Abstract
Aims
Restoration of myocardial blood flow and perfusion during percutaneous coronary intervention (PCI) measured using Thrombolysis in Myocardial Infarction (TIMI) flow grade (TFG) and perfusion grade (TMPG) is associated with improved outcomes in acute coronary syndrome (ACS). Associations between TFG/TMPG and changes in biomarkers reflecting myocardial damage/dysfunction and inflammation is unknown.
Methods and results
Among 2606 patients included, TFG was evaluated in 2198 and TMPG in 1874 with ST-segment elevation myocardial infarction (STEMI) or non-ST-segment ACS (NSTE-ACS). Biomarkers reflecting myocardial necrosis [troponin T (TnT)], myocardial dysfunction [N-terminal prohormone brain natriuretic peptide (NT-proBNP)], inflammation [interleukin-6 (IL-6) and C-reactive protein (CRP)], and oxidative stress/ageing/inflammation [growth differentiation factor-15 (GDF-15)] were measured at baseline, discharge, and 1- and 6-month post-randomization. Associations between TFG/TMPG and changes in biomarker levels were evaluated using the Mann–Whitney–Wilcoxon signed test. In total, 1423 (54.6%) patients had STEMI and 1183 (45.4%) NSTE-ACS. Complete reperfusion after PCI with TFG = 3 was achieved in 1110 (85.3%) with STEMI and in 793 (88.5%) with NSTE-ACS. Normal myocardial perfusion with TMPG = 3 was achieved in 475 (41.6%) with STEMI and in 396 (54.0%) with NSTE-ACS. Levels of TnT, NT-proBNP, IL-6, CRP, and GDF-15 were substantially lower at discharge in patients with complete vs. incomplete TFG and STEMI (P < 0.01). This pattern was not observed for patients with NSTE-ACS. Patients with normal vs. abnormal TMPG and NSTE-ACS had lower levels of NT-proBNP at discharge (P = 0.01).
Conclusions
Successful restoration of epicardial blood flow in STEMI was associated with less myocardial necrosis/dysfunction and inflammation. Attainment of normal myocardial perfusion was associated with less myocardial dysfunction in NSTE-ACS.
Keywords: Acute coronary syndrome, Percutaneous coronary intervention, TIMI flow grade, TIMI myocardial perfusion grade, Biomarkers
Graphical Abstract
Graphical Abstract.
Introduction
Early coronary angiography and revascularization are recommended for patients presenting with either ST-segment elevation myocardial infarction (STEMI) or non-ST-segment acute coronary syndrome (NSTE-ACS) in order to lower the risk of subsequent recurrent ischaemic coronary events.1–4 For patients presenting with STEMI, percutaneous coronary intervention (PCI) should be performed without delay, aiming to immediately restore coronary blood flow in order to reduce myocardial damage and thereby lower the risk of events related to myocardial dysfunction.5,6 For patients with NSTE-ACS, the timing of invasive procedures is controversial as it is uncertain whether early stabilization of the impaired coronary blood flow has any effect on the development of myocardial damage.7,8
In patients with acute coronary syndrome (ACS) undergoing PCI, the restoration of nutritional blood flow can be evaluated by measuring flow and perfusion in the epicardial coronary arteries and the myocardial tissue, respectively. The restoration of epicardial blood flow is usually evaluated using the Thrombolysis in Myocardial Infarction (TIMI) flow grade (TFG) and the restoration of myocardial tissue perfusion by TIMI myocardial perfusion grade (TMPG).9,10 Successful restoration of epicardial blood flow and myocardial microvascular perfusion after ACS and PCI are both independently, and when combined into an angiographic perfusion score (APS), associated with reduced risk of future cardiovascular events.10,11
In patients with ACS, the magnitude of myocardial damage can be estimated by biomarkers reflecting myocardial necrosis [high-sensitivity (hs)-cardiac troponin T (hs-TnT)], myocardial dysfunction [N-terminal prohormone brain natriuretic peptide (NT-proBNP)], inflammation [C-reactive protein (CRP), interleukin-6 (IL-6)], and oxidative stress/ageing/inflammation [growth differentiation factor-15 (GDF-15)]. The levels of these biomarkers increase in the acute phase of ACS, and their levels and dynamic changes are associated both with the risk of acute complications and long-term recurrent cardiovascular events.12
Understanding the relationships between early restoration of coronary and tissue blood flow and the dynamic changes in biomarker levels reflecting processes related to myocardial damage might provide insight into the optimal treatment strategies in patients with STEMI and NSTE-ACS. The purpose of this study was therefore to outline the relations between angiographic outcomes, as measured by TFG and TMPG, and dynamic changes of biomarkers reflecting myocardial damage, myocardial dysfunction, inflammation, and oxidative stress/ageing in patients with STEMI and NSTE-ACS undergoing PCI.
Methods
Study population
The PLATelet inhibition and patient Outcomes (PLATO) trial (ClinicalTrials.gov identifier NCT00391872) was a double-blind, phase III, randomized controlled trial that studied the efficacy and safety of ticagrelor vs. clopidogrel in 18 624 patients with ACS, including patients with STEMI and NSTE-ACS. The follow-up was up to 12 months, with a median follow-up of 9 months. The study design, detailed endpoint definitions and outcomes have been previously published.13 Patients were included in the PLATO trial if they were hospitalized for STEMI or NSTE-ACS with onset within 24 h. For NSTE-ACS, at least two of three criteria were required: ST-segment depression or transient elevation ≥1 mm in two or more contiguous leads, positive biomarker indicating myocardial necrosis, or one additional risk factor: age >60 years, prior myocardial infarction (MI) or coronary artery bypass grafting, carotid artery disease, previous ischaemic stroke, transient ischaemic attack, carotid stenosis or cerebral revascularization, diabetes mellitus, peripheral artery disease, or chronic kidney disease. For STEMI, inclusion required a planned primary PCI. Among exclusion criteria, the most important included fibrinolytic therapy within 24 h, need for oral anticoagulation therapy, need for dialysis, and clinically important anaemia or thrombocytopenia.14
The PLATO angiographic substudy was designed in collaboration with the academic chairs and members of the steering committee and data were reviewed by the core lab at the academic research organization Pharmacologic/Percutaneous Endoluminal Revascularization For Unstable Syndromes and its Evaluation (PERFUSE).15 All sites with accurate angiographic equipment for digital acquisition of cineangiograms were invited to participate in the angiographic substudy. Angiograms were performed according to local practice patterns and saved and sent by compact discs to the PERFUSE angiographic core laboratory for analysis. A total of 2616 patients were included in the angiography substudy and for these patients an assessment of pre- and post-PCI coronary blood flow and perfusion was evaluated. In addition, all study patients at selected sites were invited to participate in the biomarker substudy which included venous blood sampling at baseline (randomization), discharge and after 1- and 6-month post-randomization. Informed consent was obtained from all patients, and the trial complied with the Helsinki Declaration.
Clinical exposure
The epicardial blood flow during coronary angiography was recorded with a TFG score between 0 (no perfusion) and 3 (complete reperfusion).14,16 As TFG only records epicardial blood flow and not myocardial perfusion, TMPG was assessed to determine microvascular perfusion entirely based on visual angiographic assessment and was scored 0 to 3 as previously defined.12,17 TMPG 0 represents no apparent tissue-level perfusion and TMPG 3 represents normal myocardial perfusion.12,17 In addition, APS was calculated based on TFG and TMPG scores pre- and post-PCI, with APS 0–3 representing failed perfusion, APS 4–9 partial perfusion, and APS 10–12 full perfusion. In this study, characteristics and outcomes were separately reported and compared between patients with incomplete vs. complete epicardial reperfusion blood flow (TFG ≤2 vs. TFG 3), and between abnormal vs. normal myocardial perfusion (TMPG ≤2 vs. TMPG 3). In supplementary analyses, characteristics and outcomes were separately reported and compared between patients with failed-partial vs. full perfusion (APS 0–9 vs. APS 10–12). All measurements were assessed at the PERFUSE angiographic core laboratory, as previously described.15
Biomarker analyses
The endpoint for this angiographic and biomarker substudy were the temporal changes in levels of hs-TnT, NT-proBNP, CRP, IL-6, and GDF-15 during the acute phase of ACS and during follow-up. Blood samples in the PLATO trial were collected by direct venepuncture and plasma samples were frozen at −70°C and stored in aliquots in a central repository at Uppsala Biobank until biochemical analyses were performed at the Uppsala Clinical Research Center (UCR) laboratory, Uppsala, Sweden. Blood samples were obtained at baseline (median of about 10 h after admission), at discharge, and at 1- and 6-month post-randomization. Levels of hs-TnT, NT-proBNP, and GDF-15 were analysed with sandwich immunoassays on Cobas Analytics Immunoanalyzers (Roche Diagnostics). C-reactive protein was analysed using the Architect platform (Abbott Diagnostics). IL-6 was measured using Quantikine hs-IL-6 ELISA (R&D Systems Inc). All analyses were performed at UCR Laboratory at Uppsala University, Sweden.
