Abstract
Background and aims
Cirrhotic cardiomyopathy (CCM) is a term used to collectively describe abnormal structural and functional changes in heart of patients with cirrhosis. The present study was undertaken to find the prevalence of CCM in patients with liver cirrhosis and its predictors. We also followed these patients to evaluate the role of CCM in the development of hepatorenal syndrome (HRS).
Materials & methods
This was a prospective study carried out in department of Gastroenterology, Sir Ganga Ram hospital, New Delhi. A total of 104 patients with liver cirrhosis were included. Liver cirrhosis was diagnosed on basis of clinical, biochemical, and imaging features. CCM was defined based on echocardiography. Dobutamine stress echocardiography and hepatic venous pressure gradient (HVPG) were performed in patients who gave consent. HRS was defined as per standard criteria. Patients with CCM were followed for development of HRS.
Results
Fifty (48%) patients were diagnosed with CCM. All patients had diastolic dysfunction, and none had systolic dysfunction. Median age of patients with CCM was significantly higher (59 [31–78 y] vs. 52 [24–70 y], P < 0.05). Severity of liver disease (Child Turcotte Pugh score and model for end-stage liver disease score) and portal pressures (HVPG) did not differ in patients with or without CCM. Patients with CCM did not have increased incidence of HRS at the end of 6-month follow-up study.
Conclusion
The presence of CCM was not related with the severity of liver dysfunction or portal pressures. Age was a significant determinant of CCM. Diastolic cardiac dysfunction does not influence the occurrence of HRS.
Keywords: cirrhosis, cardiomyopathy, diastolic cardiac dysfunction, hepatorenal syndrome
Abbreviations: CCM, cirrhotic cardiomyopathy; CTP, Child Turcotte Pugh; DD, diastolic dysfunction; DSE, dobutamine stress echocardiography; FHVP, free hepatic venous pressure; HRS, hepatorenal syndrome; HVPG, hepatic venous pressure gradient; LVEF, left ventricular ejection fraction; MELD, model for end-stage liver disease; TDI, tissue Doppler imaging; 2D echo, two-dimensional echocardiography
The term cirrhotic cardiomyopathy (CCM) was first coined by Lee et al.1 It collectively describes the effects of cirrhosis-induced cardiovascular changes in circulation as well as mechanical and electrical functions of the heart. Initially, the hyperdynamic circulation seen in patients with cirrhosis was attributed to the effects of alcohol, as occurs in beriberi-associated cardiomyopathy.2 Reports of unexpected deaths due to heart failure after liver transplantation,3 transjugular intrahepatic porto-systemic stent shunt,4 and surgical portocaval shunts5 led to interest in investigating for cardiac dysfunction related to cirrhosis.
The pathogenesis of CCM is related to splanchnic vasodilatation as a result of portal hypertension. Redistribution of the circulating blood volume results in a reduced effective central blood volume leading to “effective hypovolemia”.6 This causes volume- and baroreceptor-mediated activation of potent vasoconstrictor systems, such as the sympathetic nervous system and renin–angiotensin–aldosterone axis,7 which further aggravates the hyperdynamic circulation and causes cardiac strain. These alterations in the hemodynamics and induced cardiac strain lead to development of CCM.6
CCM predominantly manifests as diastolic dysfunction (DD), characterized by an abnormal pattern of transmitral flow and increased atrial contribution to the late ventricular filling. On two-dimensional echocardiography (2D echo), this is seen as a reduced E wave (early diastole) to A wave (atrial contraction) ratio. Tissue Doppler imaging (TDI) is now considered a better method to estimate DD as it is less influenced by preload and afterload on the cardiac musculature.8, 9, 10
CCM can manifest as systolic dysfunction also which results in decreased cardiac reserve during physiological or pharmacological stress.11,12 Other observed defects in CCM include electrophysiological abnormalities like chronotropic incompetence, electromechanical uncoupling, prolonged QT interval, and humoral changes like elevated brain natriuretic peptide, which often correlates with prolonged QT interval.13
The exact determinants of CCM have not been clearly elucidated. Whether it is the severity of liver disease or degree of portal hypertension which determines the occurrence of cardiac dysfunction in liver cirrhosis is yet to be clearly established. Hepatic venous pressure gradient (HVPG) is the most accurate measurement of portal hypertension. There are no studies which have investigated the relationship of HVPG with CCM. Hemodynamic changes induced by cirrhosis ultimately progress to renal vasoconstriction and trigger hepatorenal syndrome (HRS). There have been very few studies which have prospectively evaluated the development of HRS in patients with CCM.14
We conducted this prospective study to determine the prevalence and predictors of CCM in patients with liver cirrhosis. We also studied the relation of CCM with HVPG and followed these patients for development of HRS.
Materials and methods
Sample Collection
This prospective study was conducted in the Department of Gastroenterology, Sir Ganga Ram Hospital, New Delhi, from 1st July 2014 to 1st January 2016. All adult patients (>18 years) diagnosed with liver cirrhosis, based on clinical, biochemical, radiological features, were evaluated. CCM was diagnosed by the consensus guidelines.15
-
1.
Abnormal systolic contractile responses to stress (systolic dysfunction) and/or
-
2.
DD at rest and
-
3.
Absence of clinically significant cardiopulmonary disease.
