Abstract
The distribution of culturable hyperthermophiles was studied in relation to environmental conditions in the Kubiki oil reservoir in Japan, where the temperature was between 50 and 58°C. Dominant hyperthermophilic cocci and rods were isolated and shown to belong to the genera Thermococcus and Thermotoga, respectively, by 16S rDNA analyses. Using the most-probable-number method, we found that hyperthermophilic cocci were widely distributed in several unconnected fault blocks in the Kubiki oil reservoir. In 1996 to 1997, their populations in the production waters from oil wells were 9.2 × 103 to 4.6 × 104 cells/ml, or 10 to 42% of total cocci. On the other hand, hyperthermophilic rods were found in only one fault block of the reservoir with populations less than 10 cells/ml. Dominant Thermococcus and Thermotoga spp. grew at reservoir temperatures and utilized amino acids and sugars, respectively, as sole carbon sources. While organic carbon was plentiful in the environment, these hyperthermophiles were unable to grow in the formation water due to lack of essential nutrients. Concentrations of some organic and inorganic substances differed among fault blocks, indicating that the movement of formation water between fault blocks was restricted. This finding suggests that the supply of nutrients via fluid current is limited in this subterranean environment and that the organisms are starved in the oil reservoir. Under starved conditions at 50°C, culturable cells of Thermococcus sp. remained around the initial cell density for about 200 days, while those of Thermotoga sp. decreased exponentially to 0.01% of the initial cell density after incubation for the same period. The difference in survivability between these two hyperthermophiles seems to reflect their populations in the fault blocks. These results indicate that hyperthermophilic cocci and rods adapt to the subterranean environment of the Kubiki oil reservoir by developing an ability to survive under starved conditions.
Many thermophilic bacteria with optimum growth temperatures from 45 to 80°C have been isolated from oil fields (4, 8, 9, 11, 12, 27, 40, 41, 43, 44). In 1993, hyperthermophiles which grew optimally over 80°C were found in the production fluids of North Sea and Alaskan oil reservoirs (48). However, it was pointed out that these organisms might have been introduced into the oil reservoir during drilling or enhanced oil recovery with water injection, because their characteristics are similar to those of organisms found in hydrothermal fields (42). The hyperthermophilic archaea Thermococcus litoralis and Archaeoglobus fulgidus were isolated from oil wells in the East Paris Basin where no water had been injected (33). While heterotrophic microorganisms have been studied in 37 crude oil reservoirs at temperatures between 37 and 148°C, hyperthermophiles have been detected only in the North Sea oil reservoir (16).
In the North Sea oil reservoir, sulfate reducers in different fault blocks were different, though no correlation was found between the seawater injection and their population densities in the production water (39). Flora of hyperthermophiles at several wells of Thistle offshore were similar (48). However, their distribution in relation to the environmental conditions has not been studied.
The ecological system of these hyperthermophiles under the ground is still unknown. We studied the distribution of hyperthermophiles and the environmental conditions in an oil reservoir in Japan. Their physiological growth characteristics, including nutritional requirements and survivability in the formation water, were also studied. Their strategies for living in the environment of the oil reservoir are discussed.
MATERIALS AND METHODS
Kubiki oil reservoir.
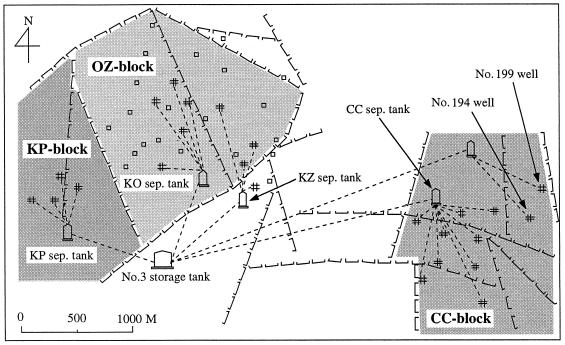
The Kubiki oil reservoir is located near the coast of the Sea of Japan in Niigata prefecture, Japan. This reservoir has many faults and is divided into many unconnected fault blocks (49) (Fig. 1). The depths of oil wells were between 1,000 and 1,200 m, with in situ temperatures between 50 and 58°C. The average in situ pressure was 6 MPa. This oil reservoir consists of three major fault blocks, the KP, OZ, and CC blocks, which are subdivided into smaller blocks (Fig. 1). In the OZ block, 650,000 kl of seawater had been injected into some wells to enhance oil recovery from 1977 to 1994 (Fig. 1). Wells in the KP and CC blocks have not been injected with seawater.
FIG. 1.
Map of studied area and sampling points in Kubiki oil reservoir. ⊔⊔⊔, fault line; #, production well; □, well injected with seawater; ----, production tubing; sep., separation.
Samples of formation waters.
To study the microbial community in formation water, production fluids were collected from the no. 3 storage tank (Fig. 1), where production fluids from all oil wells are stored for approximately 24 h. To determine the microbial distribution, we collected six production fluids from the drains of four separation tanks and two wellheads (Fig. 1). Formation water was collected in sterile glass bottles, which were then sealed with sterile silicon stoppers and plastic screw caps. Samples were chilled in a cooler box and transferred to the laboratory.
Culture conditions of the hyperthermophiles.
All culture media for hyperthermophiles (Table 1) were based on artificial seawater (ASW) (26) and supplemented, with 10 ml per liter of a Wolfe trace mineral solution (51), 10 ml of a vitamin mix solution (2), and 60 mg of resazurin. HC and HA media were flushed with an oxygen-free mixture of hydrogen and carbon dioxide (80%:20%) instead of oxygen-free nitrogen. Gas phase of the AY medium was air.
TABLE 1.
Media used for detection of hyperthermophiles from the Kubiki oil reservoir
| Mediuma | Gas phase | Carbon source |
|---|---|---|
| HC | H2:CO2 (4:1,200 kPa) | Free |
| HA | H2:CO2 (4:1,200 kPa) | Acetate (0.1%) + lactate (0.1%) |
| AY | Air (100 kPa) | Yeast extract (0.2%) |
| YE | N2 (100 kPa) | Yeast extract (0.2%) |
| AA | N2 (100 kPa) | 20 amino acidsb (total, 0.2%) |
| GS | N2 (100 kPa) | Glucose (0.1%) + starch (0.1%) |
| AL | N2 (100 kPa) | Acetate (0.1%) + lactate (0.1%) |
| CO | N2 (100 kPa) | Crude oilc (10% [vol/vol]) |
Media based on artificial seawater were incubated for 1 to 7 days at 85°C in a dry oven.
