Abstract
Most women in the United States do not meet the recommendations for healthful nutrition and weight before and during pregnancy. Women and providers often ask what a healthy diet for a pregnant woman should look like. The message should be “eat better, not more.” This can be achieved by basing diet on a variety of nutrient-dense, whole foods, including fruits, vegetables, legumes, whole grains, healthy fats with omega-3 fatty acids that include nuts and seeds, and fish, in place of poorer quality highly processed foods. Such a diet embodies nutritional density and is less likely to be accompanied by excessive energy intake than the standard American diet consisting of increased intakes of processed foods, fatty red meat, and sweetened foods and beverages. Women who report “prudent” or “health-conscious” eating patterns before and/or during pregnancy may have fewer pregnancy complications and adverse child health outcomes. Comprehensive nutritional supplementation (multiple micronutrients plus balanced protein energy) among women with inadequate nutrition has been associated with improved birth outcomes, including decreased rates of low birthweight. A diet that severely restricts any macronutrient class should be avoided, specifically the ketogenic diet that lacks carbohydrates, the Paleo diet because of dairy restriction, and any diet characterized by excess saturated fats. User-friendly tools to facilitate a quick evaluation of dietary patterns with clear guidance on how to address dietary inadequacies and embedded support from trained healthcare providers are urgently needed.
Recent evidence has shown that although excessive gestational weight gain predicts adverse perinatal outcomes among women with normal weight, the degree of prepregnancy obesity predicts adverse perinatal outcomes to a greater degree than gestational weight gain among women with obesity. Furthermore, low body mass index and insufficient gestational weight gain are associated with poor perinatal outcomes. Observational data have shown that first-trimester gain is the strongest predictor of adverse outcomes. Interventions beginning in early pregnancy or preconception are needed to prevent downstream complications for mothers and their children. For neonates, human milk provides personalized nutrition and is associated with short- and long-term health benefits for infants and mothers. Eating a healthy diet is a way for lactating mothers to support optimal health for themselves and their infants.
Keywords: adolescent pregnancy, developmental origins of disease, fetal and neonatal nutrition, gestational diabetes mellitus, lactation, macronutrients, maternal nutrition, micronutrients, nutritional requirements, pregnancy, vitamin supplementation
Introduction
The reproductive period is a critical time for establishing risks of chronic diseases in offspring later in life.1 Nutrition plays a vital role during this developmental period, and because it is a determinant of lifetime risk of disease, it is potentially a modifiable risk factor. Although the World Health Organization (WHO) provides guidelines for antenatal care,2 comprehensive guidelines detailing nutritional needs of women throughout reproduction from preconception through pregnancy and lactation are lacking.
The role of optimal nutrition for the continuum beginning at preconception, during pregnancy, at birth, and beyond extending through childhood and adolescence has received too little attention from researchers, clinicians, and policy experts in the past but has recently become a frequent topic of discussion, including a recent National Academies of Science, Engineering, and Medicine workshop.3 The need for additional well-designed research on this topic became apparent in a recent series of systematic reviews from the US Department of Agriculture (USDA): Nutrition Evidence Systematic Review, Pregnancy and Birth to 24 Months Project.4 Of note, 29 of the most important questions related to pregnancy and infant milk-feeding practices were systematically reviewed, with the highlights related to nutrition during pregnancy presented in Figure 1. Each topic was summarized by a conclusion statement and was assigned a grade based on the quality of evidence. For 5 conclusion statements, a grade was not assignable, and the remaining 3 pregnancy questions received a grade that reflected only limited available evidence.5,6 For the infant milk topics, 4 received a grade indicating moderate evidence, 10 had limited evidence, and a grade was not assignable for 21 questions.7 The uncertain conclusions of the aforementioned systematic reviews underscored the need for more well-conceived studies to address specific questions regarding the role of nutrition in pregnancy. However, the inadequate numbers of studies capable of meeting the strict criteria of the reviews, do not negate the large number of robust studies on related topics, from which the scientific community may benefit. In this review, we have included such studies that give important insight into the many aspects of nutrition for women during their reproductive years.
FIGURE 1. Dietary patterns and maternal and birth outcomes: systematic review results.

GDM, gestational diabetes mellitus; HDP, hypertensive disorders of pregnancy; HHS, Department of Health and Human Services; NESR, Nutrition Evidence Systematic Review; USDA, US Department of Agriculture.
In the past, public policy guidelines did not include pregnant or lactating women or infants under the age of 2. Fortunately, the 2020 to 2025 Dietary Guidelines for the first time include recommendations for infants, toddlers, and pregnant women that will provide added benefits for healthcare professionals and the public. The USDA released its final guideline document (USDA 2020–2025 Dietary Guidelines) in December 2020.8 Although this report was not available at the time of the Nutrition in Pregnancy: Lifelong Impact conference in 2019, which was the motivation for this review, the findings and recommendations of this document were nevertheless consistent with the new USDA guidelines.9 Other reviews on this topic bring additional clarity to the issue.10
The conclusions offered herein come from recommendations from assembled experts on (1) the health benefits of consuming nutritious food before, during, and after pregnancy, (2) the value of promoting improved nutrition among pregnant women, and (3) the gaps in knowledge regarding nutrition during reproductive years that require urgent attention. Although the meeting was largely focused on women in the United States, there were also important insights from global partners.
Points of Agreement of Authors
Comprehensive improvements in nutrition and health status of women before and during pregnancy will contribute to optimal fetal growth, favorable obstetrical outcomes, improved perinatal survival, and the potential for better long-term health in both the mother and offspring.
Poor maternal nutritional status is causally associated with abnormal fetal growth patterns, including low birthweight (LBW; <2500 g), small for gestational age (SGA) (<10% birthweight for gestational age) or fetal growth restriction (FGR), macrosomia (>4–4.5 kg), and large for gestational age (LGA) (>90% birthweight for gestational age), each of which is associated with increased risks of developing childhood and adult chronic diseases.
The dietary patterns of pregnant adolescents are generally less healthy than those of pregnant adult women and are critically important during a time of continued maternal growth and development, indicating the need for enhancing diet quality among young pregnant mothers. Many adolescent mothers face multifaceted socioeconomic and lifestyle difficulties that require professional and social support to aid in optimizing their diets and other aspects of their health and social care before, during, and after their pregnancies.
The consumption of a beneficial dietary pattern before and during pregnancy is associated with a reduced risk of disorders of pregnancy, including gestational diabetes mellitus (GDM), preterm birth, obesity-related complications, and, in some populations, preeclampsia and gestational hypertension. Nutrition therapy provides the foundation for the treatment of GDM and is especially important for pregnant women with obesity who have undergone bariatric surgery or who have preexisting diabetes mellitus (DM).
A diet with balanced macronutrient intake provides the best chance for a healthy pregnancy and optimal perinatal outcomes. Nutritious diets are those that include ample quantities of vegetables, fruits, whole grains, nuts, legumes, fish, oils enriched in monounsaturated fat, and fiber and are lower in fatty red meat and refined grains. Furthermore, healthy diets avoid simple sugars, processed foods, and trans and saturated fats.
A diet that consistently and substantially restricts any macronutrient should be avoided during pregnancy. Fad diets as promoted by the popular press are widespread and may be especially harmful during pregnancy because of the resulting nutrient imbalance and consequent nutrient deficiencies or ketosis.
Growing evidence indicates that maternal prepregnancy body mass index (BMI) impacts the influence of gestational weight gain (GWG) on complications of pregnancy. Although the optimal time to improve maternal body weight and nutrition-related lifestyle is well before conception occurs, GWG goals, including a diet that limits nonnutritive, calorie-dense foods, maybe more achievable intervention targets for some women than weight modification before pregnancy.
Human milk is uniquely suited to meet the nutritional needs of normal infants born at term for the first 4 to 6 months of life, and its consumption during infancy is associated with lower risks of chronic disease in later life. Human milk composition is influenced by maternal dietary intake during lactation and maternal adipose nutrient stores, which together influence maternal milk and nutrient production and composition. Among women with GDM, there is evidence that exclusive breastfeeding for at least 6 months decreases the risk of type 2 DM (T2DM) for the mother and is protective for the risk of childhood obesity in her offspring.
The regular consumption of multivitamin and mineral supplements that contain optimal amounts of folic acid, among other micronutrients, is recommended for all reproductive-age women to augment a balanced diet, starting at least 2 to 3 months before conception and continuing throughout pregnancy until the cessation of lactation or at least 4 to 6 weeks after delivery. Women who become pregnant after bariatric surgery need additional supplements and close monitoring before and during pregnancy.
It is imperative that healthcare providers have the time, knowledge, and means to discuss optimal nutrition and provide educational support to women of reproductive age to improve their health before, during, and after pregnancy.
A review of the scientific bases for points of agreement is explained below.
Comprehensive Improvements in Nutrition and Health Status of Women Before Conception and During Pregnancy will Contribute to Optimal Fetal Growth, Favorable Obstetrical Outcomes, Improved Perinatal Survival, and the Potential for Better Long-Term Health in Both the Mother and Offspring
Background and current status
Recent national data suggested that many women in the United States do not meet the recommendations for healthful weight and nutrition before and during pregnancy. As of 2019, 29% of women met the criteria for obesity before pregnancy, which increased by 11% from 2016. Overall, only 32% of US women gain weight within the recommended range during gestation, and the distributions of low or excessive weight gain varied accordingly by prepregnancy BMI.11 In 2015, only half of US women surveyed met the guidelines for physical activity, and 29.7% of women reported taking a vitamin or folate supplement regularly before pregnancy.12 National data on food intake in US women before and during pregnancy women are limited; however, several reports have suggested that substandard quality diets are common.13–16 For example, between 2010 and 2013, a cohort of 7500 nulliparous women from 8 large US medical centers recalled their usual diet within 3 months of conception, and researchers assessed their diet quality using the Healthy Eating Index (HEI)-2010.14 More than half of the women reported an inadequate number of servings of the component food groups. The authors estimated that 39% of calories came from foods containing added sugars, solid fats, and alcohol, and the mean HEI score was only 63 of 100 points.14 When the same index was estimated for 795 pregnant participants in the National Health and Nutrition Examination Survey (NHANES; 2003–2012), the score was lower (poorer diet quality) at 50.7.13 In another recent analysis of pregnant women in NHANES, more than a third of the women reported diets below the estimated average requirement for key nutrients, such as vitamins D and E, iron, and magnesium, even with the use of dietary supplements, whereas 99.9% of the women reported diets too high in sodium.16 Social disadvantage plays a role in food behavior, and researchers have identified characteristics of US women, such as education level below a college degree and women of color who may be at the highest risk of less healthy intakes13–15 or low levels of nutritional biomarkers.13,17
Impact on pregnancy outcomes
In the 20th century, researchers and clinicians considered the fetus to be “a perfect parasite”18 who could meet its nutritional requirements in all but extreme famine.19 This perspective encouraged pregnant women to restrict their diet and minimize GWG in the middle 20th century.20 Infants with LBW were assumed to be “skinny” but “relatively untroubled.”19 However, current evidence found that maternal body size, dietary practices, and nutritional status before and during pregnancy are important factors for fetal health. Both inadequate nutrition and excessive nutrition and weight before and during pregnancy contribute to complications related to fertility (maternal and paternal); conception; development of the placenta, embryo, and fetus; fetal size; and perinatal complications, resulting in suboptimal pregnancy consequences for the mother and infant (Table 1).14,21–29,30,31 Animal models and human studies have suggested that maternal nutrition and maternal pre-pregnancy metabolic condition regulate fetal-placental gene expression, organ structures, metabolism, and growth during critical periods of development, affecting offspring risk of cardiovascular, metabolic, respiratory, immunologic, neuropsychiatric, and other chronic conditions starting during childhood development and into adulthood, with and without LBW.28,32–34 The intrauterine environment can establish poor trajectories of health that may be increased when nutrient restriction in utero is followed by postnatal nutrient excess.35,36 To illustrate, in Holland during World War II, where the population recovered from the Dutch Hunger Winter famine relatively quickly, exposure to famine early in pregnancy was associated with a higher risk of offspring obesity and cardiovascular disease (CVD) in adulthood, whereas exposure to famine in the second half of pregnancy led more commonly to T2DM.35
TABLE 1.
| Maternal | Child |
|---|---|
| Fertility | Fetal malformations and loss |
| Oocyte and embryo quality | Preterm delivery |
| Antenatal, intrapartum, and postpartum complications | Small for gestational age |
| Cesarean or operative delivery | Stillbirth |
| Lactation performance | Infant mortality |
| Depression | Rapid infant growth |
| Immediate and long-term obesity | Asthma and allergies |
| Development of noncommunicable diseases throughout the duration of life | Childhood obesity, adolescent, and adult obesity |
| Early age of menarche | |
| Neurocognitive, mental, and behavioral health | |
| Altered DNA methylation | |
| Development of noncommunicable diseases throughout the duration of life |
Opportunities for positive impact
Unfortunately, recognition of the importance of preconception nutrition, except for micronutrients, such as folate for the prevention of neural tube defects, is limited among healthcare workers, policymakers, and the public. The WHO Report of the Commission on Ending Childhood Obesity37 recognized preconception and pregnancy care as 1 of 6 key areas of action and called for clear guidance and support for the promotion of good nutrition and dietary counseling in antenatal care. Although healthy eating and physical activity counseling for adequate weight gain is recommended, the availability of effective support during pregnancy is limited. Weight gain in pregnancy in low-to-middle–income countries (LMICs) is not monitored routinely in some countries, and prepregnancy BMI is generally unknown. In addition, culturally acceptable, affordable, nutritious food supplements are urgently needed in areas where the prevalence of maternal undernutrition and poor food quality is high. Comprehensive improvements in nutrition and health status of women before conception and during pregnancy may have immediate effects on fetal growth, obstetrical outcomes, and perinatal survival. In a recently completed multicountry trial in which the effects of a comprehensive nutrition intervention initiated before conception was compared with the same intervention initiated late in the first trimester of pregnancy (vs no intervention), birth outcomes, including birth length and birthweight, LBW, SGA, and stunting, were strongly impacted by the nutritional intervention, with the largest effects in the preconception arm.38 Nulliparity and preconception anemia were strong effect modifiers of the response to intervention with more modest effects by baseline BMI.38,39 The WHO global guidance for antenatal care recommends several central nutritional and health interventions for a healthy pregnancy, including multiple micronutrient supplements containing iron and folic acid, calcium supplementation for the prevention of preeclampsia in low intake contexts, and balanced energy and protein supplementation for undernourished populations to reduce LBW.2
For women with easy access to low-quality food and who are overweight or have obesity, evidence to support preconception nutrition is insufficient and mostly observational. Limited evidence suggested that a specific benefit of a diet higher in vegetables, fruits, whole grains, nuts, legumes, and fish and lower in red and processed meats before and during pregnancy is associated with a reduced risk of hypertensive disorders of pregnancy (HDP) and GDM.5 Overweight and obesity are a major public health problem affecting more than two-thirds of women of reproductive age.40,41 Limited studies have shown improvement in maternal diet after preconception lifestyle interventions,42,43 but the field of published preconception prospective interventional trials has remained severely lacking.44
Poor and Inappropriate Maternal Nutritional Status is Causally Associated with Abnormal Fetal Growth Patterns Including Low Birthweight (<2500 g), Small for Gestational Age (<10% Birthweight for Gestational Age) or Fetal Growth Restriction, Macrosomia (>4–4.5 kg), and Large for Gestational Age (>90% Birthweight for Gestational Age), Each of Which is Associated with Increased Risks of Developing Childhood and Adult Chronic Diseases
Background and status
Examples of the powerful influence of maternal nutrition on fetal development are demonstrated by the pregnancy outcomes associated with neonates at the extremes of birthweight: (1) neonates below the 10th percentile in weight for gestational age at birth are defined as SGA, and (2) neonates born exceeding the 90th percentile in weight for age are defined as LGA. These birthweights represent, in part, the nutritional status of the mother before and during pregnancy but do not necessarily reflect infant body composition (lean and fat mass).45 One of the WHO’s global nutrition targets calls for a 30% reduction in LBW.46 A recent Lancet paper estimated that 20.5 million infants will be born with LBW globally; thus, progress toward achieving the target has been slow.47 Maternal nutritional status, including low and high prepregnancy BMI scores, inadequate weight gain, short stature, anemia, and micronutrient deficiency, were causally associated with LBW, which may be a result of preterm birth, impaired fetal growth, or both.
