Abstract
Background
Antibodies have proven to be remarkably successful for biomedical applications. They play important roles in epidemiology and medicine from diagnostics of diseases to therapeutics, treating diseases from incessant chronic diseases such as rheumatology to pandemic outbreaks. With no end in sight for the demand for antibody products, optimizations and new techniques must be expanded to accommodate this.
Methods and Results
This review discusses optimizations and techniques for antibody production through choice of discovery platforms, expression systems, cell culture mediums, and other strategies to increase expression yield. Each system has its own merits and demerits, and the strategy chosen is critical in addressing various biological aspects.
Conclusions
There is still insufficient evidence to validate the efficacy of some of these techniques, and further research is needed to consolidate these industrial production systems. There is no doubt that more strategies, systems, and pipelines will contribute to enhance biopharmaceutical production.
Keywords: Antibody, Antibody diagnostic, Antibody therapeutic
Introduction
Currently, immunotherapeutics, especially antibody-based molecules, hold much promise for treating diseases. Compared to conventional chemotherapeutics or therapeutics of other modalities, antibodies typically show a reduced toxicity profile [1, 2]. These biologics have come a long way from their first FDA approval in 1986 to the 100th monoclonal antibody (mAb) approved for therapy in 2021 [3]. With significant revenue growth worldwide, as well as an increase in regulatory approvals to market mAbs, there is an increased emphasis in the biopharmaceutical market on R&D, as well as in developing and advancing their pipelines for various therapeutic areas [4].
With the latest technology, production of antibodies can now be done with ease and optimisation allows efficient high yield antibodies. From the starting point, choice of antibody discovery platforms plays a significant role in determining the output. Antibodies can be engineered to be highly reproducible by employing gene cloning and efficient platform-based strategies by manufacturing flexible gene of interest (GOI) from choice antibody to be cloned into eukaryotic expression vectors, which will then be transfected into various cell lines [5].
The choice of host cell plays an important role in generating quality well-characterised proteins with high sensitivity, specificity, and reproducibility. Host cells, including bacterial, yeast, insect, and mammalian are used with mammalian cell lines being the preferred mode of production. Chinese Hamster Ovary (CHO) is especially favoured for their production efficacy and reduced risk of human viral infections besides their phenotypic adaptability and plasticity [6, 7]. Usage of mammalian cell lines minimizes the risk of rejection by human bodies in therapeutic use as they are capable of undertaking appropriate protein folding, post-translational modifications, and glycosylation in a manner similar to humans and thus, to further reduce rejection risk, human cell lines such as HEK293 can be used as for human genes, human cell lines provide an environment similar to their native environment suggesting higher compatibility [8, 9].
Nonetheless, generation of recombinant antibodies using mammalian cell lines is time consuming, labour intensive, and expensive, resulting in the need for optimisation to produce antibodies in a quick and easy manner [10]. In bioproduction, cell culture medium remains a major player in protein production as it promotes cell growth and productivity, improves efficiency and quality of generated proteins, besides reducing batch-to-batch variation [11, 12]. The medium has a significant effect on research outcomes, biopharmaceutical production rates, and the outcome of assisted reproductive technology treatment, making the selection of a fitting medium principal in antibody production [13, 14].
In antibody production via transgenic cell lines, these proteins can be expressed transiently or stably. Transient expression has a relatively short period for protein harvesting but generates a lower yield as while they can express the foreign gene, it is not integrated into the genome and therefore is not replicated and lost through time [15]. Therefore, transient expression systems are only for short-term usage [16]. Besides, proteins expressed by transient cell lines are inconsistent with changes in glycan contents, and thus in the manufacturability, affinity, and efficacy [9, 17].
Stable expression is time consuming and labour intensive, but this approach enables protein expression in a stable, consistent, infinite and large amount, as well as with regulatory familiarity as the GOI is stably integrated into the host cell lines’ genome [18]. Stable cell lines can cater for the increasing demand of antibody-based products by being able to mass produce antibodies with stable formulations in a cost-effective manner. As a result, stable expression is established as the standard practise for continued genetic research or industrial manufacturing of highly specialised proteins.
However, the establishment of a stable cell line has its own hassles as it can bring about clonal variation from GOI being randomly incorporated into the genome [19]. This reduces the predictive value, complicates procedures and increases the cost of therapeutic glycoprotein production, besides making it tougher to distinguish between wild-type and mutant reporters [20]. There are numerous approaches to generate stable expressing cell lines while eliminating clonal variance. They include the use of site-specific recombinases, which comprises of Cre and Flp, as well as homologous recombination, which for example comprises of the CRISPR/Cas9 system. Although the latest technologies include combining a few of the prior mentioned systems to provide a system without any limitations that come with exploiting just a system. For instance, the novel “Cas-Pi” system, which means cascaded precise integration and the usage of hybrid enzymes such as Flp-TAL recombinases [21, 22].
Furthermore, there are strategies to be adopted to increase antibody yield. The expression vector can be designed to increase expression with high expressing promoters such as pCMV or pCNA [23]. Cell culture temperature, which is slightly hypothermic was proven to cause a rise in transgene expression [15]. Furthermore, hyperosmolality which can be achieved with either continued supplementation of feeds and build-up of metabolic waste or salt supplementation can increase gene expression levels [24]. There is an increase in specific volumetric productivity when the cells are at their largest [25]. Lastly, downstream processing is the last step in antibody production and choice of purification system determines the quality of the final antibody product.
All in all, this review highlights the importance of antibodies and several strategies to increase mass production in a time efficient and cost-effective manner. However, there is still limited data to verify the effectiveness of the aforementioned strategies and more studies can be done to consolidate these systems for industrial production. It is undeniable that there will be more strategies, systems, and pipelines expanded to further improve biopharmaceutical production.
Natural production of antibodies
By nature, antibodies are exclusive products of B cells in the presence of antigens. Each individual B cell can generate antibodies with unique amino acid sequence and antigen binding site. B cells first form antibodies as membrane-bound cell surface receptors for antigens, but when antigens and helper T cells activate naïve or memory B cells, these lymphocytes differentiate to produce antibodies as soluble secreted molecules [26].
Before antibodies are employed for use, the generated proteins must pass a series of quality checks in the endoplasmic reticulum. The endoplasmic reticulum consists of several resident cellular proteins with various functions of calcium binding and aggregation prevention of polypeptide folding intermediates amongst many others [27]. Of all of its cellular proteins, the most notable is the immunoglobulin binding protein (BiP), which is expressed in the presence of unfolded or unassembled proteins. BiP associates with the CH1 domain to direct assembly and transport of proteins, as without it, inappropriately folded or assembled proteins will be secreted [28]. It is especially important as the secretion of inappropriately folded or assembled proteins can lead to deletions to the immune response.
In the process of antibody quality check, the heavy chain is first tested for its ability to fold and assemble correctly with the light chain and once the heavy chain is certified functional, light chain rearrangements will proceed [28]. The quality check ensures that only proteins which are appropriately folded and assembled are secreted.
General overview of antibody biopharmaceuticals
In this era, where there are gradual advancements in knowledge of diseases and its formation in cellular and molecular level, as well as unrealised medical needs of chronic diseases, the growth and demand for global biopharmaceuticals and therapeutics are soaring [29]. This is principally for large supplies of cost-effective products for cost-sensitive indications such as rheumatology and asthma as they have the additional benefits of being highly specific, offering lesser side effects, and a potential cure [5].
To date, protein-based biological pharmaceuticals take the lead in biologics. These macromolecules revolutionised biotechnology while laying the foundation in molecular medicine. They are employed to treat, diagnose, prevent, cure diseases and various medical disorders. Their formats include vaccines, cellular or gene therapy drugs, as well as recombinant proteins [29]. They are valuable and essential biochemical tools for research, diagnostic and therapeutic treatment of diseases caused by pathogens and toxins, and can be produced by the human body or synthetically.
Antibodies in diagnostics
These molecules play a vital role as diagnostic tools where they ensure efficient management of diseases. Faced with an epidemic, a timely and thorough diagnosis is key in the wait of development and distribution of vaccinations or therapeutics [30]. By specific detection of proteins or other molecules, antibodies aid in recognition of infections and allergies besides factoring in the measurement of hormones and other biological markers. MAbs are indispensable in the diagnosis of pathologies and fundamental laboratory investigation as these proteins have the edge of specificity to be able to bind to specific antigenic epitopes, which facilitate prompt assessment of molecular phenotype of various tissues [31].
At present, mAbs are extensively used in technologies such as immunohistochemistry and flow cytometry. As antibodies are immune proteins, marking the evolution of the host immune system response to an infection, they are measured in a sensitive and specific way to provide a record of recent and previous infections [32]. It is superior to pathogen detection, which is only done transiently whereas antibody markers are more durable [32]. Having various advantages, including their drug safety profiles, technological advancements, development and market success are just some grounds behind its acceptance in diagnostic use [33]. Some of the latest diagnostic assays are for the novel coronavirus disease 2019 (COVID-19). These tests can detect specific biomarkers, which in this case are immunoglobulins IgM and IgG which are synthesised by the host B cells in response to infection. Some examples include chemiluminescent IA, manual ELISA, as well as rapid lateral flow IA [34].
However, in serological tests, the usage of antibodies can be traded with antibody fragments. synthesised human single-chain variable fragments (scFv) present numerous pros compared to their traditional antibody counterparts, such as reduced molecular mass, as well as enhanced thermal stability, solubility, and dispersion, besides being an antigen substitute when antigen collection is unavailable [35]. Furthermore, an antigen binding fragment (Fab) is another alternative to scFv. A Fab synthetic phage display library can be constructed as a promising tool for diagnosis to detect a selection of mAbs acting on different epitopes [36]. Alternative antibody formats do not rely on mammalian cell lines for their production, unlike traditional antibody formats but can be produced in bacterial cell lines, making it much more cost and time effective [36].
Antibodies in therapeutics
Furthermore, the pioneering of recombinant mAbs, which are synthetic mAbs with identified sequences has brought new light to therapeutics [37]. As recombinant antibodies are specified at sequence level, they offer no batch-to-batch variation and can be produced in commercially available expression systems where mammalian cell lines are commonly used [38]. Live cells are another common mode of protein production [39].
