Abstract
Geothermal soils near Amphitheater Springs in Yellowstone National Park were characterized by high temperatures (up to 70°C), high heavy metal content, low pH values (down to pH 2.7), sparse vegetation, and limited organic carbon. From these soils we cultured 16 fungal species. Two of these species were thermophilic, and six were thermotolerant. We cultured only three of these species from nearby cool (0 to 22°C) soils. Transect studies revealed that higher numbers of CFUs occurred in and below the root zone of the perennial plant Dichanthelium lanuginosum (hot springs panic grass). The dynamics of fungal CFUs in geothermal soil and nearby nongeothermal soil were investigated for 12 months by examining soil cores and in situ mesocosms. For all of the fungal species studied, the temperature of the soil from which the organisms were cultured corresponded with their optimum axenic growth temperature.
Specific genetic and/or physiological adaptations allow microorganisms to exist in many environments that experience extremes of temperature, pH, chemical content, and/or pressure (1, 10, 12, 25, 27, 33). Thermophilic organisms may belong to any of the three domains of life, but most thermophilic species that have been described are members of the Archaea or Eubacteria (5, 20). Thermophilic members of the Eukarya tolerate less heat than thermophilic members of the other domains, and it is thought that the former organisms do not grow at temperatures greater than 60°C (1, 10, 25, 27, 28). For example, fungi are considered thermophilic if they grow at 50°C or higher temperatures and do not grow at 20°C or lower temperatures (10).
Fewer than 50 species of thermophilic fungi have been described (reviewed in references 10 and 25). Many of these organisms were first isolated from guayule undergoing retting (10), a process similar to composting, during rubber production. However, in recent years thermophilic fungi have been isolated from manure composts, industrial coal mine soils, beach sands, nuclear reactor effluents, Dead Sea valley soils, and desert soils of Saudi Arabia. Although two fungi from natural geothermal soils have been mentioned previously (6, 29–31), there have been no in-depth characterizations of fungal communities in geothermal soils. In fact, in a recent publication, Yellowstone National Park soil scientists state that thermal soils are typically sterile (34).
In general, there is an inverse relationship between biological diversity and the amount of adaptation required to survive in a specific habitat (33). Thus, we expected that geothermal soils in Yellowstone National Park would have less fungal diversity and hence be more amenable to our overall goal of characterizing and monitoring soil fungal communities and their responses to environmental change. The objectives of this study were (i) to determine the diversity of culturable thermotolerant and/or thermophilic fungal species in Yellowstone National Park geothermal soils, (ii) to determine the optimal in vitro growth conditions for these species, and (iii) to characterize the temporal and spatial distribution of the culturable thermophilic and thermotolerant fungal species.
MATERIALS AND METHODS
Site characterization.
Fungi were cultured from 27 different soil samples collected at two geothermal sites (designated sites 1a and 1b) near Amphitheater Springs and at three nongeothermal sites near Amphitheater Springs or Mammoth in Yellowstone National Park. We selected Amphitheater Springs as a field site because its thermal, acidic soil and sparse vegetation suggested that the biodiversity in this ecosystem would be relatively less complex and, hence, more easily characterized. Sites 1a and 1b were separated by 800 m of nongeothermal soil and were 100 and 25 m in diameter, respectively. These sites exhibited heterogeneity with regard to temperature, pH, metal content, and the occurrence of the grass Dichanthelium lanuginosum (28; data not shown).
Soil transects.
To determine if fungal CFUs were associated with the presence of D. lanuginosum, we collected samples along two 3-m linear transects with isolated D. lanuginosum plants located several meters apart. Soil temperatures were measured every 30 cm at depths of 5, 10, 15, and 20 cm before soil cores were collected.
Soil cores.
A 2.5-cm-diameter soil corer was sterilized with 95% ethanol, and 20-cm soil cores were collected. Each soil core was divided into 5-cm portions, which were aseptically transferred to plastic bags, mixed thoroughly, stored at 4°C, and processed within hours. Soil was prepared by suspending 1.2 ml (∼1 g) of soil in 20 ml of sterile water, vortexing the suspension for 30 s, and allowing the debris to settle. After aliquots for culturing were removed, the pH values of the suspensions were determined (21).
Fungal isolation, growth, and identification.