Impact of maternal nutrition on pregnancy outcomes
During extremes of maternal undernutrition, the fetus develops chronic FGR,48,49 a prime example of “survival at the expense of growth.” This phenotype includes decreased pancreatic growth, development, and insulin secretion; increased capacity for glucose uptake in peripheral tissues (such as skeletal muscle)50; reduced utilization of amino acids for protein synthesis and cell growth; and development of hepatic insulin resistance with increased glucose production in an ovine model that produced hypoxia and reduced nutrient supply in the fetus.51 We now know that the FGR phenotype, especially when followed by later life excess caloric intake, is a risk of the development of obesity, insulin resistance, and DM later in life.52,53 Unfortunately, no strategy has emerged that improves growth and development of the FGR fetus once diagnosed in pregnancy. Previous attempts (maternal oxygen supplementation, bed rest, augmented nutrition, and medications) have either not worked or caused harm.54 Therefore, current management of pregnancies with FGR involves fetal surveillance and delivery of the fetus when adverse physiology becomes apparent, in hopes that the neonate with FGR can be treated more effectively outside the uterus.55 Although there is no direct nutritional strategy for treating FGR, recent studies in sheep have revealed that uteroplacental gene therapy involving vascular endothelial growth factors safely increased fetal growth velocity and reduced the incidence of FGR.56 In addition, recent data indicated that nutritional support and exercise before pregnancy may be more efficacious in promoting healthy placentation and fetal growth than during pregnancy.57 Moreover, the current postnatal strategy in which infant weight is a primary criterion for neonatal intensive care unit or hospital discharge may contribute to excessively rapid catch-up growth, especially for body fat mass, as parents and providers are motivated to align newborn intake and nutrition to meet weight gain targets rather than to maintain normal fetal in utero growth trajectories.58,59
At the other extreme, fetal overnutrition from maternal obesity, DM, and high fat and sugar intake may result in macrosomia or LGA.60 These conditions that present excess glucose and lipid supply to the fetus are increasingly common and associated with several complications. Fasting and pulsatile postprandial hyperglycemia promote fetal insulin secretion, contributing to excess glycogen storage and fat accretion in the fetus, especially in pregnancies complicated by T2DM, GDM, and type 1 DM (T1DM), particularly when complicated by obesity.61 Although pregnancies complicated by DM are commonly associated with macrosomia and/or LGA, most cases of these infants are born to mothers with obesity alone, which now affects up to 1 of 3 women.40 Even greater fetal fat mass accumulation occurs with the combination of high maternal plasma glucose and lipid concentrations.61–63 Recent evidence suggested that maternal triglycerides, made available to the fetus by placental lipases that hydrolyze the triglycerides to free fatty acids (FFAs), are primary drivers of fetal fat mass growth in pregnancies with obesity and contribute to accelerated fat mass accumulation in the fetus.61,64,65 Fetuses have limited capacity for fatty acid (FA) oxidation66,67 but can store fat. Excess fat mass accreted in utero might contribute to later obesity, but postnatal fat mass accretion especially during the first 1 to 2 years of life can persist into later life leading to obesity in childhood. In a nonhuman primate model, a maternal Western-style diet resulting in intermittently higher postprandial glucose and lipid exposure to the fetus resulted in the 3-year-old offspring demonstrating higher glucose excursions. Furthermore, the child’s pancreatic islets secreted more insulin, suggesting that these islets were primed before birth to hypersecrete insulin.68 In contrast, extremely high and relatively constant glucose concentrations in the fetus actually can suppress insulin production and response to glucose stimulation.69 This, along with abnormal placentation and decreases in placental perfusion, may explain why some women with long-standing T1DM complicated by vascular diseases will have neonates who are not only SGA but also at increased risk of later metabolic disease, especially when exposed to an obesogenic environment.19,69
There is increasing evidence that persistent, very high fetal glucose concentrations can inhibit fetal neuronal development, leading to reduced neuronal number, dendritic proliferation, and synapse formation, ultimately leading to reduced cognitive function in such offspring later in their lives.70 In humans, a recent study in adolescent offspring from women with T1DM showed that cognitive function was significantly diminished, with lower intelligence scores and greater learning difficulties in the offspring whose mothers had more severe hyperglycemia associated with DM.71 Moreover, the rates of congenital heart defects and major malformations of the central nervous system derived from the neural tube, such as caudal regression syndrome, were higher in offspring of mothers with both T1DM and T2DM, and the risk period during organogenesis (<8 weeks) was often before women knew they were pregnant.72 However, stillbirth risk near term is the highest in mothers with T2DM, especially when associated with obesity,73,74 both conditions associated with excess maternal caloric intake and malnutrition.
In addition, maternal overnutrition plays an important role in the early origins of childhood obesity and inflammatory diseases, such as nonalcoholic fatty liver disease (NAFLD), the most common liver disease worldwide affecting 1 in 3 youth with obesity.75 A “multiple-hit” pathogenic model has been suggested to explain the progressive liver damage that occurs among children with NAFLD.76 Data in humans demonstrated that liver fat is 68% higher in neonates born to mothers with obesity and GDM and is strongly correlated with maternal prepregnancy BMI and, perhaps, maternal triglycerides before subcutaneous fat stores are fully developed.77,78 Moreover, evidence from the national pediatric nonalcoholic steato-hepatitis (NASH) network showed that high birthweight or LBW, even when adjusting for childhood BMI, doubles the risk of advanced fibrosis in youth with biopsy-confirmed NAFLD,79 suggesting that changes at birth may precede and possibly predict the rapid onset of NASH in at-risk youth for reasons that remain poorly understood. Without effective treatments, children with NASH are at risk of developing cirrhosis and liver-related mortality in early adulthood.
Opportunities for positive impact
All women of childbearing age should receive preconception counseling and guidelines on nutrition, physical activity, and optimal GWG, with particular attention to those with undernutrition or overnutrition; those with a prepregnancy BMI that indicates underweight, overweight, or obese status; and those with medical complications, including DM, prediabetes mellitus, insulin resistance, history of GDM, chronic hypertension, and any chronic medical disease (cardiopulmonary, obstructive sleep apnea, rheumatologic, NAFLD, gastrointestinal, etc.). Medical management of any chronic condition should be optimized before pregnancy, and women should be provided with options for effective contraception until the timing of pregnancy is optimal.
The Dietary Patterns of Pregnant Adolescents are Generally Less Healthy than Those of Pregnant Adult Women and are Critically Important During a Time of Continued Growth and Development, Indicating the Need for Enhancing Diet Quality Among Young Pregnant Mothers. Many Adolescent Mothers Face Multifaceted Socioeconomic and Lifestyle Difficulties that Require Professional and Social Support to Aid in Optimizing their Diets and Other Aspects of their Health and Social Care Before, During, and After their Pregnancies
Background and current status
The physiology of pregnancy may differ in adolescents from that in adult pregnant women. Young maternal age (particularly <16 years) is a significant risk factor for stillbirth, preterm delivery, LBW, and neonatal mortality.80–84 The probability of these adverse outcomes is the greatest when pregnancy coincides with continuing and/or incomplete growth of the adolescent mother.85,86 Sheep paradigms involving nutritional management of weight and adiposity in young biologically immature adolescents have replicated this competition for nutrients between the mother and offspring in the womb.87–89 Although poor nutrient reserves at conception play a modest role, dietary manipulation of the maternal growth trajectory during pregnancy has the most profound impact on pregnancy outcomes. Overfeeding adolescent sheep to promote rapid maternal growth during pregnancy is particularly detrimental. It leads to abnormal placental growth and development, reduced uteroplacental blood flow, and reduced fetal nutrient delivery.90 In the sheep model, these led to high rates of premature delivery of LBW lambs and increased rates of intrauterine growth restriction.91 In addition, initial lactation was impaired, and neonatal morbidity was high. In contrast, when maternal growth after conception is prevented by underfeeding adolescent sheep, the progressive depletion of the mother’s nutrient reserves resulted in only a small reduction in birthweight independent of any change in placental size or length of gestation.88 Appropriate caloric intake maintains maternal adiposity throughout gestation, and this optimizes fetoplacental growth and birth outcomes. Maternal and placental endocrine systems are differentially altered in both overnutrition and undernutrition with downstream effects on fetal endocrine systems, organ development, and body composition.88 Approaches to reverse these effects in sheep have been explored: notably, improving nutrition during late gestation in the undernourished model restores fetal nutrient supply, normalizes fetal adiposity, and partially restores birthweight.87,92 Following delivery, growth-restricted lambs of both sexes born to overfed adolescents and who are fed according to appetite have altered metabolic and body composition phenotypes, which persist into adulthood,93 whereas offspring of underfed adolescent sheep were largely unaffected.
Impact on pregnancy outcomes
This body of work using sheep models has public health implications for human adolescents living in both low- and high-income countries. Adolescents have been found to consume higher levels of snack and processed foods, less fruit and vegetables, and take fewer nutritional supplements than adult women.94 Irrespective of geographic location, both nutrient reserves at conception and gestational dietary intake are likely to be powerful determinants of fetal growth in very young girls whose own growth is still ongoing or incomplete.95 Data from human pregnancies in adolescent mothers concerning overnutrition and undernutrition remain limited.38,96
Opportunities for positive impact
In settings where women have chronically inadequate diets, intervening during pregnancy has shown limited benefit in perinatal outcomes.97,98 Intervention strategies among poorly nourished women are more effective if initiated months before conception.99 Adolescent nutrition has been neglected, particularly in LMICs.96 The Lancet Commission on Adolescent Health drew attention to both overnutrition and undernutrition burdens in this age group that composed approximately 18% of the world’s population.100 As adolescent girls have not historically been prioritized in global research, there is a significant data gap regarding the burden of underweight and stunting within adolescent girls in LMICs and the knowledge of interventions needed to optimize this period of rapid growth and development.95,101
High-energy nutritional intakes that promote rapid maternal growth during pregnancy in adolescents may constrain placental development and function and are potentially more detrimental than restricted nutritional intakes that prevent maternal growth.102 In areas where early marriage soon after menarche is the norm, there is evidence that girls with a low BMI should be advised to gain weight and achieve a normal BMI before conception to decrease the risk of preterm birth and neonatal underweight.103 Thereafter, dietary intakes should be sufficient to maintain maternal nutrient reserves throughout pregnancy. Where pregnancies are unplanned and food is readily available, biologically immature mothers and caregivers should be aware of the potential consequences of excessive GWG concerning placental development. Monitoring of placental size and uteroplacental blood flow may help identify those at risk of perinatal complications but is not yet of proven efficacy.
The Consumption of a Beneficial Dietary Pattern Before and During Pregnancy is Associated with a Reduced Risk of Disorders of Pregnancy, Including Gestational Diabetes Mellitus, Preterm Birth, Obesity-Related Complications, and, in Some Populations, Preeclampsia and Gestational Hypertension. Nutrition Therapy Provides the Foundation for the Treatment of Gestational Diabetes Mellitus and is Especially Important for Pregnant Women with Obesity Who have Undergone Bariatric Surgery or Who have Preexisting Diabetes Mellitus
Gestational diabetes mellitus
Nutrition therapy is the foundation for the treatment of GDM. Rooted in carbohydrate restriction, the rationale for this approach can be traced to the pre-insulin era, when restriction of carbohydrates to ≤ 10% of calories was among the few interventions that could prolong life in those with T1DM.104,105 Pioneers in the field of DM in pregnancy recognized that in utero environmental conditions that influence fetal growth are shaped by maternal nutrition.106 Moreover, contemporary evidence in the previous 2 to 3 decades has supported associations between fasting and postprandial glucose and infant birthweight, solidifying the need for control of maternal glucose to prevent fetal overgrowth in pregnancies affected by DM.107 The restriction of dietary carbohydrates leads to the risk of increasing dietary fat intake because of the replacement of carbohydrates with fat calories, particularly in obesogenic environments influenced by the easy availability of processed foods and low-carbohydrate fad diets promoted by the popular press.108 At the same time, mounting evidence supports that high saturated fat diets result in elevated FFAs, which inhibit insulin signaling and result in insulin resistance,109 which may increase fetal exposure to excess nutrients. Furthermore, fetal exposure to excess maternal lipids, especially triglycerides, is linked with fetal overgrowth and excess adiposity, both potent predictors of later childhood obesity and metabolic disorders.61,105,110 In 2005, the American Diabetes Association acknowledged the concern for excess fetal lipid exposure secondary to maternal diet, de-emphasizing restriction of carbohydrate,111 and worldwide, there is no consensus on the optimal approach to the treatment of GDM with nutrition therapy.112,113
When a woman receives a diagnosis of GDM, regardless of the exact diagnostic criteria, nutrition therapy is the first line of treatment.114 There is high hope across the field that nutrition therapy alone in the absence of adjunct treatment with insulin or oral diabetes agents can effectively and economically treat the growing number of women with GDM. More recent data underscored the metabolic similarities in patterns of glycemia and lipidemia between diet-controlled GDM and maternal obesity without GDM.115,116 This highlights an opportunity to more thoughtfully target women with obesity outside of GDM for treatment with nutrition. Moreover, the importance of good nutrition for all pregnant women was recently highlighted by data demonstrating strong associations between fasting and postprandial triglycerides and neonatal adiposity in both women with normal weight and women with obesity (without GDM), further supporting a role for targeting these nutrition-sensitive indicators.63 Unfortunately, randomized controlled trials (RCTs) using diet and lifestyle changes, although resulting in slightly less GWG, have overall not been successful in preventing GDM.117
Currently, the evidence does not support 1 particular nutritional approach to the treatment of GDM. It was recently shown that, globally, advice for nutrition in GDM is mixed between carbohydrate restriction and more liberal carbohydrate intake, with the focus on choosing low glycemic index foods, consuming more complex carbohydrates, increasing dietary fiber, and limiting consumption of saturated fats.118 The quality of the available evidence is poor, with high heterogeneity across studies, lack of control for confounding medications, poor reporting of GWG, and low dietary compliance.112,114 Very recently, a controlled trial in which women with GDM were randomized to a lower carbohydrate and higher fat (40% carbohydrate and 45% fat) diet vs a higher-complex carbohydrate diet (60% carbohydrate and 25% fat) (both eucaloric and all meals provided for the duration of pregnancy) found no difference in birthweight, newborn adiposity by Peapod, or cord C-peptide supporting that complex carbohydrate can be liberalized by 20% above conventional recommendations and similarly normalize fetal growth, expanding nutrition options in GDM.119 A recent meta-analysis across 18 RCTs and 8 diet patterns for nutrition in GDM demonstrated that any modification that improves nutritional quality and intake after GDM diagnosis is effective in reducing fasting and postprandial glucose and lowering infant birthweight.120
Pregnancy after bariatric-metabolic surgery
Preconception weight loss for women with obesity holds great promise to improve maternal and fetal health but is difficult to achieve through lifestyle alone. In contrast, bariatric-metabolic surgery (currently the most common being Roux-en-Y gastric bypass and, recently, sleeve gastrectomy) can result in total weight loss averages that approach 25% to 30% with accompanying benefits in, and often resolution of, most obesity-related comorbidities, including GDM.121 Greater numbers of women with severe obesity are now undergoing bariatric-metabolic surgeries and subsequently are becoming pregnant. Although meta-analyses of study outcomes of this population of mothers have typically demonstrated favorable outcomes concerning lower rates of HDP (62% lower), GDM (80% lower), and fewer babies born who are LGA (69% fewer); furthermore, they have reported a slight increase in preterm delivery (odds ratio [OR], 1.35) and a higher likelihood of SGA (OR, 2.16), especially when compared with women matched for presurgical BMI.122 The close timing of bariatric surgery concerning subsequent pregnancies in addition to the type of surgery is likely an important risk factor for SGA. Because women are in an active weight loss phase during the first year after bariatric surgery, pregnancy should be avoided.123 Furthermore, micronutrient deficiencies, such as iron, vitamin D, and vitamin B12, are common in patients who have undergone bariatric surgery, especially with Roux-en-Y gastric bypass, and must be adequately resolved before and during pregnancy. Less clear are the longer-term ramifications of postsurgical maternal weight, metabolic, and micronutrient changes on infant and childhood development and their risk of chronic diseases of adulthood (eg, obesity, DM, and CVD); however, the lower risk of GDM and LGA would seem to confer a benefit.
Preeclampsia and preterm delivery
The pathophysiology of preeclampsia is believed to be related to poor placentation accompanied by oxidative and endoplasmic reticulum stress in placental cells in addition to abnormal angiogenesis.124 These processes maybe modifiable by nutrition, and hence, a good deal of attention has been directed to the role of nutrition in preeclampsia. Unfortunately, these concepts have not been well studied, and in many cases, conclusions have been opposed (overnutrition125 vs undernutrition,126 too much127 vs too litde128 dietary salt, etc.). Current information on diet has recently been thoroughly reviewed (Figure 1),5 and the role of micronutrients in preeclampsia has been a subject of increasing scrutiny. However, because of the challenges of studying the role of nutrition in the prevention of preeclampsia, their relationship remains largely unresolved. In 4 studies of nutrition before and during pregnancy to modify preeclampsia and gestational hypertension risk, limited data suggested a reduced risk with a diet higher in vegetables, fruits, whole grains, nuts, legumes, fish, and vegetable oils and lower in meat and refined grains. This information was from healthy White European women with access to medical care.5 Data were insufficient to estimate this relationship in minority women or women of low socioeconomic status.
