Abstract
Introduction
Hepatitis A virus infections are mostly asymptomatic or mildly symptomatic, and generally this disease has a benign course and resolves spontaneously. However, intrahepatic and rarer extrahepatic manifestations can complicate typical cases of acute hepatitis. Pleural effusion is an extremely rare extrahepatic entity with 20 cases reported in literature.
Case presentation
We report herein a recent case of both pleural effusion and ascites accompanying hepatitis A infection in a 5-year-old middle eastern child, diagnosed using serological testing and imaging studies, who was treated with supportive management with full resolution after 2 weeks. In addition, we review available literature regarding hepatitis A virus associated with pleural effusion using PubMed and summarize all reported cases in a comprehensive table.
Results
Literature contains 20 reported cases of serology-confirmed hepatitis A virus presenting with pleural effusion, most in the pediatric population with average age at presentation of 9 years 8 months. The majority of reported patients had right-sided pleural effusion (50%) or bilateral effusion (45%), while only 5% presented with pleural effusion on the left side. Hepatomegaly and ascites occurred concurrently in 80% and 70% respectively. Supportive treatment without invasive procedures (except one chylothorax case) yielded complete recovery in 95% of cases, while only one case progressed to fulminant liver failure followed by death.
Conclusion
Acute hepatitis A virus rarely presents with pleural effusion, usually following a benign course with spontaneous resolution in most patients. Pleural effusion does not change the prognosis or require any invasive treatment. Thus, further invasive procedures are not recommended and would only complicate this self-resolving benign condition.
Keywords: Pleural effusion, Ascites, Acute hepatitis, Acute hepatitis A virus, HAV associated with self-limited pleural effusion, Unusual manifestation, Conservative management
Introduction
Hepatitis A virus (HAV) is a positive-stranded Ribonucleic acid (RNA) virus that is stable at moderate temperatures and low pH, allowing for prolonged survival in the environment and fecal–oral transmission. It is known to circulate among children, especially in developing countries due to poor hygiene and lack of sanitation [1]. Although hepatitis A is usually asymptomatic or presents with mild symptoms in children, extrahepatic manifestations and, particularly, pleural effusions are rare [2, 3]. The first case of pleural effusion caused by hepatitis A as underlying infection was described as early as 1971 by Gross and Gerding [4], but this association has been scarcely reported in medical literature, with no more than 20 cases [5]. We provide herein a comprehensive literature review of 20 published cases and also report a new case, to clarify this rare entity.
Case presentation
A previously healthy 5-year-old middle eastern boy with no known history of any medical diseases presented to the emergency department with jaundice and scleral icterus, in addition to dark-colored urine, abdominal pain and distention, and slight shortness of breath beginning 4 days previously after contact history with individuals having acute hepatitis A symptoms.
He had no previous history of traveling, blood transfusion, bleeding, or previous medical, drug, or surgical treatment.
Upon presentation, during physical examination, the patient had high fever (39 °C), abdominal distention, hepatomegaly with normal spleen size, unilateral basal right-sided decreased breathing sound and dullness, as well as tachycardia and tachypnea. The rest of the examination was normal, including normal mental status.
The patient was admitted, and laboratory investigations were carried out (Tables 1, 2). HAV serology testing was positive. Chest x-ray showed unilateral right-sided pleural effusion. Chest contrast computed tomography (CT) scan delineated right effusion with significant lung collapse, plus negligible amount on the left side and clear left lung field (Figs. 1, 2). Abdominal sonography and abdominal–pelvic CT scan identified hepatomegaly and ascites. Echocardiography was free of any abnormality.
Table 1.
Laboratory analysis
| Laboratory analysis | Result |
|---|---|
| Hb | 11.7 g/dl |
| WBC | 6.5 × 103 cells/mm3 |
| PLT | 230 × 103/mm3 |
| Serology HAV Igm |
+ve HAV IgM −ve indirect Coombs |
| PT | 14.4 (12.3 control) |
| PTT | 25 (26 control) |
| INR | 1.17 |
| Albumin | 2.9 g/dl |
| GGT | 101 U/l |
| ALP | 410 IU/l |
| Serum ammonia | 115 UG/dl |
| Coombs test | Negative |
Hb: Hemoglobin, WBC: White blood cells, PLT: platelets, PT: Prothrombin Time, PTT: Partial Thromboplastin Time, INR: International normalized ratio, GGT: amma-glutamyl transferase, ALP: alkaline phosphatase, BUN: Blood urea nitrogen
Urinalysis also normal
Table 2.
