensitivity and specificity for polyp and cancer detection as those proven to be effective in previously conducted randomized trials should have similar or better longitudinal impact on CRC incidence and mortality37. Randomized trial data demonstrating reduced incidence and mortality are only available for a strategy of sigmoidoscopy-based screening, as well as for the older generation of guaiac-based stool tests. A range of ages groups were included in reported randomized controlled trials and observational studies, from individuals age 45 to 80, with all including individuals age 55 to 64.
Randomize Trials
gFOBT
Four out of 5 reported large randomized trials of gFOBT (utilizing the Hemoccult II, Beckman Coulter) have demonstrated that screening vs no screening reduces incidence and mortality38. Meta-analyses of these studies report relative risk reduction in mortality of as much as 18% (RR 0.82, 95% CI 0.73–0.9238,39). Across available studies, as much as a 20% relative reduction in incidence (RR range 0.80 to 1.11) has been reported38. Notably, 440–43 out of 5 available trials utilized biennial testing, and one44 had arms with annual as well as biennial testing. In current practice, the gFOBT used in these trials, Hemoccult II, is no longer available for use, and has been supplanted by high sensitivity gFOBTs, such as Hemoccult Sensa (which has higher sensitivity, but lower specificity), or by FIT (which has higher sensitivity and specificity for colonic neoplasia).
Sigmoidoscopy
Four large randomized trials encompassing data from over 450,000 subjects all showed that a strategy of screening with sigmoidoscopy vs no screening reduces incidence and mortality38,45–50. Three47–50 out of 4 of these studies utilized “once only” sigmoidoscopy, while one45 offered sigmoidoscopy every 3–5 years, and one of the studies also included an arm offering FIT. Meta-analyses have reported relative risk reductions in CRC incidence of 22% (RR 0.78, 95% CI 0.74–0.83), CRC mortality of 28% (0.65–0.80), and all cause mortality of 3% (0.97, 95% CI 0.96–0.99) in intention to screen analyses38,51.
Randomized Trials Underway
Randomized trials aiming to establish the impact of colonoscopy vs no screening, or colonoscopy vs FIT, on CRC incidence and mortality are currently underway across the world38. These studies may provide the most rigorous evidence regarding the relative effectiveness of available screening modalities.
Observational Studies
FIT
Cohort studies have reported that exposure to biennial FIT vs no screening reduces CRC incidence by a relative 10 to 22% (HR, 0.78; 95% CI, 0.65–0.9352; IRR 0.90; 95% CI 0.83–0.9753), and CRC mortality by a relative 36 to 72% (Standardized Mortality Ratio 0.59; 95% CI: 0.37–0.3952; IRR 0.64, 95% CI 0.52–0.7853; RR 0.38, 95% CI 0.35–0.4254). The wide range of reported reductions in incidence and mortality may be attributed to different study design features (populations invited, length of follow up). Meta-analyses including a range of observational study designs have reported that FIT is associated with pooled relative reductions of 21% in CRC incidence (RR, 0.79; 95% CI, 0.69–0.92), and 59% in CRC mortality (RR, 0.41; 95% CI, 0.29–0.5938,55).
Colonoscopy
Cohort studies report exposure to colonoscopy vs no colonoscopy reduces CRC incidence by a relative 46 to 69% (HR 0.54, 95%CI: 0.31–0.9456; HR 0.44, 95% CI: 0.38–0.5257; SIR 0.33; 95% CI: 0.10–0.6258; OR 0.31, 95% CI: 0.16–0.5959), and CRC mortality by a relative 68 to 88% (HR 0.32, 95% CI: 0.24–0.4557; OR 0.12, 95% CI: 0.010.9359; HR 0.12, 95%CI: 0.02–0.8256; RR 0.40, 95% CI: 0.30–0.5560). Among older adults, one study showed a significant relative reduction for CRC for individuals age 70 to 74 (RR 0.84; 95% CI: 0.76–0.91), but not those age 75 to 79 (RR 0.95, 95% CI: 0.87–1.05)61. A more recent study utilized data from the Nurses’ Health Study and Health Professionals Follow-up Study, and found exposure to screening lower endoscopy (sigmoidoscopy or colonoscopy) after age 75 was associated with a nearly 40% relative reduction in CRC incidence (HR 0.61; 95%CI: 0.51–0.74) and 40% relative reduction in CRC mortality (HR 0.60; 95%CI: 0.46–0.78) compared to no screening62. Interestingly, reduced incidence and mortality were observed for individuals exposed, as well as those unexposed, to screening lower endoscopy prior to age 75.
Meta-analyses including a range of observational studies have reported exposure to colonoscopy vs no exposure is associated with a pooled relative 69% reduction in CRC incidence (RR, 0.31; 95% CI, 0.12–0.77), and a 68% reduction in CRC mortality (RR, 0.32; 95% CI, 0.23–0.4338,63). In pooled analyses, the relative reduction in CRC mortality was stronger for distal vs proximal CRC (distal RR, 0.18; 95% CI, 0.10–0.31; proximal RR, 0.47; 95% CI, 0.29–0.7638,63). Of note, this meta-analysis did not include all of the cohort studies evaluating colonoscopy mentioned above.