Micronutrient studies have provided a few helpful insights regarding their role in the prevention of preeclampsia. Calcium supplementation has been shown to be useful in settings with low calcium intake, leading to the conclusion that replacement, not supplementation, is relevant.129 Therapy with vitamins C and E as administered in several large studies has not proven effective to prevent preeclampsia.130,131 Several other micronutrients, including folic add,132,133 vitamins A and D,133 zinc,134 iodine,135 omega-3 FAs,136 and arginine,137 are supported by some, but not all, supplementation studies. A meta-analysis restricted to LMICs reported a significant effect of omega-3 supplementation on preeclampsia (risk ratio [RR], 0.40; 95% confidence interval [CI], 0.21–0.77; I2, 0%; 6 studies, n=1343), but there was no difference in severe preeclampsia, eclampsia, or gestational hypertension.136 Moreover, some intriguing possibilities deserve further studies, including periconceptional vitamins,138–140 dietary nitrates,141 reduced sodium intake,142 and antioxidants other than vitamins C and E, but thus far, none has been of proven benefit.143
Omega-3 supplementation reduced the risk of early preterm delivery at <34 weeks of gestation (RR, 0.42; 95% CI, 0.27–0.66; P=.0002; 6 studies, n=4193) and any preterm birth (RR, 0.83; 95% CI, 0.70–0.98; P=.03; 9 studies, n=5980) according to a systematic review. The effect persisted on sensitivity analysis when restricted to women with spontaneous preterm birth (RR, 0.44; 95% CI, 0.25–0.78; P=.005).144
A Diet with Balanced Macronutrient Intake Provides the Best Chance for a Healthy Pregnancy and Optimal Perinatal Outcomes. Nutritious Diets are Those that Include Ample Quantities of Vegetables, Fruits, Whole Grains, Nuts, Legumes, Fish, Oils Enriched in Monounsaturated Fats, and Fiber and are Lower in Fatty red Meat and Refined Grains. Furthermore, Healthy Diets Avoid Simple Sugars, Processed Foods, and Trans and Saturated Fats
Systematic reviews suggested that compared with women who follow the standard American diet consisting of highly processed foods, fatty red meat, and sweetened foods and beverages, women who report “prudent” or “health-conscious” patterns before and/or during pregnancy (seafood, poultry, whole grains, legumes, healthy fats, and fruits and vegetables) have fewer pregnancy complications and adverse infant and child health outcomes.5,6,29,145–149 Of note, 1 study of couples who consumed a Mediterranean diet during in vitro fertilization cycles found an increased probability of pregnancy (OR, 1.4; 95% CI, 1.0–1.9).150 However, large RCTs would add more specific recommendations, although it is improper to randomize women to diets before conception and during pregnancy if 1 diet is viewed as less healthy.
As there continue to be significant misconceptions about the safety of seafood intake during pregnancy, leading some pregnant women to avoid seafood altogether, it is important to emphasize the 2015 to 2020 Dietary Guidelines for Americans, supported by the US Food and Drug Administration and the Environmental Protection Agency, which recommend that women who are pregnant or breastfeeding consume between 8 to 12 ounces of a variety of seafood per week from choices that are lower in mercury (Figure 2).151 Low mercury fish choices include salmon, pollock, flounder, cod, tilapia, shrimp, oysters, clams, scallops, and clams. Fish provides important nutrients, including proteins, healthy omega-3 fats, iron, and vitamins B12 and D.
FIGURE 2.

Fish choice guide for pregnancy
In addition, omega-3 FFAs can be obtained through algae-based supplements, flax, hemp, and walnuts. Although seaweed is another source, iodine content can vary and may be excessive,152,151 and seaweed can contain environmental contaminants depending on where it is grown.153
A Diet that Consistently and Substantially Restricts any Macronutrient Should be Avoided During Pregnancy. Fad Diets as Promoted by the Popular Press are Widespread and may be Especially Harmful During Pregnancy Because of Resulting Micronutrient Deficiency or Ketosis
Significant imbalance of macronutrient intake may be associated with harm. For example, prepregnancy carbohydrate restriction has been associated with higher odds of neural tube defects (adjusted OR [aOR], 1.30; 95% CI, 1.02–1.67), although the data were limited by design.154 Furthermore, restriction of dietary carbohydrates elevates the risk of increasing dietary fat intake to replace calories lost, and high levels of saturated fats increase FFAs and insulin resistance.108,119,155 Offspring of mothers on a low-carbohydrate diet may be prone to gain weight in childhood, which may be epigenetically driven.156 Fetal exposure to excess lipids is linked to fetal overgrowth and excess adiposity, predictors of later childhood obesity and metabolic disorders.61–63 A ketogenic diet often minimizes carbohydrates and promotes the consumption of high protein, high-fat foods that maybe harmful114; extremes of protein intake have been associated with LBW.157 Placental-fetal glucose demands are thought to approach 150 g/day in later pregnancy, and recent data suggest that placental glucose consumption is higher than previously understood.158 Low-carbohydrate diets promote increased lipolysis and may promote starvation ketosis in pregnancy with unknown consequences to the fetus.108,110,114,115 The Paleo diet promotes consumption of excess saturated fats and restricts the consumption of dairy-based foods, which may contribute to deficiencies in calcium and vitamin D; moreover, the single published study of 76 women in pregnancy suggested possible improvements in glucose tolerance and anemia but was associated with lower birthweight.159 Maternal diet quality was recently shown to have some effect on infant adiposity at birth160; however, further studies and biomarkers are needed to better characterize maternal diet quality and its effect on newborn body composition.
Growing Evidence Indicates that Maternal Prepregnancy Body Mass Index Impacts the Influence of Gestational Weight Gain on Complications of Pregnancy. Although the Optimal Time to Improve Maternal Body Weight and Nutrition-Related Lifestyle is Well Before Conception Occurs, Gestational Weight Gain May be a More Achievable Intervention Target for Some Women than Weight Modification Before Pregnancy
Gestational weight gain: a critical appraisal of the Institute of Medicine guidelines
In 2009, the US Institute of Medicine (IOM) released evidence-based recommendations for optimal weight gain across pregnancy according to maternal prepregnancy weight status that have been broadly adopted by both clinicians and researchers in the United States and elsewhere. Recent evidence has shown that among women with obesity, the degree of prepregnancy obesity predicts adverse outcomes for pregnancy to a greater degree than does GWG.161 This adds more urgency to targeting interventions to help women achieve the healthiest possible weight before and between conceptions.21 Nonetheless, there is also evidence that low weight gain, especially in women who are underweight or women with normal weight, or excessive GWG is associated with adverse maternal and child outcomes.162,163 Offering opportunities during prenatal care for pregnant women may be a more feasible intervention than helping women optimize their weight before pregnancy.25
In the United States, the measurement of weight is routine at each prenatal care visit. However, this practice is not consistently found in all other countries,164 and even in the United States, real-time patient feedback and counseling related to weight tracking are not routinely practiced.165 Clinicians continue to identify insufficient time and knowledge related to counseling best practices as barriers to improving weight-related tracking and counseling.166 Furthermore, in generating weight gain guidelines, the IOM committee did not have adequate evidence to identify specific advice by subclasses of obesity. Therefore, they recommended at least 5 kg of weight gain for all women entering pregnancy with a BMI of >30 kg/m2, regardless of obesity class. Epidemiologic data published since then suggest that the ideal GWG varies by obesity class. For obesity grades I (BMI, 30–34.9 kg/m2) and II (BMI, 35.0–39.9 kg/m2), studies suggested that maternal gains less than the lower limit of the IOM recommendation may not increase adverse outcomes and may decrease LGA and GDM,161,163,167 although other studies indicated an increased risk of SGA and infant mortality with weight loss and very low weight gain.168–170 However, for women with obesity grade III (BMI, ≥40.0 kg/m2), lower levels of gain, or even weight loss, may be optimal, but the current evidence is observational and based on weight alone, not maternal diet or lifestyle behaviors.168,170,171
Because of insufficient evidence at the time, the 2009 guidelines did not provide evidence-based recommendations regarding diet or physical activity changes that would best help women to achieve recommended gains. The recent evidence report and systematic review for the US Preventive Services Task Force (USPSTF) found that counseling and active behavioral interventions to limit GWG were associated with lower risk of GDM, macrosomia, LGA and emergency cesarean delivery and reduced GWG of −1.02 kg172 The led the USPSTF to issue a new recommendation statement that clinicians offer pregnant persons effective behavioral counseling interventions aimed at promoting healthy weight gain and preventing excessive GWG in pregnancy (B recommendation).173
As the IOM guidelines focused on high-resource settings, low-resource settings may need different standards to support women who are underweight and have low GWG. In LMICs, improved GWG (100 g/wk) was associated with significantly improved birthweight and length, as was baseline prepregnancy BMI, early weight gain, and GWG from 12 to 32 weeks of gestation.174
Human Milk is Uniquely Suited to Meet the Nutritional Needs of Normal Infants Born at Term for The First 4 to 6 Months of Life, and Its Consumption During Infancy is Associated With Lower Risks of Chronic Disease in Later Lite. Human Milk Composition is Influenced By Maternal Dietary Intake During Lactation And Maternal Adipose Nutrient Stores, Which Together Determine Maternal Milk and Nutrient Production and Composition. Among Women With Gestational Diabetes Mellitus, There is Evidence That Exclusive Breastfeeding For At Least 6 Months Decreases The Risk of Type 2 Diabetes Mellitus For The Mother And is Protective For The Risk of Childhood Obesity in Her Offspring
Human milk provides personalized nutrition and is associated with long-term health benefits for infants and mothers.175,176 According to the 2012 American Academy of Pediatrics policy statement, “Given the documented short- and long-term medical and neurodevelopmental advantages of breastfeeding, infant nutrition should be considered a public health issue and not only a lifestyle choice.”176 Milk composition is influenced by maternal dietary intake during lactation and maternal adipose nutrient stores, which together are responsible for the nutrients available for milk biosynthesis and ultimately maternal milk and nutrient production.177 To meet all infant nutritional needs, human milk is constantly changing, composition varies by infant age, between breasts, within a feed, throughout the day, during lactation, among women, and among populations. In light of these significant variations, accurate assessment of milk composition remains a challenge for researchers. The evaluation of donor milk pools assumed to come from women not only with adequate milk production to meet their infants’ nutritional needs but also with sufficient quantity to nourish other infants showed significant variations in composition. Among donor pools from the first and third quartiles, milk demonstrated up to a 33% difference in fat content, 22% difference in protein, and 16% difference in energy content.178 Importantly, individual women showed a greater difference in milk composition compared with variation by age of infant or length of time breastfeeding.178,179
Regarding individual macronutrients, maternal diet does not have a major impact on milk protein content or the total amount of fat in human milk, but it affects the types of FAs present in breast milk.180,181 Maternal adipose stores remain an important source of nutrients for human milk, although women with greater fat mass do not produce more or higher fat milk.182 Different lipids are the most variable component of human milk. Lipid composition variability is inversely related to the degree of breast fullness and milk volume. In addition to macronutrients and essential micronutrients, there is moderate evidence that flavors from the maternal diet during lactation are transferred into breast milk and that infants can detect diet-transmitted flavors,183 which may impact future taste preferences. The ratio of omega-6 vs omega-3 FAs in human breast milk seemed to promote postnatal fat development; however, this relationship requires further study.184 Interestingly, human milk from women with obesity or T2DM does not seem to expose the infant to a different macronutrient composition; however, it has been shown to have higher insulin levels and to influence the early infant microbiome population; moreover, any effects on infant appetite or growth remain unclear.185,186
Women with GDM who breastfeed have decreased risk of developing T2DM, with a longer duration and increased intensity of breastfeeding associated with a lower 2-year incidence of T2DM.187,188 Furthermore, breastfeeding is associated with a decreased maternal risk of metabolic syndrome,189 CVD,190 and cancer.191 Unfortunately, women who are overweight or women with obesity commonly experience difficulties in lactation and are less likely to meet exclusive breastfeeding goals,192 which suggests additional physiological barriers.
The Regular Consumption of Multiple Micronutrient Supplements That Contain Optimal Amounts of Folic Acid, Among Other Micronutrients, Is Recommended for All Reproductive-Age Women to Augment A Balanced Diet, Starting At Least 2 To 3 Months Before Conception And Continuing Throughout Pregnancy Until The Cessation of Lactation Or At Least 4 To 6 Weeks After Delivery
Evidence supports the benefit of comprehensive nutritional supplementation (multiple micronutrients plus balanced protein energy) associated with improved birth outcomes of major public health interest (eg, stunting, LBW, and SGA).193 This is supported by the 2020 WHO recommendation stating “antenatal multiple micronutrient supplements that include iron and folic acid are recommended in the context of rigorous research”2 for pregnant women and adolescent girls. Preconception folic acid is recommended for the prevention of neural tube defects.30 Routine supplementation is adequate regardless of the methylenetetrahydrofolate reductase geneotype.194 A recent Cochrane systematic review provided evidence that a daily, multiple micronutrient supplement containing iron-folic acid vs iron-folic acid alone significantly reduces the risk of LBW and SGA in LMIC.31 Well-nourished women who consume an adequate diet may not require additional multivitamin supplementation, but in the absence of comprehensive evaluation by a dietitian, routine supplementation is encouraged in the United States.195 Subgroups that particularly warrant targeted interventions for improving nutrition include nulliparous women and those who are anemic. Anemia in nonpregnant women has recently increased in the United States196 and is estimated to impact 38% of women on a global basis; the prevalence is much higher (> 50%) in certain regions, including South Asia and Central and West Africa.197 Anemia before pregnancy and in the first trimester of pregnancy has been associated with preterm delivery and LBW.198,199 Recently, a growing number of randomized controlled studies suggested that the supplementation of choline, especially in women with a history of alcohol use, may improve neurodevelopmental outcomes.200–203 These findings underscored the need for revising the current policy and recommendations for supplement use in pregnancy as an adjunct to the nutritious diet described previously, as supplements alone cannot substitute for a healthy diet.
It is Imperative That Healthcare Providers Have The Time And Means to Provide Educational Support And to Discuss Optimal Nutrition With Women of Reproductive Age to Improve Their Health
Transformative change is needed for addressing women and girls’ nutrition as they hold roles in their communities that make them drivers of development as individuals and influencers of the health and well-being of their families. Optimal reproductive health can be achieved when maternal nutritional well-being exists. This occurs only when known nutrition interventions are integrally linked to health programs and delivered at scale. Global commitment and political will are needed for driving this agenda forward. Conference experts emphasized the need for preventive health services for women, including nutrition advice during the reproductive cycle. They cited a linkage of individual health behavior change and supportive policy and healthcare environment.204 Thus, greater efforts supporting interventions that provide wholesome nutrition and total micronutrient support are needed. This support will ensure that more women who will become pregnant will experience robust placentation and embryogenesis, resulting in lower risks of diseases in their offspring because of optimal epigenetic regulation of organs.205
Key Questions
Question 1: What are the unique nutritional requirements of a normal pregnant woman and what unique features of diets produce optimal health and growth of her fetus and infant?
The WHO defines good nutrition as “intake of food necessary for optimal growth, function, and health. Good nutrition is defined as a well-balanced diet that provides all essential nutrients in optimal amounts and proportions, whereas poor nutrition is defined as a diet that lacks nutrients (either from imbalance or [from] overall insufficient food intake) or one in which some components are present in excess.”26 Additional features of a healthy diet include foods that are accessible, acceptable, affordable, safe, culturally appropriate, and composed primarily of whole foods consumed in moderation.
