This randomized clinical trial investigates the use of emergency coronary angiogram vs delayed coronary angiogram in patients who experience out-of-hospital cardiac arrest without ST-segment elevation.
Key Points
Question
Does performing an emergency coronary angiogram (CAG) vs a delayed CAG after an out-of-hospital cardiac arrest (OHCA) without ST-segment elevation on the postresuscitation electrocardiogram (ECG) lead to a better outcome?
Findings
In this randomized clinical trial including 279 patients, there was no difference in the 180-day survival rate with Cerebral Performance Category 1 or 2, which included 34.1% of patients in the emergency CAG group and 30.7% of patients in the delayed CAG group. However, the study was underpowered; a combined analysis on pooled published data of 4 randomized trials on the same topic revealed the same conclusion.
Meaning
Results of this study do not support emergency CAG after OHCA without ST-segment elevation.
Abstract
Importance
Although an emergency coronary angiogram (CAG) is recommended for patients who experience an out-of-hospital cardiac arrest (OHCA) with ST-segment elevation on the postresuscitation electrocardiogram (ECG), this strategy is still debated in patients without ST-segment elevation.
Objective
To assess the 180-day survival rate with Cerebral Performance Category (CPC) 1 or 2 of patients who experience an OHCA without ST-segment elevation on ECG and undergo emergency CAG vs delayed CAG.
Design, Setting, and Participants
The Emergency vs Delayed Coronary Angiogram in Survivors of Out-of-Hospital Cardiac Arrest (EMERGE) trial randomly assigned survivors of an OHCA without ST-segment elevation on ECG to either emergency or delayed (48 to 96 hours) CAG in 22 French centers. The trial took place from January 19, 2017, to November 23, 2020. Data were analyzed from November 24, 2020, to July 30, 2021.
Main Outcomes and Measures
The primary outcome was the 180-day survival rate with CPC of 2 or less. The secondary end points were occurrence of shock, ventricular tachycardia, and/or fibrillation within 48 hours, change in left ventricular ejection fraction between baseline and 180 days, CPC scale at intensive care unit discharge and day 90, survival rate, and hospital length of stay.
Results
A total of 279 patients (mean [SD] age, 64.7 [14.6] years; 195 men [69.9%]) were enrolled, with 141 (50.5%) in the emergency CAG group and 138 (49.5%) in the delayed CAG group. The study was underpowered. The mean (SD) time delay between randomization and CAG was 0.6 (3.7) hours in the emergency CAG group and 55.1 (37.2) hours in the delayed CAG group. The 180-day survival rates among patients with a CPC of 2 or less were 34.1% (47 of 141) in the emergency CAG group and 30.7% (42 of 138) in the delayed CAG group (hazard ratio [HR], 0.87; 95% CI, 0.65-1.15; P = .32). There was no difference in the overall survival rate at 180 days (emergency CAG, 36.2% [51 of 141] vs delayed CAG, 33.3% [46 of 138]; HR, 0.86; 95% CI, 0.64-1.15; P = .31) and in secondary outcomes between the 2 groups.
Conclusions and Relevance
In this randomized clinical trial, for patients who experience an OHCA without ST-segment elevation on ECG, a strategy of emergency CAG was not better than a strategy of delayed CAG with respect to 180-day survival rate and minimal neurologic sequelae.
Trial Registration
ClinicalTrials.gov Identifier: NCT02876458
Introduction
Sudden cardiac death remains a major public health issue. The rate of patients discharged from the hospital with no or minimal neurologic sequelae is low, varying between 2% and 18%.1,2,3,4,5 The most common cause of sudden cardiac death is ischemic cardiovascular disease, mostly acute coronary artery occlusion.6,7,8,9 However, selection of patients for an invasive strategy is still debated. Several retrospective studies show that the probability of finding an acute coronary artery lesion during an early coronary angiogram (CAG) is high (70%-80%) if ST-segment elevation on the postresuscitation electrocardiogram (ECG) is present. Therefore, guidelines recommend performing an emergent CAG in survivors of sudden cardiac death with no obvious noncardiac cause of arrest and ST-segment elevation. In patients with cardiac arrest without ST-segment elevation on postresuscitation ECG, the benefit of an emergency CAG is still a matter of debate. In these patients, the rate of acute coronary artery lesion is much lower (15%-20%).8 Three recent randomized studies10,11,12 performed in patients without ST-segment elevation after an out-of-hospital cardiac arrest (OHCA) showed no difference in survival whether CAG was performed early or at a deferred time. However, 1 study excluded patients with nonshockable rhythm10 and the second trial was underpowered.11 There is, therefore, still a need for data from randomized trials on this issue. The Emergency vs Delayed Coronary Angiogram in Survivors of Out-of-Hospital Cardiac Arrest (EMERGE) trial tested the hypothesis that the 180-day survival rate of patients with no or minimal neurologic sequelae who experience OHCA without ST-segment elevation on the postresuscitation ECG and no obvious noncardiac cause of arrest would be improved when they underwent emergency CAG compared with those who received delayed CAG.
Methods
Study Design
The EMERGE trial was a national, multicenter, randomized open-label parallel-group trial, in which survivors of OHCA were randomly assigned (1:1) to either emergency CAG or delayed CAG (within 48 to 96 hours). The trial was conducted from January 19, 2017, to November 23, 2020, and the trial protocol has been published previously.13 Survivors of OHCA are usually comatose after resuscitation, and they cannot provide an informed consent for participation in a trial. From January 2017 to 2019, if proxies were present on the site of cardiac arrest, they were asked to provide an informed consent before inclusion and randomization. Owing to difficulties in obtaining such a consent during prehospital care, an amendment was added to the protocol on September 2019 stating that a signed consent was no longer required for inclusion. The patient and/or family members (or next of kin) were informed as soon as possible, and their consent was sought for research to be continued (Supplements 1, 2, and 3).