The amino acids Gly, Ala, Ser, Thr, Cys, Asn, Gln, Leu, Ile, Val, Met, Phe, Tyr, Trp, Pro, Asp, Glu, His, Lys, and Arg were added at a final concentration of 0.01% (wt/vol) each.
Crude oil was collected from the no. 3 storage tank and autoclaved at 121°C for 30 min.
Isolation and cultivation of the hyperthermophiles.
One milliliter of the formation water was inoculated into 9 ml of culture medium in a Hungate tube and incubated at 85°C for 1 to 7 days. When the hyperthermophiles grew, the cell suspension was transferred to fresh medium.
The hyperthermophiles were isolated on Gelrite plates. Gelrite (Kelco Division of Merck & Co., Rahway, N.J.) was dissolved at 0.8% (wt/vol) in ASW medium containing 0.6% (wt/vol) yeast extract. After autoclaving at 121°C for 20 min, the solution was dispensed into glass petri dishes in an anaerobic glove box (ANX-1; Hirasawa, Tokyo, Japan) and solidified at room temperature. In the glove box, 10 to 100 μl of the enrichment culture for hyperthermophiles was placed on the plates. Plates were then placed in a Gas Pack jar (AJ9023; MART Jar Systems, Lichtenvoorde, The Netherlands) and incubated at 85°C for 1 to 7 days. This procedure was repeated at least three times.
Electron microscopy.
The isolates were fixed with 2.5% (vol/vol) glutaraldehyde for 20 min at room temperature and stained with 4% (wt/vol) uranyl acetate as described by Kurr et al. (32). They were observed in a transmission electron microscope (H-7000; Hitachi, Tokyo, Japan) operated at 75 kV.
Growth characteristics of the isolates.
Effects of temperature, pH, and NaCl on microbial growth were examined on YE-based medium. The temperature was controlled with a mineral oil bath (OH-16; Taitec, Saitama, Japan). Buffers for the pH experiments are described elsewhere (19). The cultures were incubated at the optimum growth temperatures for each isolate.
Nutritional growth requirements of the isolates.
To determine nutritional requirements, the cultures were incubated at 80°C unless otherwise stated. Growth on various carbon sources was examined in 10 ml of ASW medium (pH 7.0) containing 0.1% (wt/vol) yeast extract, peptone, casein, 20 amino acids, glucose, sucrose, maltose, starch, acetate, lactate, formate, propionate, or pyruvate without shaking. Growth on hydrocarbons was also examined in the presence of 10% (vol/vol) kerosene, light oil, or A-heavy oil (Nisseki Oil Co., Tokyo, Japan) in 30 ml of ASW medium (pH 7.0) with shaking.
The amino acid requirements of the isolates were determined as described previously (19, 21, 26).
Metabolic products of the isolates.
After growth of the isolates in YE medium, low-molecular-weight (C2 to C5) organic acids in the culture were analyzed on a high-pressure liquid chromatograph (HPLC; Hitachi L-6000) equipped with a UV detector at 210 nm. Production of H2 and CO2 was assessed by gas chromatography (Hitachi 263-30 chromatograph with a thermal conductivity detector). Hydrogen gas was analyzed with a molecular sieve 13X-S column (3-mm inside diameter by 2 m) with N2 as the carrier gas, at a flow rate of 30 ml/min (injection temperature, 82°C; column temperature, 42°C). Carbon dioxide was analyzed by using a WG-100 column (0.25 in. [outside diameter] by 1.8 m) with He as the carrier gas, at a flow rate of 40 ml/min (injection temperature, 50°C; column temperature, 50°C).
Sulfur requirements of the isolates.
One gram of steam-autoclaved elemental sulfur was added to 100 ml of YE medium in 125-ml bottles. Cultures of the isolates, with or without elemental sulfur, were incubated with shaking at the optimum growth temperature. Hydrogen sulfide production was analyzed with a gas analysis kit (Gastec, Kanagawa, Japan).
Sensitivities of the isolates to antibiotics.
Growth of the isolates was examined after incubation at 70°C for 7 days in the presence of 100 μg of rifampin, streptomycin, vancomycin, or chloramphenicol per milliliter.
Isolation of DNA.
Genomic DNA was extracted from the isolate by using a procedure described elsewhere (1), with slight modification. RNA was digested with DNase-free RNase (20 μg/ml) at 37°C for 1 h after extraction with chloroform-isoamyl alcohol.
DNA base composition (GC content).
Nucleotide composition was determined with an HPLC equipped with an UV detector (UV 8010; Tosoh, Tokyo, Japan) at 270 nm with a Develosil ODS-HG-5 column (4.6 by 250 mm) after digestion of DNA with nuclease P1 (53).
16S rDNA analysis.
PCR was used to amplify 16S rDNA from the genomic DNA of the isolates by using a pair of primers (A3R [Escherichia coli positions 518 to 536, Thermococcus celer positions 461 to 479; 5′CAGCCGCCGCGGTAATACC3′] and A8 [E. coli positions 1387 to 1406, T. celer positions 1337 to 1356; 5′GACGGGCGGTGTGTGCAAGG3′]) for archaea and another pair of primers (B0R [E. coli positions 8 to 27; 5′AGAGTTTGATCCTGGCTCAG3′] and B9 [E. coli positions 1491 to 1512; 5′TACGGCTACCTTGTTACGACTT3′]) for bacteria. Amplification consisted of 35 cycles of 1 min at 94°C, 1 min at 58°C, and 2 min at 72°C. After purification of the PCR products with a QIAEX II gel extraction kit (Qiagen, Hilden, Germany), the 16S rDNA sequences were determined with an ABI Big Dye Terminator cycle sequencing ready reaction kit (Perkin-Elmer Biosystems, Norwalk, Conn.) and an ABI 377 DNA sequencer (Perkin-Elmer). The determined sequences were aligned with the reported 16S rRNA gene sequences collected from DDBJ (DNA data bank of Japan), and the sequence similarities were determined with GENETYX-MAC 8.0.
Chemical analysis of formation water.
Prior to chemical analysis, the samples were passed through a sterile filter (Disk Capsule, 0.2-μm pore size; Fuji, Tokyo, Japan). Concentrations of Cl− and SO42− were analyzed with an HPLC equipped with a TSKgel IC-Anion-PW (4.6-mm [inside diameter] by 5 cm) column, using 4 mM potassium phosphate buffer (pH 9.1) as an elution buffer, at a flow rate of 1.0 ml/min at 40°C. They were detected with a conductivity detector (Tosoh CM 8010). Concentrations of Na+ and Mg2+ in the samples were measured with an inductively coupled sequential plasma spectrometer (model ICPS-4000; Shimadzu, Kyoto, Japan). Dissolved organic carbon (DOC) was analyzed with a total organic carbon analyzer (Shimadzu TOC 5000). Amino acids were analyzed with a reverse-phase HPLC equipped with an AccQ-Tag column (Nihon Millipore Kogyo, Tokyo, Japan) and a fluorescence detector (Tosoh FS8010). The reducing sugars were analyzed by a modified fluorescence HPLC method based on the postcolumn reaction with benzamidine (28).