A few notable challenges exist in interpreting currently available studies with respect to the potential impact of colonoscopy on colorectal cancer screening. Some studies were unable to ascertain whether the indication for the procedure was for screening or non-screening, and others have excluded individuals with prevalent cancers at baseline in the colonoscopy exposure group, which biases towards finding favorable outcomes of colonoscopy. Thus, it is possible that available observational studies may not offer a precise estimate of the potential impact of colonoscopy-based CRC screening38.
Modelling Studies
Because of the lack of randomized trials assessing the long-term impact of currently available screening tests on CRC incidence and mortality, including a lack of trials with head-to-head comparisons of strategies, modelling studies have been employed to estimate the potential benefits and harms of proposed screening strategies. For example, the analyses used to inform current USPSTF guidelines simulated the impact of >100 different strategies for CRC screening on cases and CRC-specific deaths averted, life-years gained, and screening-related complications19. Incidence and mortality outcomes for a sample of strategies which were recommended by the USPSTF are shown in Figure 6. Overall, per 1,000 average-risk persons screened, all recommended strategies were shown to result in similar CRC cases and deaths averted. The main variation across strategies was in complications, which are directly related to the proportion of individuals in each strategy predicted to need follow up colonoscopy for an abnormal test, or for primary screening, with higher rates of complications predicted when colonoscopy is used as the first CRC screening test.
Figure 6.
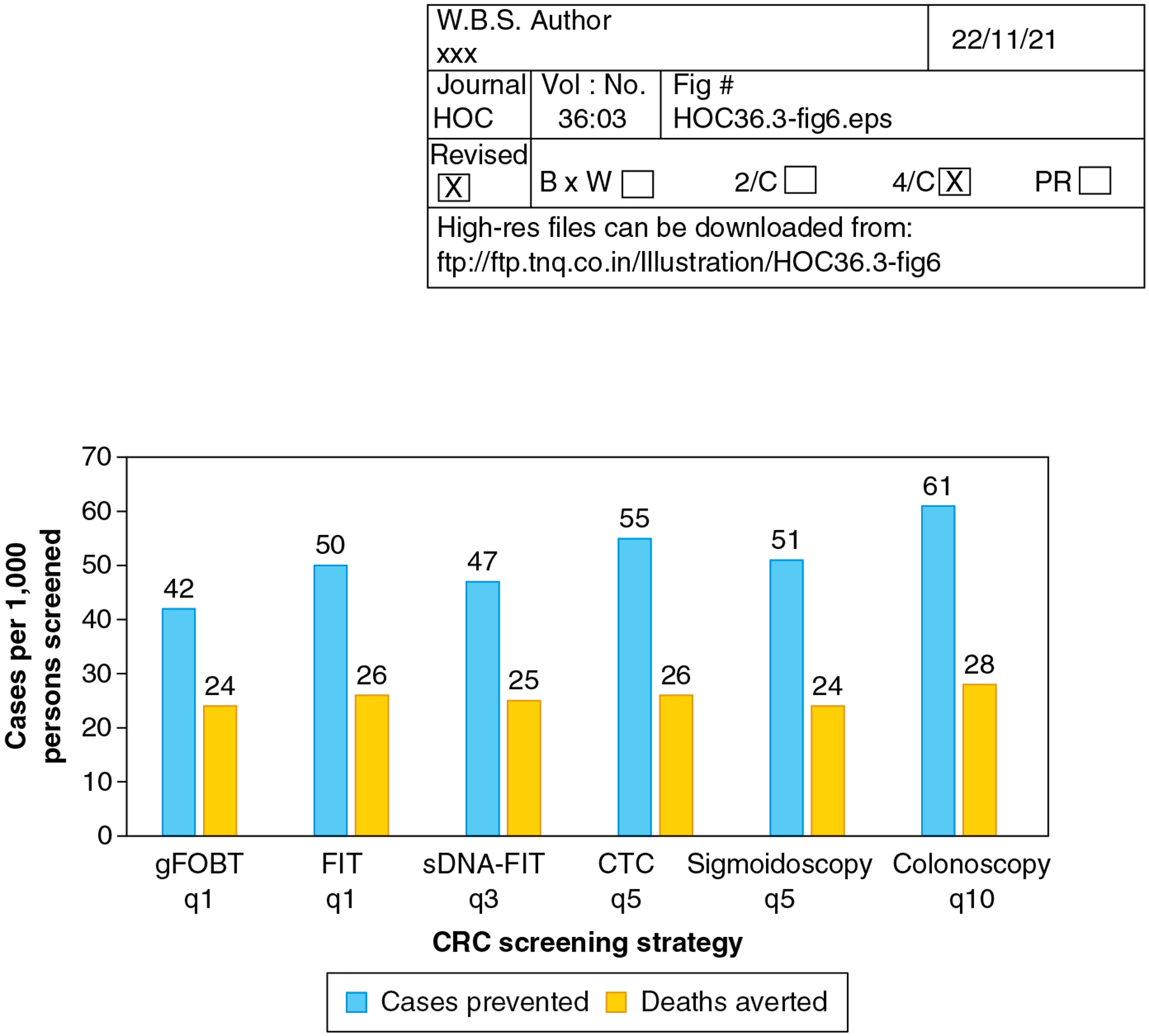
Modeled Colorectal Cancer Cases Prevented and Deaths Averted Across Screening Strategies. The number of CRC cases prevented and CRC deaths averted per 1,000 persons screened is shown for a range of strategies recommended by the US Preventive Services Task Force for screening, beginning at age 45 and continuing through age 75 years. Data show similar numbers of cases and deaths averted across all strategies. Note that the Task Force also recommended consideration of a strategy of annual sDNA-FIT, as well as sigmoidoscopy q 10 years combined with annual FIT (data not shown). CRC, colorectal cancer; gFOBT, guaiac FOBT; FIT, fecal immunochemical test; sDNA, stool DNA; CTC, CT colonography. Reference: Davidson et al. JAMA 2021.