There is growing evidence that diet and nutritional status at preconception, starting as early as childhood and adolescence, seems to be equally or even more important26 than during pregnancy, because of growing evidence that nutrition affects fertility and the early development of the placenta and fetus, which occur well before a woman recognizes that she is pregnant21 About half of US women of childbearing age consume unhealthy diets that are too high in processed ingredients, fat, sugar, and other refined carbohydrates and do not meet current nutritional recommendations.13,14,16
Although it is commonly said that pregnant women are “eating for two,” for most women, average energy requirements increase only modestly.206 In contrast, assuming that the preconception diet was adequate, prenatal needs for some micronutrients, for example, folate and iron, increase by one-third to one-half, respectively.207 The WHO has declared iodine deficiency as the single most common cause of brain damage, after starvation, and mild iodine deficiency is still a public health concern in both developing countries and Western industrialized nations, especially Europe.208 Iodine requirements are increased during pregnancy and in nursing mothers from 250 to 300 μg per day (compared with 150 μg outside of pregnancy). These requirements begin very early because of the fetal need to synthesize thyroid hormone, critical for early neurogenesis, proliferation migration, differentiation, neurite outgrowth and guidance, synaptogenesis, and myelination. The fetal thyroid begins to concentrate iodine at 10 to 12 weeks of gestation and begins making thyroid hormone with complete independence from maternal thyroid hormone production by 18 weeks of gestation.209,210 It has been demonstrated that mild-moderate iodine deficiency, which is common in pregnancy, is associated with a 10-point decrease in total intelligence quotient score and an increase in attention-deficit and hyperactivity disorder in the offspring.211
Women and providers commonly ask what a healthy diet for a pregnant woman should look like and the message to US women should be “eat better, not more.” This can be achieved by basing the diet on a variety of nutrient-dense, whole foods, including fish, fruits, vegetables, omega-3 FAs, and whole grains, in place of poorer quality processed foods and beverages to enhance nutritional quality without excessive energy intake (USDA Dietary Guidelines) (Figure 3; Table 2). Maternal requirements vary by individual characteristics, and in addition to considering dietary quality before pregnancy, factors, such as maternal body size, age, gestational age, multiple gestation, activity level, and medical conditions, should be considered. The USDA provides interactive online tools for health professionals to tailor dietary recommendations for women before and during pregnancy and the MyPlate interactive tool that women can use to plan their diets (https://www.choosemyplate.gov/browse-by-audience/view-all-audiences/adults/moms-pregnancy-breastfeeding). Table 3 shows the primary features of a healthy diet for discussing a healthy diet with patients, and Tables 4 and 5 shows questions as conversation starters for healthcare providers when talking to patients.
FIGURE 3.
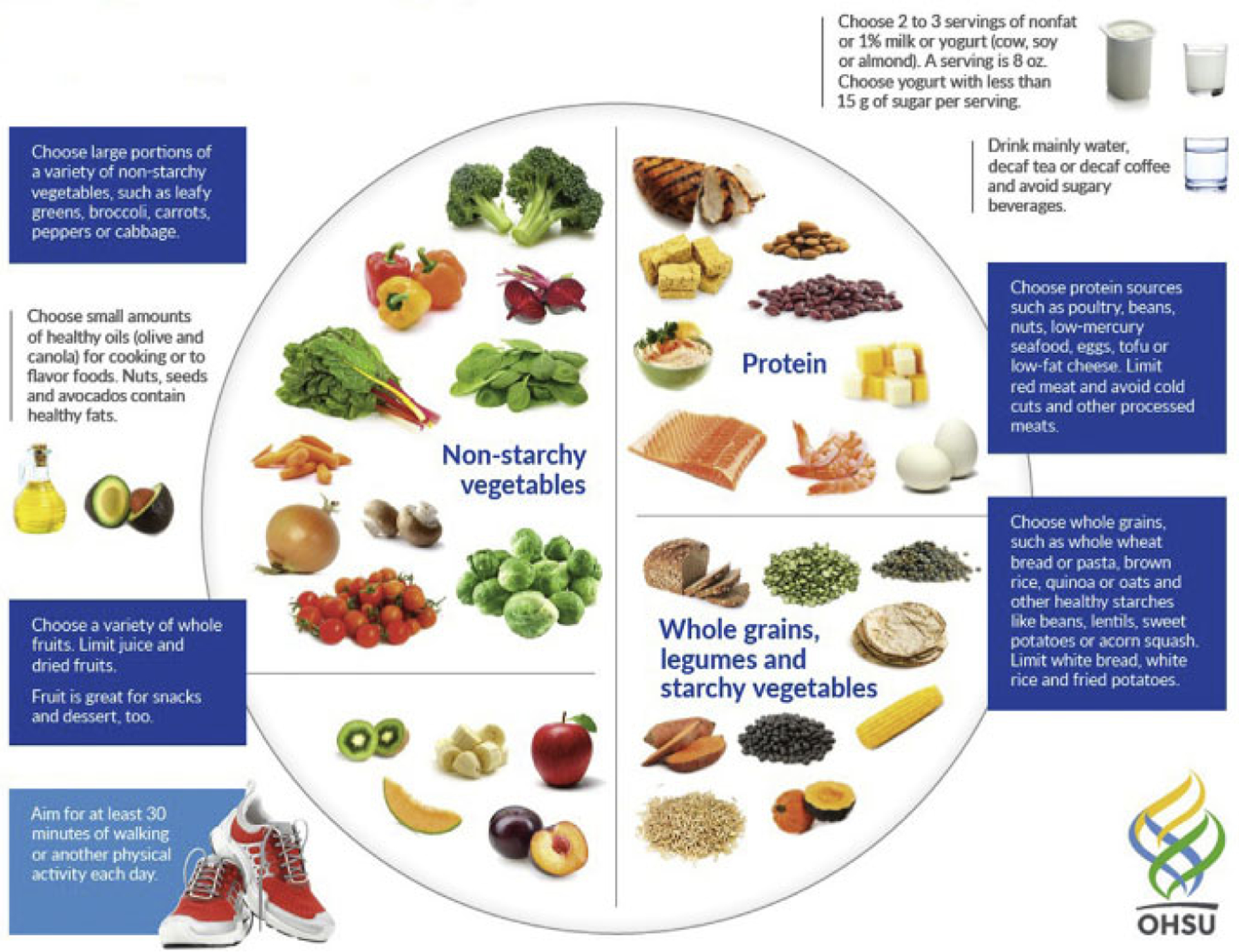
My pregnancy plate
TABLE 2.
Recommended reproductive diet patterns
| Name | Includes | Excludes | Benefits | Risks |
|---|---|---|---|---|
| Optimal diets | ||||
| Mediterranean diet | Plant-based foods—vegetables, fruits, whole grains, legumes, nuts, herbs, spices, olive oil, fish, poultry, and red wine Up to 40% calories from fat |
Limits red meat a few times per month | Reduces the risk of CVD, mortality, cancers, and cognitive diseases | |
| Dietary approaches to stop hypertension | Balanced complex carbohydrates (58%), lower fat (28%), and moderate protein (18%) High in fiber, calcium, phosphorus, magnesium, and potassium |
Low in cholesterol, fat, and sodium | Reduces weight, lowers BP and cholesterol, and reduces the risk of CVD and bone loss | Needs vitamin D supplementation |
| Flexitarian diet | Vegetarian most of the time, more vegetables, whole grains, plant-based or nonmeat proteins (“new meat”), dairy, and “sugar and spice” Focus on home prepared food with <5 ingredients |
Meat and dairy in moderation if at all | Lowers BP and cholesterol, reduces weight, and reduces the risk of heart disease, stroke, and diabetes mellitus | May need calcium, vitamin B12, and iron supplementation |
| Nordic diet | Fruits, vegetables, legumes, potatoes, whole grains, nuts, seeds, rye bread, fish, seafood, low-fat dairy, herbs, spices, and canola oil | Rare red meat and animal fats No sugar-sweetened beverages, added sugars, processed meats, and refined fast foods | Reduces weight and lowers BP and inflammatory markers | |
| Diets to avoid during pregnancy | ||||
| Atkins diet 20-40-100 | Low carbohydrate (20 g), high fat, beef, pork, poultry, fish, eggs, cheese, and sources of fat | Limit starchy vegetables, grains, legumes, simple sugars, and milk | Needs vitamin C, B vitamins, folate, calcium, and magnesium | |
| Paleo diet | Lean meats, fish, eggs, nuts, seeds, fruits, vegetables, and oils | Processed foods, wheat, other grains, legumes, dairy, potatoes, refined sugar, salt, and refined oils | Reduces weight and lowers the risk of diabetes mellitus, heart disease, and cancer | Needs calcium, B vitamins, andwhole grain nutrients |
| Ketogenic diet | Extreme carbohydrate restriction ketosis, skin-on poultry, fattier beef, pork, fish, green leafy vegetables, oils, and solid fats | Avoid starchy root vegetables, bread, pasta, other grains, and fruit | Reduces weight | Not recommended in pregnancy because of altered neonatal brain development Needs vitamin C, B vitamins, folic acid, calcium, and fiber |
BP, blood pressure; CVD, cardiovascular disease.
TABLE 3.
Common ground for healthy dietary patterns
| • Whole, unprocessed foods and beverages |
| • Rich in fruits and vegetables |
| • Whole grains and complex carbohydrates, including ancient grains |
| • Healthy fats (monounsaturated and polyunsaturated), including nuts and seeds |
| • Healthy fish |
| • Plant-based protein |
| • Drink more water |
| • Lean meats and dairy products |
TABLE 4.
Nutrition conversation starters
| • Tell me about the foods you usually eat. |
| • Are there foods that you tend to avoid? |
| • Do you prepare your own food? |
| • How many times a week do you eat foods that you did not prepare yourself? |
| • Do you think you eat a healthy diet? Why or why not? |
| • When asked about GWG, respond with “How do you feel about your food intake?” Strategize about ways to improve nutrition for maternal and fetal health and appropriate GWG. |
| Food insecurity statements to ask |
| • Within the past 12 mo, the food we bought just did not last and we did not have the money to get more. |
GWG, gestational weight gain.
TABLE 5.
Glossary
| Healthy Eating Index | A measure of diet quality used to assess how well a set of foods aligns with key recommendations of the Dietary Guidelines for American (https://www.fns.usda.gov/resource/healthy-eating-index-hei) |
| Estimated average requirement | A nutrient intake value that is estimated to meet the requirement of half the healthy individuals in a group (https://www.ncbi.nlm.nih.gov/books/NBK45182/) |
| Ketosis | A metabolic state in which fat provides most of the fuel for the body |
| Micronutrients | Vitamins and minerals required in trace amounts for the normal growth and development of living organisms |
| Malnutrition | Imbalanced nutrition |
| Nutrient dense | Food relatively rich in nutrients for the number of calories contained |
| Undernutrition | Lack of proper nutrition, caused by not having enough food or not eating enough food containing substances necessary for growth and health |
| Overnutrition | A form of malnutrition arising from excessive intake of nutrients, leading to an accumulation of body fat that impairs health |
| Prebiotics | Foods that act as food for human microflora |
| Probiotics | Foods or supplements that contain live microorganisms intended to maintain or improve the normal microflora in the body |
| Processed food | A food item that has had a series of mechanical or chemical operations performed on it to change or preserve it |
| Whole foods | Food with little or no refining or processing and containing no artificial additives or preservatives; natural or organic food |
Question 2: What is the optimal balance of macronutrients during pregnancy and lactation to support and maintain appropriate nutrient supply to the infant through lactation?
Diet planning is especially important for women planning to conceive, during pregnancy, and during lactation. Dietary patterns are an evolving area of research involving the entirety of the diet rather than focusing on individual nutrients or foods. There is limited but consistent evidence primarily in healthy White women with access to healthcare that dietary patterns before and during pregnancy higher in vegetables, fruits, whole grains, nuts, legumes, fish, and vegetable oils and lower in meat and refined grains are associated with a reduced risk of disorders of pregnancy, including preeclampsia, gestational hypertension, GDM, and preterm birth.4–6 Conclusions about the association between dietary patterns during pregnancy and birthweight outcomes is less consistent and restricted by inadequate adjustment of birthweight for gestational age and sex variation in study design, lack of measures of infant body composition, dietary assessment methodology that rely on recall rather than robust biomarkers of nutritional intake, and adjustment of key confounding factors.6 However, in a recent cohort of 354 fully breastfeeding mother-infant dyads, in utero exposure to a higher-quality maternal diet, based on the HEI-2015,212 was inversely associated with the percentage of infant fat mass.213 Avoiding simple sugars, processed foods, and trans and saturated fats and limiting red and processed meats are recommended. A diet that severely restricts any food group should be avoided, specifically the ketogenic diet that lacks carbohydrates, the Paleo diet because of dairy restriction (promoting deficiencies in calcium and vitamin D), and any diet containing excess saturated fats. Foods with a high saturated fat content containing high omega-6 vs omega-3 FAs constitute a proinflammatory diet, with some evidence of impact on obesity in the offspring and increased postnatal adipose tissue development.184,214
Literature on the effects of nutrition of lactating women on their infants’ later health is scarce. Eating a healthy diet is a way for lactating mothers to support their health and the health of their infants during lactation. The quality of a mother’s diet, particularly dietary fats, including saturated vs unsaturated fat, is an important way for mothers to ensure a healthy start for infant growth and development.
What is the best way to ensure that the embryo and fetus have adequate micronutrients throughout gestation?
A balanced diet that includes fish twice a week for docosahexaenoic acid and whole grains for folate, vitamin B12, iron, and choline is recommended as is supplementation of iodine by 150 μg to ensure that pregnant women have a total intake of 250 μg/day.209 For women with dietary restrictions, consultation with a nutritionist is recommended. A daily multivitamin that contains optimal amounts of folic add and iron is recommended for women who are planning to become pregnant. Because only a small percentage of women will eat a complete diet that includes all required nutrients, appropriate dietary supplementation with optimal levels of iron and folic add and other micronutrients is recommended. A dietitian may recommend other supplementation regimens for women at risk of insufficiency of specific micronutrients.
Question #3: Should all pregnant women receive dietary assessment and personalized advice? If so, how can we educate a professional workforce to provide such advice?
The relevance and practicality of routine nutritional assessment for all pregnant women have been the focus of debate among healthcare professionals, researchers, and policymakers. The reluctance in providing universal nutritional assessment is partly owing to resource and cost implications, but it is, in part, owing to the lack of robust evidence of effectiveness4 or ambiguity about the acceptability of current nutritional assessment techniques among pregnant mothers and their care providers.215
However, in addition to the widely recognized critical role of nutritional health during gestation, pregnancy is seen as an opportune period to influence not only the women’s lifestyle but also their families’ lifestyle for healthier outcomes.216 Therefore, it is important to assess the pregnant women’s nutritional status before or early in pregnancy to provide optimized care for mothers and their families.
Nutritional assessment during pregnancy
Nutritional assessments are carried out in various ways, including anthropometric measurements, dietary intake evaluations, and assessment of blood biomarkers. Each approach provides specific information on certain aspects of nutritional status, and all approaches have their limitations and advantages. Biochemical markers are direct ways of assessing the adequacy of maternal nutrients and minerals. Although some biomarkers, such as plasma hemoglobin levels, are commonly screened as an indication of anemia, these tests are not routinely performed for all nutrients and minerals because of not being universally cost-effective or practical in most settings. Ferritin can be used to screen for iron deficiency, although the recommended cutoff for supplementation varies.217 Furthermore, the assessment of dietary FA ingestion reflected by mass spectrometry red blood cell (RBC) FA analyses, which reflect 24 FA species and polyunsaturated essential omega-6 and omega-3 FA intakes, is not usually performed in most studies, although the analysis can now be done on a single blood spot and cost has become reasonable.218–220 Assessing the mothers’ dietary intakes are challenging, requiring a considerable amount of additional time, resource, knowledge, and skills for practitioners. In addition to these challenges, most healthcare professionals who care for women during pregnancy are ill prepared to analyze dietary intake information and to provide appropriate advice and support to women when dietary inadequacies are identified.221 Until there are user-friendly tools to facilitate a quick evaluation of dietary patterns with clear guidance on how to address dietary inadequacies and embedded support from trained healthcare providers, it is unlikely that nutritional assessments will become routine in clinical practice.
