Abstract
Nitric oxide, carbon monoxide and hydrogen sulfide are three endogenous gasotransmitters that serve a role in regulating normal and pathological cellular activities. They can stimulate or inhibit cancer cell proliferation and invasion, as well as interfere with cancer cell responses to drug treatments. Understanding the molecular pathways governing the interactions between these gases and the tumor microenvironment can be utilized for the identification of a novel technique to disrupt cancer cell interactions and may contribute to the conception of effective and safe cancer therapy strategies. The present review discusses the effects of these gases in modulating the action of chemotherapies, as well as prospective pharmacological and therapeutic interfering approaches. A deeper knowledge of the mechanisms that underpin the cellular and pharmacological effects, as well as interactions, of each of the three gases could pave the way for therapeutic treatments and translational research.
Keywords: gasotransmitter, NO, H2S, TME, cancer chemotherapy
1. Introduction
Cancer is one of the most dreaded diseases and is a major threat to human life. Among different clinical disorders, cancer is the second most common cause of death after cardiovascular diseases (1). Different approaches and strategies, such as chemotherapy, radiotherapy, surgery, immunotherapy and small molecule-targeted therapy, have been studied and applied to target and treat cancer (2,3).
Chemotherapeutic drugs work by targeting fast-growing and proliferating cells, leading to cell death and shrinking of the tumors. The conventional cancer chemotherapy, ‘the standard treatment’, is not always successful, even after 50–100 years of research and clinical experience, although cases of lymphocytic leukemia and Hodgkin's lymphoma have been treated successfully in this manner (1). Conventional chemotherapy indiscriminately delivers the toxic anticancer agent to tumors and normal tissues simultaneously (4). Therefore, cancer-selective drug delivery approaches are required to avoid undesirable systemic side effects. One way of tackling these problems is to deliver anticancer drugs selectively to the tumor site (5). One of the different approaches is using gasotransmitters to selectively provide anticancer drugs to the tumor site (6).
The three small, diffusible gaseous mediators nitric oxide (NO), carbon monoxide (CO) and hydrogen sulfide (H2S) serve multiple roles in normal physiology and the pathogenesis of numerous diseases. Several studies have emphasized the roles of NO, CO and H2S in cancer (7–10); however, there are numerous puzzles and controversies. Some studies have demonstrated that these mediators are pro-tumorigenic, while others have reported that they have an antitumorigenic effect (11–13). It is now recognized that these three gases exhibit bell-shaped (also termed ‘biphasic’, ‘bimodal’ or ‘Janus-faced’) pharmacological characteristics in cancer (6). An improved understanding of the complicated pharmacological nature of these mediators has far-reaching consequences. It also tackles some of the difficulties of the field, enabling the development of novel therapeutic techniques based on pharmacologically suppressing mediator production (6). The present review discusses the important roles of NO, CO and H2S in tumor pathophysiology, addressing how different levels of these gases can affect tumor growth, angiogenesis and survival. Furthermore, it highlights the potential therapeutic value of the gasotransmitters in cancer chemotherapy.
2. Chemotherapy
History of chemotherapy
The use of chemicals to treat a disease is called chemotherapy. This therapeutic model was conceptually born in the early 20th century when the German physician Paul Ehrlich adopted chemicals to treat infectious diseases (14,15). Ehrlich stepped into the field of oncology with great ambition, trying to explore de novo pharmacological bullets to shoot cancer cells (16). The net findings of all his experiments were disappointing since none of the proposed drugs worked on cancer cells (17).
Cancer chemotherapy remained indistinct for >30 years, and scientists continued to follow Ehlrich's fishing strategy after his death. Certain researchers studied the effect of mustard gas or its derivatives on bone marrow eradication; an idea that was obtained from using the gases during the First World War (18,19). Others, such as Sidney Farber, used anti-folates, such as aminopterin and 6-mercaptopurine, to treat childhood cancers (20). In 1950, 6-mercaptopurine was selected for a clinical trial investigating the treatment of acute lymphatic leukemia in children. Despite the promising initial results leading to cancer remission, all investigated chemicals had significant adverse effects indicated by quick relapse a few weeks after treatment (21). The chemotherapeutic drug screening mission was continued. By 1964, ~215,000 chemicals, plant derivatives and fermentation products were studied, and several million mice were included in these studies (22). The challenges encountered in the discovery and delivery of the proper anticancer chemotherapeutics were developing a convenient model to reduce the vast repertoire of chemicals into a considerable list that could have efficiency against cancer, obtaining suitable funds to support the suggested studies and treatment modalities, and admission to clinical facilities to examine the impact of the selected substances. Therefore, different organizations, funding agencies and research centers were established to support scientists and oncologists economically, in order to defeat cancer.
After all these chemotherapeutic screening failures, scientists turned the view back, asking what makes cancer cells switch their response to treatment from sensitive to resistant. Scientists examined if it would be better to employ dual chemotherapy rather than the conventional monotherapy approach used, and this idea of using multiple chemical combinations immediately appeared promising. Freireich et al (23) were the first scientists who combined a four-drug regime (vincristine, amethopterin, mercaptopurine and prednisone) to treat leukemia in children. Despite full cancer remission for several months, they observed severe brain metastasis and death, and thus, stopped this chemotherapeutic regimen. The outcome of tetra-combinatorial therapeutic approaches, including mechlorethamine, oncovin, procarbazine and prednisone (MOPP), and mechlorethamine, oncovin, methotrexate and prednisone, in treating Hodgkin's diseases was surprising, as the complete remission rate increased to 80% in the USA (24). Furthermore, ~60% of patients with Hodgkin's treated with MOPP never relapsed (25). MOPP, ‘the miracle’, made the concept of cancer curability possible. Indications from combination chemotherapies in treating certain types of advanced hematological malignancies motivated scientists to consider a similar therapeutic regime for solid tumors; however, the primary method for treating solid tumors was surgery (26). By the early 1970s, the adjuvant chemotherapy approach was introduced, where chemotherapy was used after surgery to target microscopic tumors and reduce cancer recurrence (26). Bonadonna et al (27) introduced the first combinational chemotherapeutic-postoperative approach, called cyclophosphamide, methotrexate, fluorouracil-adjuvant therapy, to treat early-stage breast cancer in women. The concept of combinational adjuvant chemotherapy was popular in the USA. Fisher et al (28) examined L-phenylalanine mustard to target breast cancer and other solid tumors, such as colorectal cancer. Depending on the type and size of the tumor, an additional approach, called neoadjuvant chemotherapy, is currently used. In this approach, chemotherapy is applied before the surgery or the primary therapy (29).
Most, if not all, solid tumors acquire drug resistance after a few cycles of chemotherapy, and thus, an efficient chemotherapeutic approach has not been developed yet. This is mainly due to dynamic phenotypic and genotypic changes in cancer cells and their surrounding microenvironment. Despite the common non-curative effect of chemotherapy, the disease progression-free survival curves have been markedly improved (30). Any effective therapeutic approach requires systematic knowledge regarding the drug's mechanism of action, primary pharmacologic metabolites, the differences in pharmacokinetics and pharmacodynamics, and the behavior of cancer cells and their crosstalk with the tumor microenvironment (TME) (31). This knowledge has markedly progressed during the last 20–30 years upon the emergence of novel technical avenues in genomic and proteomic analysis. As a result, novel treatment modalities, such as immunotherapy and targeted therapy, have been introduced and suggested to be applied either separately or in combination with chemotherapy.
Mechanism of action and classification of chemotherapy
Chemotherapeutic drugs are clustered into subgroups according to their structure and overall mechanisms of action. Each subgroup is subdivided into several cytostatic drugs, which are used to treat different types of cancer (32). Table SI lists the most prominent types of drugs, their mechanism of action, the targeted cancer types and the number of clinical trials for each drug.
Microenvironment and chemotherapy
In non-hematological malignancy, a tumor is a disorganized, miscommunicated aberrant tissue, where tumor cells are surrounded by stroma and they all interact unsystematically within one unit. The stroma consists of cellular and non-cellular compartments, and altogether they are referred to as the TME. The TME is made up of different types of cells, such as cancer-associated fibroblasts (CAFs), tumor-associated macrophages (TAMs), different sub-types of anti- and pro-inflammatory immune cells, adipocytes and tumor-associated vasculature (endothelial cells and pericytes), and extracellular matrix (ECM) (33). These compartments interact with each other and with tumor cells, initiating various biochemical and cellular signals, which drive cancer cell proliferation, invasion and the response to treatment (34). Chemotherapy eliminates and reduces tumor growth primarily, whereas a small population of cancer cells shift their survival machinery and do not respond to the treatment, as they become more aggressive cells, which serve as the source of relapse. The TME has the potential to drive the anti-chemotherapeutic effect of cancer cells by interfering with different survival mechanisms and cellular signaling pathways (35). This is evident in different types of cancer, such as breast and ovarian cancer, in which enriched TME signatures associated with a treatment-resistant phenotype are observed (34). Among the different signatures, the hypoxic nature of the TME decreases the proliferation rate and induces survival of cancer cells, thus reducing their response to chemotherapy (36–38). The hypoxic TME triggers angiogenic switch by inducing aberrant blood vessel formation in cancer, and due to the leaky properties of cancer-associated vasculature, the drugs that circulate in the blood will not be delivered efficiently to the core of the tumor (39,40). Additionally, the pharmacokinetic action of certain chemotherapeutic drugs depends on the availability of free radicals. Therefore, the cytotoxic activity of those drugs is reduced in the absence or presence of low oxygen (O2) levels (40,41).
The architecture of the TME, characterized by its phenotypic plasticity and heterogenic properties, is essential to allow or prevent drug delivery to the tumor (42). The reorganization of the ECM due to the interaction of cancer cells with CAFs and TAMs leads to drug sequestration, preventing them from reaching the cancer cells (34,43,44).
3. Gasotransmitters
In the last decades, three gaseous molecules have been identified as gasotransmitters: NO, CO and H2S. These particular gases are similar to each other in their production and function, but exert their functions in unique ways in the human body (45). NO is produced endogenously in endothelial cells from L-Arg by a family of enzymes, called NO synthases (NOS), in the vasculature, which modulates vascular tone by activating soluble guanylyl cyclase (sGC) enzyme and producing cyclic GMP (46). Endogenous CO is produced by the enzyme heme oxygenase (HO), which converts free heme to biliverdin (47). CO has a vasorelaxant and an antiproliferative action on vascular smooth muscles cells (VSMCs), making it an important determinant of vascular tone in several pathophysiological conditions (48). H2S is produced endogenously in mammalian tissues from L-cysteine by cystathionine-β-synthase (CBS), cystathionine γ-lyase and another mitochondrial enzyme, 3-mercaptopyruvate sulfurtransferase (49). It regulates vascular diameter, and protects the endothelium from oxidative stress, ischemia reperfusion injury and chronic inflammation by activating several K+ channels in VSMCs (50,51). According to Wang et al (52), other molecules, such as sulfur dioxide, methane, hydrogen gas, ammonia and carbon dioxide, are also considered to be potential gasotransmitter candidates, despite the fact that they have not been adequately explored or do not completely fit the diagnostic criteria for endogenous gasotransmitters.
History of gasotransmitters
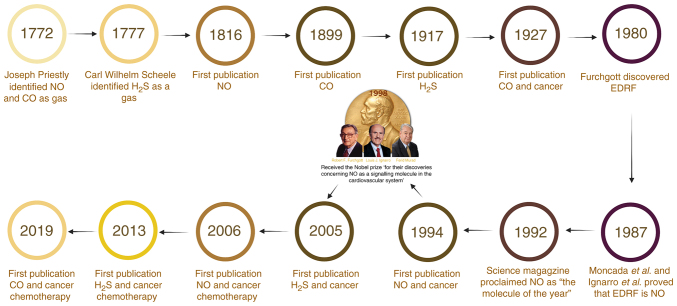
NO was discovered in 1772 by Joseph Priestley as a clear, colorless gas with a half-life of 6–10 sec (53). In 1979, Gruetter et al (54) found that adding NO in a mixture with nitrogen or argon gases into an organ bath vessel containing isolated pre-contracted strips of a bovine coronary artery induces vascular smooth muscle relaxation. In 1980, Furchgott and Zawadzki (55) revealed that endothelial cells produce endothelium-derived relaxing factor (EDRF) in response to stimulation by acetylcholine in vessels with intact endothelium. After 7 years and in two unrelated studies, both Ignarro et al (56,57) and Palmer et al (56,57) demonstrated that EDRF is NO. Moncada et al (58) demonstrated that NO is synthesized from the amino acid L-arginine. Earlier, Murad et al (59) reported that nitro vasodilators, such as nitroglycerin (GTN) and sodium nitroprusside, induce vascular tissue relaxation, stimulate sGC expression and increase cGMP levels in tissues. All these studies contributed to the establishment of a signaling molecule in the cardiovascular system. In 1992, the cover of Science magazine proclaimed NO as the molecule of the year (60). Furthermore, 6 years later, Pfizer, Inc. introduced Viagra, a drug that inhibits phosphodiesterase-5 via the NO-cGMP signaling cascade, which revolutionized the management of erectile dysfunction (61). In the same year, the importance of the NO discovery was acknowledged by awarding the Nobel Prize in Physiology and Medicine to Furchgott, Ignarro and Murad (62).
Few discoveries have had the type of impact on biology that NO has had since it was discovered (63). The first scientific article described NO in 1816 (64), while in 1994, Thomsen, et al (48,65) were the first to report a link between NO and cancer action. In 1993, there were >1,000 new publications on the biology of NO. At the end of the 20th century, the rate of NO publications approached a plateau at ~6,000 papers per year, spanning almost every area of biomedicine (63). The number of published articles in PubMed (pubmed.ncbi.nlm.nih.gov/) reached 58,848 by the end of 2020.
In the late 1200s, a poisonous gas produced by the incomplete combustion of wood similar to CO was described by the Spanish alchemist Arnold of Villanova (66). Between 1772 and 1799, an English chemist, Joseph Priestley, recognized and characterized CO (53). The first scientific article described CO in 1899 (67), and subsequently, the ‘first paper linking CO to cancer was published in 1927 (68). Between 1920 and 1960, Roughton performed several kinetic studies on CO and hemoglobin (69–71). In 1944, he revealed that CO bound to hemoglobin changed the oxyhemoglobin dissociation curve, and demonstrated that CO was produced in the body during the metabolism of the hemoglobin molecule (72). Subsequently, Tenhunen et al (73) described and characterized HO as the enzyme responsible for breaking down heme in the body, demonstrating that heme catalysis resulted in the subsequent release of CO and free iron as by-products.
H2S was first discovered in 1777 by Carl Wilhelm Scheele (74), and the first paper related to H2S was published in 1917 (75). The importance of H2S in cell physiology was highlighted in the mid-1990s, and the first link between H2S and cancer was reported in 2005. H2S at physiological concentrations can reduce the apoptotic effects of chemopreventative drugs and play an important role in the response of colonic epithelial cells of the human adenocarcinoma cell line HCT116 to both beneficial and toxic chemicals (76). It is clear that H2S, similar to other endogenous gases, has now been identified as a gasotransmitter (77). It was initially regarded as highly poisonous in the environment; however, this perception has changed as a growing number of studies have illustrated H2S as a cytoprotective and cardioprotective agent (78,79). Fig. 1 depicts a timeline of important scientific developments in the history of gasotransmitter research and therapeutic usage.
Figure 1.
Timeline of key scientific advances during the history of gasotransmitters research and its therapeutic use. CO, carbon monoxide; EDRF, endothelium-derived relaxing factor; H2S, hydrogen sulfide; NO, nitric oxide.
Role of NO in cancer
NO is a small biomolecule that exerts different effects on tumor growth and invasion. It is a pleiotropic regulator and serves essential roles in various intercellular or intracellular processes, including vasodilatation, neurotransmission and macrophage-mediated immunity (7). Vascular endothelial cells can synthesize NO from L-arginine, and this biosynthetic pathway has been thoroughly documented in numerous other cell types, including nervous and immune cells (80,81). It can display a cytotoxic property at higher concentrations as generated by activated macrophages and endothelial cells (7). A total of three different isoforms of the NOS family synthases have been identified: Endothelial NOS (eNOS), neuronal NOS (nNOS) and inducible NOS (iNOS). The gene symbol nomenclatures are NOS1 for nNOS, NOS2 for iNOS and NOS3 for eNOS (7). However, the role of NO in cancer biology, particularly in breast cancer, only started to be elucidated in 1994 (82). It has been detected that NOS expression is increased in various types of cancer, such as breast, cervical, brain, laryngeal, and head and neck cancer (83) (Table I). NO exhibits a pro- or antitumorigenic effect (84). NO appears to enhance tumor growth and cell proliferation at measurable concentrations in different clinical samples from different cancer types (85).
Table I.
Studies on the involvement of NO, H2S and CO synthesis enzymes in the modulation of cancer.
| First author/s, year | Gasotransmitters | Enzyme | Type of cancer | (Refs.) |
|---|---|---|---|---|
| Aaltoma, 2001, Aaltomaa, 2000, Bhowmick and Girotti, 2014, Coulter, 2010, Cronauer, 2007, Erlandsson, 2018, McCarthy, 2007, Fahey and Girotti, 2015, D'Este, 2020, Ryk, 2015, Uotila, 2001 | NO | iNOS | Prostate | (170–180) |
| Abdelmagid, 2011, Abdelmagid and Too, 2008, Ambs and Glynn, 2011, Alalami and Martin, 1998, Basudhar, 2017, Bentrari, 2005, Bing, 2001, Deepa, 2013, Duenas-Gonzalez, 1997, Evig, 2004, Flaherty, 2019, Garrido, 2017, Hsu, 2000, Kanugula, 2014, Kotamraju, 2007, López-Sánchez, 2020, Loibl, 2005a, Lahiri and Martin, 2004, Oktem, 2004, Martin, 1999, Nafea, 2020, Oktem, 2006, Zeybek, 2011, Walsh, 2016, Wink, 2015, Tschugguel, 1999 | Breast | (181–206) | ||
| Abadi and Kusters, 2013, Chen, 2006, Doi, 1999, Goto, 1999, Goto, 2006, Hirst and Robson, 2010, Hirahashi, 2014, Huang, 2012, Ikeguchi, 2002, Jun, 1999, Jung, 2002; Rafiei, 2012, Rajnakova, 2001, Zhang, 2011, Sawa, 2008, Shen, 2004, Son, 2001, Song, 2004 | Gastric | (207–224) | ||
| Ambs, 1998b, Bing, 2001, Bocca, 2010, Dabrowska, 2018, Deepa, 2013, Narayanan, 2004, Narayanan, 2003, Kwak, 2000, Kapral, 2015, Puglisi, 2015, Williams, 2003, Tong, 2007; Sasahara, 2002, Shen, 2015, Spiegel, 2005 | Colon | (187,188,225–237) | ||
| Eijan, 2002, Hosseini, 2006, Klotz, 1999, Koskela, 2012, Langle, 2020, Lin, 2003, Ryk, 2010, Ryk, 2011b, Wolf, 2000, Sandes, 2005, Sandes, 2012 | Bladder | (238–248) | ||
| Jung, 2002, Ambs and Harris, 1999, Chung, 2003, Cianchi, 2004, Cianchi, 2003, Fransen, 2005, Gallo, 1999, Ropponen, 2000, Zafirellis, 2010, Wang, 2004 | Colorectal | (217,249–257) | ||
| Delarue, 2001, Hu, 1998, Li, 2014, Liu, 1998, Liu, 2010, Zhang, 2014a, Ye, 2013, Sim, 2012 | Lung | (258–265) | ||
| Franco, 2004, Kong, 2001, Kong, 2002, Wang, 2016a, Vickers, 1999, Takahashi, 2008 | Pancreatic | (266–271) | ||
| Chen, 2005, Dong, 2014, Jiang, 2015, Lee, 2007, Ryu, 2015, Sowjanya, 2016 | Cervical | (272–277) | ||
| Li, 2017b, Malone, 2006, Martins Filho, 2014, Raspollini, 2004, Zhao, 2016, Xu, 1998, Trinh, 2015, Sanhueza, 2016b | Ovarian | (278–285) | ||
| Jayasurya, 2003 | Nasopharyngeal | (286) | ||
| Lukes, 2008 | Tonsillar | (287) | ||
| Soler, 2000 | Thyroid | (288) | ||
| Rafa, 2017 | Colitis | (289) | ||
| Amasyali, 2012, Klotz, 1999, Lin, 2003, Polat, 2015, Ryk, 2011a | eNOS | Bladder | (240,243,290–292) | |
| Abedinzadeh, 2020, Brankovic, 2013, Diler and Oden, 2016, Fu, 2015, Medeiros, 2003, Medeiros, 2002a, Medeiros, 2002b, Nikolic, 2015, Marangoni, 2006, Martin, 1999, Polytarchou, 2009, Re, 2011, Re, 2018, Ziaei, 2013, Zhao, 2014, Yu, 2013, Wu, 2014, Safarinejad, 2013, Sanli, 2011 | Prostate | (200,293–310) | ||
| Bayraktutan, 2020, Cheon, 2000, Fujita, 2010, Kocer, 2020, Peddireddy, 2018 | Lung | (311–315) | ||
| Chen, 2018, Crucitta, 2019, Basaran, 2011, Hao, 2010, Hefler, 2006, Kafousi, 2012, Lahiri and Martin, 2004, Loibl, 2006, Loibl, 2005b, Lu, 2006, Mortensen, 1999a, Mortensen, 1999b, Oktem, 2006, Martin, 2000b, Ramirez-Patino, 2013, Zintzaras, 2010, Zhang, 2016a, Zeybek, 2011 | Breast | (198,202,203,316–330) | ||
| Carkic, 2020, Karthik, 2014 | Oral | (331,332) | ||
| Chen, 2014, Mortensen, 2004, Penarando, 2018, Yeh, 2009 | Colorectal | (333–336) | ||
| Dabrowska, 2018 | Colon | (227) | ||
| Hefler, 2002 | Ovarian | (337) | ||
| Doi, 1999, Krishnaveni, 2015, Wang, 2005, Tecder Unal, 2010 | Gastric | (209,338–340) | ||
| de Fatima, 2008 | Renal | (341) | ||
| Lukes, 2008 | Tonsillar | (287) | ||
| Hung, 2019, Waheed, 2019 | Uterine and cervical | (342,343) | ||
| Riener, 2004 | Vulvar | (344) | ||
| Wang, 2016b | Pancreatic | (345) | ||
| Yanar, 2016 | Larynx | (346) | ||
| Chen, 2013 | nNOS | Neural stem cell | (347) | |
| de Fatima, 2008 | Renal | (341) | ||
| Karihtala, 2007 | Breast | (348) | ||
| Lewko, 2001 | Lung | (349) | ||
| Ahmed, 2012, Al Dhaheri, 2013, Avtandilyan, 2018, Bani, 1995, Avtandilyan, 2019, Alagol, 1999, Bhattacharjee, 2012, Cendan, 1996a, Cendan, 1996b, Chakraborty, 2004, Coskun, 2003, Coskun, 2008, Dave, 2014, De Vitto, 2013, Deliconstantinos, 1995, Demircan, 2020, Duan, 2014, El Hasasna, 2016, Engelen, 2018, Erbas, 2007, Gaballah, 2001, Gajalakshmi, 2013, Ganguly Bhattacharjee, 2012, Ganguly Bhattacharjee, 2014, Gaspari, 2020, Guha, 2002, Gunel, 2003, Kampa, 2001, Khalkhali-Ellis and Hendrix, 2003, Konukoglu, 2007, Kumar and Kashyap, 2015, Lahiri and Martin, 2009, Lee, 2014a, Hewala, 2010, Finkelman, 2017, Jadeski, 2002, McCurdy, 2011, Metwally, 2011, Mishra, 2020, Nakamura, 2006, Negroni, 2010, Martin, 2000a, Pervin, 2001b, Pervin, 2007, Martin, 2003, Nath, 2015, Pance, 2006, Parihar, 2008, Pervin, 2001a, Pervin, 2008, Pervin, 2003, Punathil, 2008, Radisavljevic, 2004, Rashad, 2014, Ray, 2001, Reveneau, 1999, Ridnour, 2012, Zhu, 2015, Zheng, 2020, Zeillinger, 1996, Wang, 2010, Thamrongwittawatpong, 2001, Toomey, 2001, Salaroglio, 2018, Sen, 2013a, Sen, 2013b, Shoulars, 2008, Simeone, 2003, Simeone, 2002, Simeone, 2008, Singh and Katiyar, 2011, Smeda, 2018, Song, 2002, Switzer, 2012, Switzer, 2011 | Non- specific | Breast | (350–424) | |
| Ai, 2015, Babykutty, 2012, Bessa, 2002, Cendan, 1996a, Chattopadhyay, 2012b, Edes, 2010, Ishikawa, 2003, Jarry, 2004, Jenkins, 1994, Jeon, 2005, Kim, 2020, Kopecka, 2011, Liu, 2003, Liu, 2008, Mojic, 2012, Millet, 2002, Oláh, 2018, Prevotat, 2006, Rao, 2004, Rao, 2006, Riganti, 2005, Wang and MacNaughton, 2005, Xie, 2020, Wenzel, 2003, Williams, 2001, Voss, 2019, Tesei, 2007, Thomsen, 1995, Stettner, 2018 | Colon | (357,425–452) | ||
| Ambs, 1998, Cembrzynska-Nowak, 1998, Cembrzyńska-Nowak, 2000, Chen, 2019b, Deliu, 2017, Fu, 2019, Fujimoto, 1997, Gao, 2019a, Hwang, 2017, Kaynar, 2005, Koizumi, 2001, Munaweera, 2015, Maciag, 2011, Maiuthed, 2020, Lee, 2006; Liaw, 2010, Liu, 2018, Luanpitpong and Chanvorachote, 2015, Muto, 2017, Masri, 2010, Masri, 2005, Matsunaga, 2014, McCurdy, 2011, Powan and Chanvorachote, 2014, Punathil and Katiyar, 2009, Zhou, 2000, Zhou, 2020, Zhang, 2014, Yongsanguanchai, 2015, Wongvaranon, 2013, Saisongkorh, 2016, Saleem, 2011, Sanuphan, 2013, Sanuphan, 2015, Su, 2010, Suzuki, 2019, Szejniuk, 2019 | Lung | (386,453–488) | ||
| Arif, 2010, Beevi, 2004, Korde Choudhari, 2012, Patel, 2009 | Oral | (489–492) | ||
| Brankovic, 2017, El-Mezayen, 2012, Gao, 2019b, Gao, 2019c, Jiang, 2013, Krzystek-Korpacka, 2020, Liu, 2004, McIlhatton, 2007, Moochhala, 1996, Muscat, 2005, Mandal, 2018, Yagihashi, 2000, Yu, 2005, Wei, 2008, Siedlar, 1999 | Colorectal | (493–507) | ||
| Arora, 2018, Atala, 2019, Dillioglugil, 2012, Della Pietra, 2015, Huh, 2006, Laschak, 2012, Lee, 2009; | Prostate | (94,152,176,508–519) | ||
| McCarthy, 2007, Rezakhani, 2017, Royle, 2004, Wang, 2007, Tan, 2017, Thomas, 2012, Siemens, 2009, Soni, 2020 | ||||
| Caneba, 2014, El-Sehemy, 2013, El-Sehemy, 2016, Kielbik, 2013, Leung, 2008, Thomsen, 1998, Salimian Rizi, 2015, Stevens, 2010 | Ovarian | (520–527) | ||
| Camp, 2006, Chen, 2020; Fujita, 2014, Fujita, 2019, Zhou, 2009, Wang, 2003, Wang and Xie, 2010, Thomas, 2002, Stewart, 2011, Sugita, 2010 | Pancreatic | (528–537) | ||
| Caygill, 2011, Sun, 2013 | Esophageal | (538,539) | ||
| Gecit, 2012, Gecit, 2017, Jansson, 1998, Kilic, 2006, Khalifa, 1999, Morcos, 1999, Wang, 2001, Thiel, 2014, Saygili, 2006 | Bladder | (540–548) | ||
| Bakan, 2002, Dincer, 2006, Holian, 2002, Eroglu, 1999, Koh, 1999, Oshima, 2001, Rajnakova, 1997, Yao, 2015, Sang, 2011, Sang, 2010 | Gastric | (549–558) | ||
| Li, 2017b, Muntane, 2013; Sayed-Ahmad and Mohamad, 2005 | Liver | (559–561) | ||
| Li, 2017a, Park, 2003, Wei, 2011, Sudhesh, 2013, Sundaram, 2020 | Cervical | (562–566) | ||
| Menendez, 2007, Yang, 2016 | Bone | (567,568) | ||
| Giusti, 2003, Ragot, 2015 | Thyroid | (569,570) | ||
| Gallo, 1998, Kawakami, 2004, Wu, 2020, Utispan and Koontongkaew, 2020 | Head and neck | (571–574) | ||
| Taysi, 2003 | Larynx | (575) | ||
| Thomsen, 1994, Sanhueza, 2016 | Gynecological | (65,576) | ||
| Chattopadhyay, 2012a, Dong, 2019, Li, 2020, Lv, 2014 | H2S | CSE, CBS and 3-MST | Breast | (577–580) |
| Choi, 2012, Kawahara, 2020b, Sekiguchi, 2016, Wang, 2019, Ye, 2020, Zhang, 2015 | Gastric | (126,581–585) | ||
| Chattopadhyay, 2012b, Chen, 2019a, Cai, 2010, Hale, 2018, Hong, 2014, Kodela, 2015, Rose, 2005, Oláh, 2018, Szabo, 2013, Chen, 2017 | Colon | (76,113,428,440,586–591) | ||
| Chen, 2017, Elsheikh, 2014, Faris, 2020, Malagrinò, 2019, Sakuma, 2019 | Colorectal | (591–595) | ||
| Liu, 2017 | Bladder | (596) | ||
| Pei, 2011, Liu, 2016 | Prostate | (96,597) | ||
| Wang, 2020 | Lung | (110) | ||
| Zhang, 2016 | Oral | (598) | ||
| Zhuang, 2018 | Bone | (599) | ||
| Xu, 2018 | Thyroid | (600) | ||
| Huang, 2020, Kawahara, 2017, Kim, 2018, Kourti, 2019, Lee, 2014 | CO | HO | Breast | (601–605) |
| Kawahara, 2020a, Kawahara, 201 | Ovarian | (606,607) | ||
| Khasag, 2014, Liptay, 2009, Nemeth, 2016, Shao, 2018, Shirabe, 1970, Zarogoulidis, 2012 | Lung | (608–613) | ||
| Lian, 2016 | Gastric | (614) | ||
| Lv, 2019, Yin, 2014 | Colorectal | (615,616) | ||
| Nitta, 2019 | Testicular | (617) | ||
| Oláh, 2018 | Colon | (440) | ||
| van Osch, 2019 | Bladder | (618) | ||
| Vítek, 2014 | Pancreatic | (619) |
3-MST, 3-mercaptopyruvate sulfurtransferase; CBS, cystathionine-β-synthase; CO, carbon monoxide; CSE, cystathionine-γ-lyase; eNOS, endothelial NOS; H2S, hydrogen sulfide; HO, heme oxygenase; iNOS, inducible NOS; nNOS, neuronal NOS; NO, nitric oxide.
In contrast to conventional signaling molecules that act by binding to specific receptor molecules, NO exerts its biological actions via a wide range of chemical reactions (86). The NO concentration and minor differences in the composition of the intracellular and extracellular environment determine the exact reactions attained. Under normal physiological conditions, cells produce small but significant amounts of NO, contributing to the regulation of anti-inflammatory effects and its antioxidant properties (83). However, in tissues with a high NO output, iNOS is activated, and nitration (addition of NO2), nitrosation (addition of NO+) and oxidation will be dominant (87). The interaction of NO with O2 or superoxide (O2−) results in the formation of reactive nitrogen species (RNS). The RNS, dinitrogen trioxide (N2O3) and peroxynitrite (ONOO), can induce two types of chemical stresses: Nitrosative and oxidative (88). N2O3 effectively nitrosates various biological targets to yield potentially carcinogenic nitrosamines and nitrosothiol derivatives, and N-nitrosation may have essential implications in the known association between chronic inflammation and malignant transformation (88). O2− and NO may rapidly interact to produce the potent cytotoxic oxidants ONOO− and its conjugated acid, peroxynitrous acid. In natural solutions, ONOO is a powerful oxidant, oxidizing thiols or thioethers, nitrating tyrosine residues, nitrating and oxidizing guanosine, degrading carbohydrates, initiating lipid peroxidation, and cleaving DNA, which has important implications in cancer (83).
Effect of NO on the TME
The effects of NO in a multistage model of cancer have been reported previously, it can drive angiogenesis, apoptosis, the cell cycle, invasion and the metastatic process (83,85). NO also serves a role in cellular transformation, the onset of neoplastic lesion formation, and the monitoring of invasion and colonization throughout metastasis (89). Therefore, understanding its role in promoting TME elements is crucial as it will reduce the ambiguity, and aid the development of NO-based cancer therapeutics, which will be effective in the prevention and treatment of a range of human cancer types.