A limitation of the modeling analysis used to inform USPSTF recommendations is that cost-effectiveness could not be reported, and that each strategy was compared under the assumption that 100% of people offered the test would complete the test and all required follow up. Cost-effectiveness analyses have been extensively done by other groups. Compared to no screening, strategies including screening with FIT, sigmoidoscopy, colonoscopy, CT colonography, sDNA-FIT, and methylated serum septin 9 have been shown to be cost effective at common thresholds for cost per quality adjusted life years gained, with some analyses demonstrating that FIT and colonoscopy could even be cost-saving64,65. Across all strategies, FIT and colonoscopy have been consistently shown to be the most cost effective. The most recent analysis available suggests that a strategy of colon capsule is not cost effective64,65. Notably, if sDNA-FIT or colon capsule were shown to be much more acceptable than other tests, the cost-effectiveness relative to other modalities could shift38,66.
Overall, modeling studies suggest comparable reductions in CRC incidence and mortality can be achieved across a wide range of screening strategies, offering the opportunity for patients, physicians, and other healthcare providers to engage in shared decision-making regarding pros and cons of various strategies, and for promotion of the concept that “the best screening test is the one that gets done”67.
Age to Initiate Screening: 45 is the new 5068
The age to initiate screening has been informed by data from epidemiologic studies, observational studies of CRC test yield, and modeling studies. Traditionally in the US, 50 has been the recommended age to initiate screening, based on prior observations that age specific incidence begins to rise substantially at this age, as well as modeling studies which suggested that initiating screening at age 50 appeared to offer the best balance of benefits and harms across the population. As mentioned previously, and covered in detail elsewhere in this issue, recent epidemiologic trends have shown that incidence under age 50 has been rising. Modeling studies taking into account these trends have suggested that initiating screening at a younger age can improve outcomes, with acceptable harms and costs. For example, the modeling study used to support current USPSTF guidelines found that initiation of screening at age 45 instead of 50 could result in additional prevention of 2 to 3 CRC cases and 1 CRC death per 1,000 persons screened. A cost-effectiveness analysis has also suggested that initiation at age 45 instead of 50 could be cost effective across a range of currently available strategies69. As a result of available evidence, the USPSTF, the American Cancer Society, and the three major gastroenterological societies in the United States (American College of Gastroenterology, American Society for Gastrointestinal Endoscopy, and American Gastroenterological Association) now recommend initiation of screening at age 45 instead of 5016,19,70.
Age to Stop Screening
Similar to for initiation of screening, the recommended age to stop CRC screening has generally been informed by epidemiologic data on age specific incidence, observational data, and modeling studies. Additionally, for older adults (defined here as individuals >75 years of age), consideration of “lag time to benefit”, in which the projected time required to benefit from an intervention (such as screening) is assessed relative to an individual’s life expectancy based on comorbidity and functional status, has been promoted as a strategy for aiding decisions on when to stop screening71. Modeling studies have suggested little to no benefit for continuing screening beyond age 75 for individuals with prior exposure to screening, and continued benefit for screening for older adults with good life expectancy who have never been exposed to screening. As mentioned previously, new evidence suggests that screening after age 75 with lower endoscopy, even among individuals with prior exposure to screening, may reduce CRC incidence and mortality62. Currently, all guidelines recommend screening for individuals through age 75, with the USPSTF recommending careful consideration of the pros and cons among individuals age 76 to 85, and against screening after age 85 years19.
Screening Guidelines
Current USPSTF guidelines for colorectal cancer screening are summarized in Table 3. Screening from age 50 to 75 is recommended as Grade A based on high certainty that the net benefit is substantial. Screening from ages 45 to 49 is recommended as Grade B based on moderate certainty that the net benefit is moderate. Screening age 76 to 85 is recommended as Grade C based on moderate certainty that continuing screening among individuals in this age group previously screened has small net benefit, and assessment that those who have never been screened in this age group are more likely to benefit. Notably, the USPSTF guidelines do not include recommendations for screening and surveillance for higher than average risk populations such as those with a family history of CRC, or a personal history of polyps, CRC, or inflammatory bowel disease; those are covered in detail elsewhere17,32,72,73.
Table 3.
2021 US Preventive Services Task Force Recommendations for Colorectal Cancer Screening
| Age Group | Recommendation | Grade of Recommendation* | Recommended Strategies |
|---|---|---|---|
| 45 to 49 years | Screen | B |
|
| 50 to 75 years | Screen | A | |
| 76 to 85 years | Selectively offer screening. Net benefit may be small, and overall health, prior screening history, and preferences should be considered. |
C |
For CRC screening, Grade A for age 50 to 75 years was based on high certainty that the net benefit is substantial, Grade B for ages 45 to 49 years was based on moderate certainty that the net benefit is moderate, and Grade C for ages 76 to 85 years was based on moderate certainty that continuing screening among individuals in this age group previously screened has small net benefit, and assessment that those who have never been screened in this age group are more likely to benefit. gFOBT, high sensitivity guaiac fecal occult blood test; FIT, fecal immunochemical test; sDNA, stool DNA; CT, computed tomographic
Challenges to Effectiveness of Screening
In current clinical and public health practice, there are three key challenges to optimizing effectiveness of CRC screening: test uptake, abnormal test follow up, and colonoscopy quality.