The protocol was approved by the Agence Nationale de Sécurité du Médicament et des Produits de Santé and the French Ethics Committee (Comité de Protection des Personnes Ile de France IV). This study followed the Consolidated Standards of Reporting Trials (CONSORT) reporting guidelines.
Population
Eligible for inclusion were patients older than 18 years who experienced an OHCA with return of spontaneous circulation (ROSC), without an obvious noncardiac cause of arrest14 and who were admitted to a center with an intensive care unit and a 24/7 interventional cardiology department. Exclusion criteria were as follows: patients younger than 18 years, those with in-hospital cardiac arrest, patients without ROSC, the presence of ST-segment elevation on the postresuscitation ECG according to European Society of Cardiology guidelines,15 suspected noncardiac etiology, presence of comorbidities with life expectancy of less than 1 year, pregnancy, adults subject to a legal protection measure (guardianship or curatorship), and participation in another interventional trial.
Randomization
In France, management of OHCA is performed by the mobile intensive care unit system, with 1 physician specialized in emergency medicine, 1 nurse, and 1 ambulance driver. Calls for OHCA are received by a physician at the medical dispatch center. The included patients were from 22 French centers and were monitored for up to 180 days. Randomization was performed electronically, with a centralized randomization system (Cleanweb software; Telemedicine Technologies SAS) by the dispatch center. Randomization could also be done at hospital arrival if it had not been performed before. In this case, randomization had to be performed within the hour after the patient’s admission. Patients allocated to the emergency CAG group were transferred directly to the catheterization laboratory. Patients randomly assigned to the delayed CAG group were admitted to the intensive care unit, and a CAG was planned 48 to 96 hours after admission.
Study Outcomes
The primary study outcome was the 180-day survival rate with no or minimal neurologic sequelae. All survivors were evaluated by an independent physician who was blinded to the randomization to assess the Cerebral Performance Category (CPC) level at 180 days. A patient was assigned CPC categories 1 or 2 if they had no or minimal neurologic sequelae 180 days after the event.
Secondary end points were shock, tachycardia, and fibrillation episodes during the first 48 hours after hospital admission, changes in left ventricular ejection fraction between baseline and 180 days as assessed by echocardiogram, major neurologic sequelae defined by CPC score 3 or 4 (assessed at intensive care unit discharge, 90 days, and 180 days), all-cause death, and hospital length of stay. Definitions of the trial outcomes are presented in the eAppendix in Supplement 4.
Management in the Intensive Care Unit
The treatments implemented in the intensive care unit were in accordance with European guidelines, adjusted to each unit’s practices, particularly regarding targeted temperature management (between 32 °C and 36 °C), and hemodynamic management.15 Investigators were allowed to perform a CAG sooner than 48 to 96 hours in the delayed group if the following events appeared: ST-segment elevation or new left bundle branch block on the ECG, shock unresponsive to inotropes, electrical storm (repeated ventricular fibrillation or tachycardia), or new segmental hypokinesia or akinesia on an echocardiogram.
Coronary Angiogram and Percutaneous Coronary Interventions
The CAG and percutaneous coronary intervention (PCI) were performed according to the strategies described in the consensus statement from the European Association for Percutaneous Cardiovascular Interventions/Stent for Life groups.16 In summary, use of the radial approach was encouraged. PCI was performed when a culprit lesion was clearly identified using angiographic criteria (recent occlusion, angiographic features suggesting an unstable lesions). Revascularization of nonculprit lesions was discouraged. The use of drug-eluting stents and preprocedure unfractionated heparin was recommended. The postprocedure regimen included aspirin and an antiplatelet P2Y12 inhibitor.
Follow-up Visits and Data Gathering
The first patient was randomly assigned on January 19, 2017, and the last 6-month follow-up was performed on November 23, 2020. Follow-up was conducted during outpatient clinic visits scheduled at 90 (range, 80-100) days and 180 (range, 170-190) days after cardiac arrest, during a medical consultation in the center which had included the patient. Patients for whom no outpatient visit was possible were contacted by telephone or followed up through contact with the patient’s relatives to determine neurologic status with the CPC score.
Sample Size Calculation
Based on results of the Parisian Region Out-of-Hospital Cardiac Arrest (PROCAT) study,8 the 180-day survival rate with no or minimal sequelae in the delayed CAG group was estimated at 36%. Assuming an absolute difference of 10% between the 2 groups, ie, a 180-day survival rate with CPC 1 or 2 of 46% in the emergency CAG group, with a 2-sided 5% type I error, 80% power, and a 180-day maximum length of follow-up, a total number of 678 patients was required. A total of 970 patients included were planned to account for a 10% lost-to-follow-up rate and a crossover rate of 30%.
Statistical Analysis
All analyses were performed on an intention-to-treat basis. Clinical event rates and other categorical data are summarized as percentages. Continuous data are presented as mean (SD) or as median (IQR). Kaplan-Meier plots were constructed for time-to-event outcomes, with treatment effects estimated using Cox models and results presented as hazard ratios (HRs) with their 95% CIs. A Schoenfeld test was used to check the proportional-hazards assumption. Secondary outcomes are presented with HRs, risk ratios, and 95% CIs. The widths of the CIs have not been adjusted for multiplicity, and any inferences drawn from these intervals may not be reproducible. Analyses were performed using SAS software, version 9.2 (SAS Institute) and R statistical software, version 4.0.2 (R Core Team).