When designing recombinant antibodies, many molecular formats are considered. Human immunoglobulin isotypes consist of IgA, which subtypes to IgA1 and IgA2, IgD, IgE, IgG that subtypes to IgG1, IgG2, IgG3, and IgG4, as well as IgM. IgG is the leading molecular format used in today’s antibody drugs as it neutralizes infectious mediums and engage immune cells by activating the complement system [37, 40]. In preclinical stages, there are more non-IgG formats used although IgG is still the most dominant despite this, interest in testing alternative CH variants as therapeutic antibodies is growing [41]. This is largely contributed to the fact that the effector functions of antibodies are regulated by their isotypes. IgM is more pro-inflammatory since it stimulates complement more effectively [42]. IgM also exhibits specialised immune functions besides possessing higher avidity and steric hindrance, which neutralizes viruses as well as serving as the primary antibody defence against new antigens [37, 43, 44]. Furthermore, early clinical studies have introduced the potential of IgA isotype antibodies as anti-cancer therapeutics as it can reduce tumours [40]. Additionally, IgE antibody molecules, despite being the least abundant CH variant, can utilize FcRs from monocytes and macrophages to activate different effector cell population which defines its role in allergy and parasitic infections [45]. Then, there is the IgD CH variant where it’s function is obscure besides being the least characterised but is reported to induce the release of immunoactivity, pro-inflammatory, and antimicrobial mediators [37, 46].
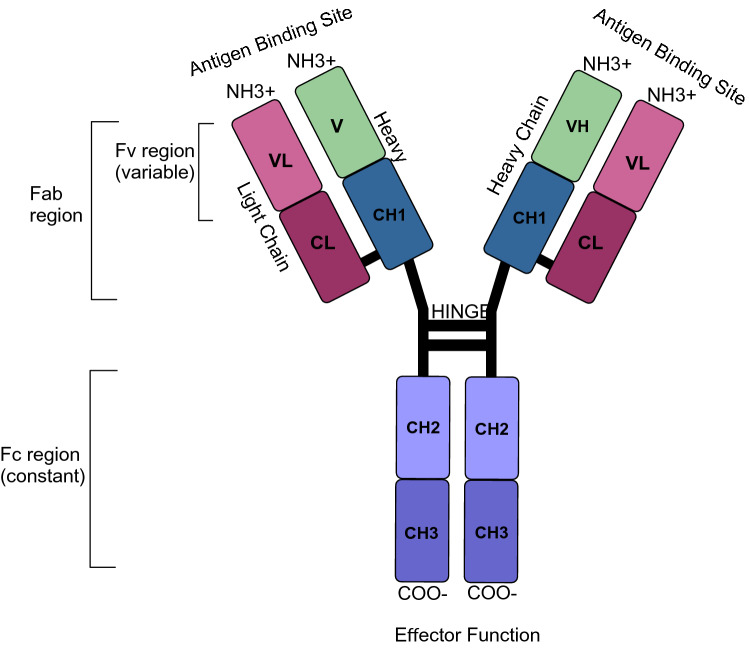
Lastly, there is IgG which accounts for most of the immunoglobulin in human sera and its subclasses influence immune responses by interacting with various FcRs [37]. This immunoglobulin possesses a well-defined biochemical structure. The schematic structure of an IgG monoclonal antibody is shown in Fig. 1 and as illustrated, it consists of constant and variable regions where the heavy chain section consists of CH1, CH2, CH3 and VH while the light chain consists of CL and VL. IgG’s subclasses are highly conserved, while possessing differing constant regions. This effect the effector function, giving each subclass unique ones influencing half-life, placental transport, antigen binding, immune complex formation, complement activation, and triggering of effector cells IgG2 provides defence against bacterial pathogens as a decreased level increases susceptibility to bacterial infections, IgG3 induces inflammation but its short half-life prevents excessive inflammatory response while refraining its usage in therapeutics, and IgG4 is induced by repeated, extensive antigen exposure in non-infectious setting [47, 48].]. Lastly, the most dominant human subtype in blood, IgG1, which is induced by antibody responses to soluble protein antigens and thus, a reduced level of IgG1 results in a decreased total IgG level, bringing about recurrent infections [47]. This isotype has mediating functions such as complement activation or deposition (CDC), which lyses virions or infected cells and antibody-dependant cell-mediated cytotoxicity (ADCC), contributing to its popularity in cancer treatments [48, 49]. All of the above-mentioned reasons appoint IgG1 as the default model for therapeutic monoclonal antibodies [48].
Fig. 1.
Schematic structure of a human IgG1 antibody. IgG consists of two heavy and two light chains. The variable domains are variable light (VL) and variable heavy (VH), which forms the antigen binding site. The constant domains are CL (constant light) and CH1–3 (constant heavy). IgG can be furthermore divided into Fab (fragment antigen binding) which consists of Fv (fragment variable) and Fc (fragment crystallizable) which induce effector functions
In therapeutics, Fc fusion proteins are commonly used as well. These proteins are created by fusing the antibody Fc region with a fusion partner, which may be in form of receptor’s extracellular domains, soluble cytokines, ligands, enzymes, engineered domains, or peptides [50]. The Fc region is dimeric and IgG is the common format used for this bioengineered protein due to its effector function and increased plasma half-life [51]. The crystal structure of the IgG1 Fc region is shown in Fig. 2. The Fc region of this protein interacts with the FcRn receptor on endothelial cells to prevent intracellular degradation and extend its half-life [52].
Fig. 2.
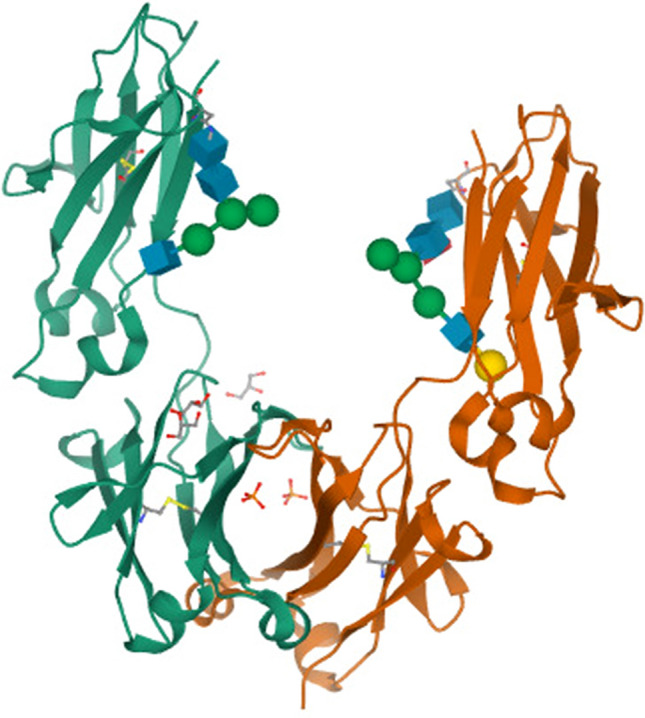
Crystal structure of human IgG1-Fc. Image from the RCSB PDB (rcsb.org) of PBD ID 5JII [53]
Another common format for therapeutics is bispecific antibodies (BsAbs), which have the advantage over traditional mAbs that are monospecific as they can bind two distinct epitopes or antigens in a single molecule. Thus, bsAbs has increased binding specificity, novel mechanisms of action, and higher clinical efficacy [54]. The two most popular formats of bsAbs are non-IgG-like or scFv-based and IgG-like, which is the full-length IgG-like asymmetric antibody [55]. The IgG-like formats have improved solubility, stability, serum half-life, and affinity, contributing to increased biological activity, while non-IgG-like formats have the advantage of productivity ease and low immunogenicity [56]. The unique format of BsAbs increases its mechanisms of action to enhance its success as a therapeutic, and its functionality can be modified to modulate its immunogenicity, effector functions, and half-life [57]. Although bsAbs comes with its own difficulties, particularly in development of correct matching of light (VL) and heavy (VH) chains, compromising its quantity, quality, and stability [55, 56].
Therapeutic antibodies are classified by their functionality, binding activity and affinity to their target, molecular characteristics as well as effector function. Effector function is critical for their efficacy by contributing to their mechanism of action (MoA), such as specific binding of soluble antigen or a target-bound antigen and specific agonizing or antagonizing with receptors for modulating immune responses [39, 48]. The effector functions of antibodies are reliant on the interaction between Fc domains with FcγRs and complement components and they include ADCC and ADCP via the recruitment of FcγRs63 and CDC via the recruitment of C1q [1].
These immunotherapeutic are extremely valuable because of their rapid discovery process, incredible specificity, and low toxicity and their high specificity, which stems from their unique maturation process [58]. With antibody engineering, recombinant antibodies can be optimised at will and with ease of production as GOI is easily amenable to efficient, reproducible platform-based approaches, making recombinant antibodies a marketable biological product [5, 59]. These technologies massively boost process yields and minimise production costs. Besides, antibodies are “safer” compared to other therapeutic products in human clinical trials as antibodies tend to be well-tolerated and highly specific [5]. Therapeutic antibodies can neutralize toxins or cytokines, block receptors, bind to cells and modulate the host immune system or any combination [60]. In terms of cancer treatments, more significance is placed on single-domain antibodies (sdAbs) or nanobodies as they possess high-binding attributes, production ease, reduced immunogenicity while being highly stable [61]. In the FDA approval rates of drugs, antibody products take the lead by accounting for a fifth of approvals annually [3, 5].
With their innate advantage of possessing extreme specificity and regulatable precision, mAb therapeutics are one of the most prominent and rapidly growing clinically licensed drugs existing in this timeline where they are so commonly used that four of the top five therapeutic proteins are mAbs besides accounting for nearly half of all therapeutic protein sales [41, 62]. Antibody-based immunotherapies are well accepted in oncology, autoimmunity, chronic inflammatory diseases and in the near future infectious diseases as well [17]. MAbs can locate and attack viral proteins where they are vulnerable, resulting in mAbs being the preferred medium for treating viral diseases [17]. Furthermore, their high specificity aids in understanding interactions between host and virus besides mechanisms of viral pathogenesis which in turn aids discovery of cures to viral infections and diseases [58]. These proteins are feasible therapeutic modality for infectious diseases such as Ebola, human cytomegalovirus and many more [58]. However, the overabundance of approved antibody drugs created a large overlap of therapeutic targets where dissimilar drugs occupy identical routes, for instance a receptor or its reciprocal ligand [41]. Furthermore, the approval of too many antibody drugs caused a widespread overlap in approved indications for antibodies [41].