Statistics
Data management and statistical analyses were performed at UCR, Uppsala, Sweden. Patient characteristics including demographics, risk factors, medical history, and management were assessed as medians with 25th–75th percentiles for continuous variables and as frequencies with percentages for categorical variables. Characteristics were reported separately for patients with STEMI and NSTE-ACS, and separately for TFG ≤2 (incomplete reperfusion) vs. TFG 3 (complete reperfusion), and for TMPG ≤2 (abnormal myocardial perfusion) vs. TMPG 3 (normal myocardial perfusion). In supplementary analyses, characteristics were reported separately for patients with APS 0–9 (failed-partial perfusion) vs. APS 10–12 (full perfusion). Changes of biomarker levels over time (at baseline, discharge, 1 month and 6 months post-randomization) were presented in a tabular format and illustrated using line graphs indicating median values with confidence intervals. Statistical differences in biomarker levels over time in patients with TFG ≤2 vs. TFG 3, TMPG ≤2 vs. TMPG 3 and APS 0–9 vs. APS 10–12 were evaluated using the Mann–Whitney test within the STEMI (n = 1423) and NSTE-ACS (n = 1183) cohorts. In addition, statistical differences between biomarker levels at baseline and at discharge, 1 and 6 months [delta (Δ) statistics] were calculated using the Wilcoxon signed rank test. No adjustments were made for clinical characteristics or randomized treatment. Two-sided P-value of <0.05 was considered statistically significant and no correction was made for multiplicity, so the results are viewed as hypothesis-generating. Statistical analyses were performed with R version 3.6.0 (The R Foundation for Statistical Computing).
Results
Baseline characteristics
The PLATO angiographic substudy included 2616 patients of whom 2606 (99.6%) had biomarker samples collected at any time point and were therefore included in the present study. Among those included, 2198 (84.3%) and 1874 (71.9%) had TFG and TMPG measurements collected. Baseline characteristics and angiographic measurements in patients with STEMI and NSTE-ACS according to TFG and TMPG post-PCI are summarized in Table 1. Baseline characteristics and angiographic measurements according to APS are presented in Supplementary material online, Table S1. The median age of the population was 61 years, and 1910 (73.3%) were men. Amongst those included, 1423 (54.6%) had a diagnosis of STEMI and 1183 (45.4%) NSTE-ACS. Median time from symptom onset to PCI was 5.2 h for patients with STEMI and 24.3 h for patients with NSTE-ACS, and median time from diagnostic electrocardiogram to PCI was 1.7 h for patients with STEMI and 21.0 h for patients with NSTE-ACS. Complete reperfusion after PCI with TFG 3 was achieved in 1110 (85.3%) with STEMI and in 793 (88.5%) with NSTE-ACS. Amongst patients with available TMPG measurement, normal myocardial perfusion with TMPG 3 was achieved in 475 (41.6%) with STEMI and in 396 (54.0%) with NSTE-ACS after PCI. Patients with complete vs. incomplete reperfusion were in general younger and were more like to be habitual smokers and have risk factors such as dyslipidaemia, diabetes mellitus, peripheral artery disease, heart failure, and history of MI. In comparison, patients with normal vs. abnormal myocardial perfusion were more likely to be younger and habitual smokers (Table 1). Compared to patients with NSTE-ACS, patients with STEMI had higher concentrations of hs-TnT, NT-proBNP, CRP, IL-6, and GDF-15 at discharge (Tables 2 and 3).
Table 1.
Baseline characteristics
| Characteristic | STEMI | NSTE-ACS | ||||||
|---|---|---|---|---|---|---|---|---|
| (n = 1423) | (n = 1183) | |||||||
| TFG ≤ 2 | TFG = 3 | TMPG ≤ 2 | TMPG = 3 | TFG ≤ 2 | TFG = 3 | TMPG ≤ 2 | TMPG = 3 | |
| (n = 192) | (n = 1110) | (n = 666) | (n = 475) | (n = 103) | (n = 793) | (n = 337) | (n = 396) | |
| Demographics | ||||||||
| Age (median, IQR) | 61 (53–69) | 58 (51–67) | 59 (52–66) | 58 (51–68) | 66 (58–73) | 64 (55–71) | 65 (55–73) | 64 (56–71) |
| Sex, male | 143 (74.5) | 819 (73.8) | 481 (72.2) | 345 (72.6) | 81 (78.6) | 571 (72.0) | 248 (73.6) | 279 (70.5) |
| BMI kg/m2 (median, IQR), n = 2600 | 27.8 (25.2–30.8) | 27.7 (25.1–30.7) | 27.7 (24.9–30.4) | 27.6 (25–30.8) | 27.3 (24.9–29.7) | 27.7 (25.2–30.9) | 27.1 (24.5–30.4) [3] | 27.8 (25.4–31.) |
| Risk factors | ||||||||
| Habitual smoker | 82 (42.7) | 556 (50.1) | 329 (49.4) | 218 (45.9) | 27 (26.2) | 280 (35.3) | 129 (38.3) | 128 (32.3) |
| Hypertension | 110 (57.3) | 634 (57.1) | 377 (56.6) | 274 (57.7) | 56 (54.4) | 536 (67.6) | 219 (65.0) | 266 (67.2) |
| Dyslipidaemia | 59 (30.7) | 398 (35.9) | 235 (35.3) | 165 (34.7) | 45 (43.7) | 404 (50.9) | 169 (50.1) | 211 (53.3) |
| Diabetes mellitus | 29 (15.1) | 188 (16.9) | 114 (17.1) | 79 (16.6) | 20 (19.4) | 189 (23.8) | 77 (22.8) | 92 (23.2) |
| Medical history | ||||||||
| Myocardial infarction | 15 (7.8) | 128 (11.5) | 74 (11.1) | 43 (9.1) | 19 (18.4) | 165 (20.8) | 67 (19.9) | 81 (20.5) |
| Heart failure | 2 (1.0) | 23 (2.1) | 10 (1.5) | 8 (1.7) | 1 (1.0) | 31 (3.9) | 13 (3.9) | 13 (3.3) |
| PCI | 8 (4.2) | 75 (6.8) | 39 (5.9) | 27 (5.7) | 12 (11.7) | 150 (18.9) | 61 (18.1) | 63 (15.9) |
| CABG | 3 (1.6) | 18 (1.6) | 12 (1.8) | 4 (0.8) | 11 (10.7) | 69 (8.7) | 37 (11.0) | 27 (6.8) |
| Non-haemorrhagic stroke | 4 (2.1) | 24 (2.2) | 16 (2.4) | 9 (1.9) | 0 (0) | 18 (2.3) | 8 (2.4) | 7 (1.8) |
| Peripheral arterial disease | 3 (1.6) | 53 (4.8) | 30 (4.5) | 19 (4.0) | 4 (3.9) | 53 (6.7) | 19 (5.6) | 28 (7.1) |
| Chronic renal disease | 4 (2.1) | 23 (2.1) | 12 (1.8) | 10 (2.1) | 2 (1.9) | 34 (4.3) | 11 (3.3) | 18 (4.5) |
| Randomized treatment | ||||||||
| Ticagrelor | 97 (50.5) | 556 (51.5) | 330 (49.5) | 247 (52.0) | 48 (46.6) | 401 (50.6) | 174 (51.6) | 194 (49.0) |
| Clopidogrel | 95 (49.5) | 544 (49.0) | 336 (50.5) | 228 (48.0) | 55 (53.4) | 392 (49.4) | 163 (48.4) | 202 (51.0) |
| Angiographic measurements, post-PCI | ||||||||
| TFG | ||||||||
| TFG = 0 | 18 (9.4) | 0 (0) | 17 (2.6) | 0 (0.0) | 22 (21.4) | 0 (0.0) | 20 (6.0) | 1 (0.3) |
| TFG = 1 | 25 (13) | 0 (0) | 21 (3.2) | 3 (0.6) | 14 (13.6) | 0 (0.0) | 12 (3.6) | 1 (0.3) |
| TFG = 2 | 149 (77.6) | 0 (0) | 89 (13.4) | 47 (9.9) | 67 (65.0) | 0 (0.0) | 26 (7.8) | 27 (6.9) |
| TFG = 3 | 0 (0.0) | 1110 (100) | 535 (80.8) | 424 (89.5) | 0 (0.0) | 793 (100.0) | 274 (82.5) | 365 (92.6) |
| TMPG | ||||||||
| TMPG = 0 | 43 (24.3) | 20 (2.1) | 65 (9.8) | 0 (0.0) | 20 (23.0) | 22 (3.4) | 44 (13.1) | 0 (0.0) |
| TMPG = 1 | 80 (45.2) | 444 (46.3) | 526 (79.0) | 0 (0.0) | 31 (35.6) | 202 (31.6) | 236 (70.0) | 0 (0.0) |
| TMPG = 2 | 4 (2.3) | 71 (7.4) | 75 (11.3) | 0 (0.0) | 7 (8.0) | 50 (7.8) | 57 (16.9) | 0 (0.0) |
| TMPG = 3 | 50 (28.2) | 424 (44.2) | 0 (0.0) | 475 (100.0) | 29 (33.3) | 365 (57.1) | 0 (0.0) | 396 (100.0) |
BMI, body mass index; CABG, coronary artery bypass grafting; NSTE-ACS, non-ST-segment elevation acute coronary syndrome; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction; TFG, Thrombolysis in Myocardial Infarction (TIMI) flow grade; TMPG, TIMI myocardial perfusion grade.