Liver biochemical markers tracing during hospital stay
| Liver biochemical markers tracing during hospital stay | 1st day | 2nd day | 3rd day |
|---|---|---|---|
| AST (U/l) | 470 | 396 | 300 |
| ALT (U/l) | 883 | 654 | 578 |
| Total bilirubin (mg/dl) | 8.9 | 4.1 | 4.1 |
| Direct bilirubin (mg/dl) | 6.6 | 2.7 | 3 |
AST: Aspartate aminotransferase, ALT: alanine aminotransferase
On day 5 post-discharge, follow-up LFTs normalized
Fig. 1.
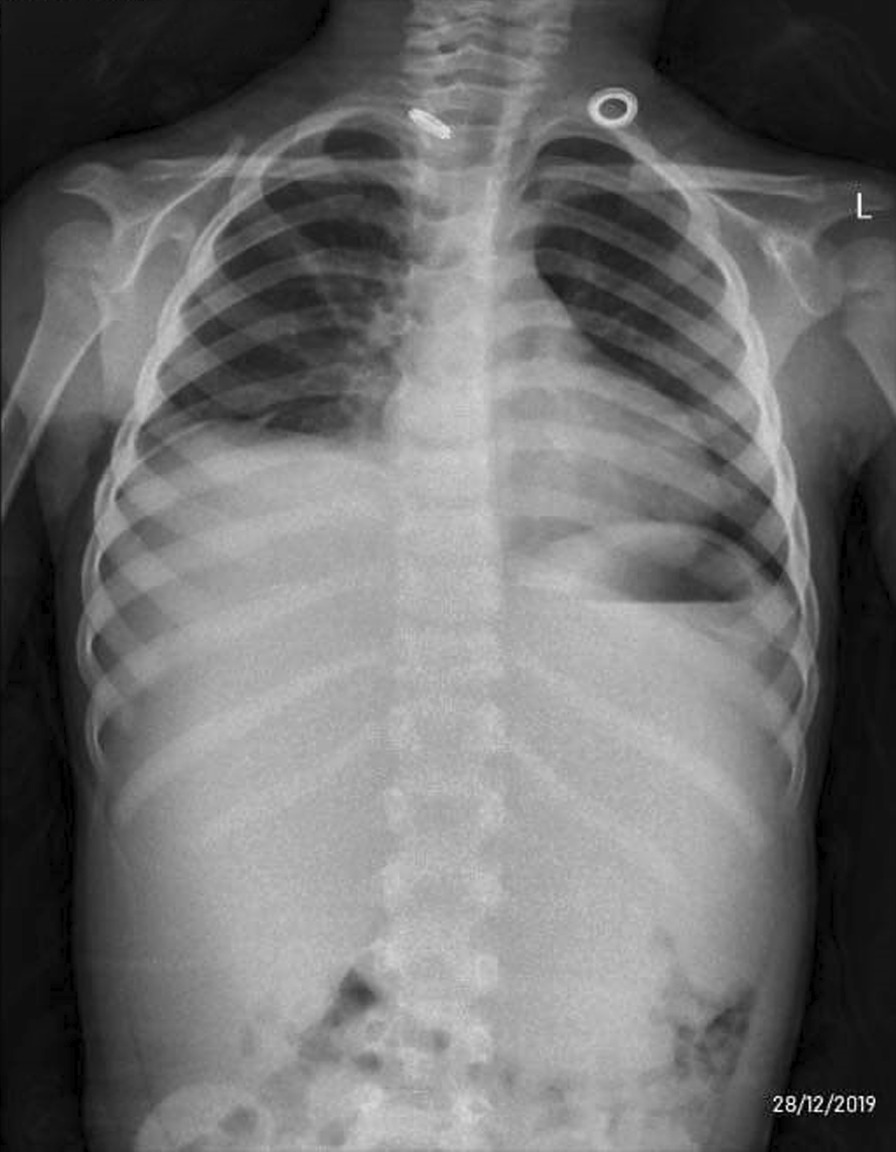
Chest x-ray showing right-sided pleural effusion
Fig. 2.