Some of the most recently approved therapies include Isatuximab and Belantamab mafodotin for multiple myeloma, Atoltivimab, Maftivimab, and Odesivimab for Zaire Ebolavirus, Sacituzumab govitecan and Margetuximab for breast cancer, as well as the cocktail REGEN-COV of casirivimab with imdevimab and bamlanivimab with etesevimab for COVID-19 [63, 64]. Different monoclonal antibody therapeutics tend to be administrated together to reduce risk of treatment failure as they can work broadly against viral variants which may be resistant to an antibody therapeutic as viruses can accumulate mutations. Presenting an alternative to vaccines, therapeutics antibodies offer protection against the virus for those who respond poorly to vaccines, for instance, the aged and the immunocompromised, as well as those unwilling to be vaccinated for religious reasons or otherwise [63].
However, for all its potential, antibody therapeutics has its weaknesses such as deficient pharmacokinetics, and tissue attainability, as well as a diminished relationship with the immune system [59]. Antibody-based vaccines and therapeutics can cause antibody-dependent enhancement (ADE) which may aggravate the severity of the disease [65]. ADE from antibody-based interventions has been recorded in human immunodeficiency virus (HIV), Ebola, dengue, influenza and flaviviruses as well as human respiratory virus infections including SARS, MERS, and measles [42, 65]. The threat of ADE can be reduced by utilising higher dosage of neutralizing antibodies for reduced concentrations increases the risk of ADE [65]. ADE can also be diminished using mAbs instead of vaccine-induced antibodies as mAbs can be engineered with molecular precision [42]. Additionally, the generation of mAb therapies are rather costly, and necessitates certain skills unfamiliar to generic-drug makers, making it inaccessible to most of the population [66].
All in all, to meet the growing clinical demand, process intensification focussing on the development of novel systems and techniques as well as a solid production platform must be considered in order to achieve the objective of enhanced production while reducing power consumption, waste creation, and curtailing manufacturing cost in the process to achieve more effective, cheaper, and more sustainable technologies [29, 62]. This can be done through optimisation of biopharmaceutical manufacturing process such as through protein expression systems, and mediums as well as continuous production instead of batch production. Technological innovation is imperative in the passage to discover cheaper, easier methods to generate large number of antibodies [66].
Antibody discovery platforms
In antibody discovery, some of the most common approaches used are the conventional animal immunisation and surface display technologies. Animal immunisation can be performed on either wild-type or transgenic animals and while it is effective, the antigens can be toxic or well conserved to the animal species, inhibiting neutralizing antibodies from being generated [67]. Then there are surface display technologies, an in vitro method able to overcome immune tolerance limitations faced by animal immunisation besides being able to regulate selection and screening conditions, giving rise to selecting specificities.
Surface display technologies can be divided into prokaryotic and eukaryotic systems and they are categorised as phage, bacterial, yeast, mammalian, cell, ribosomal, mRNA, and DNA amongst others. The most established surface display technology is the phage display, which is a phagemid system with advantages of easy library generation, large diversities, while being able to obtain variants of high specificity and affinity. Phage display can display full length antibodies although antibody fragments are more commonly applied with robust reliable processing by filamentous bacteriophages such as M13 in Escherichia coli [68, 69]. However, phage display has its downsides where its antibodies are not glycosylated and thus impacts its expression in mammalian cells in terms of binding, biodistribution, or pharmacokinetics [70]. Then there is bacterial display which is popular for its attributes of fast growth, employment ease, cost effectiveness, its ability for cell sorting in real time multiparameter fluorescence analysis, and its large library sizes but bacterial display is unable to produce human-like post-translational modifications [68]. Yeast surface display utilises Saccharomyces cerevisiae to express antibodies in both full length and antibody fragments although the latter is more popular in the forms of scFvs or Fabs [67]. This system has the added benefits of being fluorescence-activated cell sorting compatible allowing analysis of library candidates online and in real time while excising control on selection quantifiably, and its advanced protein folding and quality control machinery allows more complex molecules to be generated in comparison in prokaryotic systems besides allowing antigen-specific antibodies to be isolated from universal libraries, although this system has a downside of confined library sizes [68]. Mammalian display can isolate antibodies with prescribed specificities and affinities besides producing antibodies with appropriate folding, human-like glycosylation, and post-translation modifications [67]. The aforementioned eukaryotic platforms of yeast and mammalian can isolate high affinity antibodies with definitive biological characteristics and this is able to generate glycosylated antibodies allowing for proper protein expression in mammalian cells [70]. Although compared to bacteria or yeast systems, the library size from mammalian display is more limited [68]. Furthermore, there is the cell free ribosomal display and as it is without a host, the library size is not restricted by the host’s transformation efficiency, thus, allowing generation and screening of larger diversity [68]. Additionally, there is B cell immunisation, which is the most natural genotype phenotype coupling and thus not exactly considered an engineered platform technology. It can retain the natural heavy and light chain combination, which is advantageous as during random assembly of library generation, there may be specificity and affinity loss as well as induce self-reactive molecules [68].
Antibody expression systems
Mammalian and non-mammalian cell lines
Growing numbers of revolutionary biological products and biosimilars have thus intensified the demand for high productivity host cell lines as the choice of host cells is the most notable and influential method to increase volumetric cell yield while minimising production cost [71]. However, while achieving high product yield at low cost is much coveted, it is important to consider the structural quality of the produced molecules as the selection of expression system can heavily influence the type and degree of the product’s structure, particularly glycosylation [29]. Some main traits of a good host for therapeutic production are rapid development, known organelles and functions for protein processing and secretion, as well as possessing strain engineering toolkits to integrate recombinant protein into host genome and engineer post-translational modifications [72]. Additionally, intrinsic qualities such as proven specific productivities, lack of co-secreted proteases, proteome’s amount of host cell proteins, process flexibility, and absence of mammalian viral infectivity, in addition to other factors including usage of cheaper, less-refined feeds, along with robustness to shear stress, pH, and temperature can direct the selection of a prospective host as well [71]. Recombinant proteins such as antibodies can be expressed using various platforms and there have been multiple expression systems identified.
The most common strategy for producing recombinant proteins is using bacteria, typically Escherichia coli but, the foreign protein being toxic to the host, besides, protein folding machinery differences, and the absence of post-translational modifications in bacteria makes them inapt hosts for expression of functional eukaryotic proteins [8]. Yeast, insect, and plant cell-based expression systems are applicable as well but unfortunately, they form glycan structures, unlike a human’s which carries the risk of undesired immunogenicity for therapeutics [29]. Additionally, the development of the aforementioned systems are fragmented, making it challenging to compare them to mammalian cell lines for potential benefits [71].
As a result, the primary framework for generation for therapeutic recombinant proteins is utilising mammalian cell lines as for ectopically expressed mammalian genes, they are the nearest likeness to the native environment and in this way have an inherent capability to create functional mammalian proteins [8]. Therein, mammalian cells are the largest contributors to recombinant proteins on the market at about 70% [73]. They can perform the proper and necessary folding, post-translational modifications and glycosylation in patterns similar to those in humans which reduces the risk of rejection besides being able adapt to large-scale production systems and produce large volumes of therapeutic antibodies [74, 75]. Glycosylation in antibodies is an important factor to note as it has incredible diversity and complexity of which antibody structure and function is crucially dependent on. Glycosylation patterns can determine structure and effector functions of antibodies including ADCC, CDC and its pharmacokinetic/pharmacodynamics properties [4]. Despite that, recombinant proteins expressed in mammalian cell lines can encounter complications in expression yields, time required to produce over-expressing lines, along with increased cost [8].
The most popular cell line used in industrial therapeutic production is CHO cell lines. CHO cell lines can expand rapidly to large densities in bioreactors, generate high quantities of recombinant protein, increased resistance to human virus transmissions, and generate human compatible post-translational modifications, besides possessing a genome with considerable plasticity which increases its flexibility to being genetically modified and manufacturing process scales [7]. Despite these many advantages, their characteristics of being genomically unstable and heterogeneous results in regular genotypic variation and phenotypic instability, which brings about cell line instability and therefore, concerns about the process reproducibility and consistency [6]. This issue generates genomic instability over time, causing a drop in protein production and thus fabricates titer and product quality instability [7]. Expression of mAbs from mouse and hamster-derived cell lines has its downsides as nonhuman cell lines express proteins with glycan structures which differ substantially from humans, and they are N-glycolylneuraminic acid and galactose-alpha 1,3-galactose group which elicits immunogenic responses in human cells [76]. Furthermore, the α-1,3-galactose linkage induce anaphylaxis reactions at specific levels [77]. These cell lines secrete fucosylated IgG-G0 glycoforms which is able to activate complement through MBL binding when clustered [78]. It has been well identified that nonhuman glycan structures are produced by rodent cell lines [79]. Sialic acid production differs as well. In human cells, the formation of Neu5Gc is inhibited, but it is identified in murine derived proteins which triggers antibody production to contribute to serum sickness in humans and many other pathophysiological implications [80, 81]. Additionally, murine derived proteins may consist of xenoantigenic gal-a-gal linkages which can cause hypersensitivity reaction and as a result increases risk of murine derived antibody-based therapeutics [82]. Consequently, product quality, chiefly glycan structures of murine derived biologics must be screened but this result in acceptable proteins being discarded from these concerns.
To circumvent immunogenicity, human cell lines can be exploited. Upsides of employing human cell lines are that they form immunogenic glycans, but the downsides are that they are vulnerable to human viral infection. Thus, viral inactivation on human cell lines is essential and HEK293 cell line which stands for Human Embryonic Kidney 293 is a viral DNA transfected human cell line widely used for protein biologics production [29]. It is the leading human cell line platform for protein expression with diverse variants although its efficiency is restricted by low transgene expression levels in these cells [76].
Both CHO and HEK cell lines are extremely stable, amenable to genetic manipulations, and have rapid growth and proliferation rates, indicating a high protein synthesis ability [10].
Cell culture medium
In another note, although choosing high expressing host cell clone is vital in bioproduction, developing cell culture medium is critical for cost-effectively promoting cell growth and productivity, as well as for reducing batch-to-batch variance during production [12]. Furthermore, to improve cell lines and cell culture processes with high efficiency and quality, it's crucial to use the right media [11]. Variations in cell culture media can influence therapeutic behaviour of the produced antibodies through glycosylation or otherwise [4]. The importance of cell culture medium stems from the fact that it is able to promote cell viability and proliferation along with cellular function which indicates that medium nature has a direct impact on research findings, biopharmaceutical output rates, and assisted reproductive technology treatment outcome [13]. As a result, the selection of an appropriate medium is critical.