Variation in Test Uptake
Across the US population, marked variation and inequities in CRC screening uptake exist by age group, race/ethnicity, socioeconomic position, and insurance status. As is covered in detail elsewhere in this issue, addressing these inequities is critical to ensuring that all populations can benefit from CRC screening. Notably, health insurance is necessary to ensure access, but is insufficient for optimizing participation74,75. If access to screening is available, a number of evidence-based interventions for improving uptake exist, including mailed outreach offering FIT, patient navigation, and clinic-based strategies such as provider reminders76. Mailed FIT has been shown in meta-analyses of multiple randomized trials to result in a 28% absolute increase in proportion of individuals up-to-date with screening compared to usual care visit-based offers for screening77, and in one large health system implementation of this strategy was associated with a marked reduction in CRC incidence and mortality over time78.
Abnormal Test Follow Up
All non-colonoscopy tests require a follow up colonoscopy to complete the screening episode after an abnormal result, but achieving high rates of completion is a challenge. For example, colonoscopy completion after abnormal gFOBT or FIT ranges from 22–83%79–84, and has been particularly challenging for underserved populations. Patient navigation and putting systems in place to ensure abnormal test review, colonoscopy orders, colonoscopy scheduling, and reminders may achieve high rates of completion84. As the number of non-colonoscopy tests available for screening increases, the importance of ensuring colonoscopy completion after abnormal test results becomes increasingly paramount and should be regarded as a quality factor for ensuring individual level and population-level effectiveness of CRC screening efforts.
Colonoscopy Quality
The quality of a colonoscopy has a marked impact on risk for incident and fatal CRC. For example, a colonoscopist’s adenoma detection rate, defined as the proportion of their patients undergoing screening colonoscopy where at least one adenoma was found, has been closely linked with CRC outcomes. In one study, compared to patients who had colonoscopies performed by colonoscopists in the lowest quintile of adenoma detection, patients who had colonoscopies performed by colonoscopists in the highest quintile had a 48% relative reduction in risk for incident cancer, and a 62% relative reduction in risk for fatal colorectal cancer85. These data underscore the importance of quality insurance programs, and guiding patients to colonoscopists who measure and report quality metrics such as adenoma detection rate.
Evidence Gaps and Opportunities
Evidence gaps and opportunities with implications for CRC screening and prevention include:
Data on relative effectiveness of available CRC strategies, compared head to head
Need for novel strategies for promoting optimal uptake of CRC screening and follow up of abnormal tests
Need for policies that require measurement and public reporting of colonoscopy completion after an abnormal non-colonoscopy test
Need for highly sensitive and specific, highly acceptable non-invasive CRC screening tests
Strategies to optimize quality of colonoscopy in practice
Need for policies which promote equity in access and opportunities to complete CRC screening
Key points:
Colorectal cancer is a major public health problem
Screening reduces incidence and mortality
Current guidelines recommend routine screening from ages to 45 to 75
Recommended strategies for screening include annual guaiac fecal occult blood testing, annual fecal immununochemical testing, multitarget stool DNA-fecal immunochemical testing every 1–3 years, computed tomographic colonography every 5 years, sigmoidoscopy every 5 years alone or every 10 years paired with annual fecal immuonochemical testing, and colonoscopy every 10 years
Synopsis:
Colorectal cancer (CRC) is the 2nd leading cause of cancer death in the United States (US). Screening reduces CRC incidence and mortality. 2021 US Preventive Service Task Force (USPSTF) guidelines and available evidence support routine screening from ages 45 to 75, and individualized consideration of screening age 76 to 85. USPSTF guidelines recommend annual guaiac fecal occult blood testing, annual fecal immunochemical testing (FIT), annual to every 3-year multi-target stool DNA-FIT, every 5 year sigmoidoscopy, every 10 year sigmoidoscopy with annual FIT, every 5 year computed tomographic colonography, and every 10 year colonoscopy as options for screening, such that the “best test is the one that gets done.”
Clinical Care Points.
Colorectal cancer (CRC) is the 2nd leading cause of cancer death in the US
The natural history of CRC, which includes a precursor polyp stage, offers an opportunity for early detection and prevention of colorectal cancer
Available evidence demonstrates that screening reduces CRC incidence and mortality, and that a wide variety of CRC screening tests can achieve these outcomes
2021 US Preventive Service Task Force guidelines and available evidence support routine screening from ages 45 to 75, and individualized consideration of screening age 76 to 85
- The “best test is the one that gets done,” as 2021 US Preventive Services Task Force guidelines recommend multiple options for screening, including:
- gFOBT every year
- FIT every year
- sDNA-FIT every 1 to 3 years
- Sigmoidoscopy every 5 years or every 10 years when combined with FIT every year
- CT colonography every 5 years
- Colonoscopy every 10 years
Challenges to individual and population effectiveness of screening include variable access and uptake of screening, inconsistent rates of colonoscopy completion after abnormal non-colonoscopy tests, and variation in colonoscopy quality
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
- I serve as a local site investigator for two trials investigating blood-based biomarkers for colorectal cancer screening sponsored by Freenome Holdings, Inc, and Epigenomics AG.