The most commonly used method of anthropometries includes measuring weight and height at the first antenatal (booking) visit to calculate maternal BMI as an indicator of nutritional status. However, in some countries, follow-up weight assessments are not recommended or practiced, citing a lack of evidence on the benefits of routine weighing during pregnancy, a lack of time or equipment, and concerns about anxiety that it may cause for mothers.164 In a thematic analysis of 400 posts made in a UK-based parenting Internet forum in the week after the publication of the National Institute for Health and Clinical Excellence (NICE) guidance on weight management in pregnancy in July 2010,222 concerns were expressed by women about feeling patronized if just being told about the risks of obesity and excessive GWG without clear guidance and support to manage it effectively.223 Thus, the value of routine weight measurements during pregnancy without sensitive and helpful follow-up is a matter of debate by many healthcare professionals and investigators.224,225
Changes in the policies of governmental and professional organizations to improve nutritional health of women before, during and after pregnancy
The importance of maternal nutrition concerning pregnancy health and intrauterine fetal growth and beyond is widely recognized. However, there is a great deal of variation in policies and practices within and among countries concerning nutritional assessment and related care of women during the perinatal period. Several initiatives and organizations across the globe have attempted to address the growing nutritional challenges among maternity populations, including the National Academy of Medicine (formerly the IOM), NICE,222 and Think Nutrition First.26 Furthermore, there are initiatives, such as those in the United Kingdom “Every Contact Counts,” with the aim of promoting a healthy lifestyle at every opportunity in which patients and mothers attend clinics or visit healthcare providers.226 However, there are evident inconsistencies in recommendations and practices that are counterproductive in achieving optimum lifestyle and nutritional health during the reproductive period. A lack of sufficient evidence in clinically meaningful and/or locally sensitive and effective gestational weight management4 approaches has been cited as the main reason for variation in current nutritional assessment and relevant care and management. Providing nutritional education and introducing interventions before pregnancy particularly from adolescent stages227,228 through pregnancy and using digital sources for wider engagements are suggested.229
There is an urgent need for further research in providing culturally sensitive and effective interventions in promoting a healthy lifestyle and reliable nutritional assessment during the perinatal period.
Question #4: How should we address the nutritional needs of special populations of women, including those regularly consuming too many or too few calories or inadequate nutrients?
Special populations of women and pregnant women with unique nutritional needs have been identified to include adolescent girls, women with GDM, women with pregestational DM, women who are overweight, women with obesity (particularly severe obesity), women with preeclampsia, and women who are underweight. It is strongly recommended to develop strategies for targeting these special populations by individual groups to address the uniqueness of each condition.
Major Research Gaps and Recommendations for Future Directions
The huge body of animal work, human observational studies, and a growing number of experimental trials aimed at understanding how maternal nutrition matters are exciting, but there are many challenges in conducting research in the field of nutrition among pregnant women. We are still in the early stages of accruing the kind of causal, consistent, nuanced data, preferentially based on more robust dietary biomarkers that are needed to confidently create interventions and policies.28,32,33,230 Animal studies provide mechanistic explanations for the developmental features of imparting disease risk and associated epigenetic changes, but animal findings require demonstration in humans before they are applied clinically. This is particularly the case in adolescent pregnancies in which the human data are limited. Observational epidemiologic studies with retrospective designs, such famine cohort studies and prospectively collected cohorts, have contributed tantalizing evidence supporting the animal work, but at best, they demonstrated correlative relationships because nutritional exposures often track with other social and environmental exposures. Diet is difficult to measure accurately, and the essential window of nutritional exposure may not be known or may vary by outcome studied. Even for well-designed prospective cohort studies, nutritional measures may begin after the critical window of exposure or may be limited to a single point in time, confounded by subjective measures of dietary recall, which does not reliably capture the full extent of a woman’s intake or body weight. Maternal and child outcomes are multifactorial, making it difficult to identify the role of a particular exposure or the modifying effect of other exposures. Observational studies can be strengthened by new methods of analysis, including the use of sibling analyses that allow better control for shared genetics and environments. These approaches can better control for confounding but still do not rise to the level of establishing clear causality.230 RCTs do allow for causal inference but are often insufficient because the timing of the intervention that is feasible, such as the beginning of the second trimester of pregnancy, may miss the essential critical window of susceptibility. Moreover, studies are only capable of focusing on specific exposures during a limited time frame and are unable to control for subsequent exposures. Currently, it is not possible to conduct experimental studies that allow sufficient time to follow participants throughout their lifetimes, and it is not possible to randomize women of childbearing age to what are perceived to be less healthy diets. It is equally difficult to demonstrate the chain of events from a maternal intervention (such as reducing excessive GWG) throughout the child’s life (assessing metabolic markers) to the outcome of obesity or chronic disease development during childhood, adolescence, and adulthood, given the innumerable and heterogeneous developmental exposures throughout life. Multigenerational studies should be established to ensure that such information is available for the next generation.
Future studies will bring new information on the roles of newly recognized areas of medicine, such as the microbiome and diet, to the health of human offspring. The maternal microbiome is recognized as a key determinant of a range of important maternal and child health outcomes and, together with perinatal factors, influences infant health.231 The composition of the microbiome acquired in early infancy is critical for shaping infant and adult immune function and metabolic status.232 The mechanisms by which intestinal dysbiosis in early life contribute to postnatal inflammation and progression of the disease remain unclear. Metabolic plasticity has been proposed to underlie the observation that microbes critically contribute to the initiation of inflammation and progression to childhood noncommunicable diseases, including obesity, T1DM and T2DM, fatty liver disease, and even autism.233 Personalized medicine is likely to substantially challenge the assumption that 1 diet fits all and demonstrates that recommendations on macronutrients and micronutrients do not affect every individual in the same way.234 Given that diet is an environmental exposure that interacts with the genome, the epigenome, and a person’s metabolism, the field of personalized nutrition that takes into account the interactions among diet, genes, and health using the approaches of nutrigenomics and nutrigenetics may become feasible to identify the optimal diet for an individual.234
Some studies suggest that prebiotics and probiotics in pregnancy provide benefits235; however, the quality of evidence is weak as diet is simultaneously modified. To date, randomized clinical trials of probiotics or prebiotics in pregnant women with obesity have shown little benefit on infant outcomes.236 However, remodeling metabolic pathways during pregnancy or lactation because of changes in the microbiome holds the promise for diminishing adverse developmental programming in the next generation.
Priority recommendations related to nutrition in pregnancy and lactation requiring the immediate attention of funding agencies were identified below.
Specific major gaps
Funding agencies should take an active role in directing the generation of high-quality evidence relevant to nutrition in pregnancy. This may be accomplished through funding opportunities that require standardization across studies, control of confounders, and collection of common measures across studies and within specific populations while ensuring enrollment of diverse populations for optimal generalizability. Funding agencies can request investigators to include specific variables and direct how they will be measured to increase the ability to compare studies or pool data in the future.
Research projects that are designed to identify and validate nutritional biochemical markers to assess nutritional status are strongly recommended.
Research is needed to understand the development of nutrigenomic-based approaches to identify ways to individualize nutrition recommendations.
Research on the influence of the gut microbiome on maternal and infant health is emerging as a highly important area to increase our understanding of how manipulation through diet, human milk oligosaccharides, and prebiotics or probiotics could influence perinatal outcomes.
High-quality studies of adolescent pregnancy and multiple gestations are needed to develop and implement effective nutritional strategies.
A national policy group should be formed that includes representation from governmental, professional, academic, and charity organizations to develop approaches that will promote the consumption of nutritious food among women before and during pregnancy and lactation.
The following strategies are recommended:
Design evidence-based educational approaches that focus on nutrition across academic clinical programs for all providers in training, emphasizing a multidisciplinary team approach to management.
Design evidence-based education interventions focused on nutrition aimed at the public, both consumers and pregnant women and their families.
Leverage advocacy groups to infuse evidence-based nutrition knowledge across public platforms.
Leverage the use of technology to disseminate appropriate nutrition education (Apps) and create connections between scientists, policy-makers, and the general population.
Fill a major gap in our understanding of fetal growth: to move beyond the limitations of our current ultrasound techniques to develop a simple approach during pregnancy to measure accurately, repetitively, and noninvasively fetal growth, including length, head circumference, “weight,” and body composition—as a minimum, lean vs fat mass. Such methods would allow essential approaches to develop norms of fetal growth, to make such clinical measurements locally within unique populations and institutions, and to measure responses to different maternal diets and environmental conditions.
Institute measures of maternal glucose concentration throughout pregnancy and for longer periods (eg, continuous glucose monitors) to establish glycemic patterns. A major gap in the capacity to improve glucose metabolism and concentrations in the pregnant mother and to avoid or reduce the adverse impact on fetal growth and development is the lack of more continuous daytime and nocturnal measurements, which could help maintain maternal glucose concentrations in the “normal” range and at “reasonably” constant levels for much longer periods. Considerably more studies are now in progress, indicating that this approach has the potential to improve current pregnancy management.237–241
Implement better methods to assess lipid availability and use by the fetus, which measure maternal lipid concentrations and profiles throughout pregnancy and for longer periods, including both fasting and post-prandial triglycerides and RBC FA profiles, the latter to characterize dietary intake of FA, omega-6, and omega-3 polyunsaturated essential FAs. Moreover, it is important to determine how to maintain maternal lipid concentrations in the “normal” range and at relatively constant concentrations with appropriate and successful maternal diets. Essential omega-3 FA in the maternal diet should be emphasized with sufficient omega-6 FAs for fetal growth, ensuring optimal neuronal development and brain growth. The longer-term consequences for later life metabolism and risk of adiposity and neurologic development and cognitive capacity remain high priorities for future research.
More clearly discern the normal amino acid and protein nutrition of the fetus given that the optimal maternal dietary protein content is not known. The regulatory roles of individual amino acids in the fetus need a much clearer definition.
Differentiate how to best feed the mother whose fetus and its placenta are showing signs of growth restriction (undernutrition) and how to feed the mother whose fetus and placenta are showing signs of overgrowth of fat mass (ovenutrition) as early as possible in pregnancy.242
Gestational diabetes mellitus and obesity
The following strategies are suggested to improve the outcome of pregnancies in women with GDM and/or obesity:
Establish consensus across the field for diagnostic criteria and common priority measures in RCTs, such as measurement of neonatal adiposity.
Promote prospective planning with agreed-on, clearly defined outcomes and adequate power estimates for multicenter trials.
Establish consistent BMI reporting, including linking electronic medical record data for utilization of measured rather than reported maternal prepregnancy weight.
Encourage consistent GWG reporting to include both total and pattern of GWG.
Include, as appropriate, studies of women within all major ethnic groups and analyze as distinct groups (ethnicity, degrees of glucose intolerance, insulin resistance, and abnormalities in insulin secretion) to mitigate metabolic heterogeneity.
Use standardized treatments. The overall goal is balanced nutrition that is affordable and culturally acceptable for women with GDM,114 as it should be for all pregnant women, which normalizes maternal weight and blood glucose concentrations. There should be an increased representation of ethnically diverse women and women of lower socioeconomic status.
Ascertain how individuals with different metabolic capacities might respond differently to dietary manipulation utilizing nutrigenomics specific to the mother, her microbiome, and potentially her fetus (personalized nutrition).
Future directions for preconception bariatric-metabolic surgery research include the following:
Pregnancy outcomes by racial and ethnic status
Impact of micronutrient supplementation on maternal-fetal outcomes
Impact of specific bariatric surgeries, timing of surgery, and GWG and subsequent maternal-fetal outcomes
Mechanisms of increased risk of growth-restricted infants
Effects on childhood development, growth, and expression of chronic diseases of adulthood
Preeclampsia studies presented special challenges, in addition to the usual challenges of nutritional studies. These suggested that successful studies are as follows:
Studies should be done before pregnancy and in the periconceptional period. Many of the relevant nutritionally related developments of preeclampsia are present in very early pregnancy and some may be present before pregnancy.
Studies require an accurate medical diagnosis that avoids confusion among the HDP and self-reporting, as self-reporting of preeclampsia is very inaccurate (50%–59% positive predictive value).243
Studies should recognize the heterogeneous character and risk factors for preeclampsia and strive to consider “subsets.”
Conclusions
There is consistent agreement that a woman’s nutrition and weight should be assessed and improved before, during, and after pregnancy to encourage and promote health of the woman and her offspring.26,244,245 Thus, we must not wait for definitive scientific proof of mechanisms that underlie the potential beneficial effects of quality nutrition in a childbearing woman before recommending nutrient-rich diets before and during pregnancy and during lactation. It is the view of the authors that now is the time to assess our current knowledge of nutritional needs of women during their reproductive years, to apply what we know, to generate public health policies that ensure nutritious food availability, and to strongly encourage funding agencies to prioritize nutritional research that will address the several knowledge gaps to improve health benefits to all populations.
ACKNOWLEDGMENTS
We offer deep appreciation to the Vitamix Foundation and Bob’s Red Mill for the contribution toward a conference and to the experts who were speakers at the 2019 Nutrition in Pregnancy conference. Their contributions were important in the writing of this manuscript. The following experts involved in this review were as follows: Jennifer Barber, PhD, University of Michigan; Andrew Bremer, MD, PhD, Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), National Institutes of Health (NIH); Romy Gaillard, PhD, Erasmus University, The Netherlands; Kelle Moley, MD, National March of Dimes; Kripa Raghavan, DrPH, MPH, MSc, US Department of Agriculture, Center for Nutrition Policy and Promotion; Daniel Raiten, PhD, NICHD, NIH; Usha Ramakrishnan, PhD, Emory University; Leanne Redman, PhD, FTOS, Pennington Biomedical Research Center; Roberto Romero, MD, NICHD, NIH; Kartik Shankar, PhD, DABT, Anshutz Medical Campus, University of Colorado; Diane Stadler, PhD, RD, Oregon Health & Science University; Alison Steiber, PhD, RDN, Academy of Nutrition and Dietetics; Chittaranjan Yajnik, MD, FRCP, King Edward Memorial Hospital Research Centre, India.
The Vitamix Foundation and Bob’s Red Mill provided funding for the 2019 Nutrition in Pregnancy: Lifelong Consequences conference, which stimulated the idea of this report. The funders had no role in the planning of the conference or writing of the manuscript.
Footnotes
The authors report no conflict of interest.