We performed a search for randomized clinical trials comparing early vs delayed CAG in patients who experience cardiac arrest without ST-segment elevation. Databases including PubMed, Embase, and Google Scholar were used to obtain all relevant studies up to October 2021, using key words including “cardiac arrest” and “coronary angiography” or “coronary angiogram” and “randomized controlled trial,” which worked as Medical Subject Headings and free-style words in searching retrieval. All publications containing the relevant key words were included in this study. Risk ratios and corresponding 95% CIs as well of the summary risk ratio (random-effects model) were obtained using the R-package metaphor (metafor 2021), version 3.0-2 (R Foundation). Statistical heterogeneity was tested using the Q statistic generated from the χ2 test and was considered statistically significant for a 2-sided P value < .05. Data were analyzed from November 24, 2020, to July 30, 2021.
Results
Enrollment and Patient Characteristics
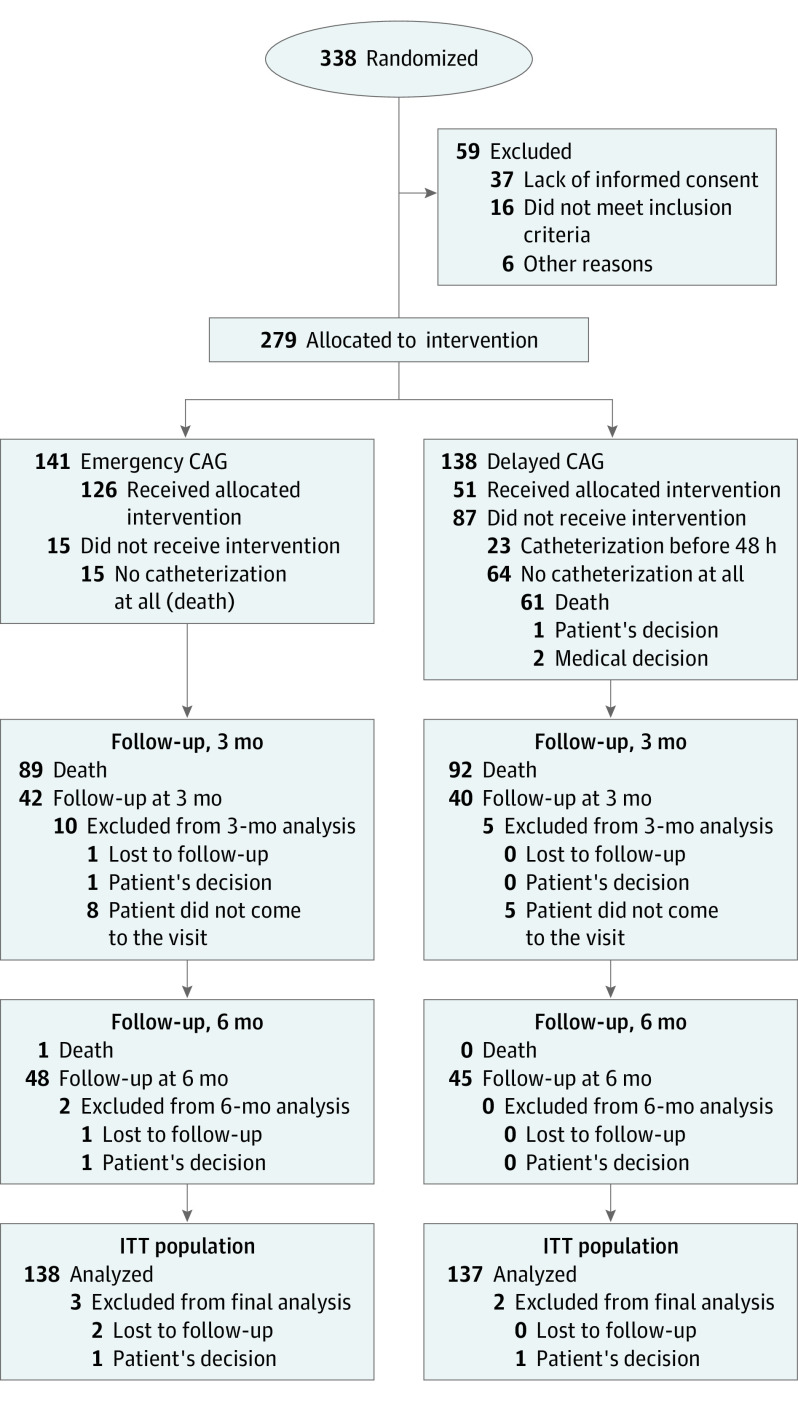
A total of 279 patients (mean [SD] age, 64.7 [14.6] years; 195 men [69.9%]; 84 women [30.1%]) were enrolled during the study period from January 2017 to June 2019. At that time, funding was terminated according to prespecified rules regarding the rate of inclusions, requiring at least 50% of inclusions in 2 years. The enrollment objective was therefore not achieved. A total of 141 patients (50.5%) were randomly assigned to the emergency CAG group, and 138 patients (49.5%) were assigned to the delayed CAG group (Figure 1).
Figure 1. Patient Flow Diagram.
CAG indicates coronary angiogram; ITT, intention to treat.
There were no differences between the 2 groups regarding cardiopulmonary resuscitation characteristics (Table 1). The initial rhythm was nonshockable in 65.2% of patients (90 of 138) in the early CAG group and 69.9% of patients (93 of 133) in the delayed CAG group.