Table 2.
Temporal changes in biomarker levels in patients with STEMI or NSTE-ACS with complete vs. incomplete TFG
| Biomarker | STEMI | NSTE-ACS | ||||||
|---|---|---|---|---|---|---|---|---|
| (n = 1302) | (n = 896) | |||||||
| TFG ≤ 2 | TFG = 3 | P-value | Δ P-value | TFG ≤ 2 | TFG = 3 | P-value | Δ P-value | |
| (n = 192) | (n = 1110) | (n = 103) | (n = 793) | |||||
| Hs-troponin T (ng/L) | ||||||||
| Baseline | 180.0 (134.0–255.0) | 98.2 (88.9–110.0) | <0.001 | Ref | 250.0 (162.0–326.0) | 214.5 (186.0–239.0) | 0.51 | Ref |
| (n = 182) | (n = 1083) | (n = 97) | (n = 760) | |||||
| Discharge | 3114.5 (2194.0–3601.0) | 1736.0 (1613.0–1918.0) | <0.001 | <0.001 | 509.0 (325.0–756.0) | 366.0 (310.0–446.0) | 0.04 | 0.07 |
| (n = 172) | (n = 1052) | (n = 89) | (n = 687) | |||||
| 1 month | 16.4 (15.1–20.2) | 12.0 (11.4–12.7) | <0.001 | 0.001 | 10.6 (9.5–13.0) | 10.9 (10.4–11.4) | 0.50 | 0.47 |
| (n = 165) | (n = 1005) | (n = 85) | (n = 622) | |||||
| 6 months | 9.2 (8.2–11.7) | 8.2 (7.7–8.8) | 0.07 | 0.004 | 8.2 (6.7–10) | 8.8 (8.0–9.8) | 0.60 | 0.82 |
| (n = 71) | (n = 442) | (n = 39) | (n = 316) | |||||
| NT-proBNP (ng/L) | ||||||||
| Baseline | 232.5 (151.0–319.0) | 172.0 (152.0–192.0) | 0.03 | Ref | 337.0 (245.0–466.0) | 530.0 (463.0–584.0) | 0.003 | Ref |
| (n = 184) | (n = 1087) | (n = 98) | (n = 764) | |||||
| Discharge | 1120.0 (960.0–1474.0) | 710.0 (658.0–778.0) | <0.001 | <0.001 | 532.0 (314.0–695.0) | 432.0 (393.0–483.0) | 0.49 | 0.003 |
| (n = 172) | (n = 1051) | (n = 89) | (n = 687) | |||||
| 1 month | 1027.0 (892.0–1284.0) | 593.0 (561.0–645.0) | <0.001 | 349.0 (214.0–484.0) | 321.0 (284.0–359.0) | 0.92 | 0.11 | |
| (n = 165) | (n = 1005) | <0.001 | (n = 85) | (n = 622) | ||||
| 6 months | 229.0 (183.0–409.0) | 188.0 (170.0–224.0) | 0.07 | 0.83 | 128.0 (79.0–202.0) | 141.0 (120.0–164.0) | 0.68 | 0.32 |
| (n = 71) | (n = 442) | (n = 39) | (n = 316) | |||||
| CRP (mg/L) | ||||||||
| Baseline | 2.9 (2.4–4.2) | 2.8 (2.5–3.0) | 0.11 | Ref | 2.7 (2.3–3.3) | 3.7 (3.4–4.1) | 0.07 | Ref |
| (n = 181) | (n = 1046) | (n = 90) | (n = 690) | |||||
| Discharge | 31.0 (27.0–36.0) | 18.0 (17.0–19.0) | <0.001 | <0.001 | 16.0 (9.2–20.0) | 12.0 (11.0–14.0) | 0.30 | 0.03 |
| (n = 171) | (n = 1051) | (n = 89) | (n = 684) | |||||
| 1 month | 2.9 (2.4–3.4) | 2.2 (2.0–2.4) | 0.09 | 0.42 | 1.8 (1.3–2.4) | 2.1 (1.9–2.3) | 0.18 | 0.36 |
| (n = 165) | (n = 1004) | (n = 85) | (n = 622) | |||||
| 6 months | 2.0 (1.4–3.0) | 2.0 (1.7–2.2) | 0.89 | 0.14 | 1.8 (1.0–2.3) | 1.8 (1.5–2.0) | 0.74 | 0.38 |
| (n = 70) | (n = 442) | (n = 39) | (n = 315) | |||||
| IL-6 (ng/L) | ||||||||
| Baseline | 3.4 (3.0–3.6) | 3.0 (2.8–3.2) | 0.01 | Ref | 3.0 (2.5–3.6) | 3.3 (3.0–3.6) | 0.20 | Ref |
| (n = 185) | (n = 1094) | (n = 96) | (n = 753) | |||||
| Discharge | 8.9 (7.7–9.7) | 6.0 (5.6–6.4) | <0.001 | <0.001 | 6.5 (4.9–8.3) | 5.4 (5.2–6.1) | 0.24 | 0.03 |
| (n = 180) | (n = 1072) | (n = 94) | (n = 728) | |||||
| 1 month | 2.1 (1.9–2.4) | 2.1 (1.9–2.2) | 0.46 | 0.01 | 1.7 (1.5–2.1) | 2.0 (1.9–2.2) | 0.05 | 0.49 |
| (n = 176) | (n = 1039) | (n = 92) | (n = 702) | |||||
| 6 months | 1.8 (1.6–2.1) | 1.9 (1.8–2.0) | 0.46 | 0.008 | 1.9 (1.5–2.6) | 1.9 (1.7–2.0) | 0.74 | 0.25 |
| (n = 70) | (n = 442) | (n = 39) | (n = 315) | |||||
| GDF-15 (ng/L) | ||||||||
| Baseline | 1467.7 (1315.5–1601.5) | 1464.8 (1398.9–1507.8) | 0.70 | Ref | 1461.1 (1281.8–1600.8) | 1518.2 (1452.8–1570.4) | 0.36 | Ref |
| (n = 183) | (n = 1084) | (n = 97) | (n = 767) | |||||
| Discharge | 1511.7 (1431.1–1660.2) | 1409.6 (1380.6–1447.8) | 0.005 | 0.002 | 1799.2 (1412.7–1930.3) | 1600.0 (1532.0–1661.2) | 0.55 | 0.08 |
| (n = 172) | (n = 1051) | (n = 89) | (n = 687) | |||||
| 1 month | 1277.8 (1212.9–1417.6) | 1244.8 (1198.8–1286.1) | 0.32 | 0.64 | 1311.8 (1212.0–1606.0) | 1411.7 (1355.8–1487.1) | 0.74 | 0.69 |
| (n = 165) | (n = 1005) | (n = 85) | (n = 622) | |||||
| 6 months | 1190.9 (1048.7–1311.4) | 1223.7 (1168.9–1289.9) | 0.40 | 0.27 | 1273.0 (1072.3–1582.3) | 1356.8 (1295.2–1502.1) | 0.48 | 0.85 |
| (n = 71) | (n = 442) | (n = 39) | (n = 318) | |||||
CRP, C-reactive protein; GDF-15, growth differentiation factor 15; Hs-Troponin T, high-sensitivity Troponin T; IL-6, interleukin-6; NSTE-ACS, non-ST-segment elevation acute coronary syndrome; NT-proBNP, N-terminal prohormone brain natriuretic peptide; STEMI, ST-segment elevation myocardial infarction; TFG, Thrombolysis in Myocardial Infarction (TIMI) flow grade.