Estimation of total cell density.
Total cell density of microorganisms was determined by the acridine orange direct count method (22). Cells were fixed with 1% (vol/vol) glutaraldehyde, stained with 0.05% acridine orange (vol/vol), and then filtrated through polycarbonate filters (Nuclepore Track-Etch membrane; 0.2-μm pore size; Corning Costar, Boston, Mass.). The filter was observed under an epifluorescence microscope (BX60; Olympus, Tokyo, Japan).
Estimation of culturable cell density.
Culturable cell density was determined by the three-tube most-probable-number method (10). Hyperthermophilic cocci were detected by using YE medium containing vancomycin (100 μg/ml) at 85°C; for hyperthermophilic rods, we used GC medium at 80°C. Growth was monitored by turbidity of the cultures. In this study, cells that grew in YE medium at 45 and 20°C were defined as anaerobic heterotrophic thermophiles and mesophiles, respectively.
Growth ability of isolates in the Kubiki oil reservoir.
Growth ability of the isolates in the oil reservoir was estimated by using KFW medium containing the formation water and crude oil from the Kubiki oil reservoir. Filtrate obtained after passage through a membrane filter (Fuji Disk Capsule; 0.2 μm pore size) (90 ml) was added, along with sterile resazurin (60 mg/liter) and 10 ml of autoclaved crude oil, into sterile 125-ml vials. These vials were flushed with oxygen-free nitrogen, and sterile Na2S solution was added at a final concentration of 1 mM. Limitation of the nutrients in the formation water was examined with KFW medium containing 20 amino acids (0.1% each amino acid) or 2% sucrose. The isolates were inoculated into the vials with the initial cell density at approximately 105 cells/ml. They were incubated at their optimum growth temperatures for 10 days, and growth was monitored by the acridine orange direct count method (22).
Survivability of isolates in the Kubiki oil reservoir.
To estimate survivability in the Kubiki oil reservoir, the isolates were inoculated in KFW medium at an initial cell density of approximately 105 cells/ml. After incubation at 50°C for about 200 days, cell densities were monitored by the three-tube most-probable-number method.
Nucleotide sequence accession numbers.
The 16S rDNA sequences of strains CKU-1 and RKU-1 were registered at the DDBJ under accession no. AB027014 and AB027016, respectively. The sequence of strain CKU-199 (isolated from the no. 199 well) was registered at the DDBJ under accession no. AB027015.
RESULTS
Isolation of hyperthermophiles.
Two types of hyperthermophiles were isolated from the Kubiki oil reservoir after cultivation at 85°C. Cocci grew in both YE and AA media. Rods with togas at both ends grew in YE and GS media. No hyperthermophiles grew in HC, HA, AY, AL, or CO media.
Eleven cocci and six rods were isolated from the enrichment cultures which had been inoculated with the sample from the no. 3 storage tank. The partial 16S rDNA sequences were determined for these isolates. Homologies between the 16S rDNA sequences (455 bp) of the isolated 11 cocci and that of T. celer were 98.5 to 99.1%, which indicated that they belonged to the genus Thermococcus. Homologies between the 16S rDNA sequences (440 bp) of the isolated six rods and that of Thermotoga maritima were 98.6 to 99.3%, which indicated that they belonged to the genus Thermotoga.
Characteristics of isolated coccus and rod.
Strain CKU-1 was one of the 11 cocci isolated from the formation water of the Kubiki oil reservoir, and strain RKU-1 was one of the six rods (Fig. 2; Table 2). Identity of the 16S rDNA sequence (857 bp) between strain CKU-1 and T. celer was 98.7%. Identity of the 16S rDNA sequence (1482 bp) between strain RKU-1 and Thermotoga maritima was 99.3%.
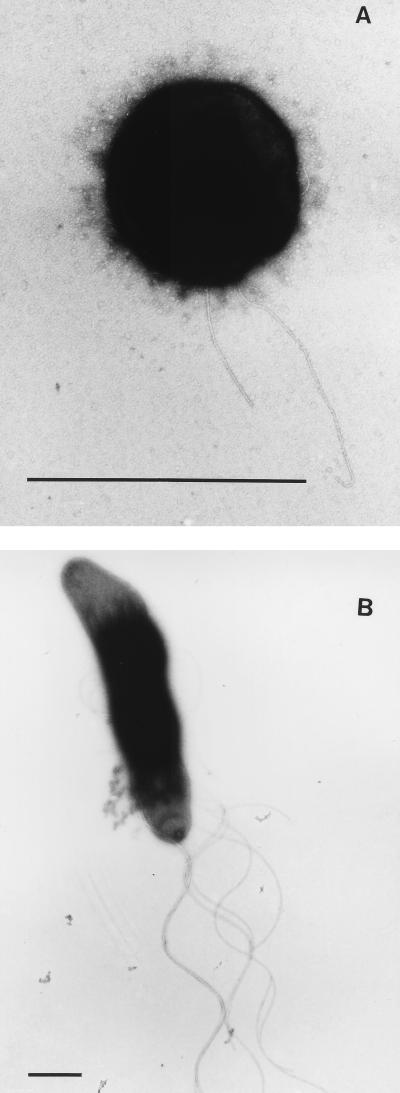
FIG. 2.
Transmission electron micrographs of strains CKU-1 (A) and RKU-1 (B); negative staining with 4% uranyl acetate. Bars, 1 μm.
TABLE 2.
Characteristics of strains CKU-1 and RKU-1
| Strain | Diam (μm) | Flagella | Temp (°C)
|
pH
|
NaCl concn (%)
|
Doubling time (h) at:
|
||||
|---|---|---|---|---|---|---|---|---|---|---|
| Range | Optimum | Range | Optimum | Range | Optimum | Optimum temp | In situ temp (52°C) | |||
| CKU-1 | 0.5–3.0 | + | 46–95 | 85 | 5.2–9.0 | 6.0 | 0.1–6.0 | 2.0 | 0.6 | 11.4 |
| RKU-1 | 2.0–7.0 × 0.7–1.0 | + | 47–88 | 80 | 5.2–9.0 | 7.0 | 0.1–5.5 | 1.0 | 0.9 | 22.1 |
| Substrates used | Metabolites | Growth response to elemental sulfur | Antibiotic sensitivitya | G+C content (%) |
|---|---|---|---|---|
| Yeast extract, proteinaceous substrates,b amino acids | Acetate, lactate, H2, CO2 | No effect | R | 37.8 |
| Yeast extract, peptone, sugars,c cellulose | Acetate, lactate, H2, CO2 | Inhibit | R, S, V, C | 46.8 |
R, rifampin; S, streptomycin; V, vancomycin; C, chloramphenicol.