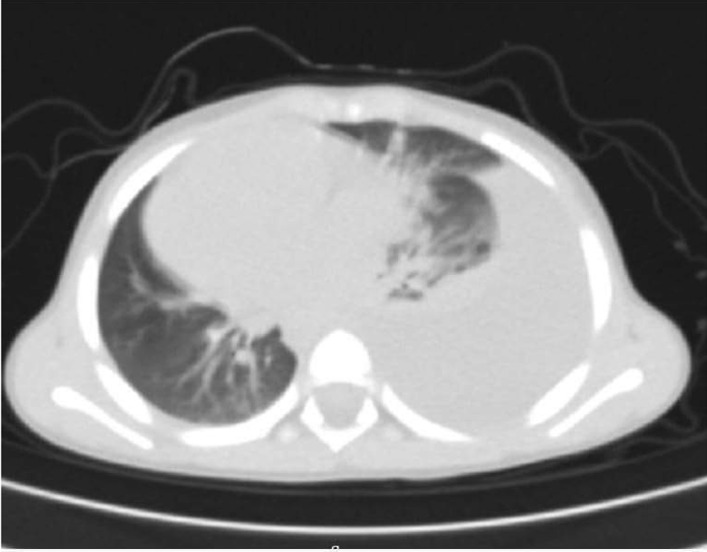
Computed tomography showing right-sided pleural effusion
Our patient was diagnosed with HAV acute hepatitis associated with right-side pleural effusion and ascites, confirmed by CT scan. Treatment consisted of supportive parenteral fluid and carbohydrate-enriched diet, while no diuretics or antibiotics were used. The patient was discharged on day 4 after significant improvement, achieving full clinical and biochemical recovery 5 days postdischarge with normal liver function tests and normal lung and abdominal imaging.
Discussion
Acute hepatitis caused by hepatitis A virus infection can manifest with a variety of symptoms and severities. One important factor is age, as disease severity is inversely proportional to age, with more than 80% of children having a less severe course and complete recovery within 3 months, usually being asymptomatic and anicteric. However, severity and mortality rates increase with advancing age [6].
Onset of symptoms follows a mean incubation period of approximately 30 days. Common signs and symptoms include fever, jaundice, fatigue, abdominal pain, nausea, and emesis.
Infectivity and viral shedding last from the beginning of the incubation period until 1 week after jaundice resolution, during which the virus is capable of fecal–oral spread [7].
HAV acute hepatitis may be associated with many complications, including:
Intrahepatic: such as cholestatic hepatitis, relapsing hepatitis, and autoimmune hepatitis. Rarely, hepatitis A can progress to acute liver failure.
Extrahepatic manifestations are infrequently reported in HAV acute hepatitis (6.4–8%) and may include: urticarial and maculopapular rash, acute kidney injury, autoimmune hemolytic anemia, aplastic anemia, acute pancreatitis, mononeuritis, reactive arthritis, Guillain–Barre syndrome and pleural or pericardial effusion, ascites, glomerulonephritis, polyarteritis nodosa, cryoglobulinemia, and thrombocytopenia [2, 3, 8].
Among those complications, pleural effusion is an extremely rare co-occurring condition that is scarcely reported in literature. The exact mechanism is unknown and could be multifactorial. There are many theories regarding the suspected pathogenesis of this entity:
Kurt et al. suggested direct viral invasion of pleura, immune complex deposition, or inflammatory response as probable cause, since HAV ribonucleic acid was found in the pleural fluid of a HAV viral hepatitis patient by polymerase chain reaction [9].
Dhakal et al. postulated that copresenting ascites could contribute to the pleural effusion via small diaphragmatic defects or diaphragmatic lymphatics [5, 10].
Also, a decrease in the plasma oncotic pressure as well as a transient rise of the pressure in the portal vein and/or lymphatics due to compression by the hepatic sinusoids may be a contributory factor in some cases developing ascites and pleural effusion [5, 11].