The following patients were excluded from the study.
-
1)
Any history of cardiovascular disease or on active treatment for cardiac illness (beta blockers were stopped at least 7 days before evaluation)
-
2)
Active sepsis requiring organ support like ventilator or inotropic support.
-
3)
Severe pulmonary hypertension (systolic pulmonary artery pressure >40 mm Hg)
-
4)
Patients with hemochromatosis/glycogen storage disorders.
-
5)
Patients with alcoholic cirrhosis with abstinence of less than 6 months.
-
6)
Patients with diabetes mellitus on treatment or newly diagnosed diabetes (fasting blood glucose >126 gm%, postprandial blood sugar >200 gm%, HbA1c > 6.5%)
-
7)
Known cases of systemic hypertension on treatment or newly diagnosed hypertensives (≥3 consecutive blood pressure (BP) readings >140/90 mm Hg).
-
8)
Child score ≥13, grade 2 encephalopathy, severe degree of ascites not controlled with medical management.
-
9)
Patients not willing to sign informed written consent.
All patients underwent routine biochemical and hematological investigations and investigation for etiology of liver cirrhosis. 2D echo with TDI was performed for all patients. HVPG and dobutamine stress echocardiography (DSE) were performed for patients who consented for the same.
2D echo examinations were carried out by an experienced cardiologist (RM) using Vivid System Seven (GE,Vingmed, Milwaukee,USA) at 3.5 MHz. The procedure was performed with patients in left lateral decubitus, and the evaluation was made through parasternal long and short axes and apical four-chamber views. Left ventricular end-diastolic diameter, interventricular septal thickness, and left atrial diameter were measured with the 2D method in the parasternal long axis. Left atrial volume and area were calculated in the apical four-chamber views. Left ventricular volumes and ejection fraction were estimated using Simpson's biplane method. A left ventricular ejection fraction (LVEF) above 50% was considered normal. Spectral Doppler measurements were obtained with the transducer in the apical four-chamber view and the Doppler beam aligned perpendicularly to the plane of the mitral annulus.
DD was diagnosed when at least one of the following was present:
-
1.
E/A ratio (age corrected) < 1.0 or
-
2.
Prolonged deceleration time (>200 ms) or
-
3.
Prolonged isovolumic relaxation time (>80 ms)
DD was categorized into 3 grades based on E (early diastolic) and A (late diastolic) velocities. Grades of DD was classified as grade 1 (mild DD or impaired relaxation phase: E/A <0.8, DT > 200 ms), grade 2 (moderate DD or pseudonormal phase: E/A 0.8–1.5, DT 160–200 ms), and grade 3 (severe DD or restrictive filling phase: E/A ≥2, DT < 160 ms).
Systolic dysfunction was diagnosed when at least one of the following was present:
-
1.
Blunted increase in cardiac output with pharmacological stimuli (dobutamine). An abnormal response was defined as a decrease of less than 10% in LV end-diastolic volume, a decrease of less than 20% in end-systolic volume, and an increase of less than 10% in LVEF at peak dobutamine infusion (40 μg/kg/min) or
-
2.
Resting LVEF <50%
HVPG was performed by the standard technique.16 A balloon catheter was introduced into the right hepatic vein under fluoroscopic guidance. The zero-reference point was set at the mid-axillary point. The free hepatic venous pressure (FHVP) was obtained by keeping the catheter free into the lumen of the hepatic vein. The balloon of the catheter was then inflated to wedge the lumen of the hepatic vein. The pressure tracing at this juncture showing the absence of wave forms was labeled as wedged hepatic venous pressure (WHVP). HVPG was obtained by subtracting FHVP from WHVPs (HVPG = WHVP – FHVP). The normal value of HVPG in our hemodynamic laboratory is between 1 and 4 mm Hg.
The presence of HRS was diagnosed based on criteria proposed by the International Ascites Club's Diagnostic Criteria of HRS.17
Patient Follow-up
Patients who were included in the study were followed up for six months from the date of inclusion in the study.
Statistical Analysis
Statistical testing was conducted with a statistical package for the social science system (SPSS), version 20.0. Continuous variables were presented as mean (standard deviation) or median (range) if the data were unevenly distributed. Categorical variables were expressed as frequencies and percentages. The comparison of normally distributed continuous variables between the groups was performed using the Student’s T-test. Nominal categorical data between the groups were compared using the chi-square test or Fischer's exact test as appropriate. Non-normal distribution continuous variables were compared using the Mann Whitney U test. A multivariate logistic regression model was used if required to identify independent risk factors for cardiac complications. A stepwise approach was used to enter new terms into the model with a limit of P < 0.05 to enter the terms. For all statistical tests, a P-value of less than 0.05 was taken to indicate a significant difference.