Synthetic media are currently divided into various categories depending on the form of ingredients used, such as serum-containing media, serum free media (SFM), protein-free media, and chemically defined media. Serum-containing media consist of various serum-derived compounds, thus obscuring the medium makeup. Thereby, causing proportions to vary from batch-to-batch and because of this, the culture findings are less predictable and there is a threat of contamination whether in form of bacteria, viruses or prions which also impedes further optimisation and standardisation of systems [83]. This unfortunately limits its use for therapeutic protein production as serum content increases the risk of contamination such as pathogen transmission and additionally, its high protein content can interfere with product purification during the downstream process [11, 15]. On the bright side, this media is customizable to increase cell proliferation for a broad variety of mammalian cell types since serum contains a large number of active substances required for animal cell survival and development [84].
Serum, the supernatant of clotted blood, is an integral part of mammalian cell culture as it is a source of numerous compounds, reduces shear stress, improves the medium’s pH-buffering capacity and modify the culture substratum to allow proliferation of adherent cells [13, 15]. There are various serums to date, with some examples such as fetal bovine serum (FBS), calf serum (CF), and horse serum. FBS is the go-to for most applications as it is abundant in growth factors and lacking in γ‐globulins that stimulate cell proliferation but comparatively, CS has a reduced ability to promote cell growth and there is a high degree of homogeneity between lots in horse serums [13]. FBS owns its popularity to its ability to promote cell proliferation, metabolism, and differentiation, support cellular functions, supply necessary molecules, buffer pH, and inhibit protease of which their combined role is to promote cell development [85]. However, in terms of biosafety, FBS, like most other serums has unknown biochemical makeup, varying between lots, hence hindering culture medium standardisation. An alternative is knockout serum replacement, with a specified composition of organic components, trace elements, insulin, transferrin, and albumin [86].
Serum-free media (SFM), on the other hand, have a standardised formulation, leading to high reliability of results and the ability to evaluate the cultivation process. The development of conventional SFM to protein-free and chemically defined medium stems from increasingly defined components and nutrients necessary to improve cell proliferation [85, 87]. Subgroups of SFM, protein-free media comprises of zero protein whatsoever and chemically defined media which contains only known ingredients. These factors improve stability and reproducibility, easing cellular secretion detection while reducing microbial contamination. The general makeup of this media are amino acids, vitamins, glucose, inorganic salts, anti-shear protectants and nuclei acids with additions of supplements such as chemical components and growth factors [87].
Component in cell culture media is vital for its unique functions [87–89]. Energy sources, i.e. glucose plays a vital role as glycosylation precursors and thus can affect glycosylation profiles of the produced mAbs. Amino acids serve as important energy and nitrogen sources. Vitamins are essential components of SFM because they help maintain cell growth and regulate and monitor cell metabolism. Lipids are essential components of cell membrane structure that plays a role in cell signalling, serves as energy storage in cells as well as increase protein expression and glycosylation. Trace elements regulate cell metabolism and enzyme activity. Lastly, inorganic salts modulate osmotic pressure during cell culture phase besides regulating the cells’ metabolism.
The design of culture media and supplements influences mAb production as it can improve bioprocesses where certain pathways are favoured and lipid limitations are eradicated [90]. However, the downsides to serum free media are weaker growth performance when compared to their counterparts besides the difficulty of design as to date, there are very limited cell lines blessed with serum free media [84].
Transient and stable expression
The procedure of producing a mammalian recombinant protein expressing system starts with transfection of recombinant gene vector into cells and followed by selection using selection markers to select cells with successful plasmid integration to generate clones [29]. Antibodies can be expressed in mammalian cell lines transiently or stably by integrating expression constructs into the host’s genome. Choice of expression systems can be done solely based on quantity of protein required and convenience as this does not affect their biochemical characteristics as reported in these studies where there is no structural discrepancies between proteins produced via transient and stable expression systems [54, 55, 91, 92]. This is vital as biochemical properties influences the affinity and thus stoichiometry, stability, and expressibility of antibodies, making them are essential parameters for antibodies in research and medical tools [93].This study reports no biochemical distinction from stable and transient expressed antibodies with the only difference being expression levels which was reasoned to be from dissimilar promoters employed [92]. Based on this report, antibodies from both systems exhibit similar properties from antibody folding, assembly, and affinity [54]. A challenge faced by bispecific antibody expression is misassembly but as reported in this study, both systems generate similar amounts of misassembled bispecific antibodies although most expressed proteins were correctly assembled [91]. Furthermore, this study reports no biochemical discrepancies in proteins expressed from transient and stable systems but antibodies produced from stable has a slightly high level of proper assembly and expression, the authors noted co-expression in stable cells remains the predominant approach for IgG-like bispecific antibody production [55]. Additionally, the review from Wang reports on numerous bispecific antibodies expressed from various hosts and expression systems where both transient and stable systems were utilized, indicating no particular desirable mode of expression with both reporting high stable formulations [55]. All in all, proteins from stable cell lines and transient cells are similar biochemically with no reported differences on its stoichiometry.
Transient gene expression has a short time frame for protein harvesting but a short production time frame. Transiently transfected cells generate a lower yield. Transient expression is based on transgene-carrying plasmids transfected into the cell nuclei are transcribed as non-integrated plasmids rather than being permanently incorporated into the genome and thus the foreign gene is not replicated and gets diluted over time as it is lost through cell division, degradation or other factors [15]. Transient gene expression is most frequently and successfully employed technique for functional studies such as biochemical and preclinical assessments as well as initial screening for new expression strategy, especially when examining the design of the vector construct and molecular candidates [16]. It is unfortunately unapt for most structural studies and large-scaled productions, and thus as a result, transiently transfected cell lines are only used in short-term experiments [16]. However, as of recent years, through engineering of new expression vectors, transfection optimizations, and development of new culture processes, transient expression systems have advanced to the point where milligram to gram quantities of recombinant proteins can now be generated and although the yield is still comparatively lower than yield from stable gene expression systems, this is an incredible progress [15]. HEK 293 cells are the preferred mammalian cell-based transient protein expression systems for their flexible conversion to suspension cultivation in SFM, good transfection efficiency, high productivity, ability to grow to high cell densities and the inclusion of an episomal replication mechanism [16, 94].
Furthermore, transiently transfected cells may express inconsistently throughout the population. A dated notion believes that all in vitro generated antibodies will possess similar profiles to those produced in vivo but time has proven otherwise and minor variation in culture conditions, cell quality, medium content as well as transfection conditions cause major differences in glycan content regardless of being produced in one and the same production laboratory [17]. Using stable cell lines, the disparity and variation in expression levels associated with repeated transient transfection can be eradicated. Another downside to transient gene expression is that in transient transfection assays, several gene regulatory regions or inducible promoters work incorrectly which may be as many long-range enhancer and silencer components do not operate unless incorporated into the genome or a lack of sufficient chromatin formation, therefore transient transfection assay is not a suitable platform for determining the full impact of regulatory regions on a gene’s expression [19]. Stable expression is especially important and thus the standard practise for continued genetic research or industrial protein manufacturing.
Meanwhile stable clones may take several months to be generated and but it is able to express large amount of consistent, stable quality protein of interest with regulatory familiarity. Stable cell lines can continually express the transgene as the expression construct is integrated stably into the host cell’s genome and thus stable cell lines are able to carry and pass on the same modified genetic characteristics to its future generations [18, 73]. Although it can be noted that as stable cell lines consist of a single gene copy number of the integrated DNA, it results in a lower level of protein expression as compared to their transient counterparts with a higher copy number [95]. In contemporary era, the overflowing market for antibody medicines induces a need for their much-improved counterparts and the patent expiration of many “blockbuster” antibody drugs that drove the production of several biosimilar antibodies has all in all increases the demand for time effective, quality and quantity of antibodies [41]. Stable cell lines are able to metaphorically tick all these boxes as for long term production. These cell lines are cost-effective and manufactures stable antibodies in high yield to cater for the demand.
Principally, stable cell lines are generated using strong viral or cellular promoters and enhancers to drive expression by genomic integrating the GOI through an expression vector encoding the recombinant gene [18]. Based on integrant quantity and their sites, there may be variation in protein synthesis levels in the same cell batch [8]. A selective advantage to the transgenic cells such as antibiotic resistance in the genome can be introduced for selection of cell lines. Gene transfer to stable mammalian cells lines can be done virally and non-virally. Nonetheless, the expression levels are dependent on transgene integration position, quantity of integrations per cell, and individual cell variations amongst many others. Consequently, screening for high producer cell lines is necessary to obtain a cell line of fast growth and high productivity.
Position effect is a phenomenon from randomly integrated transgene, interfering with expression monitoring as it is dependent on the differential influences of its genomic surroundings [96]. This is due to clonal variation which brings about integration site concerns as the current human cell line generation processes are based on random integration of GOI into the genome which limits predictive value, process streamline, and cost-effective therapeutic glycoprotein production which also makes it impossible to directly compare between wild-type and mutant reporters [19, 20]. Besides, an integrated gene’s copy number tend to be vague and differs between cells, making the cell population heterogenous. To preserve the transgene, continuous selection pressure is necessary, as cells prefer to eliminate randomly inserted alien genetic elements, which may alter genome integrity [18]. The encoded resistance markers are not able to completely neutralize such selective agents i.e., antibiotics, which will then lead to cumulative mutations in the host cells, and thus must be removed from the proteins before usage [97]. Robust exogenous promoters such as pCMV elevate and express the recombinant gene non-physiologically [98]. Recombinant protein over-expression from multiple integrants can impose a significant strain on host cells, hence negatively impacting proper protein production [18].