Tables 3 and 4 present a comprehensive review of available published cases of HAV with pleural effusion. All 20 patients were diagnosed by positive serum anti-HAV IgM antibodies and pleural effusion on imaging studies, regardless of the underlying nature of effusion (transudative or exudative). Most patients were from the pediatric population with average age of 9 years 8 months, while 80% (16 patients) were younger than 12 years old, with a male-to-female ratio of 9:11. Most patients presented with usual symptoms of acute hepatitis including fever, vomiting, abdominal pain, jaundice, icterus, and fatigue. Also, abdominal and chest examination revealed hepatomegaly, abdominal distention, chest dullness, and decreased airway entry and normal mental status in all patients. Laboratory testing showed an average of 3.1 albumin g/dl, with average total and direct bilirubin of 5.2 and 4 mg/dl respectively. Chest x-ray, ultrasonography, and in some patients computerized tomography or magnetic resonance imaging all generally showed similar results: most patients had right-sided (ten patients) or bilateral pleural effusion (nine patients), while one case had effusion on the left side. Also, the majority copresented with hepatomegaly (16 patients) and ascites (14 patients). Finally, thickened gallbladder wall was seen in only three patients. Moreover, pleural fluid analysis mostly showed a transudative nature of the effusion, while one patient had exudative effusion resulting from Salmonella paratyphi A superinfection, and one case had chylothorax, but pleural fluid analysis was carried out in a limited number of patients (nine).
Table 3.
Literature review
| Study | Sex | Age | Chief compliant | Physical examination | Management | Diuretics | Outcome |
|---|---|---|---|---|---|---|---|
| Saha [15] | M | 3 years | Generalized body swelling | Icterus, bilateral pitting edema, abdominal distention, hepatomegaly | Supportive management | N/A | Spontaneous resolution after 4 days |
|
Roy [16] Case 1 |
F | 6 years | Fever, vomiting, fatigue | Icterus, abdominal distention, hepatomegaly, decreased breath sounds on the right side of chest | Supportive management, B-complex, ursodeoxycholic acid, oral lactulose | ++ | Spontaneous resolution after 1 week |
|
Roy [16] Case 2 |
M | 4 years | Fever, jaundice | Abdominal distention, hepatomegaly | Supportive management | ++ | Spontaneous resolution |
| Owen [17] | M | 42 years | Fever, malaise, pleuritic pain | Dullness on the base of right lung | Supportive management | N/A | Spontaneous resolution |
| Dalai et al. [14] | F | 3 years | Fever, abdominal pain | Icterus, hepatomegaly | Supportive management, IV vit. K, oral antibiotic | N/A | Spontaneous resolution after 3 weeks |
| Nagarajan et al. [18] Case 1 | F | 7 years | Abdominal pain, jaundice | Icterus, hepatomegaly, decreased breath sounds on the right side of chest | Supportive management | N/A | Spontaneous resolution after 3 weeks |
| Nagarajan et al. [18] Case 2 | F | 10 years | Fever, vomiting, abdominal pain | Icterus, hepatomegaly | Supportive management | N/A | Spontaneous resolution |
| Allen et al. [19] | F | 30 years | Flu like symptoms, fatigue, myalgia | Icterus, right upper quadrant abdominal tenderness | Supportive management | N/A | Spontaneous resolution |
| Selimoğlu et al. [20] | M | 8 years | Fever, jaundice, anorexia, abdominal pain | Icterus, hepatomegaly, dullness on the base of right lung | Supportive management | N/A | Spontaneous resolution |
| Mehta et al. [13] | M | 3 years | Fever, vomiting, abdominal pain, jaundice | Icterus, hepatomegaly, dullness on the base of right lung | Supportive management, IV amoxicillin–clavulanic acid, thoracotomy with chest tube insertion | N/A | Complete resolution after 1 week |