Results
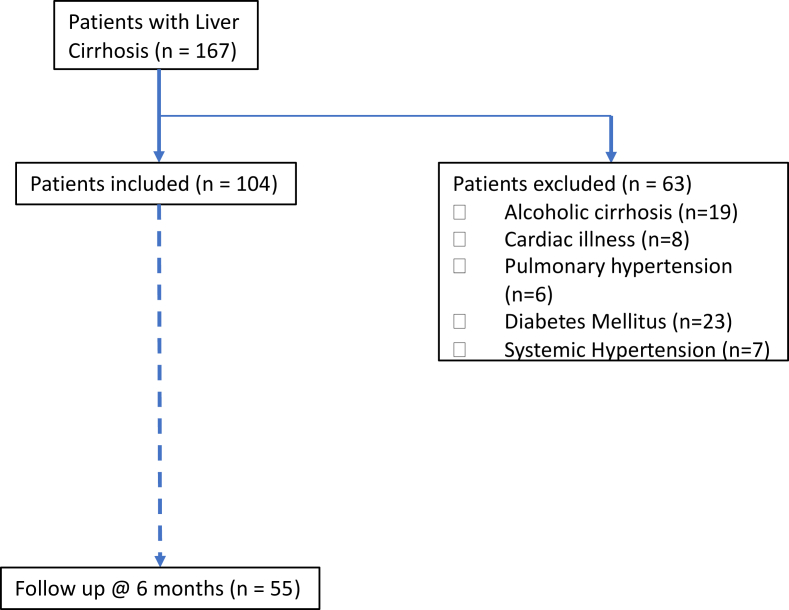
A total of 167 patients with cirrhosis were screened; of these, 104 patients were included in the analysis after exclusion (Figure 1). The median age of patients was 55 years (range 24–78 years), and majority were men (n = 80, 77%). The most common etiology of cirrhosis was alcohol (n = 82, 79%) followed by viral Hepatitiis B Virus and Hepatitis C Virus (HBV/HCV) etiology (n = 17, 16%). The median Child Turcotte Pugh (CTP) score was 8 (range 5–13), and the median model for end-stage liver disease (MELD) score was 12.9 (range 6.4–27.9). The highest proportion of patients belonged to Child class B (n = 56, 54%). Various biochemical parameters of the cohort are shown in Table 1. HVPG was performed in 42 patients who gave consent. The median HVPG was 15 mm of Hg (range 5–30 mmHg).
Figure 1.
Patient enrollment.
Table 1.
Patient Demography.
| Parameter | Value [(median/range); (no./percentage)] |
|---|---|
| Age (years) | 55 (24–78) |
| Sex | |
| Males | 80 (77%) |
| Females | 24 (23%) |
| Etiology | |
| Alcohol | 82 (79%) |
| NAFLD/cryptogenic | 5 (5%) |
| Viral | 17 (16%) |
| Biochemical parameters | |
| Hemoglobin (gm/dL) | 9.3 (8–16.5) |
| Platelet count (x103/mm3) | 85.5 (14–335) |
| Serum creatinine (mg%) | 0.89 (0.4–1.5) |
| Serum sodium (mEq/L) | 137.5 (121–155) |
| Total bilirubin (mg%) | 1.58 (0.3–5.7) |
| Serum albumin (gm%) | 2.5 (1.7–4.5) |
| PT-INR | 1.3 (0.9–3.3) |
| Child class (overall) | |
| Child A | 21 (20%) |
| Child B | 56 (54%) |
| Child C | 27 (26%) |
| CTP score | 8 (5, 6, 7, 8, 9, 10, 11, 12, 13) |
| MELD score | 12.9 (6.4–27.9) |
| HVPG (n=42) mmHg | 15 (4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30) |
Abbreviations: NAFLD: nonalcoholic fatty liver disease; no.: number; gm: gram; dL: deciliter; mg: milligram; mEq: milliequivalent; PT-INR: prothrombin time international normalized ratio; CTP: Child Turcotte Pugh; MELD: model for end-stage liver disease; HVPG: hepatic venous pressure gradient.
CCM was seen in 50 of 104 (48%) patients, and all these patients had DD. Eighty-four percent (n = 42) of the patients had grade 1; 14% patients (n = 7) had grade 2, whereas only 2% patients (n = 1) had grade 3 DD. DSE was performed in 29 patients, and none of them showed features of systolic dysfunction.
Predictors of CCM
Median age of patients with DD was significantly higher than that of those without DD (59 [31–78 y] vs. 52 [24–70 y], P < 0.05). There was no gender predilection in patients with CCM versus those without CCM (M; F, 43:11 vs. 38:12, P = 0.64). There was no statistical difference in the hematological and biochemical parameters between the two groups (Table 2).
Table 2.
Comparison of Patients With and Without CCM.
| Parameters | CCM absent (n = 54) | CCM present (n = 50) | P-value |
|---|---|---|---|
| Age (median) (years) | 59 | 52 | <0.05 |
| Gender | |||
| Males | 40 (74%) | 40 (80%) | 0.64 |
| Females | 14 (26%) | 10 (20%) | |
| Hemoglobin (gm/dL) | 9 (7–16.5) | 9.7 (7–14.6) | 0.14 |
| Platelet count (x103/mm3) | 85 (24–279) | 86.5 (14–335) | 0.66 |
| Serum creatinine (mg%) | 0.88 (0.4–1.5) | 0.91 (0.5–1.5) | 0.47 |
| Serum sodium (mEq/L) | 138 (123–147) | 137 (121–155) | 0.72 |
| Total bilirubin (mg%) | 1.6 (0.3–5.5) | 1.5 (0.4–5.7) | 0.88 |
| Serum albumin (gm%) | 2.4 (1.7–4.3) | 2.6 (1.9–4.5) | 0.19 |
| PT-INR | 1.3 (0.9–3.0) | 1.3 (1–3.3) | 0.77 |
| CTP score | |||
| A | 9 (16%) | 12 (24%) | 0.06 |
| B and C | 45 (84%) | 38 (76%) | |
Abbreviations: CCM: cirrhotic cardiomyopathy; gm: gram; dL: deciliter; mg: milligram; mEq: milliequivalent; PT-INR: prothrombin time international normalized ratio; CTP: Child Turcotte Pugh.