To simulate the low-nutrient-content conditions in geothermal soils, fungi were isolated by plating soil suspensions on 0.1× potato dextrose agar (PDA) (pH 5) (Difco Laboratories, Detroit, Mich.) and minimal medium containing (per liter) 0.5 g of l-glutamic acid HCl, 0.5 g of citric acid · H2O, 0.5 g of malic acid (disodium salt), 0.5 g of glucose, 0.1 g of yeast extract, and 15 g of agar. The pH of the minimal medium was adjusted to 4.0 with HCl. After autoclaving, 10 ml of a salts solution (containing 50 g of K2HPO4 per liter, 10 g of NaCl per liter, 1 g of FeCl3 · 6H2O per liter, 10 g of CaCl2 · 2H2O per liter, and 25 g of MgCl2 per liter) per liter was added to the minimal medium. All media contained 50 mg of streptomycin per liter and 50 mg of ampicillin per liter, which were added to cooled media after autoclaving to inhibit bacterial growth. Soil suspensions were diluted to obtain 20 to 30 CFUs per plate. Ten plates (subsamples) prepared from each soil sample were incubated for 24 to 96 h at 40°C, and each morphologically unique fungal colony was subcultured on 0.1× PDA, corn meal agar (pH 5.0) (Difco), minimal medium, and 0.1× Sabouraud dextrose (Difco) agar (pH 4.0). All of the fungal species observed grew on 0.1× PDA, which was used for subsequent experiments. Fungal species were identified by microscopic analysis by using taxonomic guides and standard procedures (2, 4, 8–11, 13–18, 23–25, 32). Unidentified Penicillium species were distinguished on the basis of morphological variations and were designated Penicillium species 1 through Penicillium species 5. The absence of species designations for some of the isolates (see Table 2) does not mean that the organisms are new species but rather reflects difficulties in taxonomic identification. Isolates may be obtained from us with the permission of the Yellowstone National Park Center for Resources.
TABLE 2.
Temperature and pH ranges of cores and mesocosms in Yellowstone National Parka
| Mesocosm or core(s) | Plant(s) associated with mesocosm or core(s) | Initial pH range | Initial temp (°C) atb:
|
Annual temp range (°C) atc:
|
||
|---|---|---|---|---|---|---|
| 5 cm | 15 cm | 5 cm | 15 cm | |||
| Mesocosm 31 | D. lanuginosum | 4.5–7.5 | 31 | 46 | 5–44 | 19–56 |
| Mesocosm 32 | D. lanuginosum | 4.4–6.3 | 35 | 50 | 2–44 | 12–58 |
| Mesocosm 33 | D. lanuginosum | 4.7–5.4 | 24 | 36 | 1–38 | 11–45 |
| Cores 36A-D | D. lanuginosum | 4.6–4.9 | 34 | 41 | 9–34 | 14–41 |
| Cores 37A-Cd | Mixed grasses | 6.1–6.3 | 22 | 19 | <0–22e | <0–14e |
| Cores 4A-D | D. lanuginosum | 3.9–4.5 | 42 | 56 | 10–42 | 18–56 |
| Cores 6A-D | D. lanuginosum | 4.2–5.0 | 28 | 38 | 11–30 | 18–38 |
| Cores 8A-D | D. lanuginosum | 4.3–4.9 | 27 | 33 | 8–30 | 15–33 |
| Cores 11A-D | Decaying log | 2.7–4.0 | 98 | 107 | 55–98 | 91–107 |
| Cores 13A-D | D. lanuginosum | 4.2–4.7 | 43 | 47 | 8–43 | 14–47 |
| Cores 25A-D | D. lanuginosum | 3.9–4.2 | 20 | 23 | 2–24 | 10–24 |
| Cores 27A-Dd | Lodgepole pine | NDf | 9 | 7 | ND | ND |
| Core 28Dd | Mixed grasses | ND | 12 | 10 | ND | ND |
| Core 29Ad | Sagebrush | ND | 12 | 11 | ND | ND |
| Core 34g | D. lanuginosum | 4.2–4.4 | 3 | 8c | 3–29 | 8–32 |
| Core 35 | D. lanuginosum | 5.1–5.2 | 32 | 47 | 10–32 | 18–42 |
The ambient air temperature at Amphitheater Springs ranged from −36°C in January 1997 to 32°C in August 1997.
Temperatures were determined in September 1996 by using A cores.
Temperatures were determined by using cores A through D.
Nongeothermal core(s).
When the ground was frozen, the temperature was not recorded (<0°C).
ND, not determined.
The first core was collected in February 1997.