Peptone or casein.
Glucose, sucrose, maltose, or starch.
Formation waters in the fault blocks.
To estimate the movement of formation water in the reservoir, concentrations of organic and inorganic components in the formation waters from several fault blocks were analyzed (Table 3). All samples were collected from the same oil layer, but the oil wells in the KP block were a little deeper than those in the other blocks (Table 3). The in situ temperature was between 50 and 58°C and increased with depth. The pHs in all of the formation waters were about 8. Concentrations of DOC were between 36 and 755 mg/liter and differed among the samples even in the same fault block. Concentrations of Cl−, Na+, Mg2+, and SO42− in the OZ block were higher than those in the KP block. The higher concentrations of these ions in the OZ block were possibly caused by seawater injection. Concentrations of these ions in KP block were similar to those in the CC block.
TABLE 3.
Properties of formation waters in fault blocks
| Source | Sampling site | Depth (m) | Temp (°C) | pH | DOC (mg/liter) | Cl− (g/liter) | Na+ (g/liter) | Mg2+ (mg/liter) | SO42− (mg/liter) |
|---|---|---|---|---|---|---|---|---|---|
| Formation water from: | |||||||||
| KP block | KP separation tank | 1,210–1,245 | 57–58 | 7.9 | 43 | 7.8 | 3.1 | 9 | NDb |
| OZ block | KO separation tank | 1,022–1,126 | 51–54 | 7.9 | 112 | 12.0 | 4.6 | 137 | 161 |
| KZ separation tank | 1,004–1,094 | 50–53 | 7.9 | 527 | 11.4 | 4.2 | 28 | ND | |
| CC block | CC separation tank | 1,048–1,118 | 51–54 | 8.1 | 96 | 7.5 | 3.0 | 7 | ND |
| No. 194 well | 1,080 | 53 | 7.9 | 755 | 7.0 | 2.9 | 15 | ND | |
| No. 199 well | 1,051 | 52 | 7.9 | 36 | 5.4 | 2.1 | 7 | ND | |
| Seawatera | 8.0 | 19.4 | 10.8 | 1,300 | 2,700 |
Data from reference 30.
ND, not detected (<1 mg/liter).
Population change of microorganisms in the Kubiki oil reservoir.
Total cell densities and culturable cell densities of hyperthermophilic, thermophilic, and mesophilic anaerobic heterotrophs in the formation water of the no. 3 storage tank were investigated from June 1996 to September 1997 (Table 4). In the Kubiki oil reservoir, only cocci were observed after incubation at 85°C in YE medium containing 100 μg of vancomycin per ml. Only rods with togas were observed after incubation in GS medium at 80°C. No coccus was observed after incubation in YE medium at 45 or 20°C. Total cell densities of cocci and rods were 8.0 × 104 to 2.0 × 105 cells/ml and 6.5 × 105 to 2.5 × 106 cells/ml, respectively in this period. Culturable cell densities of hyperthermophilic cocci, hyperthermophilic rods, thermophiles, and mesophiles did not change much and were 9.2 × 103 to 4.6 × 104, <0.4 × 100 to 9.3 × 100, 2.4 × 104 to 1.1 × 105, and 1.1 × 104 to 4.0 × 104 cells/ml, respectively in this study period. Culturable cell densities of hyperthermophilic cocci were 10 to 42% of total cocci. Culturable cell densities of rods, including hyperthermophilic rods, thermophiles, and mesophiles, were 1.8 to 9.9% of total rods.
TABLE 4.
Total and culturable cell densities in formation waters of no. 3 storage tank
| Time of collection | Total cell density (cells/ml)
|
Culturable cell density (cells/ml)
|
||||
|---|---|---|---|---|---|---|
| Cocci | Rods | Hyperthermophilic cocci | Hyperthermophilic rods | Thermophiles | Mesophiles | |
| June 1996 | 8.0 × 104 | 2.5 × 106 | 9.2 × 103 | NDa | 3.5 × 104 | 1.1 × 104 |
| November 1996 | 2.0 × 105 | 1.5 × 106 | 2.4 × 104 | 9.3 × 100 | 1.1 × 105 | 2.1 × 104 |
| March 1997 | 1.1 × 105 | 6.5 × 105 | 4.6 × 104 | ND | 2.4 × 104 | 4.0 × 104 |
| September 1997 | 9.0 × 104 | 1.7 × 106 | 9.2 × 103 | 0.4 × 100 | 4.6 × 104 | 2.4 × 104 |
| Avg | 1.2 × 105 | 3.1 × 106 | 2.2 × 104 | 2.4 × 100 | 5.4 × 104 | 2.4 × 104 |
ND, not detected (<0.4 × 100 cells/ml).
Distribution of hyperthermophiles and other anaerobic heterotrophs.
Hyperthermophilic cocci were found in all of the blocks examined (Table 5). Their culturable cell densities were 1.5 × 100 to 1.1 × 105 cells/ml. Hyperthermophilic rods were detected only in the KP block, with culturable cell density of 1.5 × 100 cells/ml (Table 5). Anaerobic heterotrophic thermophiles and mesophiles were found in all samples (Table 5). Average culturable cell densities (9.1 × 104 cells/ml) of thermophiles in all samples were about the same as those in the no. 3 storage tank (5.4 × 104 cells/ml [Table 4]). Culturable cell densities of mesophiles in the production fluid collected at wellheads were much lower than those in separation tanks.
TABLE 5.
Total and culturable cell densities in formation waters in fault blocks
| Fault block | Sampling site | Total cell density (cells/ml)
|
Culturable cell density (cells/ml)
|
||||
|---|---|---|---|---|---|---|---|
| Cocci | Rods | Hyperthermophilic cocci | Hyperthermophilic rods | Thermophiles | Mesophiles | ||
| KP | KP separation tank | 4.1 × 104 | 1.1 × 105 | 1.5 × 100 | 1.5 × 100 | 4.8 × 103 | 1.3 × 103 |
| OZ | KO separation tank | 1.7 × 105 | 1.7 × 106 | 1.1 × 105 | NDa | 4.6 × 105 | 4.6 × 104 |
| KZ separation tank | 1.7 × 105 | 5.0 × 105 | 4.6 × 103 | ND | 4.6 × 104 | 3.3 × 102 | |
| CC | CC separation tank | 3.8 × 105 | 9.5 × 105 | 2.4 × 104 | ND | 2.4 × 104 | 4.6 × 103 |
| no. 194 well | 1.3 × 105 | 2.1 × 106 | 1.5 × 101 | ND | 1.1 × 104 | 1.5 × 101 | |
| no. 199 well | 3.4 × 104 | 1.6 × 105 | 1.5 × 103 | ND | 2.4 × 103 | 9.3 × 100 | |
ND, not detected (<0.4 × 100 cells/ml).