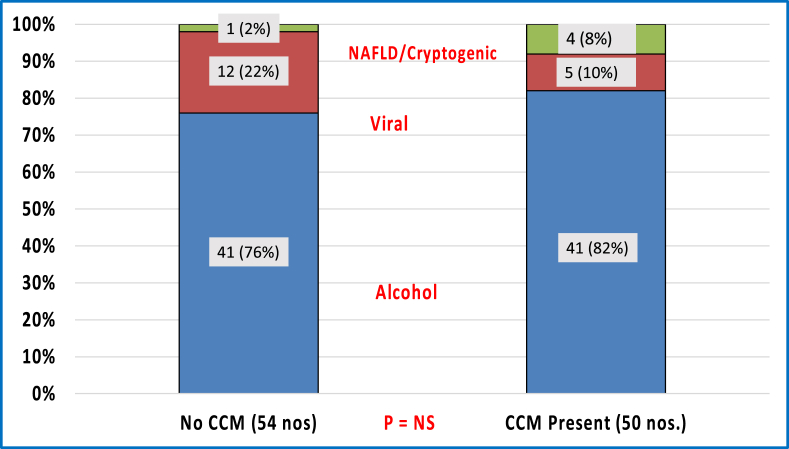
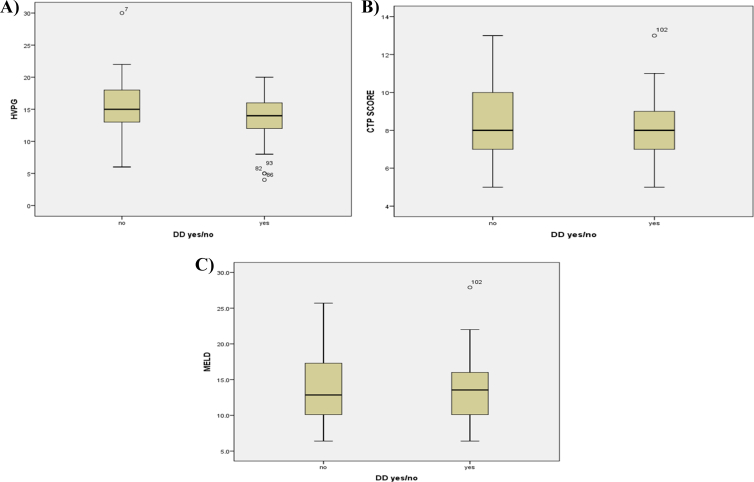
Prevalence of CCM was same across all the etiologies of liver cirrhosis (Figure 2). The median CTP scores in patients with CCM (8, range 5–13) and without CCM (8, range 5–13) were not different statistically (P = 0.79). The median MELD score in patients with CCM was higher than that in those without CCM, but it was not significant statistically (13.5 [6.4–27.9] vs. 12.8 [6.4–25.7]) (P = 0.95) (Figure 3). Although the differences were not statistically significant, median HVPG in patients with DD was lower than that in those without DD (14 [6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20] vs. 15 [6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30] mm Hg, P = 0.32), and patients with ascites had higher prevalence of DD (60%) than those without ascites (40%) (P = 0.33).
Figure 2.
Cirrhotic etiology in patients with and without CCM. Abbreviations: NAFLD: nonalcoholic fatty liver disease; CCM: cirrhotic cardiomyopathy; NS: not significant.
Figure 3.
Comparison of HVPG, CTP, and MELD scores in patients with and without CCM; (A) HVPG, (B) CTP score, and (C) MELD score. None of the parameters showed any statistical difference between the two groups. Abbreviations: CTP: Child Turcotte Pugh; MELD: model for end-stage liver disease; HVPG: hepatic venous pressure gradient.
Comparison of the 2D echo parameters between the patients with or without DD showed a significant difference in the left ventricular end diastolic diameters (48 vs. 45 mm, P = 0.01). Aortic, left atrial, and left ventricular end systolic diameters and LVEFs showed no statistical difference in patients with or without CCM (Table 3).
Table 3.
2D Echo Parameters in Patients With and Without CCM.
| Parameter | No CCM (absent DD) | CCM present (DD present) | P value |
|---|---|---|---|
| Aorta (ed), mm | 30 (22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35) | 30.5 (25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39) | 0.17 |
| Lt atrium (es), mm | 35 (22–52) | 35 (26–50) | 0.75 |
| LVID, mm | 45 (27–57) | 48 (38–57) | 0.01 |
| LVIS, mm | 26 (20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37) | 27 (20–40) | 0.13 |
| LVEF, % | 65 (60–70) | 65 (55–70) | 0.57 |
Abbreviations: LVID: left ventricular end diastolic diameter; LVIS: left ventricular end systolic diameter; LVEF: ejection fraction; ed: end-diastolic; es: end-systolic; mm: millimeter; CCM: cirrhotic cardiomyopathy.