The TME is characterized by hypoxia and acidity. Small pH drops (−0.6 U) favor the production of bioactive NO from nitrite, as evidenced by a higher degree of cyclic guanosine 3′,5′-monophosphate-dependent vasorelaxation in arterioles. A small dose of nitrite may make tumors more sensitive to radiation, resulting in a considerable growth delay and improved survival in mice (90). Therefore, low pH has been revealed to be an ideal setting for tumor-selective NO generation in response to nitrite systemic injection (90). The generation of NO by iNOS inhibits C-X-C motif chemokine ligand 10 expression in melanoma cells, resulting in a protumorigenic TME (91). Furthermore, eNOS upregulation in the TME reduced both the frequency and size of tumor implants in a surgical model of pancreatic cancer liver metastasis (92) and the influence of NO on tumor cell protease expression since tumor cell anoikis and invasion are both regulated by myofibroblast-derived matrix. Within tumor cells, eNOS-dependent downregulation of the matrix protease cathepsin B was detected, and cathepsin B silencing reduced tumor cell invasiveness in a manner comparable to eNOS upregulation. Therefore, an NO gradient within the TME influences tumor progression through orchestrated molecular interactions between tumor cells and stroma.
The role of NO in the complex interactions between the TME and the immune response is a good example of how complicated the molecular and cellular mechanisms determining the involvement of NO in cancer biology are. Although the activities of NO in the TME are varied and context-dependent, the evidence suggests that NO is an immunosuppressive mediator (93). By targeting tumors in a cell nonautonomous manner, S-nitroso glutathione (GSNO), a NO donor, reduced the tumor burden in a mouse model of castration-resistant prostate cancer (CRPC). Both the abundance of anti-inflammatory M2 macrophages and protein kinase R-like endoplasmic reticulum kinase expression were decreased by GSNO, indicating that NO influences TAM activity. GSNO also reduced IL-34, indicating that TAM differentiation was suppressed. This demonstrates the importance of NO in CRPC tumor inhibition via the TME (94).
Role of H2S in cancer
H2S is a novel gasotransmitter, which regulates cell proliferation and other cellular functions (95). It has been revealed that H2S serves an essential role as a signal molecule in regulating cell survival (95). It seems paradoxical that, on one hand, H2S acts as a physiological intercellular messenger to stimulate cell proliferation, and on the other hand, it may display cytotoxic activity (96). H2S, at physiologically relevant concentrations, hyperpolarizes the cell membrane, regulates cell proliferation, relaxes blood vessels and modulates neuronal excitability (95). Increased expression of various H2S-producing enzymes in cancer cells of different tissues has been reported, and novel roles of H2S in the pathophysiology of cancer have emerged (9). This is mainly observed in some cancer types, such as breast, lung, gastric, colorectal, bladder, prostate, oral, bone and thyroid cancer, where the malignant cells both express high levels of CBS and produce increased amounts of H2S, which results in enhanced tumor growth and spread by stimulating cellular bioenergetics, activating proliferative, migratory and invasive signaling pathways, and enhancing tumor angiogenesis (97), as indicated in Table I, which highlights the research on the involvement of NO, H2S, and CO production enzymes in cancer regulation. Importantly, in preclinical models of these cancer types, either pharmacological inhibition or genetic silencing of CBS was sufficient to suppress cancer cell bioenergetics in vitro, and to inhibit tumor growth and metastasis in vivo (9,98). This enhances the antitumor efficacy of frontline chemotherapeutic agents, providing a strong rationale for the development of CBS-targeted inhibitors as anticancer therapies (99). However, the observation that inhibition of H2S biosynthesis exerts anticancer effects is contradicted by another study which demonstrated that increasing H2S with exogenous donors also exerts antitumor actions (100). H2S stimulates the cytoprotective PI3K-AKT, p38-MAPK and nuclear factor erythroid 2 (NRF2) signaling pathways when present at low concentrations (101). Sulfhydration partially promotes a number of biological functions of H2S, including ATP-sensitive potassium (KATP) channel opening (101). At physiological concentrations, H2S can also serve a role in stimulating the cellular bioenergetic function by donating electrons to the mitochondrial electron transport chain at complex II, leading to increased mitochondrial levels of cyclic AMP (102). At higher concentrations, H2S inhibits oxidation of cytochrome c, which results in disruption of mitochondrial electron transport, and it can also exert pro-oxidant and DNA-damaging effects (103).
Effect of H2S on the TME
H2S acts as a gaseous signaling molecule and is endogenously generated by three H2S-producing enzymes, namely CBS, cystathionine γ-lyase and 3-mercaptopyruvate sulfur transferase. Imbalances in H2S-producing enzymes as well as H2S levels are associated with malfunctional H2S metabolism, which has been increasingly associated with several human pathological disorders (104). Several cancer cell lines and specimens exhibit upregulation of one or more of the H2S-synthesizing enzymes, and this aberrant expression is suggested to be a tumor enhancer (105). By modulation of the expression of the H2S-producing enzyme, the amount of tumor-derived H2S is altered, thereby modifying the TME and affecting tumor expansion and metastasis (106).
Numerous mechanisms contribute to the pro-tumor effect of H2S, including the induction of angiogenesis, regulation of mitochondrial bioenergetics, acceleration of cell cycle progression and anti-apoptotic functions (107). Furthermore, hypoxia is a common feature of the TME in a number of solid tumors, which affects the level of H2S by preventing H2S catabolism and consequently stimulating cystathionine γ-lyase gene expression (107). Furthermore, under the influence of hypoxia in the microenvironment, the levels of H2S-producing enzymes are upregulated, and the H2S-producing enzymes are transferred toward the mitochondria, which results in increased H2S production (106). Angiogenesis, which is an important process in cancer progression, is stimulated by paracrine signaling between stroma in the TME and epithelial tumor cells (108).
Previous evidence has demonstrated that H2S is an endogenous stimulator of ischemic-induced angiogenesis by promoting the upregulation of hypoxia-inducible factor 1 (HIF-1)-α via activation of different pathways, including the VEGFR2/mTOR and PI3K/AKT signaling pathways (105,109,110). H2S appears to support tumor cell proliferation by increasing vascular endothelial growth factor (a critical growth factor in angiogenesis) expression in kidney and ischemic tissues (111,112). An in vivo study conducted on nude mice revealed that silencing of CBS expression markedly decreased tumor growth. The researchers concluded that the reduction in tumor growth was associated with both the suppression of cancer cell signaling and metabolism, as well as the paracrine mechanism in the tumor environment (113).
In colon cancer, CBS-derived H2S promotes angiogenesis and vasorelaxation, thereby supporting tumor growth (113). In ovarian cancer, CBS knockdown reduces the number of blood vessels, resulting in tumor growth (97). Taken together, these results indicate that CBS serves an essential role in promoting angiogenesis and tumor growth. Therefore, CBS could be a promising molecular target for cancer therapy. Recently, researchers have developed a novel strategy to improve chemotherapy in patients with colorectal cancer by remodeling the TME through reduction of the high levels of H2S in colon tissues using copper iron oxide nanoparticles (114). Another strategy for cancer treatment is destroying the tumor metabolism symbiosis. This method successfully affects cancer cells with minimum impairment to healthy cells by using a zero-waste zwitterion-based H2S-driven nanomotor that generates acidosis in cancer cells within the TME, and consequently, the tumor growth will be suppressed (115).
One of the important characteristics of cancer cells is the acidic microenvironment (reduced intracellular pH) due to accumulation of lactic acid that results from a high rate of glycolysis. H2S donors trigger the activation of cellular transporters, such as glutamine transporter-1 (GLT-1) and ATP-binding cassette transporter A1, which directly regulates the aerobic glycolysis, which is a metabolic indicator of cancer (116). Nevertheless, activation of GLT-1 has both a promoting and an inhibiting effect depending on the cancer cell type, and thus, further studies are required to clarify the consequent responses (8).
It has been demonstrated that most cancer cells exhibit increased uptake of glucose and high lactate production, known as the Warburg effect, due to glycolysis that causes the acidic TME, which enhances tumor progression (117). Previous studies have demonstrated that continuous exposure of cancer cells to a low concentration of H2S results in inhibition of cancer progression. This anticancer effect is mainly due to an increase in metabolic lactic acid production by H2S and diminishes the pH regulatory system, which consequently leads to intense intracellular acidification and eventually drives cancer cell death (107,118).
Within the TME, there are key proteins and enzymes, such as matrix metalloproteinase, adhesive enzymes (E-cadherin) and integrins, which serve an essential role in the migration and metastasis of cancer cells (119,120). Tumor cells that enter the stroma within the TME after detaching from the main tumor move into the blood vessels and ultimately reach the other organs in the body (121). H2S donors have been used in different studies and have been demonstrated to successfully prevent migration and invasion by decreasing proteins and enzymes involved in migration and invasion in different cancer types (8,122,123). For example, it has been reported that treatment of hepatocellular carcinoma cells with 600–1,000 µM sodium hydrosulfide (NaHS), which is an H2S donor, efficiently reduces migration and invasion in a concentration-dependent manner via modulation of the EGFR/ERK/MMP-2 and PTEN/AKT signaling pathways (124). Similarly, NaHS treatment prevents migratory activity in thyroid cancer cells by deactivating the PI3K/AKT/mTOR and MAPK signaling pathways (125). Furthermore, NaHS reduces the MMP-2 protein levels in gastric cancer (126). Additionally, H2S serves a role in a different stage of cancer development and is involved in modulation of the TME, which regulates the rate of cancer progression and the effectiveness of therapy (106).
Role of CO in cancer
CO is best recognized for being a toxic gas produced by the burning of fossil fuels. On the one hand, CO poisoning is associated with high mortality rates, and thus, it attracts a lot of attention (127). On the other hand, CO has been conclusively demonstrated to be a gasotransmitter with physiological activities in mammals (128,129). CO is now accepted as a potential therapeutic agent along with its physiological roles and has entered multiple clinical trials (130,131). CO is produced in all cells by HO-1 and HO-2 (132). Each possesses strong cytoprotective functions for the cell, evidenced by the fact that the absence of either, particularly the stress-response isoform HO-1, is detrimental to the cell and organism (133,134). The inducible HO isoform (HO-1) can be upregulated in response to various stimuli, including heme, oxidative stress, ultraviolet irradiation, heat shock, hypoxia and NO (135). The constitutive HO isoform (HO-2) is expressed in several tissues, including the brain, kidney, liver and spleen (6). Low CO concentrations also activate KATP channels and influence various intracellular kinase pathways, including the PI3K-AKT and p38 MAPK signaling pathways (128). CO exerts adverse biological effects at higher concentrations, which, in vivo, are mainly attributed to the binding of CO to hemoglobin. The resulting carboxyhemoglobin reduces the O2-carrying capacity of the blood and leads to tissue hypoxia. In vitro, CO inhibits mitochondrial electron transport by irreversibly inhibiting cytochrome c oxidase (128).
Cellular and animal pharmacological experiments suggest numerous therapeutic indications where HO-1 or CO administration imparts benefits in treating conditions such as sepsis, bacterial infection, cancer, inflammation, circadian clock regulation, stroke, erectile dysfunction and heart attack (131). Some of the best-characterized physiological effects of CO include anti-inflammatory, antiproliferative, anti-apoptotic and anticoagulative responses. By contrast, at higher concentrations, CO becomes cytotoxic (136). In contrast to NO, the cytoprotective and cytotoxic effects of CO are intimately intertwined. For example, a low level of CO-mediated inhibition of mitochondrial activity, followed by a slight increase in intracellular reactive O2 species production, is important in CO-mediated cytoprotective signaling events (137,138). In a way, the cytoprotective effects of CO resemble the protective effects of pharmacological preconditioning. A short, relatively mild insult triggers a secondary cytoprotective phenotype via activation of the prototypical antioxidant response element NRF2-related factor. Thus, a protective cellular phenotype is maintained in the cell for a long time after CO has already been cleared from the biological system (6).
Gasotransmitter signaling significance
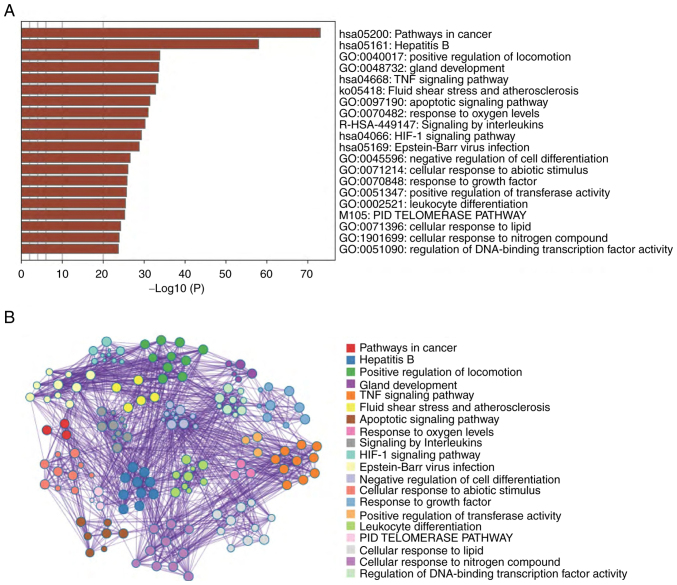
To highlight the significance of gasotransmitter signaling cascades in tumor growth and the chemotherapeutic response, network analysis approaches were utilized to identify the gasotransmitter-tumor signaling signature. Utilizing the PubMed (https://pubmed.ncbi.nlm.nih.gov/) and Web of Science (https://clarivate.com/webofsciencegroup/solutions/web-of-science/) databases, ~127 candidates (genes and proteins) were identified, which were significantly related to the gasotransmitters and tumorigenesis simultaneously. Using the selected list of candidates, the present network analyses were applied using the Enrich R (https://maayanlab.cloud/Enrichr/) and Metascape (https://metascape.org/gp/index.html#/main/step1) databases to identify all possible genes, proteins and pathways that may represent tumor-gasotransmitters interrelated signaling. As shown in Fig. 2A and B, the most relevant enriched pathways in the present analysis were ones related to cancer, which in turn, justifies the relevance and accuracy of the selected candidate list and highlights the significance of gasotransmitter signaling cascades in tumor development and growth. Additionally, different essential cellular signaling pathways were significantly enriched, such as the ‘positive regulation of locomotion’, ‘TNF signaling pathway’, ‘apoptotic signaling pathway’ and ‘HIF-1 signaling pathway’. These pathways serve an essential role in driving the fate of cancer cells and their response to different treatment modalities (139). Therefore, it is reasonably relevant to investigate the crosstalk between gasotransmitters and tumor cells.
Figure 2.
Pathway enrichment analysis. (A) Bar graph of enriched pathways across input gene lists, the top 20 clusters are arranged according to the degree of significance (P-value). (B) Network of the top 20 enriched pathways. The members with the best P-value from each of the 20 clusters were selected with the constraint that there are not >15 members per cluster and not >250 terms in total. Each node represents an enriched member and is colored accordingly. GO, Gene Ontology; HIF-1, hypoxia-inducible factor 1; PID, primary immunodeficiency.
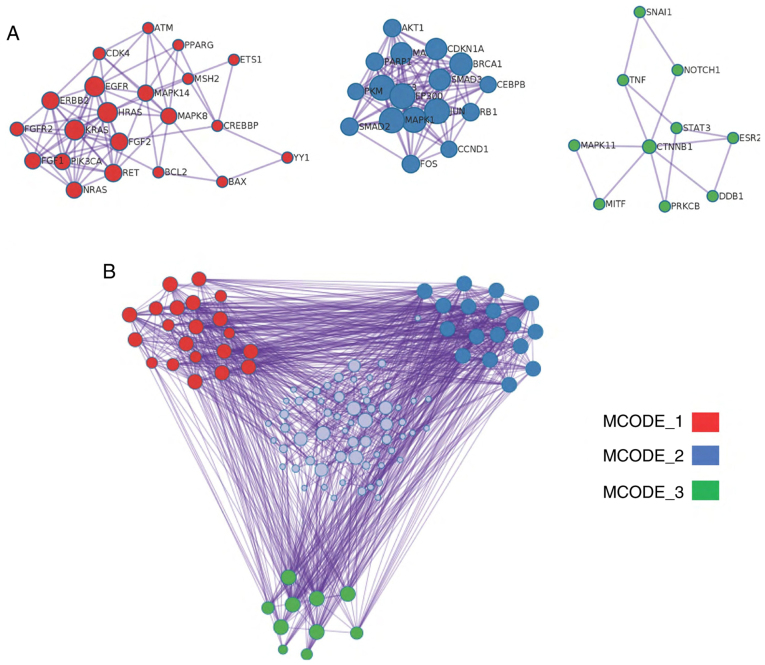
This pathway analysis was further validated using the EviNet database (https://www.evinet.org/). The detected enrichment signature was similar to the one identified using Enrich R and Metascape (data not shown). Furthermore, a deeper enrichment analysis revealing protein-protein interactions was performed considering three direct and physical connections at the minimum (Fig. 3A and B). Accordingly, the candidates were clustered into three densely connected networks upon applying the molecular complex detection algorithm using the Metascape annotation database. Each group or individual candidate within the group may represent a platform for molecular-mechanistic studies to investigate the interaction between the group members and gasotransmitters and their impact on cancer cell proliferation and response to treatment.
Figure 3.
Protein-protein interaction enrichment analysis using the BioGrid, InWeb_IM and OmniPath databases. The network contains the subset of proteins that form physical interactions with at least one other member in the list. The MCODE algorithm was applied for each network containing at least three proteins to identify densely connected network components. (A) Three MCODE clusters and corresponding protein members. Red, proteins enriched in the two best scoring pathways by P-value (‘Cancer Pathways’ and ‘Endocrine Resistance’); blue, two best scoring pathways by P-value (‘Colorectal Cancer’ and ‘Hepatitis B’), and the green cluster represents three pathways: ‘regulation of hematopoiesis’, ‘regulation of myeloid cell differentiation’ and ‘Hepatitis B’. (B) Protein-protein interaction network showing proteins in the three enriched MCODE modules and other candidate proteins, which have one physical contact with each other at the minimum. MCODE, molecular complex detection algorithm.
The present review investigated links between the current candidate list and a drug signature database containing annotations regarding drug induction or inhibition of gene expression. As shown in Table SII, the present candidate list was significantly enriched and associated with different anticancer or cancer-related drugs. The odds ratio ranking method is simply the odds ratio; however, the combined score is the odds ratio multiplied by the negative natural log of the P-value derived from Fisher's exact test and the Enrichr z-score (combined score=log(p)*z). Overall, this suggested that gasotransmitters serve essential roles in drug response signaling by cancer cells. Therefore, well-designed mechanistic studies are required to elucidate such roles and open novel avenues for drug discovery and cancer treatment modalities.
4. Effectiveness of gasotransmitters in chemotherapeutic drug treatment
Following the crucial discovery of gasotransmitters as fundamental biological molecules, their physiological significance has become a debated topic in recent decades. Utilizing gasotransmitters as therapeutic aids is justified by their roles in carcinogenesis, including enhancement of apoptotic stimuli, inhibition of metastasis and inhibition of angiogenesis. Therefore, using them alone or in combination with cytotoxic agents is an essential research platform for researchers and clinicians in cancer therapy (6,140–142).
Platinum compounds have been investigated extensively, and several studies have demonstrated that tumor cells are sensitized to cisplatin compounds by NO donors (143,144). In vitro, combination of cisplatin with natural NO gas or the NO donors diethylamine NONOate (DEA NONOate) or 1-propanamine, 3-(2-hydroxy-2-nitroso-1-propylhydrazino) NONOate increased the killing efficacy of cisplatin by 50–1,000 times compared with cisplatin alone, and the effect lasted for a number of hours (145). Furthermore, the combination treatment of cisplatin and diethylenetriamine NONOate reverses resistance and induces apoptosis in prostate cancer cell lines (146) and metastatic human colon carcinoma cell lines (147). NO-producing aspirin compounds that can emit NO for several hours have also been investigated. For example, in a clonogenic assay, nitroaspirin exhibited dose-dependent cytotoxicity and greatly boosted cisplatin cytotoxicity in both resistant and susceptible cells (148).
Carmustine is a chemotherapeutic drug that is combined with a NO source (the donor drug DEA NONOate), and the combination of chlorotoxin-NO, carmustine, or temozolomide enhances glioma cell death. Two variables that contributed to the enhanced cytotoxic activity of these cells were the production of active levels of the cytoprotective enzyme O6-methylguanine-DNA methyltransferase activity and altered p53 activity (149).
In the same year, Shami, Saavedra, Wang, Bonifant, Diwan, Singh, Gu, Fox, Buzard, Citro, Waterhouse, Davies, Ji and Keefer (150) created glutathione/glutathione S-transferase-activated nitric oxide (JS-K), a selective targeted NO donor that was active in vitro and in vivo against human HL60 leukemia cells, following its reaction with glutathione to produce NO in vivo. JS-K acts as a chemosensitizer for doxorubicin-induced cytotoxicity in renal (151), prostate (152) and bladder cancer cells (153).
NF-κB and NOS activation make HT29 human colon cancer cells more sensitive to doxorubicin cytotoxicity (154). Simvastatin increases NF-κB activity and NO production, while also increasing doxorubicin intracellular accumulation and cytotoxicity (154). The enhanced intracellular accumulation of doxorubicin is caused by tyrosine nitration in P-glycoprotein and multidrug resistance protein 1 by NO (155). In mice with triple-negative breast cancer, NO-releasing nanoparticles in combination with doxorubicin or a doxorubicin nanoparticle carrier decreased cell survival, caused apoptosis, elevated doxorubicin intracellularly, compromised lysosomal membrane integrity and suppressed tumor growth (156). Subsequently, an S class nanocarrier of NO (Nano-NO) was developed, and successfully targeted NO to hepatocellular cancer (157). Nano-NO has improved the administration and efficacy of chemotherapy. Additionally, combining nanomaterials with NO donors, as shown by Housein et al (78) and others (158), has improved the method of NO delivery. The aforementioned Nano-NO make tumor cells more susceptible to chemotherapy.
GTN in combination with vinorelbine and cisplatin increases the response in patients with lung cancer and reduces the median time to tumor progression (159). Additionally, the combination treatment of GTN and valproic acid results in the inhibition of Bcl-2 as well as the expression of Bax and caspase-3 in human K562 cells (160). STAT3 is associated with a number of the substituted NO-releasing quinolone-1,2,4-triazole/oxime derivatives (161). In melanoma with the B-Raf V600E mutation and vemurafenib-resistant melanoma, STAT3 inhibitors have shown efficacy (161). Poly-S nitrosylated human albumin alters colon cancer cell resistance to bevacizumab (162). Furthermore, the combination of bevacizumab with S-nitrosylated human albumin exhibits antitumor effects both in vitro and in vivo (163).
Long-term (3–5 days) exposure of cancer cells to low levels of H2S (30 M; sustained >7 days) using the slow-releasing H2S donor GYY4137 causes cancer cell death in vitro by activating caspase activity and causing apoptosis (164,165). In a mouse xenograft model, GYY4137 caused a reduction in tumor volume, and this had no apparent deleterious effects on physiological functions (165). A previous study, which was performed on 11 cancer cell lines, revealed that H2S-releasing non-steroidal anti-inflammatory drugs inhibited the proliferation of all 11 cancer cell lines that were tested (102), providing further evidence of the potential of H2S as an anticancer agent. Using sulfide salt NaHS, which releases large amounts of H2S instantaneously in an aqueous solution (≤400 µM; detected within first 1.5 h), caused only a minimal growth inhibitory effect in cancer cell lines, indicating the possibility that a long period of continuous, low-level H2S exposure is required for its efficient anticancer function (118). Based on these findings, it is hypothesized that the anti-proliferative effect of H2S is selective, meaning it affects cancer cells but not normal cells.
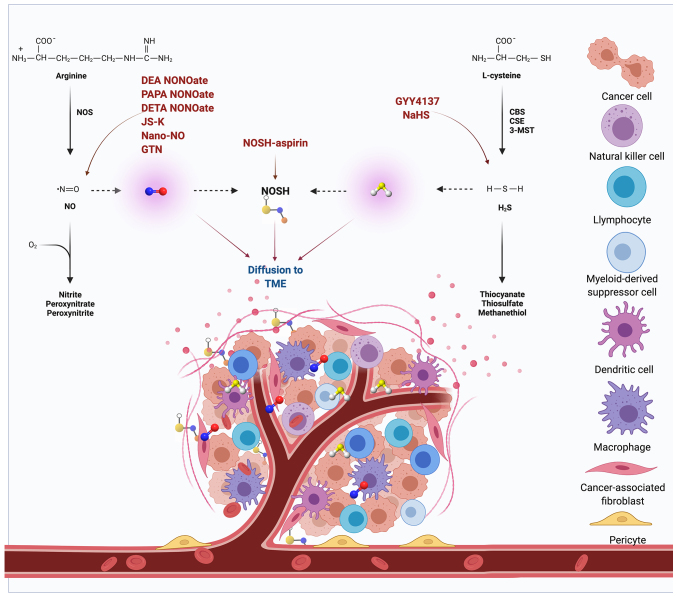
One must remember that the three gasotransmitters do not work alone. Instead, they work together. This cooperation occasionally occurs using overlapping signaling pathways (for instance, both NO and CO stimulate the sGC pathway). NO directly stimulates the sGC pathway, and H2S concurrently blocks cGMP via inhibition of cGMP phosphodiesterase (166). One of the few studies of this contest demonstrated the anticancer effect of a combined NO- and H2S-donating compound, nitric oxide and hydrogen sulfide-releasing hybrid-aspirin, both in vitro and in vivo (6). The impact of NO and H2S on the TME is displayed in Fig. 4, and several gasotransmitter-based drugs targeting the TME are currently being investigated in clinical studies (167–169). To further investigate and understand the nature of these interactions, more comprehensive studies are required, mainly in the context of cancer, which may be utilized for therapeutic benefits in the future.
Figure 4.
Impact of NO and H2S on the TME. Several gasotransmitter-based drugs targeting the TME are currently being tested in clinical trials. DEA NONOate, PAPA NONOate, DETA NONOate, JS-K, Nano-NO and GTN are NO-releasing drugs, GYY4137 and NaHS are H2S-releasing drugs, and NOSH-aspirin releases NOSH into the TME. When used in combination, these drugs increase the cytotoxic activity of various chemotherapeutic drugs. 3-MST, 3-mercaptopyruvate sulfurtransferase; CBS, cystathionine-β-synthase; CSE, cystathionine-γ-lyase; DEA NONOate, diethylamine NONOate; DETA NONOate, diethylenetriamine NONOate; GTN, nitroglycerin; H2S, hydrogen sulfide; JS-K, glutathione/glutathione S-transferase-activated nitric oxide; NaHS, sodium hydrosulfide; Nano-NO, nitric oxide nanoparticles; NO, nitric oxide; NOS, nitric oxide synthase; NOSH, nitric oxide and hydrogen sulfide-releasing hybrid; PAPA NONOate, 1-propanamine, 3-(2-hydroxy-2-nitroso-1-propylhydrazino); TME, tumor microenvironment.
5. Conclusions
More than three decades of studies in the field of the three gasotransmitters NO, CO and H2S have resulted in the identification of several pathophysiological paradigms and associated experimental therapeutic approaches that may be ultimately suitable for clinical translation. In particular, the initial perplexing observation that both gasotransmitter-synthesis inhibitors and donors appear to have anticancer effects, which the complex biology and bell-shaped pharmacology of NO, CO and H2S can explain, should not be considered as a barrier to translation into clinical settings. Their critical functions in normal cells compared with cancer cells open avenues for combinatorial treatment approaches together with chemotherapeutic drugs, aiming for improved clinical significance.
Supplementary Material
Acknowledgements
Not applicable.