P-value: Statistical differences in biomarkers levels over time in patients with TFG ≤2 vs. TFP = 3 evaluated using the Mann–Whitney test.
Δ P-value: Statistical differences in biomarkers levels over time in comparison to baseline levels in patients with TFG ≤2 vs. TFP = 3 evaluated using the Wilcoxon signed rank test.
Table 3.
Temporal changes in biomarker levels in patients with STEMI or NSTE-ACS with normal vs. abnormal TMPG
| Biomarker | STEMI | NSTE-ACS | ||||||
|---|---|---|---|---|---|---|---|---|
| (n = 1141) | (n = 733) | |||||||
| TMPG ≤ 2 | TMPG = 3 | P-value | Δ P-value | TMPG ≤ 2 | TMPG = 3 | P-value | Δ P-value | |
| (n = 666) | (n = 475) | (n = 337) | (n = 396) | |||||
| hs-Troponin T (ng/L) | ||||||||
| Baseline | 97.1 (79.4–118.0) | 108.0 (92.0–129.0) | 0.37 | Ref | 252.0 (195.0–316.0) | 181.5 (150.0–220.0) | 0.08 | Ref |
| (n = 645) | (n = 463) | (n = 319) | (n = 378) | |||||
| Discharge | 2154.5 (1916.0–2352.0) | 1602.0 (1440.0–1845.0) | <0.001 | 0.001 | 470.0 (346.0–560.0) | 338.0 (272.0–446.0) | 0.04 | 0.07 |
| (n = 622) | (n = 459) | (n = 284) | (n = 351) | |||||
| 1 month | 13.0 (12.0–13.6) | 11.7 (10.9–12.7) | 0.01 | 0.19 | 10.9 (10.4–11.9) | 10.6 (9.3–11.6) | 0.41 | 0.08 |
| (n = 598) | (n = 435) | (n = 263) | (n = 321) | |||||
| 6 months | 8.7 (8.1–9.1) | 7.7 (7.2–8.4) | 0.17 | 0.66 | 9.2 (7.4–10.0) | 8.6 (7.1–10.0) | 0.87 | 0.02 |
| (n = 250) | (n = 199) | (n = 121) | (n = 158) | |||||
| NT-proBNP (ng/L) | ||||||||
| Baseline | 151.0 (134.0–174.0) | 192.0 (162.0–249.0) | 0.06 | Ref | 475.0 (414.0–585.0) | 515.0 (425.0–584.0) | 0.81 | Ref |
| (n = 649) | (n = 465) | (n = 321) | (n = 381) | |||||
| Discharge | 728.0 (674.0–808.0) | 809.0 (691.0–898.0) | 0.51 | 0.67 | 501.0 (410.0–620.0) | 411.0 (326.0–466.0) | 0.01 | 0.12 |
| (n = 621) | (n = 459) | (n = 284) | (n = 351) | |||||
| 1 month | 678.5 (600.0–760.0) | 598.0 (522.0–698.0) | 0.03 | 0.003 | 370.0 (297.0–464.0) | 282.0 (240.0–330.0) | 0.01 | 0.17 |
| (n = 598) | (n = 435) | (n = 263) | (n = 321) | |||||
| 6 months | 210.0 (184.0–263.0) | 175.0 (142.0–216.0) | 0.08 | 0.31 | 162.0 (123.0–224.0) | 121.0 (104.0–146.0) | 0.08 | 0.55 |
| (n = 250) | (n = 199) | (n = 121) | (n = 158) | |||||
| CRP (mg/L) | ||||||||
| Baseline | 2.7 (2.4–3.0) | 2.8 (2.4–3.2) | 0.60 | Ref | 3.0 (2.6–3.6) | 3.7 (3.3–4.5) | 0.41 | Ref |
| (n = 627) | (n = 451) | (n = 294) | (n = 343) | |||||
| Discharge | 21.0 (19.0–22.0) | 18.0 (16.0–20.0) | 0.05 | 0.06 | 15.0 (12.0–18.0) | 11.5 (9.4–13.0) | 0.03 | 0.001 |
| (n = 620) | (n = 459) | (n = 284) | (n = 348) | |||||
| 1 month | 2.3 (2.0–2.6) | 2.2 (2.0–2.6) | 0.64 | 0.86 | 2.2 (1.8–2.4) | 2.0 (1.7–2.3) | 0.37 | 0.18 |
| (n = 598) | (n = 434) | (n = 263) | (n = 321) | |||||
| 6 months | 1.9 (1.6–2.2) | 2.1 (1.8–2.5) | 0.44 | 0.002 | 1.8 (1.5–2.3) | 1.6 (1.1–2.0) | 0.59 | 0.42 |
| (n = 249) | (n = 199) | (n = 121) | (n = 157) | |||||
| IL-6 (ng/L) | ||||||||
| Baseline | 3.0 (2.7–3.3) | 3.0 (2.7–3.3) | 0.97 | Ref | 3.3 (3.0–3.6) | 3.0 (2.7–3.5) | 0.42 | Ref |
| (n = 654) | (n = 468) | (n = 312) | (n = 377) | |||||
| Discharge | 6.7 (6.1–7.3) | 6.4 (5.7–7.1) | 0.51 | 0.45 | 6.3 (5.1–7.1) | 5.6 (5.2–6.4) | 0.30 | 0.23 |
| (n = 638) | (n = 466) | (n = 301) | (n = 368) | |||||
| 1 month | 2.1 (1.9–2.2) | 2.0 (1.8–2.1) | 0.36 | 0.67 | 2.0 (1.8–2.3) | 2.0 (1.9–2.2) | 0.71 | 0.35 |
| (n = 626) | (n = 442) | (n = 293) | (n = 358) | |||||
| 6 months | 2.0 (1.8–2.1) | 1.8 (1.6–2.0) | 0.63 | 0.45 | 1.9 (1.5–2.1) | 1.8 (1.7–2.0) | 0.86 | 0.74 |
| (n = 249) | (n = 199) | (n = 121) | (n = 157) | |||||
| GDF-15 (ng/L) | ||||||||
| Baseline | 1482.4 (1415.9–1545.1) | 1420.4 (1331.9–1511.9) | 0.45 | Ref | 1520.9 (1432.9–1591.8) | 1472.2 (1389.4–1572.0) | 0.60 | Ref |
| (n = 646) | (n = 464) | (n = 323) | (n = 380) | |||||
| Discharge | 1430.0 (1382.1–1481.7) | 1412.4 (1363.9–1481.4) | 0.84 | 0.71 | 1646.9 (1530.0–1796.3) | 1596.8 (1462.9–1733.0) | 0.16 | 0.10 |
| (n = 621) | (n = 459) | (n = 284) | (n = 351) | |||||
| 1 month | 1240.4 (1190.7–1277.8) | 1257.5 (1163.8–1325.3) | 0.69 | 0.32 | 1403.8 (1314.1–1546.8) | 1395.8 (1314.5–1489.9) | 0.57 | 0.73 |
| (n = 598) | (n = 435) | (n = 263) | (n = 321) | |||||
| 6 months | 1223.9 (1167.4–1337.8) | 1211.1 (1109.5–1285.4) | 0.22 | 1351.1 (1258.2–1617.6) | 1373.9 (1270.3–1512.0) | 0.79 | 0.42 | |
| (n = 250) | (n = 199) | 0.56 | (n = 122) | (n = 159) | ||||
CRP, C-reactive protein; GDF-15, growth differentiation factor 15; hs-Troponin T, high-sensitivity Troponin T; IL-6, interleukin-6; NSTE-ACS, non-ST-segment elevation acute coronary syndrome; NT-proBNP, N-terminal prohormone brain natriuretic peptide; STEMI, ST-segment elevation myocardial infarction; TMPG, Thrombolysis in Myocardial Infarction (TIMI) myocardial perfusion grade.