Temperature and pH optima of fungi.
The temperature optima of fungal isolates were determined by measuring colony diameters after incubation of 0.1× PDA plates for 24 to 96 h at 20, 25, 30, 35, 40, 50, and 55°C. The species that grew at 50°C but not at 20°C were considered thermophilic (10). The mesophilic species were the species that grew at temperatures between 20 and 40°C and could not tolerate 55°C (see below). The pH optima for fungal growth were determined by measuring colony diameters after 0.1× PDA plates adjusted to pH 3 to 7 were incubated for 24 to 96 h at 35 or 45°C. Growth rates were determined with the following equation: g = d2 − d1/24, where g is the growth rate and d1 and d2 are the colony diameters measured over a 24-h period at 24 and 48 h after inoculation.
Thermotolerance of fungi.
Mycelial plugs without spores from cultures grown at 35°C were transferred to 0.1× PDA and incubated at 55°C for 7 days; then they were incubated at 35°C again. Colony diameters were measured prior to the temperature shift and at 3, 4, and 5 days after the shift. The organisms that grew after the shift back to 35°C were considered thermotolerant organisms, and the organisms that did not grow after the shift back to 35°C were considered mesophilic organisms.
In situ mesocosm construction and establishment.
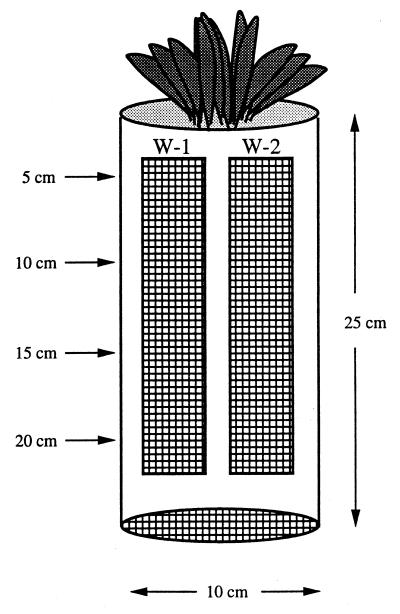
Mesocosms were constructed from polyvinyl chloride (PVC) pipe as follows. A 25-cm length of 10-cm-diameter PVC pipe was cut in half along its length, and two windows (4.5 by 20 cm) were cut out of each half of the pipe, as shown in Fig. 1. The two halves of pipe were placed together so that a tube was reformed, and the inner circumference was lined with 1.5-mm-mesh nylon screen that was fixed with nylon zip straps to the walls between the windows. Two T type thermocouple probes were attached with nylon zip straps to the inside walls on each half of the mesocosm at depths of 5 and 15 cm, and these probes were monitored with a Campbell Scientific model CR10 data logger. The top of the tube was secured with a nylon zip strap, and the bottom of the tube was covered with nylon screen and secured with nylon zip straps.
FIG. 1.
In situ mesocosm design. The plant at the surface is D. lanuginosum. The cross-hatching represents nylon mesh over windows from which samples were obtained at depths of 5, 10, 15, and 20 cm. Thermocouple probes were placed at depths of 5 and 15 cm and used to record temperatures every 2 h.
Three mesocosms were constructed and placed in the geothermal soil at site 1b approximately 1 m apart. A large soil core (10 by 25 cm) containing one D. lanuginosum plant was removed with a shovel and placed into each PVC tube. The mesocosms were placed into the original soil core sites and packed into position. Every 6 months the data logger storage modules were downloaded and replaced.
Community structure and dynamics.
The numbers of fungal CFUs were determined four times between September 1996 and September 1997. We sampled a mesocosm by lifting it out of the ground and making a small incision in each screened window in order to extract soil. Two windows, representing replicates, at each depth (5, 10, 15, and 20 cm) were sampled, and all subsequent samples were removed from the same incisions in the windows. In addition, the surface soil near the plant crown was sampled. The mesocosms were returned to their locations and packed into position. We also determined the numbers of fungal CFUs in soil cores collected near the mesocosms (cores 36A through C) and in adjacent cool-soil cores (cores 37A through C).
Soil sample suspensions were prepared as described above and plated onto 0.1× PDA, which yielded 20 to 30 CFUs per plate; 10 plates (subsamples) per sample were prepared. The minimum number of CFUs detected by this plating procedure was 40 CFUs/g of soil. The plates were incubated at 40°C for 48 to 96 h. The numbers of CFUs of each species obtained from two windows (replicates) at each depth were determined.