Bold values are representes the P < 0.05.
Development of HRS
Of the total 104 patients enrolled in the study, follow-up data based on repeat admissions or outpatient follow-up were available in 55 patients (28 patients with DD and 27 without DD) (Table 3). Among these, the occurrence of at least one episode of HRS was documented in 27 patients. HRS was seen in 14 of 28 (50%) patients with DD and in 13 of 27 (48%) patients without DD (P = NS). Patients who developed HRS had a higher CTP score and MELD score (10 [7, 8, 9, 10, 11, 12, 13] vs. 8 [6, 7, 8, 9, 10, 11], P = 0.002; MELD 15.9 [9.3–27.9] vs. 12.1 [7.4–17.6], P = 0.001). HVPG values were significantly higher in patients with HRS (15.5 [13–20.5] vs. 13 [3.5–18] P = 0.007) (Table 4).
Table 4.
Comparison of Patients With and Without HRS.
| Parameter | No HRS (n = 28) | HRS present (n = 27) | P value |
|---|---|---|---|
| Age | 56 (28–71) | 55 (27–72) | NS |
| CTP score | 8 (6, 7, 8, 9, 10, 11) | 10 (7, 8, 9, 10, 11, 12, 13) | 0.002 |
| MELD score | 12.1 (7.4–17.6) | 15.9 (9.3–27.9) | 0.001 |
| HVPG | 13 (3.5–18) | 15.5 (13–20.5) | 0.007 |
| Diastolic dysfunction | 14 (50%) | 14 (52%) | NS |
Abbreviations: CTP: Child Turcotte Pugh; MELD: model for end-stage liver disease; HVPG: hepatic venous pressure gradient; HRS: hepatorenal syndrome; NS: not significant.
Bold values are representes the P < 0.05.
Discussion
CCM is a distinct syndrome of cardiac and hemodynamic changes in patients with liver cirrhosis. Although it has been described since 1980s, there are limited studies about its prevalence, determinants, and clinical significance.
One hundred and four patients (median age 55 years, 77% males) were included in this study. Alcohol was the most common etiology of cirrhosis (79%), followed by viral (16%). The number of patients with nonalcoholic steatohepatitis-related cirrhosis was low because of the rigorous exclusion of patients with metabolic risk factors such as diabetes and systemic hypertension.
The prevalence of CCM in our study was 48%, and most of the patients (84%) had a mild DD. These results are almost similar to earlier reported studies9,18,19 While mild DD is common especially in elderly, more severe forms of DD (grade 2/3) are not frequently encountered. None of the patients who underwent DSE showed evidence of systolic cardiac dysfunction. The role of DSE in detecting CCM is questionable.15 The European Association for the Study of the Liver–endorsed guidelines at present do not recommend DSE for diagnosis of CCM.20
The presence of DD is the result of actual structural abnormalities such as ventricular hypertrophy, subendocardial edema, and altered collagen structure, which would be related to long-standing pathophysiological and anatomical alterations.21 Our study found that left ventricular diastolic diameter and left atrial volume were significantly higher in patients with CCM which has been reported earlier by Moller et al.22 Pozzi et al.23 found that in cirrhotic patients with tense ascites, the A wave velocity is markedly increased, the E/A ratio is markedly reduced, and the deceleration time is significantly prolonged. Finucci et al.10 reported increased stroke volume, left ventricular end-diastolic volume, left atrial volume, peak A velocity, deceleration time of the E wave, and decreased E/A ratio in 42 Child-Pugh class B and C cirrhotic patients.
Wong et al.24 found an increased interventricular septal and posterior wall thickness, as well as increased diameter of the left atrium in a mixed population of 39 preascitic and ascitic patients. All patients also had some degree of DD. They found isovolumic relaxation time to be prolonged in cirrhotic patients irrespective of the presence of ascites, with significantly reduced the E/A ratio only in patients with ascites. Accordingly, they concluded that patients with ascites had a thicker left ventricular wall and a lower E/A ratio, indicating greater impedance to venous return than preascitic cirrhotic patients. In our study, patients with ascites had higher prevalence of DD than those without ascites, but this difference was not significant. Some studies have found that patients with ascites have greater impairment of cardiac function;25 however, this may not be true for patients with minimal or mild ascites. The presence of ascites impairs cardiac contractility by a predominantly mechanical effect in which the elevated diaphragm and increased intrathoracic pressures hamper cardiac function. Most of our patients at the time of echocardiographic examination had mild to moderate ascites; we had excluded patients who had tense and refractory ascites. The American Society of Echocardiography and the European Association of Cardiovascular Imaging have recommended the use of more specific criteria for diagnosing CCM derived from pulsed-wave TDI.21 We investigated the presence of DD using TDI which is a better indicator of DD and less dependent on transient shear and strain.