| Alhan et al. [14] | M | 3 years | Fever, vomiting, jaundice | Febrile, icterus, hepatomegaly, dullness on the base of right lung | Supportive management | N/A | Death after 2 weeks due to fulminant liver failure, increased intracranial pressure |
| Erdem et al. [10] | M | 12 years | Nausea, vomiting, fatigue | Icterus, febrile, hepatomegaly | Supportive management, vit. K, protein/lipid–restricted and carbohydrate-enriched diet | N/A | Spontaneous resolution after 10 days |
| Ghosh and Kundu [12] | F | 4 years | Fever, jaundice, cough, dyspnea | Hepatomegaly, splenomegaly, abdominal distention, dullness on the base of right lung | Supportive management, IV vit. K, IV cefotaxime | N/A | Complete resolution after 1 week |
| Gürkan et al. [10] | M | 4 years | Jaundice, abdominal pain, vomiting, headache | Jaundice, febrile, abdominal distention, hepatomegaly | Supportive management | N/A | Spontaneous resolution after 15 days |
| Kaman et al. [21] | F | 4 years | Fever, fatigue, abdominal pain | Icterus, decreased breath sounds on the right side of chest | Supportive management, vit. K | N/A | Spontaneous resolution after 1 week |
| Vaidya et al. [22] | F | 7 years | Vomiting, nausea | Icterus, hepatomegaly | Supportive management | N/A | Spontaneous resolution after 2 weeks |
| Bukulmez et al. [23] | F | 7 years | Fever, jaundice, abdominal pain | Icterus, hepatomegaly, dullness on the base of right lung, abdominal distention | Supportive management | N/A | Spontaneous resolution after 2 week |
| Dhakal et al. [5] | F | 2.5 years | Abdominal pain, scleral icterus | Icterus, hepatomegaly, dullness on the base of right lung | Supportive management | N/A | Spontaneous resolution after 2 week |
| Hadgu et al. [24] | M | 4.8 years | Fever, abdominal pain, nausea and vomiting, cough | Bilateral dullness and decreased air entry, hepatomegaly, anicteric | Supportive treatment | N/A | Spontaneous resolution after 1 month |
| Iza et al. [25] | F | 32 years | Jaundice epigastric pain, nausea, vomiting, dark urine | Icteric, abdominal tenderness, abdominal distension positive Murphy sign, decreased air entry on right chest | Supportive treatment | N/A | Spontaneous resolution after 4 months |
F: Female, M: Male, Y: Years, M: months, N/A: not available, USG: ultrasonography, CXR: chest x-ray, CT: Computed tomography, MRI: Magnetic resonance imaging
Table 4.
Literature review
| Study | Imaging finding | HAV serology | INR | Albumin (g/dl) | Total/direct bilirubin (mg/dl) | Pleural fluid analysis |
|---|---|---|---|---|---|---|
| Saha [15] |
USG: bilateral pleural effusion, ascites CXR: left-sided pleural effusion |
Serum anti-HAV IgM positive | N/A | 2.9 | 5.6/5 | NA |
|
Roy [16] Case 1 |
USG: hepatomegaly, ascites, bilateral pleural effusion CXR: bilateral pleural effusion (right > left) |
Serum and pleural fluid anti-HAV IgM positive | N/A | 3.4 | 2.6/1.4 | Total cell count 1500, glucose 99 mg/dl and protein 4.1 g/dl, negative culture |
|
Roy [16] Case 2 |
USG: hepatomegaly, ascites, bilateral pleural effusion CXR: bilateral pleural effusion (right > left) |
Serum anti-HAV IgM positive | N/A | 3.2 | 6.2/6.2 | NA |
| Owen [17] | CXR: right pleural effusion | NA | N/A | N/A | N/A | NA |
| Dalai et al. [14] | USG: right-sided pleural effusion, ascites, hepatomegaly | Serum anti-HAV IgM positive | 2 | N/A | 3.5/1.5 | NA |
| Nagarajan et al. [17] Case 1 |
USG: hepatomegaly, ascites, bilateral pleural effusion CXR: bilateral pleural effusion (right > left) |
Serum and pleural fluid anti-HAV IgM positive | N/A | 2.5 | 5.4/4.8 | Total cell count 0, protein 20 g/dl |