- I serve as a paid consultant to Guardant Health and CellMax Life, which are developing blood-based biomarkers for colorectal cancer screening.
References
- 1.Kuipers EJ, Grady WM, Lieberman D, et al. Colorectal cancer. Nature Reviews Disease Primers. 2015;1(1):15065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Goding Sauer A, et al. Colorectal cancer statistics, 2020. CA: A Cancer Journal for Clinicians. 2020;0:1–20. [DOI] [PubMed] [Google Scholar]
- 3.Colon and Rectum SEER Incidence and U.S. Mortality Rates by Age at Diagnosis, 2014–2018. In: SEER*Explorer: An interactive website for SEER cancer statistics [Internet]. Surveillance, Epidemiology, and End Results Program, National Cancer Institute. Available at:https://seer.cancer.gov/explorer/application.html?site=20&data_type=9&graph_type=3&compareBy=rate_type&chk_rate_type_2=2&chk_rate_type_3=3&sex=1&race=1&advopt_precision=1&advopt_show_ci=on. Accessed August 27, 2021. [Google Scholar]
- 4.Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal cancer. Lancet. 2019;394(10207):1467–1480. [DOI] [PubMed] [Google Scholar]
- 5.Risk factors and protective factors. In: IARC (2019). Colorectal cancer screening. IARC Handb Cancer Prev. 17:1–300. Available from: http://publications.iarc.fr/573. [Google Scholar]
- 6.Bailie L, Loughrey MB, Coleman HG. Lifestyle risk factors for serrated colorectal polyps: a systematic review and meta-analysis. Gastroenterology. 2017;152(1):92–104. [DOI] [PubMed] [Google Scholar]
- 7.Brenner H, Altenhofen L, Stock C, Hoffmeister M. Incidence of colorectal adenomas: birth cohort analysis among 4.3 million participants of screening colonoscopy. Cancer Epidemiol Biomarkers Prev. 2014;23(9):1920–1927. [DOI] [PubMed] [Google Scholar]
- 8.Wong MCS, Huang J, Huang JLW, et al. Global Prevalence of Colorectal Neoplasia: A Systematic Review and Meta-Analysis. Clin Gastroenterol Hepatol. 2020;18(3):553–561 e510. [DOI] [PubMed] [Google Scholar]
- 9.Meester RGS, van Herk M, Lansdorp-Vogelaar I, Ladabaum U. Prevalence and clinical features of sessile serrated polyps: a systematic review. Gastroenterology. 2020;159(1):105–118 e125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Colon and Rectum SEER 5-Year Relative Survival Rates, 2011–2017. In: SEER*Explorer: An interactive website for SEER cancer statistics [Internet]. Surveillance, Epidemiology, and End Results Program, National Cancer Institute. Available at:https://seer.cancer.gov/explorer/application.html?site=20&data_type=4&graph_type=5&compareBy=stage&chk_stage_104=104&chk_stage_105=105&chk_stage_106=106&chk_stage_107=107&series=9&sex=1&race=1&age_range=1&advopt_precision=1&advopt_show_ci=on#tableWrap. Accessed August 27, 2021. [Google Scholar]
- 11.Vogelaar I, van Ballegooijen M, Schrag D, et al. How much can current interventions reduce colorectal cancer mortality in the U.S.? Cancer. 2006;107(7):1624–1633. [DOI] [PubMed] [Google Scholar]
- 12.Colon and Rectum Recent Trends in SEER Incidence and U.S. Mortality Rates, 2000–2018. In: SEER*Explorer: An interactive website for SEER cancer statistics [Internet]. Surveillance, Epidemiology, and End Results Program, National Cancer Institute. Available at:https://seer.cancer.gov/explorer/application.html?site=20&data_type=9&graph_type=2&compareBy=rate_type&chk_rate_type_2=2&chk_rate_type_3=3&sex=1&race=1&age_range=1&hdn_stage=101&advopt_precision=1&advopt_show_ci=on&advopt_display=1. Accessed August 27, 2021. [Google Scholar]
- 13.Subramanian S, Amonkar MM, Hunt TL. Use of colonoscopy for colorectal cancer screening: evidence from the 2000 National Health Interview Survey. Cancer Epidemiol Biomarkers Prev. 2005;14(2):409–416. [DOI] [PubMed] [Google Scholar]
- 14.QuickStats: Percentage of Adults Aged 50–75 Years Who Met Colorectal Cancer (CRC) Screening Recommendations — National Health Interview Survey, United States, 2018. MMWR Morb Mortal Wkly Rep 2020;69:314. DOI: 10.15585/mmwr.mm6911a7external. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wilson JMG, Jungner G. Principles and practice of screening for disease. Geneva: WHO; 1968. www.who.int/bulletin/volumes/86/4/07-050112BP.pdf. [Google Scholar]
- 16.Smith RA, Andrews KS, Brooks D, et al. Cancer screening in the United States, 2019: a review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J Clin. 2019;69(3):184–210. [DOI] [PubMed] [Google Scholar]
- 17.Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Am J Gastroenterol. 2017;112(7):1016–1030. [DOI] [PubMed] [Google Scholar]
- 18.Provenzale D, Ness RM, Llor X, et al. NCCN guidelines insights: colorectal cancer screening, version 2.2020. J Natl Compr Canc Netw. 2020;18(10):1312–1320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.U. S. Preventive Services Task Force, Davidson KW, Barry MJ, et al. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965–1977. [DOI] [PubMed] [Google Scholar]
- 20.IARC (2019). Colorectal cancer screening. IARC Handb Cancer Prev. 17:1–300. Available from: http://publications.iarc.fr/573. [Google Scholar]