RESULTS
Relationship between fungal CFUs and D. lanuginosum.
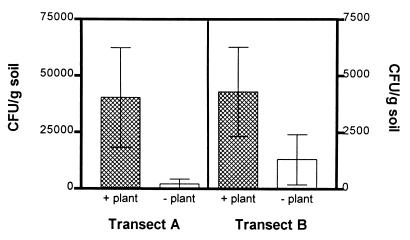
We monitored the number of fungal CFUs in soil cores along two linear transects to determine if the number of fungal propagules increased in the vicinity of the only plant present, D. lanuginosum. D. lanuginosum occurs in North America, the West Indies, and northern South America, but at elevations such as those that occur in Yellowstone National Park (>6,000 ft), it is restricted to geothermally heated ground (26). The transects which we used were at site 1a, where single plants were usually several meters apart. The mean numbers of CFUs were 20 and 3.2 times higher in soil below D. lanuginosum than in soil devoid of surface vegetation along transect A (P = 0.008, as determined by Student’s t test) and transect B (P = 0.02, as determined by Student’s t test), respectively (Fig. 2 [only 5-cm data are shown]). There also was an inverse relationship between the number of fungal CFUs and soil depth; fewer CFUs were obtained from lower depths in all transect cores. At depths of 15 to 20 cm, where D. lanuginosum roots were no longer visible, the numbers of fungal CFUs did not differ from the numbers of CFUs in cores that lacked D. lanuginosum (data not shown).
FIG. 2.
Total fungal CFUs obtained at 5 cm along two thermal soil transects with sparse vegetation. Core samples were collected every 30 cm in May 1996 (transect A) and September 1996 (transect B) and either had D. lanuginosum on the surface (+ plant) or were devoid of vegetation (− plant). The error bars indicate standard deviations (n ≥ 3; P = 0.008 for transect A and P = 0.02 for transect B, as determined by Student’s t tests).
Culturable fungi in geothermal soils.
We cultured 16 different species of fungi from geothermal soils plated at 40°C (Table 1), although we observed other species when soil samples were plated at room temperature (data not shown). All of the species that grew at 40°C were members of the Deuteromycetes, Ascomycetes, or Zygomycetes; no member of the Basidiomycetes was identified. The optimal temperature for laboratory growth for most of these species was 35°C, and the optimal pH for most species was pH 5 when the organisms were grown on 0.1× PDA at the optimal temperature (Table 1). Two organisms, Dactylaria constrictum var. gallopava and Acremonium alabamense, grew at 55°C but not at 20°C and were classified as thermophiles (10). Six other species (Absidia cylindrospora, Aspergillus fumigatus, Aspergillus niger, Penicillium species 1, Penicillium species 2, and Penicillium species 3) had thermotolerant profiles because they did not grow at 55°C but grew when they were incubated at 35°C after exposure to 55°C for 1 week. The remaining species were mesophilic since they did not grow at 35°C after exposure to 55°C for 1 week.
TABLE 1.
Optimal pH values and temperatures for fungi
| Organism | Optimal pHa | Optimal in vitro growth temp (°C)b | Growth rate (cm/day)b | Temp classc | In vitro temp range (°C) | % of CFUs atd:
|
Total CFUse | ||
|---|---|---|---|---|---|---|---|---|---|
| 0–20°C | 20–40°C | 40–70°C | |||||||
| Absidia cylindrospora | 5–6 | 35 | 0.83 | TT | 20–45 | ||||
| Acremonium alabamense | 5 | 45 | 0.93 | TP | 30–55 | 13 | 39 | 48 | 1.9 × 105 |
| Acremonium ochraceum | 6 | 25–35 | 0.14–0.29 | M | 20–45 | ||||
| Aspergillus fumigatus | 4 | 35 | 0.64 | TT | 20–45 | ||||
| Aspergillus niger | 5 | 35 | 0.48 | TT | 20–45 | ||||
| Chaetomium erraticum | 6 | 35 | 0.5 | M | 20–45 | ||||
| Cunninghamella elegans | 5 | 35 | 0.38 | M | 20–45 | 19 | 71 | 11 | 2.2 × 105 |
| Penicillium piceum | 5 | 35 | 0.33 | M | 20–45 | ||||
| Penicillium species 1 | 5 | 35 | 0.35 | TT | 20–45 | ||||
| Penicillium species 2 | 5 | 35 | TT | 20–45 | |||||
| Penicillium species 3 | 4 | 35 | 0.36 | TT | 20–45 | ||||
| Penicillium species 4 | 5 | 25 | 0.24 | M | 20–45 | 89 | 11 | 1 | 9.8 × 104 |
| Penicillium species 5 | 6 | 35 | 0.24 | M | 20–45 | ||||
| Pseudeurotium sp. | 6 | 35 | M | 20–45 | |||||
| Dactylaria constricta var. gallopava | 6 | 45 | 0.26 | TP | 25–55 | 20 | 56 | 25 | 4.2 × 105 |
| Torula sp. | 5 | 35 | 0.24 | M | 20–45 | ||||
The highest growth rate on 0.1× PDA.