Glossary
Abbreviations
- CAF
cancer-associated fibroblast
- CBS
cystathionine-β-synthase
- CO
carbon monoxide
- CRPC
castration-resistant prostate cancer
- DEA NONOate
diethylamine NONOate
- ECM
extracellular matrix
- EDRF
endothelium-derived relaxing factor
- eNOS
endothelial NOS
- GLT-1
glutamine transporter-1
- GSNO
S-nitroso glutathione
- GTN
nitroglycerin
- H2S
hydrogen sulfide
- HIF-1
hypoxia-inducible factor 1
- HO
heme oxygenase
- iNOS
inducible NOS
- MOPP
mechlorethamine, oncovin, procarbazine and prednisone
- N2O3
dinitrogen trioxide
- NaHS
sodium hydrosulfide
- nNOS
neuronal NOS
- NO
nitric oxide
- NOS
NO synthases
- NRF2
nuclear factor erythroid 2
- ONOO
peroxynitrite
- RNS
reactive nitrogen species
- sGC
soluble guanylyl cyclase
- TAM
tumor-associated macrophage
- TME
tumor microenvironment
Funding Statement
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
MAAN, ZOK, ZH, HAH, RMA and BMH wrote the manuscript and designed the figures. AS and TA wrote the manuscript and critically revised the paper. Data authentication is not applicable. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Leaf C. Why we're losing the war on cancer (and how to win it) Fortune. 2004;149:76–82. 84–86, 88. [PubMed] [Google Scholar]
- 2.Debela DT, Muzazu SG, Heraro KD, Ndalama MT, Mesele BW, Haile DC, Kitui SK, Manyazewal T. New approaches and procedures for cancer treatment: Current perspectives. SAGE Open Med. 2021;9:20503121211034366. doi: 10.1177/20503121211034366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ke X, Shen L. Molecular targeted therapy of cancer: The progress and future prospect. Front Laboratory Med. 2017;1:69–75. doi: 10.1016/j.flm.2017.06.001. [DOI] [Google Scholar]
- 4.Liang XJ, Chen C, Zhao Y, Wang PC. Circumventing tumor resistance to chemotherapy by nanotechnology. Methods Mol Biol. 2010;596:467–488. doi: 10.1007/978-1-60761-416-6_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Iyer AK, Khaled G, Fang J, Maeda H. Exploiting the enhanced permeability and retention effect for tumor targeting. Drug Discov Today. 2006;11:812–818. doi: 10.1016/j.drudis.2006.07.005. [DOI] [PubMed] [Google Scholar]
- 6.Szabo C. Gasotransmitters in cancer: From pathophysiology to experimental therapy. Nat Rev Drug Discov. 2016;15:185–203. doi: 10.1038/nrd.2015.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Xu W, Liu LZ, Loizidou M, Ahmed M, Charles IG. The role of nitric oxide in cancer. Cell Res. 2002;12:311–320. doi: 10.1038/sj.cr.7290133. [DOI] [PubMed] [Google Scholar]
- 8.Ngowi EE, Afzal A, Sarfraz M, Khattak S, Zaman SU, Khan NH, Li T, Jiang QY, Zhang X, Duan SF, et al. Role of hydrogen sulfide donors in cancer development and progression. Int J Biol Sci. 2021;17:73–88. doi: 10.7150/ijbs.47850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hellmich MR, Szabo C. Hydrogen sulfide and cancer. Handb Exp Pharmacol. 2015;230:233–241. doi: 10.1007/978-3-319-18144-8_12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wegiel B, Gallo D, Csizmadia E, Harris C, Belcher J, Vercellotti GM, Penacho N, Seth P, Sukhatme V, Ahmed A, et al. Carbon monoxide expedites metabolic exhaustion to inhibit tumor growth. Cancer Res. 2013;73:7009–7021. doi: 10.1158/0008-5472.CAN-13-1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vannini F, Kashfi K, Nath N. The dual role of iNOS in cancer. Redox Biol. 2015;6:334–343. doi: 10.1016/j.redox.2015.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kashfi K. The dichotomous role of H2S in cancer cell biology? Déjà vu all over again. Biochem Pharmacol. 2018;149:205–223. doi: 10.1016/j.bcp.2018.01.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tien Vo TT, Vo QC, Tuan VP, Wee Y, Cheng HC, Lee IT. The potentials of carbon monoxide-releasing molecules in cancer treatment: An outlook from ROS biology and medicine. Redox Biol. 2021;46:102124. doi: 10.1016/j.redox.2021.102124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gensini GF, Conti AA, Lippi D. The contributions of Paul Ehrlich to infectious disease. J Infection. 2007;54:221–224. doi: 10.1016/j.jinf.2004.05.022. [DOI] [PubMed] [Google Scholar]
- 15.Riddell S. The emperor of all maladies: A biography of cancer. J Clin Invest. 2011;121:5. doi: 10.1172/JCI45710. [DOI] [Google Scholar]
- 16.Bosch F, Rosich L. The contributions of Paul Ehrlich to pharmacology: A tribute on the occasion of the centenary of his Nobel Prize. Pharmacology. 2008;82:171–179. doi: 10.1159/000149583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Valent P, Groner B, Schumacher U, Superti-Furga G, Busslinger M, Kralovics R, Zielinski C, Penninger JM, Kerjaschki D, Stingl G, et al. Paul Ehrlich (1854–1915) and his contributions to the foundation and birth of translational medicine. J Innate Immun. 2016;8:111–120. doi: 10.1159/000443526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Faguet GB. A brief history of cancer: Age-old milestones underlying our current knowledge database. Int J Cancer. 2015;136:2022–2036. doi: 10.1002/ijc.29134. [DOI] [PubMed] [Google Scholar]
- 19.Baselga J, Bhardwaj N, Cantley LC, DeMatteo R, DuBois RN, Foti M, Gapstur SM, Hahn WC, Helman LJ, Jensen RA, et al. AACR cancer progress report 2015. Clin Cancer Res. 2015;21((Suppl 19)):S1–S128. doi: 10.1158/1078-0432.CCR-15-1846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Farber S, Diamond LK. Temporary remissions in acute leukemia in children produced by folic acid antagonist, 4-aminopteroyl-glutamic acid. N Engl J Med. 1948;238:787–793. doi: 10.1056/NEJM194806032382301. [DOI] [PubMed] [Google Scholar]
- 21.Kotecha RS, Gottardo NG, Kees UR, Cole CH. The evolution of clinical trials for infant acute lymphoblastic leukemia. Blood Cancer J. 2014;4:e200. doi: 10.1038/bcj.2014.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kinjo J, Nakano D, Fujioka T, Okabe H. Screening of promising chemotherapeutic candidates from plants extracts. J Nat Med. 2016;70:335–360. doi: 10.1007/s11418-016-0992-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Freireich EJ, Karon M, Frei III E. Quadruple combination therapy (VAMP) for acute lymphocytic leukemia of childhood. Proc Am Assoc Cancer Res. 1964;5:20. [Google Scholar]
- 24.Liebman HA, Hum GJ, Sheehan WW, Ryden VM, Bateman JR. Randomized study for the treatment of adult advanced Hodgkin's disease: Mechlorethamine, vincristine, procarbazine, and prednisone (MOPP) versus lomustine, vinblastine, and prednisone. Cancer Treat Rep. 1983;67:413–419. [PubMed] [Google Scholar]
- 25.Bonadonna G, Zucali R, Monfardini S, De Lena M, Uslenghi C. Combination chemotherapy of Hodgkin's disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide versus MOPP. Cancer. 1975;36:252–259. doi: 10.1002/1097-0142(197507)36:1<252::AID-CNCR2820360128>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 26.Arruebo M, Vilaboa N, Sáez-Gutierrez B, Lambea J, Tres A, Valladares M, González-Fernández A. Assessment of the evolution of cancer treatment therapies. Cancers (Basel) 2011;3:3279–3330. doi: 10.3390/cancers3033279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bonadonna G, Valagussa P, Moliterni A, Zambetti M, Brambilla C. Adjuvant cyclophosphamide, methotrexate, and fluorouracil in node-positive breast cancer: The results of 20 years of follow-up. N Engl J Med. 1995;332:901–906. doi: 10.1056/NEJM199504063321401. [DOI] [PubMed] [Google Scholar]
- 28.Fisher B, Sherman B, Rockette H, Redmond C, Margolese R, Fisher ER. 1-phenylalanine mustard (L-PAM) in the management of premenopausal patients with primary breast cancer: lack of association of disease-free survival with depression of ovarian function. National Surgical Adjuvant Project for Breast and Bowel Cancers. Cancer. 1979;44:847–857. doi: 10.1002/1097-0142(197909)44:3<847::AID-CNCR2820440309>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- 29.Early Breast Cancer Trialists' Collaborative Group (EBCTCG), corp-author Long-term outcomes for neoadjuvant versus adjuvant chemotherapy in early breast cancer: Meta-analysis of individual patient data from ten randomised trials. Lancet Oncol. 2018;19:27–39. doi: 10.1016/S1470-2045(17)30777-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mansoori B, Mohammadi A, Davudian S, Shirjang S, Baradaran B. The different mechanisms of cancer drug resistance: A Brief Review. Adv Pharm Bull. 2017;7:339–348. doi: 10.15171/apb.2017.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Roma-Rodrigues C, Mendes R, Baptista PV, Fernandes AR. Targeting tumor microenvironment for cancer therapy. Int J Mol Sci. 2019;20:840. doi: 10.3390/ijms20040840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bagnyukova TV, Serebriiskii IG, Zhou Y, Hopper-Borge EA, Golemis EA, Astsaturov I. Chemotherapy and signaling: How can targeted therapies supercharge cytotoxic agents? Cancer Biol Ther. 2010;10:839–853. doi: 10.4161/cbt.10.9.13738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Whiteside TL. The tumor microenvironment and its role in promoting tumor growth. Oncogene. 2008;27:5904–5912. doi: 10.1038/onc.2008.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Alkasalias T, Moyano-Galceran L, Arsenian-Henriksson M, Lehti K. Fibroblasts in the Tumor Microenvironment: Shield or Spear? Int J Mol Sci. 2018;19:1532. doi: 10.3390/ijms19051532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Klemm F, Joyce JA. Microenvironmental regulation of therapeutic response in cancer. Trends Cell Biol. 2015;25:198–213. doi: 10.1016/j.tcb.2014.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vaupel P, Multhoff G. Hypoxia-/HIF-1α-Driven factors of the tumor microenvironment impeding antitumor immune responses and promoting malignant progression. Adv Exp Med Biol. 2018;1072:171–175. doi: 10.1007/978-3-319-91287-5_27. [DOI] [PubMed] [Google Scholar]
- 37.Jing X, Yang F, Shao C, Wei K, Xie M, Shen H, Shu Y. Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol Cancer. 2019;18:157. doi: 10.1186/s12943-019-1089-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Luo W, Wang Y. Hypoxia mediates tumor malignancy and therapy resistance. Adv Exp Med Biol. 2019;1136:1–18. doi: 10.1007/978-3-030-12734-3_1. [DOI] [PubMed] [Google Scholar]
- 39.Jarosz-Biej M, Smolarczyk R, Cichoń T, Kułach N. Tumor Microenvironment as A ‘Game Changer’ in cancer radiotherapy. Int J Mol Sci. 2019;20:3212. doi: 10.3390/ijms20133212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Befani C, Liakos P. The role of hypoxia-inducible factor-2 alpha in angiogenesis. J Cell Physiol. 2018;233:9087–9098. doi: 10.1002/jcp.26805. [DOI] [PubMed] [Google Scholar]
- 41.Senthebane DA, Rowe A, Thomford NE, Shipanga H, Munro D, Mazeedi MAMA, Almazyadi HAM, Kallmeyer K, Dandara C, Pepper MS, et al. The role of tumor microenvironment in chemoresistance: to survive, keep your enemies closer. Int J Mol Sci. 2017;18:1586. doi: 10.3390/ijms18071586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hass R, von der Ohe J, Ungefroren H. Impact of the tumor microenvironment on tumor heterogeneity and consequences for cancer cell plasticity and stemness. Cancers (Basel) 2020;12:3716. doi: 10.3390/cancers12123716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Vitale I, Manic G, Coussens LM, Kroemer G, Galluzzi L. Macrophages and Metabolism in the Tumor Microenvironment. Cell Metab. 2019;30:36–50. doi: 10.1016/j.cmet.2019.06.001. [DOI] [PubMed] [Google Scholar]
- 44.Bu L, Baba H, Yoshida N, Miyake K, Yasuda T, Uchihara T, Tan P, Ishimoto T. Biological heterogeneity and versatility of cancer-associated fibroblasts in the tumor microenvironment. Oncogene. 2019;38:4887–4901. doi: 10.1038/s41388-019-0765-y. [DOI] [PubMed] [Google Scholar]
- 45.Wang R. The Evolution of Gasotransmitter Biology and Medicine. In: Wang R, editor. Signal Transduction and the Gasotransmitters: NO, CO, and H2S in Biology and Medicine. Humana Press; Totowa, NJ: 2004. pp. 3–31. [DOI] [Google Scholar]
- 46.Andrew PJ, Mayer B. Enzymatic function of nitric oxide synthases. Cardiovasc Res. 1999;43:521–531. doi: 10.1016/S0008-6363(99)00115-7. [DOI] [PubMed] [Google Scholar]
- 47.Ryter SW, Choi AMK. Heme oxygenase-1/carbon monoxide: From metabolism to molecular therapy. Am J Respir Cell Mol Biol. 2009;41:251–260. doi: 10.1165/rcmb.2009-0170TR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Naik JS, O'Donaughy TL, Walker BR. Endogenous carbon monoxide is an endothelial-derived vasodilator factor in the mesenteric circulation. Am J Physiol Heart Circ Physiol. 2003;284:H838–H845. doi: 10.1152/ajpheart.00747.2002. [DOI] [PubMed] [Google Scholar]
- 49.Wang R. The gasotransmitter role of hydrogen sulfide. Antioxid Redox Signal. 2003;5:493–501. doi: 10.1089/152308603768295249. [DOI] [PubMed] [Google Scholar]
- 50.Tang G, Wu L, Liang W, Wang R. Direct stimulation of K(ATP) channels by exogenous and endogenous hydrogen sulfide in vascular smooth muscle cells. Mol Pharmacol. 2005;68:1757–1764. doi: 10.1124/mol.105.017467. [DOI] [PubMed] [Google Scholar]
- 51.Salihi A. Activation of inward rectifier potassium channels in high salt impairment of hydrogen sulfide-induced aortic relaxation in rats. Physiol Pharmacol. 2016;19:263–273. [Google Scholar]
- 52.Wang L, Xie X, Ke B, Huang W, Jiang X, He G. Recent advances on endogenous gasotransmitters in inflammatory dermatological disorders. J Adv Res. doi: 10.1016/j.jare.2021.08.012. (In Press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.West JB. Joseph Priestley, oxygen, and the enlightenment. Am J Physiol Lung Cell Mol Physiol. 2014;306:L111–L119. doi: 10.1152/ajplung.00310.2013. [DOI] [PubMed] [Google Scholar]
- 54.Gruetter CA, Barry BK, McNamara DB, Gruetter DY, Kadowitz PJ, Ignarro L. Relaxation of bovine coronary artery and activation of coronary arterial guanylate cyclase by nitric oxide, nitroprusside and a carcinogenic nitrosoamine. J Cyclic Nucleotide Res. 1979;5:211–224. [PubMed] [Google Scholar]
- 55.Furchgott RF, Zawadzki JV. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature. 1980;288:373–376. doi: 10.1038/288373a0. [DOI] [PubMed] [Google Scholar]
- 56.Ignarro LJ, Buga GM, Wood KS, Byrns RE, Chaudhuri G. Endothelium-derived relaxing factor produced and released from artery and vein is nitric oxide. Proc Natl Acad Sci USA. 1987;84:9265–9269. doi: 10.1073/pnas.84.24.9265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Palmer RM, Ferrige AG, Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature. 1987;327:524–526. doi: 10.1038/327524a0. [DOI] [PubMed] [Google Scholar]
- 58.Moncada S, Palmer RM, Higgs EA. The discovery of nitric oxide as the endogenous nitrovasodilator. Hypertension. 1988;12:365–372. doi: 10.1161/01.HYP.12.4.365. [DOI] [PubMed] [Google Scholar]
- 59.Murad F, Mittal CK, Arnold WP, Katsuki S, Kimura H. Guanylate cyclase: Activation by azide, nitro compounds, nitric oxide, and hydroxyl radical and inhibition by hemoglobin and myoglobin. Adv Cyclic Nucleotide Res. 1978;9:145–158. [PubMed] [Google Scholar]
- 60.Koshland DE., Jr The molecule of the year. Science. 1992;258:1861. doi: 10.1126/science.1470903. [DOI] [PubMed] [Google Scholar]
- 61.Marsh N, Marsh A. A short history of nitroglycerine and nitric oxide in pharmacology and physiology. Clin Exp Pharmacol Physiol. 2000;27:313–319. doi: 10.1046/j.1440-1681.2000.03240.x. [DOI] [PubMed] [Google Scholar]
- 62.Xu WM, Liu LZ. Nitric Oxide: Nitric oxide: From a mysterious labile factor to the molecule of the Nobel Prize. Recent progress in nitric oxide research. Cell Res. 1998;8:251–258. doi: 10.1038/cr.1998.25. [DOI] [PubMed] [Google Scholar]
- 63.Yetik-Anacak G, Catravas JD. Nitric oxide and the endothelium: History and impact on cardiovascular disease. Vascul Pharmacol. 2006;45:268–276. doi: 10.1016/j.vph.2006.08.002. [DOI] [PubMed] [Google Scholar]
- 64.Kennedy J. Account of the phænomena produced by a large quantity of nitric oxide of quicksilver, swallowed by mistake; and of the means employed to counteract its deleterious influence. Med Chir J Rev. 1816;1:189–196. [PMC free article] [PubMed] [Google Scholar]
- 65.Thomsen LL, Lawton FG, Knowles RG, Beesley JE, Riveros-Moreno V, Moncada S. Nitric oxide synthase activity in human gynecological cancer. Cancer Res. 1994;54:1352–1354. [PubMed] [Google Scholar]
- 66.Hidy GM, Mueller PK, Altshuler SL, Chow JC, Watson JG. Air quality measurements-From rubber bands to tapping the rainbow. J Air Waste Manag Assoc. 2017;67:637–668. doi: 10.1080/10962247.2017.1308890. [DOI] [PubMed] [Google Scholar]
- 67.Kinnicutt LP, Sanford GR. The iodometric determination of small quantities of carbon monoxide. Public Health Pap Rep. 1899;25:600–604. [PMC free article] [PubMed] [Google Scholar]
- 68.Luden G. THE carbon monoxide menace and the cancer problem. Can Med Assoc J. 1927;17:43–48. [PMC free article] [PubMed] [Google Scholar]
- 69.Roughton FJW. The Kinetics of Haemoglobin V-The Combination of Carbon Monoxide with Reduced Haemoglobin. Proc R Soc London Series B, Containing Papers Biol Character. 1934;115:464–473. doi: 10.1098/rspb.1934.0053. [DOI] [Google Scholar]
- 70.Roughton FJW, Darling RC. The effect of carbon monoxide on the oxyhemoglobin dissociation curve. Am J Physiol Legacy Content. 1944;141:17–31. doi: 10.1152/ajplegacy.1944.141.5.737-s. [DOI] [Google Scholar]
- 71.Roughton FJ. The equilibrium between carbon monoxide and sheep haemoglobin at very high percentage saturations. J Physiol. 1954;126:359–383. doi: 10.1113/jphysiol.1954.sp005215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Sjostrand T. Endogenous formation of carbon monoxide in man. Nature. 1949;164:580. doi: 10.1038/164580a0. [DOI] [PubMed] [Google Scholar]
- 73.Tenhunen R, Marver HS, Schmid R. Microsomal heme oxygenase. Characterization of the enzyme. J Biol Chem. 1969;244:6388–6394. doi: 10.1016/S0021-9258(18)63477-5. [DOI] [PubMed] [Google Scholar]
- 74.Zhao Y, Biggs TD, Xian M. Hydrogen sulfide (H2S) releasing agents: Chemistry and biological applications. Chem Commun (Camb) 2014;50:11788–11805. doi: 10.1039/C4CC00968A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Tanner FW. Studies on the bacterial metabolism of sulfur: I. Formation of hydrogen sulfide from certain sulfur compounds under aerobic conditions. J Bacteriol. 1917;2:585–593. doi: 10.1128/jb.2.5.585-593.1917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Rose P, Moore PK, Ming SH, Nam OC, Armstrong JS, Whiteman M. Hydrogen sulfide protects colon cancer cells from chemopreventative agent beta-phenylethyl isothiocyanate induced apoptosis. World J Gastroenterol. 2005;11:3990–3997. doi: 10.3748/wjg.v11.i26.3990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Wang R. Signaling pathways for the vascular effects of hydrogen sulfide. Curr Opin Nephrol Hypertens. 2011;20:107–112. doi: 10.1097/MNH.0b013e3283430651. [DOI] [PubMed] [Google Scholar]
- 78.Housein Z, Kareem TS, Salihi A. In vitro anticancer activity of hydrogen sulfide and nitric oxide alongside nickel nanoparticle and novel mutations in their genes in CRC patients. Sci Rep. 2021;11:2536. doi: 10.1038/s41598-021-82244-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Shen Y, Shen Z, Luo S, Guo W, Zhu YZ. The cardioprotective effects of hydrogen sulfide in heart diseases: From molecular mechanisms to therapeutic potential. Oxid Med Cell Longev. 2015;2015:925167. doi: 10.1155/2015/925167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Coleman JW. Nitric oxide in immunity and inflammation. Int Immunopharmacol. 2001;1:1397–1406. doi: 10.1016/S1567-5769(01)00086-8. [DOI] [PubMed] [Google Scholar]
- 81.Esplugues JV. NO as a signalling molecule in the nervous system. Br J Pharmacol. 2002;135:1079–1095. doi: 10.1038/sj.bjp.0704569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Sen S, Jensen K, Brennan K, Ramadoss S, Chaudhuri G. Chapter 10-Chemoprotective and chemosensitizing effects of nitric oxide and other biologically active gases in breast cancer chemotherapy: Potential implications. In: Bonavida B, editor. Nitric Oxide (Donor/Induced) in Chemosensitizing. Academic Press; 2017. pp. 169–178. [DOI] [Google Scholar]
- 83.Choudhari SK, Chaudhary M, Bagde S, Gadbail AR, Joshi V. Nitric oxide and cancer: A review. World J Surg Oncol. 2013;11:118. doi: 10.1186/1477-7819-11-118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Burke AJ, Sullivan FJ, Giles FJ, Glynn SA. The yin and yang of nitric oxide in cancer progression. Carcinogenesis. 2013;34:503–512. doi: 10.1093/carcin/bgt034. [DOI] [PubMed] [Google Scholar]
- 85.Ying L, Hofseth LJ. An Emerging role for endothelial nitric oxide synthase in chronic inflammation and cancer. Cancer Res. 2007;67:1407–1410. doi: 10.1158/0008-5472.CAN-06-2149. [DOI] [PubMed] [Google Scholar]
- 86.Tennyson Andrew G, Lippard Stephen J. Generation, translocation, and action of nitric oxide in living systems. Chem Biol. 2011;18:1211–1220. doi: 10.1016/j.chembiol.2011.09.009. [DOI] [PubMed] [Google Scholar]
- 87.Grisham MB, Jourd'Heuil D, Wink DA. Nitric oxide. I. Physiological chemistry of nitric oxide and its metabolites: Implications in inflammation. Am J Physiol. 1999;276:G315–G321. doi: 10.1152/ajpgi.1999.276.2.G315. [DOI] [PubMed] [Google Scholar]
- 88.Subapriya R, Kumaraguruparan R, Ramachandran CR, Nagini S. Oxidant-antioxidant status in patients with oral squamous cell carcinomas at different intraoral sites. Clin Biochem. 2002;35:489–493. doi: 10.1016/S0009-9120(02)00340-5. [DOI] [PubMed] [Google Scholar]
- 89.Cheng H, Wang L, Mollica M, Re AT, Wu S, Zuo L. Nitric oxide in cancer metastasis. Cancer Lett. 2014;353:1–7. doi: 10.1016/j.canlet.2014.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Frérart F, Sonveaux P, Rath G, Smoos A, Meqor A, Charlier N, Jordan BF, Saliez J, Noël A, Dessy C, et al. The acidic tumor microenvironment promotes the reconversion of nitrite into nitric oxide: Towards a new and safe radiosensitizing strategy. Clin Cancer Res. 2008;14:2768–2774. doi: 10.1158/1078-0432.CCR-07-4001. [DOI] [PubMed] [Google Scholar]
- 91.Tanese K, Grimm EA, Ekmekcioglu S. The role of melanoma tumor-derived nitric oxide in the tumor inflammatory microenvironment: Its impact on the chemokine expression profile, including suppression of CXCL10. Int J Cancer. 2012;131:891–901. doi: 10.1002/ijc.26451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Decker NK, Abdelmoneim SS, Yaqoob U, Hendrickson H, Hormes J, Bentley M, Pitot H, Urrutia R, Gores GJ, Shah VH. Nitric oxide regulates tumor cell cross-talk with stromal cells in the tumor microenvironment of the liver. Am J Pathol. 2008;173:1002–1012. doi: 10.2353/ajpath.2008.080158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.PeNarando J, Aranda E, RodrIguez-Ariza A. Immunomodulatory roles of nitric oxide in cancer: Tumor microenvironment says ‘NO’ to antitumor immune response. Transl Res. 2019;210:99–108. doi: 10.1016/j.trsl.2019.03.003. [DOI] [PubMed] [Google Scholar]
- 94.Arora H, Panara K, Kuchakulla M, Kulandavelu S, Burnstein KL, Schally AV, Hare JM, Ramasamy R. Alterations of tumor microenvironment by nitric oxide impedes castration-resistant prostate cancer growth. Proc Natl Acad Sci USA. 2018;115:11298–11303. doi: 10.1073/pnas.1812704115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Yang G. Hydrogen sulfide in cell survival: A double-edged sword. Exp Rev Clin Pharmacol. 2011;4:33–47. doi: 10.1586/ecp.10.131. [DOI] [PubMed] [Google Scholar]
- 96.Pei Y, Wu B, Cao Q, Wu L, Yang G. Hydrogen sulfide mediates the anti-survival effect of sulforaphane on human prostate cancer cells. Toxicol Appl Pharmacol. 2011;257:420–428. doi: 10.1016/j.taap.2011.09.026. [DOI] [PubMed] [Google Scholar]
- 97.Bhattacharyya S, Saha S, Giri K, Lanza IR, Nair KS, Jennings NB, Rodriguez-Aguayo C, Lopez-Berestein G, Basal E, Weaver AL, et al. Cystathionine beta-synthase (CBS) contributes to advanced ovarian cancer progression and drug resistance. PLoS One. 2013;8:e79167. doi: 10.1371/journal.pone.0079167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Hellmich MR, Coletta C, Chao C, Szabo C. The therapeutic potential of cystathionine β-synthetase/hydrogen sulfide inhibition in cancer. Antioxid Redox Signal. 2015;22:424–448. doi: 10.1089/ars.2014.5933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zhu H, Blake S, Chan KT, Pearson RB, Kang J. Cystathionine β-synthase in physiology and cancer. Biomed Res Int. 2018;2018:3205125. doi: 10.1155/2018/3205125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Hellmich MR, Szabo C. Chemistry, Biochemistry and Pharmacology of Hydrogen Sulfide. Springer; New York, NY: 2015. Hydrogen sulfide and cancer; pp. 233–241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Sen N. Functional and molecular insights of hydrogen sulfide signaling and protein sulfhydration. J Mol Biol. 2017;429:543–561. doi: 10.1016/j.jmb.2016.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Chattopadhyay M, Kodela R, Nath N, Dastagirzada YM, Velázquez-Martínez CA, Boring D, Kashfi K. Hydrogen sulfide-releasing NSAIDs inhibit the growth of human cancer cells: A general property and evidence of a tissue type-independent effect. Biochem Pharmacol. 2012;83:715–722. doi: 10.1016/j.bcp.2011.12.018. [DOI] [PubMed] [Google Scholar]
- 103.Szabo C, Ransy C, Módis K, Andriamihaja M, Murghes B, Coletta C, Olah G, Yanagi K, Bouillaud F. Regulation of mitochondrial bioenergetic function by hydrogen sulfide. Part I. Biochemical and physiological mechanisms. Br J Pharmacol. 2014;171:2099–2122. doi: 10.1111/bph.12369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Rose P, Moore PK, Zhu YZ. H2S biosynthesis and catabolism: New insights from molecular studies. Cell Mol Life Sci. 2017;74:1391–1412. doi: 10.1007/s00018-016-2406-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Cao X, Ding L, Xie ZZ, Yang Y, Whiteman M, Moore PK, Bian JS. A review of hydrogen sulfide synthesis, metabolism, and measurement: Is modulation of hydrogen sulfide a novel therapeutic for cancer? Antioxid Redox Signal. 2019;31:1–38. doi: 10.1089/ars.2017.7058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Giuffrè A, Tomé CS, Fernandes DGF, Zuhra K, Vicente JB. Hydrogen sulfide metabolism and signaling in the tumor microenvironment. Adv Exp Med Biol. 2020;1219:335–353. doi: 10.1007/978-3-030-34025-4_17. [DOI] [PubMed] [Google Scholar]
- 107.Wang RH, Chu YH, Lin KT. The hidden role of hydrogen sulfide metabolism in cancer. Int J Mol Sci. 2021;22:6562. doi: 10.3390/ijms22126562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Watnick RS. The role of the tumor microenvironment in regulating angiogenesis. Cold Spring Harb Perspect Med. 2012;2:a006676. doi: 10.1101/cshperspect.a006676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Zhou Y, Li XH, Zhang CC, Wang MJ, Xue WL, Wu DD, Ma FF, Li WW, Tao BB, Zhu YC. Hydrogen sulfide promotes angiogenesis by downregulating miR-640 via the VEGFR2/mTOR pathway. Am J Physiol Cell Physiol. 2016;310:C305–C317. doi: 10.1152/ajpcell.00230.2015. [DOI] [PubMed] [Google Scholar]
- 110.Wang M, Yan J, Cao X, Hua P, Li Z. Hydrogen sulfide modulates epithelial-mesenchymal transition and angiogenesis in non-small cell lung cancer via HIF-1α activation. Biochem Pharmacol. 2020;172:113775. doi: 10.1016/j.bcp.2019.113775. [DOI] [PubMed] [Google Scholar]
- 111.Szabo C, Hellmich MR. Endogenously produced hydrogen sulfide supports tumor cell growth and proliferation. Cell Cycle. 2013;12:2915–2916. doi: 10.4161/cc.26064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Holwerda KM, Burke SD, Faas MM, Zsengeller Z, Stillman IE, Kang PM, van Goor H, McCurley A, Jaffe IZ, Karumanchi SA, Lely AT. Hydrogen sulfide attenuates sFlt1-Induced hypertension and renal damage by upregulating vascular endothelial growth factor. J Am Soc Nephrol. 2014;25:717–725. doi: 10.1681/ASN.2013030291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Szabo C, Coletta C, Chao C, Módis K, Szczesny B, Papapetropoulos A, Hellmich MR. Tumor-derived hydrogen sulfide, produced by cystathionine-β-synthase, stimulates bioenergetics, cell proliferation, and angiogenesis in colon cancer. Proc Natl Acad Sci USA. 2013;110:12474–12479. doi: 10.1073/pnas.1306241110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Liu D, Liu M, Wan Y, Zhou X, Yang S, An L, Huang G, Tian Q. Remodeling endogenous H2S microenvironment in colon cancer to enhance chemodynamic therapy. Chem Engineering J. 2021;422:130098. doi: 10.1016/j.cej.2021.130098. [DOI] [Google Scholar]
- 115.Wan M, Liu Z, Li T, Chen H, Wang Q, Chen T, Tao Y, Mao C. Zwitterion-Based hydrogen sulfide nanomotors induce multiple acidosis in tumor cells by destroying tumor metabolic symbiosis. Angew Chem Int Ed Engl. 2021;60:16139–16148. doi: 10.1002/anie.202104304. [DOI] [PubMed] [Google Scholar]
- 116.Li D, Xiong Q, Peng J, Hu B, Li W, Zhu Y, Shen X. Hydrogen Sulfide Up-Regulates the expression of ATP-Binding cassette transporter A1 via promoting nuclear translocation of PPARα. Int J Mol Sci. 2016;17:635. doi: 10.3390/ijms17050635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Schulze A, Harris AL. How cancer metabolism is tuned for proliferation and vulnerable to disruption. Nature. 2012;491:364. doi: 10.1038/nature11706. [DOI] [PubMed] [Google Scholar]