Thus, there are strategies to eliminate clonal variance while generating stable high expressing cell lines expressing protein of interest in a controllable, quantifiable, and efficient fashion. Site-specific targeting based on recombinases manipulate genomic DNA with high efficiency and fine spatiotemporal control by inserting the GOI into the chromatin in a transcriptionally active region, or genomic hot spot [99, 100]. These systems do not possess preference for specific cell types but require genetically engineered parental cell lines with “landing pads” which are suitably arranged recombinase recognition sites as chromosomal targets [101]. Site-specific recombination is a precise genome editing tool to produce more controllable and predictable stable cell clones with high productivity and is likely to dramatically reduce the time needed for cell line screening, thus making them indispensable for biomedical research [79]. Due to their exceptional ability to excise, insert, inverse, and translocate genomic DNA in living organisms, site-specific recombinases are vital genome engineering techniques [100]. Anyhow, there are some technical issues that comes with the use of site-specific recombinase. The systematics of temporal and spatially regulated recombination must be improved, the expression of a recombination reporter does not necessarily signify target gene deletion, traditional lineage tracing for cell fate mapping is solely dependent on recognized cell type markers, and hence to perform regional and prompt gene expression and deletion, novel strategies are required [100]. Some examples of the genetic manipulation toolbox are the Cre/Loxp and Flp/Frt, which depend on a reporter gene’s random integration to identify genomic hot spots [79].
The most widely used site-specific recombinase, Cre (causes/cyclisation recombinase) has been used to understand gene function and modify many forms of DNA, in any cellular setting and in several species in vivo [100]. In site-specific recombination, the two necessary components are the transacting function, which in this case is Cre and a site which is loxp of which the Cre recombinase catalyses the recombination between the loxp recognition sites [100]. The expression and activity of the Cre recombinase, which is encoded by the bacteriophage P1 gene Cre is regulated by tissue-specific promoters and ligand tamoxifen to allow conditional genetic modifications [102]. However, there are some setbacks in the usage of this system as they possess an inconsistent nature and the epigenetic silencing of the promoters used can create highly variable expression patterns and alien phenotypes, additionally, as the event is invisible, there may be variable recombination or gene deletion efficiency which is why novel technologies such as iSuRe-Cre can be used [102].
A similar, straightforward and recurrent technology is the Flp-FRT recombination system derived from Saccharomyces cerevisiae where the flippase (Flp) recombinase catalyses DNA rearrangements through engineered genomic Flp recognition target (FRT) sites to achieve precise gene integration [21, 103]. This technology is used to modify genomes both within the germline and somatic tissue where gene expression is manipulated by utilising previously integrated FRT sites in the genome or extrachromosomal arrays to modify DNA elements at specific sites [104]. However, there is a range of possible shortcomings to this current system. The existence of bacterial sequences that are CpG-rich and thus vulnerable to methylation in mammalian cells, which could result in the reporter being silenced [19]. Additionally, the Flp-FRT systems integrate entire vector sequences, including vector backbone, into the FRT site [21].
Homologous recombination is another strategy to be adopted for eradicating clonal variation where targeted transgene insertions are site-specific using designer nucleases which allows considerable flexibility in target site selection in comparison to site-specific recombination and is known as biological scissors for genome editing [101]. An example is the genome editing RNA guided system, CRISPR/Cas9. This system is made up of two elements, Cas9 enzyme and a guide RNA (gRNA), where Cas9, a RNA guided endonuclease is used to form targeted double-strand breaks (DSBs) in mammalian cell genome which will be patched by non-homologous end-joining (NHEJ) or, to a lower degree, homology-directed repair (HDR) [100, 105]. Utilising clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein (Cas9)-mediated targeted integration combined with recombinase-mediated cassette exchange (RMCE), this system is a platform for producing isogenic cell lines [20].
As of recent years, new systems consisting of two or more of the aforementioned techniques are designed to offer the best of all worlds where the limitations of each system can be countered with another. An example is a new integration system where large DNA constructs can be inserted into specific genome sites by using a combination of recombinases, Flp and Cre as regular methods as the application of CRISPR technology is laborious and time consuming [104]. Another novel technology is “Cas-Pi”, which stands for cascaded precise integration, where large, complex, exogenous, and repetitive DNA sequences can be precisely integrated into genomic loci in cultured cells [21]. This is especially unique as these DNA sequences are difficult to integrate with contemporary genome targeting technology and this protocol came to be with a combination of Cre-loxp and Flp-FRT recombination events as well as CRISPR technology [21]. In genomic engineering, there is also the use of different hybrid enzymes such as the development of Flp-TAL recombinases, which comprises of Flp, a site-specific recombinase and DNA-binding site of transcription activator-like effector (TAL) to integrate and delete genes [22].
Nonetheless, the design of these systems and concurrent necessity expertise as well as the cost impedes their deployment.
Other strategies in increasing antibody yield
Low protein yield is a major drawback of producing recombinant therapeutic proteins from mammalian cell lines such as HEK293 cells. Optimizing the design of the expression vector components is one of the many efficient strategies for increasing transgene expression levels [106]. Use of promoters such as EF1α, SV40, TK, and CMV promoters can regulate mammalian cell line expression [20]. The most popular promotor is pCMV for its high expression but yield of transgenes powered by the CMV promotor diminish with culture time due to complex transcriptional silencing and DNA methylation issues from its many potential methylation sites [76]. Therefore, artificial promoters, such as the fusion of CMV with chicken beta-actin promoter (CAG) are preferred instead [76]. Besides, while the transgene expression from pCMV promoter is high in initial stages after transduction or transfection, it decreases soon after during prolonged culturing of the transduced or transfected cells and instead the transgene expression was noticeably more stable and increased under the control of the pCNA promoter which was noted through this study as a potential universal promoter for gene therapeutic constructs [23]. CMV may be the choice promotor for short-term expression while for long-term expression, CMV-CAG hybrid or CNA may be chosen instead. Thus, the choice of promoters is especially important as the promoter is a crucial component of an expression vector, and choosing the right one will boost the expression and stability of a GOI.
Another strategy is to reduce cell culture temperature to a slightly hypothermic temperature to increase transgene expression. This will empirically extend the active life of the culture by slowing apoptosis and nutrient degradation, and, for certain proteins, this technique will also improve the yield of correctly folded protein [10]. Besides improving the assembly and secretion rates of the expressed protein, lower temperatures reduce product aggregation due to improved endoplasmic reticulum machinery [107]. Hypothermia causes a rise in transgene expression, as well as an accumulation of cells in the G1 process and a decrease in cellular metabolism [15]. However, while reducing culture temperature does not impact protein function or activity, it decreases cell growth rate and impedes cell division [108].
Furthermore, specific recombinant protein productivity is correlated with culture osmolality as it influences cell size which in turn influences biosynthetic capacity and nutrient exchange [24]. Hyperosmolar conditions which are customarily induced from repeated feed addition and metabolic by-product build-up or salt supplementation with sodium chloride or potassium chloride brings on increased nucleus mitochondria, endoplasmic reticulum, as well as protein and gene expression [24]. However, the quality and biopotency of mAb production are guided by application time, hyperosmolality level and culture mode [88]. It is important to note that hyperosmolar conditions can suppress cell growth and apoptosis [24, 88].
Moreover, increase specific productivity can be obtained by cell cycle arrest when the cells are at the largest size. Harvesting of antibodies can be optimised to be done with the highest productivity during or prolonging the stationary phase and late-stage culture by starving the cells or adding molecule regulators as they are the key development phases in recombinant protein production where there is a rise in cell percentage in the G1 process and an increase in cell diameter [24]. Without increase cell density, this strategy is makes it possible to obtain and increase specific volumetric productivity as cell size is correlated to extracellular nutrient conditions which enables mammalian cell size to be manipulated through media and feeds to improve cell growth and specific productivity [25]. Correlating with osmolarity, there is an accumulation of extracellular metabolites which can be further intensifies by overfeeding during the late-stage culture contributing to an increase in osmolarity [24].
Downstream processing
The recovery and purification of biosynthetic is critical in assuring its quality. After the proteins are produced and harvested, the next step is purification. The principal concern for purification is purity, followed by speed, overall yield, cost, and process throughput [52]. However, although purification will remove most unwanted molecules, heterogeneities will still occur [39]. Various modifications such as incomplete disulphide bond formation, glycosylation, N-terminal pyroglutamine cyclisation, C-terminal lysine processing, deamidation, isomerization, and oxidation, over the protein lifespan amass to heterogeneity [109]. Furthermore, heterogeneity may be caused by subsequent exposure to culture media, stress from pH and temperature as well as post-translational modifications, degradation, and other chemical modifications during preparation and storing [39].
A purification system is chosen on the basis of its robustness, reliability, and scalability. Affinity purification are often the choice method for antibodies although its effectiveness is reliant on the antibody’s ability to bind to an affinity adsorbent [110]. For process scale purification, Protein A affinity chromatography derived by the cell wall of Staphylococcus aureus is the benchmark as it utilises specific ligand interactions between mAb Fc region and immobilised Protein A to achieve high selectivity at over 95% [52]. For pharmaceutically accepted purity levels, Protein A affinity chromatography is followed up by cation-exchange chromatography and then anion-exchange chromatography [52]. It is then proceeded with virus inactivation and ultrafiltration/diafiltration. Other purification systems include histidine-ligand-affinity chromatography, metal-affinity chromatography, thiophilic interaction, lectin-affinity chromatography, and purification of recombinant antibodies using affinity tags [110].
Conclusion
Over the last few decades, antibodies are extremely prevalent with the never-ending advancement and commercialisation of this biologics. Knowledge from antibody product development over the years has aided in advancement and invention of novel technologies as well as increased production to satisfy the vast demand. It has and still is piloting the way for ground breaking therapeutic advancements by being the primary therapeutic modality among many others. Over this time, discovery and production of antibody drugs has gone through significant change and polishing, including our understanding of target biology. Up to this point, the conventional industry method for mammalian cell hosts’ protein development is the traditional generation of stable cell lines. In events where protein of interest is well-defined and large quantities are required, investment of time and resources in the development of such cell lines to significantly increase cell productivity is a given. This calls for site-specific recombination technologies which have enabled improved editing of genomic DNA in a variety of biomedical research fields to fulfil the ever-increasing demands. In a nutshell, each and every system possesses its respective set of pros and cons, therefore, the choice of approach is important and used to answer different biological concerns. Although more evidence is necessary to consolidate the effectiveness of these strategies, there is much hope and it is beyond doubt that there will be expansion on strategies, systems, and pipelines to offer additional advancement in molecular medicine.
Author contributions
JKKM has the idea for the article, who performed the literature search and data analysis, and GJT drafted and critically revised the work. CSYL and FN revised and critique the work.
Funding
This study was funded by “Ministry of Higher Education Malaysia for Fundamental Research Grant Scheme with Project Code: FRGS/1/2020/STG01/USM/02/12" and "Ministry of Higher Education Malaysia for Higher Institution Centre of Excellence (Grant No. HICoE: 311/CIPPM/4401005)".
Declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kang TH, Jung ST. Boosting therapeutic potency of antibodies by taming Fc domain functions. Exp Mol Med. 2019 doi: 10.1038/s12276-019-0345-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.De Cecco M, Galbraith DN, McDermott LL. What makes a good antibody–drug conjugate? Expert Opin Biol Ther. 2021;21:841–847. doi: 10.1080/14712598.2021.1880562. [DOI] [PubMed] [Google Scholar]
- 3.Mullard A. FDA approves 100th monoclonal antibody product. Nat Rev Drug Discov. 2021;20:491–495. doi: 10.1038/d41573-021-00079-7. [DOI] [PubMed] [Google Scholar]
- 4.Kaur H. Characterization of glycosylation in monoclonal antibodies and its importance in therapeutic antibody development. Crit Rev Biotechnol. 2021;41:300–315. doi: 10.1080/07388551.2020.1869684. [DOI] [PubMed] [Google Scholar]
- 5.Ecker DM, Jones SD, Levine HL. The therapeutic monoclonal antibody market. MAbs. 2015;7:9–14. doi: 10.4161/19420862.2015.989042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Baik JY, Lee KH. A framework to quantify karyotype variation associated with CHO cell line instability at a single-cell level. Biotechnol Bioeng. 2017;114:1045–1053. doi: 10.1002/bit.26231. [DOI] [PubMed] [Google Scholar]
- 7.Dahodwala H, Lee KH. The fickle CHO: a review of the causes, implications, and potential alleviation of the CHO cell line instability problem. Curr Opin Biotechnol. 2019;60:128–137. doi: 10.1016/j.copbio.2019.01.011. [DOI] [PubMed] [Google Scholar]
- 8.Assur Z, Hendrickson WA, Mancia F. Tools for co-expressing multiple proteins in mammalian cells. Methods Mol Biol. 2012;801:161–172. doi: 10.1007/978-1-61779-352-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ryu J, Kim E-J, Kim J-K, Park TH, Kim B-G, Jeong H-J. Development of a CHO cell line for stable production of recombinant antibodies against human MMP9. BMC Biotechnol. 2022;22:1–8. doi: 10.1186/s12896-022-00738-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Roobol A, Roobol J, Smith ME, Carden MJ, Hershey JWB, Willis AE, Smales CM. Engineered transient and stable overexpression of translation factors eIF3i and eIF3c in CHOK1 and HEK293 cells gives enhanced cell growth associated with increased c-Myc expression and increased recombinant protein synthesis. Metab Eng. 2020;59:98–105. doi: 10.1016/j.ymben.2020.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kim YJ, Han SK, Yoon S, Kim CW. Rich production media as a platform for CHO cell line development. AMB Express. 2020 doi: 10.1186/s13568-020-01025-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Obaidi I, Mota LM, Quigley A, Butler M. The role of protein hydrolysates in prolonging viability and enhancing antibody production of CHO cells. Appl Microbiol Biotechnol. 2021 doi: 10.1007/s00253-021-11244-8. [DOI] [PubMed] [Google Scholar]
- 13.Yao T, Asayama Y. Animal-cell culture media: history, characteristics, and current issues. Reprod Med Biol. 2017;16:99–117. doi: 10.1002/rmb2.12024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Combe M, Sokolenko S. Quantifying the impact of cell culture media on CHO cell growth and protein production. Biotechnol Adv. 2021;50:107761. doi: 10.1016/j.biotechadv.2021.107761. [DOI] [PubMed] [Google Scholar]
- 15.Lee J, Hansen HG, Park S, Park J, Kim Y. Transient gene expression-based protein production in recombinant mammalian cells. Cell Cult Eng. 2019 doi: 10.1002/9783527811410.ch3. [DOI] [Google Scholar]
- 16.Park SH, Park JH, Lee JH, Lee HM, Kang YJ, Lee EJ, Shin S, Lee GM, Kim YG. Amplification of EBNA-1 through a single-plasmid vector-based gene amplification system in HEK293 cells as an efficient transient gene expression system. Appl Microbiol Biotechnol. 2021;105:67–76. doi: 10.1007/s00253-020-11001-3. [DOI] [PubMed] [Google Scholar]
- 17.Saphire EO, Schendel SL, Gunn BM, Milligan JC, Alter G. Antibody-mediated protection against Ebola virus. Nat Immunol. 2018;19:1169–1178. doi: 10.1038/s41590-018-0233-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lo CA, Greben AW, Chen BE. Generating stable cell lines with quantifiable protein production using CRISPR/Cas9-mediated knock-in. Biotechniques. 2017;62:165–174. doi: 10.2144/000114534. [DOI] [PubMed] [Google Scholar]
- 19.Jensen O, Ansari S, Gebauer L, Müller SF, Lowjaga KAAT, Geyer J, Tzvetkov MV, Brockmöller J. A double-Flp-in method for stable overexpression of two genes. Sci Rep. 2020;10:1–14. doi: 10.1038/s41598-020-71051-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shin S, Kim SH, Shin SW, Grav LM, Pedersen LE, Lee JS, Lee GM. Comprehensive analysis of genomic safe harbors as target sites for stable expression of the heterologous gene in HEK293 cells. ACS Synth Biol. 2020;9:1263–1269. doi: 10.1021/acssynbio.0c00097. [DOI] [PubMed] [Google Scholar]
- 21.Li J, Li Y, Pawlik KM, Napierala JS, Napierala M. A CRISPR-Cas9, Crelox, and Flp-FRT cascade strategy for the precise and efficient integration of exogenous DNA into cellular genomes. Cris J. 2020;3:470–486. doi: 10.1089/crispr.2020.0042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Voziyanova E, Li F, Shah R, Voziyanov Y. Genome targeting by hybrid Flp-TAL recombinases. Sci Rep. 2020;10:1–15. doi: 10.1038/s41598-020-74474-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Antonova DV, Alekseenko IV, Siniushina AK, Kuzmich AI, Pleshkan VV. Searching for promoters to drive stable and long-term transgene expression in fibroblasts for syngeneic mouse tumor models. Int J Mol Sci. 2020;21:1–18. doi: 10.3390/ijms21176098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Alhuthali S, Kotidis P, Kontoravdi C. Osmolality effects on cho cell growth, cell volume, antibody productivity and glycosylation. Int J Mol Sci. 2021 doi: 10.3390/ijms22073290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pan X, Alsayyari AA, Dalm C, Hageman JA, Wijffels RH, Martens DE. Transcriptome analysis of CHO cell size increase during a fed-batch process. Biotechnol J. 2019;14:1–12. doi: 10.1002/biot.201800156. [DOI] [PubMed] [Google Scholar]
- 26.Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P. B cells and antibodies. In: Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P, editors. Molecular biology of the cell. 4. New York: Garland Science; 2002. [Google Scholar]
- 27.Morris JA, Dorner AJ, Edwards CA, Hendershott LM, Kaufman RJ. Immunoglobulin binding protein (BiP) function is required to protect cells from endoplasmic reticulum stress but is not required for the secretion of selective proteins. J Biol Chem. 1997;272:4327–4334. doi: 10.1074/jbc.272.7.4327. [DOI] [PubMed] [Google Scholar]
- 28.Feige MJ, Hendershot LM, Buchner J. How antibodies fold. Trends Biochem Sci. 2010;35:189–198. doi: 10.1016/j.tibs.2009.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.O’Flaherty R, Bergin A, Flampouri E, Mota LM, Obaidi I, Quigley A, Xie Y, Butler M. Mammalian cell culture for production of recombinant proteins: a review of the critical steps in their biomanufacturing. Biotechnol Adv. 2020;43:107552. doi: 10.1016/j.biotechadv.2020.107552. [DOI] [PubMed] [Google Scholar]
- 30.Ejazi SA, Ghosh S, Ali N. Antibody detection assays for COVID-19 diagnosis: an early overview. Immunol Cell Biol. 2021;99:21–33. doi: 10.1111/imcb.12397. [DOI] [PubMed] [Google Scholar]
- 31.Weiner GJ. Building better monoclonal antibody-based therapeutics. Nat Rev Cancer. 2015;15:361–370. doi: 10.1038/nrc3930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Alter G, Seder R. The power of antibody-based surveillance. N Engl J Med. 2020;383:1782–1784. doi: 10.1056/NEJMe2028079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Siddiqui MZ. Monoclonal antibodies as diagnostics; an appraisal. Indian J Pharm Sci. 2010;72:12–17. doi: 10.4103/0250-474X.62229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vashist SK, et al. In vitro diagnostic assays for covid-19: recent advances and emerging trends. Diagnostics. 2020;10:202. doi: 10.3390/diagnostics10040202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Xu X, Zhang R, Chen X. Application of a single-chain fragment variable (scFv) antibody for the confirmatory diagnosis of hydatid disease in non-endemic areas. Electron J Biotechnol. 2017;29:57–62. doi: 10.1016/j.ejbt.2017.07.003. [DOI] [Google Scholar]
- 36.Kim YJ, Lee MH, Lee SR, Chung HY, Kim K, Lee TG, Kim DY. Neutralizing human antibodies against severe acute respiratory syndrome coronavirus 2 isolated from a human synthetic fab phage display library. Int J Mol Sci. 2021;22:1–18. doi: 10.3390/ijms22041913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lua WH, Ling WL, Yeo JY, Poh JJ, Lane DP, Gan SKE. The effects of antibody engineering CH and CL in trastuzumab and pertuzumab recombinant models: Impact on antibody production and antigen-binding. Sci Rep. 2018;8:1–9. doi: 10.1038/s41598-017-18892-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Panawala L (2017) Difference between monoclonal and polyclonal antibodies. Available online: https://www.researchgate.net/publication/320707210_Difference_Between_Monoclonal_and_Polyclonal_Antibodies. Accessed on 23 Dec 2021