In our study, we did not find systolic dysfunction in the patients who underwent DSE. This was possibly due to the small number of patients who underwent DSE and the relatively compensated patients in the evaluated cohort. There are many studies in literature which have reported systolic cardiac dysfunction in patients with cirrhosis.26, 27, 28 Wong et al.24 demonstrated an abnormal and inverse systolic pressure to end-systolic volume relationship, an index of myocardial contractility in both alcoholic and nonalcoholic cirrhotic patients, with or without ascites. The same researchers also demonstrated that both the absolute and the percentage increases in cardiac output in the cirrhotic patients were significantly decreased compared with the controls29 during the graded exercise test by using upright bicycle ergometry. The contractile abnormality appeared to be more severe in the ascitic cirrhotic patients, suggesting a correlation between the degree of cardiac dysfunction and the severity of liver disease.
In our study, age of patients was found to be the only predictor of DD in liver cirrhosis. It is a well-known fact that incidence of diastolic cardiac dysfunction increases with age and mild forms of DD are very common in the elderly. The definition of CCM rightly includes an age adjustment for measuring DD.8 We did not find any significant relationship between the severity of liver disease as measured by Child and MELD scores with DD. These findings are similar to recent other publications.30, 31, 32 There have been studies which have reported relation between severity of liver disease and CCM. Braillon et al.33 showed a direct linear correlation with the degree of liver dysfunction with the magnitude of hemodynamic change. Henriksen et al.34 showed a direct relationship with increased levels of natriuretic factors with increasing severity of liver cirrhosis.
We effectively excluded all confounding factors which may influence cardiac function including diabetes and hypertension which are strong individual risk factors for DD. In addition, we investigated the presence of DD using TDI which is a better indicator of DD and less dependent on transient shear and strain.
We did not find any relationship between HVPG and cardiac dysfunction. To the best of our knowledge, this is the first study to assess the relationship between HVPG and CCM. Genovesi et al.35 found a relationship between higher HVPG pressures and low serum calcium with prolongation of the QT interval especially in alcoholic subjects.
HRS is a functional renal dysfunction occurring in cirrhosis which occurs due to progressive systemic vasodilation and central (effective) hypovolemia. The hemodynamic changes proportionally deteriorate with worsening liver function and portal hypertension.14,16 There was no correlation between the presence of diastolic cardiac dysfunction and the development of HRS at 6-month follow-up. Similar observations have been reported as per previous studies.36,37 Majority of patients with CCM in our cohort only had a mild DD, and previous studies have shown that HRS develops in patients with systolic dysfunction, as these patients have low cardiac output.14,38 Patients who developed HRS in our study had higher CTP, MELD, and HVPG scores.
Among the limitations of our study, first was a relatively small cohort of patients which was due to rigorous exclusion criteria, which may lead to type II error. However, we had a homogenous cohort of patients in whom most of the confounders for cardiac dysfunction were excluded. Alcohol was the most common etiology of cirrhosis in our study; however, all these patients were abstinent for more than 6 months. Most of the patients had Child B cirrhosis because of exclusion of patients with tense ascites. Previous studies have used more complex echocardiographic parameters to define diastolic and systolic dysfunction. We analyzed this using basic, yet most efficient and important simple parameters like E/A ratio or Δ LVEF to define cardiac dysfunction which are easily reproducible and interpretable from a gastroenterologist's viewpoint. In addition, recently, an expert CCM consensus panel suggested revision of definitions of CCM.39 We could not include these revised criteria as the study was conducted much before these revisions.
To conclude, CCM was seen in about half of the patients with liver cirrhosis and was characterized predominantly by mild DD. Age of the patient was seen to be a stronger indicator for occurrence of diastolic cardiac dysfunction, rather than severity of liver dysfunction or higher portal pressures. There was no relation between diastolic cardiac dysfunction and the development of HRS in our study, although this has to be interpreted with caution as patients with higher grades of DD (grade 2/3) were relatively few. Prospective studies with larger sample size maybe needed to clarify this issue.
Credit authorship contribution statement
S.A. contributed to conceptualization, manuscript writing, data collection, and methodology. P.R. contributed to conceptualization, manuscript editing, supervision, and investigation. M.K. contributed to supervision and manuscript editing. R.M. contributed to conceptualization, manuscript review, and data collection.
Conflicts of interest
The authors have none to declare.
Funding
None.