- 21.Lin JS, Perdue LA, Henrikson NB, Bean SI, Blasi PR. Screening for colorectal cancer: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2021;325(19):1978–1998. [DOI] [PubMed] [Google Scholar]
- 22.Ibuka T, Adachi S, Horibe Y, et al. Effects of antithrombotic drugs on the results of fecal immunochemical test in colorectal neoplasms screening. Sci Rep. 2021;11(1):4348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Niikura R, Yamada A, Fujishiro M, et al. The effects of direct oral anticoagulants, warfarin, aspirin and thienopyridine on the performance of immunochemical, faecal, occult blood tests. Digestion. 2019;100(2):117–126. [DOI] [PubMed] [Google Scholar]
- 24.Tsuji Y, Gunji T, Sato H, et al. Antithrombotic drug does not affect the positive predictive value of an immunochemical fecal occult blood test. Dig Endosc. 2014;26(3):424–429. [DOI] [PubMed] [Google Scholar]
- 25.Levi Z, Rozen P, Hazazi R, et al. Sensitivity, but not specificity, of a quantitative immunochemical fecal occult blood test for neoplasia is slightly increased by the use of low-dose aspirin, NSAIDs, and anticoagulants. Am J Gastroenterol. 2009;104(4):933–938. [DOI] [PubMed] [Google Scholar]
- 26.Randel KR, Botteri E, Romstad KMK, et al. Effects of oral anticoagulants and aspirin on performance of fecal immunochemical tests in colorectal cancer screening. Gastroenterology. 2019;156(6):1642–1649 e1641. [DOI] [PubMed] [Google Scholar]
- 27.Bujanda L, Sarasqueta C, Lanas A, et al. Effect of oral anticoagulants on the outcome of faecal immunochemical test. Br J Cancer. 2014;110(5):1334–1337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sawhney MS, McDougall H, Nelson DB, Bond JH. Fecal occult blood test in patients on low-dose aspirin, warfarin, clopidogrel, or non-steroidal anti-inflammatory drugs. Dig Dis Sci. 2010;55(6):1637–1642. [DOI] [PubMed] [Google Scholar]
- 29.Imperiale TF, Ransohoff DF, Itzkowitz SH, et al. Multitarget stool DNA testing for colorectal-cancer screening. New England Journal of Medicine. 2014;370(14):1287–1297. [DOI] [PubMed] [Google Scholar]
- 30.Knudsen AB, Rutter CM, Peterse EFP, et al. Colorectal cancer screening: an updated modeling study for the US Preventive Services Task Force. JAMA. 2021;325(19):1998–2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pickhardt PJ, Hassan C, Halligan S, Marmo R. Colorectal cancer: CT colonography and colonoscopy for detection--systematic review and meta-analysis. Radiology. 2011;259(2):393–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gupta S, Lieberman D, Anderson JC, et al. Recommendations for follow-up after colonoscopy and polypectomy: a consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2020;158(4):1131–1153 e1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cash BD, Fleisher MR, Fern S, et al. Multicentre, prospective, randomised study comparing the diagnostic yield of colon capsule endoscopy versus CT colonography in a screening population (the TOPAZ study). Gut. 2020. [DOI] [PubMed] [Google Scholar]
- 34.Voska M, Zavoral M, Grega T, et al. Accuracy of colon capsule endoscopy for colorectal neoplasia detection in individuals referred for a screening colonoscopy. Gastroenterol Res Pract. 2019;2019:5975438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rex DK, Adler SN, Aisenberg J, et al. Accuracy of capsule colonoscopy in detecting colorectal polyps in a screening population. Gastroenterology. 2015;148(5):948–957 e942. [DOI] [PubMed] [Google Scholar]
- 36.Potter NT, Hurban P, White MN, et al. Validation of a real-time PCR-based qualitative assay for the detection of methylated SEPT9 DNA in human plasma. Clin Chem. 2014;60(9):1183–1191. [DOI] [PubMed] [Google Scholar]
- 37.Young GP, Senore C, Mandel JS, et al. Recommendations for a step-wise comparative approach to the evaluation of new screening tests for colorectal cancer. Cancer. 2016;122(6):826–839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Studies of colorectal cancer screening. In: IARC (2019). Colorectal cancer screening. IARC Handb Cancer Prev. 17:1–300. Available from: http://publications.iarc.fr/573. [Google Scholar]
- 39.Fitzpatrick-Lewis D, Ali MU, Warren R, Kenny M, Sherifali D, Raina P. Screening for colorectal cancer: a systematic review and meta-analysis. Clin Colorectal Cancer. 2016;15(4):298–313. [DOI] [PubMed] [Google Scholar]
- 40.Hardcastle JD, Chamberlain JO, Robinson MH, et al. Randomised controlled trial of faecal-occult-blood screening for colorectal cancer. Lancet. 1996;348(9040):1472–1477. [DOI] [PubMed] [Google Scholar]
- 41.Kronborg O, Jorgensen OD, Fenger C, Rasmussen M. Randomized study of biennial screening with a faecal occult blood test: results after nine screening rounds. Scand J Gastroenterol. 2004;39(9):846–851. [DOI] [PubMed] [Google Scholar]
- 42.Lindholm E, Brevinge H, Haglind E. Survival benefit in a randomized clinical trial of faecal occult blood screening for colorectal cancer. Br J Surg. 2008;95(8):1029–1036. [DOI] [PubMed] [Google Scholar]