Determined on 0.1× PDA (pH 5).
TP, thermophilic (organisms grew at temperatures greater than 50°C); TT, thermotolerant (organisms survived incubation at 55°C for 7 days); M, mesophilic (organisms did not survive incubation at 55°C).
Percentages of fungal CFUs obtained from all three mesocosms and cores 36A through C at different temperatures.
Total number of CFUs obtained from all three mesocosms and cores 36A through C.
When nongeothermal soil samples (e.g., cores 37A through C) were plated at 40°C, only A. fumigatus and two Penicillium species were recovered. None of the other fungi listed in Table 1 were isolated from four adjacent nongeothermal soils during this study (cores 37A through C and data not shown).
In situ soil mesocosms and characterization of geothermal soils.
The soils at sites 1a and 1b are acid-altered rhyolite and siliceous sinter geothermal soils (34), and they varied significantly with respect to temperature, pH, organic carbon content, and metal content both within and between cores (Table 2 and data not shown). The temperature increased with depth in all of the geothermal soils (Table 2, mesocosms 31 through 33 and cores 36A through C and 74 other cores, including the transect cores, from geothermal soils [data not shown]). In contrast, the temperature in adjacent nongeothermal soil decreased with depth (Table 2, cores 37A through C and three other nongeothermal soil cores [data not shown]). There was no apparent relationship between geothermal soil depth and pH. The temperatures of geothermal soil cores near Amphitheater Springs varied substantially (Table 2, cores 4 through 35). In addition, cores could not be sampled more than once and could not be used to assess changes in propagule frequency with time. To avoid these complications, we placed three in situ mesocosms (Fig. 1) at geothermal site 1b and collected samples from them during September 1996 and February, May, and September 1997. We also took core samples on the same collection dates between the mesocosms (cores 36A through C) and from nearby nongeothermal soil (cores 37A through C) for comparison. The geothermal soils never had snow cover and contained more water in the winter than in the summer (data not shown).
As observed with all of the geothermal soils examined at sites 1a and 1b, the temperatures in the mesocosms increased with depth (Table 2). In addition, when temperatures were monitored every hour over a 12-h period, the data revealed that there were daily temperature cycles at depths of 5 and 15 cm in all three mesocosms and that the temperatures ranged from 0.5 to 60°C. A nongeothermal soil, monitored over a 24-h period in September 1996, did not exhibit this diurnal temperature fluctuation; that is, the temperature at depths of 10 to 20 cm was stable at ∼8°C (data not shown). The lowest and highest temperatures in the mesocosms occurred in the winter and the summer, respectively. During the year, the temperature in a mesocosm varied up to 46°C at each depth. There was no apparent relationship between the season and the soil pH; core samples were almost always acidic, and the pH values were within the ranges listed in Table 2.
Numbers of fungal CFUs in mesocosms and nearby soil cores.
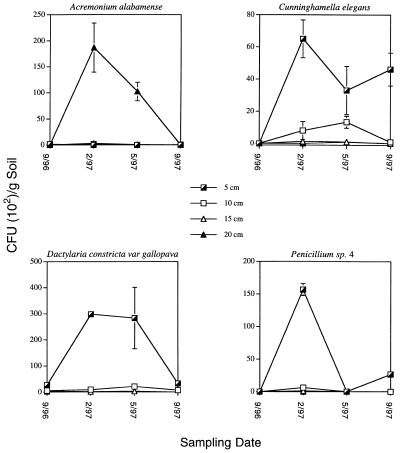
In general, the numbers of fungal CFUs in the three mesocosms and adjacent core samples (cores 36A through C) were similar, and we isolated the species listed in Table 1 from all mesocosms and adjacent cores. The numbers of CFUs of all species changed with the season and increased in the winter and spring, when soil moisture levels were greatest. The numbers of culturable CFUs of four representative organisms, A. alabamense, D. constricta var. gallopava, C. elegans, and Penicillium species 4, from one representative mesocosm (mesocosm 32) are shown in Fig. 3. Adjacent cores 36A through C and mesocosms exhibited similar trends, and the standard deviations were similar.