- 39.Wang X, An Z, Luo W, Xia N, Zhao Q. Molecular and functional analysis of monoclonal antibodies in support of biologics development. Protein Cell. 2018;9:74–85. doi: 10.1007/s13238-017-0447-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.van Tetering G, Evers M, Chan C, Stip M, Leusen J. Fc engineering strategies to advance IgA antibodies as therapeutic agents. Antibodies. 2020;9:70. doi: 10.3390/antib9040070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Carter PJ, Lazar GA. Next generation antibody drugs: Pursuit of the “high-hanging fruit”. Nat Rev Drug Discov. 2018;17:197–223. doi: 10.1038/nrd.2017.227. [DOI] [PubMed] [Google Scholar]
- 42.Iwasaki A, Yang Y. The potential danger of suboptimal antibody responses in COVID-19. Nat Rev Immunol. 2020;20:339–341. doi: 10.1038/s41577-020-0321-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shen C, Zhang M, Chen Y, Zhang L, Wang G, Chen J, Chen S, Li Z, Wei F, Chen J, Yang K, Guo S, Wang Y, Zheng Q, Yu H, Luo W, Zhang J, Chen H, Chen Y, Xia N. An IgM antibody targeting the receptor binding site of influenza B blocks viral infection with great breadth and potency. Theranostics. 2019;9:210–231. doi: 10.7150/thno.28434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Klingler J, Weiss S, Itri V, Liu X, Oguntuyo KY. Role of IgM and IgA antibodies in the neutralization of SARS-CoV-2. medRxiv. 2020 doi: 10.1101/2020.08.18.20177303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Karagiannis SN, Josephs DH, Bax HJ, Spicer JF. Therapeutic IgE antibodies: harnessing a macrophage-mediated immune surveillance mechanism against cancer. Cancer Res. 2017;77:2779–2783. doi: 10.1158/0008-5472.CAN-17-0428. [DOI] [PubMed] [Google Scholar]
- 46.Chen K, Cerutti A. The function and regulation of immunoglobulin D. Curr Opin Immunol. 2011;23:345–352. doi: 10.1016/j.coi.2011.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Vidarsson G, Dekkers G, Rispens T. IgG subclasses and allotypes: from structure to effector functions. Front Immunol. 2014;5:1–17. doi: 10.3389/fimmu.2014.00520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gogesch P, Dudek S, van Zandbergen G, Waibler Z, Anzaghe M. The role of FC receptors on the effectiveness of therapeutic monoclonal antibodies. Int J Mol Sci. 2021 doi: 10.3390/ijms22168947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Baba TW, Liska V, Hofmann-Lehmann R, Vlasak J, Xu W, Ayehunie S, Cavacini LA, Posner MR, Katinger H, Stiegler G, Bernacky BJ, Rizvi TA, Schmidt R, Hill LR, Keeling ME, Lu Y, Wright JE, Chou T-C, Ruprecht RM. Human neutralizing monoclonal antibodies of the IgG1 subtype protect against mucosal simian–human immunodeficiency virus infection. Nat Med. 2000;6:200–206. doi: 10.1038/72309. [DOI] [PubMed] [Google Scholar]
- 50.Yang C, Gao X, Gong R. Engineering of Fc fragments with optimized physicochemical properties implying improvement of clinical potentials for Fc-based therapeutics. Front Immunol. 2018 doi: 10.3389/fimmu.2017.01860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rath T, Baker K, Dumont JA, Peters RT, Jiang H, Qiao SW, Lencer WI, Pierce GF, Blumberg RS. Fc-fusion proteins and FcRn: Structural insights for longer-lasting and more effective therapeutics. Crit Rev Biotechnol. 2015;35:235–254. doi: 10.3109/07388551.2013.834293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Shukla AA, Hubbard B, Tressel T, Guhan S, Low D. Downstream processing of monoclonal antibodies-Application of platform approaches. J Chromatogr B. 2007;848:28–39. doi: 10.1016/j.jchromb.2006.09.026. [DOI] [PubMed] [Google Scholar]
- 53.Lobner E, Humm AS, Göritzer K, Mlynek G, Puchinger MG, Hasenhindl C, Rüker F, Traxlmayr MW, Djinović-Carugo K, Obinger C. Fcab-HER2 interaction: a ménage à trois. lessons from X-ray and solution studies. Structure. 2017;25:878–889.e5. doi: 10.1016/j.str.2017.04.014. [DOI] [PubMed] [Google Scholar]
- 54.Mason M, Sweeney B, Cain K, Stephens P, Sharfstein ST. Identifying bottlenecks in transient and stable production of recombinant monoclonal-antibody sequence variants in chinese hamster ovary cells. Biotechnol Prog. 2012;28:846–855. doi: 10.1002/btpr.1542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wang Q, Chen Y, Park J, Liu X, Hu Y, Wang T, McFarland K, Betenbaugh MJ. Design and production of bispecific antibodies. Antibodies. 2019;8:43. doi: 10.3390/antib8030043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ma J, Mo Y, Tang M, Shen J, Qi Y, Zhao W, Huang Y, Xu Y, Qian C. Bispecific antibodies: from research to clinical application. Front Immunol. 2021 doi: 10.3389/fimmu.2021.626616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lim SM, Pyo KH, Soo RA, Cho BC. The promise of bispecific antibodies: clinical applications and challenges. Cancer Treat Rev. 2021;99:102240. doi: 10.1016/j.ctrv.2021.102240. [DOI] [PubMed] [Google Scholar]
- 58.Salazar G, Zhang N, Fu TM, An Z. Antibody therapies for the prevention and treatment of viral infections. npj Vaccines. 2017;2:1–12. doi: 10.1038/s41541-017-0019-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Chames P, Van Regenmortel M, Weiss E, Baty D. Therapeutic antibodies: successes, limitations and hopes for the future. Br J Pharmacol. 2009;157:220–233. doi: 10.1111/j.1476-5381.2009.00190.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kuhn P, Fühner V, Unkauf T, Moreira GMSG, Frenzel A, Miethe S, Hust M. Recombinant antibodies for diagnostics and therapy against pathogens and toxins generated by phage display. Proteomics. 2016;10:922–948. doi: 10.1002/prca.201600002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Xenaki KT, Oliveira S, van Bergen en Henegouwen PMP. Antibody or antibody fragments: Implications for molecular imaging and targeted therapy of solid tumors. Front Immunol. 2017 doi: 10.3389/fimmu.2017.01287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yang O, Prabhu S, Ierapetritou M. Comparison between batch and continuous monoclonal antibody production and economic analysis. Ind Eng Chem Res. 2019;58:5851–5863. doi: 10.1021/acs.iecr.8b04717. [DOI] [Google Scholar]
- 63.Kaplon H, Reichert JM. Antibodies to watch in 2021. MAbs. 2021;13:1–34. doi: 10.1080/19420862.2020.1860476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.FDA News Release (2021) Coronavirus (COVID-19) Update: FDA Revokes Emergency Use Authorization for Monoclonal Antibody Bamlanivimab. U.S. Food Drugs Adm. 1
- 65.Lee WS, Wheatley AK, Kent SJ, DeKosky BJ. Antibody-dependent enhancement and SARS-CoV-2 vaccines and therapies. Nat Microbiol. 2020;5:1185–1191. doi: 10.1038/s41564-020-00789-5. [DOI] [PubMed] [Google Scholar]
- 66.Ledford H. Antibody therapies could be a bridge to a coronavirus vaccine—but will the world benefit? Nature. 2020;584:333–334. doi: 10.1038/d41586-020-02360-y. [DOI] [PubMed] [Google Scholar]
- 67.Doerner A, Rhiel L, Zielonka S, Kolmar H. Therapeutic antibody engineering by high efficiency cell screening. FEBS Lett. 2014;588:278–287. doi: 10.1016/j.febslet.2013.11.025. [DOI] [PubMed] [Google Scholar]
- 68.Valldorf B, Hinz SC, Russo G, Pekar L, Mohr L, Klemm J, Doerner A, Krah S, Hust M, Zielonka S. Antibody display technologies: selecting the cream of the crop. Biol Chem. 2021 doi: 10.1515/hsz-2020-0377. [DOI] [PubMed] [Google Scholar]
- 69.Almagro JC, Pedraza-Escalona M, Arrieta HI, Pérez-Tapia SM. Phage display libraries for antibody therapeutic discovery and development. Antibodies. 2019;8:44. doi: 10.3390/antib8030044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Alfaleh MA, Alsaab HO, Mahmoud AB, Alkayyal AA, Jones ML, Mahler SM, Hashem AM. Phage display derived monoclonal antibodies: from bench to bedside. Front Immunol. 2020 doi: 10.3389/fimmu.2020.01986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Jiang H, Horwitz AA, Wright C, Tai A, Znameroski EA, Tsegaye Y, Warbington H, Bower BS, Alves C, Co C, Jonnalagadda K, Platt D, Walter JM, Natarajan V, Ubersax JA, Cherry JR, Love JC. Challenging the workhorse: comparative analysis of eukaryotic micro-organisms for expressing monoclonal antibodies. Biotechnol Bioeng. 2019;116:1449–1462. doi: 10.1002/bit.26951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Matthews CB, Wright C, Kuo A, Colant N, Westoby M, Love JC. Reexamining opportunities for therapeutic protein production in eukaryotic microorganisms. Biotechnol Bioeng. 2017;114:2432–2444. doi: 10.1002/bit.26378. [DOI] [PubMed] [Google Scholar]
- 73.Lalonde ME, Durocher Y. Therapeutic glycoprotein production in mammalian cells. J Biotechnol. 2017;251:128–140. doi: 10.1016/j.jbiotec.2017.04.028. [DOI] [PubMed] [Google Scholar]
- 74.Wells E, Robinson AS. Cellular engineering for therapeutic protein production: product quality, host modification, and process improvement. Biotechnol J. 2017;12:1–12. doi: 10.1002/biot.201600105. [DOI] [PubMed] [Google Scholar]
- 75.Dangi AK, Sinha R, Dwivedi S, Gupta SK, Shukla P. Cell line techniques and gene editing tools for antibody production: a review. Front Pharmacol. 2018;9:1–12. doi: 10.3389/fphar.2018.00630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Dou Y, Lin Y, Wang T, yun, Wang XY, Jia Y long, Zhao C peng, The CAG promoter maintains high-level transgene expression in HEK293 cells. FEBS Open Bio. 2021;11:95–104. doi: 10.1002/2211-5463.13029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Bosques CJ, Collins BE, Meador JW, III, Sarvaiya H, Murphy JL, DelloRusso G, Bulik DA, Hsu IH, Washburn N, Sipsey SF, Myette JR, Raman R, Shriver Z, Sasisekharan R, Venkataraman G. Chinese hamster ovary cells can produce galactose-α-1,3-galactose antigens on proteins. Nat Biotechnol. 2010;28:1153–1156. doi: 10.1038/nbt1110-1153.Chinese. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Arnold JN, Wormald MR, Sim RB, Rudd PM, Dwek RA. The impact of glycosylation on the biological function and structure of human immunoglobulins. Annu Rev Immunol. 2007;25:21–50. doi: 10.1146/annurev.immunol.25.022106.141702. [DOI] [PubMed] [Google Scholar]