Table 1. Patients’ Characteristics and Procedural Data.
| Variable | Emergency CAG (n = 141) | Delayed CAG (n = 138) |
|---|---|---|
| Age, mean (SD), y | 65.4 (13.8) | 63.9 (15.4) |
| Median (IQR) | 67 (55.0-76.0) | 66.5 (55.0-75.8) |
| Male sex, No./total No. (%) | 103/141 (73.1) | 92/138 (66.7) |
| Female sex, No./total No. (%) | 38/141 (26.9) | 46/138 (33.3) |
| Presence of coronary graft, No./total No. (%) | 4/126 (3.2) | 1/74 (1.4) |
| OHCA witnessed, No./total No. (%) | 125/141 (88.7) | 127/137 (92.7) |
| Bystander CPR, No./total No. (%) | 93/124 (75.0) | 98/123 (79.7) |
| Time from OHCA to basic life support, total No.; median (IQR), min | 118; 3 (1-6) | 113; 2 (1-5) |
| Time from OHCA to ROSC, total No.; median (IQR), min | 127; 27 (16.5-36.5) | 120; 25 (18-35.3) |
| Place of cardiac arrest, No./total No. (%) | ||
| Public place | 58/141 (41.1) | 58/138 (42.0) |
| Home | 73/141 (51.8) | 74/138 (53.6) |
| Other | 10/141 (7.1) | 6/138 (4.4) |
| Initial rhythm, No./total No. (%) | ||
| Nonshockable rhythm | 90/138 (65.2) | 93/133 (69.9) |
| Shockable rhythm | 48/138 (34.8) | 40/133 (30.1) |
| Cumulative dose of IV epinephrine, total No.; median (IQR), mg | 135; 2 (0.0-3.0) | 137; 2 (0.0-4.0) |
| Use of platelet antiaggregant at the time of resuscitation, No./total No. (%) | 17/137 (12.4) | 17/138 (12.3) |
| Use of anticoagulation at the time of resuscitation, No./total No. (%) | 10/137 (7.3) | 13/138 (9.4) |
| GCS after resuscitation, total No.; median (IQR) | 130; 3 (3.0-3.0) | 130; 3 (3.0-3.0) |
| TTM after resuscitation, No./total No. (%) | 130; 52/101 (51.5) | 130; 51/95 (53.7) |
| Signs of potential ischemia on ECG, No./total No. (%) | 66/133 (49.6) | 62/129 (48.1) |
| LVEF at inclusion, total No.; median (IQR), % | 85; 45 (30.0-55.0) | 92; 50 (30.0-60.0) |
| Procedural data | ||
| CAG performed, No./total No. (%) | 126/141 (89.4) | 74/138 (53.6) |
| Reason for not performing CAG, No./total No. (%) | ||
| Unstable hemodynamic status | 0/15 (0.0) | 8/64(12.5) |
| Severe brain damage | 0/15 (0.0) | 13/64 (20.3) |
| Deceased | 7/15 (46.7) | 40/64 (62.5) |
| Other | 8/15 (53.3) | 3/64 (4.7) |
| Time from arrest to CAG, total No.; median (IQR), h | 111; 2 (2-3) | 66; 65.5 (40.8-74.8) |
| Time from randomization to CAG, total No.; median (IQR), h | 123; 1 (1-1) | 72; 63 (39.8-74.5) |
| Urgent CAG before the planned procedure, No./total No. (%) | NA | 23/74 (31.1) |
| Radial/femoral approach, No./total No. (%) | ||
| Radial | 63/125 (50.4) | 56/73 (76.7) |
| Femoral | 60/125 (48.0) | 17/73 (23.3) |
| Combined radial and femoral approach | 2/125 (1.6) | 0/73 (0.0) |
| Unknown | 1/126 | 1/74 |
| Unknown coronary status | 15 (10.6) | 64 (46.4) |
| Severity of coronary artery disease, No./total No. (%) | ||
| No significant disease | 57/126 (45.2) | 41/74 (55.4) |
| 1-Vessel disease | 22/126 (17.5) | 11/74 (14.9) |
| 2-Vessel disease | 26/126 (20.6) | 10/74 (13.5) |
| 3-Vessel disease | 21/126 (16.7) | 12/74 (16.2) |
| Stent previously implanted, No./total No. (%) | 7/126 (5.5) | 4/74 (5.4) |
| Percutaneous coronary intervention, No./total No. (%) | ||
| Yes | 38/126 (30.2) | 17/74 (23.0) |
| No | 88/126 (69.8) | 57/74 (77.0) |
| If not performed, No./total No. (%) | ||
| No significant lesion | 35/88 (39.8) | 20/57 (35.1) |
| No clear culprit lesion | 37/88 (42.1) | 24/57 (42.1) |
| Clear culprit lesion not treated | 1/88 (1.1) | 1/57 (1.8) |
| Other reason | 15/88 (17.1) | 12/57 (21.1) |
| Stent implanted, No./total No. (%) | 35/38 (92.1) | 14/17 (82.4) |
Abbreviations: CAG, coronary angiography; CPR, cardiopulmonary resuscitation; ECG, electrocardiography; GCS, Glasgow Coma Scale score; IV, intravenous; LVEF, left ventricular ejection fraction; MAP, median arterial pressure; NA, not applicable; OHCA, out-of-hospital cardiac arrest; ROSC, return of spontaneous circulation; TTM, targeted temperature management.