REFERENCES
- 1.Barker DJ. The developmental origins of adult disease. J Am Coll Nutr 2004;23:588S–95S. [DOI] [PubMed] [Google Scholar]
- 2.World Health Organization. WHO antenatal care recommendations for a positive pregnancy experience. Nutritional interventions update: multiple micronutrient supplements during pregnancy. Geneva, Switzerland: World Health Organization; 2020. [PubMed] [Google Scholar]
- 3.National Academies of Sciences, Engineering, and Medicine, Health and Medicine Division, Food and Nutrition Board In: Harrison M, ed. Nutrition during pregnancy and lactation: exploring new evidence: proceedings of a workshop. Washington, DC: National Academies Press; 2020. [PubMed] [Google Scholar]
- 4.Stoody EE, Spahn JM, Casavale KO. The Pregnancy and Birth to 24 Months Project: a series of systematic reviews on diet and health. Am J Clin Nutr 2019;109:685–697S. [DOI] [PubMed] [Google Scholar]
- 5.Raghavan R, Dreibelbis C, Kingshipp BL, et al. Dietary patterns before and during pregnancy and maternal outcomes: a systematic review. Am J Clin Nutr 2019;109:705–728S. [DOI] [PubMed] [Google Scholar]
- 6.Raghavan R, Dreibelbis C, Kingshipp BL, et al. Dietary patterns before and during pregnancy and birth outcomes: a systematic review. Am J Clin Nutr 2019;109:729–756S. [DOI] [PubMed] [Google Scholar]
- 7.Güngör D, Nadaud P, LaPergola CC, et al. Infant milk-feeding practices and diabetes outcomes in offspring: a systematic review. Am J Clin Nutr 2019;109:817–837S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.US Department of Agriculture. US Department of Health and Human Services. Dietary Guidelines for Americans, 2020–2025. 9th Edition. Dietary Guidelines for Americans. 2020. Available at: https://www.dietaryguidelines.gov/sites/default/files/2020-12/Dietary_Guidelines_for_Americans_2020-2025.pdf. Accessed November 11, 2021. [Google Scholar]
- 9.Dietary Guidelines Advisory Committee. Scientific report of the 2020 Dietary Guidelines Advisory Committee: advisory report to the Secretary of Agriculture and the Secretary of Health and Human Services. Dietary Guidelines for Americans. 2020. Available at: https://www.dietaryguidelines.gov/2020-advisory-committee-report. Accessed November 11, 2021. [Google Scholar]
- 10.Koletzko B, Godfrey KM, Poston L, et al. Nutrition during pregnancy, lactation and early childhood and its implications for maternal and long-term child health: the early nutrition project recommendations. Ann Nutr Metab 2019;74:93–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Deputy NP, Sharma AJ, Kim SY, Hinkle SN. Prevalence and characteristics associated with gestational weight gain adequacy. Obstet Gynecol 2015;125:773–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Robbins CL, Zapata LB, Farr SL, et al. Core state preconception health indicators - pregnancy risk assessment monitoring system and behavioral risk factor surveillance system, 2009. MMWR Surveill Summ 2014;63:1–62. [PubMed] [Google Scholar]
- 13.Shin D, Lee KW, Song WO. Pre-pregnancy weight status is associated with diet quality and nutritional biomarkers during pregnancy. Nutrients 2016;8:162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bodnar LM, Simhan HN, Parker CB, et al. Racial or ethnic and socioeconomic inequalities in adherence to national dietary guidance in a large cohort of US pregnant women. J Acad Nutr Diet 2017;117:867–77.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Carmichael SL, Ma C, Feldkamp ML, Shaw GM; National Birth Defects Prevention Study. Comparing usual dietary intakes among subgroups of mothers in the year before pregnancy. Public Health Rep 2019;134:155–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bailey RL, Pac SG, Fulgoni VL 3rd, Reidy KC, Catalano PM. Estimation of total usual dietary intakes of pregnant women in the United States. JAMA Netw Open 2019;2:e195967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rai D, Bird JK, McBurney Ml, Chapman-Novakofski KM. Nutritional status as assessed by nutrient intakes and biomarkers among women of childbearing age—is the burden of nutrient inadequacies growing in America? Public Health Nutr 2015;18:1658–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Almond D, Currie J. Killing me softly: the fetal origins hypothesis. J Econ Perspect 2011;25:153–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hytten F Nutritional requirements in pregnancy. What happens if they are not met? Midwifery 1990;6:140–5. [DOI] [PubMed] [Google Scholar]
- 20.Abrams B, Altman SL, Pickett KE. Pregnancy weight gain: still controversial. Am J Clin Nutr 2000;71:1233–1241S. [DOI] [PubMed] [Google Scholar]
- 21.Stephenson J, Heslehurst N, Hall J, et al. Before the beginning: nutrition and lifestyle in the preconception period and its importance for future health. Lancet 2018;391:1830–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fleming TP, Watkins AJ, Velazquez MA, et al. Origins of lifetime health around the time of conception: causes and consequences. Lancet 2018;391:1842–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Godfrey KM, Matthews N, Glazier J, Jackson A, Wilman C, Sibley CP. Neutral amino acid uptake by the microvillous plasma membrane of the human placenta is inversely related to fetal size at birth in normal pregnancy. J Clin Endocrinol Metab 1998;83:3320–6. [DOI] [PubMed] [Google Scholar]
- 24.Institute of Medicine (US) and National Research Council (US) Committee to Reexamine IOM Pregnancy Weight Guidelines In: Rasmussen KM, Yaktine AL, eds. Weight gain during pregnancy: reexamining the guidelines. Washington (DC): National Academies Press (US); 2009. [PubMed] [Google Scholar]
- 25.Kominiarek MA, Peaceman AM. Gestational weight gain. Am J Obstet Gynecol 2017;217:642–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hanson MA, Bardsley A, De-Regil LM, et al. The International Federation of Gynecology and Obstetrics (FIGO) recommendations on adolescent, preconception, and maternal nutrition: “Think Nutrition First.” Int J Gynaecol Obstet 2015;131:S213–53. [DOI] [PubMed] [Google Scholar]
- 27.Ramakrishnan U, Grant F, Goldenberg T, Zongrone A, Martorell R. Effect of women’s nutrition before and during early pregnancy on maternal and infant outcomes: a systematic review. Paediatr Perinat Epidemiol 2012;26:285–301. [DOI] [PubMed] [Google Scholar]
- 28.Lindsay KL, Buss C, Wadhwa PD, Entringer S. The interplay between nutrition and stress in pregnancy: implications for fetal programming of brain development. Biol Psychiatry 2019;85:135–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Borge TC, Aase H, Brantsæter AL, Biele G. The importance of maternal diet quality during pregnancy on cognitive and behavioural outcomes in children: a systematic review and meta-analysis. BMJ Open 2017;7:e016777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.De-Regil LM, Peña-Rosas JP, Fernández-Gaxiola AC, Rayco-Solon P. Effects and safety of periconceptional oral folate supplementation for preventing birth defects. Cochrane Database Syst Rev 2015:CD007950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Keats EC, Haider BA, Tam E, Bhutta ZA. Multiple-micronutrient supplementation for women during pregnancy. Cochrane Database Syst Rev 2019;3:CD004905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fall CHD, Kumaran K. Metabolic programming in early life in humans. Philos Trans R Soc Lond B Biol Sci 2019;374:20180123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Aris IM, Fleisch AF, Oken E. Developmental origins of disease: emerging prenatal risk factors and future disease risk. Curr Epidemiol Rep 2018;5:293–302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lassance L, Haghiac M, Leahy P, et al. Identification of early transcriptome signatures in placenta exposed to insulin and obesity. Am J Obstet Gynecol 2015;212:647.e1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Calkins K, Devaskar SU. Fetal origins of adult disease. Curr Probl Pediatr Adolesc Health Care 2011;41:158–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Stanner SA, Bulmer K, Andrès C, et al. Does malnutrition in utero determine diabetes and coronary heart disease in adulthood? Results from the Leningrad siege study, a cross sectional study. BMJ 1997;315:1342–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.World health Organization. Report of the Commission on Ending Childhood Obesity: implementation plan: executive summary. World Health Organization. 2017. Available at: https://apps.who.int/iris/handle/10665/259349. Accessed November 11, 2021. [Google Scholar]
- 38.Hambidge KM, Bann CM, McClure EM, et al. Maternal characteristics affect fetal growth response in the women first preconception nutrition trial. Nutrients 2019;11:2534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hambidge KM, Westcott JE, Garcés A, et al. A multicountry randomized controlled trial of comprehensive maternal nutrition supplementation initiated before conception: the Women First trial. Am J Clin Nutr 2019;109:457–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.ACOG Practice Bulletin No 156: obesity in pregnancy. Obstet Gynecol 2015;126:e112–26. [DOI] [PubMed] [Google Scholar]
- 41.Flegal KM, Kruszon-Moran D, Carroll MD, Fryar CD, Ogden CL. Trends in obesity among adults in the United States, 2005 to 2014. JAMA 2016;315:2284–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.van Elten TM, Karsten MDA, Geelen A, et al. Preconception lifestyle intervention reduces long term energy intake in women with obesity and infertility: a randomised controlled trial. Int J Behav Nutr Phys Act 2019;16:3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.van Elten TM, Karsten MDA, Geelen A, et al. Effects of a preconception lifestyle intervention in obese infertile women on diet and physical activity; a secondary analysis of a randomized controlled trial. PLoS One 2018;13:e0206888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Opray N, Grivell RM, Deussen AR, Dodd JM. Directed preconception health programs and interventions for improving pregnancy outcomes for women who are overweight or obese. Cochrane Database Syst Rev 2015;7:CD010932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Catalano PM, Tyzbir ED, Allen SR, McBean JH, McAuliffe TL. Evaluation of fetal growth by estimation of neonatal body composition. Obstet Gynecol 1992;79:46–50. [PubMed] [Google Scholar]
- 46.WHO recommendations on antenatal care for a positive pregnancy experience. Geneva, Switzerland: World Health Organization; 2016. [PubMed] [Google Scholar]
- 47.Blencowe H, Krasevec J, de Onis M, et al. National, regional, and worldwide estimates of low birthweight in 2015, with trends from 2000: a systematic analysis. Lancet Glob Health 2019;7:e849–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Salam RA, Das JK, Ali A, Lassi ZS, Bhutta ZA. Maternal undernutrition and intra-uterine growth restriction. Expert Rev Obstet Gynecol 2013;8:559–67. [Google Scholar]
- 49.Bergmann RL, Bergmann KE, Dudenhausen JW. Undernutrition and growth restriction in pregnancy. Nestle Nutr Workshop Ser Pediatr Program 2008;61:103–21. [DOI] [PubMed] [Google Scholar]
- 50.Thorn SR, Rozance PJ, Brown LD, Hay WW Jr. The intrauterine growth restriction phenotype: fetal adaptations and potential implications for later life insulin resistance and diabetes. Semin Reprod Med 2011;29:225–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Thorn SR, Brown LD, Rozance PJ, Hay WW Jr, Friedman JE. Increased hepatic glucose production in fetal sheep with intra-uterine growth restriction is not suppressed by insulin. Diabetes 2013;62:65–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gluckman PD, Hanson MA, Buklijas T, Low FM, Beedle AS. Epigenetic mechanisms that underpin metabolic and cardiovascular diseases. Nat Rev Endocrinol 2009;5:401–8. [DOI] [PubMed] [Google Scholar]
- 53.Ross MG, Beall MH. Adult sequelae of intrauterine growth restriction. Semin Perinatol 2008;32:213–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Morrison JL, Regnault TR. Nutrition in pregnancy: optimising maternal diet and fetal adaptations to altered nutrient supply. Nutrients 2016;8:342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins—Obstetrics and the Society for Maternal-Fetal Medicine. ACOG Practice Bulletin No. 204: fetal growth restriction. Obstet Gynecol 2019;133:e97–109. [DOI] [PubMed] [Google Scholar]
- 56.Carr DJ, Wallace JM, Aitken RP, et al. Uteroplacental adenovirus vascular endothelial growth factor gene therapy increases fetal growth velocity in growth-restricted sheep pregnancies. Hum Gene Ther 2014;25:375–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Potdar RD, Sahariah SA, Gandhi M, et al. Improving women’s diet quality preconceptionally and during gestation: effects on birth weight and prevalence of low birth weight—a randomized controlled efficacy trial in India (Mumbai Maternal Nutrition Project). Am J Clin Nutr 2014;100:1257–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Embleton ND, Skeath T. Catch-up growth and metabolic and cognitive outcomes in adolescents born preterm. Nestlé Nutr Inst Workshop Ser 2015;81:61–71. [DOI] [PubMed] [Google Scholar]
- 59.Embleton ND, Korada M, Wood CL, Pearce MS, Swamy R, Cheetham TD. Catch-up growth and metabolic outcomes in adolescents born preterm. Arch Dis Child 2016;101:1026–31. [DOI] [PubMed] [Google Scholar]
- 60.Heude B, Thiébaugeorges O, Goua V, et al. Pre-pregnancy body mass index and weight gain during pregnancy: relations with gestational diabetes and hypertension, and birth outcomes. Matern Child Health J 2012;16:355–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Barbour LA, Hernandez TL. Maternal lipids and fetal overgrowth: making fat from fat. Clin Ther 2018;40:1638–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hernandez TL, Van Pelt RE, Anderson MA, et al. A higher-complex carbohydrate diet in gestational diabetes mellitus achieves glucose targets and lowers postprandial lipids: a randomized crossover study. Diabetes Care 2014;37:1254–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Barbour LA, Farabi SS, Friedman JE, et al. Postprandial triglycerides predict newborn fat more strongly than glucose in women with obesity in early pregnancy. Obesity (Silver Spring) 2018;26:1347–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kulkarni SR, Kumaran K, Rao SR, et al. Maternal lipids are as important as glucose for fetal growth: findings from the Pune Maternal Nutrition Study. Diabetes Care 2013;36:2706–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Adank MC, Benschop L, Kors AW, et al. Maternal lipid profile in early pregnancy is associated with foetal growth and the risk of a child born large-for-gestational age: a population-based prospective cohort study : maternal lipid profile in early pregnancy and foetal growth. BMC Med 2020;18:276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Herrera E, Desoye G. Maternal and fetal lipid metabolism under normal and gestational diabetic conditions. Horm Mol Biol Clin Investig 2016;26:109–27. [DOI] [PubMed] [Google Scholar]
- 67.Kim SR, Kubo T, Kuroda Y, et al. Comparative metabolome analysis of cultured fetal and adult hepatocytes in humans. J Toxicol Sci 2014;39:717–23. [DOI] [PubMed] [Google Scholar]
- 68.Elsakr JM, Dunn JC, Tennant K, et al. Maternal western-style diet affects offspring islet composition and function in a non-human primate model of maternal over-nutrition. Mol Metab 2019;25:73–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Carver TD, Anderson SM, Aldoretta PA, Esler AL, Hay WW Jr. Glucose suppression of insulin secretion in chronically hyperglycemic fetal sheep. Pediatr Res 1995;38:754–62. [DOI] [PubMed] [Google Scholar]
- 70.Jing YH, Song YF, Yao YM, Yin J, Wang DG, Gao LP. Retardation of fetal dendritic development induced by gestational hyperglycemia is associated with brain insulin/IGF-I signals. Int J Dev Neurosci 2014;37:15–20. [DOI] [PubMed] [Google Scholar]
- 71.Bytoft B, Knorr S, Vlachova Z, et al. Long-term cognitive implications of intrauterine hyperglycemia in adolescent offspring of women with type 1 diabetes (the EPICOM Study). Diabetes Care 2016;39:1356–63. [DOI] [PubMed] [Google Scholar]
- 72.∅yen N, Diaz LJ, Leirgul E, et al. Prepregnancy diabetes and offspring risk of congenital heart disease: a nationwide cohort study. Circulation 2016;133:2243–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mackin ST, Nelson SM, Wild SH, et al. Factors associated with stillbirth in women with diabetes. Diabetologia 2019;62:1938–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Browne K, Park BY, Goetzinger KR, Caughey AB, Yao R. The joint effects of obesity and pregestational diabetes on the risk of still-birth. J Matern Fetal Neonatal Med 2021;34:332–8. [DOI] [PubMed] [Google Scholar]
- 75.Anderson EL, Howe LD, Jones HE, Higgins JP, Lawlor DA, Fraser A. The prevalence of non-alcoholic fatty liver disease in children and adolescents: a systematic review and meta-analysis. PLoS One 2015;10:e0140908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wesolowski SR, Kasmi KC, Jonscher KR, Friedman JE. Developmental origins of NAFLD: a womb with a clue. Nat Rev Gastroenterol Hepatol 2017;14:81–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Brumbaugh DE, Tearse P, Cree-Green M, et al. Intrahepatic fat is increased in the neonatal offspring of obese women with gestational diabetes. J Pediatr 2013;162:930–6.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hernandez TL, Farabi SS, Hirsch NM, et al. Maternal triglycerides in gestational diabetes are strongly associated with increased newborn hepatic fat independent of subcutaneous fat. Diabetes 2019;68. [Google Scholar]
- 79.Newton KP, Feldman HS, Chambers CD, et al. Low and high birth weights are risk factors for nonalcoholic fatty liver disease in children. J Pediatr 2017;187:141–6.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Conde-Agudelo A, Belizán JM, Lammers C. Maternal-perinatal morbidity and mortality associated with adolescent pregnancy in Latin America: cross-sectional study. Am J Obstet Gynecol 2005;192:342–9. [DOI] [PubMed] [Google Scholar]