References
- 1.Lee S.S. Cardiac abnormalities in liver cirrhosis. West J Med. 1989;151:530–551. [PMC free article] [PubMed] [Google Scholar]
- 2.Kowalski H.J., Abelmann W.H. The cardiac output at rest in Laennec's cirrhosis. J Clin Invest. 1953;32:1025–1033. doi: 10.1172/JCI102813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rayes N., Bechstein W.O., Keck H., Blumhardt G., Lohmann R., Neuhaus P. Cause of death after liver transplantation: an analysis of 41 cases in 382 patients. Zentralblatt Für Chir. 1995;120:435–438. [PubMed] [Google Scholar]
- 4.Lebrec D., Giuily N., Hadengue A., et al. Transjugular intrahepatic portosystemic shunts: comparison with paracentesis in patients with cirrhosis and refractory ascites: a randomized trial. French Group of Clinicians and a Group of Biologists. J Hepatol. 1996 Aug;25:135–144. doi: 10.1016/s0168-8278(96)80065-1. [DOI] [PubMed] [Google Scholar]
- 5.Franco D., Vons C., Traynor O., de Smadja C. Should portosystemic shunt be reconsidered in the treatment of intractable ascites in cirrhosis? Arch Surg Chic Ill 1960. 1988 Aug;123:987–991. doi: 10.1001/archsurg.1988.01400320073015. [DOI] [PubMed] [Google Scholar]
- 6.Iwakiri Y., Groszmann R.J. The hyperdynamic circulation of chronic liver diseases: from the patient to the molecule. Hepatol Baltim Md. 2006;43:S121–S131. doi: 10.1002/hep.20993. [DOI] [PubMed] [Google Scholar]
- 7.Møller S., Iversen J.S., Henriksen J.H., Bendtsen F. Reduced baroreflex sensitivity in alcoholic cirrhosis: relations to hemodynamics and humoral systems. Am J Physiol Heart Circ Physiol. 2007 Jun;292:H2966–H2972. doi: 10.1152/ajpheart.01227.2006. [DOI] [PubMed] [Google Scholar]
- 8.Nagueh S.F., Smiseth O.A., Appleton C.P., et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American society of echocardiography and the European association of cardiovascular imaging. J Am Soc Echocardiogr. 2016;29:277–314. doi: 10.1016/j.echo.2016.01.011. [DOI] [PubMed] [Google Scholar]
- 9.Falletta C., Filì D., Nugara C., et al. Diastolic dysfunction diagnosed by tissue Doppler imaging in cirrhotic patients: prevalence and its possible relationship with clinical outcome. Eur J Intern Med. 2015;26:830–834. doi: 10.1016/j.ejim.2015.10.009. [DOI] [PubMed] [Google Scholar]
- 10.Finucci G., Desideri A., Sacerdoti D., et al. Left ventricular diastolic function in liver cirrhosis. Scand J Gastroenterol. 1996;31:279–284. doi: 10.3109/00365529609004879. [DOI] [PubMed] [Google Scholar]
- 11.Gould L., Shariff M., Zahir M., Di Lieto M. Cardiac hemodynamics in alcoholic patients with chronic liver disease and a presystolic gallop. J Clin Invest. 1969;48:860–868. doi: 10.1172/JCI106044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Møller S., Henriksen J.H. Cardiovascular dysfunction in cirrhosis. Pathophysiological evidence of a cirrhotic cardiomyopathy. Scand J Gastroenterol. 2001;36:785–794. doi: 10.1080/003655201750313289. [DOI] [PubMed] [Google Scholar]
- 13.Henriksen J.H., Fuglsang S., Bendtsen F., Christensen E., Møller S. Dyssynchronous electrical and mechanical systole in patients with cirrhosis. J Hepatol. 2002;36 doi: 10.1016/s0168-8278(02)00010-7. 513–2. [DOI] [PubMed] [Google Scholar]
- 14.Krag A., Bendtsen F., Henriksen J.H., Møller S. Low cardiac output predicts development of hepatorenal syndrome and survival in patients with cirrhosis and ascites. Gut. 2010;59:105–110. doi: 10.1136/gut.2009.180570. [DOI] [PubMed] [Google Scholar]
- 15.Møller S., Henriksen J.H. Cirrhotic cardiomyopathy. J Hepatol. 2010 Jul;53:179–190. doi: 10.1016/j.jhep.2010.02.023. [DOI] [PubMed] [Google Scholar]
- 16.Kumar A., Khan N.M., Anikhindi S.A., et al. Correlation of transient elastography with hepatic venous pressure gradient in patients with cirrhotic portal hypertension: a study of 326 patients from India. World J Gastroenterol. 2017;23:687–696. doi: 10.3748/wjg.v23.i4.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Acevedo J.G., Cramp M.E. Hepatorenal syndrome: update on diagnosis and therapy. World J Hepatol. 2017;9:293–299. doi: 10.4254/wjh.v9.i6.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Salari A., Shafaghi A., Ofoghi M., Saeidinia A., Mansour-Ghanaei F. Diastolic dysfunction and severity of cirrhosis in nonalcoholic cirrhotic patients. Int J Hepatol. 2013;2013:892876. doi: 10.1155/2013/892876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bokarvadia R., Jain M., Kedarisetty C., Varghese J., Venkataraman J. Prevalence and clinical presentation of cirrhotic cardiomyopathy: a single centre experience from southern India. Indian J Gastroenterol. 2019;38:150–157. doi: 10.1007/s12664-019-00946-7. [DOI] [PubMed] [Google Scholar]
- 20.Ruiz-del-Arbol L., Monescillo A., Arocena C., et al. Circulatory function and hepatorenal syndrome in cirrhosis. Hepatol Baltim Md. 2005;42:439–447. doi: 10.1002/hep.20766. [DOI] [PubMed] [Google Scholar]