Coronary Angiogram
CAG was performed in 126 of 141 patients (89.4%) in the emergency CAG group vs 74 of 138 patients (53.6%) in the delayed CAG group. The mean (SD) time delay between randomization and CAG was 0.6 (3.7) hours in the emergency CAG group and 55.1 (37.2) hours in the delayed CAG group. CAG was performed within a median of 65.5 (IQR, 40.8-74.8) hours after the OHCA in the delayed CAG group compared with 2 (IQR, 2-3) hours in the emergency CAG group. The main reason for not performing CAG in the delayed CAG group was early death from a neurologic cause in 14 patients, a cardiorespiratory etiology in 25 patients, and an unknown cause in 1 patient. In the delayed CAG group, CAG was performed urgently in 23 patients among the 138 patients (16.7%) of this group within 14.7 hours, and 7 underwent a percutaneous intervention. No significant coronary artery disease was found in 45.2% of patients (57 of 126) in the emergency CAG group and in 55.4% of patients (41 of 74) in the delayed CAG group. The rate of PCI among patients who underwent CAG was higher in the emergency CAG group (38 [30.2%]) than in the delayed CAG group (17 [23%]). The main reason for not performing PCI was the absence of significant lesion (35 of 88 patients [39.8%] in the early CAG group and 20 of 57 patients [35.1%] in the delayed CAG group) or the absence of clear culprit lesion (42.1% in both groups).
End Points
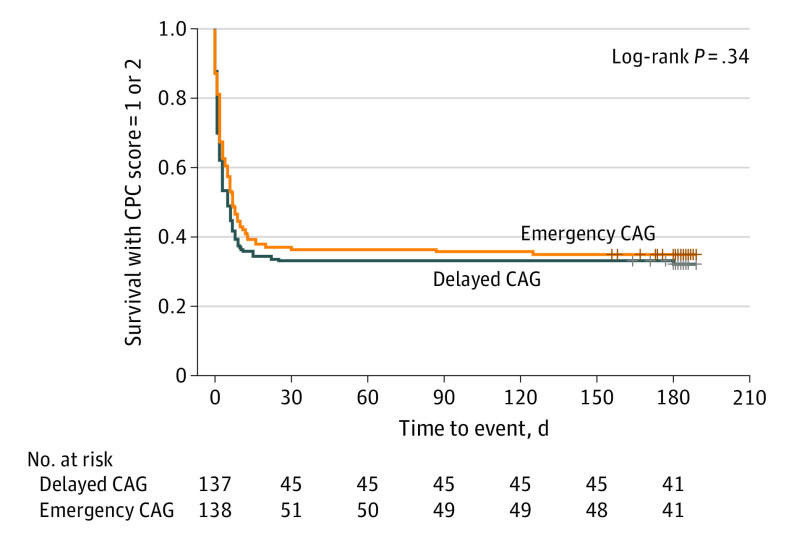
There was no difference in the 180-day survival rates with CPC 1 or 2: 34.1% (47 of 141) in the emergency CAG group and 30.7% (42 of 138) in the delayed CAG group (HR, 0.87; 95% CI, 0.65-1.15; P = .32) (Table 2, Figure 2). There was no difference in the overall survival rate at 180 days (emergency CAG, 36.2% [51 of 141] vs delayed CAG, 33.3% [46 of 138]; HR, 0.86; 95% CI, 0.64-1.15; P = .31) and in secondary outcomes between the 2 groups (Table 2).
Table 2. Outcomes.
| Outcome | Emergency CAG (n = 141) | Delayed CAG (n = 138) | Hazard ratioa,b (95% CI) | P value |
|---|---|---|---|---|
| Primary outcome at 180 d,a,c No./total No. (%) | ||||
| CPC = 1 or 2 | 47/141 (34.1) | 42/138 (30.7) | 0.87 (0.65-1.15) | .32 |
| CPC = 3, 4, or 5 | 91/141 (65.9) | 95/138 (69.3) | ||
| Unknown CPC status | 3/141 (2.1) | 1/138 (0.7) | ||
| Secondary outcomes | ||||
| Overall survival rate at 180 da | 51/141(36.2) | 46/138 (33.3) | 0.86 (0.64-1.15) | .31 |
| At ICU discharge,a No./total No. (%) | ||||
| CPC = 1 or 2 | 43/141(30.5) | 40/138 (29.0) | 0.85 (0.64-1.13) | .25 |
| CPC = 3, 4, or 5 | 92/141 (65.2) | 96/138 (69.6) | ||
| Unknown CPC status | 6/141 (4.3) | 2/138 (1.5) | ||
| Secondary outcome at 90 d,a No./total No. (%) | ||||
| CPC = 1 or 2 | 40/141 (28.4) | 34/138 (24.6) | 0.86 (0.64-1.14) | .29 |
| CPC = 3, 4, or 5 | 92/141 (65.2) | 94/138 (68.1) | ||
| Unknown CPC status | 9/141 (6.4) | 0/138 (0.0) | ||
| Occurrence of shock during, No./total No. (%) | ||||
| First 48 hb | 50/129 (38.8) | 53/133 (39.8) | 1.03 (0.76-1.39) | .86 |
| Death before 48 h | 9/129 (7.0) | 5/133 (3.8) | ||
| Unknown | 2/129 (1.6) | 0/133 (0.0) | ||
| Occurrence of VT/VF during the first 48 h, No. (%)b | 10/141 (7.1) | 5/138 (3.6) | 0.51(0.18-1.46) | .21 |
| LVEF at 180 d, median, (IQR), % | 60 (50.0-63.0) | 57.5 (51.0-6.00) | NA | .26 |
| Evolution of LVEFd from baseline to 180 d, median (IQR), % | 10.5 (0-24.0) | 9.5 (1.5-18.0) | NA | .94e |
| Length of hospital stay, median (IQR), d | 7 (2.0-13.0) | 5 (1.0-11.0) | NA | .75 |
| Withdrawal of care,b No./total No. (%) | 56/141 (39.7) | 65/138 (47.1) | 1.19 (0.91-1.55) | .22 |
Abbreviations: CAG, coronary angiography; CPC, Cerebral Performance Category; ICU, intensive care unit; LVEF, left ventricular ejection fraction; NA, not available; VF, ventricular fibrillation; VT, ventricular tachycardia.