- 118.Lee ZW, Teo XY, Tay EY, Tan CH, Hagen T, Moore PK, Deng LW. Utilizing hydrogen sulfide as a novel anti-cancer agent by targeting cancer glycolysis and pH imbalance. Br J Pharmacol. 2014;171:4322–4336. doi: 10.1111/bph.12773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Niland S, Riscanevo AX, Eble JA. Matrix metalloproteinases shape the tumor microenvironment in cancer progression. Int J Mol Sci. 2021;23:146. doi: 10.3390/ijms23010146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Harjunpää H, Llort Asens M, Guenther C, Fagerholm SC. Cell adhesion molecules and their roles and regulation in the immune and tumor microenvironment. Front Immunol. 2019;10:1078. doi: 10.3389/fimmu.2019.01078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Mao Y, Keller ET, Garfield DH, Shen K, Wang J. Stromal cells in tumor microenvironment and breast cancer. Cancer Metastasis Rev. 2013;32:303–315. doi: 10.1007/s10555-012-9415-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Zhen Y, Pan W, Hu F, Wu H, Feng J, Zhang Y, Chen J. Exogenous hydrogen sulfide exerts proliferation/anti-apoptosis/angiogenesis/migration effects via amplifying the activation of NF-κB pathway in PLC/PRF/5 hepatoma cells. Int J Oncol. 2015;46:2194–2204. doi: 10.3892/ijo.2015.2914. [DOI] [PubMed] [Google Scholar]
- 123.Li H, Xu F, Gao G, Gao X, Wu B, Zheng C, Wang P, Li Z, Hua H, Li D. Hydrogen sulfide and its donors: Novel antitumor and antimetastatic therapies for triple-negative breast cancer. Redox Biol. 2020;34:101564. doi: 10.1016/j.redox.2020.101564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Wu D, Li M, Tian W, Wang S, Cui L, Li H, Wang H, Ji A, Li Y. Hydrogen sulfide acts as a double-edged sword in human hepatocellular carcinoma cells through EGFR/ERK/MMP-2 and PTEN/AKT signaling pathways. Sci Rep. 2017;7:5134. doi: 10.1038/s41598-017-05457-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Wu D, Li J, Zhang Q, Tian W, Zhong P, Liu Z, Wang H, Wang H, Ji A, Li Y. Exogenous hydrogen sulfide regulates the growth of human thyroid carcinoma cells. Oxid Med Cell Longev. 2019;2019:6927298. doi: 10.1155/2019/6927298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Zhang L, Qi Q, Yang J, Sun D, Li C, Xue Y, Jiang Q, Tian Y, Xu C, Wang R. An anticancer role of hydrogen sulfide in human gastric cancer cells. Oxid Med Cell Longev. 2015;2015:636410. doi: 10.1155/2015/636410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Rose JJ, Wang L, Xu Q, McTiernan CF, Shiva S, Tejero J, Gladwin MT. Carbon monoxide poisoning: Pathogenesis, management, and future directions of therapy. Am J Respir Crit Care Med. 2017;195:596–606. doi: 10.1164/rccm.201606-1275CI. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Motterlini R, Otterbein LE. The therapeutic potential of carbon monoxide. Nat Rev Drug Discov. 2010;9:728–743. doi: 10.1038/nrd3228. [DOI] [PubMed] [Google Scholar]
- 129.Wegiel B, Nemeth Z, Correa-Costa M, Bulmer AC, Otterbein LE. Heme Oxygenase-1: A Metabolic Nike. Antioxid Redox Signal. 2014;20:1709–1722. doi: 10.1089/ars.2013.5667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Ismailova A, Kuter D, Bohle DS, Butler IS. An overview of the potential therapeutic applications of CO-Releasing molecules. Bioinorg Chem Appl. 2018;2018:8547364. doi: 10.1155/2018/8547364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Ji X, Damera K, Zheng Y, Yu B, Otterbein LE, Wang B. Toward carbon monoxide-based therapeutics: Critical drug delivery and developability issues. J Pharm Sci. 2016;105:406–416. doi: 10.1016/j.xphs.2015.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.McCoubrey WK, Jr, Huang T, Maines MD. Isolation and characterization of a cDNA from the rat brain that encodes hemoprotein heme oxygenase-3. Eur J Biochem. 1997;247:725–732. doi: 10.1111/j.1432-1033.1997.00725.x. [DOI] [PubMed] [Google Scholar]
- 133.Otterbein LE, Soares MP, Yamashita K, Bach FH. Heme oxygenase-1: Unleashing the protective properties of heme. Trends Immunol. 2003;24:449–455. doi: 10.1016/S1471-4906(03)00181-9. [DOI] [PubMed] [Google Scholar]
- 134.Ryter SW, Alam J, Choi AMK. Heme Oxygenase-1/Carbon monoxide: From basic science to therapeutic applications. Physiol Rev. 2006;86:583–650. doi: 10.1152/physrev.00011.2005. [DOI] [PubMed] [Google Scholar]
- 135.Chen S, Wang X, Nisar MF, Lin M, Zhong JL. Heme Oxygenases: Cellular multifunctional and protective molecules against UV–Induced oxidative stress. Oxid Med Cell Longev. 2019;2019:5416728. doi: 10.1155/2019/5416728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Goebel U, Wollborn J. Carbon monoxide in intensive care medicine-time to start the therapeutic application? Intensive Care Med Exp. 2020;8:2. doi: 10.1186/s40635-020-0292-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Zuckerbraun BS, Chin BY, Bilban M, d'Avila JC, Rao J, Billiar TR, Otterbein LE. Carbon monoxide signals via inhibition of cytochrome c oxidase and generation of mitochondrial reactive oxygen species. FASEB J. 2007;21:1099–1106. doi: 10.1096/fj.06-6644com. [DOI] [PubMed] [Google Scholar]
- 138.Chin BY, Jiang G, Wegiel B, Wang HJ, Macdonald T, Zhang XC, Gallo D, Cszimadia E, Bach FH, Lee PJ, Otterbein LE. Hypoxia-inducible factor 1alpha stabilization by carbon monoxide results in cytoprotective preconditioning. Proc Natl Acad Sci USA. 2007;104:5109–5114. doi: 10.1073/pnas.0609611104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Hanahan D, Weinberg RA. Hallmarks of cancer: The next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 140.Shefa U, Yeo SG, Kim MS, Song IO, Jung J, Jeong NY, Huh Y. Role of gasotransmitters in oxidative stresses, neuroinflammation, and neuronal repair. Biomed Res. 2017;2017:1689341. doi: 10.1155/2017/1689341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Yang C, Jeong S, Ku S, Lee K, Park MH. Use of gasotransmitters for the controlled release of polymer-based nitric oxide carriers in medical applications. J Control Release. 2018;279:157–170. doi: 10.1016/j.jconrel.2018.04.025. [DOI] [PubMed] [Google Scholar]
- 142.Hassan AY, Maulood IM, Salihi A. The vasodilatory mechanism of nitric oxide and hydrogen sulfide in the human mesenteric artery in patients with colorectal cancer. Exp Ther Med. 2021;21:214. doi: 10.3892/etm.2021.9646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Konovalova NP, Goncharova SA, Volkova LM, Rajewskaya TA, Eremenko LT, Korolev AM. Nitric oxide donor increases the efficiency of cytostatic therapy and retards the development of drug resistance. Nitric Oxide. 2003;8:59–64. doi: 10.1016/S1089-8603(02)00142-8. [DOI] [PubMed] [Google Scholar]
- 144.Weyerbrock A, Baumer B, Papazoglou A. Growth inhibition and chemosensitization of exogenous nitric oxide released from NONOates in glioma cells in vitro. J Neurosurg. 2009;110:128–136. doi: 10.3171/2008.6.17607. [DOI] [PubMed] [Google Scholar]
- 145.Wink DA, Cook JA, Christodoulou D, Krishna MC, Pacelli R, Kim S, DeGraff W, Gamson J, Vodovotz Y, Russo A, Mitchell JB. Nitric oxide and some nitric oxide donor compounds enhance the cytotoxicity of cisplatin. Nitric Oxide. 1997;1:88–94. doi: 10.1006/niox.1996.0108. [DOI] [PubMed] [Google Scholar]
- 146.Huerta-Yepez S, Vega M, Escoto-Chavez SE, Murdock B, Sakai T, Baritaki S, Bonavida B. Nitric oxide sensitizes tumor cells to TRAIL-induced apoptosis via inhibition of the DR5 transcription repressor Yin Yang 1. Nitric Oxide. 2009;20:39–52. doi: 10.1016/j.niox.2008.08.001. [DOI] [PubMed] [Google Scholar]
- 147.Huerta S, Baay-Guzman G, Gonzalez-Bonilla CR, Livingston EH, Huerta-Yepez S, Bonavida B. In vitro and in vivo sensitization of SW620 metastatic colon cancer cells to CDDP-induced apoptosis by the nitric oxide donor DETANONOate: Involvement of AIF. Nitric Oxide. 2009;20:182–194. doi: 10.1016/j.niox.2008.11.006. [DOI] [PubMed] [Google Scholar]
- 148.Bratasz A, Selvendiran K, Wasowicz T, Bobko A, Khramtsov VV, Ignarro LJ, Kuppusamy P. NCX-4040, a nitric oxide-releasing aspirin, sensitizes drug-resistant human ovarian xenograft tumors to cisplatin by depletion of cellular thiols. J Transl Med. 2008;6:9. doi: 10.1186/1479-5876-6-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Safdar S, Payne CA, Tu NH, Taite LJ. Targeted nitric oxide delivery preferentially induces glioma cell chemosensitivity via altered p53 and O(6)-methylguanine-DNA methyltransferase activity. Biotechnol Bioengering. 2013;110:1211–1220. doi: 10.1002/bit.24775. [DOI] [PubMed] [Google Scholar]
- 150.Shami PJ, Saavedra JE, Wang LY, Bonifant CL, Diwan BA, Singh SV, Gu Y, Fox SD, Buzard GS, Citro ML. JS-K, a glutathione/glutathione S-transferase-activated nitric oxide donor of the diazeniumdiolate class with potent antineoplastic activity. Mol Cancer Ther. 2003;2:409–417. [PubMed] [Google Scholar]
- 151.Qiu M, Ke L, Zhang S, Zeng X, Fang Z, Liu J. JS-K, a GST-activated nitric oxide donor prodrug, enhances chemo-sensitivity in renal carcinoma cells and prevents cardiac myocytes toxicity induced by Doxorubicin. Cancer Chemother Pharmacol. 2017;80:275–286. doi: 10.1007/s00280-017-3359-9. [DOI] [PubMed] [Google Scholar]
- 152.Laschak M, Spindler KD, Schrader AJ, Hessenauer A, Streicher W, Schrader M, Cronauer MV. JS-K, a glutathione/glutathione S-transferase-activated nitric oxide releasing prodrug inhibits androgen receptor and WNT-signaling in prostate cancer cells. BMC Cancer. 2012;12:130. doi: 10.1186/1471-2407-12-130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Qiu M, Chen L, Tan G, Ke L, Zhang S, Chen H, Liu J. A reactive oxygen species activation mechanism contributes to JS-K-induced apoptosis in human bladder cancer cells. Sci Rep. 2015;5:15104. doi: 10.1038/srep15104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Riganti C, Doublier S, Costamagna C, Aldieri E, Pescarmona G, Ghigo D, Bosia A. Activation of nuclear factor-kappa B pathway by simvastatin and RhoA silencing increases doxorubicin cytotoxicity in human colon cancer HT29 cells. Mol Pharmacol. 2008;74:476–484. doi: 10.1124/mol.108.045286. [DOI] [PubMed] [Google Scholar]
- 155.De Boo S, Kopecka J, Brusa D, Gazzano E, Matera L, Ghigo D, Bosia A, Riganti C. iNOS activity is necessary for the cytotoxic and immunogenic effects of doxorubicin in human colon cancer cells. Mol Cancer. 2009;8:108. doi: 10.1186/1476-4598-8-108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Alimoradi H, Greish K, Barzegar-Fallah A, Alshaibani L, Pittalà V. Nitric oxide-releasing nanoparticles improve doxorubicin anticancer activity. Int J Nanomedicine. 2018;13:7771–7787. doi: 10.2147/IJN.S187089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Sung YC, Jin PR, Chu LA, Hsu FF, Wang MR, Chang CC, Chiou SJ, Qiu JT, Gao DY, Lin CC, et al. Delivery of nitric oxide with a nanocarrier promotes tumour vessel normalization and potentiates anti-cancer therapies. Nat Nanotechnol. 2019;14:1160–1169. doi: 10.1038/s41565-019-0570-3. [DOI] [PubMed] [Google Scholar]
- 158.Pieretti JC, Pelegrino MT, Nascimento MHM, Tortella GR, Rubilar O, Seabra AB. Small molecules for great solutions: Can nitric oxide-releasing nanomaterials overcome drug resistance in chemotherapy? Biochem Pharmacol. 2020;176:113740. doi: 10.1016/j.bcp.2019.113740. [DOI] [PubMed] [Google Scholar]
- 159.Yasuda H, Nakayama K, Watanabe M, Suzuki S, Fuji H, Okinaga S, Kanda A, Zayasu K, Sasaki T, Asada M, et al. Nitroglycerin treatment may enhance chemosensitivity to docetaxel and carboplatin in patients with lung adenocarcinoma. Clin Cancer Res. 2006;12:6748–6757. doi: 10.1158/1078-0432.CCR-06-1124. [DOI] [PubMed] [Google Scholar]
- 160.Aalaei S, Mohammadzadeh M, Pazhang Y. Synergistic induction of apoptosis in a cell model of human leukemia K562 by nitroglycerine and valproic acid. EXCLI J. 2019;18:619–630. doi: 10.17179/excli2019-1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Kaoud TS, Mohassab AM, Hassan HA, Yan C, Van Ravenstein SX, Abdelhamid D, Dalby KN, Abdel-Aziz M. NO-releasing STAT3 inhibitors suppress BRAF-mutant melanoma growth. Eur J Med Chem. 2020;186:111885. doi: 10.1016/j.ejmech.2019.111885. [DOI] [PubMed] [Google Scholar]
- 162.Ishima Y, Kragh-Hansen U, Maruyama T, Otagiri M. Poly-s-nitrosated albumin as a safe and effective multifunctional antitumor agent: Characterization, biochemistry and possible future therapeutic applications. Biomed Res Int. 2013;2013:353892. doi: 10.1155/2013/353892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Ishima Y, Inoue A, Fang J, Kinoshita R, Ikeda M, Watanabe H, Maeda H, Otagiri M, Maruyama T. Poly-S-nitrosated human albumin enhances the antitumor and antimetastasis effect of bevacizumab, partly by inhibiting autophagy through the generation of nitric oxide. Cancer Sci. 2015;106:194–200. doi: 10.1111/cas.12577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Li L, Whiteman M, Guan YY, Neo KL, Cheng Y, Lee SW, Zhao Y, Baskar R, Tan CH, Moore PK. Characterization of a novel, water-soluble hydrogen sulfide-releasing molecule (GYY4137): New insights into the biology of hydrogen sulfide. Circulation. 2008;117:2351–2360. doi: 10.1161/CIRCULATIONAHA.107.753467. [DOI] [PubMed] [Google Scholar]
- 165.Lee ZW, Zhou J, Chen CS, Zhao Y, Tan CH, Li L, Moore PK, Deng LW. The slow-releasing hydrogen sulfide donor, GYY4137, exhibits novel anti-cancer effects in vitro and in vivo. PLoS One. 2011;6:e21077. doi: 10.1371/journal.pone.0021077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Nalli AD, Bhattacharya S, Wang H, Kendig DM, Grider JR, Murthy KS. Augmentation of cGMP/PKG pathway and colonic motility by hydrogen sulfide. Am J Physiol Gastrointest Liver Physiol. 2017;313:G330–G341. doi: 10.1152/ajpgi.00161.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Liu B, Huang X, Li Y, Liao W, Li M, Liu Y, He R, Feng D, Zhu R, Kurihara H. JS-K, a nitric oxide donor, induces autophagy as a complementary mechanism inhibiting ovarian cancer. BMC Cancer. 2019;19:645. doi: 10.1186/s12885-019-5619-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Fonseca MD, Cunha FQ, Kashfi K, Cunha TM. NOSH-aspirin (NBS-1120), a dual nitric oxide and hydrogen sulfide-releasing hybrid, reduces inflammatory pain. Pharmacol Res Perspect. 2015;3:e00133. doi: 10.1002/prp2.133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Rose P, Dymock BW, Moore PK. GYY4137, a novel water-soluble, H2S-releasing molecule. Methods Enzymol. 2015;554:143–167. doi: 10.1016/bs.mie.2014.11.014. [DOI] [PubMed] [Google Scholar]
- 170.Aaltoma SH, Lipponen PK, Kosma VM. Inducible nitric oxide synthase (iNOS) expression and its prognostic value in prostate cancer. Anticancer Res. 2001;21:3101–3106. [PubMed] [Google Scholar]
- 171.Aaltomaa SH, Lipponen PK, Viitanen J, Kankkunen JP, Ala-Opas MY, Kosma VM. The prognostic value of inducible nitric oxide synthase in local prostate cancer. BJU Int. 2000;86:234–239. doi: 10.1046/j.1464-410x.2000.00787.x. [DOI] [PubMed] [Google Scholar]
- 172.Bhowmick R, Girotti AW. Pro-survival and pro-growth effects of stress-induced nitric oxide in a prostate cancer photodynamic therapy model. Cancer Lett. 2014;343:115–122. doi: 10.1016/j.canlet.2013.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Coulter JA, Page NL, Worthington J, Robson T, Hirst DG, McCarthy HO. Transcriptional regulation of inducible nitric oxide synthase gene therapy: Targeting early stage and advanced prostate cancer. J Gene Med. 2010;12:755–765. doi: 10.1002/jgm.1495. [DOI] [PubMed] [Google Scholar]
- 174.Cronauer MV, Ince Y, Engers R, Rinnab L, Weidemann W, Suschek CV, Burchardt M, Kleinert H, Wiedenmann J, Sies H, et al. Nitric oxide-mediated inhibition of androgen receptor activity: Possible implications for prostate cancer progression. Oncogene. 2007;26:1875–1884. doi: 10.1038/sj.onc.1209984. [DOI] [PubMed] [Google Scholar]
- 175.Erlandsson A, Carlsson J, Andersson SO, Vyas C, Wikström P, Andrén O, Davidsson S, Rider JR. High inducible nitric oxide synthase in prostate tumor epithelium is associated with lethal prostate cancer. Scand J Urol. 2018;52:129–133. doi: 10.1080/21681805.2017.1421261. [DOI] [PubMed] [Google Scholar]
- 176.McCarthy HO, Coulter JA, Worthington J, Robson T, Hirst DG. Human osteocalcin: A strong promoter for nitric oxide synthase gene therapy, with specificity for hormone refractory prostate cancer. J Gene Med. 2007;9:511–520. doi: 10.1002/jgm.1045. [DOI] [PubMed] [Google Scholar]
- 177.Fahey JM, Girotti AW. Accelerated migration and invasion of prostate cancer cells after a photodynamic therapy-like challenge: Role of nitric oxide. Nitric Oxide. 2015;49:47–55. doi: 10.1016/j.niox.2015.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.D'Este F, Della Pietra E, Badillo Pazmay GV, Xodo LE, Rapozzi V. Role of nitric oxide in the response to photooxidative stress in prostate cancer cells. Biochem Pharmacol. 2020;182:114205. doi: 10.1016/j.bcp.2020.114205. [DOI] [PubMed] [Google Scholar]
- 179.Ryk C, de Verdier P, Montgomery E, Peter Wiklund N, Wiklund F, Gronberg H. Polymorphisms in the nitric-oxide synthase 2 gene and prostate cancer pathogenesis. Redox Biol. 2015;5:419. doi: 10.1016/j.redox.2015.09.029. [DOI] [PubMed] [Google Scholar]
- 180.Uotila P, Valve E, Martikainen P, Nevalainen M, Nurmi M, Harkonen P. Increased expression of cyclooxygenase-2 and nitric oxide synthase-2 in human prostate cancer. Urol Res. 2001;29:23–28. doi: 10.1007/s002400000148. [DOI] [PubMed] [Google Scholar]
- 181.Abdelmagid SA, Rickard JA, McDonald WJ, Thomas LN, Too CK. CAT-1-mediated arginine uptake and regulation of nitric oxide synthases for the survival of human breast cancer cell lines. J Cell Biochem. 2011;112:1084–1092. doi: 10.1002/jcb.23022. [DOI] [PubMed] [Google Scholar]
- 182.Abdelmagid SA, Too CK. Prolactin and estrogen up-regulate carboxypeptidase-d to promote nitric oxide production and survival of mcf-7 breast cancer cells. Endocrinology. 2008;149:4821–4828. doi: 10.1210/en.2008-0145. [DOI] [PubMed] [Google Scholar]
- 183.Ambs S, Glynn SA. Candidate pathways linking inducible nitric oxide synthase to a basal-like transcription pattern and tumor progression in human breast cancer. Cell Cycle. 2011;10:619–624. doi: 10.4161/cc.10.4.14864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Alalami O, Martin JH. ZR-75-1 human breast cancer cells: Expression of inducible nitric oxide synthase and effect of tamoxifen and phorbol ester on nitric oxide production. Cancer Lett. 1998;123:99–105. doi: 10.1016/S0304-3835(97)00404-7. [DOI] [PubMed] [Google Scholar]
- 185.Basudhar D, Somasundaram V, de Oliveira GA, Kesarwala A, Heinecke JL, Cheng RY, Glynn SA, Ambs S, Wink DA, Ridnour LA. Nitric oxide synthase-2-derived nitric oxide drives multiple pathways of breast cancer progression. Antioxid Redox Signal. 2017;26:1044–1058. doi: 10.1089/ars.2016.6813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Bentrari F, Arnould L, Jackson AP, Jeannin JF, Pance A. Progesterone enhances cytokine-stimulated nitric oxide synthase II expression and cell death in human breast cancer cells. Lab Invest. 2005;85:624–632. doi: 10.1038/labinvest.3700267. [DOI] [PubMed] [Google Scholar]
- 187.Bing RJ, Miyataka M, Rich KA, Hanson N, Wang X, Slosser HD, Shi SR. Nitric oxide, prostanoids, cyclooxygenase, and angiogenesis in colon and breast cancer. Clin Cancer Res. 2001;7:3385–3392. [PubMed] [Google Scholar]
- 188.Deepa M, Sureshkumar T, Satheeshkumar PK, Priya S. Antioxidant rich Morus alba leaf extract induces apoptosis in human colon and breast cancer cells by the downregulation of nitric oxide produced by inducible nitric oxide synthase. Nutr Cancer. 2013;65:305–310. doi: 10.1080/01635581.2013.748924. [DOI] [PubMed] [Google Scholar]
- 189.Duenas-Gonzalez A, Isales CM, del Mar Abad-Hernandez M, Gonzalez-Sarmiento R, Sangueza O, Rodriguez-Commes J. Expression of inducible nitric oxide synthase in breast cancer correlates with metastatic disease. Mod Pathol. 1997;10:645–649. [PubMed] [Google Scholar]
- 190.Evig CB, Kelley EE, Weydert CJ, Chu Y, Buettner GR, Burns CP. Endogenous production and exogenous exposure to nitric oxide augment doxorubicin cytotoxicity for breast cancer cells but not cardiac myoblasts. Nitric Oxide. 2004;10:119–129. doi: 10.1016/j.niox.2004.03.006. [DOI] [PubMed] [Google Scholar]
- 191.Flaherty RL, Intabli H, Falcinelli M, Bucca G, Hesketh A, Patel BA, Allen MC, Smith CP, Flint MS. Stress hormone-mediated acceleration of breast cancer metastasis is halted by inhibition of nitric oxide synthase. Cancer letters. 2019;459:59–71. doi: 10.1016/j.canlet.2019.05.027. [DOI] [PubMed] [Google Scholar]
- 192.Garrido P, Shalaby A, Walsh EM, Keane N, Webber M, Keane MM, Sullivan FJ, Kerin MJ, Callagy G, Ryan AE, Glynn SA. Impact of inducible nitric oxide synthase (iNOS) expression on triple negative breast cancer outcome and activation of EGFR and ERK signaling pathways. Oncotarget. 2017;8:80568–80588. doi: 10.18632/oncotarget.19631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Hsu JT, Ying C, Chen CJ. Regulation of inducible nitric oxide synthase by dietary phytoestrogen in MCF-7 human mammary cancer cells. Reprod Nutr Dev. 2000;40:11–18. doi: 10.1051/rnd:2000116. [DOI] [PubMed] [Google Scholar]
- 194.Kanugula AK, Gollavilli PN, Vasamsetti SB, Karnewar S, Gopoju R, Ummanni R, Kotamraju S. Statin-induced inhibition of breast cancer proliferation and invasion involves attenuation of iron transport: Intermediacy of nitric oxide and antioxidant defence mechanisms. FEBS J. 2014;281:3719–3738. doi: 10.1111/febs.12893. [DOI] [PubMed] [Google Scholar]
- 195.Kotamraju S, Williams CL, Kalyanaraman B. Statin-induced breast cancer cell death: Role of inducible nitric oxide and arginase-dependent pathways. Cancer Res. 2007;67:7386–7394. doi: 10.1158/0008-5472.CAN-07-0993. [DOI] [PubMed] [Google Scholar]
- 196.López-Sánchez LM, Mena R, Guil-Luna S, Mantrana A, Peñarando J, Toledano-Fonseca M, Conde F, De la Haba-Rodríguez JR, Aranda E, Rodríguez-Ariza A. Nitric oxide-targeted therapy inhibits stemness and increases the efficacy of tamoxifen in estrogen receptor-positive breast cancer cells. Lab Invest. 2021;101:292–303. doi: 10.1038/s41374-020-00507-z. [DOI] [PubMed] [Google Scholar]
- 197.Loibl S, Buck A, Strank C, von Minckwitz G, Roller M, Sinn HP, Schini-Kerth V, Solbach C, Strebhardt K, Kaufmann M. The role of early expression of inducible nitric oxide synthase in human breast cancer. Eur J Cancer. 2005;41:265–271. doi: 10.1016/j.ejca.2004.07.010. [DOI] [PubMed] [Google Scholar]
- 198.Lahiri M, Martin JH. Reduced expression of endothelial and inducible nitric oxide synthase in a multidrug resistant variant of the MCF-7 human breast cancer cell line. Oncol Rep. 2004;12:1007–1011. doi: 10.3892/or.12.5.1007. [DOI] [PubMed] [Google Scholar]
- 199.Oktem G, Karabulut B, Selvi N, Sezgin C, Sanli UA, Uslu R, Yurtseven ME, Omay SB. Differential effects of doxorubicin and docetaxel on nitric oxide production and inducible nitric oxide synthase expression in MCF-7 human breast cancer cells. Oncol Res. 2004;14:381–386. doi: 10.3727/0965040041292305. [DOI] [PubMed] [Google Scholar]
- 200.Martin JH, Alalami O, van den Berg HW. Reduced expression of endothelial and inducible nitric oxide synthase in a human breast cancer cell line which has acquired estrogen independence. Cancer Lett. 1999;144:65–74. doi: 10.1016/S0304-3835(99)00198-6. [DOI] [PubMed] [Google Scholar]
- 201.Nafea H, Youness RA, Abou-Aisha K, Gad MZ. LncRNA HEIH/miR-939-5p interplay modulates triple-negative breast cancer progression through NOS2-induced nitric oxide production. J Cell Physiol. 2021;236:5362–5372. doi: 10.1002/jcp.30234. [DOI] [PubMed] [Google Scholar]
- 202.Oktem G, Bilir A, Selvi N, Yurtseven ME, Vatansever S, Ates U, Uysal A, Omay SB. Chemotherapy influences inducible nitric oxide synthase (iNOS) and endothelial nitric oxide synthase (eNOS) activity on 3D breast cancer cell line. Oncol Res. 2006;16:195–203. doi: 10.3727/000000006783981107. [DOI] [PubMed] [Google Scholar]
- 203.Zeybek ND, Inan S, Ekerbicer N, Vatansever HS, Karakaya J, Muftuoglu SF. The effects of Gemcitabine and Vinorelbine on inducible nitric oxide synthase (iNOS) and endothelial nitric oxide synthase (eNOS) distribution of MCF-7 breast cancer cells. Acta Histochem. 2011;113:62–67. doi: 10.1016/j.acthis.2009.07.006. [DOI] [PubMed] [Google Scholar]
- 204.Walsh EM, Keane MM, Wink DA, Callagy G, Glynn SA. Review of triple negative breast cancer and the impact of inducible nitric oxide synthase on tumor biology and patient outcomes. Crit Rev Oncog. 2016;21:333–351. doi: 10.1615/CritRevOncog.2017021307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Wink DA, Ridnour LA, Cheng R, Switzer CW, Glynn S, Ambs S. The oncogenic properties of the redox inflammatory protein inducible nitric oxide synthase in ER(−) breast cancer. Redox Biol. 2015;5:413. doi: 10.1016/j.redox.2015.09.012. [DOI] [PubMed] [Google Scholar]
- 206.Tschugguel W, Schneeberger C, Unfried G, Czerwenka K, Weninger W, Mildner M, Gruber DM, Sator MO, Waldhör T, Huber JC. Expression of inducible nitric oxide synthase in human breast cancer depends on tumor grade. Breast Cancer Res Treat. 1999;56:145–151. doi: 10.1023/A:1006288526311. [DOI] [PubMed] [Google Scholar]
- 207.Abadi AT, Kusters JG. Association of inducible nitric oxide synthetase genotype and Helicobacter pylori infection gastric cancer risk may be due to faulty primer design. World J Gastroenterol. 2013;19:429–430. doi: 10.3748/wjg.v19.i3.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Chen CN, Hsieh FJ, Cheng YM, Chang KJ, Lee PH. Expression of inducible nitric oxide synthase and cyclooxygenase-2 in angiogenesis and clinical outcome of human gastric cancer. J Surg Oncol. 2006;94:226–233. doi: 10.1002/jso.20372. [DOI] [PubMed] [Google Scholar]
- 209.Doi C, Noguchi Y, Marat D, Saito A, Fukuzawa K, Yoshikawa T, Tsuburaya A, Ito T. Expression of nitric oxide synthase in gastric cancer. Cancer Lett. 1999;144:161–167. doi: 10.1016/S0304-3835(99)00222-0. [DOI] [PubMed] [Google Scholar]
- 210.Goto T, Haruma K, Kitadai Y, Ito M, Yoshihara M, Sumii K, Hayakawa N, Kajiyama G. Enhanced expression of inducible nitric oxide synthase and nitrotyrosine in gastric mucosa of gastric cancer patients. Clin Cancer Res. 1999;5:1411–1415. [PubMed] [Google Scholar]
- 211.Goto Y, Ando T, Naito M, Goto H, Hamajima N. Inducible nitric oxide synthase polymorphism is associated with the increased risk of differentiated gastric cancer in a Japanese population. World J Gastroenterol. 2006;12:6361–6365. doi: 10.3748/wjg.v12.i39.6361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Hirst D, Robson T. Nitric oxide in cancer therapeutics: Interaction with cytotoxic chemotherapy. Curr Pharm Des. 2010;16:411–420. doi: 10.2174/138161210790232185. [DOI] [PubMed] [Google Scholar]
- 213.Hirahashi M, Koga Y, Kumagai R, Aishima S, Taguchi K, Oda Y. Induced nitric oxide synthetase and peroxiredoxin expression in intramucosal poorly differentiated gastric cancer of young patients. Pathol Int. 2014;64:155–163. doi: 10.1111/pin.12152. [DOI] [PubMed] [Google Scholar]
- 214.Huang FY, Chan AO, Rashid A, Wong DK, Cho CH, Yuen MF. Helicobacter pylori induces promoter methylation of E-cadherin via interleukin-1β activation of nitric oxide production in gastric cancer cells. Cancer. 2012;118:4969–4980. doi: 10.1002/cncr.27519. [DOI] [PubMed] [Google Scholar]
- 215.Ikeguchi M, Ueta T, Kaibara N. Expression of inducible nitric oxide synthase during cisplatin-based chemotherapy for gastric cancer. In Vivo. 2002;16:185–190. [PubMed] [Google Scholar]
- 216.Jun Y, Fei G, Ebert MP, Malfertheiner P. Expression of inducible nitric oxide synthase in human gastric cancer. World J Gastroenterol. 1999;5:430–431. doi: 10.3748/wjg.v5.i5.430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Jung ID, Lee JS, Yun SY, Park CG, Han JW, Lee HW, Lee HY. Doxorubicin inhibits the production of nitric oxide by colorectal cancer cells. Arch Pharm Res. 2002;25:691–696. doi: 10.1007/BF02976946. [DOI] [PubMed] [Google Scholar]
- 218.Rafiei A, Hosseini V, Janbabai G, Fazli B, Ajami A, Hosseini-Khah Z, Gilbreath J, Merrell DS. Inducible nitric oxide synthetase genotype and helicobacter pylori infection affect gastric cancer risk. World J Gastroenterol. 2012;18:4917–4924. doi: 10.3748/wjg.v18.i35.4917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Rajnakova A, Moochhala S, Goh PM, Ngoi S. Expression of nitric oxide synthase, cyclooxygenase, and p53 in different stages of human gastric cancer. Cancer Lett. 2001;172:177–185. doi: 10.1016/S0304-3835(01)00645-0. [DOI] [PubMed] [Google Scholar]
- 220.Zhang W, He XJ, Ma YY, Wang HJ, Xia YJ, Zhao ZS, Ye ZY, Tao HQ. Inducible nitric oxide synthase expression correlates with angiogenesis, lymphangiogenesis, and poor prognosis in gastric cancer patients. Hum Pathol. 2011;42:1275–1282. doi: 10.1016/j.humpath.2010.09.020. [DOI] [PubMed] [Google Scholar]
- 221.Sawa T, Mounawar M, Tatemichi M, Gilibert I, Katoh T, Ohshima H. Increased risk of gastric cancer in Japanese subjects is associated with microsatellite polymorphisms in the heme oxygenase-1 and the inducible nitric oxide synthase gene promoters. Cancer Lett. 2008;269:78–84. doi: 10.1016/j.canlet.2008.04.015. [DOI] [PubMed] [Google Scholar]
- 222.Shen J, Wang RT, Wang LW, Xu YC, Wang XR. A novel genetic polymorphism of inducible nitric oxide synthase is associated with an increased risk of gastric cancer. World J Gastroenterol. 2004;10:3278–3283. doi: 10.3748/wjg.v10.i22.3278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Son HJ, Kim YH, Park DI, Kim JJ, Rhee PL, Paik SW, Choi KW, Song SY, Rhee JC. Interaction between cyclooxygenase-2 and inducible nitric oxide synthase in gastric cancer. J Clin Gastroenterol. 2001;33:383–388. doi: 10.1097/00004836-200111000-00008. [DOI] [PubMed] [Google Scholar]