- 81.Ganchimeg T, Ota E, Morisaki N, et al. Pregnancy and childbirth outcomes among adolescent mothers: a World Health Organization multicountry study. BJOG 2014;121 (Suppl1):40–8. [DOI] [PubMed] [Google Scholar]
- 82.Neal S, Channon AA, Chintsanya J. The impact of young maternal age at birth on neonatal mortality: evidence from 45 low and middle income countries. PLoS One 2018;13:e0195731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Salihu HM, Sharma PP, Ekundayo OJ, et al. Childhood pregnancy (10–14 years old) and risk of stillbirth in singletons and twins. J Pediatr 2006;148:522–6. [DOI] [PubMed] [Google Scholar]
- 84.Marvin-Dowle K, Soltani H. A comparison of neonatal outcomes between adolescent and adult mothers in developed countries: a systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol X 2020;6:100109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Frisancho AR, Matos J, Leonard WR, Yaroch LA. Developmental and nutritional determinants of pregnancy outcome among teenagers. Am J Phys Anthropol 1985;66:247–61. [DOI] [PubMed] [Google Scholar]
- 86.Scholl TO, Hediger ML, Schall JI. Maternal growth and fetal growth: pregnancy course and outcome in the Camden Study. Ann N Y Acad Sci 1997;817:292–301. [DOI] [PubMed] [Google Scholar]
- 87.Luther J, Aitken R, Milne J, et al. Maternal and fetal growth, body composition, endocrinology, and metabolic status in undernourished adolescent sheep. Biol Reprod 2007;77:343–50. [DOI] [PubMed] [Google Scholar]
- 88.Wallace JM. Competition for nutrients in pregnant adolescents: consequences for maternal, conceptus and offspring endocrine systems. J Endocrinol 2019;242:T1–19. [DOI] [PubMed] [Google Scholar]
- 89.Wallace JM, Aitken RP, Cheyne MA. Nutrient partitioning and fetal growth in rapidly growing adolescent ewes. J Reprod Fertil 1996;107:183–90. [DOI] [PubMed] [Google Scholar]
- 90.Wallace JM, Aitken RP, Milne JS, Hay WW Jr. Nutritionally mediated placental growth restriction in the growing adolescent: consequences for the fetus. Biol Reprod 2004;71:1055–62. [DOI] [PubMed] [Google Scholar]
- 91.Wallace JM, Luther JS, Milne JS, et al. Nutritional modulation of adolescent pregnancy outcome – a review. Placenta 2006;27(SupplA):S61–8. [DOI] [PubMed] [Google Scholar]
- 92.Adam CL, Williams PA, Milne JS, Aitken RP, Wallace JM. Orexigenic gene expression in late gestation ovine foetal hypothalamus is sensitive to maternal undernutrition and realimentation. J Neuroendocrinol 2015;27:765–71. [DOI] [PubMed] [Google Scholar]
- 93.Wallace JM, Milne JS, Aitken RP, Horgan GW, Adam CL. Ovine prenatal growth restriction impacts glucose metabolism and body composition throughout life in both sexes. Reproduction 2018;156:103–19. [DOI] [PubMed] [Google Scholar]
- 94.Marvin-Dowle K, Kilner K, Burley V, Soltani H. Differences in dietary pattern by maternal age in the Born in Bradford cohort: a comparative analysis. PLoS One 2018;13:e0208879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Das JK, Salam RA, Thornburg KL, et al. Nutrition in adolescents: physiology, metabolism, and nutritional needs. Ann N Y Acad Sci 2017;1393:21–33. [DOI] [PubMed] [Google Scholar]
- 96.Krebs N, Bagby S, Bhutta ZA, et al. International summit on the nutrition of adolescent girls and young women: consensus statement. Ann N Y Acad Sci 2017;1400:3–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Goldstein RF, Abell SK, Ranasinha S, et al. Association of gestational weight gain with maternal and infant outcomes: a systematic review and meta-analysis. JAMA 2017;317:2207–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.da Silva Lopes K, Ota E, Shakya P, et al. Effects of nutrition interventions during pregnancy on low birth weight: an overview of systematic reviews. BMJ Glob Health 2017;2:e000389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Dhaded SM, Hambidge KM, Ali SA, et al. Preconception nutrition intervention improved birth length and reduced stunting and wasting in newborns in South Asia: the Women First randomized controlled trial. PLoS One 2020;15:e0218960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Patton GC, Sawyer SM, Santelli JS, et al. Our future: a Lancet commission on adolescent health and wellbeing. Lancet 2016;387:2423–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Christian P, Smith ER. Adolescent undernutrition: global burden, physiology, and nutritional risks. Ann Nutr Metab 2018;72:316–28. [DOI] [PubMed] [Google Scholar]
- 102.Marvin-Dowle K, Burley VJ, Soltani H. Nutrient intakes and nutritional biomarkers in pregnant adolescents: a systematic review of studies in developed countries. BMC Pregnancy Childbirth 2016;16:268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Hoellen F, Hornemann A, Haertel C, et al. Does maternal underweight prior to conception influence pregnancy risks and outcome? In Vivo 2014;28:1165–70. [PubMed] [Google Scholar]
- 104.Mestman JH. Historical notes on diabetes and pregnancy. Endocrinologist 2002;12:224–42. [Google Scholar]
- 105.Hernandez TL, Brand-Miller JC. Nutrition therapy in gestational diabetes mellitus: time to move forward. Diabetes Care 2018;41:1343–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Freinkel N Banting Lecture 1980. Of pregnancy and progeny. Diabetes 1980;29:1023–35. [DOI] [PubMed] [Google Scholar]
- 107.Hernandez TL. Glycemic targets in pregnancies affected by diabetes: historical perspective and future directions. Curr Diab Rep 2015;15:565. [DOI] [PubMed] [Google Scholar]
- 108.Hernandez TL. Carbohydrate content in the GDM diet: two views: view 1: nutrition therapy in gestational diabetes: the case for complex carbohydrates. Diabetes Spectr 2016;29:82–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Hernandez TL, Friedman JE, Barbour LA. Insulin resistance in pregnancy: implications for mother and offspring. Cham, Switzerland: Springer Nature Switzerland AG; 2019. [Google Scholar]
- 110.Barbour LA, Hernandez TL. Maternal non-glycemic contributors to fetal growth in obesity and gestational diabetes: spotlight on lipids. Curr Diab Rep 2018;18:37. [DOI] [PubMed] [Google Scholar]
- 111.Metzger BE, Buchanan TA, Coustan DR, et al. Summary and recommendations of the Fifth International Workshop-Conference on Gestational Diabetes Mellitus. Diabetes Care 2007;30(Suppl2):S251–60. [DOI] [PubMed] [Google Scholar]
- 112.Han S, Middleton P, Shepherd E, Van Ryswyk E, Crowther CA. Different types of dietary advice for women with gestational diabetes mellitus. Cochrane Database Syst Rev 2017;2:CD009275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Tsirou E, Grammatikopoulou MG, Theodoridis X, et al. Guidelines for medical nutrition therapy in gestational diabetes mellitus: systematic review and critical appraisal. J Acad Nutr Diet 2019;119:1320–39. [DOI] [PubMed] [Google Scholar]
- 114.Hernandez TL, Mande A, Barbour LA. Nutrition therapy within and beyond gestational diabetes. Diabetes Res Clin Pract 2018;145:39–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Barbour LA, Hernandez TL, Friedman JE. Metabolic changes during normal, obese, and GDM pregnancies. In Reece A, Coustan D, ed. Diabetes and obesity in women. Philadelphia, PA: Wolters Kluwer. https://www.amazon.com/diabetes-obesity-women-albert-reece/dp/1496390547. Accessed September 26, 2018. [Google Scholar]
- 116.Barbour LA. Metabolic culprits in obese pregnancies and gestational diabetes mellitus: big babies, big twists, big picture: the 2018 Norbert Freinkel award lecture. Diabetes Care 2019;42:718–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Simmons D Prevention of gestational diabetes mellitus: where are we now? Diabetes Obes Metab 2015;17:824–34. [DOI] [PubMed] [Google Scholar]
- 118.García-Patterson A, Balsells M, Yamamoto JM, et al. Usual dietary treatment of gestational diabetes mellitus assessed after control diet in randomized controlled trials: subanalysis of a systematic review and meta-analysis. Acta Diabetol 2019;56:237–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Hernandez TL, Farabi SS, Jaron A, et al. 97-OR: randomization to a higher–complex carbohydrate vs. conventional diet in GDM improves glucose tolerance and results in similar cord blood insulin and newborn adiposity. Diabetes 2020;69. [Google Scholar]
- 120.Yamamoto JM, Kellett JE, Balsells M, et al. Gestational diabetes mellitus and diet: a systematic review and meta-analysis of randomized controlled trials examining the impact of modified dietary interventions on maternal glucose control and neonatal birth weight. Diabetes Care 2018;41:1346–61. [DOI] [PubMed] [Google Scholar]
- 121.Courcoulas AP, King WC, Belle SH, et al. Seven-year weight trajectories and health outcomes in the longitudinal assessment of bariatric surgery (LABS) study. JAMA Surg 2018;153:427–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kwong W, Tomlinson G, Feig DS. Maternal and neonatal outcomes after bariatric surgery; a systematic review and meta-analysis: do the benefits outweigh the risks? Am J Obstet Gynecol 2018;218:573–80. [DOI] [PubMed] [Google Scholar]
- 123.Dutton H, Borengasser SJ, Gaudet LM, Barbour LA, Keely EJ. Obesity in pregnancy: optimizing outcomes for mom and baby. Med Clin North Am 2018;102:87–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Burton GJ, Yung HW, Murray AJ. Mitochondrial - endoplasmic reticulum interactions in the trophoblast: stress and senescence. Placenta 2017;52:146–55. [DOI] [PubMed] [Google Scholar]
- 125.Chesly LC. Hyperensive disorders of pregnancy. New York, NY: Appleton-Century-Crofts; 1978. [Google Scholar]
- 126.Brewer T Metabolic toxemia of late pregnancy: a disease entity. Gynaecologia 1969;167:1–8. [DOI] [PubMed] [Google Scholar]
- 127.Chesley LC. Hypertensive disorders in pregnancy. J Nurse Midwifery 1985;30:99–104. [DOI] [PubMed] [Google Scholar]
- 128.Robinson M Salt in pregnancy. Lancet 1958;1:178–81. [DOI] [PubMed] [Google Scholar]
- 129.Hofmeyr GJ, Lawrie TA, Atallah AN, Torloni MR. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database Syst Rev 2018;10:CD001059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Rumbold A, Ota E, Hori H, Miyazaki C, Crowther CA. Vitamin E supplementation in pregnancy. Cochrane Database Syst Rev 2015;9:CD004069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Rumbold A, Ota E, Nagata C, Shahrook S, Crowther CA. Vitamin C supplementation in pregnancy. Cochrane Database Syst Rev 2015;2015:CD004072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Bulloch RE, Lovell AL, Jordan VMB, McCowan LME, Thompson JMD, Wall CR. Maternal folic acid supplementation for the prevention of preeclampsia: a systematic review and meta-analysis. Paediatr Perinat Epidemiol 2018;32:346–57. [DOI] [PubMed] [Google Scholar]
- 133.Achamrah N, Ditisheim A. Nutritional approach to preeclampsia prevention. Curr Opin Clin Nutr Metab Care 2018;21:168–73. [DOI] [PubMed] [Google Scholar]
- 134.Wilson RL, Grieger JA, Bianco-Miotto T, Roberts CT. Association between maternal zinc status, dietary zinc intake and pregnancy complications: a systematic review. Nutrients 2016;8:641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Xiao Y, Sun H, Li C, et al. Effect of iodine nutrition on pregnancy outcomes in an iodine-sufficient area in China. Biol Trace Elem Res 2018;182:231–7. [DOI] [PubMed] [Google Scholar]
- 136.Kinshella MW, Omar S, Scherbinsky K, et al. Effects of maternal nutritional supplements and dietary interventions on placental complications: an umbrella review, meta-analysis and evidence map. Nutrients 2021;13:472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Smeeth L, Williams D. Can a dietary supplement prevent pre-eclampsia? BMJ 2011;342:d2777. [DOI] [PubMed] [Google Scholar]
- 138.Bodnar LM, Tang G, Ness RB, Harger G, Roberts JM. Periconceptional multivitamin use reduces the risk of preeclampsia. Am J Epidemiol 2006;164:470–7. [DOI] [PubMed] [Google Scholar]
- 139.Catov JM, Nohr EA, Bodnar LM, Knudson VK, Olsen SF, Olsen J. Association of periconceptional multivitamin use with reduced risk of preeclampsia among normal-weight women in the Danish National Birth Cohort. Am J Epidemiol 2009;169:1304–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Vanderlelie J, Scott R, Shibl R, Lewkowicz J, Perkins A, Scuffham PA. First trimester multivitamin/mineral use is associated with reduced risk of pre-eclampsia among overweight and obese women. Matern Child Nutr 2016;12:339–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Ormesher L, Myers JE, Chmiel C, et al. Effects of dietary nitrate supplementation, from beetroot juice, on blood pressure in hypertensive pregnant women: a randomised, double-blind, placebo-controlled feasibility trial. Nitric Oxide 2018;80:37–44. [DOI] [PubMed] [Google Scholar]
- 142.Rakova N, Muller DN, Staff AC, Luft FC, Dechend R. Novel ideas about salt, blood pressure, and pregnancy. J Reprod Immunol 2014;101–102:135–9. [DOI] [PubMed] [Google Scholar]
- 143.Cindrova-Davies T The therapeutic potential of antioxidants, ER chaperones, NO and H2S donors, and statins for treatment of preeclampsia. Front Pharmacol 2014;5:119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Kar S, Wong M, Rogozinska E, Thangaratinam S. Effects of omega-3 fatty acids in prevention of early preterm delivery: a systematic review and meta-analysis of randomized studies. Eur J Obstet Gynecol Reprod Biol 2016;198:40–6. [DOI] [PubMed] [Google Scholar]
- 145.Amati F, Hassounah S, Swaka A. The impact of Mediterranean dietary patterns during pregnancy on maternal and offspring health. Nutrients 2019;11:1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Biagi C, Nunzio MD, Bordoni A, Gori D, Lanari M. Effect of adherence to Mediterranean diet during pregnancy on children’s health: a systematic review. Nutrients 2019;11:997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Chen X, Zhao D, Mao X, Xia Y, Baker PN, Zhang H. Maternal dietary patterns and pregnancy outcome. Nutrients 2016;8:351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Kibret KT, Chojenta C, Gresham E, Tegegne TK, Loxton D. Maternal dietary patterns and risk of adverse pregnancy (hypertensive disorders of pregnancy and gestational diabetes mellitus) and birth (preterm birth and low birth weight) outcomes: a systematic review and meta-analysis. Public Health Nutr 2018:1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zhang Y, Lin J, Fu W, Liu S, Gong C, Dai J. Mediterranean diet during pregnancy and childhood for asthma in children: a systematic review and meta-analysis of observational studies. Pediatr Pulmonol 2019;54:949–61. [DOI] [PubMed] [Google Scholar]
- 150.Vujkovic M, de Vries JH, Lindemans J, et al. The preconception Mediterranean dietary pattern in couples undergoing in vitro fertilization/intracytoplasmic sperm injection treatment increases the chance of pregnancy. Fertil Steril 2010;94:2096–101. [DOI] [PubMed] [Google Scholar]
- 151.US Food and Drug Administration. FDA issues revised “Advice about eating fish: for women who are or might become pregnant, breastfeeding mothers, and young children.” Food and Drug Administration. 2019. Available at: https://www.fda.gov/food/cfsan-constituent-updates/fda-issues-revised-advice-about-eating-fish-women-who-are-or-might-become-pregnant-breastfeeding. Accessed November 11, 2021. [Google Scholar]
- 152.Vlaardingerbroek H Unusual cause of congenital hypothyroidism in a term infant. BMJ Case Rep 2021;14:e237930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Mise N, Ohtsu M, Ikegami A, et al. Hijiki seaweed consumption elevates levels of inorganic arsenic intake in Japanese children and pregnant women. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 2019;36:84–95. [DOI] [PubMed] [Google Scholar]
- 154.Desrosiers TA, Siega-Riz AM, Mosley BS, Meyer RE. National Birth Defects Prevention Study. Low carbohydrate diets may increase risk of neural tube defects. Birth Defects Res 2018;110:901–9. [DOI] [PubMed] [Google Scholar]
- 155.Hernandez TL, Van Pelt RE, Anderson MA, et al. Women with gestational diabetes mellitus randomized to a higher-complex carbohydrate/low-fat diet manifest lower adipose tissue insulin resistance, inflammation, glucose, and free fatty acids: a pilot study. Diabetes Care 2016;39:39–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Godfrey KM, Sheppard A, Gluckman PD, et al. Epigenetic gene promoter methylation at birth is associated with child’s later adiposity. Diabetes 2011;60:1528–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Barker DJP, Thornburg KL. The obstetric origins of health for a lifetime. Clin Obstet Gynecol 2013;56:511–9. [DOI] [PubMed] [Google Scholar]
- 158.Holme AM, Roland MC, Lorentzen B, Michelsen TM, Henriksen T. Placental glucose transfer: a human in vivo study. PLoS One 2015;10:e0117084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Lavie M, Lavie I, Maslovitz S. Paleolithic diet during pregnancy-a potential beneficial effect on metabolic indices and birth weight. Eur J Obstet Gynecol Reprod Biol 2019;242:7–11. [DOI] [PubMed] [Google Scholar]