Hazard ratio estimated by survival model.
Risk ratio estimated by log-binomial regression model.
The distribution of the primary end point was as follows: in emergency group CPC = 1: 41/141 (29.7), CPC = 2:6/141 (4.3), CPC = 3:1 /141 (0.7), CPC = 4: 0/141 (0.0), and CPC = 5: 90/141(63.8); in delayed group CPC = 1 41/138 (29.9), CPC = 2: 1/138 (0.7), CPC = 3: 3/138 (2.1), CPC = 4: 0/138 (0.0), and CPC = 5: 92/138 (66.7).
Difference between LVEF at 6 months and LVEF at baseline.
Analysis of covariance test.
Figure 2. Patient Survival With a Cerebral Performance Category (CPC) Score of 1 or 2.
CAG indicates coronary angiogram.
Meta-analysis
The search for randomized clinical trials comparing early vs delayed CAG in patients who experienced cardiac arrest without ST-segment elevation found 3 studies10,11,12 published in the literature, performed with similar methods. We summarized their results with those of the EMERGE study. As shown in Figure 3, the results of this combined analysis, comprising a total of 1446 patients, are in accordance with those of the EMERGE study alone, showing no benefit of an early CAG in this population.
Figure 3. Summary of Randomized Clinical Trials Comparing Emergency vs Delayed Coronary Angiogram (CAG) in Patients With Cardiac Arrest Without ST-Segment Elevation .
COACT indicates Coronary Angiography After Cardiac Arrest; EMERGE, Emergency vs Delayed Coronary Angiogram in Survivors of Out-of-Hospital Cardiac Arrest; PEARL, Randomized Pilot Clinical Trial of Early Coronary Angiography vs No Early Coronary Angiography for Postcardiac Arrest Patients Without ST-Segment Elevation; TOMAHAWK, Immediate Unselected Coronary Angiography vs Delayed Triage in Survivors of Out-of-Hospital Cardiac Arrest Without ST-Segment Elevation.
Discussion
In the EMERGE randomized multicenter clinical trial, 2 strategies were compared: emergency CAG vs delayed CAG, in survivors of an OHCA with no obvious noncardiac cause of arrest and no ST-segment elevation on ECG. No significant difference was found between the 2 groups in the 180-day survival rate with CPC 1 or 2. However, the study patient enrollment target goal was not achieved, and thus, the EMERGE study was underpowered to adequately assess the primary end point.
Nonrandomized studies suggested a benefit of an emergency invasive strategy in survivors of OHCA regardless of ECG findings. Spaulding et al7 showed that coronary artery disease was the cause of more than 70% of OHCA and that performing an emergency CAG seemed to improve patient outcome. The Larsen and Ravkilde meta-analysis17 showed that the prevalence of an acute coronary artery lesion ranged from 59% to 71% in patients with OHCA and without an obvious noncardiac cause of arrest. In 2015, the European Resuscitation Council and European Society of Intensive Care Medicine defined in their guidelines for postresuscitation care that all survivors of OHCA should be sent to a specialized center that can perform an emergent CAG in the event of an ST-segment elevation and can consider performance of a CAG as soon as possible in the event of a non–ST-segment elevation.15 However, several recent studies have challenged those recommendations in survivors of an OHCA without ST-segment elevation on the post-ROSC ECG, who represent the majority of OHCA survivors.18
Our results are in line with 3 recent randomized clinical trials, even though the Coronary Angiography After Cardiac Arrest (COACT) trial used survival at 90 days as a primary end point,10 the Randomized Pilot Clinical Trial of Early Coronary Angiography vs No Early Coronary Angiography for Postcardiac Arrest Patients Without ST-Segment Elevation (PEARL) study used a composite primary outcome,11 and the Immediate Unselected Coronary Angiography vs Delayed Triage in Survivors of Out-of-Hospital Cardiac Arrest Without ST-Segment Elevation (TOMAHAWK) study used all-cause death at 30 days as the primary end point.12 Because these studies were performed with similar methods, we summarized their results with those of the EMERGE study. The combined analysis on 1446 patients shows no benefit of an early strategy.
Our study adds new insights as EMERGE included mostly patients with a nonshockable rhythm at presentation. These patients were excluded from randomization in the COACT trial and represented only 23.5% of patients in the PEARL study. The EMERGE trial randomized a nonselected population that did not exclude patients with shock after ROSC in contrast to COACT, which could have lowered the expected benefit of an emergency CAG. Indeed, patients with cardiogenic shock and myocardial infarction are known to benefit the most from early revascularization.19,20 Of note, in France, the first medical evaluation is done very early in the course of the OHCA, as the first aid unit is led by a physician. Data from EMERGE suggest that in this specific population, even when medical evaluation and randomization between emergency vs delayed CAG is done early, a direct admission to a catheterization laboratory is debatable and may challenge strategy in terms of organization and resources. The absence of superiority of emergency CAG has important consequences on the organization of the chain of survival.
Future research should focus on identifying the subgroup of patients who experience an OHCA with non–ST-segment elevation who would benefit from an early CAG. The PROCAT 2 cohort study21 found that younger age, a shorter resuscitation length (<20 minutes), an initial shockable rhythm, and low-dose epinephrine were significantly associated with a better neurologic outcome after a PCI in an OHCA population with non–ST segment elevation on ECG after ROSC but that the subgroup of older men (50 years or older) resuscitated from an initially shockable rhythm mostly benefitted from an emergent CAG strategy.