- 43.Pitkaniemi J, Seppa K, Hakama M, et al. Effectiveness of screening for colorectal cancer with a faecal occult-blood test, in Finland. BMJ Open Gastroenterol. 2015;2(1):e000034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Shaukat A, Mongin SJ, Geisser MS, et al. Long-term mortality after screening for colorectal cancer. N Engl J Med. 2013;369(12):1106–1114. [DOI] [PubMed] [Google Scholar]
- 45.Schoen RE, Pinsky PF, Weissfeld JL, et al. Colorectal-cancer incidence and mortality with screening flexible sigmoidoscopy. N Engl J Med. 2012;366(25):2345–2357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Atkin WS, Edwards R, Kralj-Hans I, et al. Once-only flexible sigmoidoscopy screening in prevention of colorectal cancer: a multicentre randomised controlled trial. Lancet. 2010;375(9726):1624–1633. [DOI] [PubMed] [Google Scholar]
- 47.Atkin W, Wooldrage K, Parkin DM, et al. Long term effects of once-only flexible sigmoidoscopy screening after 17 years of follow-up: the UK Flexible Sigmoidoscopy Screening randomised controlled trial. Lancet. 2017;389(10076):1299–1311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Holme O, Loberg M, Kalager M, et al. Effect of flexible sigmoidoscopy screening on colorectal cancer incidence and mortality: a randomized clinical trial. JAMA. 2014;312(6):606–615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Holme O, Loberg M, Kalager M, et al. Long-term effectiveness of sigmoidoscopy screening on colorectal cancer incidence and mortality in women and men: a randomized trial. Ann Intern Med. 2018;168(11):775–782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Segnan N, Armaroli P, Bonelli L, et al. Once-only sigmoidoscopy in colorectal cancer screening: follow-up findings of the Italian Randomized Controlled Trial--SCORE. J Natl Cancer Inst. 2011;103(17):1310–1322. [DOI] [PubMed] [Google Scholar]
- 51.Tinmouth J, Vella ET, Baxter NN, et al. Colorectal cancer screening in average risk populations: evidence summary. Can J Gastroenterol Hepatol. 2016;2016:2878149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ventura L, Mantellini P, Grazzini G, et al. The impact of immunochemical faecal occult blood testing on colorectal cancer incidence. Dig Liver Dis. 2014;46(1):82–86. [DOI] [PubMed] [Google Scholar]
- 53.Giorgi Rossi P, Vicentini M, Sacchettini C, et al. Impact of screening program on incidence of colorectal cancer: a cohort study in Italy. Am J Gastroenterol. 2015;110(9):1359–1366. [DOI] [PubMed] [Google Scholar]
- 54.Chiu HM, Chen SL, Yen AM, et al. Effectiveness of fecal immunochemical testing in reducing colorectal cancer mortality from the One Million Taiwanese Screening Program. Cancer. 2015;121(18):3221–3229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zhang J, Cheng Z, Ma Y, et al. Effectiveness of screening modalities in colorectal cancer: a network meta-analysis. Clin Colorectal Cancer. 2017;16(4):252–263. [DOI] [PubMed] [Google Scholar]
- 56.Lee JK, Jensen CD, Levin TR, et al. Long-term risk of colorectal cancer and related deaths after a colonoscopy with normal findings. JAMA Intern Med. 2019;179(2):153–160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Nishihara R, Wu K, Lochhead P, et al. Long-term colorectal-cancer incidence and mortality after lower endoscopy. N Engl J Med. 2013;369(12):1095–1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kahi CJ, Imperiale TF, Juliar BE, Rex DK. Effect of screening colonoscopy on colorectal cancer incidence and mortality. Clin Gastroenterol Hepatol. 2009;7(7):770–775; quiz 711. [DOI] [PubMed] [Google Scholar]
- 59.Manser CN, Bachmann LM, Brunner J, Hunold F, Bauerfeind P, Marbet UA. Colonoscopy screening markedly reduces the occurrence of colon carcinomas and carcinoma-related death: a closed cohort study. Gastrointest Endosc. 2012;76(1):110–117. [DOI] [PubMed] [Google Scholar]
- 60.Eldridge RC, Doubeni CA, Fletcher RH, et al. Uncontrolled confounding in studies of screening effectiveness: an example of colonoscopy. J Med Screen. 2013;20(4):198–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Garcia-Albeniz X, Hsu J, Bretthauer M, Hernan MA. Effectiveness of screening colonoscopy to prevent colorectal cancer among medicare beneficiaries aged 70 to 79 years: a prospective observational study. Ann Intern Med. 2017;166(1):18–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ma W, Wang K, Nguyen LH, et al. Association of screening lower endoscopy with colorectal cancer incidence and mortality in adults older than 75 years. JAMA Oncology. 2021;7(7):985–992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Brenner H, Stock C, Hoffmeister M. Effect of screening sigmoidoscopy and screening colonoscopy on colorectal cancer incidence and mortality: systematic review and meta-analysis of randomised controlled trials and observational studies. BMJ. 2014;348:g2467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Peterse EFP, Meester RGS, de Jonge L, et al. Comparing the cost-effectiveness of innovative colorectal cancer screening tests. J Natl Cancer Inst. 2021;113(2):154–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ladabaum U Cost-effectiveness of current colorectal cancer screening tests. Gastrointest Endosc Clin N Am. 2020;30(3):479–497. [DOI] [PubMed] [Google Scholar]