| Nagarajan et al. [17] case 2 |
USG: hepatomegaly, bilateral pleural effusion, ascites CXR: bilateral pleural effusion |
Serum anti-HAV IgM positive | N/A | 3.2 | 6.9/5.9 | NA |
| Allen et al. [18] |
USG: ascites, diffuse gallbladder wall thickening CT: ascites, right-side pleural effusion, gallbladder wall thickening |
Serum anti-HAV IgM positive | N/A | 3.6 | 6/2.4 | NA |
| Selimoğlu et al. [19] | USG: hepatomegaly CXR: right lower lung consolidation | Serum and pleural fluid anti-HAV IgM positive | 1.25 | 3.5 | 6/3.5 | Total cell count 0, glucose 70 mg/dl and protein 4.5 g/dl, negative culture |
| Mehta et al. [13] | CXR: right-side pleural effusion | Serum and pleural fluid anti-HAV IgM positive | N/A | 2.8 | 5.3/5.2 | Total cell count 18200, glucose 94 mg/dl and protein 7.7 g/dl, negative culture |
| Alhan et al. [14] | USG: hepatomegaly, right-side pleural effusion | Serum and pleural fluid anti-HAV IgM positive | N/A | 4.0 | 3.9/2.6 | Total cell count 0, transudate |
| Erdem et al. [20] | USG: ascites, right-side pleural effusion, thickened gallbladder wall; CXR: right-sided pleural effusion | Serum and pleural fluid anti-HAV IgM, positive | 1.1 | 1.9 | 6.3/5.6 | Total cell count 0, transudate |
| Ghosh and Kundu [12] |
CXR: middle and lower zones of left lung opacity MRI: pleural effusion, hepatosplenomegaly, thickened gallbladder wall |
Serum and pleural fluid anti-HAV IgM positive | 1.9 | N/A | 5.6/5.5 | Exudative pleural effusion |
| Gürkan et al. [10] |
USG: ascites CXR: bilateral pleural effusion |
Serum anti-HAV IgM positive | N/A | 3.6 | 6/2.5 | NA |
| Kaman et al. [10] |
USG: ascites, pleural effusion CXR: right-side pleural effusion |
Serum anti-HAV IgM positive | N/A | 2.5 | 6.6/4.8 | Glucose 90 mg/dl, negative culture |
| Vaidya et al. [21] |
USG: ascites, hepatomegaly, bilateral pleural effusion CXR: left-side pleural effusion |
Serum anti-HAV IgM positive | 1.1 | 4 | 5.2/4.2 | NA |
| Bukulmez et al. [22] |
USG: hepatomegaly, right-side pleural effusion CT: right pleural effusion |
Serum anti-HAV IgM positive | 1 | 3.3 | 8.2/6.7 | NA |
| Dhakal et al. [5] |
USG: ascites, bilateral pleural effusion CXR: right-side pleural effusion |
Serum anti-HAV IgM positive | N/A | N/A | 5.8/4.5 | NA |
| Hadgu et al. [24] | USG: mild ascites, hepatosplenomegaly, and small bilateral pleural effusion | Serum anti-HAV IgM positive | 1.5 | 3.8 | 1.5/0.5 | No cells, lactic acid dehydrogenase 15 IU/l, negative TB, negative bacterial culture |
| Iza et al. [25] | USG: right pleural effusion, ascites and acalculous cholecystitis | Serum anti-HAV IgM positive | Normal | 3.5 | 2.6/2.5 | N/A |
F: Female, M: Male, Y: Years, M: months, N/A: not available, USG: ultrasonography, CXR: chest x-ray, CT: Computed tomography, MRI: Magnetic resonance imaging
All patients were managed supportively. Furthermore, no invasive additional treatments were used in five cases, including intravenous fluids, vitamin K, oral lactulose, and antibiotics prophylactically for bacterial superinfection [12, 14], while thoracostomy and chest tube insertion were only needed in one patient of chylothorax [13].
Of these patients, 95% (19 out of 20 patients) had complete recovery and resolution of pleural effusion and ascites, while one patient (5% of patients) suffered from fulminant liver failure and refractory intracranial pressure increase leading to death 2 weeks after diagnosis [14]
HAV infection is usually self-limited and does not progress to a chronic or latent state, being managed supportively, and the same applies to associated pleural effusions. Pleural effusions do not change the prognosis or require any invasive treatment.