FIG. 3.
Numbers of CFUs of representative species obtained from one mesocosm (mesocosm 32) at depths of 5 to 20 cm from September 1996 to September 1997. Each point represents the average of two replicate sample values, and the error bars indicate standard deviations (not visible if the deviation was small). The temperature ranges in this mesocosm at depths of 5 to 20 cm at different sampling times were as follows: 31 to 63°C in September 1996, 7 to 24°C in February 1997, 21 to 50°C in May 1997, and 29 to 67°C in September 1997.
In the winter, there were few CFUs (<40 CFUs/g of soil) of the thermophile A. alabamense in the cooler soil samples near the surface, or this organism was undetectable (Fig. 3 and Table 1). Only 13% of the A. alabamense CFUs were cultured from soils with temperatures below 20°C, whereas 48% were cultured from soils with temperatures above 40°C. In contrast, the other thermophile, D. constricta, was cultured from soils with a broader temperature range; 20% of the D. constricta CFUs were cultured from <20°C soil, and 56% were cultured from 20 to 40°C soil (Fig. 3 and Table 1). The mesophile Penicillium species 4 was present only in cooler soils (89% of the CFUs were obtained from soils with temperatures less than 20°C) and was not detectable in the warmest soil samples (Fig. 3 and Table 1). However, another representative mesophile, Cunninghamella elegans, was cultured from soils with a broader range of temperatures and was present at all depths in the winter and spring, but it was isolated only from the cooler September soil samples (Fig. 3 and Table 1). The frequencies of all 16 species cultured from thermal soils were consistent with the laboratory growth temperature ranges and growth optima of the organisms (Table 1 and data not shown).
DISCUSSION
Physical analyses of soil cores revealed that significant variations in soil temperature and pH occurred at geothermal soil sites in Yellowstone National Park. Soil near Amphitheater Springs is acidic because of sulfuric acid produced by the oxidation of sulfides, such as hydrogen sulfide (H2S) and pyrite (FeS2), that are present at high concentrations in most geothermal areas of the park (3, 7). The relationships between temperature and soil depth also were due to geothermal activity.
Only two fungi isolated from geothermal soil grew at 55°C, but six other species were thermotolerant. All of the isolates tolerated a wide range of pH values and grew at pH 3 to 6. The highest numbers of fungal CFUs occurred at soil depths of 5 cm, and more fungal CFUs were isolated from soil cores containing roots of D. lanuginosum. These findings suggested that the presence of plants is a primary factor for fungal CFU production, at least at this geothermal site. Very little is known about the nature of interactions between root-associated fungi and plants adapted to geothermal environments. Perhaps D. lanuginosum ameliorates the effects of this extreme environment. This plant, like the plants of temperate soils, may also provide fungal nutrients via root exudates, or alternatively, the fungi may utilize the plants as a nutrient source by establishing symbiotic or saprophytic associations (12).
The frequencies of the species which we identified corresponded with soil temperatures and were consistent with the optimal laboratory growth temperatures. More CFUs of all species were cultured from soils with higher moisture contents in the winter and spring. However, moisture alone was not sufficient for obtaining higher numbers of CFUs, as the numbers of CFUs of all species increased only in moist soils whose temperatures were within the optimal ranges. We cultured two true thermophilic fungi from the geothermal soil samples, and these fungi were never isolated from cooler soils. Although both A. alabamense and D. constricta were thermophilic, a greater percentage of D. constricta CFUs than of A. alabamense CFUs was cultured from soils with moderate temperatures (20 to 40°C). This finding is consistent with the axenic growth temperature ranges of these organisms, as A. alabamense did not grow axenically at 25°C, whereas D. constricta did (Table 1). The mesophilic organism Penicillium species 4 was most prevalent at temperatures below 20°C, whereas another mesophilic species, C. elegans, was frequently cultured from soils with temperatures between 20 and 40°C. Again, the laboratory growth temperature optima of the organisms (Table 1) were correlated with the frequencies at different soil temperatures. The numbers of CFUs of other mesophilic and thermotolerant species were similarly consistent with the axenic growth temperature ranges of the organisms.