- 66.Deibel A, Deng L, Cheng CY, et al. Evaluating key characteristics of ideal colorectal cancer screening modalities: the microsimulation approach. Gastrointest Endosc. 2021;94(2):379–390 e377. [DOI] [PubMed] [Google Scholar]
- 67.Lieberman D Challenges for colorectal cancer screening decision modeling. Gastrointest Endosc. 2021;94(2):391–393. [DOI] [PubMed] [Google Scholar]
- 68.Ng K, May FP, Schrag D. US Preventive Services Task Force recommendations for colorectal cancer screening: forty-five is the new fifty. JAMA. 2021;325(19):1943–1945. [DOI] [PubMed] [Google Scholar]
- 69.Ladabaum U, Mannalithara A, Meester RGS, Gupta S, Schoen RE. Cost-effectiveness and national effects of initiating colorectal cancer screening for average-risk persons at age 45 years instead of 50 years. Gastroenterology. 2019;157(1):137–148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.GI societies support starting CRC screening in mid-to-late 40s. In: American Gastroenterological Association. 2021. Available at: https://gastro.org/press-releases/gi-societies-support-starting-crcscreening-in-mid-to-late-40s/. Accessed August 31, 2021. [Google Scholar]
- 71.May FP, Gupta S. When should screening stop for elderly individuals at average and increased risk for colorectal cancer? Clin Gastroenterol Hepatol. 2018;16(2):178–180 e171. [DOI] [PubMed] [Google Scholar]
- 72.Kahi CJ, Boland CR, Dominitz JA, et al. Colonoscopy surveillance after colorectal cancer resection: recommendations of the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2016;150(3):758–768 e711. [DOI] [PubMed] [Google Scholar]
- 73.Murthy SK, Feuerstein JD, Nguyen GC, Velayos FS. AGA clinical practice update on endoscopic surveillance and management of colorectal dysplasia in inflammatory bowel diseases: expert review. Gastroenterology. 2021;161(3):1043–1051 e1044. [DOI] [PubMed] [Google Scholar]
- 74.May FP, Yang L, Corona E, Glenn BA, Bastani R. Disparities in colorectal cancer screening in the United States before and after implementation of the Affordable Care Act. Clin Gastroenterol Hepatol. 2020;18(8):1796–1804 e1792. [DOI] [PubMed] [Google Scholar]
- 75.Demb J, Gupta S. Racial and ethnic disparities in colorectal cancer screening pose persistent challenges to health equity. Clin Gastroenterol Hepatol. 2020;18(8):1691–1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Dougherty MK, Brenner AT, Crockett SD, et al. Evaluation of interventions intended to increase colorectal cancer screening rates in the United States: a systematic review and meta-analysis. JAMA Intern Med. 2018;178(12):1645–1658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Jager M, Demb J, Asghar A, et al. Mailed outreach is superior to usual care alone for colorectal cancer screening in the USA: a systematic review and meta-analysis. Dig Dis Sci. 2019;64(9):2489–2496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Levin TR, Corley DA, Jensen CD, et al. Effects of organized colorectal cancer screening on cancer incidence and mortality in a large community-based population. Gastroenterology. 2018;155(5):1383–1391 e1385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Jimbo M, Myers RE, Meyer B, et al. Reasons patients with a positive fecal occult blood test result do not undergo complete diagnostic evaluation. Ann Fam Med. 2009;7(1):11–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Humphrey LL, Shannon J, Partin MR, O’Malley J, Chen Z, Helfand M. Improving the follow-up of positive hemoccult screening tests: an electronic intervention. J Gen Intern Med. 2011;26(7):691–697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Rao SK, Schilling TF, Sequist TD. Challenges in the management of positive fecal occult blood tests. J Gen Intern Med. 2009;24(3):356–360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Chubak J, Garcia MP, Burnett-Hartman AN, et al. Time to colonoscopy after positive fecal blood test in four U.S. health care systems. Cancer Epidemiol Biomarkers Prev. 2016;25(2):344–350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Bharti B, May FFP, Nodora J, et al. Diagnostic colonoscopy completion after abnormal fecal immunochemical testing and quality of tests used at 8 Federally Qualified Health Centers in Southern California: Opportunities for improving screening outcomes. Cancer. 2019;125(23):4203–4209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Selby K, Jensen CD, Zhao WK, et al. Strategies to improve follow-up after positive fecal immunochemical tests in a community-based setting: a mixed-methods study. Clin Transl Gastroenterol. 2019;10(2):e00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Corley DA, Jensen CD, Marks AR, et al. Adenoma detection rate and risk of colorectal cancer and death. N Engl J Med. 2014;370(14):1298–1306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.American Cancer Society. Cancer Facts & Figures 2021. Atlanta: American Cancer Society; 2021. [Google Scholar]