Conclusion
Pleural effusion is a benign, rare, extrahepatic complication of HAV acute hepatitis, mostly present in juveniles as early right-sided effusion. It resolves spontaneously with supportive management. Thus, further invasive procedures would only complicate this self-resolving benign condition and should be minimized.
Acknowledgements
Not applicable.
Author contributions
JSZ, TZA, IM, LZT, AAZA, and BMS were involved in the study concept and design. Data collection was performed by KJA, AHY, KJF, MASA, MRM, and AKH. JSZ, TZA, YIMZ, and FMA wrote the paper. RHT, HIAA, and YIMZ reviewed the literature. All authors read and approved the final manuscript.
Funding
No funding or grant support.
Availability of data and materials
The data used to support the findings of this study are available from the corresponding author upon reasonable request.
Declarations
Ethics approval and consent to participate
The study is exempt from ethical approval in our institution, and consent has been obtained.
Consent for publication
Written informed consent was obtained from the patient’s legal guardian for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Competing interests
The authors declare that they have no conflicts of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Jihad Samer Zalloum, Email: Jihad.s.zalloum@gmail.com.
Tareq Z. Alzughayyar, Email: tareq-20007@hotmail.com
Fawzy M. Abunejma, Email: fawzimazen49@gmail.com
Ibba Mayadma, Email: eeba18796@gmail.com.
Layan Ziad Tomeh, Email: tomehlayan3@gmail.com.
Karim Jamal Abulaila, Email: Kareem.laila.med@gmail.com.
Asil Husam Yagmour, Email: aseel.yaghmour@hotmail.com.
Khalid Jamal Faris, Email: khald199998@gmail.com.
Mohammed A. S. Aramin, Email: mohammadarbr@hotmail.com
Mo’min Ra’id Mesk, Email: mumn202@gmail.com.
Asala Khalil Hasani, Email: asalahasani20@gmail.com.
Balqis Mustafa Shawer, Email: Balkees202@hotmail.com.
Rawand Hisham Titi, Email: 180138@ppu.edu.ps.
Ayat A. Z. Aljuba, Email: ayatjubeh2000@gmail.com
Hussam I. A. Alzeerelhouseini, Email: Hussam.z@live.com
Yousef I. M. Zatari, Email: yousef_zat_1995@hotmail.com
References
- 1.Martin A, Lemon SM. Hepatitis A virus: from discovery to vaccines. Hepatology. 2006;43(S1):S164–S172. doi: 10.1002/hep.21052. [DOI] [PubMed] [Google Scholar]
- 2.Amarapurkar DN, Amarapurkar AD. Extrahepatic manifestations of viral hepatitis. Ann Hepatol. 2002;1(4):192–195. doi: 10.1016/S1665-2681(19)32165-9. [DOI] [PubMed] [Google Scholar]
- 3.Willner IR, et al. Serious hepatitis A: an analysis of patients hospitalized during an urban epidemic in the United States. Ann Internal Med. 1998;128(2):111–114. doi: 10.7326/0003-4819-128-2-199801150-00006. [DOI] [PubMed] [Google Scholar]
- 4.Gross PA, Gerding DN. Pleural effusion associated with viral hepatitis. Gastroenterology. 1971;60(5):898–902. doi: 10.1016/S0016-5085(71)80090-2. [DOI] [PubMed] [Google Scholar]
- 5.Dhakal AK, et al. An unusual association of pleural effusion with acute viral hepatitis A infection. Pediatr Health Med Ther. 2014;5:149–153. doi: 10.2147/PHMT.S70869. [DOI] [Google Scholar]
- 6.Ciocca M. Clinical course and consequences of hepatitis A infection. Vaccine. 2000;18:S71–S74. doi: 10.1016/S0264-410X(99)00470-3. [DOI] [PubMed] [Google Scholar]
- 7.Koff RS. Clinical manifestations and diagnosis of hepatitis A virus infection. Vaccine. 1992;10:S15–S17. doi: 10.1016/0264-410X(92)90533-P. [DOI] [PubMed] [Google Scholar]
- 8.Nagarajan VP, et al. Pleural effusion—an unusual cause. Australas Med J. 2012;5(7):369. doi: 10.4066/AMJ.2012.1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kurt AN, et al. Pleural effusion associated with hepatitis A. J Pediatr Inf. 2008;2:25–26. [Google Scholar]
- 10.Gürkan F. Ascites and pleural effusion accompanying hepatitis A infection in a child. Clin Microbiol Infect. 2000;6(5):286–287. doi: 10.1046/j.1469-0691.2000.00077-3.x. [DOI] [PubMed] [Google Scholar]
- 11.Goyal S, et al. Hepatitis A infection: an unusual cause of pleural effusion. 2015.