In randomized trials, patients are selected based on predefined variables but not on clinical history, warning signs, and OHCA circumstances. A meta-analysis of the 3 randomized trials and EMERGE, based on such individual factors, could help to define more precisely the subpopulation that could benefit from an early CAG after an OHCA without an extracardiac cause and without ST-segment elevation on ECG. These patients could be triaged and admitted directly to the catheterization laboratory. However, if CAG and PCI can benefit a specific population, the best timing of this procedure remains unknown.
In EMERGE, the main reason for not performing PCI was the absence of a significant or clear culprit lesion. Fifty percent of patients presented with potential signs of ischemia on the ECG. Ischemic ECG changes alone seem to be a poor marker of the presence of a culprit lesion.22The absence of benefit of an emergent CAG strategy may, therefore, be attributable to the low rate of acute lesions. Furthermore, the main cause of death in OHCA is neurologic damage. Of interest, the higher rate of PCI in the emergency CAG group suggests that death occurred in patients with significant coronary artery disease despite emergency CAG and PCI when indicated. Future research could focus on diagnostic methods and algorithms to select survivors of OHCA with no ST-segment elevation who present with unstable lesions and who may benefit from an early invasive strategy. A strategy using ECG findings has been proposed but has not been evaluated on a large number of patients.23
Limitations
This study had several limitations. Preplanned sample size was not achieved, and thus, the EMERGE study was underpowered to adequately assess the primary and secondary end points. Overestimation of the number of cardiac arrests meeting the inclusion and exclusion criteria and difficulties in obtaining informed consent were the main reasons for not achieving the preplanned sample size. Similar studies are ongoing and are having similar recruitment difficulties.24,25 The physicians were not blinded to randomized treatment allocation but were not involved in the research process. Determination of culprit lesions is subjective, and the angiograms were not analyzed by a core laboratory. Patients with refractory cardiac arrest were excluded, and therefore, no conclusion on the value of CAG in this group of patients can be drawn. Finally, the echocardiograms and the follow-up visits, including neurologic assessment, were not evaluated by a core laboratory.
Conclusions
In this randomized clinical trial of patients successfully resuscitated after an OHCA without ST-segment elevation, a strategy of emergency CAG was not found to be better than a strategy of delayed CAG with respect to the 180-day survival rate with no or minimal neurologic sequelae. The study patient enrollment goal was not achieved, and therefore, the study was underpowered to adequately assess the primary and secondary end points. However, our results are consistent with previously published studies and do not support emergency CAG in survivors of OHCA without ST elevation.
EMERGE Protocol
EMERGE Modifications
EMERGE Statistical Analysis Plan
eAppendix. Study Outcomes Definitions
Nonauthor Collaborators
Data Sharing Statement
References
- 1.Lloyd-Jones D, Adams RJ, Brown TM, et al. ; WRITING GROUP MEMBERS; American Heart Association Statistics Committee and Stroke Statistics Subcommittee . Heart disease and stroke statistics—2010 update: a report from the American Heart Association. Circulation. 2010;121(7):e46-e215. doi: 10.1161/CIRCULATIONAHA.109.192667 [DOI] [PubMed] [Google Scholar]
- 2.Writing Group Members; Mozaffarian D, Benjamin EJ, Go AS, et al. ; American Heart Association Statistics Committee; Stroke Statistics Subcommittee . Heart disease and stroke statistics—2016 update: a report from the American Heart Association. Circulation. 2016;133(4):e38-e360. doi: 10.1161/CIR.0000000000000350 [DOI] [PubMed] [Google Scholar]
- 3.Kiguchi T, Okubo M, Nishiyama C, et al. Out-of-hospital cardiac arrest across the world: first report from the International Liaison Committee on Resuscitation (ILCOR). Resuscitation. 2020;152:39-49. doi: 10.1016/j.resuscitation.2020.02.044 [DOI] [PubMed] [Google Scholar]
- 4.Nichol G, Baker D. The epidemiology of sudden death. In: Paradis NA, Halperin HR, Kern KB, Wenzel V, Chamberlain DA, eds. Cardiac Arrest—The Science and Practice of Resuscitation Medicine. Cambridge University Press; 2007:26-48. [Google Scholar]
- 5.Sasson C, Rogers MAMM, Dahl J, Kellermann AL. Predictors of survival from out-of-hospital cardiac arrest: a systematic review and meta-analysis. Circ Cardiovasc Qual Outcomes. 2010;3(1):63-81. doi: 10.1161/CIRCOUTCOMES.109.889576 [DOI] [PubMed] [Google Scholar]
- 6.Byrne R, Constant O, Smyth Y, et al. Multiple source surveillance incidence and aetiology of out-of-hospital sudden cardiac death in a rural population in the West of Ireland. Eur Heart J. 2008;29(11):1418-1423. doi: 10.1093/eurheartj/ehn155 [DOI] [PubMed] [Google Scholar]
- 7.Spaulding CM, Joly LM, Rosenberg A, et al. Immediate coronary angiography in survivors of out-of-hospital cardiac arrest. N Engl J Med. 1997;336(23):1629-1633. doi: 10.1056/NEJM199706053362302 [DOI] [PubMed] [Google Scholar]