- 224.Song ZY, Wen SQ, Peng JP, Huang X, Qian KD. Significance of vascular endothelial growth factor expression and its correlation with inducible nitric oxide synthase in gastric cancer. World J Gastroenterol. 2004;10:1250–1255. doi: 10.3748/wjg.v10.i9.1250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Ambs S, Merriam WG, Bennett WP, Felley-Bosco E, Ogunfusika MO, Oser SM, Klein S, Shields PG, Billiar TR, Harris CC. Frequent nitric oxide synthase-2 expression in human colon adenomas: Implication for tumor angiogenesis and colon cancer progression. Cancer Res. 1998;58:334–341. [PubMed] [Google Scholar]
- 226.Bocca C, Bozzo F, Bassignana A, Miglietta A. Antiproliferative effect of a novel nitro-oxy derivative of celecoxib in human colon cancer cells: Role of COX-2 and nitric oxide. Anticancer Res. 2010;30:2659–2666. [PubMed] [Google Scholar]
- 227.Dabrowska M, Uram L, Zielinski Z, Rode W, Sikora E. Oxidative stress and inhibition of nitric oxide generation underlie methotrexate-induced senescence in human colon cancer cells. Mech Ageing Dev. 2018;170:22–29. doi: 10.1016/j.mad.2017.07.006. [DOI] [PubMed] [Google Scholar]
- 228.Narayanan BA, Narayanan NK, Desai D, Pittman B, Reddy BS. Effects of a combination of docosahexaenoic acid and 1,4-phenylene bis(methylene) selenocyanate on cyclooxygenase 2, inducible nitric oxide synthase and beta-catenin pathways in colon cancer cells. Carcinogenesis. 2004;25:2443–2449. doi: 10.1093/carcin/bgh252. [DOI] [PubMed] [Google Scholar]
- 229.Narayanan BA, Narayanan NK, Simi B, Reddy BS. Modulation of inducible nitric oxide synthase and related proinflammatory genes by the omega-3 fatty acid docosahexaenoic acid in human colon cancer cells. Cancer Res. 2003;63:972–979. [PubMed] [Google Scholar]
- 230.Kwak JY, Han MK, Choi KS, Park IH, Park SY, Sohn MH, Kim UH, McGregor JR, Samlowski WE, Yim CY. Cytokines secreted by lymphokine-activated killer cells induce endogenous nitric oxide synthesis and apoptosis in DLD-1 colon cancer cells. Cell Immunol. 2000;203:84–94. doi: 10.1006/cimm.2000.1682. [DOI] [PubMed] [Google Scholar]
- 231.Kapral M, Wawszczyk J, Sosnicki S, Weglarz L. Down-regulation of inducible nitric oxide synthase expression by inositol hexaphosphate in human colon cancer cells. Acta Pol Pharm. 2015;72:705–711. [PubMed] [Google Scholar]
- 232.Puglisi MA, Cenciarelli C, Tesori V, Cappellari M, Martini M, Di Francesco AM, Giorda E, Carsetti R, Ricci-Vitiani L, Gasbarrini A. High nitric oxide production, secondary to inducible nitric oxide synthase expression, is essential for regulation of the tumour-initiating properties of colon cancer stem cells. J Pathol. 2015;236:479–490. doi: 10.1002/path.4545. [DOI] [PubMed] [Google Scholar]
- 233.Williams JL, Nath N, Chen J, Hundley TR, Gao J, Kopelovich L, Kashfi K, Rigas B. Growth inhibition of human colon cancer cells by nitric oxide (NO)-donating aspirin is associated with cyclooxygenase-2 induction and beta-catenin/T-cell factor signaling, nuclear factor-kappaB, and NO synthase 2 inhibition: Implications for chemoprevention. Cancer Res. 2003;63:7613–7618. [PubMed] [Google Scholar]
- 234.Tong X, Zheng S, Jin J, Zhu L, Lou Y, Yao H. Triptolide inhibits cyclooxygenase-2 and inducible nitric oxide synthase expression in human colon cancer and leukemia cells. Acta Biochim Biophys Sin (Shanghai) 2007;39:89–95. doi: 10.1111/j.1745-7270.2007.00254.x. [DOI] [PubMed] [Google Scholar]
- 235.Sasahara Y, Mutoh M, Takahashi M, Fukuda K, Tanaka N, Sugimura T, Wakabayashi K. Suppression of promoter-dependent transcriptional activity of inducible nitric oxide synthase by sodium butyrate in colon cancer cells. Cancer Lett. 2002;177:155–161. doi: 10.1016/S0304-3835(01)00778-9. [DOI] [PubMed] [Google Scholar]
- 236.Shen X, Chen J, Qiu R, Fan X, Xin Y. Effect of camptothecin on inducible nitric oxide synthase expression in the colon cancer SW480 cell line. Oncol Lett. 2015;10:3157–3160. doi: 10.3892/ol.2015.3658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Spiegel A, Hundley TR, Chen J, Ouyang N, Liu X, Go MF, Tsioulias GJ, Kashfi K, Rigas B. NO-donating aspirin inhibits both the expression and catalytic activity of inducible nitric oxide synthase in HT-29 human colon cancer cells. Biochem Pharmacol. 2005;70:993–1000. doi: 10.1016/j.bcp.2005.06.027. [DOI] [PubMed] [Google Scholar]
- 238.Eijan AM, Piccardo I, Riveros MD, Sandes EO, Porcella H, Jasnis MA, Sacerdote De Lustig E, Malagrino H, Pasik L, Casabé AR. Nitric oxide in patients with transitional bladder cancer. J Surg Oncol. 2002;81:203–208. doi: 10.1002/jso.10170. [DOI] [PubMed] [Google Scholar]
- 239.Hosseini A, Koskela LR, Ehren I, Aguilar-Santelises M, Sirsjo A, Wiklund NP. Enhanced formation of nitric oxide in bladder carcinoma in situ and in BCG treated bladder cancer. Nitric Oxide. 2006;15:337–343. doi: 10.1016/j.niox.2006.01.007. [DOI] [PubMed] [Google Scholar]
- 240.Klotz T, Bloch W, Jacobs G, Niggemann S, Engelmann U, Addicks K. Immunolocalization of inducible and constitutive nitric oxide synthases in human bladder cancer. Urology. 1999;54:416–419. doi: 10.1016/S0090-4295(99)00212-5. [DOI] [PubMed] [Google Scholar]
- 241.Koskela LR, Poljakovic M, Ehren I, Wiklund NP, de Verdier PJ. Localization and expression of inducible nitric oxide synthase in patients after BCG treatment for bladder cancer. Nitric Oxide. 2012;27:185–191. doi: 10.1016/j.niox.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 242.Langle YV, Balarino NP, Belgorosky D, Cresta Morgado PD, Sandes EO, Marino L, Bilbao ER, Zambrano M, Lodillinsky C, Eiján AM. Effect of nitric oxide inhibition in Bacillus Calmette-Guerin bladder cancer treatment. Nitric Oxide. 2020;98:50–59. doi: 10.1016/j.niox.2020.03.003. [DOI] [PubMed] [Google Scholar]
- 243.Lin Z, Chen S, Ye C, Zhu S. Nitric oxide synthase expression in human bladder cancer and its relation to angiogenesis. Urol Res. 2003;31:232–235. doi: 10.1007/s00240-003-0302-9. [DOI] [PubMed] [Google Scholar]
- 244.Ryk C, Steineck G, Wiklund NP, Nyberg T, de Verdier PJ. The (CCTTT)n microsatellite polymorphism in the nitric oxide synthase 2 gene may influence bladder cancer pathogenesis. J Urol. 2010;184:2150–2157. doi: 10.1016/j.juro.2010.06.091. [DOI] [PubMed] [Google Scholar]
- 245.Ryk C, Wiklund NP, Nyberg T, De Verdier PJ. Ser608Leu polymorphisms in the nitric oxide synthase-2 gene may influence urinary bladder cancer pathogenesis. Scand J Urol Nephrol. 2011;45:319–325. doi: 10.3109/00365599.2011.584901. [DOI] [PubMed] [Google Scholar]
- 246.Wolf H, Haeckel C, Roessner A. Inducible nitric oxide synthase expression in human urinary bladder cancer. Virchows Arch. 2000;437:662–666. doi: 10.1007/s004280000296. [DOI] [PubMed] [Google Scholar]
- 247.Sandes EO, Faletti AG, Riveros MD, Vidal Mdel C, Gimenez L, Casabé AR, Eiján AM. Expression of inducible nitric oxide synthase in tumoral and non-tumoral epithelia from bladder cancer patients. Nitric Oxide. 2005;12:39–45. doi: 10.1016/j.niox.2005.03.007. [DOI] [PubMed] [Google Scholar]
- 248.Sandes EO, Lodillinsky C, Langle Y, Belgorosky D, Marino L, Gimenez L, Casabé AR, Eiján AM. Inducible nitric oxide synthase and PPARγ are involved in bladder cancer progression. J Urol. 2012;188:967–973. doi: 10.1016/j.juro.2012.04.099. [DOI] [PubMed] [Google Scholar]
- 249.Ambs S, Harris CC. RESPONSE: Re: Relationship between p53 mutations and inducible nitric oxide synthase expression in human colorectal cancer. J Natl Cancer Inst. 1999;91:1510–1511. doi: 10.1093/jnci/91.17.1510. [DOI] [PubMed] [Google Scholar]
- 250.Chung P, Cook T, Liu K, Vodovotz Y, Zamora R, Finkelstein S, Billiar T, Blumberg D. Overexpression of the human inducible nitric oxide synthase gene enhances radiation-induced apoptosis in colorectal cancer cells via a caspase-dependent mechanism. Nitric Oxide. 2003;8:119–126. doi: 10.1016/S1089-8603(02)00147-7. [DOI] [PubMed] [Google Scholar]
- 251.Cianchi F, Cortesini C, Fantappie O, Messerini L, Sardi I, Lasagna N, Perna F, Fabbroni V, Di Felice A, Perigli G, et al. Cyclooxygenase-2 activation mediates the proangiogenic effect of nitric oxide in colorectal cancer. Clin Cancer Res. 2004;10:2694–2704. doi: 10.1158/1078-0432.CCR-03-0192. [DOI] [PubMed] [Google Scholar]
- 252.Cianchi F, Cortesini C, Fantappie O, Messerini L, Schiavone N, Vannacci A, Nistri S, Sardi I, Baroni G, Marzocca C, et al. Inducible nitric oxide synthase expression in human colorectal cancer: Correlation with tumor angiogenesis. Am J Pathol. 2003;162:793–801. doi: 10.1016/S0002-9440(10)63876-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Fransen K, Elander N, Soderkvist P. Nitric oxide synthase 2 (NOS2) promoter polymorphisms in colorectal cancer. Cancer Lett. 2005;225:99–103. doi: 10.1016/j.canlet.2005.02.006. [DOI] [PubMed] [Google Scholar]
- 254.Gallo O, Sardi I, Masini M, Franchi A. Re: Relationship between p53 mutations and inducible nitric oxide synthase expression in human colorectal cancer. J Natl Cancer Inst. 1999;91:1509–1511. doi: 10.1093/jnci/91.17.1509. [DOI] [PubMed] [Google Scholar]
- 255.Ropponen KM, Kellokoski JK, Lipponen PK, Eskelinen MJ, Alanne L, Alhava EM, Kosma VM. Expression of inducible nitric oxide synthase in colorectal cancer and its association with prognosis. Scand J Gastroenterol. 2000;35:1204–1211. doi: 10.1080/003655200750056709. [DOI] [PubMed] [Google Scholar]
- 256.Zafirellis K, Zachaki A, Agrogiannis G, Gravani K. Inducible nitric oxide synthase expression and its prognostic significance in colorectal cancer. APMIS. 2010;118:115–124. doi: 10.1111/j.1600-0463.2009.02569.x. [DOI] [PubMed] [Google Scholar]
- 257.Wang Z, Cook T, Alber S, Liu K, Kovesdi I, Watkins SK, Vodovotz Y, Billiar TR, Blumberg D. Adenoviral gene transfer of the human inducible nitric oxide synthase gene enhances the radiation response of human colorectal cancer associated with alterations in tumor vascularity. Cancer Res. 2004;64:1386–1395. doi: 10.1158/0008-5472.CAN-03-1307. [DOI] [PubMed] [Google Scholar]
- 258.Delarue FL, Taylor BS, Sebti SM. Ras and RhoA suppress whereas RhoB enhances cytokine-induced transcription of nitric oxide synthase-2 in human normal liver AKN-1 cells and lung cancer A-549 cells. Oncogene. 2001;20:6531–6537. doi: 10.1038/sj.onc.1204801. [DOI] [PubMed] [Google Scholar]
- 259.Hu C, Li G, Wu E. A study on the activity of nitric oxide in alveolar macrophages from patients with lung cancer. Hunan Yi Ke Da Xue Xue Bao. 1998;23:157–160. (In Chinese) [PubMed] [Google Scholar]
- 260.Li Y, Ma C, Shi X, Wen Z, Li D, Sun M, Ding H. Effect of nitric oxide synthase on multiple drug resistance is related to Wnt signaling in non-small cell lung cancer. Oncol Rep. 2014;32:1703–1708. doi: 10.3892/or.2014.3351. [DOI] [PubMed] [Google Scholar]
- 261.Liu CY, Wang CH, Chen TC, Lin HC, Yu CT, Kuo HP. Increased level of exhaled nitric oxide and up-regulation of inducible nitric oxide synthase in patients with primary lung cancer. Br J Cancer. 1998;78:534–541. doi: 10.1038/bjc.1998.528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Liu CY, Wang YM, Wang CL, Feng PH, Ko HW, Liu YH, Wu YC, Chu Y, Chung FT, Kuo CH, et al. Population alterations of L-arginase- and inducible nitric oxide synthase-expressed CD11b+/CD14-/CD15+/CD33+ myeloid-derived suppressor cells and CD8+ T lymphocytes in patients with advanced-stage non-small cell lung cancer. J Cancer Res Clin Oncol. 2010;136:35–45. doi: 10.1007/s00432-009-0634-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Zhang J, Li B, Ding X, Sun M, Li H, Yang M, Zhou C, Yu H, Liu H, Yu G. Genetic variants in inducible nitric oxide synthase gene are associated with the risk of radiation-induced lung injury in lung cancer patients receiving definitive thoracic radiation. Radiother Oncol. 2014;111:194–198. doi: 10.1016/j.radonc.2014.03.001. [DOI] [PubMed] [Google Scholar]
- 264.Ye S, Yang W, Wang Y, Ou W, Ma Q, Yu C, Ren J, Zhong G, Shi H, Yuan Z, et al. Cationic liposome-mediated nitric oxide synthase gene therapy enhances the antitumor effects of cisplatin in lung cancer. Int J Mol Med. 2013;31:33–42. doi: 10.3892/ijmm.2012.1171. [DOI] [PubMed] [Google Scholar]
- 265.Sim SH, Ahn YO, Yoon J, Kim TM, Lee SH, Kim DW, Heo DS. Influence of chemotherapy on nitric oxide synthase, indole-amine-2,3-dioxygenase and CD124 expression in granulocytes and monocytes of non-small cell lung cancer. Cancer Sci. 2012;103:155–160. doi: 10.1111/j.1349-7006.2011.02158.x. [DOI] [PubMed] [Google Scholar]
- 266.Franco L, Doria D, Bertazzoni E, Benini A, Bassi C. Increased expression of inducible nitric oxide synthase and cyclooxygenase-2 in pancreatic cancer. Prostaglandins Other Lipid Mediat. 2004;73:51–58. doi: 10.1016/j.prostaglandins.2003.12.001. [DOI] [PubMed] [Google Scholar]
- 267.Kong G, Kim E, Kim W, Lee YW, Lee JK, Paik SW, Rhee JC, Choi KW, Lee KT. Inducible nitric oxide synthase (iNOS) immunoreactivity and its relationship to cell proliferation, apoptosis, angiogenesis, clinicopathologic characteristics, and patient survival in pancreatic cancer. Int J Pancreatol. 2001;29:133–140. doi: 10.1385/IJGC:29:3:133. [DOI] [PubMed] [Google Scholar]
- 268.Kong G, Kim EK, Kim WS, Lee KT, Lee YW, Lee JK, Paik SW, Rhee JC. Role of cyclooxygenase-2 and inducible nitric oxide synthase in pancreatic cancer. J Gastroenterol Hepatol. 2002;17:914–921. doi: 10.1046/j.1440-1746.2002.02829.x. [DOI] [PubMed] [Google Scholar]
- 269.Wang J, He P, Gaida M, Yang S, Schetter AJ, Gaedcke J, Ghadimi BM, Ried T, Yfantis H, Lee D, et al. Inducible nitric oxide synthase enhances disease aggressiveness in pancreatic cancer. Oncotarget. 2016;7:52993–53004. doi: 10.18632/oncotarget.10323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Vickers SM, MacMillan-Crow LA, Green M, Ellis C, Thompson JA. Association of increased immunostaining for inducible nitric oxide synthase and nitrotyrosine with fibroblast growth factor transformation in pancreatic cancer. Arch Surg. 1999;134:245–251. doi: 10.1001/archsurg.134.3.245. [DOI] [PubMed] [Google Scholar]
- 271.Takahashi M, Kitahashi T, Ishigamori R, Mutoh M, Komiya M, Sato H, Kamanaka Y, Naka M, Maruyama T, Sugimura T, Wakabayashi K. Increased expression of inducible nitric oxide synthase (iNOS) in N-nitrosobis(2-oxopropyl)amine-induced hamster pancreatic carcinogenesis and prevention of cancer development by ONO-1714, an iNOS inhibitor. Carcinogenesis. 2008;29:1608–1613. doi: 10.1093/carcin/bgn152. [DOI] [PubMed] [Google Scholar]
- 272.Chen HH, Su WC, Chou CY, Guo HR, Ho SY, Que J, Lee WY. Increased expression of nitric oxide synthase and cyclooxygenase-2 is associated with poor survival in cervical cancer treated with radiotherapy. Int J Radiat Oncol Biol Phys. 2005;63:1093–1100. doi: 10.1016/j.ijrobp.2005.03.062. [DOI] [PubMed] [Google Scholar]
- 273.Dong J, Cheng M, Sun H. Function of inducible nitric oxide synthase in the regulation of cervical cancer cell proliferation and the expression of vascular endothelial growth factor. Mol Med Rep. 2014;9:583–589. doi: 10.3892/mmr.2013.1838. [DOI] [PubMed] [Google Scholar]
- 274.Jiang N, Tian Z, Tang J, Ou R, Xu Y. Granulocyte macrophage-colony stimulating factor (GM-CSF) downregulates the expression of protumor factors cyclooxygenase-2 and inducible nitric oxide synthase in a GM-CSF receptor-independent manner in cervical cancer cells. Mediators Inflamm. 2015;2015:601604. doi: 10.1155/2015/601604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Lee TS, Jeon YT, Kim JW, Park NH, Kang SB, Lee HP, Song YS. Lack of association of the cyclooxygenase-2 and inducible nitric oxide synthase gene polymorphism with risk of cervical cancer in Korean population. Ann N Y Acad Sci. 2007;1095:134–142. doi: 10.1196/annals.1397.017. [DOI] [PubMed] [Google Scholar]
- 276.Ryu YK, Lee JW, Moon EY. Thymosin beta-4, actin-sequestering protein regulates vascular endothelial growth factor expression via hypoxia-inducible nitric oxide production in HeLa cervical cancer cells. Biomol Ther (Seoul) 2015;23:19–25. doi: 10.4062/biomolther.2014.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Sowjanya AP, Rao M, Vedantham H, Kalpana B, Poli UR, Marks MA, Sujatha M. Correlation of plasma nitrite/nitrate levels and inducible nitric oxide gene expression among women with cervical abnormalities and cancer. Nitric Oxide. 2016;52:21–28. doi: 10.1016/j.niox.2015.09.005. [DOI] [PubMed] [Google Scholar]
- 278.Li L, Zhu L, Hao B, Gao W, Wang Q, Li K, Wang M, Huang M, Liu Z, Yang Q, et al. iNOS-derived nitric oxide promotes glycolysis by inducing pyruvate kinase M2 nuclear translocation in ovarian cancer. Oncotarget. 2017;8:33047–33063. doi: 10.18632/oncotarget.16523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Malone JM, Saed GM, Diamond MP, Sokol RJ, Munkarah AR. The effects of the inhibition of inducible nitric oxide synthase on angiogenesis of epithelial ovarian cancer. Am J Obstet Gynecol. 2006;194:1110–1118. doi: 10.1016/j.ajog.2005.12.019. [DOI] [PubMed] [Google Scholar]
- 280.Martins Filho A, Jammal MP, Cobo Ede C, Silveira TP, Adad SJ, Murta EF, Nomelini RS. Correlation of cytokines and inducible nitric oxide synthase expression with prognostic factors in ovarian cancer. Immunol Lett. 2014;158:195–199. doi: 10.1016/j.imlet.2014.01.005. [DOI] [PubMed] [Google Scholar]
- 281.Raspollini MR, Amunni G, Villanucci A, Boddi V, Baroni G, Taddei A, Taddei GL. Expression of inducible nitric oxide synthase and cyclooxygenase-2 in ovarian cancer: Correlation with clinical outcome. Gynecol Oncol. 2004;92:806–812. doi: 10.1016/j.ygyno.2003.12.023. [DOI] [PubMed] [Google Scholar]
- 282.Zhao L, Yu C, Zhou S, Lau WB, Lau B, Luo Z, Lin Q, Yang H, Xuan Y, Yi T, et al. Epigenetic repression of PDZ-LIM domain-containing protein 2 promotes ovarian cancer via NOS2-derived nitric oxide signaling. Oncotarget. 2016;7:1408–1420. doi: 10.18632/oncotarget.6368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Xu L, Xie K, Fidler IJ. Therapy of human ovarian cancer by transfection with the murine interferon beta gene: Role of macrophage-inducible nitric oxide synthase. Hum Gene Ther. 1998;9:2699–2708. doi: 10.1089/hum.1998.9.18-2699. [DOI] [PubMed] [Google Scholar]
- 284.Trinh B, Ko SY, Haria D, Barengo N, Naora H. The homeoprotein DLX4 controls inducible nitric oxide synthase-mediated angiogenesis in ovarian cancer. Mol Cancer. 2015;14:97. doi: 10.1186/s12943-015-0368-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Sanhueza C, Araos J, Naranjo L, Barros E, Toledo L, Subiabre M, Toledo F, Gutiérrez J, Chiarello DI, Pardo F, et al. Are NHE1 and inducible nitric oxide synthase involved in human ovarian cancer? Pharmacol Res. 2016;105:183–185. doi: 10.1016/j.phrs.2016.01.007. [DOI] [PubMed] [Google Scholar]
- 286.Jayasurya A, Dheen ST, Yap WM, Tan NG, Ng YK, Bay BH. Inducible nitric oxide synthase and bcl-2 expression in nasopharyngeal cancer: Correlation with outcome of patients after radiotherapy. Int J Radiat Oncol Biol Phys. 2003;56:837–845. doi: 10.1016/S0360-3016(03)00122-6. [DOI] [PubMed] [Google Scholar]
- 287.Lukes P, Pacova H, Kucera T, Vesely D, Martinek J, Astl J. Expression of endothelial and inducible nitric oxide synthase and caspase-3 in tonsillar cancer, chronic tonsillitis and healthy tonsils. Folia Biol (Praha) 2008;54:141–145. doi: 10.14712/fb2008054050141. [DOI] [PubMed] [Google Scholar]
- 288.Soler MN, Bobe P, Benihoud K, Lemaire G, Roos BA, Lausson S. Gene therapy of rat medullary thyroid cancer by naked nitric oxide synthase II DNA injection. J Gene Med. 2000;2:344–352. doi: 10.1002/1521-2254(200009/10)2:5<344::AID-JGM124>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- 289.Rafa H, Benkhelifa S, AitYounes S, Saoula H, Belhadef S, Belkhelfa M, Boukercha A, Toumi R, Soufli I, Moralès O, et al. All-trans retinoic acid modulates TLR4/NF-κB signaling pathway targeting TNF-α and nitric oxide synthase 2 expression in colonic mucosa during ulcerative colitis and colitis associated cancer. Mediators Inflamm. 2017;2017:7353252. doi: 10.1155/2017/7353252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Amasyali AS, Kucukgergin C, Erdem S, Sanli O, Seckin S, Nane I. Nitric oxide synthase (eNOS4a/b) gene polymorphism is associated with tumor recurrence and progression in superficial bladder cancer cases. J Urol. 2012;188:2398–2403. doi: 10.1016/j.juro.2012.07.096. [DOI] [PubMed] [Google Scholar]
- 291.Polat F, Diler SB, Azazi İ, Öden A. T-786C, G894T, and intron 4 VNTR (4a/b) polymorphisms of the endothelial nitric oxide synthase gene in bladder cancer cases. Asian Pac J Cancer Prev. 2015;16:2199–2202. doi: 10.7314/APJCP.2015.16.6.2199. [DOI] [PubMed] [Google Scholar]
- 292.Ryk C, Wiklund NP, Nyberg T, de Verdier PJ. Polymorphisms in nitric-oxide synthase 3 may influence the risk of urinary-bladder cancer. Nitric Oxide. 2011;25:338–343. doi: 10.1016/j.niox.2011.06.003. [DOI] [PubMed] [Google Scholar]
- 293.Abedinzadeh M, Dastgheib SA, Maleki H, Heiranizadeh N, Zare M, Jafari-Nedooshan J, Kargar S, Neamatzadeh H. Association of endothelial nitric oxide synthase gene polymorphisms with susceptibility to prostate cancer: A comprehensive systematic review and meta-analysis. Urol J. 2020;17:329–337. doi: 10.22037/uj.v0i0.5445. [DOI] [PubMed] [Google Scholar]
- 294.Branković A, Brajušković G, Nikolić Z, Vukotić V, Cerović S, Savić-Pavićević D, Romac S. Endothelial nitric oxide synthase gene polymorphisms and prostate cancer risk in Serbian population. Int J Exp Pathol. 2013;94:355–361. doi: 10.1111/iep.12045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Diler SB, Oden A. The T-786C, G894T, and intron 4 VNTR (4a/b) polymorphisms of the endothelial nitric oxide synthase gene in prostate cancer cases. Genetika. 2016;52:249–254. doi: 10.7868/s0016675816020028. [DOI] [PubMed] [Google Scholar]
- 296.Fu Q, Liu X, Liu Y, Yang J, Lv G, Dong S. MicroRNA-335 and −543 suppress bone metastasis in prostate cancer via targeting endothelial nitric oxide synthase. Int J Mol Med. 2015;36:1417–1425. doi: 10.3892/ijmm.2015.2355. [DOI] [PubMed] [Google Scholar]
- 297.Medeiros R, Morais A, Vasconcelos A, Costa S, Carrilho S, Oliveira J, Lopes C. Endothelial nitric oxide synthase gene polymorphisms and the shedding of circulating tumour cells in the blood of prostate cancer patients. Cancer Lett. 2003;189:85–90. doi: 10.1016/S0304-3835(02)00118-0. [DOI] [PubMed] [Google Scholar]
- 298.Medeiros R, Morais A, Vasconcelos A, Costa S, Pinto D, Oliveira J, Lopes C. Endothelial nitric oxide synthase gene polymorphisms and genetic susceptibility to prostate cancer. Eur J Cancer Prev. 2002;11:343–350. doi: 10.1097/00008469-200208000-00005. [DOI] [PubMed] [Google Scholar]
- 299.Medeiros RM, Morais A, Vasconcelos A, Costa S, Pinto D, Oliveira J, Ferreira P, Lopes C. Outcome in prostate cancer: Association with endothelial nitric oxide synthase Glu-Asp298 polymorphism at exon 7. Clin Cancer Res. 2002;8:3433–3437. [PubMed] [Google Scholar]
- 300.Nikolic ZZ, Pavicevic D, Romac SP, Brajuskovic GN. Genetic variants within endothelial nitric oxide synthase gene and prostate cancer: A meta-analysis. Clin Transl Sci. 2015;8:23–31. doi: 10.1111/cts.12203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Marangoni K, Neves AF, Cardoso AM, Santos WK, Faria PC, Goulart LR. The endothelial nitric oxide synthase Glu-298-Asp polymorphism and its mRNA expression in the peripheral blood of patients with prostate cancer and benign prostatic hyperplasia. Cancer Detect Prev. 2006;30:7–13. doi: 10.1016/j.cdp.2005.09.004. [DOI] [PubMed] [Google Scholar]
- 302.Polytarchou C, Hatziapostolou M, Poimenidi E, Mikelis C, Papadopoulou A, Parthymou A, Papadimitriou E. Nitric oxide stimulates migration of human endothelial and prostate cancer cells through up-regulation of pleiotrophin expression and its receptor protein tyrosine phosphatase beta/zeta. Int J Cancer. 2009;124:1785–1793. doi: 10.1002/ijc.24084. [DOI] [PubMed] [Google Scholar]
- 303.Re A, Aiello A, Nanni S, Grasselli A, Benvenuti V, Pantisano V, Strigari L, Colussi C, Ciccone S, Mazzetti AP, et al. Silencing of GSTP1, a prostate cancer prognostic gene, by the estrogen receptor-β and endothelial nitric oxide synthase complex. Mol Endocrinol. 2011;25:2003–2016. doi: 10.1210/me.2011-1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Re A, Colussi C, Nanni S, Aiello A, Bacci L, Grassi C, Pontecorvi A, Farsetti A. Nucleoporin 153 regulates estrogen-dependent nuclear translocation of endothelial nitric oxide synthase and estrogen receptor beta in prostate cancer. Oncotarget. 2018;9:27985–27997. doi: 10.18632/oncotarget.25462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Ziaei SA, Samzadeh M, Jamaldini SH, Afshari M, Haghdoost AA, Hasanzad M. Endothelial nitric oxide synthase Glu298Asp polymorphism as a risk factor for prostate cancer. Int J Biol Markers. 2013;28:43–48. doi: 10.5301/JBM.2012.9585. [DOI] [PubMed] [Google Scholar]
- 306.Zhao C, Yan W, Zu X, Chen M, Liu L, Zhao S, Liu H, Hu X, Luo R, Xia Y, Qi L. Association between endothelial nitric oxide synthase 894G>T polymorphism and prostate cancer risk: A meta-analysis of literature studies. Tumour Biol. 2014;35:11727–11733. doi: 10.1007/s13277-014-2097-4. [DOI] [PubMed] [Google Scholar]
- 307.Yu S, Jia L, Zhang Y, Wu D, Xu Z, Ng CF, To KK, Huang Y, Chan FL. Increased expression of activated endothelial nitric oxide synthase contributes to antiandrogen resistance in prostate cancer cells by suppressing androgen receptor transactivation. Cancer Lett. 2013;328:83–94. doi: 10.1016/j.canlet.2012.09.006. [DOI] [PubMed] [Google Scholar]
- 308.Wu JH, Yang K, Ma HS, Xu Y. Association of endothelia nitric oxide synthase gene rs1799983 polymorphism with susceptibility to prostate cancer: A meta-analysis. Tumour Biol. 2014;35:7057–7062. doi: 10.1007/s13277-014-1870-8. [DOI] [PubMed] [Google Scholar]
- 309.Safarinejad MR, Safarinejad S, Shafiei N, Safarinejad S. Effects of the T-786C, G894T, and Intron 4 VNTR (4a/b) polymorphisms of the endothelial nitric oxide synthase gene on the risk of prostate cancer. Urol Oncol. 2013;31:1132–1140. doi: 10.1016/j.urolonc.2012.01.002. [DOI] [PubMed] [Google Scholar]
- 310.Sanli O, Kucukgergin C, Gokpinar M, Tefik T, Nane I, Seckin S. Despite the lack of association between different genotypes and the presence of prostate cancer, endothelial nitric oxide synthase a/b (eNOS4a/b) polymorphism may be associated with advanced clinical stage and bone metastasis. Urol Oncol. 2011;29:183–188. doi: 10.1016/j.urolonc.2011.04.006. [DOI] [PubMed] [Google Scholar]
- 311.Bayraktutan Z, Kiziltunc A, Bakan E, Alp HH. Determination of endothelial nitric oxide synthase gene polymorphism and plasma asymmetric dimethyl arginine concentrations in patients with lung cancer. Eurasian J Med. 2020;52:185–190. doi: 10.5152/eurasianjmed.2020.19220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Cheon KT, Choi KH, Lee HB, Park SK, Rhee YK, Lee YC. Gene polymorphisms of endothelial nitric oxide synthase and angiotensin-converting enzyme in patients with lung cancer. Lung. 2000;178:351–360. doi: 10.1007/s004080000039. [DOI] [PubMed] [Google Scholar]
- 313.Fujita S, Masago K, Hatachi Y, Fukuhara A, Hata A, Kaji R, Kim YH, Mio T, Mishima M, Katakami N. Genetic polymorphisms in the endothelial nitric oxide synthase gene correlate with overall survival in advanced non-small-cell lung cancer patients treated with platinum-based doublet chemotherapy. BMC Med Genet. 2010;11:167. doi: 10.1186/1471-2350-11-167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Kocer C, Benlier N, Balci SO, Pehlivan S, Sanli M, Nacak M. The role of endothelial nitric oxide synthase gene polymorphisms in patients with lung cancer. Clin Respir J. 2020;14:948–955. doi: 10.1111/crj.13228. [DOI] [PubMed] [Google Scholar]
- 315.Peddireddy V, Badabagni SP, Gundimeda SD, Mundluru HP. Association of eNOS and ACE gene polymorphisms and plasma nitric oxide with risk of non-small cell lung cancer in South India. Clin Respir J. 2018;12:207–217. doi: 10.1111/crj.12517. [DOI] [PubMed] [Google Scholar]
- 316.Chen CH, Wu SH, Tseng YM, Hou MF, Tsai LY, Tsai SM. Distinct role of endothelial nitric oxide synthase gene polymorphisms from menopausal status in the patients with sporadic breast cancer in Taiwan. Nitric Oxide. 2018;72:1–6. doi: 10.1016/j.niox.2017.10.009. [DOI] [PubMed] [Google Scholar]
- 317.Crucitta S, Restante G, Del Re M, Bertolini I, Bona E, Rofi E, Fontanelli L, Gianfilippo G, Fogli S, Stasi I, et al. Endothelial nitric oxide synthase c.-813C>T predicts for proteinuria in metastatic breast cancer patients treated with bevacizumab-based chemotherapy. Cancer Chemother Pharmacol. 2019;84:1219–1227. doi: 10.1007/s00280-019-03933-z. [DOI] [PubMed] [Google Scholar]
- 318.Basaran G, Turhal NS, Cabuk D, Yurt N, Yurtseven G, Gumus M, Teomete M, Dane F, Yumuk PF. Weight gain after adjuvant chemotherapy in patients with early breast cancer in Istanbul Turkey. Med Oncol. 2011;28:409–415. doi: 10.1007/s12032-010-9463-x. [DOI] [PubMed] [Google Scholar]
- 319.Hao Y, Montiel R, Huang Y. Endothelial nitric oxide synthase (eNOS) 894 G>T polymorphism is associated with breast cancer risk: A meta-analysis. Breast Cancer Res Treat. 2010;124:809–813. doi: 10.1007/s10549-010-0833-z. [DOI] [PubMed] [Google Scholar]