- 21.Krag A., Bendtsen F., Dahl E.K., Kjær A., Petersen C.L., Møller S. Cardiac function in patients with early cirrhosis during maximal beta-adrenergic drive: a dobutamine stress study. PLoS One. 2014;9 doi: 10.1371/journal.pone.0109179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Møller S., Henriksen J.H. Cirrhotic cardiomyopathy: a pathophysiological review of circulatory dysfunction in liver disease. Heart Br Card Soc. 2002;87:9–15. doi: 10.1136/heart.87.1.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pozzi M., Carugo S., Boari G., et al. Evidence of functional and structural cardiac abnormalities in cirrhotic patients with and without ascites. Hepatol Baltim Md. 1997 Nov;26:1131–1137. doi: 10.1002/hep.510260507. [DOI] [PubMed] [Google Scholar]
- 24.Wong F., Liu P., Lilly L., Bomzon A., Blendis L. Role of cardiac structural and functional abnormalities in the pathogenesis of hyperdynamic circulation and renal sodium retention in cirrhosis. Clin Sci Lond Engl 1979. 1999 Sep;97:259–267. [PubMed] [Google Scholar]
- 25.Dadhich S., Goswami A., Jain V.K., Gahlot A., Kulamarva G., Bhargava N. Cardiac dysfunction in cirrhotic portal hypertension with or without ascites. Ann Gastroenterol. 2014;27:244–249. [PMC free article] [PubMed] [Google Scholar]
- 26.Bernardi M., Calandra S., Colantoni A., et al. Q-T interval prolongation in cirrhosis: prevalence, relationship with severity, and etiology of the disease and possible pathogenetic factors. Hepatol Baltim Md. 1998 Jan;27:28–34. doi: 10.1002/hep.510270106. [DOI] [PubMed] [Google Scholar]
- 27.Grose R.D., Nolan J., Dillon J.F., et al. Exercise-induced left ventricular dysfunction in alcoholic and non-alcoholic cirrhosis. J Hepatol. 1995 Mar;22:326–332. doi: 10.1016/0168-8278(95)80286-x. [DOI] [PubMed] [Google Scholar]
- 28.Laffi G., Barletta G., La Villa G., et al. Altered cardiovascular responsiveness to active tilting in nonalcoholic cirrhosis. Gastroenterology. 1997 Sep;113:891–898. doi: 10.1016/s0016-5085(97)70184-7. [DOI] [PubMed] [Google Scholar]
- 29.Wong F., Girgrah N., Graba J., Allidina Y., Liu P., Blendis L. The cardiac response to exercise in cirrhosis. Gut. 2001 Aug;49:268–275. doi: 10.1136/gut.49.2.268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Merli M., Calicchia A., Ruffa A., et al. Cardiac dysfunction in cirrhosis is not associated with the severity of liver disease. Eur J Intern Med. 2013 Mar;24:172–176. doi: 10.1016/j.ejim.2012.08.007. [DOI] [PubMed] [Google Scholar]
- 31.Alexander J., Mishra P., Desai N., Ambadekar S., Gala B., Sawant P. Cirrhotic cardiomyopathy: Indian scenario. J Gastroenterol Hepatol. 2007 Mar;22:395–399. doi: 10.1111/j.1440-1746.2006.04507.x. [DOI] [PubMed] [Google Scholar]
- 32.Wiese S., Hove J.D., Bendtsen F., Møller S. Cirrhotic cardiomyopathy: pathogenesis and clinical relevance. Nat Rev Gastroenterol Hepatol. 2014 Mar;11:177–186. doi: 10.1038/nrgastro.2013.210. [DOI] [PubMed] [Google Scholar]
- 33.Braillon A., Cales P., Valla D., Gaudy D., Geoffroy P., Lebrec D. Influence of the degree of liver failure on systemic and splanchnic haemodynamics and on response to propranolol in patients with cirrhosis. Gut. 1986 Oct;27:1204–1209. doi: 10.1136/gut.27.10.1204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Henriksen J.H., Gøtze J.P., Fuglsang S., Christensen E., Bendtsen F., Møller S. Increased circulating pro-brain natriuretic peptide (proBNP) and brain natriuretic peptide (BNP) in patients with cirrhosis: relation to cardiovascular dysfunction and severity of disease. Gut. 2003 Oct;52:1511–1517. doi: 10.1136/gut.52.10.1511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Genovesi S., Prata Pizzala D.M., Pozzi M., et al. QT interval prolongation and decreased heart rate variability in cirrhotic patients: relevance of hepatic venous pressure gradient and serum calcium. Clin Sci (Lond). 2009;116:851–859. doi: 10.1042/CS20080325. Published 2009 May 14. [DOI] [PubMed] [Google Scholar]
- 36.Nazar A., Guevara M., Sitges M., et al. LEFT ventricular function assessed by echocardiography in cirrhosis: relationship to systemic hemodynamics and renal dysfunction. J Hepatol. 2013;58:51–57. doi: 10.1016/j.jhep.2012.08.027. [DOI] [PubMed] [Google Scholar]
- 37.Somani P.O., Contractor Q., Chaurasia A.S., Rathi P.M. Diastolic dysfunction characterizes cirrhotic cardiomyopathy. Indian Heart J. 2014;66:649–655. doi: 10.1016/j.ihj.2014.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ruiz-del-Arbol L., Urman J., Fernández J., et al. Systemic, renal, and hepatic hemodynamic derangement in cirrhotic patients with spontaneous bacterial peritonitis. Hepatol Baltim Md. 2003;38:1210–1218. doi: 10.1053/jhep.2003.50447. [DOI] [PubMed] [Google Scholar]
- 39.Izzy M., VanWagner L.B., Lin G., et al. Cirrhotic Cardiomyopathy Consortium Redefining cirrhotic cardiomyopathy for the modern era. Hepatology. 2020 Jan;71:334–345. doi: 10.1002/hep.30875. Epub 2019 Oct 11. [DOI] [PMC free article] [PubMed] [Google Scholar]