- 8.Dumas F, Cariou A, Manzo-Silberman S, et al. Immediate percutaneous coronary intervention is associated with better survival after out-of-hospital cardiac arrest: insights from the PROCAT (Parisian Region Out of Hospital Cardiac Arrest) registry. Circ Cardiovasc Interv. 2010;3(3):200-207. doi: 10.1161/CIRCINTERVENTIONS.109.913665 [DOI] [PubMed] [Google Scholar]
- 9.Deo R, Albert CM. Epidemiology and genetics of sudden cardiac death. Circulation. 2012;125(4):620-637. doi: 10.1161/CIRCULATIONAHA.111.023838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lemkes JS, Janssens GN, van der Hoeven NW, et al. Coronary angiography after cardiac arrest without ST-segment elevation. N Engl J Med. 2019;380(15):1397-1407. doi: 10.1056/NEJMoa1816897 [DOI] [PubMed] [Google Scholar]
- 11.Kern KB, Radsel P, Jentzer JC, et al. Randomized pilot clinical trial of early coronary angiography vs no early coronary angiography after cardiac arrest without ST-segment elevation: the PEARL study. Circulation. 2020;142(21):2002-2012. doi: 10.1161/CIRCULATIONAHA.120.049569 [DOI] [PubMed] [Google Scholar]
- 12.Desch S, Freund A, Akin I, et al. ; TOMAHAWK Investigators . Angiography after out-of-hospital cardiac arrest without ST-segment elevation. N Engl J Med. 2021;385(27):2544-2553. doi: 10.1056/NEJMoa2101909 [DOI] [PubMed] [Google Scholar]
- 13.Hauw-Berlemont C, Lamhaut L, Diehl JL, et al. ; EMERGE Investigators . EMERGEncy vs delayed coronary angiogram in survivors of out-of-hospital cardiac arrest with no obvious non-cardiac cause of arrest: design of the EMERGE trial. Am Heart J. 2020;222:131-138. doi: 10.1016/j.ahj.2020.01.006 [DOI] [PubMed] [Google Scholar]
- 14.Myat A, Song KJ, Rea T. Out-of-hospital cardiac arrest: current concepts. Lancet. 2018;391(10124):970-979. doi: 10.1016/S0140-6736(18)30472-0 [DOI] [PubMed] [Google Scholar]
- 15.Ibanez B, James S, Agewall S, et al. ; ESC Scientific Document Group . 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: the task force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39(2):119-177. doi: 10.1093/eurheartj/ehx393 [DOI] [PubMed] [Google Scholar]
- 16.Noc M, Fajadet J, Lassen JF, et al. ; European Association for Percutaneous Cardiovascular Interventions (EAPCI); Stent for Life (SFL) Group . Invasive coronary treatment strategies for out-of-hospital cardiac arrest: a consensus statement from the European Association for Percutaneous Cardiovascular Interventions (EAPCI)/Stent for Life (SFL) groups. EuroIntervention. 2014;10(1):31-37. doi: 10.4244/EIJV10I1A7 [DOI] [PubMed] [Google Scholar]
- 17.Larsen JM, Ravkilde J. Acute coronary angiography in patients resuscitated from out-of-hospital cardiac arrest—a systematic review and meta-analysis. Resuscitation. 2012;83(12):1427-1433. doi: 10.1016/j.resuscitation.2012.08.337 [DOI] [PubMed] [Google Scholar]
- 18.Windecker S, Kolh P, Alfonso F, et al. ; Authors/Task Force members . 2014 ESC/EACTS guidelines on myocardial revascularization: the task force on myocardial revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS) developed with the special contribution of the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J. 2014;35(37):2541-2619. doi: 10.1093/eurheartj/ehu278 [DOI] [PubMed] [Google Scholar]
- 19.Hollenbeck RD, McPherson JA, Mooney MR, et al. Early cardiac catheterization is associated with improved survival in comatose survivors of cardiac arrest without STEMI. Resuscitation. 2014;85(1):88-95. doi: 10.1016/j.resuscitation.2013.07.027 [DOI] [PubMed] [Google Scholar]
- 20.Hochman JS, Sleeper LA, Webb JG, et al. ; SHOCK Investigators . Early revascularization and long-term survival in cardiogenic shock complicating acute myocardial infarction. JAMA. 2006;295(21):2511-2515. doi: 10.1001/jama.295.21.2511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dumas F, Bougouin W, Geri G, et al. Emergency percutaneous coronary intervention in postcardiac arrest patients without ST-segment elevation pattern: insights from the PROCAT II registry. JACC Cardiovasc Interv. 2016;9(10):1011-1018. doi: 10.1016/j.jcin.2016.02.001 [DOI] [PubMed] [Google Scholar]
- 22.Baldi E, Schnaubelt S, Caputo ML, et al. Association of timing of electrocardiogram acquisition after return of spontaneous circulation with coronary angiography findings in patients with out-of-hospital cardiac arrest. JAMA Netw Open. 2021;4(1):e2032875. doi: 10.1001/jamanetworkopen.2020.32875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sideris G, Voicu S, Dillinger JG, et al. Value of postresuscitation electrocardiogram in the diagnosis of acute myocardial infarction in out-of-hospital cardiac arrest patients. Resuscitation. 2011;82(9):1148-1153. doi: 10.1016/j.resuscitation.2011.04.023 [DOI] [PubMed] [Google Scholar]
- 24.Direct or subacute coronary angiography in out-of-hospital cardiac arrest (DISCO). ClinicalTrials.gov identifier: NCT02309151. Updated January 18, 2020. Accessed September 17, 2020. https://clinicaltrials.gov/ct2/show/NCT02309151
- 25.A randomised trial of expedited transfer to a cardiac arrest centre for non-ST elevation out-of-hospital cardiac arrest (ARREST). ClinicalTrials.gov identifier: NCT03872960. Updated July 22, 2020. Accessed September 10, 2020. https://clinicaltrials.gov/ct2/show/NCT03872960
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
EMERGE Protocol
EMERGE Modifications
EMERGE Statistical Analysis Plan
eAppendix. Study Outcomes Definitions
Nonauthor Collaborators
Data Sharing Statement