- 320.Hefler LA, Grimm C, Lantzsch T, Lampe D, Koelbl H, Lebrecht A, Heinze G, Tempfer C, Reinthaller A, Zeillinger R. Polymorphisms of the endothelial nitric oxide synthase gene in breast cancer. Breast Cancer Res Treat. 2006;98:151–155. doi: 10.1007/s10549-005-9143-2. [DOI] [PubMed] [Google Scholar]
- 321.Kafousi M, Vrekoussis T, Tsentelierou E, Pavlakis K, Navrozoglou I, Dousias V, Sanidas E, Tsiftsis D, Georgoulias V, Stathopoulos EN. Immunohistochemical study of the angiogenetic network of VEGF, HIF1α, VEGFR-2 and endothelial nitric oxide synthase (eNOS) in human breast cancer. Pathol Oncol Res. 2012;18:33–41. doi: 10.1007/s12253-011-9413-8. [DOI] [PubMed] [Google Scholar]
- 322.Loibl S, Bratengeier J, Farines V, von Minckwitz G, Spänkuch B, Schini-Kerth V, Nepveu F, Strebhardt K, Kaufmann M. Investigations on the inducible and endothelial nitric oxide synthases in human breast cancer cell line MCF-7-estrogen has an influence on e-NOS, but not on i-NOS. Pathol Res Pract. 2006;202:1–7. doi: 10.1016/j.prp.2005.10.003. [DOI] [PubMed] [Google Scholar]
- 323.Loibl S, Strank C, von Minckwitz G, Sinn HP, Buck A, Solbach C, Strebhardt K, Kaufmann M. Immunohistochemical evaluation of endothelial nitric oxide synthase expression in primary breast cancer. Breast. 2005;14:230–235. doi: 10.1016/j.breast.2004.11.005. [DOI] [PubMed] [Google Scholar]
- 324.Lu J, Wei Q, Bondy ML, Yu TK, Li D, Brewster A, Shete S, Sahin A, Meric-Bernstam F, Wang LE. Promoter polymorphism (−786t>C) in the endothelial nitric oxide synthase gene is associated with risk of sporadic breast cancer in non-Hispanic white women age younger than 55 years. Cancer. 2006;107:2245–2253. doi: 10.1002/cncr.22269. [DOI] [PubMed] [Google Scholar]
- 325.Mortensen K, Holck S, Christensen IJ, Skouv J, Hougaard DM, Blom J, Larsson LI. Endothelial cell nitric oxide synthase in peritumoral microvessels is a favorable prognostic indicator in premenopausal breast cancer patients. Clin Cancer Res. 1999;5:1093–1097. [PubMed] [Google Scholar]
- 326.Mortensen K, Skouv J, Hougaard DM, Larsson LI. Endogenous endothelial cell nitric-oxide synthase modulates apoptosis in cultured breast cancer cells and is transcriptionally regulated by p53. J Biol Chemistry. 1999;274:37679–37684. doi: 10.1074/jbc.274.53.37679. [DOI] [PubMed] [Google Scholar]
- 327.Martin JH, Begum S, Alalami O, Harrison A, Scott KW. Endothelial nitric oxide synthase: Correlation with histologic grade, lymph node status and estrogen receptor expression in human breast cancer. Tumour Biol. 2000;21:90–97. doi: 10.1159/000030114. [DOI] [PubMed] [Google Scholar]
- 328.Ramírez-Patiño R, Figuera LE, Puebla-Pérez AM, Delgado-Saucedo JI, Legazpí-Macias MM, Mariaud-Schmidt RP, Ramos-Silva A, Gutiérrez-Hurtado IA, Gómez Flores-Ramos L, Zúñiga-González GM, Gallegos-Arreola MP. Intron 4 VNTR (4a/b) polymorphism of the endothelial nitric oxide synthase gene is associated with breast cancer in Mexican women. J Korean Med Sci. 2013;28:1587–1594. doi: 10.3346/jkms.2013.28.11.1587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 329.Zintzaras E, Grammatikou M, Kitsios GD, Doxani C, Zdoukopoulos N, Papandreou C, Patrikidou A. Polymorphisms of the endothelial nitric oxide synthase gene in breast cancer: A genetic association study and meta-analysis. J Hum Genet. 2010;55:743–748. doi: 10.1038/jhg.2010.136. [DOI] [PubMed] [Google Scholar]
- 330.Zhang L, Zeng M, Fu BM. Inhibition of endothelial nitric oxide synthase decreases breast cancer cell MDA-MB-231 adhesion to intact microvessels under physiological flows. Am J Physiol Heart Circ Physiol. 2016;310:H1735–H1747. doi: 10.1152/ajpheart.00109.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Carkic J, Nikolic N, Nisevic J, Lazarevic M, Kuzmanovic-Pficer J, Jelovac D, Milasin J. Endothelial nitric oxide synthase polymorphisms/haplotypes are strong modulators of oral cancer risk in Serbian population. J Oral Sci. 2020;62:322–326. doi: 10.2334/josnusd.19-0310. [DOI] [PubMed] [Google Scholar]
- 332.Karthik B, Shruthi DK, Singh J, Tegginamani AS, Kudva S. Do tobacco stimulate the production of nitric oxide by up regulation of inducible nitric oxide synthesis in cancer: Immunohistochemical determination of inducible nitric oxide synthesis in oral squamous cell carcinoma-a comparative study in tobacco habituers and non-habituers. J Cancer Res Ther. 2014;10:244–250. doi: 10.4103/0973-1482.136542. [DOI] [PubMed] [Google Scholar]
- 333.Chen Y, Li J, Guo Y, Guo XY. Nitric oxide synthase 3 gene variants and colorectal cancer: A meta-analysis. Asian Pac J Cancer Prev. 2014;15:3811–3815. doi: 10.7314/APJCP.2014.15.8.3811. [DOI] [PubMed] [Google Scholar]
- 334.Mortensen K, Christensen IJ, Nielsen HJ, Hansen U, Larsson LI. High expression of endothelial cell nitric oxide synthase in peritumoral microvessels predicts increased disease-free survival in colorectal cancer. Cancer Lett. 2004;216:109–114. doi: 10.1016/j.canlet.2004.05.025. [DOI] [PubMed] [Google Scholar]
- 335.Peñarando J, López-Sánchez LM, Mena R, Guil-Luna S, Conde F, Hernández V, Toledano M, Gudiño V, Raponi M, Billard C, et al. A role for endothelial nitric oxide synthase in intestinal stem cell proliferation and mesenchymal colorectal cancer. BMC Biol. 2018;16:3. doi: 10.1186/s12915-017-0472-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 336.Yeh CC, Santella RM, Hsieh LL, Sung FC, Tang R. An intron 4 VNTR polymorphism of the endothelial nitric oxide synthase gene is associated with early-onset colorectal cancer. Int J Cancer. 2009;124:1565–1571. doi: 10.1002/ijc.24114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Hefler LA, Ludwig E, Lampe D, Zeillinger R, Leodolter S, Gitsch G, Koelbl H, Tempfer CB. Polymorphisms of the endothelial nitric oxide synthase gene in ovarian cancer. Gynecol Oncol. 2002;86:134–137. doi: 10.1006/gyno.2002.6749. [DOI] [PubMed] [Google Scholar]
- 338.Krishnaveni D, Amar Chand B, Shravan Kumar P, Uma Devi M, Ramanna M, Jyothy A, Pratibha N, Balakrishna N, Venkateshwari A. Association of endothelial nitric oxide synthase gene T-786C promoter polymorphism with gastric cancer. World J Gastrointest Oncol. 2015;7:87–94. doi: 10.4251/wjgo.v7.i7.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Wang L, Shi GG, Yao JC, Gong W, Wei D, Wu TT, Ajani JA, Huang S, Xie K. Expression of endothelial nitric oxide synthase correlates with the angiogenic phenotype of and predicts poor prognosis in human gastric cancer. Gastric Cancer. 2005;8:18–28. doi: 10.1007/s10120-004-0310-7. [DOI] [PubMed] [Google Scholar]
- 340.Tecder Ünal M, Karabulut HG, Gümüş-Akay G, Dölen Y, Elhan A, Tükün A, Ünal AE. Endothelial nitric oxide synthase gene polymorphism in gastric cancer. Turk J Gastroenterol. 2010;21:338–344. doi: 10.4318/tjg.2010.0118. [DOI] [PubMed] [Google Scholar]
- 341.de Fatima A, Zambuzzi WF, Modolo LV, Tarsitano CA, Gadelha FR, Hyslop S, de Carvalho JE, Salgado I, Ferreira CV, Pilli RA. Cytotoxicity of goniothalamin enantiomers in renal cancer cells: Involvement of nitric oxide, apoptosis and autophagy. Chem Biol Interact. 2008;176:143–150. doi: 10.1016/j.cbi.2008.08.003. [DOI] [PubMed] [Google Scholar]
- 342.Hung WC, Wu TF, Ng SC, Lee YC, Shen HP, Yang SF, Wang PH. Involvement of endothelial nitric oxide synthase gene variants in the aggressiveness of uterine cervical cancer. J Cancer. 2019;10:2594–2600. doi: 10.7150/jca.33192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Waheed S, Cheng RY, Casablanca Y, Maxwell GL, Wink DA, Syed V. Nitric oxide donor DETA/NO inhibits the growth of endometrial cancer cells by upregulating the expression of RASSF1 and CDKN1A. Molecules. 2019;24:3722. doi: 10.3390/molecules24203722. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 344.Riener EK, Hefler LA, Grimm C, Galid A, Zeillinger R, Tong-Cacsire D, Gitsch G, Leodolter S, Tempfer CB. Polymorphisms of the endothelial nitric oxide synthase gene in women with vulvar cancer. Gynecol Oncol. 2004;93:686–690. doi: 10.1016/j.ygyno.2004.03.030. [DOI] [PubMed] [Google Scholar]
- 345.Wang J, Yang S, He P, Schetter AJ, Gaedcke J, Ghadimi BM, Ried T, Yfantis HG, Lee DH, Gaida MM, et al. Endothelial Nitric oxide synthase traffic inducer (NOSTRIN) is a negative regulator of disease aggressiveness in pancreatic cancer. Clin Cancer Res. 2016;22:5992–6001. doi: 10.1158/1078-0432.CCR-16-0511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Yanar K, Cakatay U, Aydin S, Verim A, Atukeren P, Özkan NE, Karatoprak K, Cebe T, Turan S, Ozkök E, et al. Relation between endothelial nitric oxide synthase genotypes and oxidative stress markers in larynx cancer. Oxid Med Cell Longev. 2016;2016:4985063. doi: 10.1155/2016/4985063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Chen C, Wang Y, Goh SS, Yang J, Lam DH, Choudhury Y, Tay FC, Du S, Tan WK, Purwanti YI, et al. Inhibition of neuronal nitric oxide synthase activity promotes migration of human-induced pluripotent stem cell-derived neural stem cells toward cancer cells. J Neurochem. 2013;126:318–330. doi: 10.1111/jnc.12199. [DOI] [PubMed] [Google Scholar]
- 348.Karihtala P, Soini Y, Auvinen P, Tammi R, Tammi M, Kosma VM. Hyaluronan in breast cancer: Correlations with nitric oxide synthases and tyrosine nitrosylation. J Histochem Cytochem. 2007;55:1191–1198. doi: 10.1369/jhc.7A7270.2007. [DOI] [PubMed] [Google Scholar]
- 349.Lewko B, Zółtowska A, Stepiński J, Kamiński M, Skokowski J, Roszkiewicz A, Moszkowska G. Nitric oxide synthase type 1 expression in human lung cancer and its relation to p53. Med Sci Monit. 2001;7:218–221. [PubMed] [Google Scholar]
- 350.Ahmed HH, Metwally FM, Mahdy ES, Shosha WG, Ramadan SS. Clinical value of serum hepatocyte growth factor, B-cell lymphoma-2 and nitric oxide in primary breast cancer patients. Eur Rev Med Pharmacol Sci. 2012;16:958–965. [PubMed] [Google Scholar]
- 351.Al Dhaheri Y, Attoub S, Arafat K, Abuqamar S, Viallet J, Saleh A, Al Agha H, Eid A, Iratni R. Anti-metastatic and anti-tumor growth effects of Origanum majorana on highly metastatic human breast cancer cells: Inhibition of NFκB signaling and reduction of nitric oxide production. PLoS One. 2013;8:e68808. doi: 10.1371/journal.pone.0068808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Avtandilyan N, Javrushyan H, Petrosyan G, Trchounian A. The involvement of arginase and nitric oxide synthase in breast cancer development: Arginase and NO Synthase as therapeutic targets in cancer. Biomed Res Int. 2018;2018:8696923. doi: 10.1155/2018/8696923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Bani D, Masini E, Bello MG, Bigazzi M, Sacchi TB. Relaxin activates the L-arginine-nitric oxide pathway in human breast cancer cells. Cancer Res. 1995;55:5272–5275. [PubMed] [Google Scholar]
- 354.Avtandilyan N, Javrushyan H, Karapetyan A, Trchounian A. RETRACTED ARTICLE: Inhibition of tumor progression by NG-Nitro-L-arginine Methyl Ester in 7,12-dimethylbenz(a)anthracene induced breast cancer: Nitric oxide synthase inhibition as an antitumor prevention. J Mammary Gland Biol Neoplasia. 2019;24:199. doi: 10.1007/s10911-019-09428-0. [DOI] [PubMed] [Google Scholar]
- 355.Alagol H, Erdem E, Sancak B, Turkmen G, Camlibel M, Bugdayci G. Nitric oxide biosynthesis and malondialdehyde levels in advanced breast cancer. Aust N Z J Surg. 1999;69:647–650. doi: 10.1046/j.1440-1622.1999.01656.x. [DOI] [PubMed] [Google Scholar]
- 356.Bhattacharjee KG, Bhattacharyya M, Halder UC, Jana P, Sinha AK. The ‘Cross Talk’ between the receptors of insulin, estrogen and progesterone in neutrophils in the synthesis of maspin through nitric oxide in breast cancer. Int J Biomed Sci. 2012;8:129–139. [PMC free article] [PubMed] [Google Scholar]
- 357.Cendan JC, Souba WW, Copeland EM, III, Lind DS. Increased L-arginine transport in a nitric oxide-producing metastatic colon cancer cell line. Ann Surg Oncol. 1996;3:501–508. doi: 10.1007/BF02305770. [DOI] [PubMed] [Google Scholar]
- 358.Cendan JC, Topping DL, Pruitt J, Snowdy S, Copeland EM, III, Lind DS. Inflammatory mediators stimulate arginine transport and arginine-derived nitric oxide production in a murine breast cancer cell line. J Surg Res. 1996;60:284–288. doi: 10.1006/jsre.1996.0044. [DOI] [PubMed] [Google Scholar]
- 359.Chakraborty S, Girish GV, Sinha AK. Impaired binding of insulin to erythrocyte membrane receptor and the activation of nitric oxide synthase by the hormone in human breast cancer. J Cancer Res Clin Oncol. 2004;130:294–300. doi: 10.1007/s00432-003-0532-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Coskun U, Gunel N, Sancak B, Günel U, Onuk E, Bayram O, Yílmaz E, Candan S, Ozkan S. Significance of serum vascular endothelial growth factor, insulin-like growth factor-I levels and nitric oxide activity in breast cancer patients. Breast. 2003;12:104–110. doi: 10.1016/S0960-9776(02)00214-X. [DOI] [PubMed] [Google Scholar]
- 361.Coskun U, Gunel N, Sancak B, Günel U, Onuk E, Bayram O, Yílmaz E, Candan S, Ozkan S. WITHDRAWN: Significance of serum vascular endothelial growth factor, insulin-like growth factor-I levels and nitric oxide activity in breast cancer patients. Breast. 2008 Aug 20; doi: 10.1054/brst.2002.0483. doi: 10.1054/brst.2002.0483 (Epub ahead of print) [DOI] [PubMed] [Google Scholar]
- 362.Dave B, Granados-Principal S, Zhu R, Benz S, Rabizadeh S, Soon-Shiong P, Yu KD, Shao Z, Li X, Gilcrease M, et al. Targeting RPL39 and MLF2 reduces tumor initiation and metastasis in breast cancer by inhibiting nitric oxide synthase signaling. Proc Natl Acad Sci USA. 2014;111:8838–8843. doi: 10.1073/pnas.1320769111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 363.De Vitto H, Mendonca BS, Elseth KM, Onul A, Xue J, Vesper BJ, Gallo CV, Rumjanek FD, Paradise WA, Radosevich JA. Part III. Molecular changes induced by high nitric oxide adaptation in human breast cancer cell line BT-20 (BT-20-HNO): A switch from aerobic to anaerobic metabolism. Tumour Biol. 2013;34:403–413. doi: 10.1007/s13277-012-0555-4. [DOI] [PubMed] [Google Scholar]
- 364.Deliconstantinos G, Villiotou V, Stavrides JC, Salemes N, Gogas J. Nitric oxide and peroxynitrite production by human erythrocytes: A causative factor of toxic anemia in breast cancer patients. Anticancer Res. 1995;15:1435–1446. [PubMed] [Google Scholar]
- 365.Demircan B, Yucel B, Radosevich JA. DNA methylation in human breast cancer cell lines adapted to high nitric oxide. In Vivo. 2020;34:169–176. doi: 10.21873/invivo.11758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Duan L, Danzer B, Levenson VV, Maki CG. Critical roles for nitric oxide and ERK in the completion of prosurvival autophagy in 4OHTAM-treated estrogen receptor-positive breast cancer cells. Cancer Lett. 2014;353:290–300. doi: 10.1016/j.canlet.2014.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 367.El Hasasna H, Saleh A, Al Samri H, Athamneh K, Attoub S, Arafat K, Benhalilou N, Alyan S, Viallet J, Al Dhaheri Y, et al. Rhus coriaria suppresses angiogenesis, metastasis and tumor growth of breast cancer through inhibition of STAT3, NFκB and nitric oxide pathways. Scic Rep. 2016;6:21144. doi: 10.1038/srep21144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 368.Engelen M, Klimberg VS, Allasia A, Deutz NEP. Major surgery diminishes systemic arginine availability and suppresses nitric oxide response to feeding in patients with early stage breast cancer. Clin Nutr. 2018;37:1645–1653. doi: 10.1016/j.clnu.2017.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 369.Erbas H, Aydogdu N, Usta U, Erten O. Protective role of carnitine in breast cancer via decreasing arginase activity and increasing nitric oxide. Cell Biol Int. 2007;31:1414–1419. doi: 10.1016/j.cellbi.2007.06.007. [DOI] [PubMed] [Google Scholar]
- 370.Gaballah HE, Abdel Salam I, Abdel Wahab N, Mansour OM. Plasma bcl-2 and nitric oxide: Possible prognostic role in patients with metastatic breast cancer. Med Oncol. 2001;18:171–178. doi: 10.1385/MO:18:3:171. [DOI] [PubMed] [Google Scholar]
- 371.Gajalakshmi P, Priya MK, Pradeep T, Behera J, Muthumani K, Madhuwanti S, Saran U, Chatterjee S. Breast cancer drugs dampen vascular functions by interfering with nitric oxide signaling in endothelium. Toxicol Appl Pharmacol. 2013;269:121–131. doi: 10.1016/j.taap.2013.03.011. [DOI] [PubMed] [Google Scholar]
- 372.Ganguly Bhattacharjee K, Bhattacharyya M, Halder UC, Jana P, Sinha AK. The role of neutrophil estrogen receptor status on maspin synthesis via nitric oxide production in human breast cancer. J Breast Cancer. 2012;15:181–188. doi: 10.4048/jbc.2012.15.2.181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 373.Ganguly Bhattacharjee K, Bhattacharyya M, Halder UC, Jana P, Sinha AK. Effect of progesterone receptor status on maspin synthesis via nitric oxide production in neutrophils in human breast cancer. Breast Cancer. 2014;21:605–613. doi: 10.1007/s12282-012-0422-6. [DOI] [PubMed] [Google Scholar]
- 374.Gaspari APS, da Silva RS, Carneiro ZA, de Carvalho MR, Carvalho I, Pernomian L, Ferreira LP, Ramos LCB, de Souza GA, Formiga ALB. Improving cytotoxicity against breast cancer cells by using Mixed-Ligand Ruthenium(II) Complexes of 2,2′-Bipyridine, amino acid, and nitric oxide derivatives as potential anticancer agents. Anticancer Agents Med Chem. 2021;21:1602–1611. doi: 10.2174/0929867327666201020155105. [DOI] [PubMed] [Google Scholar]
- 375.Guha M, Biswas J, Tirkey J, Sinha AK. Impairment of stimulation by estrogen of insulin-activated nitric oxide synthase in human breast cancer. Int J Cancer. 2002;100:261–265. doi: 10.1002/ijc.10482. [DOI] [PubMed] [Google Scholar]
- 376.Günel N, Coskun U, Sancak B, Hasdemir O, Sare M, Bayram O, Celenkoglu G, Ozkan S. Prognostic value of serum IL-18 and nitric oxide activity in breast cancer patients at operable stage. Am J Clin Oncol. 2003;26:416–421. doi: 10.1097/00000421-200308000-00023. [DOI] [PubMed] [Google Scholar]
- 377.Kampa M, Hatzoglou A, Notas G, Niniraki M, Kouroumalis E, Castanas E. Opioids are non-competitive inhibitors of nitric oxide synthase in T47D human breast cancer cells. Cell Death Differ. 2001;8:943–952. doi: 10.1038/sj.cdd.4400893. [DOI] [PubMed] [Google Scholar]
- 378.Khalkhali-Ellis Z, Hendrix MJ. Nitric oxide regulation of maspin expression in normal mammary epithelial and breast cancer cells. Am J Pathol. 2003;162:1411–1417. doi: 10.1016/S0002-9440(10)64274-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 379.Konukoglu D, Turhan MS, Celik V, Turna H. Relation of serum vascular endothelial growth factor as an angiogenesis biomarker with nitric oxide & urokinase-type plasminogen activator in breast cancer patients. Indian J Med Res. 2007;125:747–751. [PubMed] [Google Scholar]
- 380.Kumar S, Kashyap P. Antiproliferative activity and nitric oxide production of a methanolic extract of Fraxinus micrantha on Michigan Cancer Foundation-7 mammalian breast carcinoma cell line. J Intercult Ethnopharmacol. 2015;4:109–113. doi: 10.5455/jice.20150129102013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Lahiri M, Martin JH. Nitric oxide decreases motility and increases adhesion in human breast cancer cells. Oncol Rep. 2009;21:275–281. [PubMed] [Google Scholar]
- 382.Lee SY, Rim Y, McPherson DD, Huang SL, Kim H. A novel liposomal nanomedicine for nitric oxide delivery and breast cancer treatment. Biomed Mater Eng. 2014;24:61–67. doi: 10.3233/BME-130784. [DOI] [PubMed] [Google Scholar]
- 383.Hewala TI, Abd El-Moneim NA, Ebied SA, Sheta MI, Soliman K, Abu-Elenean A. Diagnostic and prognostic value of serum nitric oxide, tumor necrosis factor-alpha, basic fibroblast growth factor and copper as angiogenic markers in premenopausal breast cancer patients: A case-control study. Br J Biomed Sci. 2010;67:167–176. doi: 10.1080/09674845.2010.11730315. [DOI] [PubMed] [Google Scholar]
- 384.Finkelman BS, Putt M, Wang T, Wang L, Narayan H, Domchek S, DeMichele A, Fox K, Matro J, Shah P, et al. Arginine-nitric oxide metabolites and cardiac dysfunction in patients with breast cancer. J Am Coll Cardiol. 2017;70:152–162. doi: 10.1016/j.jacc.2017.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 385.Jadeski LC, Chakraborty C, Lala PK. Role of nitric oxide in tumour progression with special reference to a murine breast cancer model. Can J Physiol Pharmacol. 2002;80:125–135. doi: 10.1139/y02-007. [DOI] [PubMed] [Google Scholar]
- 386.McCurdy MR, Wazni MW, Martinez J, McAleer MF, Guerrero T. Exhaled nitric oxide predicts radiation pneumonitis in esophageal and lung cancer patients receiving thoracic radiation. Radiother Oncol. 2011;101:443–448. doi: 10.1016/j.radonc.2011.08.035. [DOI] [PubMed] [Google Scholar]
- 387.Metwally FM, El-mezayen HA, Ahmed HH. Significance of vascular endothelial growth factor, interleukin-18 and nitric oxide in patients with breast cancer: Correlation with carbohydrate antigen 15.3. Med Oncol. 2011;28((Suppl 1)):S15–S21. doi: 10.1007/s12032-010-9657-2. [DOI] [PubMed] [Google Scholar]
- 388.Mishra D, Patel V, Banerjee D. Nitric Oxide and S-Nitrosylation in Cancers: Emphasis on breast cancer. Breast Cancer (Auckl) 2020;14:1178223419882688. doi: 10.1177/1178223419882688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 389.Nakamura Y, Yasuoka H, Tsujimoto M, Yoshidome K, Nakahara M, Nakao K, Nakamura M, Kakudo K. Nitric oxide in breast cancer: Induction of vascular endothelial growth factor-C and correlation with metastasis and poor prognosis. Clin Cancer Res. 2006;12:1201–1207. doi: 10.1158/1078-0432.CCR-05-1269. [DOI] [PubMed] [Google Scholar]
- 390.Negroni MP, Fiszman GL, Azar ME, Morgado CC, Español AJ, Pelegrina LT, de la Torre E, Sales ME. Immunoglobulin G from breast cancer patients in stage I stimulates muscarinic acetylcholine receptors in MCF7 cells and induces proliferation. Participation of nitric oxide synthase-derived nitric oxide. J Clin Immunol. 2010;30:474–484. doi: 10.1007/s10875-010-9370-0. [DOI] [PubMed] [Google Scholar]
- 391.Martin JH, Alalami O, Yaqoob F. Differential effects of retinoids on nitric oxide production by promonocytic U937 cells and ZR-75-1 human breast cancer cells. Oncol Rep. 2000;7:219–223. [PubMed] [Google Scholar]
- 392.Martin JH, Symonds A, Chohan S. Down-regulation of nitric oxide production by droloxifene and toremifene in human breast cancer cells. Oncol Rep. 2003;10:979–984. doi: 10.3892/or.10.4.979. [DOI] [PubMed] [Google Scholar]
- 393.Nath N, Chattopadhyay M, Rodes DB, Nazarenko A, Kodela R, Kashfi K. Nitric Oxide-releasing aspirin suppresses NF-κB signaling in estrogen receptor negative breast cancer cells in vitro and in vivo. Molecules. 2015;20:12481–12499. doi: 10.3390/molecules200712481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 394.Pance A. Nitric oxide and hormones in breast cancer: Allies or enemies? Future Oncol. 2006;2:275–288. doi: 10.2217/14796694.2.2.275. [DOI] [PubMed] [Google Scholar]
- 395.Parihar A, Parihar MS, Ghafourifar P. Significance of mitochondrial calcium and nitric oxide for apoptosis of human breast cancer cells induced by tamoxifen and etoposide. Int J Mol Med. 2008;21:317–324. [PubMed] [Google Scholar]
- 396.Pervin S, Singh R, Chaudhuri G. Nitric oxide-induced cytostasis and cell cycle arrest of a human breast cancer cell line (MDA-MB-231): Potential role of cyclin D1. Proc Natl Acad Sci USA. 2001;98:3583–3588. doi: 10.1073/pnas.041603998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 397.Pervin S, Singh R, Chaudhuri G. Nitric oxide, N omega-hydroxy-L-arginine and breast cancer. Nitric Oxide. 2008;19:103–106. doi: 10.1016/j.niox.2008.04.016. [DOI] [PubMed] [Google Scholar]
- 398.Pervin S, Singh R, Freije WA, Chaudhuri G. MKP-1-induced dephosphorylation of extracellular signal-regulated kinase is essential for triggering nitric oxide-induced apoptosis in human breast cancer cell lines: Implications in breast cancer. Cancer Res. 2003;63:8853–8860. [PubMed] [Google Scholar]
- 399.Pervin S, Singh R, Gau CL, Edamatsu H, Tamanoi F, Chaudhuri G. Potentiation of nitric oxide-induced apoptosis of MDA-MB-468 cells by farnesyltransferase inhibitor: Implications in breast cancer. Cancer Res. 2001;61:4701–4706. [PubMed] [Google Scholar]
- 400.Pervin S, Singh R, Hernandez E, Wu G, Chaudhuri G. Nitric oxide in physiologic concentrations targets the translational machinery to increase the proliferation of human breast cancer cells: Involvement of mammalian target of rapamycin/eIF4E pathway. Cancer Res. 2007;67:289–299. doi: 10.1158/0008-5472.CAN-05-4623. [DOI] [PubMed] [Google Scholar]
- 401.Punathil T, Tollefsbol TO, Katiyar SK. EGCG inhibits mammary cancer cell migration through inhibition of nitric oxide synthase and guanylate cyclase. Biochem Biophys Res Commun. 2008;375:162–167. doi: 10.1016/j.bbrc.2008.07.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 402.Radisavljevic Z. Inactivated tumor suppressor Rb by nitric oxide promotes mitosis in human breast cancer cells. J Cell Biochem. 2004;92:1–5. doi: 10.1002/jcb.20063. [DOI] [PubMed] [Google Scholar]
- 403.Rashad YA, Elkhodary TR, El-Gayar AM, Eissa LA. Evaluation of serum levels of HER2, MMP-9, Nitric Oxide, and total antioxidant capacity in egyptian breast cancer patients: Correlation with clinico-pathological parameters. Sci Pharm. 2014;82:129–145. doi: 10.3797/scipharm.1306-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 404.Ray GN, Shahid M, Husain SA. Effect of nitric oxide and malondialdehyde on sister-chromatid exchanges in breast cancer. Br J Biomed Sci. 2001;58:169–176. [PubMed] [Google Scholar]
- 405.Reveneau S, Arnould L, Jolimoy G, Hilpert S, Lejeune P, Saint-Giorgio V, Belichard C, Jeannin JF. Nitric oxide synthase in human breast cancer is associated with tumor grade, proliferation rate, and expression of progesterone receptors. Lab Invest. 1999;79:1215–1225. [PubMed] [Google Scholar]
- 406.Ridnour LA, Barasch KM, Windhausen AN, Dorsey TH, Lizardo MM, Yfantis HG, Lee DH, Switzer CH, Cheng RY, Heinecke JL, et al. Nitric oxide synthase and breast cancer: Role of TIMP-1 in NO-mediated Akt activation. PLoS One. 2012;7:e44081. doi: 10.1371/journal.pone.0044081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 407.Zhu W, Yang B, Fu H, Ma L, Liu T, Chai R, Zheng Z, Zhang Q, Li G. Flavone inhibits nitric oxide synthase (NOS) activity, nitric oxide production and protein S-nitrosylation in breast cancer cells. Biochem Biophys Res Commun. 2015;458:590–595. doi: 10.1016/j.bbrc.2015.01.154. [DOI] [PubMed] [Google Scholar]
- 408.Zheng X, Fernando V, Sharma V, Walia Y, Letson J, Furuta S. Correction of arginine metabolism with sepiapterin-the precursor of nitric oxide synthase cofactor BH(4)-induces immunostimulatory-shift of breast cancer. Biochem Pharmacol. 2020;176:113887. doi: 10.1016/j.bcp.2020.113887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 409.Zeillinger R, Tantscher E, Schneeberger C, Tschugguel W, Eder S, Sliutz G, Huber JC. Simultaneous expression of nitric oxide synthase and estrogen receptor in human breast cancer cell lines. Breast Cancer Res Treat. 1996;40:205–207. doi: 10.1007/BF01806216. [DOI] [PubMed] [Google Scholar]
- 410.Wang HJ, Wei XF, Jiang YY, Huang H, Yang Y, Fan SM, Zang LH, Tashiro S, Onodera S, Ikejima T. Silibinin induces the generation of nitric oxide in human breast cancer MCF-7 cells. Free Radic Res. 2010;44:577–584. doi: 10.3109/10715760903300717. [DOI] [PubMed] [Google Scholar]
- 411.Thamrongwittawatpong L, Sirivatanauksorn Y, Batten JJ, Williamson RC, Kakkar AK, Mathie RT. The effect of N(G)-monomethyl-L-arginine and tamoxifen on nitric oxide production in breast cancer cells stimulated by oestrogen and progesterone. Eur J Surg. 2001;167:484–489. doi: 10.1080/110241501316914830. [DOI] [PubMed] [Google Scholar]
- 412.Toomey D, Condron C, Wu QD, Kay E, Harmey J, Broe P, Kelly C, Bouchier-Hayes D. TGF-beta1 is elevated in breast cancer tissue and regulates nitric oxide production from a number of cellular sources during hypoxia re-oxygenation injury. Br J Biomed Sci. 2001;58:177–183. [PubMed] [Google Scholar]
- 413.Salaroglio IC, Gazzano E, Abdullrahman A, Mungo E, Castella B, Abd-Elrahman GEFA, Massaia M, Donadelli M, Rubinstein M, Riganti C, Kopecka J. Increasing intratumor C/EBP-β LIP and nitric oxide levels overcome resistance to doxorubicin in triple negative breast cancer. J Exp Clin Cancer Res. 2018;37:286. doi: 10.1186/s13046-018-0967-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 414.Sen S, Kawahara B, Chaudhuri G. Mitochondrial-associated nitric oxide synthase activity inhibits cytochrome c oxidase: Implications for breast cancer. Free Radic Biol Med. 2013;57:210–220. doi: 10.1016/j.freeradbiomed.2012.10.545. [DOI] [PubMed] [Google Scholar]
- 415.Sen S, Kawahara B, Fukuto J, Chaudhuri G. Induction of a feed forward pro-apoptotic mechanistic loop by nitric oxide in a human breast cancer model. PLoS One. 2013;8:e70593. doi: 10.1371/journal.pone.0070593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 416.Shoulars K, Rodriguez MA, Thompson T, Turk J, Crowley J, Markaverich BM. Regulation of the nitric oxide pathway genes by tetrahydrofurandiols: Microarray analysis of MCF-7 human breast cancer cells. Cancer Lett. 2008;264:265–273. doi: 10.1016/j.canlet.2008.01.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 417.Simeone AM, Broemeling LD, Rosenblum J, Tari AM. HER2/neu reduces the apoptotic effects of N-(4-hydroxyphenyl)retinamide (4-HPR) in breast cancer cells by decreasing nitric oxide production. Oncogene. 2003;22:6739–6747. doi: 10.1038/sj.onc.1206786. [DOI] [PubMed] [Google Scholar]