- 79.Melville M, Estes S. Mammalian cell line developments in speed and efficiency. Adv Biochem Eng Biotechnol. 2013;123:127–141. doi: 10.1007/10_2013_260. [DOI] [PubMed] [Google Scholar]
- 80.Noguchi A, Mukuria CJ, Suzuki E, Naiki M. Immunogenicity of N-glycolylneuraminic acid-containing carbohydrate chains of recombinant human erythropoietin expressed in chinese hamster ovary cells. J Biochem. 1995;117:59–62. doi: 10.1093/oxfordjournals.jbchem.a124721. [DOI] [PubMed] [Google Scholar]
- 81.Yu C, Gao K, Zhu L, Wang W, Wang L, Zhang F, Liu C, Li M, Wormald MR, Rudd PM, Wang J. At least two Fc Neu5Gc residues of monoclonal antibodies are required for binding to anti-Neu5Gc antibody. Sci Rep. 2016;7:1–11. doi: 10.1038/srep20029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Chung CH, Mirakhur B, Chan E, Le Q-T, Berlin J, Morse M, Murphy BA, Satinover SM, Hosen J, Mauro D, Slebos RJ, Zhou Q, Gold D, Hatley T, Hicklin DJ, Platts-Mills TAE. Cetuximab-induced anaphylaxis and IgE specific for galactose-α-1,3-galactose. N Engl J Med. 2008;358:2735–2736. doi: 10.1056/NEJMc080834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Park YH, Gong SP, Kim HY, Kim GA, Choi JH, Ahn JY, Lim JM. Development of a serum-free defined system employing growth factors for preantral follicle culture. Mol Reprod Dev. 2013;80:725–733. doi: 10.1002/mrd.22204. [DOI] [PubMed] [Google Scholar]
- 84.Zhang G, Zhao X, Li X, Du G, Zhou J, Chen J. Challenges and possibilities for bio-manufacturing cultured meat. Trends Food Sci Technol. 2020;97:443–450. doi: 10.1016/j.tifs.2020.01.026. [DOI] [Google Scholar]
- 85.Devireddy LR, Myers M, Screven R, Liu Z, Boxer L. A serum-free medium formulation efficiently supports isolation and propagation of canine adipose-derived mesenchymal stem/stromal cells. PLoS ONE. 2019;14:1–21. doi: 10.1371/journal.pone.0210250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Gomes DS, Aragaõ LB, Lima Neto MF, Barroso PAA, Paulino LRFM, Silva BR, Souza ALP, Vasconcelos GL, Silva AWB, Silva JRV. Supplementation of culture medium with knockout serum replacement improves the survival of bovine secondary follicles when compared with other protein sources during in vitro culture. Zygote. 2019 doi: 10.1017/S0967199419000583. [DOI] [PubMed] [Google Scholar]
- 87.Li W, Fan Z, Lin Y, Wang TY. Serum-free medium for recombinant protein expression in chinese hamster ovary cells. Front Bioeng Biotechnol. 2021 doi: 10.3389/fbioe.2021.646363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Qin J, Wu X, Xia Z, Huang Z, Zhang Y, Wang Y, Fu Q, Zheng C. The effect of hyperosmolality application time on production, quality, and biopotency of monoclonal antibodies produced in CHO cell fed-batch and perfusion cultures. Appl Microbiol Biotechnol. 2019;103:1217–1229. doi: 10.1007/s00253-018-9555-7. [DOI] [PubMed] [Google Scholar]
- 89.Budge JD, Knight TJ, Povey J, Roobol J, Brown IR, Singh G, Dean A, Turner S, Jaques CM, Young RJ, Racher AJ, Smales CM. Data for engineering lipid metabolism of chinese hamster ovary (CHO) cells for enhanced recombinant protein production. Data Br. 2020;29:105217. doi: 10.1016/j.dib.2020.105217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Pérez-Rodriguez S, de Ramírez-Lira M, J, Trujillo-Roldán MA, Valdez-Cruz NA, Nutrient supplementation strategy improves cell concentration and longevity, monoclonal antibody production and lactate metabolism of chinese hamster ovary cells. Bioengineered. 2020;11:463–471. doi: 10.1080/21655979.2020.1744266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Rajendra Y, Peery RB, Hougland MD, Barnard GC, Wu X, Fitchett JR, Bacica M, Demarest SJ. Transient and stable CHO expression, purification and characterization of novel hetero-dimeric bispecific IgG antibodies. Biotechnol Prog. 2017;33:469–477. doi: 10.1002/btpr.2414. [DOI] [PubMed] [Google Scholar]
- 92.Li J, Menzel C, Meier D, Zhang C, Dübel S, Jostock T. A comparative study of different vector designs for the mammalian expression of recombinant IgG antibodies. J Immunol Methods. 2007;318:113–124. doi: 10.1016/j.jim.2006.10.010. [DOI] [PubMed] [Google Scholar]
- 93.Warszawski S, Katz AB, Lipsh R, Khmelnitsky L, Ben NG, Javitt G, Dym O, Unger T, Knop O, Albeck S, Diskin R, Fass D, Sharon M, Fleishman SJ. Optimizing antibody affinity and stability by the automated design of the variable light-heavy chain interfaces. PLoS Comput Biol. 2019;15:1–24. doi: 10.1371/journal.pcbi.1007207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Gutiérrez-Granados S, Cervera L, Kamen AA, Gòdia F. Advancements in mammalian cell transient gene expression (TGE) technology for accelerated production of biologics. Crit Rev Biotechnol. 2018;38:918–940. doi: 10.1080/07388551.2017.1419459. [DOI] [PubMed] [Google Scholar]
- 95.Schlicht D, Estoppey C, Macoin J, Pouleau B, Bertschinger M. Generation of homogeneous cell populations with tunable levels of transgene expression. J Biotechnol. 2021;325:288–293. doi: 10.1016/j.jbiotec.2020.10.008. [DOI] [PubMed] [Google Scholar]
- 96.Chen M, Licon K, Otsuka R, Pillus L, Ideker T. Decoupling epigenetic and genetic effects through systematic analysis of gene position. Cell Rep. 2013;3:128–137. doi: 10.1016/j.celrep.2012.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Trastoy MO, Defais M, Larminat F. Resistance to the antibiotic Zeocin by stable expression of the Sh ble gene does not fully suppress Zeocin-induced DNA cleavage in human cells. Mutagenesis. 2005;20:111–114. doi: 10.1093/mutage/gei016. [DOI] [PubMed] [Google Scholar]
- 98.Xia W, Bringmann P, McClary J, Jones PP, Manzana W, Zhu Y, Wang S, Liu Y, Harvey S, Madlansacay MR, McLean K, Rosser MP, MacRobbie J, Olsen CL, Cobb RR. High levels of protein expression using different mammalian CMV promoters in several cell lines. Protein Expr Purif. 2006;45:115–124. doi: 10.1016/j.pep.2005.07.008. [DOI] [PubMed] [Google Scholar]
- 99.Tian X, He L, Liu K, Pu W, Zhao H, Li Y, Liu X, Tang M, Sun R, Fei J, Ji Y, Qiao Z, Lui KO, Zhou B. Generation of a self-cleaved inducible Cre recombinase for efficient temporal genetic manipulation. EMBO J. 2020;39:1–12. doi: 10.15252/embj.2019102675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Tian X, Zhou B. Strategies for site-specific recombination with high efficiency and precise spatiotemporal resolution. J Biol Chem. 2021;296:100509. doi: 10.1016/j.jbc.2021.100509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Phan QV, Contzen J, Seemann P, Gossen M. Site-specific chromosomal gene insertion: Flp recombinase versus Cas9 nuclease. Sci Rep. 2017;7:1–12. doi: 10.1038/s41598-017-17651-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Fernández-Chacón M, Casquero-García V, Luo W, Francesca Lunella F, Ferreira Rocha S, Del Olmo-Cabrera S, Benedito R. iSuRe-Cre is a genetic tool to reliably induce and report Cre-dependent genetic modifications. Nat Commun. 2019;10:1–13. doi: 10.1038/s41467-019-10239-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Srivastava V. FLP-mediated site-specific gene integration in rice. In: Bandyopadhyay A, Thilmony R, editors. Rice genome engineering and gene editing. New York, NY: Humana Press; 2021. pp. 231–240. [Google Scholar]
- 104.Nonet ML. Efficient transgenesis in caenorhabditis elegans using flp recombinase-mediated cassette exchange. Genetics. 2020;215:903–921. doi: 10.1534/genetics.120.303388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Tran NT, Bashir S, Li X, Rossius J, Chu VT, Rajewsky K, Kühn R. With homologous recombination factors. Front Genet. 2019;10:1–13. doi: 10.3389/fgene.2019.00365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Bayat H, Hossienzadeh S, Pourmaleki E, Ahani R, Rahimpour A. Evaluation of different vector design strategies for the expression of recombinant monoclonal antibody in CHO cells. Prep Biochem Biotechnol. 2018;48:160–164. doi: 10.1080/10826068.2017.1421966. [DOI] [PubMed] [Google Scholar]
- 107.Wang K, Zhang T, Chen J, Liu C, Tang J, Xie Q. The effect of culture temperature on the aggregation of recombinant TNFR-Fc is regulated by the PERK-eIF2a pathway in CHO cells. Protein Pept Lett. 2018;25:570–579. doi: 10.2174/0929866525666180530121317. [DOI] [PubMed] [Google Scholar]
- 108.Fujii KK, Taga Y, Sakai T, Ito S, Hattori S, Nagata K, Koide T. Lowering the culture temperature corrects collagen abnormalities caused by HSP47 gene knockout. Sci Rep. 2019;9:1–11. doi: 10.1038/s41598-019-53962-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Liu H, Gaza-Bulseco G, Faldu D, Chumsae C, Sun J. Heterogeneity of monoclonal antibodies. J Pharm Sci. 2008;97:2426–2447. doi: 10.1002/jps.21180. [DOI] [PubMed] [Google Scholar]
- 110.Huse K, Böhme HJ, Scholz GH. Purification of antibodies by affinity chromatography. J Biochem Biophys Methods. 2002;51:217–231. doi: 10.1016/S0165-022X(02)00017-9. [DOI] [PubMed] [Google Scholar]