Six dominant hyperthermophilic cocci were isolated from four separation tanks and two wellheads, respectively (Fig. 1). The 16S rDNA sequence (455 bp) of the strain isolated from the KO separation tank was identical to that of strain CKU-1. Those of other strains isolated from the KP, KZ, and CC separation tanks and no. 194 and 199 wells were identical to each other, and identity to that of strain CKU-1 was 99.3%.
One hyperthermophilic rod was also isolated from the KP separation tank. Its 16S rDNA sequence (440 bp) was identical to that of strain RKU-1.
Growth ability of the isolates in the Kubiki oil reservoir.
Strains CKU-1 and RKU-1 grew in YE medium at the in situ temperatures of the Kubiki oil reservoir (50 to 58°C). Their doubling times at 52°C were 11.4 and 22.1 h, respectively (Tables 2 and 3). Strain CKU-1 required yeast extract, proteinaceous substrates such as peptone or casein, or amino acids as sole carbon and energy sources (Table 2). Six amino acids (Leu, Met, Phe, Thr, Trp, and Tyr) were essential for the growth of strain CKU-1, which did not grow on sugars, organic acids, or hydrocarbons. On the other hand, strain RKU-1 required a sugar (glucose, sucrose, maltose, or starch) as sole carbon and energy source (Table 2). It also grew on yeast extract, peptone, or cellulose but did not grow on casein, amino acids, organic acids, or hydrocarbons.
The DOC in formation water from the no. 3 storage tank was 480 mg of C/liter. Concentrations of low-molecular-weight organic acids, total free amino acids, and reducing sugars as carbon were 3.5, 0.14, and less than 0.01 mg of C/liter, respectively. Some amino acids (Ala, Asn, Asp, Cys, Gln, Glu, Gly, Ile, Leu, Lys, Met, Phe, Pro, Ser, Thr, Tyr, and Val) were detected between 0.001 and 0.020 mg of C/liter. However, Arg, His, and Trp were not detected (<0.001 mg of C/liter). Most of other organic components in the formation water could not be identified.
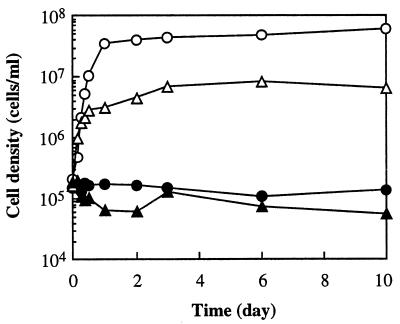
Strains CKU-1 and RKU-1 did not grow in KFW medium but did grow in the same medium supplemented with their essential nutrients, amino acids and sugars, respectively (Fig. 3). This indicated that they were unable to grow in in situ formation water due to lack of essential nutrients.
FIG. 3.
Growth curves of strains CKU-1 and RKU-1 in filtered sterile formation water with 10% (vol/vol) crude oil under anaerobic conditions (KFW medium) at their optimum temperatures. ▴, CKU-1 in KFW medium; ▵, CKU-1 in KFW medium containing 20 amino acids (each at 0.1 g/liter); ●, RKU-1 in KFW medium; ○, RKU-1 in KFW medium containing sucrose (1 g/liter).
Survivability of the isolates in the Kubiki oil reservoir.
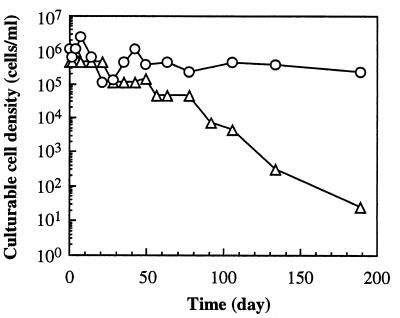
In the formation water at 50°C, the culturable cell density of strain CKU-1 fluctuated between 9.3 × 104 and 2.4 × 106 cells/ml within 50 days, but it did not change significantly for about 200 days (Fig. 4). On the other hand, the culturable cell density of strain RKU-1 decreased exponentially to 0.01% of the initial cell density after incubation for about 200 days (Fig. 4).
FIG. 4.
Survival of strains CKU-1 (○) and RKU-1 (▵) in KFW medium at 50°C.
DISCUSSION
Hyperthermophilic cocci and rods which utilized amino acids and sugars as sole carbon and energy sources, respectively, were isolated from the Kubiki oil reservoir, where the temperature was between 50 and 58°C. An unidentified hyperthermophile (48) and a thermophilic sulfate reducer (47) have been reported to degrade hydrocarbons in the oil reservoirs. Archaeoglobus spp. and many thermophilic sulfate reducers utilize organic acids in oil reservoirs (3, 4, 41, 44). However, the hyperthermophiles in the Kubiki oil reservoir did not utilize acetate, lactate, or crude oil as a sole carbon and energy source.
16S rDNA analyses of 17 hyperthermophilic cocci and rods isolated from the storage tank showed that they belonged to the genera Thermococcus and Thermotoga, respectively. The GC content of strain CKU-1 was similar to that of T. litoralis (5, 38). T. litoralis has been isolated not only from shallow submarine solfatara but also from the oil reservoirs (33, 48). However, strain CKU-1 was different from T. litoralis in some characteristics, such as possession of flagella, pyruvate utilization, and effect of elemental sulfur on the growth (Table 2). While the optimum growth temperature and GC content of strain RKU-1 were similar to those of Thermotoga maritima (23), the organism differed from T. maritima in some characteristics such as cellulose utilization, effect of elemental sulfur on growth, and sensitivity to rifampin (Table 3).
Because culturable cell densities of mesophiles in tanks were much higher than those at wellheads (Table 5), they seemed to grow in the tanks. On the other hand, hyperthermophiles were unable to grow at the temperature in the tanks. Culturable cell densities of hyperthermophilic cocci in the storage tank were much higher than those of hyperthermophilic rods. Moreover, culturable hyperthermophilic cocci in the storage tank in March 1997 comprised 42% of total cocci. This indicates that they were dominant in this reservoir.