- 418.Simeone AM, Ekmekcioglu S, Broemeling LD, Grimm EA, Tari AM. A novel mechanism by which N-(4-hydroxyphenyl)retinamide inhibits breast cancer cell growth: The production of nitric oxide. Mol Cancer Ther. 2002;1:1009–1017. [PubMed] [Google Scholar]
- 419.Simeone AM, McMurtry V, Nieves-Alicea R, Saavedra JE, Keefer LK, Johnson MM, Tari AM. TIMP-2 mediates the anti-invasive effects of the nitric oxide-releasing prodrug JS-K in breast cancer cells. Breast Cancer Res. 2008;10:R44. doi: 10.1186/bcr2095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 420.Singh T, Katiyar SK. Honokiol, a phytochemical from Magnolia spp., inhibits breast cancer cell migration by targeting nitric oxide and cyclooxygenase-2. Int J Oncol. 2011;38:769–776. doi: 10.3892/ijo.2011.899. [DOI] [PubMed] [Google Scholar]
- 421.Smeda M, Kieronska A, Adamski MG, Proniewski B, Sternak M, Mohaissen T, Przyborowski K, Derszniak K, Kaczor D, Stojak M, et al. Nitric oxide deficiency and endothelial-mesenchymal transition of pulmonary endothelium in the progression of 4T1 metastatic breast cancer in mice. Breast Cancer Res. 2018;20:86. doi: 10.1186/s13058-018-1013-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Song YK, Billiar TR, Lee YJ. Role of galectin-3 in breast cancer metastasis: Involvement of nitric oxide. Am J Pathol. 2002;160:1069–1075. doi: 10.1016/S0002-9440(10)64927-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Switzer CH, Cheng RY, Ridnour LA, Glynn SA, Ambs S, Wink DA. Ets-1 is a transcriptional mediator of oncogenic nitric oxide signaling in estrogen receptor-negative breast cancer. Breast Cancer Res. 2012;14:R125. doi: 10.1186/bcr3319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 424.Switzer CH, Glynn SA, Ridnour LA, Cheng RY, Vitek MP, Ambs S, Wink DA. Nitric oxide and protein phosphatase 2A provide novel therapeutic opportunities in ER-negative breast cancer. Trends Pharmacol Sci. 2011;32:644–651. doi: 10.1016/j.tips.2011.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 425.Ai Y, Kang F, Huang Z, Xue X, Lai Y, Peng S, Tian J, Zhang Y. Synthesis of CDDO-amino acid-nitric oxide donor trihybrids as potential antitumor agents against both drug-sensitive and drug-resistant colon cancer. J Med Chem. 2015;58:2452–2464. doi: 10.1021/jm5019302. [DOI] [PubMed] [Google Scholar]
- 426.Babykutty S, Suboj P, Srinivas P, Nair AS, Chandramohan K, Gopala S. Insidious role of nitric oxide in migration/invasion of colon cancer cells by upregulating MMP-2/9 via activation of cGMP-PKG-ERK signaling pathways. Clin Exp Metastasis. 2012;29:471–492. doi: 10.1007/s10585-012-9464-6. [DOI] [PubMed] [Google Scholar]
- 427.Bessa X, Elizalde JI, Mitjans F, Piñol V, Miquel R, Panés J, Piulats J, Piqué JM, Castells A. Leukocyte recruitment in colon cancer: Role of cell adhesion molecules, nitric oxide, and transforming growth factor beta1. Gastroenterology. 2002;122:1122–1132. doi: 10.1053/gast.2002.32369. [DOI] [PubMed] [Google Scholar]
- 428.Chattopadhyay M, Kodela R, Olson KR, Kashfi K. NOSH-aspirin (NBS-1120), a novel nitric oxide- and hydrogen sulfide-releasing hybrid is a potent inhibitor of colon cancer cell growth in vitro and in a xenograft mouse model. Biochem Biophys Res Commun. 2012;419:523–528. doi: 10.1016/j.bbrc.2012.02.051. [DOI] [PubMed] [Google Scholar]
- 429.Edes K, Cassidy P, Shami PJ, Moos PJ. JS-K, a nitric oxide prodrug, has enhanced cytotoxicity in colon cancer cells with knockdown of thioredoxin reductase 1. PLoS One. 2010;5:e8786. doi: 10.1371/journal.pone.0008786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Ishikawa T, Yoshida N, Higashihara H, Inoue M, Uchiyama K, Takagi T, Handa O, Kokura S, Naito Y, Okanoue T, Yoshikawa T. Different effects of constitutive nitric oxide synthase and heme oxygenase on pulmonary or liver metastasis of colon cancer in mice. Clin Exp Metastasis. 2003;20:445–450. doi: 10.1023/A:1025448403124. [DOI] [PubMed] [Google Scholar]
- 431.Jarry A, Charrier L, Bou-Hanna C, Devilder MC, Crussaire V, Denis MG, Vallette G, Laboisse CL. Position in cell cycle controls the sensitivity of colon cancer cells to nitric oxide-dependent programmed cell death. Cancer Res. 2004;64:4227–4234. doi: 10.1158/0008-5472.CAN-04-0254. [DOI] [PubMed] [Google Scholar]
- 432.Jenkins DC, Charles IG, Baylis SA, Lelchuk R, Radomski MW, Moncada S. Human colon cancer cell lines show a diverse pattern of nitric oxide synthase gene expression and nitric oxide generation. Br J Cancer. 1994;70:847–849. doi: 10.1038/bjc.1994.409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 433.Jeon HK, Choi SU, Jung NP. Association of the ERK1/2 and p38 kinase pathways with nitric oxide-induced apoptosis and cell cycle arrest in colon cancer cells. Cell Biol Toxicol. 2005;21:115–125. doi: 10.1007/s10565-005-0148-8. [DOI] [PubMed] [Google Scholar]
- 434.Kim MY. Sasa quelpaertensis Nakai extract induces p53-independent apoptosis via the elevation of nitric oxide production in human HCT116 colon cancer cells. Oncol Lett. 2020;19:3027–3034. doi: 10.3892/ol.2020.11379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 435.Kopecka J, Campia I, Brusa D, Doublier S, Matera L, Ghigo D, Bosia A, Riganti C. Nitric oxide and P-glycoprotein modulate the phagocytosis of colon cancer cells. J Cell Mol Med. 2011;15:1492–1504. doi: 10.1111/j.1582-4934.2010.01137.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 436.Liu Q, Chan ST, Mahendran R. Nitric oxide induces cyclooxygenase expression and inhibits cell growth in colon cancer cell lines. Carcinogenesis. 2003;24:637–642. doi: 10.1093/carcin/bgg014. [DOI] [PubMed] [Google Scholar]
- 437.Liu Q, Inoue H, Mahendran R. Transcriptional regulation of the COX-2 expression by nitric oxide in colon cancer cell lines. Oncol Rep. 2008;19:269–274. [PubMed] [Google Scholar]
- 438.Mojic M, Mijatovic S, Maksimovic-Ivanic D, Miljkovic D, Stosic-Grujicic S, Stankovic M, Mangano K, Travali S, Donia M, Fagone P, et al. Therapeutic potential of nitric oxide-modified drugs in colon cancer cells. Mol Pharmacol. 2012;82:700–710. doi: 10.1124/mol.112.077842. [DOI] [PubMed] [Google Scholar]
- 439.Millet A, Bettaieb A, Renaud F, Prevotat L, Hammann A, Solary E, Mignotte B, Jeannin JF. Influence of the nitric oxide donor glyceryl trinitrate on apoptotic pathways in human colon cancer cells. Gastroenterology. 2002;123:235–246. doi: 10.1053/gast.2002.34310. [DOI] [PubMed] [Google Scholar]
- 440.Oláh G, Módis K, Törö G, Hellmich MR, Szczesny B, Szabo C. Role of endogenous and exogenous nitric oxide, carbon monoxide and hydrogen sulfide in HCT116 colon cancer cell proliferation. Biochem Pharmacol. 2018;149:186–204. doi: 10.1016/j.bcp.2017.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 441.Prevotat L, Filomenko R, Solary E, Jeannin JF, Bettaieb A. Nitric oxide-induced down-regulation of beta-catenin in colon cancer cells by a proteasome-independent specific pathway. Gastroenterology. 2006;131:1142–1152. doi: 10.1053/j.gastro.2006.07.017. [DOI] [PubMed] [Google Scholar]
- 442.Rao CV. Nitric oxide signaling in colon cancer chemoprevention. Mutation Res. 2004;555:107–119. doi: 10.1016/j.mrfmmm.2004.05.022. [DOI] [PubMed] [Google Scholar]
- 443.Rao CV, Reddy BS, Steele VE, Wang CX, Liu X, Ouyang N, Patlolla JM, Simi B, Kopelovich L, Rigas B. Nitric oxide-releasing aspirin and indomethacin are potent inhibitors against colon cancer in azoxymethane-treated rats: Effects on molecular targets. Mol Cancer Ther. 2006;5:1530–1538. doi: 10.1158/1535-7163.MCT-06-0061. [DOI] [PubMed] [Google Scholar]
- 444.Riganti C, Miraglia E, Viarisio D, Costamagna C, Pescarmona G, Ghigo D, Bosia A. Nitric oxide reverts the resistance to doxorubicin in human colon cancer cells by inhibiting the drug efflux. Cancer Res. 2005;65:516–525. [PubMed] [Google Scholar]
- 445.Wang H, MacNaughton WK. Overexpressed beta-catenin blocks nitric oxide-induced apoptosis in colonic cancer cells. Cancer Res. 2005;65:8604–8607. doi: 10.1158/0008-5472.CAN-05-1169. [DOI] [PubMed] [Google Scholar]
- 446.Xie J, Xia L, Xiang W, He W, Yin H, Wang F, Gao T, Qi W, Yang Z, Yang X, et al. Metformin selectively inhibits metastatic colorectal cancer with the KRAS mutation by intracellular accumulation through silencing MATE1. Proc Natl Acad Sci USA. 2020;117:13012–13022. doi: 10.1073/pnas.1918845117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 447.Wenzel U, Kuntz S, De Sousa UJ, Daniel H. Nitric oxide suppresses apoptosis in human colon cancer cells by scavenging mitochondrial superoxide anions. Int J Cancer. 2003;106:666–675. doi: 10.1002/ijc.11294. [DOI] [PubMed] [Google Scholar]
- 448.Williams JL, Borgo S, Hasan I, Castillo E, Traganos F, Rigas B. Nitric oxide-releasing nonsteroidal anti-inflammatory drugs (NSAIDs) alter the kinetics of human colon cancer cell lines more effectively than traditional NSAIDs: Implications for colon cancer chemoprevention. Cancer Res. 2001;61:3285–3289. [PubMed] [Google Scholar]
- 449.Voss NCS, Kold-Petersen H, Boedtkjer E. Enhanced nitric oxide signaling amplifies vasorelaxation of human colon cancer feed arteries. Am J Physiol Heart Circ Physiol. 2019;316:H245–H254. doi: 10.1152/ajpheart.00368.2018. [DOI] [PubMed] [Google Scholar]
- 450.Tesei A, Rosetti M, Ulivi P, Fabbri F, Medri L, Vannini I, Bolla M, Amadori D, Zoli W. Study of molecular mechanisms of pro-apoptotic activity of NCX 4040, a novel nitric oxide-releasing aspirin, in colon cancer cell lines. J Transl Med. 2007;5:52. doi: 10.1186/1479-5876-5-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 451.Thomsen LL, Miles DW, Happerfield L, Bobrow LG, Knowles RG, Moncada S. Nitric oxide synthase activity in human breast cancer. Br J Cancer. 1995;72:41–44. doi: 10.1038/bjc.1995.274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 452.Stettner N, Rosen C, Bernshtein B, Gur-Cohen S, Frug J, Silberman A, Sarver A, Carmel-Neiderman NN, Eilam R, Biton I, et al. Induction of Nitric-Oxide metabolism in enterocytes alleviates colitis and inflammation-associated colon cancer. Cell Rep. 2018;23:1962–1976. doi: 10.1016/j.celrep.2018.04.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 453.Ambs S, Bennett WP, Merriam WG, Ogunfusika MO, Oser SM, Khan MA, Jones RT, Harris CC. Vascular endothelial growth factor and nitric oxide synthase expression in human lung cancer and the relation to p53. Br J Ccancer. 1998;78:233–239. doi: 10.1038/bjc.1998.470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 454.Cembrzynska-Nowak M, Bienkowska M, Szklarz E. Exogenous interleukin 2 regulates interleukin 6 and nitric oxide but not interferon gamma and tumor necrosis factor alpha production in bronchoalveolar leukocytes from patients with small cell lung cancer. Arch Immunol Ther Exp (Warsz) 1998;46:367–374. [PubMed] [Google Scholar]
- 455.Cembrzyńska-Nowak M, Bieńkowska M, Weryńska B, Dyła T, Jankowska R. The contribution of endotoxins present in the respiratory tract to overproduction of nitric oxide associated with impaired interleukin 6 release in bronchoalveolar leukocytes from lung cancer patients. Arch Immunol Ther Exp (Warsz) 2000;48:119–125. [PubMed] [Google Scholar]
- 456.Chen Z, Huang KY, Ling Y, Goto M, Duan HQ, Tong XH, Liu YL, Cheng YY, Morris-Natschke SL, Yang PC, et al. Discovery of an Oleanolic Acid/Hederagenin-nitric oxide donor hybrid as an EGFR tyrosine kinase inhibitor for non-small-cell lung cancer. J Natural Prod. 2019;82:3065–3073. doi: 10.1021/acs.jnatprod.9b00659. [DOI] [PubMed] [Google Scholar]
- 457.Deliu Z, Tamas T, Chowdhury J, Aqil M, Bassiony M, Radosevich JA. Expression of cross-tolerance to a wide range of conditions in a human lung cancer cell line after adaptation to nitric oxide. Tumour Biol. 2017;39:1010428317723778. doi: 10.1177/1010428317723778. [DOI] [PubMed] [Google Scholar]
- 458.Fu J, Han J, Meng T, Hu J, Yin J. Novel α-ketoamide based diazeniumdiolates as hydrogen peroxide responsive nitric oxide donors with anti-lung cancer activity. Chem Commun (Camb) 2019;55:12904–12907. doi: 10.1039/C9CC05266F. [DOI] [PubMed] [Google Scholar]
- 459.Fujimoto H, Ando Y, Yamashita T, Terazaki H, Tanaka Y, Sasaki J, Matsumoto M, Suga M, Ando M. Nitric oxide synthase activity in human lung cancer. Jpn J Cancer Res. 1997;88:1190–1198. doi: 10.1111/j.1349-7006.1997.tb00348.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 460.Gao X, Xuan Y, Benner A, Anusruti A, Brenner H, Schottker B. Nitric oxide metabolites and lung cancer incidence: A matched case-control study nested in the ESTHER cohort. Oxid Med Cell Longev. 2019;2019:6470950. doi: 10.1155/2019/6470950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 461.Hwang JH, Park SJ, Ko WG, Kang SM, Lee DB, Bang J, Park BJ, Wee CB, Kim DJ, Jang IS, Ko JH. Cordycepin induces human lung cancer cell apoptosis by inhibiting nitric oxide mediated ERK/Slug signaling pathway. Am J Cancer Res. 2017;7:417–432. [PMC free article] [PubMed] [Google Scholar]
- 462.Kaynar H, Meral M, Turhan H, Keles M, Celik G, Akcay F. Glutathione peroxidase, glutathione-S-transferase, catalase, xanthine oxidase, Cu-Zn superoxide dismutase activities, total glutathione, nitric oxide, and malondialdehyde levels in erythrocytes of patients with small cell and non-small cell lung cancer. Cancer Lett. 2005;227:133–139. doi: 10.1016/j.canlet.2004.12.005. [DOI] [PubMed] [Google Scholar]
- 463.Koizumi M, Yamazaki H, Toyokawa K, Yoshioka Y, Suzuki G, Ito M, Shinkawa K, Nishino K, Watanabe Y, Inoue T, et al. Influence of thoracic radiotherapy on exhaled nitric oxide levels in patients with lung cancer. Jpn J Clin Oncol. 2001;31:142–146. doi: 10.1093/jjco/hye028. [DOI] [PubMed] [Google Scholar]
- 464.Munaweera I, Shi Y, Koneru B, Patel A, Dang MH, Di Pasqua AJ, Balkus KJ., Jr Nitric oxide- and cisplatin-releasing silica nanoparticles for use against non-small cell lung cancer. J Inorg Biochem. 2015;153:23–31. doi: 10.1016/j.jinorgbio.2015.09.002. [DOI] [PubMed] [Google Scholar]
- 465.Maciag AE, Chakrapani H, Saavedra JE, Morris NL, Holland RJ, Kosak KM, Shami PJ, Anderson LM, Keefer LK. The nitric oxide prodrug JS-K is effective against non-small-cell lung cancer cells in vitro and in vivo: Involvement of reactive oxygen species. J Pharmacol Exp Ther. 2011;336:313–320. doi: 10.1124/jpet.110.174904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 466.Maiuthed A, Prakhongcheep O, Chanvorachote P. Microarray-based analysis of genes, transcription factors, and epigenetic modifications in lung cancer exposed to nitric oxide. Cancer Genom Proteomics. 2020;17:401–415. doi: 10.21873/cgp.20199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 467.Lee JJ, Kwon HK, Lee DS, Lee SW, Lee KK, Kim KJ, Kim JL. Mycelial extract of phellinus linteus induces cell death in A549 lung cancer cells and elevation of nitric oxide in raw 264.7 macrophage cells. Mycobiology. 2006;34:143–147. doi: 10.4489/MYCO.2006.34.3.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 468.Liaw YP, Ting TF, Ho CC, Chiou ZY. Cell type specificity of lung cancer associated with nitric oxide. Sci Total Environ. 2010;408:4931–4934. doi: 10.1016/j.scitotenv.2010.06.023. [DOI] [PubMed] [Google Scholar]
- 469.Liu PF, Zhao DH, Qi Y, Wang JG, Zhao M, Xiao K, Xie LX. The clinical value of exhaled nitric oxide in patients with lung cancer. Clin Respir J. 2018;12:23–30. doi: 10.1111/crj.12471. [DOI] [PubMed] [Google Scholar]
- 470.Luanpitpong S, Chanvorachote P. Nitric oxide and aggressive behavior of lung cancer cells. Anticancer Res. 2015;35:4585–4592. [PubMed] [Google Scholar]
- 471.Muto S, Takagi H, Owada Y, Inoue T, Watanabe Y, Yamaura T, Fukuhara M, Okabe N, Matsumura Y, Hasegawa T, et al. Serum nitric oxide as a predictive biomarker for bevacizumab in non-small cell lung cancer patients. Anticancer Res. 2017;37:3169–3174. doi: 10.21873/anticanres.11676. [DOI] [PubMed] [Google Scholar]
- 472.Masri F. Role of nitric oxide and its metabolites as potential markers in lung cancer. Ann Thorac Med. 2010;5:123–127. doi: 10.4103/1817-1737.65036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 473.Masri FA, Comhair SA, Koeck T, Xu W, Janocha A, Ghosh S, Dweik RA, Golish J, Kinter M, Stuehr DJ, et al. Abnormalities in nitric oxide and its derivatives in lung cancer. Am J Respir Crit Care Med. 2005;172:597–605. doi: 10.1164/rccm.200411-1523OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 474.Matsunaga T, Yamaji Y, Tomokuni T, Morita H, Morikawa Y, Suzuki A, Yonezawa A, Endo S, Ikari A, Iguchi K, et al. Nitric oxide confers cisplatin resistance in human lung cancer cells through upregulation of aldo-keto reductase 1B10 and proteasome. Free Radical Res. 2014;48:1371–1385. doi: 10.3109/10715762.2014.957694. [DOI] [PubMed] [Google Scholar]
- 475.Powan P, Chanvorachote P. Nitric oxide mediates cell aggregation and mesenchymal to epithelial transition in anoikis-resistant lung cancer cells. Mol Cell Biochem. 2014;393:237–245. doi: 10.1007/s11010-014-2066-7. [DOI] [PubMed] [Google Scholar]
- 476.Punathil T, Katiyar SK. Inhibition of non-small cell lung cancer cell migration by grape seed proanthocyanidins is mediated through the inhibition of nitric oxide, guanylate cyclase, and ERK1/2. Mol Carcinog. 2009;48:232–242. doi: 10.1002/mc.20473. [DOI] [PubMed] [Google Scholar]
- 477.Zhou J, Zhu Q, Yao H. Chemotherapy of non-small-cell lung cancer (NSCLC) and changes in serum sAPO-1/Fas and nitric oxide (NO) levels. Zhonghua Zhong Liu Za Zhi. 2000;22:225–227. (In Chinese) [PubMed] [Google Scholar]
- 478.Zhou H, Dong Y, Ma X, Xu J, Xu S. Development of a novel truncated deguelin derivative possessing nitric oxide donor as a potential anti-lung cancer agent. Fitoterapia. 2020;146:104670. doi: 10.1016/j.fitote.2020.104670. [DOI] [PubMed] [Google Scholar]
- 479.Zhang L, Liu J, Wang X, Li Z, Zhang X, Cao P, She X, Dai Q, Tang J, Liu Z. Upregulation of cytoskeleton protein and extracellular matrix protein induced by stromal-derived nitric oxide promotes lung cancer invasion and metastasis. Curr Mol Med. 2014;14:762–771. doi: 10.2174/1566524014666140724103147. [DOI] [PubMed] [Google Scholar]
- 480.Yongsanguanchai N, Pongrakhananon V, Mutirangura A, Rojanasakul Y, Chanvorachote P. Nitric oxide induces cancer stem cell-like phenotypes in human lung cancer cells. Am J Physiol Cell Physiol. 2015;308:C89–C100. doi: 10.1152/ajpcell.00187.2014. [DOI] [PubMed] [Google Scholar]
- 481.Wongvaranon P, Pongrakhananon V, Chunhacha P, Chanvorachote P. Acquired resistance to chemotherapy in lung cancer cells mediated by prolonged nitric oxide exposure. Anticancer Res. 2013;33:5433–5444. [PubMed] [Google Scholar]
- 482.Saisongkorh V, Maiuthed A, Chanvorachote P. Nitric oxide increases the migratory activity of non-small cell lung cancer cells via AKT-mediated integrin αv and β1 upregulation. Cell Oncol (Dordr) 2016;39:449–462. doi: 10.1007/s13402-016-0287-3. [DOI] [PubMed] [Google Scholar]
- 483.Saleem W, Suzuki Y, Mobaraki A, Yoshida Y, Noda S, Saitoh JI, Nakano T. Reduction of nitric oxide level enhances the radiosensitivity of hypoxic non-small cell lung cancer. Cancer Sci. 2011;102:2150–2156. doi: 10.1111/j.1349-7006.2011.02095.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 484.Sanuphan A, Chunhacha P, Pongrakhananon V, Chanvorachote P. Long-term nitric oxide exposure enhances lung cancer cell migration. Biomed Res Int. 2013;2013:186972. doi: 10.1155/2013/186972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 485.Sanuphan A, Chunhacha P, Pongrakhananon V, Chanvorachote P. Corrigendum to ‘Long-term nitric oxide exposure enhances lung cancer cell migration’. Biomed Res Int. 2015;2015:310636. doi: 10.1155/2015/310636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 486.Su X, Takahashi A, Guo G, Mori E, Okamoto N, Ohnishi K, Iwasaki T, Ohnishi T. Biphasic effects of nitric oxide radicals on radiation-induced lethality and chromosome aberrations in human lung cancer cells carrying different p53 gene status. Int J Radiat Oncol Biol Phys. 2010;77:559–565. doi: 10.1016/j.ijrobp.2009.12.059. [DOI] [PubMed] [Google Scholar]
- 487.Suzuki Y, Inui N, Karayama M, Imokawa S, Yamada T, Yokomura K, Asada K, Kusagaya H, Kaida Y, Matsuda H, et al. Effect of PD-1 inhibitor on exhaled nitric oxide and pulmonary function in non-small cell lung cancer patients with and without COPD. Int J Chron Obstruct Pulmon Dis. 2019;14:1867–1877. doi: 10.2147/COPD.S214610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 488.Szejniuk WM, Nielsen MS, Brønnum D, Takács-Szabó Z, Weinreich UM, Pilegaard Thomsen L, Bøgsted M, Jensen I, McCulloch T, Falkmer UG, et al. Fractional exhaled nitric oxide as a potential biomarker for radiation pneumonitis in patients with non-small cell lung cancer: A pilot study. Clin Transl Radiat Oncol. 2019;19:103–109. doi: 10.1016/j.ctro.2019.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 489.Arif M, Vedamurthy BM, Choudhari R, Ostwal YB, Mantelingu K, Kodaganur GS, Kundu TK. Nitric oxide-mediated histone hyperacetylation in oral cancer: Target for a water-soluble HAT inhibitor, CTK7A. Chem Biol. 2010;17:903–913. doi: 10.1016/j.chembiol.2010.06.014. [DOI] [PubMed] [Google Scholar]
- 490.Beevi SS, Rasheed AM, Geetha A. Evaluation of oxidative stress and nitric oxide levels in patients with oral cavity cancer. Jpn J Clin Oncol. 2004;34:379–385. doi: 10.1093/jjco/hyh058. [DOI] [PubMed] [Google Scholar]
- 491.Korde Choudhari S, Sridharan G, Gadbail A, Poornima V. Nitric oxide and oral cancer: A review. Oral Oncol. 2012;48:475–483. doi: 10.1016/j.oraloncology.2012.01.003. [DOI] [PubMed] [Google Scholar]
- 492.Patel JB, Shah FD, Shukla SN, Shah PM, Patel PS. Role of nitric oxide and antioxidant enzymes in the pathogenesis of oral cancer. J Cancer Res Ther. 2009;5:247–253. doi: 10.4103/0973-1482.59898. [DOI] [PubMed] [Google Scholar]
- 493.Brankovic B, Stanojevic G, Stojanovic I, Veljkovic A, Kocic G, Janosevic P, Nestorovic M, Petrovic D, Djindjic B, Pavlovic D, Krivokapic Z. Nitric oxide synthesis modulation-a possible diagnostic and therapeutic target in colorectal cancer. J BUON. 2017;22:162–169. [PubMed] [Google Scholar]
- 494.El-Mezayen HA, Toson el SA, Darwish H, El-Badry E. Discriminant function based on parameters of hyaluronic acid metabolism and nitric oxide to differentiate metastatic from non-metastatic colorectal cancer patients. Tumour Biol. 2012;33:995–1004. doi: 10.1007/s13277-012-0332-4. [DOI] [PubMed] [Google Scholar]
- 495.Gao Y, Zhou S, Pang L, Yang J, Li HJ, Huo X, Qian SY. Celastrol suppresses nitric oxide synthases and the angiogenesis pathway in colorectal cancer. Free Radic Res. 2019;53:324–334. doi: 10.1080/10715762.2019.1575512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 496.Gao Y, Zhou S, Xu Y, Sheng S, Qian SY, Huo X. Nitric oxide synthase inhibitors 1400W and L-NIO inhibit angiogenesis pathway of colorectal cancer. Nitric Oxide. 2019;83:33–39. doi: 10.1016/j.niox.2018.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 497.Jiang H, Verovski VN, Leonard W, Law KL, Vermeersch M, Storme G, Van den Berge D, Gevaert T, Sermeus A, De Ridder M. Hepatocytes determine the hypoxic microenvironment and radiosensitivity of colorectal cancer cells through production of nitric oxide that targets mitochondrial respiration. Int J Radiat Oncol Biol Phys. 2013;85:820–827. doi: 10.1016/j.ijrobp.2012.07.2359. [DOI] [PubMed] [Google Scholar]
- 498.Krzystek-Korpacka M, Szczęśniak-Sięga B, Szczuka I, Fortuna P, Zawadzki M, Kubiak A, Mierzchała-Pasierb M, Fleszar MG, Lewandowski Ł, Serek P, et al. L-Arginine/Nitric oxide pathway is altered in colorectal cancer and can be modulated by novel derivatives from oxicam class of non-steroidal anti-inflammatory drugs. Cancers (Basel) 2020;12:2594. doi: 10.3390/cancers12092594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 499.Liu Y, Borchert GL, Phang JM. Polyoma enhancer activator 3, an ets transcription factor, mediates the induction of cyclooxygenase-2 by nitric oxide in colorectal cancer cells. J Biol Chemistry. 2004;279:18694–18700. doi: 10.1016/S0021-9258(20)77093-6. [DOI] [PubMed] [Google Scholar]
- 500.McIlhatton MA, Tyler J, Burkholder S, Ruschoff J, Rigas B, Kopelovich L, Fishel R. Nitric oxide-donating aspirin derivatives suppress microsatellite instability in mismatch repair-deficient and hereditary nonpolyposis colorectal cancer cells. Cancer Res. 2007;67:10966–10975. doi: 10.1158/0008-5472.CAN-07-2562. [DOI] [PubMed] [Google Scholar]
- 501.Moochhala S, Chhatwal VJ, Chan ST, Ngoi SS, Chia YW, Rauff A. Nitric oxide synthase activity and expression in human colorectal cancer. Carcinogenesis. 1996;17:1171–1174. doi: 10.1093/carcin/17.5.1171. [DOI] [PubMed] [Google Scholar]
- 502.Muscat JE, Dyer AM, Rosenbaum RE, Rigas B. Nitric oxide-releasing medications and colorectal cancer risk: The framingham study. Anticancer Res. 2005;25:4471–4474. [PubMed] [Google Scholar]
- 503.Mandal P. Molecular signature of nitric oxide on major cancer hallmarks of colorectal carcinoma. Inflammopharmacology. 2018;26:331–336. doi: 10.1007/s10787-017-0435-z. [DOI] [PubMed] [Google Scholar]
- 504.Yagihashi N, Kasajima H, Sugai S, Matsumoto K, Ebina Y, Morita T, Murakami T, Yagihashi S. Increased in situ expression of nitric oxide synthase in human colorectal cancer. Virchows Archiv. 2000;436:109–114. doi: 10.1007/PL00008208. [DOI] [PubMed] [Google Scholar]
- 505.Yu LB, Dong XS, Sun WZ, Zhao DL, Yang Y. Effect of a nitric oxide synthase inhibitor NG-nitro-L-arginine methyl ester on invasion of human colorectal cancer cell line SL-174T. World J Gastroenterol. 2005;11:6385–6388. doi: 10.3748/wjg.v11.i40.6385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 506.Wei B, Wei HB, Qi CL, Han XY, Wang TB. Effect of a selective inducible nitric oxide synthase inhibitor on cell growth in human colorectal cancer Lovo cell line. Zhonghua Wei Chang Wai Ke Za Zhi. 2008;11:280–283. (In Chinese) [PubMed] [Google Scholar]
- 507.Siedlar M, Mytar B, Hyszko M, Zembala M. Involvement of protein kinases in signalling for nitric oxide (NO) and tumour necrosis factor alpha (TNF) production by monocytes stimulated with colorectal DeTa cancer cells: The lack of evidence for the role of TNF in the regulation of NO production. Immunol Lett. 1999;65:189–195. doi: 10.1016/S0165-2478(98)00103-5. [DOI] [PubMed] [Google Scholar]
- 508.Atala A. Re: Alterations of tumor microenvironment by nitric oxide impedes castration-resistant prostate cancer growth. J Urol. 2019;202:35–36. doi: 10.1097/JU.0000000000000251. [DOI] [PubMed] [Google Scholar]
- 509.Dillioglugil MO, Mekik H, Muezzinoglu B, Ozkan TA, Demir CG, Dillioglugil O. Blood and tissue nitric oxide and malondialdehyde are prognostic indicators of localized prostate cancer. Int Urol Nephrol. 2012;44:1691–1696. doi: 10.1007/s11255-012-0221-1. [DOI] [PubMed] [Google Scholar]
- 510.Della Pietra E, Simonella F, Bonavida B, Xodo LE, Rapozzi V. Repeated sub-optimal photodynamic treatments with pheophorbide a induce an epithelial mesenchymal transition in prostate cancer cells via nitric oxide. Nitric Oxide. 2015;45:43–53. doi: 10.1016/j.niox.2015.02.005. [DOI] [PubMed] [Google Scholar]
- 511.Huh J, Liepins A, Zielonka J, Andrekopoulos C, Kalyanaraman B, Sorokin A. Cyclooxygenase 2 rescues LNCaP prostate cancer cells from sanguinarine-induced apoptosis by a mechanism involving inhibition of nitric oxide synthase activity. Cancer Res. 2006;66:3726–3736. doi: 10.1158/0008-5472.CAN-05-4033. [DOI] [PubMed] [Google Scholar]
- 512.Lee KM, Kang D, Park SK, Berndt SI, Reding D, Chatterjee N, Chanock S, Huang WY, Hayes RB. Nitric oxide synthase gene polymorphisms and prostate cancer risk. Carcinogenesis. 2009;30:621–625. doi: 10.1093/carcin/bgp028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 513.Rezakhani L, Khazaei MR, Ghanbari A, Khazaei M. Crab shell extract induces prostate cancer cell line (LNcap) apoptosis and decreases nitric oxide secretion. Cell J. 2017;19:231–237. doi: 10.22074/cellj.2016.4879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 514.Royle JS, Ross JA, Ansell I, Bollina P, Tulloch DN, Habib FK. Nitric oxide donating nonsteroidal anti-inflammatory drugs induce apoptosis in human prostate cancer cell systems and human prostatic stroma via caspase-3. J Urol. 2004;172:338–344. doi: 10.1097/01.ju.0000132367.02834.41. [DOI] [PubMed] [Google Scholar]
- 515.Wang D, Lu S, Dong Z. Regulation of TGF-beta1 gene transcription in human prostate cancer cells by nitric oxide. Prostate. 2007;67:1825–1833. doi: 10.1002/pros.20669. [DOI] [PubMed] [Google Scholar]
- 516.Tan G, Qiu M, Chen L, Zhang S, Ke L, Liu J. JS-K, a nitric oxide pro-drug, regulates growth and apoptosis through the ubiquitin-proteasome pathway in prostate cancer cells. BMC Cancer. 2017;17:376. doi: 10.1186/s12885-017-3351-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 517.Thomas LN, Morehouse TJ, Too CK. Testosterone and prolactin increase carboxypeptidase-D and nitric oxide levels to promote survival of prostate cancer cells. Prostate. 2012;72:450–460. doi: 10.1002/pros.21446. [DOI] [PubMed] [Google Scholar]
- 518.Siemens DR, Heaton JP, Adams MA, Kawakami J, Graham CH. Phase II study of nitric oxide donor for men with increasing prostate-specific antigen level after surgery or radiotherapy for prostate cancer. Urology. 2009;74:878–883. doi: 10.1016/j.urology.2009.03.004. [DOI] [PubMed] [Google Scholar]