Although none of the fungi listed in Table 1 is a new species, fungi in Yellowstone National Park geothermal soils may have diverged from their nongeothermal soil counterparts. The adaptive strategies of these fungi may include the development of mycelia, spores, or sclerotia that tolerate prolonged exposure to elevated temperatures and/or a wide range of pH values. For example, one unidentified isolate in this study was obtained from a decaying log (core 11) that was as hot as 107°C. However, this fungus probably survived but did not grow at high temperatures, since the upper limit for eukaryotic growth is thought to be ∼60°C. Although slides buried at our site revealed that there was evidence of hyphal growth in geothermal soils, we did not identify which species grew and which species merely survived at higher soil temperatures. We cultured all of the species listed in Table 1 from soils whose temperatures were outside the growth ranges of the organisms, but additional experiments are needed to discern the temperature tolerance and adaptive mechanism(s) of these fungi.
Many other fungal species could be present in the geothermal soils examined, but they were not detected by traditional culturing methods (19). For example, species present in very low numbers were not detected because of the soil dilutions necessary to obtain countable CFUs. We also observed more total CFUs when suspensions were diluted so that they yielded fewer CFUs per plate. This finding indicates that some species may inhibit the growth of other species in more crowded culture conditions. Additional studies performed with molecular-biology-based techniques (22), such as PCR followed by denaturing gradient gel electrophoresis, should reveal whether culturable fungal species are, indeed, the predominant fungal species in these soils and whether these soils harbor uncultured fungi which can provide a more accurate assessment of the biodiversity of this unique ecosystem.
ACKNOWLEDGMENTS
We thank R. Lindstrom and the National Park Service for providing housing, information, and library facilities during this research and Richard Stout for introducing us to geothermal soils and thermotolerant plants in Yellowstone National Park. This project was made possible by the permission, cooperation, and assistance of the National Park Service and was carried out under the guidelines for research in Yellowstone National Park.
This research was supported in part by a joint NSF-DOE-USDA grant (R.J.R. was one of the principal investigators), by the U.S. Geological Survey (R.J.R. was the principal investigator), and by a U.S. Army Research Office grant (J.M.H. was the principal investigator).
REFERENCES
- 1.Aguilar A. Extremophile research in the European Union: from fundamental aspects to industrial expectations. FEMS Microbiol Rev. 1996;18:89–92. [Google Scholar]
- 2.Ames L M. A monograph of the Chaetomiaceae. US Army Res Dev Ser. 1961;2:126–251. [Google Scholar]
- 3.Barnes H L, Rose A W. Origins of hydrothermal ores. Science. 1998;279:2064–2065. [Google Scholar]
- 4.Barnett H L, Hunter B B. Illustrated genera of imperfect fungi. St. Paul, Minn: American Phytopathology Society; 1998. [Google Scholar]
- 5.Barns S M, Delwiche C F, Palmer J D, Pace N R. Perspectives on archaeal diversity, thermophily, and monophyly from environmental rRNA sequences. Proc Natl Acad Sci USA. 1996;93:9188–9193. doi: 10.1073/pnas.93.17.9188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Belly R T, Tansey M R, Brock T D. Algal excretion of 14C-labeled compounds and microbial interactions in Cyanidium caldarium mats. J Phycol. 1973;9:123–127. [Google Scholar]
- 7.Brock T D, Mosser J L. Rate of sulfuric-acid production in Yellowstone National Park. Geol Soc Am Bull. 1975;86:194–198. [Google Scholar]
- 8.Cooke J C. Morphology of Chaetomium erraticum. Am J Bot. 1969;3:335–340. [Google Scholar]
- 9.Cooke J C. Variation in isolates of Chaetomium trilaterale. Mycologia. 1973;65:1212–1220. [PubMed] [Google Scholar]