- 12.Ghosh A, Kundu P. Hepatitis A with superadded salmonella paratyphi A infection presenting with exudative pleural effusion and acalculous cholecystitis. Indian Pediatr. 2017;54(6):514–515. [PubMed] [Google Scholar]
- 13.Mehta K, et al. Hepatitis a associated with chylothorax: an uncommon presentation of a common infection. J Trop Pediatr. 2015;61(6):468–473. doi: 10.1093/tropej/fmv059. [DOI] [PubMed] [Google Scholar]
- 14.Dalai R, et al. A rare case of childhood hepatitis A infection with pleural effusion, acalculous cholecystitis, and ascites. J Fam Med Prim Care. 2018;7(6):1581. doi: 10.4103/jfmpc.jfmpc_298_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Saha S, Sengupta M. Anasarca—an atypical presentation of hepatitis A. East J Med. 2012;17(3):133. [Google Scholar]
- 16.Roy S. Hepatitis A presenting as bilateral pleural effusion in two pediatric patients. Indian J Case Rep. 2017;3:145–147. doi: 10.32677/IJCR.2017.v03.i03.011. [DOI] [Google Scholar]
- 17.Owen RL, Shapiro H. Pleural effusion, rash, and anergy in icteric hepatitis. N Engl J Med. 1974;291(18):963–963. doi: 10.1056/NEJM197410312911810. [DOI] [PubMed] [Google Scholar]
- 18.Allen O, et al. A very rare complication of hepatitis A infection: acute myocarditis—a case report with literature review. Case Rep Med. 2018 doi: 10.1155/2018/3625139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Selimoğlu MA, et al. A rare complication of hepatitis A: pleural effusion. J Emerg Med. 2005;28(2):229–230. doi: 10.1016/j.jemermed.2004.11.008. [DOI] [PubMed] [Google Scholar]
- 20.Erdem E, et al. Hepatitis A with pleural effusion, ascites and acalculous cholecystitis. Iran J Pediatr. 2010;20(4):479. [PMC free article] [PubMed] [Google Scholar]
- 21.Kaman A, et al. A case of pleural effusion and ascites associated with hepatitis A/Hepatit A Ile Iliskili Plevral Efüzyon ve Asit Olgu Sunumu. Cocuk Enfeksiyon Dergisi. 2016;10(1):33. doi: 10.5152/ced.2015.1781. [DOI] [Google Scholar]
- 22.Vaidya P, Kadam C. Hepatitis A: an unusual presentation. Indian Pediatr. 2003;40(9):910. [PubMed] [Google Scholar]
- 23.Bukulmez A, et al. Pleural effusion: a rare complication of hepatitis A. Indian J Med Microbiol. 2008;26(1):87. doi: 10.1016/S0255-0857(21)02005-3. [DOI] [PubMed] [Google Scholar]
- 24.Hadgu FB, Alemu HT. Pleural effusion associated with anicteric hepatitis A virus infection—unusual manifestation of a common disease: a case report. Pediatr Health Med Ther. 2020;11:189. doi: 10.2147/PHMT.S251393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Iza Palacios L, Espinoza-Ríos J. Hepatitis A and hepatitis E virus co-infection with right pleural effusion, ascites and acute acalculous cholecystitis. A case report. Rev Gastroenterol Perú. 2020;40(1):77–79. doi: 10.47892/rgp.2020.401.1035. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon reasonable request.