- 519.Soni Y, Softness K, Arora H, Ramasamy R. The Yin yang role of nitric oxide in prostate cancer. Am J Mens Health. 2020;14:1557988320903191. doi: 10.1177/1557988320903191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 520.Caneba CA, Yang L, Baddour J, Curtis R, Win J, Hartig S, Marini J, Nagrath D. Nitric oxide is a positive regulator of the Warburg effect in ovarian cancer cells. Cell Death Dis. 2014;5:e1302. doi: 10.1038/cddis.2014.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 521.El-Sehemy A, Chang AC, Azad AK, Gupta N, Xu Z, Steed H, Karsan A, Fu Y. Notch activation augments nitric oxide/soluble guanylyl cyclase signaling in immortalized ovarian surface epithelial cells and ovarian cancer cells. Cell Signal. 2013;25:2780–2787. doi: 10.1016/j.cellsig.2013.09.008. [DOI] [PubMed] [Google Scholar]
- 522.El-Sehemy A, Postovit LM, Fu Y. Nitric oxide signaling in human ovarian cancer: A potential therapeutic target. Nitric Oxide. 2016;54:30–37. doi: 10.1016/j.niox.2016.02.002. [DOI] [PubMed] [Google Scholar]
- 523.Kielbik M, Klink M, Brzezinska M, Szulc I, Sulowska Z. Nitric oxide donors: Spermine/NO and diethylenetriamine/NO induce ovarian cancer cell death and affect STAT3 and AKT signaling proteins. Nitric Oxide. 2013;35:93–109. doi: 10.1016/j.niox.2013.09.001. [DOI] [PubMed] [Google Scholar]
- 524.Leung EL, Fraser M, Fiscus RR, Tsang BK. Cisplatin alters nitric oxide synthase levels in human ovarian cancer cells: Involvement in p53 regulation and cisplatin resistance. Br J Cancer. 2008;98:1803–1809. doi: 10.1038/sj.bjc.6604375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 525.Thomsen LL, Sargent JM, Williamson CJ, Elgie AW. Nitric oxide synthase activity in fresh cells from ovarian tumour tissue: Relationship of enzyme activity with clinical parameters of patients with ovarian cancer. Biochem Pharmacol. 1998;56:1365–1370. doi: 10.1016/S0006-2952(98)00187-7. [DOI] [PubMed] [Google Scholar]
- 526.Salimian Rizi B, Caneba C, Nowicka A, Nabiyar AW, Liu X, Chen K, Klopp A, Nagrath D. Nitric oxide mediates metabolic coupling of omentum-derived adipose stroma to ovarian and endometrial cancer cells. Cancer Res. 2015;75:456–471. doi: 10.1158/0008-5472.CAN-14-1337. [DOI] [PubMed] [Google Scholar]
- 527.Stevens EV, Carpenter AW, Shin JH, Liu J, Der CJ, Schoenfisch MH. Nitric oxide-releasing silica nanoparticle inhibition of ovarian cancer cell growth. Mol Pharm. 2010;7:775–785. doi: 10.1021/mp9002865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 528.Camp ER, Yang A, Liu W, Fan F, Somcio R, Hicklin DJ, Ellis LM. Roles of nitric oxide synthase inhibition and vascular endothelial growth factor receptor-2 inhibition on vascular morphology and function in an in vivo model of pancreatic cancer. Clin Cancer Res. 2006;12:2628–2633. doi: 10.1158/1078-0432.CCR-05-2257. [DOI] [PubMed] [Google Scholar]
- 529.Chen X, Jia F, Li Y, Deng Y, Huang Y, Liu W, Jin Q, Ji J. Nitric oxide-induced stromal depletion for improved nanoparticle penetration in pancreatic cancer treatment. Biomaterials. 2020;246:119999. doi: 10.1016/j.biomaterials.2020.119999. [DOI] [PubMed] [Google Scholar]
- 530.Fujita M, Imadome K, Endo S, Shoji Y, Yamada S, Imai T. Nitric oxide increases the invasion of pancreatic cancer cells via activation of the PI3K-AKT and RhoA pathways after carbon ion irradiation. FEBS Lett. 2014;588:3240–3250. doi: 10.1016/j.febslet.2014.07.006. [DOI] [PubMed] [Google Scholar]
- 531.Fujita M, Somasundaram V, Basudhar D, Cheng RYS, Ridnour LA, Higuchi H, Imadome K, No JH, Bharadwaj G, Wink DA. Role of nitric oxide in pancreatic cancer cells exhibiting the invasive phenotype. Redox Biol. 2019;22:101158. doi: 10.1016/j.redox.2019.101158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 532.Zhou H, Huang L, Sun Y, Rigas B. Nitric oxide-donating aspirin inhibits the growth of pancreatic cancer cells through redox-dependent signaling. Cancer Lett. 2009;273:292–299. doi: 10.1016/j.canlet.2008.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 533.Wang B, Wei D, Crum VE, Richardson EL, Xiong HH, Luo Y, Huang S, Abbruzzese JL, Xie K. A novel model system for studying the double-edged roles of nitric oxide production in pancreatic cancer growth and metastasis. Oncogene. 2003;22:1771–1782. doi: 10.1038/sj.onc.1206386. [DOI] [PubMed] [Google Scholar]
- 534.Wang L, Xie K. Nitric oxide and pancreatic cancer pathogenesis, prevention, and treatment. Curr Pharm Design. 2010;16:421–427. doi: 10.2174/138161210790232194. [DOI] [PubMed] [Google Scholar]
- 535.Thomas WJ, Thomas DL, Knezetic JA, Adrian TE. The role of oxygen-derived free radicals and nitric oxide in cytokine-induced antiproliferation of pancreatic cancer cells. Pancreas. 2002;24:161–168. doi: 10.1097/00006676-200203000-00007. [DOI] [PubMed] [Google Scholar]
- 536.Stewart GD, Nanda J, Katz E, Bowman KJ, Christie JG, Brown DJ, McLaren DB, Riddick AC, Ross JA, Jones GD, Habib FK. DNA strand breaks and hypoxia response inhibition mediate the radiosensitisation effect of nitric oxide donors on prostate cancer under varying oxygen conditions. Biochem Pharmacol. 2011;81:203–210. doi: 10.1016/j.bcp.2010.09.022. [DOI] [PubMed] [Google Scholar]
- 537.Sugita H, Kaneki M, Furuhashi S, Hirota M, Takamori H, Baba H. Nitric oxide inhibits the proliferation and invasion of pancreatic cancer cells through degradation of insulin receptor substrate-1 protein. Mol Cancer Res. 2010;8:1152–1163. doi: 10.1158/1541-7786.MCR-09-0472. [DOI] [PubMed] [Google Scholar]
- 538.Caygill CP, Royston C, Charlett A, Wall CM, Gatenby PA, Ramus JR, Watson A, Winslet M, Hourigan CS, Dev Bardhan K. Barrett's, blood groups and progression to oesophageal cancer: Is nitric oxide the link? Eur J Gastroenterol Hepatol. 2011;23:801–806. doi: 10.1097/MEG.0b013e3283489dcf. [DOI] [PubMed] [Google Scholar]
- 539.Sun GG, Hu WN, Zhang J, Li CL, Yang CR. Effect of nitric oxide on esophageal cancer cell line TE-1. Chin Med Sci J. 2013;28:44–49. doi: 10.1016/S1001-9294(13)60018-8. [DOI] [PubMed] [Google Scholar]
- 540.Gecit I, Aslan M, Gunes M, Pirincci N, Esen R, Demir H, Ceylan K. Serum prolidase activity, oxidative stress, and nitric oxide levels in patients with bladder cancer. J Cancer Res Clin Oncol. 2012;138:739–743. doi: 10.1007/s00432-011-1136-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 541.Gecit İ, Eryılmaz R, Kavak S, Meral İ, Demir H, Pirinççi N, Güneş M, Taken K. The prolidase activity, oxidative stress, and nitric oxide levels of bladder tissues with or without tumor in patients with bladder cancer. J Membr Biol. 2017;250:455–459. doi: 10.1007/s00232-017-9971-0. [DOI] [PubMed] [Google Scholar]
- 542.Jansson OT, Morcos E, Brundin L, Lundberg JO, Adolfsson J, Söderhäll M, Wiklund NP. The role of nitric oxide in bacillus Calmette-Guerin mediated anti-tumour effects in human bladder cancer. Br J Cancer. 1998;78:588–592. doi: 10.1038/bjc.1998.545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 543.Kilic S, Bayraktar N, Beytur A, Ergin H, Bayraktar M, Egri M. Can the levels of nitric oxide in the urine, serum and tumor tissue be putative markers for bladder cancer? Int J Urol. 2006;13:1079–1085. doi: 10.1111/j.1442-2042.2006.01481.x. [DOI] [PubMed] [Google Scholar]
- 544.Khalifa A, Eissa S, Aziz A. Determination of cytosolic citrulline and nitrate as indicators of nitric oxide in bladder cancer: Possible association with basic fibroblast growth factor. Clin Biochem. 1999;32:635–638. doi: 10.1016/S0009-9120(99)00069-7. [DOI] [PubMed] [Google Scholar]
- 545.Morcos E, Jansson OT, Adolfsson J, Kratz G, Wiklund NP. Endogenously formed nitric oxide modulates cell growth in bladder cancer cell lines. Urology. 1999;53:1252–1257. doi: 10.1016/S0090-4295(99)00033-3. [DOI] [PubMed] [Google Scholar]
- 546.Wang Z, Fu S, Chen Y, Qin D. Modulation of nitric oxide on lymphokine-activated killer cells in patients with bladder cancer. Chin Med Sci J. 2001;16:213. [PubMed] [Google Scholar]
- 547.Thiel T, Ryk C, Chatzakos V, Hallén Grufman K, Bavand-Chobot N, Flygare J, Wiklund NP, de Verdier PJ. Secondary stimulation from Bacillus Calmette-Guerin induced macrophages induce nitric oxide independent cell-death in bladder cancer cells. Cancer Lett. 2014;348:119–125. doi: 10.1016/j.canlet.2014.03.014. [DOI] [PubMed] [Google Scholar]
- 548.Saygili EI, Akcay T, Dincer Y, Obek C, Kural AR, Cakalir C. Methylguanine DNA methyl transferase activities, glutathione s transferase and nitric oxide in bladder cancer patients. Cancer Invest. 2006;24:256–260. doi: 10.1080/07357900600634120. [DOI] [PubMed] [Google Scholar]
- 549.Bakan E, Taysi S, Polat MF, Dalga S, Umudum Z, Bakan N, Gumus M. Nitric oxide levels and lipid peroxidation in plasma of patients with gastric cancer. Jap J Clin Oncol. 2002;32:162–166. doi: 10.1093/jjco/hyf035. [DOI] [PubMed] [Google Scholar]
- 550.Dincer Y, Akcay T, Tortum OB, Dogusoy G. Nitric oxide and antioxidant defense in patients with gastric cancer. Dig Dis Sci. 2006;51:1367–1370. doi: 10.1007/s10620-005-9021-0. [DOI] [PubMed] [Google Scholar]
- 551.Holian O, Wahid S, Atten MJ, Attar BM. Inhibition of gastric cancer cell proliferation by resveratrol: Role of nitric oxide. Am J Physiol Gastrointest Liver Physiol. 2002;282:G809–G816. doi: 10.1152/ajpgi.00193.2001. [DOI] [PubMed] [Google Scholar]
- 552.Eroğlu A, Demirci S, Ayyildiz A, Kocaoğlu H, Akbulut H, Akgül H, Elhan HA. Serum concentrations of vascular endothelial growth factor and nitrite as an estimate of in vivo nitric oxide in patients with gastric cancer. Br J Cancer. 1999;80:1630–1634. doi: 10.1038/sj/bjc/6690573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 553.Koh E, Noh SH, Lee YD, Lee HY, Han JW, Lee HW, Hong S. Differential expression of nitric oxide synthase in human stomach cancer. Cancer Lett. 1999;146:173–180. doi: 10.1016/S0304-3835(99)00265-7. [DOI] [PubMed] [Google Scholar]
- 554.Oshima T, Imada T, Nagashima Y, Cho H, Shiozawa M, Rino Y, Takanashi Y. Role of nitric oxide in human gastric cancer cells treated with 5-fluorouracil. Oncol Rep. 2001;8:847–849. doi: 10.3892/or.8.4.847. [DOI] [PubMed] [Google Scholar]
- 555.Rajnakova A, Goh PM, Chan ST, Ngoi SS, Alponat A, Moochhala S. Expression of differential nitric oxide synthase isoforms in human normal gastric mucosa and gastric cancer tissue. Carcinogenesis. 1997;18:1841–1845. doi: 10.1093/carcin/18.9.1841. [DOI] [PubMed] [Google Scholar]
- 556.Yao X, Wu Y, Zhu M, Qian H, Chen Y. Nitric oxide/cyclic guanosine monophosphate inducers sodium nitroprusside and L-arginine inhibit the proliferation of gastric cancer cells via the activation of type II cyclic guanosine monophosphate-dependent protein kinase. Oncol Lett. 2015;10:479–484. doi: 10.3892/ol.2015.3229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 557.Sang J, Chen Y, Tao Y. Nitric oxide inhibits gastric cancer cell growth through the modulation of the Akt pathway. Mol Med Rep. 2011;4:1163–1167. doi: 10.3892/mmr.2011.535. [DOI] [PubMed] [Google Scholar]
- 558.Sang JR, Chen YC, Shao GB, Huang XJ. Effect of nitric oxide on the proliferation of AGS gastric cancer cells. Chin J Cancer. 2010;29:158–162. doi: 10.5732/cjc.009.10315. [DOI] [PubMed] [Google Scholar]
- 559.Li YH, Guo M, Shi SW, Zhang QL, Yang SP, Liu JG. A ruthenium-nitrosyl-functionalized nanoplatform for the targeting of liver cancer cells and NIR-light-controlled delivery of nitric oxide combined with photothermal therapy. J Mater Chem B. 2017;5:7831–7838. doi: 10.1039/C7TB02059G. [DOI] [PubMed] [Google Scholar]
- 560.Muntane J, De la Rosa AJ, Marin LM, Padillo FJ. Nitric oxide and cell death in liver cancer cells. Mitochondrion. 2013;13:257–262. doi: 10.1016/j.mito.2012.09.004. [DOI] [PubMed] [Google Scholar]
- 561.Sayed-Ahmad MM, Mohamad MA. Contribution of nitric oxide and epidermal growth factor receptor in anti-metastatic potential of paclitaxel in human liver cancer cell (HebG2) J Egypt Natl Canc Inst. 2005;17:35–41. [PubMed] [Google Scholar]
- 562.Li J, Rao H, Jin C, Liu J. Involvement of the Toll-like receptor/Nitric oxide signaling pathway in the pathogenesis of cervical cancer caused by high-risk human papillomavirus infection. Biomed Res Int. 2017;2017:7830262. doi: 10.1155/2017/7830262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 563.Park IC, Woo SH, Park MJ, Lee HC, Lee SJ, Hong YJ, Lee SH, Hong SI, Rhee CH. Ionizing radiation and nitric oxide donor sensitize Fas-induced apoptosis via up-regulation of Fas in human cervical cancer cells. Oncol Rep. 2003;10:629–633. [PubMed] [Google Scholar]
- 564.Wei XM, Wang Q, Gao SJ, Sui L. Relationship between nitric oxide in cervical microenvironment and different HPV types and effect on cervical cancer cells. Zhonghua Fu Chan Ke Za Zhi. 2011;46:260–265. (In Chinese) [PubMed] [Google Scholar]
- 565.Sudhesh P, Tamilarasan K, Arumugam P, Berchmans S. Nitric oxide releasing photoresponsive nanohybrids as excellent therapeutic agent for cervical cancer cell lines. ACS Appl Mater Interfaces. 2013;5:8263–8266. doi: 10.1021/am402086m. [DOI] [PubMed] [Google Scholar]
- 566.Sundaram MK, Khan MA, Alalami U, Somvanshi P, Bhardwaj T, Pramodh S, Raina R, Shekfeh Z, Haque S, Hussain A. Phytochemicals induce apoptosis by modulation of nitric oxide signaling pathway in cervical cancer cells. Eur Rev Med Pharmacol Sci. 2020;24:11827–11844. doi: 10.26355/eurrev_202011_23840. [DOI] [PubMed] [Google Scholar]
- 567.Menendez L, Juarez L, Garcia V, Hidalgo A, Baamonde A. Involvement of nitric oxide in the inhibition of bone cancer-induced hyperalgesia through the activation of peripheral opioid receptors in mice. Neuropharmacology. 2007;53:71–80. doi: 10.1016/j.neuropharm.2007.04.011. [DOI] [PubMed] [Google Scholar]
- 568.Yang Y, Zhang J, Liu Y, Zheng Y, Bo J, Zhou X, Wang J, Ma Z. Role of nitric oxide synthase in the development of bone cancer pain and effect of L-NMMA. Mol Med Rep. 2016;13:1220–1226. doi: 10.3892/mmr.2015.4647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 569.Giusti M, Valenti S, Guazzini B, Molinari E, Cavallero D, Augeri C, Minuto F. Circulating nitric oxide is modulated by recombinant human TSH administration during monitoring of thyroid cancer remnant. J Endocrinol Invest. 2003;26:1192–1197. doi: 10.1007/BF03349156. [DOI] [PubMed] [Google Scholar]
- 570.Ragot T, Provost C, Prignon A, Cohen R, Lepoivre M, Lausson S. Apoptosis induction by combination of drugs or a conjugated molecule associating non-steroidal anti-inflammatory and nitric oxide donor effects in medullary thyroid cancer models: Implication of the tumor suppressor p73. Thyroid Res. 2015;8:13. doi: 10.1186/s13044-015-0025-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 571.Gallo O, Masini E, Morbidelli L, Franchi A, Fini-Storchi I, Vergari WA, Ziche M. Role of nitric oxide in angiogenesis and tumor progression in head and neck cancer. J Natl Cancer Inst. 1998;90:587–596. doi: 10.1093/jnci/90.8.587. [DOI] [PubMed] [Google Scholar]
- 572.Kawakami K, Kawakami M, Puri RK. Nitric oxide accelerates interleukin-13 cytotoxin-mediated regression in head and neck cancer animal model. Clin Cancer Res. 2004;10:5264–5270. doi: 10.1158/1078-0432.CCR-04-0314. [DOI] [PubMed] [Google Scholar]
- 573.Wu HL, Chu YH, Tai YH, Tsou MY, Wu CH, Lo WL, Tai SK, Yeh CC, Lu CC. Stage-dependent angiopoietin-Tie2 and nitric oxide signaling of erythrocytes in response to surgical trauma in head and neck cancer. World J Surg Oncol. 2020;18:209. doi: 10.1186/s12957-020-01991-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 574.Utispan K, Koontongkaew S. High nitric oxide adaptation in isogenic primary and metastatic head and neck cancer cells. Anticancer Res. 2020;40:2657–2665. doi: 10.21873/anticanres.14236. [DOI] [PubMed] [Google Scholar]
- 575.Taysi S, Uslu C, Akcay F, Sutbeyaz MY. Malondialdehyde and nitric oxide levels in the plasma of patients with advanced laryngeal cancer. Surg Today. 2003;33:651–654. doi: 10.1007/s00595-002-2562-3. [DOI] [PubMed] [Google Scholar]
- 576.Sanhueza C, Araos J, Naranjo L, Barros E, Subiabre M, Toledo F, Gutiérrez J, Chiarello DI, Pardo F, Leiva A, Sobrevia L. Nitric oxide and pH modulation in gynaecological cancer. J Cell Mol Med. 2016;20:2223–2230. doi: 10.1111/jcmm.12921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 577.Chattopadhyay M, Kodela R, Nath N, Barsegian A, Boring D, Kashfi K. Hydrogen sulfide-releasing aspirin suppresses NF-κB signaling in estrogen receptor negative breast cancer cells in vitro and in vivo. Biochem Pharmacol. 2012;83:723–732. doi: 10.1016/j.bcp.2011.12.019. [DOI] [PubMed] [Google Scholar]
- 578.Dong Q, Yang B, Han JG, Zhang MM, Liu W, Zhang X, Yu HL, Liu ZG, Zhang SH, Li T, et al. A novel hydrogen sulfide-releasing donor, HA-ADT, suppresses the growth of human breast cancer cells through inhibiting the PI3K/AKT/mTOR and Ras/Raf/MEK/ERK signaling pathways. Cancer Lett. 2019;455:60–72. doi: 10.1016/j.canlet.2019.04.031. [DOI] [PubMed] [Google Scholar]
- 579.Li Y, Zhou J, Wang L, Xie Z. Endogenous hydrogen sulfide-triggered MOF-based nanoenzyme for synergic cancer therapy. ACS Appl Mater Interfaces. 2020;12:30213–30220. doi: 10.1021/acsami.0c08659. [DOI] [PubMed] [Google Scholar]
- 580.Lv M, Li Y, Ji MH, Zhuang M, Tang JH. Inhibition of invasion and epithelial-mesenchymal transition of human breast cancer cells by hydrogen sulfide through decreased phospho-p38 expression. Mol Med Rep. 2014;10:341–346. doi: 10.3892/mmr.2014.2161. [DOI] [PubMed] [Google Scholar]
- 581.Choi KS, Song H, Kim EH, Choi JH, Hong H, Han YM, Hahm KB. Inhibition of Hydrogen Sulfide-induced angiogenesis and inflammation in vascular endothelial cells: Potential mechanisms of gastric cancer prevention by korean red ginseng. J Ginseng Res. 2012;36:135–145. doi: 10.5142/jgr.2012.36.2.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 582.Kawahara Y, Hirashita Y, Tamura C, Kudo Y, Sakai K, Togo K, Fukuda K, Matsunari O, Okamoto K, Ogawa R, et al. Helicobacter pylori infection modulates endogenous hydrogen sulfide production in gastric cancer AGS cells. Helicobacter. 2020;25:e12732. doi: 10.1111/hel.12732. [DOI] [PubMed] [Google Scholar]
- 583.Sekiguchi F, Sekimoto T, Ogura A, Kawabata A. Endogenous hydrogen sulfide enhances cell proliferation of human gastric cancer AGS cells. Biol Pharm Bull. 2016;39:887–890. doi: 10.1248/bpb.b15-01015. [DOI] [PubMed] [Google Scholar]
- 584.Wang R, Tao B, Fan Q, Wang S, Chen L, Zhang J, Hao Y, Dong S, Wang Z, Wang W, et al. Fatty-acid receptor CD36 functions as a hydrogen sulfide-targeted receptor with its Cys333-Cys272 disulfide bond serving as a specific molecular switch to accelerate gastric cancer metastasis. EBioMedicine. 2019;45:108–123. doi: 10.1016/j.ebiom.2019.06.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 585.Ye F, Li X, Sun K, Xu W, Shi H, Bian J, Lu R, Ye Y. Inhibition of endogenous hydrogen sulfide biosynthesis enhances the anti-cancer effect of 3,3′-diindolylmethane in human gastric cancer cells. Life Sci. 2020;261:118348. doi: 10.1016/j.lfs.2020.118348. [DOI] [PubMed] [Google Scholar]
- 586.Chen S, Yue T, Huang Z, Zhu J, Bu D, Wang X, Pan Y, Liu Y, Wang P. Inhibition of hydrogen sulfide synthesis reverses acquired resistance to 5-FU through miR-215-5p-EREG/TYMS axis in colon cancer cells. Cancer Lett. 2019;466:49–60. doi: 10.1016/j.canlet.2019.09.006. [DOI] [PubMed] [Google Scholar]
- 587.Cai WJ, Wang MJ, Ju LH, Wang C, Zhu YC. Hydrogen sulfide induces human colon cancer cell proliferation: Role of Akt, ERK and p21. Cell Biol Int. 2010;34:565–572. doi: 10.1042/CBI20090368. [DOI] [PubMed] [Google Scholar]
- 588.Hale VL, Jeraldo P, Mundy M, Yao J, Keeney G, Scott N, Cheek EH, Davidson J, Greene M, Martinez C, et al. Synthesis of multi-omic data and community metabolic models reveals insights into the role of hydrogen sulfide in colon cancer. Methods. 2018;149:59–68. doi: 10.1016/j.ymeth.2018.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 589.Hong M, Tang X, He K. Effect of hydrogen sulfide on human colon cancer SW480 cell proliferation and migration in vitro. Nan Fang Yi Ke Da Xue Xue Bao. 2014;34:699–703. (In Chinese) [PubMed] [Google Scholar]
- 590.Kodela R, Nath N, Chattopadhyay M, Nesbitt DE, Velázquez-Martínez CA, Kashfi K. Hydrogen sulfide-releasing naproxen suppresses colon cancer cell growth and inhibits NF-κB signaling. Drug Design Dev Ther. 2015;9:4873–4882. doi: 10.2147/DDDT.S91116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 591.Chen S, Wang P, Liu Y. Research progress of the association of hydrogen sulfide with colorectal cancer and its associated anti-tumor drugs. Zhonghua Wei Chang Wai Ke Za Zhi. 2017;20:834–840. (In Chinese) [PubMed] [Google Scholar]
- 592.Elsheikh W, Blackler RW, Flannigan KL, Wallace JL. Enhanced chemopreventive effects of a hydrogen sulfide-releasing anti-inflammatory drug (ATB-346) in experimental colorectal cancer. Nitric Oxide. 2014;41:131–137. doi: 10.1016/j.niox.2014.04.006. [DOI] [PubMed] [Google Scholar]
- 593.Faris P, Ferulli F, Vismara M, Tanzi M, Negri S, Rumolo A, Lefkimmiatis K, Maestri M, Shekha M, Pedrazzoli P, et al. Hydrogen Sulfide-evoked intracellular Ca2+ signals in primary cultures of metastatic colorectal cancer cells. Cancers (Basel) 2020;12:3338. doi: 10.3390/cancers12113338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 594.Malagrinò F, Zuhra K, Mascolo L, Mastronicola D, Vicente JB, Forte E, Giuffrè A. Hydrogen sulfide oxidation: Adaptive changes in mitochondria of SW480 colorectal cancer cells upon exposure to hypoxia. Oxid Med Cell Longev. 2019;2019:8102936. doi: 10.1155/2019/8102936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 595.Sakuma S, Minamino S, Takase M, Ishiyama Y, Hosokura H, Kohda T, Ikeda Y, Fujimoto Y. Hydrogen sulfide donor GYY4137 suppresses proliferation of human colorectal cancer Caco-2 cells by inducing both cell cycle arrest and cell death. Heliyon. 2019;5:e02244. doi: 10.1016/j.heliyon.2019.e02244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 596.Liu H, Chang J, Zhao Z, Li Y, Hou J. Effects of exogenous hydrogen sulfide on the proliferation and invasion of human Bladder cancer cells. J Cancer Res Ther. 2017;13:829–832. doi: 10.4103/jcrt.JCRT_423_17. [DOI] [PubMed] [Google Scholar]
- 597.Liu M, Wu L, Montaut S, Yang G. Hydrogen sulfide signaling axis as a target for prostate cancer therapeutics. Prostate Cancer. 2016;2016:8108549. doi: 10.1155/2016/8108549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 598.Zhang S, Bian H, Li X, Wu H, Bi Q, Yan Y, Wang Y. Hydrogen sulfide promotes cell proliferation of oral cancer through activation of the COX2/AKT/ERK1/2 axis. Oncol Rep. 2016;35:2825–2832. doi: 10.3892/or.2016.4691. [DOI] [PubMed] [Google Scholar]
- 599.Zhuang L, Li K, Wang G, Shou T, Gao C, Mao Y, Bao M, Zhao M. Preconditioning with hydrogen sulfide prevents bone cancer pain in rats through a proliferator-activated receptor gamma/p38/Jun N-terminal kinase pathway. Exp Biol Med (Maywood) 2018;243:57–65. doi: 10.1177/1535370217740859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 600.Xu Y, Ma N, Wei P, Zeng Z, Meng J. Expression of hydrogen sulfide synthases and Hh signaling pathway components correlate with the clinicopathological characteristics of papillary thyroid cancer patients. Int J Clin Exp Pathol. 2018;11:1818–1824. [PMC free article] [PubMed] [Google Scholar]
- 601.Huang CC, Ho CH, Chen YC, Hsu CC, Lin HJ, Tian YF, Wang JJ, Guo HR. Impact of carbon monoxide poisoning on the risk of breast cancer. Sci Rep. 2020;10:20450. doi: 10.1038/s41598-020-77371-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 602.Kawahara B, Moller T, Hu-Moore K, Carrington S, Faull KF, Sen S, Mascharak PK. Attenuation of antioxidant capacity in human breast cancer cells by carbon monoxide through inhibition of cystathionine β-synthase activity: Implications in chemotherapeutic drug sensitivity. J Med Chem. 2017;60:8000–8010. doi: 10.1021/acs.jmedchem.7b00476. [DOI] [PubMed] [Google Scholar]
- 603.Kim DH, Yoon HJ, Cha YN, Surh YJ. Role of heme oxygenase-1 and its reaction product, carbon monoxide, in manifestation of breast cancer stem cell-like properties: Notch-1 as a putative target. Free Radical Res. 2018;52:1336–1347. doi: 10.1080/10715762.2018.1473571. [DOI] [PubMed] [Google Scholar]
- 604.Kourti M, Westwell A, Jiang W, Cai J. Repurposing old carbon monoxide-releasing molecules towards the anti-angiogenic therapy of triple-negative breast cancer. Oncotarget. 2019;10:1132–1148. doi: 10.18632/oncotarget.26638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 605.Lee WY, Chen YC, Shih CM, Lin CM, Cheng CH, Chen KC, Lin CW. The induction of heme oxygenase-1 suppresses heat shock protein 90 and the proliferation of human breast cancer cells through its byproduct carbon monoxide. Toxicol Appl Pharmacol. 2014;274:55–62. doi: 10.1016/j.taap.2013.10.027. [DOI] [PubMed] [Google Scholar]
- 606.Kawahara B, Gao L, Cohn W, Whitelegge JP, Sen S, Janzen C, Mascharak PK. Diminished viability of human ovarian cancer cells by antigen-specific delivery of carbon monoxide with a family of photoactivatable antibody-photoCORM conjugates. Chem Sci. 2020;11:467–473. doi: 10.1039/C9SC03166A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 607.Kawahara B, Ramadoss S, Chaudhuri G, Janzen C, Sen S, Mascharak PK. Carbon monoxide sensitizes cisplatin-resistant ovarian cancer cell lines toward cisplatin via attenuation of levels of glutathione and nuclear metallothionein. J Inorganic Biochem. 2019;191:29–39. doi: 10.1016/j.jinorgbio.2018.11.003. [DOI] [PubMed] [Google Scholar]
- 608.Khasag N, Sakiyama S, Toba H, Yoshida M, Nakagawa Y, Takizawa H, Kawakami Y, Kenzaki K, Ali AH, Kondo K, Tangoku A. Monitoring of exhaled carbon monoxide and carbon dioxide during lung cancer operation. Eur J Cardiothorac Surg. 2014;45:531–536. doi: 10.1093/ejcts/ezt395. [DOI] [PubMed] [Google Scholar]
- 609.Liptay MJ, Basu S, Hoaglin MC, Freedman N, Faber LP, Warren WH, Hammoud ZT, Kim AW. Diffusion lung capacity for carbon monoxide (DLCO) is an independent prognostic factor for long-term survival after curative lung resection for cancer. J Surg Oncol. 2009;100:703–707. doi: 10.1002/jso.21407. [DOI] [PubMed] [Google Scholar]
- 610.Nemeth Z, Csizmadia E, Vikstrom L, Li M, Bisht K, Feizi A, Otterbein S, Zuckerbraun B, Costa DB, Pandolfi PP, et al. Alterations of tumor microenvironment by carbon monoxide impedes lung cancer growth. Oncotarget. 2016;7:23919–23932. doi: 10.18632/oncotarget.8081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 611.Shao L, Gu YY, Jiang CH, Liu CY, Lv LP, Liu JN, Zou Y. Carbon monoxide releasing molecule-2 suppresses proliferation, migration, invasion, and promotes apoptosis in non-small cell lung cancer Calu-3 cells. Eur Rev Med Pharmacol Sci. 2018;22:1948–1957. doi: 10.26355/eurrev_201804_14720. [DOI] [PubMed] [Google Scholar]
- 612.Shirabe T, Mawatari S, Kuroiwa Y. Autopsy case of carbon monoxide poisoning at the Miike Coal Mine explosion-a case of lung cancer with the fatal outcome in 3 years and 4 months. Shinkei Kenkyu No Shimpo. 1970;14:321–326. [PubMed] [Google Scholar]
- 613.Zarogoulidis P, Kerenidi T, Huang H, Kontakiotis T, Tremma O, Porpodis K, Kalianos A, Rapti A, Foroulis C, Zissimopoulos A, et al. Six minute walking test and carbon monoxide diffusing capacity for non-small cell lung cancer: Easy performed tests in every day practice. J Thorac Dis. 2012;4:569–576. doi: 10.3978/j.issn.2072-1439.2012.08.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 614.Lian S, Xia Y, Ung TT, Khoi PN, Yoon HJ, Kim NH, Kim KK, Jung YD. Carbon monoxide releasing molecule-2 ameliorates IL-1β-induced IL-8 in human gastric cancer cells. Toxicology. 2016:361–362. 24–38. doi: 10.1016/j.tox.2016.07.003. [DOI] [PubMed] [Google Scholar]
- 615.Lv C, Su Q, Fang J, Yin H. Styrene-maleic acid copolymer-encapsulated carbon monoxide releasing molecule-2 (SMA/CORM-2) suppresses proliferation, migration and invasion of colorectal cancer cells in vitro and in vivo. Biochem Biophys Res Commun. 2019;520:320–326. doi: 10.1016/j.bbrc.2019.09.112. [DOI] [PubMed] [Google Scholar]
- 616.Yin H, Fang J, Liao L, Maeda H, Su Q. Upregulation of heme oxygenase-1 in colorectal cancer patients with increased circulation carbon monoxide levels, potentially affects chemotherapeutic sensitivity. BMC Cancer. 2014;14:436. doi: 10.1186/1471-2407-14-436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 617.Nitta S, Kawai K, Nagumo Y, Ikeda A, Kandori S, Kojima T, Nishiyama H. Impact of hemoglobin levels on hemoglobin-adjusted carbon monoxide diffusion capacity after chemotherapy for testicular cancer. Jpn J Clin Oncol. 2019;49:1151–1156. doi: 10.1093/jjco/hyz103. [DOI] [PubMed] [Google Scholar]
- 618.van Osch FHM, Pauwels CGGM, Jochems SHJ, Fayokun R, James ND, Wallace DMA, Cheng KK, Bryan RT, van Schooten FJ, Zeegers MP. Tar, nicotine and carbon monoxide yield of UK cigarettes and the risk of non-muscle-invasive and muscle-invasive bladder cancer. Eur J Cancer Prev. 2019;28:40–44. doi: 10.1097/CEJ.0000000000000404. [DOI] [PubMed] [Google Scholar]
- 619.Vítek L, Gbelcová H, Muchová L, Váňová K, Zelenka J, Koníčková R, Suk J, Zadinova M, Knejzlík Z, Ahmad S, et al. Antiproliferative effects of carbon monoxide on pancreatic cancer. Dig Liver Dis. 2014;46:369–375. doi: 10.1016/j.dld.2013.12.007. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Not applicable.