P-value: Statistical differences in biomarkers levels over time in patients with TMPG ≤2 vs. TMPG = 3 evaluated using the Mann–Whitney test.
Δ P-value: Statistical differences in biomarkers levels over time in comparison to baseline levels in patients with TMPG ≤2 vs. TMPG = 3 evaluated using the Wilcoxon signed rank test.
Angiographic outcomes and biomarker levels in patients with ST-segment elevation myocardial infarction
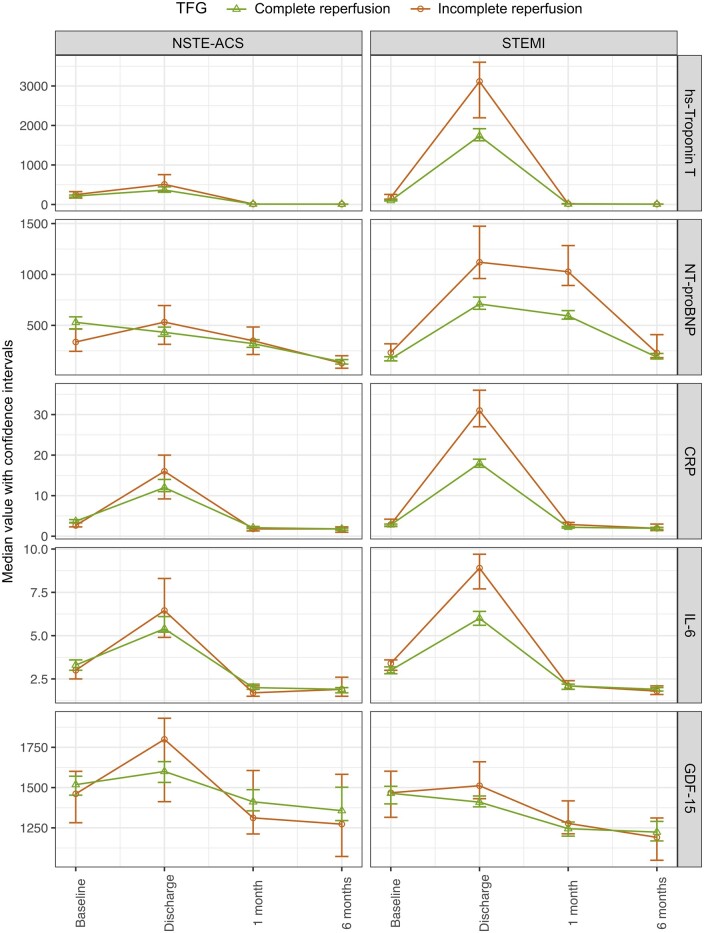
Temporal changes in biomarker levels between baseline, discharge, 1 month and 6 months post-randomization in patients with STEMI and with complete vs. incomplete TFG restoration and normal vs. abnormal TMPG after PCI are presented in Figures 1 and 2 and in Tables 2 and 3. Similarly, temporal changes in biomarker levels in patients with full vs. failed-partial perfusion according to APS are presented in Supplementary material online, Figure S1 and Table S2. In patients with STEMI and with successful reperfusion with the achievement of TFG 3, median levels of hs-TnT, NT-proBNP, and IL-6 were significantly lower at baseline prior to PCI compared to patients with TFG ≤2. Similarly, patients with TFG 3 vs. TFG ≤2 had lower levels of hs-TnT and NT-proBNP at discharge and 1-month post-randomization. Median levels of CRP, IL-6, and GDF-15 were significantly lower in patients with STEMI and with TFG 3 vs. TFG ≤2 on discharge (Figure 1 and Table 2). In comparison, patients with STEMI and TMPG 3 vs. TMPG ≤2 had lower levels of hs-TnT at discharge with no other associations observed (Figure 2 and Table 3). Analogous results were observed among patients with full APS vs. failed-partial APS (Supplementary material online, Figure S1 and Table 2).
Figure 1.
Dynamic changes in biomarker levels (median values with confidence interval) in patients with complete vs. incomplete Thrombolysis in Myocardial Infarction flow grade.
Figure 2.
Dynamic change in biomarker levels (median values with confidence interval) in patients with normal vs. abnormal Thrombolysis in Myocardial Infarction myocardial perfusion grade.
Angiographic outcomes and biomarker levels in patients with non-ST-segment acute coronary syndrome
In patients with NSTE-ACS, levels of hs-TnT were lower at discharge in patients with TFG 3 vs. TFG ≤2. No other significant associations were observed in relation to biomarker levels over time and successful reperfusion as measured using TFG (Figure 1 and Table 2). In comparison, patients with NSTE-ACS and TMPG 3 vs. TMPG ≤2 had lower levels of hs-TnT, NT-proBNP, and CRP on discharge, and also lower levels of NT-proBNP at 1 month (Figure 2 and Table 3). No other significant differences were seen over time. A similar pattern was observed among patients with NSTE-ACS and full APS vs. failed-partial APS (Supplementary material online, Figure S1 and Table 2).1
Discussion
In this PLATO substudy, we assessed angiographic markers of epicardial and tissue reperfusion/perfusion and their association with temporal changes in biomarker levels reflecting myocardial damage, myocardial dysfunction, inflammation, and oxidative stress/ageing in patients with STEMI and NSTE-ACS undergoing early PCI procedures. As expected, indicators of myocardial necrosis, myocardial dysfunction and inflammation were substantially higher in the acute phase in patients with STEMI than NSTE-ACS. Patients with STEMI and with complete vs. incomplete reperfusion as measured by TFG had less myocardial necrosis and dysfunction (TnT and NT-proBNP) on discharge and 1-month post-randomization, and lower levels of inflammatory activity (CRP and IL-6) and GDF-15 at discharge. Similarly, patients with STEMI and with normal vs. abnormal myocardial perfusion as measured by TMPG had lower levels of TnT on discharge. In patients with NSTE-ACS, improved myocardial perfusion, as measured by TMPG, was associated with lower levels of NT-proBNP on discharge and 1-month follow-up. These findings emphasize the importance of early successful and complete revascularization in patients with STEMI, as well as the importance of knowledge of timing of blood sampling when using biomarkers for prediction of cardiovascular outcomes in patients with ACS.
Previous studies have established associations between markers of myocardial necrosis (TnT), myocardial dysfunction (NT-proBNP) and cardiovascular outcomes, including cardiovascular mortality and MI in patients with ACS and in those with chronic coronary syndromes.18,19 Similarly, inflammatory biomarkers (IL-6 and CRP) have been showed to be associated with adverse cardiovascular outcomes, and anti-inflammatory drugs have shown promise in randomized clinical trials as potential treatment strategies in patients with chronic coronary syndromes.20–23 Moreover, GDF-15, both in the acute and chronic state after ACS, has in previous studies been associated with recurrent cardiovascular events and major bleeding.24–26 Based on these findings, biomarker-based risk scores such as the age-biomarkers-clinical variables (ABC) score, which incorporates TnT and NT-proBNP, have been developed for prediction of cardiovascular mortality in patients with chronic coronary syndromes.19
Successful early restoration of epicardial blood flow and improved myocardial perfusion as measured using TFG and TMPG, respectively, have been related to lower 30-day and long-term mortality in patients with STEMI, and with a trend in reduction for patients with NSTE-ACS.12,17,27–29 TMPG in addition to TFG adds long-term prognostic information and studies have shown that early complete restoration of both epicardial blood flow and microvascular perfusion are complementary when assessing risk of long-term mortality in patients with STEMI, and can be combined into a score (APS) for risk assessment11,12 However, the importance of the biomarker response in relation to angiographic restoration of epicardial blood flow and myocardial perfusion is unclear. In the current study, successful restoration of epicardial blood flow was associated with lower levels of TnT and NT-proBNP at discharge and at 1-month follow-up in patients with STEMI, but not in patients with NSTE-ACS. Similarly, restoration of epicardial blood flow was associated with lower levels of inflammatory biomarkers (CRP and IL-6) and GDF-15 at discharge in patients with STEMI, but not in NSTE-ACS. However, patients with NSTE-ACS and with normal vs. abnormal myocardial perfusion had lower levels of NT-proBNP at discharge and 1-month follow-up. Concerning patients with STEMI, similar observations have been made in the Assessment of Pexelizumab (APEX)-acute myocardial infarction biomarker substudy in which 376 patients with STEMI had biomarkers measured at the time of randomization and 24 h later, in which levels of TnT, NT-proBNP, CRP, and IL-6 were reduced in patients with complete restoration of epicardial blood.30 However, to our knowledge, there are no previous studies on the associations between TFG, TMPG, and biomarker levels in patients with NSTE-ACS.