Microbial distribution in the reservoir must be affected by the geological structure (39). In the Kubiki oil reservoir, geological investigation has shown that the oil layers between the OZ and CC blocks are completely separated by the faults (49). The difference of concentrations of organic and inorganic substances between the OZ block and the adjacent KP block indicated that the injected seawater did not move easily from one block to another through the faults. This indicates that even if microorganisms were introduced by injected seawater and grew in one block, it would be difficult for them to spread over the other blocks. Hyperthermophilic rods were found only in the KP block (Table 5). Their distribution might be restricted by unknown factors, or their densities in other fault blocks might be lower than the detection limit (<0.4 × 100 culturable cells/ml). Hyperthermophilic cocci were found in all fault blocks of the Kubiki oil reservoir, but their culturable cell densities varied from 100 to 105 cells/ml, even in the same blocks. Partial 16S rDNA sequences of the dominant hyperthermophilic cocci isolated from the three different fault blocks were almost identical. These results suggested that they were widely distributed throughout the separated fault blocks in the Kubiki oil reservoir.
It is not clear how these hyperthermophiles dwell in the oil reservoir at temperatures lower than their optima. The minimum growth temperatures of strains CKU-1 and RKU-1 were 46 and 47°C, respectively (Tables 2 and 3). None of the known Thermococcus strains (5, 7, 13, 14, 15, 19, 24, 29, 35, 36, 45, 52) except T. profundus, T. siculi, and T. barophilus (17, 31, 34) grow below 55°C. Hyperthermophilic Thermotoga species (T. maritima and T. neapolitana) did not grow below 55°C (6, 23, 25). The growth ability below 55°C may be important in the Kubiki oil reservoir.
Growth of strains CKU-1 and RKU-1 was restricted due to lack of essential nutrients in the formation water of the Kubiki oil reservoir (Fig. 3). While the cell densities of the S0-dependent heterotrophic hyperthermophiles in a shallow hydrothermal field in Kodakara-Jima, Japan, were high, concentrations of their essential nutrients, amino acids, were low (20). In these hydrothermal fields, water current may continuously supply nutrients to the microbial communities (18, 37). On the other hand, the flow rates of natural groundwater in a deep sedimentary basin were calculated to be 0.001 to 0.01 m per year (50). This indicates that the replenishment of organic carbons via fluid currents in the oil reservoir is likely to be much slower than that in the hydrothermal environment.
Under starved conditions, the culturable cell density of heterotrophic microorganisms generally decreases in a logarithmic fashion (46). While the culturable cell density of strain RKU-1 slowly decreased under starved conditions in the formation water of the Kubiki oil reservoir, that of strain CKU-1 was almost constant for about 200 days (Fig. 4). Survivability under starved conditions may be important for their existence in the oil reservoir because supplies of the essential nutrients are likely to be limited. The difference of survivability between these two isolates seems to reflect their cell densities in the oil reservoir.
Distribution and physiological characteristics of hyperthermophilic archaea and bacteria in the Kubiki oil reservoir indicate that these organisms adapt to the subterranean environment where temperatures are lower than the optimum. The existence of hyperthermophiles in the oil reservoir at low temperature and under starved conditions suggests that they are distributed widely in the ground.
ACKNOWLEDGMENTS
We thank Teikoku Oil Co., Ltd. for contribution of samples and information about the Kubiki oil reservoir. We are grateful to M. Kurihara and R. Aburaki for excellent technical assistance, to K. Kamino for analysis of amino acids, K. Adachi for analysis of reduced sugars, and to H. Taki for analysis of organic acids. B. K. Baillie is acknowledged for editing the English.
REFERENCES
- 1.Ausubel F M, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K, editors. Current protocols in molecular biology. New York, N.Y: Greene Publishing Associates and Wiley-Interscience; 1987. [Google Scholar]
- 2.Bazylinski D A, Wirsen C O, Jannasch H W. Microbial utilization of naturally occurring hydrocarbons at the Guaymas Basin hydrothermal vent site. Appl Environ Microbiol. 1989;55:2832–2836. doi: 10.1128/aem.55.11.2832-2836.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Beeder J, Nilsen R K, Rosnes J T, Torsvik T, Lien T. Archaeoglobus fulgidus isolated from hot North Sea oil field waters. Appl Environ Microbiol. 1994;60:1227–1231. doi: 10.1128/aem.60.4.1227-1231.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Beeder J, Torsivik T, Lien T. Thermodesulforhabdus morvegicus gen. nov. and sp. nov., a novel thermophilic sulfate-reducing bacterium from oil field water. Arch Microbiol. 1995;164:331–336. [PubMed] [Google Scholar]
- 5.Belkin C, Jannasch H W. A new extremely thermophilic, sulfur-reducing heterotrophic, marine bacterium. Arch Microbiol. 1985;141:181–186. [Google Scholar]
- 6.Belkin C, Wirsen C O, Jannasch H W. A new sulfur-reducing, extremely thermophilic eubacterium from a submarine thermal vent. Appl Environ Microbiol. 1986;51:1180–1185. doi: 10.1128/aem.51.6.1180-1185.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Canganella F, Jones W J, Gambacorta A, Antranikian G. Thermococcus guaymasiensis sp. nov. and Thermococcus aggregans sp. nov., two novel thermophilic archaea isolated from the Guaymas Basin hydrothermal vent site. Int J Syst Bacteriol. 1998;48:1181–1185. doi: 10.1099/00207713-48-4-1181. [DOI] [PubMed] [Google Scholar]