- 160.Shapiro ALB, Kaar JL, Crume TL, et al. Maternal diet quality in pregnancy and neonatal adiposity: the Healthy Start Study. Int J Obes (Lond) 2016;40:1056–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.LifeCycle Project-Maternal Obesity and Childhood Outcomes Study Group, Voerman E, Santos S, et al. Association of gestational weight gain with adverse maternal and infant outcomes. JAMA 2019;321:1702–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Rogozińska E, Zamora J, Marlin N, et al. Gestational weight gain outside the Institute of Medicine recommendations and adverse pregnancy outcomes: analysis using individual participant data from randomised trials. BMC Pregnancy Childbirth 2019;19:322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Nicklas JM, Barbour LA. Optimizing weight for maternal and infant health - tenable, or too late? Expert Rev Endocrinol Metab 2015;10:227–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Scott C, Andersen CT, Valdez N, et al. No global consensus: a cross-sectional survey of maternal weight policies. BMC Pregnancy Childbirth 2014;14:167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Deputy NP, Sharma AJ, Kim SY, Olson CK. Achieving appropriate gestational weight gain: the role of healthcare provider advice. J Womens Health (Larchmt) 2018;27:552–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Rogerson D, Soltani H, Copeland R. Undergraduate UK nutrition education might not adequately address weight management. Public Health Nutr 2016;19:371–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Kominiarek MA, Seligman NS, Dolin C, et al. Gestational weight gain and obesity: is 20 pounds too much? Am J Obstet Gynecol 2013;209:214.e1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Bodnar LM, Pugh SJ, Lash TL, et al. Low gestational weight gain and risk of adverse perinatal outcomes in obese and severely obese women. Epidemiology 2016;27:894–902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Catalano PM, Mele L, Landon MB, et al. Inadequate weight gain in overweight and obese pregnant women: what is the effect on fetal growth? Am J Obstet Gynecol 2014;211:137. e1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Bodnar LM, Siminerio LL, Himes KP, et al. Maternal obesity and gestational weight gain are risk factors for infant death. Obesity (Silver Spring) 2016;24:490–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Faucher MA, Barger MK. Gestational weight gain in obese women by class of obesity and select maternal/newborn outcomes: a systematic review. Women Birth 2015;28:e70–9. [DOI] [PubMed] [Google Scholar]
- 172.Cantor AG, Jungbauer RM, McDonagh M, et al. Counseling and behavioral interventions for healthy weight and weight gain in pregnancy: evidence report and systematic review for the US Preventive Services Task Force. JAMA 2021;325:2094–109. [DOI] [PubMed] [Google Scholar]
- 173.US Preventive Services Task Force, Davidson KW, Barry MJ, et al. Behavioral counseling interventions for healthy weight and weight gain in pregnancy: US Preventive Services Task Force recommendation statement. JAMA 2021;325:2087–93. [DOI] [PubMed] [Google Scholar]
- 174.Bauserman MS, Bann CM, Hambidge KM, et al. Gestational weight gain in 4 low- and middle-income countries and associations with birth outcomes: a secondary analysis of the Women First Trial. Am J Clin Nutr 2021;114:804–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Bartick MC, Schwarz EB, Green BD, et al. Suboptimal breastfeeding in the United States: maternal and pediatric health outcomes and costs. Matern Child Nutr 2017;13:e12366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Section on Breastfeeding. Breastfeeding and the use of human milk. Pediatrics 2012;129:e827–41. [DOI] [PubMed] [Google Scholar]
- 177.Rasmussen KM. The influence of maternal nutrition on lactation. Annu Rev Nutr 1992;12:103–17. [DOI] [PubMed] [Google Scholar]
- 178.John A, Sun R, Maillart L, Schaefer A, Hamilton Spence E, Perrin MT. Macronutrient variability in human milk from donors to a milk bank: implications for feeding preterm infants. PLoS One 2019;14:e0210610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Perrin MT, Fogleman AD, Newburg DS, Allen JC. A longitudinal study of human milk composition in the second year postpartum: implications for human milk banking. Matern Child Nutr 2017;13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Khan S, Hepworth AR, Prime DK, Lai CT, Trengove NJ, Hartmann PE. Variation in fat, lactose, and protein composition in breast milk over 24 hours: associations with infant feeding patterns. J Hum Lact 2013;29:81–9. [DOI] [PubMed] [Google Scholar]
- 181.Del Prado M, Villalpando S, Elizondo A, Rodríguez M, Demmelmair H, Koletzko B. Contribution of dietary and newly formed arachidonic acid to human milk lipids in women eating a low-fat diet. Am J Clin Nutr 2001;74:242–7. [DOI] [PubMed] [Google Scholar]
- 182.Khodabakhshi A, Mehrad-Majd H, Vahid F, Safarian M. Association of maternal breast milk and serum levels of macronutrients, hormones, and maternal body composition with infant’s body weight. Eur J Clin Nutr 2018;72:394–400. [DOI] [PubMed] [Google Scholar]
- 183.Spahn JM, Callahan EH, Spill MK, et al. Influence of maternal diet on flavor transfer to amniotic fluid and breast milk and children’s responses: a systematic review. Am J Clin Nutr 2019;109:1003–1026S. [DOI] [PubMed] [Google Scholar]
- 184.Rudolph MC, Young BE, Lemas DJ, et al. Early infant adipose deposition is positively associated with the n-6 to n-3 fatty acid ratio in human milk independent of maternal BMI. Int J Obes (Lond) 2017;41:510–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Lemas DJ, Young BE, Baker PR 2nd, et al. Alterations in human milk leptin and insulin are associated with early changes in the infant intestinal microbiome. Am J Clin Nutr 2016;103:1291–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Rodel RL, Carli JM, Hirsch NM, et al. Human milk composition in type 2 diabetes imparts higher insulin exposure. Diabetes 2019;68(Suppl1). 1422–P. [Google Scholar]
- 187.Gunderson EP, Hurston SR, Ning X, et al. Lactation and progression to type 2 diabetes mellitus after gestational diabetes mellitus: a prospective cohort study. Ann Intern Med 2015;163:889–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Gunderson EP, Lewis CE, Lin Y, et al. Lactation duration and progression to diabetes in women across the childbearing years: the 30-year CARDIA study. JAMA Intern Med 2018;178:328–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Choi SR, Kim YM, Cho MS, Kim SH, Shim YS. Association between duration of breast feeding and metabolic syndrome: the Korean National Health and Nutrition Examination Surveys. J Womens Health (Larchmt) 2017;26:361–7. [DOI] [PubMed] [Google Scholar]
- 190.Peters SAE, Yang L, Guo Y, et al. Breastfeeding and the risk of maternal cardiovascular disease: a prospective study of 300 000 Chinese women. J Am Heart Assoc 2017;6:e006081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Chowdhury R, Sinha B, Sankar MJ, et al. Breastfeeding and maternal health outcomes: a systematic review and meta-analysis. Acta Paediatr 2015;104:96–113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Marshall NE, Lau B, Purnell JQ, Thornburg KL. Impact of maternal obesity and breastfeeding intention on lactation intensity and duration. Matern Child Nutr 2019;15:e12732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Wilson RD, Genetics Committee, Wilson RD, et al. Pre-conception folic acid and multivitamin supplementation for the primary and secondary prevention of neural tube defects and other folic acid-sensitive congenital anomalies. J Obstet Gynaecol Can 2015;37:534–52. [DOI] [PubMed] [Google Scholar]
- 194.Guinotte CL, Burns MG, Axume JA, et al. Methylenetetrahydrofolate reductase 677C-> T variant modulates folate status response to controlled folate intakes in young women. J Nutr 2003;133:1272–80. [DOI] [PubMed] [Google Scholar]
- 195.Institute of Medicine (US) Committee on Nutritional Status During Pregnancy and Lactation. Nutrition during pregnancy: part I weight gain: part II nutrient supplements. Washington, DC: National Academies Press (US); 1990. [PubMed] [Google Scholar]
- 196.Le CH. The prevalence of anemia and moderate-severe anemia in the US population (NHANES 2003–2012). PLoS One 2016;11:e0166635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Stevens GA, Finucane MM, De-Regil LM, et al. Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995–2011: a systematic analysis of population-representative data. Lancet Glob Health 2013;1:e16–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Dewey KG, Oaks BM. U-shaped curve for risk associated with maternal hemoglobin, iron status, or iron supplementation. Am J Clin Nutr 2017;106(Suppl6):1694–1702S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Smith ER, Shankar AH, Wu LS, et al. Modifiers of the effect of maternal multiple micronutrient supplementation on stillbirth, birth outcomes, and infant mortality: a meta-analysis of individual patient data from 17 randomised trials in low-income and middle-income countries. Lancet Glob Health 2017;5:e1090–100. [DOI] [PubMed] [Google Scholar]
- 200.Caudill MA, Strupp BJ, Muscalu L, Nevins JEH, Canfield RL. Maternal choline supplementation during the third trimester of pregnancy improves infant information processing speed: a randomized, double-blind, controlled feeding study. FASEB J 2018;32:2172–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Jacobson SW, Carter RC, Molteno CD, et al. Efficacy of maternal choline supplementation during pregnancy in mitigating adverse effects of prenatal alcohol exposure on growth and cognitive function: a randomized, double-blind, placebo-controlled clinical trial. Alcohol Clin Exp Res 2018;42:1327–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Derbyshire E, Obeid R. Choline, neurological development and brain function: a systematic review focusing on the first 1000 days. Nutrients 2020:12:1731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Freedman R, Hunter SK, Law AJ, et al. Higher gestational choline levels in maternal infection are protective for infant brain development. J Pediatr 2019;208:198–206.e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.World Health Organization and knowledge translation in maternal, newborn, child and adolescent health and nutrition, Arch Dis Child 2021. Dec 30, 10.1136/archdischild-2021-323102. [DOI] [PMC free article] [PubMed]
- 205.Gernand AD, Schulze KJ, Stewart CP, West KP Jr, Christian P. Micronutrient deficiencies in pregnancy worldwide: health effects and prevention. Nat Rev Endocrinol 2016;12:274–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Most J, Dervis S, Haman F, Adamo KB, Redman LM. Energy intake requirements in pregnancy. Nutrients 2019;11:1812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Darnton-Hill I, Mkparu UC. Micronutrients in pregnancy in low- and middle-income countries. Nutrients 2015;7:1744–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Pearce EN, Lazarus JH, Moreno-Reyes R, Zimmermann MB. Consequences of iodine deficiency and excess in pregnant women: an overview of current knowns and unknowns. Am J Clin Nutr 2016;104:918–923S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Alexander EK, Pearce EN, Brent GA, et al. 2017 Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and the postpartum. Thyroid 2017;27:315–89. [DOI] [PubMed] [Google Scholar]
- 210.Miranda A, Sousa N. Maternal hormonal milieu influence on fetal brain development. Brain Behav 2018;8:e00920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Vermiglio F, Lo Presti VP, Moleti M, et al. Attention deficit and hyperactivity disorders in the offspring of mothers exposed to mild-moderate iodine deficiency: a possible novel iodine deficiency disorder in developed countries. J Clin Endocrinol Metab 2004;89:6054–60. [DOI] [PubMed] [Google Scholar]
- 212.Krebs-Smith SM, Pannucci TE, Subar AF, et al. Update of the healthy eating index: HEI-2015. J Acad Nutr Diet 2018;118:1591–602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Tahir MJ, Haapala JL, Foster LP, et al. Higher maternal diet quality during pregnancy and lactation is associated with lower infant weight-for-length, body fat percent, and fat mass in early postnatal life. Nutrients 2019;11:632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Sen S, Rifas-Shiman SL, Shivappa N, et al. Associations of prenatal and early life dietary inflammatory potential with childhood adiposity and cardiometabolic risk in Project Viva. Pediatr Obes 2018;13:292–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Widen E, Siega-Riz AM. Prenatal nutrition: a practical guide for assessment and counseling. J Midwifery Womens Health 2010;55:540–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Anderson AS. Symposium on ‘nutritional adaptation to pregnancy and lactation.’ Pregnancy as a time for dietary change? Proc Nutr Soc 2001;60:497–504. [DOI] [PubMed] [Google Scholar]
- 217.Daru J, Allotey J, Peña-Rosas JP, Khan KS. Serum ferritin thresholds for the diagnosis of iron deficiency in pregnancy: a systematic review. Transfus Med 2017;27:167–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Harris WS, Luo J, Pottala JV, Margolis KL, Espeland MA, Robinson JG. Red blood cell fatty acids and incident diabetes mellitus in the Women’s Health Initiative memory study. PLoS One 2016;11:e0147894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Baack ML, Puumala SE, Messier SE, Pritchett DK, Harris WS. Daily enteral DHA supplementation alleviates deficiency in premature infants. Lipids 2016;51:423–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Harris WS, Tintle NL, Etherton MR, Vasan RS. Erythrocyte long-chain omega-3 fatty acid levels are inversely associated with mortality and with incident cardiovascular disease: the Framingham Heart Study. J Clin Lipidol 2018;12:718–27.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Lee A, Newton M, Radcliffe J, Belski R. Pregnancy nutrition knowledge and experiences of pregnant women and antenatal care clinicians: a mixed methods approach. Women Birth 2018;31:269–77. [DOI] [PubMed] [Google Scholar]
- 222.National Institute for Health and Care Excellence. Weight management before, during and after pregnancy. National Institute for Health and Care Excellence. 2010. Available at: https://www.nice.org.uk/guidance/ph27. Accessed November 11, 2021. [Google Scholar]
- 223.Arden MA, Duxbury AM, Soltani H. Responses to gestational weight management guidance: a thematic analysis of comments made by women in online parenting forums. BMC Pregnancy Childbirth 2014;14:216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Daley AJ, Jolly K, Jebb SA, et al. Effectiveness of regular weighing, weight target setting and feedback by community midwives within routine antenatal care in preventing excessive gestational weight gain: randomised controlled trial. BMC Obes 2015;3:7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Fealy S, Davis D, Foureur M, Attia J, Hazelton M, Hure A. The return of weighing in pregnancy: a discussion of evidence and practice. Women Birth 2020;33:119–24. [DOI] [PubMed] [Google Scholar]
- 226.Percival J Promoting health: making every contact count. Nurs Stand 2014;28:37–41. [DOI] [PubMed] [Google Scholar]
- 227.Dean SV, Lassi ZS, Imam AM, Bhutta ZA. Preconception care: nutritional risks and interventions. Reprod Health 2014;11 (Suppl3):S3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Lassi ZS, Moin A, Das JK, Salam RA, Bhutta ZA. Systematic review on evidence-based adolescent nutrition interventions. Ann N Y Acad Sci 2017;1393:34–50. [DOI] [PubMed] [Google Scholar]
- 229.Sherifali D, Nerenberg KA, Wilson S, et al. The effectiveness of eHealth technologies on weight management in pregnant and postpartum women: systematic review and meta-analysis. J Med Internet Res 2017;19:e337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Gage SH, Munafò MR, Davey Smith G. Causal inference in developmental origins of health and disease (DOHaD) research. Annu Rev Psychol 2016;67:567–85. [DOI] [PubMed] [Google Scholar]
- 231.Stiemsma LT, Michels KB. The role of the microbiome in the developmental origins of health and disease. Pediatrics 2018;141:e20172437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Castanys-Muñoz E, Martin MJ, Vazquez E. Building a beneficial microbiome from birth. Adv Nutr 2016;7:323–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Pascale A, Marchesi N, Marelli C, et al. Microbiota and metabolic diseases. Endocrine 2018;61:357–71. [DOI] [PubMed] [Google Scholar]
- 234.Peña-Romero AC, Navas-Carrillo D, Marín F, Orenes-Piñero E. The future of nutrition: nutrigenomics and nutrigenetics in obesity and cardiovascular diseases. Crit Rev Food Sci Nutr 2018;58:3030–41. [DOI] [PubMed] [Google Scholar]
- 235.Han MM, Sun JF, Su XH, et al. Probiotics improve glucose and lipid metabolism in pregnant women: a meta-analysis. Ann Transl Med 2019;7:99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Jarde A, Lewis-Mikhael AM, Moayyedi P, et al. Pregnancy outcomes in women taking probiotics or prebiotics: a systematic review and meta-analysis. BMC Pregnancy Childbirth 2018;18:14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Murphy HR, Rayman G, Duffield K, et al. Changes in the glycemic profiles of women with type 1 and type 2 diabetes during pregnancy. Diabetes Care 2007;30:2785–91. [DOI] [PubMed] [Google Scholar]
- 238.Feig DS, Donovan LE, Corcoy R, et al. Continuous glucose monitoring in pregnant women with type 1 diabetes (CONCEPTT): a multicentre international randomised controlled trial. Lancet 2017;390:2347–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Stewart ZA, Wilinska ME, Hartnell S, et al. Day-and-night closed-loop insulin delivery in a broad population of pregnant women with type 1 diabetes: a randomized controlled crossover trial. Diabetes Care 2018;41:1391–9. [DOI] [PubMed] [Google Scholar]
- 240.Secher AL, Ringholm L, Andersen HU, Damm P, Mathiesen ER. The effect of real-time continuous glucose monitoring in pregnant women with diabetes: a randomized controlled trial. Diabetes Care 2013;36:1877–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Paramasivam SS, Chinna K, Singh AKK, et al. Continuous glucose monitoring results in lower HbA1c in Malaysian women with insulin-treated gestational diabetes: a randomized controlled trial. Diabet Med 2018;35:1118–29. [DOI] [PubMed] [Google Scholar]
- 242.Wallace JM, Agard JP, Horgan GW. A new customised placental weight standard redefines the relationship between maternal obesity and extremes of placental size and is more closely associated with pregnancy complications than an existing population standard. J Dev Orig Health Dis 2020;11:350–9. [DOI] [PubMed] [Google Scholar]
- 243.Stuart JJ, Bairey Merz CN, Berga SL, et al. Maternal recall of hypertensive disorders in pregnancy: a systematic review. J Womens Health (Larchmt) 2013;22:37–I7. [DOI] [PubMed] [Google Scholar]
- 244.GBD 2017 Diet Collaborators. Health effects of dietary risks in 195 countries, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019;393:1958–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 548: weight gain during pregnancy. Obstet Gynecol 2013;121:210–2. [DOI] [PubMed] [Google Scholar]