The present findings have implications both for the timing of coronary procedures and blood sampling for biomarker levels in patients with STEMI and NSTE-ACS. The demonstration of the association between early successful restoration of epicardial blood flow and immediate and long-term reduction of biomarkers of myocardial necrosis/function (TnT and NT-proBNP), inflammation (CRP and IL-6), and GDF-15 in patients with STEMI emphasizes the importance of immediate and successful coronary reperfusion to reduce the degree of myocardial damage and to avoid myocardial dysfunction and reinfarction in patients with STEMI. However, how an early and successful reperfusion might be achieved is a clinical challenge in itself with pre- and peri-hospital logistics playing a key role to minimize time between first medical contact and wire crossing. Also, optimized pre-hospital treatment of ongoing STEMI with fibrinolytic agents or glycoprotein IIb/IIIa inhibitors have in some studies been associated with improved outcomes, but there is limited evidence with respect to if early P2Y12 inhibitor initiation might improve outcome.31,32 For example, in the Ambulance for New ST Elevation Myocardial Infarction to Open the Coronary Artery (ATLANTIC) trial, pre-hospital versus in-hospital initiation of ticagrelor at a median difference of 31 minutes was associated with similar rates of pre-PCI coronary reperfusion.33 As such, early and successful reperfusion is important in patients with STEMI to improve the acute and long-term prognosis, however, the tools to further improve this are currently limited. On the other hand, the results in our study also emphasize that in patients with NSTE-ACS, coronary revascularization seems less urgent as early successful epicardial reperfusion did not influence the development of myocardial damage as assessed using biomarkers. However, still the early performance of revascularization is important based on the findings that attainment of normal myocardial perfusion was associated with lower levels of NT-proBNP, which in turn is associated with improved long-term risk for new cardiovascular events.12,17,27–29 Furthermore, our finding emphasize that timing of biomarker sampling in relation to PCI and in relation to revascularization outcomes might be of importance when using biomarkers as prognostic indicators, especially since we have shown that there are temporal changes in biomarker concentrations in relation to revascularization outcomes.
Limitations
As with all substudies, there are inherent limitations to this study that merits consideration. First, the generalizability of this substudy might be limited to the patients meeting the inclusion and exclusion criteria of the PLATO trial. However, the PLATO trial included a wide range of patients with ACS, including patients with STEMI and NSTE-ACS. Second, this study involved multiple biomarkers in a large cohort, however, only a subgroup of 2606 patients were included in this angiographic and biomarker substudy and there was a drop in number of patients providing biomarker samples during follow-up at 1 and 6 months, leading to a loss in power to detect minimal differences in biomarker levels. Furthermore, the drop in patients providing biomarkers samples at 6 months was substantial and might be due to unmeasured confounders. Finally, not all angiographic parameters were evaluable in all coronary angiograms. Still, this uniquely large international trial cohort provides important information compared to prior angiographic and biomarker studies.
Conclusions
In patients with ACS, biomarkers reflecting myocardial necrosis, myocardial dysfunction, and inflammation were substantially higher in the acute phase following STEMI compared to NSTE-ACS. In STEMI, successful restoration of epicardial blood was associated with less myocardial necrosis and dysfunction (TnT and NT-proBNP) on discharge and 1-month post-randomization, and lower levels of inflammatory activity (CRP and IL-6) and GDF-15 at discharge. In patients with NSTE-ACS, successful restoration of myocardial perfusion was associated with less myocardial dysfunction (NT-proBNP) on discharge and 1-month follow-up. These findings underline the importance of successful restoration of epicardial blood in patients with STEMI, and the importance of successful restoration of myocardial perfusion in patients with ACS.
Supplementary material
Supplementary material is available at European Heart Journal: Acute Cardiovascular Care online.
Supplementary Material
Acknowledgements
The authors thank the patients who participated in the PLATO trial. The authors thank Ida Björkgren (Uppsala Clinical Research Center, Uppsala, Sweden) for the editorial support.
Funding
The PLATO (PLATelet inhibition and patients Outcome) trial was funded by AstraZeneca.
Conflict of interest: G.B. reports honoraria for lectures and scientific advice from AstraZeneca, Boehringer Ingelheim, and Novo Nordisk. H.R. reports institutional research grant from AstraZeneca. V.K. reports personal fees from Abbott Vascular, Bayer, Amgen, and Daichii-Sankyo; grants from AstraZeneca. S.K.J. reports institutional research grant, honoraria, and consultant/advisory board fee from AstraZeneca and institutional research grant and consultant/advisory board fee from Medtronic; institutional research grants and honoraria from The Medicines Company; consultant/advisory board fees from Janssen, Bayer, Novartis, Amgen, and PhaseBio. R.F.S. reports institutional research grants, consultancy fees, and honoraria from AstraZeneca; institutional research grants and consultancy fees from Cytosorbents, GlyCardial Diagnostics, and Thromboserin; consultancy fees and honoraria from Bayer and Bristol‐Myers Squibb/Pfizer; and consultancy fees from Amgen, Hengrui, Idorsia, PhaseBio, Portola, and Sanofi Aventis; and honoraria from Intas Pharmaceuticals and Medscape. P.G.S. reports grants and speaker/consultancy fees from Amarin, Bayer, Sanofi, and Servier; speaker/consultancy fees from AstraZeneca, Amgen, Amarin, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Idorsia, Janssen, Novartis, Pfizer, and Regeneron. H.A.K. reports personal fees from AstraZeneca, Bayer Vital, and Roche Diagnostics. R.A.H. reports institutional research grants from AstraZeneca during the conduct of the study; and research grants from Janssen and consulting fees from Bristol-Myers Squibb outside the submitted work. C.M.G. reports research support from Johnson & Johnson; consulting support from AstraZeneca, Johnson & Johnson, Janssen Pharmaceuticals, and Bayer Corporation. A.B. reports personal fees and non-financial support from AstraZeneca, during the conduct of the study; personal fees and non-financial support from Bristol Myers Squibb, Sanofi Aventis, and Bayer; personal fees from Eisai, Novartis, GlaxoSmithKline, and Amgen, outside the submitted work. A.S. reports institutional research grants from AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb/Pfizer, GlaxoSmithKline, and Roche Diagnostics; consulting fees from OLINK Proteomics. L.W. reports institutional research grants from AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb/Pfizer, GlaxoSmithKline, Merck&Co, and Roche Diagnostics; consulting fees from Abbott.
Data availability
The sponsors of the PLATO trial (AstraZeneca, Cambridge, UK) are committed to responsible sharing of clinical study reports, related clinical documents, and patient level clinical study data. Researchers are invited to submit inquiries online. To submit inquiries, please visit https://astrazenecagrouptrials.pharmacm.com. All data relevant to the study are included in the article or uploaded as supplementary information.
Contributor Information
Gorav Batra, Department of Medical Sciences, Cardiology, Uppsala University, Uppsala, Sweden; Uppsala Clinical Research Center, Uppsala University, Uppsala Science Park, Hubben, Dag Hammarskjölds väg 38, 751 85 Uppsala, Sweden.
Henrik Renlund, Uppsala Clinical Research Center, Uppsala University, Uppsala Science Park, Hubben, Dag Hammarskjölds väg 38, 751 85 Uppsala, Sweden.
Vijay Kunadian, Translational and Clinical Research Institute, Faculty of Medical Sciences, Newcastle University and Cardiothoracic Centre, Freeman Hospital, Newcastle upon Tyne Hospitals NHS Foundation Trust, Newcastle upon Tyne, UK.
Stefan K James, Department of Medical Sciences, Cardiology, Uppsala University, Uppsala, Sweden; Uppsala Clinical Research Center, Uppsala University, Uppsala Science Park, Hubben, Dag Hammarskjölds väg 38, 751 85 Uppsala, Sweden.