- 8.Cayol J-L, Ollivier B, Patel B K C, Ravot G, Magot M, Ageron E, Grimont P A D, Garcia J-L. Description of Thermoanaerobacter brockii subsp. lactiethylicus subsp. nov., isolated from a deep subsurface French oil well, a proposal to reclassify Thermoanaerobacter finnii as Thermoanaerobacter brockii subsp. finnii comb. nov., and an emended description of Thermoanaerobacter brockii. Int J Syst Bacteriol. 1995;45:783–789. doi: 10.1099/00207713-45-4-783. [DOI] [PubMed] [Google Scholar]
- 9.Cochrane W J, Johne P S, Sanders P F, Holt D M, Mosley M J. Studies on the thermophilic sulfate-reducing bacteria from a souring North Sea oil field. Soc Petrol Eng Prod Eng. 1988;18368:301–316. [Google Scholar]
- 10.Eaton A D, Clesceri L S, Greenberg A E. Standard methods for the examination of water and wastewater. 19th ed. Washington, D.C.: American Public Health Association; 1995. [Google Scholar]
- 11.Fardeau M-L, Cayol J-L, Magot M, Patel B K C, Ollivier B. Effect of thiosulfate as electron acceptor on glucose and xylose oxidation by Thermoanaerobacter finnii and a Thermoanaerobacter sp. isolated from oil field water. Res Microbiol. 1996;147:159–165. doi: 10.1016/0923-2508(96)80215-4. [DOI] [PubMed] [Google Scholar]
- 12.Fardeau M-L, Ollivier B, Patel B K C, Magot M, Thomas P, Rimbault A, Rocchiccioli F, Garcia J-L. Thermotoga hypogea sp. nov., a xylanolytic thermophilic bacterium from an oil-producing well. Int J Syst Bacteriol. 1997;47:1013–1019. doi: 10.1099/00207713-47-4-1013. [DOI] [PubMed] [Google Scholar]
- 13.Godfroy A, Lesongeur F, Raguénès G, Quérellou J, Antoine E, Meunier J-R, Guezennec J, Barbier G. Thermococcus hydrothermalis sp. nov., a new hyperthermophilic archaeon isolated from a deep-sea hydrothermal vent. Int J Syst Bacteriol. 1997;47:622–626. doi: 10.1099/00207713-47-3-622. [DOI] [PubMed] [Google Scholar]
- 14.Godfroy A, Meunier J-R, Guezennec J, Lesongeur F, Raguénès G, Rimbault A, Barbier G. Thermococcus fumicolans sp. nov., a new hyperthermophilic archaeon isolated from a deep-sea hydrothermal vent in North Fiji basin. Int J Syst Bacteriol. 1996;46:1113–1119. doi: 10.1099/00207713-46-4-1113. [DOI] [PubMed] [Google Scholar]
- 15.González J M, Kato C, Horikoshi K. Thermococcus peptonophilus sp. nov., a fast-growing, extremely thermophilic archaebacterium isolated from deep-sea hydrothermal vents. Arch Microbiol. 1995;164:159–164. [PubMed] [Google Scholar]
- 16.Grassia G S, McLean K M, Glénat P, Bauld J, Sheehy A J. A systematic survey for thermophilic fermentative bacteria and archaea in high temperature petroleum reservoirs. FEMS Microbiol Ecol. 1996;21:47–58. [Google Scholar]
- 17.Grote R, Li L, Tamaoka J, Kato C, Horikoshi K, Antranikian G. Thermococcus siculi sp. nov., a novel hyperthermophilic archaeon isolated from a deep-sea hydrothermal vent at the Mid-Okinawa Trough. Extremophiles. 1999;3:55–62. doi: 10.1007/s007920050099. [DOI] [PubMed] [Google Scholar]
- 18.Gundersen J K, Jorgensen B B, Larsen E, Jannasch H W. Mats of giant sulphur bacteria on deep-sea sediments due to fluctuating hydrothermal flow. Nature. 1992;360:454–456. [Google Scholar]
- 19.Hoaki T, Nishijima M, Kato M, Adachi K, Mizobuchi S, Hanzawa N, Maruyama T. Growth requirements of hyperthermophilic sulfur-dependent heterotrophic archaea isolated from a shallow submarine geothermal system with reference to their essential amino acids. Appl Environ Microbiol. 1994;60:2898–2904. doi: 10.1128/aem.60.8.2898-2904.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hoaki T, Nishijima M, Miyashita H, Maruyama T. Dense community of hyperthermophilic sulfur-dependent heterotrophs in a geothermally heated shallow submarine biotope near Kodakara-Jima Island, Kagoshima, Japan. Appl Environ Microbiol. 1995;61:1931–1937. doi: 10.1128/aem.61.5.1931-1937.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hoaki T, Wirsen C O, Hanzawa S, Maruyama T, Jannasch H W. Amino acid requirements of two hyperthermophilic archaeal isolates from deep-sea vents, Desulfurococcus strain SY and Pyrococcus strain GB-D. Appl Environ Microbiol. 1993;59:610–613. doi: 10.1128/aem.59.2.610-613.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hobbie J E, Daley R J, Jasper S. Use of Nuclepore filters for counting bacteria by fluorescence microscopy. Appl Environ Microbiol. 1977;33:1225–1228. doi: 10.1128/aem.33.5.1225-1228.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Huber R, Langworthy T A, König H, Thomm M, Woese C R, Sleytr U B, Stetter K O. Thermotoga maritima sp. nov. represents a new genus of unique extremely thermophilic eubacteria growing up to 90°C. Arch Microbiol. 1986;144:324–333. [Google Scholar]
- 24.Huber R, Stöhr J, Honenhaus S, Rachel R, Burggraf S, Jannasch H W, Stetter K O. Thermococcus chitonophagus sp. nov., a novel, chitin-degrading, hyperthermophilic archaeum from a deep-sea hydrothermal environment. Arch Microbiol. 1995;164:255–264. [Google Scholar]
- 25.Jannasch H W, Huber R, Belkin S, Stetter K O. Thermotoga neapolitana sp. nov. of the extremely thermophilic, eubacterial genus Thermotoga. Arch Microbiol. 1988;150:103–104. [Google Scholar]
- 26.Jannasch H W, Wirsen C O, Hoaki T. Isolation and cultivation of heterotrophic hyperthermophiles from deep-sea hydrothermal vents. In: Robb F T, et al., editors. Archaea: a laboratory manual. Plainview, N.Y: Cold Spring Harbor Laboratory Press; 1995. pp. 9–13. [Google Scholar]
- 27.Jeanthon C, Reysenbach A-L, L'Haridon S, Gambacorta A, Pace N R, Glénat P, Prieur D. Thermotoga subterranea sp. nov., a new thermophilic bacterium isolated from a continental oil reservoir. Arch Microbiol. 1995;164:91–97. [PubMed] [Google Scholar]
- 28.Kai M, Tamura K, Yamaguchi M, Ohkura Y. Aromatic amines as fluorogenic reagents for reducing carbohydrates. Anal Sci. 1985;1:59–63. [Google Scholar]