- 10.Cooney D G, Emerson R. Thermophilic fungi: an account of their biology, activities, and classification. W. H. San Francisco, Calif: Freeman and Co.; 1964. [Google Scholar]
- 11.De Hoog G S. The genera Blastobotrys, Pseudeurotium, Calcarisporium. Stud Mycol. 1974;7:120–204. [Google Scholar]
- 12.Dix N J, Webster J. Fungal ecology. London, United Kingdom: Chapman & Hall; 1995. [Google Scholar]
- 13.Domson K H, Gams W, Anderson T H. Compendium of soil fungi. New York, N.Y: Academic Press; 1980. [Google Scholar]
- 14.Ellis M B. Dematiaceous hyphomycetes. Surrey, England: Commonwealth Mycological Institute; 1971. [Google Scholar]
- 15.Ellis M B. More dematiaceous hyphomycetes. Surrey, England: Commonwealth Mycological Institute; 1976. [Google Scholar]
- 16.Gilman J C. A manual of soil fungi. Ames: The Iowa State College Press; 1972. [Google Scholar]
- 17.Glenn A E, Bacon C W, Price R, Hanlin R T. Molecular phylogeny of Acremonium and its taxonomic implications. Mycologia. 1996;88:369–383. [Google Scholar]
- 18.Goh T K, Hyde K D. A revision of Dactylaria, with description of D. tunicata sp. nov. from submerged wood in Australia. Mycol Res. 1997;101:1265–1272. [Google Scholar]
- 19.Hawksworth D L, Rossman A Y. Where are all the undescribed fungi? Phytopathology. 1997;87:888–891. doi: 10.1094/PHYTO.1997.87.9.888. [DOI] [PubMed] [Google Scholar]
- 20.Hugenholtz P, Pitulle C, Hershberger K L, Pace N R. Novel division level bacterial diversity in a Yellowstone Hot Spring. J Bacteriol. 1998;180:366–376. doi: 10.1128/jb.180.2.366-376.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Klute A, Page A L. Methods of soil analysis. Madison, Wis: American Society of Agronomy, Inc.; 1982. [Google Scholar]
- 22.Kowalchuk G. New perspectives towards analysing fungal communities in terrestrial environments. Curr Opin Biotechnol. 1999;10:247–251. doi: 10.1016/S0958-1669(99)80043-3. [DOI] [PubMed] [Google Scholar]
- 23.Miliko A, Belyakova L. Genus Cunninghamella mutruchot and taxonomy of the Cunninghamellaceae. Microbiology (Engl Translation Microbiologiya) 1967;4:684–690. [Google Scholar]
- 24.Morgan-Jones G. Notes to hyphomycetes. A new thermophilic species of Acremonium. Can J Bot. 1974;52:429–431. [Google Scholar]
- 25.Mouchacca J. Thermophilic fungi: biodiversity and taxonomic status. Cryptogam Mycol. 1997;18:19–69. [Google Scholar]
- 26.Spellenberg R. Synthetic hybridization and taxonomy of western North American Dichanthelium, group lanuginosa (Poaceae) Madrono. 1995;23:134–153. [Google Scholar]
- 27.Stetter K O. Extremophiles and their adaptation to hot environments. FEBS Lett. 1999;452:22–25. doi: 10.1016/s0014-5793(99)00663-8. [DOI] [PubMed] [Google Scholar]
- 28.Stout R G, Kerstetter M, Summers M, McDermott T. Heat- and acid-tolerance of a grass commonly found in geothermal areas within Yellowstone National Park. Plant Sci. 1997;130:1–9. [Google Scholar]
- 29.Tansey M R, Brock T D. Isolation of thermophilic and thermotolerant fungi from hot spring effluents and thermal soils of Yellowstone National Park. Bacteriological Proceedings 1971. Washington, D.C.: American Society for Microbiology; 1971. p. 36. [Google Scholar]
- 30.Tansey M R, Brock T D. Dactylaria gallopava, a cause of avian encephalitis, in hot spring effluents, thermal soils and self-heated coal waste piles. Nature. 1973;242:202–203. doi: 10.1038/242202a0. [DOI] [PubMed] [Google Scholar]
- 31.Tansey M R, Brock T D. Microbial life at high temperatures: ecological aspects. In: Kushner D J, editor. Microbial life in extreme environments. London, United Kingdom: Academic Press; 1978. pp. 1–24. [Google Scholar]
- 32.Von Arx J A. The genera of fungi sporulating in pure culture. J. Vaduz, Germany: Cramer Publishing Co.; 1981. [Google Scholar]
- 33.Whitaker R H. Communities and ecosystems. New York, N.Y: MacMillan Publishing Co.; 1975. [Google Scholar]
- 34.Yellowstone National Park Soil Scientists. Soils investigation of the Reese Creek-McMinn Bench-Mammoth area, northwestern Yellowstone National Park, Wyoming. Mammouth, Wyo: National Park Service; 1991. [Google Scholar]