Robert F Storey, Department of Infection, Immunity and Cardiovascular Disease, University of Sheffield, Sheffield, UK.
P Gabriel Steg, Université de Paris, Assistance Publique-Hôpitaux de Paris, Hôpital Bichat, French Alliance for Cardiovascular Trials, and INSERM U1148, Paris, France.
Hugo A Katus, Medizinishe Klinik, Universitätsklinikum Heidelberg, Heidelberg, Germany.
Robert A Harrington, Department of Medicine, Stanford University, Stanford, CA, USA; Cardiovascular Division, Department of Medicine, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA, USA.
C Michael Gibson, Department of Cardiology, Centre of Postgraduate Medical Education, Grochowski Hospital, Warsaw, Poland; Department of Medical Sciences, Clinical Chemistry, Uppsala University, Uppsala, Sweden.
Agneta Siegbahn, Uppsala Clinical Research Center, Uppsala University, Uppsala Science Park, Hubben, Dag Hammarskjölds väg 38, 751 85 Uppsala, Sweden.
Lars Wallentin, Department of Medical Sciences, Cardiology, Uppsala University, Uppsala, Sweden; Uppsala Clinical Research Center, Uppsala University, Uppsala Science Park, Hubben, Dag Hammarskjölds väg 38, 751 85 Uppsala, Sweden.
References
- 1. O’Gara PT, Kushner FG, Ascheim DD, Casey DE Jr, Chung MK, de Lemos JA, Ettinger SM, Fang JC, Fesmire FM, Franklin BA, Granger CB, Krumholz HM, Linderbaum JA, Morrow DA, Newby LK, Ornato JP, Ou N, Radford MJ, Tamis-Holland JE, Tommaso CL, Tracy CM, Woo YJ, Zhao DX, Anderson JL, Jacobs AK, Halperin JL, Albert NM, Brindis RG, Creager MA, DeMets D, Guyton RA, Hochman JS, Kovacs RJ, Kushner FG, Ohman EM, Stevenson WG, Yancy CW; American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines . 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology foundation/American Heart Association task force on practice guidelines. J Am Coll Cardiol 2013;61:e78–e140. [DOI] [PubMed] [Google Scholar]
- 2. Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, Bueno H, Caforio ALP, Crea F, Goudevenos JA, Halvorsen S, Hindricks G, Kastrati A, Lenzen MJ, Prescott E, Roffi M, Valgimigli M, Varenhorst C, Vranckx P, Widimský P; ESC Scientific Document Group . 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J 2018;39:119–177.28886621 [Google Scholar]
- 3. Amsterdam EA, Wenger NK, Brindis RG, Casey DE Jr, Ganiats TG, Holmes DR Jr, Jaffe AS, Jneid H, Kelly RF, Kontos MC, Levine GN, Liebson PR, Mukherjee D, Peterson ED, Sabatine MS, Smalling RW, Zieman SJ.. 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association task force on practice guidelines. J Am Coll Cardiol 2014;64:e139–e228. [DOI] [PubMed] [Google Scholar]
- 4. Collet J-P, Thiele H, Barbato E, Barthélémy O, Bauersachs J, Bhatt DL, Dendale P, Dorobantu M, Edvardsen T, Folliguet T, Gale CP, Gilard M, Jobs A, Jüni P, Lambrinou E, Lewis BS, Mehilli J, Meliga E, Merkely B, Mueller C, Roffi M, Rutten FH, Sibbing D, Siontis GCM; ESC Scientific Document Group . 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: The Task Force for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J 2021;42:1289–1367. [DOI] [PubMed] [Google Scholar]
- 5. Keeley EC, Boura JA, Grines CL.. Primary angioplasty versus intravenous thrombolytic therapy for acute myocardial infarction: a quantitative review of 23 randomised trials. Lancet 2003;361:13–20. [DOI] [PubMed] [Google Scholar]
- 6. Andersen HR, Nielsen TT, Rasmussen K, Thuesen L, Kelbaek H, Thayssen P, Abildgaard U, Pedersen F, Madsen JK, Grande P, Villadsen AB, Krusell LR, Haghfelt T, Lomholt P, Husted SE, Vigholt E, Kjaergard HK, Mortensen LS.. A comparison of coronary angioplasty with fibrinolytic therapy in acute myocardial infarction. N Engl J Med 2003;349:733–742. [DOI] [PubMed] [Google Scholar]
- 7. Mehta SR, Granger CB, Boden WE, Steg PG, Bassand J-P, Faxon DP, Afzal R, Chrolavicius S, Jolly SS, Widimsky P, Avezum A, Rupprecht H-J, Zhu J, Col J, Natarajan MK, Horsman C, Fox KAA, Yusuf S.. Early versus delayed invasive intervention in acute coronary syndromes. N Engl J Med 2009;360:2165–2175. [DOI] [PubMed] [Google Scholar]
- 8. Kofoed KF, Kelbæk H, Hansen PR, Torp-Pedersen C, Høfsten D, Kløvgaard L, Holmvang L, Helqvist S, Jørgensen E, Galatius S, Pedersen F, Bang L, Saunamaki K, Clemmensen P, Linde JJ, Heitmann M, Wendelboe Nielsen O, Raymond IE, Kristiansen OP, Svendsen IH, Bech J, Dominguez Vall-Lamora MH, Kragelund C, Hansen TF, Dahlgaard Hove J, Jørgensen T, Fornitz GG, Steffensen R, Jurlander B, Abdulla J, Lyngbæk S, Elming H, Therkelsen SK, Abildgaard U, Jensen JS, Gislason G, Køber LV, Engstrøm T.. Early versus standard care invasive examination and treatment of patients with non-ST-segment elevation acute coronary syndrome. Circulation 2018;138:2741–2750. [DOI] [PubMed] [Google Scholar]
- 9. The TIMI Study Group . The thrombolysis in myocardial infarction (TIMI) trial—phase I findings. N Engl J Med 1985;312:932–936. [DOI] [PubMed] [Google Scholar]
- 10. Gibson MC, Schömig A.. Coronary and myocardial angiography: angiographic assessment of both epicardial and myocardial perfusion. Circulation 2004;109:3096–3105. [DOI] [PubMed] [Google Scholar]
- 11. Gibson CM, Murphy SA, Morrow DA, Aroesty JM, Gibbons RJ, Gourlay SG, Barron HV, Giugliano RP, Antman EM, Braunwald E.. Angiographic perfusion score: an angiographic variable that integrates both epicardial and tissue level perfusion before and after facilitated percutaneous coronary intervention in acute myocardial infarction. Am Heart J 2004;148:336–340. [DOI] [PubMed] [Google Scholar]
- 12. Gibson CM, Cannon CP, Murphy SA, Marble SJ, Barron HV, Braunwald E.. Relationship of the TIMI myocardial perfusion grades, flow grades, frame count, and percutaneous coronary intervention to long-term outcomes after thrombolytic administration in acute myocardial infarction. Circulation 2002;105:1909–1913. [DOI] [PubMed] [Google Scholar]
- 13. Eggers KM, Lindahl B.. Prognostic biomarkers in acute coronary syndromes: risk stratification beyond cardiac troponins. Curr Cardiol Rep 2017;19:29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Wallentin L, Becker RC, Budaj A, Cannon CP, Emanuelsson H, Held C, Horrow J, Husted S, James S, Katus H, Mahaffey KW, Scirica BM, Skene A, Steg PG, Storey RF, Harrington RA.. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 2009;361:1045–1057. [DOI] [PubMed] [Google Scholar]
- 15. Kunadian V, James SK, Wojdyla DM, Zorkun C, Wu J, Storey RF, Steg PG, Katus H, Emanuelsson H, Horrow J, Maya J, Wallentin L, Harrington RA, Gibson CM.. Angiographic outcomes in the PLATO trial (Platelet Inhibition and Patient Outcomes). JACC Cardiovasc Interv 2013;6:671–683. [DOI] [PubMed] [Google Scholar]
- 16. James S, Akerblom A, Cannon CP, Emanuelsson H, Husted S, Katus H, Skene A, Steg PG, Storey RF, Harrington R, Becker R, Wallentin L.. Comparison of ticagrelor, the first reversible oral P2Y12 receptor antagonist, with clopidogrel in patients with acute coronary syndromes: rationale, design, and baseline characteristics of the PLATelet inhibition and patient Outcomes (PLATO) trial. Am Heart J 2009;157:599–605. [DOI] [PubMed] [Google Scholar]