- 29.Keller M, Braun F-J, Dirmeier R, Hafenbradl D, Burggraf S, Rachel R, Stetter K O. Thermococcus alcaliphilus sp. nov., a new hyperthermophilic archaeum growing on polysulfide at alkaline pH. Arch Microbiol. 1995;164:390–395. [PubMed] [Google Scholar]
- 30.Kennish M. Practical handbook of marine science. Boca Raton, Fla: CRC Press, Inc.; 1989. [Google Scholar]
- 31.Kobayashi T, Kwak Y S, Akiba T, Kudo T, Horikoshi K. Thermococcus profundus sp. nov., a new hyperthermophilic archaeon isolated from a deep-sea hydrothermal vent. System Appl Microbiol. 1994;17:232–236. [Google Scholar]
- 32.Kurr M, Huber R, König H, Jannasch H W, Fricke H, Trincone A, Kristjansson J K, Stetter K O. Methanopyrus kandleri, gen. and sp. nov. represents a novel group of hyperthermophilic methanogens, growing at 110°C. Arch Microbiol. 1991;156:239–247. [Google Scholar]
- 33.L'Haridon S, Reysenbach A-L, Glénat P, Prieur D, Jeanthon C. Hot subterranean biosphere in a continental oil reservoir. Nature. 1995;377:223–224. [PubMed] [Google Scholar]
- 34.Marteinsson V T, Birrien J-L, Reysenbach A-L, Vernet M, Marie D, Gambacorta A, Messner P, Sleytr U B, Prieur D. Thermococcus barophilus sp. nov., a new barophilic and hyperthermophilic archaeon isolated under high hydrostatic pressure from a deep-sea hydrothermal vent. Int J Syst Bacteriol. 1999;49:351–359. doi: 10.1099/00207713-49-2-351. [DOI] [PubMed] [Google Scholar]
- 35.Miroshnichenko M L, Bonch-Osmolovskaya E A, Neuner A, Kostrikina N A, Chernych N A, Alekseev V A. Thermococcus stetteri sp. nov., a new extremely thermophilic marine sulfur-metabolizing archaebacterium. Syst Appl Microbiol. 1989;12:257–262. [Google Scholar]
- 36.Miroshnichenko M L, Gongadze G M, Rainey F A, Kostyukova A S, Lysenko A M, Chernyh N A, Bonch-Osmolovskaya E A. Thermococcus gorgonarius sp. nov. and Thermococcus pacificus sp. nov., heterotrophic extremely thermophilic archaea from New Zealand submarine hot vents. Int J Syst Bacteriol. 1998;48:23–29. doi: 10.1099/00207713-48-1-23. [DOI] [PubMed] [Google Scholar]
- 37.Nelson D C, Fisher C R. Chemoautotrophic and methanotrophic endosymbiotic bacteria at deep-sea vents and seeps. In: Karl D M, editor. The microbiology of deep-sea hydrothermal vents. Boca Raton, Fla: CRC Press Inc.; 1995. [Google Scholar]
- 38.Neuner A, Jannasch H W, Belkin S, Stetter K O. Thermococcus litoralis sp. nov.: a new species of extremely thermophilic marine archaebacteria. Arch Microbiol. 1990;153:205–207. [Google Scholar]
- 39.Nilsen R K, Beeder J, Thorstenson T, Torsvik T. Distribution of thermophilic marine sulfate reducers in North Sea oil field waters and oil reservoirs. Appl Environ Microbiol. 1996;62:1793–1798. doi: 10.1128/aem.62.5.1793-1798.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nilsen R K, Beeder J, Torsvik T. Methanococcus thermolithotrophicus isolated from North Sea oil field reservoir water. Appl Environ Microbiol. 1996;62:728–731. doi: 10.1128/aem.62.2.728-731.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nilsen R K, Torsvik T, Lien T. Desulfotomaculum thermocisternum, sp. nov., a sulfate reducer isolated from a hot North Sea oil reservoir. Int J System Bacteriol. 1996;46:397–402. [Google Scholar]
- 42.Parkes J, Maxwell J. Some like it hot (and oily) Nature. 1993;365:694–695. [Google Scholar]
- 43.Ravot G, Magot M, Fardeau M-L, Patel B K C, Prensier G, Egan A, Garcia J-L, Ollivier B. Thermotoga elfii sp. nov., a novel thermophilic bacterium from an African oil-producing well. Int J Syst Bacteriol. 1995;45:308–314. doi: 10.1099/00207713-45-2-308. [DOI] [PubMed] [Google Scholar]
- 44.Rees G N, Grassia G S, Sheeny A J, Dwivedi P P, Patel B K C. Desulfacinum infernum gen. nov., sp. nov., a thermophilic sulfate-reducing bacterium from a petroleum reservoir. Int J Syst Bacteriol. 1995;45:85–89. [Google Scholar]
- 45.Ronimus R S, Reysenbach A-L, Musgrave D R, Morgan H W. The phylogenetic position of Thermococcus isolate AN1 based on 16S rRNA gene sequence analysis: a proposal that AN1 represents a new species, Thermococcus zilligii sp. nov. Arch Microbiol. 1997;168:245–248. doi: 10.1007/s002030050495. [DOI] [PubMed] [Google Scholar]
- 46.Roszak D B, Colwell R R. Survival strategies of bacteria in the natural environment. Microbiol Rev. 1987;51:365–379. doi: 10.1128/mr.51.3.365-379.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rueter P, Rabus R, Wilkes H, Aeckersberg F, Rainey F A, Jannasch H W, Widdel F. Anaerobic oxidation of hydrocarbons in crude oil by new types of sulphate-reducing bacteria. Nature. 1994;372:455–458. doi: 10.1038/372455a0. [DOI] [PubMed] [Google Scholar]
- 48.Stetter K O, Huber R, Blochl E, Kurr M, Eden R D, Fielder M, Cash H, Vance I. Hyperthermophilic archaea are thriving in deep North Sea and Alaskan reservoirs. Nature. 1993;365:743–745. [Google Scholar]
- 49.Takeuchi K, Yoshimura T, Kato H. Geology of the Kakizaki district. Quadrangle series, scale 1:50,000, Niigata 7 no. 48, NJ-54-35-9. Geological survey of Japan; 1996. , Tsukuba. [Google Scholar]
- 50.Verweij J M. Hydrocarbon migration systems analysis. Development in petroleum science. Vol. 35. Amsterdam, The Netherlands: Elsevier; 1993. [Google Scholar]
- 51.Wolin E A, Wolin M J, Wolfe R S. Formation of methane by bacterial extracts. J Biol Chem. 1963;238:2882–2886. [PubMed] [Google Scholar]
- 52.Zillig W, Holz I, Janekovic D, Schäfer W, Reiter W D. The archaebacterium Thermococcus celer represents a novel genus within the thermophilic branch of the archaebacteria. Syst Appl Microbiol. 1983;4:88–94. doi: 10.1016/S0723-2020(83)80036-8. [DOI] [PubMed] [Google Scholar]
- 53.Zillig W, Stetter K O, Wunderl S, Schulz W, Priess H, Scholz J. The Sulforobus-“Caldariella” group: taxonomy on the basis of the structure of DNA-dependent RNA polymerases. Arch Microbiol. 1980;125:259–269. [Google Scholar]