- 17. Gibson CM, Cannon CP, Murphy SA, Ryan KA, Mesley R, Marble SJ, McCabe CH, Van de Werf F, Braunwald E.. Relationship of TIMI myocardial perfusion grade to mortality after administration of thrombolytic drugs. Circulation 2000;101:125–130. [DOI] [PubMed] [Google Scholar]
- 18. Lindholm D, James SK, Gabrysch K, Storey RF, Himmelmann A, Cannon CP, Mahaffey KW, Steg PG, Held C, Siegbahn A, Wallentin L.. Association of multiple biomarkers with risk of all-cause and cause-specific mortality after acute coronary syndromes: a secondary analysis of the PLATO biomarker study. JAMA Cardiol 2018;3:1160–1166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Lindholm D, Lindbäck J, Armstrong PW, Budaj A, Cannon CP, Granger CB, Hagström E, Held C, Koenig W, Östlund O, Stewart RAH, Soffer J, White HD, de Winter RJ, Steg PG, Siegbahn A, Kleber ME, Dressel A, Grammer TB, März W, Wallentin L.. Biomarker-based risk model to predict cardiovascular mortality in patients with stable coronary disease. J Am Coll Cardiol 2017;70:813–826. [DOI] [PubMed] [Google Scholar]
- 20. Held C, White HD, Stewart RAet al. Inflammatory biomarkers interleukin‐6 and C‐reactive protein and outcomes in stable coronary heart disease: experiences from the STABILITY (stabilization of atherosclerotic plaque by initiation of darapladib therapy) trial. J Am Heart Assoc 2019;6:e005077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, Kastelein JJP, Cornel JH, Pais P, Pella D, Genest J, Cifkova R, Lorenzatti A, Forster T, Kobalava Z, Vida-Simiti L, Flather M, Shimokawa H, Ogawa H, Dellborg M, Rossi PRF, Troquay RPT, Libby P, Glynn RJ; CANTOS Trial Group . Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med 2017;377:1119–1131. [DOI] [PubMed] [Google Scholar]
- 22. Nidorf SM, Fiolet ATL, Mosterd A, Eikelboom JW, Schut A, Opstal TSJ, The SHK, Xu X-F, Ireland MA, Lenderink T, Latchem D, Hoogslag P, Jerzewski A, Nierop P, Whelan A, Hendriks R, Swart H, Schaap J, Kuijper AFM, van Hessen MWJ, Saklani P, Tan I, Thompson AG, Morton A, Judkins C, Bax WA, Dirksen M, Alings M, Hankey GJ, Budgeon CA, Tijssen JGP, Cornel JH, Thompson PL.. Colchicine in patients with chronic coronary disease. N Engl J Med 2020;383:1838–1847. [DOI] [PubMed] [Google Scholar]
- 23. Thomas MR, James SK, Becker RC, Himmelmann A, Katus HA, Cannon CP, Steg PG, Siegbahn A, Lakic T, Storey RF, Wallentin L.. Prognostic impact of baseline inflammatory markers in patients with acute coronary syndromes treated with ticagrelor and clopidogrel. Eur Hear J Acute Cardiovasc Care 2021;10:153–163. [DOI] [PubMed] [Google Scholar]
- 24. Hagström E, James SK, Bertilsson M, Becker RC, Himmelmann A, Husted S, Katus HA, Steg PG, Storey RF, Siegbahn A, Wallentin L; PLATO Investigators . Growth differentiation factor-15 level predicts major bleeding and cardiovascular events in patients with acute coronary syndromes: results from the PLATO study. Eur Heart J 2016;37:1325–1333. [DOI] [PubMed] [Google Scholar]
- 25. Hagström E, Held C, Stewart RAH, Aylward PE, Budaj A, Cannon CP, Koenig W, Krug-Gourley S, Mohler ER, Steg PG, Tarka E, Östlund O, White HD, Siegbahn A, Wallentin L; STABILITY Investigators . Growth differentiation factor 15 predicts all-cause morbidity and mortality in stable coronary heart disease. Clin Chem 2017;63:325–333. [DOI] [PubMed] [Google Scholar]
- 26. Lindholm D, Hagström E, James SK, Becker RC, Cannon CP, Himmelmann A, Katus HA, Maurer G, López-Sendón JL, Steg PG, Storey RF, Siegbahn A, Wallentin L.. Growth differentiation factor 15 at 1 month after an acute coronary syndrome is associated with increased risk of major bleeding. J Am Heart Assoc 2020;6:e005580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Vogt A, von Essen R, Tebbe U, Feuerer W, Appel K-F, Neuhaus K-L.. Impact of early perfusion status of the infarct-related artery on short-term mortality after thrombolysis for acute myocardial infarction: retrospective analysis of four german multicenter studies. J Am Coll Cardiol 1993;21:1391–1395. [DOI] [PubMed] [Google Scholar]
- 28. Anderson JL, Karagounis LA, Becker LC, Sorensen SG, Menlove RL.. TIMI perfusion grade 3 but not grade 2 results in improved outcome after thrombolysis for myocardial infarction. Ventriculographic, enzymatic, and electrocardiographic evidence from the TEAM-3 Study. Circulation 1993;87:1829–1839. [DOI] [PubMed] [Google Scholar]
- 29. Bailleul C, Aissaoui N, Cayla G, Dillinger J-G, Jouve B, Schiele F, Ferrières J, Simon T, Danchin N, Puymirat E; FAST-MI investigators . Prognostic impact of prepercutaneous coronary intervention TIMI flow in patients with ST-segment and non-ST-segment elevation myocardial infarction: results from the FAST-MI 2010 registry. Arch Cardiovasc Dis 2018;111:101–108. [DOI] [PubMed] [Google Scholar]
- 30. van Diepen S, Alemayehu WG, Zheng Y, Theroux P, Newby LK, Mahaffey KW, Granger CB, Armstrong PW.. Temporal changes in biomarkers and their relationships to reperfusion and to clinical outcomes among patients with ST segment elevation myocardial infarction. J Thromb Thrombolysis 2016;42:376–385. [DOI] [PubMed] [Google Scholar]
- 31. Montalescot G, Barragan P, Wittenberg O, Ecollan P, Elhadad S, Villain P, Boulenc JM, Morice MC, Maillard L, Pansiéri M, Choussat R, Pinton P; ADMIRAL Investigators. Abciximab before Direct Angioplasty and Stenting in Myocardial Infarction Regarding Acute and Long-Term Follow-up . Platelet glycoprotein IIb/IIIa inhibition with coronary stenting for acute myocardial infarction. N Engl J Med 2001;344:1895–1903. [DOI] [PubMed] [Google Scholar]
- 32. Van't Hof AWJ, Ten Berg J, Heestermans T, Dill T, Funck RC, van Werkum W, Dambrink J-HE, Suryapranata H, van Houwelingen G, Ottervanger JP, Stella P, Giannitsis E, Hamm C; Ongoing Tirofiban In Myocardial infarction Evaluation (On-TIME) 2 study group . Prehospital initiation of tirofiban in patients with ST-elevation myocardial infarction undergoing primary angioplasty (On-TIME 2): a multicentre, double-blind, randomised controlled trial. Lancet 2008;372:537–546. [DOI] [PubMed] [Google Scholar]
- 33. Montalescot G, van't Hof AW, Lapostolle F, Silvain J, Lassen JF, Bolognese L, Cantor WJ, Cequier A, Chettibi M, Goodman SG, Hammett CJ, Huber K, Janzon M, Merkely B, Storey RF, Zeymer U, Stibbe O, Ecollan P, Heutz WMJM, Swahn E, Collet J-P, Willems FF, Baradat C, Licour M, Tsatsaris A, Vicaut E, Hamm CW; ATLANTIC Investigators . Prehospital ticagrelor in ST-segment elevation myocardial infarction. N Engl J Med 2014;371:1016–1027. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The sponsors of the PLATO trial (AstraZeneca, Cambridge, UK) are committed to responsible sharing of clinical study reports, related clinical documents, and patient level clinical study data. Researchers are invited to submit inquiries online. To submit inquiries, please visit https://astrazenecagrouptrials.pharmacm.com. All data relevant to the study are included in the article or uploaded as supplementary information.